Abstract
Elongation of most bones occur at the growth plate through endochondral ossification in postnatal mammals. The maturation of chondrocyte is a crucial factor in longitudinal bone growth, which is regulated by a complex network of paracrine and endocrine signaling pathways. Here, we show that a phytochemical sulfuretin can stimulate hypertrophic chondrocyte differentiation in vitro and in vivo. We found that sulfuretin stabilized nuclear factor (erythroid-derived 2)-like 2 (Nrf2), stimulated its transcriptional activity, and induced expression of its target genes. Sulfuretin treatment resulted in an increase in body length of zebrafish larvae and induced the expression of chondrocyte markers. Consistently, a clinically available Nrf2 activator, dimethyl fumarate (DMF), induced the expression of hypertrophic chondrocyte markers and increased the body length of zebrafish. Importantly, we found that chondrocyte gene expression in cell culture and skeletal growth in zebrafish stimulated by sulfuretin were significantly abrogated by Nrf2 depletion, suggesting that such stimulatory effects of sulfuretin were dependent on Nrf2, at least in part. Taken together, these data show that sulfuretin has a potential use as supporting ingredients for enhancing bone growth.
Keywords: Bone growth, Chondrocyte differentiation, Nuclear factor (erythroid-derived 2)-like 2 (Nrf2), Sulfuretin
INTRODUCTION
The precise regulation of chondrocyte differentiation and function can be achieved by multiple secreted factors, including insulin-like growth factor (IGF), thyroid hormone, parathyroid hormone-related peptide (PTHrP), Indian hedgehog (IHH), fibroblast growth factor (FGF), wingless-related integration site (Wnt), transforming growth factor-β (TGF-β), and C-type natriuretic peptide (1-3). Chondrocyte differentiation is also regulated by intracellular signaling mediators such as MAP kinase, PI3K/Akt, and GTPase and various transcriptional regulators including Sox9, β-catenin, Smad, Runx, Mef2, and histone deacetylase HDAC4 (1, 4).
Sulfuretin, a major flavonoid isolated from Rhus verniciflua Stokes (RVS), has been shown to inhibit oxidative stress, inflammation, and obesity related metabolic defects (5-7). Sulfuretin can promote osteoblast differentiation in vitro and bone regeneration in vivo through activation of BMP, mTOR, Wnt/β-catenin, and Runx2 signaling (8). Sulfuretin can also inhibit osteoclast differentiation induced by macrophage colony stimulating factor and receptor activator of NF-κB ligand released from bone marrow macrophages by inhibiting NF-kB signaling (9, 10). These findings suggest that sulfuretin can facilitate bone formation by stimulating osteoblastogenesis and concurrently inhibiting osteoclastogenesis. However, whether sulfuretin plays a role in chondrocyte differentiation remains to be elucidated.
Transcription factor nuclear factor erythroid 2 (NF-E2) p45-related factor 2 (Nrf2) is a player that can sense oxidative stress and provide antioxidant responses (11). A ubiquitously detected Nrf2 can recognize the antioxidant response element (ARE), a cis-acting sequence found in the 5’-flanking region of target genes (12). Nrf2 has been implicated in bone remodeling (6). In particular, Nrf2 is detected in chondrocyte layers of tibia from postnatal mouse (13). Nrf2 overexpression in ATDC5 cells can inhibit chondrocyte differentiation (13). Nrf2 also inhibits apoptosis in human T/C28a2 chondrocytes induced by a high shear flow (14). Plant-derived natural antioxidants andrographolide and astaxanthin can protect chondrocytes against oxidative stress through Nrf2 activation (15, 16). These data suggest a critical role of Nrf2 in chondrocytes.
Signaling cascades of bone formation in higher vertebrates are well conserved in zebrafish, rendering zebrafish a useful model to dissect underlying molecular mechanisms and to model human skeletal diseases. Zebrafish model can take advantages of tractable genetic manipulations and versatile in vivo imaging (17). It can be applied in compound screening for bone homeostasis (18).
In this study, we investigated effects of sulfuretin on bone growth using cell lines and zebrafish models. We showed that sulfuretin stimulated chondrocyte differentiation and increased body length in zebrafish in an Nrf2-dependent manner. These data suggest that sulfuretin-mediated Nrf2 activation has potential applications in bone growth and its related disorders.
RESULTS
Sulfuretin promotes chondrocyte differentiation
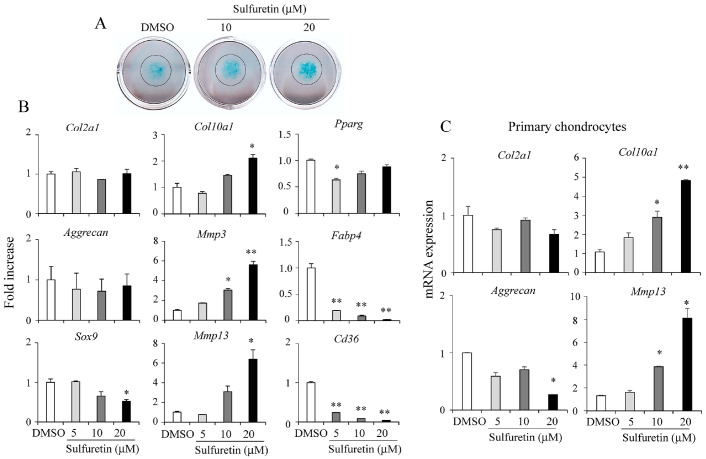
Previous findings have suggested that sulfuretin exhibits biological effects in bone cells including osteoblasts and osteoclasts (8). However, its roles in chondrocyte differentiation have not been addressed yet. To investigate effects of sulfuretin on chondrocyte differentiation, we induced a mass culture of C3H10T1/2 cells into chondrocytes for 12 days in the presence of sulfuretin. Matrix proteoglycan production evaluated by Alcian blue staining exhibited stimulatory effects of sulfuretin on chondrocyte differentiation (Fig. 1A). Sulfuretin at tolerated doses (Supplementary Fig. 1) induced the expression of late hypertrophic chondrocyte markers such as Col10a1, Mmp13, and Mmp3 without significantly changing the expression of early chondrogenic markers such as Col2a1, Aggrecan, or Sox9 (Fig. 1B). Sulfuretin suppressed the expression of adipogenic markers including Pparg, Fabp4, and Cd36 (Fig. 1B), consistent with a previous report (7). Sulfuretin also increased expression of Col10a1 and Mmp13, but not early chondrogenic markers (Col2a1 or Aggrecan) in primary chondrocytes isolated from mouse at P7 (Fig. 1C). These data show that sulfuretin can stimulate chondrocyte differentiation.
Fig. 1.
Sulfuretin stimulates chondrocyte differentiation in cell culture. (A) C3H10T1/2 cells were differentiated into chondrocytes in the presence of sulfuretin (10 or 20 μM) for 12 days followed by Alcian Blue staining. (B) Relative mRNA expression levels of early chondrocyte genes (Col2a1, Aggrecan, and Sox9), late chondrocyte genes (Col10a1, Mmp3, and Mmp13), and adipocyte genes (Pparg, Fabp4, and Cd36) after sulfuretin treatment were determined by real-time PCR. (C) Primary chondrocytes were isolated from mice at postnatal day 7 and treated with sulfuretin for 12 days. Relative mRNA expression was quantified by real-time PCR. Data shown represent mean ± SEM. Statistical significance was determined relative to a control by Student’s t-test (*P < 0.05; **P < 0.01).
Sulfuretin directly activates Nrf2 mediated transcription
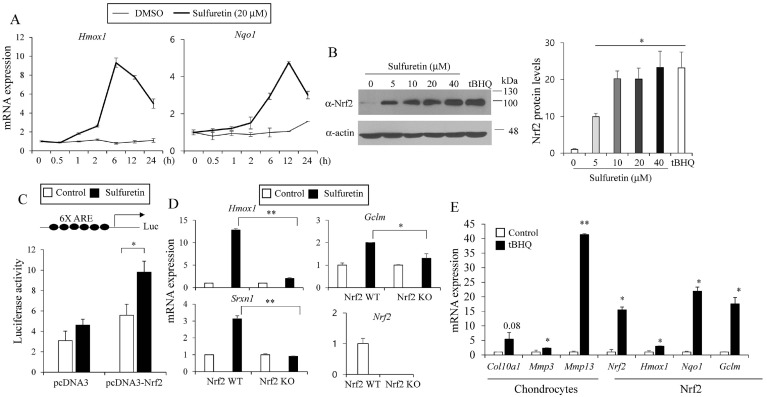
Various antioxidant phytochemicals isolated from edible or medicinal plants have been shown to increase Nrf2 activities (19). Accordingly, sulfuretin has been shown to affect Nrf2 singling pathway in human SH-SY5Y cells (20) and HepG2 cells (21). Based on this, we investigated effects of sulfuretin on the expression levels of verified Nrf2 dependent target genes, Heme oxygenase 1 (Hmox1) and NAD(P)H:quinone oxidoreductase 1 (Nqo1) in C3H10T1/2 cells (Fig. 2A). We found that sulfuretin increased the mRNA levels of Hmox1 and Nqo1 upto 9.3 and 4.6 folds respectively.
Fig. 2.
Sulfuretin stimulates Nrf2 activity. (A) C3H10T1/2 cells were treated with sulfuretin (20 μM) for 0.5, 1, 2, 6, 12, and 24 hours and relative temporal expression levels of Hmox1 and Nqo1 were measured by real-time PCR. (B) Sulfuretin treatment at 5-40 μM for 6 hours in C3H10T1/2 cells increased Nrf2 protein levels as determined by western blotting. Actin was used for loading control and tBHQ (50 μM) was used as a control. Representative blot images and relative densitometry bar graph were shown (n = 3). (C) HEK293T cells were transfected with the Nrf2 dependent-antioxidant response element (ARE) luciferase reporter construct (6XARE) and an expression vector coding for Nrf2 (pcDNA3-Nrf2). After 24 hours, cells were treated with DMSO (control) or sulfuretin (20 μM) for 24 hours and luciferase activity was measured. Luciferase signals were normalized with Renilla luciferase activity. (D) Nrf2 wild type (WT) MEF or Nrf2 knockout (KO) MEF were treated with sulfuretin (20 μM) for 12 hours and expression levels of Nrf2 target genes were measured by real-time PCR. (E) C3H10T1/2 cells were differentiated into chondrocytes in the presence of tBHQ (100 μM) and relative levels of hypertrophic genes and Nrf2 target genes were determined by real-time PCR. Data are presented as mean ± SEM (n = 3). Statistical significance was determined by comparison to the control using Student’s t-test (*P < 0.05; **P < 0.01).
Since Nrf2 stability is increased upon its activation, we measured protein levels of Nrf2. Comparable to the effects of tert-butylhydroquinone (tBHQ), a known Nrf2 activator, sulfuretin dose dependently increased Nrf2 protein levels in C3H10T1/2 cells (Fig. 2B). We transiently co-transfected HEK293T cells with Nrf2 dependent-antioxidant response element (ARE) luciferase reporter constructs along with an expression vector encoding Nrf2 and treated with DMSO or sulfuretin. Sulfuretin significantly stimulated the luciferase activity in the presence of Nrf2 (Fig. 2C).
To examine the effects of Nrf2 dependency of sulfuretin in inducing Nrf2 target gene expression, we treated sulfuretin into wild type (Nrf2 WT) and Nrf2 knockout (Nrf2 KO) mouse embryonic fibroblast (MEF) and measured Nrf2-induced gene expression. Sulfuretin induced expression levels of Nrf2 target genes (Hmox1, Gclm, and Srxn1) in WT MEF. However, these effects were significantly impaired in Nrf2 KO cells, showing that sulfuretin’s effects were Nrf2 dependent (Fig. 2D).
Having observed the effects of sulfuretin in stimulating Nrf2 activity, we tested alternative pharmacological Nrf2 activators to examine their effects on chondrocyte gene expression and bone growth. Similar to effects of sulfuretin, treatments with Nrf2 activator tBHQ in C3H10T1/2 cells for 12 days during chondrocyte differentiation induced Nrf2, Hmox1, Nqo1, and hypertrophic chondrocyte genes Col10a1, Mmp3, and Mmp13 (Fig. 2E). Taken together, these data indicate that sulfuretin can stimulate Nrf2 activity.
Sulfuretin increases bone formation in zebrafish
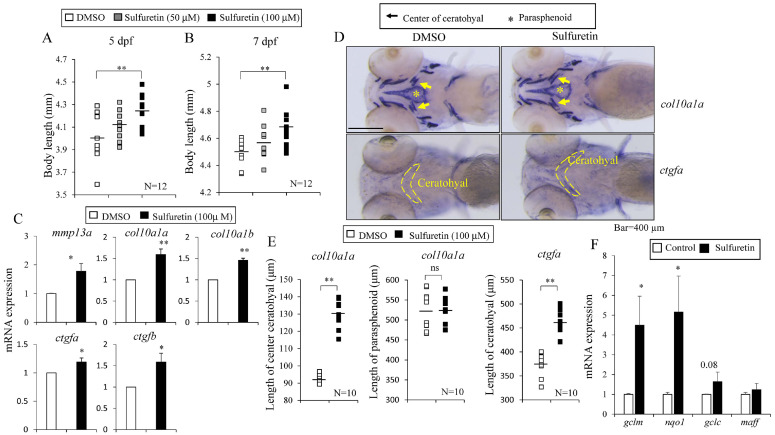
Zebrafish and mammals share conserved mechanisms of bone formation and remodeling (22). Thus, we tested the effects of sulfuretin in zebrafish. Incubating zebrafish embryos starting at 1 day postfertilization (dpf) for 4 or 6 days with sulfuretin (100 μM) significantly (P < 0.05) increased body lengths by approximately 5.6% and 3.9%, respectively, compared to control treatment (Fig. 3A, B). In addition, bone formation in the developing spine shown by alizarin red-positive numbers of small bones in the vertebral column was accelerated in sulfuretin-treated larvae than in DMSO-treated larvae (Supplementary Fig. 2). Similar to the effects in cell culture, sulfuretin treatment induced expression of hypertrophic chondrocyte genes including mmp13, col10a1a, col10a1b, ctgfa, and ctgfb in zebrafish larvae (Fig. 3C). Since ceratohyal and parasphenoid are two hallmarks of bone elements that form through endochondral ossification and intramembranous ossification, respectively, in zebrafish (23), we performed whole mount in situ hybridization (WISH) for visualizing col10a1 and ctgf as selective markers of hypertrophied chondrocyte formation (24). Compared to DMSO treatment, 4 days of treatment with sulfuretin (100 μM) increased the length of the center of ceratohyal by approximately 40% (col10a1 staining) and the total length of ceratohyal (ctgf staining) by 20%. However, no difference was observed in the parasphenoid area shown by WISH staining of col10a1 and ctgf (Fig. 3D, E). These data strongly suggest that sulfuretin can specifically stimulate endochondral ossification.
Fig. 3.
Sulfuretin treatment increases body length and Nrf2 target gene expression in zebrafish. (A) Zebrafish at 1 dpf were treated with sulfuretin (50 or 100 μM) for 4 days. Body lengths were then measured at 5 dpf. (B) Zebrafish at 1 dpf were treated with sulfuretin for 6 days and body lengths were measured at 7 dpf. (C) Total RNA was isolated from zebrafish after treatment with sulfuretin (100 μM) for 4 days and relative expression levels of mmp13a, col10a1a, col10a1b, ctgfa, and ctgfb were determined by real-time PCR. (D, E) Sulfuretin (100 μM) treatments increases the length of the ceratohyal (arrow) but not the parasphenoid (*) in zebrafish. (D) Whole mount in situ hybridization of col10a1 and ctgf. (E) Quantification of WISH staining in center of ceratohyal (col10a1), parasphenoid area (col10a1), and total length of ceratohyal (ctgf). (F) Zebrafish at 1 dpf were treated with sulfuretin for 4 days and expression levels of gclm, nqo1, gclc, and maff were measured by real-time PCR. Data are presented as mean ± SEM (n = 10 per group). Statistical significance was determined by Student’s t-test (ns, not significant; *P < 0.05; **P < 0.01).
To investigate whether sulfuretin also act on Nrf2 in zebrafish, we measured the Nrf2 target gene expression in zebrafish treated with sulfuretin. In line with the effects of sulfuretin as an Nrf2 activator, sulfuretin treatment in zebrafish consistently increased expression levels of nqo1, gclc, and gclm (Fig. 3F).
To further verify the effects of Nrf2 in body length, we treated 1 dpf zebrafish embryos with an FDA-approved cysteine-modifying drug dimethyl fumarate (DMF) (25) for 6 days and measured body lengths. Treatment with 1 or 4 μM DMF significantly increased the body length (Supplementary Fig. 3) and alizarin red-positive numbers of small bones in the vertebral column (Supplementary Fig. 4). DMF also induced the expression of hypertrophic chondrocyte markers similar to effects of sulfuretin in zebrafish (Supplementary Fig. 3). Together, these data show that sulfuretin can stimulate Nrf2 activity and increase body length in zebrafish.
Nrf2 is required for sulfuretin-induced chondrocyte gene expression in vitro and increased body length in zebrafish larvae
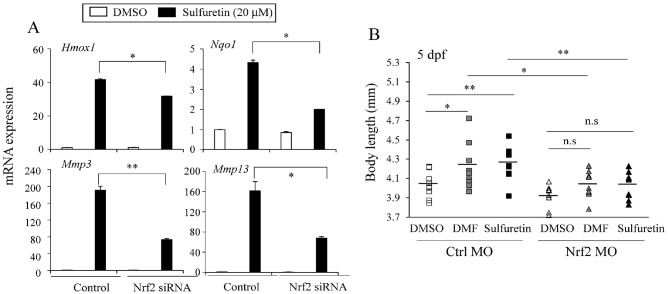
To assess Nrf2 dependent effects of sulfuretin on chondrocyte gene induction, we transfected C3H10T1/2 cells with Nrf2 small interfering RNAs (siRNA) or scramble siRNA and differentiated them into chondrocytes in the presence of sulfuretin. While the expression levels of Nrf2 target genes such as Hmox1 and Nqo1 upon sulfuretin treatment were dramatically increased in the group treated with scramble siRNA controls, such effects of sulfuretin were significantly compromised in Nrf2 siRNA transfected cells (Fig. 4A). In addition, induction of hypertrophic chondrocyte genes Mmp3 and Mmp13 by sulfuretin was also significantly impaired in Nrf2 siRNA transfected cells (Fig. 4A).
Fig. 4.
Nrf2 is necessary for effects of sulfuretin on chondrocyte and bone growth in zebrafish. (A) Induction of chondrocyte genes by sulfuretin is blunted in Nrf2 knockdown cells. C3H10T1/2 cells were transfected with siRNA targeting Nrf2 (Nrf2 siRNA) and differentiated into chondrocytes for 12 days. Expression levels of Nrf2 targets (Hmox1 and Nqo1) and chondrocyte genes (Mmp3 and Mmp13) were measured by real-time PCR. Data are presented as mean ± SEM (n = 3). (B) Effects of sulfuretin and DMF on body lengths were compromised in nrf2 morpholino injected zebrafish. Sulfuretin (100 μM) or DMF (4 μM) was used to treat control morpholino or nrf2 morpholino injected zebrafish at 1 dpf for 4 days and their lengths were determined. Data are presented as mean ± SEM (n = 10). Statistical significance was determined by comparison with the control using Student’s t-test (ns, not significant; *P < 0.05; **P < 0.01).
To further determine the Nrf2-dependent effects of sulfuretin and DMF in vivo, we treated nrf2 morpholino (MO)-injected zebrafish embryos with sulfuretin for 4 days and assessed its effects. Sulfuretin or DMF treatment significantly increased body length of zebrafish by 5.22% or 4.77%, respectively, compared to control treatments. However, such effects were significantly abrogated in nrf2 MO injected larvae by 3.0% and 3.16% (42% and 33.8% reduction), respectively, compared to effects in control MO injected larvae, corroborating the role of nrf2 activation in increased body length (Fig. 4B). Together, these data indicate that the ability of sulfuretin to increase chondrocyte differentiation and body length is dependent on Nrf2, at least in part.
DISCUSSION
Sulfuretin, a major flavonoid of Rhus verniciflua Stokes, exhibits anti-inflammatory, anti-obese, anti-oxidative, pro-osteogenic, and neuroprotective activities (5, 7, 26). It has been also shown that sulfuretin can induce bone regeneration and inhibit bone resorption (8). In this study, we provided evidence that sulfuretin could increase expression of late chondrocyte genes in C3H10T1/2 cells and primary chondrocytes. We also showed that treatment with sulfuretin increased body lengths of zebrafish larvae by 5.6% likely through activation of Nrf2 signaling. Therefore our data strongly suggest that sulfuretin stimulates chondrocyte differentiation in vitro and increases body length in zebrafish.
We presented evidence that sulfuretin could increase chondrocyte gene expression by acting on Nrf2 signaling pathway. In line with this, a previous study has shown that sulfuretin can protect Aβ25-35-induced neurotoxicity through activation of Nrf2 and PI3K/Akt signaling pathways (20). Although the detailed molecular pathways linking Nrf2 to chondrocyte differentiation remain to be determined, it is possible that Nrf2 exhibits functional connections with numerous signaling pathways mediated by nuclear factor-κB (NF-κB), p53, mTOR, adrenergic receptor, and Notch1 (27-29). Growth factors such as IGF, PTHrp, IHH, FGF, Wnt, TGF-β, and BMP signaling are also candidates for Nrf2 targeting as they can affect chondrocyte differentiation (1-3). Indeed, we found that sulfuretin treatment promoted expression of Wnt, Tgf-β, and BMP signaling target genes (Supplementary Fig. 5), suggesting possible links between sulfuretin and these growth factor signaling pathways.
In the current study, sulfuretin increased bone length possibly by acting on chondrocytes. However, we cannot exclude the possibility that sulfuretin can affect multiple cell types including osteoblasts and osteoclasts as they have known to regulate bone lengths in certain animal models (5). In addition, it has been shown that enhancing or decreasing activities of osteoblasts or osteoclasts, respectively, can increase bone length. FGFr2 inactivation in osteoblast can decrease bone growth while osteoblast expression of growth hormone can increase tibial and femoral lengths (30). Therefore, although sulfuretin affects chondrocyte differentiation in a cell-autonomous manner, a limitation of the current study lies in the lack of direct evidence linking the contribution of sulfuretin mediated osteoblast and osteoclast activities to effect on longitudinal bone length in vivo. As such, the effects of sulfuretin in bone tissues followed by histological examination on hypertrophic chondrocytes in animal models would be interesting topics for future investigation.
In conclusion, we showed that sulfuretin stimulated chondrocyte differentiation in vitro and longitudinal bone growth in zebrafish. We also show that sulfuretin-mediated Nrf2 signaling plays a role in controlling chondrocyte differentiation. Given these in vitro and in vivo effects, sulfuretin and Nrf2 activators might provide a new approach to treat bone growth problem and bone-related diseases.
MATERIALS AND METHODS
Materials and methods are available in the supplemental material.
Funding Statement
ACKNOWLEDGEMENTS This study was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Science, and Technology (NRF-2020R1A2B5B02001592 to K.W.P., NRF-2020R1I1A1A 01074938 to U.J.Y., and NRF-2022R1I1A2071529 to J.-S.L.). This study was also partly supported by KRIBB Research Initiative Program (KGM5352322, KGM2112335, KGM9992312, KGM1382312) to J.-S.L.
Footnotes
CONFLICTS OF INTEREST
The authors have no conflicting interests.
REFERENCES
- 1.Mackie EJ, Ahmed YA, Tatarczuch L, Chen KS, Mirams M. Endochondral ossification: how cartilage is converted into bone in the developing skeleton. Int J Biochem Cell Biol. 2008;40:46–62. doi: 10.1016/j.biocel.2007.06.009. [DOI] [PubMed] [Google Scholar]
- 2.Peake NJ, Hobbs AJ, Pingguan-Murphy B, Salter DM, Berenbaum F, Chowdhury TT. Role of C-type natriuretic peptide signalling in maintaining cartilage and bone function. Osteoarthritis Cartilage. 2014;22:1800–1807. doi: 10.1016/j.joca.2014.07.018. [DOI] [PubMed] [Google Scholar]
- 3.van der Eerden BC, Karperien M, Wit JM. Systemic and local regulation of the growth plate. Endocr Rev. 2003;24:782–801. doi: 10.1210/er.2002-0033. [DOI] [PubMed] [Google Scholar]
- 4.Ulici V, Hoenselaar KD, Gillespie JR, Beier F. The PI3K pathway regulates endochondral bone growth through control of hypertrophic chondrocyte differentiation. BMC Dev Biol. 2008;8:40. doi: 10.1186/1471-213X-8-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Song MY, Jeong GS, Kwon KB, et al. Sulfuretin protects against cytokine-induced beta-cell damage and prevents streptozotocin-induced diabetes. Exp Mol Med. 2010;42:628–638. doi: 10.3858/emm.2010.42.9.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kim JH, Singhal V, Biswal S, Thimmulappa RK, DiGirolamo DJ. Nrf2 is required for normal postnatal bone acquisition in mice. Bone Res. 2014;2:14033. doi: 10.1038/boneres.2014.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kim S, Song NJ, Chang SH, et al. Sulfuretin prevents obesity and metabolic diseases in diet induced obese mice. Biomol Ther (Seoul) 2019;27:107–116. doi: 10.4062/biomolther.2018.090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Auh QS, Park KR, Yun HM, et al. Sulfuretin promotes osteoblastic differentiation in primary cultured osteoblasts and in vivo bone healing. Oncotarget. 2016;7:78320–78330. doi: 10.18632/oncotarget.12460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lee YR, Hwang JK, Koh HW, et al. Sulfuretin, a major flavonoid isolated from Rhus verniciflua, ameliorates experimental arthritis in mice. Life Sci. 2012;90:799–807. doi: 10.1016/j.lfs.2012.04.015. [DOI] [PubMed] [Google Scholar]
- 10.Shin JS, Park YM, Choi JH, et al. Sulfuretin isolated from heartwood of Rhus verniciflua inhibits LPS-induced inducible nitric oxide synthase, cyclooxygenase-2, and pro-inflammatory cytokines expression via the down-regulation of NF-kappaB in RAW 264.7 murine macrophage cells. Int Immunopharmacol. 2010;10:943–950. doi: 10.1016/j.intimp.2010.05.007. [DOI] [PubMed] [Google Scholar]
- 11.Mitsuishi Y, Motohashi H, Yamamoto M. The Keap1-Nrf2 system in cancers: stress response and anabolic metabolism. Front Oncol. 2012;2:200. doi: 10.3389/fonc.2012.00200.1cf2daa998d2444089b046eee35055e6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cuadrado A, Rojo AI, Wells G, et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat Rev Drug Discov. 2019;18:295–317. doi: 10.1038/s41573-018-0008-x. [DOI] [PubMed] [Google Scholar]
- 13.Hinoi E, Fujimori S, Wang L, Hojo H, Uno K, Yoneda Y. Nrf2 negatively regulates osteoblast differentiation via interfering with Runx2-dependent transcriptional activation. J Biol Chem. 2006;281:18015–18024. doi: 10.1074/jbc.M600603200. [DOI] [PubMed] [Google Scholar]
- 14.Healy ZR, Lee NH, Gao X, et al. Divergent responses of chondrocytes and endothelial cells to shear stress: cross-talk among COX-2, the phase 2 response, and apoptosis. Proc Natl Acad Sci U S A. 2005;102:14010–14015. doi: 10.1073/pnas.0506620102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Li B, Jiang T, Liu H, et al. Andrographolide protects chondrocytes from oxidative stress injury by activation of the Keap1-Nrf2-Are signaling pathway. J Cell Physiol. 2018;234:561–571. doi: 10.1002/jcp.26769. [DOI] [PubMed] [Google Scholar]
- 16.Sun K, Luo J, Jing X, et al. Astaxanthin protects against osteoarthritis via Nrf2: a guardian of cartilage homeostasis. Aging (Albany NY) 2019;11:10513–10531. doi: 10.18632/aging.102474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Valenti MT, Marchetto G, Mottes M, Dalle Carbonare L. Zebrafish: a suitable tool for the study of cell signaling in bone. Cells. 2020;9:1911. doi: 10.3390/cells9081911.eea482ce67444bb8bf661ef01b134bb7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bergen DJ, Kague E, Hammond CL. Zebrafish as an emerging model for osteoporosis: a primary testing platform for screening new osteo-active compounds. Front Endocrinol. 2019;10:6. doi: 10.3389/fendo.2019.00006.d941afaa09c14de7b807f9dc4b6448fa [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Suraweera TL, Rupasinghe HPV, Dellaire G, Xu Z. Regulation of Nrf2/ARE pathway by dietary flavonoids: a friend or foe for cancer management? Antioxidants (Basel) 2020;9:973. doi: 10.3390/antiox9100973.8b69a98ee7604eefa03dcc4b6ff5adb6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kwon SH, Ma SX, Hwang JY, Lee SY, Jang CG. Involvement of the Nrf2/HO-1 signaling pathway in sulfuretin-induced protection against amyloid beta25-35 neurotoxicity. Neuroscience. 2015;304:14–28. doi: 10.1016/j.neuroscience.2015.07.030. [DOI] [PubMed] [Google Scholar]
- 21.Lee DS, Kim KS, Ko W, et al. The cytoprotective effect of sulfuretin against tert-butyl hydroperoxide-induced hepatotoxicity through Nrf2/ARE and JNK/ERK MAPK-mediated heme oxygenase-1 expression. Int J Mol Sci. 2014;15:8863–8877. doi: 10.3390/ijms15058863.f70b40b926e24e1692b6ab5768b8d646 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Maradonna F, Gioacchini G, Falcinelli S, et al. Probiotic supplementation promotes calcification in Danio rerio larvae: a molecular study. PLoS One. 2013;8:e83155. doi: 10.1371/journal.pone.0083155.1f010fd167674c23a96124f38b4a266f [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mitchell RE, Huitema LF, Skinner RE, et al. New tools for studying osteoarthritis genetics in zebrafish. Osteoarthritis Cartilage. 2013;21:269–278. doi: 10.1016/j.joca.2012.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tonelli F, Bek JW, Besio R, et al. Zebrafish: a resourceful vertebrate model to investigate skeletal disorders. Front Endocrinol (Lausanne) 2020;11:489. doi: 10.3389/fendo.2020.00489.358daf080da6408ca85467f2d0cc22af [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Robledinos-Anton N, Fernandez-Gines R, Manda G, Cuadrado A. Activators and inhibitors of NRF2: a review of their potential for clinical development. Oxid Med Cell Longev. 2019;2019:9372182. doi: 10.1155/2019/9372182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kim JH, Shin YC, Ko SG. Integrating traditional medicine into modern inflammatory diseases care: multitargeting by Rhus verniciflua Stokes. Mediators Inflamm. 2014;2014:154561. doi: 10.1155/2014/154561.f4395e4a2b49435da1ed8395ae13744b [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dodson M, de la Vega MR, Cholanians AB, Schmidlin CJ, Chapman E, Zhang DD. Modulating NRF2 in disease: timing is everything. Annu Rev Pharmacol Toxicol. 2019;59:555–575. doi: 10.1146/annurev-pharmtox-010818-021856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mitchell J, Lai LP, Peralta F, Xu Y, Sugamori K. β2-adrenergic receptors inhibit the expression of collagen type II in growth plate chondrocytes by stimulating the AP-1 factor Jun-B. Am J Physiol Endocrinol Metab. 2011;300:E633–E639. doi: 10.1152/ajpendo.00515.2010. [DOI] [PubMed] [Google Scholar]
- 29.Chang SH, Jang J, Oh S, et al. Nrf2 induces Ucp1 expression in adipocytes in response to beta3-AR stimulation and enhances oxygen consumption in high-fat diet-fed obese mice. BMB Rep. 2021;54:419–424. doi: 10.5483/BMBRep.2021.54.8.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Eswarakumar VP, Monsonego-Ornan E, Pines M, Antonopoulou I, Morriss-Kay GM, Lonai P. The IIIc alternative of Fgfr2 is a positive regulator of bone formation. Development. 2002;129:3783–3793. doi: 10.1242/dev.129.16.3783. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.