Abstract
Background
Type 1 diabetes (T1D) results from an autoimmune attack of the pancreatic β cells that progresses to dysglycemia and symptomatic hyperglycemia. Current biomarkers to track this evolution are limited, with development of islet autoantibodies marking the onset of autoimmunity and metabolic tests used to detect dysglycemia. Therefore, additional biomarkers are needed to better track disease initiation and progression. Multiple clinical studies have used proteomics to identify biomarker candidates. However, most of the studies were limited to the initial candidate identification, which needs to be further validated and have assays developed for clinical use. Here we curate these studies to help prioritize biomarker candidates for validation studies and to obtain a broader view of processes regulated during disease development.
Methods
This systematic review was registered with Open Science Framework (https://doi.org/10.17605/OSF.IO/N8TSA). Using PRISMA guidelines, we conducted a systematic search of proteomics studies of T1D in the PubMed to identify putative protein biomarkers of the disease. Studies that performed mass spectrometry-based untargeted/targeted proteomic analysis of human serum/plasma of control, pre-seroconversion, post-seroconversion, and/or T1D-diagnosed subjects were included. For unbiased screening, 3 reviewers screened all the articles independently using the pre-determined criteria.
Results
A total of 13 studies met our inclusion criteria, resulting in the identification of 266 unique proteins, with 31 (11.6%) being identified across 3 or more studies. The circulating protein biomarkers were found to be enriched in complement, lipid metabolism, and immune response pathways, all of which are found to be dysregulated in different phases of T1D development. We found 2 subsets: 17 proteins (C3, C1R, C8G, C4B, IBP2, IBP3, ITIH1, ITIH2, BTD, APOE, TETN, C1S, C6A3, SAA4, ALS, SEPP1 and PI16) and 3 proteins (C3, CLUS and C4A) have consistent regulation in at least 2 independent studies at post-seroconversion and post-diagnosis compared to controls, respectively, making them strong candidates for clinical assay development.
Conclusions
Biomarkers analyzed in this systematic review highlight alterations in specific biological processes in T1D, including complement, lipid metabolism, and immune response pathways, and may have potential for further use in the clinic as prognostic or diagnostic assays.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12014-023-09429-6.
Keywords: Type 1 diabetes, Biomarker, Plasma, Proteomics
Background
Our understanding of type 1 diabetes (T1D) pathophysiology has advanced significantly in the last 100 years since discovering insulin as a T1D treatment, but additional biomarkers of its earliest stages could help determine its cause and develop more refined and targeted prevention approaches. The disease begins as an autoimmune insult on pancreatic β cells (seroconversion, marked by the detection of circulating autoantibodies) that progresses to elevated blood glucose and glycated hemoglobin A1c in the body—the current gold standard for T1D diagnosis. Current biomarkers to track this evolution are limited, with the development of autoantibodies to insulin, glutamic acid decarboxylase, insulinoma-associated antigen-2, or zinc transporter 8 marking the onset of islet autoimmunity [1, 2]. Proteomics is a powerful tool to identify biomarkers, as it can detect and quantify thousands of proteins. Several proteomics studies have been carried out to identify T1D biomarkers. However, the development of biomarkers is a long process that involves identification of candidates, validation, and clinical assay development [3]. Despite all the efforts of the field, our knowledge is still concentrated in the initial biomarker candidate identification step. A deep analysis of the published reports can recognize reproducible protein expression patterns [4], leading to the identification of most promising candidates.
Here, we performed a systematic review of untargeted and targeted proteomics of serum or plasma from individuals in different stages of T1D development. We report several proteins that were differentially expressed in individuals at various stages of T1D development, and we also interpreted the findings to understand processes regulated in T1D development.
Methods
Study design and search strategy
We conducted this systematic review according to the PRISMA guidelines by searching the PubMed database with the terms “type 1 diabetes” and “proteomics” as of 08 August 2022 [5]. Articles were manually curated with the following expressions:
((Type 1 Diabetes) AND (Proteomics)) NOT ((Review [Publication Type])) OR (Systematic Review [Publication Type])) OR (Meta-analysis [Publication Type]) OR (Commentary [Publication Type])) AND ((Serum) OR (Plasma)).
Eligibility Criteria
Studies comparing the serum/plasma proteome of humans developing or having T1D and that of controls were included in the analysis. Ethnicity, study population size, sex, or disease time point were not included as exclusion criteria to minimize excluding informative biomarkers. We excluded reports of individuals with T1D without matched controls and any studies that failed to report detailed proteomic analyses. Study design (case–control, cohort, or longitudinal) was not an exclusion criterion. We excluded articles without accessible abstracts or full text, articles that were reviews, commentaries, systematic reviews, or meta-analyses.
Study selection
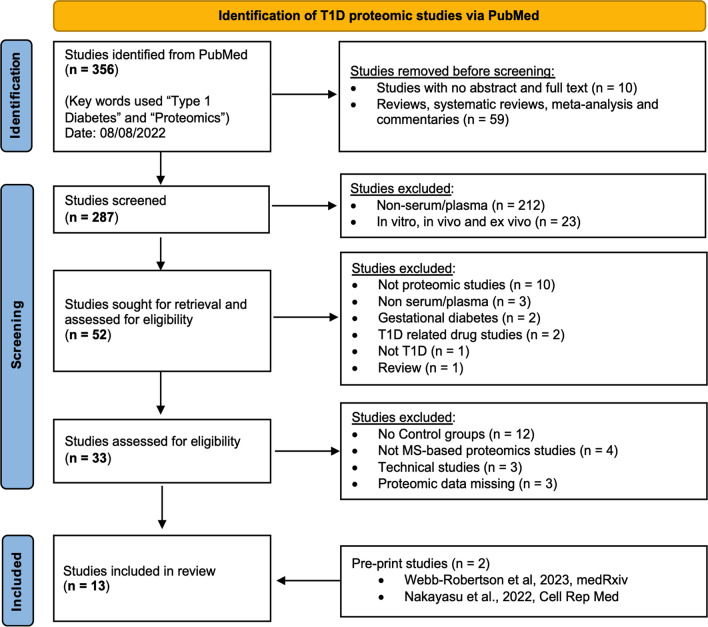
The systematic review of the literature resulted in 356 initial articles. All the studies that were excluded using the PubMed algorithm (see Eligibility Criteria) were manually verified that they did not meet the inclusion criteria. The remaining articles were manually screened to eliminate studies that did not use human serum/plasma or mass spectrometry-based proteomic analysis or were related to gestational diabetes and T1D-drug studies. Finally, the studies related to the mass spectrometry technique without a control group or with missing proteomic data were excluded from the final list after the full text was read. In addition, we added two manuscripts (unpublished at the time) by our group that met the eligibility criteria. Figure 1 outlines the study screening and selection process following the PRISMA guidelines.
Fig. 1.
PRISMA flow chart of literature search strategy, screening, and exclusion criteria
Data analysis and visualization
The final 13 articles included were screened by three reviewers independently (SS, ECE, HRH) to verify they met the initial inclusion criteria. The additional metadata of sample type, population size, mass spectrometry analysis type, data analysis method, and statistical tests were also included as factors. All the authors discussed any conflicts and were added to the analysis upon unanimous agreement. Protein data were extracted from the articles manually using Adobe/Microsoft Excel as described in the Additional file 1: Table S1. Proteins were reported based on UniProt IDs. When accession numbers/UniProt IDs were not provided or outdated databases were used for protein matches, UniProt IDs were found based on the peptide sequences or gene name. Protein abundances were manually converted to binary “–1” or “1” representing down or up-regulation, respectively, with “0” denoting not observed. In studies that reported abundance of multiple peptides for a protein, directional disagreement between them was reported as 1/-1. Studies were then grouped by the sampling time point (pre-seroconversion, post-seroconversion, and post-diagnosis). Functional-enrichment analysis was performed with Database for Annotation, Visualization, and Integrated Discovery (DAVID) using the default parameters [6]. We used the Kyoto Encyclopedia of Genes and Genomes (KEGG) output for further interpretations. Redundant pathways were consolidated by manual inspection and checking for overlap between pathways, being only the one with highest coverage kept for network analysis. Final network data were visualized with Cytoscape (v.3.9.1) and Graphpad prism software.
Results and discussion
Characteristics and description of eligible studies
The 13 articles described in our systematic review were performed across three disease developmental stages: pre-onset, further divided into (i) pre-seroconversion and (ii) post-seroconversion; and (iii) post-diagnosis. Pre/post seroconversion was defined based on the manifestation of the autoimmune response measured by the appearance of autoantibodies while post-diagnosis was defined by onset of symptomatic hyperglycemia. Details of the studies and their temporal categorization are summarized in Table 1 and the results are summarized in Additional file 1: Table S2.
Table 1.
Characteristic features of the eligible proteomic studies
| References | Study group | Control group | Sample type | Discovery method | Validation method | Number of Proteins Identified by Disease Progression | ||
|---|---|---|---|---|---|---|---|---|
| Clinical definition (Disease condition and sample size) |
Demographics (Age and sex, location/ ethnicity) |
Clinical definition (Disease condition and sample size) |
Demographics (Age and sex, location/ethnicity) |
|||||
| Pre-seroconversion | ||||||||
| Moulder et al. 2015, Diabetes | HLA + , autoantibody + . n = 19 | Age (3 m to 12y), Finland (DIPP) | HLA + ., autoantibody-, controls, n = 19 | Age, sex, sample periodicity, and risk group matched | Serum | 2DLC-MS/MS (Untargeted proteomics) | NA |
Pre-seroconversion: 3 (1, 2) Post-seroconversion Longitudinal: 55 (31, 24) Post-seroconversion: 10 (5, 5) |
| Frohnert et al. 2020, Diabetes | Family history of T1D, HLA + , autoantibody + . n = 20, T1D n = 22, |
age (AbPos-0.7–26.5y, T1D- 0.7-15y), and sex (AbPos-8F/12 M & T1D-10F/12 M), Ethnicity (AbPos-15NHW/5RND, T1D-21NHW/1RND), US (DAISY cohort) |
HLA + ., autoantibody- controls n = 25 |
Matched to HLA genotype, age (0.7-23y), sex (13F/12 M), Ethinicity (20NHW/5RND), US. (DAISY cohort) | Serum | LC-SRM-MS, (Targeted proteomics) | NA |
Pre-seroconversion: 3 (2, 1) Post-seroconversion: 2 (1, 1) |
| Webb-Robertson et al. 2023, medRxiv |
HLA + Pre-seroconversion. n = 47, Post-seroconversion. n = 131, Pre T1D n = 70, |
Age (Post-seroconversion ~ 1-23y, pre-T1D ~ 0-29y), Sex: (Pre-seroconversion-19F/28 M, Post-serconversion.- 63F/68 M & pre-T1D –31F/39 M), US (DAISY cohort) |
HLA + controls n = 40 |
Matched to HLA genotype, age (~ 0-14y), and sex (16F/24 M), US. (DAISY cohort) | Plasma | LC-SRM-MS, (Targeted proteomics) | Multiplex assay and ELISA |
Pre-seroconversion: 6 (0, 6) Post-seroconversion, Post IA: 12 (0, 12) |
| Nakayasu et al. 2023, Cell. Rep. Med |
Untargeted: IA endpoint n = 46, T1D endpoint n = 46 Targeted: IA endpoint n = 401, T1D endpoint n = 94 |
Discovery: T1D: 25F/21 M IA: 17F/29 M Validation: T1D: 43F/51 M IA: 179F/222 M (TEDDY cohort) |
Untargeted: IA Control n = 46, T1D Control n = 46 Targeted: IA Control n = 401, T1D Control n = 94 |
Matched to clinical center, gender, family history of T1D age, and HLA-DR-DQ genotypes | Plasma | 2DLC-MS/MS (Untargeted proteomics) | LC-SRM-MS (Targeted proteomics) |
Pre-seroconversion: T1: 4 (3, 1) Post-seroconversion: T2: 72 (44, 28) Pre-seroconversion, Targeted: Month-9: 22 (14, 8) Month-6: 29 (25, 4) Month-3: 25 (13, 11, 1) Post-seroconversion, Targeted: Month 0: 42 (7, 35) Month 3: 21 (6, 14, 1) Month 6: 46 (36, 9, 1) Month 9: 38 (1, 37) Month 12: 21 (10, 11) Month 15: 18 (12, 6) Month 18: 21 (8, 13) |
| Post-seroconversion | ||||||||
| Metz et al. (2008) J Proteome Res | Post-diagnosis n = 10 |
Age (< 30y) (DASP) |
HLA- controls, n = 10 |
Age (< 30y) (DASP) |
Serum & Plasma | 2DLC-MS/MS (Untargeted proteomics) | NA | Post-Diagnosis: 5 (3, 2) |
| Zhi et al. 2011, Mol. Cel. Proteom | Post-diagnosis n = 30 | Age (~ 0 to ~ 90y), US | Autoantibody-. controls, n = 30 | Age and sex Matched, US | Serum | 2-DE gel-MALDI–TOF MS (Untargeted proteomics) | Luminex and ELISA assays | Post-Diagnosis: 17 (11, 6) |
| Chen et al. 2012, J. Proteomics | Post-diagnosis n = 15 | Age and Sex not defined, Taiwan | Controls, n = 5 | Taiwan | Plasma | LC–MS/MS (Untargeted proteomics) | ELISA and Immuno-blotting | Post-Diagnosis: 36 (16, 20) |
| Zhang et al. 2013, J. Exp. Med | Post-diagnosis n = 50 |
Age (10-29y), Sex (15F/35 M), ME (DASP) |
HLA-., controls, n = 100 |
Age (18-28y), Sex (51F/49 M), ME (DASP) |
Serum & Plasma |
LC–MS/MS (Untargeted proteomics) |
LC-SRM-MS (Targeted proteomics) |
Post-Diagnosis: 24 (4, 18, 2) Post Diagnosis Targeted: 24 (8, 11, 5) |
|
Manjunatha et al. 2016, Metabolism |
T1D-PC n = 15 & T1D-GC n = 15 | Age (T1D-PC: 33.6 ± 12.97y, T1D-GC: 34.5 ± 12.48y), US | ND-PC n = 15 & ND-GC n = 15 | Matched for age, sex, and BMI, US | Serum and Plasma | LC–MS/MS (Untargeted proteomics) | NA | Post-Diagnosis: 39 (23, 16) |
| Von Toerne et al. 2017, Diabetologia | T1D family history, Post-seroconversion, rapid T1D n = 15 & slow T1D n = 15 |
Age (Rapid T1D 0.5-33y, slow T1D 9.5–17.5y), Germany (BABYDIAB/BABYDIET birth cohorts) |
T1D family history, autoantibody-. n = 15 |
Age and sex matched, Germany (BABYDIAB/BABYDIET birth cohorts) |
Serum | LC–MS/MS (Untargeted proteomics) | LC-SRM-MS (Targeted proteomics) | Post-seroconversion: 26 (13, 13) |
| do Nascimento de Oliveira et al. 2018, Diabetes Metab Syndr Obes | Post-diagnosis n = 30 | No Familiar history, Age (35.03 ± 8,6), Sex (18F/12F), Brazil | Controls n = 30 | No Familiar history, Age (31.5 ± 10.67), Sex (23F/7 M), and other clinical criteria matched, Brazil | Serum |
LC–MS/MS (Untargeted proteomics) |
NA | Post-Diagnosis: 8 (6, 2) |
| Liu et al. 2018, J. Proteomics |
HLAPos Post-seroconversion, T1D n = 11 |
Age (1-14y), 7 male and 4 female, RND (3) and NHW (8), US (DAISY cohort) |
HLA + , autoantibody- controls n = 10 |
Age 1-14y, 5 male and 5 female, RND (1) and NHW (9), US (DAISY cohort) |
Plasma | LC–MS/MS (Untargeted proteomics) | ELISA | Post-seroconversion: 12 (6, 6) |
| Gourgari et al. 2019, Cadiocasc. Diabetol | Post-Diagnosis, with high risk of cardiovascular disease, n = 26 |
12–21 years old, US |
Controls n = 13 |
Age, sex, BMI, and clinical lipid measurement matched, US |
Plasma |
LC-DIA-MS (Untargeted proteomics) |
NA | Post-Diagnosis: 8 (6, 2) |
A total of 13 studies were identified, and details regarding the various study groups, sampling, and tools for measurement and validation are listed. Italic indicates up-regulated proteins, bold indicates down-regulated proteins and bolditalic indicates conflicting detected peptide abundance. Terms used: HLA human leukocyte antigens, F Female, M Male, NHW non-Hispanic white, RND Race not defined, ME Mixed ethnicity, PC poor glycemic control, GC good glycemic control, ND non-diabetic controls, AbPos Antibody positive, y years, m months, IA Islet autoantibodies, T1D Type 1 diabetes, BMI Body mass index, NA Not applicable, DIPP Diabetes Prediction and Prevention, DAISY Diabetes Auto Immunity Study in the Young, TEDDY: The Environmental Determinants of Diabetes in the Young, DASP: Diabetes Antibody Standardization Program, ELISA enzyme-linked immunosorbent assay, LC–DIA–MS Liquid chromatography data independent-acquisition-mass spectrometry, LC–MS/MS Liquid chromatography–tandem mass spectrometry, LC–SRM–MS Liquid chromatography-selected reaction monitoring-mass spectrometry, and 2-DE gel-MALDI–TOF MS 2D gel electrophoresis matrix-assisted laser desorption/ionization time-of-flight mass spectrometry
Pre-onset proteomic profiles
Our literature search identified 6 papers that investigated the temporal protein abundance changes in individuals with T1D. Studies by Moulder et al. [7], Fronhert et al. [8], Nakayasu et al. [9], and Webb-Robertson et al. [10] looked at protein abundance changes at both pre-and post-seroconversion stages. In contrast, von Toerne et al. and Lui et al. examined the protein profile only after seroconversion [11, 12]. Moulder et al., von Toerne et al., Lui et al., and Nakayasu et al. used untargeted proteomics and identified 65, 26, 12, and 72 proteins, respectively, that were significantly different in post-seroconversion vs controls. Nakayasu et al., used targeted proteomics in plasma from individuals at -9, -6, and -3 months pre-seroconversion and 2, 6, 9, 12, 15, and 18 months post-seroconversion. Webb-Robertson et al. used targeted proteomics, investigating the expression of 19 complement proteins in plasma from pre-seroconversion, and post-seroconversion subjects, which were further validated using ELISA assays. In addition, von Toerne et al., and Fronhert et al. used targeted proteomics as a validation method to look at 3 and 5 unique proteins, respectively, whereas Liu et al. used ELISA as its validation step. In conclusion, these studies have identified many biomarker candidates, but with limited validation.
Post-diagnosis proteomic profiles
One of the first plasma/serum proteomics studies of individuals with T1D was performed by Metz et al. in 2008 [13]. They identified 5 differentially abundant proteins in recently diagnosed T1D patients compared to controls. Similarly, studies by Zhi et al. and Chen et al. utilized untargeted proteomics and identified 17 and 36 differentially abundant proteins, respectively, in sera from individuals with T1D compared to controls [14, 15]. Zhang et al. and Oliveira et al. performed untargeted proteomics of serum/plasma samples from individuals with T1D and identified 24, and 8 differentially abundant proteins, respectively [16, 17]. Zhang et al. tested the 24 proteins using targeted proteomics in 50 T1D vs. 100 controls, validating 16 proteins with high discriminating power. A subsequent blinded experiment in an independent cohort of 10 individuals with T1D and 10 controls identified the chemokine proplatelet basic factor (PPBP/CXCL7 –proteins are listed based on their gene names) and C1 inhibitor with 100% sensitivity and specificity to discriminate between the groups. Manjunatha et al. and Gourgari et al. performed untargeted proteomic analysis on high-density lipoproteins (HDL) and found a compositional but not level change of the HDL proteome in T1D individuals with a high risk of cardiovascular complications [18, 19]. Overall, these studies showed proteomic changes in plasma profiles after T1D onset, which have the potential to be developed as diagnostic biomarkers.
Potential biological functions of biomarker candidates
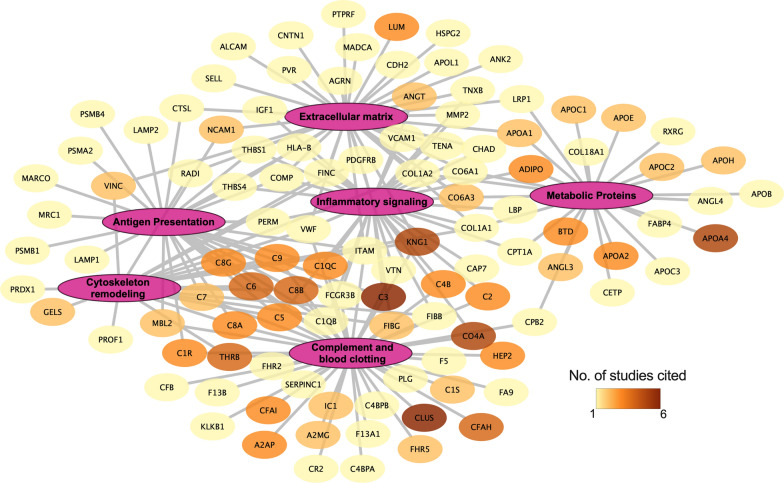
To better understand the biological relevance of these proteins, we performed a functional enrichment analysis using DAVID [6]. The KEGG annotation from DAVID mapped 157 proteins out of 266 to 26 biological pathways (Additional file 1: Table S3). Out of these pathways, the complement and coagulation pathway (44 proteins) had the most significant number of proteins mapped to it, followed by COVID-19 (23 proteins), Staphylococcus aureus infection (17 proteins), and systemic lupus erythematosus (16 proteins). These pathways were further curated down by consolidating overlapping/redundant proteins into 6 pathways, i.e., complement and coagulation, metabolic protein, inflammatory signaling, cytoskeleton remodeling, extracellular matrix, and antigen presentation (Fig. 2). Here, we further discuss these pathways in the context of T1D risk and disease evolution over time.
Fig. 2.
Pathway analysis of the protein biomarkers. This network represents proteins linked to their respective consolidated pathways from KEGG. Pathways were consolidated based on their overlap and redundancy. The nodes are colored based on the number of studies that the proteins were shown to be significantly regulated
Complement system
The complement system is a cascade of proteases making up a humoral extension of the innate immune system. Dysregulation of the complement pathway is linked to chronic and autoimmune diseases. Complement deficiencies are either inherited or acquired. Inherited deficiencies of complement proteins C1-C4 are strongly associated with bacterial infection and systemic lupus erythematosus, while inherited deficiencies of C5-C9 are associated with bacterial infection and sepsis [20]. Acquired deficiency or factor level changes arise when activation or inflammation-related acute phase responses exist, resulting in either up or downstream exhaustion of some factor [21]. They also interact with adaptive immunity by forming complexes with antibodies, including islet autoantibodies [22]. A cytotoxic effect [23] of such complexes has been controversial, but this concept has gained a renewed interest with recent discovery of β-cell surface autoantibodies [24]. In individuals with T1D, complement components C3 and C4 are highly expressed in the pancreas, including the islets [25, 26]. Studies of pancreata obtained from cadaveric T1D donors have reported C4D immunostaining in blood vessel endothelium and exocrine ducts [25], a change typically associated with activation at that site, and significant upregulation of the complement cascade (C1QA, C1QB, C1QC, C1R, C1S, C3, C4B, C5, C5AR1, C6, C7, C8A, C8B, C8G, and especially C9) [27]. Their tissue compartment localization was extrapolated from transcriptomic data but remains uncertain. This information may be of limited value for our project as the tissue or plasma status of the complement system at the time of death in a person with long-standing T1D may not resemble that at the time of the appearance of islet autoantibodies in an otherwise healthy young child.
Importantly for studies of pre-diagnosis T1D, a functional relationship has been demonstrated between activation of components C3 and C5 and improved β-cell function in mice and humans [28], suggesting direct effects on β cells. The pro-inflammatory cytokines interleukin-1β and interferon-γ increase C3 expression in rodent and human β cells [29]. C3 silencing exacerbates β-cell apoptosis. On the other hand, upregulation of the complement system improves β-cell autophagic response—a protective homeostatic response to the β-cell stress [29] that is impaired in T1D development [30]. Exogenously added C3 protects against cytokine-induced β-cell death via protein kinase B (AKT) activation and c-Jun N-terminal kinase inhibition. While locally produced C3 is an important survival mechanism in β-cells under a pro-inflammatory assault, it is not known if a C3-focused therapy could slow or abort the progression of diabetes in humans. In addition, a variant of C3 gene is associated with T1D [31], which may suggest that the response to such intervention could be genetically modified. In sum, although changes in the complement system are clearly linked to the risk of T1D development and its rapid progression, the direction of the effect and the therapeutic implications are uncertain. Thus, we need to determine whether the system should be activated or modulated, what components of the pathway are most relevant to T1D development, and at what point in the evolution of the disease should a specific change in the pathway be introduced. Regarding complement therapeutics designed to block or modulate activation, there is a range of drugs that are either available or in clinical development. For example, these therapeutics will modulate the C3 and C5 convertases, thus dampening overall activation, or be more specific to target C5, C5a, C3a, or complement receptors for activation fragments (reviewed in [32]). Conversely, activation of the pathways is being explored for the treatment of infectious diseases, cancer, and disorders of metabolism [33].
The 13 papers we examined reported some aspect of the complement system as dysregulated, with 44 out of the 266 biomarker candidates identified from our KEGG analysis (Fig. 3 and Additional file 1: Table S3). Upon closer inspection (Additional file 1: Table S2), C4 (combining C4A and C4B observations) was the most identified protein, followed by C3 in this systematic review, which was reported to be primarily downregulated in post-seroconversion and post-diagnosis compared to controls [11, 14, 16, 19]. This was further corroborated by Webb-Robertson et al., where C3 and C4 levels were consistently low in pre- and post-seroconverted subjects [10]. Interestingly, further breakdown of the disease progression by Nakayasu et. al. shows C4 levels were up until 6 months before seroconversion, with decreasing levels reported 3 months prior to autoantibody detection in patients [9]. A low abundance of C3 and C4 throughout the course of the disease was also reported by a longitudinal study conducted by Moulder et al. [34]. These results are consistent with observations of deficiencies in downstream complement components coupled with increased abundance of the Membrane attack complex (MAC) inhibitor clusterin (Clus, also known as APOJ) pre-seroconversion. However, there are conflicting reports of abundance following seroconversion. Previous ex-vivo characterization of the T1D pancreas corroborated our identification of complement dysregulation but instead found an increased abundance of complement markers following diagnosis [25, 27, 35]. Pre-clinical models of C3 knockout or receptor blockage leading to reduction in T1D development and other findings also suggest a role for the complement system in the T1D development [36–38]. While at first, the serum and tissue complement abundances appear to be at odds with one another, the low complement levels pre-seroconversion may be due to consumption through C3, C4, and MAC depositing in the pancreas throughout T1D development. Low C3 and C4 have been seen in both COVID-19 and lipodystrophy cases, and low C3 with high C5-C9 are common in glomerulopathy [39]. Evidence of lifelong complement deposits in the pancreas matching the parallel findings of lifelong low C3 levels provides further evidence of complement deposition in the pancreas as the driving force behind low blood levels of complement and, therefore, progressive loss of β cells due to increased immune response.
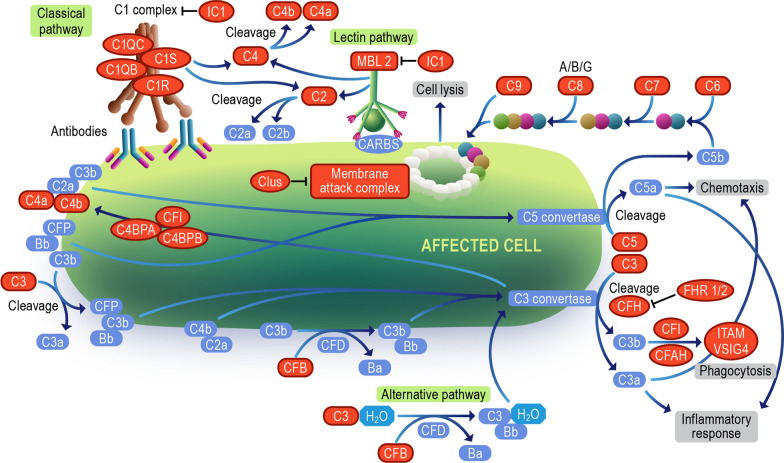
Fig. 3.
Complement cascade. The diagram represents all the complement pathway proteins denoted by orange color identified in our systematic review. The Image was modified from “Complement cascade pathway” on the Reactome website (https://reactome.org/PathwayBrowser/#/R-HSA-166658 with StableID: R-HSA-166658)
In the context of T1D, the complement system may play a part in modulating adaptive immune response against islets, with human leukocyte antigens (HLA) class II genes being associated with T1D risk [40]. Activation of the complement cascade and deposition of complement factors into the pancreas have been reported during insulitis [22, 27]. In late-stage diabetes, elevated complement levels in serum are linked to diabetic nephropathy [41]. Overall, the disruption of the complement pathway seems to be a characteristic trait of T1D, however, further studies are warranted to help navigate the path to consistent biomarker or drug development.
Immune pathways
A recurrent theme among the T1D biomarker candidates is the enrichment of proteins related to antigen processing and presentation. For instance, the complement pathway can opsonize pathogens and dead cells to be presented to the antigen-presenting cells. Other proteins related to antigen opsonization are also regulated in T1D, such as antibodies and opsonization/scavenger receptors. Once the antigen is opsonized, it is phagocyted into phagosomes. In fact, β cells infected with coxsackievirus are efficiently phagocyted by dendritic cells, making coxsackievirus infection a potential trigger of the islet autoimmune response [42, 43]. The phagocytic process requires an extensive cell cytoskeleton remodeling [44], which was another pathway enriched in our analysis. The phagosome can be next fused to lysosomes to initially process the antigens, which are further processed in the proteasome and loaded into HLA for presenting to T cells [45]. HLA alleles represent the main risk factor of T1D development, further supporting that this pathway is involved in the autoimmune response [45].
Another process that occurs in parallel is the cytokine and chemokine signaling [46]. Among the biomarker candidates, the chemokine pro-platelet basic protein/ chemokine ligand 7 (PPBP/CXCL7) has been identified along with 3 other immunoregulatory molecules (Fig. 4). Despite all the cytokines/chemokines regulated in T1D, little is known about their mechanistic roles in disease development. Cytokine/chemokine and even phagocytosis can trigger signaling cascades in the cells that further regulate these processes but also leads to the expression of other effector molecules. Among Inflammatory receptor and signaling proteins various phosphatases, phospho-binding and cytokine/growth factor proteins have been described (Fig. 4). In addition, the transcription factor TNIP1 has also been shown to be regulated in T1D (Fig. 4). Regarding the effector molecules, oxidative stress proteins myeloperoxidase, glutathione peroxidase 3, peroxiredoxin-1, and sulfhydryl oxidase 1, were also shown to be regulated in T1D. Oxidative stress has been shown to induce β-cell dysfunction and death and has also been proposed as a potential therapeutic target [47].
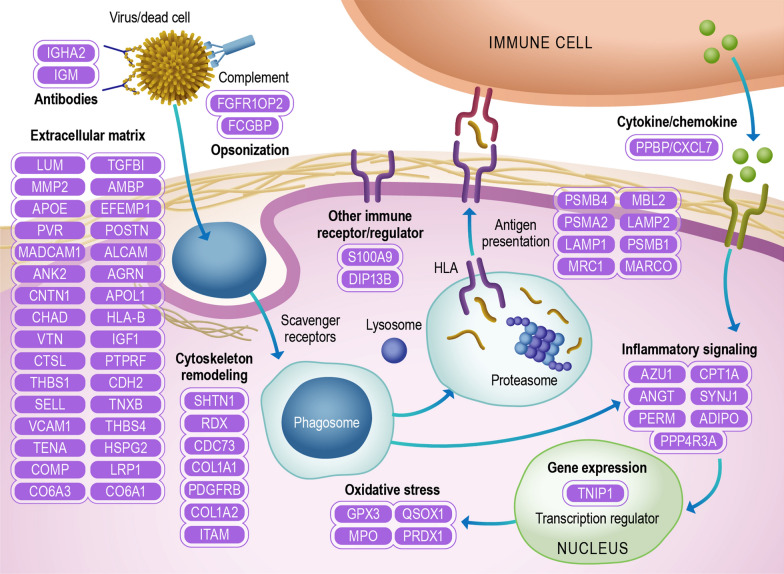
Fig. 4.
Immune pathways. The diagram represents proteins identified in the systematic review belonging to immune pathways and functions: extracellular matrix, cytoskeleton/actin filament, oxidative stress, gene expression, inflammatory signaling, antigen presentation, cytokine/chemokine, opsonization, antibodies, and other immune receptors/regulators. Proteins were annotated into different pathways with DAVID and by their function description in UniProt. Proteins are listed using their UniProt gene names
A common feature in the plasma of individuals with T1D is the regulation of extracellular matrix proteins, of which 34 have been described to be regulated (Fig. 4). The extracellular matrix is an integral part of the immune response regulated by cytokines and chemokines [48, 49]. For instance, the extracellular matrix peri-islet basement membrane serves as a barrier, protecting islets from immune cell infiltration in insulitis in mouse models of T1D [50]. In addition, preservation of the extracellular matrix by administration of dextran sulfate, a mimic molecule of the extracellular matrix proteoglycans, has been shown to protect β cells and to be a potential treatment for T1D in mice [51].
Plasma lipoproteins
The DAVID analysis showed ~ 5% of the reported proteins to be key players in lipid metabolism. Most circulating plasma lipids are packaged into lipoproteins which are traditionally classified into four common subfractions based on particle density: chylomicrons (CM), very low-density lipoproteins (VLDL), low-density lipoproteins (LDL), and high-density lipoproteins (HDL). Structurally dynamic apolipoprotein scaffolds reside at the water–lipid interface of all subclasses where they modulate particle interactions with plasma enzymes, cofactors, and cell surface receptors that continuously remodel the lipoproteins throughout their lifespan. Though traditionally defined based on their “cholesterol” content, proteomics studies over the last decade have revealed significant compositional heterogeneity exists within the lipoprotein subfractions which contain upwards of 273 different proteins [52] with the HDL subfraction accounting for > 250 of these proteins. Thus, lipoproteins are thought to consist of a variety of compositionally distinct subspecies which have now been shown to modulate a diverse array of metabolic pathways [53, 54].
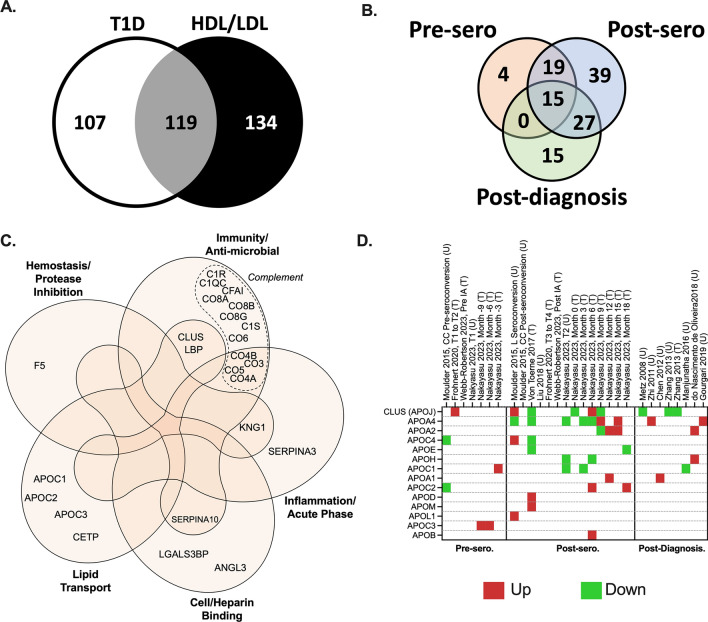
Though DAVID analysis implicated “cholesterol metabolism,” and by proxy lipoproteins, the analysis fails to capture the full lipoproteome due to the recency of lipoprotein molecular profiling studies in the literature. When compared to a lipoprotein-specific database [52], we found nearly half (119 proteins) of the protein biomarkers identified in individuals with T1D are associated with lipoproteins (Fig. 5A, Additional file 1: Table S2—Common T1D/LP proteins column), out of which 73 were specifically associated to HDL functions (Additional file 1: Table S2—T1D proteins with known HDL function column). Approximately 68% of changes in protein levels were unique to post-seroconversion and post-diagnosis indicating the most profound changes in lipoprotein metabolism occur later in the disease process (Fig. 5B). A total of 38 members were altered pre-seroconversion with most overlapping with previously discussed immune response and complement cascade (Fig. 5C). Outside of the immune and complement proteins, we noted a few well-studied apolipoprotein APOCs and clusterin were altered pre-seroconversion (Fig. 5D), hinting some changes occur in the lipoproteome prior to the onset of dysglycemia or hyperglycemia.
Fig. 5.
Overlap of T1D-relevant proteins with high- (HDL)/ low- (LDL) density lipoproteome. A Venn diagram showing common protein hits (grey) between the HDL/LDL proteome [52] (black) and those reported in our systematic review (white). B Venn diagram detailing the T1D stage as pre-seroconversion (pre-sero), post-seroconversion (post-sero) and post-diagnosis of the 119 HDL/LDL/T1D shared proteins from A. C Gene Ontology of HDL lipoprotein-known functions of pre-seroconversion reported proteins. D Heatmap of apolipoproteins with altered levels reported in at least one stage of T1D development. Up (Red color) are upregulated proteins and down (Green color) are downregulated proteins. Proteins are named using their UniProt gene names. Panel C was modified from a published figure by Davidson et al. (copyright permission was obtained from the publisher, license number: 5433910818199) [52]
Most of the lipoproteome members altered post-seroconversion and post-diagnosis have documented roles in triacylglycerol metabolism. Perhaps the most robust of these observations were associated with changes in plasma APOA4 and clusterin altered in 5 studies post-seroconversion and post-diagnosis [7, 11, 14, 18, 19]. APOA4 is well-documented to modulate the triacylglycerol packaging in the triacylglycerol-rich lipoproteins (CMs and VLDL). [55]. Additionally, APOA4 is reported to play key roles in satiety, gastric function, and glucose homeostasis [56, 57], all of which have been reported altered in individuals with T1D [57–61]. Clusterin is known to affect insulin signaling and inflammation [62, 63]. High plasma levels of clusterin have been reported in pre and diabetic patients, regarded as a marker for diabetes [64]. Five post-seroconversion and post-diagnosis studies reported increased plasma APOA2; a well-known HDL scaffold protein. While HDL has little triacylglycerol, APOA2 has been shown to be implicated in triacylglycerol metabolism [65] through a mechanism that is still poorly understood. Several studies report changes in the APOCs and APOE [7, 11, 18] which are also thought to modulate triacylglycerol lipolysis in VLDLs [66, 67]. These observations are in-line with elevated triacylglycerol levels and inhibition of lipoprotein lipase associated with the innate immune response [68].
Most of the biomarker studies were performed on whole plasma. As most apolipoproteins are exchangeable, more detailed lipoprotein speciation studies are required to determine the subclass on which these particles are located and how they are modulating particle function in the context of T1D. Though two studies attempted to speciate lipoproteins from individuals with T1D, both limited their analysis to HDLs (21, 22) thus missing most of the changes involved with the triacylglycerol-rich particles. Future studies that examine the temporal changes in the lipoproteome across all subclasses will better inform on the role of these pleiotropic particles and how they cross-communicate with the complement system and immune pathways in T1D.
Overview of the most promising biomarker candidates for clinical use
For clinical use, biomarkers need to be highly sensitive and specific. As T1D is a chronic disease in which each stage can take months to years, biomarkers with more stable changes in abundance are preferable to proteins with transient regulation. In addition, they need to be reproducible. Therefore, proteins with consistent abundance profiles across different studies would make them stronger candidates to be developed into clinical assays. Our systematic review identified 266 proteins, 80 (30%) were found across multiple papers, and 31 (11.6%) were reported three or more times (Additional file 1: Table S2 & Fig. 6). The differential expression patterns were more limited in the pre-seroconversion than post-seroconversion, with most differentially expressed proteins in the post-diagnosis group. We found 198 proteins that were only present in post-seroconversion and post-diagnosis stages compared to pre-seroconversion. From those, proteins such as BTD, ITIH2, ALS, IBP3, SAA4 were consistently upregulated in post-seroconversion and APOA4 in post-diagnosis stages, observed in ≥ 2 articles. Similarly, proteins such as IBP2, APOE, C1S, C6A3, PI16, and TETN were consistently downregulated in post-seroconversion and C4A in post-diagnosis stages. Out of the remaining 68 proteins that were detected in the pre-seroconversion stage, 38 proteins were reported as increased abundance (by < 2 articles) and 19 had decreased abundance (by < 2 articles), and 11 had conflicting data (by at least 2 articles). From the 38 proteins that were up in the pre-seroconversion group, only the C8G protein was found to be up in post-seroconversion, whereas, SEPP1 and ITIH1 were found to be down in post-seroconversion along with CLUS in post-diagnosis stages (by ≥ 2 articles). Similarly, out of 19 downregulated proteins in the pre-seroconversion group, C1R, and C4B were only downregulated in post-seroconversion and C3 was found to be downregulated in the post-seroconversion and post-diagnosis stages by ≥ 2 articles. Due to the lack of studies looking at the preseroconversion stage in patients, none of the proteins in this stage were corroborated by multiple independent studies, however, there were 17 proteins (C3, C1R, C8G, C4B, IBP2, IBP3, ITIH1, ITIH2, BTD, APOE, TETN, C1S, C6A3, SAA4, ALS, SEPP1 & PI16) in post-seroconversion group and 3 proteins (C3, CLUS, and C4A) in the post-diagnosis group that had consistent regulation in at least 2 independent studies. Upon evaluating just the directionality (i.e., up and down) of the proteins across all three temporal groups (pre-seroconversion, post-seroconversion, and post-diagnosis), C3 was found to be consistently downregulated, and C8G and ITIH3 were upregulated in cases compared to control (these proteins were observed by only one article in pre-seroconversion stage, but multiple articles in the other stages), making them and along with other proteins mentioned above as strong candidates for clinical assay development.
Fig. 6.
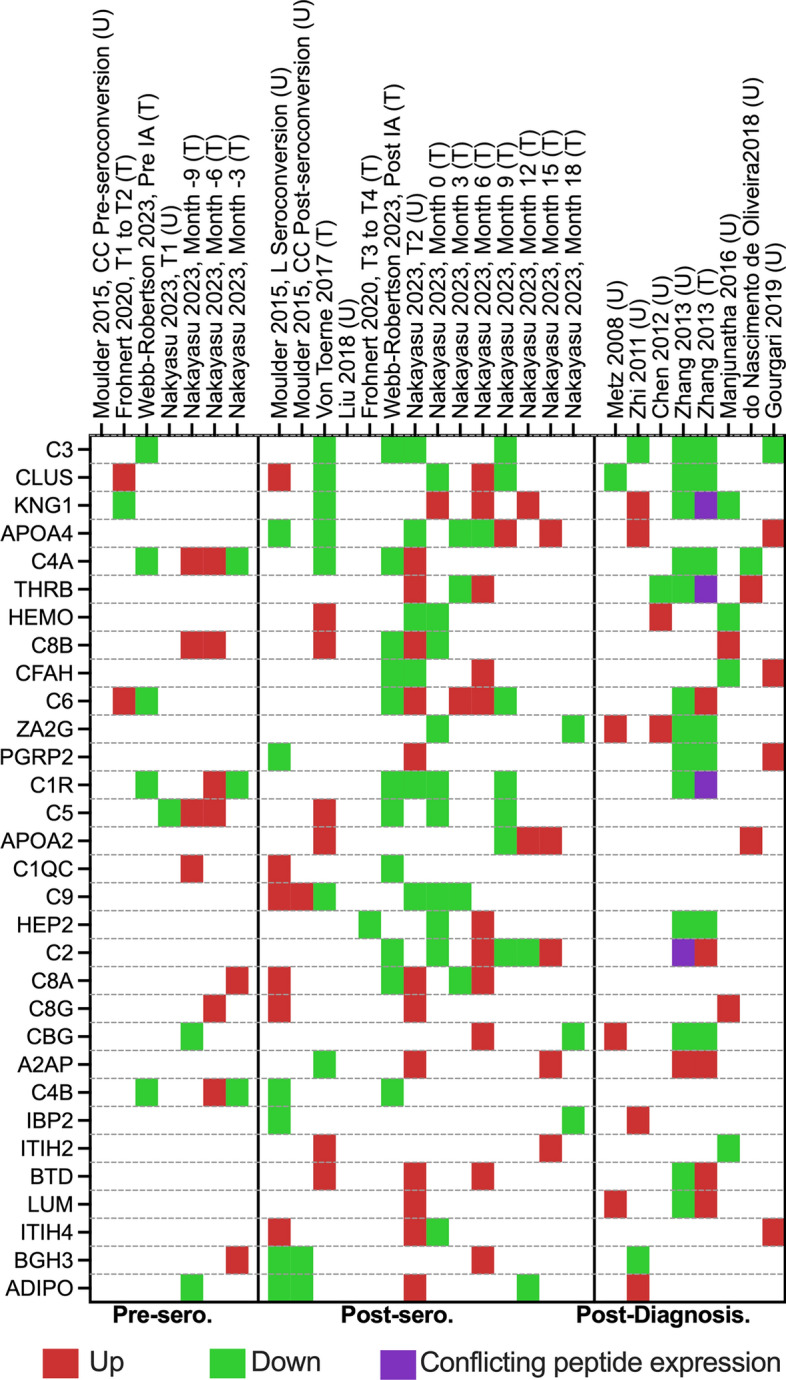
Potential biomarkers list. It is a heatmap of all the protein biomarkers identified by multiple proteomic papers (3 or more times). Proteins that the studies have not reported are represented as blank. “T” denotes the targeted proteomic approach, whereas “U” denotes the untargeted proteomics. Proteins are listed based on their gene names. Down—significantly downregulated proteins, sero. seroconversion, Up—significantly upregulated proteins
Limitations of the study
In this study, we compare data across different mass spectrometry-based proteomics approaches, which limits the ways that we can compare across datasets. For instance, isobaric labeling experiments, such as TMT and iTRAQ, have the issue of fold change compression [69]. Therefore, we only compare the directionality of the protein regulation, which lacks the information on how strong the regulation is. However, a protein being detected as differentially abundant across different platforms can arguably be considered a better biomarker candidate because of the differences in measurement errors and possible biases across platforms. Another limitation is that studies often report only significantly changing proteins. Therefore, important information that could clarify if a protein is not significant in a different study is lost. An inherited limitation/challenge is that clinical samples are not synchronized. Therefore, some of the ephemeral changes in the protein abundances would unlike to be cross-validated between studies. However, those proteins might still bring invaluable information about the disease process, even though they are poor biomarker candidates.
Conclusions and perspectives
Our systematic analysis found 266 candidate protein biomarkers of T1D, of which 80 (30%) were observed in multiple studies. This helps to prioritize the validation step of biomarker development. Despite some of the biomarkers being consistently regulated across different studies, they still need to go through an extensive validation process before moving to clinical assay development. Ideally, the biomarker candidates should be cross-validated in independent cohorts of samples and tested for sensitivity and specificity. In general, fewer signatures were identified prior to the onset of the disease. This can be partly because T1D has an almost silent developmental phase, and it is not expected that significant biochemical changes would be observed in the blood of these individuals. Alternatively, the pre-seroconversion phase may be convoluted by multiple factors, temporal regulations, and trajectories that lead to autoimmunity and hinder our ability to identify a consistent signature. In this context, machine learning can be an excellent approach to identifying multivariate panels of proteins to serve as biomarkers of T1D development. This approach has been used to combine metabolic, genetic, and autoimmune signatures to predict the onset of disease and can be easily adapted to test peptide/protein panels [8, 70–72]. Another concept that can further improve biomarkers' robustness is using ratio between protein abundance changes rather than profiling individual proteins. Ratios between oppositely regulated proteins would have much bigger differences compared to them individually, providing higher discriminatory power between cases and controls. After selecting candidates, clinical-grade assays must be developed and tested for robustness, specificity, and sensitivity in the clinical setting. In addition to biomarkers, our systematic review of proteomics studies provided insights into the pathways regulated in T1D, such as complement system, plasma lipoproteins, and immune response [70, 71]. Our systematic review also opens opportunities to study the functions of the biomarker candidates in T1D development and pathology. For instance, our group has found that PPBP/CXCL7 can reduce pro-inflammatory cytokine-mediated apoptosis in macrophage cell cultures while it enhances it in cultured β cells [73]. This may have a role in T1D development by potentiating macrophages and killing β cells in insulitis. Overall, this systematic review provides insights into processes regulated in T1D development and highlights some of the best candidates for developing clinical assays.
Supplementary Information
Additional file 1: Table S1. Studies analyzed in this systematic review. Table S2. Summary of the biomarker candidates identified across different studies. Table S3. Functional-enrichment analysis of type 1 diabetes biomarker candidates.
Acknowledgements
The authors thank Mr. Nathan Johnson of PNNL for his help drafting figures.
Abbreviations
- CM
Chylomicrons
- DAVID
Database for Annotation Visualization and Integrated Discovery
- HDL
High-density lipoproteins
- KEGG
Kyoto Encyclopedia of Genes and Genomes
- LDL
Low-density lipoprotein
- PPBP
Proplatelet basic protein
- T1D
Type 1 diabetes
- VLDL
Very low-density lipoprotein
Author contributions
Concept and idea: MJR, ESN, & TOM; Systematic review of the literature: SS, ECE, & HRH; Analysis: All authors; Writing: SS, ECE, HRH, IDL, JTM, & ESN; All authors read and approved the final version of the manuscript.
Funding
The Leona M. and Harry B. Helmsley Charitable Trust (to M.R.). National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases grants U01 DK127786 (to T.O.M. and B-J.W-R.) and U01 DK127505 (to E.S.N). Human Islet Research Network (to E.S.N).
Availability of data and materials
Supplementary material along with registration details are electronically available at Open Science Framework (OSF) website https://osf.io/n8tsa/.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Yu L, Zhao Z, Steck AK. T1D Autoantibodies: room for improvement? Curr Opin Endocrinol Diabetes Obes. 2017;24(4):285–291. doi: 10.1097/MED.0000000000000348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jacobsen LM, Bocchino L, Evans-Molina C, et al. The risk of progression to type 1 diabetes is highly variable in individuals with multiple autoantibodies following screening. Diabetologia. 2020;63(3):588–596. doi: 10.1007/s00125-019-05047-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nakayasu ES, Gritsenko M, Piehowski PD, et al. Tutorial: best practices and considerations for mass-spectrometry-based protein biomarker discovery and validation. Nat Protoc. 2021;16(8):3737–3760. doi: 10.1038/s41596-021-00566-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.MacLean E, Broger T, Yerlikaya S, Fernandez-Carballo BL, Pai M, Denkinger CM. A systematic review of biomarkers to detect active tuberculosis. Nat Microbiol. 2019;4(5):748–758. doi: 10.1038/s41564-019-0380-2. [DOI] [PubMed] [Google Scholar]
- 5.Shamseer L, Moher D, Clarke M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ. 2015 doi: 10.1136/bmj.g7647. [DOI] [PubMed] [Google Scholar]
- 6.da Huang W, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009;4(1):44–57. doi: 10.1038/nprot.2008.211. [DOI] [PubMed] [Google Scholar]
- 7.Moulder R, Bhosale SD, Erkkila T, et al. Serum proteomes distinguish children developing type 1 diabetes in a cohort with HLA-conferred susceptibility. Diabetes. 2015;64(6):2265–2278. doi: 10.2337/db14-0983. [DOI] [PubMed] [Google Scholar]
- 8.Frohnert BI, Webb-Robertson BJ, Bramer LM, et al. Predictive modeling of type 1 diabetes stages using disparate data sources. Diabetes. 2020;69(2):238–248. doi: 10.2337/db18-1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nakayasu ES, Bramer LM, Ansong C, et al. Plasma protein biomarkers predict the development of persistent autoantibodies and type 1 diabetes 6 months prior to the onset of autoimmunity. Cell Rep Med. 2023;4(7):101093. doi: 10.1016/j.xcrm.2023.101093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Webb-Robertson B-JM, Nakayasu ES, Dong F, et al. Decrease in multiple complement protein levels is associated with the development of islet autoimmunity and type 1 diabetes. MedRxiv. 2023 doi: 10.1101/2023.07.13.23292628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.von Toerne C, Laimighofer M, Achenbach P, et al. Peptide serum markers in islet autoantibody-positive children. Diabetologia. 2017;60(2):287–295. doi: 10.1007/s00125-016-4150-x. [DOI] [PubMed] [Google Scholar]
- 12.Liu CW, Bramer L, Webb-Robertson BJ, Waugh K, Rewers MJ, Zhang Q. Temporal expression profiling of plasma proteins reveals oxidative stress in early stages of Type 1 Diabetes progression. J Proteomics. 2018;172:100–110. doi: 10.1016/j.jprot.2017.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Metz TO, Qian WJ, Jacobs JM, et al. Application of proteomics in the discovery of candidate protein biomarkers in a diabetes autoantibody standardization program sample subset. J Proteome Res. 2008;7(2):698–707. doi: 10.1021/pr700606w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhi W, Sharma A, Purohit S, et al. Discovery and validation of serum protein changes in type 1 diabetes patients using high throughput two dimensional liquid chromatography-mass spectrometry and immunoassays. Mol Cell Proteomics. 2011 doi: 10.1074/mcp.M111.012203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chen CC, Lu YC, Chen YW, et al. Hemopexin is up-regulated in plasma from type 1 diabetes mellitus patients: Role of glucose-induced ROS. J Proteomics. 2012;75(12):3760–3777. doi: 10.1016/j.jprot.2012.04.047. [DOI] [PubMed] [Google Scholar]
- 16.Zhang Q, Fillmore TL, Schepmoes AA, et al. Serum proteomics reveals systemic dysregulation of innate immunity in type 1 diabetes. J Exp Med. 2013;210(1):191–203. doi: 10.1084/jem.20111843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.do Nascimento de Oliveira V, Lima-Neto ABM, van Tilburg MF, et al. Proteomic analysis to identify candidate biomarkers associated with type 1 diabetes. Diabetes Metab Syndr Obes 2018:11: 289-301. 10.2147/dmso.S162008 [DOI] [PMC free article] [PubMed]
- 18.Manjunatha S, Distelmaier K, Dasari S, Carter RE, Kudva YC, Nair KS. Functional and proteomic alterations of plasma high density lipoproteins in type 1 diabetes mellitus. Metabolism. 2016;65(9):1421–1431. doi: 10.1016/j.metabol.2016.06.008. [DOI] [PubMed] [Google Scholar]
- 19.Gourgari E, Ma J, Playford MP, et al. Proteomic alterations of HDL in youth with type 1 diabetes and their associations with glycemic control: a case-control study. Cardiovasc Diabetol. 2019;18(1):43. doi: 10.1186/s12933-019-0846-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Botto M, Kirschfink M, Macor P, Pickering MC, Wurzner R, Tedesco F. Complement in human diseases: Lessons from complement deficiencies. Mol Immunol. 2009;46(14):2774–2783. doi: 10.1016/j.molimm.2009.04.029. [DOI] [PubMed] [Google Scholar]
- 21.Ekdahl KN, Persson B, Mohlin C, Sandholm K, Skattum L, Nilsson B. Interpretation of serological complement biomarkers in disease. Front Immunol. 2018;9:2237. doi: 10.3389/fimmu.2018.02237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Radillo O, Nocera A, Leprini A, et al. Complement-fixing islet cell antibodies in type-1 diabetes can trigger the assembly of the terminal complement complex on human islet cells and are potentially cytotoxic. Clin Immunol Immunopathol. 1996;79(3):217–223. doi: 10.1006/clin.1996.0071. [DOI] [PubMed] [Google Scholar]
- 23.Hehmke B, Michaelis D, Hildmann W, Richter KV, Kohnert KD. Cytotoxic islet cell autoantibodies in newly diagnosed insulin-dependent diabetes mellitus: lack of correlation to age, residual beta cell function, HLA antigens and Coxsackie B virus antibodies. Diabetes Res. 1987;4(2):73–77. [PubMed] [Google Scholar]
- 24.Jia X, Gu Y, High H, Yu L. Islet autoantibodies in disease prediction and pathogenesis. Diabetol Int. 2020;11(1):6–10. doi: 10.1007/s13340-019-00414-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rowe P, Wasserfall C, Croker B, et al. Increased complement activation in human type 1 diabetes pancreata. Diabetes Care. 2013;36(11):3815–3817. doi: 10.2337/dc13-0203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.King BC, Kulak K, Krus U, et al. Complement component C3 is highly expressed in human pancreatic islets and prevents beta cell death via atg16l1 interaction and autophagy regulation. Cell Metab. 2019;29(1):202–210.e206. doi: 10.1016/j.cmet.2018.09.009. [DOI] [PubMed] [Google Scholar]
- 27.Woo J, Sudhir PR, Zhang Q. Pancreatic tissue proteomics unveils key proteins, pathways, and networks associated with type 1 diabetes. Proteom Clin App. 2020;14(6):e2000053. doi: 10.1002/prca.202000053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Atanes P, Ruz-Maldonado I, Pingitore A, et al. C3aR and C5aR1 act as key regulators of human and mouse beta-cell function. Cell Mol Life Sci. 2018;75(4):715–726. doi: 10.1007/s00018-017-2655-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Dos Santos RS, Marroqui L, Grieco FA, et al. Protective role of complement C3 against cytokine-mediated β-cell apoptosis. Endocrinology. 2017;158(8):2503–2521. doi: 10.1210/en.2017-00104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Muralidharan C, Conteh AM, Marasco MR, et al. Pancreatic beta cell autophagy is impaired in type 1 diabetes. Diabetologia. 2021;64(4):865–877. doi: 10.1007/s00125-021-05387-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Torn C, Liu X, Hagopian W, et al. Complement gene variants in relation to autoantibodies to beta cell specific antigens and type 1 diabetes in the TEDDY Study. Sci Rep. 2016;6:27887. doi: 10.1038/srep27887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ricklin D, Mastellos DC, Reis ES, Lambris JD. The renaissance of complement therapeutics. Nat Rev Nephrol. 2018;14(1):26–47. doi: 10.1038/nrneph.2017.156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.West EE, Afzali B, Kemper C. Unexpected Roles for Intracellular Complement in the Regulation of Th1 Responses. Adv Immunol. 2018;138:35–70. doi: 10.1016/bs.ai.2018.02.001. [DOI] [PubMed] [Google Scholar]
- 34.Moulder R, Bhosale SD, Erkkilä T, et al. Serum proteomes distinguish children developing type 1 diabetes in a cohort with HLA-conferred susceptibility. Diabetes. 2015;64(6):2265–2278. doi: 10.2337/db14-0983. [DOI] [PubMed] [Google Scholar]
- 35.Planas R, Carrillo J, Sanchez A, et al. Gene expression profiles for the human pancreas and purified islets in type 1 diabetes: new findings at clinical onset and in long-standing diabetes. Clin Exp Immunol. 2010;159(1):23–44. doi: 10.1111/j.1365-2249.2009.04053.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gao X, Liu H, He B, Fu Z. Resistance to streptozotocin-induced autoimmune diabetes in absence of complement C3: myeloid-derived suppressor cells play a role. PLoS ONE. 2013;8(6):e66334. doi: 10.1371/journal.pone.0066334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Noorchashm H, Moore DJ, Lieu YK, et al. Contribution of the innate immune system to autoimmune diabetes: a role for the CR1/CR2 complement receptors. Cell Immunol. 1999;195(1):75–79. doi: 10.1006/cimm.1999.1522. [DOI] [PubMed] [Google Scholar]
- 38.Axelgaard E, Ostergaard JA, Thiel S, Hansen TK. Diabetes is associated with increased autoreactivity of mannan-binding lectin. J Diabetes Res. 2017;2017:6368780. doi: 10.1155/2017/6368780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Medjeral-Thomas NR, O'Shaughnessy MM, O'Regan JA, et al. C3 glomerulopathy: clinicopathologic features and predictors of outcome. Clin J Am Soc Nephrol. 2014;9(1):46–53. doi: 10.2215/CJN.04700513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nejentsev S, Howson JM, Walker NM, et al. Localization of type 1 diabetes susceptibility to the MHC class I genes HLA-B and HLA-A. Nature. 2007;450(7171):887–892. doi: 10.1038/nature06406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Flyvbjerg A. The role of the complement system in diabetic nephropathy. Nat Rev Nephrol. 2017;13(5):311–318. doi: 10.1038/nrneph.2017.31. [DOI] [PubMed] [Google Scholar]
- 42.Schulte BM, Kramer M, Ansems M, et al. Phagocytosis of enterovirus-infected pancreatic beta-cells triggers innate immune responses in human dendritic cells. Diabetes. 2010;59(5):1182–1191. doi: 10.2337/db09-1071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lloyd RE, Tamhankar M, Lernmark A. Enteroviruses and type 1 diabetes: multiple mechanisms and factors? Annu Rev Med. 2022;73:483–499. doi: 10.1146/annurev-med-042320-015952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Mylvaganam S, Freeman SA, Grinstein S. The cytoskeleton in phagocytosis and macropinocytosis. Curr Biol. 2021;31(10):R619–R632. doi: 10.1016/j.cub.2021.01.036. [DOI] [PubMed] [Google Scholar]
- 45.Blander JM, Medzhitov R. On regulation of phagosome maturation and antigen presentation. Nat Immunol. 2006;7(10):1029–1035. doi: 10.1038/ni1006-1029. [DOI] [PubMed] [Google Scholar]
- 46.Eizirik DL, Colli ML, Ortis F. The role of inflammation in insulitis and beta-cell loss in type 1 diabetes. Nat Rev Endocrinol. 2009;5(4):219–226. doi: 10.1038/nrendo.2009.21. [DOI] [PubMed] [Google Scholar]
- 47.Dinic S, Arambasic Jovanovic J, Uskokovic A, et al. Oxidative stress-mediated beta cell death and dysfunction as a target for diabetes management. Front Endocrinol (Lausanne) 2022;13:1006376. doi: 10.3389/fendo.2022.1006376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Morwood SR, Nicholson LB. Modulation of the immune response by extracellular matrix proteins. Arch Immunol Ther Exp (Warsz) 2006;54(6):367–374. doi: 10.1007/s00005-006-0043-x. [DOI] [PubMed] [Google Scholar]
- 49.Sorokin L. The impact of the extracellular matrix on inflammation. Nat Rev Immunol. 2010;10(10):712–723. doi: 10.1038/nri2852. [DOI] [PubMed] [Google Scholar]
- 50.Korpos E, Kadri N, Kappelhoff R, et al. The peri-islet basement membrane, a barrier to infiltrating leukocytes in type 1 diabetes in mouse and human. Diabetes. 2013;62(2):531–542. doi: 10.2337/db12-0432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lu G, Rausell-Palamos F, Zhang J, et al. Dextran sulfate protects pancreatic beta-cells, reduces autoimmunity, and ameliorates type 1 diabetes. Diabetes. 2020;69(8):1692–1707. doi: 10.2337/db19-0725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Davidson WS, Shah AS, Sexmith H, et al. The HDL proteome watch: compilation of studies leads to new insights on HDL function. Biochim Biophys Acta Mol Cell Biol Lipids. 2022;1867(2):159072. doi: 10.1016/j.bbalip.2021.159072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Gordon SM, Remaley AT. High density lipoproteins are modulators of protease activity: Implications in inflammation, complement activation, and atherothrombosis. Atherosclerosis. 2017;259:104–113. doi: 10.1016/j.atherosclerosis.2016.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Davidson WS, Cooke AL, Swertfeger DK, Shah AS. The difference between high density lipoprotein subfractions and subspecies: an evolving model in cardiovascular disease and diabetes. Curr Atheroscler Rep. 2021;23(6):23. doi: 10.1007/s11883-021-00925-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wang F, Kohan AB, Lo CM, Liu M, Howles P, Tso P. Apolipoprotein A-IV: a protein intimately involved in metabolism. J Lipid Res. 2015;56(8):1403–1418. doi: 10.1194/jlr.R052753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Li X, Xu M, Wang F, et al. Apolipoprotein A-IV reduces hepatic gluconeogenesis through nuclear receptor NR1D1. J Biol Chem. 2014;289(4):2396–2404. doi: 10.1074/jbc.M113.511766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kohan AB, Wang F, Lo CM, Liu M, Tso P. ApoA-IV: current and emerging roles in intestinal lipid metabolism, glucose homeostasis, and satiety. Am J Physiol Gastrointest Liver Physiol. 2015;308(6):G472–481. doi: 10.1152/ajpgi.00098.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.VerHague MA, Cheng D, Weinberg RB, Shelness GS. Apolipoprotein A-IV expression in mouse liver enhances triglyceride secretion and reduces hepatic lipid content by promoting very low density lipoprotein particle expansion. Arterioscler Thromb Vasc Biol. 2013;33(11):2501–2508. doi: 10.1161/ATVBAHA.113.301948. [DOI] [PubMed] [Google Scholar]
- 59.Culnan DM, Cooney RN, Stanley B, Lynch CJ. Apolipoprotein A-IV, a putative satiety/antiatherogenic factor, rises after gastric bypass. Obesity (Silver Spring) 2009;17(1):46–52. doi: 10.1038/oby.2008.428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wang F, Kohan AB, Kindel TL, et al. Apolipoprotein A-IV improves glucose homeostasis by enhancing insulin secretion. Proc Natl Acad Sci U S A. 2012;109(24):9641–9646. doi: 10.1073/pnas.1201433109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Fraser RJ, Horowitz M, Maddox AF, Harding PE, Chatterton BE, Dent J. Hyperglycaemia slows gastric emptying in type 1 (insulin-dependent) diabetes mellitus. Diabetologia. 1990;33(11):675–680. doi: 10.1007/BF00400569. [DOI] [PubMed] [Google Scholar]
- 62.Wittwer J, Bradley D. Clusterin and its role in insulin resistance and the cardiometabolic syndrome. Front Immunol. 2021;12:612496. doi: 10.3389/fimmu.2021.612496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Shim YJ, Kang BH, Jeon HS, et al. Clusterin induces matrix metalloproteinase-9 expression via ERK1/2 and PI3K/Akt/NF-kappaB pathways in monocytes/macrophages. J Leukoc Biol. 2011;90(4):761–769. doi: 10.1189/jlb.0311110. [DOI] [PubMed] [Google Scholar]
- 64.Ha J, Moon MK, Kim H, et al. Plasma clusterin as a potential link between diabetes and Alzheimer disease. J Clin Endocrinol Metab. 2020 doi: 10.1210/clinem/dgaa378. [DOI] [PubMed] [Google Scholar]
- 65.Desai NK, Ooi EM, Mitchell PD, Furtado J, Sacks FM. Metabolism of apolipoprotein A-II containing triglyceride rich ApoB lipoproteins in humans. Atherosclerosis. 2015;241(2):326–333. doi: 10.1016/j.atherosclerosis.2015.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kowal RC, Herz J, Weisgraber KH, Mahley RW, Brown MS, Goldstein JL. Opposing effects of apolipoproteins E and C on lipoprotein binding to low density lipoprotein receptor-related protein. J Biol Chem. 1990;265(18):10771–10779. doi: 10.1016/S0021-9258(18)87014-4. [DOI] [PubMed] [Google Scholar]
- 67.Ramms B, Patel S, Nora C, et al. ApoC-III ASO promotes tissue LPL activity in the absence of apoE-mediated TRL clearance. J Lipid Res. 2019;60(8):1379–1395. doi: 10.1194/jlr.M093740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Gao Y, Vidal-Itriago A, Kalsbeek MJ, et al. Lipoprotein lipase maintains microglial innate immunity in obesity. Cell Rep. 2017;20(13):3034–3042. doi: 10.1016/j.celrep.2017.09.008. [DOI] [PubMed] [Google Scholar]
- 69.Ow SY, Salim M, Noirel J, Evans C, Rehman I, Wright PC. iTRAQ underestimation in simple and complex mixtures: “The Good, the Bad and the Ugly”. J Proteome Res. 2009;8(11):5347–5355. doi: 10.1021/pr900634c. [DOI] [PubMed] [Google Scholar]
- 70.Webb-Robertson BM, Nakayasu ES, Frohnert BI, et al. Integration of infant metabolite, genetic, and islet autoimmunity signatures to predict type 1 diabetes by age 6 years. J Clin Endocrinol Metab. 2022;107(8):2329–2338. doi: 10.1210/clinem/dgac225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Webb-Robertson BM, Bramer LM, Stanfill BA, et al. Prediction of the development of islet autoantibodies through integration of environmental, genetic, and metabolic markers. J Diabetes. 2021;13(2):143–153. doi: 10.1111/1753-0407.13093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Nakayasu ES, Bramer LM, Ansong C, et al. Plasma protein biomarkers predict both the development of persistent autoantibodies and type 1 diabetes 6 months prior to the onset of autoimmunity: the TEDDY Study. MedRxiv. 2022 doi: 10.1101/2022.12.07.22283187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Vallejo MC, Sarkar S, Elliott EC, et al. Meta-analysis refinement of plasma extracellular vesicle composition identifies proplatelet basic protein as a signaling messenger in type 1 diabetes. BioRxiv. 2022 doi: 10.1101/2022.09.28.509996. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. Studies analyzed in this systematic review. Table S2. Summary of the biomarker candidates identified across different studies. Table S3. Functional-enrichment analysis of type 1 diabetes biomarker candidates.
Data Availability Statement
Supplementary material along with registration details are electronically available at Open Science Framework (OSF) website https://osf.io/n8tsa/.