Abstract
Although prior studies have documented an association between various measures of pain and mortality, none of those studies has evaluated whether the association between pain and mortality varies significantly by age. We suspect that pain—particularly pain that interferes with the ability to lead a normal life—could be an early warning sign that may portend increased risk of physical impairment and mortality later in life. In this paper, we investigated whether pain was associated with increased mortality risk, particularly in midlife. Data came from the Midlife in the US study, which sampled non-institutionalized, English-speaking adults aged 25–74 in the contiguous United States in 1995-96. Our analysis included 4041 respondents who completed a follow-up self-administered questionnaire in 2004-05, 2703 of whom completed another self-administered questionnaire in 2013-14. We modeled mortality through December 31, 2021. In demographic-adjusted models, pain interference was more strongly associated with mortality than other pain measures, and the association was stronger at younger ages. The hazard ratio for pain interference declined from 1.39 per SD (95% CI 1.26–1.54) at age 60 to 1.14 (95% CI 1.04–1.24) at age 90. Although potential confounders accounted for more than 60% of the association with premature mortality, pain interference remained significantly associated with increased mortality rates (HR = 1.13 at age 60, 95% CI 1.02–1.26). We found no evidence that the association between pain and mortality was driven by cancer. If anything, pain interference was more strongly associated with cardiovascular than cancer mortality. At the oldest ages, physical function is likely to be a better predictor of mortality than pain. Yet, pain interference may be a useful warning sign at younger ages, when there are fewer physical limitations and mortality rates are low. It may be particularly helpful in identifying risk of premature mortality in midlife, before the emergence of severe physical limitations.
Keywords: Pain, Pain interference, Chronic pain, Mortality, Death, United States
Highlights
-
•
Pain interference may help identify risk of premature mortality in midlife.
-
•
It may be a useful warning sign that appears before severe physical limitations.
-
•
Among pain measures, pain interference is most strongly associated with mortality.
-
•
The association between pain and mortality is stronger at younger than older ages.
-
•
At the oldest ages, physical function predicts mortality better than pain.
1. Introduction
Physical function is one of the best prognostic markers of short-term mortality risk among adults (Glei et al., 2016; Goldman, Glei, & Weinstein, 2016, 2017). Pain could also have prognostic value if it represents a precursor to physical limitations that occur later in life. In light of the strong relationship between pain and physical limitations, the National Pain Strategy proposed a new construct that combines pain and physical function: high-impact chronic pain is defined as persistent pain with substantial restriction of life activities for six months or more (Interagency Pain Research Coordinating Committee, 2016; Dahlhamer, 2018; Interagency Pain Research Coordinating; Von Korff et al., 2016).
We suspect that pain—in particular pain that interferes with the ability to lead a normal life—could be an early warning sign that may appear in midlife but portends increased risk of physical impairment and mortality later in life. Such an early warning sign would be valuable because it may provide an opportunity for interventions that stave off more severe health consequences that are difficult to reverse.
Prior studies have documented an association between pain and mortality (Smith, Wilkie, Croft, Parmar, et al., 2018), particularly for severe pain (Zajacova et al., 2021), multi-site pain (Chen et al., 2021), severe chronic pain (Torrance et al., 2010), widespread chronic pain (Andersson, 2009; Macfarlane, Barnish, & Jones, 2017), or extreme pain interference (Smith, Wilkie, Croft, & McBeth, 2018). However, a meta-analysis found only a modest, but not significant, association between chronic pain and all-cause mortality (Smith et al., 2014). Although the magnitude was somewhat stronger for widespread pain, the pooled estimate was still not significant (Smith et al., 2014). A subsequent meta-analysis reported a significant association between widespread pain and mortality (Macfarlane et al., 2017). Later research evaluated the association between various measures of pain and mortality in data from two English studies; they found that the relationship with mortality was strongest for extreme pain interference, whereas the associations with any pain, widespread pain, and number of pain sites were not significant net of confounders (Smith, Wilkie, Croft, & McBeth, 2018). Smith et al. (2018, p. 242) concluded: “The impact of pain was more important than the presence or extent of pain in the relationship between pain and mortality.”
To our knowledge, no prior study has evaluated whether the association between pain and mortality varies significantly by age. That is, no one has demonstrated a significant interaction between age and pain on the risk of mortality. However, one prior study (Andersson, 2009) stratified the sample into younger (25–64) versus older (65–74) persons at baseline. They found that the association between widespread chronic pain and mortality appeared to be stronger in the younger group than in the older group, but there was no indication that they tested whether that difference was significant. More importantly, their analysis did not appear to account for age as a time-varying covariate in order to evaluate the effect on mortality by age at the time of death.
Here, we use measures of pain frequency by type (headaches, backaches, joint pain/stiffness, pain in extremities), prevalence of chronic pain, and the severity of pain interference to predict mortality over 17 years among a US national sample observed at ages 30–93. We expect the association with mortality will be strongest for pain that interferes with normal activities. We evaluate whether the association between pain and mortality is stronger at younger ages, when few people exhibit physical limitations and the mortality rate is low. In addition, the association between pain and mortality is compared with the corresponding magnitude for physical limitations, which is likely to be one of the best—albeit more proximate—predictors of mortality. Finally, we investigate the extent to which the association may result from confounding with other factors that affect both pain and mortality. Is pain merely a warning signal or could it have a causal effect on mortality?
2. Methods
2.1. Data
The Midlife in the United States (MIDUS) study targeted non-institutionalized, English-speaking adults aged 25–74 in the contiguous United States (Brim et al., 2020). Details regarding the sampling strategy and response rates are provided in Text S1. At Wave 1, the original cohort included 7108 participants who completed a phone interview (fielded January 1995‒;September 1996), 6325 of whom also completed a mail-in self-administered questionnaire (SAQ). At Wave 2, 4963 completed a follow-up interview (fielded January 2004‒;August 2005) and 4041 completed the SAQ (61% of 6628 survivors from the Wave 1 cohort). At Wave 3, 3294 (55% of survivors) completed the main phone interview (fielded May 2013‒;April 2014) and 2924 completed the SAQ.
Because the measures of chronic pain and pain interference were not asked at Wave 1, we restricted our analysis to those who completed the SAQ at Wave 2 (N = 4041 respondents aged 30–84). Among those, 2703 also completed the SAQ at Wave 3 (when they were aged 39–93), yielding a total of 6744 observations.
The MIDUS study was approved by the Educational and Social/Behavioral Science institutional review board at University of Wisconsin, Madison [#SE-2011-0350]. Informed consent was obtained from all participants.
2.2. Measures
2.2.1. Mortality
Vital status was ascertained through searches of the National Death Index, survey fieldwork, and longitudinal sample maintenance (Ryff et al., 2022). To ensure the completeness of mortality follow-up, we analyzed deaths only through December 31, 2021 (see Text S2 for details). Among the analysis sample, there were 860 deaths after Wave 2; the youngest death occurred at age 42 and the oldest at age 97.
Given the total number of deaths among our analysis sample, we had limited statistical power to model cause-specific mortality. Nonetheless, we estimated auxiliary models for broad groups of causes (see Text S2 for detailed ICD-10 codes): 1) cancers (228 deaths); 2) cardiovascular disease (273 deaths); and 3) a residual category of all other causes (345 deaths).
2.2.2. Predictors
Pain measures, physical limitations, age, and chronic conditions were specified as time-varying covariates, measured first at baseline (Wave 2) and updated at Wave 3. The remaining analysis variables were measured only at baseline. Table S1 shows descriptive statistics by survey wave for all the covariates included in the analysis.
2.2.2.1. Pain of various types
Respondents were asked how often, during the past 30 days, they experienced four types of pain: 1) headaches; 2) backaches; 3) aches or stiffness in joints; and 4) pain or aches in extremities (arms/hands/legs/feet). The six response categories for each those three questions ranged from “not at all” to “almost every day.”
2.2.2.2. Chronic pain and pain interference
They were also asked: “Do you have chronic pain, that is do you have pain that persists beyond the time of normal healing and has lasted anywhere from a few months to many years?” Those who reported any chronic pain were asked about the extent to which pain interferes with various activities during the past week: 1) general activity; 2) mood; 3) relations with other people; 4) sleep; and 5) enjoyment of life. Each item was rated on an ordinal scale ranging from 0 (“did not interfere”) to 10 (“completely interfered”). These questions represent 5 of the 7 items in the Brief Pain Inventory Short Form (Cleeland, 2009a; 2009b). The severity of pain interference index was computed as the average across the 5 items (α = 0.95 at both waves).
2.2.2.3. Index of physical limitations
Respondents were asked, “How much does your health limit you in doing each of the following? Lifting or carrying groceries; climbing several flights of stairs; bending, kneeling, or stooping; walking more than a mile; walking several blocks; walking one block; vigorous activity (e.g., running, lifting heavy objects); moderate activity (e.g., bowling, vacuuming).” The response categories for each of the 8 physical tasks were coded on a four-point scale (0 = not at all, 1 = a little, 2 = some, 3 = a lot). Based on Long and Pavalko (2004), we constructed an index by summing the 8 items (potential range 0–24), adding a constant (0.5), and taking the logarithm of the result, which allows for relative rather than absolute effects.
2.2.2.4. Demographic characteristics
All models controlled for age, sex, and race/ethnicity. Age was measured at the time of the phone interview. Race and ethnicity were based on self-identification and recoded into the following categories: non-Hispanic White, non-Hispanic Black or African American, non-Hispanic other race (including American Indian or Alaska Native, Asian, Native Hawaiian, or Pacific Islander), and Hispanic.
2.2.2.5. Other potential confounders
The other potential confounders comprised marital status, a composite measure of relative socioeconomic status (SES), smoking, obesity, and various chronic conditions. Most of the confounders were measured only at baseline (Wave 2) to avoid potential endogeneity (e.g., pain at Wave 2 may have exacerbated obesity at Wave 3). The exceptions were chronic conditions, which were treated as time-varying covariates because they were likely to be a cause rather than a consequence of pain/limitations. See Text S3 for details regarding the construction of the measures.
2.3. Analytic strategy
We used standard practices of multiple imputation to handle missing data (Rubin, 1996; Schafer, 1999); see Text S4 for details. We began by examining the age pattern across various measures of pain as well as the index of physical limitations. Next, we investigated the bivariate association between pain interference and physical limitations.
Then, we fitted Cox hazard models to test the association between pain measures and mortality, using age as the time metric to estimate age-specific mortality. A robust variance estimator was used to correct for family-level clustering. In addition to age, all models controlled for sex and race/ethnicity. For comparison, we estimated the corresponding association between physical limitations and mortality, adjusted for the same demographic characteristics. In subsequent models, we adjusted sequentially for potential confounders of the association between pain interference and mortality: marital status; SES; smoking; obesity; and chronic conditions.
We tested the proportionality assumption for each of the covariates and found evidence that the hazard ratio (HR) varied significantly by age for the following covariates: non-Hispanic Black, socioeconomic status, backaches, joint pain, extremity pain, pain interference, diabetes, and heart trouble. Thus, the final models included interactions between age and those covariates.
3. Results
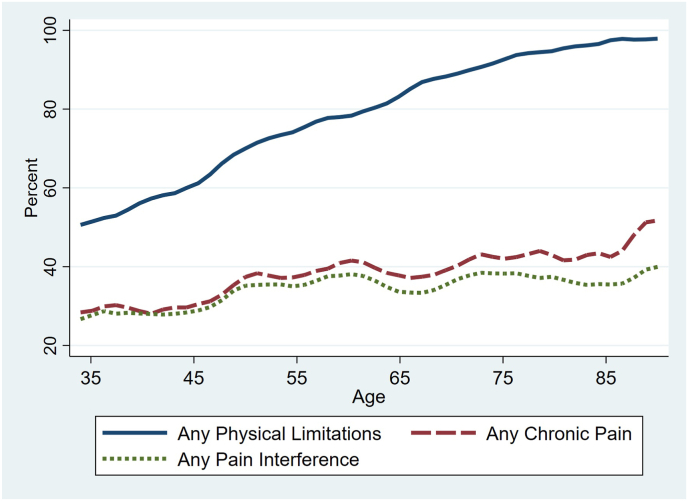
The prevalence of pain followed different age patterns depending on the type (Fig. 1). The percentage that reported any headaches declined with age, whereas backaches decreased only slightly with age and the other two types of pain (i.e., joint, extremities) increased with age, particularly between ages 40 and 50. As shown in Fig. 2, chronic pain and pain interference also rose with age, but the age-related increase was much steeper for the prevalence of a physical limitation. That is, the association with age was much weaker for pain interference than for physical limitations. For example, the correlation between age and the continuous measures of pain interference and physical limitation was only 0.02 for pain interference versus 0.40 for physical limitations.
Fig. 1.
Smoothed plot of pain prevalence (by type) across age.
Note: We plotted the percentage reporting any pain of the specified type across age for the pooled sample of observations at Waves 2 and 3 using the “lpoly” command in Stata 16.1 (StataCorp, 2019) to perform local mean smoothing—also known as the Nadaraya-Watson estimator (Nadaraya, 1964; Watson, 1964). This graph is restricted to the age range 34–90 because we have very few observations below age 34 and above age 90.
Fig. 2.
Smoothed plot of the prevalence of chronic pain, pain interference, and physical limitations across age.
Note: We plotted the percentage reporting chronic pain, pain interference, and physical limitations across age for the pooled sample of observations at Waves 2 and 3 using the “lpoly” command in Stata 16.1 to perform local mean smoothing—also known as the Nadaraya-Watson estimator (Nadaraya, 1964; Watson, 1964). This graph is restricted to the age range 34–90 because we have very few observations below age 34 and above age 90.
3.1. Relationship between pain interference and physical limitations
One-fifth of the sample reported neither pain interference nor physical limitations (Table S2), but it was higher for those aged 30–49 (36%) than for those aged 70 and older (7%). The largest share (33%) reported no pain interference but low physical limitations.
Fewer than 7% reported high levels of both pain interference and physical limitations, but that percentage increased with age: 4% at ages 30–49 versus 9% at ages 70 and older. Very few (<1%) reported high pain interference but no limitations.
Only 4% reported high limitations but no pain interference, although that value increased with age: 1% at ages 30–49 versus 9% at ages 70 and older. Among those aged 70 and older, 44% reported medium or high physical limitations, but low or no pain interference. Among that group, the percentage with at least “some” limitation was highest for vigorous activity such as running (92%), walking more than one mile (79%), bending/kneeling/stooping (65%), and climbing several flights of stairs (59%); only 40% reported at least some limitation walking one block.
3.2. Hazard models adjusted for demographic characteristics
In the demographic-adjusted model, frequency of headaches was weakly associated with mortality (HR = 1.09 per SD, 95% CI 1.01–1.17; Table S3, Model 1). As demonstrated in Fig. 3, the associations with other types of pain diminished with age. For example, the HR for extremity pain was 1.35 per SD (95% CI 1.18–1.55) at age 60, but declined to 0.98 (95% CI 0.88–1.09) by age 90.
Fig. 3.
Hazard ratios for the relative increase in mortality associated with measures of pain frequency for selected ages adjusted for demographic factors.
Note: The hazard ratios are based on the models shown in Table S3 and are plotted on the log scale. The error bars represent the 95% confidence intervals. We do not show the hazard ratios for mortality below age 60 because only 63 (7%) decedents died below that age.
Prevalence of chronic pain also exhibited a modest significant association (HR = 1.16 standardized effect size, 95% CI 1.09–1.24; Table S4, Model 5). The index for the severity of pain interference was most strongly associated with mortality at younger ages (Model 6). As shown in Fig. 4, the demographic-adjusted HR for pain interference declined from 1.39 per SD (95% CI 1.26–1.54) at age 60 to 1.14 (95% CI 1.04–1.24) at age 90. Among the pain measures, interference demonstrated the strongest relationship with mortality. Nonetheless, the association with the index of physical limitations was, by far, the strongest (HR = 1.90 per SD, 95% CI 1.69–2.12, Model 7).
Fig. 4.
Hazard ratios for pain interference by age
Note: The hazard ratios (HRs) indicate the relative increase in mortality rates per SD of pain interference and are plotted on the log scale. The demographic-adjusted HRs are based on Model 6 from Table S4, while the fully-adjusted HRs are based on Model 5 (Table 1). The error bars represent the 95% confidence intervals. We do not show the hazard ratios for mortality below age 60 because only 63 (7%) decedents died below that age.
3.3. Hazard models adjusted for additional confounders
Although pain interference predicted mortality, the relationship was not necessarily causal. The association could be spurious, resulting from confounding with other factors that affected both pain and mortality.
In Table 1, we adjusted sequentially for additional confounders. Model 1 controlled for marital status, which resulted in little change in the HR for pain interference. Model 2 further adjusted for SES, which substantially weakened the HR pain interference (1.28 at age 60, 95% CI 1.15–1.43); it also weakened the age interaction, which was no longer significant. Thus, part of the reason that pain interference was associated with premature mortality appears to be because socioeconomically disadvantaged Americans were more likely to suffer pain interference as well as higher risk of midlife mortality.
Table 1.
Hazard ratios for pain interference from Cox models predicting all-cause mortality adjusted for potential confounders.
| Model 1 | Model 2 | Model 3 | Model 4 | Model 5 | |
|---|---|---|---|---|---|
| Male | 1.510*** | 1.575*** | 1.511*** | 1.504*** | 1.411*** |
| (1.315–1.733) | (1.371–1.810) | (1.311–1.742) | (1.304–1.736) | (1.221–1.631) | |
| (Non-Hispanic White) | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| Non-Hispanic Black | 1.741 | 1.461 | 1.508 | 1.503 | 1.296 |
| (0.984–3.078) | (0.826–2.584) | (0.845–2.691) | (0.848–2.664) | (0.725–2.316) | |
| Non-Hispanic Other race | 1.435 | 1.397 | 1.341 | 1.390 | 1.201 |
| (0.969–2.125) | (0.943–2.071) | (0.906–1.984) | (0.950–2.032) | (0.803–1.794) | |
| Hispanic | 1.030 | 1.023 | 0.925 | 0.908 | 0.961 |
| (0.651–1.631) | (0.651–1.609) | (0.550–1.554) | (0.520–1.585) | (0.579–1.593) | |
| (Married) | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| Partnered | 1.079 | 1.073 | 0.950 | 0.924 | 0.896 |
| (0.734–1.588) | (0.731–1.576) | (0.646–1.396) | (0.634–1.346) | (0.622–1.292) | |
| Widowed | 1.181 | 1.117 | 1.068 | 1.067 | 1.048 |
| (0.958–1.457) | (0.908–1.375) | (0.865–1.318) | (0.861–1.321) | (0.847–1.296) | |
| Divorced/separated | 1.448*** | 1.384** | 1.251* | 1.236 | 1.254* |
| (1.163–1.802) | (1.113–1.720) | (1.007–1.554) | (0.993–1.539) | (1.004–1.566) | |
| Never married | 1.493** | 1.518** | 1.609** | 1.555** | 1.534** |
| (1.109–2.011) | (1.125–2.048) | (1.185–2.185) | (1.141–2.119) | (1.129–2.083) | |
| Socioeconomic status | NA | 0.222*** | 0.298*** | 0.321*** | 0.379*** |
| NA | (0.133–0.372) | (0.176–0.504) | (0.189–0.543) | (0.226–0.635) | |
| (Never smoked) | NA | NA | 1.000 | 1.000 | 1.000 |
| Former smoker | NA | NA | 1.236** | 1.214* | 1.177* |
| NA | NA | (1.060–1.442) | (1.041–1.417) | (1.009–1.373) | |
| Current smoker | NA | NA | 2.575*** | 2.668*** | 2.559*** |
| NA | NA | (2.077–3.192) | (2.141–3.323) | (2.061–3.178) | |
| Obesitya | NA | NA | NA | 1.191*** | 1.087* |
| NA | NA | NA | (1.103–1.286) | (1.002–1.180) | |
| Ever had cancer | NA | NA | NA | NA | 1.041 |
| NA | NA | NA | NA | (0.893–1.213) | |
| Ever had heart trouble | NA | NA | NA | NA | 2.673*** |
| NA | NA | NA | NA | (1.964–3.638) | |
| Ever had stroke | NA | NA | NA | NA | 1.769*** |
| NA | NA | NA | NA | (1.336–2.342) | |
| Diabetes | NA | NA | NA | NA | 1.597*** |
| NA | NA | NA | NA | (1.273–2.003) | |
| Lung problems | NA | NA | NA | NA | 1.405*** |
| Model 1 | Model 2 | Model 3 | Model 4 | Model 5 | |
|---|---|---|---|---|---|
| NA | NA | NA | NA | (1.178–1.677) | |
| Arthritis/rheumatism/bone/joint disease | NA | NA | NA | NA | 0.889 |
| NA | NA | NA | NA | (0.763–1.035) | |
| Severity of pain interferencea | 1.391*** | 1.284*** | 1.243*** | 1.206*** | 1.131* |
| (1.257–1.539) | (1.154–1.428) | (1.118–1.382) | (1.082–1.345) | (1.016–1.260) | |
| Interactions with (Age-60): | |||||
| Non-Hispanic Black | 0.966* | 0.972 | 0.974 | 0.973 | 0.980 |
| (0.933–1.000) | (0.939–1.006) | (0.940–1.008) | (0.941–1.007) | (0.946–1.015) | |
| Socioeconomic status | NA | 1.045*** | 1.033** | 1.031* | 1.024 |
| NA | (1.021–1.070) | (1.008–1.058) | (1.006–1.056) | (1.000–1.049) | |
| Ever had heart trouble | NA | NA | NA | NA | 0.985* |
| NA | NA | NA | NA | (0.972–0.998) | |
| Diabetes | NA | NA | NA | NA | 0.972*** |
| NA | NA | NA | NA | (0.958–0.987) | |
| Pain interferencea | 0.993** | 0.996 | 0.998 | 0.998 | 1.000 |
| (0.988–0.998) | (0.991–1.001) | (0.992–1.003) | (0.992–1.003) | (0.995–1.005) |
Note: The 95% confidence intervals are shown in parentheses below the hazard ratio. In cases where there was evidence of non-proportional hazards, we interacted the relevant variable with Age-60 so that main effect represents the hazard ratio (HR) at age 60. For example, in Model 6, the HR for pain interference at age 60 is 1.13 per SD. The corresponding HR for age x can be obtained as follows: , where is the HR for the main effect and is the HR for the interaction with age. For example, the HR for pain interference at age 85 based on Model 5 is: 1.131*0.999925 = 1.13.
Abbreviation: NA, not applicable.
Standardized (based on the pooled distribution of observations from both waves).
Model 3 added smoking, which attenuated the HR for pain interference even further (1.24 at age 60, 95% CI 1.12–1.38), but remained statistically discernible. The addition of obesity in Model 4 furtherweakened the HR for pain interference (1.21 at age 60, 95% CI 1.08–1.35).
Finally, Model 5 adjusted for chronic conditions. Net of all these potential confounders, the HR for pain interference was substantially reduced (1.13 at age 60, 95% CI 1.02–1.26). As shown in Fig. 4, there was virtually no change over age in the HR for pain interference based on the fully-adjusted model. These results suggest that more than 60% of the association between pain interference and mortality at younger ages was a result of confounding with other factors (i.e., SES, smoking, obesity, and chronic conditions).
3.4. Cause-specific mortality
In auxiliary analyses, we investigated the association between pain interference and mortality from broad causes of death. In the demographic-adjusted model (Table S5, Model 1), the association with pain interference appeared to be slightly stronger for cardiovascular (HR = 1.30 per SD, 95% CI 1.17–1.44) than for cancer (HR = 1.22, 95% CI 1.08–1.39) or the residual category (HR = 1.22, 95% CI 1.11–1.34). In the fully-adjusted model (Model 2), the HR for pain interference was substantially reduced and no longer significant for cancer and the residual category. However, the association remained significant for cardiovascular mortality (HR = 1.18, 95% CI 1.05–1.32). Confounders accounted for more than half of the association with cancer mortality and nearly half of the association with mortality from the residual set of causes, but only about one-third of the association with cardiovascular mortality.
4. Discussion
Pain interference was a notable warning signal of heightened mortality risk, particularly in midlife. The association was stronger than the corresponding relationship with the frequency of different types of pain or a binary indicator of chronic pain.
Some researchers have suggested that the association between pain and mortality may be driven by cancer (Smith et al., 2014), but we found no evidence of that. If anything, pain interference was more strongly associated with cardiovascular deaths than cancer mortality. This result was consistent with prior work suggesting that severe chronic pain was more strongly associated with cardiovascular mortality—particularly for deaths resulting from ischemic heart disease—than all-cause mortality (Torrance et al., 2010).
The weak relationship between age and pain interference may reflect age-related changes in people's expectations for pain and normal activity. At older ages, people may have adjusted their activity levels to accommodate pain and physical limitations. Someone in their 80s is likely to have some difficulty running, kneeling, or climbing stairs, but it may not interfere with their normal daily life because they avoid those activities.
Compared with pain interference, physical limitations were more strongly associated with mortality, probably because they were more proximate. However, pain interference could represent a precursor to physical limitations that do not emerge until later in life. The risk of mortality is low in midlife, but those early deaths can have an undue influence on life expectancy. An early warning signal could be valuable if that information can be used to identify modifiable factors that might delay mortality.
4.1. Warning signal vs. causal factor?
There is a difference between treating pain as a warning signal versus trying to establish that pain has a causal effect on mortality. Our results suggested that more 60% the association between pain interference and premature mortality resulted from confounding with SES, smoking (which is potentially modifiable), obesity, and chronic conditions. Nonetheless, in the fully-adjusted model, pain interference remained significantly associated with higher mortality rates. It may be particularly helpful in identifying those at risk of premature mortality in midlife, before the emergence of severe physical limitations that are difficult to reverse.
Pain interference may contribute not only to premature mortality, but also to other adverse outcomes. One study found that pain interference at age 29 increased hazards of subsequent labor force exit and health-related work limitation (Pooleri, Yeduri, Horne, Frech, & Tumin, 2023). Another study found pain interference predicted injurious falls (Cai, Leveille, Shi, Chen, & You, 2020).
4.2. If the effect is causal, what is the possible mechanism?
Zajacova et al. (2021) suggested that the mechanisms linking pain and mortality could include use of pain medication (Inoue, Ritz, & Arah, 2022) as well as the effects of pain on allostatic load, immune system suppression, and impairment of growth and tissue repair (Gatchel, Peng, Peters, Fuchs, & Turk, 2007). Torrance et al. (2010) argued that the stress could be the mechanism linking severe pain causes with mortality from ischemic heart disease: severe chronic pain induces elevated cortisol and other manifestations of the stress response, which accelerate the atheroscelerotic process.
4.3. Limitations
There are other factors that may be associated with both pain and mortality risk, but it is unclear whether they represent confounders or mediators. For example, physical activity, perceived stress, social activity, drug/alcohol abuse, and sleep quality are likely to be influenced by pain levels, which would make them mediators. However, the association could also be bidirectional (e.g., stress exposure and drug abuse could heighten pain sensitivity). Unfortunately, we could not disentangle the direction of the effects. If we wanted to adjust for these variables as confounders, we would need measures much earlier in life before pain and physical limitations were evident. If we adjusted for these variables at baseline, they would probably attenuate the association between pain interference and mortality, but we would have no way of determining whether that was because of confounding or because they acted as mediators. Pain could represent a root cause that leads to other problems, which might be more proximate determinants of mortality.
5. Conclusion
At the oldest ages, physical function is likely to be a better predictor of mortality than pain. Yet, pain interference may be a useful warning sign at younger ages, when there are fewer physical limitations. It could signal the need to look for other underlying problems that heighten mortality risk. Feel like the pain is killing you? It just might be.
CRediT author statement
D.A. Glei: Conceptualization, Methodology, Software, Formal Analysis, Investigation, Data Curation, Writing – Original Draft, Visualization. M. Weinstein: Resources, Writing – Review & Editing, Supervision, Project, Funding Acquisition.
Declaration of interests
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Provided consulting services to the University of California-Berkeley, the University of California-Riverside, and Rose Li Associates (DAG).
Acknowledgements
This work was supported by the National Institute on Aging [grant numbers P01 AG020166, U19AG051426, U19AG051426-06A1] and the Graduate School of Arts and Sciences, Georgetown University. The sponsor played no role in the study design; in the collection, analysis, and interpretation of the data; in the writing of the report; and in the decision to submit the manuscript for publication.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ssmph.2023.101513.
Contributor Information
Dana A. Glei, Email: dag77@georgetown.edu.
Maxine Weinstein, Email: weinstma@georgetown.edu.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
Data availability
The data are publicly available from https://www.icpsr.umich.edu/web/ICPSR/series/203. The most recent mortality update is available from https://midus.colectica.org.
References
- Andersson H.I. Increased mortality among individuals with chronic widespread pain relates to lifestyle factors: A prospective population-based study. Disability & Rehabilitation. 2009;31(24):1980–1987. doi: 10.3109/09638280902874154. [DOI] [PubMed] [Google Scholar]
- Brim O.G., Baltes P.B., Bumpass L.L., Cleary P.D., Featherman D.L., Hazzard W.R., et al. 2020. National survey of midlife development in the United States (MIDUS 1), 1995-1996: Description of MIDUS samples. Inter-university Consortium for Political and Social Research [distributor], Version 19. [DOI] [Google Scholar]
- Cai Y., Leveille S.G., Shi L., Chen P., You T. Chronic pain and risk of injurious falls in community-dwelling older adults. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences. 2020;76(9):e179–e186. doi: 10.1093/gerona/glaa249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L., Ferreira M.L., Nassar N., Preen D.B., Hopper J.L., Li S., et al. Association of chronic musculoskeletal pain with mortality among UK adults: A population-based cohort study with mediation analysis. EClinicalMedicine. 2021;42 doi: 10.1016/j.eclinm.2021.101202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cleeland C.S. The University of Texas MD Anderson Cancer Center; 2009. The Brief pain inventory: User guide.https://www.mdanderson.org/documents/Departments-and-Divisions/Symptom-Research/BPI_UserGuide.pdf [Google Scholar]
- Cleeland C.S. 2009. Brief pain inventory (BPI). MD anderson cancer center.https://www.mdanderson.org/research/departments-labs-institutes/departments-divisions/symptom-research/symptom-assessment-tools/brief-pain-inventory.html [Google Scholar]
- Dahlhamer J. Prevalence of chronic pain and high-impact chronic pain among adults—United States, 2016. MMWR. Morbidity and Mortality Weekly Report. 2018;67 doi: 10.15585/mmwr.mm6736a2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gatchel R.J., Peng Y.B., Peters M.L., Fuchs P.N., Turk D.C. The biopsychosocial approach to chronic pain: Scientific advances and future directions. Psychological Bulletin. 2007;133(4):581–624. doi: 10.1037/0033-2909.133.4.581. [DOI] [PubMed] [Google Scholar]
- Glei D.A., Goldman N., Risques R.A., Rehkopf D.H., Dow W.H., Rosero-Bixby L., et al. Predicting survival: Telomere length versus conventional predictors. PLoS One. 2016;11(4) doi: 10.1371/journal.pone.0152486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman N., Glei D.A., Weinstein M. What matters most for predicting survival? A multinational population-based cohort study. PLoS One. 2016;11(7) doi: 10.1371/journal.pone.0159273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman N., Glei D.A., Weinstein M. The best predictors of survival: Do they vary by age, sex, and race? Population and Development Review. 2017;43(3):541–560. doi: 10.1111/padr.12079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoue K., Ritz B., Arah O.A. Causal effect of chronic pain on mortality through opioid prescriptions: Application of the front-door formula. Epidemiology. 2022;33(4):572–580. doi: 10.1097/EDE.0000000000001490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Interagency Pain Research Coordinating Committee . US Department of Health and Human Services, National Institutes of Health; 2016. National pain strategy—a comprehensive population health -level strategy for pain; p. 84. [Google Scholar]
- Long J.S., Pavalko E. Comparing alternative measures of functional limitation. Medical Care. 2004;42(1):19–27. doi: 10.1097/01.mlr.0000102293.37107.c5. [DOI] [PubMed] [Google Scholar]
- Macfarlane G.J., Barnish M.S., Jones G.T. Persons with chronic widespread pain experience excess mortality: Longitudinal results from UK Biobank and meta-analysis. Annals of the Rheumatic Diseases. 2017;76(11):1815–1822. doi: 10.1136/annrheumdis-2017-211476. [DOI] [PubMed] [Google Scholar]
- Nadaraya E.A. On estimate regression. Theory of Probability and Its Applications. 1964;9:141–142. Journal Article. [Google Scholar]
- Pooleri A., Yeduri R., Horne G., Frech A., Tumin D. Pain interference in young adulthood and work participation. Pain. 2023;164(4):831. doi: 10.1097/j.pain.0000000000002769. [DOI] [PubMed] [Google Scholar]
- Rubin D.B. Multiple imputation after 18+ years (with discussion) Journal of the American Statistical Association. 1996;91:473–489. Journal Article) [Google Scholar]
- Ryff C., Almeida D., Ayanian J., Binkley N., Carr D.S., Coe C., et al. 2022. Documentation of mortality statistics and cause of death codes for core (non-refresher) MIDUS and Milwaukee samples.https://midus-study.github.io/public-documentation/Mortality/Core/MIDUS_Core_DocumentationOfMortality_20220316.pdf [Google Scholar]
- Schafer J.L. Multiple imputation: A primer. Statistical Methods in Medical Research. 1999;8(1):3–15. doi: 10.1177/096228029900800102. [DOI] [PubMed] [Google Scholar]
- Smith D., Wilkie R., Croft P., McBeth J. Pain and mortality in older adults: The influence of pain phenotype. Arthritis Care & Research. 2018;70(2):236–243. doi: 10.1002/acr.23268. [DOI] [PubMed] [Google Scholar]
- Smith D., Wilkie R., Croft P., Parmar S., McBeth J. Pain and mortality: Mechanisms for a relationship. Pain. 2018;159(6):1112. doi: 10.1097/j.pain.0000000000001193. [DOI] [PubMed] [Google Scholar]
- Smith D., Wilkie R., Uthman O., Jordan J.L., McBeth J. Chronic pain and mortality: A systematic review. PLoS One. 2014;9(6) doi: 10.1371/journal.pone.0099048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- StataCorp . StataCorp LLC; 2019. Stata: Release 16. Statistical software. [Google Scholar]
- Torrance N., Elliott A.M., Lee A.J., Smith B.H. Severe chronic pain is associated with increased 10 year mortality. A cohort record linkage study. European Journal of Pain. 2010;14(4):380–386. doi: 10.1016/j.ejpain.2009.07.006. [DOI] [PubMed] [Google Scholar]
- Von Korff M., Scher A.I., Helmick C., Carter-Pokras O., Dodick D.W., Goulet J., et al. United States national pain strategy for population research: Concepts, definitions, and pilot data. The Journal of Pain. 2016;17(10):1068–1080. doi: 10.1016/j.jpain.2016.06.009. [DOI] [PubMed] [Google Scholar]
- Watson G. Smooth regression analysis. Sankhya - Series A. 1964;26:359–372. Journal Article) [Google Scholar]
- Zajacova A., Grol-Prokopczyk H., Zimmer Z. Sociology of chronic pain. Journal of Health and Social Behavior. 2021 doi: 10.1177/00221465211025962. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data are publicly available from https://www.icpsr.umich.edu/web/ICPSR/series/203. The most recent mortality update is available from https://midus.colectica.org.