Abstract
Adenoviral vectors have been widely used as vaccine candidates or potential vaccine candidates against infectious diseases due to the convenience of genome manipulation, their ability to accommodate large exogenous gene fragments, easy access of obtaining high-titer of virus, and high efficiency of transduction. At the same time, adenoviral vectors have also been used extensively in clinical research for cancer gene therapy and treatment of diseases caused by a single gene defect. However, application of adenovirus also faces a series of challenges such as poor targeting, strong immune response against the vector itself, and they cannot be used repeatedly. It is believed that these problems will be solved gradually with further research and technological development in related fields. Here, we review the construction methods of adenoviral vectors, including “gutless” adenovirus and discuss application of adenoviral vectors as prophylactic vaccines for infectious pathogens and their application prospects as therapeutic vaccines for cancer and other kinds of chronic infectious disease such as human papillomavirus, hepatitis B virus, and hepatitis C virus.
Keywords: MT: Delivery Strategies, adenovirus, construction methods, application
Graphical abstract

Chen and his colleagues review the construction and application of adenoviral vectors, and discuss the progress of adenoviral vectors in basic research and clinical applications. Adenoviral vectors are currently among the most promising viral vectors for gene therapy and prophylactic vaccine development due to their various advantages.
Introduction
Adenovirus (Ads), which were first isolated in 1953,1 are widespread and endemic in nature. Studies have found that Ads have over 100 serotypes and can be divided into two genera: mammalian adenoviruses and avian adenoviruses.2,3 In this review, we focus on the human adenovirus (HAd).
HAd is a kind of double-stranded DNA virus without an envelope and susceptible to all age groups of the population, with a relatively high incidence in children, the elderly, and immunocompromised individuals, who are also at a higher risk of complications from infection.4,5,6 HAd is transmitted mainly by droplet, fecal-oral, and through contact with objects contaminated with the virus. The spread of HAd is particularly rapid in crowded or enclosed areas.7,8,9
HAd is capable of causing symptomatic infections of the upper and lower respiratory tract, gastrointestinal tract, or eyes. Most HAd-infected individuals only exhibit mild symptoms of the common cold, conjunctivitis, and tonsillitis.10,11,12,13,14 Currently, there are no specific drugs available for the treatment of HAd-infected patients, but the vast majority of infected individuals recover naturally; even severe Ad pneumonia does not cause death, except for rare outbreaks of primary pneumonia that occur in infants and immunocompromised populations. In particular, hematopoietic stem cell and solid organ transplant recipients, and people with congenital immunodeficiency or human immunodeficiency virus (HIV) infection are at a higher risk of death from Ad infection than the general population.15,16,17,18,19
It was not until the 1980s that the therapeutic potential of Ad as a gene delivery vector was discovered.20 Ad is the first viral gene delivery vector used in humans.21 Among more than 90 serotypes of HAd, serotype 5 (HAd5) is the most widely used vector.22 In the early 1990s, Ad vectors were used to express the alpha-1 antitrypsin (A1AT) gene in rat hepatocytes and lung tissue.23,24 Subsequently, Ad vectors were rapidly used in clinical trials for the treatment of various monogenic diseases. Numerous clinical trials have confirmed the safety and efficacy of Ad vectors. Since Ad vectors were applied to gene therapy, they have been continuously modified to improve their accommodation capability, transduction efficiency, exogenous gene-expressing duration, and in vivo safety. A wide variety of Ad vectors are currently being used in clinical trials, which demonstrates the promising prospects of Ad as a vaccine vector and gene therapy tool.25,26,27,28
Characteristics of Ad vectors
Ad is a non-enveloped virus with a linear double-stranded DNA genome containing four fragments encoding early gene products (E1-E4) and five fragments encoding late gene products (L1-L5).29,30 E1, E2, and E4 gene products can regulate transcription and translation of late-stage genes, which are essential for viral replication.31 E3 gene products can disrupt the immune response by altering antigen presentation, inhibiting cytokine production, and cellular apoptosis, but are not necessary for viral replication. The L1-L5 proteins encoded by late genes are associated with Ad particle assembly.32,33,34 Each end of the Ad genome is flanked by 100–150 base pair (bp) long inverted terminal repeats (ITRs), the nucleotide sequence immediately adjacent to the left ITR is the packaging signal (Ψ) of Ad, a cis-acting element necessary for Ad replication (Figure 1A).35,36,37
Figure 1.
Schematic of adenovirus genome and particle structure
(A) The length of adenovirus genome is around 35–37 kb. Each end of the Ad genome is flanked by 100–150 bp long inverted terminal repeats (ITRs), the nucleotide sequence immediately adjacent to the left ITR is the packaging signal (Ψ) of Ad. E1-E4 represent four fragments encoding early gene products on the adenovirus genome; L1-L5 represent five late genes on adenovirus genome. (B) The viral capsid of adenovirus has a diameter of 80–100 nm. The viral capsid has a typical icosahedral symmetry, consisting of 252 8–10 nm diameter capsid particles, including 240 hexons and 12 pentons.
To utilize Ad vectors more safely and effectively, researchers have continuously modified Ad vectors through genetic engineering techniques. In the first-generation Ad vector with a deletion of the E1/E3 region in the Ad genome, the deleted region was used as an insertion site for exogenous genes and the capacity is approximately up to 4.5–8 kilobases (kb).38,39 The construction method is simple and it is easy to obtain high titers of virus in helper cell lines.
The second generation of Ad vectors, with a deletion of the E2/E4 regions in addition to deletion of the E1/E3 regions, which has increased the accommodation capacity of the vector to carry exogenous genes, improved transgenic capacity and reduced the likelihood of replication-competent adenovirus (RCA) generation.40,41,42 However, titers of the second generation were significantly lower than those of the first generation, being only 1/1,000 to 1/10 of the latter. Meanwhile, although the host immune response against the vector was diminished, it still existed.43,44,45,46
The third generation of Ad vector removes all or most of the adenoviral genome, retaining only necessary cis-acting elements of ITR and packaging, hence it is also known as a gutless adenovirus vector.47 Due to deletion of most of the adenoviral genome and absence of viral protein expression, the accommodation capacity for the exogenous gene has been increased substantially to 36 kb, while immune response against the vector has been reduced and safety has been significantly enhanced.48,49 The absence of adenoviral protein expression in vivo means the protein of the vector itself will not be recognized by major histocompatibility complex class I molecules and presented to CD8+ T cells,50 thereby significantly reducing stimulation of the immune response and vector toxicity, resulting in prolonged expression of the transgene, which has been shown to last up to 10 years. However, construction of this kind of Ad vector is difficult, and the required DNA length must be supplemented with a stuffer sequence and a foreign transgenic sequence, and a helper virus and helper cell line are required during the packaging process. The subsequent purification process requires removal of helper virus contamination, which increases the difficulty and cost of purification and makes its clinical application difficult.
All three kinds of adenoviral vectors mentioned above are replication deficient and have their own advantages and disadvantages. Ad vectors have the following significant advantages: (1) their genome can be easily edited and manipulated, and can be used as delivery vectors of the exogenous genes; (2) as the first expression system that can express multiple genes in the same cell line or tissue, their genome has high accommodation capacity, up to 8–36 kb exogenous gene fragments can be inserted, and multiple genes can be expressed simultaneously; (3) they can efficiently transduce different types of human and other mammalian cells, including some highly differentiated tissues, such as muscle, heart, lung, and brain tissue, which is not limited by whether the target cells are in the division period, and is an efficient gene transmission and expression vector; (4) they do not integrate into chromosomes in almost all known cells except oocytes, so there is no potential risk of mutagenesis by insertion into the genome; (5) the adenoviral vector itself has an adjuvant effect, inducing strong and effective humoral and cellular immunity; (6) Ads can proliferate in helper cells, making it easy to obtain high titer viruses, and can be industrialized and mass produced using suspension cell culture systems. Based on the numerous advantages mentioned above, adenoviruses have been widely used in various fields such as in vitro gene transduction, in vivo gene therapy, and vaccination. At present, first-generation adenoviral vectors are still the main ones used for gene therapy and vaccine vectors.
On the other hand, adenoviral vectors also have some limitations and shortcomings: (1) Ads can infect a variety of cells and tissues, which results in a lack of targeting of the gene transduction, (2) the immunogenicity of the adenoviral vector itself often induces a strong immune response against itself, which restricts repeated use of the vector, (3) an adenoviral vector needs to encode dozens of proteins for itself, which often results in low expression efficiency of the foreign gene, (4) due to the high affinity of the Ad capsid for receptors such as coagulation factors, adenoviral vectors rapidly accumulate in the liver after immunization and have a non-negligible impact on liver metabolism, resulting in liver damage.
Methods for construction of recombinant Ads
The viral capsid of Ads has a diameter of 80–100 nm and a relative molecular mass of about 175,000 Da. The viral capsid has a typical icosahedral symmetry, consisting of 252 8–10 nm diameter capsid particles, including 240 hexons and 12 pentons (Figure 1B).51 The epitope on the hexon determines the serotype and can stimulate production of neutralizing antibodies (NAs) against Ad vectors in the host.52 Exogenous epitopes can be inserted into hexons, penton bases, fiber proteins, and peptide IX without affecting stability and function of viral structures, and can induce specific immune responses against the inserted epitope.53,54
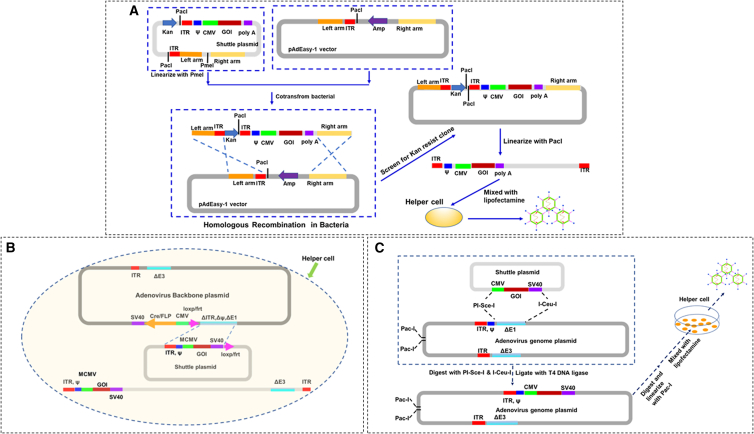
With development of molecular biology and genetic engineering techniques, the packaging technology for recombinant Ads has constantly been improved. In the original recombinant Ad packaging method, the exogenous gene is cloned between two homologous arms on the shuttle vector and co-transfected with the Ad genome into human embryonic kidney 293 (HEK293) cells or other cells supplied with E1 in trans.55 In helper cell lines, the recombination that occurs between the upper homologous arm of the shuttle vector and the nucleotide homologous region on the Ad genome replaces the E1 region with a foreign gene. However, the incidence of recombination is relatively low with this approach; obtaining recombinant Ads using this method is laborious, time consuming, and requires multiple rounds of plaque purification.
In the second method, homologous recombination occurs in Escherichia coli. The commonly used pAdEasy Ad packaging system uses homologous recombination to complete insertion of an exogenous gene into the Ad genome in E. coli, obtaining a circular recombinant Ad genome with the necessary components for replication in bacteria (Figure 2A).56 This method is efficient but more complex and requires three steps of transformation involving the use of two different competent strains of E. coli, including specific E. coli that express RecA recombinase.57 In addition, because recombination occurs in bacteria, small genomic changes are not easily detected; therefore, to obtain high-quality recombinant Ad strain (high viral yield, high target gene expression), multiple recombinant Ad plasmids packaging DNA should be selected and transfected separately in 293 cells to rescue multiple recombinant Ad viruses for screening.
Figure 2.
Packaging system for the first generation of adenoviral vectors
(A) pAd-Easy adenovirus packaging system: homologous recombination to complete the insertion of an exogenous gene into the Ad genome in Escherichia coli, obtaining a circular recombinant Ad genome with the necessary components for replication in bacteria. The adenovirus packaging plasmid was linearized with PacI and then transfected into a helper cell line. (B) The pAd-MaxTM packaging system: co-transfects 293 cells with a shuttle plasmid cloned with an exogenous gene and a packaging plasmid carrying the majority of the Ad genome, and uses the Cre/loxP-based system to achieve recombination and generate the recombinant adenovirus. (C) Direct-cloning strategy to construct the adenovirus vector: clones the adenoviral genome directly into a plasmid vector and then cuts the exogenous gene from the shuttle vector and inserts it into the adenovirus backbone vector. The final plasmid is linearized with PacI and transfected into the helper cell line, which can rescue the recombinant Ad after transfection into the packaging cell line.
The third method was developed by Ng and co-workers and is commercially available as an AdMax packaging system. This method co-transfects 293 cells with a shuttle plasmid cloned with an exogenous gene and a packaging plasmid carrying the majority of the Ad genome, and uses the Cre/loxP-based system to achieve recombination and generate the recombinant Ad (Figure 2B). Frequency of Ad vector rescue by Cre-mediated site-specific recombination is about 30-fold higher than traditional homologous recombination.58,59 This process takes place in 293 cells, thus avoiding recombination in bacteria and greatly simplifies the process of molecular cloning during the construction of recombinant Ad vector. In this system, stronger mCMV promoters are often used to obtain higher expression levels of the exogenous gene.
The fourth approach is to clone the Ad genome directly into a plasmid vector and then insert the exogenous gene into the vector, which can rescue the recombinant Ad after transfection into the packaging cell line (Figure 2C). This approach is technically demanding for molecular cloning, as the Ad genome is large (∼36 kb) and there are few restriction sites available, which makes it very difficult to clone the whole Ad genome into a plasmid vector.60 However, the great advantage of this approach is that it allows for the direct cloning and assembly of the adenoviral genome, completely eliminating potential risks in the original Ad genome and avoiding the risk of uncertainty that may exist in the homologous recombination process, which can affect the clinical application of Ad vectors.
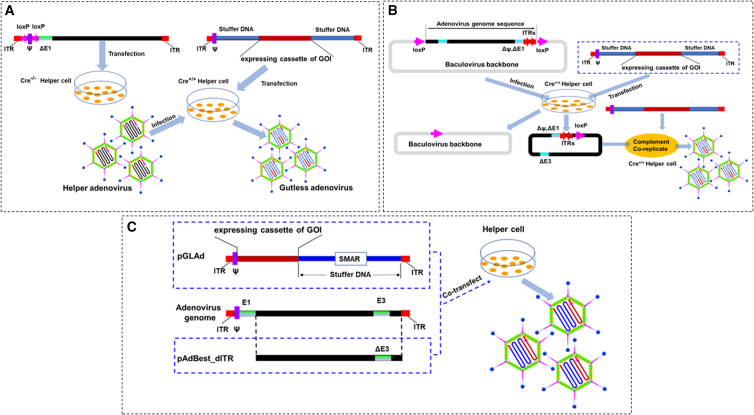
What should be particularly introduced is the packaging method for third-generation Ad vectors. Third-generation Ad vectors, also known as gutless Ad vectors, remove all viral sequences except the ITR and packaging signals. These vectors, also known as "high-capacity" adenoviral vectors (HCAds), can accommodate approximately 36 kb of exogenous genes.49 Parks et al. developed a helper adenovirus-dependent packaging system for production of gutless Ads. In the genome of the helper virus, the packaging signal is flanked by loxP sites; so, in 293 cells stably expressing Cre recombinase (293Cre), the packaging signal is effectively excised, preventing the helper virus genome from being packaged into the viral particle.44 However, the genomic DNA of the helper virus replicates in a normal manner and can therefore be expressed in trans to provide elements required for gutless Ad replication and packaging (Figure 3A). The gutless Ad can be separated from the residual helper virus fraction by cesium chloride buoyant density centrifugation. Cheshenko et al. developed an alternative system for gutless Ad production using a baculovirus gene transduction system, in which genes essential for Ad vector replication and packaging are delivered to production cells via a baculovirus delivery vector.61 They constructed a hybrid baculovirus, Bac-B4, carrying the packaging signal for the Ad genome that can be cleaved and excised by Cre recombinase. The gutless Ad obtained using this method is completely free of helper Ad and is capable of transducing cultured 293 cells (Figure 3B). This gutless Ad packaging method based on baculovirus delivery systems is also a very useful alternative method. Lee and co-workers reported the production of gutless Ad with a helper plasmid in the absence of helper Ad.62,63 With the helper plasmid, they successfully produced large amounts of recombinant gutless Ad. Importantly, the helper plasmid-based system produced only gutless Ads, but not Ads and RCA derived from the helper plasmid (Figure 3C). The generated gutless Ad was effective in delivering the transgene and showed therapeutic potential for Huntington’s chorea (HD) and Duchenne muscular dystrophy. Their data suggest the helper plasmid-based gutless Ad-producing system may become a new platform for gene therapy.
Figure 3.
Gutless packaging system
(A) Helper adenovirus-dependent gutless packaging system: in the genome of the helper virus, the packaging signal is flanked by loxP sites, so that in 293 cells stably expressing Cre recombinase (293Cre) the packaging signal is effectively excised, preventing the helper virus genome from being packaged into the viral particle. The genomic DNA of the helper virus replicates in a normal manner and can therefore be expressed in trans to provide the elements required for gutess Ad replication and packaging. (B) Helper baculovirus-dependent gutless packaging system: a hybrid baculovirus, Bac-B4, carrying the packaging signal for the Ad genome can be cleaved and excised by Cre recombinase. The gutess Ad obtained using this method is completely free of helper Ad and is capable of transducing cultured 293 cell. (C) Helper plasmid-based system produced gutess adenovirus: the adenoviral proteins that are essential for packaging and replication are provided in trans by the helper plasmid, gutess adenovirus, but not wild-type adenovirus and replication-competent adenovirus (RCA) derived from the helper plasmid.
Distribution of receptors and construction of novel Ads for receptor negative cells
Receptors on the host cell surface mediate initial contact between the virus and the cell, which in turn triggers a signaling pathway for the virus to enter the cell. Ads can transduce cells efficiently because their receptors are widely distributed on various cell types and there is a very high affinity between Ads and their receptors. In fact, Ads enter the cell using a variety of co-receptors and attachment factors of the host cell. Co-receptors and adhesion factors help Ads adhere to cells and facilitate their entry and replication.64,65,66 Other kinds of host factors, such as intracytoplasmic Ca2+ ion channel proteins and intranuclear somatic maturation factors or intracellular transport factors, such as actin and microtubule filaments, can also enhance infection (e.g., by increasing receptor expression levels on the surface of the host cell), although these kinds of factors do not directly bind to viral particles.
Coxsackievirus belongs to the enterovirus family and is made up of a single strand of ribonucleic acid (RNA). Ads belong to adenoviridae, which is a kind of double-stranded DNA virus. However, Ads and coxsackieviruses share the same receptor, the coxsackie-adenovirus receptor (CAR), to enter cells. CAR is a kind of transmembrane glycoprotein that belongs to the immunoglobulin superfamily. A variety of Ads, including HAd5, recognize CAR that is expressed on the surface of many types of cells, including hepatocytes, epithelial cells, and cardiomyocytes as its receptors.67 Published results showed that, on the cellular membrane of some malignant tumor cells, expression of CAR was downregulated to varying degrees. In addition to this, the higher the degree of malignancy of the tumor, the lower the expression of CAR.
CAR exists in the tight junctions and adherent junctions of polarized epithelial cells. It interacts with the fiber knob domain of all HAds except for those in subgenus B, and mediates virus attachment.68 Some chimpanzee adenoviruses (ChAds) also can bind to CAR to initiate the cell entry process. Ads of subgenus B mainly use CD46 and desmoglein 2 as receptors, and can also bind to CD80 or CD86, some immune molecules expressed on the surface of dendritic cells and mature B cells.69,70,71 Subgenus C can also use acetyl heparin sulfate, major histocompatibility complex class Iα2, etc., as adhesion receptors.72,73,74,75 Serotypes 8, 19, 26, and 37 human adenoviruses belong to subgenus D and recognize sialic acid receptors.76,77,78,79 Integrins are secondary receptors on the surface of HAdV-invading cells, such as integrins αvβ3 and αvβ5, which bind to the RGD motif of the penton and mediate internalization of the Ads.80,81,82,83,84
Next, take HAd5 as an example to review the process of Ads entering cells. At the initial stage of infection, the fiber protein on the surface of the Ad virion binds to the CAR and then, through the endocytosis of clathrin, the virion is internalized into the cytoplasm, and there endosomal acidification causes conformational changes of the capsid protein.85 After conformational changes, Ad virions travel through microtubules to the nuclear pore complex, where the viral genome is released into the nucleus to initiate transcription and translation (Figure 4).
Figure 4.
Cell attachment and entry of human serotype 5 adenovirus
The fiber protein on the surface of the Ad virion binds to the CAR, and then, through the endocytosis of clathrin, the virion is internalized into the cytoplasm, and there endosomal acidification causes conformational changes of the capsid protein. After conformational changes, adenovirus virions travel through microtubules to the nuclear pore complex, where the viral genome is released into the nucleus to initiate transcription and translation.
Unlike HAd5, which utilizes the CAR as the main receptor, the fiber knob proteins of several other serotypes of Ads (including HAd3, HAd11, HAd26, HAd35, HAd37, and HAd52) can bind to CD46, desmoglein 2, GD1aglycan, and/or polysialic acid receptors on the cell surface for cell entry (Figure 5).76,86,87,88,89,90,91,92 Based on this feature of Ads, the fiber knob coding genes from Ads utilizing different receptors can be used to replace the corresponding genes in HAd5, constructing a novel chimeric Ad vector that can efficiently deliver genes to CAR-negative target cells. For example, the HAd5/3 chimeric Ad was constructed by replacing the fiber knob domain of the HAd5 with the corresponding domain of the HAd3.93,94 Since the receptor of HAd3 has a higher expression on the surface of tumor cells, HAd5/3 chimeric vectors can enter the tumor cells through the Ad3 receptor.94,95 DNX-2401 is a selectively replicated chimeric adenoviral vector. The ACDCRGDCFCG peptide was inserted into the fiber protein of the Ad, which causes the modified fiber protein to be able to bind to integrin αvβ3 and αvβ5 and mediate Ad invasion into cells without the need for CAR receptors.96 The vector has been clinically tested in patients with recurrent malignant glioma. The RGDK amino acid sequence in the fiber protein of Ad vector VCN-01 was replaced by the heparin sulfate glycosaminoglycan binding site (KKTK), which causes the chimeric Ad to enter the cell through the glycosaminoglycan receptor on the cell surface.97,98
Figure 5.
Different knob receptors on the cellular membrane for adenovirus entry
Human serotype 5 adenovirus utilizes the coxsackievirus and adenovirus receptor (CAR) as the main receptor, the fiber knob proteins of several other serotypes of adenovirus (including HAd3, HAd11, HAd26, HAd35, HAd37, and HAd52) can bind to CD46, desmoglein 2 (DSG-2), polysialic acid (polySia), and/or GD1aglycan (GD1a) on the cell surface for cell entry.
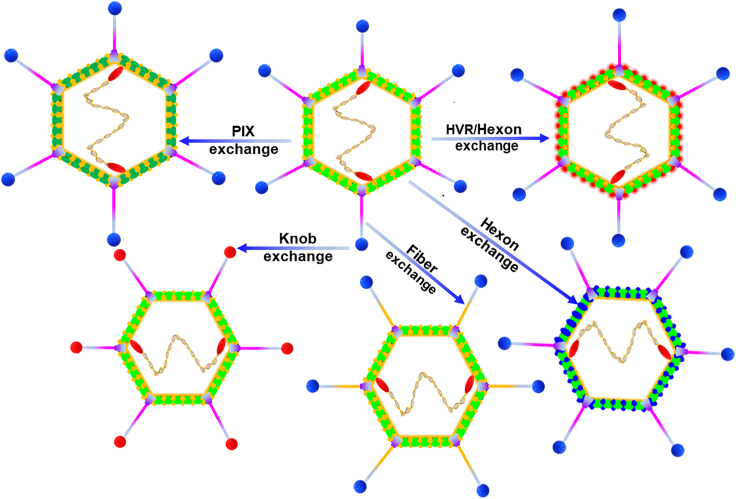
Transduction efficiency of HAd5 mainly depends on the expression level of CAR on the surface of target cells. However, in some cell types, such as primary tumor cells, stem cells, dendritic cells, etc., expression level of CAR on the surface is low.99,100,101 Therefore, the transduction efficiency of HAd5 on such cells is low, thus limiting its application. HAd35 recognizes CD46, which is highly expressed on human CD3+ T cells as its receptor. Replacing the fiber gene of HAd5 with that of HAd35 to generate a chimeric Ad vector, Ad5/F35, enhances the transduction efficiency of the Ad to T cells, resulting in broader application.102 However, modification of the fiber protein cannot completely avoid the immune response against the hexon and penton. In fact, in addition to modification of the fiber and fiber knob, other proteins on the surface of the Ad virion such as hexon and PIX can also be modified to construct chimeric Ads (Figure 6).
Figure 6.
Construction of chimeric adenovirus
The modification of fiber and fiber knob; other proteins on the surface of the Ad virion such as HVR/Hexon and PIX can also be modified to construct a chimeric Ad.
It should be pointed out that the role of receptors in cell entry of Ads remains unclear.64,66,103 For example, skeletal muscle cells are the main target cells for the adenoviral vector-based vaccines when inoculated using the intramuscular pathway. However, the expression level of CAR on the skeletal muscle is significantly lower than in other tissues.104,105 Published results showed that expression levels of receptors in target cells had no significant impact on the immune effect of the vaccine. CD46, another important receptor for HAd35, is only expressed in the testes of wild-type mice, but in CD46 transgenic mice this protein can be widely expressed in various tissues throughout the body. Nevertheless, immunizing wild-type mice and CD46 transgenic mice with Ad35-based vaccine intramuscularly caused no significant difference in the expression level of transgene or the protective effect of the vaccine between the two groups.106,107 In addition, CAR is mainly expressed on the basal lateral surface and the Ad has difficulty accessing CAR by intranasal or intratracheal immunization.108,109,110 In fact, adenoviral vector-based vaccines administered by nasal spray has achieved very good results.111,112,113,114 To further improve gene delivery efficiency and extend application of adenoviral vectors, it is of great significance to study the underlying mechanism of receptors in the process of adenovirus entry into cells.
Strategies to circumvent pre-existing antibodies against Ad vectors
The biggest obstacle limiting repeated use of the adenoviral vector is the strong immunogenicity of the vector itself, which often induces the body to produce NAs against the Ad. NAs against multiple serotypes of Ads are commonly found in the population, resulting in rapid clearance of the adenoviral vector by the host immune system, affecting transduction efficiency and expression intensity and duration of the exogenous gene.115,116 How to circumvent the inhibitory effect of NAs against the adenoviral vector is currently one of the main limiting factors for its clinical application. Although increasing the immune dose of Ad can overcome antagonism of pre-existing antibodies in vivo to some extent, increasing the immune dose will also increase related side effects and economic costs, and is therefore unlikely to be a realistic choice for adenoviral vector-based vaccine application.
Since the first discovery and successful isolation of Ad in the 1950s, more than 100 serotypes have been isolated to date, of which 52 different serotypes have been identified in humans and can be divided into 6 subgroups (A, B, C, D, E, and F).117 Human adenovirus serotype 2 (HAd2) and HAd5 are the two Ads most commonly used as gene therapy tools and vaccine delivery vectors, both belonging to subgroup C, and sharing 95% homology in DNA sequence. In fact, HAd2 and 5 infections are very common in the population, with some studies showing the pediatric population was 59.6% and 43.3% seropositive for rAd2 and rAd5, respectively. Some reports demonstrated that about 80% of the population has NAs against HAd5.118 Development of new serotypes of Ads as vectors for vaccines and gene therapy is becoming increasingly important, especially to develop rare serotypes of Ads with low prevalence in the population and Ads from other species like chimpanzees.
Tatsis and co-workers from the Wistar Institute at the University of Pennsylvania constructed multiple replication-deficient ChAd vectors with E1 deficiency including ChAd68, ChAd7, and ChAd6.119,120,121 These ChAd vectors can effectively replicate in HEK293 cells, indicating that ChAd can utilize the E1 gene of HAd5 to complete the replication cycle. Another advantage of using E1 motifs from different hosts to provide trans complementarity is that it can reduce the probability of producing RCA, as the nucleotide fragments on both sides of the E1 gene from different host sources have very low homology. Zhou and co-workers validated the gene delivery ability of these ChAds to different cells and tissues, as well as their ability to induce humoral and cellular immunity in vivo.119,122,123,124,125,126,127 Their results indicated that adenoviral vectors derived from chimpanzees have the same transport capacity and immunogenicity as HAd5.
In fact, the use of ChAd as a vaccine vector has some distinct advantages.128 First, ChAds can be cultured in human cell lines, such as HEK293 cells, which have been approved for production of multiple vaccines. In addition, ChAds have low seroprevalence in the population because they rarely circulate in humans.129,130 Its amino acid sequence in the hypervariable region of hexons, which contains most of the neutralizing antigenic determinants, differs from common serotype HAd, so ChAd can efficiently evade the NA against HAd. According to some published reports, NAs against ChAds are rare in European and US populations, with prevalence ranging from 0% to 4%, compared with up to 20% in people in sub-Saharan African countries.131 This difference may be attributed to differences in lifestyle and level of interaction with wildlife. However, 20% seroprevalence is still significantly lower than of HAd5. There is a general lack of NAs against ChAds in the population, and its powerful gene delivery ability and the ability to induce humoral and cellular immunity, make ChAd vectors another powerful weapon for humans to fight against emerging infectious diseases. In addition to HAds and ChAds, a number of other mammalian Ads and avian Ads, including porcine Ads, bovine Ads, sheep Ads, avian Ads, and canine Ads, have also been used as tools for gene therapy and vaccine research.132,133,134,135,136,137
Studies have shown that the strong immune response of the host against the adenoviral vector itself will reduce the feasibility of using the same vector for booster immunization.116 However, there is little cross-immune response between Ads of different serotypes, but they share similar genomic structures and functional proteins, which provides more possibilities and options for development of chimeric adenoviral vectors using Ads of different serotypes.54,138
In the past few decades, the vast majority of research on adenoviral vector-based vaccines or gene therapy have been based on HAd5, as its pathogenesis and replication mechanisms have been well understood. To expand the application range of adenoviral vectors, fiber protein from other serotypes (such as HAd3, HAd11, HAd35, HAd37, HAd40, etc.) are commonly used for chimerism with HAd5 fiber proteins to improve transduction efficiency and circumvent pre-existing immune responses against HAd5.139,140
Modification of the viral capsid protein structure is currently the most common targeted modification strategy.141 However, these modification strategies are not perfect. For example, modification of the fiber protein will significantly change the Ad’s tissue tropism, which may bring unknown safety risks; modification of the hexon often affects the Ad’s assembly, making it difficult for packaging of the recombinant Ad, or significantly reducing the Ad’s proliferation ability.53 Therefore, there is still great room for improvement in targeted modification of Ad.
To circumvent the inhibitory effect of pre-existing NAs on adenoviral vectors (mainly against HAd5), it is also an intentional exploration to modify the surface of the viral particle’s surface by physical or chemical means. Polymers such as polyethylene glycol (PEG) and multivalent hydrophilic polymers were used to mask certain antigenic epitopes and reduce the immune response, which can significantly prolong Ad-mediated gene expression.142,143,144 Encapsulation of HAd5 with PEG can block binding of anti-HAd5 antibodies to viral particles and avoid the inhibitory effect of anti-Ad5 NA. Covalent attachment of polymers to Ad capsid proteins can reduce the inhibitory effect of NAs on Ad transduction efficiency. Ad vectors containing poly-lysine peptide-modified fibers can reduce hepatotoxicity by decreasing IL-6 production.145,146 In addition, liposome-encapsulated HAd5 vectors can also escape the inhibitory effect of NAs, and both cationic and anionic liposomes can be used for encapsulation of HAd vectors.147,148 In fact, since Ad virions have a negative charge on their surface, they can be easily encapsulated by cationic liposomes simply by mixing Ad carriers and cationic liposomes together. Zhong et al. successfully formulated HAd5 with anionic liposomes. Their results showed that anionic liposome-formulated HAd5 could escape from the effect of NAs, while improving its transduction efficiency in CAR-deficient cells.149 In addition, recent studies have shown that Ad particles can be encapsulated by extracellular vesicles, such as exosomes, but it is unclear whether extracellular vesicles containing HAds can bypass anti-HAd5 NAs.150
Primary immunization with a low dose of adenoviral vector-based vaccine in the immunization protocol and boost with a high dose of Ad can induce a stronger specific immune response. In addition to this, intranasal immunization is significantly better than intramuscular injection in preventing respiratory infections, which means the immunization method also can affect the effectiveness of the adenoviral vector-based vaccine. In the study of the COVID-19 vaccine, it was found that intranasal administration significantly reduced side effects of the vaccine while achieving better protection.151 Studies have shown that nebulized vaccines with intranasal immunization are more effective in stimulating mucosal immunity and reducing adverse reactions and side effects of the vaccine.152,153
Adenoviral vector-based vaccine for infectious pathogens
Due to their characteristics, Ads have become the preferred vaccine vectors against infectious disease and been considered as the most promising vaccine vectors against infectious diseases. Currently, adenoviral vector-based vaccines have been developed to cover a wide range of infectious agents from malaria to HIV-1. As of March 2023, a total of 739 clinical trials of vaccines and gene therapy have used Ads as vectors (Table 1), and this number is still on the rise. Among them, adenoviral vector-based vaccines against SARS-CoV-2 and Ebola have been successfully used in clinical practice.
Table 1.
Adenoviral vector vaccines against pathogens
| Serotype of Ad | Pathogens | Status | NCT no. |
|---|---|---|---|
| Ad5 | SARS-CoV-2 | phase I | NCT05526183 |
| Ad5 and ChAd68 | SARS-CoV-2 | phase 1 | NCT05094609 |
| AdCLD (Chimeric 5/35) | SARS-CoV-2 | active | NCT04666012 |
| ChAdOx1 | SARS-CoV-2 | phase 2 | NCT05049226 |
| ChAdV68 | SARS-CoV-2 | phase 1 | NCT04776317 |
| Ad 4 | HIV | phase 1 | NCT02771730 |
| Ad5 or Ad35 | HIV | phase 1 | NCT00472719 |
| Ad26 | HIV | phase 1 | NCT00618605 |
| AdC6 and AdC7 | HIV | phase 1 | NCT05182125 |
| Ad3 | Ebola | phase 1 | NCT04041570 |
| Ad5 | Ebola | phase I/II | NCT04840992 |
| Ad26 | Ebola | phase 3 | NCT02509494 |
| Ad26 | hemorrhagic fever, Ebola | phase 2 | NCT02564523 |
| Unknown | influenza (H1N1) | phase 2 | NCT02918006 |
| Ad4 | H5N1 | phase 1 | NCT01443936 |
| Unknown | norovirus | phase 1 | NCT03125473 |
| Unknown | norovirus | phase 1 | NCT02868073 |
| ChAd3 | Marburg virus disease | phase 2 | NCT05817422 |
| ChAd63 | malaria Plasmodium vivax | phase Ia | NCT01816113 |
| Ad35 | malaria | phase 2 | NCT01366534 |
| Ad5 | malaria | phase 2 | NCT00870987 |
| ChAdOx2 | Crohn disease, Mycobacterium avium subspecies paratuberculosis | phase 1 | NCT03027193 |
| ChAdOx1 | MERS | phase 1 | NCT03399578 |
| Ad26 | RSV | phase 2 | NCT03303625 |
| Ad35 | Plasmodium falciparum | phase 1 | NCT00371189 |
| ChAd155- | hepatitis B | phase 2 | NCT03866187 |
| ChAd3 | hepatitis C | phase 1 | NCT03688061 |
| AdCh3 and Ad6 | hepatitis C | phase 1 | NCT01094873 |
| ChAdOx1 | chikungunya fever | phase 1 | NCT03590392 |
| Ad4 | anthrax infection | phase 1 | NCT01979406 |
In vaccine research, Ad is often used as delivery vectors for antigens, where protective antigen genes are inserted into the Ad genome, and a recombinant Ad is generated as a potential candidate vaccine. After immunization, the recombinant Ad will activate the body’s immune system, resulting in massive production of protective antibodies. However, the main factors limiting the prospect of adenoviral vector-based vaccine applications are the widespread presence of pre-existing NAs against the vector itself in the population, as well as robust immune-related adverse events induced by adenoviral proteins, all of which must be taken into account and solved in clinical application.
The majority of adenoviral vectors currently used as prophylactic vaccines against infectious diseases are still first-generation adenoviral vectors, i.e., those deleted in the E1 and E3 regions. Gene encoding the antigenic protein that induces protective antibodies is often inserted into the E1 region.
Adenoviral vector-based Ebola vaccine
Ebola is a deadly disease caused by the Ebola virus, formerly known as Ebola hemorrhagic fever. After Ebola virus infection in the human body, it can quickly spread to tissues and organs throughout the body, destroying connective tissue, inducing a severe inflammatory cytokine storm and causing apoptosis of the immune system, and eventually infected people will die due to multiple organ failure and extensive hemorrhage.154,155
The lyophilized EBO vaccine jointly developed by the Institute of Bioengineering of the Academy of Military Medical Sciences (CAS) and CanSino Biologics (CanSino) uses the AdMax packaging system to construct Ad5 vectors with E1 and E3 deletion, and uses HEK293SF-3F6 cell lines for mass production. Antibody titers decreased gradually 6 months after primary immunization, and antibody titers increased rapidly on day 28 after booster immunization with the same recombinant adenovirus. Another research team from China has developed a novel Ebola virus vaccine based on human serotype 2 adenovirus (HAd2).
The EBO vaccine developed by Janssen Pharmaceuticals is based on HAd26 and the vaccinia Ankara strain is used for booster immunization. In the phase I clinical study, NAs were stimulated and generated in 87%–100% of volunteers after receiving the second dose of vaccine. The ChAd3 vector-based EBO vaccine (ChAd3-EBO-Z), developed in collaboration with the US National Institute of Allergy and Infectious Diseases and GSK, has also shown good immunogenicity and safety.
Adenoviral vector-based COVID-19 vaccine
In the global war against COVID-19, messenger RNA (mRNA) vaccine saved millions of lives, while the adenoviral vector vaccine also played an extremely important role.156 Adenoviral vector vaccine and mRNA vaccine have their own advantages and disadvantages. However, adenoviral vector vaccine has advantages on storage conditions. Liquid formulations of adenoviral vector vaccine could maintain its effectiveness for more than 1 year at 2°C–8°C and its lyophilized formulation can be stored longer under the above conditions, and even can be stored for several days at 30°C, without any significant loss of vitality.157 Different from the adenoviral vector vaccine, the mRNA vaccines BNT162b2 and mRNA-1273 need to be kept at −80°C and −20°C, respectively.156 This storage condition is a challenge for the mRNA vaccine. Strict dependence on low-temperature storage and transportation of the mRNA vaccine can be attributed to instability of the LNP system. Safety concerns for mRNA vaccines, especial for LNP particles, remain.158 After many years of clinical research, adenoviral vector vaccines have proved their safety, while long-term safety of mRNA vaccine is not well understood. Meanwhile, an increasing number of studies have shown that immunization with mRNA vaccines increases the risk of vascular closure.159 In the long run, adenoviral vector vaccines are also better than mRNA vaccines in induced immune duration. BNT162b2 and mRNA-1273 vaccines can induce excellent short-term NA response and protective efficacy. However, high initial serum NA titer induced by the mRNA vaccine declines in 3–6 months, with a further decline in 8 months. Different from BNT162b2 and mRNA-1273, the initial NA titer induced by Ad26.Cov-2.S was low, but the NA response and clinical effect were quite durable, lasting at least 8 months.156 In addition, compared with the mature industrial-scale automated production of adenoviruses, production of mRNA vaccines has not been continuous and automated, which greatly limits the production capacity and increases the cost of mRNA vaccines. Therefore, although adenoviral vector vaccines also have some side effects and risks, they are still the first-line vaccines in many developing countries.
Since the outbreak of COVID-19, four kinds of adenoviral vector-based COVID-19 vaccines have been approved and widely applied among the human population. Among the four adenoviral vector-based vaccines, Ad5-nCoV was developed by the Academy of Military Sciences/CanSino in China,160 AZD1222 was developed by Oxford University/AstraZeneca in the UK,161 Ad26.CoV-2.S was developed by Johnson & Johnson in the US,162 and Sputnik V was developed by the Gamaleya Research Institute in Russia.163 All four vaccines used replication-deficient Ads to express full-length spike (S) protein, but the antigen design and vaccination dose were different. The codon of the S gene in AZD1222 has been optimized, and a tPA signal peptide has been added at the N-terminal. Its vector ChAdOx1 is a replication-deficient version of ChAdY25, and the pre-existing immunity to this vector is very low among the human population. The antigen design of Ad5 nCoV is similar to AZD1222. The optimized S gene with the tPA signal peptide is cloned into the Ad5 vector that lacks E1 and E3, and the recombinant Ad is constructed using the AdMax system. Sputnik V, also known as Gam-COVID-Vac, expresses the full-length S protein using Ad26 (first dose) and Ad5 vectors (second dose), respectively, which can bypass the inhibitory effect of anti-vector NA induced by the first-dose vaccination.
In addition to the EBO and COVID-19 vaccines, which have been approved for clinical application in the human population, there are a number of adenoviral vector-based vaccines against infectious diseases that are currently in various stages of clinical trials.
Adenoviral vector-based influenza vaccine
Compared with current influenza vaccine technology, the Ad carrier platform has its own advantages and can serve as a necessary supplement to current influenza vaccine technology. In response to the global influenza pandemic, the adenoviral vector-based vaccine platform can produce a large quantity of adenoviral vector-based vaccines at a low cost in a short period of time, which is crucial to suppress virus transmission and reduce infection-related morbidity and mortality at the early stage of the pandemic. Several human and non-human Ad vector systems have already been used to develop seasonal and universal influenza vaccines. Clinical studies have shown that human avian influenza vaccines based on HAd4 and HAd5 expressing H5N1 HA have good immunogenicity and safety.164 Adenoviral vector-based vaccine ChAdOx1 NP + M1, expressing NP and matrix protein 1 (M1), was constructed based on a ChAd Y25 isolate, which had protective effect against different subtypes of influenza viruses.165 Ad vector-based influenza vaccines that co-expressed the main influenza virus antigens (e.g., HA, NP, and M2) have been tested in clinical trials.
We constructed a novel adenovirus vector ChAdC7 utilizing the direct clone strategy and inserted the H5N1 hemagglutinin (HA) into the E1 region on its genome. Our previous study showed that a ChAd vector carrying influenza virus HA gene could be a promising vaccine candidate of multiple HA subtypes influenza virus.166
Oncolytic Ad
Oncolytic viruses (OVs) only selectively replicate within cancer cells without affecting normal cells.167,168,169,170,171 In recent years, OV therapy has been extensively studied as an emerging anti-tumor biological therapy, which can induce the body’s innate and adaptive immune response to attack tumor cells.168,172,173 As a member of the of OV family, oncolytic Ads are in a leading position in the field of research and development both in the laboratory and in clinical practice.174 Unlike replication-deficient Ad vectors that require a helper cell line that provides replication-essential gene E1 in trans to complete the replication cycle, oncolytic Ad, also known as selectively replicating recombinant Ad, which can complete the replication cycle only in cancer cells is harmless to normal cells. Oncolytic Ad replicates inside tumor cells and then releases large quantities of offspring viruses, which go on to infect surrounding tumor cells, inhibiting tumor growth, and eventually eliminating tumors. Construction of selective or conditional replication recombinant Ad mainly involves placing the E1A or E1B genes, which are essential for Ad replication, under the control of tumor tissue-specific promoters, so they are only expressed in specific tumor tissues.85
Tumor-specific promoters can be divided into two categories, one is broad-spectrum tumor-specific promoters: these promoters initiate transcription and expression of downstream genes in all tumor cells, such as survivin promoters, cyclooxygenase (COX-2) promoters, hTERT promoters, and telomerase promoter, among which the most studied is the telomerase promoter, because the vast majority of malignant tumor cells are telomerase positive; the other type of promoter is active and functional only in a specific tumor cell, such as prostate-specific antigen promoter, alpha-fetoprotein liver cancer promoter, carcinoembryonic antigen epithelial cancer promoter, and breast cancer cell-specific promoters.175,176 To further improve the effect of oncolytic Ads, anti-tumor genes and immunomodulatory genes are often inserted in its genome to enhance the ability of the OV to kill tumor cells while improving the body’s anti-tumor immune response.
Hypoxia is a characteristic feature of solid tumors that occurs during tumor development and growth and can lead to cell-cycle arrest.177 Taking advantage of this property of tumor cells, Hernandez-Alcoceba et al. designed a tumor-lysing Ad in which expression of the E1A gene is controlled by a promoter containing a hypoxia response element, and the modified oncolytic adenovirus can fully replicate under hypoxic conditions to achieve the purpose of oncolysis.178
The use of OVs to express certain cytokines with immunomodulatory function is beneficial to improve the body’s anti-tumor immune response.179,180,181,182,183,184 At present, the most studied is granulocyte macrophage colony-stimulating factor (GM-CSF). Oncolytic Ads express GM-CSF to help infiltrate DC precursor cells such as monocytes in tumors to differentiate into DC, and oncolytic Ads release a large number of tumor-associated antigens after dissolving tumor cells to provide DC with abundant tumor antigens,185,186,187,188,189,190,191,192,193,194 which stimulate the body’s cytotoxic T lymphocyte response specific to tumor antigens. In addition to GM-CSF, immune regulator genes, such as IL-12, IL-18, CD40L, 4-1BBL, and B7 are expressed by oncolytic adenovirus alone or jointly, which can also enhance the ability of anti-tumor immune response of the body to a certain extent (Table 2).167,169,172,173,179,180,182,187,195,196
Table 2.
Oncolytic adenovirus armed with immunomodulation
| Immunomodulatory molecules | Mechanism | Cancer type | References |
|---|---|---|---|
| IL-2 | stimulates NK and T cells | melanoma, renal cancer, prostate cancer, glioblastoma, other solid tumors | O'Malley et al.227; Yao et al.228; Toloza et al.229; Mulders et al.227,228,229,230 |
| IL-12 | regulates the immune response and the differentiation of T cells | melanoma, head and neck squamous cell carcinoma | Bortolanza et al.231,232,233,234; He et al.232; Nasu et al.233; Sangro et al.231,232,233,234 |
| IL-15 | activates NK and T cells | glioblastoma | Isvoranu et al.235; Morris et al.236 |
| IL-18 | promotes the development of Th1 and memory CD8 T cells | malignant melanoma | Yang et al.237; Zheng et al.238; Zheng et al.237,238,239 |
| TNF-α | stimulates the T cell compartment in tumor microenvironment | pancreatic ductal, adenocarcinoma | Sato-Dahlman et al.240; Cervera-Carrascon et al.241; Cervera-Carrascon et al.240,241,242 |
| IFN-α/β | inhibits tumor growth directly and indirectly by acting upon tumor and immune cells | hepatocellular and pancreatic carcinomas | Huang et al.243; Borden et al.244; Dinney et al.243,244,245 |
| GM-CSF | promotes differentiation, maturation, expansion, and enhances the function of DCs and macrophages | melanoma, thymic carcinoma, lung esophageal cancer | Lv et al.194; Sato-Dahlman et al.240; Tian et al.246; Wang et al.247 |
| CXCL 10 and 12 | promotes T cell activation | advanced solid tumors | Li et al.248; O'Bryan et al.249 |
| IL-24 | induces cancer cell apoptosis | lung cancer, breast cancer | Ramesh et al.128; Mao et al.250 |
| RANTES | enhances tumor infiltration and maturation of APCs in vivo | hepatocellular carcinoma | Ang et al.251; Li et al.252 |
| Bispecific T cell engager | T cell activation, upregulation of proinflammatory cytokines | prostate cancer | Li et al.252; Freedman et al.253; Huang et al.252,253,254 |
Application of adenoviral vectors for gene editing
Clustered, regularly interspaced, short palindromic repeats (CRISPR)-related protein 9 (Cas9), and guiding RNA (gRNA) gene editing technology, also called CRISPR-Cas9, has opened up new avenues for treatment of a wide range of genetic defects, and several preclinical studies and clinical results have demonstrated its powerful therapeutic effects.197,198 However, how to safely and effectively deliver genome editing components is still a major challenge for in vivo genome editing. Adenoviral vector is considered to be a promising delivery vehicle for in vivo gene editing. Palmer et al. developed a single helper-dependent Ad (HDAd), which is used to deliver all the components of CRISPR-Cas9 for efficient gene editing in transduced cells. This all-in-one HDAd provides an important platform for achieving and expanding CRISPR-Cas9 utility, particularly for difficult-to-transfect cells and in vivo applications.199 Palmer and co-workers have shown that HDAds can efficiently deliver donor DNA for homology-directed repair via spontaneous homologous recombination.200,201
Human papillomavirus (HPV) causes malignant epithelial cancers, including cervical cancer, non-melanoma skin cancer, and head and neck cancer. They drive development of tumors through expression of oncoproteins E6 and E7.202 Ehrke-Schulz and co-workers inserted the complete CRISPR-Cas9-gRNA genome-editing module into a HCAd. After the cells are transduced by this HCAd, HPV18-E6 or HPV16-E6 genes can be specifically knocked out, causing a decrease in HPV-positive viability, while HPV-negative normal cells are not affected.203,204 This indicates that the HCAd can be used for HPV-specific cancer gene therapy when combined with HPV-specific CRISPR-Cas9-gRNA.
Cystic fibrosis (CF) is caused by mutations in the CF transmembrane conduction regulatory factor gene.205 Despite the success of CRISPR-Cas9 genome editing in vitro, there are still challenges when using CRISPR-Cas9 in vivo to treat CF lung disease. HD Ad has the advantages of large capacity and low immunogenicity, coupled with the versatility of the CRISPR-Cas9 system, delivering CRISPR-Cas9 together with HD Ad to the airway for lung gene therapy and shows great potential.206
More than 240 million people worldwide are chronically infected with hepatitis B virus (HBV), leading to chronic hepatitis, cirrhosis, and hepatocellular carcinoma.207 Genome editing using CRISPR-Cas9 could offer new therapeutic approaches for chronic HBV infection as it can directly disrupt the HBV genome. Kato et al. constructed an Ad carrying eight multiplex gRNA of CRISPER-Cas9 targeting the HBV X gene. With the treatment of this Ad, the HBV X gene integrated into the chromosomes of HepG2 cells can be cleaved and destroyed multiple times. The Ad containing eight multiple gRNA units not only ensures that the target gene is knocked out multiple times, but also solves the problem of off-target effects and escape mutants in genome editing therapy.208
Application of Ads to RNAi therapy
RNA interference (RNAi) is a phenomenon of silencing gene expression through specific degradation of homologous mRNA sequences, which exists in almost all eukaryotic cells.209 Due to its high specificity and efficiency, RNAi has been widely used in basic research on gene function and human diseases, as well as antiviral research. In recent years, Ad-mediated small hairpin RNA (shRNA) has been used as a potential method for antiviral research on dengue fever virus, porcine circovirus type 2, and foot-and-mouth disease virus, as well as other kinds viruses.210
Motegi et al. developed an effective Ad vector-mediated RNAi system, in which the Ad vector carries four shRNA expression cassettes (Ad multi-shRNA vectors), which can enhance the silencing effect to the same gene by carrying four identical shRNA sequences, or effectively silence multiple target genes simultaneously, by carrying four different shRNA sequences.211 Ad multi-shRNA vectors not only have potential practicality in basic research but also in clinical gene therapy.212
In mammals, microRNAs (miRNAs) are involved in regulation of many genes. More than 1,000 human miRNAs have been identified and are estimated to regulate expression of at least 30% of human genes.213 RNAi has already been widely used as a tool for gene knockdown in basic research or clinical medicine. Ad vectors are often used to deliver shRNA or miRNA expression cassettes into target cells in vitro. In addition, because Ad vectors have a relatively large packaging capacity, they are often used to transcribe or co-express protein and shRNAs or miRNA in the same expression cassette.214,215,216,217
Our previous study suggested that artificial miRNAs targeting the inside conserved regions of influenza A virus delivered by ChAd68 could provide cross-protection against multiple subtypes of IAV challenging in ICR mice.189,218 We also found that delivering the artificial miRNA of the HA gene on the H5N1 avian influenza virus (AIV) by ChAd68 could also provide 40%–70% immediate protection against a lethal H5N1 AIV challenge in mice.219
Ad vectors are often used to deliver shRNA or miRNA expression cassettes into target cells in vitro. By co-expressing miRNAs targeting p21, the anticancer effect of Ad expressing p53 can be enhanced.214 Another beneficial exploration is to combine oncolytic Ads with RNAi expression boxes. Gürlevik et al. inserted multiple miRNAs targeting essential genes for Ad replication into the oncolytic Ad genome and controlled by the p53 promoter. The replication of Ad would be inhibited in normal cells expressing p53, while its replication would not be restricted in tumor cells with p53 dysfunction.220
Hepatitis C virus (HCV) is a hepatophilic human virus, and its infection causes chronic liver inflammation and fibrosis, which can finally lead to hepatocellular carcinoma.221 Currently, 170 million people worldwide are infected with HCV. Sakurai and co-workers developed an adenoviral vector expressing tough decoy RNA (TuD-122a) targeting miR-122a to inhibit HCV replication in hepatocytes, and their results suggested that adenoviral vector-mediated expression of TuD-122a would be a promising tool for the treatment of HCV infection.222,223
Conclusions
The outbreak of new infectious diseases such as SARS-CoV-2 and mpox has accelerated the research pace of new vaccines.224,225 The adenoviral vector-based COVID-19 vaccine has been approved and clinically applied worldwide, and shows great potential and the promising prospect of the adenoviral vector platform in dealing with emerging infectious diseases.226 In addition, recombinant Ad has also been widely used in clinical research as a delivery tool for gene therapy or been modified as an oncolytic Ad.47,174 Although there are some obstacles to application of adenoviral vectors, such as how to circumvent antagonism of the NA against the Ad vector, and how to improve targeting of adenoviral vectors, it is believed that with in-depth understanding of the biological characteristics of Ad, researchers can find ways to solve these problems, further improve immunogenicity, therapeutic effects, and safety of adenoviral vectors, and reduce their side effects.
Acknowledgments
This work was supported by special funding for basic research of Ab&B Bio-Tech CO., LTD. JS, Taizhou, Jiangsu, China. We would like to give thanks for the support from Ministry of Science and Technology of the People's Republic of China.
Author contributions
Z.C. and Y.A. conceptualized the outline of this manuscript. H.Z. and H.W. wrote and revised the manuscript. All authors made substantial, direct, and intellectual contributions to this article and approved it for publication.
Declaration of interests
The authors declare no competing interests.
References
- 1.Syyam A., Nawaz A., Ijaz A., Sajjad U., Fazil A., Irfan S., Muzaffar A., Shahid M., Idrees M., Malik K., Afzal S. Adenovirus vector system: construction, history and therapeutic applications. Biotechniques. 2022;73:297–305. doi: 10.2144/btn-2022-0051. [DOI] [PubMed] [Google Scholar]
- 2.Harrach B., Tarján Z.L., Benkő M. Adenoviruses across the animal kingdom: a walk in the zoo. FEBS Lett. 2019;593:3660–3673. doi: 10.1002/1873-3468.13687. [DOI] [PubMed] [Google Scholar]
- 3.Zhuang Q., Wang S., Zhang F., Zhao C., Chen Q., Zhao R., Guo P., Ju L., Li J., Hou G., et al. Molecular epidemiology analysis of fowl adenovirus detected from apparently healthy birds in eastern China. BMC Vet. Res. 2023;19:5. doi: 10.1186/s12917-022-03545-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Radke J.R., Cook J.L. Human adenovirus lung disease: outbreaks, models of immune-response-driven acute lung injury and pandemic potential. Curr. Opin. Infect. Dis. 2023;36:164–170. doi: 10.1097/QCO.0000000000000919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Xie L., Zhang B., Zhou J., Huang H., Zeng S., Liu Q., Xie Z., Gao H., Duan Z., Zhong L. Human adenovirus load in respiratory tract secretions are predictors for disease severity in children with human adenovirus pneumonia. Virol. J. 2018;15:123. doi: 10.1186/s12985-018-1037-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zou L., Zhou J., Li H., Wu J., Mo Y., Chen Q., Fang L., Wu D., Wu J., Ke C. Human adenovirus infection in children with acute respiratory tract disease in Guangzhou, China. APMIS: acta pathologica, microbiologica, et immunologica Scandinavica. 2012;120:683–688. doi: 10.1111/j.1600-0463.2012.02890.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cheng J., Qi X., Chen D., Xu X., Wang G., Dai Y., Cui D., Chen Q., Fan P., Ni L., et al. Epidemiology and transmission characteristics of human adenovirus type 7 caused acute respiratory disease outbreak in military trainees in East China. Am. J. Transl. Res. 2016;8:2331–2342. [PMC free article] [PubMed] [Google Scholar]
- 8.Jing S., Zhang J., Cao M., Liu M., Yan Y., Zhao S., Cao N., Ou J., Ma K., Cai X., et al. Household Transmission of Human Adenovirus Type 55 in Case of Fatal Acute Respiratory Disease. Emerg. Infect. Dis. 2019;25:1756–1758. doi: 10.3201/eid2509.181937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Paielli D.L., Wing M.S., Rogulski K.R., Gilbert J.D., Kolozsvary A., Kim J.H., Hughes J., Schnell M., Thompson T., Freytag S.O. Evaluation of the biodistribution, persistence, toxicity, and potential of germ-line transmission of a replication-competent human adenovirus following intraprostatic administration in the mouse. Mol. Ther. 2000;1:263–274. doi: 10.1006/mthe.2000.0037. [DOI] [PubMed] [Google Scholar]
- 10.Barnadas C., Schmidt D.J., Fischer T.K., Fonager J. Molecular epidemiology of human adenovirus infections in Denmark, 2011-2016. J. Clin. Virol. 2018;104:16–22. doi: 10.1016/j.jcv.2018.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Heo J.Y., Noh J.Y., Jeong H.W., Choe K.W., Song J.Y., Kim W.J., Cheong H.J. Molecular Epidemiology of Human Adenovirus-Associated Febrile Respiratory Illness in Soldiers, South Korea(1) Emerg. Infect. Dis. 2018;24:1221–1227. doi: 10.3201/eid2407.171222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lynch J.P., 3rd, Kajon A.E. Adenovirus: Epidemiology, Global Spread of Novel Types, and Approach to Treatment. Semin. Respir. Crit. Care Med. 2021;42:800–821. doi: 10.1055/s-0041-1733802. [DOI] [PubMed] [Google Scholar]
- 13.Pscheidt V.M., Gregianini T.S., Martins L.G., Veiga A.B.G.d. Epidemiology of human adenovirus associated with respiratory infection in southern Brazil. Rev. Med. Virol. 2021;31 doi: 10.1002/rmv.2189. [DOI] [PubMed] [Google Scholar]
- 14.Singh M.P., Ram J., Kumar A., Rungta T., Gupta A., Khurana J., Ratho R.K. Molecular epidemiology of circulating human adenovirus types in acute conjunctivitis cases in Chandigarh, North India. Indian J. Med. Microbiol. 2018;36:113–115. doi: 10.4103/ijmm.IJMM_17_258. [DOI] [PubMed] [Google Scholar]
- 15.Kolawole O.M., Oladosu T.O., Abdulkarim A.A., Okoh A.I. Prevalence of adenovirus respiratory tract and hiv co-infections in patients attending the University of Ilorin, teaching hospital, Ilorin, Nigeria. BMC Res. Notes. 2014;7:870. doi: 10.1186/1756-0500-7-870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Inamoto Y., Takeda W., Hirakawa T., Sakaguchi H., Nakano N., Uchida N., Doki N., Ikegame K., Katayama Y., Sawa M., et al. Adenovirus disease after hematopoietic cell transplantation: A Japanese transplant registry analysis. Am. J. Hematol. 2022;97:1568–1579. doi: 10.1002/ajh.26723. [DOI] [PubMed] [Google Scholar]
- 17.Lum E.L., Zuckerman J., Gaynor P., Bunnapradist S. Adenovirus in a Kidney Transplant Recipient. Kidney Med. 2023;5 doi: 10.1016/j.xkme.2023.100605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Michaels M.G., Green M., Wald E.R., Starzl T.E. Adenovirus infection in pediatric liver transplant recipients. J. Infect. Dis. 1992;165:170–174. doi: 10.1093/infdis/165.1.170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yagisawa T., Takahashi K., Yamaguchi Y., Teraoka S., Horita S., Toma H., Agishi T., Ota K. Adenovirus induced nephropathy in kidney transplant recipients. Transplant. Proc. 1989;21:2097–2099. [PubMed] [Google Scholar]
- 20.Douglas J.T. Adenovirus-mediated gene delivery: an overview. Methods Mol. Biol. 2004;246:3–14. doi: 10.1385/1-59259-650-9:3. [DOI] [PubMed] [Google Scholar]
- 21.Crystal R.G. Adenovirus: the first effective in vivo gene delivery vector. Hum. Gene Ther. 2014;25:3–11. doi: 10.1089/hum.2013.2527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Alonso-Padilla J., Papp T., Kaján G.L., Benkő M., Havenga M., Lemckert A., Harrach B., Baker A.H. Development of Novel Adenoviral Vectors to Overcome Challenges Observed With HAdV-5-based Constructs. Mol. Ther. 2016;24:6–16. doi: 10.1038/mt.2015.194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gilardi P., Courtney M., Pavirani A., Perricaudet M. Expression of human alpha 1-antitrypsin using a recombinant adenovirus vector. FEBS Lett. 1990;267:60–62. doi: 10.1016/0014-5793(90)80287-s. [DOI] [PubMed] [Google Scholar]
- 24.Gupta A.K., Sarin G.S., Mathur M.D., Ghosh B. Alpha 1-antitrypsin and serum albumin in tear fluids in acute adenovirus conjunctivitis. Br. J. Ophthalmol. 1988;72:390–393. doi: 10.1136/bjo.72.5.390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dummer R., Eichmüller S., Gellrich S., Assaf C., Dreno B., Schiller M., Dereure O., Baudard M., Bagot M., Khammari A., et al. Phase II clinical trial of intratumoral application of TG1042 (adenovirus-interferon-gamma) in patients with advanced cutaneous T-cell lymphomas and multilesional cutaneous B-cell lymphomas. Mol. Ther. 2010;18:1244–1247. doi: 10.1038/mt.2010.52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lin X., Huang H., Li S., Li H., Li Y., Cao Y., Zhang D., Xia Y., Guo Y., Huang W., Jiang W. A phase I clinical trial of an adenovirus-mediated endostatin gene (E10A) in patients with solid tumors. Cancer Biol. Ther. 2007;6:648–653. doi: 10.4161/cbt.6.5.4004. [DOI] [PubMed] [Google Scholar]
- 27.Makower D., Rozenblit A., Kaufman H., Edelman M., Lane M.E., Zwiebel J., Haynes H., Wadler S. Phase II clinical trial of intralesional administration of the oncolytic adenovirus ONYX-015 in patients with hepatobiliary tumors with correlative p53 studies. Clin. Cancer Res. 2003;9:693–702. [PubMed] [Google Scholar]
- 28.Xia Z.J., Chang J.H., Zhang L., Jiang W.Q., Guan Z.Z., Liu J.W., Zhang Y., Hu X.H., Wu G.H., Wang H.Q., et al. [Phase III randomized clinical trial of intratumoral injection of E1B gene-deleted adenovirus (H101) combined with cisplatin-based chemotherapy in treating squamous cell cancer of head and neck or esophagus] Ai zheng = Aizheng = Chinese journal of cancer. 2004;23:1666–1670. [PubMed] [Google Scholar]
- 29.Hall K., Blair Zajdel M.E., Blair G.E. Unity and diversity in the human adenoviruses: exploiting alternative entry pathways for gene therapy. Biochem. J. 2010;431:321–336. doi: 10.1042/BJ20100766. [DOI] [PubMed] [Google Scholar]
- 30.Kulanayake S., Tikoo S.K. Adenovirus Core Proteins: Structure and Function. Viruses. 2021;13 doi: 10.3390/v13030388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Täuber B., Dobner T. Molecular regulation and biological function of adenovirus early genes: the E4 ORFs. Gene. 2001;278:1–23. doi: 10.1016/s0378-1119(01)00722-3. [DOI] [PubMed] [Google Scholar]
- 32.Lichtenstein D.L., Toth K., Doronin K., Tollefson A.E., Wold W.S.M. Functions and mechanisms of action of the adenovirus E3 proteins. Int. Rev. Immunol. 2004;23:75–111. doi: 10.1080/08830180490265556. [DOI] [PubMed] [Google Scholar]
- 33.Farley D.C., Brown J.L., Leppard K.N. Activation of the early-late switch in adenovirus type 5 major late transcription unit expression by L4 gene products. J. Virol. 2004;78:1782–1791. doi: 10.1128/jvi.78.4.1782-1791.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hasson T.B., Soloway P.D., Ornelles D.A., Doerfler W., Shenk T. Adenovirus L1 52- and 55-kilodalton proteins are required for assembly of virions. J. Virol. 1989;63:3612–3621. doi: 10.1128/JVI.63.9.3612-3621.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tatsis N., Ertl H.C.J. Adenoviruses as vaccine vectors. Mol. Ther. 2004;10:616–629. doi: 10.1016/j.ymthe.2004.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hatfield L., Hearing P. Redundant elements in the adenovirus type 5 inverted terminal repeat promote bidirectional transcription in vitro and are important for virus growth in vivo. Virology. 1991;184:265–276. doi: 10.1016/0042-6822(91)90843-z. [DOI] [PubMed] [Google Scholar]
- 37.Ahi Y.S., Mittal S.K. Components of Adenovirus Genome Packaging. Front. Microbiol. 2016;7:1503. doi: 10.3389/fmicb.2016.01503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jiang B., Qian K., Du L., Luttrell I., Chitaley K., Dichek D.A. Helper-dependent adenovirus is superior to first-generation adenovirus for expressing transgenes in atherosclerosis-prone arteries. Arterioscler. Thromb. Vasc. Biol. 2011;31:1317–1325. doi: 10.1161/ATVBAHA.111.225516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lamartina S., Cimino M., Roscilli G., Dammassa E., Lazzaro D., Rota R., Ciliberto G., Toniatti C. Helper-dependent adenovirus for the gene therapy of proliferative retinopathies: stable gene transfer, regulated gene expression and therapeutic efficacy. J. Gene Med. 2007;9:862–874. doi: 10.1002/jgm.1083. [DOI] [PubMed] [Google Scholar]
- 40.Gao G.P., Yang Y., Wilson J.M. Biology of adenovirus vectors with E1 and E4 deletions for liver-directed gene therapy. J. Virol. 1996;70:8934–8943. doi: 10.1128/JVI.70.12.8934-8943.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ramalingam R., Rafii S., Worgall S., Hackett N.R., Crystal R.G. Induction of endogenous genes following infection of human endothelial cells with an E1(-) E4(+) adenovirus gene transfer vector. J. Virol. 1999;73:10183–10190. doi: 10.1128/JVI.73.12.10183-10190.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yeh P., Dedieu J.F., Orsini C., Vigne E., Denefle P., Perricaudet M. Efficient dual transcomplementation of adenovirus E1 and E4 regions from a 293-derived cell line expressing a minimal E4 functional unit. J. Virol. 1996;70:559–565. doi: 10.1128/JVI.70.1.559-565.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Cregan S.P., MacLaurin J., Gendron T.F., Callaghan S.M., Park D.S., Parks R.J., Graham F.L., Morley P., Slack R.S. Helper-dependent adenovirus vectors: their use as a gene delivery system to neurons. Gene Ther. 2000;7:1200–1209. doi: 10.1038/sj.gt.3301208. [DOI] [PubMed] [Google Scholar]
- 44.Parks R.J., Chen L., Anton M., Sankar U., Rudnicki M.A., Graham F.L. A helper-dependent adenovirus vector system: removal of helper virus by Cre-mediated excision of the viral packaging signal. Proc. Natl. Acad. Sci. USA. 1996;93:13565–13570. doi: 10.1073/pnas.93.24.13565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Parks R.J., Graham F.L. A helper-dependent system for adenovirus vector production helps define a lower limit for efficient DNA packaging. J. Virol. 1997;71:3293–3298. doi: 10.1128/JVI.71.4.3293-3298.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zhou H., Zhao T., Pastore L., Nageh M., Zheng W., Rao X.M., Beaudet A.L. A Cre-expressing cell line and an E1/E2a double-deleted virus for preparation of helper-dependent adenovirus vector. Mol. Ther. 2001;3:613–622. doi: 10.1006/mthe.2001.0288. [DOI] [PubMed] [Google Scholar]
- 47.Chang J. Adenovirus Vectors: Excellent Tools for Vaccine Development. Immune Network. 2021;21:e6. doi: 10.4110/in.2021.21.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Alba R., Bosch A., Chillon M. Gutless adenovirus: last-generation adenovirus for gene therapy. Gene Ther. 2005;12:S18–S27. doi: 10.1038/sj.gt.3302612. [DOI] [PubMed] [Google Scholar]
- 49.Kochanek S., Schiedner G., Volpers C. High-capacity 'gutless' adenoviral vectors. Curr. Opin. Mol. Ther. 2001;3:454–463. [PubMed] [Google Scholar]
- 50.Kron M.W., Engler T., Schmidt E., Schirmbeck R., Kochanek S., Kreppel F. High-capacity adenoviral vectors circumvent the limitations of DeltaE1 and DeltaE1/DeltaE3 adenovirus vectors to induce multispecific transgene product-directed CD8 T-cell responses. J. Gene Med. 2011;13:648–657. doi: 10.1002/jgm.1629. [DOI] [PubMed] [Google Scholar]
- 51.Reddy V.S., Natchiar S.K., Stewart P.L., Nemerow G.R. Crystal structure of human adenovirus at 3.5 A resolution. Science. 2010;329:1071–1075. doi: 10.1126/science.1187292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Harmon A.W., Moitra R., Xu Z., Byrnes A.P. Hexons from adenovirus serotypes 5 and 48 differentially protect adenovirus vectors from neutralization by mouse and human serum. PLoS One. 2018;13 doi: 10.1371/journal.pone.0192353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Capasso C., Garofalo M., Hirvinen M., Cerullo V. The evolution of adenoviral vectors through genetic and chemical surface modifications. Viruses. 2014;6:832–855. doi: 10.3390/v6020832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Miralles M., Garcia M., Tejero M., Bosch A., Chillón M. Production of chimeric adenovirus. Methods Mol. Biol. 2014;1089:231–243. doi: 10.1007/978-1-62703-679-5_16. [DOI] [PubMed] [Google Scholar]
- 55.Danthinne X., Imperiale M.J. Production of first generation adenovirus vectors: a review. Gene Ther. 2000;7:1707–1714. doi: 10.1038/sj.gt.3301301. [DOI] [PubMed] [Google Scholar]
- 56.He T.C., Zhou S., da Costa L.T., Yu J., Kinzler K.W., Vogelstein B. A simplified system for generating recombinant adenoviruses. Proc. Natl. Acad. Sci. USA. 1998;95:2509–2514. doi: 10.1073/pnas.95.5.2509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Luo J., Deng Z.L., Luo X., Tang N., Song W.X., Chen J., Sharff K.A., Luu H.H., Haydon R.C., Kinzler K.W., et al. A protocol for rapid generation of recombinant adenoviruses using the AdEasy system. Nat. Protoc. 2007;2:1236–1247. doi: 10.1038/nprot.2007.135. [DOI] [PubMed] [Google Scholar]
- 58.Aoki K., Barker C., Danthinne X., Imperiale M.J., Nabel G.J. Efficient generation of recombinant adenoviral vectors by Cre-lox recombination in vitro. Mol. Med. 1999;5:224–231. [PMC free article] [PubMed] [Google Scholar]
- 59.Hardy S., Kitamura M., Harris-Stansil T., Dai Y., Phipps M.L. Construction of adenovirus vectors through Cre-lox recombination. J. Virol. 1997;71:1842–1849. doi: 10.1128/JVI.71.3.1842-1849.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zhou D., Zhou X., Bian A., Li H., Chen H., Small J.C., Li Y., Giles-Davis W., Xiang Z., Ertl H.C.J. An efficient method of directly cloning chimpanzee adenovirus as a vaccine vector. Nat. Protoc. 2010;5:1775–1785. doi: 10.1038/nprot.2010.134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cheshenko N., Krougliak N., Eisensmith R.C., Krougliak V.A. A novel system for the production of fully deleted adenovirus vectors that does not require helper adenovirus. Gene Ther. 2001;8:846–854. doi: 10.1038/sj.gt.3301459. [DOI] [PubMed] [Google Scholar]
- 62.Lee D., Liu J., Junn H.J., Lee E.J., Jeong K.S., Seol D.W. No more helper adenovirus: production of gutless adenovirus (GLAd) free of adenovirus and replication-competent adenovirus (RCA) contaminants. Exp. Mol. Med. 2019;51:1–18. doi: 10.1038/s12276-019-0334-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Liu J., Seol D.W. Helper virus-free gutless adenovirus (HF-GLAd): a new platform for gene therapy. BMB Rep. 2020;53:565–575. doi: 10.5483/BMBRep.2020.53.11.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Arnberg N. Adenovirus receptors: implications for tropism, treatment and targeting. Rev. Med. Virol. 2009;19:165–178. doi: 10.1002/rmv.612. [DOI] [PubMed] [Google Scholar]
- 65.Sharma A., Li X., Bangari D.S., Mittal S.K. Adenovirus receptors and their implications in gene delivery. Virus Res. 2009;143:184–194. doi: 10.1016/j.virusres.2009.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zhang Y., Bergelson J.M. Adenovirus receptors. J. Virol. 2005;79:12125–12131. doi: 10.1128/JVI.79.19.12125-12131.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ramamoorthy S., Garg S., Mishra B., Radotra B.D., Saikia U.N. Coxsackievirus and Adenovirus Receptor (CAR) Expression in Autopsy Tissues: Organ-Specific Patterns and Clinical Significance. Cureus. 2023;15 doi: 10.7759/cureus.37138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Hemmi S., Geertsen R., Mezzacasa A., Peter I., Dummer R. The presence of human coxsackievirus and adenovirus receptor is associated with efficient adenovirus-mediated transgene expression in human melanoma cell cultures. Hum. Gene Ther. 1998;9:2363–2373. doi: 10.1089/hum.1998.9.16-2363. [DOI] [PubMed] [Google Scholar]
- 69.Gaggar A., Shayakhmetov D.M., Lieber A. CD46 is a cellular receptor for group B adenoviruses. Nat. Med. 2003;9:1408–1412. doi: 10.1038/nm952. [DOI] [PubMed] [Google Scholar]
- 70.Marttila M., Persson D., Gustafsson D., Liszewski M.K., Atkinson J.P., Wadell G., Arnberg N. CD46 is a cellular receptor for all species B adenoviruses except types 3 and 7. J. Virol. 2005;79:14429–14436. doi: 10.1128/JVI.79.22.14429-14436.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Short J.J., Vasu C., Holterman M.J., Curiel D.T., Pereboev A. Members of adenovirus species B utilize CD80 and CD86 as cellular attachment receptors. Virus Res. 2006;122:144–153. doi: 10.1016/j.virusres.2006.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ramos-Martínez I.E., Ramos-Martínez E., Segura-Velázquez R.Á., Saavedra-Montañez M., Cervantes-Torres J.B., Cerbón M., Papy-Garcia D., Zenteno E., Sánchez-Betancourt J.I. Heparan Sulfate and Sialic Acid in Viral Attachment: Two Sides of the Same Coin? Int. J. Mol. Sci. 2022;23 doi: 10.3390/ijms23179842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Cagno V., Tseligka E.D., Jones S.T., Tapparel C. Heparan Sulfate Proteoglycans and Viral Attachment: True Receptors or Adaptation Bias? Viruses. 2019;11 doi: 10.3390/v11070596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Tuve S., Wang H., Jacobs J.D., Yumul R.C., Smith D.F., Lieber A. Role of cellular heparan sulfate proteoglycans in infection of human adenovirus serotype 3 and 35. PLoS Pathog. 2008;4 doi: 10.1371/journal.ppat.1000189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Beier D.C., Cox J.H., Vining D.R., Cresswell P., Engelhard V.H. Association of human class I MHC alleles with the adenovirus E3/19K protein. J. Immunol. 1994;152:3862–3872. [PubMed] [Google Scholar]
- 76.Arnberg N., Edlund K., Kidd A.H., Wadell G. Adenovirus type 37 uses sialic acid as a cellular receptor. J. Virol. 2000;74:42–48. [PMC free article] [PubMed] [Google Scholar]
- 77.Baker A.T., Mundy R.M., Davies J.A., Rizkallah P.J., Parker A.L. Human adenovirus type 26 uses sialic acid-bearing glycans as a primary cell entry receptor. Sci. Adv. 2019;5:eaax3567. doi: 10.1126/sciadv.aax3567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Boulanger P.A., Houdret N., Scharfman A., Lemay P. The role of surface sialic acid in adenovirus-cell adsorption. J. Gen. Virol. 1972;16:429–434. doi: 10.1099/0022-1317-16-3-429. [DOI] [PubMed] [Google Scholar]
- 79.Rademacher C., Bru T., McBride R., Robison E., Nycholat C.M., Kremer E.J., Paulson J.C. A Siglec-like sialic-acid-binding motif revealed in an adenovirus capsid protein. Glycobiology. 2012;22:1086–1091. doi: 10.1093/glycob/cws073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Bieri M., Hendrickx R., Bauer M., Yu B., Jetzer T., Dreier B., Mittl P.R.E., Sobek J., Plückthun A., Greber U.F., Hemmi S. The RGD-binding integrins alphavbeta6 and alphavbeta8 are receptors for mouse adenovirus-1 and -3 infection. PLoS Pathog. 2021;17 doi: 10.1371/journal.ppat.1010083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Nemerow G.R., Cheresh D.A., Wickham T.J. Adenovirus entry into host cells: a role for alpha(v) integrins. Trends Cell Biol. 1994;4:52–55. doi: 10.1016/0962-8924(94)90010-8. [DOI] [PubMed] [Google Scholar]
- 82.Nemerow G.R., Stewart P.L. Insights into Adenovirus Uncoating from Interactions with Integrins and Mediators of Host Immunity. Viruses. 2016;8 doi: 10.3390/v8120337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Storm R.J., Persson B.D., Skalman L.N., Frängsmyr L., Lindström M., Rankin G., Lundmark R., Domellöf F.P., Arnberg N. Human Adenovirus Type 37 Uses alpha(V)beta(1) and alpha(3)beta(1) Integrins for Infection of Human Corneal Cells. J. Virol. 2017;91 doi: 10.1128/JVI.02019-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Venail F., Wang J., Ruel J., Ballana E., Rebillard G., Eybalin M., Arbones M., Bosch A., Puel J.L. Coxsackie adenovirus receptor and alpha nu beta3/alpha nu beta5 integrins in adenovirus gene transfer of rat cochlea. Gene Ther. 2007;14:30–37. doi: 10.1038/sj.gt.3302826. [DOI] [PubMed] [Google Scholar]
- 85.Gómez-Navarro J., Curiel D.T. Conditionally replicative adenoviral vectors for cancer gene therapy. Lancet Oncol. 2000;1:148–158. doi: 10.1016/s1470-2045(00)00030-9. [DOI] [PubMed] [Google Scholar]
- 86.Lenman A., Liaci A.M., Liu Y., Frängsmyr L., Frank M., Blaum B.S., Chai W., Podgorski I.I., Harrach B., Benkő M., et al. Polysialic acid is a cellular receptor for human adenovirus 52. Proc. Natl. Acad. Sci. USA. 2018;115:E4264–E4273. doi: 10.1073/pnas.1716900115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Arnberg N., Pring-Akerblom P., Wadell G. Adenovirus type 37 uses sialic acid as a cellular receptor on Chang C cells. J. Virol. 2002;76:8834–8841. doi: 10.1128/jvi.76.17.8834-8841.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Everitt E., Rodríguez E. Adenovirus cellular receptor site recirculation of HeLa cells upon receptor-mediated endocytosis is not low pH-dependent. Arch. Virol. 1999;144:787–795. doi: 10.1007/s007050050544. [DOI] [PubMed] [Google Scholar]
- 89.Howitt J., Anderson C.W., Freimuth P. Adenovirus interaction with its cellular receptor CAR. Curr. Top. Microbiol. Immunol. 2003;272:331–364. doi: 10.1007/978-3-662-05597-7_11. [DOI] [PubMed] [Google Scholar]
- 90.Li X., Bangari D.S., Sharma A., Mittal S.K. Bovine adenovirus serotype 3 utilizes sialic acid as a cellular receptor for virus entry. Virology. 2009;392:162–168. doi: 10.1016/j.virol.2009.06.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Segerman A., Atkinson J.P., Marttila M., Dennerquist V., Wadell G., Arnberg N. Adenovirus type 11 uses CD46 as a cellular receptor. J. Virol. 2003;77:9183–9191. doi: 10.1128/jvi.77.17.9183-9191.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Stasiak A.C., Stehle T. Human adenovirus binding to host cell receptors: a structural view. Med. Microbiol. Immunol. 2020;209:325–333. doi: 10.1007/s00430-019-00645-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Murakami M., Ugai H., Belousova N., Pereboev A., Dent P., Fisher P.B., Everts M., Curiel D.T. Chimeric adenoviral vectors incorporating a fiber of human adenovirus 3 efficiently mediate gene transfer into prostate cancer cells. Prostate. 2010;70:362–376. doi: 10.1002/pros.21070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Zafar S., Quixabeira D.C.A., Kudling T.V., Cervera-Carrascon V., Santos J.M., Grönberg-Vähä-Koskela S., Zhao F., Aronen P., Heiniö C., Havunen R., et al. Ad5/3 is able to avoid neutralization by binding to erythrocytes and lymphocytes. Cancer Gene Ther. 2021;28:442–454. doi: 10.1038/s41417-020-00226-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Kuryk L., Møller A.S.W. Chimeric oncolytic Ad5/3 virus replicates and lyses ovarian cancer cells through desmoglein-2 cell entry receptor. J. Med. Virol. 2020;92:1309–1315. doi: 10.1002/jmv.25677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.The Oncolytic Adenovirus DNX-2401 Has Antitumor Activity in Glioblastoma. Cancer Discov. 2018;8:382. doi: 10.1158/2159-8290.CD-RW2018-031. [DOI] [PubMed] [Google Scholar]
- 97.Martínez-Vélez N., Xipell E., Vera B., Acanda de la Rocha A., Zalacain M., Marrodán L., Gonzalez-Huarriz M., Toledo G., Cascallo M., Alemany R., et al. The Oncolytic Adenovirus VCN-01 as Therapeutic Approach Against Pediatric Osteosarcoma. Clin. Cancer Res. 2016;22:2217–2225. doi: 10.1158/1078-0432.CCR-15-1899. [DOI] [PubMed] [Google Scholar]
- 98.Rodríguez-García A., Giménez-Alejandre M., Rojas J.J., Moreno R., Bazan-Peregrino M., Cascalló M., Alemany R. Safety and efficacy of VCN-01, an oncolytic adenovirus combining fiber HSG-binding domain replacement with RGD and hyaluronidase expression. Clin. Cancer Res. 2015;21:1406–1418. doi: 10.1158/1078-0432.CCR-14-2213. [DOI] [PubMed] [Google Scholar]
- 99.Abdolazimi Y., Mojarrad M., Pedram M., Modarressi M.H. Analysis of the expression of coxsackievirus and adenovirus receptor in five colon cancer cell lines. World J. Gastroenterol. 2007;13:6365–6369. doi: 10.3748/wjg.v13.i47.6365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Korn W.M., Macal M., Christian C., Lacher M.D., McMillan A., Rauen K.A., Warren R.S., Ferrell L. Expression of the coxsackievirus- and adenovirus receptor in gastrointestinal cancer correlates with tumor differentiation. Cancer Gene Ther. 2006;13:792–797. doi: 10.1038/sj.cgt.7700947. [DOI] [PubMed] [Google Scholar]
- 101.Adams W.C., Bond E., Havenga M.J.E., Holterman L., Goudsmit J., Karlsson Hedestam G.B., Koup R.A., Loré K. Adenovirus serotype 5 infects human dendritic cells via a coxsackievirus-adenovirus receptor-independent receptor pathway mediated by lactoferrin and DC-SIGN. J. Gen. Virol. 2009;90:1600–1610. doi: 10.1099/vir.0.008342-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Flickinger J.C., Jr., Staudt R.E., Singh J., Carlson R.D., Barton J.R., Baybutt T.R., Rappaport J.A., Zalewski A., Pattison A., Waldman S.A., Snook A.E. Chimeric adenoviral (Ad5.F35) and listeria vector prime-boost immunization is safe and effective for cancer immunotherapy. NPJ Vaccines. 2022;7:61. doi: 10.1038/s41541-022-00483-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Nemerow G.R. Cell receptors involved in adenovirus entry. Virology. 2000;274:1–4. doi: 10.1006/viro.2000.0468. [DOI] [PubMed] [Google Scholar]
- 104.Kimura E., Maeda Y., Arima T., Nishida Y., Yamashita S., Hara A., Uyama E., Mita S., Uchino M. Efficient repetitive gene delivery to skeletal muscle using recombinant adenovirus vector containing the Coxsackievirus and adenovirus receptor cDNA. Gene Ther. 2001;8:20–27. doi: 10.1038/sj.gt.3301359. [DOI] [PubMed] [Google Scholar]
- 105.Feero W.G., Rosenblatt J.D., Huard J., Watkins S.C., Epperly M., Clemens P.R., Kochanek S., Glorioso J.C., Partridge T.A., Hoffman E.P. Viral gene delivery to skeletal muscle: insights on maturation-dependent loss of fiber infectivity for adenovirus and herpes simplex type 1 viral vectors. Hum. Gene Ther. 1997;8:371–380. doi: 10.1089/hum.1997.8.4-371. [DOI] [PubMed] [Google Scholar]
- 106.Adams W.C., Gujer C., McInerney G., Gall J.G.D., Petrovas C., Karlsson Hedestam G.B., Koup R.A., Loré K. Adenovirus type-35 vectors block human CD4+ T-cell activation via CD46 ligation. Proc. Natl. Acad. Sci. USA. 2011;108:7499–7504. doi: 10.1073/pnas.1017146108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Sakurai F., Kawabata K., Koizumi N., Inoue N., Okabe M., Yamaguchi T., Hayakawa T., Mizuguchi H. Adenovirus serotype 35 vector-mediated transduction into human CD46-transgenic mice. Gene Ther. 2006;13:1118–1126. doi: 10.1038/sj.gt.3302749. [DOI] [PubMed] [Google Scholar]
- 108.Persson B.D., Lenman A., Frängsmyr L., Schmid M., Ahlm C., Plückthun A., Jenssen H., Arnberg N. Lactoferrin-Hexon Interactions Mediate CAR-Independent Adenovirus Infection of Human Respiratory Cells. J. Virol. 2020;94 doi: 10.1128/JVI.00542-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Mokhashi N., Choi R.Y., Cicalese S., Eguchi K., Boyer M.J., Cooper H.A., Kimura Y., Akiyama T., Scalia R., Rizzo V., Eguchi S. Transduction Efficiency of Adenovirus Vectors in Endothelial Cells and Vascular Smooth Muscle Cells. J. Cardiovasc. Pharmacol. 2020;75:603–607. doi: 10.1097/FJC.0000000000000821. [DOI] [PubMed] [Google Scholar]
- 110.Hidaka C., Milano E., Leopold P.L., Bergelson J.M., Hackett N.R., Finberg R.W., Wickham T.J., Kovesdi I., Roelvink P., Crystal R.G. CAR-dependent and CAR-independent pathways of adenovirus vector-mediated gene transfer and expression in human fibroblasts. J. Clin. Invest. 1999;103:579–587. doi: 10.1172/JCI5309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Van Kampen K.R., Shi Z., Gao P., Zhang J., Foster K.W., Chen D.T., Marks D., Elmets C.A., Tang D.c.C. Safety and immunogenicity of adenovirus-vectored nasal and epicutaneous influenza vaccines in humans. Vaccine. 2005;23:1029–1036. doi: 10.1016/j.vaccine.2004.07.043. [DOI] [PubMed] [Google Scholar]
- 112.Renteria S.S., Clemens C.C., Croyle M.A. Development of a nasal adenovirus-based vaccine: Effect of concentration and formulation on adenovirus stability and infectious titer during actuation from two delivery devices. Vaccine. 2010;28:2137–2148. doi: 10.1016/j.vaccine.2009.12.025. [DOI] [PubMed] [Google Scholar]
- 113.Kim H.D., Tahara K., Maxwell J.A., Lalonde R., Fukuiwa T., Fujihashi K., Van Kampen K.R., Kong F.K., Tang D.c.C., Fukuchi K.i. Nasal inoculation of an adenovirus vector encoding 11 tandem repeats of Abeta1-6 upregulates IL-10 expression and reduces amyloid load in a Mo/Hu APPswe PS1dE9 mouse model of Alzheimer's disease. J. Gene Med. 2007;9:88–98. doi: 10.1002/jgm.993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Croyle M.A., Patel A., Tran K.N., Gray M., Zhang Y., Strong J.E., Feldmann H., Kobinger G.P. Nasal delivery of an adenovirus-based vaccine bypasses pre-existing immunity to the vaccine carrier and improves the immune response in mice. PLoS One. 2008;3 doi: 10.1371/journal.pone.0003548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Ono R., Nishimae F., Wakida T., Sakurai F., Mizuguchi H. Effects of pre-existing anti-adenovirus antibodies on transgene expression levels and therapeutic efficacies of arming oncolytic adenovirus. Sci. Rep. 2022;12 doi: 10.1038/s41598-022-26030-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Fausther-Bovendo H., Kobinger G.P. Pre-existing immunity against Ad vectors: humoral, cellular, and innate response, what's important? Hum. Vaccin. Immunother. 2014;10:2875–2884. doi: 10.4161/hv.29594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Lukashok S.A., Horwitz M.S. New perspectives in adenoviruses. Curr. Clin. Top. Infect. Dis. 1998;18:286–305. [PubMed] [Google Scholar]
- 118.Yu B., Wang Z., Dong J., Wang C., Gu L., Sun C., Kong W., Yu X. A serological survey of human adenovirus serotype 2 and 5 circulating pediatric populations in Changchun, China, 2011. Virol. J. 2012;9:287. doi: 10.1186/1743-422X-9-287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Tatsis N., Tesema L., Robinson E.R., Giles-Davis W., McCoy K., Gao G.P., Wilson J.M., Ertl H.C.J. Chimpanzee-origin adenovirus vectors as vaccine carriers. Gene Ther. 2006;13:421–429. doi: 10.1038/sj.gt.3302675. [DOI] [PubMed] [Google Scholar]
- 120.Jeyanathan M., Thanthrige-Don N., Afkhami S., Lai R., Damjanovic D., Zganiacz A., Feng X., Yao X.D., Rosenthal K.L., Medina M.F., et al. Novel chimpanzee adenovirus-vectored respiratory mucosal tuberculosis vaccine: overcoming local anti-human adenovirus immunity for potent TB protection. Mucosal Immunol. 2015;8:1373–1387. doi: 10.1038/mi.2015.29. [DOI] [PubMed] [Google Scholar]
- 121.Jenkin D., Ritchie A.J., Aboagye J., Fedosyuk S., Thorley L., Provstgaad-Morys S., Sanders H., Bellamy D., Makinson R., Xiang Z.Q., et al. Safety and immunogenicity of a simian-adenovirus-vectored rabies vaccine: an open-label, non-randomised, dose-escalation, first-in-human, single-centre, phase 1 clinical trial. Lancet. Microbe. 2022;3:e663–e671. doi: 10.1016/S2666-5247(22)00126-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Zhou D., Wu T.L., Lasaro M.O., Latimer B.P., Parzych E.M., Bian A., Li Y., Li H., Erikson J., Xiang Z., Ertl H.C.J. A universal influenza A vaccine based on adenovirus expressing matrix-2 ectodomain and nucleoprotein protects mice from lethal challenge. Mol. Ther. 2010;18:2182–2189. doi: 10.1038/mt.2010.202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Zhou D., Cun A., Li Y., Xiang Z., Ertl H.C.J. A chimpanzee-origin adenovirus vector expressing the rabies virus glycoprotein as an oral vaccine against inhalation infection with rabies virus. Mol. Ther. 2006;14:662–672. doi: 10.1016/j.ymthe.2006.03.027. [DOI] [PubMed] [Google Scholar]
- 124.Xiang Z.Q., Greenberg L., Ertl H.C., Rupprecht C.E. Protection of non-human primates against rabies with an adenovirus recombinant vaccine. Virology. 2014;450–451:243–249. doi: 10.1016/j.virol.2013.12.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Santra S., Sun Y., Korioth-Schmitz B., Fitzgerald J., Charbonneau C., Santos G., Seaman M.S., Ratcliffe S.J., Montefiori D.C., Nabel G.J., et al. Heterologous prime/boost immunizations of rhesus monkeys using chimpanzee adenovirus vectors. Vaccine. 2009;27:5837–5845. doi: 10.1016/j.vaccine.2009.07.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Lasaro M.O., Ertl H.C.J. New insights on adenovirus as vaccine vectors. Mol. Ther. 2009;17:1333–1339. doi: 10.1038/mt.2009.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.de Souza A.P.D., Haut L.H., Silva R., Ferreira S.I.A.C.P., Zanetti C.R., Ertl H.C.J., Pinto A.R. Genital CD8+ T cell response to HIV-1 gag in mice immunized by mucosal routes with a recombinant simian adenovirus. Vaccine. 2007;25:109–116. doi: 10.1016/j.vaccine.2006.07.011. [DOI] [PubMed] [Google Scholar]
- 128.Ramesh R., Ioannides C.G., Roth J.A., Chada S. Adenovirus-mediated interleukin (IL)-24 immunotherapy for cancer. Methods Mol. Biol. 2010;651:241–270. doi: 10.1007/978-1-60761-786-0_14. [DOI] [PubMed] [Google Scholar]
- 129.Dudareva M., Andrews L., Gilbert S.C., Bejon P., Marsh K., Mwacharo J., Kai O., Nicosia A., Hill A.V.S. Prevalence of serum neutralizing antibodies against chimpanzee adenovirus 63 and human adenovirus 5 in Kenyan children, in the context of vaccine vector efficacy. Vaccine. 2009;27:3501–3504. doi: 10.1016/j.vaccine.2009.03.080. [DOI] [PubMed] [Google Scholar]
- 130.Dicks M.D.J., Spencer A.J., Edwards N.J., Wadell G., Bojang K., Gilbert S.C., Hill A.V.S., Cottingham M.G. A novel chimpanzee adenovirus vector with low human seroprevalence: improved systems for vector derivation and comparative immunogenicity. PLoS One. 2012;7 doi: 10.1371/journal.pone.0040385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Xiang Z., Li Y., Cun A., Yang W., Ellenberg S., Switzer W.M., Kalish M.L., Ertl H.C.J. Chimpanzee adenovirus antibodies in humans, sub-Saharan Africa. Emerg. Infect. Dis. 2006;12:1596–1599. doi: 10.3201/eid1210.060078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.McFerran J.B., Smyth J.A. Avian adenoviruses. Rev. Sci. Tech. 2000;19:589–601. [PubMed] [Google Scholar]
- 133.Hess M. Detection and differentiation of avian adenoviruses: a review. Avian Pathol. 2000;29:195–206. doi: 10.1080/03079450050045440. [DOI] [PubMed] [Google Scholar]
- 134.Ren J., Zhang L., Cheng P., Zhang F., Liu Z., Tikoo S.K., Chen R., Du E. Generation of infectious clone of bovine adenovirus type I expressing a visible marker gene. J. Virol. Methods. 2018;261:139–146. doi: 10.1016/j.jviromet.2018.08.020. [DOI] [PubMed] [Google Scholar]
- 135.Mira F., Puleio R., Schirò G., Condorelli L., Di Bella S., Chiaramonte G., Purpari G., Cannella V., Balboni A., Randazzo V., et al. Study on the Canine Adenovirus Type 1 (CAdV-1) Infection in Domestic Dogs in Southern Italy. Pathogens. 2022;11 doi: 10.3390/pathogens11111254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Kim S.M., Kim S.K., Park J.H., Lee K.N., Ko Y.J., Lee H.S., Seo M.G., Shin Y.K., Kim B. A recombinant adenovirus bicistronically expressing porcine interferon-alpha and interferon-gamma enhances antiviral effects against foot-and-mouth disease virus. Antiviral Res. 2014;104:52–58. doi: 10.1016/j.antiviral.2014.01.014. [DOI] [PubMed] [Google Scholar]
- 137.Belák S., Pálfi V., Tury E. Experimental infection of lambs with an adenovirus isolated from sheep and related to bovine adenovirus type 2. I. Virological studies. Acta Vet. Acad. Sci. Hung. 1975;25:91–95. [PubMed] [Google Scholar]
- 138.Crenshaw B.J., Jones L.B., Bell C.R., Kumar S., Matthews Q.L. Perspective on Adenoviruses: Epidemiology, Pathogenicity, and Gene Therapy. Biomedicines. 2019;7 doi: 10.3390/biomedicines7030061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Kim J.W., Kane J.R., Young J.S., Chang A.L., Kanojia D., Morshed R.A., Miska J., Ahmed A.U., Balyasnikova I.V., Han Y., et al. A Genetically Modified Adenoviral Vector with a Phage Display-Derived Peptide Incorporated into Fiber Fibritin Chimera Prolongs Survival in Experimental Glioma. Hum. Gene Ther. 2015;26:635–646. doi: 10.1089/hum.2015.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Gaden F., Franqueville L., Magnusson M.K., Hong S.S., Merten M.D., Lindholm L., Boulanger P. Gene transduction and cell entry pathway of fiber-modified adenovirus type 5 vectors carrying novel endocytic peptide ligands selected on human tracheal glandular cells. J. Virol. 2004;78:7227–7247. doi: 10.1128/JVI.78.13.7227-7247.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Weklak D., Pembaur D., Koukou G., Jonsson F., Hagedorn C., Kreppel F. Genetic and Chemical Capsid Modifications of Adenovirus Vectors to Modulate Vector-Host Interactions. Viruses. 2021;13 doi: 10.3390/v13071300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Hoang Thi T.T., Pilkington E.H., Nguyen D.H., Lee J.S., Park K.D., Truong N.P. The Importance of Poly(ethylene glycol) Alternatives for Overcoming PEG Immunogenicity in Drug Delivery and Bioconjugation. Polymers. 2020;12 doi: 10.3390/polym12020298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Shi L., Zhang J., Zhao M., Tang S., Cheng X., Zhang W., Li W., Liu X., Peng H., Wang Q. Effects of polyethylene glycol on the surface of nanoparticles for targeted drug delivery. Nanoscale. 2021;13:10748–10764. doi: 10.1039/d1nr02065j. [DOI] [PubMed] [Google Scholar]
- 144.Fisher K.D., Stallwood Y., Green N.K., Ulbrich K., Mautner V., Seymour L.W. Polymer-coated adenovirus permits efficient retargeting and evades neutralising antibodies. Gene Ther. 2001;8:341–348. doi: 10.1038/sj.gt.3301389. [DOI] [PubMed] [Google Scholar]
- 145.Zhang W., Guo X., Yin F., Zou X., Hou W., Lu Z. Fiber modifications enable fowl adenovirus 4 vectors to transduce human cells. J. Gene Med. 2021;23:e3368. doi: 10.1002/jgm.3368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Matthews C., Jenkins G., Hilfinger J., Davidson B. Poly-L-lysine improves gene transfer with adenovirus formulated in PLGA microspheres. Gene Ther. 1999;6:1558–1564. doi: 10.1038/sj.gt.3300978. [DOI] [PubMed] [Google Scholar]
- 147.Liu S.H., Smyth-Templeton N., Davis A.R., Davis E.A., Ballian N., Li M., Liu H., Fisher W., Brunicardi F.C. Multiple treatment cycles of liposome-encapsulated adenoviral RIP-TK gene therapy effectively ablate human pancreatic cancer cells in SCID mice. Surgery. 2011;149:484–495. doi: 10.1016/j.surg.2010.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Mendez N., Herrera V., Zhang L., Hedjran F., Feuer R., Blair S.L., Trogler W.C., Reid T.R., Kummel A.C. Encapsulation of adenovirus serotype 5 in anionic lecithin liposomes using a bead-based immunoprecipitation technique enhances transfection efficiency. Biomaterials. 2014;35:9554–9561. doi: 10.1016/j.biomaterials.2014.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Zhong Z., Shi S., Han J., Zhang Z., Sun X. Anionic liposomes increase the efficiency of adenovirus-mediated gene transfer to coxsackie-adenovirus receptor deficient cells. Mol. Pharm. 2010;7:105–115. doi: 10.1021/mp900151k. [DOI] [PubMed] [Google Scholar]
- 150.Zhang Y., Wu J., Zhang H., Wei J., Wu J. Extracellular Vesicles-Mimetic Encapsulation Improves Oncolytic Viro-Immunotherapy in Tumors With Low Coxsackie and Adenovirus Receptor. Front. Bioeng. Biotechnol. 2020;8 doi: 10.3389/fbioe.2020.574007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Rubin R. COVID-19 Vaccine Nasal Spray. JAMA. 2021;326:1138. doi: 10.1001/jama.2021.14996. [DOI] [PubMed] [Google Scholar]
- 152.Deng S., Liang H., Chen P., Li Y., Li Z., Fan S., Wu K., Li X., Chen W., Qin Y., et al. Viral Vector Vaccine Development and Application during the COVID-19 Pandemic. Microorganisms. 2022;10 doi: 10.3390/microorganisms10071450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Xi J., Lei L.R., Zouzas W., April Si X. Nasally inhaled therapeutics and vaccination for COVID-19: Developments and challenges. MedComm. 2021;2:569–586. doi: 10.1002/mco2.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Feldmann H., Geisbert T.W. Ebola haemorrhagic fever. Lancet. 2011;377:849–862. doi: 10.1016/S0140-6736(10)60667-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Eurosurveillance Editorial team Ebola Public Health Emergency of International Concern, Democratic Republic of the Congo, 2019. Euro Surveill. 2019;24 doi: 10.2807/1560-7917.ES.2019.24.29.190718e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Barouch D.H. Covid-19 Vaccines - Immunity, Variants, Boosters. N. Engl. J. Med. 2022;387:1011–1020. doi: 10.1056/NEJMra2206573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Bilbao G., Contreras J.L., Zhang H.G., Pike M.J., Overturf K., Mikheeva G., Krasnykh V., Curiel D.T. Adenovirus-mediated gene expression in vivo is enhanced by the antiapoptotic bcl-2 gene. J. Virol. 1999;73:6992–7000. doi: 10.1128/JVI.73.8.6992-7000.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Ndeupen S., Qin Z., Jacobsen S., Estanbouli H., Bouteau A., Igyártó B.Z. The mRNA-LNP platform's lipid nanoparticle component used in preclinical vaccine studies is highly inflammatory. bioRxiv. 2021 doi: 10.1101/2021.03.04.430128. Preprint at. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Li J.X., Wang Y.H., Bair H., Hsu S.B., Chen C., Wei J.C.C., Lin C.J. Risk assessment of retinal vascular occlusion after COVID-19 vaccination. NPJ vaccines. 2023;8:64. doi: 10.1038/s41541-023-00661-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Zhu F.C., Li Y.H., Guan X.H., Hou L.H., Wang W.J., Li J.X., Wu S.P., Wang B.S., Wang Z., Wang L., et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: a dose-escalation, open-label, non-randomised, first-in-human trial. Lancet. 2020;395:1845–1854. doi: 10.1016/S0140-6736(20)31208-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Maaske J., Sproule S., Falsey A.R., Sobieszczyk M.E., Luetkemeyer A.F., Paulsen G.C., Riddler S.A., Robb M.L., Rolle C.P., Sha B.E., et al. Robust humoral and cellular recall responses to AZD1222 attenuate breakthrough SARS-CoV-2 infection compared to unvaccinated. Front. Immunol. 2022;13 doi: 10.3389/fimmu.2022.1062067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Daly S., Nguyen A.V., Soto J.M., Vance A.Z. Ad26.COV2.S Vaccine-Induced Thrombocytopenia Leading to Dural Sinus Thrombosis and Intracranial Hemorrhage Requiring Hemicraniectomy: A Case Report and Systematic Review. Cureus. 2022;14 doi: 10.7759/cureus.28083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Pascuale C.A., Varese A., Ojeda D.S., Pasinovich M.E., Lopez L., Rossi A.H., Rodriguez P.E., Miglietta E.A., Laboratorio SeVa Group. Mazzitelli I., et al. Immunogenicity and reactogenicity of heterologous immunization against SARS CoV-2 using Sputnik V, ChAdOx1-S, BBIBP-CorV, Ad5-nCoV, and mRNA-1273. Cell Rep. Med. 2022;3 doi: 10.1016/j.xcrm.2022.100706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Xiang K., Ying G., Yan Z., Shanshan Y., Lei Z., Hongjun L., Maosheng S. Progress on adenovirus-vectored universal influenza vaccines. Hum. Vaccin. Immunother. 2015;11:1209–1222. doi: 10.1080/21645515.2015.1016674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Vatzia E., Feest K., McNee A., Manjegowda T., Carr B.V., Paudyal B., Chrun T., Maze E.A., McCarron A., Morris S., et al. Immunization with matrix-nucleoprotein and neuraminidase protects against H3N2 influenza challenge in pH1N1 pre-exposed pigs. NPJ Vaccines. 2023;8:19. doi: 10.1038/s41541-023-00620-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Cheng T., Wang X., Song Y., Tang X., Zhang C., Zhang H., Jin X., Zhou D. Chimpanzee adenovirus vector-based avian influenza vaccine completely protects mice against lethal challenge of H5N1. Vaccine. 2016;34:4875–4883. doi: 10.1016/j.vaccine.2016.08.066. [DOI] [PubMed] [Google Scholar]
- 167.Rahman M.M., McFadden G. Oncolytic Viruses: Newest Frontier for Cancer Immunotherapy. Cancers. 2021;13 doi: 10.3390/cancers13215452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Lin D., Shen Y., Liang T. Oncolytic virotherapy: basic principles, recent advances and future directions. Signal Transduct. Target. Ther. 2023;8:156. doi: 10.1038/s41392-023-01407-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Jhawar S.R., Thandoni A., Bommareddy P.K., Hassan S., Kohlhapp F.J., Goyal S., Schenkel J.M., Silk A.W., Zloza A. Oncolytic Viruses-Natural and Genetically Engineered Cancer Immunotherapies. Front. Oncol. 2017;7:202. doi: 10.3389/fonc.2017.00202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Chiocca E.A. Oncolytic viruses. Nat. Rev. Cancer. 2002;2:938–950. doi: 10.1038/nrc948. [DOI] [PubMed] [Google Scholar]
- 171.Davola M.E., Mossman K.L. Oncolytic viruses: how "lytic" must they be for therapeutic efficacy? OncoImmunology. 2019;8 doi: 10.1080/2162402X.2019.1596006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Innao V., Rizzo V., Allegra A.G., Musolino C., Allegra A. Oncolytic Viruses and Hematological Malignancies: A New Class of Immunotherapy Drugs. Curr. Oncol. 2020;28:159–183. doi: 10.3390/curroncol28010019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Kaufman H.L., Kohlhapp F.J., Zloza A. Oncolytic viruses: a new class of immunotherapy drugs. Nat. Rev. Drug Discov. 2016;15:660. doi: 10.1038/nrd.2016.178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Tripodi L., Vitale M., Cerullo V., Pastore L. Oncolytic Adenoviruses for Cancer Therapy. Int. J. Mol. Sci. 2021;22 doi: 10.3390/ijms22052517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Cafferata E.G., Lopez M.V., Nuñez F.J., Maenza M.A.R., Podhajcer O.L. Application of Synthetic Tumor-Specific Promoters Responsive to the Tumor Microenvironment. Methods Mol. Biol. 2017;1651:213–227. doi: 10.1007/978-1-4939-7223-4_16. [DOI] [PubMed] [Google Scholar]
- 176.Hardcastle J., Kurozumi K., Chiocca E.A., Kaur B. Oncolytic viruses driven by tumor-specific promoters. Curr. Cancer Drug Targets. 2007;7:181–189. doi: 10.2174/156800907780058880. [DOI] [PubMed] [Google Scholar]
- 177.Zhang W., Zhang C., Tian W., Qin J., Chen J., Zhang Q., Fang L., Zheng J. Efficacy of an Oncolytic Adenovirus Driven by a Chimeric Promoter and Armed with Decorin Against Renal Cell Carcinoma. Hum. Gene Ther. 2020;31:651–663. doi: 10.1089/hum.2019.352. [DOI] [PubMed] [Google Scholar]
- 178.Hernandez-Alcoceba R., Pihalja M., Qian D., Clarke M.F. New oncolytic adenoviruses with hypoxia- and estrogen receptor-regulated replication. Hum. Gene Ther. 2002;13:1737–1750. doi: 10.1089/104303402760293574. [DOI] [PubMed] [Google Scholar]
- 179.Carew J.F., Kooby D.A., Halterman M.W., Kim S.H., Federoff H.J., Fong Y. A novel approach to cancer therapy using an oncolytic herpes virus to package amplicons containing cytokine genes. Mol. Ther. 2001;4:250–256. doi: 10.1006/mthe.2001.0448. [DOI] [PubMed] [Google Scholar]
- 180.Dong S., Liu B., Hu S., Guo F., Zhong Y., Cai Q., Zhang S., Qian Y., Wang J., Zhou F. A novel oncolytic virus induces a regional cytokine storm and safely eliminates malignant ascites of colon cancer. Cancer Med. 2022;11:4297–4309. doi: 10.1002/cam4.4772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Ilett E., Kottke T., Donnelly O., Thompson J., Willmon C., Diaz R., Zaidi S., Coffey M., Selby P., Harrington K., et al. Cytokine conditioning enhances systemic delivery and therapy of an oncolytic virus. Mol. Ther. 2014;22:1851–1863. doi: 10.1038/mt.2014.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Inoue T., Byrne T., Inoue M., Tait M.E., Wall P., Wang A., Dermyer M.R., Laklai H., Binder J.J., Lees C., et al. Oncolytic Vaccinia Virus Gene Modification and Cytokine Expression Effects on Tumor Infection, Immune Response, and Killing. Mol. Cancer Ther. 2021;20:1481–1494. doi: 10.1158/1535-7163.MCT-20-0863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Nguyen H.M., Guz-Montgomery K., Saha D. Oncolytic Virus Encoding a Master Pro-Inflammatory Cytokine Interleukin 12 in Cancer Immunotherapy. Cells. 2020;9 doi: 10.3390/cells9020400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Pearl T.M., Markert J.M., Cassady K.A., Ghonime M.G. Oncolytic Virus-Based Cytokine Expression to Improve Immune Activity in Brain and Solid Tumors. Mol. Ther. Oncolytics. 2019;13:14–21. doi: 10.1016/j.omto.2019.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Deng L., Fan J., Guo M., Huang B. Oncolytic and immunologic cancer therapy with GM-CSF-armed vaccinia virus of Tian Tan strain Guang9. Cancer Lett. 2016;372:251–257. doi: 10.1016/j.canlet.2016.01.025. [DOI] [PubMed] [Google Scholar]
- 186.Deng L., Yang X., Fan J., Ding Y., Peng Y., Xu D., Huang B., Hu Z. An Oncolytic Vaccinia Virus Armed with GM-CSF and IL-24 Double Genes for Cancer Targeted Therapy. OncoTargets Ther. 2020;13:3535–3544. doi: 10.2147/OTT.S249816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Kaufman H.L., Kim D.W., DeRaffele G., Mitcham J., Coffin R.S., Kim-Schulze S. Local and distant immunity induced by intralesional vaccination with an oncolytic herpes virus encoding GM-CSF in patients with stage IIIc and IV melanoma. Ann. Surg Oncol. 2010;17:718–730. doi: 10.1245/s10434-009-0809-6. [DOI] [PubMed] [Google Scholar]
- 188.Malhotra S., Kim T., Zager J., Bennett J., Ebright M., D'Angelica M., Fong Y. Use of an oncolytic virus secreting GM-CSF as combined oncolytic and immunotherapy for treatment of colorectal and hepatic adenocarcinomas. Surgery. 2007;141:520–529. doi: 10.1016/j.surg.2006.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Rangsitratkul C., Lawson C., Bernier-Godon F., Niavarani S.R., Boudaud M., Rouleau S., Gladu-Corbin A.O., Surendran A., Ekindi-Ndongo N., Koti M., et al. Intravesical immunotherapy with a GM-CSF armed oncolytic vesicular stomatitis virus improves outcome in bladder cancer. Mol. Ther. Oncolytics. 2022;24:507–521. doi: 10.1016/j.omto.2022.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Bramante S., Kaufmann J.K., Veckman V., Liikanen I., Nettelbeck D.M., Hemminki O., Vassilev L., Cerullo V., Oksanen M., Heiskanen R., et al. Treatment of melanoma with a serotype 5/3 chimeric oncolytic adenovirus coding for GM-CSF: Results in vitro, in rodents and in humans. Int. J. Cancer. 2015;137:1775–1783. doi: 10.1002/ijc.29536. [DOI] [PubMed] [Google Scholar]
- 191.Bramante S., Koski A., Kipar A., Diaconu I., Liikanen I., Hemminki O., Vassilev L., Parviainen S., Cerullo V., Pesonen S.K., et al. Serotype chimeric oncolytic adenovirus coding for GM-CSF for treatment of sarcoma in rodents and humans. Int. J. Cancer. 2014;135:720–730. doi: 10.1002/ijc.28696. [DOI] [PubMed] [Google Scholar]
- 192.Choi K.J., Kim J.H., Lee Y.S., Kim J., Suh B.S., Kim H., Cho S., Sohn J.H., Kim G.E., Yun C.O. Concurrent delivery of GM-CSF and B7-1 using an oncolytic adenovirus elicits potent antitumor effect. Gene Ther. 2006;13:1010–1020. doi: 10.1038/sj.gt.3302759. [DOI] [PubMed] [Google Scholar]
- 193.Kuryk L., Møller A.S.W., Garofalo M., Cerullo V., Pesonen S., Alemany R., Jaderberg M. Antitumor-specific T-cell responses induced by oncolytic adenovirus ONCOS-102 (AdV5/3-D24-GM-CSF) in peritoneal mesothelioma mouse model. J. Med. Virol. 2018;90:1669–1673. doi: 10.1002/jmv.25229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Lv S.Q., Ye Z.L., Liu P.Y., Huang Y., Li L.F., Liu H., Zhu H.L., Jin H.J., Qian Q.J. 11R-P53 and GM-CSF Expressing Oncolytic Adenovirus Target Cancer Stem Cells with Enhanced Synergistic Activity. J. Cancer. 2017;8:199–206. doi: 10.7150/jca.16406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Crupi M.J.F., Taha Z., Janssen T.J.A., Petryk J., Boulton S., Alluqmani N., Jirovec A., Kassas O., Khan S.T., Vallati S., et al. Oncolytic virus driven T-cell-based combination immunotherapy platform for colorectal cancer. Front. Immunol. 2022;13 doi: 10.3389/fimmu.2022.1029269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Zhao Y., Liu Z., Li L., Wu J., Zhang H., Zhang H., Lei T., Xu B. Oncolytic Adenovirus: Prospects for Cancer Immunotherapy. Front. Microbiol. 2021;12 doi: 10.3389/fmicb.2021.707290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Kirchner M., Schneider S. CRISPR-Cas: From the Bacterial Adaptive Immune System to a Versatile Tool for Genome Engineering. Angew. Chem. Int. Ed. Engl. 2015;54:13508–13514. doi: 10.1002/anie.201504741. [DOI] [PubMed] [Google Scholar]
- 198.Wang X., Huang X., Fang X., Zhang Y., Wang W. CRISPR-Cas9 System as a Versatile Tool for Genome Engineering in Human Cells. Molecular therapy. Nucleic acids. 2016;5:e388. doi: 10.1038/mtna.2016.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Palmer D.J., Turner D.L., Ng P. Production of CRISPR/Cas9-Mediated Self-Cleaving Helper-Dependent Adenoviruses. Molecular therapy. Mol. Ther. Methods Clin. Dev. 2019;13:432–439. doi: 10.1016/j.omtm.2019.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Palmer D.J., Turner D.L., Ng P. Bi-allelic Homology-Directed Repair with Helper-Dependent Adenoviruses. Molecular therapy. Mol. Ther. Methods Clin. Dev. 2019;15:285–293. doi: 10.1016/j.omtm.2019.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Palmer D.J., Turner D.L., Ng P. A Single "All-in-One" Helper-Dependent Adenovirus to Deliver Donor DNA and CRISPR/Cas9 for Efficient Homology-Directed Repair. Molecular therapy. Mol. Ther. Methods Clin. Dev. 2020;17:441–447. doi: 10.1016/j.omtm.2020.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Gupta S.M., Warke H., Chaudhari H., Mavani P., Katke R.D., Kerkar S.C., Mania-Pramanik J. Human Papillomavirus E6/E7 oncogene transcripts as biomarkers for the early detection of cervical cancer. J. Med. Virol. 2022;94:3368–3375. doi: 10.1002/jmv.27700. [DOI] [PubMed] [Google Scholar]
- 203.Ehrke-Schulz E., Schiwon M., Hagedorn C., Ehrhardt A. Establishment of the CRISPR/Cas9 System for Targeted Gene Disruption and Gene Tagging. Methods Mol. Biol. 2017;1654:165–176. doi: 10.1007/978-1-4939-7231-9_11. [DOI] [PubMed] [Google Scholar]
- 204.Ehrke-Schulz E., Heinemann S., Schulte L., Schiwon M., Ehrhardt A. Adenoviral Vectors Armed with PAPILLOMAVIRUs Oncogene Specific CRISPR/Cas9 Kill Human-Papillomavirus-Induced Cervical Cancer Cells. Cancers. 2020;12 doi: 10.3390/cancers12071934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Endres T.M., Konstan M.W. What Is Cystic Fibrosis? JAMA. 2022;327:191. doi: 10.1001/jama.2021.23280. [DOI] [PubMed] [Google Scholar]
- 206.Donnelley M., Parsons D.W. Gene Therapy for Cystic Fibrosis Lung Disease: Overcoming the Barriers to Translation to the Clinic. Front. Pharmacol. 2018;9:1381. doi: 10.3389/fphar.2018.01381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Jiang Y., Han Q., Zhao H., Zhang J. The Mechanisms of HBV-Induced Hepatocellular Carcinoma. J. Hepatocell. Carcinoma. 2021;8:435–450. doi: 10.2147/JHC.S307962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Kato Y., Tabata H., Sato K., Nakamura M., Saito I., Nakanishi T. Adenovirus Vectors Expressing Eight Multiplex Guide RNAs of CRISPR/Cas9 Efficiently Disrupted Diverse Hepatitis B Virus Gene Derived from Heterogeneous Patient. Int. J. Mol. Sci. 2021;22 doi: 10.3390/ijms221910570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Pecot C.V., Calin G.A., Coleman R.L., Lopez-Berestein G., Sood A.K. RNA interference in the clinic: challenges and future directions. Nat. Rev. Cancer. 2011;11:59–67. doi: 10.1038/nrc2966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Chen W., Liu M., Jiao Y., Yan W., Wei X., Chen J., Fei L., Liu Y., Zuo X., Yang F., et al. Adenovirus-mediated RNA interference against foot-and-mouth disease virus infection both in vitro and in vivo. J. Virol. 2006;80:3559–3566. doi: 10.1128/JVI.80.7.3559-3566.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Motegi Y., Katayama K., Sakurai F., Kato T., Yamaguchi T., Matsui H., Takahashi M., Kawabata K., Mizuguchi H. An effective gene-knockdown using multiple shRNA-expressing adenovirus vectors. J. Control. Release. 2011;153:149–153. doi: 10.1016/j.jconrel.2011.04.009. [DOI] [PubMed] [Google Scholar]
- 212.Mowa M.B., Crowther C., Arbuthnot P. Therapeutic potential of adenoviral vectors for delivery of expressed RNAi activators. Expert Opin. Drug Deliv. 2010;7:1373–1385. doi: 10.1517/17425247.2010.533655. [DOI] [PubMed] [Google Scholar]
- 213.Alles J., Fehlmann T., Fischer U., Backes C., Galata V., Minet M., Hart M., Abu-Halima M., Grässer F.A., Lenhof H.P., et al. An estimate of the total number of true human miRNAs. Nucleic Acids Res. 2019;47:3353–3364. doi: 10.1093/nar/gkz097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Idogawa M., Sasaki Y., Suzuki H., Mita H., Imai K., Shinomura Y., Tokino T. A single recombinant adenovirus expressing p53 and p21-targeting artificial microRNAs efficiently induces apoptosis in human cancer cells. Clin. Cancer Res. 2009;15:3725–3732. doi: 10.1158/1078-0432.CCR-08-2396. [DOI] [PubMed] [Google Scholar]
- 215.Yan M., Chen J., Jiang H., Xie Y., Li C., Chen L., Yang B., Cao J. Effective inhibition of cancer cells by recombinant adenovirus expressing EGFR-targeting artificial microRNA and reversed-caspase-3. PLoS One. 2020;15 doi: 10.1371/journal.pone.0237098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Lou W., Chen Q., Ma L., Liu J., Yang Z., Shen J., Cui Y., Bian X.W., Qian C. Oncolytic adenovirus co-expressing miRNA-34a and IL-24 induces superior antitumor activity in experimental tumor model. J. Mol. Med. 2013;91:715–725. doi: 10.1007/s00109-012-0985-x. [DOI] [PubMed] [Google Scholar]
- 217.Kang S., Kang D., Bakhtiar Ul Islam S.M., Je S., Kim J.H., Song J.J. Silencing Daxx increases the anti-tumor activity of a TRAIL/shRNA Bcl-xL-expressing oncolytic adenovirus through enhanced viral replication and cellular arrest. Cell. Signal. 2015;27:1214–1224. doi: 10.1016/j.cellsig.2015.02.028. [DOI] [PubMed] [Google Scholar]
- 218.Zhang H., Tang X., Zhu C., Song Y., Yin J., Xu J., Ertl H.C.J., Zhou D. Adenovirus-mediated artificial MicroRNAs targeting matrix or nucleoprotein genes protect mice against lethal influenza virus challenge. Gene Ther. 2015;22:653–662. doi: 10.1038/gt.2015.31. [DOI] [PubMed] [Google Scholar]
- 219.Tang X., Zhang H., Song Y., Zhou D., Wang J. Hemagglutinin-targeting Artificial MicroRNAs Expressed by Adenovirus Protect Mice From Different Clades of H5N1 Infection. Molecular therapy. Nucleic acids. 2016;5:e311. doi: 10.1038/mtna.2016.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Gürlevik E., Woller N., Strüver N., Schache P., Kloos A., Manns M.P., Zender L., Kühnel F., Kubicka S. Selectivity of oncolytic viral replication prevents antiviral immune response and toxicity, but does not improve antitumoral immunity. Mol. Ther. 2010;18:1972–1982. doi: 10.1038/mt.2010.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Morozov V.A., Lagaye S. Hepatitis C virus: Morphogenesis, infection and therapy. World J. Hepatol. 2018;10:186–212. doi: 10.4254/wjh.v10.i2.186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Ledgerwood J.E., DeZure A.D., Stanley D.A., Coates E.E., Novik L., Enama M.E., Berkowitz N.M., Hu Z., Joshi G., Ploquin A., et al. Chimpanzee Adenovirus Vector Ebola Vaccine. N. Engl. J. Med. 2017;376:928–938. doi: 10.1056/NEJMoa1410863. [DOI] [PubMed] [Google Scholar]
- 223.Sakurai F., Furukawa N., Higuchi M., Okamoto S., Ono K., Yoshida T., Kondoh M., Yagi K., Sakamoto N., Katayama K., Mizuguchi H. Suppression of hepatitis C virus replicon by adenovirus vector-mediated expression of tough decoy RNA against miR-122a. Virus Res. 2012;165:214–218. doi: 10.1016/j.virusres.2012.02.003. [DOI] [PubMed] [Google Scholar]
- 224.Rudan I. The COVID-19 pandemic: SARS-CoV-2, childhood hepatitis and monkeypox raise five new questions for the global health research community. J. Glob. Health. 2022;22 doi: 10.7189/jogh.12.01002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Jiang F., Liu Y., Xue Y., Cheng P., Wang J., Lian J., Gong W. Developing a multiepitope vaccine for the prevention of SARS-CoV-2 and monkeypox virus co-infection: A reverse vaccinology analysis. Int. Immunopharmacol. 2023;115 doi: 10.1016/j.intimp.2023.109728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Jacob-Dolan C., Barouch D.H. COVID-19 Vaccines: Adenoviral Vectors. Annu. Rev. Med. 2022;73:41–54. doi: 10.1146/annurev-med-012621-102252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.O'Malley B.W., Jr., Sewell D.A., Li D., Kosai K., Chen S.H., Woo S.L., Duan L. The role of interleukin-2 in combination adenovirus gene therapy for head and neck cancer. Mol. Endocrinol. 1997;11:667–673. doi: 10.1210/mend.11.6.0012. [DOI] [PubMed] [Google Scholar]
- 228.Yao M.Z., Gu J.F., Wang J.H., Sun L.Y., Liu H., Liu X.Y. Adenovirus-mediated interleukin-2 gene therapy of nociception. Gene Ther. 2003;10:1392–1399. doi: 10.1038/sj.gt.3301992. [DOI] [PubMed] [Google Scholar]
- 229.Toloza E.M., Hunt K., Swisher S., McBride W., Lau R., Pang S., Rhoades K., Drake T., Belldegrun A., Glaspy J., Economou J.S. vivo cancer gene therapy with a recombinant interleukin-2 adenovirus vector. Cancer Gene Ther. 1996;3:11–17. [PubMed] [Google Scholar]
- 230.Mulders P., Tso C.L., Pang S., Kaboo R., McBride W.H., Hinkel A., Gitlitz B., Dannull J., Figlin R., Belldegrun A. Adenovirus-mediated interleukin-2 production by tumors induces growth of cytotoxic tumor-infiltrating lymphocytes against human renal cell carcinoma. J. Immunother. 1998;21:170–180. doi: 10.1097/00002371-199805000-00002. [DOI] [PubMed] [Google Scholar]
- 231.Bortolanza S., Bunuales M., Otano I., Gonzalez-Aseguinolaza G., Ortiz-de-Solorzano C., Perez D., Prieto J., Hernandez-Alcoceba R. Treatment of pancreatic cancer with an oncolytic adenovirus expressing interleukin-12 in Syrian hamsters. Mol. Ther. 2009;17:614–622. doi: 10.1038/mt.2009.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.He X.Z., Wang L., Zhang Y.Y. An effective vaccine against colon cancer in mice: use of recombinant adenovirus interleukin-12 transduced dendritic cells. World J. Gastroenterol. 2008;14:532–540. doi: 10.3748/wjg.14.532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Nasu Y., Ebara S., Kumon H. [Adenovirus-mediated interleukin-12 gene therapy for prostate cancer] Nihon Rinsho. 2004;62:1181–1191. [PubMed] [Google Scholar]
- 234.Sangro B., Mazzolini G., Ruiz J., Herraiz M., Quiroga J., Herrero I., Benito A., Larrache J., Pueyo J., Subtil J.C., et al. Phase I trial of intratumoral injection of an adenovirus encoding interleukin-12 for advanced digestive tumors. J. Clin. Oncol. 2004;22:1389–1397. doi: 10.1200/JCO.2004.04.059. [DOI] [PubMed] [Google Scholar]
- 235.Isvoranu G., Surcel M., Munteanu A.N., Bratu O.G., Ionita-Radu F., Neagu M.T., Chiritoiu-Butnaru M. Therapeutic potential of interleukin-15 in cancer (Review) Exp. Ther. Med. 2021;22:675. doi: 10.3892/etm.2021.10107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Morris J.C., Ramlogan-Steel C.A., Yu P., Black B.A., Mannan P., Allison J.P., Waldmann T.A., Steel J.C. Vaccination with tumor cells expressing IL-15 and IL-15Ralpha inhibits murine breast and prostate cancer. Gene Ther. 2014;21:393–401. doi: 10.1038/gt.2014.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Yang C., Cao H., Liu N., Xu K., Ding M., Mao L.J. Oncolytic adenovirus expressing interleukin-18 improves antitumor activity of dacarbazine for malignant melanoma. Drug Des. Devel. Ther. 2016;10:3755–3761. doi: 10.2147/DDDT.S115121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Zheng J.N., Pei D.S., Mao L.J., Liu X.Y., Sun F.H., Zhang B.F., Liu Y.Q., Liu J.J., Li W., Han D. Oncolytic adenovirus expressing interleukin-18 induces significant antitumor effects against melanoma in mice through inhibition of angiogenesis. Cancer Gene Ther. 2010;17:28–36. doi: 10.1038/cgt.2009.38. [DOI] [PubMed] [Google Scholar]
- 239.Zheng J.N., Pei D.S., Sun F.H., Liu X.Y., Mao L.J., Zhang B.F., Wen R.M., Xu W., Shi Z., Liu J.J., Li W. Potent antitumor efficacy of interleukin-18 delivered by conditionally replicative adenovirus vector in renal cell carcinoma-bearing nude mice via inhibition of angiogenesis. Cancer Biol. Ther. 2009;8:599–606. doi: 10.4161/cbt.8.7.7914. [DOI] [PubMed] [Google Scholar]
- 240.Sato-Dahlman M., LaRocca C.J., Yanagiba C., Yamamoto M. Adenovirus and Immunotherapy: Advancing Cancer Treatment by Combination. Cancers. 2020;12 doi: 10.3390/cancers12051295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Cervera-Carrascon V., Quixabeira D.C.A., Santos J.M., Havunen R., Milenova I., Verhoeff J., Heiniö C., Zafar S., Garcia-Vallejo J.J., van Beusechem V.W., et al. Adenovirus Armed With TNFa and IL2 Added to aPD-1 Regimen Mediates Antitumor Efficacy in Tumors Refractory to aPD-1. Front. Immunol. 2021;12 doi: 10.3389/fimmu.2021.706517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Cervera-Carrascon V., Siurala M., Santos J.M., Havunen R., Tähtinen S., Karell P., Sorsa S., Kanerva A., Hemminki A. TNFa and IL-2 armed adenoviruses enable complete responses by anti-PD-1 checkpoint blockade. OncoImmunology. 2018;7 doi: 10.1080/2162402X.2017.1412902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Huang H., Xiao T., He L., Ji H., Liu X.Y. Interferon-beta-armed oncolytic adenovirus induces both apoptosis and necroptosis in cancer cells. Acta Biochim. Biophys. Sin. 2012;44:737–745. doi: 10.1093/abbs/gms060. [DOI] [PubMed] [Google Scholar]
- 244.Borden E.C. Interferons alpha and beta in cancer: therapeutic opportunities from new insights. Nat. Rev. Drug Discov. 2019;18:219–234. doi: 10.1038/s41573-018-0011-2. [DOI] [PubMed] [Google Scholar]
- 245.Dinney C.P.N., Fisher M.B., Navai N., O'Donnell M.A., Cutler D., Abraham A., Young S., Hutchins B., Caceres M., Kishnani N., et al. Phase I trial of intravesical recombinant adenovirus mediated interferon-alpha2b formulated in Syn3 for Bacillus Calmette-Guerin failures in nonmuscle invasive bladder cancer. J. Urol. 2013;190:850–856. doi: 10.1016/j.juro.2013.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Tian Y., Xie D., Yang L. Engineering strategies to enhance oncolytic viruses in cancer immunotherapy. Signal Transduct. Target. Ther. 2022;7:117. doi: 10.1038/s41392-022-00951-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Wang L., Qi X., Sun Y., Liang L., Ju D. Adenovirus-mediated combined P16 gene and GM-CSF gene therapy for the treatment of established tumor and induction of antitumor immunity. Cancer Gene Ther. 2002;9:819–824. doi: 10.1038/sj.cgt.7700502. [DOI] [PubMed] [Google Scholar]
- 248.Li X., Lu M., Yuan M., Ye J., Zhang W., Xu L., Wu X., Hui B., Yang Y., Wei B., et al. CXCL10-armed oncolytic adenovirus promotes tumor-infiltrating T-cell chemotaxis to enhance anti-PD-1 therapy. OncoImmunology. 2022;11 doi: 10.1080/2162402X.2022.2118210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.O'Bryan S.M., Mathis J.M. CXCL12 Retargeting of an Oncolytic Adenovirus Vector to the Chemokine CXCR4 and CXCR7 Receptors in Breast Cancer. J. Cancer Ther. 2021;12:311–336. doi: 10.4236/jct.2021.126029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Mao L.J., Ding M., Xu K., Pan J., Yu H., Yang C. Oncolytic Adenovirus Harboring Interleukin-24 Improves Chemotherapy for Advanced Prostate Cancer. J. Cancer. 2018;9:4391–4397. doi: 10.7150/jca.26437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Ang L., Li J., Dong H., Wang C., Huang J., Li M., Zhao M., Su C., Wu Q. Chimeric Oncolytic Adenovirus Armed Chemokine Rantes for Treatment of Breast Cancer. Bioengineering (Basel) 2022;9 doi: 10.3390/bioengineering9080342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Li J., Liu H., Li L., Wu H., Wang C., Yan Z., Wang Y., Su C., Jin H., Zhou F., et al. The combination of an oxygen-dependent degradation domain-regulated adenovirus expressing the chemokine RANTES/CCL5 and NK-92 cells exerts enhanced antitumor activity in hepatocellular carcinoma. Oncol. Rep. 2013;29:895–902. doi: 10.3892/or.2012.2217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Freedman J.D., Duffy M.R., Lei-Rossmann J., Muntzer A., Scott E.M., Hagel J., Campo L., Bryant R.J., Verrill C., Lambert A., et al. An Oncolytic Virus Expressing a T-cell Engager Simultaneously Targets Cancer and Immunosuppressive Stromal Cells. Cancer Res. 2018;78:6852–6865. doi: 10.1158/0008-5472.CAN-18-1750. [DOI] [PubMed] [Google Scholar]
- 254.Huang Q., Cai W.Q., Han Z.W., Wang M.Y., Zhou Y., Cheng J.T., Zhang Y., Wang Y.Y., Xin Q., Wang X.W., et al. Bispecific T cell engagers and their synergistic tumor immunotherapy with oncolytic viruses. Am. J. Cancer Res. 2021;11:2430–2455. [PMC free article] [PubMed] [Google Scholar]