Abstract
Antibody‒drug conjugates (ADCs), which combine the advantages of monoclonal antibodies with precise targeting and payloads with efficient killing, show great clinical therapeutic value. The ADCs’ payloads play a key role in determining the efficacy of ADC drugs and thus have attracted great attention in the field. An ideal ADC payload should possess sufficient toxicity, low immunogenicity, high stability, and modifiable functional groups. Common ADC payloads include tubulin inhibitors and DNA damaging agents, with tubulin inhibitors accounting for more than half of the ADC drugs in clinical development. However, due to clinical limitations of traditional ADC payloads, such as inadequate efficacy and the development of acquired drug resistance, novel highly efficient payloads with diverse targets and reduced side effects are being developed. This perspective summarizes the recent research advances of traditional and novel ADC payloads with main focuses on the structure–activity relationship studies, co-crystal structures, and designing strategies, and further discusses the future research directions of ADC payloads. This review also aims to provide valuable references and future directions for the development of novel ADC payloads that will have high efficacy, low toxicity, adequate stability, and abilities to overcome drug resistance.
Key words: Antibody‒drug conjugates, Dual payloads, Tubulin inhibitors, DNA damaging agents, PROTACs, RNA targeting agents
Graphical abstract
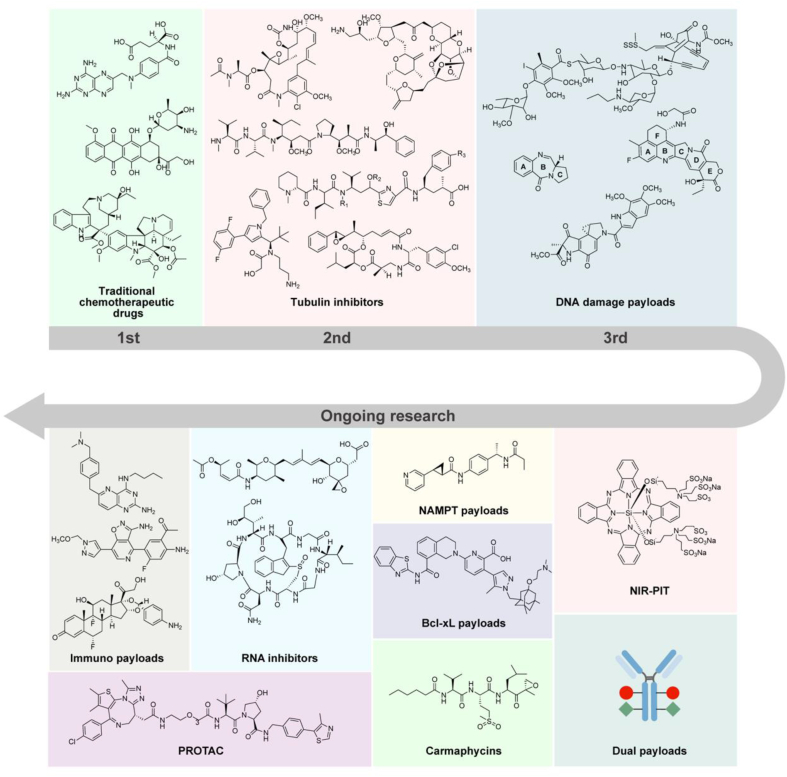
The evolution of antibody–drug conjugate (ADC) payloads. From traditional payloads that are currently used in clinics to the ongoing development efforts to incorporate novel payloads with improved therapeutic indexes.
1. Introduction
Despite the recent advancements in immunotherapy and cell therapies, chemotherapy remains to be the most used strategy in cancer treatment. However, due to their low therapeutic index, conventional chemotherapeutics, despite showing potent cytotoxicity against cancer cells, often show toxic effects on healthy tissues, which significantly limits their clinical efficacy1,2. Thus, developing drug delivery systems with high efficiency and limited systemic toxicity for the treatment of cancers may be an effective strategy to solve the problem3. Hence, a novel concept, antibody‒drug conjugates (ADCs) has been conceived and developed (Fig. 1). Typically, an ADC is formed by an antibody targeting a tumor-specific antigen or a related antigen and several payloads through appropriate linkers. An ADC combines the high targeting capabilities of a monoclonal antibody (mAb) and the high potency of the payloads in tumor tissues. ADCs have become one of the fastest-developing drug classes in oncology in recent years, because of their lower side effects, broader treatment applications, and higher therapeutic index4,5.
Figure 1.
Key structures and mechanisms of action of ADCs. (A) The general mechanism of action of ADCs; (B) The mechanism of DNA inhibitors as ADC payloads; (C) The mechanism of Splicing inhibitors as ADC payloads; (D) The mechanism of tubulin inhibitors as ADC payloads; (E) The mechanism of PROTAC molecules as ADC payloads; (F) The mechanism of Bcl-xL inhibitors and proteasome inhibitors as ADC payloads; (G) The mechanism of NAMPT inhibitors as ADC payloads; (H) The mechanism of NIR-PIT ADC.
The concept of ADC drugs was first proposed by German Nobel Laureate Paul Ehrlich in 19136. But not until 1975, when the development of hybridoma technology started being used to produce monoclonal antibodies really began the era of developing ADC drugs. ADC drugs have undergone three generations of innovation, driven by increasingly mature technologies (Fig. 2). In the first generation of ADC drugs, traditional chemotherapeutics such as methotrexate, vinblastine, and doxorubicin were used as cytotoxic payloads7. However, because of their insufficient cytotoxicity against cancer cells, lack of tumor selectivity, and low accumulation in target cells, these first-generation ADCs showed even poorer efficacy than their parental payloads, which resulted in their clinical fails8, 9, 10, 11. Subsequently, novel and highly cytotoxic compounds that showed 100 to 1000 times more potent than the traditional chemotherapeutics used in the first-generation ADCs attracted strong interest. They often have unbearable side effects when used as a single drug for killing tumors. For example, tubulin inhibitors maytansine showed extremely powerful antiproliferative activity against tumor cells. However, its toxic side effects, such as neurotoxicity and gastrointestinal reactions, were also severe. Hence, it was not approved for cancer therapy as a single agent12. Interestingly, these highly cytotoxic compounds are ideal payloads for ADCs. The microtubules, composed of α- and β-tubulin, are the main components of cytoskeleton. Tubulin inhibitors can kill tumor cells by disrupting their cytoskeleton structures and interfering with mitosis. Thus, tumor cells, which divide faster, are more sensitive to tubulin inhibitors compared to normal cells13. Most of the second-generation ADCs used significantly more potent tubulin inhibitors as payloads. Unfortunately, while tubulin inhibitors are very effective against actively dividing tumor cells, they are much less effective against static cancer cells14. To potentially overcome this limitation, DNA damaging agents, which can target the whole cell cycle, were selected as the cytotoxic payloads for most of the third-generation ADCs. DNA damaging agents can destroy DNA structures to kill tumor cells through double-strand breaking, alkylation, chimerism, and cross-linking. Representative DNA damaging payloads include enediyne, topoisomerase I inhibitors, and pyrrolo benzodiazepines (PBD)15. Currently, 15 ADC drugs have been approved, and a large number of ADC drugs are undergoing clinical trials (Table 1)16. Their payloads are mainly of natural origin, and tubulin inhibitors account for more than half of them.
Figure 2.
Milestones in the development of ADCs payloads.
Table 1.
ADC drugs approved by FDA (up to March 2023).
| No. | ADC name | Brand name | Company | Target | Payload | Indication | Launch year |
|---|---|---|---|---|---|---|---|
| 1 | Gemtuzumab ozogamicin (GO) | Mylotarg | Pfizer/Wyeth | CD33 | Calicheamicin | Acute myeloid leukemia | 2000.05 Relaunched in 2017.09 |
| 2 | Brentuximab vedotin (SGN-35) | Adcetris | Seattle Genetics Millennium/Takeda | CD30 | MMAE | Hodgkin lymphoma, large cell lymphoma | 2011.08 |
| 3 | Trastuzumab emtansine (T-DM1) | Kadcyla | Genentech/Roche | HER2 | DM1 | HER2+ breast cancer | 2013.02 |
| 4 | Inotuzumab ozogamicin | Besponsa | Pfizer/Wyeth | CD22 | Calicheamicin | Acute lymphoblastic leukemia | 2017.08 |
| 5 | Moxetumomab pasudotox | Lumoxiti | AstraZeneca | CD22 | Pseudomonas exotoxin | Relapsed or refractory hairy cell leukemia | 2018.09 |
| 6 | Polatuzumab vedotin | Polivy | Genentech/Roche | CD79b | MMAE | Diffuse large B-cell lymphoma | 2019.07 |
| 7 | Enfortumab vedotin | Padcev | Astellas/Seattle Genetics | Nectin-4 | MMAE | Advanced or metastatic urothelial cancer | 2019.12 |
| 8 | Fam-trastuzumab deruxtecan (DS-8201) | Enhertu | AstraZeneca/Daiichi Sankyo | HER2 | Dxd | Metastatic breast cancer | 2019.12 |
| 9 | Sacituzumab govitecan | Trodelvy | Immunomedics | Trop-2 | SN-38 | Triple-negative breast cancer | 2020.04 |
| 10 | Belantamab mafodotin | Blenrep | GlaxoSmithKline | BCMA | MMAF | Relapsed or refractory multiple myeloma | 2020.08 |
| 11 | Cetuximab saratolacan | Akalux | RakutenMedical | EGFR | IRDye700DX | Head and neck cancer | 2020.09 |
| 12 | Loncastuximab tesirine-lpyl | Zynlonta | ADC Therapeutics | CD19 | SG3199 | Large B-cell lymphoma | 2021.04 |
| 13 | Disitamab vedotin | Aidixi | RemeGen | HER2 | MMAE | HER2+ gastric carcinoma | 2021.06 |
| 14 | Tisotumab vedotin-tftv | Tivdak | Seagen | Tissue factor | MMAE | Cervical cancer | 2021.09 |
| 15 | Mirvetuximab soravtansine-gyxn | ELAHERE™ | ImmunoGen, Inc. | Fra | DM4 | Ovarian cancer | 2022.11 |
Although ADCs have gone through three generations, current payloads still have clinical limitations such as severe side effects and the development of drug resistance. There is still a strong unmet medical need to develop more potent ADC payloads, ideally with better therapeutic indexes17. Accordingly, additional novel ADC payloads, including RNA inhibitors, Bcl-xL inhibitors, NAMPT inhibitors, and carmaphycins18, 19, 20, are being designed. In addition, immune ADCs using immunomodulators as payloads have attracted significant attention because of their key roles in tumor immunotherapy21. Instead of using single simple molecules as their payloads, several novel strategies for designing ADCs using more complex payloads have emerged. For example, PROTACs or photosensitizers have been used as ADC payloads, and methods to incorporate several payloads, each with a different target, into a single antibody have also been developed22, 23, 24. These groundbreaking strategies can lead to the next generation of ADCs.
Based on the importance and main characteristics of the ADC payloads, this review summarizes the discovery and main structural modifications of various traditional ADC payloads from the perspective of their crystal structures and the related structure–activity relationships (SAR) between ADC payloads and their targets. In addition, a variety of novel ADC payloads based on new drug targets and innovative design strategies will also be summarized in this review. Finally, in view of the limitations of the current payloads, the main research directions for developing future ADC payloads are discussed, with the hope to provide a reference for the future design of novel ADC payloads with ideal characteristics.
2. Importance and characteristics of the ADC payload
After an ADC drug enters the blood circulation and binds to the target antigen receptor on the surface of the tumor cell, the newly formed ADC‒antigen complex will be degraded by lysosomes after internalization, releasing the payload and inducing the tumor cell death (Fig. 1). Therefore, payloads are an important part of the ADC design25. The activity and physicochemical properties of the payload have a direct impact on the antitumor efficacy of ADC drugs. The mechanism of action of the payload is an important factor determining the performance of the ADC (e.g., adverse reactions). Besides, certain other characteristics of ADC payloads, such as cytotoxicity, immunogenicity, stability of storage during preparation and circulation, water solubility, and modifiability are also important26.
The ideal payloads should have the following characteristics. First, they should have adequately high cytotoxicity. Tumor-specific antigens are very limited, especially in solid tumors. Moreover, due to the low permeability and poor internalization activity of monoclonal antibodies, the number of ADC payloads that can be endocytosed into tumor cells via antibody antigen binding is very low27. Second, ADC payloads should have sufficiently low immunogenicity. Protein drugs have the risk to induce immunogenicity, which may negatively affect the ADC efficacy or even lead to mortality of the treated patients. Although ADCs currently use human or humanized monoclonal antibodies and small molecule payloads, they still may increase the risk of immunogenicity compared with therapeutic monoclonal antibodies. To address this issue, some highly toxic payloads are extracted from plants, animals or microorganisms, ensuring the immunogenicity of the payload in humans is small enough to be negligible. Using smaller molecular payloads is also a way to reduce the risk of immunogenicity. Third, ADC payloads should have high stability. Because antibodies have a long half-life in circulation, ADCs should remain stable in the blood circulation to avoid release or decomposition. The payload should also remain stable in the cytoplasm and lysosome without significant degradations under low pH conditions3. Fourth, ADC payloads should have functional groups that can be modified without significantly affecting their potency. The payload must have a modifiable functional group or a site that can conjugate to the monoclonal antibody. The site of the modification must be carefully selected to preserve the potency of the parental drug. More importantly, when using non-cleavable linkers, the payload must retain its potency even after the antibody degrades28. Fifth, the ADC payloads should have bystander killing effects. Some ADC drugs are internalized and release small, uncharged, permeable membrane hydrophobic molecules, which spread through the cell membrane and kill tumor cells with negative expression of adjacent antigens. This process is known as the “bystander killing effect” and has important implications on tumor cells with uneven antigen-expression29. In general, payloads that promote bystander killing effects in cancer cells are better suited for cancers with a low or heterogeneous expression of target antigens. To construct ADC drugs with the bystander effect, their structures must meet the conditions that the linker can be cleaved, and the payload is permeable to the cell membranes. Hydrophobic molecules with good membrane penetration have a strong bystander killing effect, but this may also make the drugs easily absorbed by healthy tissues, resulting in serious systemic toxicity. Therefore, the balance between the two is very important for the development of ADCs. Sixth, ADC payloads should have proper water solubility. Payloads must be appropriately water-soluble to facilitate conjugation with the antibody and ensure sufficient solubility of the conjugate under physiological conditions. When excessive hydrophobic payloads conjugate to an antibody, the resulting ADC tends to aggregate and becomes unstable. In addition, the hydrophilicity of the payload affects the cell membrane permeability of the parental ADC or its metabolite, and thus its bystander killing activity. Finally, the payloads’ target should be intracellular as most ADCs need to enter tumor cells to release their payloads. Many potent payloads from microorganisms, plants, and animals that have membrane targeting, such as those on neurons that work primarily by blocking ion channels or disrupting clotting, are not suitable for use as ADC payloads30.
Among the above seven characteristics of the ADC payload, high potency, low immunogenicity, stability in drug preparation and blood circulation, and modifiable functional groups are the important factors that must be considered in the selection of the ADC payload. Other characteristics including proper water solubility or bystander killing effect, though generally less essential, still have significant impacts on the effectiveness of an ADC. Based on these characteristics, we summarized the effectiveness of existing ADC payloads, including payloads targeting tubulin/DNA/RNA, immune ADC payloads, novel potential ADC payloads, and the payloads used in the design of ADC drugs under the guidance of various new strategies. By analyzing the binding mode and the related SAR between various payloads and proteins, we hope to clarify how the structures determine their biological activity and characteristics and provide a valuable reference for the generation of more new ADC payloads in the future.
3. The various payloads of ADC
3.1. Microtubules targeting payloads
Microtubules are an important part of the cytoskeleton in eukaryotic cells. They are dynamic polar polymers composed of α,β tubulin heterodimers parallel to the cylindrical axis. Microtubules play an important role in maintaining various cellular functions such as cell morphology, signal transduction, organelle transportation, cell motility, cell division, and mitosis, and are important targets for tumor therapy. Tubulin is the composition of microtubules, and tubulin inhibitors interfere with the dynamic combination of microtubules by binding to tubulin, arresting cells in the G2/M phase of the cell cycle, and ultimately leading to apoptosis31. Because tubulin inhibitors destroy the mitotic spindle and play an anti-mitosis role, they have stronger toxicity against rapidly dividing cancer cells than most slow-growing normal cells, making them popular ADC payloads. Among them, the tubulin polymerization enhancers, such as auristatin compounds MMAE and MMAF, work on the β-subunit of the α,β tubulin dimer, so that the growth of the microtubules is not regulated. In contrast, tubulin polymerization inhibitors, such as maytansine compounds DM1 and DM4, block the polymerization of tubulin dimers by inhibiting the formation of mature microtubules. These tubulin inhibitors are the most common second-generation payloads used in many clinical ADC drugs and have been structurally modified by multiple teams as lead compounds to further improve the physicochemical properties of such ADC payloads, making them more suitable for the development of ADC drugs32.
3.1.1. Maytansinoids
Maytansine (1) (Fig. 3A) was originally isolated from the bark of the African shrub Maytenus ovatus and is a class of benzoansamacrolide antibiotics that bind to tubulin and inhibit the assembly of microtubules. In the in vitro cell activity assay, its IC50 falls into the picomolar range, demonstrating its strong ability to inhibit tumor cell proliferation. It also shows good stability and solubility in other experiments. However, due to its narrow treatment window and toxic side effects such as neurotoxicity and gastrointestinal reactions due to the lack of selectivity, it has been clinically prohibited from direct use in human treatment12. However, the high cytotoxicity of maytansine perfectly meets the requirements of ADC payloads, making maytansine a strong candidate as an ADC payload. Since maytansine does not have a reactive functional group, it cannot bind to the antibodies. Structure−activity studies with maytansinoids had identified the C3 N-acyl-N-methyl-l-alanyl ester side chain, the C4–C5 epoxide moiety, the C9 carbinol function, and the position of the conjugated C11 and C13 double bonds as critical elements for activity33. This left the phenyl ring and the N-acyl group as chemically modifiable entities. Replacing the N-acetyl group in maytansine with a 3-methyldithiopropionyl group, maytansine derivative DM1 (2) (Fig. 3A) containing disulfide can be obtained. And DM4 (3) is obtained by adding two methyl groups around the DM1 disulfide bond. Derived DM1 and DM4, with methylthiopropionyl group, can be conjugated to the linker by disulfide bonds. DM1 and DM4 are the most commonly used two maytansine-like ADC payloads in clinical practical applications, such as Trastuzumab-SMCC-DM1, the first ADC drug based on maytansine derivatives approved for marketing (ClinicalTrials.gov identifier: NCT04158947, NCT01983501, NCT02562378, etc.)34. Currently, 20% of the ADCs in development use maytansine derivatives as a payload.
Figure 3.
Design and SAR analysis of maytansinoids. (A) Chemical structures of maytansinoids 1–5; (B) Binding mode of maytansine (1) in complex with tubulin (PDB code 4TV8); (C) Binding mode of compound 5 in complex with tubulin (PDB code 5SBA).
In 2014, Michel O. Steinmetz's team35 studied maytansine and its role with microtubules and found that on β-tubulin, the maytansine class has three key interaction points with tubulin (Fig. 3B), which include hydrogen bonds between the carbonyl groups at positions 1 of maytansine, and residues Asn102 and Lys105 of β-tubulin; hydrogen bonds between the hydroxyl/carbonyl oxygens at positions 1 and Val181 of β-tubulin; and hydrophobic interactions between the methyl groups at positions 6a of the ligands and a pocket shaped by residues Asn101, Asn102, Val182, Phe404, and Tyr408 of β-tubulin. These conserved tubulin interaction points constitute a common pharmacophore. Maytansine is the most commonly used ADC payload, but its synthesis, isolation and purification of intermediate diastereomer products are difficult. At the same time, it is a substrate of multidrug-resistant protein 1 (MDR1), a transporter that reduces the potency of some antibody-maytansine-like conjugates. So Jinliang Yang's team36 explored the crystal structure of L-DM1-SMe (C3-position methyl-l configuration DM1), D-DM1-SMe (C3-position methyl-d configuration DM1) and β tubulin, and found that the carbonyl oxygen atoms in the L-DM1-SMe ester group and the sulfur methyl at the tail of the C3 side chain form a strong intramolecular interaction with the hydroxyl at position 9 and benzene rings, respectively, which contributes to the enhancement of binding affinity. The C3 side chain of D-DM1-SMe swings in the opposite direction, unable to form intramolecular interactions, explaining the detailed mechanism by which the chirality of methyl groups at the C3 position affects anti-cancer activity. The study provides a basis for the design of the next generation of maytansine binding site inhibitors with novel scaffolds to improve synthetic strategies and reduce MDR1-mediated resistance.
Maytansinol (4) (Fig. 3A) was first obtained by chemically removing the acyl group from the hydroxyl group at the C3 position of an extract from Putterlickia verrucose. It showed weaker inhibitory activity on tubulin polymerization than maytansine, implying that the ester moiety at the C3 position of maytansine and maytansinoids plays an important role in biological activity and cell permeability. The carbonyl oxygen atom of the ester moiety forms a strong intramolecular interaction with the hydroxy group at the 9 position, fixing the bioactive conformation. Maytansinol has been regarded as a valuable lead compound. A variety of different natural and semisynthetic maytansinoids with various ester side chains can be easily prepared by the acylation of the hydroxyl group at C3 position37,38. Daniele Passarella's group39 obtained a new maytansine-like compound 5 through the acylation of maytansinol (4), which has a strong ability to inhibit microtubule assembly, with an IC50 of 0.07–1.2 nmol/L on A549 cells. The crystal structure (Fig. 3C) shows that the binding of the maytansinoids has no effect on the overall conformation of tubulin, the compounds form hydrogen bonds between the C1–O and the main chain nitrogen atom of Val181 and between the C24–O and the side chains of Lys105 and Asn102. Additionally, compound 5 establishes a hydrogen bond to the main chain carbonyl group of Gly100 via their C9–OH group. In the studied maytansinoids, all the modifications introduced at the C3 position point towards the solvent and do not interfere with the close environment of the maytansine site. Thus, acylation enhances the full biological activity of maytansine, and the modifications introduced at the C3 positions do not interfere with the binding state, affinity, or effectiveness of the compound to tubulin dimers. Regeneron's team40 studied the effects of nitrogen substitution in N-methylalanine by changing the length of the upper side chains of the ring and the linkers connected by primary amine and secondary amines. EGFRvIII targeted ADC was obtained using compound 6 as payload, with IC50 of 0.3–0.4 nmol/L for HEK293, U251, and MMT cell lines, and tumor regression was achieved in U251/EGFRvIII and MMT/EGFRvIII mice models.
3.1.2. Auristatin
Dolastatin 10 (7), isolated from Dolabella auricularia in 1987, has strong anti-proliferative activity against a variety of cancer cells and can strongly inhibit microtubule assembly, leading to cell cycle arrest and apoptosis, making it a promising anti-cancer drug41 (Fig. 4-1A). Its water-soluble synthetic analogs are called auristatin. Among them, the most widely used are monomethyl auristatin E (MMAE) (8) and monomethyl auristatin F (MMAF) (9). Each of them contains a functional handle capable of the subsequent conjugation, further improving the efficacy in vivo42. MMAE comprises four amino acids: monomethylvaline (MeVal), valine (Val), dolaisoleuine (Dil) and dolaproine (Dap), and the carboxy-terminal amine norephedrine. In MMAF, the C-terminus of monomethylvaline is replaced by phenylalanine, and its cellular activity is significantly reduced. Andrea E. Prota's team43 found that the binding affinity of MMAF to free tubulin increased nearly 5 times compared to MMAE, mainly due to the critical Arg278 residue on the β1 tubulin subunit, which is exposed to MMAF through ordered water molecules. They also showed that detailed structural interactions that extend the vinca domain to the peptide site are not only functionally distinct by inhibiting nucleotide exchange, but also indicate how peptide site antimitotics attain increased potency over the vinca alkaloids. The analysis may provide a structural explanation of why the cellular activity of MMAF is reduced.
Figure 4-1.
Design and SAR analysis of auristatin analogs. (A) Chemical structures of auristatin analogs 7–10, 21–24; (B) Binding mode of compound 10 in complex with tubulin (PDB code 4X1I).
Jinliang Yang's team44 defined an essential pharmacodynamic group through a structural study of the binding domain of MMAE and tubulin in the vinca domain. The pharmacophore consists of two hydrophobic regions I and II and two hydrogen bonding regions that interact with Asp179β and Asn329α, laying the foundation for the rational design of ADC payloads with high specificity, strong affinity, and high efficiency. In 2020, Christopher J. O Donnell's45 team designed and synthesized an auristatin analog 10 with N-terminal modifications (IC50 = 0.2 nmol/L) and for the first time achieved a detailed study of its binding patterns by having a higher resolution auristatin crystal structure. The crystal structure shows that the N-terminal dimethyl of compound 10 fits well in the receptor pocket and does not affect the overall binding affinity of the molecule (Fig. 4-1B). In addition, key polar interactions were identified, including the hydrogen bond network between terminal protonated amino groups and Asp β197 and Phe α351 aminocarbonyl groups located on the T5 ring, bifocal interaction between N-2 valine and Asn α329, and critical hydrogen bond interaction between Tyr β224's main chain amide and the terminal carbonyl groups of Dap and Doe in compound 10. This latter hydrogen bond network arranges the aromatic ring in Tyr β224 in a π-stacking interaction with the nucleobase of GDP, preventing its dissociation from β-tubulin. The trapping of GDP is believed to be a key mechanism by which auristatins exert their tubulin-interfering effect. In addition, all analyzed analogs have a cis-configuration at the Val-Dil amide bond in their functionally relevant tubulin-bound state, whereas in solution this bond is exclusively in the trans-configuration. This reveals the preferred binding mode of auristatin and provides a valuable tool for structure-based drug design.
Figure 4-2.
Chemical structures of auristatin analogs 11–20.
Most auristatin analogs studies have been focusing on C-terminal or N-terminal modifications46. In the C-terminal modification, Doronina et al.47 described MMAF analogs 11 and 12 where the carboxylic acid of phenylalanine was replaced by tetrazoles and phosphonates. Compound 11 had an IC50 of 6.0 nmol/L on H3396 cells and compound 12 had an IC50 of 0.09 nmol/L on HCT116 cells. Both 11 and 12 (free acid type) have already been used to form active ADCs by binding to H1F6 antibody against CD70 via the VC-linker, with potency similar to that of MMAF as the payload (IC50: 4–10 nmol/L) (Fig. 4-2B). Pettit et al.48 designed auristatin analogs 13–15 modified with C-terminal aromatic phosphate and quinoline, with an IC50 of 0.81, 0.19 and 6.35 nmol/L on McF-7 cells, respectively. Among them, 13 has excellent water solubility (>236 mg/mL), two quinoline analogs 14 and 15 showed good activity, and 14 was significantly more potent (Fig. 4-2A). Lerchen et al.49 further illustrates the degree of modification allowed in the C-terminal binding domain of auristatin. They obtained effective auristatin analogs 16–18 with an IC50 of 0.5, 1.2, and 1.2 nmol/L on 768O cells by adding substituents at the C-terminal (Fig. 4-2B). For N-terminal modification, Satomaa et al.50 demonstrated that proparyl alcohol and azide derivatives 19 and 20 retained considerable cellular potency despite the presence of hydrophilic side chains, respectively, with an IC50 of 1.0 nmol/L and <1.0 nmol/L on SKOV3 cells for 19 and 20. Their azides and alkynes can be used in subsequent cyclization reactions to form more complex hydrophilic "sugar crosslinkers” that enable binding to various antibodies (lysine, cysteine, glycan, and transglutaminase conjugates). Based on this, trastuzumab and EGFR1 ADC were prepared, which performed well in vivo with the potency of 1.5–10 mg/kg (DAR = 3) (Fig. 4-2A); Doronina obtained auristatin analogs 21 by substituting p-aminobenzoic acid to N-terminal amino acids and showed an IC50 of 0.12 nmol/L in Karpas 299 cells51. An ADC with payload 21 reduced tumor volume in the Karpas 299 human ALCL xenograft model at a dose of 1 mg/kg. However, this ADC is not as effective as the related VC-MMAE ADC (Fig. 4-1A); In 2020, Svetlana O. Doronina's team52 designed a series of alkyl-MMAF analogs to investigate the possibility of developing auristatins with tunable membrane permeability, activity, and resistance to efflux pumps. The ability of MMAF to tolerate considerable substituents at the N-terminus provides a convenient way to modulate free drug hydrophobicity without significantly affecting cell activity. They selected the branched and non-branched aliphatic groups from C1 to C12 as substituents and generated MMAF N-terminal alkyl derivatives 22. The cytotoxicity of this series of derivatives increased with the increase of alkyl chain length, and neither the spatial volume of substituents nor the increase of hydrophobicity resulted in a decrease in the activity. The novel analogs combine the characteristics of MMAE and MMAF: high efficiency, bystander activity, and overcoming MDR. These findings further expand the role of Auristatins as ADC payloads. Agensys recently introduced the azide group to the P2 and P4 subunits, resulting in a compound 23 with an IC50 of 0.057 nmol/L in MOLM13 cells. As the P2 or P4 sites serve as conjugate sites for ADC drugs, this provides a new direction for the next generation of auristatin derivatives53. Prof. Luke R. Odell's team54 developed azastatin (24), an auristatin derivative, which was more cytotoxic (IC50: 0.13–3.0 nmol/L) than MMAE (IC50: 0.47–6.5 nmol/L), suggesting that the N-terminal dimethylamine groups increased the potency of the compound. In addition, the derivative contains a central amine side chain antibody binding site, suitable for ADC development.
3.1.3. Eribulin
The natural product of polyether macrolide, Halichondrin B (25), originally isolated from Halichondraria Okadai et al.56, has been proved to have good anti-proliferation activity55 (Fig. 5A). Its structurally simplified novel non-taxane fully synthetic analog eribulin mesylate (26), a microtubule dynamics inhibitor with anti-mitotic effects, has been approved in 2010 for the treatment of patients with locally advanced and metastatic breast cancer (MBC). Michel O. Steinmetz's team57 found through the crystal structure that the binding site of eribulin to tubulin is shaped by the hydrophobic and polar residues of helices H1, H6, H7, loops H6–H7, S3–H3 (T3), and S5–H5 (T5) of β-tubulin (Fig. 5B). In addition, the “cage” structure of eribulin is in contact with the ribose moiety of the guanosine nucleotide. In the tubulin‒eribulin complex, the ligand wraps around the side chain of Tyr224. When eribulin binds in the context of two longitudinally aligned tubulin dimers as found in microtubules, the strand S9, helix H10, and loop T7 of α-tubulin from the neighboring tubulin dimer will clash into the main body of the eribulin molecule. These results suggest that eribulin can sequester tubulin dimers into assembling incompetent tubulin‒eribulin complexes at high concentrations, disturbing the extension of the primordial thread at the end of the microtubule at low concentrations, which can destabilize the microtubules or inhibit microtubule dynamics.
Figure 5.
Design and SAR analysis of eribulin analogs. (A) Chemical structures of eribulin analogs 25–27; (B) Binding mode of eribulin 26 in complex with tubulin (PDB code 5JH7).
Eribulin'‘s strong anti-mitotic activity in tumor biology makes it promising as an ADC payload. Earl F. Albone's team58 designed an ADC with Eribulin as the payload by adding a linker to the C-35 primary amine group, which had a strong effect on ovarian cancer cell IGBOV1 (IC50 is 20 pmol/L). In a non-small cell lung cancer cell NCI-H2110 xenograft model, a dose of 5 mg/kg ADC induces complete tumor elimination, suggesting that the microtubule targeting agent eribulin can be used as an ADC payload. In addition, as early as 2011, Sridhar Narayan's laboratory59 synthesized and evaluated several second-generation eribulin analogs with neutral C32 side chains. These side-chain-modified eribulin analogs are significantly more potent on U-251 and SF-295 cells than eribulin. By introducing low alkalinity amines into the C32 side chain or increasing the lipophilic activity of eribulin, the obtained compound 27 has an inhibitory effect on a variety of xenograft tumors in vitro and in vivo and has oral bioutilization value. It can increase exposure in the cerebrospinal fluid and may provide drug candidates for the treatment of a wider variety of human cancers such as brain tumors (Fig. 5B).
3.1.4. Tubulysins
Tubulysins (28) is a natural anti-mitotic peptide isolated from the culture medium of myxomycetes by Hofle et al. (Fig. 6A), which is a linear tetrapeptide consisting of N-methyl-d-pipecolic acid (Mep), Lisoleuine (L-Ile), Tubuvaline (Tuv) and Tububealanine (Tup) or the microtubule hemolysin Tut (Tut)60. It can inhibit the polymerization of tubulin and induce cell apoptosis and has a strong anti-proliferation activity on cancer cells including multi-drug resistant KB-V1 cells (IC50 = 0.08 nmol/L), which has a good prospect in the development of anti-cancer drugs. In 2018, K. C. Nicolaou's team61 synthesized highly potent tubulysins analogs 29 and 30 with IC50 of 6 and 3 pmol/L, respectively, for HEK293T cells. In 2021, the team conducted crystallographic studies of compound 30 with T2R-TTL and found that compound 30 bound to the Vinca domain, forming an elongated conformation within the pocket of the β- and α-tubulin molecular interfaces near the nucleotide-binding site, where the phenyl carbamate moiety of the molecule localized on a hydrophobic bag of amino acid residue L248 of the α2-tubulin subunit (Fig. 6B). According to the combination mode, they designed an ADC with good stability in plasma and significant effectiveness on NCI-N87 mouse tumor model, which has potential in the research of targeted cancer therapy.
Figure 6.
Design and SAR analysis of tubulysin analogs. (A) Chemical structures of tubulysin analogs 28–34; (B) Binding mode of compound 30 in complex with tubulin (PDB code 6Y4N).
An SAR study of tubulysin demonstrated the feasibility of key residue modifications in tubulysins: at the N-terminus, the 6-membered ring of the Mep residue can be efficiently replaced by 5-membered and acyclic residues N-methyl-d-proline, N-methylsarcosine, and N,N-dimethyl-d-alanine with minimal loss of activity in most cases; C11-acetyl modification of the Tuv moiety can have a significant effect on the activity: acetyl hydrolysis into secondary alcohols will result in a loss of activity up to 1000 times, though it can tolerate a wide range of changes, especially to N,O-acetal changes; In contrast, the Tup residue is a commonly targeted dosing conjugate site that can be modified; The substituent on C13 is not tolerant to change, and replacing the isopropyl group into cyclohexyl or aryl (phenyl and p-methoxylphenyl) group will result in the loss of activity; The carboxylic acid end is a common conjugate site and is therefore often modified62. In order to overcome the instability caused by hydrolysis of tubulysin C-11 acetate, Patrick J. Burke63 designed a C-11 alkoxy analog 31, which showed similar biological activity to tubulysin M and significantly improved plasma stability (Fig. 6A). Chakrapani Subramanyam64 identified a potent tubulysin analog 32 which contains an α-methyl tertiary amine. The resulting ADC showed good potency both in vivo and in vitro. Heng Cheng65 prepared a carbamate-containing tubulysin analog 33, whose carbamate group is stable in vivo. Its IC50 on OVCAR3 and N87 cells is 0.20 and 0.23 nmol/L, respectively, and 0.01 μmol/L of an ADC formed by conjugating compound 33 and anti-meso antibodies can induce a significant delay in tumor growth. Derivative 34 containing non-hydrolysable N-substituents on tubuvaline (Tuv), with an IC50 < 0.28 nmol/L in HT-29 cells, also has the potential to act as ADC payloads66.
3.1.5. Cryptophycins
Cryptophycin is a natural macrocyclic polypeptide produced by cyanobacteria with a 16-member ring. It contains two hydroxy acids (A and D units) and two amino acids (B and C units). Cryptophycin irreversibly inhibits β-tubulin polymerization during mitosis, which can lead to cell cycle blockade in the G2/M phase and activate the apoptosis pathway, thereby producing picomolar antiproliferative capacity in vitro. The first synthetic and evaluated cryptophycin analog, cryptophycin-1 (35), had an IC50 of 4.58 pmol/L for KB cells and 7.63 pmol/L for LoVo cells67(Fig. 7A). However, due to the instability of macrolide in blood circulation, its in vivo antitumor activity is not significant. After further structural modification, cryptophycin-52 (36) with one methyl more than cryptophycin-1 at the α position of C unit β-alanine was obtained, which increased steric hindrance and significantly improved the in vivo stability. Very low concentrations of cryptophycin-52 (36) inhibited cell proliferation during mitosis (IC50 = 11 pmol/L) without significant changes in microtubule length or quality. The binding of cryptophycin-52 (36) to tubulin is very tight, and the affinity of the cryptophycin-52 (36)-tubulin complex to microtubules is very high (Kd = 47 nmol/L). Although cryptophycin-52 is effective, it was terminated in phase II due to dose-dependent toxicity. Using this compound as the payload of the ADC may reduce its toxicity. Recently, Paul T. Wingfield's team68 identified the binding site of cryptophycin-52 (36) and its parent compound cryptophycin-1 (35) on tubulin at the tubulin dimer interface, partially overlapping with maytansine (Fig. 7B). The conformations of both α-tubulin and β-tubulin have changed, especially in helix H8 and H10. There are significant differences between α,β monomers and between cryptophycin-52 (36) bound, cryptophycin-1 bound tubulin. Then, the suitable sites on cryptophycin were analyzed: In unit A, para-substituted phenyls are more potent than ortho-substituted or meta-substituted analogs, and the para-substituted phenyl could be used as the conjugate point; Unit B is sensitive to slight modifications, such as 5-fluoro substitution on benzene rings. In unit C, large substituents such as benzyl or isopropyl at the C6 position strongly reduce its potency; However, the activity only slightly changes after modifications on unit D, and the isobutyl group in unit D can be used as the conjugate point. The isobutyl conjugate site of unit D may be better than that of unit A. This analysis provides a reference for the subsequent design of ADC with cryptophycin as a payload to find suitable conjugation sites.
Figure 7.
Design and SAR analysis of cryptophycin analogs. (A) Chemical structures of cryptophycin analogs 35–40; (B) Binding mode of cryptophycin-52 (36) in complex with tubulin (PDB code 7M20).
The analog 37 of cryptophycin-52 (36) (IC50 = 33 pmol/L) is a very promising clinical drug candidate and has been patented by Eli Lilly (Fig. 7A). The aromatic ring of unit A in cryptophycin-52 can be modified in place without significantly affecting its biological activity. These modifications are used by Sanofi-Aventis as conjugating points for antibody conjugation. Cryptophycin 52-chlorohydrin compounds such as cryptophycin-55 (38) are 100–1000 times more bioactive in vivo. Cryptophycin-55 glycine (39) showed the highest cell activity in vitro. Cryptophycin-55 glycine is conjugated to trastuzumab targeting HER2 receptor via a cleavable linker, and the resulting ADC exhibits nanomolar activity against SK-BR3 cells. Genentech converts benzene from cryptophycin-52 to benzylamine to obtain a payload 40 suitable for connection via carbamate bonds69. Jinliang Yang's team70 conjugated cryptophycin-55, a prodrug of cryptophycin-52 (CR55, which can be reincarnated into cryptophycin-52 under physiological conditions), with trastuzumab. Its IC50 were at low nanomolar levels (0.58–1.19 nmol/L) in HER2-positive tumor cell lines, and 10 mg/kg of the corresponding ADCs showed significant anti-tumor activity in xenograft models of ovarian cancer (SKOV3) and gastric cancer (NCIeN87). It provides ideas for the development of ADC antineoplastic drugs based on cryptophycin-55 and cryptophycin-52.
3.1.6. EG5 inhibitors
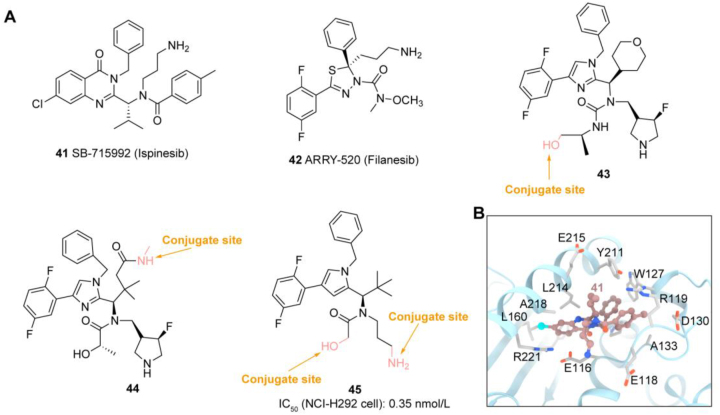
Kinesin (KSP/EG5/KIF11) is an ATP-dependent protein that participates in the isolation of centrosomes and the production of bipolar spindles during the G2/M phase of the cell cycle, playing an important role in mitosis. High expression of EG5 in blood tumors (e.g., AML parent cell and diffuse large B-cell lymphoma (DLBCL)) and solid tumors (e.g., breast cancer, bladder and pancreatic cancers) is associated with their poor prognosis, making it an attractive cancer treatment target71. Several EG5 inhibitors, such as SB-715992 (Ispinesib) (41) and Filanesib (ARRY-520) (42), have already been in clinical trials (Fig. 8A). However, the most common side effects of EG5 inhibitors, such as neutropenia, mucositis, and stomatitis, as well as their narrow treatment windows, limit their therapeutic effectiveness. By taking advantage of ADCs however, healthy tissues can be protected from exposure to cytotoxic compounds, potentially reducing their overall toxic side effects and expanding the therapeutic window for compounds. Dimitrios A. Skoufias's team72 compared the structure of the Eg5 Arry-520 complex with that of the Eg5 ispinesib complex (PDB ID:4AP0) (Fig. 8B). The superposition of the two complexes shows that the quinazolinone of ispinesib occupies a similar position to the difluorophenyl ring of Arry-520 and has a similar interaction. In ispinesib, the two aromatic substituents p-toluene and benzyl are stacked on top of each other, stabilizing the skeleton with Glu118, Arg119 and further stacking of Arg119, Trp127 and Asp130 side chains. However, in the Eg5 Arry-520 complex, this skeleton stacking is much weaker. In addition, for Arry-520, there is a direct hydrophobic stacking interaction between difluorophenyl and isobutyl side chain residues, while in ispinesib complexes, the side chain moves 2 Å to accommodate benzyl and p-toluene, thus completely weakening or losing this interaction. Arry-520 and ispinesib form hydrogen bond interactions between the primary amines of Gly117 and the side chains of the main chain oxygen and Glu116. The oxygen of the methoxy group in Arry-520 may interact weakly with the hydroxyl groups of Tyr211 (3.63 Å). In addition, one fluorine in the phenyl ring interacts with the main chain NH (3.03 Å) of Gly217 and weakly interacts with the side chain of Arg221 (3.69 Å). In general, the tight binding of Arry-520 is achieved through a series of strong or weak hydrogen bond interactions and hydrophobic interactions with the amino acid side chains in the inhibitor binding bag. In addition, both D130A and L214A mutations in the allosteric binding site of Eg5 can make Array-520 resistant, while ispinesib is still sensitive to L214A mutant. Based on this, subsequent modifications of the compounds may be considered to avoid resistance to certain mutations that lead to reduced compound inhibition, producing new inhibitors that can overcome these potential clinically relevant problems.
Figure 8.
Design and SAR analysis of EG5 inhibitors. (A) Chemical structures of EG5 inhibitors 41–45; (B) Binding mode of ispinesib (41) in complex with EG5 (PDB code 4AP0).
Novartis developed EG5 inhibitors 43, 44 as payloads conjugating with an antibody to target HER2 using non-lysable linkers (Fig. 8A). In a HER2 expressing mouse xenograft model (SK-OV-3ip), a single intravenous ADC dose (5 mg/kg) can cause tumor growth to stop for 3 weeks, better than Kadcyla (5 mg/kg)73. Using a non-cellular permeable EG5 pyridine inhibitor as a payload, Anette Sommer74 developed a novel IL3RA targeting ADC that demonstrated effective and selective antiproliferative effects. Carsten Terjung75 investigated the applicability of a new of EG5 pyrrole subclass inhibitor 45 as a novel payload for ADC. In a xenograft model of urothelial cell carcinoma (UCC), this ADC showed a high efficacy which results in complete tumor eradication.
Disruption of microtubules can induce a cell cycle arrest in the G2/M phase, which makes microtubules an attractive target for drug discovery. In many cases, the narrow therapeutic window and lack of tumor specificity can lead to the failure of potent tubulin inhibitors as anticancer agents alone. However, using ADCs as targeted therapies may be a promising way to address the limitations of tubulin inhibitor monotherapy. More than half of the ADCs currently in clinical development have tubulin inhibitors (auristatins and maytansinoids) as payloads. While tubulin inhibitors have had some commercial success as ADC payloads, they are limited by various shortcomings in the development process. For example, it can only target the tumor cells in the dividing phase, but not the cells in the non-dividing and static phase. In addition, tubulin inhibitors in tumor cells have a higher number of targets, which means that a larger dose of the drug is required to stop tubulin. As a result, these ADCs with tubulin inhibitors as payloads are still far from the ideal targeted therapeutic concept.
3.2. DNA targeting payloads
Compared to tubulin inhibitors, DNA inhibitors can act on the whole cell cycle by destroying DNA through double-strand breakage, alkylation, chimerism, crosslinking, etc., causing cytotoxic effects, and having a good therapeutic effect on solid tumors76. In addition, DNA inhibitors have far fewer targets than tubulin inhibitors, and DNA inhibitors can show a better killing effect when ADCs carry the same amount of payloads into the cells. Besides, ADCs with DNA inhibitors as payloads can target tumor cells with low antigen expression, which explains why DNA inhibitors have been chosen as payloads in many next-generation ADCs.
3.2.1. Enediyne
Enediyne isolated from bacterial sources is one of the most cytotoxic natural products found so far. The uptake of which will result in cell death by breaking the single or double strands of DNA in living cells, and it also has extremely strong activity against various bacteria and tumor cells. Enediyne can be divided into two subfamilies: Cal-like enediyne and anthraquinone fusion enediyne. The highly toxic enediyne compounds are not suitable for direct use as anticancer agents, but the potency and mechanism of action of these antitumor antibiotics make them attractive ADC payloads77. The most commonly used ADC payloads of natural enediyne products are calciheamicin γI1 (46) from the Cal enediyne class and the anthraquinone fused enediyne Uncialamycin (47) (Fig. 9A).
Figure 9.
Design and SAR analysis of enediyne. (A) Chemical structures of enediyne 46–52; (B) Binding mode of calicheamicin γI1 (46) in complex with DNA (PDB code 2PIK).
Calicheamicins target small grooves that bind to DNA and specifically induce DNA double-strand breakage, leading to apoptosis. Although the initial experiments showed strong activity, the destruction of normal cell DNA by calicheamicins impeded their clinical progression. Nonetheless, high cytotoxicity, small molecule size, and a well-defined mechanism of action make calicheamicins attractive ADC payloads78. Calicheamicin γI1 (46) is currently the most studied calicheicins. Dinshaw J. Patel and his team79 investigated the high-resolution structure of calicheamicin γI1-DNA double-stranded complex and found that the directional alignment of the aryltetrasaccharide of calicheamicin deep in the minor groove centered about the oligopyrimidine oligopurine tract, in turn, orients the attached enediyne ring to span both strands of the duplex in the intensity refined structures of the complex (Fig. 9B). The enediyne ring which is tilted relative to the helix axis adopts an orientation deep in the minor groove which allows abstraction of DNA sugar-phosphate backbone protons following activation by the exposed trisulfide trigger and cycloaromatization, which ultimately results in DNA strand cleavage. There is a close complementarity at the interface between the drug and the DNA minor groove which spans the entire binding site in the refined structures of the complex. The study provides a database to address two issues in the field associated with the identification and cutting of DNA of the calicheamicin class. Calicheamicin γI1 (46) has a strong cellular effect, and the emergence of ADCs represented by Mylotarg largely depends on the discovery of calicheamicin γI1 (46), whose N-acetyl derivative becomes the payload of Mylotarg. Nicolaou et al.80 synthesized Calicheamicin θI1 (48), an analog of calicheamicin γI1, which has an IC50 value of <1 pmol/L for a variety of tumor cells and is also suitable for use as an ADC payload. In addition, the FDA-approved Inotuzumab Ozogamicin (Besponsa) also has calicheamicin as a payload.
Uncialamycin is a natural enediyne product isolated from lichens, which can cause DNA cleavage and shows strong cellular activity. One of the common structural features of the potent analogs of uncialamycin is the presence of a primary or secondary phenylamine group on the A ring. If the amine is converted to the corresponding benzoamide and the terminal arylamine is introduced, the instability problem can be solved and the activity remains81. Sanjeev Gangwar82 used the phenol alkylation method and designed a highly potent and chemically stable uncialamycin analog 49 (Fig. 9A). The resulting ADC by binding the payload with the anti-mesothelin (meso) antibody has an IC50 of 0.2 nmol/L in H226 cells, exhibiting specific and lasting tumor growth inhibition at tolerated doses. Julia Gavrilyuk et al.83 selected the methylamine analog 50 as the uncialamycin ADC payload, and ADCs targeting TL and CD46 exhibited low picomole potency in HEK293T cells. Emmanuel N. Pitsinos84 modified uncialamycin to remove a carbon from the aromatic part of the molecule to obtain the compound 51 (the cell line under test: IC50 (H226 cell) = 28 pmol/L; IC50 (N87 cell) = 11 pmol/L; IC50 (OVCAR3 cell) = 316 pmol/L; IC50 (Adr cell) = 20 pmol/L). Sanjeev Gangwar85 conjugated the uncialamycin analog 52 to an anti-mesothelin (meso) antibody. The resulting ADC showed an IC50 value of 0.98 nmol/L and an IC50 value of 0.88 nmol/L in H226 lung cancer cells.
3.2.2. Topoisomerase I inhibitors
Topoisomerase I (TOPO-I), an important ribozyme for genomic stability and DNA structure preservation, has become a popular target for ADCs. TOPO-I inhibitors are associated with innate and adaptive immune responses, suggesting that ADCs targeting TOPO-I may also contribute to antitumor immunotherapy86. Lance Stewart et al.87 reported the X-ray crystal structure of TOPO-I covalently linked to double-stranded DNA in combination with the clinically approved anticancer drug topotecan (53) (Fig. 10A). Topotecan (53) and the human TOPO I-DNA covalent complex suggest that topotecan is a non-competitive inhibitor that binds to an enzyme–substrate complex by inserting bases between the two strands of DNA at an enzyme-induced notch. The binding mode of topotecan is stacking interactions with the DNA, a hydrogen bond contacting Asp-533, and water-bridged contacts to the active site between phosphotyrosine and Asn-722. The natural pentacyclic product camptothecin (CPT) (54) (Fig. 10B), an analog of topotecan (53), is a particularly attractive class of ADC payloads that induce double-stranded DNA breakage in cells in the S phase by binding to TOPO-I and DNA to form a stable complex, causing apoptosis. However, the extremely low solubility of camptothecin prevents its widespread use as a cancer treatment drug. SN-38 (56), the active ingredient of irinotecan CPT-11 (55), is a semisynthetic camptothecin. Although SN-38 has the desired potency, the preparation of active conjugates is challenging due to its highly hydrophobic nature and the limited number of conjugation sites available. Still, SN-38 has been conjugated to a humanized antibody against trophoblast cell-surface antigen 2 (Trop-2), resulting in the ADC sacituzumab govitecan, which is in clinical investigation. Exatecan mesylate (DX-8951f) (57) is a water-soluble CPT derivative with stronger TOPO-I inhibitory and antitumor activity than other CPT analogs. DXd (DX-8951 derivative) (58) is a novel, highly membrane-permeable topoisomerase I inhibitor with a cell IC50 of 2.05–17.88 nmol/L, which can overcome P-glycoprotein-mediated multidrug resistance and has stronger efficacy for various tumor transplant models, including in vivo CPT-11 resistant tumors88.
Figure 10.
Design and SAR analysis of Topoisomerase I inhibitors. (A) Binding mode of Topotecan (53) in complex with TOPO I-DNA (PDB code 1K4T); (B) Chemical structures of Topoisomerase I inhibitors 53–60.
DXd (58) has an F ring that contains a second chiral center, complicating its synthesis and derivatization. To simplify its structure, Wayne C. Widdison's89 SAR study showed that fluorine substitution at the C11 position usually increases cytotoxicity several times, and the C20 center must be in S configuration for the molecule to remain active. The improved stability of exatecan lactone is attributed to the action of the C11 fluorine substituent and the F ring. ADCs derived from camptothecin derivatives 58 and 59 can effectively release part of the metabolites of camptothecin containing sulfide or mercaptan. These metabolites have a strong inhibitory effect on targeted Ag + cells and show similar dose-dependent antitumor activity in both HSC-2 and H1703 EGFR + xenotransplantation models. There is no F ring in payload 59, but its conjugates have similar efficacy in vitro and in vivo as those containing DXd. This simplification makes the derivatization of camptothecin A and B rings easier to use for structure‒activity relationship studies and payload optimization. Dongke Yu90 obtained a novel HER2-targeting ADC DS-8201a by conjugating DXd to trastuzumab. The SAR showed that hydroxyl substitution of C-10 could enhance the anti-tumor activity of CPT, and the dissubstitution of C-7 and C-10 could stabilize the lactone ring and improve the molecular biological activity. DXd is cyclized at the C-7 and C-9 sites, thereby increasing the inhibitory activity. DS-8201a avoids drug resistance and is very effective in treating heterogeneous tumors through the bystander killing effect. Scott C. Jeffrey91 developed a novel hydrophilic and protease lytic glutlysine tripeptide ligand, using a highly cytotoxic 7-aminomethyl-10,11-methylene dioxygen CPT analog 60 as the payload. The conjugated ADC showed effective antitumor activity in both CD30 highly expressed strains and MDR-positive 786-O renal cell carcinoma nude mice.
3.2.3. PBD
Pyrrolo[2,1-c][1,4] benzodiazepines (PBD) (61) found in Streptomyces are a class of natural products with antitumor activity (Fig. 11). Such compounds consist of an aromatic A ring, a 1-4-diaza-5-1 B ring, and a pyrrolidine C ring. Their mode of action is selective alkylation in small grooves in DNA, where N2 of guanine forms a covalent bond with the electrophilic N10/C11 imine of PBD. Continuous DNA damage is caused by cross-linking between DNA chains, which leads to cell cycle arrest in the G2/M phase and cell apoptosis, thus exhibiting a powerful cytotoxic effect92. NMR structural data from Klaus Weisz's team93 confirmed the strong binding of PBD heterozygotes to small grooves of DNA double strands, forming covalent bonds between the PBD moiety and the outer ring guanine amino group.
Figure 11.
Design and SAR analysis of PBD analogs 61–72.
PBD dimers have the potential to be used as ADC payloads, such as the ROR1-targeting ADC CS5001 designed with PBD as a payload, which has shown strong selectivity in a variety of ROR1-expressing tumor cell lines and has shown significant in vivo anti-tumor activity in blood and solid tumor xenograft mouse models. Clinical trials are currently underway. To further expand more PBD-like compounds as ADC payloads, Rahman and colleagues modified the C8 position of PBD to obtain compound 62, whose conjugates have preclinical activity. Based on compound 62, Khondaker Miraz. Rahman94 synthesized PBD core and constructed C8 hybrid 63 using the combination of benzofuran and N-methylpyrrole (Fig. 11). Subsequently, a series of compounds were obtained by systematically shortening the C8 linked monomer containing the pyrrole benzofuran motif (e.g., the C8 linked monomer of compound 64 was shortened by removing of amide). It was found that a relatively short side chain at the C8 position of PBD was sufficient to confer cytotoxicity. Therefore, the shortened PBD monomer could be used as a new ADC payload. The dihydrobenzofuran compound 64 was highly toxic in primary CLL cells and opening the furan ring also maintained low nanomolar activity in primary CLL cells and JN-3 cell lines. The change of benzene element in benzofuran had a more significant effect on cytotoxicity, and the cytotoxicity was significantly reduced after removing the aromatic properties of the ring. Jagath R. Junutula95 identified an isoquinoline benzodiazepines (IQB) payload 65 whose (S,S) stereochemistry at the C6a position is critical for optimal IQB activity. This is a new class of PBD dimer DNA damage payloads, showing high activity in HEPG2 A704 cell line. Cytotoxic pyrrolobenzodiazepine (PBD)-dimer molecules are frequently utilized as payloads for ADCs. In order to further explore this ADC payload class, Peter S. Dragovich96 modified their physicochemical properties by the systematic introduction of acidic and basic moieties into their chemical structures. The results suggested that the PBD-dimer payloads containing acidic functional groups (including highly ionizable moieties such as the sulfonic acid present in compound 66) were able to efficiently transport from the cellular lysosome to the cytosol and/or nucleus following antibody-mediated delivery. The modified PBD dimer compound 66 was conjugated with CD22 antibody to obtain an ADC with an IC50 of 12 nmol/L against BJAB cells, significantly improving the anti-proliferation activity of the unconjugated PBD dimer. The PBD dimer payload 66 will contribute to the design of new ADCs with higher therapeutic potential. Peter S. Dragovich explored the dimer of PBD containing disulphin prodrugs as the payload of antibody-drug conjugates and found that many PBD monomers containing disulfide prodrugs exhibit strong antiproliferative activity against cells (such as KPL-4 cells) with relatively high intracellular GSH levels. Multiple disulfide precursors are stable in physiological concentrations of Cys, but unstable in the presence of GSH in tumor cells. Increasing the terminal space volume of compounds 67 and 68 can significantly improve their stability. For example, the obtained compounds 69 and 70 containing two methyl groups are highly stable to both GSH and Cys. The stability of thiophene-derived disulfide was improved in vivo97. In these common PBD payloads, the conjugate sites on PBD dimers are divided into two categories: C2 and N10. An example of a C2 conjugation is payload 71, which contains a phenyl ring attached to the PBD C2 position. The payload SG3199 (72) of the CD19-targeting ADCT-402 is conjugated through its N10 position98. Since the N10 site on the PBD B ring is involved in covalent binding to DNA, the conjugate of the linker to the N10 site is safer than that to the C2 site99. In the subsequent work of PBD payloads, its conjugate sites can also be considered as a factor affecting the direction of modification.
3.2.4. Duocarmycins
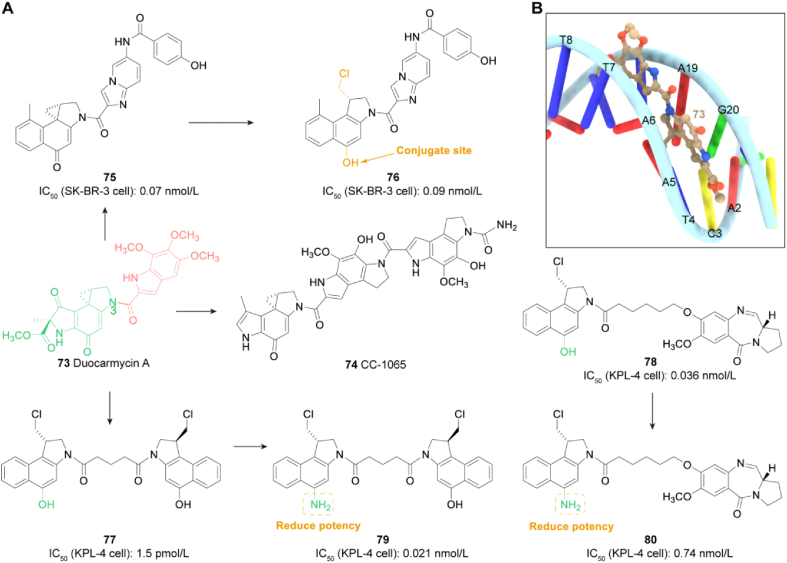
Duocarmycin A (73) is a strong DNA alkylation agent isolated from Streptomyces, consisting of a DNA alkylation portion and a binding portion (Fig. 12A). The first discovered natural member of duocarmycins, CC-1065 (74), binds to the DNA microgroove through its highly active propane ring and alkylates adenine at the N3 position, ultimately leading to cell death. Although CC-1065 (74) is highly potent in vitro, it has shown only moderate in vivo activity and irreversible hepatotoxicity in animal models. To improve the biological characteristics of these compounds, using them in ADCs may be a viable choice100. Walter J. Chazin et al.101 described the structure of unnatural enantiomers and DNA adducts of duocarmycin SA and found that both enantiomers of duocarmycin SA can share the same binding site and the same orientation of binding on the AATTA duplex yet alkylate different sites (Fig. 12B). Alkylation of the complementary strands by the two enantiomers occurs from the same bound orientation but is offset by one base-pair for the unnatural enantiomers. This difference has important implications for the structures of the two adducts, which as a result, will affect their relative alkylation efficiencies. Analysis of this new structure, bound to the same AATTA duplex explains the observed similarities and differences in DNA site selectivity for natural versus unnatural enantiomers. Inter-subunit twist has been observed in all structures of DNA adducts of the duocarmycin/CC-1065 family. Since DNA has a left-handed helical twist, the natural tendency for the trimethoxyindole subunit of DSA is to twist in a counterclockwise fashion relative to the alkylation subunit when bound in the minor groove. Thus, the handedness of the DNA molecule itself imposes an important constraint on the (+) enantiomer agent that is not present for the (−) enantiomer agent. The bulky C24 methoxy group of both DSA enantiomers is located in approximately the same position on the floor of the minor groove, in the cleft between the A16 and T17 bases. This research provides a new understanding of the factors controlling DNA alkylation rates by these compounds and the importance of DNA-binding-induced activation of the alkylation reaction.
Figure 12.
Design and SAR analysis of duocarmycins analogs. (A) Chemical structures of duocarmycins analogs 73–80; (B) Binding mode of Duocarmycins A (73) in complex with DNA (PDB code 1DSM).
Patrick H. Beusker102 developed a novel payload for reducing interchain disulfide based on duocarmycins. It was combined with trastuzumab, an anti-HER2 antibody, to produce ADC SYD985 with favorable properties in vitro and in vivo. Duocarmycins DUBA (75) with imidazole [1,2-a] pyridinyl is incorporated into the conjugated drug in its inactive prodrug form, seco-DIBA (76) (75, formed through spontaneous spirocyclization of 76) (Fig. 12A). Compound 76 contains two hydroxyl groups, each of which can be conjugated to the antibody via a linker. Moana Tercel used a Pd-catalyzed amination method to convert seco-CBI (seco-1,2,9,9a-tetrahydrocyclopropa[c]benz[e]indol-4-one), a synthetic analog of the alkylating subunit of the duocarmycin natural products, from the phenol to amino form. Then obtain phenol CBI analogs 77, 78 and amino CBI analogs 79, 80. Compound 77 has an IC50 in the range of 0.1–2 pmol/L for six human tumor cell lines. Replacing compound 77 or 78 phenol CBI with corresponding compounds 79 and 80 amino CBI results in a significant reduction in potency and a greater sensitivity to P-gp overexpression. Nevertheless, compound 79 maintained significantly low picomolar cytotoxicity in most of the cell lines tested, and, like compounds 77, 78 and 80, completely inhibited proliferation of all cell lines at sufficiently high concentrations103.
A clear trend in the ADC field is that the therapeutic target is moving from hematological tumors to solid tumors. This trend may shift the choice of payload from a microtubule-targeted inhibitor to a DNA damaging agent, which typically exhibits higher potency and meets the criteria that the new generation of payloads requires a higher therapeutic index than the current payloads. In addition, using DNA-damaging agents as payloads can target tumor-specific/associated surface receptors at relatively low levels of expression. The use of DNA-damaging agents as payloads for ADCs is well established. However, since DNA damage is difficult to repair, this may lead to potential toxic side effects. Currently, only one ADC with PDB as the payload is in clinical use. Therefore, their safety/drug resistance issues still require further study, and more new generations of ADC payloads need to be developed in the future.
3.3. RNA targeting payloads
There are still many tumors that do not respond to ADCs or develop resistance to drugs. Slow-growing tumor cells, typically do not rely on tubulin-mediated cellular processes as much as rapidly dividing cells. Therefore, in order to further expand the range of ADC payloads and identify other types of ADC payloads that are effective in both fast and slow-proliferating cells and can escape MDR-mediated resistance, attention has turned to small molecular payloads targeting RNA. Small molecule inhibitors that target RNA can kill both dividing and dormant tumor cells, and being used as an ADC payload is expected to solve the problem of tumor drug resistance and tumor recurrence and spread caused by ineffective tumor dormant cells104. There are currently two main types of RNA inhibitors available for ADC payloads: RNA splicing inhibitors (Thailanstatin and its analogs) and RNA polymerase II inhibitors (Amatoxins).
3.3.1. Thailanstatin
RNA splicing controls metabolism, angiogenesis, cancer cell proliferation and metastasis mainly by excision of introns and exons that are responsible for the complex cellular mechanism of transforming RNA to mRNA. They can also directly control the initiation, elongation, and termination of transcription, which is the biological target of cancer inhibition. Abdel-Wahab et al.105 found that under the interference of RNA splicing regulatory drugs, tumor cells can produce neoantigens, which could be presented as neoantigen epitopes through MHC I, thereby stimulating anti-tumor immunity. It showed that RNA splicing regulation can be used as a potential source of tumor antigens, with the potential to be applied to tumor immune checkpoint therapy. Spliceosome inhibitors are potent antiproliferative drugs capable of targeting cells that are actively dividing and quiescent. RNA spliceosome inhibitors are therefore a promising class of ADC payloads. Thailanstatin is a natural product originally isolated from Brucella Thailand msmb43 and activated by binding to the SF3b subunit of the spliceosome U2 snRNA subcomplex. The thailanstatin family has a strong binding and inhibitory effect on spliceosomes by inhibiting the eukaryotic mRNA splicing pathway, resulting in low nanomolar IC50 values for a variety of cancer cell lines, which can be used as a potential payload for ADCs106.
Yi-Qiang Cheng106 reported three new thailanstatin compounds isolated from MSMB43: thailanstatin A (81), thailanstatin B (82), and thailanstatin C (83) (Fig. 13). Thailanstatin A (81) is a potential natural anti-cancer drug candidate. The overall structure of the thailanstatin drug is similar to that of the spliceosome inhibitor candidate FR901464 (84), except that the C1 position of thailanstatin lacks an unstable hydroxyl group, while the C17 position has an additional carboxyl moiety, so thailanstatin has better stability. The highly active epoxides at thailanstatin A (81) and FR901464 (84) C3 sites are essential in inhibiting pre-mRNA splicing and cancer cell proliferation. The biological activity of the compound is reduced by halogenation modifications thailanstatin B (82) and thailanstatin C (83) on epoxides. The addition of methyl to thailanstatin C (83) (as compared to thailanstatin B (82)) had no significant effect on the bioactivity of the compound. Stephan Rigol107 has synthesized the thailanstatin analogs 85 and 86, which have the IC50 value of picomoles. Pfizer conducted the first exploration of RNA splicing inhibitors as antibody drug conjugate payloads. The carboxylic acid-containing thailanstatin semi-synthetic analog 87 was directly coupled to trastuzumab surface lysine to obtain a very effective thailanstatin ADC. Compound 87 and its corresponding ADCs are effective in both HER2 highly expressed gastric cancer cell line N87 and MDR1 overexpressed cell lines (compound 87 in N87 cells IC50 is 1.8 nmol/L). In addition, the exposure of the ADCs was sufficient to result in excellent potency in a gastric cancer xenograft model at doses as low as 1.5 mg/kg, which was superior to the clinically approved ADC T-DM1108.
Figure 13.
Design and SAR analysis of thailanstatin analogs 81–87.
3.3.2. Amatoxins
Amatoxins were first isolated by Heinrich Wieland and Rudolf Hallermayer in 1941. Amatoxins is a toxic dicyclic octapeptides synthesized by ribosomes and is a selective inhibitor of eukaryotic RNA polymerase II that can lead to apoptosis. Amatoxins contain a large ring of eight l-configuration amino acids, connected between tryptophan and cysteine residues through the sulfoxide moiety, whose three side chains are hydroxylated and thus have good water solubility. The most in-depth studies of amatoxins are the α-amanitin (88) containing 6-hydroxytryptophan (Htp), trans-4-hydroxyproline (Hyp), and (3R,4R)-4,5-dihydroxyloleucine (Dhil), which is the most effective and specific RNA polymerase II inhibitor known to date, with the potential to break through drug resistance or destroy silent phase tumor cells, effectively preventing tumor metastasis and recurrence109 (Fig. 14A). Roger D. Kornberg's team110 analyzed the crystal structure of RNA polymerase II-α-amanitin and found that the position of the inhibitor is adjacent to the Bridge Helix and the Trigger Loo is predominantly in a stable conformation away from the addition site (Fig. 14B). The α-amanitin cyclic peptide backbone forms a pocket that allows specific complementary hydrogen bonds to the Rpb1 His1085 side chain imidazole ring, creating a point of restraint for the Trigger Loo. The specificity for imidazole by the α-amanitin pocket is achieved primarily through the interaction of amide NH of Gly7 to Rpb1 His1085 N (D1) and the carbonyl of Asn1 to Rpb1 His1085 NH (E2). The study showed that α-amanitin inhibits the extension of RNA polymerase II by directly interfering with the Trigger Loo of RNA polymerase II and exhibits slowly decreased substrate selectivity.
Figure 14.
Design and SAR analysis of amatoxins. (A) Chemical structures of amatoxins 88, 89; (B) Binding mode of α-Amanitin (88) in complex with RNA (PDB code 3CQZ).
Toxicity such as apoptosis and necrosis caused by amatoxins has hindered its clinical application. But due to its small molecular weight, good solubility in aqueous solutions, and inhibition of RNA polymerase II, it has received increasing attention in cancer studies in recent years as an antibody‒drug conjugate payload. α-Amanitin ADC is highly likely to act on both proliferating and quiescent tumor cells111. In the ADC field, the use of amanitin as a payload is a relatively new approach. HDP-101 (ClinicalTrials.gov identifier: NCT04879043), a B cell maturation antigen (BCMA)-targeted ADC developed by Heidelberg Pharma with amatoxins as payloads, is currently in phase I/IIa clinical trials for the treatment of relapsed/refractory multiple myeloma, and the first patient administration was completed on February 15, 2022. HDP-102 is an ADC drug that targets CD37 and is targeted for non-Hodgkin lymphoma (NHL). HDP-103 targets prostate-specific membrane antigen (PSMA) with the target indication for metastatic castration-resistant prostate cancer (mCRPC)112. Steffen Goletz113 conjugated the carboxyl group of β-amanitin (89) to the amino group of IgG lysine, resulting in an ADC with good plasma stability and high cytotoxicity (Fig. 14A).
ADCs with thailanstatin and amatoxins as payloads are able to avoid treatment resistance and be effective against dormant tumor cells, whereas existing standard therapies rarely achieve this effect and often lead to tumor recurrence and drug resistance. Therefore, in addition to tubulin inhibitors and DNA damage agents as payloads, the new generation of RNA inhibitors as payloads of ADCs has received more and more attention. Although there are no RNA-payload ADCs on the market, their great potential warrants further explorations.
3.4. Immune ADC payloads
Initially, ADCs were limited to oncology. However, their clinical success has encouraged the application of ADCs in other therapeutic areas, most notably immunology. Tumor immunity involves T cells, macrophages, dendritic cells, etc., which transform cold tumors into hot tumors through immunomodulation, and ultimately enhance the effect of immunotherapy. Immune checkpoint inhibitors enhance the anti-cancer immune response of T cells by eliminating tumor inhibition of T cells. However, for cancer patients (whose tumors are commonly known as cold tumors) who do not have a spontaneous T cell response and T cell infiltration around the tumor, immune checkpoint inhibitors do not work well. Therefore, one of the focuses of immunotherapy research and development is to develop anti-cancer therapies that can stimulate the patients’ innate immune response and promote tumor-specific T cell recruitment114. Unlike traditional ADCs, immune-stimulating antibody conjugates (ISACs) combine the precision of antibody navigated targeting and the power of small molecule-based modulation of the innate and adaptive immune systems. On the one hand, antibodies conjugate small molecules of the immuno-agonist type and play a role in enhancing the immune response. Tumor targeting of antibodies, on the other hand, reduces the toxicity of small molecule immuno agonists. In terms of target selection, traditional ADCs must strictly limit tumor-specific antigens due to payload toxicity, while immune-modulating ADCs can expand more tumor-related antigens. In terms of endocytosis, traditional ADCs are targeted antigen-mediated endocytosis, which directly kills cancer cells. Immune-modulating ADCs activate immune cells through endocytosis mediated by FcγR on the surface of immune cells21. At present, a variety of immune-modulating ADC drugs are under development, the new payloads mainly include Toll-like receptor (TLR) agonists and interferon gene stimulators (STING) agonists, and this field has great potential for development.
3.4.1. Toll-like receptor agonists
Toll-like receptors (TLRs) are an important class of protein molecules involved in innate immunity and are the first barrier for the body to resist infectious diseases. Activation of TLR receptors is able to improve antigen presentation of dendritic cells and macrophages in the tumor microenvironment and promote the proliferation of CD8+ T cells. Toll-like receptor agonists act as an immune enhancer that induces an enhanced anti-tumor immune response in lung cancer, melanoma, leukemia, and glioma, directly or indirectly, thereby effectively inhibiting tumor growth. Therefore, the application of TLR agonists to tumor immunotherapy is expected to activate the innate immune response, mediate the acquired immune response, make cold tumors hot, solve the problem of low response rate of single immune checkpoint inhibitors, improve the effectiveness of immunotherapy, and achieve the purpose of reshaping the tumor microenvironment. At present, TLR has become one of the research hotspots in the field of immunotherapy, and among the members of the TLR family, researchers pay more attention to TLR7, TLR8, and TLR9. TLR8 agonists (90) (91) activate DC cells and enhance the killing of tumor cells by T cells (Fig. 15). However, immunotherapy is often accompanied by side effects, such as cytokine release syndrome, a common condition that causes systemic inflammation. Systemic administration of TLR8 agonists also presents associated toxicity, limiting the amount of TLR8 agonists that patients can take. Therefore, the application of TLR8 agonists to ADC can simultaneously combine the targeting effect of antibodies and the immune regulatory effect of TLR8 to reduce toxic and side effects115. Toshiyuki Shimizu's team116 found that the TLR7 TM helix is aligned with the TLR7-TLR signal conditioner (UNC93B1) TM3 and TM6 helices and the juxtamembrane region, and the LRR-CT motif interact with the luminal side of the UNC93B1 N-terminal six-helix bundle. The dimerization of the TLR7–UNC93B1 complex is mediated by both TLR7‒TLR7 and UNC93B1‒UNC93B1 interactions, which occur at the top and bottom regions of the 2:2 complex, respectively. The protruding loop region of LRR11 from the middle region of the TLR7 ECD mainly contributes to the dimerization of TLR7, while the intracellular helices in the regions connecting the TM6 and TM7 from the two protomers of UNC93B1 interact with each other in an antiparallel manner. This study elucidated the structural basis of TLR's interaction with UNC93B1 and facilitated the subsequent design of compounds against autoimmune diseases.
Figure 15.
Design and SAR analysis of TLR agonists 90–92.
Silverback's SBT6050 is an anti-HER2 antibody conjugate with TLR8 agonist 91 (Fig. 15) as its payload, which drives a variety of anti-tumor immune mechanisms in a HER2-dependent manner in vitro and has shown good anti-tumor effects in mouse tumor models117 (Fig. 15). A new class of TLR7/8 dual agonists has been reported to promote tumor response to immune checkpoint antibodies, especially in cold tumors. Although the small molecules found by the team have significant anti-tumor effects, and significant synergistic effects can be observed in combination with PD-1 and PD-L1 antibodies, similar to other reported TLR agonists, small molecules are still overexposed in the peripheral blood, resulting in more obvious immune-related adverse reactions (irAEs), such as weight loss and influenza-like symptoms118. In order to avoid the adverse reactions that can be caused by direct administration of TLR7/8 agonists, the team began experimenting with antibody conjugate drug strategies in 2015. After several years of exploration, a new class of immunomodulatory antibody conjugate drug HE-S2 was successfully constructed with TLR7 agonist (92) as the payload. Animal experiment results showed that HE-S2 (150 μg/per mouse) had a very significant anti-tumor effect, most colon cancer mice's tumors basically disappeared after administration, and in the melanin tumor model, the HE-S2 treatment effect is also better than the control group. Importantly, there was no significant weight loss in the HE-S2 treatment group. In addition, although the tumor of mice was shrinking after administration, the expression of PD-L1 on the surface of tumor cells and immune cells in the tumor microenvironment was significantly increased, which may be conducive to improving the targeting of subsequent HE-S2 administration. The novel ADC HE-S2 not only induces a powerful anti-tumor immune response by blocking the PD-1/PD-L1 interaction and activating the TLR7/8 signaling pathway, but also induces upregulation of its targeted antigen PD-L1 through epigenetic regulation and IFN-γ, thereby endows PD-1/PD-L1 blockade with more sensitivity119. Michael N. Alonso's120 team conjugated a TLR7/8 agonist with HER2 antibody, resulting in an ADC drug that recognizes Fcγ receptor-dependent phagocytosis and TLR-mediated activation through tumor antigens, driving myeloid cell tumor killing and subsequent T-cell-mediated antitumor immunity, suitable for systemic administration, safe and effective. In addition, more and more companies are beginning to use TLR agonists for antibody drug conjugation: Bolt's BCC-1001 (ClinicalTrials.gov identifier: NCT04278144), which is used for the treatment of HER2+ positive solid tumors, derived from TLR7/8 agonist conjugation of HER2 has entered clinical studies. Sutro's IADC technology can be used to conjugate antibodies with TLR agonists. Tallac's TRAAC technique was used for site-specific antibody conjugation with TLR9 agonist. ALX Oncology's SIRPα antibody, developed in collaboration with Tallac, can be conjugated with TLR9 agonists. Hengrui Pharmaceutical uses TLR7 agonist, BeiGene uses TLR7/8 agonist, Innovent Bio introduces Bolt technology and uses TLR7/8 agonist for conjugating. Silverback's ImmunoTAC technology selects TLR8 agonist as the payload. These studies provide a novel ADC strategy that enhances the anti-tumor immune response of immune checkpoint blockade therapy. At the same time, this strategy also provides an idea to solve the problem of poor drug quality in the direct use of immuno-agonists.
3.4.2. STING agonists
The stimulator of interferon genes (STING) is a key regulator in the natural immune signaling pathway and has the function of initiating the body's natural immune defense response and promoting the formation of adaptive immunity by T cells. The STING-mediated type I interferon signaling pathway is a major discovery in the field of natural immunity, providing a new target for tumor immunotherapy. Activating the STING pathway can induce the expression and secretion of type I interferons and a variety of other cytokines, activate the natural immune response, promote the anti-tumor immune response, and achieve the purpose of treating tumors. STING agonists are widely used in a variety of research fields. In addition to inducing natural immune responses, they play roles in other DNA or RNA sensor signaling pathways, autophagy, ER stress and apoptosis. With the characteristics of antitumor and immunogenicity, STING agonists have also become a new immunotherapy test drug and vaccine adjuvant. The existing clinical research data have preliminarily proved that the combination of STING agonists and immune checkpoint inhibitors can significantly enhance the efficacy of immunotherapy and is expected to become an innovative therapy121. Non-nucleoside small molecule STING agonists had good stability and could be given orally or by injection in animal models and showed promising anticancer activity. However, since STING proteins are also present in healthy cells, one of the problems that systemic STING agonists need to solve is how to stimulate the anti-tumor immune response without causing an excessive inflammatory response in healthy cells. If these agonists can be further optimized, they have the potential to revolutionize the field of immunotherapy122.
Given that systemic administration of free STING agonists may be limited by toxicity and widespread biological distribution may not be ideal, it can be considered to achieve tumor-targeted delivery of STING agonists through ADC to reduce toxicity. The cyclic dinucleotides (CDNs) (93) have been extensively studied as natural ligands of STING proteins (Fig. 16A). Lichuan Gu's team determined the CBD (CDN binding site) of the porcine STING complex with four CDNs: 2,3 cGAMP; 3,3 cGAMP; c-di-GMP and c-di-AMP. Among them, the asymmetric ligand 2,3 cGAMP has the highest binding affinity with STING (Fig. 16B). Structural analysis of porcine STING CBD 2′,3′-cGAMP complex was carried out and found that the asymmetric unit contains two STING CBD molecules (protomer A and B) that form the dimer. The individual protomers make extensive interactions with each other through the α1, α2, α3, and central β-sheet region, which forms a deep U-shaped cleft. The 2′,3′-cGAMP ligand is located at the cleft using its purines pointing upward and the ribose ring sticking in the downward position (relevant to the membrane). The base, phosphate, and ribose groups all contribute to binding to STING. The purine rings of 2′,3′-cGAMP stack against Tyr167 and Arg238 from two promoters. The phosphate moieties of 2′,3′-cGAMP are recognized by Arg238 through electrostatic interactions and hydrogen bonds. Arg238 also forms hydrogen bonds at the Hoogsteen edge of the nucleoside moieties. Besides the stacking interactions, the adenosine and guanosine moieties have different interaction modes with STING. For both, the interactions involve their Watson‒Crick, Hoogsteen, and sugar edges. All the nitrogen atoms (except the position-9 amino) and the oxygen atom in the purines interact directly or indirectly (water-mediated) with the STING protein. These extensive interactions are consistent with the high binding affinity between ligands and porcine STING CBD. A strikingly distinctive feature of the porcine STING CBD-2′,3′-cGAMP complex is that the two protomers adopt different conformations, resulting in an asymmetric ligand-binding pocket. Consequently, 2′,3′-cGAMP, which is also asymmetric, is held in one well-defined conformation in the ligand-binding pocket. This unique binding mode is in sharp contrast with that seen in previous structures involving STING proteins from other species, where 2′,3′-cGAMP binds the symmetric STING dimer in two alternative modes with the adenosine and guanosine moieties switching positions. In conclusion, the study found that porcine STING uses asymmetrical conformations to bind CDNs. Analysis of this asymmetric conformation reveals the mechanisms of ligand recognition and identification of STING123.
Figure 16.
Design and SAR analysis of STING agonists. (A) Chemical structures of STING agonists 93–96; (B) Binding mode of CDN (93) in complex with STING (PDB code 6A05).
With CDN as ADC payload, CDN and antibody can be conjugated through phosphate ester, sugar ring, and base. SPEROVIE (WO2021046426A1, 2021.03.11) formed ADC drugs by conjugating thiophosphate or formate esters in CDN with PD-1 or CTLA-4 antibodies; Immunesensor (WO2021016204A1, 2021.01.28) elicited amino or mercapto from the ribose of the CDN at position 3′ and then forms an ADC drug by conjugating carbamate or disulfide bond to the antibody; MIT and Millennium (WO2021216572A1, 2021.10.28) innovatively conjured phospholipid molecules to phosphate esters of CDN. The resulting conjugates can also further form liposome structures to improve their ability to fuse with cell membranes. Beijing Xuanyi Pharmaceutical (WO2021178818A2, 2021.09.10) connected the TLR9 agonist CpG-DNA with the CDN to form a fusion molecule that could activate both TLR and STING immune signaling pathways to enhance immune activity. However, diphosphate esters in the CDN structure will cause low bioavailability of drugs. Therefore, with the discovery of benzimidazole compounds, research on STING agonists with non-nucleic acid structure has been started. ADC drugs based on dipolybenzimidazole have also been designed and synthesized. The ADC candidate XMT-2056 obtained by Mersana using STING agonist 94 as the payload is already in preclinical studies (Fig. 16A). It aims to activate the innate immune system by stimulating STING signals in tumor tissue immune cells and tumor cells. The potency of XMT-2056 was increased by more than 100-fold compared to the STING agonist payload. In preclinical models, XMT-2056 showed potent antitumor activity as a single agent in models with both high and low HER2 expression, and its efficacy was enhanced when it was used in combination with multiple approved agents, including trastuzumab, pertuzumab, anti-PD-1 mAb, or DS-8201. Preclinical data suggested that XMT-2056 has the potential to achieve immunologic memory to prolong antitumor activity. In May, the FDA granted the drug orphan drug designation for the treatment of stomach cancer. Mersana plans to initiate a phase I clinical trial of XMT-2056 later this year to investigate its therapeutic potential in a range of HER2-positive tumors, such as breast cancer, gastric cancer, and non-small cell lung cancer; Ono Pharmaceutical of Japan developed a non-nucleic acid STING agonist 95 as the payload, which was conjugated with the linker through the pyrazole ring in the structure of the drug. RYVU is based on the company's development of a class of nitroheterocyclic amines STING agonists 96 that form ADC drugs with antibodies.
3.4.3. Glucocorticoid receptor modulators
Glucocorticoid receptor modulators (GRMs) are commonly used to treat inflammation associated with various diseases. It is possible to minimize the GR reverse transcription effect (considered to be the cause of undesirable side effects) while still maintaining the GR reverse transcription effect (thought to be the cause of the expected anti-inflammatory effect). However, GRM can also cause musculoskeletal, endocrine, and gastrointestinal side effects and other toxicities, limiting their therapeutic effects (especially in long-term use). Using GRM as a payload for immune-type ADCs may be an ideal option, as targeted administration will minimize systemic exposure to the GRM payload, which can provide significant efficacy at doses that do not cause harmful side effects and enable chronic dosing. Such an ADC would combine the efficacy of a monoclonal antibody (targeting an inflammatory target) with a selective glucocorticoid receptor modulator124. Glucocorticoid payloads contain amine functional groups that can be used to bind to lysosomal cleavable antibody linkers. The first reports of GRMs as ADC payloads used the known dexamethasone (97) (Fig. 17A), the GRM ADC antibody component selected α-TNF, and the linker was linked to the C21 hydroxyl group of dexamethasone. ABBV-3373, which AbbVie is developing, is also an ADC drug composed of α-TNF and GRM, that can precisely target the regulation of activated immune cells, modulate TNF-mediated inflammatory signaling pathways, significantly reduce glucocorticoid-related systemic side effects, and potentially treat rheumatoid arthritis (RA). Recently, Adrian D. Hobson125 initiated the α-TNF-GRM ADC project and obtained an α-TNF-GRM ADC with a DAR of 4 using maleimide-Gly-Ala-ALA as the linker. Analysis of P1NP and corticosterone biomarkers showed that the ADC had an adequate therapeutic window between efficacy and adverse effects. In chronic mouse arthritis models, α-TNF-GRM ADCs are more effective than α-TNF monoclonal antibodies and homotype controls with the same GRM payload.
Figure 17.
Design and SAR analysis of glucocorticoid receptor modulators (GRMs). (A) Chemical structures of GRMs 97–102; (B) Binding mode of compound 97 in complex with GR (PDB code 4UDC); (C) Binding mode of compound 98 in complex with GR (PDB code 4UDD).
The group of Victor Gualla126 determined the crystal structure of GR with dexamethasone (97) and 98 (the active metabolite of the prodrug ciclesonide). The structure of GR and dexamethasone (97) used an open conformation, and GR exhibited a higher mutation frequency in the H6–H7 region. An evaluation of PDB cocrystal structures, where ligands are complexed in the ligand-binding domain of the GR (residues 500–777), revealed the structure of desisobutyrylciclesonide complexed with GR (PDB code 4UDC, 4UDD) (Fig. 17B and C). Peter S. Dragovich127 synthetically tested a variety of analogs containing different amine substituent acetal groups. Among them, piperazine compound 99 exhibits cellular activity like dexamethasone (97) and compound 98 (GRE cell, + mTNF, IC50 = 33 nmol/L). However, compound 99 was poorly permeable, which might be due to the presence of alkaline amines in the chemical structure and thus weakens its cell efficacy. Compound 99 is conjugated to the anti-TNF mAb using a dipeptide-based protease (Ala–Ala) cleavable linker, and the resulting conjugate is only weakly active in cell-based evaluation (IC50 = 8.1 μmol/L). Further exploration identified aniline-containing compound 100 that was more potent than compound 99 and had GR binding and cell activity similar to or better than dexamethasone (97) and compound 98 (GRE cell, + mTNF, IC50 = 4.0 nmol/L). Subsequently, by conjugating compound 100 to the anti-TNF monoclonal antibody by a linker containing a protease cleavable dipeptide moiety but lacking an aminobenzyloxycarbonyl spacer group, the resulting conjugate showed strong GR activity in cells containing membrane-bound TNF, while exhibited minimal activity in cells externally lacking this protein (GRE cell, + mTNF, IC50 = 0.44 μmol/L; GRE cell, mTNF, IC50 >50 μmol/L). Intraperitoneal injections of the conjugate at doses of 1, 3, and 10 mg/kg in mice in a collagen-induced arthritis (CIA) model showed a dose-dependent decrease in the degree of paw swelling within 21 days. Importantly, this conjugate has lower toxic side effects. Therefore, the delivery of glucocorticoids by antibody conjugate targeting can minimize unwanted side effects while improving the anti-inflammatory activity of unbound compounds.
William Olson's128 team described the new glucocorticoid class. Their strategy for developing GC conjugates (GC‒ADCs) is to identify the regions in budesonide that can be modified to improve the stability and titer of the conjugates for new free loads. Two modification methods of busonide were then evaluated: First, the substitution of C21 hydroxyl groups with amines or anilines, and second, the substitution of C9 and C6 hydrogen with fluorine. Payloads containing amines and anilines can be conjugated to the antibody via carbamate bonds, which are more stable than carbonate or phosphate bonds provided by hydroxyl groups. The team obtained two lead compounds 101 and 102, by SAR analysis of Budesonide molecules and molecular simulation. Efficacy testing of compounds 101, 102 found that the two compounds were more selective to glucocorticoid receptors (GR) than other nuclear hormone receptors, had better or the same potency as busesonide in cell-based assays, had no cardiotoxicity in hERG, no genotoxicity in Ames, and showed potency in in vivo pro-inflammatory mouse models. In lipopolysaccharide (LPS)-stimulated mice, the release of tumor necrosis factor-α (TNF-α) was significantly reduced. The GC–ADC obtained afterward by histase cleavable linker is highly stable in plasma and specifically releases GCs in antigen-positive cells, suggesting that these novel GCs can act as ADC payloads to treat autoimmune and inflammatory diseases.
Immuno-agonists activate non-specific immune responses when administered systemically, and, in severe cases, can trigger a deadly storm of cytokines. Conjugating immuno-agonists to tumor-targeting antibodies can deliver immune-agonists to the tumor microenvironment and release them locally, alleviating the serious toxicity of systemic administration of immunostimulants and helping to avoid systemic side effects in anti-tumor therapy. However, although the idea of immune-stimulating ADC drugs to achieve an anti-tumor effect by inducing immune mechanisms is very clear, and the preclinical data are relatively good, it is difficult to solve the problem of dose-dependent toxicity in clinical trials. At the same time as the efficacy of the agonists increases, the toxicity will also increase, which makes it difficult to ensure that the efficacy can be improved in a safe and reliable manner. For example, SBT6050, which exhibits good anti-tumor effects in mouse tumor models, is currently not working well in clinical studies due to its toxicity problems. Therefore, the most important consideration in subsequent studies is the balance between toxicity and efficacy. A redesign to weaken the toxicity and improve the delivery window is also highly favorable.
3.5. Novel potential ADC payloads
3.5.1. Bcl-xL inhibitors
Bcl-xL is an anti-apoptotic protein that plays an important role in tumor formation, metastasis, and drug resistance. Apoptosis of cancer cells can theoretically be triggered by drugs blocking the BH3 binding domain on Bcl-xL. However, Bcl-xL is critical for platelet survival, and pan-inhibitors of Bcl-xL may produce platelet toxicity129. Considering Bcl-xL as a target of payloads of ADC drugs may retain the activity of Bcl-xL inhibitors while reducing their toxicity to platelets. Tomoyasu Ishikawa performed a crystal structure analysis of Bcl-xL inhibitors ABT-737 (103) co-crystalized with Bcl-xL, and the results showed that the compound's biphenyl moiety (unit W) was forming lipophilic interactions in a pocket consist of Phe146, Leu130, Leu108, and Phe105 (western region; h1 H2), while the alkylsulfonamide moiety (unit E) of the compound was forming lipophilic interactions in another pocket composed of Phe97, Tyr195, and Tyr101 (eastern region; h4) (Fig. 18). In unit E, three aromatic rings form intramolecular π‒π interactions, forming a unique rigid U-shaped conformation. This conformation may enhance the intermolecular binding of ABT-737 (103) to Bcl-xL. In addition, the structure‒activity relationship of a series of small molecule inhibitors was studied, and it was found that the inhibitory effect of unit E on Bcl-xL was more important than unit W, especially ring B. The analysis of crystal structure and structure‒activity relationship will provide the basis for designing more efficient inhibitors for ADC payload identification130 In 2017, AbbVie first conjugated Bcl-xL inhibitor 104 as a payload to an anti-B7H3 antibody in the form of an ADC to obtain ABBV-155 (ClinicalTrials.gov identifier: NCT03595059) targeting EGFR, which is still in clinical trials (WO2017214301A1, WO2017214282Al).
Figure 18.
Design and SAR analysis of Bcl-xL inhibitors. (A) Chemical structures of Bcl-xL inhibitors 103, 104; (B) Binding mode of ABT-737 (103) in complex with Bcl-xL (PDB code 2YXJ).
3.5.2. NAMPT inhibitors
Niacinamide phosphate ribose transferase (NAMPT), which converts nicotinamide into nicotinamide mononucleotide, is a rate-limiting enzyme that controls the concentration of NAD+ within cells. When NAMPT is suppressed, NAD + levels drop below the level required for metabolism, leading to an energy crisis and eventually, cell death. Although NAMPT inhibitors are potent cytotoxic molecules, their use as chemotherapeutic drugs in cancer treatment is hampered by their toxicity. Several NAMPT inhibitors such as FK-866 (105) (Fig. 19A) have discontinued the clinical trials due to dose-dependent toxicity. However, NAMPT inhibitors have a simple structure and high potency, and their activation mechanism is different from the current ADC payloads, so NAMPT inhibitors can be used as ADC payloads with good activity and tolerance, which is expected to improve the treatment window of NAMPT inhibitors and then be applied to the clinical environment131. Chris Tse and his teams19 identified novel nonsubstrate NAMPT inhibitors with robust preclinical efficacy and pharmacokinetics properties that enable oral dosing. The lead molecule in this series, A-1293201 (106), contains an isoindoline “head group”, which can effectively inhibit recombinant human NAMPT. Examination of the interactions between A-1293201 and NAMPT by X-ray crystallography revealed several distinct sites for cooperative interactions (Fig. 19B). The sites include the PRPP-binding subsite A, which is hydrophilic in nature and unoccupied by nicotinamide, the nicotinamide binding subsite B, where important pi-stacking and hydrogen bond interactions with nicotinamide are made, the tether subsite C, which is narrow, lipophilic, and not occupied by bound nicotinamide, and the distal opening subsite D, which widens and provides several bound water molecules and hydrophobic surfaces. Similar to FK866, the isoindoline urea portion of A-1293201 is engaged in important pi-stacking interactions in subsite B as well as hydrogen bonds and hydrophobic interaction in subsite C. In contrast to FK866 as well as recent second-generation NAMPT inhibitors, the isoindoline urea compounds are the first known as potent nonphosphoribosylated NAMPT inhibitors. The novel NAMPT inhibitor 107 designed by Carl Uli Bialucha132 is effective in the nmol/L range for multiple cell lines including NCI-H526, MDA-MB453, and NCI-N87. The structure‒activity relationship shows that pyridine nitrogen can be phosphorylated by NAMPT enzyme and is an important part of the pharmacophore. In addition, the whole 3-pyridine (S,S) cyclopropyl carboxamide group is essential for high levels of cellular activity. The R3 region can tolerate a variety of substituents, and the ethyl substituent compound 108 is obtained by replacing a more rigid unsubstituted phenyl ring, which has a similar effect on A2780 and CORL23 cell lines to compound 107. Benzene rings can be further derived in positions to provide the best carrier for ligation, and the addition of piperazine substituents to the phenyl ring's para position is well-tolerated compound 109. The free secondary amines on the piperazine group can bind to the antibody as the conjugation point of the linker. Phenyl piperazine substitution at R3 and substitution of R1 and R2 to F and H, respectively, resulted in a slightly improved analog 110 that was effective against C-Kit and HER2-expressing cell lines at nmol/L concentration. The anti-C-Kit conjugates obtained using non-cleavable linkers with payloads 110 not only have low aggregation, but also have optimal anti-CKIT activity (IC50 is 9 and 40 pmol/L, respectively), and are shown to be targeted in the GIST-T1 xenograft model of c-Kit-positive gastrointestinal stromal tumors. The payload for ADCs needs to contain active heteroatoms to bind the cleavable linker, while FK-866 lacks an amino, hydroxy, or mercapto functional group. To obtain a payload that can be easily integrated and retain potency, Peter D. Senter133 prepared a series of FK-866 analogs with aniline substitution at different locations on the aromatic tail. The compounds were further evaluated for cytotoxicity against L540cy, A549, and HepG2 cell lines. Compound 111 displayed similar potency to FK-866 in both assays. The crystals of NAMPT and compound 111 showed that the newly incorporated aniline group interacts with the backbone amide of Glu376 via a hydrogen bond, potentially offsetting any desolvation penalty for the addition of a polar functional group. Compound 112 prepared with a potent inhibitor chs-828 found in the pyridyl cyanoguanidine group and compound 113 prepared with pyridyl square amide groups retain a strong binding to NAMPT and have strong cytotoxicity. These compounds have been advanced into the evaluation as ADC drug payloads. At well-tolerated doses, significant levels of activity were shown in several tumor models.
Figure 19.
Design and SAR analysis of NAMPT inhibitors. (A) Chemical structures of NAMPT inhibitors 105–113; (B) Binding mode of A-1293201 (106) in complex with NAMPT (PDB code 5U2M).
3.5.3. Carmaphycins
Proteasome activity inhibitors are an emerging class of anticancer drugs that are super cytotoxic to certain cancer cells, and the epoxyketone proteasome inhibitor carmaphycins is a representative of this class. Carmaphycins A (114) and B (115) isolated and are identified from marine cyanobacteria Symploca sp., and their α,β-epoxyone are directly attached to methylthionine sulfoxide or methylthionine sulfone, inhibiting the β5 subunit of the proteasome at low nanomolar concentrations, effective against many cancer cell lines (Fig. 20). However, due to their poor selectivity, they often exhibit toxic side effects. Using a highly toxic carmaphycins derivative as an ADC payload maintains the desired potency and achieves better tolerance134. Carmaphycins can be divided into four distinct parts (P1–P4). William H. Gerwick20 synthesized the first generation analog 116 with amine groups at the distal end of P4 by substituting 6-amino hexanoic acid chain, which significantly reduced the killing effect on NCI-H460 cells (IC50 = 860 nmol/L). The second-generation carmaphycins analogs contain a 2-aminoethyl homocysteine residue at P2. The straight-chain alkyl chain is substituted with an aromatic phenyl or pyridine ring to give an analog 117, which has a lower cell-killing effect on HCT116 and NCI-H460 than carmaphycins B (115), because the fatty amine of P2 is alkaline and positively charged at physiological pH, reducing its cell permeability. To reduce the alkalinity at P2, the team replaced the P2 methyl sulfone moiety of carmaphycin B (115) with 4-thioaniline or 4-sulfonylaniline to obtain compounds 118 or 119. Compound 119 is more effective than 118, which means less alkaline amine potency is better. After that, an analog 120 containing two leucine-epoxyketone drug clusters at P1 and P2 was synthesized, and its IC50 in HCT116 and NCI-H460 cell lines was 0.2 and 5.4 nmol/L, respectively. The epoxyketones at P1 were critical to the potent cytotoxic activity of compound 120. Analog 121 contains aniline-containing side chains of P3, and the IC50 for SKBR3 cells is 21 nmol/L. Placing the aniline functional group in the P4 position in the analog 122 is more effective for cell growth inhibition compared to compound 121. Compound 119 in the series of analogs maintained high cellular potency, but its current conjugated ADC did not show superior cell-killing ability for the cancer cell lines tested compared to free antibodies.
Figure 20.
Design of carmaphycins analogs 114–122.
3.6. New strategy guided ADC payloads
3.6.1. ADC with PROTAC molecules as payloads
The main challenge in the development of ADCs is related to dose-limiting toxicity (DLT), which indicates the difficulties to balance the efficacy of drug therapy with off-target toxicity. The dose of the drug that ADC actually delivers to the tumor is very small, which means that the drug molecule must be extremely cytotoxic, although this can lead to toxic side effects. PROTACs are a group of bifunctional compounds consisting of a ligand targeting a protein of interest (POI), a ligand of the E3 ligase and a linker. PROTACs bring the POI and E3 ligase closer together, labeling the POI with ubiquitination which will then be degraded by the proteasome. PROTAC is catalytic and therefore effectively degrades the protein of interest at lower doses135. PROTACs may be ideal payloads for ADCs. The antibody PROTAC conjugations formed by conjugating antibodies and PROTAC molecules can specifically degrade target proteins in specific cells, realizing the selectivity of PROTAC technology at the cell or tissue level. Antibody‒PROTAC conjugates combine the catalytic properties of PROTAC with the tissue specificity of ADCs, thereby overcoming the traditional limitations of targeted degraders and ADCs, with great potential for targeting novel targets.
Connecting a PROTAC molecule 123 that targets BRD4 protein to a HER2 antibody can form an antibody PROTAC conjugate with a molecular weight of about 150 kD, with an average of 4 PROTAC molecules on each antibody molecule (Fig. 21). The antibody PROTAC conjugate can effectively degrade the BRD4 protein in HER2 high-expression cells SK-BR-3 and has no degradation effect on the BRD4 protein in HER2 low-expression cells MCF-723. BRD4 inhibitors 124 are conjugated to interchain Cys residues of mAb targeting HER2 using maleimide-based conjugation, resulting in ADC conjugates that reduce BRD4 levels in BT-474 (HER2-positive) cell lines. Two ADCs degrading ERα have also recently been described in the patent literature. The first is to attach the VHL-based PROTAC molecule 125 to the mAb targeting HER2 via the carbonate section; The second ADC is connected to the VHL-binding region of the same PROTAC using an enzyme-containing cleavable pyrophosphate linker. In MCF7-neo/HER2 cells, both conjugates exhibited efficient antigen-dependent ERα degradation, and both conjugates showed acceptable results in mouse stability and pharmacokinetic tests. PROTAC molecule 126, which has a CRBN as E3 ligase and targets the TGFβR2 protein, can be conjugated to an anti-HER2 antibody via a non-cleavable linker. In HEK293 cells transfected with HER2 receptors, ADCs of 0.5 and 1.0 μmol/L degraded the TGFβR2 protein after 24 and 48 h. In addition, a single IV injection of 1 mg/kg of ADC using PROTAC molecule 127, a VHL-based degradable BRM protein, as a payload achieved a strong, antigen-dependent reduction of BRM protein in BJAB lymphoma xenograft tumors known to express high CD22 surface receptors. This conjugate exhibits the desired biological activity in vivo and further diversifies the target protein that can be successfully modulated by ADC mode, expanding the possibility of targeting PROTAC to cells expressing this surface protein136.
Figure 21.
Chemical structures of PROTAC molecules 123–127.
Although the antibody‒PROTAC conjugate field is still in its infancy, a diverse group of such entities has already been created and its members subsequently have shown to exhibit meaningful in vitro and/or in vivo biological activities. Many of these new entities employed higher payload loadings (DAR values of six) relative to the majority of known cytotoxic ADCs (DAR = two to four), but it remains to be determined whether such increases are generally required for typical antibody–PROTAC conjugate applications. Multiple examples of antigen-dependent activity were achieved both in vivo and in vitro with the described conjugates, and these outcomes demonstrated the ability of the antibody‒PROTAC conjugate modalities to deliver their respective PROTAC payloads to specific tumors and/or cells of interest. Given these promising initial results, antibody‒PROTAC conjugates appear well-positioned for future growth and application. It is expected that in the future, antibody–PROTAC conjugates will be applied to tissues or living organisms, providing new ideas for drug therapy or biological research.
3.6.2. ADC with NIR-PIT drugs as payloads
Near-infrared photoimmunotherapy (NIR-PIT) drugs are generally composed of tumor-specific monoclonal antibodies that target tumors and a photoactivating chemical through connecting linkers, which are essentially ADC drugs. NIR-PIT drugs can form a new targeted anti-cancer platform with devices that irradiate infrared light to tumor sites. The platform enables antibody-mediated targeted delivery to achieve a high degree of tumor specificity, while using infrared light to activate the biophysical mechanism of the drug to accurately induce rapid death of cancer cells without harming the surrounding normal tissues. The NIR-PIT drug-induced polyclonal immune response can eliminate tumor cells that survive the first step of NIR-PIT drugs, even if the NIR-PIT drugs are insufficient, unevenly delivered, or dosed insufficiently due to the uneven expression of target antigens, as the subsequent second-step polyclonal immune response will also kill residual tumor cells. In addition, NIR-PIT drugs can also be used as a useful supplement to pre-existing PD-1 monoclonal antibodies, PD-L1 monoclonal antibodies, or CTLA-4 monoclonal antibodies, enhancing their tumor immune response137. The payload in NIR-PIT drugs is not a cytotoxic substance, but a water-soluble phthalocyanine derivative (e.g., the silicon phthalocyanine derivative IR700 (IRDye700DX) (128)) (Fig. 22A). IR700 is a small, photoactivated molecule that can be excited by near-infrared light and has no phototoxicity or biotoxicity. When the antibody binds to the tumor surface antigen, under the stimulation of near-infrared light, IR700 undergoes a photo-induced ligand release reaction, releasing the hydrophilic side chains, resulting in a significant increase in the hydrophobicity of the remaining part. This, in turn, destroys cell membranes, triggering rapid and highly selective immunogenic cell death (ICD) that targets cancer cells (Fig. 22B). While directly killing cancer cells, NIR-PIT-induced ICD can lead to rapid maturation of immature dendritic cells nearing death of cancer cells, initiate the host anti-cancer immune response, promote the re-formation of CD8-positive T cells against antigens released by dead cancer cells, and further amplify the therapeutic effect of NIR-PIT138.
Figure 22.
Design and analysis of IRDye700DX (128). (A) Chemical structures of IRDye700DX (128); (B) The mechanism of NIR-PIT ADC.
Approved by PMDA acceleration in 2020 under the trade name Akalux, Rakuten Medical's Cetuximab Sarotalocan is an antibody–conjugated drug composed of cetuximab and the water-soluble silicon phthalocyanine derivative IRDye700DX, which targets epidermal growth factor receptors for the treatment of unresectable locally advanced or locally recurrent head and neck cancer, making it the world's first approved light immunotherapy drug. After 24 h of administration, the drug can be specifically aggregated on the surface of EGFR-positive tumor cells. The use of 690 nm wavelength near-infrared light to illuminate the tumor site induces Cetuximab sarotalocan to kill cancer cells and activate the immune response22. The fastest progress in researching NIR-PIT drugs is Rakuten Medical's ASP-1929 (also known as RM-1929). ASP-1929 is a conjugated drug of cetuximab and IR700 that targets the epidermal growth factor receptor EGFR and can be used to treat unresectable locally advanced or recurrent head and neck cancer. In addition to ASP-1929, Rakuten Medical also had a NIR-PIT drug that demonstrated excellent antitumor activity in preclinical studies in combination with PD-1 monoclonal antibody in advanced tumor animal models. A few of other NIR-PIT drug studies, including 131I (PcMAb) (CD38 monoclonal conjugated NIR-PIT drug), TROP-2-IR700, ramucirumab-IR700, conjugate AvIR-mediated PIT, anti-TF antibody 1849-ICG conjugate, MN-14-700DX, Avelumab-IR700 and the like are all taking IR700 as payloads.
As an “unpopular track” in the field of ADC, the clinical and market prospects of optoimmune ADCs still need to undergo the test of time. However, the development of new targets and new photoactivated effector molecules based on relevant principles may become a new breakthrough point in the ADC track.
3.6.3. ADC with dual payloads
With the advent of ADC therapy resistance, binding one antibody to two or even more different cytotoxic payloads provides an attractive option for the development of next-generation ADCs. To demonstrate the possible benefits of the dual-payloads drug ADC, Levengood et al.24 prepared a class of ADCs containing two different tubulin polymerization inhibitors in 2017. The team linked MMAE and MMAF, which have different physical and chemical properties, to the same anti-CD30 antibody to perform complementary anticancer activities. This work clearly demonstrates a potentially effective approach to the construction of a potent two-drug ADC and how delivery of multiple cytotoxic payloads improves ADC activity. Later, Kumar et al.139 developed a heterogeneous trifunctional linker that conjugated MMAE and PBD dimer payloads to produce a dual-payload drug ADC. Nilchan et al.140 used a dual antibody conjugating method to generate a dual-payload ADC containing the targeted tubulin payload MMAF and DNA topoisomerase II inhibitor PNU-159682 by specific conjugation of HER2 antibodies at the engineering selenized cysteine and cysteine residue sites. PNU-159682 caused S-phase cell cycle arrest due to its DNA-damaging activity, while MMAF simultaneously inhibited tubulin polymerization and caused G2/M-phase cell cycle arrest. The dual-drug ADC showed selective and potent cytotoxicity against HER2-expressing cell lines and exhibited dual mechanisms of action consistent with the attached drugs.
ADC drugs often encounter problems associated with heterogeneity within tumors. Breast cancer, for example, is usually made up of multiple cells with different gene expression profiles, and its heterogeneity is a major factor in drug resistance, recurrence, and metastasis after chemotherapy. To solve this problem, Kyoji Tsuchikama efficiently constructed a series of dual-payload ADCs with a clear drug‒antibody ratio (DAR) (DAR combinations of 2+2, 4+2, and 2+4, respectively) by chemical enzyme linking method, and the flexibility of DAR adjustment is conducive to fine-adjustment of the physicochemical properties, efficacy and toxicity of the ADC according to the disease target and payload combination (Fig. 23). Among them, the MMAE/MMAF dual-payload ADC with DAR of (4+2) is more effective than the MMAE single-drug ADCs with DAR of 4 or 6, and has specific HER2 cell killing ability, ideal pharmacokinetic properties, minimal inflammatory response, and marginal toxicity at therapeutic doses. In addition, in the mouse model of JIMT-1/MDA-MB-231 mixed xenograft with HER2 heterogeneity and increased resistance to TDM1, dual-payload ADCs had greater therapeutic effects and survival benefits than the two single-agent combinations. The reason may be that the stronger MMAF molecules kill JIMT-1 cells, helping the conjugated MMAE molecules to effectively exert a bystander effect on neighboring MDA-MB-231 cells. Importantly, this significant efficacy cannot be achieved through a 1:1 combination of MMAF ADC and MMAE ADC alone. The enhanced efficacy of the dual-payload ADC was also demonstrated in a hCC1954-TDR xenograft mouse model of another type of breast cancer with low HER2, intra-tumor heterogeneity, and T-DM1 resistance. The results highlight the potential of dual-payload ADCs to overcome breast cancer heterogeneity and drug resistance, suggesting that simultaneous delivery of two payloads with different drug properties is a promising approach for ADC-based treatment of other refractory cancers with heterogeneity and drug resistance141. Using their Synthemer platform, the Cambridge researchers designed a dual-payload ADC that combines both auristatin F hydroxypropylamide (AF-HPA) and DNA monoalkyllating agent (I-BiP), exhibiting strong cytotoxicity and can exert both mechanisms of action. However, while multi-payload ADC drugs can improve efficacy and reduce the possibility of drug resistance, the side effects caused by their treatment are also worth considering. For example, Upifitamab Rilsodotin (UpRi, XMT-1536), a novel ADC drug developed by Mersana that targets NaPi2b, is conjugated to the auristatin F-hydroxypropyl amide cytotoxic drug through a cleaved linker of the anti-HER2 monoclonal antibody HT-19, DAR to 12. XMT-1536 has extremely strong anti-tumor activity, especially for cancers with low expression of HER2. However, the interim phase 1 clinical data of ovarian cancer showed that XMT-1536 could cause serious treatment-related side effects, with an incidence of up to 48% (47/97). The most common side effects were gastrointestinal obstruction (7%), and the incidence of fever, pneumonia, and abdominal pain was 5%. Although Mersana tries to reduce the dose, the range of treatment window options is narrow, and the future is not optimistic. Therefore, for multi-payload ADC, safety control is an important issue that needs to be paid attention to when multi-class payload assembly is carried out. The key is to find and deal with the cause of the narrow treatment window. Future research should also continue to move towards a balance between efficacy and toxic side effects.
Figure 23.
The mode pattern of dual-payloads drug ADC (The red and green shapes mean different payloads).
3.6.4. Payloads of PDC
Peptide drug conjugates (PDCs) are a novel targeted therapy consisting of the linker, homing peptide, and cell-toxic payloads142. Compared to ADC drugs, PDC drugs have advantages such as small molecular weight, strong tumor penetration, low immunogenicity, large-scale synthesis by solid-phase synthesis, low production cost, relatively good pharmacokinetics, and relatively uniform batch products. They are the next generation of targeted anti-tumor drugs after small molecule targeted drugs, monoclonal antibodies, and ADCs. However, the application of PDC as a therapeutic drug also has certain limitations, such as low oral bioavailability, short half-life, incomplete cleavage of some payloads leading to significantly reduced bioactivity compared to the prototype drug, and possibly inferior targeting compared to ADCs143, 144, 145. The mechanism of action of PDC varies depending on the linker and homing peptide. One is that PDC is internalized and releases the payload inside the cell. The other is that PDC is cleaved in the tumor microenvironment and the payloads are internalized to exert its effect (Fig. 24A). Most payloads can not only kill tumor cells with high expression of the target but also kill surrounding tumor cells with low expression or no expression of the target through bystander effects in the tumor microenvironment146,147. Peptides can affect the efficiency of PDC internalization. The ideal PDC peptide should have strong target binding affinity, high stability, low immunogenicity, high internalization rate, and a relatively long plasma half-life. The choice of linker is one of the key factors in PDC design. The linker needs to balance stability and release efficiency. Cleavable linkers have higher release efficiency, while non-cleavable linkers are more stable. The toxicity and physicochemical properties of the payload can directly affect the drug's ability to kill tumors and thus affect its efficacy. The ideal payload should have high cell toxicity (usually low IC50), low immunogenicity, high stability, appropriate hydrophobicity, and good solubility148, 149, 150.
Figure 24.
Key structures and mechanisms of action of PDCs. (A) The general mechanism of action of PDCs; (B) Chemical structures of the common PDC payloads.
The PDC payloads can be divided into chemical drugs (Fig. 24B), protein drugs, and peptide drugs. Among them, chemical drugs and protein drugs are more common. Chemical drugs represented by DM1(2), MMAE (8), KSP inhibitors (45), camptothecin (54), doxorubicin (129), paclitaxel (130), methotrexate (131), daunorubicin (132), and gemcitabine (133) are commonly used. Protein drugs mainly include interferon and tumor necrosis factor151, 152, 153, 154. Radioisotopes are also commonly used as payloads for PDCs155. When PDCs are used for cancer diagnosis, they can be labeled with positron-emitting radioisotopes (fluorine-18 (18F), copper-64 (64Cu), and gallium-68 (68Ga)) to generate PET imaging. By binding to targeted receptors on tumor cells, malignant tissues can be accurately located. Radioisotope-labeled PDCs can also be used for treatment. Peptide receptor radionuclide therapy (PRRT) targets and tissue-specific radiation of tumor cells based on their overexpressed receptors. The most commonly used radioisotopes are indium-111 (111In), yttrium-90 (90Y), and lutetium-177 (177Lu).
As of April 2023, there are only two PDC drugs approved for marketing worldwide, Lutathera® and Pepaxto®156,157. Lutathera® is a PDC targeting the growth hormone-inhibiting hormone receptor developed by Advanced Accelerator Applications S.A, a subsidiary of Novartis. After entering the cell, it releases the radioisotope 177Lu to damage tumor cells through radiation. It was first approved by the European Medicines Agency (EMA) in September 2017. In patients with advanced neuroendocrine tumors, Lutathera® has a longer progression-free survival and higher response rate compared to high-dose long-acting octreotide. It was approved by the FDA on January 26, 2018. Pepaxto® is a PDC targeting aminopeptidase developed by Oncopeptides, consisting of the DNA alkylating agent Melflufen (134) covalently linked to a targeting peptide of aminopeptidase. Melflufen has high lipophilicity and can be rapidly absorbed by myeloma cells. Once inside the cell, the conjugated peptide of Melflufen is immediately cleaved by aminopeptidase, releasing the hydrophilic alkylating agent Melphalan (135), causing DNA damage in tumor cells and leading to tumor cell death. Pepaxto® was first approved by the FDA in February 2021, in combination with dexamethasone, for the treatment of adult relapsed or refractory multiple myeloma. However, in October of the same year, Oncopeptides announced that it would withdraw Pepaxto® from the US market mainly due to an increased mortality rate in the confirmatory OCEAN study. In August 2022, Pepaxto® was approved by the EMA. Currently, there are a total of 17 PDC drugs in clinical development worldwide, with 3 in phase III, 9 in phase II, and 5 in phase I158. The main targets are tubulin and CA9, with MMAE (8) being the main payload. PDC is an equivalent antibody–drug conjugate that overcomes some of the limitations of ADC and has many unique advantages. Although there are currently few PDC drugs on the market, there have been cases of withdrawal due to serious safety issues. However, with the resolution of related technical issues and breakthroughs in supporting technologies, it is believed that PDC drugs will also become a hot spot for research and investment in the crowded racetrack in the next decade.
4. Conclusions
ADC drug development has broad prospects, since they integrate specific antibodies for selective tumor cell targeting, conjugate a wide range of potent cytotoxic payloads, and employ diverse linker technologies to generate effective cancer therapeutics. From a technical point of view, ADC drugs have undergone three generations of changes so far, and there have been breakthroughs in antibodies, drug carriers, connectors, etc., especially in the use of payloads.
Although the specificity and cytotoxicity of the new generation ADC payloads have shown significantly improved characteristics than those in the early stages, the current payloads still have some limitations. First, they generally have limited solid tumor permeability and toxicity, which limits their applications to solid tumors. Second, some tumors are insensitive to current ADC drugs. Third, the complexity of payload pharmacokinetics, tumor targeting, and insufficient release can all affect the efficacy of ADC drugs. Fourth, like other antitumor drugs, ADC drugs can also develop resistance. Finally, earlier payload selection tends to be toxic but effective drugs, but their higher toxicity limits the systematic application of the payload. Although the antibody–drug conjugates show effectiveness in treatment, the combination with highly toxic payloads is not necessarily excellent. Therefore, other types of payloads such as RNA inhibitors, immuno-agonists, and apoptosis-promoting Bcl-xL inhibitors are being developed. Especially with the approval of the FDA for immune checkpoint blockade therapy, the era of tumor immunotherapy began. Immuno-agonist ADCs have also been developed, which can use antibodies to deliver immune agonists to the tumor microenvironment and release them locally, alleviating the serious toxicity problem with the systemic administration of immune agonists. Immuno-agonists (such as Toll-like receptors, STING agonists, etc.) are also becoming more and more popular in ADCs as new payloads, and immune-modulating ADCs are expected to further enhance the anti-tumor immune response of immune checkpoint blocking therapy. Although the immune ADC is still in the initial stage of development and has not yet achieved satisfactory clinical results, the immune-modulating ADC has the potential to greatly broaden the treatment window of the ADC, improve its anti-tumor activity, and have ideal safety and efficacy. It is believed that the design of ADCs that target the tumor microenvironment through immunomodulatory payload modification is still a hot topic in the field of future research, especially the use of payloads that are not directly toxic to cells (such as payloads that can guide the immune system to attack cancer cells). In addition, using PROTAC molecules as ADC payload can fully combine the catalytic properties of PROTAC with the tissue-specific advantages of ADC, and overcome the traditional limitations of targeted degradation agents and ADC (such as drug resistance, toxic side effects, etc.). Given the promising results, PROTAC‒ADC is an important direction for future ADC drug development. The antibody binding payloads also include protein-derived enzymes from plants (e.g., saponin, ricin A chain) or bacterial toxins (PE, pseudomonas exotoxin, DT, diphtheria toxin) that induce cell death by catalytically irreversibly inhibiting protein synthesis159, 160, 161, 162. Novel payload categories also include oligonucleotides, siRNA, proteins, and more. In order to solve the shortcomings of traditional ADC payloads, in addition to developing and modifying traditional and novel potential payloads, many new strategies are emerging for ADC design. For example, the design of ADC drugs by combining the same antibody with different payloads that have unrelated mechanisms can achieve synergetic effects by minimizing the possibility of resistance, and simultaneously targeting different targets, which opens a new approach for the treatment of other refractory cancers with heterogeneity and drug resistance, which is particularly important for solid tumors. Also, when designing hybrid payloads, the balance between efficacy and toxicity should be emphasized. In addition, by using antibody-mediated targeted delivery, the ADCs composed of photoactivating chemicals as payloads and specific monoclonal antibodies can achieve high tumor specificity and can accurately induce the rapid death of cancer cells, avoid harming the surrounding normal tissues by using the laser-activated biophysical mechanism, and realize the requirements of precise treatment, which is also a development direction worth exploring and looking forward to in the future.
Although ADCs have evolved to date, the types of payload skeletons available to them have not increased, and most cytotoxic drugs are derivatives of the precore backbone. Only a limited number of highly cytotoxic natural compounds, derivatives, or synthetic analogs currently have the potential to be used as payloads for ADCs and enter the clinic. Although novel ADC payloads and new strategies to guide the construction of ADCs continue to emerge, these are still in the initial stage of development, and the future direction needs to be verified by more experiments. Therefore, in the next generation of ADCs, it is necessary to develop structurally novel high-efficiency payloads (e.g., NAMPT inhibitors with simple structure, high activity, and special mechanism of action) with multiple mechanisms of action, higher efficacy, and fewer side effects by screening natural product libraries, chemical synthesis, and direct derivatization of natural products to improve the activity and its tolerance. However, new scaffold discoveries are challenging and require a lot of work in areas such as natural product identification, high-throughput screening, or structure-based drug design. In contrast, structural modifications to approved or well-studied payloads can identify novel payloads more quickly. After obtaining the parent nuclear compound, the structure–activity relationship and crystallographic techniques should be fully utilized to explore the binding mode of the target with the parent nuclear compound, and the effective action site and the effective functional group of the compound. Only then will the subsequent optimization and transformation of the parent nuclear compound be efficiently carried out. When modifying the payload, adjusting its physicochemical properties, especially its polarity, can overcome some problems faced in the preparation and application of ADCs. Connecting too many hydrophobic payloads can alter the conformational stability of the antibody, increase its tendency to aggregate and precipitate, and ultimately affect its maximum DAR, plasma stability, bystander effect, and more. Hydrophobic payloads can easily penetrate the cell membrane and kill surrounding antigen-negative tumor cells through the bystander effect while hydrophilic payloads diffuse out of cancer cells at a slower rate. In addition, a major problem faced by most ADCs is the large toxic side effects, among which the toxic side effects caused by the payload are mainly reflected in the damage to rapidly proliferating healthy cells, such as the reduction of lymphocytes and gastrointestinal reactions. Therefore, the payload with small molecular weight, good tissue penetration, and a short half-life can be selected, which can be rapidly enriched at the target position after release and quickly perform the function of killing cancer cells. At the same time, the drug molecules in the blood can be rapidly removed, which is conducive to reducing the systemic toxic side effects. It is also possible to mutate important sites of the target, and then modify and screen out more well-tolerated compounds.
In summary, there are some differences between novel ADC payloads and traditional payloads: 1) The traditional ADC payloads mostly choose small molecules with strong pharmacological effects but also have relatively large toxic side effects, such as PBD, while the novel payloads tend to choose small molecules with moderate pharmacological effects and toxic side effects, such as Dxd. 2) The traditional ADC payloads mainly consist of microtubule inhibitors and DNA damaging agents, while the novel ADC payloads have a richer variety of target types, more choices, and different mechanisms of action compared to traditional payloads. For example, using immune stimulants as ADC payloads. 3) The structure of traditional ADC small molecule payloads is generally complex and difficult to synthesize. The novel ADC payloads tend to choose small molecules with simple structures and low molecular weights, greatly reducing the difficulty of synthesis and expanding the application market of ADC payloads. 4) The traditional ADC payloads generally use a single type of small molecule compound directly linked to the antibody through a linker, while the new type of ADC can combine different types of payloads, achieving a 1+1>2 effect. In addition to traditional small molecule compounds, the novel payloads also select novel types such as PROTAC and photoactivated molecules as effective payloads. Although ADC drug has been developed so far and their efficacy has been greatly improved, there are still some problems such as drug resistance, adverse reactions, and insufficient stability in the blood waiting to be solved. The payload occupies an important position in the efficacy of ADC drugs, so we summarized the ADC small molecule payloads and its future trend in combination with the relevant crystal structure and structure‒activity relationship research, in order to provide more new references for the future development directions of ADC drug payloads and the subsequent optimization and transformation, so as to construct more ADC drugs to improve their therapeutic effect, overcome drug resistance, reduce adverse reactions, and promote the development of ADC drug research.
Acknowledgments
This work was provided by the National Natural Science Foundation of China (82073318), the Fundamental Research Funds for the Central Universities (SCU2022D025, 0082604151345, China), and Sichuan Science and Technology Program Projects (2019YFS0003, China) to Yuxi Wang. Additional support was provided by the University of Tennessee College of Pharmacy Drug Discovery Center to Wei Li.
Author contributions
Yuxi Wang conceived and supervised the project. Zhijia Wang, Hanxuan Li, and Lantu Gou reviewed the literature and drafted the manuscript. Wei Li provided additional supervision and revised the manuscript. All authors approved the final manuscript.
Conflicts of interest
The authors have no conflicts of interest to declare.
Footnotes
Peer review under the responsibility of Chinese Pharmaceutical Association and Institute of Materia Medica, Chinese Academy of Medical Sciences.
Contributor Information
Wei Li, Email: wli@uthsc.edu.
Yuxi Wang, Email: yuxiwang@scu.edu.cn.
References
- 1.Weingart S.N., Zhang L., Sweeney M., Hassett M. Chemotherapy medication errors. Lancet Oncol. 2018;19:e191–e199. doi: 10.1016/S1470-2045(18)30094-9. [DOI] [PubMed] [Google Scholar]
- 2.Zraik I.M., Hess-Busch Y. Management of chemotherapy side effects and their long-term sequelae. Urologe. 2021;60:862–871. doi: 10.1007/s00120-021-01569-7. [DOI] [PubMed] [Google Scholar]
- 3.Ponziani S., Di Vittorio G., Pitari G., Cimini A.M., Ardini M., Gentile R., et al. Antibody‒drug conjugates: the new frontier of chemotherapy. Int J Mol Sci. 2020;21:5510. doi: 10.3390/ijms21155510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.McKertish C.M., Kayser V. Advances and limitations of antibody drug conjugates for cancer. Biomedicines. 2021;9:872. doi: 10.3390/biomedicines9080872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chau C.H., Steeg P.S., Figg W.D. Antibody‒drug conjugates for cancer. Lancet. 2019;394:793–804. doi: 10.1016/S0140-6736(19)31774-X. [DOI] [PubMed] [Google Scholar]
- 6.Strebhardt K., Ullrich A. Paul Ehrlich's magic bullet concept: 100 years of progress. Nat Rev Cancer. 2008;8:473–480. doi: 10.1038/nrc2394. [DOI] [PubMed] [Google Scholar]
- 7.Trail P.A., Willner D., Lasch S.J., Henderson A.J., Hofstead S., Casazza A.M., et al. Cure of xenografted human carcinomas by BR96-doxorubicin immunoconjugates. Science. 1993;261:212–215. doi: 10.1126/science.8327892. [DOI] [PubMed] [Google Scholar]
- 8.Shen W.C., Ballou B., Ryser H.J., Hakala T.R. Targeting, internalization, and cytotoxicity of methotrexate-monoclonal anti-stage-specific embryonic antigen-1 antibody conjugates in cultured F-9 teratocarcinoma cells. Cancer Res. 1986;46:3912–3916. [PubMed] [Google Scholar]
- 9.Johnson D.A., Laguzza B.C. Antitumor xenograft activity with a conjugate of a Vinca derivative and the squamous carcinoma-reactive monoclonal antibody PF1/D. Cancer Res. 1987;47:3118–3122. [PubMed] [Google Scholar]
- 10.Dillman R.O., Johnson D.E., Shawler D.L., Koziol J.A. Superiority of an acid-labile daunorubicin-monoclonal antibody immunoconjugate compared to free drug. Cancer Res. 1988;48:6097–6102. [PubMed] [Google Scholar]
- 11.Tolcher A.W., Sugarman S., Gelmon K.A., Cohen R., Saleh M., Isaacs C., et al. Randomized phase II study of BR96-doxorubicin conjugate in patients with metastatic breast cancer. J Clin Oncol. 1999;17:478–484. doi: 10.1200/JCO.1999.17.2.478. [DOI] [PubMed] [Google Scholar]
- 12.Remillard S., Rebhun L.I., Howie G.A., Kupchan S.M. Antimitotic activity of the potent tumor inhibitor maytansine. Science. 1975;189:1002–1005. doi: 10.1126/science.1241159. [DOI] [PubMed] [Google Scholar]
- 13.Dumontet C., Jordan M.A. Microtubule-binding agents: a dynamic field of cancer therapeutics. Nat Rev Drug Discov. 2010;9:790–803. doi: 10.1038/nrd3253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Florian S., Mitchison T.J. Anti-microtubule drugs. Methods Mol Biol. 2016;1413:403–421. doi: 10.1007/978-1-4939-3542-0_25. [DOI] [PubMed] [Google Scholar]
- 15.Fu Y., Ho M. DNA damaging agent-based antibody‒drug conjugates for cancer therapy. Antib Ther. 2018;1:33–43. doi: 10.1093/abt/tby007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Drago J.Z., Modi S., Chandarlapaty S. Unlocking the potential of antibody‒drug conjugates for cancer therapy. Nat Rev Clin Oncol. 2021;18:327–344. doi: 10.1038/s41571-021-00470-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Collins D.M., Bossenmaier B., Kollmorgen G., Niederfellner G. Acquired resistance to antibody‒drug conjugates. Cancers. 2019;11:394. doi: 10.3390/cancers11030394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hennessy E.J. Selective inhibitors of Bcl-2 and Bcl-xL: balancing antitumor activity with on-target toxicity. Bioorg Med Chem Lett. 2016;26:2105–2114. doi: 10.1016/j.bmcl.2016.03.032. [DOI] [PubMed] [Google Scholar]
- 19.Wilsbacher J.L., Cheng M., Cheng D., Trammell S.A.J., Shi Y., Guo J., et al. Discovery and characterization of novel nonsubstrate and substrate NAMPT inhibitors. Mol Cancer Ther. 2017;16:1236–1245. doi: 10.1158/1535-7163.MCT-16-0819. [DOI] [PubMed] [Google Scholar]
- 20.Almaliti J., Miller B., Pietraszkiewicz H., Glukhov E., Naman C.B., Kline T., et al. Exploration of the carmaphycins as payloads in antibody drug conjugate anticancer agents. Eur J Med Chem. 2019;161:416–432. doi: 10.1016/j.ejmech.2018.10.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gerber H.P., Sapra P., Loganzo F., May C. Combining antibody‒drug conjugates and immune-mediated cancer therapy: what to expect?. Biochem Pharmacol. 2016;102:1–6. doi: 10.1016/j.bcp.2015.12.008. [DOI] [PubMed] [Google Scholar]
- 22.do Pazo C., Nawaz K., Webster R.M. The oncology market for antibody‒drug conjugates. Nat Rev Drug Discov. 2021;20:583–584. doi: 10.1038/d41573-021-00054-2. [DOI] [PubMed] [Google Scholar]
- 23.Maneiro M.A., Forte N., Shchepinova M.M., Kounde C.S., Chudasama V., Baker J.R., et al. Antibody–PROTAC conjugates enable HER2-dependent targeted protein degradation of BRD4. ACS Chem Biol. 2020;15:1306–1312. doi: 10.1021/acschembio.0c00285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Levengood M.R., Zhang X., Hunter J.H., Emmerton K.K., Miyamoto J.B., Lewis T.S., et al. Orthogonal cysteine protection enables homogeneous multi-drug antibody‒drug conjugates. Angew Chem Int Ed Engl. 2017;56:733–737. doi: 10.1002/anie.201608292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kalim M., Chen J., Wang S., Lin C., Ullah S., Liang K., et al. Intracellular trafficking of new anticancer therapeutics: antibody‒drug conjugates. Drug Des Devel Ther. 2017;11:2265–2276. doi: 10.2147/DDDT.S135571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Widdison W.C.R. antibody‒drug conjugates and immunotoxins; New York: 2013. Factors involved in the design of cytotoxic payloads for antibody–drug conjugates. [Google Scholar]
- 27.Ducry L. Humana Press; 2013. Antibody‒drug conjugates. [Google Scholar]
- 28.Birrer M.J., Moore K.N., Betella I., Bates R.C. Antibody‒drug conjugate-based therapeutics: state of the science. J Natl Cancer Inst. 2019;111:538–549. doi: 10.1093/jnci/djz035. [DOI] [PubMed] [Google Scholar]
- 29.Li F., Emmerton K.K., Jonas M., Zhang X., Miyamoto J.B., Setter J.R., et al. Intracellular released payload influences potency and bystander-killing effects of antibody–drug conjugates in preclinical models. Cancer Res. 2016;76:2710–2719. doi: 10.1158/0008-5472.CAN-15-1795. [DOI] [PubMed] [Google Scholar]
- 30.Chen H., Lin Z., Arnst K.E., Miller D.D., Li W. Tubulin inhibitor-based antibody‒drug conjugates for cancer therapy. Molecules. 2017;22:1281. doi: 10.3390/molecules22081281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Janke C., Magiera M.M. The tubulin code and its role in controlling microtubule properties and functions. Nat Rev Mol Cell Biol. 2020;21:307–326. doi: 10.1038/s41580-020-0214-3. [DOI] [PubMed] [Google Scholar]
- 32.Barreca M., Stathis A., Barraja P., Bertoni F. An overview on anti-tubulin agents for the treatment of lymphoma patients. Pharmacol Ther. 2020;211 doi: 10.1016/j.pharmthera.2020.107552. [DOI] [PubMed] [Google Scholar]
- 33.Cassady J.M., Chan K.K., Floss H.G., Leistner E. Recent developments in the maytansinoid antitumor agents. Chem Pharm Bull (Tokyo) 2004;52:1–26. doi: 10.1248/cpb.52.1. [DOI] [PubMed] [Google Scholar]
- 34.Lewis Phillips G.D., Li G., Dugger D.L., Crocker L.M., Parsons K.L., Mai E., et al. Targeting HER2-positive breast cancer with trastuzumab-DM1, an antibody–cytotoxic drug conjugate. Cancer Res. 2008;68:9280–9290. doi: 10.1158/0008-5472.CAN-08-1776. [DOI] [PubMed] [Google Scholar]
- 35.Prota A.E., Bargsten K., Diaz J.F., Marsh M., Cuevas C., Liniger M., et al. A new tubulin-binding site and pharmacophore for microtubule-destabilizing anticancer drugs. Proc Natl Acad Sci U S A. 2014;111:13817–13821. doi: 10.1073/pnas.1408124111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Li W., Huang M., Li Y., Xia A., Tan L., Zhang Z., et al. C3 ester side chain plays a pivotal role in the antitumor activity of Maytansinoids. Biochem Biophys Res Commun. 2021;566:197–203. doi: 10.1016/j.bbrc.2021.05.071. [DOI] [PubMed] [Google Scholar]
- 37.Ikeyama S., Takeuchi M.J.B.P. Antitubulin activities of ansamitocins and maytansinoids. Biochem Pharmacol. 1981;30:2421–2425. doi: 10.1016/0006-2952(81)90336-1. [DOI] [PubMed] [Google Scholar]
- 38.Widdison W.C., Wilhelm S.D., Cavanagh E.E., Whiteman K.R., Leece B.A., Kovtun Y., et al. Semisynthetic maytansine analogues for the targeted treatment of cancer. J Med Chem. 2006;49:4392–4408. doi: 10.1021/jm060319f. [DOI] [PubMed] [Google Scholar]
- 39.Marzullo P., Boiarska Z., Perez-Pena H., Abel A.C., Alvarez-Bernad B., Lucena-Agell D., et al. Maytansinol derivatives: side reactions as a chance for new tubulin binders. Chemistry. 2022;28 doi: 10.1002/chem.202103520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nittoli T., Kelly M.P., Delfino F., Rudge J., Kunz A., Markotan T., et al. Antibody drug conjugates of cleavable amino-alkyl and aryl maytansinoids. Bioorg Med Chem. 2018;26:2271–2279. doi: 10.1016/j.bmc.2018.02.025. [DOI] [PubMed] [Google Scholar]
- 41.Pettit G.R., Srirangam J.K., Barkoczy J., Williams M., Boyd M.R., Hamel E., et al. Antineoplastic agents 365. Dolastatin 10 SAR probes. Anti Cancer Drug Des. 1998;13:243–277. [PubMed] [Google Scholar]
- 42.Gao G., Wang Y., Hua H., Li D., Tang C. Marine antitumor peptide dolastatin 10: biological activity, structural modification and synthetic chemistry. Mar Drugs. 2021;19:363. doi: 10.3390/md19070363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Waight A.B., Bargsten K., Doronina S., Steinmetz M.O., Sussman D., Prota A.E. Structural basis of microtubule destabilization by potent Auristatin anti-mitotics. PLoS One. 2016;11 doi: 10.1371/journal.pone.0160890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wang Y., Benz F.W., Wu Y., Wang Q., Chen Y., Chen X., et al. Structural insights into the pharmacophore of vinca domain inhibitors of microtubules. Mol Pharmacol. 2016;89:233–242. doi: 10.1124/mol.115.100149. [DOI] [PubMed] [Google Scholar]
- 45.Maderna A., Doroski M., Subramanyam C., Porte A., Leverett C.A., Vetelino B.C., et al. Discovery of cytotoxic dolastatin 10 analogues with N-terminal modifications. J Med Chem. 2014;57:10527–10543. doi: 10.1021/jm501649k. [DOI] [PubMed] [Google Scholar]
- 46.Maderna A., Leverett C.A. Recent advances in the development of new auristatins: structural modifications and application in antibody drug conjugates. Mol Pharm. 2015;12:1798–1812. doi: 10.1021/mp500762u. [DOI] [PubMed] [Google Scholar]
- 47.Doronina S.O., Kline T.B., Jeffrey A., Senter P.D., Meyer D. US patent WO2007008848(A3); 2006-07-07. inventors. Seagen Inc., assignee. Monomethylvaline compounds having phenylalanine carboxy modifications at the C-terminus. [Google Scholar]
- 48.Pettit G.R., Hogan F., Toms S. Antineoplastic agents. 592. Highly effective cancer cell growth inhibitory structural modifications of dolastatin 10. J Nat Prod. 2011;74:962–968. doi: 10.1021/np1007334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lerchen H.G., Stelte-Ludwig B., Golfier S., Schuhmacher J., Krenz U. US patent WO2011154359(A1); 2011-06-06. inventors. Seagen Inc., assignee. Novel auristatin derivatives and use thereof. [Google Scholar]
- 50.Satomaa T., Helin J., Ekholm F.S., inventors . US patent WO2014177771(A1); 2014-05-02. Conjugates of a glycoprotein or a glycan with a toxic payload. [Google Scholar]
- 51.Doronina S.O., Mendelsohn B.A., inventors . US patent WO2006132670(A3); 2005-11-14. Seagen Inc., assignee. Auristatins having an aminobenzoic acid unit at the N terminus. [Google Scholar]
- 52.Moquist P.N., Bovee T.D., Waight A.B., Mitchell J.A., Miyamoto J.B., Mason M.L., et al. Novel Auristatins with high bystander and cytotoxic activities in drug efflux-positive tumor models. Mol Cancer Ther. 2021;20:320–328. doi: 10.1158/1535-7163.MCT-20-0618. [DOI] [PubMed] [Google Scholar]
- 53.Akaiwa M., Dugal-Tessier J., Mendelsohn B.A. Antibody‒drug conjugate payloads; study of Auristatin derivatives. Chem Pharm Bull (Tokyo) 2020;68:201–211. doi: 10.1248/cpb.c19-00853. [DOI] [PubMed] [Google Scholar]
- 54.Hartmann R.W., Fahrner R., Shevshenko D., Fyrknas M., Larsson R., Lehmann F., et al. Rational design of Azastatin as a potential ADC payload with reduced bystander killing. ChemMedChem. 2020;15:2500–2512. doi: 10.1002/cmdc.202000497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Towle M.J., Salvato K.A., Budrow J., Wels B.F., Kuznetsov G., Aalfs K.K., et al. In vitro and in vivo anticancer activities of synthetic macrocyclic ketone analogues of halichondrin B. Cancer Res. 2001;61:1013–1021. [PubMed] [Google Scholar]
- 56.Dybdal-Hargreaves N.F., Risinger A.L., Mooberry S.L. Eribulin mesylate: mechanism of action of a unique microtubule-targeting agent. Clin Cancer Res. 2015;21:2445–2452. doi: 10.1158/1078-0432.CCR-14-3252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Doodhi H., Prota A.E., Rodriguez-Garcia R., Xiao H., Custar D.W., Bargsten K., et al. Termination of protofilament elongation by eribulin induces lattice defects that promote microtubule catastrophes. Curr Biol. 2016;26:1713–1721. doi: 10.1016/j.cub.2016.04.053. [DOI] [PubMed] [Google Scholar]
- 58.Cheng X., Li J., Tanaka K., Majumder U., Milinichik A.Z., Verdi A.C., et al. MORAb-202, an antibody‒drug conjugate utilizing humanized anti-human FRalpha farletuzumab and the microtubule-targeting agent eribulin, has potent antitumor activity. Mol Cancer Ther. 2018;17:2665–2675. doi: 10.1158/1535-7163.MCT-17-1215. [DOI] [PubMed] [Google Scholar]
- 59.Narayan S., Carlson E.M., Cheng H., Condon K., Du H., Eckley S., et al. Novel second generation analogs of eribulin. Part II: orally available and active against resistant tumors in vivo. Bioorg Med Chem Lett. 2011;21:1634–1638. doi: 10.1016/j.bmcl.2011.01.097. [DOI] [PubMed] [Google Scholar]
- 60.Park Y., Bae S.Y., Hah J.M., Lee S.K., Ryu J.S. Synthesis of stereochemically diverse cyclic analogs of tubulysins. Bioorg Med Chem. 2015;23:6827–6843. doi: 10.1016/j.bmc.2015.10.003. [DOI] [PubMed] [Google Scholar]
- 61.Nicolaou K.C., Pan S., Pulukuri K.K., Ye Q., Rigol S., Erande R.D., et al. Design, synthesis, and biological evaluation of tubulysin analogues, linker-drugs, and antibody‒drug conjugates, insights into structure‒activity relationships, and tubulysin-tubulin binding derived from X-ray crystallographic analysis. J Org Chem. 2021;86:3377–3421. doi: 10.1021/acs.joc.0c02755. [DOI] [PubMed] [Google Scholar]
- 62.Murray B.C., Peterson M.T., Fecik R.A. Chemistry and biology of tubulysins: antimitotic tetrapeptides with activity against drug resistant cancers. Nat Prod Rep. 2015;32:654–662. doi: 10.1039/c4np00036f. [DOI] [PubMed] [Google Scholar]
- 63.Courter J.R., Hamilton J.Z., Hendrick N.R., Zaval M., Waight A.B., Lyon R.P., et al. Structure‒activity relationships of tubulysin analogues. Bioorg Med Chem Lett. 2020;30 doi: 10.1016/j.bmcl.2020.127241. [DOI] [PubMed] [Google Scholar]
- 64.Leverett C.A., Sukuru S.C., Vetelino B.C., Musto S., Parris K., Pandit J., et al. Design, synthesis, and cytotoxic evaluation of novel tubulysin analogues as ADC payloads. ACS Med Chem Lett. 2016;7:999–1004. doi: 10.1021/acsmedchemlett.6b00274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Cheng H., Cong Q., Dervin D., Stevens A., Vemuri K., Huber M., et al. Synthesis and biological evaluation of a carbamate-containing tubulysin antibody‒drug conjugate. Bioconjugate Chem. 2020;31:2350–2361. doi: 10.1021/acs.bioconjchem.0c00429. [DOI] [PubMed] [Google Scholar]
- 66.Sani M., Lazzari P., Folini M., Spiga M., Zuco V., De Cesare M., et al. Synthesis and superpotent anticancer activity of tubulysins carrying non-hydrolysable N-substituents on tubuvaline. Chemistry. 2017;23:5842–5850. doi: 10.1002/chem.201700874. [DOI] [PubMed] [Google Scholar]
- 67.Weiss C., Figueras E., Borbely A.N., Sewald N. Cryptophycins: cytotoxic cyclodepsipeptides with potential for tumor targeting. J Pept Sci. 2017;23:514–531. doi: 10.1002/psc.3015. [DOI] [PubMed] [Google Scholar]
- 68.Eren E., Watts N.R., Sackett D.L., Wingfield P.T. Conformational changes in tubulin upon binding cryptophycin-52 reveal its mechanism of action. J Biol Chem. 2021;297 doi: 10.1016/j.jbc.2021.101138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Verma V.A., Pillow T.H., DePalatis L., Li G., Phillips G.L., Polson A.G., et al. The cryptophycins as potent payloads for antibody drug conjugates. Bioorg Med Chem Lett. 2015;25:864–868. doi: 10.1016/j.bmcl.2014.12.070. [DOI] [PubMed] [Google Scholar]
- 70.Lai Q., Wu M., Wang R., Lai W., Tao Y., Lu Y., et al. Cryptophycin-55/52 based antibody‒drug conjugates: synthesis, efficacy, and mode of action studies. Eur J Med Chem. 2020;199 doi: 10.1016/j.ejmech.2020.112364. [DOI] [PubMed] [Google Scholar]
- 71.Good J.A., Skoufias D.A., Kozielski F. Elucidating the functionality of kinesins: an overview of small molecule inhibitors. Semin Cell Dev Biol. 2011;22:935–945. doi: 10.1016/j.semcdb.2011.09.023. [DOI] [PubMed] [Google Scholar]
- 72.Indorato R.L., Talapatra S.K., Lin F., Haider S., Mackay S.P., Kozielski F., et al. Is the fate of clinical candidate Arry-520 already sealed? Predicting resistance in Eg5‒inhibitor complexes. Mol Cancer Ther. 2019;18:2394–2406. doi: 10.1158/1535-7163.MCT-19-0154. [DOI] [PubMed] [Google Scholar]
- 73.Karpov A.S., Nieto-Oberhuber C.M., Abrams T., Beng-Louka E., Blanco E., Chamoin S., et al. Discovery of potent and selective antibody‒drug conjugates with Eg5 inhibitors through linker and payload optimization. ACS Med Chem Lett. 2019;10:1674–1679. doi: 10.1021/acsmedchemlett.9b00468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Kirchhoff D., Stelte-Ludwig B., Lerchen H.G., Wengner A.M., Ahsen O.V., Buchmann P., et al. IL3RA-targeting antibody‒drug conjugate BAY-943 with a kinesin spindle protein inhibitor payload shows efficacy in preclinical models of hematologic malignancies. Cancers. 2020;12:3464. doi: 10.3390/cancers12113464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lerchen H.G., Wittrock S., Stelte-Ludwig B., Sommer A., Berndt S., Griebenow N., et al. Antibody‒drug conjugates with pyrrole-based KSP inhibitors as the payload class. Angew Chem Int Ed Engl. 2018;57:15243–15247. doi: 10.1002/anie.201807619. [DOI] [PubMed] [Google Scholar]
- 76.Amani J., Gorjizadeh N., Younesi S., Najafi M., Ashrafi A.M., Irian S., et al. Cyclin-dependent kinase inhibitors (CDKIs) and the DNA damage response: the link between signaling pathways and cancer. DNA Repair. 2021;102 doi: 10.1016/j.dnarep.2021.103103. [DOI] [PubMed] [Google Scholar]
- 77.Adhikari A., Shen B., Rader C. Challenges and opportunities to develop enediyne natural products as payloads for antibody‒drug conjugates. Antib Ther. 2021;4:1–15. doi: 10.1093/abt/tbab001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Zein N., Sinha A.M., McGahren W.J., Ellestad G.A. Calicheamicin gamma 1I: an antitumor antibiotic that cleaves double-stranded DNA site specifically. Science. 1988;240:1198–1201. doi: 10.1126/science.3240341. [DOI] [PubMed] [Google Scholar]
- 79.Kumar R.A., Ikemoto N., Patel D.J. Solution structure of the calicheamicin gamma 1I-DNA complex. J Mol Biol. 1997;265:187–201. doi: 10.1006/jmbi.1996.0718. [DOI] [PubMed] [Google Scholar]
- 80.Ricart A.D. Antibody‒drug conjugates of calicheamicin derivative: gemtuzumab ozogamicin and inotuzumab ozogamicin. Clin Cancer Res. 2011;17:6417–6427. doi: 10.1158/1078-0432.CCR-11-0486. [DOI] [PubMed] [Google Scholar]
- 81.Davies J., Wang H., Taylor T., Warabi K., Huang X.H., Andersen R.J. Uncialamycin, a new enediyne antibiotic. Org Lett. 2005;7:5233–5236. doi: 10.1021/ol052081f. [DOI] [PubMed] [Google Scholar]
- 82.Poudel Y.B., Rao C., Kotapati S., Deshpande M., Thevanayagam L., Pan C., et al. Design, synthesis and biological evaluation of phenol-linked uncialamycin antibody‒drug conjugates. Bioorg Med Chem Lett. 2020;30 doi: 10.1016/j.bmcl.2019.126782. [DOI] [PubMed] [Google Scholar]
- 83.Nicolaou K.C., Rigol S., Pitsinos E.N., Das D., Lu Y., Rout S., et al. Uncialamycin-based antibody–drug conjugates: unique enediyne ADCs exhibiting bystander killing effect. Proc Natl Acad Sci U S A. 2021;118 doi: 10.1073/pnas.2107042118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Nicolaou K.C., Wang Y., Lu M., Mandal D., Pattanayak M.R., Yu R., et al. Streamlined total synthesis of uncialamycin and its application to the synthesis of designed analogues for biological investigations. J Am Chem Soc. 2016;138:8235–8246. doi: 10.1021/jacs.6b04339. [DOI] [PubMed] [Google Scholar]
- 85.Chowdari N.S., Pan C., Rao C., Langley D.R., Sivaprakasam P., Sufi B., et al. Uncialamycin as a novel payload for antibody drug conjugate (ADC) based targeted cancer therapy. Bioorg Med Chem Lett. 2019;29:466–470. doi: 10.1016/j.bmcl.2018.12.021. [DOI] [PubMed] [Google Scholar]
- 86.Dong W., Shi J., Yuan T., Qi B., Yu J., Dai J., et al. Antibody‒drug conjugates of 7-ethyl-10-hydroxycamptothecin: sacituzumab govitecan and labetuzumab govitecan. Eur J Med Chem. 2019;167:583–593. doi: 10.1016/j.ejmech.2019.02.017. [DOI] [PubMed] [Google Scholar]
- 87.Staker B.L., Hjerrild K., Feese M.D., Behnke C.A., Burgin A.B., Jr., Stewart L. The mechanism of topoisomerase I poisoning by a camptothecin analog. Proc Natl Acad Sci U S A. 2002;99:15387–15392. doi: 10.1073/pnas.242259599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Nakada T., Masuda T., Naito H., Yoshida M., Ashida S., Morita K., et al. Novel antibody drug conjugates containing exatecan derivative-based cytotoxic payloads. Bioorg Med Chem Lett. 2016;26:1542–1545. doi: 10.1016/j.bmcl.2016.02.020. [DOI] [PubMed] [Google Scholar]
- 89.Li W., Veale K.H., Qiu Q., Sinkevicius K.W., Maloney E.K., Costoplus J.A., et al. Synthesis and evaluation of camptothecin antibody‒drug conjugates. ACS Med Chem Lett. 2019;10:1386–1392. doi: 10.1021/acsmedchemlett.9b00301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Xu Z., Guo D., Jiang Z., Tong R., Jiang P., Bai L., et al. Novel HER2-targeting antibody‒drug conjugates of trastuzumab beyond T-DM1 in breast cancer: trastuzumab deruxtecan (DS-8201a) and (Vic-)trastuzumab duocarmazine (SYD985) Eur J Med Chem. 2019;183 doi: 10.1016/j.ejmech.2019.111682. [DOI] [PubMed] [Google Scholar]
- 91.Lyski R.D., Bou L.B., Lau U.Y., Meyer D.W., Cochran J.H., Okeley N.M., et al. Development of novel antibody–camptothecin conjugates. Mol Cancer Ther. 2021;20:329–339. doi: 10.1158/1535-7163.MCT-20-0526. [DOI] [PubMed] [Google Scholar]
- 92.Wells G., Martin C.R., Howard P.W., Sands Z.A., Laughton C.A., Tiberghien A., et al. Design, synthesis, and biophysical and biological evaluation of a series of pyrrolobenzodiazepine-poly(N-methylpyrrole) conjugates. J Med Chem. 2006;49:5442–5461. doi: 10.1021/jm051199z. [DOI] [PubMed] [Google Scholar]
- 93.Rettig M., Weingarth M., Langel W., Kamal A., Kumar P.P., Weisz K. Solution structure of a covalently bound pyrrolo[2,1-c][1,4]benzodiazepine-benzimidazole hybrid to a 10mer DNA duplex. Biochemistry. 2009;48:12223–12232. doi: 10.1021/bi901655t. [DOI] [PubMed] [Google Scholar]
- 94.Corcoran D.B., Lewis T., Nahar K.S., Jamshidi S., Fegan C., Pepper C., et al. Effects of systematic shortening of noncovalent C8 side chain on the cytotoxicity and NF-kappaB inhibitory capacity of pyrrolobenzodiazepines (PBDs) J Med Chem. 2019;62:2127–2139. doi: 10.1021/acs.jmedchem.8b01849. [DOI] [PubMed] [Google Scholar]
- 95.Smith S.W., Jammalamadaka V., Borkin D., Zhu J., Degrado S.J., Lu J., et al. Design and synthesis of isoquinolidinobenzodiazepine dimers, a novel class of antibody‒drug conjugate payload. ACS Med Chem Lett. 2018;9:56–60. doi: 10.1021/acsmedchemlett.7b00436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Staben L.R., Chen J., Cruz-Chuh J.D., Del Rosario G., Go M.A., Guo J., et al. Systematic variation of pyrrolobenzodiazepine (PBD)-dimer payload physicochemical properties impacts efficacy and tolerability of the corresponding antibody‒drug conjugates. J Med Chem. 2020;63:9603–9622. doi: 10.1021/acs.jmedchem.0c00691. [DOI] [PubMed] [Google Scholar]
- 97.Pei Z., Chen C., Chen J., Cruz-Chuh J.D., Delarosa R., Deng Y., et al. Exploration of pyrrolobenzodiazepine (PBD)-dimers containing disulfide-based prodrugs as payloads for antibody‒drug conjugates. Mol Pharm. 2018;15:3979–3996. doi: 10.1021/acs.molpharmaceut.8b00431. [DOI] [PubMed] [Google Scholar]
- 98.Zammarchi F., Corbett S., Adams L., Tyrer P.C., Kiakos K., Janghra N., et al. ADCT-402, a PBD dimer-containing antibody drug conjugate targeting CD19-expressing malignancies. Blood. 2018;131:1094–1105. doi: 10.1182/blood-2017-10-813493. [DOI] [PubMed] [Google Scholar]
- 99.Flynn M.J., Zammarchi F., Tyrer P.C., Akarca A.U., Janghra N., Britten C.E., et al. ADCT-301, a pyrrolobenzodiazepine (PBD) dimer-containing antibody‒drug conjugate (ADC) targeting CD25-expressing hematological malignancies. Mol Cancer Ther. 2016;15:2709–2721. doi: 10.1158/1535-7163.MCT-16-0233. [DOI] [PubMed] [Google Scholar]
- 100.Ghosh N., Sheldrake H.M., Searcey M., Pors K. Chemical and biological explorations of the family of CC-1065 and the duocarmycin natural products. Curr Top Med Chem. 2009;9:1494–1524. doi: 10.2174/156802609789909812. [DOI] [PubMed] [Google Scholar]
- 101.Smith J.A., Bifulco G., Case D.A., Boger D.L., Gomez-Paloma L., Chazin W.J. The structural basis for in situ activation of DNA alkylation by duocarmycin SA. J Mol Biol. 2000;300:1195–1204. doi: 10.1006/jmbi.2000.3887. [DOI] [PubMed] [Google Scholar]
- 102.Elgersma R.C., Coumans R.G., Huijbregts T., Menge W.M., Joosten J.A., Spijker H.J., et al. Design, synthesis, and evaluation of linker-duocarmycin payloads: toward selection of HER2-targeting antibody‒drug conjugate SYD985. Mol Pharm. 2015;12:1813–1835. doi: 10.1021/mp500781a. [DOI] [PubMed] [Google Scholar]
- 103.Giddens A.C., Lee H.H., Lu G.L., Miller C.K., Guo J., Lewis Phillips G.D., et al. Analogues of DNA minor groove cross-linking agents incorporating aminoCBI, an amino derivative of the duocarmycins: synthesis, cytotoxicity, and potential as payloads for antibody‒drug conjugates. Bioorg Med Chem. 2016;24:6075–6081. doi: 10.1016/j.bmc.2016.09.068. [DOI] [PubMed] [Google Scholar]
- 104.Yin W., Rogge M. Targeting RNA: a transformative therapeutic strategy. Clin Transl Sci. 2019;12:98–112. doi: 10.1111/cts.12624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Lu S.X., De Neef E., Thomas J.D., Sabio E., Rousseau B., Gigoux M., et al. Pharmacologic modulation of RNA splicing enhances anti-tumor immunity. Cell. 2021;184:4032–4047 e31. doi: 10.1016/j.cell.2021.05.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Liu X., Biswas S., Berg M.G., Antapli C.M., Xie F., Wang Q., et al. Genomics-guided discovery of thailanstatins A, B, and C As pre-mRNA splicing inhibitors and antiproliferative agents from Burkholderia thailandensis MSMB43. J Nat Prod. 2013;76:685–693. doi: 10.1021/np300913h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Nicolaou K.C., Rigol S. Total synthesis in search of potent antibody‒drug conjugate payloads. From the fundamentals to the translational. Acc Chem Res. 2019;52:127–139. doi: 10.1021/acs.accounts.8b00537. [DOI] [PubMed] [Google Scholar]
- 108.Puthenveetil S., Loganzo F., He H., Dirico K., Green M., Teske J., et al. Natural product splicing inhibitors: a new class of antibody‒drug conjugate (ADC) payloads. Bioconjugate Chem. 2016;27:1880–1888. doi: 10.1021/acs.bioconjchem.6b00291. [DOI] [PubMed] [Google Scholar]
- 109.Wieland T., Faulstich H. Fifty years of amanitin. Experientia. 1991;47:1186–1193. doi: 10.1007/BF01918382. [DOI] [PubMed] [Google Scholar]
- 110.Kaplan C.D., Larsson K.M., Kornberg R.D. The RNA polymerase II trigger loop functions in substrate selection and is directly targeted by alpha-amanitin. Mol Cell. 2008;30:547–556. doi: 10.1016/j.molcel.2008.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Siegert M.J., Knittel C.H., Sussmuth R.D. A convergent total synthesis of the death cap toxin alpha-amanitin. Angew Chem Int Ed Engl. 2020;59:5500–5504. doi: 10.1002/anie.201914620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Figueroa-Vazquez V., Ko J., Breunig C., Baumann A., Giesen N., Palfi A., et al. HDP-101, an Anti-BCMA antibody‒drug conjugate, safely delivers amanitin to induce cell death in proliferating and resting multiple myeloma cells. Mol Cancer Ther. 2021;20:367–378. doi: 10.1158/1535-7163.MCT-20-0287. [DOI] [PubMed] [Google Scholar]
- 113.Danielczyk A., Stahn R., Faulstich D., Loffler A., Marten A., Karsten U., et al. PankoMab: a potent new generation anti-tumour MUC1 antibody. Cancer Immunol Immunother. 2006;55:1337–1347. doi: 10.1007/s00262-006-0135-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Zhang Q., Bi J., Zheng X., Chen Y., Wang H., Wu W., et al. Blockade of the checkpoint receptor TIGIT prevents NK cell exhaustion and elicits potent anti-tumor immunity. Nat Immunol. 2018;19:723–732. doi: 10.1038/s41590-018-0132-0. [DOI] [PubMed] [Google Scholar]
- 115.Kutikhin A.G. Association of polymorphisms in TLR genes and in genes of the Toll-like receptor signaling pathway with cancer risk. Hum Immunol. 2011;72:1095–1116. doi: 10.1016/j.humimm.2011.07.307. [DOI] [PubMed] [Google Scholar]
- 116.Ishida H., Asami J., Zhang Z., Nishizawa T., Shigematsu H., Ohto U., et al. Cryo-EM structures of Toll-like receptors in complex with UNC93B1. Nat Struct Mol Biol. 2021;28:173–180. doi: 10.1038/s41594-020-00542-w. [DOI] [PubMed] [Google Scholar]
- 117.Yu J., Fang T., Yun C., Liu X., Cai X. Antibody‒drug conjugates targeting the human epidermal growth factor receptor family in cancers. Front Mol Biosci. 2022;9 doi: 10.3389/fmolb.2022.847835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Wang Z., Gao Y., He L., Sun S., Xia T., Hu L., et al. Structure-based design of highly potent Toll-like receptor 7/8 dual agonists for cancer immunotherapy. J Med Chem. 2021;64:7507–7532. doi: 10.1021/acs.jmedchem.1c00179. [DOI] [PubMed] [Google Scholar]
- 119.He L., Wang L., Wang Z., Li T., Chen H., Zhang Y., et al. Immune modulating antibody‒drug conjugate (IM-ADC) for cancer immunotherapy. J Med Chem. 2021;64:15716–15726. doi: 10.1021/acs.jmedchem.1c00961. [DOI] [PubMed] [Google Scholar]
- 120.Ackerman S.E., Pearson C.I., Gregorio J.D., Gonzalez J.C., Kenkel J.A., Hartmann F.J., et al. Immune-stimulating antibody conjugates elicit robust myeloid activation and durable antitumor immunity. Nat Can (Que) 2021;2:18–33. doi: 10.1038/s43018-020-00136-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Marcus A., Mao A.J., Lensink-Vasan M., Wang L., Vance R.E., Raulet D.H. Tumor-derived cGAMP triggers a STING-mediated interferon response in non-tumor cells to activate the NK cell response. Immunity. 2018;49:754–763 e4. doi: 10.1016/j.immuni.2018.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Ramanjulu J.M., Pesiridis G.S., Yang J., Concha N., Singhaus R., Zhang S.Y., et al. Design of amidobenzimidazole STING receptor agonists with systemic activity. Nature. 2018;564:439–443. doi: 10.1038/s41586-018-0705-y. [DOI] [PubMed] [Google Scholar]
- 123.Cong X., Yuan Z., Du Y., Wu B., Lu D., Wu X., et al. Crystal structures of porcine STING(CBD)-CDN complexes reveal the mechanism of ligand recognition and discrimination of STING proteins. J Biol Chem. 2019;294:11420–11432. doi: 10.1074/jbc.RA119.007367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Li P., Zheng Y., Chen X. Drugs for autoimmune inflammatory diseases: from small molecule compounds to anti-TNF biologics. Front Pharmacol. 2017;8:460. doi: 10.3389/fphar.2017.00460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Hobson A.D., McPherson M.J., Waegell W., Goess C.A., Stoffel R.H., Li X., et al. Design and development of glucocorticoid receptor modulators as immunology antibody‒drug conjugate payloads. J Med Chem. 2022;65:4500–4533. doi: 10.1021/acs.jmedchem.1c02099. [DOI] [PubMed] [Google Scholar]
- 126.Edman K., Hosseini A., Bjursell M.K., Aagaard A., Wissler L., Gunnarsson A., et al. Ligand binding mechanism in steroid receptors: from conserved plasticity to differential evolutionary cconstraints. Structure. 2015;23:2280–2290. doi: 10.1016/j.str.2015.09.012. [DOI] [PubMed] [Google Scholar]
- 127.Dragovich P.S. Antibody‒drug conjugates for immunology. J Med Chem. 2022;65:4496–4499. doi: 10.1021/acs.jmedchem.2c00339. [DOI] [PubMed] [Google Scholar]
- 128.Han A., Olsen O., D'Souza C., Shan J., Zhao F., Yanolatos J., et al. Development of novel glucocorticoids for use in antibody‒drug conjugates for the treatment of inflammatory diseases. J Med Chem. 2021;64:11958–11971. doi: 10.1021/acs.jmedchem.1c00541. [DOI] [PubMed] [Google Scholar]
- 129.Youle R.J., Strasser A. The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol. 2008;9:47–59. doi: 10.1038/nrm2308. [DOI] [PubMed] [Google Scholar]
- 130.Tanaka Y., Aikawa K., Nishida G., Homma M., Sogabe S., Igaki S., et al. Discovery of potent Mcl-1/Bcl-xL dual inhibitors by using a hybridization strategy based on structural analysis of target proteins. J Med Chem. 2013;56:9635–9645. doi: 10.1021/jm401170c. [DOI] [PubMed] [Google Scholar]
- 131.Holen K., Saltz L.B., Hollywood E., Burk K., Hanauske A.R. The pharmacokinetics, toxicities, and biologic effects of FK866, a nicotinamide adenine dinucleotide biosynthesis inhibitor. Invest N Drugs. 2008;26:45–51. doi: 10.1007/s10637-007-9083-2. [DOI] [PubMed] [Google Scholar]
- 132.Karpov A.S., Abrams T., Clark S., Raikar A., D'Alessio J.A., Dillon M.P., et al. Nicotinamide phosphoribosyltransferase inhibitor as a novel payload for antibody‒drug conjugates. ACS Med Chem Lett. 2018;9:838–842. doi: 10.1021/acsmedchemlett.8b00254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Neumann C.S., Olivas K.C., Anderson M.E., Cochran J.H., Jin S., Li F., et al. Targeted delivery of cytotoxic NAMPT inhibitors using antibody‒drug conjugates. Mol Cancer Ther. 2018;17:2633–2642. doi: 10.1158/1535-7163.MCT-18-0643. [DOI] [PubMed] [Google Scholar]
- 134.Pereira A.R., Kale A.J., Fenley A.T., Byrum T., Debonsi H.M., Gilson M.K., et al. The carmaphycins: new proteasome inhibitors exhibiting an alpha,beta-epoxyketone warhead from a marine cyanobacterium. Chembiochem. 2012;13:810–817. doi: 10.1002/cbic.201200007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Tinworth C.P., Lithgow H., Churcher I.J.M. Small molecule-mediated protein knockdown as a new approach to drug discovery. Med Chem Commun. 2016;7:2206–2216. [Google Scholar]
- 136.Dragovich P.S. Degrader–antibody conjugates. Chem Soc Rev. 2022;51:3886–3897. doi: 10.1039/d2cs00141a. [DOI] [PubMed] [Google Scholar]
- 137.Kobayashi H., Choyke P.L. Near-infrared photoimmunotherapy of cancer. Acc Chem Res. 2019;52:2332–2339. doi: 10.1021/acs.accounts.9b00273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Gomes-da-Silva L.C., Kepp O., Kroemer G. Regulatory approval of photoimmunotherapy: photodynamic therapy that induces immunogenic cell death. OncoImmunology. 2020;9 doi: 10.1080/2162402X.2020.1841393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Kumar A., Kinneer K., Masterson L., Ezeadi E., Howard P., Wu H., et al. Synthesis of a heterotrifunctional linker for the site-specific preparation of antibody‒drug conjugates with two distinct warheads. Bioorg Med Chem Lett. 2018;28:3617–3621. doi: 10.1016/j.bmcl.2018.10.043. [DOI] [PubMed] [Google Scholar]
- 140.Nilchan N., Li X., Pedzisa L., Nanna A.R., Roush W.R., Rader C. Dual-mechanistic antibody‒drug conjugate via site-specific selenocysteine/cysteine conjugation. Antib Ther. 2019;2:71–78. doi: 10.1093/abt/tbz009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Yamazaki C.M., Yamaguchi A., Anami Y., Xiong W., Otani Y., Lee J., et al. Antibody‒drug conjugates with dual payloads for combating breast tumor heterogeneity and drug resistance. Nat Commun. 2021;12:3528. doi: 10.1038/s41467-021-23793-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Cooper B.M., Iegre J., Dh O.D., Olwegard Halvarsson M., Spring D.R. Peptides as a platform for targeted therapeutics for cancer: peptide‒drug conjugates (PDCs) Chem Soc Rev. 2021;50:1480–1494. doi: 10.1039/d0cs00556h. [DOI] [PubMed] [Google Scholar]
- 143.Liu X., Wu F., Ji Y., Yin L. Recent advances in anti-cancer protein/peptide delivery. Bioconjugate Chem. 2019;30:305–324. doi: 10.1021/acs.bioconjchem.8b00750. [DOI] [PubMed] [Google Scholar]
- 144.Li C.M., Haratipour P., Lingeman R.G., Perry J.J.P., Gu L., Hickey R.J., et al. Novel peptide therapeutic approaches for cancer treatment. Cells. 2021;10:2908. doi: 10.3390/cells10112908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Gong L., Zhao H., Liu Y., Wu H., Liu C., Chang S., et al. Research advances in peptide‒drug conjugates. Acta Pharm Sin B. 2023;13:3659–3677. doi: 10.1016/j.apsb.2023.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Zhu Y.S., Tang K., Lv J. Peptide–drug conjugate-based novel molecular drug delivery system in cancer. Trends Pharmacol Sci. 2021;42:857–869. doi: 10.1016/j.tips.2021.07.001. [DOI] [PubMed] [Google Scholar]
- 147.Ma L., Wang C., He Z., Cheng B., Zheng L., Huang K. Peptide‒drug conjugate: a novel drug design approach. Curr Med Chem. 2017;24:3373–3396. doi: 10.2174/0929867324666170404142840. [DOI] [PubMed] [Google Scholar]
- 148.Zhou J., Li Y., Huang W., Shi W., Qian H. Source and exploration of the peptides used to construct peptide‒drug conjugates. Eur J Med Chem. 2021;224 doi: 10.1016/j.ejmech.2021.113712. [DOI] [PubMed] [Google Scholar]
- 149.Alas M., Saghaeidehkordi A., Kaur K. Peptide‒drug conjugates with different linkers for cancer therapy. J Med Chem. 2021;64:216–232. doi: 10.1021/acs.jmedchem.0c01530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Yang Y., Wang S., Ma P., Jiang Y., Cheng K., Yu Y., et al. Drug conjugate-based anticancer therapy‒current status and perspectives. Cancer Lett. 2023;552 doi: 10.1016/j.canlet.2022.215969. [DOI] [PubMed] [Google Scholar]
- 151.Wang L., Chen H., Wang F., Zhang X. The development of peptide‒drug conjugates (PDCs) strategies for paclitaxel. Expet Opin Drug Deliv. 2022;19:147–161. doi: 10.1080/17425247.2022.2039621. [DOI] [PubMed] [Google Scholar]
- 152.Ziaei E., Saghaeidehkordi A., Dill C., Maslennikov I., Chen S., Kaur K. Targeting triple negative breast cancer cells with novel cytotoxic peptide‒doxorubicin conjugates. Bioconjugate Chem. 2019;30:3098–3106. doi: 10.1021/acs.bioconjchem.9b00755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Gregorc V., Cavina R., Novello S., Grossi F., Lazzari C., Capelletto E., et al. NGR-hTNF and doxorubicin as second-line treatment of patients with small cell lung cancer. Oncol. 2018;23:1133. doi: 10.1634/theoncologist.2018-0292. e112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Jonasch E., Haluska F.G. Interferon in oncological practice: review of interferon biology, clinical applications, and toxicities. Oncol. 2001;6:34–55. doi: 10.1634/theoncologist.6-1-34. [DOI] [PubMed] [Google Scholar]
- 155.Dash A., Chakraborty S., Pillai M.R., Knapp F.F., Jr. Peptide receptor radionuclide therapy: an overview. Cancer Biother Radiopharm. 2015;30:47–71. doi: 10.1089/cbr.2014.1741. [DOI] [PubMed] [Google Scholar]
- 156.Hennrich U., Kopka K. Lutathera®: the first FDA- and EMA-approved radiopharmaceutical for peptide receptor radionuclide therapy. Pharmaceuticals. 2019;12:114. doi: 10.3390/ph12030114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Mateos M.V., Blade J., Bringhen S., Ocio E.M., Efebera Y., Pour L., et al. Melflufen: a peptide‒drug conjugate for the treatment of multiple myeloma. J Clin Med. 2020;9:3120. doi: 10.3390/jcm9103120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Fu C., Yu L., Miao Y., Liu X., Yu Z., Wei M. Peptide‒drug conjugates (PDCs): a novel trend of research and development on targeted therapy, hype or hope?. Acta Pharm Sin B. 2023;13:498–516. doi: 10.1016/j.apsb.2022.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Giansanti F., Flavell D.J., Angelucci F., Fabbrini M.S., Ippoliti R. Strategies to improve the clinical utility of saporin-based targeted toxins. Toxins. 2018;10:82. doi: 10.3390/toxins10020082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Capone E., Giansanti F., Ponziani S., Lamolinara A., Iezzi M., Cimini A., et al. EV20-Sap, a novel anti-HER-3 antibody‒drug conjugate, displays promising antitumor activity in melanoma. Oncotarget. 2017;8:95412–95424. doi: 10.18632/oncotarget.20728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Kaplan G., Mazor R., Lee F., Jang Y., Leshem Y., Pastan I. Improving the in vivo efficacy of an anti-Tac (CD25) immunotoxin by pseudomonas exotoxin A domain II engineering. Mol Cancer Ther. 2018;17:1486–1493. doi: 10.1158/1535-7163.MCT-17-1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Kaplan G., Lee F., Onda M., Kolyvas E., Bhardwaj G., Baker D., et al. Protection of the furin cleavage site in low-toxicity immunotoxins based on pseudomonas exotoxin A. Toxins. 2016;8:217. doi: 10.3390/toxins8080217. [DOI] [PMC free article] [PubMed] [Google Scholar]