Abstract
The olive oil industry faces a major problem of treating the wastewater with high organic content and safe disposal. Olive oil industrial wastewater (OOIWW) consists of highly toxic environmental pollutants with high salinity. Saline olive oil industrial wastewater was treated using halophilic consortium in UMFC (upflow microbial fuel cell) mobilized with carbon felt as electrode. Total and soluble COD (chemical oxygen demand), total suspended solids and phenol content removal were studied at different organic loads (0.56, 0.77, 1.05, 1.26, 1.52 and 1.8 gCOD/L). UMFC with OOIWW was optimized at 1.52 gCOD/L for high organic removal and corresponding electricity production. Total COD, soluble COD, TSS and phenol removal were 91%, 89%, 78%, and complete removal of phenol was accomplished at the optimized organic load (1.52 gCOD/L). Correspondingly, the maximum bioenergy yield was 784 mV with 439 mW/m2 (power density) and 560 mA/m2 (current density), respectively. The presence of prominent halophilic exo-electrogens such as Ochrobactrum, Marinobacter, Rhodococcus and Bacillus potently treated the OOIWW and exhibited high energy yield.
Keywords: Microbial fuel cell, Olive oil wastewater, Halophiles, Bioelectricity, Salinity, Bioenergy
Introduction
The major difficulty in the treatment of olive oil industrial wastewater occurs due to the presence of high quantity of organic pollutants (phenolic compounds), tannins, black to brown colour effluent, phytotoxic and antimicrobial nature (Evci et al. 2019; Shabir et al. 2023). Industrial wastewater treatment needed an efficient treatment technology to solve the environmental problems associated with wastewater discharge (Mao et al. 2021). However, irrespective of the technology performance, key factors such as high-energy requirements, capital cost and problems after waste disposal to be accounted (Mekki et al. 2013). Environmental agencies intensified rules of the waste disposal escalated the wastewater treatment to control pollution with energy production and recovery of useful products from waste (Jayashree et al. 2016). Consumption of olive oil was increased drastically in the kingdom during the recent decades. Kingdom of Saudi Arabia produce olive oil in the northern region such as Tabuk and Al-Jouf (Aly et al. 2014). Al Jouf project area in the kingdom covers 60,000 hectares of land producing different variety of food products, namely wheat, onion, corn, olive and olive oil (Al Jouf 2023). The kingdom produce olive oil industrial wastewater (OOIWW) 160,000 m3 from 23,000 ton olives (Aly et al. 2014) and keep raising based on the olive oil production to 45,000 tons in 2021 (Argaam 2021). The olive oil industries release high amount of effluent, which has the challenge to be treated efficiently to avoid toxic effects on the ecosystem (Zagklis et al. 2013). OOIWW with toxic organic substances (polyphenols), high pH, fatty acids, unpleasant odour, inorganic materials pollutes the environment and causes drastic effect on plants and humans (Shabir et al. 2023). Olive oil wastewater consists of high amount of phenolic compounds, sugars, minerals, tannins and lipids (Peri 2014; Caporaso et al. 2017). The wastewater from olive oil industry is mostly treated by employing evaporation ponds which is a low cost method. The ponds face adequate problems such as leaking, odour and low evaporation rate (Kapellakis et al. 2012). Physicochemical treatment such as coagulant, flocculant, electrocoagulation (Salameh 2015), advanced oxidation process (Agabo-García et al. 2021; Domingues et al. 2022), reverse osmosis (Coskun et al. 2010), membrane filtration (Alfano et al. 2018) and cryogenesis was used to treat olive oil wastewater (Paraskeva et al. 2007; Salameh et al. 2015). The physicochemical treatment methods used reduced the organic pollutant load in the wastewater, which is not economical and still challenging to meet the acceptable discharge limit to reduce the toxic effect of pollutants on the environment (Azzam and Hazaimeh 2021). In addition, the treatment methods require highly skilled technicians to perform the treatment process, which is a complex process and time-consuming and produces high amount of sludge (Shahawy et al. 2021). Olive oil wastewater was treated with biological treatment methods such as aerobic, anaerobic digestion and composting methods (Rharrabti and El Yamani 2019). The existing biological methods was limited by high COD (chemical oxygen demand) content of the phenol-contaminated olive oil industrial wastewater. High phenol concentration in the wastewater inhibits the microbial activity and reduces the secondary treatment efficiency (Al-Qodah et al. 2022). Secondary treatment of OOIWW is unable to achieve complete mineralization of pollutants, when this wastewater released into the water bodies causes drastic effects on the ecosystem (Shahawy et al. 2021). High operational cost and high sludge production are the main drawbacks in implementation of existing OOIWW treatment technology (Shabir et al. 2023). Among different biological methods used, microbial fuel cells (MFC) gained immense attention due to the integrated process of wastewater treatment with low sludge generation and high-energy output (Pugazhendi et al. 2022). Microbial fuel cells are definition for reactors of biological electrochemical processes by microorganisms, intercede with the immediate transformation of artificial vitality in biological substrates and produce electrical vitality. In addition, it has earned extraordinary significance because of its synchronous usage in treatment of wastewater with reducing sludge increase and energy yield vitality (Cao et al. 2019; Pugazhendi et al. 2022). The present research was intended to treat saline OOIWW integrated with bioelectricity generation using halophiles in upflow microbial fuel cell (UMFC). Additionally, the study details the occupancy of electrogenic strains in the consortium during the bioenergy generation process. The study is projected to reveal the application of halophiles in UMFC equipped with carbon felt, which possess high surface area for the treatment of saline OOIWW to meet the discharge limit provided by the environmental agency. Also, energy yield during the wastewater treatment using MFC achieves economical and eco-friendly technology to the industrial sector.
Materials and methods
Sample collection and culture enrichment
OOIWW samples were acquired from Al Jouf Company, Jeddah, Saudi Arabia. Physicochemical wastewater parameters were analysed immediately after the collection of sample from the industry. Salinity of the olive oil industrial wastewater exerted from fermentation in brine (immersion of olive in brine solution for preservation) was in the range of 4 to 5% (40 to 50g/L NaCl concentration). Electro-efficient bacterial strains play vital role in MFC to treat the wastewater coupled with significant energy output (Borole et al. 2011). OOIWW salinity inhibits the growth of non-halophiles and reduces the treatment efficiency (Ferrer-Polonio et al. 2017; Guo et al. 2023). Halophiles tolerated water stress and ionic strength using either osmolytic mechanism or salt in cytoplasm under saline condition (Arulazhagan et al. 2017). Thus, in the present research halophiles enriched from desalination plant were deployed for the OOIWW treatment in MFC.
Physicochemical characterization of wastewater
UMFC reactor with OOIWW was operated at different OLs (organic loads) 0.56, 0.77, 1.05, 1.26, 1.52 and 1.8 gCOD/L. During the operation key wastewater parameters, such as total and soluble chemical oxygen demand (COD) and total suspended solids (TSS), were analysed using standard methods as stated in APHA (2005). Removal of phenol during the treatment of OOIWW in UMFC was analysed using high-performance liquid chromatography (Agilent, USA) as illustrated by Pugazhendi et al. (2022). Raw OOIWW characteristics were pH (5.28), total COD (32 g/L), dissolved solids (5.6 g/L), salinity (46.5 g/L) and phenol (2.5 g/L). Hydraulic retention time (HRT) was varied and identified 20 days HRT as the optimum condition for the wastewater treatment. All the analysis was performed in duplicates and compared with control.
Upflow MFC reactor design and operational conditions
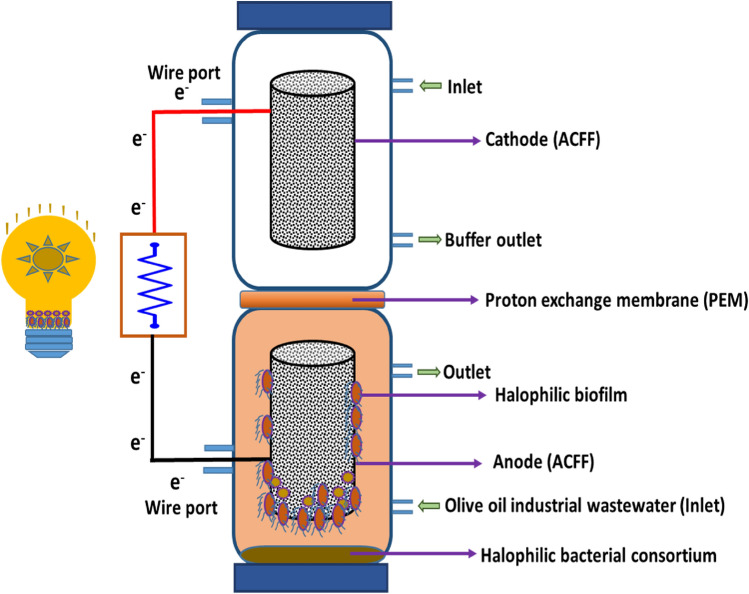
Upflow MFC reactor was incorporated with activated carbon fibre felt with surface area 180 cm2 as anode and cathode for OOIWW treatment and eco-energy yield. UMFC reactor volume was 950 to 1000 mL (Fig. 1). Nafion proton exchange membrane (5 × 5 cm) was used to separate (2 cm distance) anode loaded with OOIWW and phosphate buffer in cathode. Activated carbon fibre felt electrode with high surface area possessed vast number of micropores enhanced organic adsorption capacity, and strong biofilm formation was employed in the UMFC for the treatment of OOIWW (Jayashree et al. 2016).
Fig. 1.
Upflow MFC design for OOIWW treatment and energy production
Bioenergy production measurements
Power and current density
Surface area of the activated carbon felt was accounted to determine the energy (power and current) density in UMFC. Estimation of energy yield by PD (power density) versus CD (current density) plot provides the complete detail on energy output. External resistance from 16,000 to 100 Ω was varied to plot the polarization curve as mentioned by Ghorab et al. (2022)
The below equation was used to quantify the energy production (power and current)
| 1 |
| 2 |
where I = current (A), E = voltage (V), R = external resistance (Ω) and P = Power (W).
Columbic efficiency (CE)
Columbic efficiency is an important parameter to be evaluated during bioenergy production. CE illustrates the electrons derived from substrate mineralization by the electrogenic bacterial strains (Ismail and Jaeel 2013). Thus, CE was estimated using the following equation (Ghorab et al. 2022)
where 8 is the COD constant (4 electrons exchanged per mole of oxygen), I—current (A), F—Faraday constant (96,500 C/mol-electrons), q—flow rate (m3/sec) and ΔCOD—COD removal (g/L).
Phylogenetic analysis
Qiagen DNA extraction kit was used to extract bacterial DNA and thermal cycler was used to amplify the DNA (Applied Biosystems, USA). PCR (polymerase chain reaction) was executed with primers (27F and 1492R). PCR amplification was accomplished by using PCR supermix following Arulazhagan and Vasudevan’s (2009) protocol details. The high-throughput sequencing (HTS) was performed as stated by Pugazhendi et al. (2022). The nucleotide sequence from HTS was analysed in BLASTN search for the bacterial strains genus confirmation. Further accession number for the strains was obtained by submitting the results to NCBI (National Center for Biotechnology Information).
Results and discussion
Effect of OL on the treatment of OOIWW
Total COD removal efficacy at various OLs
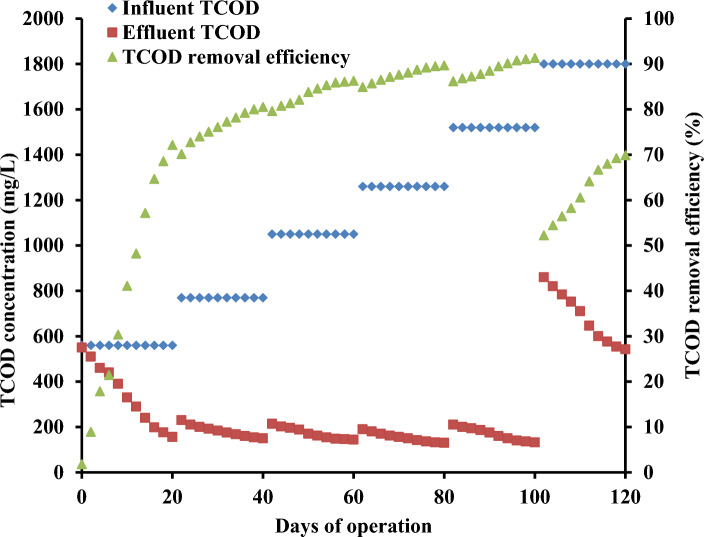
UMFC provided with carbon felt in anode and cathode chamber is separated by proton exchange membrane. The flow of wastewater occurs in upward direction in the anode chamber. Anode with biofilm treats the wastewater and releases electrons towards energy acceptor terminal for energy production (Jayashree et al. 2016). COD is the key wastewater parameter represent the organic substrate in wastewater influence the dissolved oxygen, release of high COD wastewater harm the environment (Das and Calay 2022). Figure 2 illustrates the efficacy of total COD removal in the UMFC system operated with OOIWW at different OL values. Initially, 0.56 gCOD/L OL established 72% total COD removal efficacy (Fig. 2). The total COD removal of 81%, 86% and 90% was displayed at 0.77 gCOD/L, 1.05 gCOD/L and 1.26 gCOD/L OL, respectively. Significant total COD removal of 91% was attained at 1.52 gCOD/L OL. Main factors such as quality of the substrate and adsorption of bacterial strains on anode were responsible for eminent COD removal efficiency in MFC (Rahmani et al. 2023). Bermek et al. (2014) detected 65% COD removal efficiency in air cathode MFC during the treatment of olive mill wastewater. Subsequent increment in OL to 1.8 gCOD/L displayed 70% total COD removal potential in UMFC. The drop in total COD removal efficiency is due to insufficient retention time required to mineralize the substrates in the wastewater (Buitron and Moreno-Andrade 2014). Aaron et al. (2010) revealed that flow rate and ionic strength highly influence treatment efficiency and power generation ability in MFC. High OL with elevated phenol concentration affects the bacterial growth and suspends transfer of electrons in the reactor (Jayashree et al. 2015; Pugazhendi et al. 2022).
Fig. 2.
Total COD removal efficiency
Soluble COD removal efficacy at various OLs
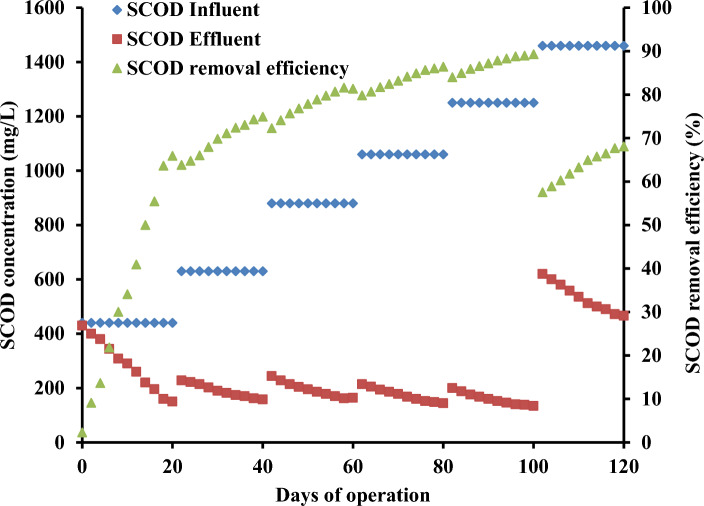
Soluble COD removal in the wastewater was used to demonstrate the UMFC treatment efficiency. Figure 3 illustrates 66%, 75%, 81% and 86% soluble COD removal efficiency at 0.56, 0.77, 1.05 and 1.26 gCOD/L OL, respectively. Magnificent soluble COD removal of 89% was displayed at 1.52 gCOD/L (Fig. 3). Efficient soluble COD removal in UMFC evidently authenticates the adaptation of halophilic strains to phenol-contaminated OOIWW. Soluble COD removal dropped to 68% was observed at increased OL of 1.8 gCOD/L during the treatment of OOIWW. The drop in soluble COD removal is attained due to the non-electrogens compete with electrogens and incomplete mineralization of substrates (Li et al. 2018; Subha et al. 2019; Pugazhendi et al. 2021). Previous researchers employed olive mill wastewater as co-substrate to promote the wastewater treatment and energy generation in MFC (Sciarria et al. 2013; Akgul et al. 2021).
Fig. 3.
SCOD removal efficiency
TSS removal efficacy at various OLs
TSS removal in UMFC demonstrated an increasing pattern along with increase in OL. TSS removal was 47%, 55%, 60% and 71% at 0.56, 0.77, 1.05 and 1.26 gCOD/L OL (Fig. 4). Prominent TSS removal of 78% was exhibited at 1.52 gCOD/L. TSS removal efficiency was in accordance with previous research on saline wastewater treatment in MFC (Pugazhendi et al. 2021; Ghorab et al. 2022). Tsekouras et al. (2022) demonstrated adhesive biofilm formation on anode lead to reduce the organic suspended solids. Karlikanovaite-Balıkci and Ozgun (2020) identified removal of organic solids by electrogens in MFC resulted in low sludge generation. Decrement in TSS removal of 63% was exhibited at increased OL of 1.8 gCOD/L. Previous researches illustrated high TSS proportion in wastewater and fermentation intermediates restricted the growth of electrogens in MFC (Ismail and Radeef 2020; Pugazhendi et al. 2022).
Fig. 4.
TSS removal efficiency
Phenol removal efficacy at various OLs
Phenol as one of the key pollutants present in OOIWW is to be removed from the wastewater. The halophilic consortium efficiently utilized phenol as carbon substrate for bioelectricity production. Phenol was completely removed in all the OL, whereas the time required for degradation varied with the OL (Table 1). UMFC operated at 0.56 and 0.77 gCOD/L OL displayed complete removal in 2 days. Time required for complete phenol degradation at OL 1.05 gCOD/L OL increased to 3 days. Raise in OL to 1.26, 1.52 and 1.8 gCOD/L disclosed 100%, 96% and 85% phenol removal, and time taken for degradation increased to 4, 5 and 7 days, respectively. Previous study by Bermek et al. (2014) observed 49% phenol removal in 3 days during the treatment of OOIWW in upflow MFC. Compared to the previous study, the present study revealed complete removal of phenol (2 and 3 days) at different OLs under saline condition.
Table 1.
Phenol removal efficiency at various OLs in UMFC operated with OOIWW
| OL (gCOD/L) | Phenol concentration (mg/L) | Phenol removal (%) | Time (Days) |
|---|---|---|---|
| 0.56 | 242 | 100 | 2 |
| 0.77 | 385 | 100 | 2 |
| 1.05 | 440 | 100 | 3 |
| 1.26 | 618 | 100 | 4 |
| 1.52 | 766 | 96 | 5 |
| 1.80 | 900 | 85 | 7 |
Energy yield from UMFC with OOIWW
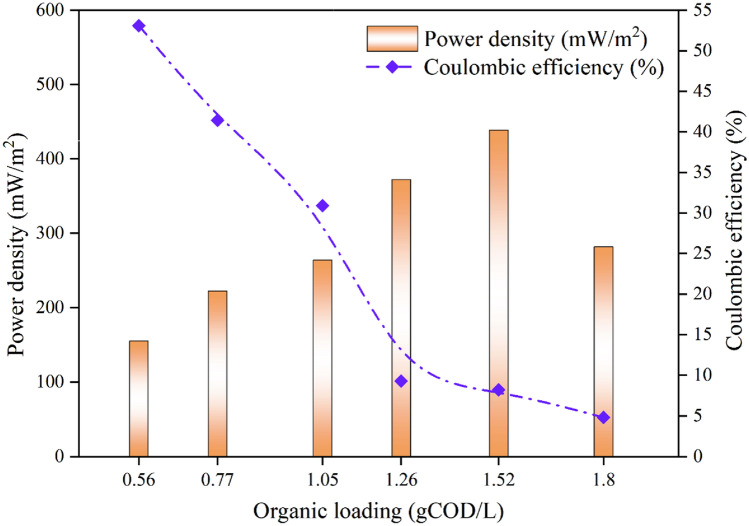
Energy yield of 155 mW/m2 PD and 333 mA/m2 of CD was accounted at 0.56 gCOD/L OL in UMFC with OOIWW (Fig. 5a, b). OL incremented to 0.77 gCOD/L acquired energy yield of 222 mW/m2 and 399 mA/m2. PD (264 mW/m2 and 372 mW/m2) and CD (434 mA/m2 and 516 mA/m2) energy yield was accounted at 1.05 gCOD/L and 1.26 gCOD/L OL, respectively. Considerable amount of energy (PD-439 mW/m2 and CD-560 mA/m2) was generated at 1.52 gCOD/L OL. Decrease in energy yield of 282 mW/m2 (PD) and 440 mA/m2 (CD) was observed at increased OL of 1.8 gCOD/L. Kondavetti et al. (2014) and Karuppiah et al. (2018) described clearly that the decline in energy yield was due to ohmic losses associated with high organic load and space between the electrodes. Miyahara et al. (2015) reported that salinity plays major role in exo-electrogenic bacterial growth, which is responsible for energy output from substrate utilization.
Fig. 5.
a Current density Vs power density in UMFC with OOIWW. b Current density Vs voltage in UMFC with OOIWW
CE quantifies the electrons gained by microbial substrate mineralization in UMFC with OOIWW. Initial OL (0.56 gCOD/L) achieved 53% of CE. OL gradually raised to 0.77, 1.05, 1.26, 1.52 and 1.8 gCOD/L disclosed a decline pattern in CE of 41.5%, 31%, 9.3%, 8.7% and 4.8%, respectively (Fig. 6). Reduction in CE was reported because (i) at high OL electrogens compete with other bacterial strains (Pugazhendi et al. 2022), (ii) substrate mineralization restricted to bacterial growth without energy yield at high external resistance (Li et al. 2016), (iii) loading MFC with real wastewater enhances methanogenesis or fermentation process (Karuppiah et al. 2018) and (iv) oxidation (oxygen–electron acceptor) during substrate mineralization by bacterial strain (Pugazhendi et al. 2020).
Fig. 6.
Columbic efficiency in UMFC with OOIWW
Identification of electrogens
The presence and dominance of electroactive bacterial strains in the anode region of the upflow MFC were investigated at different organic loadings (OLs). Ochrobactrum, Marinobacter, Rhodococcus and Bacillus exhibited high dominance in all the OL (Table 2). Further Stenotrophomonas, Pseudomonas, Acinetobacter, Sphingomonas and Sedimentibacter were present at low percent (< 8%) in the anode chamber. Organic substrates concentration and substrate mineralization capacity of the bacterial strains determine the bacterial occupancy percent in bacterial profile (Pugazhendi et al. 2022). Electron transfer for energy yield in MFC was executed by two ways: i) bacterial pili attachment with the anode (Lienemann et al. 2018) and ii) direct electrode contact with bacterial outer membrane cytochromes facilitating electron transport (Wang et al. 2019; Yasri et al. 2019; Roy et al. 2022). The microbes mineralize the organic substrates in wastewater and harvest electrons, and pili bounded with bacterial cytochromes allow the electrons to transfer towards the electron acceptor terminal for energy production. C-type cytochromes with redox proteins present in the outer membrane of the bacterial strains facilitate the electron towards electron acceptor terminal to produce energy (Aiyer 2020). Ochrobactrum possess peritrichous flagella and execute energy production by adhesion to the anode in MFC for electron transfer (Arulmani et al. 2020). Marinobacter and Rhodococcus with pili attach to the anode for electron transfer (Mounier et al. 2014; Ivshina et al. 2022). Bacillus strains form biofilm by direct anode contact and also production of biosurfactant facilitates the direct electron transfer (Gomaa et al. 2021). Thus in the present study, the presence of pili and direct contact of the strains accelerate the electron transport in UMFC. At optimized OL (1.52 gCOD/L), the dominant electroactive strains were Ochrobactrum (52%), Marinobacter (35%), Rhodococcus (5%) and Bacillus (3.5%), respectively. Ochrobactrum and Marinobacter were widely reported for biodegradation of organic pollutants (Jayashree et al. 2016; Pugazhendi et al. 2017, 2022; Rimboud et al. 2021) and energy generation in air cathode and upflow MFC under saline condition (Jamal and Pugazhendi 2021). Thus, the electrogenic strains (Ochrobactrum and Marinobacter) with fastidious growth on organic substrates exhibited maximum occupancy at optimized OL with corresponding energy yield in UMFC. Previous research by Miyahara et al. (2015) revealed that at 0.1 M salinity Geobacter strain was dominant in the anode region, and increase in salinity to 0.3 M salinity inhibited Geobacter growth. The study at 0.3 M salinity exhibited the dominance of Pseudomonas, Stenotrophomonas, Rhodococcus and Ochrobactrum acted as potential exo-electrogenic bacterial strains involved in electricity production in single-chamber MFC. Thus, the presence of potential exo-electrogens in UMFC was in accordance with previous studies on saline wastewater treatment and energy yield.
Table 2.
Dominance profile of electrogenic bacterial strains in energy production at different OLs
| Bacterial strain | Bacterial community occupancy at different OLs (%) | ||||||
|---|---|---|---|---|---|---|---|
| Organic Load (gCOD/L) | Accession Number | ||||||
| 0.56 | 0.77 | 1.05 | 1.26 | 1.52 | 1.8 | ||
| Ochrobactrum | 32 | 36 | 41 | 45 | 52 | 35.5 | OQ996124 |
| Marinobacter | 21 | 25 | 28 | 32 | 35 | 23.5 | OQ996125 |
| Rhodococcus | 12 | 10.5 | 11 | 8 | 5 | 11.5 | OQ996126 |
| Bacillus | 11 | 11 | 12 | 5 | 3.5 | 12 | OQ996127 |
| Stenotrophomonas | 7 | 5.5 | 3.5 | 4.5 | 2 | 5.5 | OQ996128 |
| Pseudomonas | 7 | 5 | 1.5 | 1.5 | 1 | 6 | OQ996129 |
| Acinetobacter | 7 | 5 | 2 | 3 | 1.5 | 4.5 | OQ996130 |
| Sphingomonas | 2 | 1 | 1 | 1 | 0 | 1.5 | OQ996131 |
| Sedimentibacter | 1 | 1 | 0 | 0 | 0 | 0 | OQ996132 |
MFC technology undergone several improvements in the past two decades for treating real wastewater under large scale (Tsekouras et al. 2022). Transfer of MFC technology still requires several factors to be considered such as electrode, HRT, reactor design, organic flow rate and potential exo-electrogenic strains (Nidheesh et al. 2022). Electrode selected for the anode should possess large surface area (for biofilm formation) and survive for longer period for the wastewater treatment process (Rousseau et al. 2020). Platinum, nickel, cobalt and stainless steel alloys with high porosity were used as cathode for cathodic reaction in MFC. Mostly carbon-based electrodes are selected to meet the desired quality and energy production (Chandrasekhar 2019; Nidheesh et al. 2022). Sorgato et al. (2023) reported HRT, the time required for the bacterial strains to mineralize the organic substrate that acts as key factor to decide the fate of wastewater treatment with removal of toxic pollutants (COD removal efficiency) in MFC. Parallel plate reactors and concentric cylinder-type designs with high space to accommodate electrodes were mostly employed to treat wastewater in large scale in MFC (Alonso et al. 2020; Wu et al. 2020). Organic flow rate in large-scale MFC should be optimized prior to operation. The organic flow highly influences the MFC performance, and increase or decrease in flow rate saturates the electrode and inhibits the bacterial growth (Ye et al. 2019). Exo-electrogens selection acts as the key factor to accelerate the MFC to harvest high energy yield (Logan 2009). MFC is employed in the secondary wastewater treatment with parallel plate connections feasible to operate for high-energy yield (Alonso et al. 2020). Implementation of MFC in domestic and industrial wastewater treatment with corresponding energy yield required to account various key factors, which evaluates the capital and operational cost.
Conclusions
Treatment of saline OOIWW in UMFC at optimized OL (1.52 gCOD/L) reduced TCOD (91%), SCOD (89%), TSS (78%) and phenol (96%), respectively. Eminent energy yield of 784 mV with respective PD (439 mW/m2) and CD (560 mA/m2) was detected at optimized OL. Dominance of exo-electrogens (Ochrobactrum, Marinobacter, Rhodococcus and Bacillus) authenticates the mineralization of organic pollutants in saline OOIWW integrated with efficient energy yield. Thus, the research concludes to assign exo-electrogenic halophiles for saline OOIWW treatment with corresponding energy harvest in MFC. Further studies on application of halophilic consortium at pilot-scale-level MFC will enhance the transfer of technology to the industry.
Accession number
The authors submitted the nucleotide sequence of bacterial strains and obtained the respective accession number from NCBI (National Center for Biotechnology Information) as mentioned below:
Ochrobactrum (OQ996124), Marinobacter (OQ996125), Rhodococcus (OQ996126), Bacillus (OQ996127), Stenotrophomonas (OQ996128), Pseudomonas (OQ996129), Acinetobacter (OQ996130), Sphingomonas (OQ996131) and Sedimentibacter (OQ996132).
Acknowledgements
This project was funded by Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah, under grant no. G-157-150-1441. The authors, therefore, acknowledge with thanks DSR for technical and financial support.
Author contribution
AP contributed to conceptualization, investigation methodology, data curation, validation, writing—original draft, and writing—review and editing. MTJ was involved in project administration, funding acquisition, methodology, data curation, and validation.
Data availability
The accession number obtained from the NCBI (National Center for Biotechnology) for the bacterial strains involved in the research was provided as per Journal guidelines. The data on bacterial strains and accession number is included in Table 2 of the article. With the accession number the readers can obtain the nucleotide sequence data from NCBI website using BLASTN search online software.
Declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships and report no commercial or proprietary interest in any product or concept that could have appeared to influence the work reported in this paper.
Research involves human or animals participation
This research work did not involve any human participation and/or animals.
Informed consent
All authors take responsibility for the integrity of the work as a whole, from inception to finished article. All the authors approve the submission of the article in 3-Biotech Journal.
References
- Aaron D, Tsouris C, Hamilton CY, Borole AP. Assessment of the effects of flow rate and ionic strength on the performance of an air-cathode microbial fuel cell using electrochemical impedance spectroscopy. Energies. 2010;3:592–606. doi: 10.3390/en3040592. [DOI] [Google Scholar]
- Agabo-García C, Calderon N, Hodaifa G. Heterogeneous photo-fenton reaction for olive mill wastewater treatment-case of reusable catalyst. Catalysts. 2021;11(5):557. doi: 10.3390/catal11050557. [DOI] [Google Scholar]
- Aiyer KS. How does electron transfer occur in microbial fuel cells? World J Microbiol Biotechnol. 2020;36:19. doi: 10.1007/s11274-020-2801-z. [DOI] [PubMed] [Google Scholar]
- Akgul V, Cirik K, Duyar A, Koroglu EO. The effect of olive oil mill and molasses wastewater as a co-substrate during simultaneous textile wastewater treatment and energy generation. Int J Global Warm. 2021;23:30–41. doi: 10.1504/IJGW.2021.112486. [DOI] [Google Scholar]
- Alfano A, Corsuto L, Finamore R, Savarese M, Ferrara F, Falco S, Santabarbara G, De Rosa M, Schiraldi C. Valorization of olive mill wastewater by membrane processes to recover natural antioxidant compounds for cosmeceutical and nutraceutical applications or functional foods. Antioxidants (basel) 2018;7(6):72. doi: 10.3390/antiox7060072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alonso RM, San-Martin MI, Mateos R, Moran A, Escapa A. Scale-up of bioelectrochemical systems for energy valorization of waste streams. In: Tiquia-Arashiro SM, Pant D, editors. Microb Electrochem Technol. Boca Raton: CRC Press; 2020. pp. 447–459. [Google Scholar]
- Al-Qodah Z, Al-Zoubi H, Hudaib B, Omar W, Soleimani M, Abu-Romman S, Frontistis Z. Sustainable vs. conventional approach for olive oil wastewater management: a review of the state of the art. Water. 2022;14:1695. doi: 10.3390/w14111695. [DOI] [Google Scholar]
- Aly AA, Hasan YNY, Al-Farraj AS. Olive mill wastewater treatment using a simple zeolite-based low-cost method. J Environ Manag. 2014;145:341–348. doi: 10.1016/j.jenvman.2014.07.012. [DOI] [PubMed] [Google Scholar]
- APHA (American Public Health Association) Standard methods for the examination of water and wastewater. 21. Washington: APHA; 2005. [Google Scholar]
- Argaam (2021) Al-Jouf Agricultural CEO says Saudi olive oil market sized at 45,000 tons https://www.argaam.com/en/article/articledetail/id/1471856#:~:text=Saudi%20Arabia's%20olive%20oil%20industry,Allied%20Industries%20(Expoliva%202021)
- Arulazhagan P, Vasudevan N. Role of moderately halophilic bacterial consortium in biodegradation of polyaromatic hydrocarbons. Mar Poll Bull. 2009;58:256–262. doi: 10.1016/j.marpolbul.2008.09.017. [DOI] [PubMed] [Google Scholar]
- Arulazhagan P, Mnif S, Rajesh Banu J, Huda Q, Jalal MAB. HC-0B-01: biodegradation of hydrocarbons by extremophiles. In: Heimann K, Karthikeyan O, Muthu S, editors. Biodegradation and bioconversion of hydrocarbons. Environmental footprints and eco-design of products and processes. Singapore: Springer; 2017. pp. 137–162. [Google Scholar]
- Arulmani SRB, Ganamuthu HL, Ashokkumar V, Govindarajan G, Kandasamy S, Zhang SH. Biofilm formation and electrochemical metabolic activity of Ochrobactrum Sp JSRB-1 and Cupriavidus Sp JSRB-2 for energy production. Environ Technol Innov. 2020;20:101145. doi: 10.1016/j.eti.2020.101145. [DOI] [Google Scholar]
- Azzam MOJ, Hazaimeh SA. Olive mill wastewater treatment and valorization by extraction/concentration of hydroxytyrosol and other natural phenols. Proc Saf Environ Protec. 2021;148:495–523. doi: 10.1016/j.psep.2020.10.030. [DOI] [Google Scholar]
- Bermek H, Catal T, Akan SS, Ulutas MS, Kumru M, Ozguven M, Liu H, Ozcelik B, Akarsubasi AT. Olive mill wastewater treatment in single-chamber air-cathode microbial fuel cells. World J Microbiol Biotechnol. 2014;30:1177–1185. doi: 10.1007/s11274-013-1541-8. [DOI] [PubMed] [Google Scholar]
- Borole AP, Reguera G, Ringeisen B, Wang ZW, Feng Y, Kim BH. Electroactive biofilms: current status and future research needs. Ener Environ Sci. 2011;4(12):4813–4834. doi: 10.1039/C1EE02511B. [DOI] [Google Scholar]
- Buitron G, Moreno-Andrade I. Performance of a single-chamber microbial fuel cell degrading phenol: effect of phenol concentration and external resistance. Appl Biochem Biotechnol. 2014;174:2471–2481. doi: 10.1007/s12010-014-1195-5. [DOI] [PubMed] [Google Scholar]
- Cao Y, Mu H, Liu W, Zhang R, Guo J, Xian M, Liu H. Electricigens in the anode of microbial fuel cells: pure cultures versus mixed communities. Microb Cell Fact. 2019;18:39. doi: 10.1186/s12934-019-1087-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caporaso N, Formisano D, Genovese A. Use of phenolic compounds from olive mill wastewater as valuable ingredients for functional foods. Crit Rev Food Sci Nutrit. 2017;58(16):2829–2841. doi: 10.1080/10408398.2017.1343797. [DOI] [PubMed] [Google Scholar]
- Chandrasekhar K. Effective and nonprecious cathode catalysts for oxygen reduction reaction in microbial fuel cells. In: Mohan SV, Varjani S, Pandey A, editors. Microb Electrochem Technol. Amsterdam: Elsevier; 2019. pp. 485–501. [Google Scholar]
- Coskun T, Debik E, Demir NM. Treatment of olive mill wastewaters by nanofiltration and reverse osmosis membranes. Desalination. 2010;259:65–70. doi: 10.1016/j.desal.2010.04.034. [DOI] [Google Scholar]
- Das S, Calay RK. Experimental study of power generation and cod removal efficiency by air cathode microbial fuel cell using Shewanella baltica 20. Energies. 2022;15:4152. doi: 10.3390/en15114152. [DOI] [Google Scholar]
- Domingues E, Fernandes E, Gomes J, Castro-Silva S, Martins RC. Advanced oxidation processes at ambient conditions for olive oil extraction industry wastewater degradation. Chem Eng Sci. 2022;263:118076. doi: 10.1016/j.ces.2022.118076. [DOI] [Google Scholar]
- Evci E, Yılmaz Ö, Bekfelavi EY, Kus NS. Degradation of olive mill wastewater by different methods and antioxidant activity of olive mill wastewater extraction. SN Appl Sci. 2019;1:1206. doi: 10.1007/s42452-019-1210-0. [DOI] [Google Scholar]
- Ferrer-Polonio E, Carbonell-Alcaina C, Mendoza-Roca JA, Iborra-Clar A, Álvarez-Blanco S, Bes-Piá A, Pastor-Alcañiz L. Brine recovery from hypersaline wastewaters from table olive processing by combination of biological treatment and membrane Technologies. J Clean Prod. 2017;142:1377–1386. doi: 10.1016/j.jclepro.2016.11.169. [DOI] [Google Scholar]
- Ghorab REA, Pugazhendi A, Jamal MT, Jeyakumar RB, Godon JJ, Mathew DK. Tannery wastewater treatment coupled with bioenergy production in upflow microbial fuel cell under saline condition. Environ Res. 2022;212:113304. doi: 10.1016/j.envres.2022.113304. [DOI] [PubMed] [Google Scholar]
- Gomaa OM, Selim N, Fathy R, Maghrawy HH, Gamal M, El Kareem HA, Kyazze G, Keshavarz T. Characterization of a biosurfactant producing electroactive Bacillus sp for enhanced Microbial Fuel Cell dye decolourisation. Enz Microb Technol. 2021;147:109767. doi: 10.1016/j.enzmictec.2021.109767. [DOI] [PubMed] [Google Scholar]
- Guo L, Xie Y, Sun W, Xu Y, Sun Y. Research progress of high-salinity wastewater treatment technology. Water. 2023;15(4):684. doi: 10.3390/w15040684v. [DOI] [Google Scholar]
- Ismail ZZ, Jaeel AJ. Sustainable power generation in continuous flow microbial fuel cell treating actual wastewater: influence of biocatalyst type on electricity production. Sci World J. 2013 doi: 10.1155/2013/713515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ismail ZZ, Radeef AY. The effect of total suspended solids on the electricity generation in microbial fuel cell treating actual potato chips processing wastewater. J Eng. 2020;26:55–62. doi: 10.31026/j.eng.2020.01.06. [DOI] [Google Scholar]
- Ivshina IB, Krivoruchko AV, Kuyukina MS, Peshkur TA, Cunningham CJ. Adhesion of Rhodococcus bacteria to solid hydrocarbons and enhanced biodegradation of these compounds. Sci Rep. 2022;12:21559. doi: 10.1038/s41598-022-26173-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jamal MT, Pugazhendi A. Treatment of fish market wastewater and energy production using halophiles in air cathode microbial fuel cell. J Environ Manag. 2021;292:112752. doi: 10.1016/j.jenvman.2021.112752. [DOI] [PubMed] [Google Scholar]
- Jayashree C, Sweta S, Arulazhagan P, Yeom IT, Iqbal MII, Banu JR. Electricity generation from retting wastewater consisting of recalcitrant compounds using continuous upflow microbial fuel cell. Biotechnol Bioproc Eng. 2015;20:753–759. doi: 10.1007/s12257-015-0017-0. [DOI] [Google Scholar]
- Jayashree C, Tamilarasan K, Rajkumar M, Arulazhagan P, Yogalakshmi KN, Srikanth M, Rajesh Banu J. Treatment of seafood processing wastewater using upflow microbial fuel cell for power generation and identification of bacterial community in anodic biofilm. J Environ Manag. 2016;180:351–358. doi: 10.1016/j.jenvman.2016.05.050. [DOI] [PubMed] [Google Scholar]
- Al Jouf (2023) https://ajcci.org.sa/for-al-jouf?lang=en
- Kapellakis IE, Paranychianakis NV, Tsagarakis KP, Angelakis AN. Treatment of olive mill wastewater with constructed wetlands. Water. 2012;4:260–271. doi: 10.3390/w4010260. [DOI] [Google Scholar]
- Karlikanovaite-Balıkci A, Ozgun OK (2020) Generation of electricity and sludge reduction in a microbial fuel cell. J Int Environ Appl Sci 15:141–151. https://dergipark.org.tr/en/pub/jieas/issue/55894/752164
- Karuppiah T, Pugazhendi A, Subramanian S, Jamal MT, Jeyakumar RB. Deriving electricity from dye processing wastewater using single chamber microbial fuel cell with carbon brush anode and platinum nano coated air cathode. Biotech. 2018;8:437. doi: 10.1007/s13205-018-1462-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kondavetti S, Lee J, Kakarla R, Kim HS, Min B. Low-cost separators for enhanced power production and field application of microbial fuel cells (MFCs) Electrochim Acta. 2014;132:434–440. doi: 10.1016/j.electacta.2014.03.046. [DOI] [Google Scholar]
- Li N, Kakarla R, Min B. Effect of influential factors on microbial growth and the correlation between current generation and biomass in an air cathode microbial fuel cell. Int J Hyd Ener. 2016;41:20606–20614. doi: 10.1016/j.ijhydene.2016.09.094. [DOI] [Google Scholar]
- Li M, Zhou M, Tian X, Tan C, McDaniel CT, Hassett DJ, Gu T. Microbial fuel cell (MFC) power performance improvement through enhanced microbial electrogenicity. Biotechnol Adv. 2018;36:1316–1327. doi: 10.1016/j.biotechadv.2018.04.010. [DOI] [PubMed] [Google Scholar]
- Lienemann M, TerAvest MA, Pitkänen JP, Stuns I, Penttilä M, Ajo-Franklin CM, Jäntti J. Towards patterned bioelectronics: facilitate dimmobilization of exoelectrogenic Escherichia coli with heterologous pili. Microbial Biotechnol. 2018;11(6):1184–1194. doi: 10.1111/1751-7915.13309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Logan B. Exoelectrogenic bacteria that power microbial fuel cells. Nat Rev Microbiol. 2009;7:375–381. doi: 10.1038/nrmicro2113. [DOI] [PubMed] [Google Scholar]
- Mao G, Hu H, Liu X, Crittenden J, Huang N. A bibliometric analysis of industrial wastewater treatments from 1998 to 2019. Environ Poll. 2021;275:115785. doi: 10.1016/j.envpol.2020.115785. [DOI] [PubMed] [Google Scholar]
- Mekki A, Dhouib A, Sayadi S. Review: effects of olive mill wastewater application on soil properties and plants growth. Int J Recyc Organic Waste Agric. 2013;2:15. doi: 10.1186/2251-7715-2-15. [DOI] [Google Scholar]
- Miyahara M, Kouzuma A, Watanabe K. Effects of NaCl concentration on anode microbes in microbial fuel cells. AMB Express. 2015;5(1):123. doi: 10.1186/s13568-015-0123-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mounier J, Camus A, Mitteau I, Vaysse PJ, Goulas P, Grimaud R, Sivadon The marine bacterium Marinobacter hydrocarbonoclasticus SP17 degrades a wide range of lipids and hydrocarbons through the formation of oleolytic biofilms with distinct gene expression profiles. FEMS Microbiol Ecol. 2014;90(3):816–831. doi: 10.1111/1574-6941.12439. [DOI] [PubMed] [Google Scholar]
- Nidheesh PV, Ganiyu SO, Kuppam C, Mousset E, Samsudeen N, Olvera-Vargas H, Kumar G. Bioelectrochemical cells as a green energy source for electrochemical treatment of water and wastewater. J Water Proc Eng. 2022;50:103232. doi: 10.1016/j.jwpe.2022.103232. [DOI] [Google Scholar]
- Paraskeva CA, Papadakis VG, Tsarouchi E, Kanellopoulou DG, Koutsoukos PG. Membrane processing for olive mill wastewater fractionation. Desalination. 2007;213(1–3):218–229. doi: 10.1016/j.desal.2006.04.087. [DOI] [Google Scholar]
- Peri C. The extra-virgin olive oil handbook. Chichester: Wiley; 2014. [Google Scholar]
- Pugazhendi A, Qari H, Basahi JMA, Godon JJ, Dhavamani J. Role of a halothermophilic bacterial consortium for the biodegradation of PAHs and the treatment of petroleum wastewater at extreme conditions. Int Biodeter Biodeg. 2017;121:44–54. doi: 10.1016/j.ibiod.2017.03.015. [DOI] [Google Scholar]
- Pugazhendi A, Al-Mutairi AE, Jamal MT, Jeyakumar RB, Palanisamy K. Treatment of seafood industrial wastewater coupled with electricity production using air cathode microbial fuel cell under saline condition. Int J Ener Res. 2020;44:12535–12545. doi: 10.1002/er.5774. [DOI] [Google Scholar]
- Pugazhendi A, Alreeshi GG, Jamal MT, Karuppiah T, Jeyakumar RB. Bioenergy production and treatment of aquaculture wastewater using saline anode microbial fuel cell under saline condition. Environ Technol Innovat. 2021;21:101331. doi: 10.1016/j.eti.2020.101331. [DOI] [Google Scholar]
- Pugazhendi A, Jamal MT, Al-Mur BA, Jeyakumar RB. Bioaugmentation of electrogenic halophiles in the treatment of pharmaceutical industrial wastewater and energy production in microbial fuel cell under saline condition. Chemosphere. 2022;288:132515. doi: 10.1016/j.chemosphere.2021.132515. [DOI] [PubMed] [Google Scholar]
- Rahmani AR, Navidjouy N, Rahimnejad M, Alizadeh S, Samarghandi MR, Nematollahi D. Effect of different concentrations of substrate in microbial fuel cells toward bioenergy recovery and simultaneous wastewater treatment. Environ Technol. 2023;43:1–9. doi: 10.1080/09593330.2020.1772374. [DOI] [PubMed] [Google Scholar]
- Rharrabti Y, Yamani EI, M, Olive mill wastewater: Treatment and valorization technologies. In: Hussain C, editor. Handbook of environmental materials management. Cham: Springer; 2019. pp. 1659–1686. [Google Scholar]
- Rimboud M, Etcheverry L, Barakat M, Achouak W, Bergel A, Délia ML. Hypersaline microbial fuel cell equipped with an oxygen-reducing microbial cathode. Bioresour Technol. 2021;337:125448. doi: 10.1016/j.biortech.2021.125448. [DOI] [PubMed] [Google Scholar]
- Rousseau R, Etcheverry L, Roubaud E, Basseguy R, Delia ML, Bergel A. Microbial electrolysis cell (MEC): strengths, weaknesses and research needs from electrochemical engineering standpoint. Appl Ener. 2020;257:113938. doi: 10.1016/j.apenergy.2019.113938. [DOI] [Google Scholar]
- Roy AS, Sharma A, Thapa BS, Pandit S, Lahiri D, Nag M, Sarkar T, Pati S, Ray RR, Shariati MA, Wilairatana P, Mubarak MS. Microbiomics for enhancing electron transfer in an electrochemical system. Front Microbiol. 2022;13:868220. doi: 10.3389/fmicb.2022.868220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salameh WKB. Treatment of olive mill wastewater by ozonation and electrocoagulation processes. Glob Adv Res J Eng Technol Innov. 2015;7:80–91. [Google Scholar]
- Salameh WKB, Ahmad H, Al-Shannag M. Treatment of olive mill wastewater by electrocoagulation processes and water resources management. Int J Environ Chem Ecol Geol Geophy Eng. 2015;9:296–300. [Google Scholar]
- Sciarria TP, Tenca A, D’Epifanio A, Mecheri B, Merlino G, Barbato M, Borin S, Licoccia S, Garavaglia V, Adani F. Using olive mill wastewater to improve performance in producing electricity from domestic wastewater by using single-chamber microbial fuel cell. Bioresour Technol. 2013;147:246–253. doi: 10.1016/j.biortech.2013.08.033. [DOI] [PubMed] [Google Scholar]
- Shabir S, Ilyas N, Saeed M, Bibi F, Sayyed RZ, Almalki WH. Treatment technologies for olive mill wastewater with impacts on plants. Environ Res. 2023;216:114399. doi: 10.1016/j.envres.2022.114399. [DOI] [PubMed] [Google Scholar]
- Shahawy AE, Ahmed IA, Nasr M, Ragab AH, Al-Mhyawi SR, Elamin KMA. Organic pollutants removal from olive mill wastewater using electrocoagulation process via central composite design (CCD) Water. 2021;13:3522. doi: 10.3390/w13243522. [DOI] [Google Scholar]
- Sorgato AC, Jeremias TC, Lobo FL, Lapolli FR. Microbial fuel cell: interplay of energy production, wastewater treatment, toxicity assessment with hydraulic retention time. Environ Res. 2023;231:116159. doi: 10.1016/j.envres.2023.116159. [DOI] [PubMed] [Google Scholar]
- Subha C, Kavitha S, Abisheka S, Tamilarasan K, Arulazhagan P, Jeyakumar R. Bioelectricity generation and effect studies from organic rich chocolaterie wastewater using continuous upflow anaerobic microbial fuel cell. Fuel. 2019;251:224–232. doi: 10.1016/j.fuel.2019.04.052. [DOI] [Google Scholar]
- Tsekouras GJ, Deligianni PM, Kanellos FD, Kontargyri V, Kontaxis PA, Manousakis NM, Elias CN. Microbial fuel cell for wastewater treatment as power plant in smart grids: utopia or reality? Front Ener Res. 2022;10:843768. doi: 10.3389/fenrg.2022.843768. [DOI] [Google Scholar]
- Wang F, Gu YJ, O’Brien JP, Yi SM, Yalcin SE, Srikanth V, Shen C, Vu D, Ing NL, Hochbaum AI, Egelman EH, Malvankar NS. Structure of microbial nanowires reveals stacked hemes that transport electrons over micrometers. Cell. 2019;177:361–369.e10. doi: 10.1016/j.cell.2019.03.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu W, Zhang D, Fang P (2020) Advance in Improving the Electrical Performance of Microbial Fuel Cell. IOP Conf. Series: Earth and Environ Sci 555:012004 IOP Publishing 10.1088/1755-1315/555/1/012004
- Yasri N, Roberts EPL, Gunasekaran S. The electrochemical perspective of bioelectrocatalytic activities in microbial electrolysis and microbial fuel cells. Ener Rep. 2019;5:1116–1136. doi: 10.1016/j.egyr.2019.08.007. [DOI] [Google Scholar]
- Ye Y, Ngo HH, Guo W, Chang SW, Nguyen DD, Liu Y, Nghiem LD, Zhang X, Wang J. Effect of organic loading rate on the recovery of nutrients and energy in a dual-chamber microbial fuel cell. Biores Technol. 2019;281:367–373. doi: 10.1016/j.biortech.2019.02.108. [DOI] [PubMed] [Google Scholar]
- Zagklis DP, Arvaniti EC, Papadakis VG, Paraskeva CA. Sustainability analysis and benchmarking of olive mill wastewater treatment methods. J Chem Technol Biotechnol. 2013;88:742–750. doi: 10.1002/jctb.4036. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The accession number obtained from the NCBI (National Center for Biotechnology) for the bacterial strains involved in the research was provided as per Journal guidelines. The data on bacterial strains and accession number is included in Table 2 of the article. With the accession number the readers can obtain the nucleotide sequence data from NCBI website using BLASTN search online software.