Abstract
Ionically bound cell wall proteins were extracted from 7 day old etiolated pea (Pisum sativum L. cv Alaska) epicotyls with 3 molar LiCl. Polyclonal antiserum was raised in rabbits against the cell wall proteins. Growth assays showed that treatment of growing region segments (5-7 millimeters) of peas with either dialyzed serum, serum globulin fraction, affinity purified immunoglobulin, or papain-cleaved antibody fragments had no effect on growth. Immunofluorescence microscopy confirmed antibody binding to cell walls and penetration of the antibodies into the tissues. Western blot analysis, immunoassay results, and affinity chromatography utilizing Sepharose-bound antibodies confirmed recognition of the protein preparation by the antibodies. Experiments employing in vitro extension as a screening measure indicated no effect upon extension by antibodies, by 50 millimolar LiCl perfusion of the apoplast or by 3 molar LiCl extraction. Addition of cell wall protein to protease pretreated segments did not restore extension nor did addition of cell wall protein to untreated segments increase extension. It is concluded that, although evidence suggests that protein is responsible for the process of extension, the class(es) of proteins which are extracted from pea cell walls with 3 molar LiCl are probably not involved in this process.
Full text
PDF

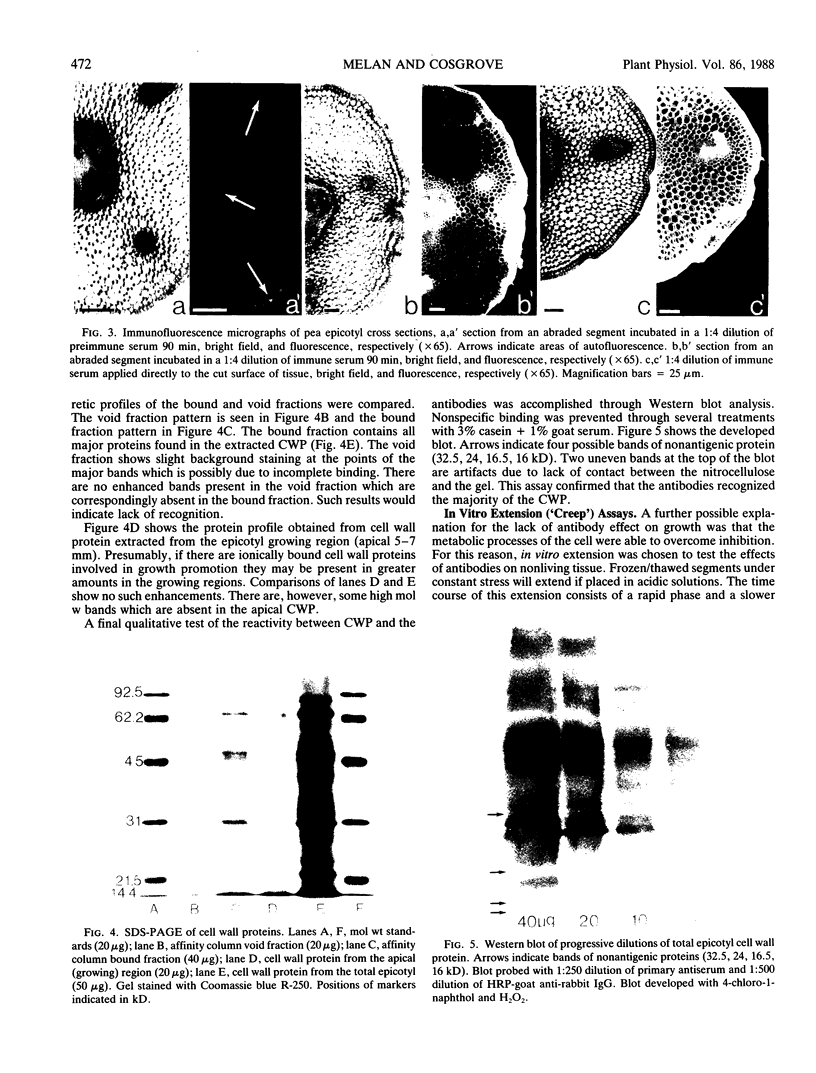

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Cais M. Metalloimmunoassay: principles and practice. Methods Enzymol. 1983;92:445–458. doi: 10.1016/0076-6879(83)92038-4. [DOI] [PubMed] [Google Scholar]
- Carpita N., Sabularse D., Montezinos D., Delmer D. P. Determination of the pore size of cell walls of living plant cells. Science. 1979 Sep 14;205(4411):1144–1147. doi: 10.1126/science.205.4411.1144. [DOI] [PubMed] [Google Scholar]
- Chard T. Ammonium sulfate and polyethylene glycol as reagents to separate antigen from antigen-antibody complexes. Methods Enzymol. 1980;70(A):280–291. doi: 10.1016/s0076-6879(80)70057-5. [DOI] [PubMed] [Google Scholar]
- Cleland R. E., Cosgrove D., Tepfer M. Long-term acid-induced wall extension in an in-vitro system. Planta. 1987;170:379–385. [PubMed] [Google Scholar]
- Cosgrove D. J., Cleland R. E. Solutes in the free space of growing stem tissues. Plant Physiol. 1983 Jun;72(2):326–331. doi: 10.1104/pp.72.2.326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cosgrove D. J. Wall relaxation in growing stems: comparison of four species and assessment of measurement techniques. Planta. 1987;171:266–278. [PubMed] [Google Scholar]
- Hawkes R., Niday E., Gordon J. A dot-immunobinding assay for monoclonal and other antibodies. Anal Biochem. 1982 Jan 1;119(1):142–147. doi: 10.1016/0003-2697(82)90677-7. [DOI] [PubMed] [Google Scholar]
- Hayashi T., Wong Y. S., Maclachlan G. Pea Xyloglucan and Cellulose : II. Hydrolysis by Pea Endo-1,4-beta-Glucanases. Plant Physiol. 1984 Jul;75(3):605–610. doi: 10.1104/pp.75.3.605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huber R., Deisenhofer J., Colman P. M., Matsushima M., Palm W. Crystallographic structure studies of an IgG molecule and an Fc fragment. Nature. 1976 Dec 2;264(5585):415–420. doi: 10.1038/264415a0. [DOI] [PubMed] [Google Scholar]
- Jacobs M., Gilbert S. F. Basal localization of the presumptive auxin transport carrier in pea stem cells. Science. 1983 Jun 17;220(4603):1297–1300. doi: 10.1126/science.220.4603.1297. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Löbler M., Klämbt D. Auxin-binding protein from coleoptile membranes of corn (Zea mays L.). II. Localization of a putative auxin receptor. J Biol Chem. 1985 Aug 15;260(17):9854–9859. [PubMed] [Google Scholar]
- Midgley A. R., Jr, Hepburn M. R. Use of the double-antibody method to separate antibody bound from free ligand in radioimmunoassay. Methods Enzymol. 1980;70(A):266–274. doi: 10.1016/s0076-6879(80)70055-1. [DOI] [PubMed] [Google Scholar]
- PORTER R. R. The hydrolysis of rabbit y-globulin and antibodies with crystalline papain. Biochem J. 1959 Sep;73:119–126. doi: 10.1042/bj0730119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson G. L. A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal Biochem. 1977 Dec;83(2):346–356. doi: 10.1016/0003-2697(77)90043-4. [DOI] [PubMed] [Google Scholar]
- Poljak R. J., Amzel L. M., Avey H. P., Becka L. N. Structure of Fab' New at 6 A resolution. Nat New Biol. 1972 Feb 2;235(57):137–140. doi: 10.1038/newbio235137a0. [DOI] [PubMed] [Google Scholar]
- Tepfer M., Taylor I. E. The permeability of plant cell walls as measured by gel filtration chromatography. Science. 1981 Aug 14;213(4509):761–763. doi: 10.1126/science.213.4509.761. [DOI] [PubMed] [Google Scholar]
- Vaitukaitis J. L. Production of antisera with small doses of immunogen: multiple intradermal injections. Methods Enzymol. 1981;73(Pt B):46–52. doi: 10.1016/0076-6879(81)73055-6. [DOI] [PubMed] [Google Scholar]