Abstract
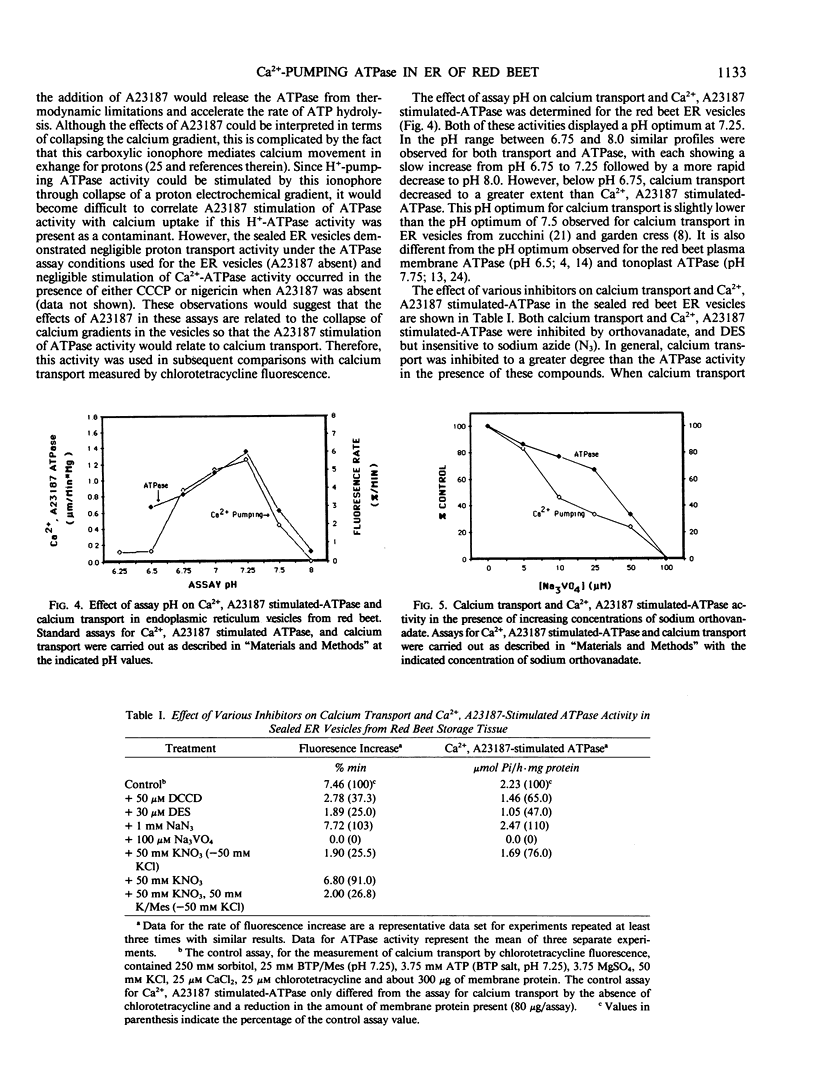
Calcium transport was examined in microsomal membrane vesicles from red beet (Beta vulgaris L.) storage tissue using chlorotetracycline as a fluorescent probe. This probe demonstrates an increase in fluorescence corresponding to calcium accumulation within the vesicles which can be collapsed by the addition of the calcium ionophore A23187. Calcium uptake in the microsomal vesicles was ATP dependent and completely inhibited by orthovanadate. Centrifugation of the microsomal membrane fraction on a linear 15 to 45% (w/w) sucrose density gradient revealed the presence of a single peak of calcium uptake which comigrated with the marker for endoplasmic reticulum. The calcium transport system associated with endoplasmic reticulum vesicles was then further characterized in fractions produced by centrifugation on discontinous sucrose density gradients. Calcium transport was insensitive to carbonylcyanide m-chlorophenylhydrazone indicating the presence of a primary transport system directly linked to ATP utilization. The endoplasmic reticulum vesicles contained an ATPase activity that was calcium dependent and further stimulated by A23187 (Ca2+, A23187 stimulated-ATPase). Both calcium uptake and Ca2+, A23187 stimulated ATPase demonstrated similar properties with respect to pH optimum, inhibitor sensitivity, substrate specificity, and substrate kinetics. Treatment of the red beet endoplasmic reticulum vesicles with [γ-32P]-ATP over short time intervals revealed the presence of a rapidly turning over 96 kilodalton radioactive peptide possibly representing a phosphorylated intermediate of this endoplasmic reticulum associated ATPase. It is proposed that this ATPase activity may represent the enzymic machinery responsible for mediating primary calcium transport in the endoplasmic reticulum linked to ATP utilization.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blumwald E., Poole R. J. Kinetics of Ca/H Antiport in Isolated Tonoplast Vesicles from Storage Tissue of Beta vulgaris L. Plant Physiol. 1986 Mar;80(3):727–731. doi: 10.1104/pp.80.3.727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Briskin D. P., Poole R. J. Characterization of a k-stimulated adenosine triphosphatase associated with the plasma membrane of red beet. Plant Physiol. 1983 Feb;71(2):350–355. doi: 10.1104/pp.71.2.350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briskin D. P., Poole R. J. Plasma membrane ATPase of red beet forms a phosphorylated intermediate. Plant Physiol. 1983 Mar;71(3):507–512. doi: 10.1104/pp.71.3.507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briskin D. P., Thornley W. R., Wyse R. E. Membrane transport in isolated vesicles from sugarbeet taproot : I. Isolation and characterization of energy-dependent, h-transporting vesicles. Plant Physiol. 1985 Aug;78(4):865–870. doi: 10.1104/pp.78.4.865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckhout T. J. Characterization of Ca Transport in Purified Endoplasmic Reticulum Membrane Vesicles from Lepidium sativum L. Roots. Plant Physiol. 1984 Dec;76(4):962–967. doi: 10.1104/pp.76.4.962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bush D. R., Sze H. Calcium transport in tonoplast and endoplasmic reticulum vesicles isolated from cultured carrot cells. Plant Physiol. 1986 Feb;80(2):549–555. doi: 10.1104/pp.80.2.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dieter P., Marmé D. Calmodulin activation of plant microsomal Ca uptake. Proc Natl Acad Sci U S A. 1980 Dec;77(12):7311–7314. doi: 10.1073/pnas.77.12.7311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drøbak B. K., Ferguson I. B. Release of Ca2+ from plant hypocotyl microsomes by inositol-1,4,5-trisphosphate. Biochem Biophys Res Commun. 1985 Aug 15;130(3):1241–1246. doi: 10.1016/0006-291x(85)91747-4. [DOI] [PubMed] [Google Scholar]
- Giannini J. L., Briskin D. P. Proton Transport in Plasma Membrane and Tonoplast Vesicles from Red Beet (Beta vulgaris L.) Storage Tissue : A Comparative Study of Ion Effects on DeltapH and DeltaPsi. Plant Physiol. 1987 Jul;84(3):613–618. doi: 10.1104/pp.84.3.613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giannini J. L., Ruiz-Cristin J., Briskin D. P. Calcium Transport in Sealed Vesicles from Red Beet (Beta vulgaris L.) Storage Tissue : II. Characterization of Ca Uptake into Plasma Membrane Vesicles. Plant Physiol. 1987 Dec;85(4):1137–1142. doi: 10.1104/pp.85.4.1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gimble J. M., Gustin M., Goodman D. B., Rasmussen H. Studies on the Ca2+ transport mechanism of human erythrocyte inside-out plasma membrane vesicles. V. Chlortetracycline fluorescence. Biochim Biophys Acta. 1982 Mar 8;685(3):253–259. doi: 10.1016/0005-2736(82)90065-7. [DOI] [PubMed] [Google Scholar]
- Gross J., Marmé D. ATP-dependent Ca uptake into plant membrane vesicles. Proc Natl Acad Sci U S A. 1978 Mar;75(3):1232–1236. doi: 10.1073/pnas.75.3.1232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasselbach W., Waas W. Energy coupling in sarcoplasmic reticulum Ca2+ transport: an overview. Ann N Y Acad Sci. 1982;402:459–469. doi: 10.1111/j.1749-6632.1982.tb25768.x. [DOI] [PubMed] [Google Scholar]
- Hodges T. K., Leonard R. T. Purification of a plasma membrane-bound adenosine triphosphatase from plant roots. Methods Enzymol. 1974;32:392–406. doi: 10.1016/0076-6879(74)32039-3. [DOI] [PubMed] [Google Scholar]
- Lew R. R., Briskin D. P., Wyse R. E. Ca uptake by endoplasmic reticulum from zucchini hypocotyls : the use of chlorotetracycline as a probe for ca uptake. Plant Physiol. 1986 Sep;82(1):47–53. doi: 10.1104/pp.82.1.47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohnishi T., Gall R. S., Mayer M. L. An improved assay of inorganic phosphate in the presence of extralabile phosphate compounds: application to the ATPase assay in the presence of phosphocreatine. Anal Biochem. 1975 Nov;69(1):261–267. doi: 10.1016/0003-2697(75)90585-0. [DOI] [PubMed] [Google Scholar]
- Poole R. J., Briskin D. P., Krátký Z., Johnstone R. M. Density gradient localization of plasma membrane and tonoplast from storage tissue of growing and dormant red beet : characterization of proton-transport and ATPase in tonoplast vesicles. Plant Physiol. 1984 Mar;74(3):549–556. doi: 10.1104/pp.74.3.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pressman B. C. Biological applications of ionophores. Annu Rev Biochem. 1976;45:501–530. doi: 10.1146/annurev.bi.45.070176.002441. [DOI] [PubMed] [Google Scholar]
- Rincon M., Boss W. F. myo-Inositol Trisphosphate Mobilizes Calcium from Fusogenic Carrot (Daucus carota L.) Protoplasts. Plant Physiol. 1987 Feb;83(2):395–398. doi: 10.1104/pp.83.2.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schumaker K. S., Sze H. A Ca/H Antiport System Driven by the Proton Electrochemical Gradient of a Tonoplast H-ATPase from Oat Roots. Plant Physiol. 1985 Dec;79(4):1111–1117. doi: 10.1104/pp.79.4.1111. [DOI] [PMC free article] [PubMed] [Google Scholar]