Abstract
Background
Anopheles funestus, the main malaria vector, prefer to oviposit in permanent and/or semi-permanent breeding habitats located far from human dwellings. Difficulties in identifying and accessing these habitats jeopardize the feasibility of conventional larviciding. In this way, a semi-field study was conducted to assess the potential of autodissemination of pyriproxyfen (PPF) by An. funestus for its control.
Methods
The study was conducted inside a semi-field system (SFS). Therein, two identical separate chambers, the treatment chamber with a PPF-treated clay pot (0.25 g AI), and the control chamber with an untreated clay pot. In both chambers, one artificial breeding habitat made of a plastic basin with one litre of water was provided. Three hundred blood-fed female An. funestus aged 5–9 days were held inside untreated and treated clay pots for 30 min and 48 h before being released for oviposition. The impact of PPF on adult emergence, fecundity, and fertility through autodissemination and sterilization effects were assessed by comparing the treatment with its appropriate control group.
Results
Mean (95% CI) percentage of adult emergence was 15.5% (14.9–16.1%) and 70.3% (69–71%) in the PPF and control chamber for females exposed for 30 min (p < 0.001); and 19% (12–28%) and 95% (88–98%) in the PPF and control chamber for females exposed for 48 h (p < 0.001) respectively. Eggs laid by exposed mosquitoes and their hatch rate were significantly reduced compared to unexposed mosquitoes (p < 0.001). Approximately, 90% of females exposed for 48 h retained abnormal ovarian follicles and only 42% in females exposed for 30 min.
Conclusion
The study demonstrated sterilization and adult emergence inhibition via autodissemination of PPF by An. funestus. Also, it offers proof that sterilized An. funestus can transfer PPF to prevent adult emergence at breeding habitats. These findings warrant further assessment of the autodissemination of PPF in controlling wild population of An. funestus, and highlights its potential for complementing long-lasting insecticidal nets.
Keywords: Anopheles funestus, Autodissemination, Pyriproxyfen, Sterilization, Semi-field settings
Background
Long-lasting insecticidal nets (LLINs) and indoor spray with residual insecticides (IRS) have remained the core interventions for malaria control [1, 2]. However, the gains achieved with these indoor-based interventions are threatened by the on-going development of insecticide resistance within targeted malaria vector populations [3, 4]. Worryingly, increased outdoor biting as a result of mosquito behavioural adaptation, and change in human behaviour by spending more time outdoors altogether makes these tools less effective in sustaining the gains [5–9]. Despite the urgent call for additional vector control tools to complement LLINs and IRS to accelerate the efforts toward malaria elimination, the additional tools need to align with the local context of the specific malaria endemic countries [10].
Larval source management (LSM) particularly chemical or microbial larviciding, is one of the promising tools that can be used in conjunction with adult-based interventions for effective control of malaria vectors [11–13]. Larviciding has additional benefits because it targets the aquatic stage of vectors and thus controls the population of exophilic, endophagic, and resistant mosquitoes that are associated with malaria transmission [13, 14]. It is recommended by the World Health Organization (WHO) to be implemented as a supplementary intervention in areas where breeding habitats are only few, findable, and easy to map and treat [15].
The potential of using larviciding for malaria control in urban settings was first demonstrated in Tanzania as part of urban malaria control programme, resulting to a 32% reduction in annual mean vector densities and sporozoite prevalence in three malaria vectors: Anopheles funestus, Anopheles coustani and Anopheles gambiae [16]. The government of Tanzania, leveraged these successes, and established the Tanzania Biotech Products Limited (TBPL), a bio-larvicide plant. This initiative has enabled the piloting of the larviciding intervention to 11 municipals in 2016 and gradually scaled up to all municipal councils in 2020 [17–19].
Effective implementation of larviciding depends on the accurate identification and targeting of productive mosquito breeding habitats [20]. This necessitates a sustainable surveillance system for monitoring the availability and distribution of breeding habitats before implementation [21–23]. Recently, the application of geospatial technology and deployment of unmanned aerial vehicles (UAV) have proven effective in identifying and targeting water bodies in a wide space, which would not have been possible by only relying on personnel [24–26]. On the other hand, both UAV and geospatial technology are resource demanding, and require high operational and analysis skills. In addition, they are both limited in distinguishing the mosquito productive breeding habitats from merely water bodies [25, 26]. On this basis, the high cost and limitations associated with both the conventional larviciding and deployment of UAV highlight the need for alternative larviciding strategies that are cost-effective and complementary to LLINs and IRS, such as the autodissemination technique [27–29].
The autodissemination technique, also known as “Mosquito-assisted larviciding” is a technique that exploit females mosquito oviposition behaviour, to transfer insecticides from a contaminated resting station to its breeding habitat and results in mortality or prevents emergence of the immature mosquito therein [28]. Several studies have demonstrated the effectiveness of the autodissemination technique, mainly with pyriproxyfen, an insect growth regulator, in controlling Aedes, Culex, and Anopheles mosquitoes under controlled and field settings [28–34]. The autodissemination technique interrupts malaria transmission by preventing the emergence of adult vectors from contaminated breeding habitats resulting in the reduction of malaria vector density [35, 36]. By relying on female mosquitoes that know suitable places to breed, this technique can precisely enhance high coverage of targeted breeding habitat during field application and overcome the need for widespread application of insecticide and excessive use of labour [32, 37, 38].
Pyriproxyfen a juvenile hormone mimic is an insect growth regulator (IGR) that has been demonstrated to effectively control disease-carrying mosquitoes of different species [29, 39–41]. Pyriproxyfen works by mimicking the action of a naturally occurring juvenile hormone by interfering with the growth and development of the target insect resulting in either sterilizing the contaminated mosquitoes [42, 43] or inhibiting adult emergence [44]. In addition, the compound is highly specific and effective at a ultralow concentration [45]. To date, there is no evidence of pyriproxyfen resistance in malaria vectors with practical implications, with exception of animal model experiments that suggest pyriproxyfen can be metabolized in the same way as pyrethroid [46]. Another study highlighted a partial increase in the level of mosquito tolerance to pyrethroids when used in sub-lethal doses [47].
Of importance, pyriproxyfen is the safe compound, with allowable amount of 300 parts per billion in human drinking water, which is 6 times higher than amount recommended by the WHO for effective mosquito control [45]. While the autodissemination technique and sterilization impact of pyriproxyfen has been demonstrated with An. gambiae and An. arabiensis [29, 34], the ability of An. funestus, a dominant malaria vector, to perform autodissemination of pyriproxyfen remains unknown. Therefore, this study was conducted in a semi-field setting in rural Tanzania to evaluate the efficacy of pyriproxyfen to control the An. funestus via autodissemination and sterilization effects.
Methods
Study site
The study was conducted at Kining’ina village (8.11417 S, 36.67484 E), in rural southern Tanzania from May to October 2022 inside a partitioned netted cage with dimensions of 6 m long, 2.25 m wide, 2 m high, built within the semi-field system. The partitioned, netted cage made with fine mesh was installed inside the semi-field chamber to prevent An. funestus from escaping upon release, due to its small body size [48, 49].
Mosquitoes rearing
Insectary-reared female An. funestus aged 5–9 days post eclosion were used for the experiments. These colonies originated from individual wild An. funestus mosquitoes collected from three villages (Tulizamoyo, Ikwambi and Sululu) within the Kilombero valley in 2019. The standardized rearing procedures of the An. funestus mosquito colonies has already been described [48]. It has been reported that the insemination rate increases as the age of male mosquitoes increases [50]. Therefore, it was considered essential to mix a batch of 5–9 days old females with male An. funestus of older age, 10–14 days in the same cages to increase the chance of mating. Female mosquitoes were starved for 12 h prior to blood feeding, by placing arm of consented human inside the cage for 30 min. These procedures were repeated on two consecutive days to ensure that the mosquitoes were fully engorged.
Test insecticide
Sumilarv dust containing pyriproxyfen of 10% active ingredient and 12 μm particle size, supplied Sumitomo Chemicals, Japan, was used in all experimental replicates.
Preparation of contamination station
In all experimental replicates, a clay pot of 10-litre volume was used as a contamination station. It was prepared by lining the inside with damp black cotton cloth dusted with 2.5 g of 10% pyriproxyfen powder using a paint brush [29]. Prior to mosquito exposure, the dusted clay pot was left to dry for 24 h to allow the pyriproxyfen powder to attach to the cotton fibers and enhance pick-up by female mosquitoes upon landing.
Experimental procedures and study design
Experiment 1: assessment of pyriproxyfen transfer from pyriproxyfen-treated clay pot to the breeding habitat by female Anopheles funestus
Two chambers of the semi-field system which were 1.5 m apart were used as the treatment and control chambers for this experiment. In the treatment chamber, a PPF-dusted clay pot and a breeding container of 1 L of water were installed at a distance of 5 m apart. Two experiments (30 min and 48 h exposure) of five replicates each were performed by exposing mosquitoes to PPF inside the clay pot. In each replicate, a batch of 300 blood-fed mosquitoes were separately held inside treated clay pot for 30 min and 48 h before were released for oviposition. The breeding habitat container was monitored for the presence of eggs for two consecutive days before being removed from the chamber.
Because the number of eggs that were naturally deposited in the breeding containers were few, the presence of PPF was confirmed via larval bioassay, during which 20 instar three insectary-reared larvae of An. funestus were introduced in a container that was removed from the chamber, and monitored for daily mortality and emergence success until all were dead or emerged to adults. A similar setup was adopted for the control chamber.
Experiment 2: assessment of fertility, reduction in egg laying, and eggs viability as a proxy indicator for pyriproxyfen sterilization effect on exposed female Anopheles funestus
Few numbers of eggs signaled the possibility of sterilization effect to exposed females. Hence, the sterilizing effect of PPF on exposed An. funestus via pyriproxyfen autodissemination was assessed using a sub sample of mosquitoes from experiment 1 (60 mosquitoes per replicate). The samples were gently aspirated on day three post their first blood meal. Of 60 mosquitoes, 30 were dissected for examination of ovary development, and 30 were transferred into a separate net cage (15 × 15 × 15 cm) and monitored for oviposition events over three gonotrophic cycles.
Prior dissection, An. funestus females were anesthetized by freezing at − 20°C for 10 min. Mosquitoes were dissected by gently pulling out the last two segments of the abdomen under the stereoscopic microscope at 0.7x magnification and the extracted ovaries were further observed under a compound microscope for a focused view of ovary appearance. The ovaries’ development status was recorded as normal when they appeared fully developed at stage V or previtellogenic resting stage and abnormal when underdeveloped at IV. The classification and interpretation of ovary appearance were based on the Christopher stage of egg development [51].
During oviposition monitoring, an oviposition substrate made of a petri dish and wet/damp cotton lined with filter paper was placed inside the net cage. The An. funestus were maintained with 10% glucose solution ad libitum. The oviposition substrate was then followed up for a maximum of five days to observe the presence of eggs. If the eggs were spotted, then the filter paper containing the eggs was removed and eggs were counted and transferred into a 300 ml plastic cup filled with tap water. Therein, eggs were monitored for daily hatching for up to seven days consecutive until all eggs hatched to larvae or died [42, 52, 53].
Following the first egg-laying cycle, the remaining An. funestus were offered a second blood meal to assess the effect of PPF on the second gonotrophic cycle. Three days post blood feeding, the oviposition substrate was supplied for egg laying. Similar monitoring procedures for egg development and hatching as described above were followed. Following the second egg-laying cycle, similar procedures were repeated to assess the effect of PPF on the third gonotrophic cycle [54].
Statistical analysis
Statistical software, R version 4.2.1, was used to analyze the data. Generalized linear mixed-effects models (GLMM) were implemented using functions within the lme4 package [55]. The difference in the total number of laid eggs between the control and treatment chambers was determined with Poisson distribution using the best fit Generalized linear mixed model. The difference in the adult emergence rate of third instar larvae previously introduced into water basins was determined with a binomial GLMM using the logit function. The difference in the total number of eggs laid by the batch of female mosquitoes in the net cage and their hatch rate was also analysed using Poisson and binomial GLMM, respectively. In the model, the experiment chamber (with or without PPF) was classified as a fixed effect, whereas the experiment replicate was classified as a random effect variable. In addition, Percentage reduction of fertility per gonotrophic cycle was calculated using the following formulation. Reduction in fertility (%) per gonotrophic cycle = (C − T)/ C × 100 where C = hatch rate in the unexposed group; T = hatch rate in the exposed group.
Results
Pyriproxyfen transfer from pyriproxyfen-treated clay pot to the breeding habitat by female Anopheles funestus
Both exposure times (30 min and 48 h) resulted in the transfer of pyriproxyfen from the PPF-treated clay pot to the provided breeding habitats. In all five experimental replicates, at 30 min of exposure, the mean (95% CI) percentage of adult emergence was 70% (69–71%) in the control chamber compared to 15.5% (14.916.1%) in the treatment chamber (p < 0.001). Similarly, at 48 h of exposure, the mean (95% CI) percentage of adult emergence was 95% (88 – 98%) in the control chamber compared to 19% (12 –28%) in the treatment chamber (p < 0.001) (Table 1). The significantly lower percentage of adult emergence in the treatment chamber suggest that provided breeding habitats were contaminated with pyriproxyfen by ovipositing female An. funestus.
Table 1.
Percentage of adult emerged from the third instar larvae in the breeding habitats
| Exposure time | Section | Proportion % [95% CI] | OR [95% CI] | P-values |
|---|---|---|---|---|
| 30 min | Control | 70.3 [69.3, 71.2] | 1 | < 0.001 |
| Pyriproxyfen | 15.5 [14.9, 16.1] | 0.07 [0.03, 0.21] | ||
| 48 h | Control | 95.0 [88.4, 97.9] | 1 | < 0.001 |
| Pyriproxyfen | 18.9 [12.2, 28.2] | 0.01 [0.004, 0.03] |
OR odds ratio, CI confidence interval, min minutes, hrs hours
Effect of autodissemination of pyriproxyfen on the Anopheles funestus reproductive capacity
As summarized in Table 2, female mosquitoes contaminated with pyriproxyfen laid significantly fewer eggs (estimated from recorded larvae) compared to the uncontaminated mosquitoes in the control chamber (p < 0.001). For 30 min and 48 h exposure, the mean number (95%CI) of mosquito larvae resulting from laid eggs in the control chamber were 89.7 ± 25.56 and 249.5 ± 15.56 respectively. The number of larvae in their respective treatment groups were significantly low, 26.7 ± 3.72 for 30 min and 49.6 ± 3.42 for 48 h exposure (p < 0.001).
Table 2.
Mean number of larvae hatched from laid eggs by An. funestus in the control and pyriproxyfen-contaminated breeding habitats
| Exposure time | Section | Predicted mean [95% CI] | RR [95% CI] | P-value |
|---|---|---|---|---|
| 30 min | Control | 189.7 [139.6, 257.9] | 1 | < 0.001 |
| Pyriproxyfen | 26.7 [19.4, 36.8] | 0.14 [0.13, 0.16] | ||
| 48 h | Control | 249.4 [218.9, 284.1] | 1 | < 0.001 |
| Pyriproxyfen | 49.6 [42.9, 57.4] | 0.20 [0.18, 0.22] |
RR relative risk, CI confidence interval, min minutes, hrs hours
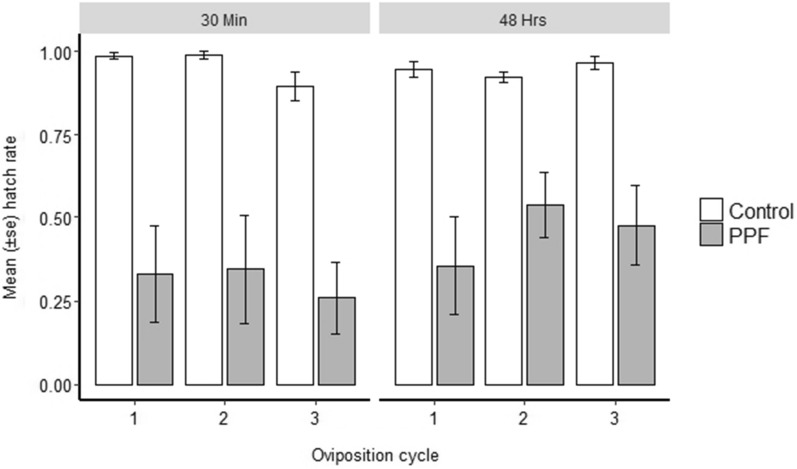
Compared to the control group, the hatch rate was significantly lower in the treatment group across all three gonotrophic cycles for both exposure times (p < 0.001) (Fig. 1). In a treatment group, hatchability decreases with increase in gonotrophic cycles for both exposure times. Overall, the reduction of hatch rate at 30 min exposure was 15.4%, 52.9%, and 60.6% for the first, second, and third gonotrophic cycles respectively, whereas at 48 h exposure were 20.8%, 45% and 51.5%, respectively.
Fig. 1.
The hatch rate of the eggs laid by exposed and unexposed female Anopheles funestus across three oviposition cycles
Effect of pyriproxyfen exposure on female Anopheles funestus sterility/fertility
Pyriproxyfen exposure adversely affected female mosquitoes eggs development (Fig. 2; Table 3). Of the 590 exposed and unexposed female An. funestus that were dissected to observe the effect of PPF on eggs development, 35% (n = 205) had abnormal eggs, and the remaining 65% (n = 385) had normal eggs. During 30 min exposure, 4% (n = 6) of 141 females in a control group had abnormal eggs compared to 42% (n = 63) of 149 in the treatment group. Whereas for 48 h exposure, 4% (n = 6) of 150 females in a a control group had abnormal eggs compared to 87% (n = 130) of 150 females in the treatment group. However, normal eggs of the females exposed for 48 h were observed to have fewer abnormal eggs arrested at stage IV.
Fig. 2.

Morphological features of an eggs of PPF-exposed and unexposed blood-fed female Anopheles funestus at 72 h post blood meal. A and C showing observed abnormal eggs (undetachable, oval shape with no floats). B and D showing observed normal eggs (detachable, boat/sausage shape with floats)
Table 3.
Effect of pyriproxyfen exposure on female An. funestus fertility
| Effect of pyriproxyfen on An. funestus fertility | 30 min Exposure | 48 h Exposure | ||
|---|---|---|---|---|
| PPF | Control | PPF | Control | |
| Mosquito exposed | 1500 | 1500 | 1500 | 1500 |
| Mosquitoes dissected | 149 | 141 | 150 | 150 |
| Percentage of sterilized females (with 100% abnormal eggs) | 42% | 4% | 87% | 4% |
| Percentage of fertile females | 58% | 96% | 13% | 96% |
PPF treatment chamber, min minutes, hrs hours
Discussion
The current study has proven that sterilized female An. funestus exposed one day post blood meal, can transfer a lethal dose of pyriproxyfen to the breeding habitat located 5 m from a contaminated clay pot. Overall, forced contaminated An. funestus with pyriproxyfen resulted to 78% and 81% adult emergence inhibition of its filial at 30 min and 48 h of exposure respectively. These findings are corroborated by previous studies in Anopheles that documented successful autodissemination events by An. arabiensis, An. gambiae and Anopheles quadrimaculatus, via either self or forceful mosquito contamination [29, 31, 33, 34, 56]. The recorded similarity in emergence inhibition at 30 min and 48 h might be due to loss of the picked pyriproxyfen particles because of their grooming behaviour when mosquitoes are exposed longer, its absorption to mosquito cuticle, and during flight to breeding habitat [57–59].
In this study, the females were held in presence of pyriproxyfen for 30 min and 48 h to mimic possible minimum and maximum resting time for rest seeking blood fed mosquitoes in the field environment [60–64]. In a situation where mosquitoes are transiting the contamination stations, the success of autodissemination events might be impaired [28, 57, 65].
Of importance, this study documented An. funestus vulnerability to pyriproxyfen sterilization after being exposed one day post blood meal, and confirmed significant reduction in eggs laid (fecundity). Overall, at 30 min and 48 h of pyriproxyfen exposure, the mean number of eggs laid by the exposed group was reduced by 85.9% and 80.1% respectively compared to the control group. Similarly, negative effect of pyriproxyfen on mosquito fecundity has been also shown in several studies [34, 42, 54, 66, 67]. Consistent with previous study [54], the effect of pyriproxyfen on fecundity and fertility (eggs hatchability) in exposed An. funestus was observed up to third gonotrophic cycle, suggesting that this effect might be irreversible during mosquito lifespan.
Previous studies have reported that pyriproxyfen interferes with the balance of hormones levels between juvenile hormone and ecdysone hormone, and disrupt the hormonal pathways responsible for egg’s maturation [44, 54, 66, 68]. Similarly, in this currently study, the dissection of PPF exposed female mosquito revealed that pyriproxyfen sterilization effect was via retention of under developed (unmatured) eggs. Longer exposure time resulted to high proportion of mosquitoes that retain underdeveloped eggs compared to shorter exposure time. Many underdeveloped eggs were arrested at Christopher stage IV, a proxy indication for sterilization effect [68]. It has been documented in other studies that the sterilization effect interferes with the desire of contaminated female to find a place for oviposition. [34, 58, 66]. This depends on the time of pyriproxyfen exposure relative to when the female obtains a blood meal. While Mbare and others reported unlikelihood of contaminated female mosquito to visit the oviposition habitat after being exposed to pyriproxyfen within 24 h before and after the blood meal [34], Itoh et al., reported the frequency of visiting the oviposition habitat to be lower for female exposed to pyriproxyfen before blood meal and higher for female exposed to pyriproxyfen after blood meal [58].
Furthermore, Yadav et al. [66], when assessing surface treated with a range of pyriproxyfen concentrations, reported a lower frequency of visiting oviposition habitat to a female exposed to a lower concentration of pyriproxyfen at 24 h before blood meal and higher to the females exposed at 24 h after blood. But the frequency of visiting the oviposition habitat was the same only for the female exposed to higher concentration [66]. In this current study, female mosquitoes exposed 24 h post blood meal were capable of visiting oviposition habitat. The difference in oviposition behaviour for contaminated female mosquitoes across different studies might be due to differences in pyriproxyfen exposure methods, pyriproxyfen formulation (e.g., powder or suspension), pyriproxyfen doses and environments under which the study was conducted.
It has been reported in previous studies that environmental factors, such as wind speed, temperature, and relative humidity are responsible for triggering oviposition flights of gravid female mosquitoes [69]. Because the current study was conducted in a semi-field environment, the observed oviposition behaviour in sterilized An. funestus is more representative to what might happen under actual field settings compared to similar studies that were conducted under laboratory conditions [34, 58, 66].
Overall, the findings of this study further support the potential of autodissemination of pyriproxyfen in controlling primary susceptible and resistant malaria vectors. More striking, is the fact that sterilized mosquitoes were capable to autodisseminate pyriproxyfen enough to cause adult emergence inhibition at the breeding habitats. Therefore, its potential use could be aligned with the current recommended integrated vectors control approach, which focuses on controlling and eliminating outdoor and residual malaria transmission [15, 70, 71]. Furthermore, this presents an opportunity of scaling up this technique along recently recommended next generation bed nets co-treated with pyriproxyfen and pyrethroid [71]. It was envisaged that, host-seeking resistant mosquitoes sterilized by pyriproxyfen nets might transfer pyriproxyfen upon successful access to a bloodmeal and resting in a contaminated station. In addition, the combined effect of these two modes of actions of pyriproxyfen can be mathematically modelled to assess its additive or synergistic effect on malaria transmission interruption.
The appropriate time for deploying autodissemination of pyriproxyfen is mainly during the dry season [72, 73] characterized by few but stable breeding habitats. This season provides ideal condition to attain optimal doses to prevent adult emergence in the breeding habitats. On the contrary, implementing autodissemination of pyriproxyfen during rainy season, associated with flooding, hence dilution of PPF in the breeding habitats, might amplify resistance levels of the already pyrethroid resistant mosquito population as the results of the sub-lethal doses in the habitats [47].
Despite achieving the main objective of this study, some limitations were observed. The effect of autodissemination of pyriproxyfen was not directly monitored at the provided breeding habitat but through larval bioassays. All experiment were conducted in presence of small water volumes (1 L), which was important to prove the principle, but not representative of actual habitats found in the field environment [74]. Lastly, the resistance status of the exposed mosquitoes was not assessed, instead the supposition that they were resistant was based on the most recent reports from the same study area [75–78].
Conclusion
The present study demonstrates sterilization and adult emergence inhibition effects in An. funestus via autodissemination of pyriproxyfen. It also offers proof that sterilized An. funestus can transfer pyriproxyfen sufficient to prevent adult emergence at its habitats. These promising findings warrant further assessment of the autodissemination of pyriproxyfen in controlling wild population of An. funestus, and emphasize its potential in complementing LLINs.
Acknowledgements
We would like to express our appreciation to Miss Monica Mpingwa, Miss Nuru Nchimbi, Mr Dickson Mwasheshi, and Mr Daniel Mathias Mabula for their support and cooperation throughout the study. All persons acknowledged here have been informed and have consented to the acknowledgement.
Abbreviations
- AI
Active ingredient
- GTS
Global technical strategy
- IRS
Indoor residual spray
- IGR
Insecticide growth regulator
- LSM
Larval source management
- LC
Lethal concentration
- LLINs
Long Lasting Insecticidal nets
- NMSP
National Malaria Strategic Plan
- SFS
Semi field system
- PPF
Pyriproxyfen
- TBPL
Tanzania Biotechnology Product Limited
- WHO
World Health Organization
Author contributions
HJK and DL conceptualized the idea, designed the study protocol, study design, conducted the experiment and contributed to the analysis, result interpretation and discussion.HN, AA, YAM performed data analysis, interpreted the results. HJK drafted the manuscript. AJN, NU, MJ, JS, FO, AM, FST and DL provided the comments to the manuscript draft.All authors read and approved the final draft of the manuscript.
Funding
We do also acknowledge funding support from the National Institute for Health Research (NIHR) (using the UK’s Official Development Assistance (ODA) Funding) and Wellcome [218776/Z/19/Z] under the NIHR-Wellcome Partnership for Global Health Research.
Availability of data and materials
All data underlying the presented results in this study will be available upon request.
Declarations
Ethics approval and consent to participate
This study was approved by Ifakara Health Institute Review Board (IHI-IRB) with ethical clearance certificate number IHI/IRB/No:11-2022.
Consent for publication
This manuscript has been approved for publication by the National Institute for Medical Research in Tanzania, NIMR/HQ/P.12 VOL.XXXVI/ 16.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Hamisi J. Kunambi, Email: hkunambi@ihi.or.tz
Dickson Lwetoijera, Email: dwilson@ihi.or.tz.
References
- 1.Eisele TP, Larsen DA, Walker N, Cibulskis RE, Yukich JO, Zikusooka CM et al. Estimates of child deaths prevented from malaria prevention scale-up in Africa 2001–2010. Malar J. 2012;11(93). 10.1186/1475-2875-11-93. [DOI] [PMC free article] [PubMed]
- 2.Bhatt S, Weiss DJ, Cameron E, Bisanzio D, Mappin B, Dalrymple U, et al. The effect of malaria control on Plasmodium falciparum in Africa between 2000 and 2015. Nature. 2015;526:207–211. doi: 10.1038/nature15535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Matowo NS, Munhenga G, Tanner M, Coetzee M, Feringa WF, Ngowo HS, et al. Fine-scale spatial and temporal heterogeneities in insecticide resistance profiles of the malaria vector, Anopheles arabiensis in rural south-eastern Tanzania. Wellcome Open Res. 2017;2:96. doi: 10.12688/wellcomeopenres.12617.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kisinza NS, Nkya TE, Kabula B, Overgaard HJ, Massue DJ, Mageni Z, et al. Multiple insecticide resistance in Anopheles gambiae from Tanzania: a major concern for malaria vector control. Malar J. 2017;16:439. doi: 10.1186/s12936-017-2087-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Govella NJ, Chaki PP, Killeen GF. Entomological surveillance of behavioural resilience and resistance in residual malaria vector populations. Malar J. 2013;12:124. doi: 10.1186/1475-2875-12-124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Russell TL, Govella NJ, Azizi S, Drakeley CJ, Kachur SP, Killeen GF. Increased proportions of outdoor feeding among residual malaria vector populations following increased use of insecticide-treated nets in rural Tanzania. Malar J. 2011;10:80. doi: 10.1186/1475-2875-10-80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mutuku FM, King CH, Mungai P, Mbogo C, Mwangangi J, Muchiri EM, et al. Impact of insecticide-treated bed nets on malaria transmission indices on the south coast of Kenya. Malar J. 2011;10:356. doi: 10.1186/1475-2875-10-356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mwangangi JM, Mbogo CM, Orindi BO, Muturi EJ, Midega JT, Nzovu J, et al. Shifts in malaria vector species composition and transmission dynamics along the kenyan coast over the past 20 years. Malar J. 2013;12:13. doi: 10.1186/1475-2875-12-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kreppel KS, Viana M, Main BJ, Johnson PCD, Govella NJ, Lee Y, et al. Emergence of behavioural avoidance strategies of malaria vectors in areas of high LLIN coverage in Tanzania. Sci Rep. 2020;10:14527. doi: 10.1038/s41598-020-71187-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.WHO. Global technical strategy for malaria 2016–2030. Geneva, World Heal. Organ. 2015. Available from: http://apps.who.int/iris/bitstream/10665/176712/1/9789241564991_eng.pdf?ua=1.
- 11.Beier JC, Keating J, Githure JI, MacDonald MB, Impoinvil DE, Novak RJ. Integrated vector management for malaria control. Malar J. 2008;7. Suppl: S4. 10.1186/1475-2875-7-S1-S4. [DOI] [PMC free article] [PubMed]
- 12.Chanda E, Masaninga F, Coleman M, Sikaala C, Katebe C, MacDonald M, et al. Integrated vector management: the zambian experience. Malar J. 2008;7:164. doi: 10.1186/1475-2875-7-164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Choi L, Majambere S, Wilson AL. Larviciding to prevent malaria transmission. Cochrane Database Syst Rev. 2019;8:CD012736. doi: 10.1002/14651858.CD012736.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Killeen GF. Characterizing, controlling and eliminating residual malaria transmission. Malar J. 2014;13:330. doi: 10.1186/1475-2875-13-330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Interim Position WHO. Statement. 2012. Available from: https://apps.who.int/iris/bitstream/handle/10665/337991/WHO-HTM-GMP-2012.06-eng.pdf.
- 16.Geissbühler Y, Kannady K, Chaki PP, Emidi B, Govella NJ, Mayagaya V, et al. Microbial larvicide application by a large-scale, community-based program reduces malaria infection prevalence in urban Dar Es Salaam, Tanzania. PLoS ONE. 2009;4:e5107. doi: 10.1371/journal.pone.0005107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mapua SA, Finda MF, Nambunga IH, Msugupakulya BJ, Ukio K, Chaki PP, et al. Addressing key gaps in implementation of mosquito larviciding to accelerate malaria vector control in southern Tanzania: results of a stakeholder engagement process in local district councils. Malar J. 2021;20:123. doi: 10.1186/s12936-021-03661-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Presidents office regional admistration and local government. Implementation of larviciding for killing malaria mosquito larva. 2018. Available from http://hssrc.tamisemi.go.tz/storage/app/uploads/public/5b8/7d9/9e2/5b87d99e28b3e080525154.pdf.
- 19.Matindo AY, Kapalata SN, Katalambula LK, Meshi EB, Munisi DZ. Biolarviciding for malaria vector control: acceptance and associated factors in southern Tanzania. Curr Res Parasit Vector-Borne Dis. 2021;1:1–7. doi: 10.1016/j.crpvbd.2021.100038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gu W, Novak RJ. Habitat-based modeling of impacts of mosquito larval interventions on entomological inoculation rates, incidence, and prevalence of malaria. Am J Trop Med Hyg. 2005;73:546–552. [PubMed] [Google Scholar]
- 21.Vanek MJ, Shoo B, Mtasiwa D, Kiama M, Lindsay SW, Fillinger U, et al. Community-based surveillance of malaria vector larval habitats: a baseline study in urban Dar es Salaam, Tanzania. BMC Public Health. 2006;6:154. doi: 10.1186/1471-2458-6-154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fillinger U, Kannady K, William G, Vanek MJ, Dongus S, Nyika D, et al. A tool box for operational mosquito larval control: preliminary results and early lessons from the Urban Malaria Control Programme in Dar es Salaam, Tanzania. Malar J. 2008;7:20. doi: 10.1186/1475-2875-7-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dongus S, Nyika D, Kannady K, Mtasiwa D, Mshinda H, Fillinger U, et al. Participatory mapping of target areas to enable operational larval source management to suppress malaria vector mosquitoes in Dar es Salaam, Tanzania. Int J Health Geogr. 2007;6:37. doi: 10.1186/1476-072X-6-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fillinger U, Lindsay SW. Suppression of exposure to malaria vectors by an order of magnitude using microbial larvicides in rural Kenya. Trop Med Int Health. 2006;11:1629–1642. doi: 10.1111/j.1365-3156.2006.01733.x. [DOI] [PubMed] [Google Scholar]
- 25.Hardy A, Makame M, Cross D, Majambere S, Msellem M. Using low-cost drones to map malaria vector habitats. Parasit Vectors. 2017;10:29. doi: 10.1186/s13071-017-1973-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Stanton MC, Kalonde P, Zembere K, Hoek Spaans R, Jones CM. The application of drones for mosquito larval habitat identification in rural environments: a practical approach for malaria control? Malar J. 2021;20:244. doi: 10.1186/s12936-021-03759-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Majambere S, Lindsay SW, Green C, Kandeh B, Fillinger U. Microbial larvicides for malaria control in the Gambia. Malar J. 2007;6:76. doi: 10.1186/1475-2875-6-76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Devine GJ, Perea EZ, Killeen GF, Stancil JD, Clark SJ, Morrison AC. Using adult mosquitoes to transfer insecticides to Aedes aegypti larval habitats. Proc Natl Acad Sci U S A. 2009;106:11530–11534. doi: 10.1073/pnas.0901369106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lwetoijera DW, Harris C, Kiware S, Devine DJ, McCall PJ, et al. Effective autodissemination of pyriproxyfen to breeding sites by the exophilic malaria vector Anopheles arabiensis in semi-field settings in Tanzania. Malar J. 2014;13:161. doi: 10.1186/1475-2875-13-161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Abad-Franch F, Zamora-Perea E, Luz SLB. Mosquito-disseminated insecticide for citywide vector control and its potential to block arbovirus epidemics: entomological observations and modeling results from amazonian Brazil. PLoS Med. 2017;14:e1002213. doi: 10.1371/journal.pmed.1002213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Swale DR, Li Z, Kraft JZ, Healy K, Liu M, David CM, et al. Development of an autodissemination strategy for the deployment of novel control agents targeting the common malaria mosquito, Anopheles quadrimaculatus Say (Diptera: Culicidae) PLoS Negl Trop Dis. 2018;12:e0006259. doi: 10.1371/journal.pntd.0006259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.McKemey A, Adey R. Autodissemination of insecticides for Mosquito control. 2018. Available from: https://www.ivcc.com/wp-content/uploads/2019/10/Autodissemination-Report-Adey-and-McKemey-Sep-2018.pdf.
- 33.Lwetoijera DW, Kiware S, Okumu F, Devine GJ, Majambere S. Autodissemination of pyriproxyfen suppresses stable populations of Anopheles arabiensis under semi-controlled settings. Malar J. 2019;18:166. doi: 10.1186/s12936-019-2803-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mbare O, Lindsay SW, Fillinger U. Pyriproxyfen for mosquito control: female sterilization or horizontal transfer to oviposition substrates by Anopheles gambiae sensu stricto and Culex quinquefasciatus. Parasit Vectors. 2014;7:280. doi: 10.1186/1756-3305-7-280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Macdonald G. The analysis of the sporozoite rate. Trop Dis Bull. 1952;49:569–586. [PubMed] [Google Scholar]
- 36.Macdonald G. The objectives of residual insecticide campaigns. Trans R Soc Trop Med Hyg. 1952;46:227–235. doi: 10.1016/0035-9203(52)90070-9. [DOI] [PubMed] [Google Scholar]
- 37.Unlu I, Rochlin I, Suman DS, Wang Y, Chandel K, Gaugler R. Large-scale operational pyriproxyfen autodissemination deployment to suppress the immature asian tiger mosquito (Diptera: Culicidae) populations. J Med Entomol. 2020;57:1120–1130. doi: 10.1093/jme/tjaa011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chandel K, Suman DS, Wang Y, Unlu I, Williges E, Williams GM, et al. Targeting a hidden enemy: pyriproxyfen autodissemination strategy for the control of the container mosquito Aedes albopictus in cryptic habitats. PLoS Negl Trop Dis. 2016;10:e0005235. doi: 10.1371/journal.pntd.0005235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lupenza ET, Kihonda J, Limwagu AJ, Ngowo HS, Sumaye RD, Lwetoijera DW. Using pastoralist community knowledge to locate and treat dry-season mosquito breeding habitats with pyriproxyfen to control Anopheles gambiae s.l. and Anopheles funestus s.l in rural Tanzania. Parasitol Res. 2021;120:1193–1202. doi: 10.1007/s00436-020-07040-4. [DOI] [PubMed] [Google Scholar]
- 40.Mbare O. Novel insecticides and application strategies for malaria vector control. Thesis. 2015;1–229. Available from: http://researchonline.lshtm.ac.uk/2374462/1/PhDthesis_OscarOchiengMbareLSH314576.pdf .
- 41.Pallett K. Can we expect new herbicides with novel modes of action in the foreseable future? Outlooks Pest Manag. 2016;27:39–43. [Google Scholar]
- 42.Harris C, Lwetoijera DW, Dongus S, Matowo NS, Lorenz LM, Devine GJ, et al. Sterilising effects of pyriproxyfen on Anopheles arabiensis and its potential use in malaria control. Parasit Vectors. 2013;6:144. doi: 10.1186/1756-3305-6-144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lwetoijera DW, Harris C, Kiware SS, Killeen GF, Dongus S, Devine GJ, et al. Comprehensive sterilization of malaria vectors using pyriproxyfen: a step closer to malaria elimination. Am J Trop Med Hyg. 2014;90:852–855. doi: 10.4269/ajtmh.13-0550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dhadialla TS, Carlson GR, Le DP. New insecticides with ecdysteroidal and juvenile hormone activity. Annu Rev Entomol. 1998;43:545–569. doi: 10.1146/annurev.ento.43.1.545. [DOI] [PubMed] [Google Scholar]
- 45.WHO. Pyriproxyfen in drinking-water: use for vector control in drinking-water sources and containers. Backgr Doc Dev WHO Guidel Drink Qual. 2008; WHO/HSE/AMR/08.03/9.
- 46.Yunta C, Grisales N, Nász S, Hemmings K, Pignatelli P, Voice M, et al. Pyriproxyfen is metabolized by P450s associated with pyrethroid resistance in an. gambiae. Insect Biochem Mol Biol. 2016;78:50–57. doi: 10.1016/j.ibmb.2016.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Opiyo MA, Ngowo HS, Mapua SA, Mpingwa M, Nchimbi N, Matowo NS, et al. Sub-lethal aquatic doses of pyriproxyfen may increase pyrethroid resistance in malaria mosquitoes. PLoS ONE. 2021;16:e0248538. doi: 10.1371/journal.pone.0248538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ngowo HS, Hape EE, Matthiopoulos J, Ferguson HM, Okumu FO. Fitness characteristics of the malaria vector Anopheles funestus during an attempted laboratory colonization. Malar J. 2021;20:148. doi: 10.1186/s12936-021-03677-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Meza FC, Muyaga LL, Limwagu AJ, Lwetoijera DW. The ability of Anopheles funestus and Anopheles arabiensis to penetrate LLINs and its effect on their mortality. Wellcome Open Res. 2022;7:265. doi: 10.12688/wellcomeopenres.18242.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Verhoek BA, Takken W. Age effects on the insemination rate of Anopheles gambiae s.1. in the laboratory. Entomol Exp Appl. 1994;1994(72):167–172. [Google Scholar]
- 51.Clements AN, Boocock MR. Ovarian development in mosquitoes: stages of growth and arrest, and follicular resorption. Physiol Entomol. 1984;9:1–8. [Google Scholar]
- 52.Djènontin A, Ahoua Alou LP, Koffi A, Zogo B, Duarte E, N’Guessan R, et al. Insecticidal and sterilizing effect of Olyset Duo®, a permethrin and pyriproxyfen mixture net against pyrethroid-susceptible and -resistant strains of Anopheles gambiae s.s.: a release-recapture assay in experimental huts. Parasite. 2015;22:27. doi: 10.1051/parasite/2015027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Suman DS, Wang Y, Bilgrami AL, Gaugler R. Ovicidal activity of three insect growth regulators against Aedes and Culex mosquitoes. Acta Trop. 2013;128:103–109. doi: 10.1016/j.actatropica.2013.06.025. [DOI] [PubMed] [Google Scholar]
- 54.Koama B, Namountougou M, Sanou R, Ndo S, Ouattara A, Dabiré RK, et al. The sterilizing effect of pyriproxyfen on the malaria vector Anopheles gambiae: physiological impact on ovaries development. Malar J. 2015;14:101. doi: 10.1186/s12936-015-0609-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bates D, Mächler M, Bolker BM, Walker SC. Fitting linear mixed-effects models using lme4. J Stat Softw. 2015;67:1–48. [Google Scholar]
- 56.Mbare O, Lindsay SW, Fillinger U. Testing a pyriproxyfen auto-dissemination station attractive to gravid Anopheles gambiae sensu stricto for the development of a novel attract-release -and-kill strategy for malaria vector control. BMC Infect Dis. 2019;19:800. doi: 10.1186/s12879-019-4438-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gaugler R, Suman D, Wang Y. An autodissemination station for the transfer of an insect growth regulator to mosquito oviposition sites. Med Vet Entomol. 2012;26:37–45. doi: 10.1111/j.1365-2915.2011.00970.x. [DOI] [PubMed] [Google Scholar]
- 58.Itoh T. Utilization of bloodfed females of Aedes aegypti as a vehicle for the transfer of the insect growth regulator, pyriproxyfen, to larval habitats. Trop Med. 1994;36:243–248. [PubMed] [Google Scholar]
- 59.Zhukovskaya M, Yanagawa A, Forschler B. Grooming behavior as a mechanism of Insect Disease Defense. Insects. 2013;4:609–630. doi: 10.3390/insects4040609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gillies MT. Studies in house leaving and outside resting of Anopheles gambiae giles and Anopheles funestus giles in East Africa.: II.—The exodus from houses and the house resting population. Bull Entomol Res. 1954;45:375–387. [Google Scholar]
- 61.Smith A. Studies on domestic habits of A. gambiae that affect its vulnerability to insecticides. I. resting places in huts. East Afr Med J. 1962;39:15–24. [PubMed] [Google Scholar]
- 62.Smith A. The preferential indoor resting habits of Anopheles gambiae in the Umbugwe area of Tanganyika. East Afr Med J. 1962;39:631–635. [PubMed] [Google Scholar]
- 63.Msugupakulya BJ, Kaindoa EW, Ngowo HS, Kihonda JM, Kahamba NF, Msaky DS, et al. Preferred resting surfaces of dominant malaria vectors inside different house types in rural south-eastern Tanzania. Malar J. 2020;19:22. doi: 10.1186/s12936-020-3108-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Fereda DE. Mating behavior and gonotrophic cycle in Anopheles gambiae complex and their significance in Vector competence and Malaria Vector Control. J Biomed Res Environ Sci. 2022;3:31–43. [Google Scholar]
- 65.Caputo B, Ienco A, Cianci D, Pombi M, Petrarca V, Baseggio A, et al. The auto-dissemination approach: a novel concept to fight Aedes albopictus in urban areas. PLoS Negl Trop Dis. 2012;6:e1793. doi: 10.1371/journal.pntd.0001793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Yadav K, Dhiman S, Acharya B, Ghorpade RR, Sukumaran D. Pyriproxyfen treated surface exposure exhibits reproductive disruption in dengue vector aedes aegypti. PLoS Negl Trop Dis. 2019;13:e0007842. doi: 10.1371/journal.pntd.0007842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ponlawat A, Fansiri T, Kurusarttra S, Pongsiri A, McCardle PW, Evans BP, et al. Development and evaluation of a pyriproxyfen-treated device to control the dengue vector, Aedes aegypti (L.) (Diptera: Culicidae) Southeast Asian J Trop Med Public Health. 2013;44:167–178. [PubMed] [Google Scholar]
- 68.Rowland M, Soto A, Messenger LA, Kirby M, Mosha FW, Manjurano A, Protopopoff N. Protopopoff N. Ovary Dissection Is a Sensitive Measure of Sterility in Anopheles gambiae Exposed to the Insect Growth Regulator Pyriproxyfen. Insects. 2023 Jun 14;14(6):552. 10.3390/insects14060552. PMID: 37367368; PMCID: PMC10299475. [DOI] [PMC free article] [PubMed]
- 69.Przigoda N, Wille R, Przigoda J, Drechsler R. behavioral aspects. Automated Validation & Verification of UML/OCL Models Using Satisfiability Solvers. 2018;125–57. 10.1007/978-3-319-72814-8_5.
- 70.WHO. Position statement on integrated vector management. Geneva, World Health Organization. 2015. Available from: https://apps.who.int/iris/bitstream/handle/10665/241146/WER8320_177-181.PDF.
- 71.WHO. Guideline for malaria 2023. Geneva, World Health Organization.
- 72.Mmbaga AT, Lwetoijera DW. Current and future opportunities of autodissemination of pyriproxyfen approach for malaria vector control in urban and rural Africa. Wellcome Open Res. 2023;8:119. doi: 10.12688/wellcomeopenres.19131.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Devine GJ, Killeen GF. The potential of a new larviciding method for the control of malaria vectors. Malar J. 2010;9:142. doi: 10.1186/1475-2875-9-142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Nambunga IH, Ngowo HS, Mapua SA, Hape EE, Msugupakulya BJ, Msaky DS, et al. Aquatic habitats of the malaria vector Anopheles funestus in rural south-eastern Tanzania. Malar J. 2020;19:219. doi: 10.1186/s12936-020-03295-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kaindoa EW, Matowo NS, Ngowo HS, Mkandawile G, Mmbando A, Finda M, et al. Interventions that effectively target Anopheles funestus mosquitoes could significantly improve control of persistent malaria transmission in south-eastern Tanzania. PLoS ONE. 2017;12:e0177807. doi: 10.1371/journal.pone.0177807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Urio NH, Pinda PG, Ngonzi AJ, Muyaga LL, Msugupakulya BJ, Finda M, et al. Effects of agricultural pesticides on the susceptibility and fitness of malaria vectors in rural south-eastern Tanzania. Parasit Vectors. 2022;15:213. doi: 10.1186/s13071-022-05318-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Pinda PG, Eichenberger C, Ngowo HS, Msaky DS, Abbasi S, Kihonda J, et al. Comparative assessment of insecticide resistance phenotypes in two major malaria vectors, Anopheles funestus and Anopheles arabiensis in south-eastern Tanzania. Malar J. 2020;19:408. doi: 10.1186/s12936-020-03483-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lwetoijera DW, Harris C, Kiware SS, Dongus S, Devine GJ, McCall PJ, et al. Increasing role of Anopheles funestus and Anopheles arabiensis in malaria transmission in the Kilombero Valley, Tanzania. Malar J. 2014;13:331. doi: 10.1186/1475-2875-13-331. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data underlying the presented results in this study will be available upon request.