Abstract
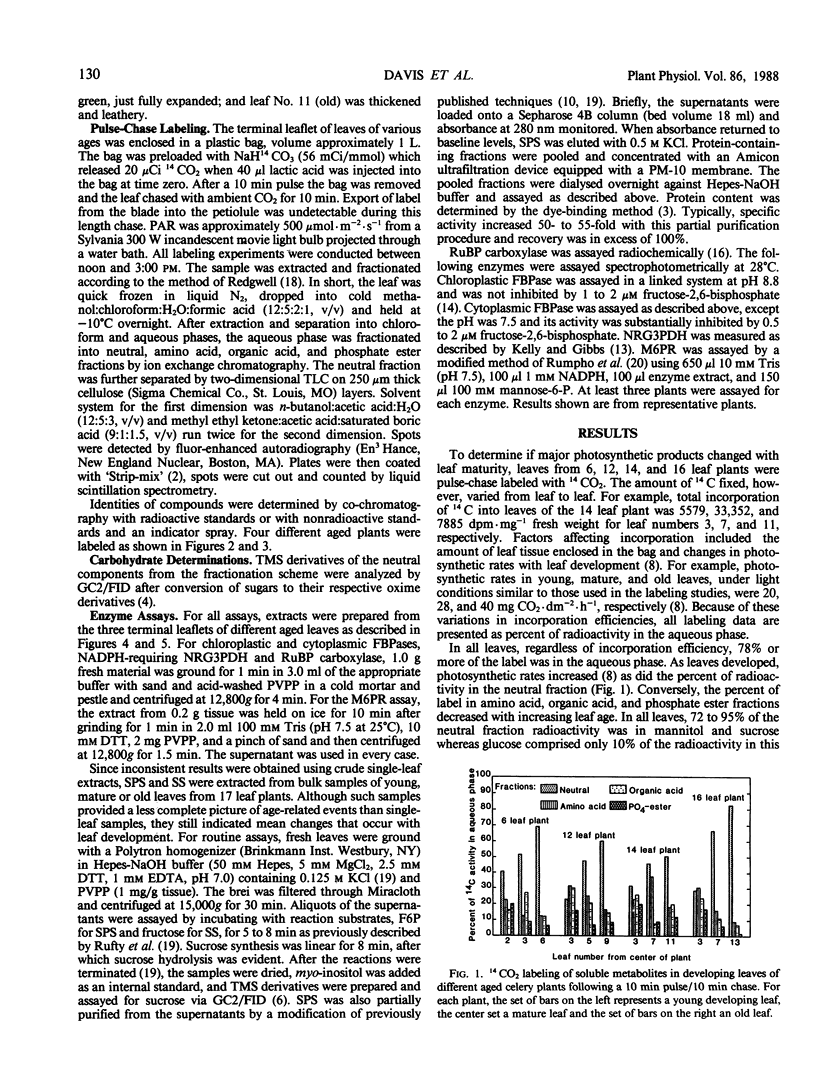
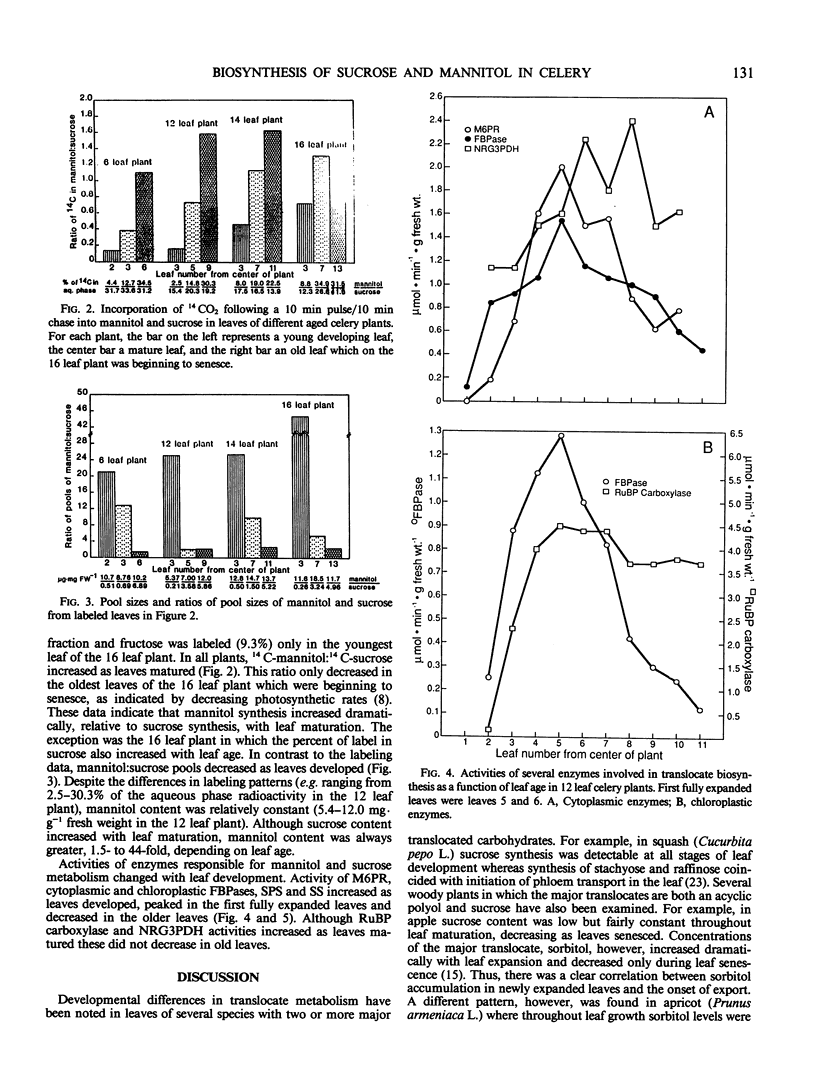
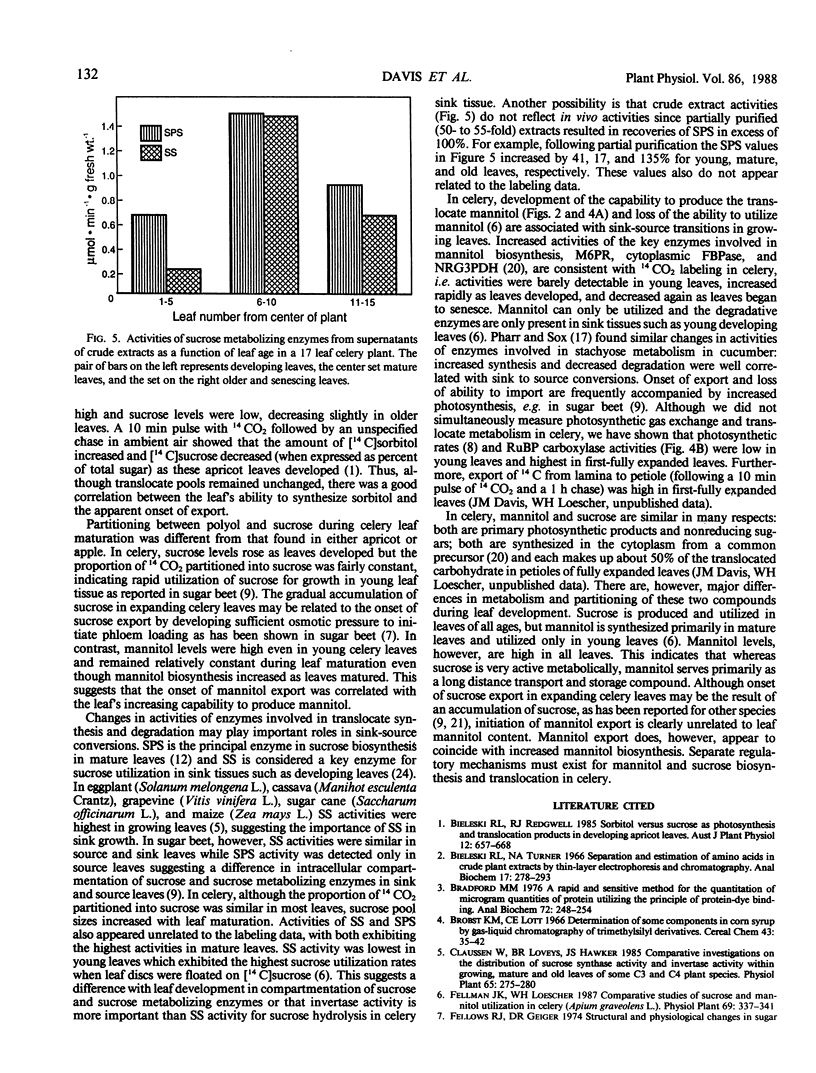
In celery (Apium graveolens L.), the two major translocated carbohydrates are sucrose and the acyclic polyol mannitol. Their metabolism, however, is different and their specific functions are uncertain. To compare their roles in carbon partitioning and sink-source transitions, developmental changes in 14CO2 labeling, pool sizes, and key enzyme activities in leaf tissues were examined. The proportion of label in mannitol increased dramatically with leaf maturation whereas that in sucrose remained fairly constant. Mannitol content, however, was high in all leaves and sucrose content increased as leaves developed. Activities of mannose-6-P reductase, cytoplasmic and chloroplastic fructose-1,6-bisphosphatases, sucrose phosphate synthase, and sucrose synthase increased with leaf maturation and decreased as leaves senesced. Ribulose bisphosphate carboxylase and nonreversible glyceraldehyde-3-P dehydrogenase activities rose as leaves developed but did not decrease. Thus, sucrose is produced in all photosynthetically active leaves whereas mannitol is synthesized primarily in mature leaves and stored in all leaves. Onset of sucrose export in celery may result from sucrose accumulation in expanding leaves, but mannitol export is clearly unrelated to mannitol concentration. Mannitol export, however, appears to coincide with increased mannitol biosynthesis. Although mannitol and sucrose arise from a common precursor in celery, subsequent metabolism and transport must be regulated separately.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bieleski R. L., Turner N. A. Separation and estimation of amino acids in crude plant extracts by thin-layer electrophoresis and chromatography. Anal Biochem. 1966 Nov;17(2):278–293. doi: 10.1016/0003-2697(66)90206-5. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Fellows R. J., Geiger D. R. Structural and Physiological Changes in Sugar Beet Leaves during Sink to Source Conversion. Plant Physiol. 1974 Dec;54(6):877–885. doi: 10.1104/pp.54.6.877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox T. C., Kennedy R. A., Loescher W. H. Developmental Changes in Photosynthetic Gas Exchange in the Polyol-Synthesizing Species, Apium graveolens L. (Celery). Plant Physiol. 1986 Sep;82(1):307–311. doi: 10.1104/pp.82.1.307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giaquinta R. Source and sink leaf metabolism in relation to Phloem translocation: carbon partitioning and enzymology. Plant Physiol. 1978 Mar;61(3):380–385. doi: 10.1104/pp.61.3.380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harbron S., Foyer C., Walker D. The purification and properties of sucrose-phosphate synthetase from spinach leaves: the involvement of this enzyme and fructose bisphosphatase in the regulation of sucrose biosynthesis. Arch Biochem Biophys. 1981 Nov;212(1):237–246. doi: 10.1016/0003-9861(81)90363-5. [DOI] [PubMed] [Google Scholar]
- Kelly G. J., Gibbs M. Nonreversible d-Glyceraldehyde 3-Phosphate Dehydrogenase of Plant Tissues. Plant Physiol. 1973 Aug;52(2):111–118. doi: 10.1104/pp.52.2.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loescher W. H., Marlow G. C., Kennedy R. A. Sorbitol metabolism and sink-source interconversions in developing apple leaves. Plant Physiol. 1982 Aug;70(2):335–339. doi: 10.1104/pp.70.2.335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McFadden B. A., Denend A. R. Ribulose diphosphate carboxylase from autotrophic microorganisms. J Bacteriol. 1972 May;110(2):633–642. doi: 10.1128/jb.110.2.633-642.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Redgwell R. J. Fractionation of plant extracts using ion-exchange Sephadex. Anal Biochem. 1980 Sep 1;107(1):44–50. doi: 10.1016/0003-2697(80)90489-3. [DOI] [PubMed] [Google Scholar]
- Rufty T. W., Kerr P. S., Huber S. C. Characterization of diurnal changes in activities of enzymes involved in sucrose biosynthesis. Plant Physiol. 1983 Oct;73(2):428–433. doi: 10.1104/pp.73.2.428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rumpho M. E., Edwards G. E., Loescher W. H. A pathway for photosynthetic carbon flow to mannitol in celery leaves : activity and localization of key enzymes. Plant Physiol. 1983 Dec;73(4):869–873. doi: 10.1104/pp.73.4.869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silvius J. E., Kremer D. F., Lee D. R. Carbon assimilation and translocation in soybean leaves at different stages of development. Plant Physiol. 1978 Jul;62(1):54–58. doi: 10.1104/pp.62.1.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turgeon R. Termination of nutrient import and development of vein loading capacity in albino tobacco leaves. Plant Physiol. 1984 Sep;76(1):45–48. doi: 10.1104/pp.76.1.45. [DOI] [PMC free article] [PubMed] [Google Scholar]


