Abstract
Background:
Angiotensin II dependent hypertension (AngII-HTN) causes comparable elevations of blood pressure (BP), aldosterone levels and renal epithelial Na+ channel (ENaC) activity in male and female rodents. Mineralocorticoid receptor (MR) antagonism has a limited antihypertensive effect associated with insufficient suppression of renal ENaC in male rodents with AngII-HTN. While MR blockade effectively reduces BP in female mice with salt-sensitive and leptin-induced hypertension, MR antagonism has not been studied in female rodents with AngII-HTN. We hypothesize that overstimulation of renal MR signaling drives redundant ENaC-mediated Na+ reabsorption and BP increase in female rats with AngII-HTN.
Methods:
We employ a combination of physiological, pharmacological, biochemical and biophysical approaches to compare the effect of MR inhibitors on BP and ENaC activity in AngII-infused male and female Sprague Dawley rats.
Results:
MR blockade markedly attenuates AngII-HTN in female rats but has only a marginal effect in males. Spironolactone increases urinary sodium excretion and urinary sodium-to-potassium ratio in AngII-infused female, but not male, rats. The expression of renal MR and 11β-hydroxysteroid dehydrogenase type 2 that determines the availability of MR to aldosterone is significantly higher in AngII-infused female rats than in males. ENaC activity is approximately two times lower in spironolactone-treated AngII-infused female rats than in males. Reduced ENaC activity in AngII-infused female rats on spironolactone correlates with increased interaction with ubiquitin ligase Nedd4-2, targeting ENaC for degradation.
Conclusions:
MR-ENaC axis is the primary determinant of excessive renal sodium reabsorption and an attractive antihypertensive target in female rats with AngII-HTN, but not in males.
Keywords: aldosterone, ENaC, sodium reabsorption, mineralocorticoid, angiotensin II, hypertension, sex differences
Graphical Abstract

Introduction
Despite the abundance of available therapeutic options hypertension remains a global public health challenge and a major risk factor for cardiovascular and chronic kidney disease1,2. Elevation of BP results from intricate interactions between various environmental and genetic factors3. Due to the complex nature of hypertension, there is no single effective treatment. Identification of the pathological mechanisms underlying hypertension in specific cohorts would allow optimizing treatment by matching the therapeutics to the disease mechanism. This strategy is imperative to reduce the burden of hypertension.
Biological sex is now recognized as a pivotal factor affecting BP control. Males and females employ the same systems to regulate BP, yet considerable sex-specific variations in the levels of volume regulatory hormones, expression and activity of renin-angiotensin-aldosterone system components and other determinants of cardiovascular homeostasis have been reported4–7. A better understanding of sex differences pertaining to BP regulation would improve the effectiveness of antihypertensive regimen in both sexes.
The kidney plays a central role in chronic BP control through regulation of sodium reabsorption. Fine-tuning of renal Na+ transport takes place in the aldosterone-sensitive distal nephron (ASDN). ENaC is a key Na+ transporter in this nephron segment and the activity of the channel is under tight control by volume regulatory hormones, such as aldosterone8,9. Excessive ENaC-dependent renal Na+ reabsorption has been clearly linked to volume expansion and hypertension10–12. Sex disparities in renal ENaC expression, circulating aldosterone levels and aldosterone signaling have been reported5,13–15. However, the interplay between biological sex, renal ENaC and aldosterone in hypertension remains to be fully understood.
Cumulative evidence suggests that aldosterone antagonism may be more effective at reducing hypertension in females than in males4,7,16–18. However, the underlying mechanisms mediating this remain unclear. Inhibition of the aldosterone-MR axis is insufficient to effectively suppress ENaC activity and excessive Na+ reabsorption in male mice and rats with AngII-dependent hypertension19. L-NAME/AngII-induced hypertension in female rats, salt sensitive BP increase in Balb/C and obesity-induced hypertension in agouti yellow hyperleptinemic female mice are characterized by inappropriately elevated aldosterone levels4,7,17. MR blockade reduces BP in hypertensive female mice4,17 and prevents cardiovascular and renal damage in rats7. Nevertheless, the effect of MR blockade on ENaC-dependent Na+ reabsorption has never been tested in hypertensive female rodents.
In this study, we test the hypothesis that MR activation is the primary determinant of ENaC-dependent renal Na+ reabsorption and a dominant BP regulatory mechanism in AngII-infused female, but not male, Sprague Dawley (SD) rats. To test this hypothesis at the molecular, cellular and systemic levels, we combined patch-clamp electrophysiology, immunoblotting, quantitative PCR with metabolic cage studies and BP assessment. Our findings indicate that MR blockade is more effective at reducing ENaC-mediated sodium retention and lowering BP in female rats with AngII-HTN, when compared to males. Our study reconciles the existing data on poor efficacy of MR antagonism in male rodent models with the available reports showing that females can benefit from MR inhibition. This suggests that MR antagonism may be the preferred therapeutic intervention to optimize treatment of hypertension in females.
Methods
Experimental animals
Male and female SD rats were used in all studies. All animal procedures were approved by the Institutional Animal Care and Use Committee of the Medical College of Georgia at Augusta University (AUP 2017-0844) and were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
BP monitoring
BP telemetry was performed, as described earlier20, in 4 groups of SD rats: 1) AngII-infused males (MA), 2) AngII-infused males on spironolactone (MAS), 3) AngII-infused females (FA), 4) AngII-infused females on spironolactone (FAS). To exclude off-target effects of spironolactone, the antihypertensive effect of MR blockade was validated by treatment with a more specific MR antagonist, eplerenone. BP was measured on a weekly basis by tail-cuff plethysmography in the rats randomized into 4 groups: 1) AngII-infused males (MA), 2) AngII-infused males on eplerenone (MAE), 3) AngII-infused females (FA), 4) AngII-infused females on eplerenone (FAE).
Statistical analysis
Results are presented as mean ± SEM. Following the published guidelines for reporting statistics21, longitudinal BP data were analyzed by a two-way mixed-design ANOVA with the Greenhouse-Geisser correction and the endpoint data – by a two-way ANOVA. Pairwise comparisons to identify significant differences between specific group means were performed by the Holm-Sidak multiple comparison test. P<0.05 was considered significant. Statistical analysis was performed using OriginPro 2022 (OriginLab Corp., Northampton, MA, USA).
Further details pertaining to animal studies, experimental procedures and statistical analysis are provided in the Online Supplement.
Data supporting the findings of this study are available from the corresponding author upon a reasonable request.
Results
MR blockade results in a pronounced antihypertensive effect in female AngII-infused rats, but not in males
Consistent with the existing data6, infusion of AngII at 400 ng·kg−1·min−1 for 2 weeks resulted in a significant elevation of mean arterial pressure (MAP) in male SD rats from 111±1 to 173±7 mmHg (Fig. 1A). Males, administered with an aldosterone antagonist, spironolactone (30 mg·kg−1·day−1 in drinking water), exhibited a comparable increase of MAP (111±2 to 165±7 mmHg). MAP was not significantly different between the two groups. In contrast, the magnitude of AngII-driven MAP elevation was approximately 2 times lower in female SD rats supplemented with spironolactone (from 104±2 to 128±8 mmHg), when compared to the vehicle-treated AngII-infused females (from 104±1 to 156±4 mmHg, Fig. 1B). Significant differences in MAP between vehicle- and spironolactone-treated AngII-infused females were observed after 6 days of treatment. AngII infusion blunted the diurnal rhythmicity of MAP in females and administration of spironolactone resulted in a noticeable improvement of the diurnal MAP rhythm, reaching statistical significance at the end of the 14-day observation period (Figure S1 C, D). In contrast, spironolactone did not affect the amplitude of the diurnal MAP rhythm in AngII-infused males (Figure S1 A, B). Thus, spironolactone markedly attenuated AngII-induced hypertension in female rats, but not in males.
Figure 1. MR blockade attenuates hypertension in AngII-infused female rats, but not in males.
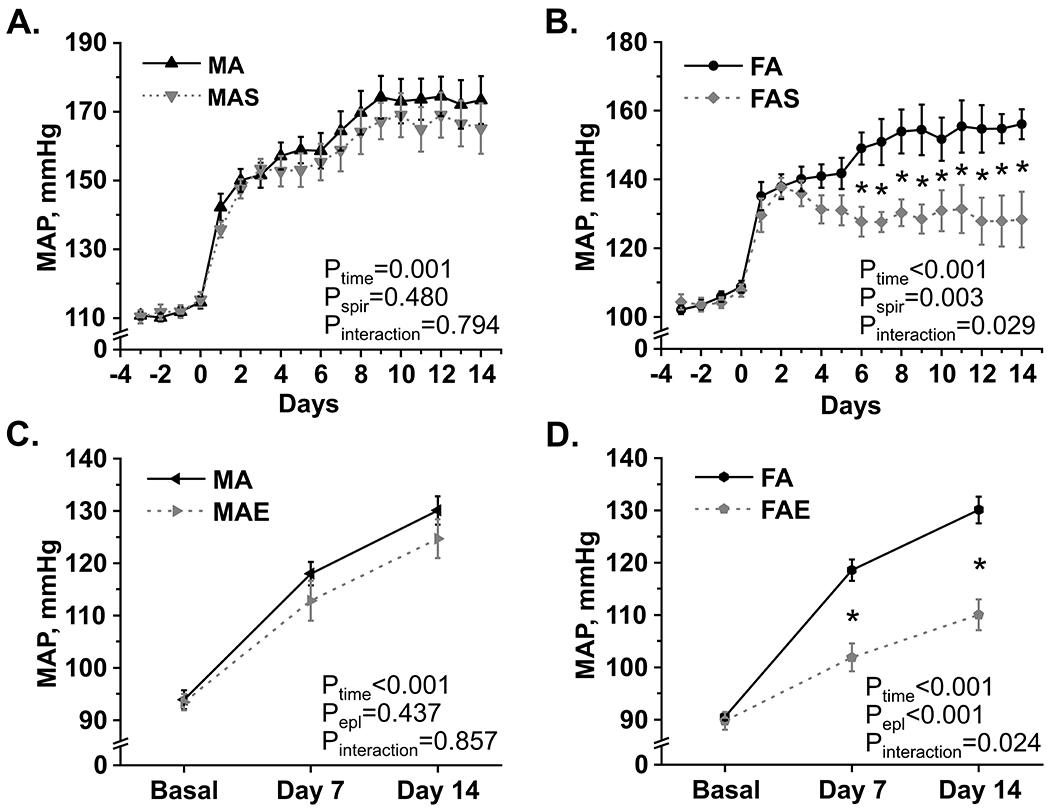
(A, B) Daily mean arterial pressure (MAP) averages recorded by telemetry at baseline and after AngII infusion in male and female SD rats treated with vehicle (MA, FA) or spironolactone (MAS, FAS). N=9 per group. Data were analyzed by a two-way mixed-design ANOVA. * – denotes P<0.05 for pairwise comparisons using the Holm-Sidak multiple comparisons test. (C, D) Daily mean arterial pressure (MAP) averages recorded by tail-cuff plethysmography at baseline, day 7 and day 14 of AngII infusion in male and female SD rats treated with vehicle (MA, FA) or eplerenone (MAE, FAE). N=8 per group. Data were analyzed by a two-way mixed-design ANOVA. * – denotes P<0.05 for pairwise comparisons using the Holm-Sidak post hoc test.
To circumvent potential progestational and antiandrogenic effects of spironolactone, we verified our findings in AngII-infused rats treated with eplerenone, a more selective MR antagonist. After 2 weeks of treatment, MAP increase was significantly blunted in AngII-infused females receiving eplerenone, when compared to vehicle-treated AngII-infused females (110±3 vs 130±3 mmHg, respectively, after 2 weeks of treatment). This difference became significant after 1 week of treatment (102±3 vs 119±2 mmHg, Fig. 1D). MAP was only marginally lower in AngII-infused males on eplerenone than in vehicle-treated male rats (125±4 vs 130±3 mmHg, respectively, after 2 weeks, Fig. 1C). Thus, the antihypertensive effect of MR blockade is considerably more pronounced in AngII-infused female rats, when compared to males.
Spironolactone increases urinary Na:K ratio in AngII-infused female rats, but not in males
Distal nephron, responsible for final adjustments to Na+ reabsorption and K+ secretion, is a universally recognized site affected by aldosterone antagonism. Thus, next we assessed if spironolactone alters urinary Na+ and K+ excretion in AngII-infused SD rats of both sexes. MR blockade did not appreciably alter renal Na+ excretion in AngII-infused males. Urinary Na+ excretion was significantly higher in spironolactone-treated AngII-infused females when compared to vehicle-treated females or males on spironolactone (Fig. 2A). Importantly, AngII-infused females on spironolactone maintain urinary Na+ excretion at significantly lower BP, indicative of improved pressure-natriuresis response. Consistent with the recognized potassium-sparing effect of MR inhibitors, K+ excretion was moderately, but significantly lower in AngII-infused rats receiving spironolactone (Pspir=0.047). AngII-infused females excreted more K+ in urine when compared to males, regardless of spironolactone intervenion (Fig. 2B). The effect of spironolactone on urinary Na+ to K+ ratio, indicative of electrolyte transport in the distal nephron, was significant (Pspir=0.013) and strongly dependent on the animal’s sex (Pinteraction=0.001; Fig. 2C). Na+:K+ ratio was significantly higher in AngII-infused females on spironolactone than in vehicle-treated females (P<0.001). Administration of spironolactone resulted in a marginal elevation of serum K+ levels in AngII-infused rats of both sexes that did not reach statistical significance and did not appreciably affect serum Na+ and Cl- in either sex (Table 1). MR blockade did not change body weight or gross readouts of renal function, such as kidney to body weight ratio and serum creatinine. While spironolactone did not improve microalbuminuria, we found that urinary albumin levels were significantly lower in AngII-infused females than in males (Table 1). Overall, a stronger antihypertensive effect of MR antagonism in AngII-infused females is, at least in part, determined by improved electrolyte handling by the kidney.
Figure 2. Spironolactone is more effective at improving renal electrolyte handling in AngII-infused females than in males.

Endpoint (day 14) urinary sodium excretion, UNaV (A); urinary potassium excretion, UKV (B), and urinary Na+ to K+ ratio (C) in AngII-infused male and female SD rats treated with vehicle (MA and FA) or spironolactone (MAS and FAS). N=8 per group. The results of a two-way ANOVA are summarized below the graphs. * – indicates a significant difference (P<0.05) between respective group means determined by the Holm-Sidak test.
Table 1. Endpoint physiological readouts for the experimental rats of both sexes.
A two-way ANOVA was used for statistical comparisons. Data are presented as mean values ± the standard error of mean (SEM). AngII – AngII-infused rats receiving vehicle for 14 days, AngII + Spir – AngII-infused rats administered with spironolactone for 14 days. N≥8 for each group.
| Parameter | Males | Females | Significance | ||||
|---|---|---|---|---|---|---|---|
| AngII | AngII+spir | AngII | AngII+spir | Psex | Pspir | Pinteraction | |
| Body weight, g | 323±7.8 | 322±5.8 | 228±4.2 | 232±2.2 | <0.001 | 0.826 | 0.665 |
| Total kidney to body weight, % | 0.72±0.01 | 0.74±0.01 | 0.71±0.02 | 0.71±0.02 | 0.225 | 0.560 | 0.710 |
| Serum creatinine, mg/dL | 0.26±0.03 | 0.26±0.03 | 0.25±0.03 | 0.24±0.03 | 0.574 | 0.800 | 0.912 |
| Urinary albumin, mg/dL·24h | 20.6±4.1 | 24.0±4.0 | 4.28±1.94 | 4.19±1.12 | <0.001 | 0.648 | 0.518 |
| Serum Na+, mmol/L | 138±0.7 | 139±0.6 | 136±0.5 | 138±0.6 | 0.005 | 0.115 | 0.409 |
| Serum K+, mmol/L | 5.1±0.23 | 5.4±0.24 | 5.2±0.2 | 5.3±0.24 | 0.159 | 0.787 | 0.900 |
| Serum Cl-, mmol/L | 96±0.7 | 96±0.7 | 96±0.8 | 98±0.95 | 0.220 | 0.178 | 0.174 |
Renal expression of MR and 11β-hydroxysteroid dehydrogenase type 2 is higher in AngII-infused females than in males
Cumulative experimental evidence identifies excessive circulating aldosterone levels and elevated MR expression in the tissues as critical sex-specific determinants of hypertension. Consistent with the previous report6, we found that serum aldosterone concentrations are significantly higher in AngII-infused male and female SD rats (~3000 pg/ml) than those typically observed at baseline without AngII-infusion (<100 pg/ml). After 2 weeks of AngII infusion circulating aldosterone levels are comparable in AngII-infused rats of both sexes (Fig. 3A). Interestingly, quantitative RT-PCR demonstrated that expression of mRNA encoding MR (Nr3c2) was significantly higher in the kidneys isolated from AngII-infused females, when compared to males (Psex=0.001; Fig. 3B), regardless of spironolactone intervention. The availability of MR to aldosterone in the ASDN is controlled by an enzyme, 11β-hydroxysteroid dehydrogenase type 2 (HSD11β2), that inactivates highly abundant corticosterone and protects MR from binding to glucocorticoids. We found that Hsd11b2 expression is significantly higher in AngII-infused female rats than in males (Psex<0.001; Fig. 3C). This points to a higher sensitivity of renal epithelium to stimulation by aldosterone or MR antagonism in female rats with AngII-dependent hypertension, when compared to males.
Figure 3. Renal expression of mRNA encoding MR and hydroxysteroid 11β-hydroxysteroid dehydrogenase type 2 is higher in females with AngII-induced hypertension, while circulating aldosterone levels are comparable in both sexes.

Endpoint serum aldosterone concentrations (A), relative expression of Nr3c2 mRNA, encoding MR (B), and Hsd11b2 mRNA (C) in the renal tissue isolated from male and female AngII-infused rats receiving vehicle (MA and FA) or spironolactone (MAS and FAS) for 2 weeks. Relative mRNA expresion values were normalized to that in AngII-infused vehicle-treated males. The results of a two-way ANOVA are summarized below the graphs. * – indicates a significant difference (P<0.05) between respective group means determined by a post-hoc Holm-Sidak test. N≥6 per group.
Spironolactone is more effective at reducing ENaC activity in AngII-infused females, when compared to males
We have previously reported that ENaC activity was similar in SD males and females at baseline conditions, and almost tripled after AngII infusion without any apparent sex differences6. Our patch-clamp experiments in freshly isolated split-open ASDNs revealed that spironolactone inhibited ENaC-mediated currents in AngII-infused female rats to a significantly larger extent than in males (Fig. 4). ENaC activity was significantly lower in AngII-infused female rats on spironolactone, when compared to the respective male group (fNPo of 0.37±0.06 vs 0.75±0.08, P=0.002, Fig. 4A). This difference was associated with significantly reduced levels of functional channels on the membrane (fN of 1.11±0.10 vs 1.76±0.15, P=0.007, Fig. 4B) and ENaC open probability (Po of 0.27±0.02 vs 0.41±0.03, P<0.001, Fig. 4C) in spironolactone-treated females when compared to males. Consistently, α-ENaC subunit reporting signal is markedly lower in ASDNs isolated from spironolactone-treated AngII-infused females than in males (Fig. 4E). Moreover, in spironolactone-treated females α-ENaC is retained inside the cells, further away from the luminal surface, strongly indicative of decreased channel functionality. Overall, we conclude that renal ENaC is more sensitive to MR blockade in AngII-infused females than in males.
Figure 4. ENaC activity and α-ENaC subunit abundance are significantly lower in the collecting ducts isolated from AngII-infused females, when compared to males.

Summary graphs of ENaC activity (fNPo; A), functional channels (fN; B) and open probability (Po; C) in AngII-infused male and female SD rats receiving vehicle (MA and FA) or spironolactone (MAS and FAS) for 2 weeks. The results of a two-way ANOVA are summarized below the graphs. * – indicates a significant difference (P<0.05) between the respective groups determined by the post-hoc Holm-Sidak test. Each group included at least 8 rats and 66 tested cells. (D) Representative single-channel ENaC current traces recorded in the ASDN principal cells from AngII-infused SD rats on spironolactone. Patches were held at a test potential of Vh=−Vp=−60 mV. Inward currents are downward. Dashed lines indicate the respective open (oi) and closed (c) states of the channel. Areas under the traces marked in blue (male) or pink (female) visualize ENaC activity. (E) Representative immunofluorescent micrographs of rat kidney sections showing the abundance and localization of α-ENaC subunit (pseudocolor green) in the AQP2-positive (pseudocolor red) collecting duct principal cells. Nuclear DAPI staining is shown in pseudocolor blue.
Sodium-chloride cotransporter, NCC, localized to the distal convoluted tubule, is another important determinant of AngII-induced hypertension22. NCC can be activated via MR, although, likely, independently of aldosterone23,24. We found that spironolactone did not affect the abundance of total or phosphorylated (active) forms of NCC in the renal tissue isolated from AngII-infused rats of both sexes (Figure S2). Thus, the effect of spironolactone appears to be mediated through aldosterone-MR cascade.
Nedd4-2 abundance is significantly higher in spironolactone-treated females than in males
Ubiquitination of ENaC subunits by the E3 ubiquitin ligase Nedd4-2 results in subsequent retrieval of the channel from the plasma membrane and is a key converging point for the regulation of ENaC surface density by aldosterone. Aldosterone-induced phosphorylation of Nedd4-2 prevents its binding to ENaC, consequently, increasing the abundance of the channel on the membrane. Here, we used a ratio of total to phosphorylated Nedd4-2 protein in kidney homogenates to gauge the interaction of Nedd4-2 with ENaC (Fig. 5). The ratio was significantly higher in the renal tissue homogenates isolated from spironolactone-treated AngII-infused female rats, when compared to AngII-infused males on spironolactone (1.6 times, P<0.001) or AngII-infused females receiving vehicle (1.4 times, P=0.002). This is indicative of a higher Nedd4-2 activity, greater ENaC internalization and reduced ENaC-dependent renal sodium reabsorption in spironolactone-treated AngII-infused females, when compared to males.
Figure 5. Nedd4-2 activity is remarkably higher in the kidneys of spironolactone-treated AngII-infused females than in males.

(A) Representative western blots from whole kidney homogenates of AngII-infused male and female SD rats receiving vehicle (MA and FA) or spironolactone (MAS and FAS), showing the total Nedd4-2 protein and its inactive form phosphorylated at serine 328. (B) A summary graph comparing Nedd4-2 to pS328Nedd4-2 ratio (indicative of Nedd4-2 interaction with ENaC) in AngII-infused male and female rats. The relative Nedd4-2 to pS328Nedd4-2 ratio, normalized to vehicle-treated AngII-infused males = 1, is shown. The results of a two-way ANOVA are summarized below the graph. * – indicates a significant difference (P<0.05) between respective group means determined by the post-hoc Holm-Sidak test. N=6 per group.
Discussion
This study allowed us to make several novel observations: 1) BP in female rats with AngII-dependent hypertension is more responsive to MR antagonism than in males; 2) spironolactone increases urinary Na+ excretion and urinary Na+ to K+ ratio specifically in AngII-infused females; 3) renal expression of mRNA encoding MR and HSD11β2 is significantly higher in AngII-infused female rats than in males; 4) MR blockade is markedly more potent at decreasing ENaC activity in hypertensive female rats than in males. Our findings indicate that elevated expression of the mineralocorticoid receptor in the kidney determines higher sensitivity of renal ENaC and BP to aldosterone antagonism in female rats with AngII-dependent hypertension, when compared to males. Overstimulation of MR signaling drives BP increase via excessive ENaC-dependent renal Na+ reabsorption in females with AngII-dependent hypertension, but not in males. The data reveal that ENaC sensitivity to MR antagonism is an unrecognized sex-specific approach to combat hypertension and provides pre-clinical evidence supporting the use of MR antagonists in hypertensive women to improve BP control rates.
We found that MR blockade is significantly more effective at attenuating hypertension in AngII-infused female SD rats than in males. This observation is consistent with the emerging clinical evidence and basic science studies showing that females may receive additional antihypertensive and cardiovascular benefits from MR inhibition, when compared to males. MR blockade effectively attenuates salt-sensitive hypertension in female Balb/C mice and decreases BP in agouti yellow obese female mice with leptin-induced hypertension4,17. In contrast, only a marginal hypotensive effect was detected in male AngII-infused rats and mice administered with MR blockers25–28. Clinically, aldosterone antagonism may be more effective at reducing BP in African American women, women with salt sensitive and resistant hypertension, when compared to men7,16,29. To the best of our knowledge, our study is the first to directly compare the antihypertensive effects of MR antagonism in male and female rats with AngII-induced hypertension.
Our study primarily employs spironolactone to explore sex-specific effects of MR blockade in AngII-infused rats. Spironolactone is a potent antimineralocorticoid and its action in the body is enhanced by long-lived therapeutically active metabolites30. Unfortunately, spironolactone is a nonselective MR antagonist with discernable antiandrogenic and progestogenic properties30,31. We confirmed that AngII-infused females derive their antihypertensive benefits specifically from inhibition of MR, using a highly selective second-generation MR antagonist, eplerenone. However, eplerenone is extensively metabolized to inactive derivatives and has a 2-4 times shorter elimination half-life than spironolactone31,32. Due to its pharmacokinetic properties and low cost to the patient, spironolactone is still widely used in clinic, despite its off-target side effects33,34. Moreover, studies in rodents showed that spironolactone and its active derivatives are accumulated in the kidney with remarkably lower levels found in the cardiac tissue30,31,35–38. Thus, upon careful consideration, we chose to pursue our experimental strategy using spironolactone.
In line with the potassium-sparing effect of MR antagonists, we observed a mild, although statistically insignificant, increase in serum potassium levels in spironolactone-treated AngII-infused rats. Hyperkalemia is, perhaps, the most alarming side effect of MR blockade in the clinical setting31,39. Often, the risk-to-benefit ratio for this intervention is defined by the magnitude of serum potassium change. Nevertheless, the incidence and severity of hyperkalemia strongly depends on the dose of the MR antagonist and the level of renal function39. Spironolactone concentrations that we used in this study are in the lower dosage range for rodents and the duration of intervention was only 2 weeks. Our findings and previously published data indicate that renal function is largely preserved in SD rats after 2 weeks of AngII infusion. It is not, therefore, surprising that spironolactone treatment does not cause major disruptions in K+ homeostasis in AngII-infused rats in our experimental setting. Perhaps, future long-term experiments could better inform of potential cumulative side-effects of MR antagonism related to potassium balance.
Our data indicate that treatment with spironolactone improves the diurnal rhythmicity of BP in AngII-infused female rats, but not in males. Blunted diurnal BP variation is a known risk factor for deterioration in renal function, cardiovascular morbidity and mortality, worsening the long-term prognosis for hypertensives with a non-dipping BP pattern40–44. Nocturnal dipping has been associated with the ability to excrete sodium during the day in individuals of African descent45. A mounting body of evidence coming from rodent models and cell lines suggests that a crosstalk between circadian clock, aldosterone and ENaC is an important determinant of systemic Na+ balance, circulating volume and BP46–48. Recent studies reveal sex differences in the circadian gating of Na+ homeostasis and BP control49,50. Yet, the interplay between sex, circadian clock, aldosterone-MR axis, renal Na+ handling and BP regulation is far from being fully understood. Our findings open an interesting avenue for future investigations exploring the contribution of sexual and temporal variability in aldosterone-MR signaling to the pathophysiology of AngII-dependent hypertension and efficacy of BP control interventions.
An important observation of this study is that MR expression is significantly higher in the kidneys of AngII-infused female rats, when compared to males. For many years, studies in the field of hypertension have been focused on circulating or tissue-specific levels and production of aldosterone, a primary salt-retaining hormone, while the implications of renal sensitivity to mineralocorticoid remained largely unexplored. Plasma aldosterone concentration is not always sufficient to reliably predict adverse cardiovascular or prohypertensive outcomes. High circulating aldosterone levels during chronic sodium deficiency (Yanomami population) or pregnancy allow for efficient sodium reabsorption in the kidney, but do not result in any cardiovascular morbidities or hypertension51–53. Animal data and clinical guidelines on normal circulating aldosterone levels vary considerably, often by orders of magnitude54,55. Studies exploring sex, race and age differences add to the existing controversy, and raise a question of what should be regarded as a normal or elevated aldosterone level in a given group7,56–59. It appears that this question cannot be answered without considering both the ligand and its receptor. Inordinate renal Na+ reabsorption is a recognized determinant of hypertension10,12. The existing evidence indicates that elevated sensitivity to aldosterone might have a detrimental effect on sodium balance and BP56,60. There is a gap in knowledge between aldosterone-MR signaling in the distal nephron and differential BP sensitivity to aldosterone or its antagonists in hypertensive patients61. Our study bridges this gap and provides a mechanistic explanation of how increased renal sensitivity to MR antagonists in AngII-infused females translates into higher BP responsiveness to aldosterone antagonism.
Our study reveals that a stronger antihypertensive effect of MR blockade is paralleled by a higher expression of Hsd11b2 in the renal tissue of AngII-infused female rats, when compared to males. This observation is particularly interesting in light of several clinical and basic science studies showing that the effects of MR inhibition on BP, renal Na+ and K+ handling, cardiovascular and renal health do not always result from aldosterone antagonism24,62–64. Aldosterone synthase-deficient mice generally exhibit a milder phenotype than kidney-specific MR knockouts24. While substantial MR effects are reported to be mediated independently of aldosterone in the distal convoluted tubule, the cortical collecting duct is much more reliant upon aldosterone-MR signaling, largely, due to a higher activity of HSD11β224,64. Higher Hsd11b2 expression that we observed in the kidneys of AngII-infused female rats points to a more prominent role of aldosterone in stimulation of renal Na+ reabsorption in this model of hypertension. Interestingly, spironolactone did not affect the abundance of NCC transporter, that is localized to the DCT and is much more likely to be regulated independently of aldosterone. Thus, our findings indicate that higher sensitivity of BP and renal ENaC activity to MR inhibitors in females with AngII-dependent hypertension stems from the blockade of aldosterone actions on MR.
Here, we demonstrate that ENaC activity is significantly lower in the ASDNs isolated from spironolactone-treated AngII-infused female rats than in males. MR inhibition reduces ENaC activity in AngII-infused females not only through lowering the number of functional channels on the membrane, but also through decreasing ENaC open probability. The former is likely a genomic effect of MR inhibition that involves canonical aldosterone signaling pathways. A downstream convergent element of these cascades is Nedd4-2 – a ubiquitin ligase, facilitating the clearance of ENaC from the membrane. Nedd4-2 activity is inhibited by phosphorylation, thereby increasing ENaC surface expression65. Loss of Need4-2 in mice causes salt-sensitive hypertension and enhanced renal ENaC expression66 resistant to eplerenone67. We observed that spironolactone markedly increased the ratio of total over phosphorylated form of Nedd4-2 in the kidney, indicative of higher protein activity and greater ENaC internalization in AngII-infused females, but not in males. This suggests lower ENaC activity due to higher protein degradation in spironolactone-treated females. In addition to increasing ENaC expression and abundance on the membrane, aldosterone was shown to stimulate the activity of the channel68–71, an effect that can be blocked by spironolactone. The exact mechanisms mediating aldosterone-induced increase in ENaC activity remain uncertain, but may employ non-canonical signaling cascades involving membrane phospholipids72,73, methylation of the channel71,74 or generation of reactive oxygen species75. Overall, our results suggest that spironolactone-driven inhibition of ENaC activity in AngII-infused females employs both canonical and noncanonical MR-dependent pathways. This provides a strong impetus for further studies addressing the effect of non-canonical aldosterone signaling pathways on renal sodium handling and BP control.
In our study a stronger BP lowering effect of MR blockade in AngII-infused females is attributed to a more pronounced reduction of renal ENaC activity, when compared to males. A mounting body of evidence indicates that MR antagonism can reduce BP in hypertensive female rodents through mechanisms that are extrinsic to the kidney76. Increased endothelial cell MR activity reportedly contributes to endothelial dysfunction and BP increase in salt sensitive Balb/C and agouti yellow obese hyperleptinemic female mice4,17,77. Activation of MR in endothelial cells increases membrane abundance of endothelial cell sodium channel resulting in reduced endothelium nitric oxide (NO) synthase 3 activity, NO levels and excessive arterial stiffness78,79. ENaC activity in dendritic cells is an important determinant of the sodium-induced proinflammatory response and hypertension in male C57BL/6 mice80. MR in T cells appear to be critical for development of AngII-induced hypertension in C57Bl/6 mice, although the sex of the animals used in this study is not specified81. The relevance of these mechanisms in hypertensive female animals remains to be elucidated, given that the research performed in female rodents mostly points to an anti-inflammatory role of immune system resulting in attenuation of hypertension. Interestingly, inflammatory cytokines, such as interleukin 6 or interferon-γ, can affect MR and ENaC-dependent sodium transport in ASDN cross-linking the inflammatory immune response and renal sodium transport82–84. Overall, the non-epithelial effects of MR observed in vasculature, immune cells and other tissues are undoubtedly important in the pathophysiology of hypertension. Further studies are necessary to integrate the renal and extrarenal effects of MR antagonism pertaining to BP control and achieve a more complete understanding of MR biology in both sexes.
In summary, our study uncovers a sex-divergence in the sensitivity of renal ENaC to MR blockade in hypertensive rats. We and others have previously reported that overstimulation of ENaC is largely independent of aldosterone, primarily driven by intrarenal AngII generation and effectively inhibited by angiotensin converting enzyme blockade in AngII-infused males. Here we show that activation of MR is a primary factor driving excessive ENaC-dependent renal Na+ reabsorption in hypertensive females. Our findings provide the mechanistic rationale to optimize the use of MR antagonists in hypertensive women to improve BP control rates.
Supplementary Material
Perspectives.
Biological sex has become increasingly recognized as a pivotal factor affecting the development and pathophysiological outcomes of hypertension, however, limited understanding of the sex-specific mechanisms regulating BP poses a critical barrier to the optimization of antihypertensive therapies. Our study differentiates the pathophysiological mechanisms governing Na+ retention in hypertensive males and females and improves our understanding of BP control mechanisms in both sexes. Our findings provide the missing pre-clinical evidence to optimize the use of MR antagonists in hypertensive patients of both sexes.
Novelty and Relevance.
What Is New?
Female rats with AngII-dependent hypertension benefit from MR antagonism, exhibiting a stronger BP reduction, when compared to males. MR blockade is also much more potent at decreasing renal ENaC activity in AngII-infused female rats than in males. Higher sensitivity of renal ENaC activity and BP to MR antagonism in female rats with AngII-dependent hypertension is determined by the elevated expression of mRNA encoding MR and HSD11β2 in the kidney.
What Is Relevant?
Identification of the mechanisms responsible for elevation of BP in specific patient cohorts is critical to effectively combat hypertension and related diseases. Our study provides mechanistic evidence for a beneficial therapeutic effect of MR inhibitors in females with AngII-dependent hypertension.
Clinical/Pathophysiological Implications?
Our findings facilitate the development of more effective stratified treatment strategies based on biological sex as a critical covariate.
Acknowledgements
We would like to thank Rabei Alaisami and Katherine Hatcher for their help with the measurement of urinary electrolytes, Simone Kennard for her invaluable help with the PCR machine setup and protocol design and Dr. Aleksandr Taraskin for his insightful suggestions on the analysis of BP diurnal rhythmicity.
Sources of Funding
This work was supported by the National Institute of Diabetes and Digestive and Kidney Disease (NIDDK) R01DK125464 grant to MM, AHA Pre-Doctoral Fellowship 917057 to VB, the National Institute of Heart, Lung and Blood Institute (NHLBI) 1P01HL134604 grant to POC, NHLBI P01HL134604 grant to JS, NIDDK R01DK124303 and NHLBI P01HL134604 grants to MB.
Abbreviations list:
- ASDN
aldosterone-sensitive distal nephron
- AngII
Angiotensin II
- AngII-HTN
Angiotensin II dependent hypertension
- BP
blood pressure
- DAPI
4’,6-diamidino-2-phenylindole
- ENaC
epithelial Na+ channel
- HSD11β2
11β-hydroxysteroid dehydrogenase type 2
- MR
mineralocorticoid receptor
- NO
nitric oxide
- SD
Sprague Dawley
Footnotes
Disclosures
None.
References
- 1.Forouzanfar MH, Afshin A, Alexander LT, Anderson HR, Bhutta ZA, Biryukov S, Brauer M, Burnett R, Cercy K, Charlson FJ, et al. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. The Lancet. 2016;388:1659–1724. doi: 10.1016/S0140-6736(16)31679-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Whelton PK, Carey RM, Aronow WS, Casey DE Jr., Collins KJ, Dennison Himmelfarb C, DePalma SM, Gidding S, Jamerson KA, Jones DW, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2018;71:e13–e115. doi: 10.1161/HYP.0000000000000065 [DOI] [PubMed] [Google Scholar]
- 3.Zanchetti A Genetic and environmental factors in development of hypertension. Journal of Hypertension. 2016;34:2109–2110. doi: 10.1097/hjh.0000000000001102 [DOI] [PubMed] [Google Scholar]
- 4.Faulkner JL, Harwood D, Bender L, Shrestha L, Brands MW, Morwitzer MJ, Kennard S, Antonova G, Belin de Chantemele EJ. Lack of Suppression of Aldosterone Production Leads to Salt-Sensitive Hypertension in Female but Not Male Balb/C Mice. Hypertension. 2018;72:1397–1406. doi: 10.1161/HYPERTENSIONAHA.118.11303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Veiras LC, Girardi ACC, Curry J, Pei L, Ralph DL, Tran A, Castelo-Branco RC, Pastor-Soler N, Arranz CT, Yu ASL, et al. Sexual Dimorphic Pattern of Renal Transporters and Electrolyte Homeostasis. Journal of the American Society of Nephrology. 2017;28:3504. doi: 10.1681/ASN.2017030295 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Veiras LC, McFarlin BE, Ralph DL, Buncha V, Prescott J, Shirvani BS, McDonough JC, Ha D, Giani J, Gurley SB, et al. Electrolyte and transporter responses to angiotensin II induced hypertension in female and male rats and mice. Acta Physiol (Oxf). 2020;229:e13448. doi: 10.1111/apha.13448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shukri MZ, Tan JW, Manosroi W, Pojoga LH, Rivera A, Williams JS, Seely EW, Adler GK, Jaffe IZ, Karas RH, et al. Biological Sex Modulates the Adrenal and Blood Pressure Responses to Angiotensin II. Hypertension. 2018;71:1083–1090. doi: 10.1161/HYPERTENSIONAHA.117.11087 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Eaton DC, Malik B, Saxena NC, Al-Khalili OK, Yue G. Mechanisms of aldosterone’s action on epithelial Na + transport. The Journal of membrane biology. 2001;184:313–319. doi: 10.1007/s00232-001-0098-x [DOI] [PubMed] [Google Scholar]
- 9.Stockand JD. New ideas about aldosterone signaling in epithelia. American journal of physiology Renal physiology. 2002;282:F559–576. doi: 10.1152/ajprenal.00320.2001 [DOI] [PubMed] [Google Scholar]
- 10.Lifton RP, Gharavi AG, Geller DS. Molecular mechanisms of human hypertension. Cell. 2001;104:545–556. doi: 10.1016/s0092-8674(01)00241-0 [DOI] [PubMed] [Google Scholar]
- 11.Pavlov TS, Staruschenko A. Involvement of ENaC in the development of salt-sensitive hypertension. American journal of physiology Renal physiology. 2017;313:F135–F140. doi: 10.1152/ajprenal.00427.2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pratt JH. Central role for ENaC in development of hypertension. J Am Soc Nephrol. 2005;16:3154–3159. doi: 10.1681/ASN.2005050460 [DOI] [PubMed] [Google Scholar]
- 13.Kienitz T, Allolio B, Strasburger CJ, Quinkler M. Sex-specific regulation of ENaC and androgen receptor in female rat kidney. Hormone and metabolic research = Hormon- und Stoffwechselforschung = Hormones et metabolisme. 2009;41:356–362. doi: 10.1055/s-0029-1192033 [DOI] [PubMed] [Google Scholar]
- 14.Tiwari S, Li L, Riazi S, Halagappa VKM, Ecelbarger CM. Sex and age result in differential regulation of the renal thiazide-sensitive NaCl cotransporter and the epithelial sodium channel in angiotensin II-infused mice. Am J Nephrol. 2009;30:554–562. doi: 10.1159/000252776 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ramirez LA, Sullivan JC. Sex Differences in Hypertension: Where We Have Been and Where We Are Going. Am J Hypertens. 2018;31:1247–1254. doi: 10.1093/ajh/hpy148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Clemmer JS, Faulkner JL, Mullen AJ, Butler KR, Hester RL. Sex-specific responses to mineralocorticoid receptor antagonism in hypertensive African American males and females. Biol Sex Differ. 2019;10:24. doi: 10.1186/s13293-019-0238-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Huby AC, Otvos L, Jr., Belin de Chantemele EJ. Leptin Induces Hypertension and Endothelial Dysfunction via Aldosterone-Dependent Mechanisms in Obese Female Mice. Hypertension. 2016;67:1020–1028. doi: 10.1161/HYPERTENSIONAHA.115.06642 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kanashiro-Takeuchi RM, Heidecker B, Lamirault G, Dharamsi JW, Hare JM. Sex-specific impact of aldosterone receptor antagonism on ventricular remodeling and gene expression after myocardial infarction. Clin Transl Sci. 2009;2:134–142. doi: 10.1111/j.1752-8062.2009.00094.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mamenko M, Zaika O, Prieto MC, Jensen VB, Doris PA, Navar LG, Pochynyuk O. Chronic angiotensin II infusion drives extensive aldosterone-independent epithelial Na+ channel activation. Hypertension. 2013;62:1111–1122. doi: 10.1161/hypertensionaha.113.01797 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gillis EE, Brands MW, Sullivan JC. Adverse Maternal and Fetal Outcomes in a Novel Experimental Model of Pregnancy after Recovery from Renal Ischemia-Reperfusion Injury. J Am Soc Nephrol. 2021;32:375–384. doi: 10.1681/ASN.2020020127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mamenko M, Lysikova DV, Spires DR, Tarima SS, Ilatovskaya DV. Practical notes on popular statistical tests in renal physiology. American journal of physiology Renal physiology. 2022;323:F389–F400. doi: 10.1152/ajprenal.00427.2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Castaneda-Bueno M, Cervantes-Perez LG, Vazquez N, Uribe N, Kantesaria S, Morla L, Bobadilla NA, Doucet A, Alessi DR, Gamba G. Activation of the renal Na+:Cl- cotransporter by angiotensin II is a WNK4-dependent process. Proc Natl Acad Sci U S A. 2012;109:7929–7934. doi: 10.1073/pnas.1200947109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fujita T Mechanism of salt-sensitive hypertension: focus on adrenal and sympathetic nervous systems. J Am Soc Nephrol. 2014;25:1148–1155. doi: 10.1681/ASN.2013121258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Maeoka Y, Su XT, Wang WH, Duan XP, Sharma A, Li N, Staub O, McCormick JA, Ellison DH. Mineralocorticoid Receptor Antagonists Cause Natriuresis in the Absence of Aldosterone. Hypertension. 2022;79:1423–1434. doi: 10.1161/HYPERTENSIONAHA.122.19159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ortiz RM, Graciano ML, Seth D, Awayda MS, Navar LG. Aldosterone receptor antagonism exacerbates intrarenal angiotensin II augmentation in ANG II-dependent hypertension. American journal of physiology Renal physiology. 2007;293:F139–147. doi: 10.1152/ajprenal.00504.2006 [DOI] [PubMed] [Google Scholar]
- 26.Rocha R, Martin-Berger CL, Yang P, Scherrer R, Delyani J, McMahon E. Selective aldosterone blockade prevents angiotensin II/salt-induced vascular inflammation in the rat heart. Endocrinology. 2002;143:4828–4836. doi: 10.1210/en.2002-220120 [DOI] [PubMed] [Google Scholar]
- 27.Ashek A, Menzies RI, Mullins LJ, Bellamy COC, Harmar AJ, Kenyon CJ, Flatman PW, Mullins JJ, Bailey MA. Activation of thiazide-sensitive co-transport by angiotensin II in the cyp1a1-Ren2 hypertensive rat. PLoS One. 2012;7:e36311–e36311. doi: 10.1371/journal.pone.0036311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhao D, Seth DM, Navar LG. Enhanced distal nephron sodium reabsorption in chronic angiotensin II-infused mice. Hypertension (Dallas, Tex : 1979). 2009;54:120–126. doi: 10.1161/HYPERTENSIONAHA.109.133785 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chapman N, Dobson J, Wilson S, Dahlof B, Sever PS, Wedel H, Poulter NR, Anglo-Scandinavian Cardiac Outcomes Trial I. Effect of spironolactone on blood pressure in subjects with resistant hypertension. Hypertension. 2007;49:839–845. doi: 10.1161/01.HYP.0000259805.18468.8c [DOI] [PubMed] [Google Scholar]
- 30.Kolkhof P, Barfacker L. 30 YEARS OF THE MINERALOCORTICOID RECEPTOR: Mineralocorticoid receptor antagonists: 60 years of research and development. J Endocrinol. 2017;234:T125–T140. doi: 10.1530/JOE-16-0600 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sica DA. Pharmacokinetics and pharmacodynamics of mineralocorticoid blocking agents and their effects on potassium homeostasis. Heart Fail Rev. 2005;10:23–29. doi: 10.1007/s10741-005-2345-1 [DOI] [PubMed] [Google Scholar]
- 32.Brown NJ. Eplerenone. Circulation. 2003;107:2512–2518. doi: doi: 10.1161/01.CIR.0000071081.35693.9A [DOI] [PubMed] [Google Scholar]
- 33.Sato A Mineralocorticoid receptor antagonists: their use and differentiation in Japan. Hypertension Research. 2013;36:185–190. doi: 10.1038/hr.2012.182 [DOI] [PubMed] [Google Scholar]
- 34.Funder JW. Aldosterone and Mineralocorticoid Receptors-Physiology and Pathophysiology. Int J Mol Sci. 2017;18:1032. doi: 10.3390/ijms18051032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.LaCagnin LB, Lutsie P, Colby HD. Conversion of spironolactone to 7 alpha-thiomethylspironolactone by hepatic and renal microsomes. Biochem Pharmacol. 1987;36:3439–3444. doi: 10.1016/0006-2952(87)90323-6 [DOI] [PubMed] [Google Scholar]
- 36.Los LE, Colby HD. Binding of spironolactone metabolites in vivo to renal mineralocorticoid receptors in guinea pigs. Pharmacology. 1994;48:86–92. doi: 10.1159/000139166 [DOI] [PubMed] [Google Scholar]
- 37.Hawkins UA, Gomez-Sanchez EP, Gomez-Sanchez CM, Gomez-Sanchez CE. The ubiquitous mineralocorticoid receptor: clinical implications. Current hypertension reports. 2012;14:573–580. doi: 10.1007/s11906-012-0297-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kolkhof P, Borden SA. Molecular pharmacology of the mineralocorticoid receptor: prospects for novel therapeutics. Mol Cell Endocrinol. 2012;350:310–317. doi: 10.1016/j.mce.2011.06.025 [DOI] [PubMed] [Google Scholar]
- 39.Sica DA. Mineralocorticoid Receptor Antagonists for Treatment of Hypertension and Heart Failure. Methodist Debakey Cardiovasc J. 2015;11:235–239. doi: 10.14797/mdcj-11-4-235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ohkubo T, Hozawa A, Yamaguchi J, Kikuya M, Ohmori K, Michimata M, Matsubara M, Hashimoto J, Hoshi H, Araki T, et al. Prognostic significance of the nocturnal decline in blood pressure in individuals with and without high 24-h blood pressure: the Ohasama study. J Hypertens. 2002;20:2183–2189. doi: 10.1097/00004872-200211000-00017 [DOI] [PubMed] [Google Scholar]
- 41.Higashi Y, Nakagawa K, Kimura M, Noma K, Hara K, Sasaki S, Goto C, Oshima T, Chayama K, Yoshizumi M. Circadian variation of blood pressure and endothelial function in patients with essential hypertension:a comparison of dippers and non-dippers. J Am Coll Cardiol. 2002;40:2039–2043. doi: 10.1016/s0735-1097(02)02535-4 [DOI] [PubMed] [Google Scholar]
- 42.Cuspidi C, Meani S, Salerno M, Valerio C, Fusi V, Severgnini B, Lonati L, Magrini F, Zanchetti A. Cardiovascular target organ damage in essential hypertensives with or without reproducible nocturnal fall in blood pressure. J Hypertens. 2004;22:273–280. doi: 10.1097/00004872-200402000-00010 [DOI] [PubMed] [Google Scholar]
- 43.de la Sierra A, Segura J, Gorostidi M, Banegas JR, de la Cruz JJ, Ruilope LM. Diurnal blood pressure variation, risk categories and antihypertensive treatment. Hypertens Res. 2010;33:767–771. doi: 10.1038/hr.2010.111 [DOI] [PubMed] [Google Scholar]
- 44.Davidson MB, Hix JK, Vidt DG, Brotman DJ. Association of impaired diurnal blood pressure variation with a subsequent decline in glomerular filtration rate. Arch Intern Med. 2006;166:846–852. doi: 10.1001/archinte.166.8.846 [DOI] [PubMed] [Google Scholar]
- 45.Bankir L, Bochud M, Maillard M, Bovet P, Gabriel A, Burnier M. Nighttime blood pressure and nocturnal dipping are associated with daytime urinary sodium excretion in African subjects. Hypertension. 2008;51:891–898. doi: 10.1161/HYPERTENSIONAHA.107.105510 [DOI] [PubMed] [Google Scholar]
- 46.Douma LG, Costello HM, Crislip GR, Cheng KY, Lynch IJ, Juffre A, Barral D, Masten S, Roig E, Beguiristain K, et al. Kidney-specific KO of the circadian clock protein PER1 alters renal Na(+) handling, aldosterone levels, and kidney/adrenal gene expression. American journal of physiology Renal physiology. 2022;322:F449–F459. doi: 10.1152/ajprenal.00385.2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Doi M, Takahashi Y, Komatsu R, Yamazaki F, Yamada H, Haraguchi S, Emoto N, Okuno Y, Tsujimoto G, Kanematsu A, et al. Salt-sensitive hypertension in circadian clock-deficient Cry-null mice involves dysregulated adrenal Hsd3b6. Nat Med. 2010;16:67–74. doi: 10.1038/nm.2061 [DOI] [PubMed] [Google Scholar]
- 48.Gumz ML, Stow LR, Lynch IJ, Greenlee MM, Rudin A, Cain BD, Weaver DR, Wingo CS. The circadian clock protein Period 1 regulates expression of the renal epithelial sodium channel in mice. J Clin Invest. 2009;119:2423–2434. doi: 10.1172/JCI36908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Crislip GR, Douma LG, Masten SH, Cheng KY, Lynch IJ, Johnston JG, Barral D, Glasford KB, Holzworth MR, Verlander JW, et al. Differences in renal BMAL1 contribution to Na(+) homeostasis and blood pressure control in male and female mice. American journal of physiology Renal physiology. 2020;318:F1463–F1477. doi: 10.1152/ajprenal.00014.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Douma LG, Solocinski K, Holzworth MR, Crislip GR, Masten SH, Miller AH, Cheng KY, Lynch IJ, Cain BD, Wingo CS, et al. Female C57BL/6J mice lacking the circadian clock protein PER1 are protected from nondipping hypertension. Am J Physiol Regul Integr Comp Physiol. 2019;316:R50–R58. doi: 10.1152/ajpregu.00381.2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Oliver WJ, Cohen EL, Neel JV. Blood pressure, sodium intake, and sodium related hormones in the Yanomamo Indians, a “no-salt” culture. Circulation. 1975;52:146–151. doi: doi: 10.1161/01.CIR.52.1.146 [DOI] [PubMed] [Google Scholar]
- 52.Escher G, Cristiano M, Causevic M, Baumann M, Frey FJ, Surbek D, Mohaupt MG. High aldosterone-to-renin variants of CYP11B2 and pregnancy outcome. Nephrology Dialysis Transplantation. 2009;24:1870–1875. doi: 10.1093/ndt/gfn763 [DOI] [PubMed] [Google Scholar]
- 53.Lindheimer MD, August P. Aldosterone, maternal volume status and healthy pregnancies: a cycle of differing views. Nephrology Dialysis Transplantation. 2009;24:1712–1714. doi: 10.1093/ndt/gfp093 [DOI] [PubMed] [Google Scholar]
- 54.Funder JW. Sensitivity to Aldosterone. Hypertension. 2014;63:1168–1170. doi: doi: 10.1161/HYPERTENSIONAHA.114.03127 [DOI] [PubMed] [Google Scholar]
- 55.Manolopoulou J, Bielohuby M, Caton SJ, Gomez-Sanchez CE, Renner-Mueller I, Wolf E, Lichtenauer UD, Beuschlein F, Hoeflich A, Bidlingmaier M. A highly sensitive immunofluorometric assay for the measurement of aldosterone in small sample volumes: validation in mouse serum. Journal of Endocrinology. 2008;196:215–224. doi: 10.1677/joe-07-0134 [DOI] [PubMed] [Google Scholar]
- 56.Tu W, Li R, Bhalla V, Eckert GJ, Pratt JH. Age-Related Blood Pressure Sensitivity to Aldosterone in Blacks and Whites. Hypertension. 2018;72:247–252. doi: doi: 10.1161/HYPERTENSIONAHA.118.11014 [DOI] [PubMed] [Google Scholar]
- 57.Pratt JH, Jones JJ, Miller JZ, Wagner MA, Fineberg NS. Racial differences in aldosterone excretion and plasma aldosterone concentrations in children. N Engl J Med. 1989;321:1152–1157. doi: 10.1056/NEJM198910263211703 [DOI] [PubMed] [Google Scholar]
- 58.Szmuilowicz ED, Adler GK, Williams JS, Green DE, Yao TM, Hopkins PN, Seely EW. Relationship between Aldosterone and Progesterone in the Human Menstrual Cycle. The Journal of Clinical Endocrinology & Metabolism. 2006;91:3981–3987. doi: 10.1210/jc.2006-1154 [DOI] [PubMed] [Google Scholar]
- 59.Toering TJ, Gant CM, Visser FW, van der Graaf AM, Laverman GD, Danser AHJ, Faas MM, Navis G, Lely AT. Sex differences in renin-angiotensin-aldosterone system affect extracellular volume in healthy subjects. American journal of physiology Renal physiology. 2018;314:F873–f878. doi: 10.1152/ajprenal.00109.2017 [DOI] [PubMed] [Google Scholar]
- 60.Tu W, Eckert GJ, Hannon TS, Liu H, Pratt LM, Wagner MA, DiMeglio LA, Jung J, Pratt JH. Racial Differences in Sensitivity of Blood Pressure to Aldosterone. Hypertension. 2014;63:1212–1218. doi: doi: 10.1161/HYPERTENSIONAHA.113.02989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Gray Z, Tu W, Chertow GM, Bhalla V. Aldosterone sensitivity: an opportunity to explore the pathogenesis of hypertension. American Journal of Physiology-Renal Physiology. 2021;320:F325–F335. doi: 10.1152/ajprenal.00415.2020 [DOI] [PubMed] [Google Scholar]
- 62.Williams B, MacDonald TM, Morant SV, Webb DJ, Sever P, McInnes GT, Ford I, Cruickshank JK, Caulfield MJ, Padmanabhan S, et al. Endocrine and haemodynamic changes in resistant hypertension, and blood pressure responses to spironolactone or amiloride: the PATHWAY-2 mechanisms substudies. Lancet Diabetes Endocrinol. 2018;6:464–475. doi: 10.1016/S2213-8587(18)30071-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Luther JM, Luo P, Wang Z, Cohen SE, Kim HS, Fogo AB, Brown NJ. Aldosterone deficiency and mineralocorticoid receptor antagonism prevent angiotensin II-induced cardiac, renal, and vascular injury. Kidney Int. 2012;82:643–651. doi: 10.1038/ki.2012.170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Nesterov V, Bertog M, Canonica J, Hummler E, Coleman R, Welling PA, Korbmacher C. Critical role of the mineralocorticoid receptor in aldosterone-dependent and aldosterone-independent regulation of ENaC in the distal nephron. American journal of physiology Renal physiology. 2021;321:F257–F268. doi: 10.1152/ajprenal.00139.2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Rotin D, Staub O. Nedd4–2 and the regulation of epithelial sodium transport. Front Physiol. 2012;3:212. doi: 10.3389/fphys.2012.00212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Minegishi S, Ishigami T, Kino T, Chen L, Nakashima-Sasaki R, Araki N, Yatsu K, Fujita M, Umemura S. An isoform of Nedd4–2 is critically involved in the renal adaptation to high salt intake in mice. Sci Rep. 2016;6:27137. doi: 10.1038/srep27137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kino T, Ishigami T, Murata T, Doi H, Nakashima-Sasaki R, Chen L, Sugiyama M, Azushima K, Wakui H, Minegishi S, et al. Eplerenone-Resistant Salt-Sensitive Hypertension in Nedd4–2 C2 KO Mice. Int J Mol Sci. 2017;18. doi: 10.3390/ijms18061250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kemendy AE, Kleyman TR, Eaton DC. Aldosterone alters the open probability of amiloride-blockable sodium channels in A6 epithelia. American Journal of Physiology-Cell Physiology. 1992;263:C825–C837. doi: 10.1152/ajpcell.1992.263.4.C825 [DOI] [PubMed] [Google Scholar]
- 69.Frindt G, Palmer LG. Acute effects of aldosterone on the epithelial Na channel in rat kidney. American Journal of Physiology-Renal Physiology. 2015;308:F572–F578. doi: 10.1152/ajprenal.00585.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Shabbir W, Topcagic N, Aufy M. Activation of autosomal recessive Pseudohypoaldosteronism1 ENaC with aldosterone. European Journal of Pharmacology. 2021;901:174090. doi: 10.1016/j.ejphar.2021.174090 [DOI] [PubMed] [Google Scholar]
- 71.Becchetti A, Kemendy AE, Stockand JD, Sariban-Sohraby S, Eaton DC. Methylation Increases the Open Probability of the Epithelial Sodium Channel in A6 Epithelia *. Journal of Biological Chemistry. 2000;275:16550–16559. doi: 10.1074/jbc.M000954200 [DOI] [PubMed] [Google Scholar]
- 72.Helms MN, Liu L, Liang Y-Y, Al-Khalili O, Vandewalle A, Saxena S, Eaton DC, Ma H-P. Phosphatidylinositol 3,4,5-Trisphosphate Mediates Aldosterone Stimulation of Epithelial Sodium Channel (ENaC) and Interacts with γ-ENaC *. Journal of Biological Chemistry. 2005;280:40885–40891. doi: 10.1074/jbc.M509646200 [DOI] [PubMed] [Google Scholar]
- 73.Tong Q, Booth RE, Worrell RT, Stockand JD. Regulation of Na+ transport by aldosterone: signaling convergence and cross talk between the PI3-K and MAPK1/2 cascades. American journal of physiology Renal physiology. 2004;286:F1232–1238. doi: 10.1152/ajprenal.00345.2003 [DOI] [PubMed] [Google Scholar]
- 74.Edinger RS, Yospin J, Perry C, Kleyman TR, Johnson JP. Regulation of epithelial Na+ channels (ENaC) by methylation: a novel methyltransferase stimulates ENaC activity. J Biol Chem. 2006;281:9110–9117. doi: 10.1074/jbc.M509232200 [DOI] [PubMed] [Google Scholar]
- 75.Yu L, Bao HF, Self JL, Eaton DC, Helms MN. Aldosterone-induced increases in superoxide production counters nitric oxide inhibition of epithelial Na channel activity in A6 distal nephron cells. American journal of physiology Renal physiology. 2007;293:F1666–1677. doi: 10.1152/ajprenal.00444.2006 [DOI] [PubMed] [Google Scholar]
- 76.Barris CT, Faulkner JL, Belin de Chantemele EJ. Salt Sensitivity of Blood Pressure in Women. Hypertension. 2023;80:268–278. doi: 10.1161/HYPERTENSIONAHA.122.17952 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Faulkner JL, Kennard S, Huby AC, Antonova G, Lu Q, Jaffe IZ, Patel VS, Fulton DJR, Belin de Chantemele EJ. Progesterone Predisposes Females to Obesity-Associated Leptin-Mediated Endothelial Dysfunction via Upregulating Endothelial MR (Mineralocorticoid Receptor) Expression. Hypertension. 2019;74:678–686. doi: 10.1161/HYPERTENSIONAHA.119.12802 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Druppel V, Kusche-Vihrog K, Grossmann C, Gekle M, Kasprzak B, Brand E, Pavenstadt H, Oberleithner H, Kliche K. Long-term application of the aldosterone antagonist spironolactone prevents stiff endothelial cell syndrome. FASEB J. 2013;27:3652–3659. doi: 10.1096/fj.13-228312 [DOI] [PubMed] [Google Scholar]
- 79.Kusche-Vihrog K, Sobczak K, Bangel N, Wilhelmi M, Nechyporuk-Zloy V, Schwab A, Schillers H, Oberleithner H. Aldosterone and amiloride alter ENaC abundance in vascular endothelium. Pflugers Arch. 2008;455:849–857. doi: 10.1007/s00424-007-0341-0 [DOI] [PubMed] [Google Scholar]
- 80.Barbaro NR, Foss JD, Kryshtal DO, Tsyba N, Kumaresan S, Xiao L, Mernaugh RL, Itani HA, Loperena R, Chen W, et al. Dendritic Cell Amiloride-Sensitive Channels Mediate Sodium-Induced Inflammation and Hypertension. Cell Rep. 2017;21:1009–1020. doi: 10.1016/j.celrep.2017.10.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Sun XN, Li C, Liu Y, Du LJ, Zeng MR, Zheng XJ, Zhang WC, Liu Y, Zhu M, Kong D, et al. T-Cell Mineralocorticoid Receptor Controls Blood Pressure by Regulating Interferon-Gamma. Circ Res. 2017;120:1584–1597. doi: 10.1161/CIRCRESAHA.116.310480 [DOI] [PubMed] [Google Scholar]
- 82.Kamat NV, Thabet SR, Xiao L, Saleh MA, Kirabo A, Madhur MS, Delpire E, Harrison DG, McDonough AA. Renal transporter activation during angiotensin-II hypertension is blunted in interferon-gamma−/− and interleukin-17A−/− mice. Hypertension. 2015;65:569–576. doi: 10.1161/HYPERTENSIONAHA.114.04975 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Li K, Guo D, Zhu H, Hering-Smith KS, Hamm LL, Ouyang J, Dong Y. Interleukin-6 stimulates epithelial sodium channels in mouse cortical collecting duct cells. Am J Physiol Regul Integr Comp Physiol. 2010;299:R590–595. doi: 10.1152/ajpregu.00207.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Wynne BM, Samson TK, Moyer HC, van Elst HJ, Moseley AS, Hecht G, Paul O, Al-Khalili O, Gomez-Sanchez C, Ko B, et al. Interleukin 6 mediated activation of the mineralocorticoid receptor in the aldosterone-sensitive distal nephron. Am J Physiol Cell Physiol. 2022;323:C1512–C1523. doi: 10.1152/ajpcell.00272.2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


