Abstract
Background
The capacity of an individual to respond to changes in food intake so that postprandial metabolic perturbations are resolved, and metabolism returns to its pre-prandial state, is called phenotypic flexibility. This ability may be a more important indicator of current health status than metabolic markers in a fasting state.
Aim
In this parallel randomized controlled trial study, an energy-restricted healthy diet and 2 dietary challenges were used to assess the effect of weight loss on phenotypic flexibility.
Methods
Seventy-two volunteers with overweight and obesity underwent a 12-wk dietary intervention. The participants were randomized to a weight loss group (WLG) with 20% less energy intake or a weight-maintenance group (WMG). At weeks 1 and 12, participants were assessed for body composition by MRI. Concurrently, markers of metabolism and insulin sensitivity were obtained from the analysis of plasma metabolome during 2 different dietary challenges—an oral glucose tolerance test (OGTT) and a mixed-meal tolerance test.
Results
Intended weight loss was achieved in the WLG (−5.6 kg, P < 0.0001) and induced a significant reduction in total and regional adipose tissue as well as ectopic fat in the liver. Amino acid-based markers of insulin action and resistance such as leucine and glutamate were reduced in the postprandial phase of the OGTT in the WLG by 11.5% and 28%, respectively, after body weight reduction. Weight loss correlated with the magnitude of changes in metabolic responses to dietary challenges. Large interindividual variation in metabolic responses to weight loss was observed.
Conclusion
Application of dietary challenges increased sensitivity to detect metabolic response to weight loss intervention. Large interindividual variation was observed across a wide range of measurements allowing the identification of distinct responses to the weight loss intervention and mechanistic insight into the metabolic response to weight loss.
Keywords: phenotypic flexibility, weight loss, insulin sensitivity, meal challenges, metabolites
Introduction
The maintenance of energy homeostasis in postprandial and postabsorptive periods requires different metabolic processes to be activated either to store excess energy from food intake or to mobilize stored substrates. This dynamic shift between negative and positive energy balance with a transfer of molecules between tissues and regulation of metabolic pathways, is to a large extent coordinated by the dynamic interplay between different hormones such as insulin, glucagon, cortisol, and incretins. The sensitivity of tissues to insulin is very important for regulating the metabolism of carbohydrates, fatty acids, and amino acids. Insulin-dependent regulation of metabolism involves multiple organs, signaling pathways, and metabolites derived from all 3 macronutrients [1]. The concentrations of these metabolites and signaling molecules in biofluids constitute dynamic phenotypic traits.
Although insulin sensitivity has become a marker of overall metabolic health, it does not describe how the metabolic networks respond to particular cues. To achieve homeostasis, physiology maintains a well-orchestrated machinery allowing the organism to adapt to the continuously changing environment, in which food plays a major role [1]. The ability to respond adequately to transient changes in substrate availability induced by food intake may predict the health status of the organism [2,3]. Homeostasis might render relatively insensitive biomarkers in biofluids that are being sampled in the absence of stressors (i.e., after overnight fasting). On the contrary, assessing the same biomarkers in a challenged state might provide more sensitive metabolic information [1,3,4]. As an example, in the absence of a challenging stimulus plasma glucose concentration is tightly regulated by several mechanisms to maintain its levels within a narrow range. Changes in plasma glucose concentrations induced by a meal may allow the detection of early problems in glucose homeostasis because the meal promotes metabolic changes involved in restoring preprandial plasma glucose concentrations [5]. Phenotypic flexibility refers to the response capacity to environmental cues triggering changes in plasma levels of metabolites and signaling molecules.
The NutriTech project aimed to integrate emerging and established technologies to develop a deeper understanding of phenotypic flexibility. In this study, we used a weight loss intervention coupled with a healthy diet in an overweight cohort—an established methodology that has been shown to have a positive effect on insulin sensitivity and phenotypic flexibility [6]. We used 2 dietary challenges to assess phenotypic flexibility: an OGTT and a mixed-meal tolerance test (MMTT), followed by a thorough characterization of body composition and plasma metabolites.
Subjects and Methods
Recruitment and study population
Research ethics was granted by the West London Ethic Committee (12/LO/0139) and the study is registered at ClinicalTrials.gov NCT01684917. The Supplementary Methods section provides information on recruitment, the screening process, and inclusion and exclusion criteria. All participants provided written, informed consent at the screening visit. The participants were recruited between June 2012 and July 2014 and the intervention run between July 2012 and October 2014. The primary outcome of the study was a change in insulin sensitivity. In total, 72 subjects completed the study. Recruitment numbers and flow are presented in Supplemental Figure 1.
Dietary intervention study
A randomized comparison of a 20% energy-restricted diet for 12 wk compared with a 12-wk weight-maintenance diet (based on average energy intake in the EU), was conducted in a cohort of adults classified as overweight and obese (average BMI: 29.2; range: 24.7–35.6). Inclusion and exclusion criteria are listed in Supplemental Table 1. Subjects in the energy restriction group are referred to as the weight-loss group (WLG) in contrast to the subjects in the weight-maintenance group (WMG). There was a similar sex distribution in both groups: 16 males and 16 females in WMG compared with 19 males and 21 females in the WLG. There were no significant differences in means of weight, height, waist, hip, waist-to-hip ratio, fasting plasma glucose and insulin concentrations, and blood pressure at baseline (TABLE 1, TABLE 2). Our approach aimed to compare the impact of dietary profile in the WLG that aligns with health guidelines and would be expected to have a positive effect on insulin sensitivity and phenotypic flexibility. We compared this to a dietary macronutrient profile commonly consumed in Europe in the WMG that would be aligned with a deterioration in insulin sensitivity and phenotypic flexibility.
TABLE 1.
Changes in body composition and biomarkers induced by weight loss
| WLG before |
WLG after |
WMG before |
WMG after |
P value | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SEM | N | Mean | SEM | N | Mean | SEM | N | Mean | SEM | N | |||
| Energy intake | kcal/d | 1834.3 | 88 | 39 | 1343.1 | 80 | 36 | 1999.7 | 118 | 32 | 1795.7 | 90 | 31 | 0.005 |
| BW | kg | 84.29 | 2.0 | 40 | 78.66 | 1.92 | 40 | 83.40 | 2.40 | 32 | 83.46 | 2.35 | 32 | <0.0001 |
| BMI | kg/m2 | 29.31 | 0.47 | 40 | 27.36 | 0.47 | 40 | 29.03 | 0.44 | 32 | 29.07 | 0.45 | 32 | <0.0001 |
| Hip | cm | 109.37 | 1.28 | 40 | 105.81 | 1.27 | 40 | 107.80 | 0.97 | 32 | 107.47 | 1.08 | 31 | <0.0001 |
| Waist | cm | 100.0 | 1.71 | 40 | 95.07 | 1.80 | 40 | 99.03 | 1.86 | 32 | 98.24 | 1.92 | 31 | 0.006 |
| Total AT | L | 35.83 | 1.71 | 38 | 30.49 | 1.51 | 37 | 33.02 | 1.63 | 30 | 33.40 | 1.66 | 30 | <0.0001 |
| Subcutaneous AT | L | 27.77 | 1.55 | 38 | 23.70 | 1.33 | 37 | 25.38 | 1.52 | 30 | 25.55 | 1.56 | 30 | <0.0001 |
| Internal AT | L | 8.06 | 0.45 | 38 | 6.79 | 0.41 | 37 | 7.64 | 0.41 | 30 | 7.85 | 0.42 | 30 | <0.0001 |
| Nonabdominal internal AT | L | 3.47 | 0.18 | 38 | 3.09 | 0.17 | 37 | 3.38 | 0.14 | 30 | 3.47 | 0.16 | 30 | <0.0001 |
| Nonabdominal subcutan. AT | L | 19.53 | 1.06 | 38 | 16.84 | 0.92 | 37 | 17.96 | 1.01 | 30 | 18.06 | 1.04 | 30 | <0.0001 |
| Intra-abdominal AT | L | 4.59 | 0.29 | 38 | 3.71 | 0.26 | 37 | 4.26 | 0.31 | 30 | 4.38 | 0.32 | 30 | <0.0001 |
| Abdom. subcutan. AT | L | 8.24 | 0.52 | 38 | 6.86 | 0.46 | 37 | 7.42 | 0.54 | 30 | 7.48 | 0.55 | 30 | <0.0001 |
| Periphery AT | L | 23.00 | 1.14 | 38 | 19.40 | 1.11 | 38 | 21.34 | 1.08 | 30 | 21.54 | 1.10 | 30 | 0.0035 |
| Trunk AT | L | 12.83 | 0.66 | 38 | 10.57 | 0.60 | 37 | 11.68 | 0.63 | 30 | 11.86 | 0.64 | 30 | <0.0001 |
| Liver fat | AU | 4.35 | 0.79 | 38 | 2.31 | 0.32 | 33 | 4.81 | 0.94 | 29 | 5.06 | 1.02 | 30 | 0.0008 |
| DBP | mmHg | 79.53 | 1.23 | 40 | 71.85 | 1.57 | 39 | 75.14 | 1.65 | 32 | 76.06 | 1.71 | 32 | 0.0006 |
| SBP | mmHg | 128.10 | 2.08 | 40 | 121.08 | 1.80 | 39 | 124.92 | 2.02 | 32 | 126.09 | 2.04 | 30 | 0.0036 |
| s-E-Selectin | ng/mL | 37.48 | 3.64 | 40 | 29.72 | 2.66 | 39 | 32.06 | 3.93 | 32 | 31.23 | 3.09 | 31 | 0.0291 |
| Leptin | ng/mL | 15.97 | 1.36 | 40 | 9.98 | 1.20 | 39 | 14.63 | 1.46 | 32 | 14.68 | 1.63 | 31 | <0.0001 |
| Total cholesterol | mmol/L | 5.40 | 0.17 | 39 | 4.97 | 0.15 | 39 | 5.16 | 0.17 | 32 | 5.36 | 0.16 | 32 | 0.0016 |
| LDL cholesterol | mmol/L | 3.12 | 0.16 | 39 | 2.82 | 0.14 | 39 | 2.89 | 0.16 | 31 | 3.02 | 0.15 | 32 | 0.0174 |
AT, adipose tissue; WLG, weight-lowering group; WMG, weight-maintaining group; DBP, diastolic blood pressure; SBP, systolic blood pressure. Results are presented as means ± SEM. P values refer to the interaction between group (WLG × WMG) and time (before × after intervention) in a mixed model analysis followed by multiple comparisons (Sídák).
TABLE 2.
Weight loss–induced changes in plasma levels of markers of insulin sensitivity at fasting and during dietary challenges
| WLG before |
WLG after |
WMG before |
WMG after |
P value WLG before × after | Pvalue WMG before × after | P value interaction arm × visit | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SEM | Mean | SEM | Mean | SEM | Mean | SEM | |||||
| Markers measured during OGTT at t=0 min | ||||||||||||
| Insulin | mIU/L | 15.27 | 0.92 | 13.56 | 1.10 | 16.38 | 1.51 | 16.28 | 1.01 | 0.175 | 0.989 | 0.303 |
| Glucose | mmol/L | 5.20 | 0.09 | 5.02 | 0.08 | 5.13 | 0.10 | 5.02 | 0.09 | 0.018 | 0.275 | 0.483 |
| Triglycerides | mmol/L | 1.31 | 0.07 | 1.12 | 0.09 | 1.24 | 0.09 | 1.31 | 0.11 | 0.013 | 0.487 | 0.009 |
| Ala | μmol/L | 326.8 | 12.2 | 312.4 | 11.59 | 322.3 | 15.1 | 331.2 | 17.0 | 0.448 | 0.782 | 0.223 |
| Glu | μmol/L | 52.74 | 5.12 | 48.58 | 4.09 | 54.66 | 4.9 | 57.11 | 5.40 | 0.658 | 0.871 | 0.374 |
| Leu | μmol/L | 126.8 | 4.03 | 123.2 | 3.54 | 131.3 | 5.0 | 133.6 | 6.30 | 0.657 | 0.849 | 0.356 |
| Trp | μmol/L | 54.43 | 1.54 | 52.38 | 1.57 | 54.66 | 1.64 | 54.25 | 2.28 | 0.220 | 0.948 | 0.399 |
| Tyr | μmol/L | 62.28 | 1.91 | 56.87 | 1.93 | 61.98 | 1.61 | 61.38 | 2.47 | 0.008 | 0.948 | 0.077 |
| Val | μmol/L | 210.2 | 5.87 | 205.8 | 5.46 | 216.7 | 7.62 | 216.0 | 9.56 | 0.719 | 0.992 | 0.686 |
| Propionylcarnitine | μmol/L | 0.29 | 0.02 | 0.28 | 0.01 | 0.33 | 0.02 | 0.34 | 0.02 | 0.349 | 0.442 | 0.087 |
| Ser | μmol/L | 108.4 | 3.70 | 121.5 | 4.45 | 107.2 | 3.30 | 109.1 | 5.21 | 0.004 | 0.900 | 0.067 |
| Gly | μmol/L | 239.4 | 12.1 | 267.9 | 14.30 | 234.1 | 12.1 | 232.6 | 15.4 | 0.003 | 0.983 | 0.019 |
| Markers measured during the OGTT expressed as area under the curve | ||||||||||||
| Insulin | AUC | 15,062 | 1373 | 12238 | 1246 | 15,018 | 1669 | 13939 | 1322 | 0.003 | 0.963 | 0.046 |
| Glucose | AUC | 1474 | 55 | 1375 | 48 | 1489 | 63 | 1466 | 62 | 0.024 | 0.830 | 0.192 |
| Triglycerides | AUC | 306 | 18 | 271 | 23 | 306 | 23 | 309 | 26 | 0.044 | 0.984 | 0.096 |
| Ala | AUC | 77,695 | 2073 | 71,432 | 2506 | 78,956 | 2806 | 79,333 | 2412 | 0.008 | 0.984 | 0.039 |
| Glu | AUC | 8655 | 887 | 6726 | 626 | 9266 | 719 | 9502 | 885 | 0.001 | 0.910 | 0.010 |
| Leu | AUC | 23,146 | 988 | 20,758 | 734 | 23,485 | 784 | 24,682 | 949 | 0.003 | 0.262 | 0.001 |
| Trp | AUC | 11,916 | 273 | 11,227 | 366 | 11,803 | 336 | 12,070 | 305 | 0.024 | 0.608 | 0.019 |
| Tyr | AUC | 11,577 | 389 | 10,553 | 367 | 11,827 | 406 | 12,078 | 362 | 0.005 | 0.739 | 0.010 |
| Val | AUC | 43,243 | 1089 | 40,448 | 1360 | 44,359 | 1400 | 45,005 | 1420 | 0.013 | 0.810 | 0.024 |
| Propionylcarnitine | AUC | 64 | 3 | 57 | 3 | 73 | 5 | 73 | 4 | 0.048 | 0.999 | 0.126 |
| Ser | AUC | 22,100 | 687 | 23,339 | 913 | 21,968 | 785 | 22,460 | 755 | 0.002 | 0.576 | 0.115 |
| Gly | AUC | 54,242 | 2738 | 57,676 | 3241 | 52,949 | 2867 | 53,873 | 2783 | 0.032 | 0.803 | 0.234 |
| Markers measured during the MMTT expressed as area under the curve | ||||||||||||
| Insulin | AUC | 19,533 | 1575 | 16,687 | 1348 | 18,960 | 1414 | 17,693 | 1572 | 0.005 | 0.259 | 0.335 |
| Glucose | AUC | 2403 | 42 | 2316 | 38 | 2419 | 43 | 2378 | 50 | 0.102 | 0.654 | 0.506 |
| Triglycerides | AUC | 855 | 53 | 764 | 44 | 933 | 85 | 814 | 64 | 0.067 | 0.382 | 0.665 |
| Ala | AUC | 156,275 | 3480 | 150,327 | 4296 | 160,771 | 5695 | 157,140 | 5694 | 0.565 | 0.518 | 0.889 |
| Glu | AUC | 19,606 | 1172 | 16,422 | 1098 | 21,014 | 1734 | 20,524 | 1702 | 0.004 | 0.747 | 0.107 |
| Leu | AUC | 67,793 | 1683 | 65,719 | 1615 | 70,621 | 2049 | 71,130 | 2587 | 0.399 | 0.954 | 0.314 |
| Trp | AUC | 24,033 | 537 | 23,213 | 587 | 23,587 | 535 | 23,704 | 714 | 0.202 | 0.984 | 0.234 |
| Tyr | AUC | 30,425 | 709 | 28,943 | 843 | 30,528 | 918 | 30,677 | 1036 | 0.096 | 0.999 | 0.181 |
| Val | AUC | 101,730 | 2250 | 99,049 | 2339 | 103,753 | 3066 | 103,553 | 3642 | 0.252 | 0.801 | 0.582 |
| Propionylcarnitine | AUC | 138 | 6 | 129 | 6 | 156 | 10 | 150 | 9 | 0.160 | 0.707 | 0.550 |
| Ser | AUC | 51,436 | 1845 | 56,298 | 2020 | 51,718 | 2311 | 49,508 | 2410 | 0.003 | 0.388 | 0.003 |
| Gly | AUC | 111,582 | 5468 | 122,677 | 6082 | 110,783 | 5792 | 107,325 | 5039 | 0.002 | 0.553 | 0.003 |
MMTT, mixed-meal tolerance test; WLG, weight-lowering group (N = 40); WMG, weight-maintaining group (N = 32); Ala, alanine; Glu, glutamine; Leu, leucine; Trp, tryptophan; Tyr, tyrosine; Val, valine; Ser, serine; Gly, glycine. Results are presented as means ± SEM. Metabolites in highlighted lines are positively correlated to insulin sensitivity. P values refer to multiple comparisons (Sídák) between WLG before and after the intervention, WMG before and after the intervention, and for the interaction arm (group) × visit (before and after intervention) that followed a mixed model analysis.
The dietary macronutrient content in the WLG reflected nutritional recommendations in the United Kingdom. Approximately 50% of energy derived is from carbohydrates, of which more than 18 g of dietary fiber, 35% from fat, and 15% from protein. Energy intake was 20% less than the estimated energy expenditure [7,8]. The diet was based on 5 main food groups (grains, fruit and vegetables, meat and fish, dairy, and fats), with a dietary profile aligned to Dietary Approaches to Stop Hypertension (DASH) recommendations [9]. This diet has been demonstrated to improve insulin sensitivity and cardiovascular disease risk factors [10]. The WMG followed a diet matching their usual energy expenditure. The diet was based on the average intake in the EU and included approximately 45% energy derived from carbohydrates, 40% from fat, and 15% from protein [10].
Both groups were contacted individually at weeks 0, 4, 8, and 12, and with telephonic interviews on weeks 2, 6, and 10. The aim was to encourage dietary compliance and reduce variability in weight loss. Diets were individually composed to comply with the personal dietary habits of everyone. Both groups completed 7-d food diaries during weeks 1–13 to monitor dietary changes. Both groups were instructed to keep exercise at habitual levels. More details about the intervention study can be found in the Supplementary Methods.
Randomization
Randomization was carried out using an online system for clinical trials called Sealed Envelope (https://www.sealedenvelope.com/). Stratified randomization based on sex, age, and BMI was used to allocate volunteers to each group.
Dietary compliance
Dietary compliance was monitored by estimating the change in fat-free and fat mass over time [11]. We also estimated change in diet quality using urinary metabolomic dietary model [12]. The urine samples were prepared with a pH 7.4 phosphate buffer for 1H-NMR spectroscopy as previously described [13] and were analyzed at 300 K on a 600 MHz spectrometer (Bruker BioSpin) using a standard 1-dimensional pulse sequence with water presaturation [13]. The urinary metabolic profiles were projected into a previously validated urinary metabolomic dietary model [12].
Challenge tests
A major aspect of phenotypic flexibility is the ability to recover from transient metabolic perturbations reflected in changes in plasma levels of metabolites and signaling molecules when challenged with food intake. Two dietary challenges were carried out after overnight fasting at baseline and after 12 wk of the intervention to assess changes in metabolism:
-
1.
OGTT, when blood samples were taken at t=0, 15, 30, 60, 120, and 240 min after the intake of 75 g glucose in 200 mL filtered water. This allows a standardized method to assess glucose homeostasis.
-
2.
MMTT, based on a liquid, high-fat, and high-glucose drink [4,5]. Blood samples were taken at t=0, 30, 60, 120, 240, 360, and 480 min after intake of a 200 mL test meal containing 75 g glucose, 60 g palm oil, and 20 g casein. This allows for the assessment of a standard dose of carbohydrates, fat, and protein on metabolism.
Further details about the dietary challenges are presented in Supplementary Methods.
Measurement of biomarkers
To assess changes in metabolism, multiple biomarkers including glucose, insulin, HbA1c, TG, LDL-cholesterol, total cholesterol (tChol), GGT, creatinine, uric acid, nonesterified fatty acids, glucagon, intercellular adhesion molecule 1 (ICAM1), vascular cell adhesion protein (VCAM), s-E-selectin, GLP-1, peptide YY (PYY), IL 1 beta (IL-1β), IL-6, IL-8, IL-10, IL-18, and IL-1 receptor antagonist (IL-1RA) were quantified in plasma using antibody-based and enzymatic methods, following manufacturer’s instructions. Insulin sensitivity indices such as HOMA-IR and HOMA for β-cell function (HOMA-β), and Matsuda index (MI) were calculated [[14], [15], [16], [17], [18]]. Detailed information about these measurements is presented in Supplementary Methods.
Targeted and untargeted metabolomics platforms
Acylcarnitines (24) amino acids (22), biogenic amines (12), glycerophospholipids (90), and sphingolipids (15) were quantified using the LC with tandem MS (LC-MS/MS)-based AbsoluteIDQ p180 Kit (Biocrates Life Sciences AG), following the manufacturer’s protocol. Additional acylcarnitines (26) were quantified after extraction with methanol in the presence of deuterated standards and butylated before analysis using liquid chromatography coupled to a Sciex 5500 mass spectrometer (Sciex) (LC-MS/MS) following a previously described method [19]. The 13 most abundant bile acids (BA) in plasma were quantified using an adapted method [20]. Briefly, 10 μL plasma was mixed with deuterated internal standards. After deproteinization with methanol, samples were evaporated to dryness, reconstituted in methanol:water (1:1), and injected into the LC-MS/MS system [21].
For the GC-MS analysis, metabolites were extracted from 40 μL plasma by ice-cold methanol:H2O (8:1) in a sample:solvent ratio of 1:10. After centrifugation (13,200 × g, 4 min, 4°C), 200 μL of supernatant was dried under vacuum. A 2-step derivatization was performed using an autosampler (Agilent 7693, Agilent Technologies) by incubating the samples with methoxyamine hydrochloride (20 mg/mL in pyridine) for 30 min at 45°C, followed by the addition of N-methyl-N-trimethylsilyl-triflouroacetamide, and a second incubation for 30 min at 45°C. Immediately after derivatization, each sample was submitted to GC-MS analysis (Agilent 6890N GC coupled to an Agilent 5975C inert XL; Agilent Technologies). The gas chromatograph was equipped with a 30-m DB-35MS capillary column (Agilent J&W GC Column). Metabolites were eluted by a temperature gradient from 80°C and rising by 11°C/minute to 325°C with 5 min hold at 325°C. Metabolite identification and quantification were accomplished using the Metabolite Detector software. Metabolites were identified according to their retention time and spectra similarity against the Golm metabolome database.
Plasma samples were randomized to exclude batch variation, ensuring a proportional number of samples from the 2 dietary challenges, collected before and after the weight loss intervention and from men and women. Quality control plasma samples (Recipe chemicals and instruments) were included in each batch of samples to control for analytical drifting.
Magnetic resonance imaging for determination of body composition
Detailed methodology of MRI measurements has been reported elsewhere [[22], [23], [24]] to quantify total and regional adipose tissue (AT) depots, as well as the fat content of the liver, pancreas, and soleus and tibialis muscles. A typical example of these measures from a single volunteer is shown in Supplemental Figure 2.
Sample size estimation
Our primary outcome was insulin sensitivity. Data for the power analysis were taken from Blumenthal et al. [10]. This was the only trial that reported the impact of weight loss and change in dietary quality on insulin sensitivity when the protocol was developed. In this trial, weight loss and improved dietary quality were associated with improved insulin sensitivity assessed by OGTT and lower total cholesterol. The difference between the means in the Blumenthal et al. [10] study was 0.18 with a standard deviation of 0.2 (a decrease that was also associated with significant improvement in several plasma risk factors such as total cholesterol and triglycerides), assuming an alpha of 5% and power of 90 and 2-tailed test to detect a change in insulin sensitivity. Thus, we needed 37 participants per group. Allowing for a dropout of 30%, we aimed to recruit 50 volunteers per group.
Data integration and statistical analyses
All NutriTech data were collected in a distributed database specifically designed to handle multi-omics human nutritional intervention studies performed at multiple sites, the “nutritional phenotype database” (dbNP http://www.dbnp.org/). Data were checked for normality using Shapiro–Wilks and all results are presented as means ± SEM. In case of missing data, the imputation was done by averaging the nearest neighbors. Because most of the missing data were derived from time series analyses (the dietary challenges), this was the method of choice. A mixed model analysis of variance was applied to fasting data including intervention (energy restriction/weight maintenance) and term (baseline/follow-up) as fixed factors and their interaction. The subject was used as a random factor. Detailed statistical procedures are described in the Supplementary Material. Pearson correlation coefficients were calculated and reported together with P values as well as the number of samples used in the calculation. Partial least squares discriminant analysis (PLS-DA) was used to build a model to identify discriminating metabolites in a subset of study subjects. Monte Carlo cross-validation was used to train 1000 models and obtain an aggregate predicted score for each sample when part of the test set with one-fifth of the data held out at random in each iteration.
Results
Response to intervention
Weight loss.
The 12 wk energy restriction promoted a significant reduction in body weight (BW) in the WLG displaying a mean reduction of 5.6 kg (P < 0.0001), whereas the WMG did not display a reduction in BW (Table 1). Despite meeting the target weight loss, there was a wide individual variation in the response to the intervention, with weight loss in WLG ranging from 0.1 to 17.5 kg (Supplemental Figure 3).
Impact of the intervention of dietary intake.
We observed a significant reduction in self-reported energy intake in the WLG group (average energy intake reduction 503.9±85.3 kcal/d, P < 0.0001) as compared with a nonsignificant alteration in the WMG (average energy intake reduction 171.6±98.3 kcal/d, P = 0.08; Table 1). Participants in the WLG also profited from improved dietary quality assessed by DASH score (2.2±0.21 compared with 3.6±0.22, P < 0.001), which was not seen in the WMG (1.9±0.2 compared with 1.8±0.19, P < 0.68). Waist and hip circumferences were reduced in the WLG, although the waist:hip ratio was unaffected. Systolic and diastolic blood pressure decreased upon weight loss in the WLG (5.5% and 8.4% decreases, P = 0.005 and 0.0007, respectively), whereas no effect was observed in the WMG (Table 1).
Dietary compliance.
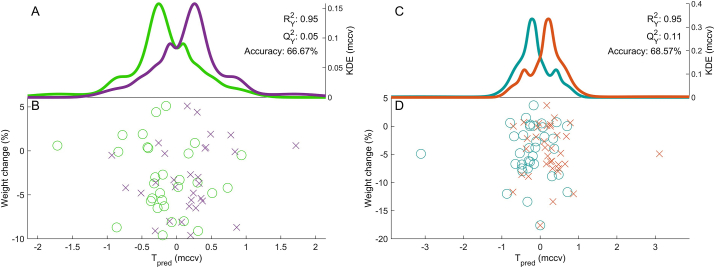
There was a strong relationship between percentage weight loss and estimated daily dietary energy reduction based on actual weight loss (r = 0.83, P < 0.001). In the WLG 10/36 volunteers had estimated daily energy reduction based on actual weight loss <75% of the prescribed diet. The heterogeneity of the weight loss appeared to be due to dietary compliance. Urine metabolomics assessment of diet quality showed a slight improvement in the diet quality for WLG but not in the WMG (Figure 1).
FIGURE 1.
Dietary predictions using urinary metabolomics in the repeated measures design. (A, B) weight-maintaining group (WMG) diet week 1 is green; week 12 is purple. (C, D) weight-lowering group (WLG) diet – week 1 is cyan; week 12 is orange. (A, C) Kernel density estimates (KDEs) of the Monte Carlo Cross Validation (MCCV) Repeated Measures Partial Least Squares (RM-PLS) predictions. (B, D) Predicted scores (Tpred) against the weight change (in %). Using a one-sample t-test to assess the difference between weeks 1 and 12, there was no significant difference in the weight-maintenance group at P = 0.06 but there was in the weight loss group toward the healthy eating profile at P = 0.03.
Markers of insulin sensitivity and phenotypic flexibility during dietary challenges
There was no significant difference in fasting glucose, insulin, HOMA IR, or HOMA %β during the 12 wk intervention between the WLG and the WMG in the OGTT (Table 3). However, there was a significant decrease in fasting insulin and HOMA IR in the MMTT. The estimation of insulin sensitivity using the MI suggests an increase in insulin sensitivity after weight loss in the OGTT but not in the MMTT.
TABLE 3.
Fasting and mean postprandial values for glucose, insulin, HOMA-IR, Β-cell function, and the Matsuda Index fasting and postprandial parameters from OGTT and MMTT tests
| Variable | WLG |
WMG |
ANCOVA |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Week 1 |
Week 12 |
Week 1 |
Week 12 |
||||||||
| Mean | SEM | Mean | SEM | Δ Change | Mean | SEM | Mean | SEM | Δ Change | P value | |
| OGTT | |||||||||||
| Fasting glucose, mmol/L | 5.1 | 0.1 | 5.0 | 0.1 | -0.17 (-0.3,-0.04) | 5.1 | 0.1 | 5.0 | 0.1 | -0.1 (-0.3,0.04) | 0.610 |
| Fasting insulin, μU/mL | 15.2 | 0.9 | 13.6 | 1.1 | -1.6 (-3.5,0.4) | 16.4 | 1.5 | 16.3 | 1.0 | -0.15 (-2.2,1.9) | 0.240 |
| HOMA IR | 1.7 | 0.1 | 1.5 | 0.1 | 0.19 (-0.4,0.02) | 1.8 | 0.2 | 1.8 | 0.1 | 0.02 (-0.3,0.2) | 0.090 |
| HOMA %β | 127.5 | 5.8 | 125.5 | 7.3 | -1.94 (-12.8,8.9) | 134.0 | 5.4 | 141.8 | 6.7 | 7.8 (-3.4,19.4) | 0.170 |
| Matsuda Index | 4.0 | 0.3 | 4.8 | 0.3 | 0.8 (0.3,1.3) | 3.9 | 0.3 | 4.0 | 0.3 | 0.15 (-0.2,0.6) | 0.030 |
| MMTT | |||||||||||
| Fasting glucose, mmol/L | 5.1 | 0.1 | 5.0 | 0.1 | -0.17 (-0.3,-0.04) | 5.0 | 0.1 | 5.0 | 0.1 | 0.02 (-0.9,1.0) | 0.100 |
| Fasting insulin, μU/mL | 18.5 | 1.3 | 14.8 | 0.1 | -3.6 (-27.7,8.8) | 18.0 | 1.2 | 19.1 | 1.9 | 1.1 (-13.2,23.9) | 0.003 |
| HOMA IR | 2.5 | 0.1 | 1.9 | 0.1 | -0.5 (-3.2, 1.1) | 2.4 | 0.1 | 2.5 | 0.1 | 0.13 (-1.7,3.0) | 0.003 |
| HOMA %β | 167.2 | 2.5 | 153.5 | 1.8 | -13 (-144,88) | 170.0 | 2.1 | 176.0 | 3.3 | 4.6 (-118,161) | 0.053 |
| Matsuda Index | 4.4 | 0.6 | 5.0 | 0.4 | 0.5 (-13,4.5) | 3.9 | 0.3 | 4.4 | 0.4 | 0.5 (-3.4,8.7) | 0.510 |
MMTT, mixed-meal tolerance test; WLG, weight-lowering group; WMG, weight-maintaining group. Values expressed mean and standard error and delta change from baseline. Between-subject effects reported in tables were assessed using ANCOVA with post values as dependent variables, type of intervention as a fixed factor (0 = weight loss, 1 = maintenance), and baseline data as a covariate.
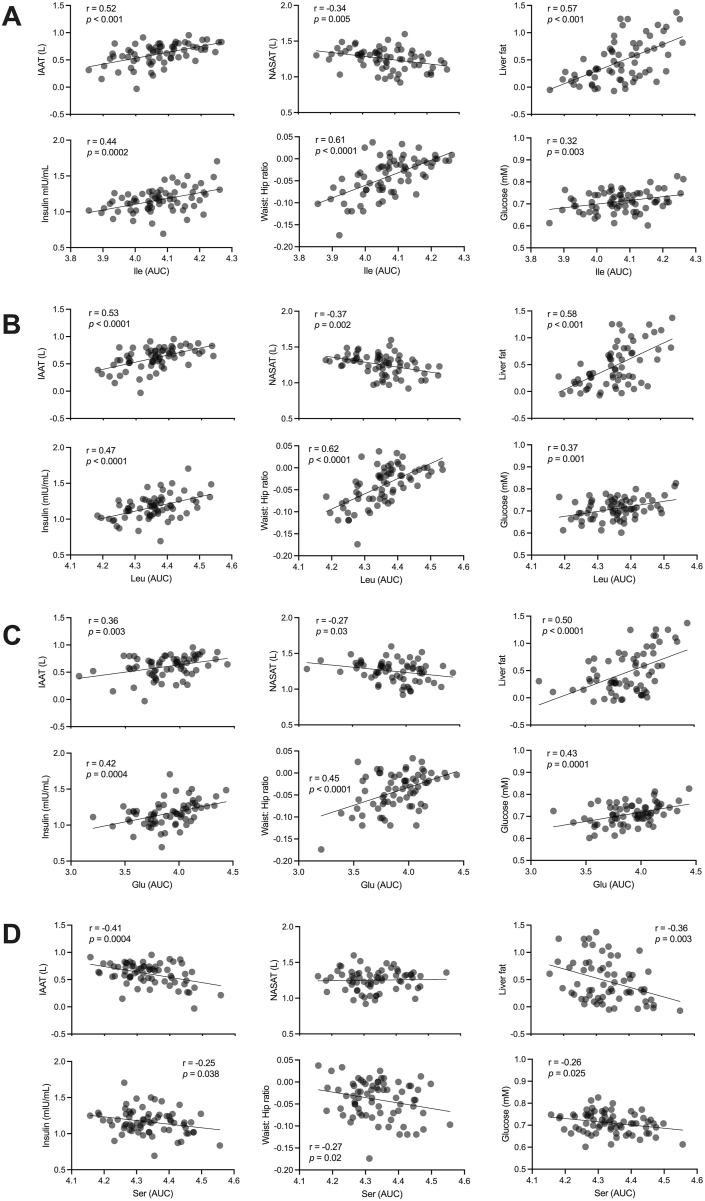
In our present study, we confirm previous observations on the relationship between body composition and markers of insulin sensitivity, although the study population primarily included normoglycemic individuals according to WHO definitions [25]. Established metabolomics markers of obesity and insulin sensitivity showed strong correlations with the volume of specific fat depots, waist:hip ratio, and glucose and insulin postprandial curves. For instance, the AUC the OGTT (OGTT AUC) of isoleucine, leucine, and glutamate was positively correlated with intra-abdominal AT, waist:hip ratio, and liver fat content. They were also positively correlated with the OGTT AUC of glucose and insulin (Figure 2). However, the OGTT AUC of these amino acids displayed a negative correlation with nonabdominal subcutaneous AT, confirming previous reports that deposition of subcutaneous fat might promote a smaller metabolic burden than the intra-abdominal deposition of AT [26,27]. The OGTT AUC of serine, an amino acid reported in association with insulin sensitivity, displayed negative correlations with intra-abdominal AT, waist:hip ratio, and hepatic fat content. The plasma concentration of serine during the OGTT was also negatively correlated with the OGTT AUC of glucose and insulin (Figure 2D). When using the same correlations for fasting data rather than OGTT AUC, similar observations were made (data not shown).
FIGURE 2.
Associations between markers of insulin sensitivity with adipose tissue and plasma glucose and insulin levels before the weight-loss intervention. (A) Pearson correlations of different markers with isoleucine. (B) Pearson correlations of different markers with leucine. (C) Pearson correlations of different markers with glutamate. (D) Pearson correlations of different markers with serine. The area under the curve data were derived from OGTT. Data were Log-transformed. Abbreviations: IAAT, intra-abdominal adipose tissue; NASAT, nonabdominal subcutaneous adipose tissue. N = 72.
Dietary challenges as a tool to assess weight loss–induced metabolic improvements
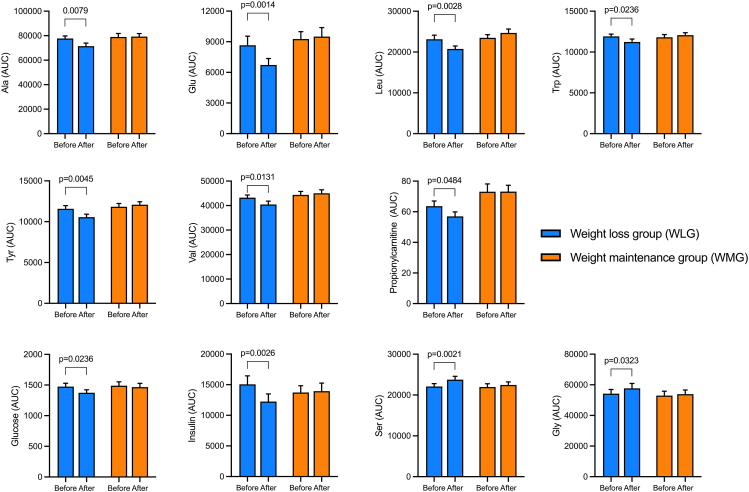
Weight loss failed to induce changes in fasting plasma concentrations of insulin, valine, and leucine, as well as other amino acid-based markers of insulin sensitivity (Table 2). However, during the OGTT, individuals from the WLG displayed lower plasma concentrations of these markers after the weight loss. The OGTT AUC of alanine, glutamate, leucine, tryptophan, tyrosine, and valine – markers of insulin resistance reported in previous studies [28,29] – were reduced after weight loss in the WLG, whereas no changes were observed in the WMG (Table 2 and Figure 3). On the contrary, the AUC of serine and glycine – metabolites previously associated with insulin sensitivity – were increased during the OGTT in the WLG after the dietary intervention by ∼5% (P < 0.04). The AUC of insulin during OGTT responded to weight loss with a 19% reduction (P = 0.007) in contrast to fasting insulin, which remained unchanged (Table 2 and Figure 3). During the MMTT, these effects were mostly unnoticed. Only insulin and glutamate exhibited reduced plasma concentrations during MMTT after weight loss, whereas serine and glycine had increased AUC during this dietary challenge (Table 2 and Figure 3). These results suggest that a dietary challenge can help detect subtle metabolic changes after weight loss, and OGTT seems to perform better than MMTT in this regard.
FIGURE 3.
Weight loss–induced changes in plasma concentration of markers of insulin sensitivity during an OGTT. Data are presented as means ± SEM. The adjusted P value is given for statistically significant differences after a mixed model analysis followed by multiple comparisons (Sídák); Weight-lowering group (WLG), N = 40. Weight-maintaining group (WMG), N = 32.
Weight loss intervention improves body composition and alters metabolic biomarkers
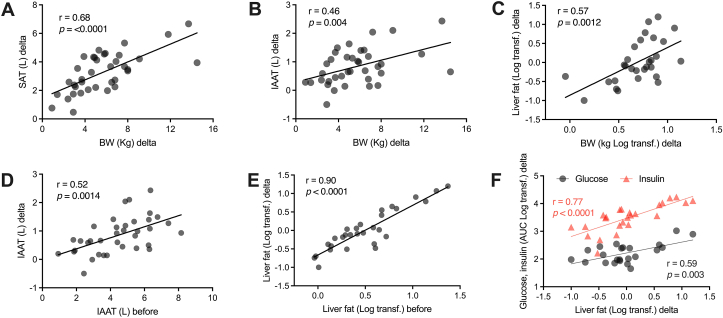
Following the weight loss intervention there were significant reductions in total and regional AT depots in the WLG, without effects in the WMG (interaction between group and time, P < 0.0001; Table 1). The mean total AT loss following the intervention was 5.3 L. The magnitude of AT reduction was greatest for the intra-abdominal AT (19.2%), and the smallest reductions were observed in the nonabdominal internal AT (10.9%), suggesting a preferential reduction of abdominal fat. The weight loss correlated positively with the reduction in the volume of subcutaneous AT (r = 0.68, P < 0.0001), intra-abdominal AT (r = 0.46, P = 0.004), and liver fat (r = 0.57, P = 0.0012) (Figure 4A–C). Although large interindividual differences were observed, there was a reduction in liver fat content in the WLG group (46.8%, P = 0.0008, Table 1).
FIGURE 4.
Associations between weight loss–induced changes in adipose tissues, ectopic fat, and markers of glucose metabolism. Two-tailed Pearson correlation analyses. When indicated, an area under the curve was derived from the OGTT. Some variables were log-transformed to facilitate visualization of the associations. Delta was calculated by subtracting the value after weight loss intervention from the value before intervention. Abbreviations: IAAT, intra-abdominal adipose tissue; BW, body weight; SAT, subcutaneous adipose tissue. Only individuals from weight-lowering group (WLG) are included (n≈40).
The amount of intra-abdominal AT at baseline correlated with the reduction in this AT depot following weight loss (r = 0.52, P = 0.0014; Figure 4D); no such relationship was observed for total AT. The baseline liver fat content was even more strongly correlated with the reduction in liver fat (r = 0.90, P < 0.0001, Figure 4E). The reduction in liver fat content was also correlated with a reduction in the AUC of glucose (r = 0.59, P = 0.0026) and insulin (r = 0.77, P < 00001) during the OGTT (Figure 4F).
In parallel to changes in body composition, fasting plasma concentrations of glucose, TG, TC, and LDL cholesterol presented small but significant reductions (P < 0.03) in the WLG upon the 12 wk of energy restriction (TABLE 1, TABLE 2). Blood pressure, leptin, and s-E-selectin also had their plasma concentrations reduced after weight loss in the WLG (Table 1).
Metabolic effects of weight loss related to insulin sensitivity and dietary quality
The effect of energy restriction on metabolic parameters and phenotypic flexibility was surprisingly small, despite the homogeneous study population and the successful weight loss. This may be due to the large difference in responses to weight loss among subjects in WLG, with weight loss ranging from 1.1% to 13.4%. In addition to this wide spectrum of responses to the weight loss intervention, some volunteers of the WLG experienced improved insulin sensitivity assessed with the MI, whereas others had no change in insulin sensitivity, despite considerable weight loss.
We investigated this phenomenon using information derived from the metabolomics analysis. To ensure that all subjects complied with the energy restriction, only volunteers who had a minimum weight loss of 5% were selected (26/40 volunteers). In the volunteers, variations in the MI after weight loss ranged from −15% to 274% and served to rank the participants into 2 groups: one including subjects with an increase of ≥35% in MI, named improvement group (n = 13), and a second group with <35% of increase or even a decrease in MI – the no-improvement group (n = 13). These 2 groups had similar changes in BW and reduction in different adipose depots in response to the intervention, losing 16%–20% of total adipose mass, but had very different changes in insulin sensitivity as measured by the MI (Table 4).
TABLE 4.
Weight loss–induced changes in anthropometric data, clinical chemistry, and the Matsuda Index in participants with or without improved insulin sensitivity during the OGTT
| Improvement |
No improvement |
P values |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Βefore interv. |
Αfter interv. |
Βefore interv. |
Αfter interv. |
Interaction | Improvement (before ×after) | No improvement (before × after) | |||||
| Αverage | SEM | Αverage | SEM | Αverage | SEM | Αverage | SEM | ||||
| Matsuda Index (0–120 min) | 2.86 | 0.35 | 4.90 | 0.50 | 3.91 | 0.65 | 3.97 | 0.57 | <0.0001 | <0.0001 | 0.971 |
| BMI (kg/m2) | 29.14 | 0.86 | 26.55 | 0.88 | 29.83 | 0.92 | 27.56 | 0.90 | 0.351 | <0.0001 | <0.0001 |
| Body weight (kg) | 83.16 | 3.15 | 75.68 | 2.90 | 86.59 | 3.40 | 79.96 | 3.20 | 0.428 | <0.0001 | <0.0001 |
| Total adipose tissue (L) | 31.91 | 2.99 | 26.59 | 2.91 | 38.55 | 3.47 | 30.46 | 2.40 | 0.872 | <0.0001 | <0.0001 |
| Subcutaneous adipose tissue (L) | 24.20 | 2.79 | 20.37 | 2.64 | 30.45 | 3.17 | 23.95 | 2.10 | 0.640 | <0.0001 | <0.0001 |
| Intra-abdominal adipose tissue (L) | 4.62 | 0.46 | 3.53 | 0.42 | 4.57 | 0.38 | 3.45 | 0.28 | 0.670 | <0.0001 | <0.0001 |
| Liver fat (arbitrary units) | 6.52 | 1.92 | 2.81 | 0.70 | 3.79 | 1.35 | 1.78 | 0.55 | 0.397 | 0.012 | 0.248 |
| Diastolic blood pressure (mmHg) | 83.23 | 2.21 | 74.85 | 2.94 | 75.46 | 2.06 | 70.50 | 2.81 | 0.441 | 0.024 | 0.246 |
| Systolic blood pressure (mmHg) | 131.92 | 3.56 | 126.31 | 2.78 | 125.31 | 2.93 | 119.33 | 3.09 | 0.960 | 0.259 | 0.248 |
| Triglycerides (mM) | 1.39 | 0.15 | 1.21 | 0.13 | 1.32 | 0.17 | 1.05 | 0.08 | 0.605 | 0.282 | 0.071 |
| LDL-cholesterol (mM) | 2.92 | 0.27 | 2.53 | 0.23 | 2.67 | 0.24 | 2.78 | 0.19 | 0.066 | 0.088 | 0.793 |
| Glucose (mM) | 5.46 | 0.14 | 5.24 | 0.13 | 5.23 | 0.10 | 5.03 | 0.09 | 0.833 | 0.064 | 0.118 |
| Total cholesterol (mM) | 5.17 | 0.31 | 4.70 | 0.24 | 4.98 | 0.20 | 4.90 | 0.19 | 0.171 | 0.048 | 0.904 |
| GGT (U/L) |
25.08 |
6.16 |
16.54 |
3.19 |
33.15 |
11.65 |
17.81 |
2.44 |
0.577 |
0.546 |
0.161 |
| Average |
SEM |
Average |
SEM |
P values t-test |
|||||||
| Matsuda index delta (absolute) | 2.04 | 0.30 | 0.05 | 0.17 | <0.0001 | ||||||
| Matsuda index delta (%) | 81.23 | 17.57 | 5.37 | 4.51 | 0.0003 | ||||||
| Body weight delta (kg) |
-7.48 |
0.82 |
-6.63 |
0.66 |
0.427 |
||||||
|
N |
N |
χ2P value |
|||||||||
| Male | 8 | 5 | 0.239 | ||||||||
| Female | 5 | 8 | |||||||||
OGTT, oral glucose tolerance test; MMTT, mixed-meal tolerance test. Data are presented as means ± SEM. N = 13 in each group. P values refer to the interaction between the terms “group” and “time” after a mixed model analysis. The P values from the multiple comparisons (Sídák) “before × after” for the Improvement group. A t-test was performed between the average values for the changes in Matsuda index and body weight. A Chi-square analysis indicates that there is no significant difference between the proportion of males and females in each group.
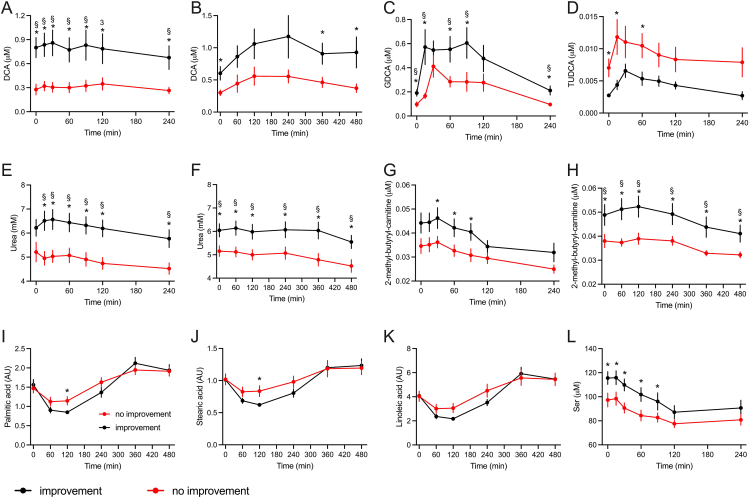
A PLS-DA model was built on results from the metabolomics analyses of plasma sampled during the MMTT and OGTT and body composition data to identify metabolic differences between the improvement and no-improvement groups. The generated model had 3 components, R2 = 0.97 and Q2 = 0.29, and did not survive cross-validation, indicating that globally there were no differences between the plasma metabolomes of these 2 groups. This outcome is probably influenced by the small number of samples in each group (n = 13). Nevertheless, the model allows the identification of metabolites at different time points during the OGTT and MMTT and body composition parameters most important for the discrimination between the 2 groups (variables with higher VIP values) (Supplemental Table 2). Urea, BA, amino acids, acylcarnitines derived from the degradation of branched-chain amino acids (BCAA), as well as different glycerophospholipids and sphingomyelins, were the most discriminating metabolites for individuals who did or did not show improved insulin sensitivity after weight loss and included glycocholate (GCA), deoxycholate (DCA), tauroursodeoxycholate (TUDCA), palmitate, stearate, linoleate, urea, 2-methyl-butyryl-carnitine, and serine (Figure 5).
FIGURE 5.
Metabolites discriminating participants with or without improved insulin sensitivity after weight loss. Data derived from the OGTT (A, C, D, E, G, and L) or mixed-meal tolerance test (MMTT) (B, F, H–K) carried out before the weight-loss intervention. DCA, deoxycholic acid; GDCA, glycodeoxycholic acid; TUDCA, tauroursodeoxycholic acid. Data are presented as means ± SEM. Differences between the 2 groups were analyzed by multiple, unpaired t-tests. False discovery rate approach by the 2-stage step-up method of Benjamin, Krieger, and Yekutieli. ∗ = P < 0.05. § = q-value < 0.05. n = 13 in each group.
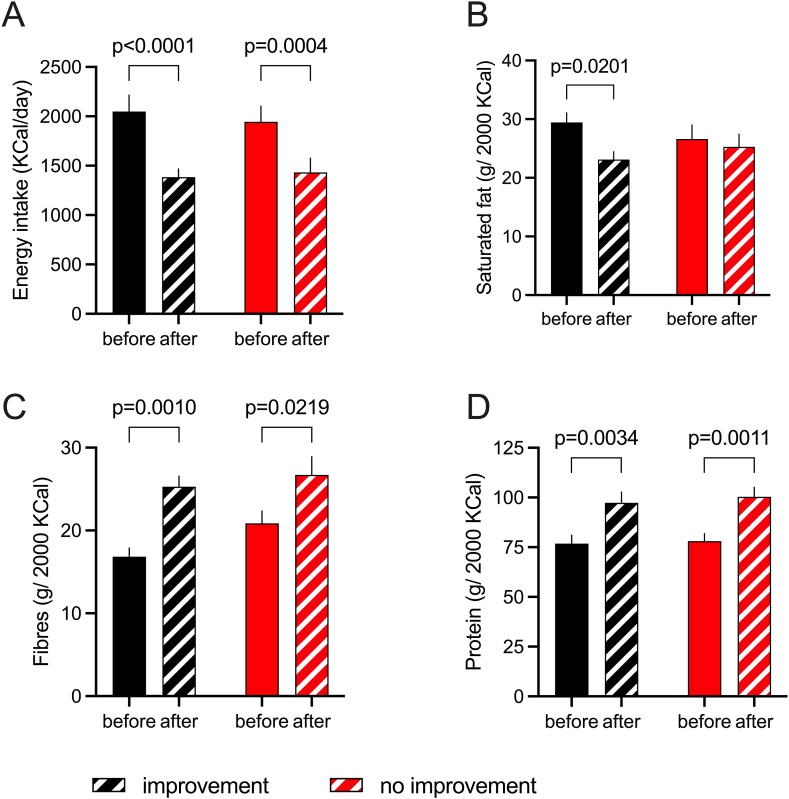
The food intake records indicate that when adjusted for total energy intake, volunteers with improved insulin sensitivity had a 20% lower intake of fiber compared with the no-improvement group before the weight loss intervention, reaching the same level of intake after the intervention, which increased in both groups (P = 0.001 in improvement compared with P = 0.0219 in no-improvement) indicating a higher chance of fiber intake in the group with improved insulin sensitivity. Moreover, only the improvement group had a 22% reduced intake of saturated fat during the intervention (P = 0.02), indicating additional improvement in diet quality (Figure 6).
FIGURE 6.
Dietary changes induced by the weight loss intervention. (A) Energy intake. (B) Intake of saturated fat. (C) Intake of fiber. (D) Protein intake. The P value of the Student’s t-test is indicated when significant. Data are presented as means ± SEM. The adjusted P value is given for statistically significant differences after a mixed model analysis followed by multiple comparisons (Sídák). n = 13 in each group.
Discussion
In the NutriTech study, whole-body MRI and plasma biomarker analyses were used to assess the effects of energy restriction on insulin sensitivity and metabolic health of overweight subjects. The use of dietary challenges aimed to assess whether changes in phenotypic flexibility could be more effectively detected by measuring postprandial markers rather than fasting samples. The outcome of this study indicates that despite an average BW loss of 6.4% in the WLG, only very few classical metabolic effects were observed. Although an estimated 60% of the cohort adhered to 75% of the dietary prescription, there were significant interindividual differences in visceral fat reduction induced by the weight loss intervention, which matches differences in response to the 2 dietary challenge tests. Another important finding was the variation in parameters that report insulin sensitivity. Although fasting insulin significantly decreased and HOMA-IR significantly increased after the intervention in the WLG during the MMTT, this was not observed during the OGTT. Moreover, the change in the postprandial estimation of insulin sensitivity by the MI following weight loss was also different between the OGTT and MMTT. This variation in response has been previously commented on by others [30]. We believe that some of the variability may be due to the individual phenotype response outlined below.
The dietary intervention resulted in an average weight loss of over 5 kg, accompanied by significant reductions in adipose mass, as well as waist and hip circumference, and ectopic fat mass. Consistent with previous studies on weight loss [[31], [32], [33]] and exercise interventions [34], the greatest reduction in AT mass was observed in visceral AT, which was associated with reductions in liver fat content. The baseline liver fat content for our overall cohort was low (mean 4.42 ± 6.64%). Although only a quarter of the subjects had liver fat greater than the level suggestive of nonalcoholic fatty liver disease, the WLG exhibited a substantial percentage change in liver fat (46%). This is comparable to results seen in other studies of weight loss through lifestyle modifications [31,32,[35], [36], [37], [38]], indicating that liver fat is responsive to negative energy balance [39]. However, the mean weight loss masks a wide variability in individual weight loss, which might have affected the group comparison.
The effects of weight loss on muscle lipid content are still debated [40], as some studies have reported a reduction in intramuscular cellular lipid (IMCL) following weight loss [31,32,41], whereas others have shown no changes [36,42].
The reduction in fasting plasma glucose was modest (−3,5% P = 0.02), and the change in fasting plasma insulin (−11%) was not statistically significant (Table 2). This may be because, despite their overweight/obese phenotype (mean BMI = 29.2 kg/m2), our middle-aged volunteers were normoglycemic according to the WHO criteria with only 3 individuals having fasting plasma glucose > 6.1 mmol/L [25].
The dietary challenges (OGTT and MMTT) performed before and after 12 wk of energy restriction revealed subtle changes in phenotypic flexibility when postprandial changes in plasma levels of glucose, insulin, and amino acids markers of insulin resistance (Ile, Leu, Val, Trp, Glu, and Ala) were considered [43]. After the weight loss intervention, the AUC of these biomarkers were more sensitive than their fasting values for detecting the beneficial effects of weight loss (Table 2 and Figure 3). It has been suggested that challenge tests can detect more subtle and earlier changes in metabolism than fasting parameters [[44], [45], [46], [47]] mostly based on studies where insulin sensitivity decreased either because of weight gain (energy excess) or in observational studies comparing different degrees of insulin sensitivity. A careful evaluation of the OGTT during a 4-wk weight gain study revealed that metabolic parameters (glucose, triglycerides, IL-6, and IL-18) remained stable in the fasting state, whereas the postprandial responses were different, revealing metabolic derailing, especially when insulin, C-peptide, and glucagon were considered [47]. In the NutriTech study as with recently published results, only subtle changes in response to the dietary challenges after weight loss were observed [39]. Possibly, the inclusion of solely healthy subjects (as assessed by HbA1c, fasting glucose, and insulin at recruitment) had an impact on the magnitude of metabolic improvements after weight loss, and in turn the assessment of phenotypic flexibility. Previously in the NutriTech study, we reported the presence of metabotypes, and we observed that 2 distinct subgroups had varying levels of phenotypic flexibility at baseline and exhibited different responses to the weight loss intervention [46]. The subgroup with reduced phenotypic flexibility at baseline was the only one to demonstrate improvements in multiple health-related biomarkers due to the weight loss intervention [46].
The weight loss intervention had its most striking effect on an individual level in the WLG, as demonstrated by the correlations between changes in various parameters, in particular, changes where fat distribution showed strong associations with changes in multiple metabolic parameters (FIGURE 2, FIGURE 4). This approach does not necessitate a control group or a homogeneous population but rather benefits from a heterogeneous population. With the emergence of extensive phenotyping, homogeneous grouping becomes challenging as stratification on primary objectives may lead to significant variations in several other intervention-related outcomes. Multi-omics studies fully utilize correlational analyses to determine the “connectivity” of the biological processes [48]. Our findings suggest that broadening inclusion criteria would enhance the strength of the correlational analyses while reducing the strength of the group-wise comparisons.
Although the number of subjects was limited, an analysis aimed at identifying a metabolic signature associated with individual susceptibility to improved metabolism following a weight loss intervention successfully identified plasma metabolites during the OGTT and MMTT that served this purpose, as demonstrated by a PLS-DA (Table 2). The BCAA-derived intermediate 2-methyl-butyryl-carnitine is among the top discriminating metabolites, displaying plasma concentrations at least 30% higher at the baseline in subjects who displayed improved insulin sensitivity after weight loss. This metabolite as well as the BCAA precursors are often reported with higher concentrations in the plasma of obese or insulin-resistant subjects [28,29]. Shah et al. [49] reported similar findings with a linear relationship between the level of plasma BCAA at baseline and improved insulin sensitivity induced by weight loss [50]. Moreover, a novel observation in our study was that plasma concentration of DCA was at least 100% higher throughout the OGTT and MMTT in participants with improved insulin sensitivity after weight loss. In these subjects, glycodeoxycholic acid (GDCA) levels were also elevated although the concentration of TUDCA was significantly lower in comparison with individuals that did not experience improved insulin sensitivity, despite having lost a similar amount of BW (Figure 5). As part of the results from the NutriTech study, our group recently described a higher concentration of DCA and GDCA during the OGTT among individuals with improved glucose homeostasis [51]. Plasma urea concentration was also among the variables most implicated in separating the 2 groups in the PLS-DA model. Urea levels were 15–20% higher in participants with improved insulin sensitivity during the weight loss intervention. It is known since the 1950s that the urease activity of gut microbiota plays an important role in nitrogen homeostasis with the microbiota consuming ∼15%–30% of all urea produced by the liver [52]. The participation of intestinal microbiota in the metabolism of BA is also known for decades, being the microbial community responsible to produce secondary BA, such as those that are elevated in the plasma of individuals with improved insulin sensitivity after weight loss. Thus, our results suggest an involvement of gut microbiota in the differential composition of the plasma metabolome among individuals with different responses to the weight loss intervention, generating a hypothesis that remains open for investigation. As a counterargument, diet could also be a determinant of urea concentration in plasma, although our results suggest that both groups of volunteers had similar protein intake before and during the intervention (Figure 6).
The analysis of dietary diaries suggests that the group with improved insulin sensitivity following weight loss was also one with a more positive change in diet quality during the intervention. This group demonstrated a larger increase in fiber intake and a pronounced reduction in the intake of saturated fat, which was not observed in the group with any improvement in insulin sensitivity (Figure 6). Although this may partly explain the better metabolic outcome in these subjects, the data set does not support the claim that better dietary quality was associated with metabolic outcomes.
As with all studies investigating the relationship between dietary change and metabolism in overweight and obese cohorts, self-reported intake should be cautiously interpreted. Although we estimated compliance with the energy restrictions in the WLG to be good, the urine metabolomics analyses suggest a careful interpretation of the diet quality. We observed a small but significant improvement in dietary quality in the WLG, which was not seen in the WMG. This may explain why the difference in metabolic outcomes was less clear than expected.
In conclusion, we demonstrate that moderate weight loss affected some metabolic parameters with a large interindividual variation. We provide novel evidence that a dietary challenge may be a sensitive tool to detect subtle changes in the metabolism induced by weight loss, offering an advantage to conventional plasma analysis after overnight fasting. OGTT was more sensitive than MMTT in detecting changes in markers of insulin sensitivity. We also observed that some participants exhibited improved insulin sensitivity whereas others did not, despite similar decreases in BW. A metabolic signature for such predisposition was proposed, suggesting that extensive phenotyping and dietary challenges are useful tools for the development of personalized nutrition. Full interpretation of complex nutritional data at an individual level, such as that presented in this study, needs new methodologies of interpreting and visualizing such as those offered by machine learning, AI, and modeling.
Acknowledgments
This project was supported by the Imperial NIHR Clinical Research Facility at Imperial College Healthcare NHS Trust. The NutriTech consortium is very thankful to Biocrates Life Sciences (Innsbruck, Austria) for the donation of the kits for LC-MS/MS-based analysis of plasma metabolites such as amino acids, glycerophospholipids, and acylcarnitines.
Author contributions
The authors’ responsibilities were as follows – GF, BO, HD, JB, ELT, LB, SW, DI, CD: designed the research program. All authors: contributed to the research leading to the findings reported here; GF, MR, JF: lead the writing of the manuscript with input from all authors. MR, JF, ELT, SW, LAA, LB, CAD, TEG, HD, IGP, JMP, DGI, JDB, BVO, GF: read and commented on the final version of the manuscript.
Conflict of interest
IG-P, JMP, and GF hold shares in Melico Sciences Ltd, and IG-P and GF are directors in the company. Melico has developed a quantitative method of assessing dietary intake using the same type of technology (NMR) as was used here to analyze urine samples. Melico was not involved in, or benefits from, this study. TEG is CEO and stock owner, and CAD is the consultant, board member, and stock owner in the analytical laboratory Vitas Ltd, Oslo, Norway.
Funding
This study was funded by the EU 7th Framework Grant number KBBE-20115-289511.
The study is registered at ClinicalTrials.gov with the registration ID NCT01684917 and can be viewed at https://clinicaltrials.gov/ct2/show/NCT01684917.
Data Availability
Data described in the manuscript, code book, and analytic code will be made available upon request to the corresponding author.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ajcnut.2023.07.002.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.van Ommen B., van der Greef J., Ordovas J.M., Daniel H. Phenotypic flexibility as key factor in the human nutrition and health relationship. Genes Nutr. 2014;9(5):423. doi: 10.1007/s12263-014-0423-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.van Ommen B., Keijer J., Heil S.G., Kaput J. Challenging homeostasis to define biomarkers for nutrition related health. Mol. Nutr. Food Res. 2009;53(7):795–804. doi: 10.1002/mnfr.200800390. [DOI] [PubMed] [Google Scholar]
- 3.Stroeve J.H.M., van Wietmarschen H., Kremer B.H.A., van Ommen B., Wopereis S. Phenotypic flexibility as a measure of health: the optimal nutritional stress response test. Genes Nutr. 2015;10(3):13. doi: 10.1007/s12263-015-0459-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.van Ommen B., Keijer J., Kleemann R., Elliot R., Drevon C.A., McArdle H., et al. The challenges for molecular nutrition research 2: quantification of the nutritional phenotype. Genes Nutr. 2008;3(2):51–59. doi: 10.1007/s12263-008-0084-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.van den Broek T.J., Baker G.C.M., Rubingh C.M., Bijlsma S., Stroeve J.H.M., van Ommen B., et al. Ranges of phenotypic flexibility in healthy subjects. Genes Nutr. 2017;12:1–14. doi: 10.1186/s12263-017-0589-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Corpeleijn E., Saris W.H.M., Blaak E.E. Metabolic flexibility in the development of insulin resistance and type 2 diabetes: effects of lifestyle: etiology and pathophysiology. Obesity Rev. 2009;10(2):178–193. doi: 10.1111/j.1467-789X.2008.00544.x. [DOI] [PubMed] [Google Scholar]
- 7.Lee S., Norheim F., Langleite T.M., Noreng H.J., Storås T.H., Afman L.A., et al. Effect of energy restriction and physical exercise intervention on phenotypic flexibility as examined by transcriptomics analyses of mRNA from adipose tissue and whole body magnetic resonance imaging. Physiol Rep. 2016;4(21):13019. doi: 10.14814/phy2.13019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Food Standards Agency . 2010. The Eatwell Guide. The Eatwell Guide. Helping you eat a healthy, balanced diet 1–12 Preprint at. [Google Scholar]
- 9.National Institutes of Health . NIH Publication Preprint at; 2006. Lowering Your Blood Pressure With DASH. [Google Scholar]
- 10.Blumenthal J.A., Babyak M.A., Sherwood A., Craighead L., Lin P.H., Johnson J., et al. Effects of the dietary approaches to stop hypertension diet alone and in combination with exercise and caloric restriction on insulin sensitivity and lipids. Hypertension. 2010;55(5):1199–1205. doi: 10.1161/HYPERTENSIONAHA.109.149153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pieper C., Redman L., Racette S., Roberts S., Bhapkar M., Rochon J., et al. Development of adherence metrics for caloric restriction interventions. Clin. Trials. 2011;8(2):155–164. doi: 10.1177/1740774511398369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Garcia-Perez I., Posma J.M., Gibson R., Chambers E.S., Hansen T.H., Vestergaad H., et al. Objective assessment of dietary patterns by use of metabolic phenotyping: a randomized, controlled, crossover trial. Lancet Diabetes Endocrinol. 2017;5(3):184–195. doi: 10.1016/S2213-8587(16)30419-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dona A.C., Jiménez B., Schäfer H., Humpfer E., Spraul M., Lewis M.R., et al. Precision high-throughput proton NMR spectroscopy of human urine, serum, and plasma for large-scale metabolic phenotyping. Anal. Chem. 2014;86(19):9887–9894. doi: 10.1021/ac5025039. [DOI] [PubMed] [Google Scholar]
- 14.Matsuda M., DeFronzo R.A. Insulin sensitivity indices obtained from oral glucose tolerance testing. Diabetes Care. 1999;22(9):1462–1470. doi: 10.2337/diacare.22.9.1462. [DOI] [PubMed] [Google Scholar]
- 15.Matthews D.R., Hosker J.P., Rudenski A.S., Naylor B.A., Treacher D.F., Turner R.C. Homeostasis model assessment: insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28(7):412–419. doi: 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]
- 16.Abdul-Ghani M.A., Matsuda M., Balas B., Defronzo R.A. Muscle and liver insulin resistance indexes derived from the oral glucose tolerance test. Diabetes Care. 2007;30(1):89–94. doi: 10.2337/dc06-1519. [DOI] [PubMed] [Google Scholar]
- 17.Bergman R.N., Phillips L.S., Cobelli C. Physiologic evaluation of factors controlling glucose tolerance in man. J. Clin. Invest. 1981;68(6):1456–1467. doi: 10.1172/jci110398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Seltzer H.S., Allen E.W., Herron A.L., Brennan M.T. Insulin secretion in response to glycemic stimulus: relation of delayed initial release to carbohydrate intolerance in mild diabetes mellitus. J. Clin. Invest. 1967;46(3):323–325. doi: 10.1172/JCI105534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Giesbertz P., Ecker J., Haag A., Spanier B., Daniel H. An LC-MS/MS method to quantify acylcarnitine species including isomeric and odd-numbered forms in plasma and tissues. J. Lipid Res. 2015;56(10):2029–2039. doi: 10.1194/jlr.D061721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tagliacozzi D., Mozzi A.F., Casetta B., Bertucci P., Bernardini S., Di Ilio C., et al. Quantitative analysis of bile acids in human plasma by liquid chromatography-electrospray tandem mass spectrometry: a simple and rapid one-step method. Clin. Chem. Lab. Med. 2003;41(12):1633–1641. doi: 10.1515/CCLM.2003.247. [DOI] [PubMed] [Google Scholar]
- 21.Fiamoncini J., Yiorkas A.M., Gedrich K., Rundle M., Alsters S.I., Roeselers G., et al. Determinants of postprandial plasma bile acid kinetics in human volunteers. Am. J. Physiol. Gastrointest. Liver Physiol. 2017;313(4):G300–G312. doi: 10.1152/ajpgi.00157.2017. [DOI] [PubMed] [Google Scholar]
- 22.Thomas E.L., Saeed N., Hajnal J.V., Brynes A., Goldstone A.P., Frost G., Bell J.D. Magnetic resonance imaging of total body fat. J. Appl. Physiol. 1998;85(5):1778–1785. doi: 10.1152/jappl.1998.85.5.1778. [DOI] [PubMed] [Google Scholar]
- 23.Thomas E.L., Hamilton G., Patel N., O’Dwyer R., Doré C.J., Goldin R.D., et al. Hepatic triglyceride content and its relation to body adiposity: a magnetic resonance imaging and proton magnetic resonance spectroscopy study. Gut. 2005;54(1):122–127. doi: 10.1136/gut.2003.036566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rico-Sanz S., Thomas E.L., Jenkinson G., Mierisová S., Iles R., Bell J.D. Diversity in levels of intracellular total creatine and triglycerides in human skeletal muscles observed by 1 H-MRS. J. Appl. Physiol. 1999;87(6):2068–2072. doi: 10.1152/jappl.1999.87.6.2068. [DOI] [PubMed] [Google Scholar]
- 25.Alberti K.G.M.M., Zimmet P.Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. 1998;15(7):539–553. doi: 10.1002/(SICI)1096-9136(199807)15:7<539::AID-DIA668>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- 26.Kelley D.E., Thaete F.L., Troost F., Huwe T., Goodpaster B.H. Subdivisions of subcutaneous abdominal adipose tissue and insulin resistance. Am. J. Physiol. Endocrinol. Metab. 2000;278(5):E941–E948. doi: 10.1152/ajpendo.2000.278.5.E941. [DOI] [PubMed] [Google Scholar]
- 27.Preis S.R., Massaro J.M., Robins S.J., Hoffmann U., Vasan R.S., Irbleck T., et al. Abdominal subcutaneous and visceral adipose tissue and insulin resistance in the Framingham heart study. Obesity. 2010;18(11):2191–2198. doi: 10.1038/oby.2010.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tai E.S., Tan M.L., Stevens R., Low Y.L., Muehlbauer M.J., et al. Insulin resistance is associated with a metabolic profile of altered protein metabolism in Chinese and Asian-Indian men. Diabetologia. 2010;53(5):757–767. doi: 10.1007/s00125-009-1637-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Newgard C.B., An J., Bain J.R., Muehlbauer M.J., Stevens R., Lein L.F., et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab. 2009;9(4):311–326. doi: 10.1016/j.cmet.2009.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tareen S.H.K., Kutmon M., de Kok T.M., Mariman E.C.M., van Baak M.A., Evelo C.T., et al. Stratifying cellular metabolism during weight loss: an interplay of metabolism, metabolic flexibility and inflammation. Sci Rep. 2020;10(1):1651. doi: 10.1038/s41598-020-58358-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Thomas E.L., Brynes A.E., Hamilton G., Patel N., Spong A., Goldin R.D., et al. Effect of nutritional counselling on hepatic, muscle and adipose tissue fat content and distribution in non-alcoholic fatty liver disease. World J Gastroenterol. 2006;12(36):5813–5819. doi: 10.3748/wjg.v12.i36.5813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Machann J., Thamer C., Stefan N., Schwenzer N.F., Kantartzis K., Häring H.U., et al. Follow-up whole-body assessment of adipose tissue compartments during a lifestyle intervention in a large cohort at increased risk for type 2 diabetes 1. Radiology. 2010;257(2):353–363. doi: 10.1148/radiol.10092284. [DOI] [PubMed] [Google Scholar]
- 33.Ross R., Dagnone D., Jones P.J., Smith H., Paddags A., Hudson R., et al. Reduction in obesity and related comorbid conditions after diet-induced weight loss or exercise-induced weight loss in men: a randomized, controlled trial. Ann. Intern. Med. 2000;133(2):92–103. doi: 10.7326/0003-4819-133-2-200007180-00008. [DOI] [PubMed] [Google Scholar]
- 34.Langleite T.M., Jensen J., Norheim F., Gulseth H.L., Tangen D.S., Kolnes K.J., et al. Insulin sensitivity, body composition and adipose depots following 12 w combined endurance and strength training in dysglycemic and normoglycemic sedentary men. Arch. Physiol. Biochem. 2016;122(4):167–179. doi: 10.1080/13813455.2016.1202985. [DOI] [PubMed] [Google Scholar]
- 35.Tiikkainen M., Bergholm R., Vehkavaara S., Rissanen A., Häkkinen A.M., Tamminen M., et al. Effects of identical weight loss on body composition and features of insulin resistance in obese women with high and low liver fat content. Diabetes. 2003;52(3):701–707. doi: 10.2337/diabetes.52.3.701. [DOI] [PubMed] [Google Scholar]
- 36.Larson-Meyer D.E., Heilbronn L.K., Redman L.M., Newcomer B.R., Frisard M.I., Anton S., et al. Effect of calorie restriction with or without exercise on insulin sensitivity, beta-cell function, fat cell size, and ectopic lipid in overweight subjects. Diabetes Care. 2006;29(6):1337–1344. doi: 10.2337/dc05-2565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sato F., Tamura Y., Watada H., Kumashiro N., Igarashi Y., Uchino H., et al. Effects of diet-induced moderate weight reduction on intrahepatic and intramyocellular triglycerides and glucose metabolism in obese subjects. J. Clin. Endocrinol. Metab. 2007;92(8):3326–3329. doi: 10.1210/jc.2006-2384. [DOI] [PubMed] [Google Scholar]
- 38.Rossi A.P., Fantin F., Zamboni G.A., Mazzali G., Zoico E., Bambace C., et al. Effect of moderate weight loss on hepatic, pancreatic and visceral lipids in obese subjects. Nutr. Diabetes. 2012;2(3):e32. doi: 10.1038/nutd.2012.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Schutte S., Esser D., Siebelink E., Michielsen C.J.R., Daanje M., Matualatupauw J.C., et al. Diverging metabolic effects of 2 energy-restricted diets differing in nutrient quality: a 12-week randomized controlled trial in subjects with abdominal obesity. Am. J. Clin. Nutr. 2022;116(1):132–150. doi: 10.1093/ajcn/nqac025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Li Y., Lee S., Langleite T., Norheim F., Pourteymour S., Jensen J., et al. Subsarcolemmal lipid droplet responses to a combined endurance and strength exercise intervention. Physiol. Rep. 2014;2(11) doi: 10.14814/phy2.12187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Petersen K.F., Dufour S., Morino K., Yoo P.S., Cline G.W., Shulma G.I. Reversal of muscle insulin resistance by weight reduction in young, lean, insulin-resistant offspring of parents with type 2 diabetes. Proc. Natl. Acad. Sci. U S A. 2012;109(21):8236–8240. doi: 10.1073/pnas.1205675109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Malenfant P., Tremblay A., Doucet E., Imbeault P., Simoneau J.A., Joanisse D.R. Elevated intramyocellular lipid concentration in obese subjects is not reduced after diet and exercise training. Am. J. Physiol. Endocrinol. Metab. 2001;280(4):E632–E639. doi: 10.1152/ajpendo.2001.280.4.E632. [DOI] [PubMed] [Google Scholar]
- 43.Wang T.J., Larson M.G., Vasan R.S., Cheng S., Rhee E.P., McCabe E., et al. Metabolite profiles and the risk of developing diabetes. Nat. Med. 2001;17(4):448–453. doi: 10.1038/nm.2307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bakker G.C., van Erk M.J., Pellis L., Wopereis S., Rubingh C.M., Cnubben N.H., et al. An antiinflammatory dietary mix modulates inflammation and oxidative and metabolic stress in overweight men: a nutrigenomics approach. Am. J. Clin. Nutr. 2010;91(4):1044–1059. doi: 10.3945/ajcn.2009.28822. [DOI] [PubMed] [Google Scholar]
- 45.Krug S., Kastenmüller G., Stückler F., Rist M.J., Skurk T., Sailer M., et al. The dynamic range of the human metabolome revealed by challenges. FASEB J. 2012;26(6):2607–2619. doi: 10.1096/fj.11-198093. [DOI] [PubMed] [Google Scholar]
- 46.Fiamoncini J., Rundle M., Gibbons H., Thomas E.L., Geillinger-Kästle K., Bunzel D., et al. Plasma metabolome analysis identifies distinct human metabotypes in the postprandial state with different susceptibility to weight loss-mediated metabolic improvements. FASEB J. 2018;32(10):5447–5448. doi: 10.1096/fj.201800330R. [DOI] [PubMed] [Google Scholar]
- 47.Kardinaal A.F.M., van Erk M.J., Dutman A.E., Stroeve J.H., van de Steeg E., Bijlsma S., et al. Quantifying phenotypic flexibility as the response to a high-fat challenge test in different states of metabolic health. FASEB J. 2015;29(11):4600–4613. doi: 10.1096/fj.14-269852. [DOI] [PubMed] [Google Scholar]
- 48.Price N.D., Magis A.T., Earls J.C., Glusman G., Levy R., Lausted C., et al. A wellness study of 108 individuals using personal, dense, dynamic data clouds. Nat. Biotechnol. 2017;35(8):747–756. doi: 10.1038/nbt.3870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Shah S.H., Crosslin D.R., Haynes C.S., Nelson S., Turer C.B., Stevens R.D., et al. Branched-chain amino acid levels are associated with improvement in insulin resistance with weight loss. Diabetologia. 2012;55(2):321–330. doi: 10.1007/s00125-011-2356-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fiamoncini J., Donado-Pestana C.M., Duarte G.B.S., Rundle M., Thomas E.L., Kiselova-Kaneva Y., et al. Plasma metabolic signatures of healthy overweight subjects challenged with an oral glucose tolerance test. Front. Nutr. 2022;9 doi: 10.3389/fnut.2022.898782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Walsert M., Bodenlost L.J. Urea metabolism in man. J. Clin. Invest. 1959;38(9):1617–1626. doi: 10.1172/JCI103940. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data described in the manuscript, code book, and analytic code will be made available upon request to the corresponding author.