Abstract
Introduction
Plants are a source of natural ingredients with retinol-like properties that can deliver anti-aging benefits without the side effects typically associated with retinoid use. We hypothesized that by combining two such analogs, bakuchiol (BAK) and Vigna aconitifolia extract (VAE), with the potent retinoid retinal (RAL), the anti-photoaging potential of RAL could be enhanced without compromising its skin irritation profile. The purpose of this study was to demonstrate that BAK and VAE potentiate the anti-photoaging activity of RAL.
Methods
Gene expression profiling of full-thickness reconstructed skin was first used to examine the impact of BAK or VAE in combination with RAL on skin biology. Next, the irritative potential of this combination, and its capacity to reverse key signs of photoaging in an ex vivo model was assessed. Finally, a proof-of-concept open label clinical study was performed to evaluate the anti-photoaging capacity and skin compatibility of a cosmetic formulation (tri-retinoid complex; 3RC) containing this complex in combination with other well characterized anti-photoaging ingredients.
Results
In vitro profiling suggested that combining 0.1% RAL with BAK or VAE potentiates the effect of RAL on keratinocyte differentiation and skin barrier function without affecting its skin irritation profile. When formulated with other anti-photoaging ingredients, such as niacinamide and melatonin, 3RC reversed ultraviolet radiation-induced deficits in structural components of the dermal extracellular matrix, including hyaluronic acid and collagen. In vivo, it led to a reversal of clinical signs of age and photodamage, with statistically significant improvement to skin firmness (+5.6%), skin elasticity (+13.9%), wrinkle count (−43.2%), and skin tone homogeneity (+7.0%), observed within 28 days of once nightly use. Notably, the number of crow’s feet wrinkles was reduced in 100% of subjects. Furthermore, 3RC was very well tolerated.
Conclusion
These data suggest that 3RC is a highly effective and well-tolerated treatment for photoaging.
Keywords: Anti-aging, Bakuchiol, Photoaging, Retinal, Retinaldehyde, Retinoid, Skin aging, Vigna aconitifolia
Key Summary Points
| Why carry out this study? |
| • Skin aging is a complex process that causes aesthetic changes in skin and has a significant emotional impact on those affected. |
| • Current retinoid-based treatments are compromised by the time required to see clinical improvements and by their side effects. |
| What was learned from the study? |
| • The natural retinol analogs, bakuchiol and Vigna aconitifolia extract potentiate the anti-photoaging effects of retinal without affecting its skin irritation profile. |
| • This leads to rapid improvement of the clinical signs of age and photodamage. |
| • This unique combination of ingredients is a highly effective and well-tolerated treatment for photoaging. |
Introduction
The discovery that the damaging effects of ultraviolet radiation (UVR) on skin can be partially reversed by the application of retinoic acid (RA) [1, 2] led to its rapid adoption as the drug treatment of choice for photoaging. RA is a low-molecular-weight lipophilic metabolite of vitamin A that controls pleiotropic cellular functions in the skin, including keratinocyte differentiation, lipid metabolism, cell proliferation, apoptosis, and epidermal barrier function [3]. RA exerts its effects through a family of nuclear RA receptors (RARα, RARβ, and RARγ). Once activated by RA, RARs associate with the retinoid X receptors (RXRα, RXRβ, and RXRγ) to form RAR-RXR heterodimers which regulate transcription following binding at the promoter regions of their target genes [4, 5].
The beneficial effects of RA on photoaged skin, however, are gradual and progressive [6]. Short-term use of RA (i.e., < 1 month) induces changes such as increased smoothness, and reduction of hyperpigmentation [7]. New deposition of collagen also appears to occur at this time [8], but only by continuing treatment for at least 12 weeks are significant reductions to fine wrinkles observed [9]. Increasing the RA concentration can lead to earlier resolution of skin damage but at a cost of increased risk of side-effects such as erythema and desquamation [10]. Because of these side effects, RA is prohibited for use in cosmetic products in Europe and the US. Instead, precursors of endogenous RA, such as retinal [retinaldehyde (RAL)] and retinol (ROL) are used. To be biologically active, these precursors must be first converted to RA. As the immediate precursor of RA, RAL is converted by keratinocytes to RA in a single step, while conversion of ROL necessitates an additional step, with RAL as an intermediate [11]. In bypassing the first of these two rate-limiting oxidation steps, RAL has been shown to be around three-fold more biologically active than ROL [12]. Its conversion to RA is also estimated to occur at least 11 times faster [11]. Moreover, transformation of RA from RAL occurs in both differentiating and non-differentiating keratinocytes, unlike ROL which occurs only in the former [11]. Taken together, these data suggest that RA transformation and retinoid activity in cosmetic formulations can be maximized using RAL.
Topical application of RAL induces changes in photoaged skin like those of RA [13], with a reduction of fine lines and wrinkles [14, 15] and hyperpigmentation [15], smoothing of the skin [14], increased hydration levels [15], thickening of the epidermis [13], and increased elasticity [13]. Crucially, however, it is much better tolerated [16, 17]. Like RA, improvements to the clinical signs of photodamage following RAL treatment are slow, and typically at least 18 weeks of daily treatment is required to see clinical benefit [14]. Increasing the dose of RAL also appears to have little effect. A comparative study of 0.1% and 0.05% RAL, for example, revealed no significant differences between both concentrations in all measured parameters except the melanin index [18].
Considering this, we hypothesized that the retinoid activity of RAL might be potentiated by formulating it with natural retinol-like ingredients that have functionally analogous effects on skin. Because such retinol-like ingredients do not appear to act via classical retinoid signaling pathways, they may also have lower irritation potentials. Bakuchiol (1-(4-hydroxyphenyl)-3,7-dimethyl-3-vinyl-1,6-octa-diene) (BAK), a meroterpene phenol from seeds of the plant Psoralea corylifolia L. [19], is perhaps the best known of these and has been widely used in cosmetic formulations for its anti-aging properties [20–24]. BAK modulates numerous retinoid-responsive genes in reconstructed full-thickness skin, including upregulating genes encoding collagen subunits (i.e., COL1A2, COL4A6, COL9A2, COL9A3, COL4A6, and COL17A1), enzymes involved in hyaluronic acid synthesis (i.e., HAS3), and key modulators of skin hydration (i.e., AQP3) [20]. Notably, it has no effect on genes encoding the RA receptors (RARB and RARG) [20], suggesting that it does not act via classical RA signaling pathways, thereby avoiding the potential for the typical irritant reactions caused by RA overload. Clinically, 0.5% BAK has been demonstrated to significantly improve fine lines and wrinkles, elasticity, firmness, and signs of photodamage after 12 weeks of treatment to a level comparable to 0.5% ROL [20, 24]. Vigna aconitifolia (Jacq.) Marechàl (moth bean) is an annual herb native to India and Pakistan belonging to the Leguminosae family. Its seeds contain mainly carbohydrates (56–62%), proteins (22–24%), water (11%), lipids (1.9%), polyphenols (1.3%), and an array of bioactive compounds with antioxidant and enzyme inhibitory properties [25]. A protein extract of Vigna aconitifolia (VAE) has been shown to deliver retinol-like effects in skin and reduce crow’s feet wrinkles with an efficacy comparable to 2.5% encapsulated ROL (information from supplier; data on file).
In this study we sought to determine if BAK and VAE potentiate the anti-photoaging activity of 0.1% RAL without compromising its skin irritation profile, and whether, by formulating 0.1% RAL, BAK, and VAE with other anti-aging ingredients, clinical signs of photodamage could be reversed. Here melatonin and niacinamide were added for their additional anti-aging benefits. Melatonin is an indolic compound derived from serotonin that is produced and metabolized in the skin [26]. Melatonin and its metabolites are free radical scavengers that help maintain genomic, cellular, and tissue integrity after UV exposure [27, 28]. Niacinamide is a member of the vitamin B family that has multi-factorial benefits in aged skin, including enhancing barrier function [29], normalizing hyperpigmentation [30], and boosting the production of dermal matrix components that can smooth wrinkles [31]. The clinical efficacy and safety of this cosmetic formulation [tri-retinoid complex (3RC)] was assessed in a proof-of-concept open-label study.
Methods
Test Materials
Several formulations were prepared for in vitro, ex vivo, and clinical studies:
For gene expression analysis, liposome-encapsulated RAL was mixed in a lipophilic base with or without BAK (Sytenol® A; Biesterfeld AG, Hamburg, Germany) (RAL/BAK), or VAE (Vit-A-Like™; BASF, Ludwigshafen, Germany) (RAL/VAE) at a final concentration of 0.05% and 0.1%. The lipophilic base alone was used as the reference (vehicle).
For comparative irritation and RAL metabolism studies, 0.1% liposome-encapsulated RAL alone and 0.1% liposome-encapsulated RAL in combination with BAK and VAE (RAL/BAK/VAE) were mixed in a lipophilic base. The lipophilic base alone was used as a control (vehicle).
Ex vivo and clinical anti-photoaging studies were performed with 3RC, a biphasic extemporaneous serum with an oil phase containing 0.1% liposome-encapsulated RAL, BAK, and melatonin physically separated from a cream phase containing VAE and niacinamide. For all clinical studies, 3RC was dosed with a pump containing 2 chambers that ensured the same quantity of each phase was delivered per application (data not shown). Stability of the formulation for the duration of the studies had been established previously (data not shown). For the ex vivo studies, a placebo formulation containing all ingredients, except RAL/BAK/VAE, was used as a control.
Gene Expression Analysis
A three-dimensional (3D) full-thickness in vitro skin model (FT skin) containing normal human epidermal keratinocytes and normal human dermal fibroblasts (EpiDerm FT™ EFT-400, MatTek Corp., Ashland, MA) was used to determine the effect of 0.05% and 0.1% RAL alone and in combination with BAK (RAL/BAK) or VAE (RAL/VAE). 0.5% ROL, 1.0% ROL, and 0.05% RA were included as additional positive controls. Gene expression was assessed using a qPCR-based gene expression panel consisting of five endogenous control genes and 107 target genes that play important roles in skin biology (Standard skin panel; Genemarkers, Kalamazoo, MI).
Briefly, 25 µl of each test substance was applied to the surface of each tissue and distributed evenly using a sterile glass spreader. Following topical application, cultures were incubated at 37 °C, 5% CO2, ~ 95% relative humidity for 24 h. After 24 h, the surface of each tissue was washed with sterile DPBS to remove the test substance, cut in half, and placed in RNAlater™ (Invitrogen, ThermoFisher Scientific, Waltham, MA) for 1–2 h at room temperature, and then at 4 °C for 4 days. RNA was isolated from each tissue using a Maxwell® 16 LEV simplyRNA tissue purification kit (Promega, Madison, WI) following the manufacturer’s instructions. cDNA was generated from 2000 ng RNA using a High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems) and qPCR reactions performed using TaqMan® gene expression assays in an OpenArray® format (ThermoFisher Scientific, Waltham, MA) using a QuantStudio 12 K Flex qPCR system (Applied Biosystems, ThermoFisher Scientific, Waltham, MA). Each gene was analyzed in duplicate, and four tissues included for each treatment group. Linear fold-change values were calculated for each gene using TIBCO Spotfire® Analyst software (TIBCO Software Inc., Palo Alto, CA). Unpaired t-tests to evaluate differences between the vehicle control and each treatment group were performed using TIBCO Spotfire® Analyst software. Differences were considered significant at p < 0.05.
In Vitro Skin Irritation Potential
Skin irritation potentials of 0.1% RAL and RAL/BAK/VAE were determined by calculating ET50 values [defined as the exposure time, in minutes, for the test item to reduce the viability of reconstructed human epidermis (RHE) by 50%]. Briefly, 100 μl of 0.1% RAL or RAL/BAK/VAE were applied to the surface of RHE (EPI-200, EpiDerm™; MatTek Corporation, Ashland MA) for 4, 8, 16, and 24 h. Cell viability by 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide (MTT) reduction at each contact time was determined and the regression line calculated to generate the ET50 value.
Ex Vivo Assessment of Retinoid Activity and Anti-Photoaging Activity
All skin explants used in this study were obtained from surgical residues of abdominal skin obtained from a Caucasian woman [53 years of age; individual typology angle (ITA°) 30°] after written informed consent from the donor and in full respect of the Declaration of Helsinki and article L.1245–2 of the French Public Health Code. The latter does not require any prior authorization by an ethics committee for use of surgical waste.
Retinoid activity of RAL/BAK/VAE and 3RC was determined by examining the expression of Cellular retinoid acid-binding protein 2 (CRABP-II) in human skin explants.
Anti-photoaging activity of 3RC was determined by examining the expression of hyaluronic acid, procollagen type I (PC1), total collagen, matrix metalloproteinase 1 (MMP-1), and levels of degraded collagen.
Briefly, explants of an average size of 8 mm × 3 mm (ø × thickness) were prepared. Skin samples were cultured in an air–liquid interface in a perforated ring of stainless steel in contact with culture medium (modified Williams’ E medium).
To determine their effect on photodamaged skin, test items were applied to skin that had previously been exposed to UVR. Briefly, skin was first exposed to 0.8 BED (Biologically Efficient Dose) [32] of UV daylight (UVDL 5.78 J/cm2 UVA + 0.22 J/cm2 UVB) using a BIO-SUN system (Vilber Lourmat, Marne-la-Vallée, France). Immediately following UV exposure, skin was treated topically with 10 mg/cm2 of each test item and covered with a 6 ø mm delivery membrane to ensure its homogeneous distribution. Irradiation and treatment were repeated each day for 5 days (six exposure treatment cycles). On day 3 (D3), 12 skin sections (two sections per skin sample per treatment group) were immunostained with hyaluronic acid binding protein (HABP, Bovine Nasal Cartilage, Biotinylated; Calbiochem® #385911; Calbiochem-Novabiochem Corp., San Diego, CA). On D6, 24 h after the final treatment, 12 skin sections were immunostained with anti-CRABP-II antibody (#PA5-23213; ThermoFisher Scientific, Waltham, MA,), anti-PC1 antibody (Abcam #64409, Cambridge, UK), collagen hybridizing peptide (CHP, Biotin Conjugate [B-CHP]; #BIO300; 3Helix, Salt Lake City, UT), or anti-MMP-1 antibody (Abcam #52631, Cambridge, UK). Total collagen was stained with picrosirius red histochemical stain. Expression of each marker was evaluated by estimating its intensity and distribution in the region of interest (epidermis for CRABP-II; papillary dermis for HABP, total collagen, CHP, MMP-1, and PC1) using ImageJ software (NIH, Bethesda, MD). Differences between groups were analyzed by one-way ANOVA with permutation test, followed by Tukey’s test with permutation. Differences were considered significant at p < 0.05.
Clinical Anti-Photoaging Efficacy
Anti-photoaging efficacy of 3RC was assessed in an open-label single-center non-comparative study involving 34 healthy women aged 40–70 years with skin phototypes I–IV (Fitzpatrick) and moderate to severe photodamage (grade II or higher according to the Glogau photodamage scale; Table 1). Participants were instructed to apply 3RC to the face and neck once a day, at night (two pumps, mean 0.47 g ± 0.13 g per application) for 1 month (28 days).
Table 1.
Glogau photodamage scale
| Grade I | Grade II | Grade III | Grade IV | |
|---|---|---|---|---|
| Classification | Mild | Moderate | Advanced | Severe |
| Description | No wrinkles | Wrinkles in motion | Wrinkles at rest | Only wrinkles |
| Skin characteristics | Early photoaging | Early to moderate photoaging | Advanced photoaging | Severe photoaging |
Ethical Approval
The study protocol reference PT.06.01 V04 of 11 December 2019 was approved on 27 December 2019 by the Independent Ethics Committee of Clinica Dr Carlos Ramos, Lisbon, Portugal. Prior written informed consent was obtained from all subjects and all studies were performed in full respect of the Declaration of Helsinki (1964) and its subsequent amendments and following COLIPA guidelines for the Evaluation of the Efficacy of Cosmetic Products. Good Clinical Practice was maintained throughout. All images are used with prior informed consent from the participants.
Exclusion Criteria
Exclusion criteria included: (i) use of topical retinoids during the 3 months prior to the onset of the study; (ii) having undertaken anti-aging or aesthetic treatment within 6 months of entry into the study; (iii) the initiation or change of hormonal treatment during the study or in the 3 months preceding it; (iv) pregnancy and nursing; (v) intensive sun or UVA exposure during the test period; and (vi) presence of a disease or use of medications that might directly interfere with the study. Concomitant use of products that contain vitamin C, AHAs/BHAs, or other retinoids, and use of exfoliating treatments was also prohibited throughout the study.
Evaluation of Treatment Efficacy
Efficacy was assessed: (1) by measurement of skin firmness (R0) and (2) elasticity (R2) in the face (left or right malar area) using a Cutometer® Dual MPA 580 equipped with a 2 mm probe (Courage + Khazaka electronic GmbH, Köln, Germany); (3) by measurement of crow’s feet wrinkles using Primos® -CR (Canfield Scientific, Parsippany, NJ); and (4) by measurement of skin tone by VISIA® -CR (Canfield Scientific, Parsippany, NJ). All data were analyzed using a Student’s t- or Wilcoxon signed-rank test to evaluate changes between the baseline (D0) and D28 using SPSS software (version 20.0; SPSS Inc., Chicago, IL). Differences were considered significant at p < 0.05.
Skin Compatibility
Skin compatibility was determined on D0 and D28 by (1) measurement of red spots by VISIA® -CR (Canfield Scientific, Parsippany, NJ); (2) dermatologist evaluation of erythema/redness using a 5-point scale where 0 = no erythema, 1: slight erythema/redness, 2: medium erythema/redness, 3: moderate erythema/redness, 4: severe erythema/redness; and (3) by evaluation of burning and stinging by visual analogue scale (VAS) assessment using a continuous scale from 0 (no sensation) to 10 (severe sensation).
Results
Gene Expression Analysis
To understand if the anti-photoaging efficacy of RAL might be enhanced in the presence of natural retinoid analogs such as BAK and VAE, we first examined the impact of combining 0.05% and 0.1% RAL with BAK or VAE on the expression of key genes involved in skin biology using a 3D full-thickness in vitro skin model.
Statistically significant changes in gene expression for each treatment group are shown in Table 2. Notably, BAK enhanced the effect of RAL on genes involved in keratinocyte differentiation (i.e., CALML5, CASP14, KRT14, TP63) and epidermal barrier function (i.e., CDSN, CLDN1, FLG, GRHL3, TGM1, DSG1). Moreover, gene expression changes tended to be greater for the combination of 0.1% RAL with BAK than they were for 0.05% with BAK. In many cases the amplitude of expression with this combination was greater than that observed for 0.05% RA (i.e., for CASP14, KRT14, CDSN, FLG, KRT1, KRT10, DSG1).
Table 2.
Statistically significant changes in gene expression versus the vehicle control group
| Function | Gene | Name | Fold change versus vehicle | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| RAL 0.05% | RAL 0.05%/BAK | RAL 0.05%/VAE | RAL 0.1% | RAL 0.1%/BAK | RAL 0.1%/VAE | ROL 0.5% | ROL 1% | RA 0.05% | |||
| Keratinocyte differentiation | CALML5 | Calmodulin-like 5 | –1.59 | –1.04 | –1.04 | –1.14 | –2.18 | –1.52 | –1.40 | –1.47 | –1.28 |
| CASP14 | Caspase 14 | –1.32 | –1.83 | –1.81 | –1.47 | –2.53 | –1.96 | –1.67 | –1.54 | –1.76 | |
| KRT1 | Keratin 1 | –2.77 | –2.71 | –2.80 | –3.79 | –2.96 | –3.39 | –2.93 | –2.16 | –2.42 | |
| KRT14 | Keratin 14 | –1.54 | –2.03 | –1.88 | –1.87 | –2.90 | –1.89 | –1.82 | –1.92 | –1.99 | |
| KRT10 | Keratin 10 | –2.08 | –2.78 | –2.13 | –2.95 | –2.35 | –2.78 | –2.43 | –1.89 | –1.88 | |
| TP63 | Tumor protein p63 | –1.15 | –1.50 | –1.42 | –1.37 | –3.07 | –1.47 | –2.14 | –1.87 | –1.56 | |
| Barrier function | CDSN | Corneodesmosin | –1.50 | –1.69 | –1.61 | –2.00 | –3.80 | – 1.64 | –1.63 | –1.83 | –1.89 |
| CLDN1 | Claudin 1 | –1.15 | –1.99 | –1.71 | –1.08 | –2.35 | –2.20 | –1.26 | –2.18 | –2.66 | |
| CLDN7 | Claudin 7 | 8.19 | 8.36 | 4.94 | 8.55 | 6.82 | 10.21 | 5.17 | 7.49 | 13.29 | |
| DSC1 | Desmocollin 1 | –4.23 | –4.74 | –2.93 | –5.44 | –4.56 | –7.87 | –4.25 | –4.05 | –4.55 | |
| DSG1 | Desmoglein 1 | –2.26 | –2.41 | –1.95 | –2.59 | –2.81 | –3.54 | –2.48 | –2.55 | –2.60 | |
| FLG | Filaggrin | –2.71 | –4.05 | –3.61 | –2.89 | –5.98 | –3.48 | –4.44 | –3.66 | –4.75 | |
| GRHL3 | Grainyhead-like transcription factor 3 | –1.79 | –2.12 | –1.90 | –1.74 | –2.21 | –2.31 | –1.83 | –2.32 | –2.35 | |
| TGM1 | Transglutaminase 1 | –1.08 | –1.22 | –1.22 | –1.21 | –2.13 | –1.08 | 1.01 | –1.21 | –1.27 | |
| Desquamation | KLK7 | Kallikrein related peptidase 7 | 2.01 | 1.70 | 1.27 | 1.44 | 1.23 | 1.37 | 1.23 | 1.42 | 1.69 |
| Extracellular matrix breakdown | MMP9 | Matrix metallopeptidase 9 | 4.38 | 4.81 | 3.21 | 3.22 | 2.89 | 2.64 | 4.79 | 3.72 | 6.93 |
| Growth factor/pigmentation | BMP4 | Bone morphogenetic protein 4 | –1.54 | –1.15 | –1.23 | –1.21 | –1.38 | –1.79 | –1.58 | –1.76 | –2.18 |
| Growth factor/wound healing | HBEGF | Heparin binding EGF like growth factor | 1.85 | 2.11 | 2.28 | 3.09 | 2.45 | 2.23 | 3.04 | 2.25 | 3.16 |
| Hydration | AQP3 | Aquaporin 3 (Gill blood group) | 3.46 | 2.64 | 1.83 | 2.28 | 1.50 | 1.98 | 1.93 | 1.74 | 2.74 |
| Immune response | DEFB1 | Defensin beta 1 | 1.64 | 1.46 | 1.42 | 1.71 | 1.44 | 1.39 | 1.45 | 1.46 | 2.09 |
| Inflammation | IL1B | Interleukin 1 beta | 2.56 | 3.11 | 4.15 | 4.11 | 6.54 | 2.25 | 6.50 | 1.54 | 1.93 |
| TNF | Tumor necrosis factor | –3.63 | –3.73 | –1.41 | –1.74 | –1.76 | –3.45 | –1.52 | –3.16 | –4.49 | |
RAL retinal, BAK bakuchiol, VAE Vigna aconitifolia extract, ROL retinol, RA retinoic acid
Statistically significant changes (p < 0.05) are listed in bold text. Genes are categorized by skin function and then alphabetically by gene symbol
Similar effects were seen with VAE, with the combination of 0.1% RAL and VAE more strongly enhancing the effect of RAL on numerous genes involved in epidermal barrier function (i.e., CLDN1, CLDN7, GRHL3, DSC1, DSG1) than the combination of 0.05% RAL and VAE.
These data suggest that combining 0.1% RAL with BAK (RAL/BAK) or VAE (RAL/VAE) can potentiate the effect of RAL, especially on biological processes related to keratinocyte differentiation and skin barrier function.
In Vitro Skin Irritation
Given that BAK and VAE appear to enhance the effects of 0.1% RAL, we wanted to ensure that they do not compromise its skin irritation profile. We therefore compared the irritation profiles of 0.1% RAL alone and 0.1% RAL in combination with BAK and VAE (RAL/BAK/VAE) in vitro in RHE by calculating their respective ET50 values. The ET50 for 0.1% RAL and RAL/BAL/VAE were both > 24 h (Table 3), suggesting that they have similar irritation potentials.
Table 3.
In vitro skin irritation potentials of 0.1% RAL and RAL/BAK/VAE
| Sample | Cell viability 4 h (%) | Cell viability 8 h (%) | Cell viability 16 h (%) | Cell viability 24 h (%) | ET50 |
|---|---|---|---|---|---|
| RAL 0.1% | 133 ± 12.9 | 140 ± 13.4 | 161 ± 12.6 | 140 ± 7.4 | > 24 h |
| RAL/BAK/VAE | 133 ± 9.1 | 137 ± 8.5 | 142 ± 30.0 | 138 ± 8.5 | > 24 h |
RAL retinal, BAK bakuchiol, VAE Vigna aconitifolia extract
Data represent the mean cell viability [± standard variation (SD)] of three RHE tissues per timepoint
Ex Vivo Assessment of Retinoid Activity
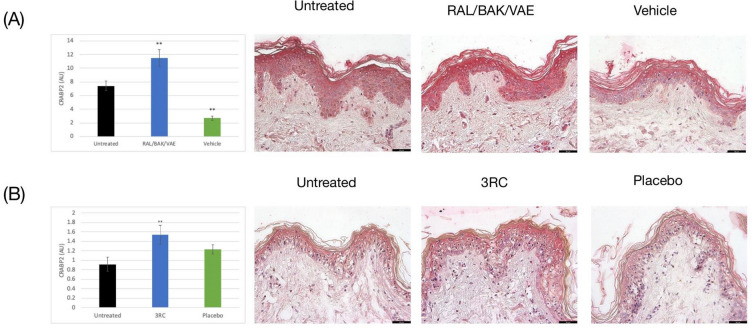
Having established that BAK and VAE enhance RAL activity we next sought to confirm that RAL is effectively converted into RA following topical application to human skin. To assess this, we measured expression levels of CRABP-II, a highly specific marker of RA activity [12]. Treatment of UV-exposed skin with RAL/BAK/VAE increased CRABP-II levels by 55% versus UV exposed skin (p < 0.01; Fig. 1A). Levels were also increased by 33% with respect to non-exposed skin (p < 0.05; data not shown). Conversely, the placebo formulation without RAL, BAK, or VAE had no effect and even appeared to repress CRABP-II expression (−60% with respect to untreated skin; p < 0.01; Fig. 1A).
Fig. 1.
Retinaldehyde is transformed into retinoic acid and activates cellular retinoid acid-binding protein 2 (CRABP-II). A CRABP-II expression following RAL/BAK/VAE treatment. Untreated UV-exposed skin, black bars; RAL/BAK/VAE-treated UV-exposed skin, blue bars; placebo-treated UV-exposed skin, green bars. B CRABP-II expression following 3RC treatment. Untreated UV-exposed skin, black bars; 3RC-treated UV-exposed skin, blue bars; placebo-treated UV-exposed skin, green bars. Graphs show the mean area of CRABP-II expression [± standard error of the mean (SEM)] in 12 independent images per condition. **p < 0.01 versus untreated UV-exposed skin. The black scale bar represents 50 μm
We next sought to confirm that RA was still efficiently produced when RAL/BAK/VAE were complexed with niacinamide and melatonin in an anti-photoaging formulation (3RC). Following topical application of 3RC to UV-exposed skin, CRABP-II levels were increased by 68% (p < 0.05; Fig. 1B). An equivalent placebo formulation without RAL increased CRABP-II expression by only 34% (not significant; Fig. 1B). These data suggest that RAL was efficiently transformed by epidermal enzymes into active RA.
Ex Vivo Anti-Photoaging Effect
Having established that 3RC possesses bona fide retinoid activity, we next examined its effect on key markers of photoaging in an ex vivo model.
Effect on Hyaluronic Acid Levels
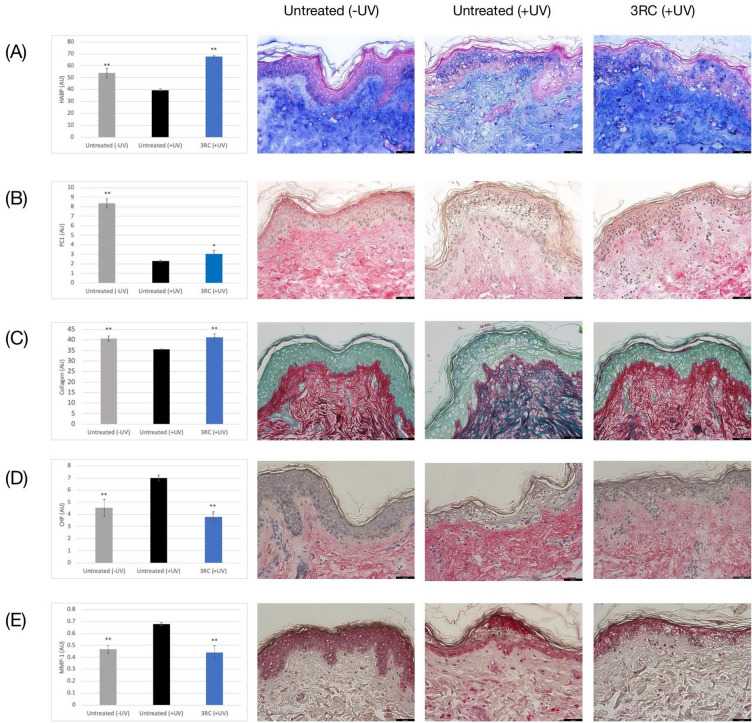
Hyaluronic acid (HA) is a naturally occurring polysaccharide that is highly abundant in the skin [33]. It plays a critical role in retaining moisture and acts as a scaffold for other structural components of the extracellular matrix, thereby helping skin to maintain its hydration, turgor, and flexibility [33]. In our skin model, UV exposure reduced dermal HA levels by 27% (p < 0.01; Fig. 2A). However, treatment with 3RC increased HA expression by 71% with respect to UV-exposed skin, and by 26% with respect to unexposed skin (p < 0.01 both; Fig. 2A). This suggests that 3RC strongly promotes HA expression, which may contribute to wrinkle reduction.
Fig. 2.
Anti-photoaging effect of 3RC. A Effect of 3RC on hyaluronic acid binding protein (HABP) expression. B Effect of 3RC on procollagen I expression. C Effect of 3RC on Total collagen levels. D Effect of 3RC on degraded collagen levels (determined using CHP). E Effect of 3RC on matrix metalloproteinase 1 (MMP-1) expression. Untreated unexposed skin, gray bars; untreated UV-exposed skin, black bars; 3RC-treated UV-exposed skin, blue bars; graphs show the mean area of positive staining (± SEM) in 12 independent images per condition. **p < 0.01 versus untreated UV-exposed skin. The black scale bar represents 50 μm
Effect on Collagen Levels
The anti-photoaging effects of RA can be partly ascribed to its ability to stimulate dermal collagen synthesis and inhibit the production and activity of collagen-degrading matrix metalloproteinases (MMPs) in UV-exposed skin [34–38]. We first determined the effect of 3RC on collagen synthesis by examining its effect on levels of the type I collagen precursor, procollagen type I (PC1). PC1 levels were increased by 33% following 3RC treatment in comparison with untreated UV-exposed skin (p < 0.05; Fig. 2B). Next, we examined the effect of 3RC on its capacity to limit collagen degradation by examining total dermal collagen levels. UV exposure reduced total collagen levels by 13% (p < 0.05; Fig. 2C), but treatment with 3RC completely prevented collagen loss (+16% with respect to UV-exposed skin; p < 0.01; Fig. 2C), and levels were comparable to unexposed skin (Fig. 2C). Degraded collagen was detected using a synthetic collagen hybridizing peptide (CHP) that specifically binds to denatured collagen strands. Not surprisingly, CHP levels were increased by 55% in UV-exposed untreated skin (p < 0.05; Fig. 2D), indicating high levels of UV-induced collagen damage, but treatment with 3RC reduced CHP staining by 46% (p < 0.01; Fig. 2D). In parallel, MMP-1 levels were upregulated by 46% upon UV exposure (p < 0.01; Fig. 2E) but were reduced by 36% upon 3RC treatment (p < 0.01; Fig. 2E). Together these data demonstrate that 3RC helps maintain dermal collagen levels and promotes de novo collagen synthesis, contributing to wrinkle reduction.
Anti-Photoaging Efficacy of 3RC
We next sought to determine the effect of short-term use of 3RC in subjects with clear signs of photoaging and all skin types (including sensitive skin).
Efficacy and skin compatibility were assessed in 32 subjects. A summary of the characteristics of the study population is shown in Table 4.
Table 4.
Characteristic of the study population
| Demographic data | |
|---|---|
| Number (n) | 32 (female 32; 100%) |
| Mean age (min–max) | 53.5 (41–70) |
| Fitzpatrick phototype |
II, 5 (15.6%) III, 26 (81.3%) 1 (3.1%) |
| Skin reactivity |
Normal, 11 (34.4%) Sensitive, 21 (65.6%) |
| Skin condition |
Normal, 6 (18.8%) Dry, 12 (37.5%) Mixed, 13 (40.6%) Oily, 1 (3.1%) |
| Glogau grade |
2 = 10 (31.3%) 3 = 22 (68.8%) |
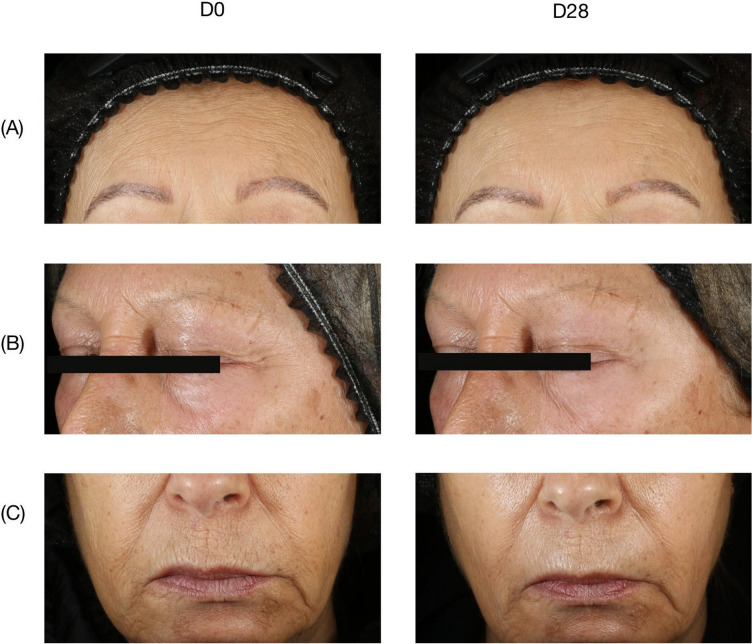
Following 28 days of use, statistically significant improvements to skin firmness (−5.6%; p < 0.05), skin elasticity (+13.9%; p < 0.001), wrinkles count (−43.2%; p < 0.001), and skin tone uniformity (−7.0%; p < 0.05) were observed (Fig. 3; Table 5). Notably, the number of crow’s feet wrinkles was reduced in 100% of subjects. Wrinkle volume and depth, however, were not affected (Table 5).
Fig. 3.
Wrinkle evolution from D0 to D28. A Forehead wrinkles on D0 (left) and D28 (right). B Periocular wrinkles on D0 (left) and D28 (right). C Perioral wrinkles on D0 (left) and D28 (right). All images are of the same subject (subject 11)
Table 5.
Change in aging parameters from baseline (D0) to D28. Data represent the mean values (± SD) at each time point (n = 32)
| Parameter | D0 | D28 | ΔD0–D28 | Change (%) | Responders |
|---|---|---|---|---|---|
| Firmness R0 (mm) | 0.311 (± 0.042) | 0.293 (± 0.050) | −0.018 (± 0.039)* | −5.6 (± 12.9)* | 24/32 (75.0%) |
| Elasticity R2 (%) | 0.553 (± 0.099) | 0.619 (± 0.091) | 0.066 (± 0.090)*** | +13.9 (± 19.9)*** | 25/32 (78.1%) |
| Wrinkles count (n) | 349.2 (± 162.8) | 197.9 (± 96.2) | −151.2 (± 79.2)*** | −43.2 (± 8.5)*** | 32/32 (100.0%) |
| Wrinkles volume (mm3) | 16.2 (± 7.6) | 15.1 (± 6.0) | −1.2 (± 3.5) | −3.8 (± 18.4) | 19/32 (59.4%) |
| Wrinkles depth (μm) | 113.6 (± 45.8) | 120.4 (± 45.2) | 6.4 (± 22.5) | +9.7 (± 23.2) | 11/32 (34.4%) |
| Skin tone uniformity (Arbitrary units) | 0.136 (± 0.035) | 0.121 (± 0.032) | −0.015 (± 0.035)* | −7.0 (± 28.1)* | 22/32 (68.8%) |
The number of responders to 3RC, defined as any positive change in the specified parameter on D28 versus D0, is indicated. *p < 0.05; ***p < 0.001
Skin Compatibility
Given that the irritating effects of retinoids are one of the principal reasons for non-compliance and thus lack of efficacy [16], skin compatibility of 3RC was determined in the same study population using a combination of instrumental measurement and clinical scoring. No significant change in erythema/redness, as determined by measuring red spots number and area by VISIA® -CR, and by clinical scoring, was observed (Table 6). Burning and stinging were also not significantly increased after treatment on D0 or D28 (Table 7). Notably, no subject was required to deviate from the recommended application protocol (i.e., once per day at night) throughout the course of the study.
Table 6.
Effect of 3RC on erythema/redness
| Parameter | D0 | D28 | ΔD0–D28 | Change (%) |
|---|---|---|---|---|
| Red spots count (n) | 97.5 (± 35.4) | 102.1 (± 34.4) | 4.6 (± 17.2) | 6.9% (± 16.8) |
| Red spots area (%) | 8.9 (± 3.6) | 9.4 (± 3.0) | 0.4 (± 1.6) | 9.8% (± 22.5) |
| Erythema/redness | 0.67 (± 0.69) | 0.73 (± 0.70) | 0.06 (± 0.21) | 11.7% (± 31.7) |
Data represent the mean visual analogue scale (VAS) scores (± SD) at each time point (n = 32)
Table 7.
Effect of 3RC on burning and stinging
| D0 | D28 | ||
|---|---|---|---|
| Pre | Post | ||
| VAS burning | 0.03 (± 0.18) | 0.12 (± 0.41) | 0.15 (± 0.49) |
| VAS stinging | 0 (± 0) | 0.18 (± 0.53) | 0.07 (± 0.23) |
Discussion
As the immediate precursor to RA, RAL is best placed of the retinoids to deliver anti-photoaging benefits. RAL has proven equally as effective as RA at reducing wrinkles and roughness in aged and photoaged skin, but with a lower irritation potential [13, 15, 39, 40]. The effects of RAL, however, are not instant, with months of continuous use needed to see significant benefit. Considering this, we asked whether faster anti-photoaging effects could be delivered by combining 0.1% RAL with natural retinol-like ingredients, without compromising RAL’s safety profile.
In this study we show that two such retinol-like ingredients appear to potentiate the effects of RAL. Combination of 0.1% RAL with either BAK or VAE enhances the effect of RAL on genes involved in keratinocyte differentiation and epidermal barrier function. There are several possible explanations as to why this occurs. The first, and simplest, is that it is an additive effect of the active ingredients in isolation. Several studies, however, allude to alternative mechanisms. Gene expression studies have shown that BAK modulates the expression of genes involved in retinoid binding and metabolism, including upregulating the gene encoding CRABP-II, responsible for the nuclear translocation of RA [41–43], upregulating DHRS9, the gene encoding one of the retinol dehydrogenases (dehydrogenase/reductase SDR family member 9) responsible for the production of RAL from ROL, and upregulating LRAT, encoding the retinyl esterase (lecithin retinol acyltransferase) responsible for esterification of ROL to retinyl esters [20]. This suggests that BAK might potentiate RA signaling firstly by increasing its translocation to the nucleus through CRABP-II, and secondly by helping to ensure that the skin maintains a high cellular pool of retinyl esters that can be readily converted back to RA later. Interestingly, we observed a 34% increase in CRABP-II expression following application of a formulation containing all components of 3RC except RAL. Although this increase in CRABP-II expression is not statistically significant, it suggests that BAK or some other component of 3RC may potentiate RA signaling by promoting CRABP-II expression. Studies have also shown that BAK dose-dependently increases the stability of ROL [44]. All retinoids are liable to light, heat, and oxidative degradation, rendering them ineffective [45], so preventing this should help maintain optimum efficacy. Whether BAK stabilizes RAL is not known, but given the structural similarities of ROL and RAL, it seems plausible. The biological mechanisms of VAE are less well characterized and further studies are required.
Despite the enhanced activity of RAL when combined with BAK or VAE, its irritation potential proved similar. This good safety profile was subsequently confirmed when a cosmetic formulation, 3RC, containing RAL/BAK/VAE was tested clinically. Irritation remains the single biggest obstacle to successful retinoid therapy, and frequently necessitates an adaptation period, when the skin is conditioned, or “retinized” to permit long-term use. With 3RC, however, such measures proved unnecessary, and all subjects were able to successfully apply 3RC daily for the duration of the study, irrespective of their skin type and condition.
3RC was also able to deliver rapid anti-photoaging benefits after 28 days of application, with statistically significant changes to all measured parameters, including skin firmness and elasticity, skin tone, and wrinkle counts. Remarkably the number of wrinkles was decreased in 100% of subjects, but surprisingly no effect on wrinkle volume or depth was seen. Previous profilometric evaluation of skin treated with 0.05% RAL saw significant changes to the depth of fine wrinkles only after 18 weeks of treatment [14, 15], suggesting that such changes might only occur with longer term use.
The results of the gene expression and anti-photoaging studies give further insight into the possible mechanisms behind the efficacy of 3RC. One of the best-known effects of RA in the epidermis is to inhibit keratinocyte differentiation [3, 46, 47], and BAK, and to a lesser extent VAE, enhances the effect of RAL on this process, and indeed appears as potent as RA in doing so. By regulating keratinocyte proliferation and differentiation, retinoids increase stratum granulosum thickness, helping to reverse age-associated skin atrophy [48]. Furthermore, we show that the 3RC reverses UV-induced deficits in important structural components of the dermal extracellular matrix, including HA and collagen, helping to explain why wrinkles were reduced and skin’s firmness and viscoelastic properties were restored clinically upon 3RC treatment. Epidermal retinyl esters have also been shown to absorb UVB radiation and prevent erythema and DNA damage [49]. Thus, the increased levels of these esters in the skin following 3RC application may also help mitigate further photodamage through their UV filter effects.
Cosmetic formulations often include other ingredients with complementary actions to increase their efficacy or bring additional benefits, and melatonin and niacinamide were included in 3RC for these reasons. Both melatonin and niacinamide help protect skin against environmental damage to maintain its homeostasis and mitigate the clinical signs of aging [50, 51]. The diverse anti-aging effects of niacinamide are principally a result of it serving as a precursor to the enzymatic cofactor nicotinamide adenine dinucleotide (NAD+) [51]. Interestingly, formation of RA from RAL is strongly increased in the presence of NAD+ [11]. On this basis, it would be interesting to examine RAL metabolism in the presence and absence of niacinamide to demonstrate if this is the case with 3RC.
3RC was also able to improve skin tone uniformity. Niacinamide, melatonin, BAK, and RAL all have an inhibitory effect on hyperpigmentation, explaining the improvements seen: niacinamide inhibits the transfer of melanosomes from melanocytes to keratinocytes [30], melatonin inhibits the crosstalk between keratinocytes and melanocytes that initiates melanogenesis [52] and can prevent melanin synthesis and melanocyte proliferation [53], BAK has been shown to decrease melanin production by inhibiting the expression of tyrosinase [54], and preventing melanin transfer to keratinocytes [54], and RAL inhibits melanin production [55] and promotes epidermal turnover and desquamation [56].
Anti-photoaging effects of 3RC were seen within 1 month of use, which was sooner than that typically observed using 0.1% RAL alone [18]. Without a comparison with 0.1% RAL alone, it is obviously impossible to claim with any certainty that the presence of BAK and VAE are responsible for this effect, but combination of RAL with other ingredients has also previously been reported to deliver anti-aging benefits in a short time. When 0.1% RAL is combined with 0.1% delta-tocopherol glucoside, a precursor of the antioxidant vitamin E, and 0.1% glycylglycine oleamide, a small amphiphilic molecule that protects the connective tissue of the skin from glycation and elastosis, significant improvements to periocular fine lines and crow’s feet are seen within 2 months of treatment [57]. Similarly, combining 0.05% RAL with 0.5% or 1% intermediate-size HA fragments significantly improved the overall photoaging score, reduced facial wrinkles, and improved clinical signs of aging following 1 month of use [58]. Combining 0.05% RAL with the vitamin E precursor pre-tocopheryl has also been shown to protect ex vivo human skin from UV-mediated damage better than 0.05% RAL alone [59]. These data suggest that synergism between RAL and other anti-aging components does occur and can deliver significant benefits over RAL alone, so comparative studies of 3RC against 0.1% RAL alone would be valuable.
The limitation of this proof-of-concept clinical study is the absence of a control or placebo group that would permit objective evaluation of its efficacy. Future longer-term studies would also be useful to evaluate its potential on wrinkle volume and depth. Moreover, by extending this study to 3 months, it would allow a direct comparison of the efficacy of 3RC with other studies using 0.01% RAL alone for the treatment of photoaging, such as that of Kwon et al. [18]. It would also be interesting to evaluate the effect of RAL in combination with both BAK and VAE on keratinocyte function in vitro to determine the effect of this combination.
Conclusion
Using a rational approach to cosmetic product development we have shown how the anti-photoaging effects of RAL can be enhanced by combining it with the natural retinol-like ingredients BAK and VAE, without affecting its preferential safety profile. Reductions in clinical signs of aging and photodamage are observed within one month of once daily use, making 3RC a highly effective and well-tolerated alternative treatment for photoaging.
Acknowledgements
The authors thank all subjects for their participation in the study; Cutech, Padova, Italy, who performed the ex vivo study; and PhD Trials® , Lisbon, Portugal who performed the clinical studies.
Author Contributions
Anthony Brown, Marta Furmanczyk, Corinne Granger, and David Ramos designed the studies. Anthony Brown wrote the paper. Anthony Brown, Marta Furmanczyk, David Ramos, Adria Ribes, Laia Pons, Javier Bustos, Antonio R. Fernandez de Henestrosa, Corinne Granger, and Eric Jourdan revised the manuscript.
Funding
All studies and the journal’s rapid service fee was wholly funded by ISDIN, the manufacturer of the formulation under study.
Medical Writing and Editorial Assistance
No medical writing or editorial assistance was received.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Ethical Approval
The clinical study protocol reference PT.06.01 V04 of 11 December 2019 was approved on 27 December 2019 by the Independent Ethics Committee of Clinica Dr Carlos Ramos, Lisbon, Portugal. Prior written informed consent was obtained from all subjects and all studies were performed in full respect of the Declaration of Helsinki (1964) and its subsequent amendments and following COLIPA guidelines for the Evaluation of the Efficacy of Cosmetic Products. Good Clinical Practice was maintained throughout. All images are used with prior informed consent from the participants.
All skin explants used in this study were obtained from surgical residues of abdominal skin obtained from a Caucasian woman (53 years of age; ITA° = 30°) after written informed consent from the donor and in full respect of the Declaration of Helsinki and article L.1245–2 of the French Public Health Code. The latter does not require any prior authorization by an ethics committee for use of surgical waste.
Conflict of Interest
Anthony Brown, Marta Furmanczyk, David Ramos, Adria Ribes, Laia Pons, Javier Bustos, Antonio R. Fernandez de Henestrosa, and Eric Jourdan are employees of ISDIN, the manufacturer of the formulation under study. Corinne Granger was employed by ISDIN at the time of the studies and acts as a paid consultant to ISDIN.
References
- 1.Kligman LH, Duo CH, Kligman AM. Topical retinoic acid enhances the repair of ultraviolet damaged dermal connective tissue. Connect Tissue Res. 1984;12(2):139–150. doi: 10.3109/03008208408992779. [DOI] [PubMed] [Google Scholar]
- 2.Kligman AM, Grove GL, Hirose R, Leyden JJ. Topical tretinoin for photoaged skin. J Am Acad Dermatol. 1986;15(4 Pt 2):836–859. doi: 10.1016/S0190-9622(86)70242-9. [DOI] [PubMed] [Google Scholar]
- 3.Lee DD, Stojadinovic O, Krzyzanowska A, Vouthounis C, Blumenberg M, Tomic-Canic M. Retinoid-responsive transcriptional changes in epidermal keratinocytes. J Cell Physiol. 2009;220(2):427–439. doi: 10.1002/jcp.21784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Giguere V, Ong ES, Segui P, Evans RM. Identification of a receptor for the morphogen retinoic acid. Nature. 1987;330(6149):624–629. doi: 10.1038/330624a0. [DOI] [PubMed] [Google Scholar]
- 5.Xiao JH, Durand B, Chambon P, Voorhees JJ. Endogenous retinoic acid receptor (RAR)-retinoid X receptor (RXR) heterodimers are the major functional forms regulating retinoid-responsive elements in adult human keratinocytes. Binding of ligands to RAR only is sufficient for RAR-RXR heterodimers to c. J Biol Chem. 1995;270(7):3001–11. doi: 10.1074/jbc.270.7.3001. [DOI] [PubMed] [Google Scholar]
- 6.Leyden JJ, Grove GL, Grove MJ, Thorne EG, Lufrano L. Treatment of photodamaged facial skin with topical tretinoin. J Am Acad Dermatol. 1989;21(3 Pt 2):638–644. doi: 10.1016/S0190-9622(89)70231-0. [DOI] [PubMed] [Google Scholar]
- 7.Shukuwa T, Kligman A, Stoudemayer T. The effect of short-term (one-month) topical tretinoin on photodamaged forearm skin. J Dermatol Treat. 1993;4(3):139–143. doi: 10.3109/09546639309080553. [DOI] [Google Scholar]
- 8.Kong R, Cui Y, Fisher GJ, Wang X, Chen Y, Schneider LM, et al. A comparative study of the effects of retinol and retinoic acid on histological, molecular, and clinical properties of human skin. J Cosmet Dermatol. 2016;15(1):49–57. doi: 10.1111/jocd.12193. [DOI] [PubMed] [Google Scholar]
- 9.Lever L, Kumar P, Marks R. Topical retinoic acid for treatment of solar damage. Br J Dermatol. 1990;122(1):91–98. doi: 10.1111/j.1365-2133.1990.tb08244.x. [DOI] [PubMed] [Google Scholar]
- 10.Kligman DE, Sadiq I, Pagnoni A, Stoudemayer T, Kligman AM. High-strength tretinoin: a method for rapid retinization of facial skin. J Am Acad Dermatol. 1998;39(2 Pt 3):S93–S97. doi: 10.1016/S0190-9622(98)70454-2. [DOI] [PubMed] [Google Scholar]
- 11.Siegenthaler G, Saurat JH, Ponec M. Retinol and retinal metabolism. Relationship to the state of differentiation of cultured human keratinocytes. Biochem J. 1990;268(2):371–8. doi: 10.1042/bj2680371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Saurat JH, Didierjean L, Masgrau E, Piletta PA, Jaconi S, Chatellard-Gruaz D, et al. Topical retinaldehyde on human skin: biologic effects and tolerance. J Invest Dermatol. 1994;103(6):770–774. doi: 10.1111/1523-1747.ep12412861. [DOI] [PubMed] [Google Scholar]
- 13.Diridollou S, Vienne MP, Alibert M, Aquilina C, Briant A, Dahan S, et al. Efficacy of topical 0.05% retinaldehyde in skin aging by ultrasound and rheological techniques. Dermatology. 1999;199:37–41. doi: 10.1159/000051377. [DOI] [PubMed] [Google Scholar]
- 14.Creidi P, Vienne MP, Ochonisky S, Lauze C, Turlier V, Lagarde JM, et al. Profilometric evaluation of photodamage after topical retinaldehyde and retinoic acid treatment. J Am Acad Dermatol. 1998;39(6):960–965. doi: 10.1016/S0190-9622(98)70270-1. [DOI] [PubMed] [Google Scholar]
- 15.Creidi P, Humbert P. Clinical use of topical retinaldehyde on photoaged skin. Dermatology. 1999;199(Suppl):49–52. doi: 10.1159/000051379. [DOI] [PubMed] [Google Scholar]
- 16.Fluhr JW, Vienne MP, Lauze C, Dupuy P, Gehring W, Gloor M. Tolerance profile of retinol, retinaldehyde and retinoic acid under maximized and long-term clinical conditions. Dermatology. 1999;199(Suppl):57–60. doi: 10.1159/000051381. [DOI] [PubMed] [Google Scholar]
- 17.Sachsenberg-Studer EM. Tolerance of topical retinaldehyde in humans. Dermatology. 1999;199(Suppl):61–63. doi: 10.1159/000051382. [DOI] [PubMed] [Google Scholar]
- 18.Kwon HS, Lee JH, Kim GM, Bae JM. Efficacy and safety of retinaldehyde 0.1% and 0.05% creams used to treat photoaged skin: a randomized double-blind controlled trial. J Cosmet Dermatol. 2018;17(3):471–6. doi: 10.1111/jocd.12551. [DOI] [PubMed] [Google Scholar]
- 19.Mehta G, Nayak UR, Dev S. Meroterpenoids—I: Psoralea corylifolia Linn.—1. Bakuchiol, a novel monoterpene phenol. Tetrahedron. 1973;29(8):1119–25. doi: 10.1016/0040-4020(73)80071-7. [DOI] [Google Scholar]
- 20.Chaudhuri RK, Bojanowski K. Bakuchiol: a retinol-like functional compound revealed by gene expression profiling and clinically proven to have anti-aging effects. Int J Cosmet Sci. 2014;36(3):221–230. doi: 10.1111/ics.12117. [DOI] [PubMed] [Google Scholar]
- 21.Draelos ZD, Gunt H, Zeichner J, Levy S. Clinical evaluation of a nature-based bakuchiol anti-aging moisturizer for sensitive skin. J Drugs Dermatol. 2020;19(12):1181–1183. doi: 10.36849/JDD.2020.5522. [DOI] [PubMed] [Google Scholar]
- 22.Goldberg DJ, Robinson DM, Granger C. Clinical evidence of the efficacy and safety of a new 3-in-1 anti-aging topical night serum-in-oil containing melatonin, bakuchiol, and ascorbyl tetraisopalmitate: 103 females treated from 28 to 84 days. J Cosmet Dermatol. 2019;18(3):806–814. doi: 10.1111/jocd.12896. [DOI] [PubMed] [Google Scholar]
- 23.Bacqueville D, Maret A, Noizet M, Duprat L, Coutanceau C, Georgescu V, et al. Efficacy of a dermocosmetic serum combining bakuchiol and vanilla tahitensis extract to prevent skin photoaging in vitro and to improve clinical outcomes for naturally aged skin. Clin Cosmet Investig Dermatol. 2020;13:359–370. doi: 10.2147/CCID.S235880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dhaliwal S, Rybak I, Ellis SR, Notay M, Trivedi M, Burney W, et al. Prospective, randomized, double-blind assessment of topical bakuchiol and retinol for facial photoageing. Br J Dermatol. 2019;180(2):289–296. doi: 10.1111/bjd.16918. [DOI] [PubMed] [Google Scholar]
- 25.Bhadkaria A, Srivastava N, Bhagyawant SS. A prospective of underutilized legume moth bean (Vigna aconitifolia (Jacq.) Marechàl): phytochemical profiling, bioactive compounds and in vitro pharmacological studies. Food Biosci. 2021;42:101088. doi: 10.1016/j.fbio.2021.101088. [DOI] [Google Scholar]
- 26.Slominski A, Wortsman J, Tobin DJ. The cutaneous serotoninergic/melatoninergic system: securing a place under the sun. FASEB J [Internet]. 2005 Feb [Cited 2023 Jul 27];19(2):176–94. Available from: https://pubmed.ncbi.nlm.nih.gov/15677341/ [DOI] [PubMed]
- 27.Skobowiat C, Brożyna AA, Janjetovic Z, Jeayeng S, Oak ASW, Kim TK, et al. Melatonin and its derivatives counteract the ultraviolet B radiation-induced damage in human and porcine skin ex vivo. J Pineal Res [Internet]. 2018 Sep 1 [Cited 2023 Jul 27];65(2). Available from: https://pubmed.ncbi.nlm.nih.gov/29702749/ [DOI] [PMC free article] [PubMed]
- 28.Slominski AT, Hardeland R, Zmijewski MA, Slominski RM, Reiter RJ, Paus R. Melatonin: A cutaneous perspective on its production, metabolism, and functions. J Invest Dermatol [Internet]. 2018 Mar 1 [Cited 2023 Jul 27];138(3):490–9. Available from: https://pubmed.ncbi.nlm.nih.gov/29428440/ [DOI] [PMC free article] [PubMed]
- 29.Tanno O, Ota Y, Kitamura N, Katsube T, Inoue S. Nicotinamide increases biosynthesis of ceramides as well as other stratum corneum lipids to improve the epidermal permeability barrier. Br J Dermatol. 2000;143(3):524–531. doi: 10.1111/j.1365-2133.2000.03705.x. [DOI] [PubMed] [Google Scholar]
- 30.Hakozaki T, Minwalla L, Zhuang J, Chhoa M, Matsubara A, Miyamoto K, et al. The effect of niacinamide on reducing cutaneous pigmentation and suppression of melanosome transfer. Br J Dermatol. 2002;147(1):20–31. doi: 10.1046/j.1365-2133.2002.04834.x. [DOI] [PubMed] [Google Scholar]
- 31.Philips N, Chalensouk-Khaosaat J, Gonzalez S, Philips N, Chalensouk-Khaosaat JGS. Stimulation of the fibrillar collagen and heat shock proteins by nicotinamide or its derivatives in non-irradiated or UVA radiated fibroblasts, and direct anti-oxidant activity of nicotinamide derivatives. Cosmetics. 2015;2(2):146–161. doi: 10.3390/cosmetics2020146. [DOI] [Google Scholar]
- 32.Del Bino S, Sok J, Bessac E, Bernerd F. Relationship between skin response to ultraviolet exposure and skin color type. Pigment Cell Res. 2006;19(6):606–614. doi: 10.1111/j.1600-0749.2006.00338.x. [DOI] [PubMed] [Google Scholar]
- 33.Šínová R, Pavlík V, Ondrej M, Velebný V, Nešporová K. Hyaluronan: a key player or just a bystander in skin photoaging? Exp Dermatol. 2022;31(4):442–458. doi: 10.1111/exd.14491. [DOI] [PubMed] [Google Scholar]
- 34.Griffiths CE, Russman AN, Majmudar G, Singer RS, Hamilton TA, Voorhees JJ. Restoration of collagen formation in photodamaged human skin by tretinoin (retinoic acid) N Engl J Med. 1993;329(8):530–535. doi: 10.1056/NEJM199308193290803. [DOI] [PubMed] [Google Scholar]
- 35.Fisher GJ, Datta SC, Talwar HS, Wang ZQ, Varani J, Kang S, et al. Molecular basis of sun-induced premature skin ageing and retinoid antagonism. Nature. 1996;379(6563):335–339. doi: 10.1038/379335a0. [DOI] [PubMed] [Google Scholar]
- 36.Fisher GJ, Talwar HS, Lin J, Voorhees JJ. Molecular mechanisms of photoaging in human skin in vivo and their prevention by all-trans retinoic acid. Photochem Photobiol. 1999;69(2):154–157. doi: 10.1562/0031-8655(1999)069<0154:mmopih>2.3.co;2. [DOI] [PubMed] [Google Scholar]
- 37.Fisher GJ, Datta S, Wang Z, Li XY, Quan T, Chung JH, et al. c-Jun-dependent inhibition of cutaneous procollagen transcription following ultraviolet irradiation is reversed by all-trans retinoic acid. J Clin Invest. 2000;106(5):663–670. doi: 10.1172/JCI9362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fisher GJ, Choi HC, Bata-Csorgo Z, Shao Y, Datta S, Wang ZQ, et al. Ultraviolet irradiation increases matrix metalloproteinase-8 protein in human skin in vivo. J Invest Dermatol. 2001;117(2):219–226. doi: 10.1046/j.0022-202x.2001.01432.x. [DOI] [PubMed] [Google Scholar]
- 39.Mukherjee S, Date A, Patravale V, Korting HC, Roeder A, Weindl G. Retinoids in the treatment of skin aging: an overview of clinical efficacy and safety. Clin Interv Aging. 2006;1(4):327–348. doi: 10.2147/ciia.2006.1.4.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Darlenski R, Surber C, Fluhr JW. Topical retinoids in the management of photodamaged skin: from theory to evidence-based practical approach. Br J Dermatol. 2010;163(6):1157–1165. doi: 10.1111/j.1365-2133.2010.09936.x. [DOI] [PubMed] [Google Scholar]
- 41.Schug TT, Berry DC, Shaw NS, Travis SN, Noy N. Opposing effects of retinoic acid on cell growth result from alternate activation of two different nuclear receptors. Cell. 2007;129(4):723–733. doi: 10.1016/j.cell.2007.02.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sessler RJ, Noy N. A ligand-activated nuclear localization signal in cellular retinoic acid binding protein-II. Mol Cell. 2005;18(3):343–353. doi: 10.1016/j.molcel.2005.03.026. [DOI] [PubMed] [Google Scholar]
- 43.Sanquer S, Eller MS, Gilchrest BA. Retinoids and state of differentiation modulate CRABP II gene expression in a skin equivalent. J Invest Dermatol. 1993;100(2):148–153. doi: 10.1111/1523-1747.ep12462785. [DOI] [PubMed] [Google Scholar]
- 44.Chaudhuri RK. Cosmetics & Toiletries. 2015. Bakuchiol to Stabilize Retinol and Polyunsaturated Lipids. Available from: https://www.cosmeticsandtoiletries.com/cosmetic-ingredients/article/21835331/bakuchiol-to-stabilize-retinol-and-polyunsaturated-lipids. Accessed June 2023.
- 45.Temova Rakuša Ž, Škufca P, Kristl A, Roškar R. Retinoid stability and degradation kinetics in commercial cosmetic products. J Cosmet Dermatol. 2021;20(7):2350–2358. doi: 10.1111/jocd.13852. [DOI] [PubMed] [Google Scholar]
- 46.Fuchs E, Green H. Regulation of terminal differentiation of cultured human keratinocytes by vitamin A. Cell. 1981;25(3):617–625. doi: 10.1016/0092-8674(81)90169-0. [DOI] [PubMed] [Google Scholar]
- 47.Jetten AM. Multi-stage program of differentiation in human epidermal keratinocytes: regulation by retinoids. J Invest Dermatol. 1990;95(5 Suppl):44S–46S. doi: 10.1111/1523-1747.ep12505757. [DOI] [PubMed] [Google Scholar]
- 48.Roeder A, Schaller M, Schäfer-Korting M, Korting HC. Tazarotene: therapeutic strategies in the treatment of psoriasis, acne and photoaging. Skin Pharmacol Physiol. 2004;17(3):111–118. doi: 10.1159/000077236. [DOI] [PubMed] [Google Scholar]
- 49.Antille C, Tran C, Sorg O, Carraux P, Didierjean L, Saurat JH. Vitamin A exerts a photoprotective action in skin by absorbing ultraviolet B radiation. J Invest Dermatol [Internet]. 2003 [Cited 2023 Jul 27];121(5):1163–7. Available from: https://pubmed.ncbi.nlm.nih.gov/14708621/ [DOI] [PubMed]
- 50.Rusanova I, Martínez-Ruiz L, Florido J, Rodríguez-Santana C, Guerra-Librero A, Acuña-Castroviejo D, et al. Protective effects of melatonin on the skin: future perspectives. Int J Mol Sci. 2019;20(19):4948. doi: 10.3390/ijms20194948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Oblong JE. The evolving role of the NAD+/nicotinamide metabolome in skin homeostasis, cellular bioenergetics, and aging. DNA Repair (Amst) 2014;23:59–63. doi: 10.1016/j.dnarep.2014.04.005. [DOI] [PubMed] [Google Scholar]
- 52.Yang S, Zeng H, Jiang L, Fu C, Gao L, Zhang L, et al. Melatonin reduces melanogenesis by inhibiting the paracrine effects of keratinocytes. Exp Dermatol. 2023;32(4):511–520. doi: 10.1111/exd.14743. [DOI] [PubMed] [Google Scholar]
- 53.Kim TK, Lin Z, Tidwell WJ, Li W, Slominski AT. Melatonin and its metabolites accumulate in the human epidermis in vivo and inhibit proliferation and tyrosinase activity in epidermal melanocytes in vitro. Mol Cell Endocrinol. 2015;404:1–8. doi: 10.1016/j.mce.2014.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kang MC, Lee JW, Lee TH, Subedi L, Wahedi HM, Do SG, et al. UP256 inhibits hyperpigmentation by tyrosinase expression/dendrite formation via rho-dependent signaling and by primary cilium formation in melanocytes. Int J Mol Sci. 2020;21(15):5341. doi: 10.3390/ijms21155341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sorg O, Kasraee B, Salomon D, Saurat JH. The potential depigmenting activity of retinaldehyde. Dermatology. 2013;227(3):231–237. doi: 10.1159/000354294. [DOI] [PubMed] [Google Scholar]
- 56.Ebanks JP, Wickett RR, Boissy RE. Mechanisms regulating skin pigmentation: the rise and fall of complexion coloration. Int J Mol Sci. 2009;10(9):4066–4087. doi: 10.3390/ijms10094066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Rouvrais C, Bacqueville D, Bogdanowicz P, Haure MJ, Duprat L, Coutanceau C, et al. A new dermocosmetic containing retinaldehyde, delta-tocopherol glucoside and glycylglycine oleamide for managing naturally aged skin: results from in vitro to clinical studies. Clin Cosmet Investig Dermatol. 2017;10:35–42. doi: 10.2147/CCID.S123575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cordero A, Leon-Dorantes G, Pons-Guiraud A, Di Pietro A, Asensi SV, Walkiewicz-Cyraska B, et al. Retinaldehyde/hyaluronic acid fragments: a synergistic association for the management of skin aging. J Cosmet Dermatol. 2011;10(2):110–117. doi: 10.1111/j.1473-2165.2011.00552.x. [DOI] [PubMed] [Google Scholar]
- 59.Boisnic S, Branchet-Gumila MC, Nocera T. Comparative study of the anti-aging effect of retinaldehyde alone or associated with pretocopheryl in a surviving human skin model submitted to ultraviolet A and B irradiation. Int J Tissue React. 2005;27(3):91–99. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.