Abstract
People with booster vaccinations appear more prone to reinfection than those with primary series vaccination only; however, selection bias in the study design complicates interpretation.
After several years of lockdowns, social distancing, and other infection control measures, the COVID-19 pandemic was finally brought under control in most places in 2022 and the World Health Organization formally declared an end to the global health emergency on 5 May 2023. (1). In large part, this was due to the exceptionally rapid development, trialing, and rollout of the SARS-CoV-2 vaccines, some of which had proven to offer upward of 90% effectiveness in randomized controlled trials (2). In 2021, immunity against the original SARS-CoV-2 viral strain became increasingly widespread as vaccines were administered worldwide and the virus continued to spread. However, before the end of the year, evolutionary selective pressure propelled the highly contagious Omicron variant around the globe (3). Genetically different from the ancestral strain, this new variant spread rapidly, including in vaccinated people, due to its increased capacity to evade immune responses trained on the original SARS-CoV-2 strain. It quickly became apparent that, faced with Omicron, the original vaccines were no longer as effective as they had been previously (4). Some immunological studies suggested that the vaccines may cause an immune imprinting effect that hampers protection against new virus strains (5).
The study by Chemaitelly et al. (6), reported in this issue of Science Advances, analyzes population-based health data from Qatar to investigate the epidemiological evidence for such immune imprinting. The study compares the risk of reinfection with Omicron, after an earlier Omicron infection, among three groups of people with different mRNA vaccination histories before any documented natural infection. The three groups are (i) those with no vaccinations at all; (ii) those with primary series vaccinations only, i.e., two doses in total; and (iii) those with primary series vaccination followed by a booster dose, i.e., three doses in total.
Using matched study designs to ensure that the comparison groups were similar with respect to a number of potential confounders (sex, age, nationality, number of comorbidities, week of primary infection, testing method, and reason at the time of the primary infection), individuals were followed over time starting 3 months after their primary Omicron infection. The rates of reinfection during follow-up were then compared across the three groups. The study found that those with a history of two vaccine doses before their primary infection were at lower risk of reinfection with Omicron compared with unvaccinated individuals. Those with a history of primary plus a booster dose before the primary infection were also at lower risk of reinfection compared with unvaccinated individuals. For the final comparison, however, the study reports that those with a primary plus a booster dose were nearly 50% more likely to experience an Omicron reinfection than those with only two vaccine doses. It is this finding, the authors argue, that possibly points to an imprinting effect.
Individuals with only two doses benefit from having had fewer vaccine-induced immunological events that train the immune system to react narrowly against the original SARS-CoV-2 strain. According to the study, this group is therefore better equipped to mount an effective immune response targeted at Omicron, whereas the three-dose group produces a less effective response. If confirmed, then this could be a critical finding and would suggest that the administration of (non-bivalent) booster doses is not necessarily helpful and may even be harmful in certain situations. The findings should however be interpreted with caution.
The study necessarily uses an observational design as it is not feasible to conduct an experiment that would randomly allocate individuals to primary series vaccination only, or to primary series plus booster vaccination, and, further, to induce a natural Omicron infection after vaccination. Ideally, a comparison would involve people who, before vaccination, are similarly predisposed immunologically to form an effective defense against infection; however, this is not possible with the design used in this study as also pointed out by Monge et al. (7) in a recent article in The BMJ.
The comparison between the three-dose group and the two-dose group is prone to bias as those included in the three-dose group are more susceptible on average to infection because of a selection effect imposed by the design. Participants included in the three-dose group all received a booster dose relatively shortly before becoming infected with Omicron, typically about 1 month earlier. By comparison, those less susceptible to infection after their booster, who did not have a primary Omicron infection, were ineligible for study inclusion.
Of course, individuals in the two-dose group also experienced an infection before inclusion in the study despite vaccination; however, they were vaccinated considerably longer ago. Much of the protective effect of the primary vaccination series had presumably waned by the time they experienced their first Omicron infection which was typically about 8 months after the second dose. Put differently, the study effectively included individuals in the three-dose group who had demonstrated poor ability to mount an effective immune response, while those included in the two-dose group reflected a broader range of the population.
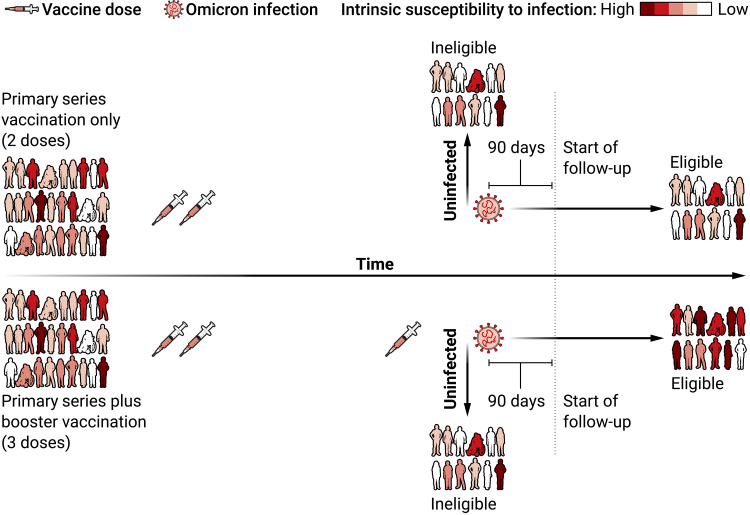
To illustrate the point further, consider the hypothetical scenario in which individuals included in the two-dose group proceeded with a booster vaccination many months after the second vaccine dose. Many would become nonsusceptible to the primary Omicron infection in the first few months after their booster dose and would now therefore be excluded from the study. The remaining individuals, who stayed susceptible and became infected despite recent booster vaccination would be included in the three-dose group. The notion that these two types of individuals, susceptible and nonsusceptible, are comparable with respect to their predisposition to reinfection seems highly unlikely, yet this is the assumption implied by the study design: The two-dose group consists of people with a broad mix of susceptibility levels, whereas the three-dose group consists of those who remained susceptible despite booster vaccination (Fig. 1).
Fig. 1. Study design comparing rates of reinfection among people with booster vaccinations and people with primary series vaccination only.
Because of the nonrandomized design, individuals who experienced a primary infection in the three-dose group, shortly after their booster dose, are more susceptible to infection than their counterparts in the two-dose group. Illustration credit: Ashley Mastin/Science Advances.
Monge et al. (7) provide a detailed methodological evaluation of the problem and illustrate through causal diagrams how the study selection procedures induce “collider bias” that creates an artifactual association between booster vaccination and reinfection (8). They then go on to replicate the biasing effect, using both simulated and empirical data, and produce estimates for the effect that is close in magnitude to the effect presented and attributed to imprinting in the study by Chemaitelly et al.
The discussion section of Chemaitelly et al. mentions a preprint version of the study by Monge et al. but largely rejects the suggestion that the imprinting effect could be a spurious finding. Chemaitelly et al. point to the fact that matching was used to balance infection exposure risk across the groups; however, matching would not have helped control for unobservable differences across the groups in the intrinsic immunological susceptibility to infection. They also argue that the imprinting effect identified in the study is not constant throughout the time of follow-up (although the hazard ratios presented by month of follow-up do not appear significantly different) and that this is consistent with an imprinting rather than a biasing effect. Last, the authors argue that their analysis comparing primary series vaccination to no vaccination should equally have produced attenuated effect estimates in the presence of selection bias, yet this analysis showed evidence of a protective vaccine effect.
Ultimately, it is hard in an observational cohort study such as Chemaitelly et al. to judge the effect of the many interacting biases that may contribute to the observed effect estimates. It is well established that infection rate comparisons in observational vaccine effectiveness studies are more likely to be valid soon after vaccination begins. This is because patterns observed long after vaccination are more highly influenced by selection effects and other biases that compound over time (9). In the study by Chemaitelly et al., rates of infection in the comparison cohorts were determined from SARS-CoV-2 test results recorded in national databases. The observed infection rates are therefore a consequence not only of incidence in the relevant comparison group but also of testing behavior which may differ between groups and over time; however, the study did adjust for testing frequency.
The study by Chemaitelly et al. addresses an important question. In an era where the circulating SARS-CoV-2 strains are almost exclusively derived from Omicron, the possibility of negative immunological imprinting after vaccination with a non-bivalent booster dose remains an important hypothesis to investigate. There is some immunological evidence to support this hypothesis; however, more robust epidemiological evidence is still warranted. Most importantly, however, there should be no question that the rollout of the vaccines, including boosters, has been central to reducing the burden of COVID-19 on mortality, morbidity, and health care systems across much of the world.
REFERENCES
- 1.World Health Organization, WHO Director-General's Opening Remarks at the Media Briefing – 5 May 2023 (World Health Organization, 2023); https://www.who.int/news-room/speeches/item/who-director-general-s-opening-remarks-at-the-media-briefing---5-may-2023.
- 2.Polack F. P., Thomas S. J., Kitchin N., Absalon J., Gurtman A., Lockhart S., Perez J. L., Marc G. P., Moreira E. D., Zerbini C., Bailey R., Swanson K. A., Roychoudhury S., Koury K., Li P., Kalina W. V., Cooper D., Frenck R. W. Jr., Hammitt L. L., Türeci Ö., Nell H., Schaefer A., Ünal S., Tresnan D. B., Mather S., Dormitzer P. R., Şahin U., Jansen K. U., Gruber W. C.; C4591001 Clinical Trial Group , Safety and efficacy of the BNT162b2 mRNA COVID-19 vaccine. N. Engl. J. Med. 383, 2603–2615 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Viana R., Moyo S., Amoako D. G., Tegally H., Scheepers C., Althaus C. L., Anyaneji U. J., Bester P. A., Boni M. F., Chand M., Choga W. T., Colquhoun R., Davids M., Deforche K., Doolabh D., du Plessis L., Engelbrecht S., Everatt J., Giandhari J., Giovanetti M., Hardie D., Hill V., Hsiao N.-Y., Iranzadeh A., Ismail A., Joseph C., Joseph R., Koopile L., Pond S. L. K., Kraemer M. U. G., Kuate-Lere L., Laguda-Akingba O., Lesetedi-Mafoko O., Lessells R. J., Lockman S., Lucaci A. G., Maharaj A., Mahlangu B., Maponga T., Mahlakwane K., Makatini Z., Marais G., Maruapula D., Masupu K., Matshaba M., Mayaphi S., Mbhele N., Mbulawa M. B., Mendes A., Mlisana K., Mnguni A., Mohale T., Moir M., Moruisi K., Mosepele M., Motsatsi G., Motswaledi M. S., Mphoyakgosi T., Msomi N., Mwangi P. N., Naidoo Y., Ntuli N., Nyaga M., Olubayo L., Pillay S., Radibe B., Ramphal Y., Ramphal U., San J. E., Scott L., Shapiro R., Singh L., Smith-Lawrence P., Stevens W., Strydom A., Subramoney K., Tebeila N., Tshiabuila D., Tsui J., van Wyk S., Weaver S., Wibmer C. K., Wilkinson E., Wolter N., Zarebski A. E., Zuze B., Goedhals D., Preiser W., Treurnicht F., Venter M., Williamson C., Pybus O. G., Bhiman J., Glass A., Martin D. P., Rambaut A., Gaseitsiwe S., von Gottberg A., de Oliveira T., Rapid epidemic expansion of the SARS-CoV-2 Omicron variant in Southern Africa. Nature 603, 679–686 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Andrews N., Stowe J., Kirsebom F., Toffa S., Rickeard T., Gallagher E., Gower C., Kall M., Groves N., O’Connell A.-M., Simons D., Blomquist P. B., Zaidi A., Nash S., Aziz N. I. B. A., Thelwall S., Dabrera G., Myers R., Amirthalingam G., Gharbia S., Barrett J. C., Elson R., Ladhani S. N., Ferguson N., Zambon M., Campbell C. N. J., Brown K., Hopkins S., Chand M., Ramsay M., Bernal J. L., COVID-19 vaccine effectiveness against the Omicron (B.1.1.529) variant. N. Engl. J. Med. 386, 1532–1546 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Reynolds C. J., Pade C., Gibbons J. M., Otter A. D., Lin K.-M., Sandoval D. M., Pieper F. P., Butler D. K., Liu S., Joy G., Forooghi N., Treibel T. A., Manisty C., Moon J. C.; COVIDsortium Investigators; COVIDsortium Immune Correlates Network, Semper A., Brooks T., Mcknight Á., Altmann D. M., Boyton R. J., Abbass H., Abiodun A., Alfarih M., Alldis Z., Altmann D. M., Amin O. E., Andiapen M., Artico J., Augusto J. B., Baca G. L., Bailey S. N. L., Bhuva A. N., Boulter A., Bowles R., Boyton R. J., Bracken O. V., O’Brien B., Brooks T., Bullock N., Butler D. K., Captur G., Carr O., Champion N., Chan C., Chandran A., Coleman T., de Sousa J. C., Couto-Parada X., Cross E., Cutino-Moguel T., D’Arcangelo S., Davies R. H., Douglas B., Genova C. D., Dieobi-Anene K., Diniz M. O., Ellis A., Feehan K., Finlay M., Fontana M., Forooghi N., Francis S., Gibbons J. M., Gillespie D., Gilroy D., Hamblin M., Harker G., Hemingway G., Hewson J., Heywood W., Hickling L. M., Hicks B., Hingorani A. D., Howes L., Itua I., Jardim V., Lee W.-Y. J., Jensen M., Jones J., Jones M., Joy G., Kapil V., Kelly C., Kurdi H., Lambourne J., Lin K.-M., Liu S., Lloyd A., Louth S., Maini M. K., Mandadapu V., Manisty C., Knight Á. M., Menacho K., Mfuko C., Mills K., Millward S., Mitchelmore O., Moon C., Moon J., Sandoval D. M., Murray S. M., Noursadeghi M., Otter A., Pade C., Palma S., Parker R., Patel K., Pawarova M., Petersen S. E., Piniera B., Pieper F. P., Rannigan L., Rapala A., Reynolds C. J., Richards A., Robathan M., Rosenheim J., Rowe C., Royds M., West J. S., Sambile G., Schmidt N. M., Selman H., Semper A., Seraphim A., Simion M., Smit A., Sugimoto M., Swadling L., Taylor S., Temperton N., Thomas S., Thornton G. D., Treibel T. A., Tucker A., Varghese A., Veerapen J., Vijayakumar M., Warner T., Welch S., White H., Wodehouse T., Wynne L., Zahedi D., Chain B., Moon J. C., Immune boosting by B.1.1.529 (Omicron) depends on previous SARS-CoV-2 exposure. Science 377, eabq1841 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chemaitelly H., Ayoub H. H., Tang P., Coyle P. V., Yassine H. M., Al Thani A. A., Al-Khatib H. A., Hasan M. R., Al-Kanaani Z., Al-Kuwari E., Jeremijenko A., Hassan Kaleeckal A., Latif A.N., Shaik R. M., Abdul-Rahim H. F., Nasrallah G. K., Al-Kuwari M. G., Butt A. A., Al-Romaihi H., Al-Thani M. H., Al-Khal A., Bertollini R., Abu-Raddad L. J., History of primary-series and booster vaccination and protection against Omicron reinfection. Sci. Adv. 9, eadh0761 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Monge S., Pastor-Barriuso R., Hernán M. A., The imprinting effect of covid-19 vaccines: An expected selection bias in observational studies. BMJ 381, e074404 (2023). [DOI] [PubMed] [Google Scholar]
- 8.Catalogue of Bias Collaboration; H. Lee, J. K. Aronson, D. Nunan, Collider Bias (2019); https://catalogofbias.org/biases/collider-bias/.
- 9.World Health Organization, Evaluation of COVID-19 vaccine effectiveness: Interim Guidance (World Health Organization, 2021); https://www.who.int/publications/i/item/WHO-2019-nCoV-vaccine_effectiveness-measurement-2021.1.