Abstract
Early-life adversities are associated with altered defensive responses. Here, we demonstrate that the repeated cross-fostering (RCF) paradigm of early maternal separation is associated with enhancements of distinct homeostatic reactions: hyperventilation in response to hypercapnia and nociceptive sensitivity, among the first generation of RCF-exposed animals, as well as among two successive generations of their normally reared offspring, through matrilineal transmission. Parallel enhancements of acid-sensing ion channel 1 (ASIC1), ASIC2, and ASIC3 messenger RNA transcripts were detected transgenerationally in central neurons, in the medulla oblongata, and in periaqueductal gray matter of RCF-lineage animals. A single, nebulized dose of the ASIC-antagonist amiloride renormalized respiratory and nociceptive responsiveness across the entire RCF lineage. These findings reveal how, following an early-life adversity, a biological memory reducible to a molecular sensor unfolds, shaping adaptation mechanisms over three generations. Our findings are entwined with multiple correlates of human anxiety and pain conditions and suggest nebulized amiloride as a therapeutic avenue.
ASIC channels and harm detection are enhanced transgenerationally while inhaled amiloride reverses the responses.
INTRODUCTION
How living organisms respond and adapt to adversities, how they encode a biological memory of past hardships, and which molecular mechanisms allow for transferring the resulting adaptations to the next generations are key questions that remain only partially answered. Addressing and solving these questions yield ramifications spanning the fields of evolutionary biology, genetics, and health sciences (1–3), with early-life adversities (ELAs) bearing particular salience to both the contexts of general life sciences (4) and human medicine (5).
Preclinical models of ELAs provide powerful settings to investigate the elicitation and enhancement of defensive responses (6). Because mammals are crucially dependent on offspring-caregiver interaction, some protocols were designed to model early maternal separation and study the enhancement of defensive reactions to harmful stimuli and the biological changes that are associated with this specific form of ELA (7, 8).
The neonatal maternal separation and the repeated cross-fostering (RCF) protocols of early maternal separation share the ability of evoking enhanced, longitudinally stable respiratory and behavioral reactions to air mixtures enriched with CO2 (9, 10). The blood-acidifying, suffocative stimulus of inhaled CO2, hence, hypercapnia, is aversive to most mammals including humans (11). Physiological adaptive responses to continuously inhaled air enriched with high [CO2] (between 4 and 6%, compared to 0.04% atmospheric [CO2]) consist of enhanced ventilation, arousal, anxiety, and avoidant behavior and are comparable in rodents and humans (12, 13). Through the key mechanism of tidal volume (Vt) augmentation (13), the respiratory responses to increased [CO2] buffer against blood and brain fluids’ acidification that derive from transient hypercapnia (13), with anxiety and/or avoidant behavior promoting escape from suffocative, metabolically unfavorable high [CO2] environments (13, 14). Similarly to other paradigms of early maternal separation such as the neonatal maternal deprivation procedure (15), the RCF is also associated with enhanced reactivity to a major class of harmful probes, namely, painful stimuli, as indicated by increased reactivity to formalin injection into, and heating of, the forepaw surface (16, 17).
In addition to studying how ELAs associate with changes in physiology and behavior (18), work has been conducted on ELA-associated alterations in brain circuitry and connectivity (18, 19), cellular architecture (20, 21), and gene expression (22). Stemming from the finding of hypothalamic-pituitary-adrenal (HPA) axis variations associated with some models of ELAs, numerous studies focused on specific glucocorticoid receptor signaling genes (23), while a smaller proportion of investigations addressed at a genome-wide level the enrichments, biological pathways, and transcriptional (24) patterns that are associated with different types of ELAs.
Genome-wide searches into the medulla oblongata (MO; a brain structure critical for detection of extracellular pH changes, respiratory control, and nociception) (13, 25–27) of RCF-exposed mice revealed H3Ac, H3K27me3, and H3K4me3 histone marks’ modifications that point to the enhancement of several neurodevelopmental genes (16). This revealed the enrichment of the acid-sensing ion channel gene-1 (Asic1), with reverse transcription polymerase chain reaction and serial analysis of gene expression analyses confirming increased MO expression of ASIC1 channels among adult animals that had been exposed to RCF as pups [i.e., RCF-exposed first generation (F0-RCF) mice] (16).
ASICs are activated by H+ in extracellular fluids and are crucial to detect hypercapnia-driven central nervous system (CNS) acidification (28, 29), drive respiratory chemoreception (30, 31), and coordinate nociception (32). Brain neurons express ASIC1a, ASIC2a, and ASIC2b subunits, as well as ASIC3, in variable and overlapping distributions (31–33). Specifically, the ASIC1 and ASIC2 form dimers that drive pH detection (31, 32), with ASIC subunits’ interplays and H+-gated currents bridging anxiety physiology to pathophysiology (33–35). Coherently, loss of ASIC2/ASIC1 reduces unconditioned (“freezing” responses to CO2) and conditioned (contextual fear) anxious responses (34, 35). Consistent with these findings, a single dose of ASIC-antagonist amiloride (nebulized to cross the blood-brain barrier), but not intraperitoneal injections, redirects adult F0-RCF mice’s respiratory responses to CO2 and thermal nociception to normality, indicating a role for altered CNS ASICs (17) in these responses. The role of ASICs in nociception is best supported by studies of ASIC3 (31, 32).
Consistent with their implication in chemoreception, respiration, and nociception (31), ASIC1, ASIC2, and ASIC3 channels are well represented in the nervous system. In addition to the MO (36), the periaqueductal gray (PAG), which is fundamental to coordinate respiratory and nociceptive responses in both rodents and humans (37–39), large portions of the cortex (40), the cerebellum (40), the dorsal root ganglion (DRG) (41, 42), and the amygdala (35) [a more recently identified chemosensor that detects CO2 and acidosis to elicit fear behavior (35)], share a representation of ASIC channels. ASIC1, ASIC2, and ASIC3 are expressed in both neuronal and nonneuronal cells of the MO, PAG, and DRG; both neuronal and nonneuronal cells in these structures subserve defensive responses to aversive stimuli such as hypercapnia and nociceptive stimulation via chemoreception (34, 36, 43, 44), although the role of ASICs in nonneuronal, compared to neuronal, cells is less understood (45).
Independent investigations provide support to the association between ASICs’ enrichment and ELAs exemplified by the RCF paradigm (16): These studies showed the association between early maternal deprivation, enhanced nervous system expression of ASIC1, and enhanced visceral pain sensitivity (41), as well as the association between prenatal maternal stress and offspring’s altered methylation of Asic1 via CpG island demethylation and gastric hypersensitivity (another form of enhanced sensitivity to visceral pain) (42). Furthermore, crossings of F0-RCF and normally reared control (CT) animals (F0-CT) showed the intergenerational matrilineal transmission of enhanced respiratory responses to CO2 stimulation (46); this went from females of the F0-RCF to their normally reared F1 progeny (F1-RCF) of both sexes. This intergenerational transmission also went hand in hand with clusters of differentially methylated DNA regions that included the Asic2 gene (46) for both F0-RCF and F1-RCF animals, as revealed by MO genome-wide intergenerational investigations of multiple F0-F1 RCF versus F0-F1 CT lineages (46). Thus, following different forms of early interference with maternal care including the RCF paradigm, a biological memory appears to arise: This can be mapped and further clarified by assessing harm-detecting traits (i.e., respiratory reactivity to CO2 and nociception) and the expression of ASIC channels in the nervous system.
Several crucial, unexplored questions arise here. First, to support a role for ASICs as a molecular substrate of the biological memory for ELA and for RCF specifically, it becomes essential to demonstrate that both the transcripts’ and the phenotypes’ enhancements can be documented trigenerationally, whereby a F0 has experienced RCF and the two successive generations (F1 and F2) have been raised normally. This means testing whether the environmental adversity epitomized through RCF affects not only the neural system of the exposed F0-RCF generation but also the functions of the successive unexposed F1-RCF generation (indicating intergenerational transmission by an impact on the F1 generation’s germ) and the functions of the following unexposed F2-RCF generation (indicating transgenerational transmission) (47, 48). Second, it becomes essential to trigenerationally map the transcripts of the three different types of ASICs across several different neural regions so that a sensible functional connection between transcripts’ enhancements and brain localization can emerge. Third, it is important to show that the aforementioned trigenerational enhancements can be modulated by appropriate molecular tools, such as pharmacological agents.
Here, we investigated the respiratory response to 6% CO2 stimulation, thermal nociception, mechanical sensitivity, the distribution of Asic1, Asic2, and Asic3 gene transcripts by RNAscope, and the modulatory effects of ASIC-antagonist amiloride in three generations of outbred mice, whereby the F0 had experienced RCF, and the two successive generations (F1-RCF and F2-RCF) had been raised normally by their biological mothers. The Asic transcripts were examined in several regions involved in pH detection, respiration, and nociception and encompassed the MO, the PAG, the V2 visual cortex, the cerebellar Purkinje cell layer, and the DRG. We hypothesized that after application of the RCF paradigm to F0 animals, a transgenerational matrilineal transmission of CO2-driven hyperventilation and enhanced nociception would become observable, in association with transgenerational enhancements of ASIC transcripts in brain areas that are essential for chemoreception and that the responses would be reversible by application of nebulized ASIC-antagonist amiloride.
RESULTS
RCF-induced altered homeostatic responses across three generations
In both animals and humans exposed to ELAs (49), multiple homeostatic responses and related interoceptive functions—encompassing respiratory and nociceptive parameters—become enhanced (49). Thus, given previous results with respiratory and nociceptive challenges among RCF-exposed animals (10, 16, 17, 46, 50), respiratory reactions to 6% CO2-air breathing and nociceptive mechanical and thermal reactions are grouped here under the overarching category of homeostatic responses. We hypothesized that homeostatic responses would be enhanced in all three generations of RCF mice.
Our results indeed show that compared to animals in a trigenerational (CT-F0, CT-F1, and CT-F2) normally reared lineage, RCF-exposed (F0-RCF) animals and their normally reared offspring (F1-RCF and F2-RCF) share enhanced homeostatic reactivity, as they are more sensitive to the aversive stimuli of hypercapnia, mechanosensory stimulation, and thermal stimulation (Fig. 1).
Fig. 1. RCF is associated with altered homeostatic responses trigenerationally (data are represented as means + SEM).
(A) Association (n = 77) between 6% CO2-air breathing and higher Vt compared to air breathing (Hotelling’s trace F1,65 = 159.0, P ≤ 0.0001), with interaction between lineage (RCF/CT) and Vt during 6% CO2 breathing (Hotelling’s trace, F1,65 = 21.82, P ≤ 0.0001) by repeated-measures analyses of variance (ANOVA-R). There were no other significant associations owing to generation (F0, F1, and F2), sex, or any possible combinations of the predictors. (B) Association (n = 102) between shorter latency of paw withdrawal at thermal stimulation (Hargreaves’ test) and the RCF lineage (F1 =199.70, P ≤ 0.001) by univariate GLM (F11 = 22.36, P ≤ 0.001). There was also an association with generation (F0, F1, and F2: F2 = 8.09, P ≤ 0.001): Tests of within- and between-groups’ variance decomposition showed that the latter, generation effect was caused by a local increase in paw withdrawal latency among F1-CT animals and revealed no lineage-by-generation interaction and no significant effect in multiple comparisons of between-generations’ effect (by Scheffe’s tests). No other significant associations owing to sex or to interactions among any of the independent variables emerged, including lineage-by-generation interaction. (C) Association (n = 81) between shorter latency of paw withdrawal at mechanosensitive stimulation [simplified up-down (SUDO)–von Frey’s test] and the RCF lineage (F1 = 62.26, P ≤ 0.001) by univariate GLM (F11 = 7.99, P ≤ 0.001). *P < 0.05, ***P < 0.001. There was also an association (D) with female sex (F1 = 4.01, P ≤ 0.05) but no other significant associations owing to generation (F0, F1, and F2) or interactions among any of the independent (lineage, generation, and sex) variables.
Respiratory response
Consistent with previous evidence that the RCF procedure does not alter baseline respiratory parameters during normal air breathing (10, 16, 17, 46, 50), a univariate preliminary general linear model (GLM) analysis of Vt during baseline (air) showed no significant effect (linear or in any combination of interactions) of lineage (RCF/CT), generation (F0/F1/F2), or sex.
During 6% CO2-air breathing, Vt was consistently enhanced among animals of the RCF lineage, without differences between generations (Fig. 1A). This means that while Vt during baseline air breathing was comparable among animals of different lineages, generations, and sexes, a larger compensatory respiratory enhancement during 6% CO2-air breathing was present, consistent and comparable, across three generations of the RCF lineage.
Nociceptive response
We found a shorter latency of paw withdrawal in animals of the RCF lineage during thermal stimulation (Fig. 1B). Further analyses showed that, despite a local increase in paw withdrawal latency among F1-CT animals, there was no lineage-by-generation interaction nor any other significant effects across multiple between-generations’ comparisons (Fig. 1B). We also found a shorter latency of paw withdrawal during mechanosensitive stimulation in animals of the RCF lineage, without differences owing to generation (Fig. 1C). There was also a smaller association with shorter latency of paw withdrawal and the female sex (Fig. 1D) but no other significant association owing to generation (F0/F1/F2) or any interaction among any of the independent variables (see also Fig. 1, C and D).
Enhanced Asic transcripts in specific brain areas across three generations
The findings of enhanced respiratory reactivity to hypercapnia and enhanced nociception across three generations of the RCF lineage, as well as previous work with ELAs, RCF, and ASICs (16, 46), lead to three specific and nested hypotheses, i.e., that in association with RCF: (i) Asic transcripts become enhanced, (ii) in brain areas that detect pH changes (hence, in the regulation of respiration and nociception), and (iii) both (i) and (ii) manifest with a trigenerational transmission pattern.
To tackle these three nested hypotheses, we analyzed Asic transcripts in three generations of RCF and CT mice in two CNS regions (MO and PAG) that play preeminent roles in pH detection (hence, in the regulation of respiration and nociception), two CNS regions (visual cortex and cerebellum) that play integrative roles in interoception (51), and one peripheral station of interoception (DRG).
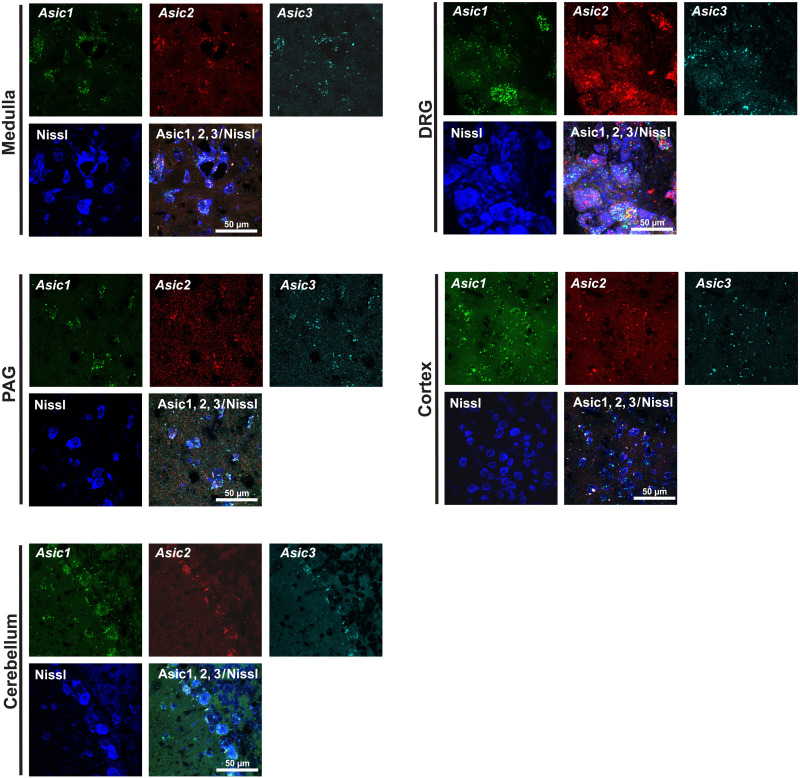
Across the aforementioned five structures, neuronal and nonneuronal cells were identified by the 4′,6-diamidino-2-phenylindole (DAPI) and Nissl methods (see Materials and Methods for further details). Figure 2 depicts the Asic1, Asic2, and Asic3 transcripts’ visualizations across all five structures. Preliminary analyses by GLMs (see also the “Statistical analyses” section) showed no differences in number of cells (neuronal or nonneuronal) between lineages (RCF/CT), between sexes, or among generations (F0/F1/F2) across the five structures analyzed here.
Fig. 2. ASC1, ASC2, and ASC3 transcripts, as revealed by in situ hybridization RNAscope across central and peripheral nervous system structures.
The figure is divided by structure (medulla, PAG, cerebellum, DRG, and cortex) and shown by Asic subtype (Asic1, Asic2, and Asic 3) with nuclear (DAPI) and neuronal (Nissl NeuroTrace) staining.
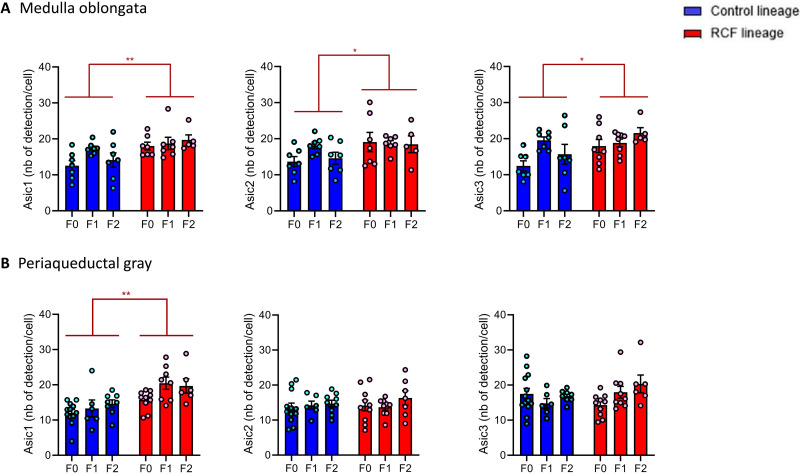
We found a pattern of trigenerational enhancement of Asic transcripts associated with the RCF lineage in the neuronal populations of both the MO and the PAG (Fig. 3). Specifically, Fig. 3A shows that, compared to animals belonging to a trigenerational (CT-F0, CT-F1, and CT-F2) normally reared lineage, RCF-exposed (F0-RCF) animals and their normally reared offspring (F1-RCF and F2-RCF) had enhanced Asic1, Asic2, and Asic3 transcripts in neurons but not in nonneuronal cells (fig. S1) within the MO, without significant differences owing to which generation they belonged to. Similar changes were observed for the PAG, with the exception that the multivariate trigenerational effect was sustained solely by the Asic1 transcripts [see legends of Fig. 3 (A and B) for further details].
Fig. 3. RCF is associated with enhanced ASIC expression in neurons of the MO and PAG matter trigenerationally (data are represented as means + SEM).
(A) Multivariate (n = 38) association between ASIC transcripts and the RCF lineage by multivariate analysis of variance (MANOVA) (Hotelling’s trace, F3,30 = 4.5, P ≤ 0.01) in the neuronal population of the MO. Tests of between-subjects’ effects showed significant univariate effects for all three ASIC populations (ASIC1: F5 = 4.00, P ≤ 0.007; ASIC2: F5 = 2.50, P ≤ 0.05; ASIC3: F5 = 3.20, P ≤ 0.02) and the significant univariate effect of RCF lineage on all three ASIC populations (ASIC1: F1 = 11.35, P ≤ 0.002; ASIC2: F1 = 7.60, P ≤ 0.01; ASIC3: F1 = 7.25, P ≤ 0.01). There was no multivariate effect of generation [Hotelling’s trace, F6,58 = 1.2, P = not significant (NS)] or lineage-by-generation interaction; there was no univariate effect of generation on any of the three ASIC subtypes or any significant lineage-by-generation univariate interaction on any of the three ASIC populations. No significant multivariate effects of lineage, generation, or lineage-by-generation interaction were found on ASIC1, ASIC2, or ASIC3 transcripts of nonneuronal MO cells by MANOVA. See also the Supplementary Materials for follow-up analyses of MANOVA results. (B) Multivariate association (n = 47) between ASIC transcripts and the RCF lineage by MANOVA (Hotelling’s trace, F3,39 = 5.80, P ≤ 0.002) in the neuronal population of the PAG. Tests of between-subjects’ effects showed only the significant univariate effect for ASIC1 (ASIC1: F5 = 5.60, P ≤ 0.001) and the significant univariate effect of RCF lineage (F1 = 17.52, P ≤ 0.001) on ASIC1. *P < 0.05, **P < 0.01. There were no multivariate effects of generation (Hotelling’s trace, F6,76 = 1.60, P = NS) or lineage-by-generation interactions. No significant multivariate effect of lineage, generation, or lineage-by-generation interaction was found on ASIC1, ASIC2, or ASIC3 transcript of nonneuronal PAG cells by MANOVA.
Follow-up analyses via discriminant analysis and planned multivariate comparisons (see the Supplementary Materials) confirmed these multivariate analysis of variance (MANOVA) results, with MO ASIC1 providing the strongest contribution to differentiate RCF from CT mice and no evidence of significant generational effects to discriminate among animals in the experiments.
To assess the extensiveness of these changes, we measured Asic transcripts levels in neuronal and nonneuronal cells of other brain areas. In contrast to the altered levels found in MO and PAG neurons, there were no significant changes in Asic1, Asic2, or Asic3 in neurons of the V2 visual cortex or the cerebellum, owing to lineage, generation, or lineage-by-generation interaction, according to multivariate GLMs (fig. S2), indicating a link between enrichment in Asic expression and brain regions with primary homeostatic functions.
To test whether changes in Asic expression and, in particular, nociceptive hypersensitivity were central or peripheral, we measured the mRNA levels in primary afferents neurons at the level of the DRG. There were no significant differences in Asic1, Asic2, or Asic3 transcripts in DRG cells owing to lineage (figs. S1 and S2). A multivariate effect of “generation” was detected, sustained by a local trough in Asic3 transcript signals yielded by F1 animals of both the RCF and CT lineages, as shown by successive tests of within- and between-group variance (see also fig. S1). These results with the DRG indicate that the enhanced sensory responses observed in the RCF lineage (Fig. 1) involved primarily central rather than peripheral nervous system adaptations.
Single-dose nebulized amiloride renormalization of homeostatic responses across three generations
Amiloride is an antagonist of ASICs (31) and a relatively large molecule that accesses the brain better when nebulized (17). To establish a causal link between enhanced Asic expression across generations and heightened homeostatic sensitivities, we assessed the ability of single-dose nebulized amiloride versus saline to modulate the threshold of response to aversive stimuli in the RCF lineage. We found that the enhanced homeostatic responses to hypercapnia, as well as mechanical and thermal stimulation in F0-RCF animals and their normally reared offspring (F1-RCF and F2-RCF), were reduced by nebulized amiloride compared to saline, in that after amiloride, the responses of the RCF lineage became indistinguishable from those of animals belonging in a trigenerational (F0, F1, and F2) normally reared (CT) lineage.
Specifically, we found that, compared to saline, a single dose of amiloride nebulized before 6% CO2-air breathing reduced Vt consistently in RCF animals, without differences owing to generation (Fig. 4A). Similarly, we found that a single dose of nebulized amiloride increased the paw withdrawal threshold response to mechanical stimulation and to thermal stimulation in RCF animals (Fig. 4, B and C).
Fig. 4. Single-dose pretreatment with nebulized amiloride normalizes homeostatic responses in RCF animals trigenerationally (data are represented as means + SEM).
(A) Association (n = 46) between Vt during 6% CO2 breathing and amiloride/saline pretreatment (F1,40 = 15.3, P ≤ 0.001) and interaction between RCF lineage and amiloride pretreatment (Hotelling’s trace, F1,40 = 23.93, P ≤ 0.001) by ANOVA-R. There were no other significant associations owing to generation (F0, F1, and F2) or any possible combinations of the predictors. (B) Association (n = 130) between latency of paw withdrawal at mechanosensitive stimulation (SUDO–von Frey’s test) and the RCF lineage (F1 = 31.2, P ≤ 0.001), pretreatment with amiloride (F1 = 15.0, P ≤ 0.001), and the interaction between the former two factors (F1 = 9.70, P ≤ 0.002) by univariate GLM (F23 = 5.30, P ≤ 0.001). No other significant effects (linear or interactive) emerged from these analyses. (C) Association (n = 152) between latency of paw withdrawal at thermal stimulation (Hargreaves’ test) and the RCF lineage (F1 = 26.80, P ≤ 0.0001), pretreatment with amiloride (F1 = 263.14, P ≤ 0.0001), and the interaction between the former two factors (F1 = 37.00, P ≤ 0.0001) by univariate GLM (F21 = 27.60, P ≤ 0.0001). **P < 0.01, ***P < 0.001. No other significant effects (linear or interactive) emerged from these analyses. See also the Supplementary Materials for follow-up analyses of MANOVA results.
Multivariate contrasts via discriminant analysis and planned multivariate comparisons (see the Supplementary Materials) confirmed the MANOVA results with the Hargreaves’ thermal stimulation test, including the lack of significant intralineage differences attributable to generation, to discriminate among animals in the experiment and showed that amiloride exerted a comparable effect on Hargreaves’ responses among RCF and CT animals (see also the Supplementary Materials). These findings indicate that ASICs are necessary to explain the enhanced homeostatic responses proper of the RCF lineage across all three generations and link the transgenerational transmission of the traits to the enhanced ASIC expression in the MO and PAG.
DISCUSSION
Here, we report trigenerational associations between the RCF lineage and (i) the homeostatic harm detection responses of heightened respiratory reactivity to CO2-enriched air mixtures, heightened sensitivity to mechanical stimulation, and heightened sensitivity to thermal stimulation; (ii) the enhancement of Asic1, Asic2, and Asic3 mRNA transcripts, in the neurons of two brain structures (MO and PAG) that are cornerstones of respiratory and pain physiology; (iii) the reversibility of all the three abovementioned homeostatic harm detection phenotypes to normalcy by administration of a single dose of ASIC-antagonist amiloride, nebulized immediately before respiratory and nociceptive testing. The associations with the RCF lineage occurred in a steady pattern, as indicated by the absence of relevant, among-generation differences and the absence of lineage-by-generation interactions. This means that across all the three levels (phenotypic, mRNA transcripts, and pharmacologic modulation of homeostatic responses) of investigation, (i) normally reared RCF offspring (F1-RCF and F2-RCF) were reciprocally comparable and did not differ significantly from their F0-RCF parents and grandparents and (ii) results with the RCF lineage were systematically different from those yielded by three generations (F0-CT, F1-CT, and F2-CT) of normally reared animals.
The trigenerational effects reported here cannot be ascribed to suboptimal nursing, licking, or grooming among RCF surrogate mothers, as all these fundamental components of the maternal environment proved to be indistinguishable from those of CT females across multiple cycles and generations of RCF procedures and studies (10, 16, 46, 50). Likewise, inasmuch as the RCF protocol is unrelated to changes in circulating corticosterone levels (10), expression of glucocorticoid or mineralcorticoid receptors (50), or significant enrichments of HPA axis–associated genes, (16, 46), these results cannot be ascribed to HPA alterations.
Because epigenetic transmission is constrained in mammals (2, 52) and may be misattributed to some types of genetic transmission (2), we interpret our findings as being indicatively, not incontrovertibly, explainable by epigenetic mechanisms. We nonetheless offer the parsimonious notion that following early interference with maternal care via the RCF paradigm, a biological memory is evoked, as it appears to be matrilineally transmitted from the first F0-RCF to the two successive normally reared F1-RCF and F2-RCF. This could be substantiated at the phenotypic level via three distinct harm-detecting homeostatic responses (CO2-evoked hyperventilation, thermal nociception, and mechanosensitivity). Coherently, distinct between-lineage (RCF versus CT) and stable within-generations (F0-F1-F2) patterns of Asic1, Asic2, and Asic3 mRNA transcripts differentiated F0-RCF, F1-RCF, and F2-RCF from F0-CT, F1-CT, and F2-CT animals in the neurons of the MO and PAG, two brain structures that are pivotal mediators of chemoreception, respiration, nociception, and anxiety (11, 13, 25, 28, 37–39). Consistent with the notion that brain ASICs mediate the harm-detecting homeostatic responses measured in this study, a single dose of ASIC-antagonist amiloride (nebulized to easily access the brain) promptly reestablished normal responses among RCF-lineage animals and consistently so across generations. Together with the inability of intraperitoneally administered amiloride to restore normal ventilatory responses to 6% CO2 in F0-RCFanimals (17), the lack of significant DRG Asic mRNA transcript signals supports a central, not peripheral, role of ASIC channels in the responses we studied.
In support to our findings, enhanced expression of ASIC1 and enhanced visceral pain sensitivity have been reported in association with early maternal deprivation (41), and prenatal maternal stress is associated with offspring ASIC1 enhancement (via Asic1 CpG island demethylation) and gastric hypersensitivity (another form of enhanced sensitivity to visceral pain) (42); ASIC1 enhancements in those studies (41, 42) were, however, reported for only one generation of rats and circumscribed to the DRG and the spinal dorsal horn.
Compared to the more intensively investigated ASIC1 and ASIC2, the pathophysiology of ASIC3 has been less explored and appears relatively less clear. Our results broadly align with the roles described for ASIC3 that are mostly in nociception (31, 32). Moreover, while amiloride modulated RCF-enhanced thermal and mechanical nociception, it also moderated control animals’ Hargreaves’ responses, suggesting differential relevance of ASIC-driven mechanisms in these two different types of nociception.
Inasmuch as fundamental processes such as respiratory regulation, response to hypercapnia, and nociception rely on multiple CNS regions, pathways, and neurotransmitters, our results are relevant to one specific facet of these complex and interrelated functions and should not be considered immediately or necessarily transferable to other systems. For instance, neurotransmitters such as serotonin (13, 53) and acetylcholine (13, 54, 55) are important for respiratory regulation including CO2-driven hyperventilation, glial cells are important mediators of chemoreception (56), and pH-sensing astrocytes (57) adapt flexibly to some environmental adversities (58). Thus, while our experiments did not detect significant changes in Asic mRNA transcripts of the nonneuronal populations, the role of some nonneuronal cells in chemoreception (56, 57) (including pH sensing, hence, respiration and nociception) has been documented, confirming the multilayered complexity of these systems. By the same token, variability has been reported for thermal nociception among offspring exposed to different maternal separation protocols, with operational differences among varying protocols (59) reflecting the heterogeneity of offspring responses that may result in enhanced/reduced sensitivities compared to control animals across different maternal separation protocols. By providing a viable molecular basis (i.e., ASICs enhancement) for the finding of RCF-associated increase in phenotypic variance and heritability of Vt during hypercapnia (10), our present results align with the broad prediction that the enhancement of interindividual variation (60, 61) is a key adaptive mechanism of response to environmental challenges.
In our particular experimental context, this takes place through the transgenerational enrichment of specific genes in dedicated brain areas that detect suffocative and painful stimuli and promote related defensive responses. The ultimate value of these surges in phenotypic variability in response to a specific ELA is not necessarily interpretable as adaptive, as this is eventually dictated by the environmental niches where an organism dwells. These concepts need to be borne in mind before discussing the many intersections between these findings and human behavioral neurophysiology (49, 62, 63) and the potential for translational applications and health care.
In humans, a continuum connects (i) childhood parental separation/loss, (ii) childhood separation anxiety, (iii) emotional and respiratory hypersensitivity to CO2-enriched air mixtures, and (iv) adult panic disorder (64). This developmental trajectory is underlain by shared genetic risk factors, with childhood parental separation/loss significantly contributing to increase an individual’s liability to all three (ii) to (iv) phenotypes, and ELAs exerting the effect of heightening genetic variance—hence, heritability—for CO2 hypersensitivity (65–68). Consistent with these human data, F0-RCF youngster and adult mice yield higher heritability for Vt responses to 6% CO2, and F0-RCF pups display higher separation anxiety indices (ultrasound vocalizations) during isolation tests than their normally reared F0-CT counterparts (10).
Human brain responses to CO2 stimulation follow a pattern of activation that originates from the brainstem’s MO and are a source of individual differences, with panic disorder patients at one extreme of the distribution of response to hypercapnia (11, 69). Consistently, brain extracellular acidic environment is linked to human liability to panic and related conditions (70, 71).
Human brain imaging studies also documented a largely shared, emotion-related brain network that is common to anxiety, dyspnea, and pain (63), and human behavioral genetic studies indicate shared genetic and unique environmental factors—rather than direct phenotypic causation (i.e., pain causes anxiety or vice versa)—as the most likely explanation for the high comorbidity between anxiety disorders and chronic primary pain (72–74).
Human Asic1 and Asic2 gene polymorphisms are associated with panic-like responses to CO2 respiratory challenges (75, 76), panic disorder (77, 78), amygdala volume and activity (78), and nociception (79). While data associating Asic polymorphisms to human anxiety disorders and key respiratory areas such as the MO or the PAG are still lacking, a brain imaging study linked the human ortholog of Asic1a to panic disorder and amygdala function (78).
The Asic1 gene was found associated with panic disorder (7, 47) in a genome-wide association study. Among monozygotic twin pairs discordant for anxious responses to CO2 stimulation, differential patterns of Asic2 gene DNA methylation discriminate responders from nonresponders (46), and the Asic2 gene yielded one of the strongest signals in the recent, first genome-wide study of human nociception (79).
In addition to the MO and PAG, other ASIC-expressing brain areas relevant in stress/emotional responses such as the amygdala or the hippocampus (32, 33, 35) could affect breathing and nociception, and these brain regions have not been investigated here. Future experiments extended to other brain areas, using cell-specific approaches and applying regional deletion of ASICs, may further clarify the effect of RCF across generations and anatomical structures.
Moreover, the nonsignificant results with ASIC transcripts in nonneuronal cell types/glia do not rule out the possible involvement of these cellular populations in the respiratory and nociceptive effects reported here. Inasmuch as transcripts measurements capture a single time point, altered expression patterns may still reveal, if measured at points in time other than those we adopted in our protocol.
The possible confounding effects of altered HPA function (10, 16, 46) or maternal nursing, licking, or grooming among RCF surrogate mothers were a priori ruled out here, in the light of multiple replicated proofs—which included intergenerational assessments (10, 16, 46, 50)—of their lack of association with the RCF paradigm. While de novo, transgenerational effects of these confounders appear unlikely, they were not formally assessed here.
In conclusion, our data point at the enhancement of ASICs as a molecular basis for responses to ELA in mammals, which could be extended to man; this mechanism has an explanatory value for both adaptive and maladaptive responses, depending on an individual’s biological setup, developmental history, and their environmental niche. In this context, our findings, together with the safety and distribution properties of amiloride nasal spray in humans (80–82), support this formulation as a viable individualized therapeutic avenue for panic disorder and related conditions, as well as for some pain states, in the foreseeable future.
MATERIALS AND METHODS
Animals
Swiss outbred mice (Harlan, Canada) were used in all experiments. All animals took part in only one of the various experiments outlined in this article so that they were naive. To prevent litter effects, a maximum of two animals per litter were allowed in the same experimental group. All experiments were conducted in accordance with the guidelines approved by the Canadian Council of Animal Care. All protocols were approved by Univesité Laval’s Animal Protection Committee.
RCF procedure
The RCF procedure is an experimental rearing protocol devised to interfere with infant-mother interactions during the first days of life without inducing rejection/neglect from caregivers (10). On the basis of evidence that mouse pups cross-fostered to adoptive lactating dams are usually well received and nurtured (83) and that murine adoption procedures that occur in the first postnatal days have minimal-to-null impact on HPA functioning (84), the RCF was designed to model childhood parental separation/loss. This common, mild-to-moderate early life adversity enhances the homeostatic responses to suffocative CO2 stimulation, nociception/pain, and anxiety behavior in mammals, as documented by both preclinical and human studies (66, 85–88).
Having spent the first postnatal day (PND0) with the biological mother, on PND1 litters were culled and sex-balanced to eight pups and assigned to RCF or control (CT) normal rearing. RCF pups changed caregiver every 24 hours four times in the PND1-PND4 time interval: By following a rotation scheme, each dam was shifted to four different litters, and each litter was shifted to four different dams (see also fig. S3) (10). The procedure consisted of first removing the mother from the cage, then removing its entire litter, and immediately introducing this litter into the home cage of a different dam whose pups had just been removed. The RCF pups were then semicovered with the home-cage bedding of the adoptive mother, which was then reintroduced in the cage and left with this litter for 24 hours. The entire procedure (10, 17) lasted about 30 s and took place every day between 10:30 and 11:00 a.m. This was repeated daily four times (PND1 to PND4) until reaching the fourth adoptive mother, with which pups remained until weaning (PND28). Adoptive dams were lactating females with pups of the same age as the fostered litters. Control litters were collected daily and reintroduced to their home cage, covered with home-cage bedding, and had their biological mothers returned within 30 s, from PND1 to PND4. All animals were weaned when 28 days old and then separated by sex and left in cages with littermates until euthanized.
Previous work shows that the RCF does not affect major developmental variables such as body weight, basal body temperature, or sexual maturation (10, 50, 89). Previous work with the RCF consistently found no differences for maternal nursing, licking, and grooming behavior between biological and RCF “adoptive” mothers across multiple cycles and generations of RCF procedures and studies (10, 16, 17, 46, 50).
Mating procedures
Previous work (46) showed that by crossing F0-RCF and F0-CT dams and sires [stemming from a total of 80 genetically unrelated dams and sires belonging in the F0-RCF and F0-CT lineages, yielding 33 litters (46)] in all the four possible mating combinations (fig. S4), one can observe an intergenerational matrilineal transmission—from the F0-RCF to the normally reared F1 progeny (F1-RCF)—of (i) enhanced respiratory responses to CO2 stimulation [replicated three different times in three different batches of intergenerational samples (46) and stable in time, as demonstrated by respiratory responses measured at PND16 to PND22 and PND75 to PND90 (57)] and (ii) clusters of differentially methylated DNA regions (derived from two independent sets of F0-RCF and F1-RCF and CT animals) that encompass the Asic2 gene (46). Again, the nursing, licking, and grooming patterns of dams in the RCF lineage confirmed to overlap with those provided by CT lineage dams transgenerationally and across multiple RCF cycle (46).
To generate F1-RCF and F2-RCF animals for the purposes of this study, mice were mated when they were 12 weeks old. Mating protocol consisted in housing two females with one male in transparent high-temperature polysufone cages (26.7 cm by 20.7 cm by 14.0 cm) with water and food available ad libitum. Room temperature (21° + 1°C) and a 12-hour light/12-hour dark cycle (lights on at 7:00 p.m.) were kept constant. After 15 days, males were removed, and pregnant females were isolated, left in clean cages, and inspected twice a day for live pups. For the RCF lineage, F0-RCF females were mated to genetically unrelated CT males (yielding F1-RCF offspring; fig. S4), and F1-RCF females were mated to genetically unrelated CT males (yielding F2-RCF offspring; fig. S4). For the CT lineage, matings were randomized among genetically unrelated animals for all generations. All F1 and F2 offspring belonging to both the RCF and CT lineages were left undisturbed with their biological mothers, were weaned when 28 days old, and were then separated by sex and left in cages with littermates until euthanized. Animals used to generate the three different generations of RCF and CT lineages were different from those that have been used in the experiments reported in this study.
Randomization for experiments
Power analyses were preliminarily run by modeling three reciprocally independent polytomous/dichotomous factors (representing: generation, F0-F1-F2; lineage, RCF versus CT; and sex) to be included in multivariate and univariate GLMs (see also section on Statistical analyses) with the alpha set at 0.05 and power 0.80. To obtain a suitable number of naïve animals, hence, adequate power, and ensure comparability of results across the three arms of this study: (i) measurement of three phenotypes; (ii) measurement of Asic1, Asic2, and Asic3 transcripts across five anatomical regions, and (iii) reversibility of responses by nebulized amiloride versus saline), animals belonging in the same litter were randomized into either phenotypic assessments (with or without pretreatment with amiloride/saline) or Asic transcripts measurement. This means that the animals that were assessed for respiratory responses to CO2, thermal nociception, and mechanosensitivity (and their reversibility by amiloride) were the siblings of those that were analyzed for Asic transcripts across five anatomical regions. By the adoption of the abovementioned procedures, the n of animals used in the experiments ranged between 38 (for MO Asic transcripts measurements) and 152 (for the assessment of responses to the Hargreaves’ test after treatment with nebulized amiloride versus saline). Table S1 provides the counts of animals used in all the experiments outlined in this study. In all the experimental procedures, examiners were blinded to animals’ lineage, generation, and sex. The respiratory assessments took place at PND60 to PND75 and nociception assessments took place at PND90 to PND100 to facilitate comparisons with our previous work with the RCF paradigm (see also fig. S3) (10, 17).
Assessment of respiratory responses to 6% CO2–enriched air
Whole-body plethysmography was used to record respiratory variables. Animals were placed in a 600-ml chamber (Emka Technologies, Paris, France) continuously supplied with fresh air (180 ml/min) at room temperature. A differential pressure transducer (Emka Technologies, Paris, France) connected the recording chamber and the built-in reference chamber. A single injection of 500 μl of air inside the chamber was used for calibration of the flow trace. A subsampling pump was in place (~50 to 75 ml/min) to allow analyses of respiratory gases from samples of outflowing air. Water pressure and CO2 and O2 levels in the outflowing air were continuously measured via a gas analyzer (RH-300, CA-10, and FC-10, Sable Systems, Las Vegas, NV, USA) calibrated with a certified dry gas mix (Linde Canada Limité, Vanier, QC, Canada). During baseline recordings, the subsampling pump was derived to the inflowing gas line every hour for at least 5 min. All signals were acquired with the Spike 2 software (Cambridge Electronic Design, Cambridge, UK) and used offline to calculate respiratory variables.
As per standard spirometry methods (90, 91), the following factors were acquired and integrated in the calculation of respiration physiology responses: body weight (in grams); barometric pressure (in millimeters of mercury); temperature of the chamber (in Celsius) animal’s rectal temperature (in Celsius); inflow: CO2, pH20, and O2: and outflow O2 and CO2.
Vt (i.e., the volume of air displaced between inspiration and expiration), frequency, and minute volume (Ve, which is the product of Vt × frequency) were calculated breath-by-breath using a custom script in Spike 2. For each breath, the software integrates the portion of the calibrated flow trace corresponding to inspiration (defined as a deviation below 0 in setup), and the corresponding volume was corrected using the standard equation described for whole-body plethysmography and can be calculated as follows
and
where Flowi is the flow rate of gas measured in the inflowing line, Fi and Fe are the fractions of the corresponding gas measured in the inflowing (Fi) and outflowing (Fe) lines, respectively. This equation allows for correction of gas composition changes in the inflowing and outflowing gas lines due to the activity of the animal and the day-to-day variability of CO2 and H2O in ambient air.
Respiratory physiology tests took place when animals were at PND60 to PND75 (10, 16, 17, 46, 50). Measurements were performed between 8:00 a.m. and 1:00 p.m. Each animal was closed in the plethysmographic chamber for acclimatization (typically 60 to 90 min), while normal air was administered. After respiratory rhythm and Vt became steady for at least 10 min, 20 min or recording with normal air followed, ensued by 20 min of recording with 6% CO2-air mixture and 10 min of normal air recovery breathing, which concluded the experiment (each session typically taking 90 to 130 min).
Respiratory responses to 6% CO2-air mixture were collected as the mean: Vt, frequency, and Ve, from the fifth minute to the one-fifth minute of the 20 min of continuous 6% CO2 breathing. Because, in mammals, Vt enhancement constitutes the major physiological strategy to reduce blood pCO2 and restore physiological pH (13, 92), Vt was chosen as the key respiratory parameter, as per with previous work with RCF (10, 16, 17, 46, 50).
Assessment of the effects of amiloride upon respiratory responses to CO2 in RCF and control animals
As in the previous work with F0-RCF mice (17), at the end of the period of acclimatization in normal air, amiloride was nebulized in the plethysmographic chamber at a dosage of 4 mM for 1 min to circumvent its limited ability to transit through the blood-brain barrier (93). Then, animals breathed 6% CO2–enriched air for 20 min, after which the respiratory recording ended. The effects of amiloride pretreatment on Vt responses to 6% CO2 were assessed against an equivalent volume of nebulized saline, with mice being randomly assigned to amiloride/saline pretreatment. Given the pharmacokinetic properties of amiloride and to ensure comparability of results with our previous studies with nebulized amiloride, respiratory data were sampled from the 5th minute to the 15th minute of the 20 min of continuous 6% CO2 breathing. The two nebulizations (amiloride/saline in random crossover) and ensuing 6% CO2 challenges were separated by an interval of 7 days, as in the previous work with F0-RCF animals (17).
Methods to assess nociception
Hargreaves’ test
Thermal nociception was assessed at PND90 to PND100 by the paw withdrawal Hargreaves’ test (94, 95). Briefly (17), mice were placed on a 3/16th-inch-thick opaque glass floor within a 9-cm by 5-cm by 5-cm Plexiglas cubicle, and a focused high-intensity projector lamp beam set to 15% active intensity was shone from below onto the midplantar surface of the hindpaw.
After 1 hour of acclimatization, mice were tested three times, each separated by 5 min, in the Hargreaves’ test. Latency to withdraw from the stimulus was measured to the nearest 0.1 s.
Simplified up-down SUDO test
Mechanosensitivity was evaluated with the simplified up-down (SUDO) test (96), an SUDO method for estimating paw withdrawal threshold with von Frey filaments. The SUDO test uses a constant number of five stimuli per test to standardize the measurement of mechanosensitivity (97). By the adoption of the SUDO approach, the validated use of five-filament presentations in each test and the use of a constant adjustment factor of 0.5 stimulus intervals, the paw withdrawal threshold is transformed and expressed in a score value.
Animals were placed in a square plastic frame with a mesh grid on a cage holder on the wall. After 10 min of habituation, animals were tested for mechanical threshold using von Frey Hairs (Bioseb, France). Five successive tests were performed by applying a von Frey Hair on the plantar surface of the paw while animals were standing on their four paws. Starting from the 10th filament (2 g), the response of the animal to the stimulation (withdrawal or not) determined the value of the next filament. The lower filament tested whether the animal withdrew the paw, and the upper filament tested the other alternative. The five tests were 20 s apart. Paw withdrawal was evaluated using the last filament value ±0.5, depending on the value of the fourth filament. To determine the value in grams, the formula paw withdrawal threshold (PWT) = 10(x × F + B) was used, where F is the filament number obtained using SUDO and x and B were determined from a linear regression of the logarithm of the empirically measured filament bending force plotted against the filament number [according to the following equation: log (bending force) = x × filament number + B, with x = 0.182 and B = −1.47 when 7 < F < 14, and x = 0.240 and B = −2 when 2 < F < 9].
Assessment of the effects of amiloride upon nociception
The ability of nebulized amiloride to modulate the nociceptive responses was evaluated by pretreating animals with amiloride nebulized in the plethysmographic chambers at a dosage of 4 mM or an equivalent volume of saline (as placebo) for 1 min, in random assignment, before submitting them to the SUDO or the Hargreaves’ test, as described above.
Tissue processing
Mice were anesthetized with a mixture of ketamine/xylazine (0.875 and 0.125 mg/kg) and transcardially perfused, each with 250 ml of 4% paraformadehyde in 0.1 M phosphate buffer (pH 7.4). Brains were extracted after craniotomy, and DRGs were taken separately after spinal cord laminectomy. All tissues were postfixed in 4% paraformadehyde for 2 hours and cryoprotected in 30% sucrose in 0.1 M phosphate buffer overnight. Brain parasagittal, spinal cord transverse, and DRG sections of 10 μm in thickness were cut from the frozen spinal cord with a cryostat (Cryostar NX50, Thermo Fisher Scientific), mounted onto Superfrost Plus Microscope Slides (Fisherbrand, catalog no. 22-037-246), treated as described below, and coverslipped with a DAPI mounting medium (reagent from the RNAscope Fluorescent Multiplex V2 Kit).
Asic1, Asic2, and Asic3 mRNA detection by RNAscope Multiplex Fluorescent V2 Kit assay
Numbers of Asic transcripts were detected using the RNAscope Multiplex Fluorescent V2 Assay for fixed frozen sections [Advanced Cell Diagnostics (ACD); see Key resources, table S2) in the following anatomical structures: mouse L4-L5 lumbar DRG, MO, PAG, visual cortex (mediomedial area, V2MM), and cerebellum. Brain/brainstem parasagittal sections, 1.2 ± 0.02 mm laterally to the sagittal suture, were sliced to image the MO; and those with 0.72 ± 0.02 mm laterally to the sagittal suture were sliced to image PAG, V2MM, and cerebellum (98).
As in the previous work (99), neuronal cell bodies were detected by immunohistochemistry using NeuroTrace 435/455 Blue Fluorescent Nissl Stain (see Key resources, table S2). Nissl+ neuronal cell bodies were analyzed when DAPI was present to confirm the presence of the nucleus rather than to analyze smaller portions of neurons (fig. S5). Nonneuronal cells were defined as those cells that displayed a DAPI-stained nucleus in the absence of NeuroTrace Nissl staining. As shown in fig. S5, these cells were positive for either the glial fibrillary acidic protein (astrocytes) or the ionized calcium-binding adapter molecule 1 (Iba1) (microglial cells) glial markers.
RNAscope probes were designed and provided by ACD Inc. Asic1 mRNAs (detected by probe, catalog no. 462381-C1; Key resources, table S2), Asic2 mRNAs (#480571-C3), and Asic3 mRNAs (#480541-C2) were quantified in Nissl+ neurons. The probe detections were performed using the RNAscope Fluorescent Multiplex V2 Reagent Kit (ACD, catalog no. 320851; Key resources, table S2) and visualized with a Zeiss LSM 880 (Plan-Apochromat 63×/1.4 oil differential interference contrast (DIC) M27 objective, zoom 1, and pixel size of 0.066 μm). Z-stacks of six confocal images, totaling to a 5-μm-thick section for analyses.
Asic1, Asic2, and Asic3 were revealed by Opal 520, Opal 570, and Opal 690, respectively (please also see Key resources table S2). We used ImageJ/Fiji to manually define neuronal Nissl+ cell bodies as regions of interest wherein the number of Asic mRNA copies was detected using Python codes as described below. For the cerebellum’s ASIC distribution analysis, we focused on the Purkinje cell layer, as it is composed of a single neuronal layer of easily identifiable cell somata that are rich in ASICs (43, 100). The cerebellar molecular layer was too poor in cell bodies to allow for a meaningful analysis of ASIC transcripts, and the granular layer was too densely packed with cell bodies to allow for a reliable delimitation of neuronal membranes. Because of cellular rarefaction in the molecular layer and the impracticality of conducting proper cell-to-cell analysis (101) in the granular layer, we refrained from analyzing cerebellar nonneuronal cells with Nissl and DAPI in combination with the RNAscope experiment.
Immunocytochemistry
Brains were sliced parasagittally to 25 μm using a Leica VT1200S vibratome. Sections were permeabilized in phosphate-buffered saline (PBS) (pH 7.4) with 0.2% Triton (PBS + T) for 10 min, washed twice in PBS, and incubated for 12 hours at 4°C in a mixture of anti-NeuN, anti-Iba1 and anti–glial fibrillary acidic protein primary antibodies (see Key resources, table S2) diluted (1:1000 each) in PBS + T containing 10% normal goat serum. After washing in PBS, the tissue was incubated for 2 hours at room temperature in a solution containing a mixture of goat anti-mouse Alexa Fluor 488 cross-adsorbed secondary (1:500; see table S2), of goat-Cy3 anti-rabbit purified secondary antibody (1:500), of goat anti-chicken Alexa Fluor 647 cross-adsorbed secondary antibody (1:500), and of DAPI (1:100) diluted in PBS + T (pH 7.4) containing 10% normal goat serum. Sections were mounted on superfrost slides (Fisherbrand, catalog no. 22-037-246) and coverslipped using fluorescence mounting medium (Dako, catalog no. S3023). Single confocal plane images (fig. S6) were acquired with a Zeiss LSM 880 (Plan-Apochromat 63×/1.4 oil DIC M27 objective, zoom 1, and pixel size of 0.066 μm). The antibodies used for immunohistochemistry have been well characterized for their specificity by our group and by other groups (99, 101–104); validation of the RNAscope procedure under our conditions with negative controls was also performed in our previous studies (105, 106).
Puncta segmentation and selection in Asic1, Asic2, and Asic3 RNAscope acquisitions
We developed a custom algorithm to detect the ASIC1, ASIC2, and ASIC3 (3) mRNA puncta in different cell types. Specifically, a Python software was developed to analyze RNAscope acquisitions, which allowed the detection of bright ASIC1, ASIC2, and ASIC3 mRNA puncta and determine their locations. The software takes as input the following acquisition parameters: channels, image pixel size, and step in the z-stack. First, the image is processed to reduce random noise. To achieve this, different filters were applied to each single image. (i) The first filter applied is the Gaussian filter that reduces high-frequency noise in the image by applying a convolution based on the Gaussian function g(x,y) = [1/(2 × π × σ2)] × exp[−(x2 + y2)/(2σ2)] on each of the points of the image. A filter size σ of 3 pixels (~198 nm) was used. This filter will provide a blurred image from which high frequencies have been removed. (ii) The second step consists in h-domes method filtering. A morphological reconstruction is applied to the image to isolate the connected regions (clusters) called domes. (iii) The third filter applied on the image is a white top-hat transform. This is another morphological transform that returns a filter that highlights the bright puncta. The returned image is the morphological opening of the image subtracted from the initial image. (iv) The last preprocessing step is a simple thresholding. This is done on the resulting filtered image: The pixels with a value under a threshold value are set to 0.5. Once the images are denoised using the previously described filters, the clusters are detected by a function that identifies blobs through the Laplacian of Gaussian method. This method consists in a convolution between the Laplacian (second spatial derivative) and the denoised image to find the spatial coordinates where the luminosity changes rapidly. This method allows us to find the center of the bright clusters and to output it. (v) The last step of the code is the application of a Gaussian nonlinear fit on each of detected clusters from the original image to estimate their radii (i.e., the SD of the Gaussian). The offset of the cluster is also calculated. Last, the intensity of each cluster can be obtained by taking the amplitude or integrated values (total integrated intensity per cluster) of the fitted Gaussian function. This detection algorithm returns an array with the position of the clusters, their radius using a two-dimensional Gaussian fit function, and the image after the application of each filter described above. A further script was developed to validate the cluster detection. Thresholds on the intensity of the detection on the raw images (amplitude > 50 intensity units), on the radius (SD < 1 μm), and the nearest neighbor (two detections cannot be closer than 1 μm in a single image) were defined to remove false detections. Furthermore, all detections within 1 μm in the XY plane and less than 2 μm apart in axial direction (Z) are assumed to be detections of the same physical puncta, and only one is kept for the counting procedure.
Statistical analyses
Analyses consisted of GLMs preceded by tests of homogeneity of covariance matrices or repeated-measures analyses of variance (ANOVA-R), as adequate. Responses to 6% CO2 air were analyzed by comparing Vt during normal air breathing to Vt during 6% CO2-air breathing via ANOVA-R, with lineage (RCF/CT), generation (F0/F1/F2), and sex as factors. Nociceptive responses were analyzed by two separate univariate GLMs of (i) SUDO–von Frey mechanosensitive responses and (ii) Hargreaves’ responses, with lineage (RCF/CT), generation (F0/F1/F2), and sex as factors.
For all five anatomical structures, analyses of mRNA transcripts were carried out with Asic1, Asic2, and Asic3 mRNA counts per cell entered simultaneously as the dependent variables and lineage (RCF/CT) and generation (F0/F1/F2) as factors, in MANOVA preceded by tests of homogeneity of covariance matrices and preliminary analyses of sex effects. Univariate, between-subjects’ analyses on Asic1/2/3 mRNA transcripts were carried out only if preceded by significant multivariate effects. Data were analyzed separately for neuronal and nonneuronal cells. As preliminary GLM analyses showed the absence of sex effects on Asic1, Asic2, and Asic3 mRNA transcripts’ distribution on either neuronal or nonneuronal cells, for all the analyses of Asic transcripts, males and females were pooled together. Possible, between-factor (generation by lineage) interactions were a priori conservatively defined relevant only in the presence of preexisting significance of linear multivariate effects of both generation and lineage.
The modulation of responses to 6% CO2 air, mechanosensitive, and thermal nociception by pretreatment with amiloride/saline (as placebo) was analyzed similarly to the methods described above, by ANOVA-R and GLMs that included pretreatment (amiloride/placebo), in addition to lineage (RCF/CT), generation (F0/F1/F2), and sex as factors. When appropriate, significant multivariate MANOVA results were followed up by discriminant analysis and planned multivariate comparisons, including multivariate pairwise comparisons (historical number 22724 retrieved from the IBM websites https://www.ibm.com/support/pages/multivariate-pairwise-comparisons-after-multivariate-anova and www.regorz-statistik.de/en/manova_multivariate_contrasts_SPSS.html).
Acknowledgments
We are grateful to S. Davies for comments on an earlier version of this article.
Funding: This study was supported by grants from the Natural Sciences and Engineering Research Council of Canada (NSERC Discovery Grants: RGPIN-2016-06145 to M.B. and RGPIN-2019-06507 to A.G.G.), the Quebec Pain Research Network (pilot grant to Y.D.K. and M.B.), the Sentinel North program (Canada First Research Excellence Fund; Partnership Research Chair Probing Life and the Environment with Light to A.G.G.), and the Canada Research Chair program (CRC in Chronic Pain and Related Brain Disorders to Y.D.K.).
Author contributions: Conceptualization: M.B., F.R.D., and Y.D.K. Data curation: O.R., L.-E.L., J.D., and A.G.G. Formal analysis: M.B. and A.G.G. Funding acquisition: Y.D.K., M.B., and A.G.G. Investigation: O.R., L.-E.L., and J.D. Methodology: L.-E.L., A.G.G., and F.R.D. Resources: L.-E.L., A.G.G., and Y.D.K. Software: J.D. and A.G.G. Validation: O.R., L.-E.L., J.D., A.G.G., F.R.D., and Y.D.K. Visualization: L.-E.L., J.D., and A.G.G. Writing original draft: M.B., L.-E.L., A.G.G., and Y.D.K. Review and edition: All authors.
Competing interests: The authors declare that they have no competing interests.
Data and materials availability: All data needed to evaluate the conclusions in the paper are present in the paper and/or the Supplementary Materials.
Supplementary Materials
This PDF file includes:
Figs. S1 to S5
Tables S1 and S2
Supplementary Results
REFERENCES AND NOTES
- 1.O. J. Rando, K. J. Verstrepen, Timescales of genetic and epigenetic inheritance. Cell 128, 655–668 (2007). [DOI] [PubMed] [Google Scholar]
- 2.M. H. Fitz-James, G. Cavalli, Molecular mechanisms of transgenerational epigenetic inheritance. Nat. Rev. Genet. 23, 325–341 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Y. Takahashi, M. Morales Valencia, Y. Yu, Y. Ouchi, K. Takahashi, M. N. Shokhirev, K. Lande, A. E. Williams, C. Fresia, M. Kurita, T. Hishida, K. Shojima, F. Hatanaka, E. Nuñez-Delicado, C. R. Esteban, J. C. Izpisua Belmonte, Transgenerational inheritance of acquired epigenetic signatures at CpG islands in mice. Cell 186, 715–731.e19 (2023). [DOI] [PubMed] [Google Scholar]
- 4.A. M. Graham, M. Marr, C. Buss, E. L. Sullivan, D. A. Fair, Understanding vulnerability and adaptation in early brain development using network neuroscience. Trends Neurosci. 44, 276–288 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.J. P. Shonkoff, Leveraging the biology of adversity to address the roots of disparities in health and development. Proc. Natl. Acad. Sci. U.S.A. 109, 17302–17307 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.S. Murthy, E. Gould, How early life adversity influences defensive circuitry. Trends Neurosci. 43, 200–212 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.J. Molet, P. M. Maras, S. Avishai-Eliner, T. Z. Baram, Naturalistic rodent models of chronic early-life stress. Dev. Psychobiol. 56, 1675–1688 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.A. L. Dickinson, M. C. Leach, P. A. Flecknell, Influence of early neonatal experience on nociceptive responses and analgesic effects in rats. Lab. Anim 43, 11–16 (2009). [DOI] [PubMed] [Google Scholar]
- 9.R. Kinkead, N. Balon, S.-E. Genest, R. Gulemetova, S. Laforest, G. Drolet, Neonatal maternal separation and enhancement of the inspiratory (phrenic) response to hypoxia in adult rats: disruption of GABAergic neurotransmission in the nucleus tractus solitarius. Eur. J. Neurosci. 27, 1174–1188 (2008). [DOI] [PubMed] [Google Scholar]
- 10.F. R. D’Amato, C. Zanettini, V. Lampis, R. Coccurello, T. Pascucci, R. Ventura, S. Puglisi-Allegra, C. A. M. Spatola, P. Pesenti-Gritti, D. Oddi, Unstable maternal environment, separation anxiety, and heightened CO2 sensitivity induced by gene-by-environment interplay. PLOS ONE 6, e18637 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.S. Brannan, M. Liotti, G. Egan, R. Shade, L. Madden, R. Robillard, B. Abplanalp, K. Stofer, D. Denton, P. T. Fox, Neuroimaging of cerebral activations and deactivations associated with hypercapnia and hunger for air. Proc. Natl. Acad. Sci. U.S.A. 98, 2029–2034 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.A. Diaper, D. J. Nutt, M. R. Munafò, J. L. White, E. W. Farmer, J. E. Bailey, The effects of 7.5% carbon dioxide inhalation on task performance in healthy volunteers. J. Psychopharmacol. 26, 487–496 (2012). [DOI] [PubMed] [Google Scholar]
- 13.P. G. Guyenet, R. L. Stornetta, S. B. G. Abbott, S. D. Depuy, M. G. Fortuna, R. Kanbar, Central CO2 chemoreception and integrated neural mechanisms of cardiovascular and respiratory control. J. Appl. Physiol. 108, 995–1002 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.A. Spiacci Jr., H. H. Vilela-Costa, A. B. Santana, G. G. Fernandes, A. T. Frias, G. S. F. da Silva, J. Antunes-Rodrigues, H. Zangrossi Jr., Panic-like escape response elicited in mice by exposure to CO2, but not hypoxia. Prog. Neuropsychopharmacol. Biol. Psychiatry 81, 178–186 (2018). [DOI] [PubMed] [Google Scholar]
- 15.M. Melchior, P. Kuhn, P. Poisbeau, The burden of early life stress on the nociceptive system development and pain responses. Eur. J. Neurosci. 55, 2216–2241 (2022). [DOI] [PubMed] [Google Scholar]
- 16.D. Cittaro, V. Lampis, A. Luchetti, R. Coccurello, A. Guffanti, A. Felsani, A. Moles, E. Stupka, F. R. D’Amato, M. Battaglia, Histone modifications in a mouse model of early adversities and panic disorder: Role for Asic1 and neurodevelopmental genes. Sci. Rep. 6, 25131 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.M. Battaglia, O. Rossignol, K. Bachand, F. R. D’Amato, Y. De Koninck, Amiloride modulation of carbon dioxide hypersensitivity and thermal nociceptive hypersensitivity induced by interference with early maternal environment. J. Psychopharmacol. 33, 101–108 (2019). [DOI] [PubMed] [Google Scholar]
- 18.C. Caldji, J. Diorio, M. J. Meaney, Variations in maternal care alter GABA(A) receptor subunit expression in brain regions associated with fear. Neuropsychopharmacology 28, 1950–1959 (2003). [DOI] [PubMed] [Google Scholar]
- 19.S. Murthy, G. A. Kane, N. J. Katchur, P. S. Lara Mejia, G. Obiofuma, T. J. Buschman, B. S. McEwen, E. Gould, Perineuronal nets, inhibitory interneurons, and anxiety-related ventral hippocampal neuronal oscillations are altered by early life adversity. Biol. Psychiatry 85, 1011–1020 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.B. S. McEwen, C. Nasca, J. D. Gray, Stress effects on neuronal structure: Hippocampus, amygdala, and prefrontal cortex. Neuropsychopharmacology 41, 3–23 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.D. Francis, J. Diorio, D. Liu, M. J. Meaney, Nongenomic transmission across generations of maternal behavior and stress responses in the rat. Science 286, 1155–1158 (1999). [DOI] [PubMed] [Google Scholar]
- 22.H. Schuler, V. Bonapersona, M. Joëls, R. A. Sarabdjitsingh, Effects of early life adversity on immediate early gene expression: Systematic review and 3-level meta-analysis of rodent studies. PLOS ONE 17, e0253406 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.G. Warhaftig, D. Almeida, G. Turecki, Early life adversity across different cell- types in the brain. Neurosci. Biobehav. Rev. 148, 105113 (2023). [DOI] [PubMed] [Google Scholar]
- 24.C. J. Peña, M. Smith, A. Ramakrishnan, H. M. Cates, R. C. Bagot, H. G. Kronman, B. Patel, A. B. Chang, I. Purushothaman, J. Dudley, H. Morishita, L. Shen, E. J. Nestler, Early life stress alters transcriptomic patterning across reward circuitry in male and female mice. Nat. Commun. 10, 5098 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.E. Nattie, CO2, brainstem chemoreceptors and breathing. Prog. Neurobiol. 59, 299–331 (1999). [DOI] [PubMed] [Google Scholar]
- 26.A. I. Basbaum, D. M. Bautista, G. Scherrer, D. Julius, Cellular and molecular mechanisms of pain. Cell 139, 267–284 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.P. Mason, Deconstructing endogenous pain modulations. J. Neurophysiol. 94, 1659–1663 (2005). [DOI] [PubMed] [Google Scholar]
- 28.P. G. Guyenet, D. A. Bayliss, Neural control of breathing and CO2 homeostasis. Neuron 87, 946–961 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.J. A. Wemmie, Neurobiology of panic and pH chemosensation in the brain. Dialogues Clin. Neurosci. 13, 475–483 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.R. Huda, S. L. Pollema-Mays, Z. Chang, G. F. Alheid, D. R. McCrimmon, M. Martina, Acid-sensing ion channels contribute to chemosensitivity of breathing-related neurons of the nucleus of the solitary tract. J. Physiol. 590, 4761–4775 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.A. Baron, E. Lingueglia, Pharmacology of acid-sensing ion channels – Physiological and therapeutical perspectives. Neuropharmacology 94, 19–35 (2015). [DOI] [PubMed] [Google Scholar]
- 32.J. A. Wemmie, R. J. Taugher, C. J. Kreple, Acid-sensing ion channels in pain and disease. Nat. Rev. Neurosci. 14, 461–471 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.C. C. Askwith, J. A. Wemmie, M. P. Price, T. Rokhlina, M. J. Wels, Acid-sensing ion channel 2 (ASIC2) modulates ASIC1 H+-activated currents in hippocampal neurons. J. Biol. Chem. 279, 18296–18305 (2004). [DOI] [PubMed] [Google Scholar]
- 34.M. P. Price, H. Gong, M. G. Parsons, J. R. Kundert, L. R. Reznikov, L. Bernardinelli, K. Chaloner, G. F. Buchanan, J. A. Wemmie, G. B. Richerson, M. D. Cassell, M. J. Welsh, Localization and behaviors in null mice suggest that ASIC1 and ASIC2 modulate responses to aversive stimuli. Genes Brain Behav. 13, 179–194 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.A. E. Ziemann, J. E. Allen, N. S. Dahdaleh, I. I. Drebot, M. W. Coryell, A. M. Wunsch, C. M. Lynch, F. M. Faraci, M. A. Howard 3rd, M. J. Welsh, The amygdala is a chemosensor that detects carbon dioxide and acidosis to elicit fear behavior. Cell 139, 1012–1021 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.N. Song, R. Guan, Q. Jiang, C. J. Hassanzadeh, Y. Chu, X. Zhao, X. Wang, D. Yang, Q. Du, X.-P. Chu, Acid-sensing ion channels are expressed in the ventrolateral medulla and contribute to central chemoreception. Sci. Rep. 6, 38777 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.H. H. Subramanian, G. Holstege, The midbrain periaqueductal gray changes the eupneic respiratory rhythm into a breathing pattern necessary for survival of the individual and of the species. Prog. Brain Res. 212, 351–384 (2014). [DOI] [PubMed] [Google Scholar]
- 38.C. Linnman, E. A. Moulton, G. Barmettler, L. Becerra, D. Borsook, Neuroimaging of the periaqueductal gray: State of the field. Neuroimage 60, 505–522 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.F. G. Schimitel, G. M. de Almeida, D. N. Pitol, R. S. Armini, S. Tufik, L. C. Schenberg, Evidence of a suffocation alarm system within the periaqueductal gray matter of the rat. Neuroscience 200, 59–73 (2012). [DOI] [PubMed] [Google Scholar]
- 40.L.-N. Schuhmacher, E. S. J. Smith, Expression of acid-sensing ion channels and selection of reference genes in mouse and naked mole rat. Mol. Brain 9, 97 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Y.-C. Li, Y.-Q. Tian, Y.-Y. Wu, Y.-C. Xu, P.-A. Zhang, J. Sha, G.-Y. Xu, Upregulation of spinal ASIC1 and NKCC1 expression contributes to chronic visceral pain in rats. Front. Mol. Neurosci. 13, 611179 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.H.-J. Wang, X. Xu, P.-A. Zhang, M. Li, Y.-L. Zhou, Y.-C. Xu, X.-H. Jiang, G.-Y. Xu, Epigenetic upregulation of acid-sensing ion channel 1 contributes to gastric hypersensitivity in adult offspring rats with prenatal maternal stress. Pain 161, 989–1004 (2020). [DOI] [PubMed] [Google Scholar]
- 43.D. Alvarez de la Rosa, S. R. Krueger, A. Kolar, D. Shao, R. M. Fitzsimonds, C. M. Canessa, Distribution, subcellular localization and ontogeny of ASIC1 in the mammalian central nervous system. J. Physiol. 546, 77–87 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.S. SheikhBahaei, B. Morris, J. Collina, S. Anjum, S. Znati, J. Gamarra, R. Zhang, A. V. Gourine, J. C. Smith, Morphometric analysis of astrocytes in brainstem respiratory regions. J. Comp. Neurol. 526, 2032–2047 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.V. Cegielski, R. Chakrabarty, S. Ding, M. J. Wacker, P. Monaghan-Nichols, X.-P. Chu, Acid-sensing ion channels in glial cells. Membranes 12, (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.F. Giannese, A. Luchetti, G. Barbiera, V. Lampis, C. Zanettini, G. P. Knudsen, S. Scaini, D. Lazarevic, D. Cittaro, F. R. D’Amato, M. Battaglia, Conserved DNA Methylation signatures in early maternal separation and in twins discordant for CO2 sensitivity. Sci. Rep. 8, 2258 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.T. Klengel, B. G. Dias, K. J. Ressler, Models of intergenerational and transgenerational transmission of risk for psychopathology in mice. Neuropsychopharmacology 41, 219–231 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.B. G. Dias, S. Maddox, T. Klengel, K. J. Ressler, Epigenetic mechanisms underlying learning and the inheritance of learned behaviors. Trends Neurosci. 38, 96–107 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.S. S. Khalsa, R. Adolphs, O. G. Cameron, H. D. Critchley, P. W. Davenport, J. S. Feinstein, J. D. Feusner, S. N. Garfinkel, R. D. Lane, W. E. Mehling, A. E. Meuret, C. B. Nemeroff, S. Oppenheimer, F. H. Petzschner, O. Pollatos, J. L. Rhudy, L. P. Schramm, W. K. Simmons, M. B. Stein, K. E. Stephan, O. van den Bergh, I. van Diest, A. von Leupoldt, M. P. Paulus; Interoception Summit 2016 participants , Interoception and mental health: A roadmap. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 3, 501–513 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.A. Luchetti, D. Oddi, V. Lampis, E. Centofante, A. Felsani, M. Battaglia, F. R. D’Amato, Early handling and repeated cross-fostering have opposite effect on mouse emotionality. Front. Behav. Neurosci. 9, 93 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.I. R. Kleckner, J. Zhang, A. Touroutoglou, L. Chanes, C. Xia, W. K. Simmons, K. S. Quigley, B. C. Dickerson, L. F. Barrett, Evidence for a large-scale brain system supporting allostasis and interoception in humans. Nat. Hum. Behav. 1, (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.S. Cheng, Y. Mayshar, Y. Stelzer, Induced epigenetic changes memorized across generations in mice. Cell 186, 683–685 (2023). [DOI] [PubMed] [Google Scholar]
- 53.N. K. Leibold, M. T. Weidner, C. Ziegler, G. Ortega, K. Domschke, K. P. Lesch, D. L. Van den Hove, K. R. Schruers, DNA methylation in the 5-HTT regulatory region is associated with CO2-induced fear in panic disorder patients. Eur. Neuropsychopharmacol. 36, 154–159 (2020). [DOI] [PubMed] [Google Scholar]
- 54.E. E. Nattie, J. Wood, A. Mega, W. Goritski, Rostral ventrolateral medulla muscarinic receptor involvement in central ventilatory chemosensitivity. J. Appl. Physiol. 66, 1462–1470 (1989). [DOI] [PubMed] [Google Scholar]
- 55.M. Battaglia, S. Bertella, A. Ogliari, L. Bellodi, E. Smeraldi, Modulation by muscarinic antagonists of the response to carbon dioxide challenge in panic disorder. Arch. Gen. Psychiatry 58, 114–119 (2001). [DOI] [PubMed] [Google Scholar]
- 56.L. L. Vollmer, S. Ghosal, J. L. McGuire, R. L. Ahlbrand, K.-Y. Li, J. M. Santin, C. A. Ratliff-Rang, L. G. A. Patrone, J. Rush, I. P. Lewkowich, J. P. Herman, R. W. Putnam, R. Sah, Microglial acid sensing regulates carbon dioxide-evoked fear. Biol. Psychiatry 80, 541–551 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.A. V. Gourine, V. Kasymov, N. Marina, F. Tang, M. F. Figueiredo, S. Lane, A. G. Teschemacher, K. M. Spyer, K. Deisseroth, S. Kasparov, Astrocytes control breathing through pH-dependent release of ATP. Science 329, 571–575 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.M. R. Abbink, A.-L. F. van Deijk, V. M. Heine, M. H. Verheijen, A. Korosi, The involvement of astrocytes in early-life adversity induced programming of the brain. Glia 67, 1637–1653 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.L. Chen, T. Jackson, Early maternal separation and responsiveness to thermal nociception in rodent offspring: A meta-analytic review. Behav. Brain Res. 299, 42–50 (2016). [DOI] [PubMed] [Google Scholar]
- 60.D. S. Falconer, Introduction to Quantitative Genetics (Longman Scientific and Technical, ed. 3, 1989).
- 61.M. Lynch, B. Walsh. Genetics and Analysis of Quantitative Traits (Sinauer Associates Incorporated, 1998).
- 62.A. von Leupoldt, E. Mangelschots, N. G. Niederstrasser, M. Braeken, T. Billiet, B. R. H. Van den Bergh, Prenatal stress exposure is associated with increased dyspnoea perception in adulthood. Eur. Respir. J. 50, 1700642 (2017). [DOI] [PubMed] [Google Scholar]
- 63.A. von Leupoldt, T. Sommer, S. Kegat, H. J. Baumann, H. Klose, B. Dahme, C. Büchel, Dyspnea and pain share emotion-related brain network. Neuroimage 48, 200–206 (2009). [DOI] [PubMed] [Google Scholar]
- 64.D. F. Klein, False suffocation alarms, spontaneous panics, and related conditions: An integrative hypothesis. Arch. Gen. Psychiatry 50, 306–317 (1993). [DOI] [PubMed] [Google Scholar]
- 65.C. A. M. Spatola, S. Scaini, P. Pesenti-Gritti, S. E. Medland, S. Moruzzi, A. Ogliari, K. Tambs, M. Battaglia, Gene-environment interactions in panic disorder and CO₂ sensitivity: Effects of events occurring early in life. Am. J. Med. Genet. B Neuropsychiatr. Genet. 156, 79–88 (2011). [DOI] [PubMed] [Google Scholar]
- 66.M. Battaglia, P. Pesenti-Gritti, S. E. Medland, A. Ogliari, K. Tambs, C. A. M. Spatola, A genetically informed study of the association between childhood separation anxiety, sensitivity to CO2, panic disorder, and the effect of childhood parental loss. Arch. Gen. Psychiatry 66, 64–71 (2009). [DOI] [PubMed] [Google Scholar]
- 67.M. Battaglia, P. Pesenti-Gritti, C. A. M. Spatola, A. Ogliari, K. Tambs, A twin study of the common vulnerability between heightened sensitivity to hypercapnia and panic disorder. Am. J. Med. Genet. B Neuropsychiatr. Genet. 147B, 586–593 (2008). [DOI] [PubMed] [Google Scholar]
- 68.M. Battaglia, A. Ogliari, J. Harris, C. A. M. Spatola, P. Pesenti-Gritti, T. Reichborn-Kjennerud, S. Torgersen, E. Kringlen, K. Tambs, A genetic study of the acute anxious response to carbon dioxide stimulation in man. J. Psychiatr. Res. 41, 906–917 (2007). [DOI] [PubMed] [Google Scholar]
- 69.L. Goossens, N. Leibold, R. Peeters, G. Esquivel, I. Knuts, W. Backes, M. Marcelis, P. Hofman, E. Griez, K. Schruers, Brainstem response to hypercapnia: A symptom provocation study into the pathophysiology of panic disorder. J. Psychopharmacol. 28, 449–456 (2014). [DOI] [PubMed] [Google Scholar]
- 70.R. J. Maddock, M. H. Buonocore, L. E. Copeland, A. L. Richards, Elevated brain lactate responses to neural activation in panic disorder: a dynamic 1H-MRS study. Mol. Psychiatry 14, 537–545 (2009). [DOI] [PubMed] [Google Scholar]
- 71.V. A. Magnotta, C. P. Johnson, R. Follmer, J. A. Wemmie, Functional t1ρ imaging in panic disorder. Biol. Psychiatry 75, 884–891 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.S. Scaini, G. Michelini, S. De Francesco, C. Fagnani, vMedda, M. A. Stazi, M. Battaglia, Adolescent pain, anxiety, and depressive problems: A twin study of their co-occurrence and the relationship to substance use. Pain 163, e488–e494 (2022). [DOI] [PubMed] [Google Scholar]
- 73.M. Battaglia, G. Garon-Carrier, L. Rappaport, M. Brendgen, G. Dionne, F. Vitaro, R. E. Tremblay, M. Boivin, Adolescent pain: Appraisal of the construct and trajectory prediction-by-symptom between age 12 and 17 years in a Canadian twin birth cohort. Pain 163, e1013–e1020 (2022). [DOI] [PubMed] [Google Scholar]
- 74.W. U. Khan, G. Michelini, M. Battaglia, Twin studies of the covariation of pain with depression and anxiety: A systematic review and re-evaluation of critical needs. Neurosci. Biobehav. Rev. 111, 135–148 (2020). [DOI] [PubMed] [Google Scholar]
- 75.J. E. Savage, O. McMichael, E. I. Gorlin, J. R. Beadel, B. Teachman, V. I. Vladimirov, J. M. Hettema, R. Roberson-Nay, Validation of candidate anxiety disorder genes using a carbon dioxide challenge task. Biol. Psychol. 109, 61–66 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.N. K. Leibold, D. L. A. van den Hove, W. Viechtbauer, G. Kenis, L. Goossens, I. Lange, I. Knuts, H. J. Smeets, I. Myin-Germeys, H. W. Steinbusch, K. R. J. Schruers, Amiloride-sensitive cation channel 2 genotype affects the response to a carbon dioxide panic challenge. J. Psychopharmacol. 31, 1294–1301 (2017). [DOI] [PubMed] [Google Scholar]
- 77.N. Gregersen, H. A. Dahl, H. N. Buttenschøn, M. Nyegaard, A. Hedemand, T. D. Als, A. G. Wang, S. Joensen, D. P. Woldbye, P. Koefoed, A. S. Kristensen, T. A. Kruse, A. D. Børglum, O. Mors, A genome-wide study of panic disorder suggests the amiloride-sensitive cation channel 1 as a candidate gene. Eur. J. Hum. Genet. 20, 84–90 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.J. W. Smoller, P. J. Gallagher, L. E. Duncan, L. M. McGrath, S. A. Haddad, A. J. Holmes, A. B. Wolf, S. Hilker, S. R. Block, S. Weill, S. Young, E. Y. Choi, J. F. Rosenbaum, J. Biederman, S. V. Faraone, J. L. Roffman, G. G. Manfro, C. Blaya, D. R. Hirshfeld-Becker, M. B. Stein, M. van Ameringen, D. F. Tolin, M. W. Otto, M. H. Pollack, N. M. Simon, R. L. Buckner, D. Öngür, B. M. Cohen, The human ortholog of acid-sensing ion channel gene ASIC1a is associated with panic disorder and amygdala structure and function. Biol. Psychiatry 76, 902–910 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.P. Fontanillas, A. Kless, J. Bothmer, J. Y. Tung; 23 and Me Research Team , Genome-wide association study of pain sensitivity assessed by questionnaire and the cold pressor test. Pain 163, 1763–1776 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.M. Azzeh, M. Battaglia, S. Davies, J. Strauss, P. Dogra, V. Yellepeddi, Novel intranasal treatment for anxiety disorders using amiloride, an acid-sensing ion channel antagonist: Pharmacokinetic modeling and simulation. Int. J. Clin. Pharmacol. Ther. 60, 253–263 (2022). [DOI] [PubMed] [Google Scholar]
- 81.V. Yellepeddi, C. Sayre, A. Burrows, K. Watt, S. Davies, J. Strauss, M. Battaglia, Stability of extemporaneously compounded amiloride nasal spray. PLOS ONE 15, e0232435 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.V. K. Yellepeddi, M. Battaglia, S. J. C. Davies, J. Alt, S. Ashby, P. Shipman, D. J. Anderson, J. E. Rower, C. Reilly, M. Voight, S. R. Mim, Pharmacokinetics of intranasal amiloride in healthy volunteers. Clin. Transl. Sci. 16, 1075–1084 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.H. Shoji, K. Kato, Maternal care affects the development of maternal behavior in inbred mice. Dev. Psychobiol. 51, 345–357 (2009). [DOI] [PubMed] [Google Scholar]
- 84.A. Barbazanges, M. Vallée, W. Mayo, J. Day, H. Simon, M. Le Moal, S. Maccari, Early and later adoptions have different long-term effects on male rat offspring. J. Neurosci. 16, 7783–7790 (1996). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.J. W. Quintino-dos-Santos, C. J. T. Müller, C. S. Bernabé, C. A. Rosa, S. Tufik, L. C. Schenberg, Evidence that the periaqueductal gray matter mediates the facilitation of panic-like reactions in neonatally-isolated adult rats. PLOS ONE 9, e90726 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.M. Battaglia, A. Ogliari, F. D’Amato, R. Kinkead, Early-life risk factors for panic and separation anxiety disorder: insights and outstanding questions arising from human and animal studies of CO2 sensitivity. Neurosci. Biobehav. Rev. 46, 455–464 (2014). [DOI] [PubMed] [Google Scholar]
- 87.K. S. Kendler, M. C. Neale, R. C. Kessler, A. C. Heath, L. J. Eaves, Childhood parental loss and adult psychopathology in women. A twin study perspective. Arch. Gen. Psychiatry 49, 109–116 (1992). [DOI] [PubMed] [Google Scholar]
- 88.D. S. You, S. Albu, H. Lisenbardt, M. W. Meagher, Cumulative childhood adversity as a risk factor for common chronic pain conditions in young adults. Pain Med. 20, 486–494 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.R. Ventura, R. Coccurello, D. Andolina, E. C. Latagliata, C. Zanettini, V. Lampis, M. Battaglia, F. R. D’Amato, A. Moles, Postnatal aversive experience impairs sensitivity to natural rewards and increases susceptibility to negative events in adult life. Cereb. Cortex 23, 1606–1617 (2013). [DOI] [PubMed] [Google Scholar]
- 90.J. E. Drorbaugh, W. O. Fenn, A barometric method for measuring ventilation in newborn infants. Pediatrics 16, 81–87 (1955). [PubMed] [Google Scholar]
- 91.D. Bartlett Jr., S. M. Tenney, Control of breathing in experimental anemia. Respir. Physiol. 10, 384–395 (1970). [DOI] [PubMed] [Google Scholar]
- 92.R. Kinkead, G. Montandon, A. Bairam, Y. Lajeunesse, R. Horne, Neonatal maternal separation disrupts regulation of sleep and breathing in adult male rats. Sleep 32, 1611–1620 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.K. K. Tai, D. D. Truong, Amiloride but not memantine reduces neurodegeneration, seizures and myoclonic jerks in rats with cardiac arrest-induced global cerebral hypoxia and reperfusion. PLOS ONE 8, e60309 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.K. Hargreaves, R. Dubner, F. Brown, C. Flores, J. Joris, A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 32, 77–88 (1988). [DOI] [PubMed] [Google Scholar]
- 95.J. S. Mogil, J. Ritchie, S. G. Sotocinal, S. B. Smith, S. Croteau, D. J. Levitin, A. K. Naumova, Screening for pain phenotypes: analysis of three congenic mouse strains on a battery of nine nociceptive assays. Pain 126, 24–34 (2006). [DOI] [PubMed] [Google Scholar]
- 96.R. P. Bonin, C. Bories, Y. De Koninck, A simplified up-down method (SUDO) for measuring mechanical nociception in rodents using von Frey filaments. Mol. Pain 10, 26 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.S. R. Chaplan, F. W. Bach, J. W. Pogrel, J. M. Chung, T. L. Yaksh, Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 53, 55–63 (1994). [DOI] [PubMed] [Google Scholar]
- 98.K. B. J. Franklin, G. Paxinos. The Mouse Brain in Stereotaxic Coordinates (Academic Press, 2008).
- 99.L.-E. Lorenzo, A. G. Godin, F. Ferrini, K. Bachand, I. Plasencia-Fernandez, S. Labrecque, A. A. Girard, D. Boudreau, I. Kianicka, M. Gagnon, N. Doyon, A. Ribeiro-da-Silva, Y. de Koninck, Enhancing neuronal chloride extrusion rescues α2/α3 GABAA-mediated analgesia in neuropathic pain. Nat. Commun. 11, 869 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.J. Wu, Y. Xu, Y.-Q. Jiang, J. Xu, Y. Hu, X.-M. Zha, ASIC subunit ratio and differential surface trafficking in the brain. Mol. Brain 9, 4 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.G. G. Consalez, D. Goldowitz, F. Casoni, R. Hawkes, Origins, development, and compartmentation of the granule cells of the cerebellum. Front. Neural Circuits 14, 611841 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.L.-E. Lorenzo, M. Ramien, M. St Louis, Y. De Koninck, A. Ribeiro-da-Silva, Postnatal changes in the Rexed lamination and markers of nociceptive afferents in the superficial dorsal horn of the rat. J. Comp. Neurol. 508, 592–604 (2008). [DOI] [PubMed] [Google Scholar]
- 103.S. Echeverry, X. Q. Shi, M. Yang, H. Huang, Y. Wu, L.-E. Lorenzo, J. Perez-Sanchez, R. P. Bonin, Y. De Koninck, J. Zhang, Spinal microglia are required for long-term maintenance of neuropathic pain. Pain 158, 1792–1801 (2017). [DOI] [PubMed] [Google Scholar]
- 104.F. Ferrini, T. Trang, T.-A. M. Mattioli, S. Laffray, T. Del’Guidice, L.-E. Lorenzo, A. Castonguay, N. Doyon, W. Zhang, A. G. Godin, Morphine hyperalgesia gated through microglia-mediated disruption of neuronal Cl− homeostasis. Nat. Neurosci. 16, 183–192 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.M. Papalampropoulou-Tsiridou, S. Labrecque, A. G. Godin, Y. De Koninck, F. Wang, Differential expression of acid-sensing ion channels in mouse primary afferents in naïve and injured conditions. Front. Cell. Neurosci. 14, 103 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.M. Papalampropoulou-Tsiridou, S. Shiers, F. Wang, A. G. Godin, T. J. Price, Y. De Koninck, Distribution of acid-sensing ion channel subunits in human sensory neurons contrasts with that in rodents. Brain Commun. 4, fcac256 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figs. S1 to S5
Tables S1 and S2
Supplementary Results