Abstract
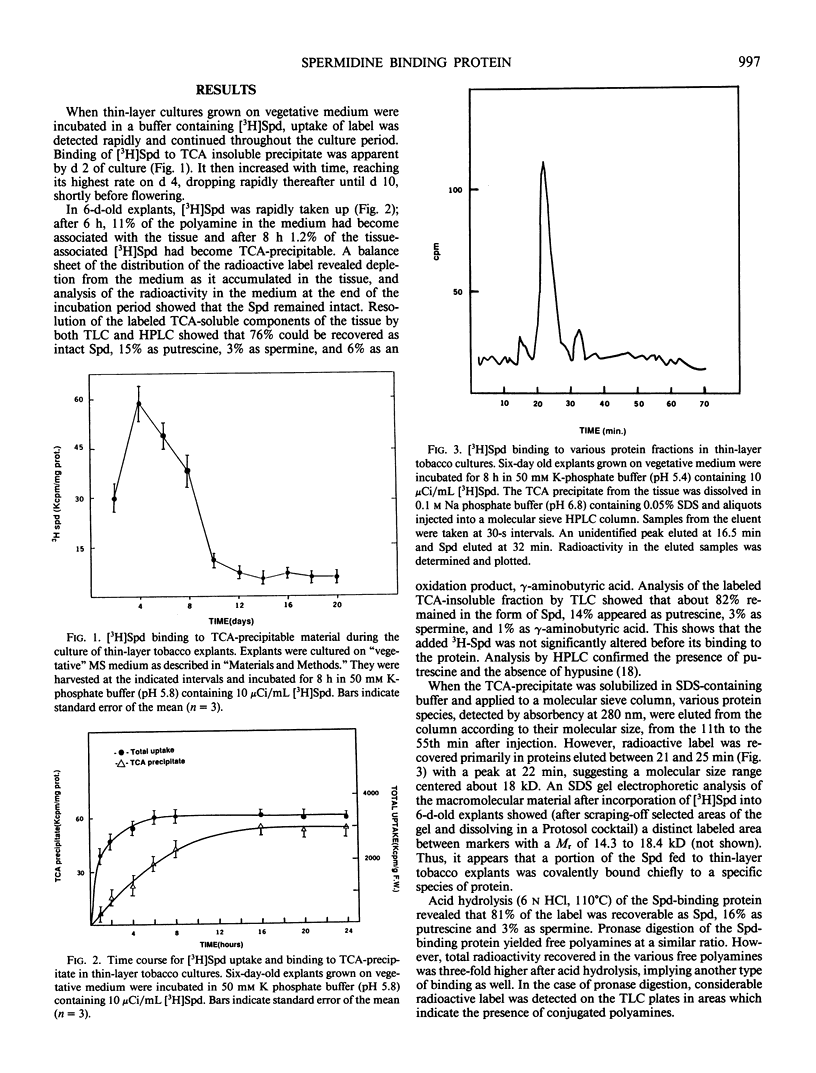
The mechanism by which spermidine induces the appearance of floral buds in thin-layer tobacco (Nicotiana tabacum) tissue culture was studied by following the fate of the radioactive compound. [3H]Spermidine was taken up rapidly by the tissue, and after a brief lag, a portion was bound to trichloroacetic acid precipitable macromolecules. Such binding increased to a maximum on day 4 of culture, coinciding with the onset of bud differentiation, and declined thereafter until shortly before flowering. About 82% of the label in the trichloroacetic acid precipitate remained as spermidine, 14% was metabolized to putrescine, 3% to spermine, and 1% to γ-aminobutyric acid. Spermidine was covalently bound to a protein with a molecular size of about 18 kilodaltons. Hydrolysis of this protein and analysis of the labeled entities revealed 81% spermidine, 16% putrescine, and 3% spermine. This post-translational modification of a unique protein by attachment of spermidine may be causally connected to the appearance of flower buds in thin-layer tobacco cultures.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Birecka H., Garraway M. O., Baumann R. J., McCann P. P. Inhibition of Ornithine Decarboxylase and Growth of the Fungus Helminthosporium maydis. Plant Physiol. 1986 Mar;80(3):798–800. doi: 10.1104/pp.80.3.798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Canellakis Z. N., Bondy P. K., Infante A. A. Spermidine is bound to a unique protein in early sea urchin embryos. Proc Natl Acad Sci U S A. 1985 Nov;82(22):7613–7615. doi: 10.1073/pnas.82.22.7613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cariello L., Wilson J., Lorand L. Activation of transglutaminase during embryonic development. Biochemistry. 1984 Dec 18;23(26):6843–6850. doi: 10.1021/bi00321a087. [DOI] [PubMed] [Google Scholar]
- Feirer R. P., Mignon G., Litvay J. D. Arginine decarboxylase and polyamines required for embryogenesis in the wild carrot. Science. 1984 Mar 30;223(4643):1433–1435. doi: 10.1126/science.223.4643.1433. [DOI] [PubMed] [Google Scholar]
- Icekson I., Apelbaum A. Evidence for transglutaminase activity in plant tissue. Plant Physiol. 1987 Aug;84(4):972–974. doi: 10.1104/pp.84.4.972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaur Sawhney R., Shekhawat N. S., Galston A. W. Polyamine levels as related to growth, differentiation and senescence in protoplast-derived cultures of Vigna aconitifolia and Avena sativa. Plant Growth Regul. 1985;3:329–337. doi: 10.1007/BF00117590. [DOI] [PubMed] [Google Scholar]
- Montague M. J., Koppenbrink J. W., Jaworski E. G. Polyamine Metabolism in Embryogenic Cells of Daucus carota: I. Changes in Intracellular Content and Rates of Synthesis. Plant Physiol. 1978 Sep;62(3):430–433. doi: 10.1104/pp.62.3.430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park M. H., Cooper H. L., Folk J. E. Identification of hypusine, an unusual amino acid, in a protein from human lymphocytes and of spermidine as its biosynthetic precursor. Proc Natl Acad Sci U S A. 1981 May;78(5):2869–2873. doi: 10.1073/pnas.78.5.2869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajam M. V., Galston A. W. The effects of some polyamine biosynthetic inhibitors on growth and morphology of phytopathogenic fungi. Plant Cell Physiol. 1985;26(4):683–692. doi: 10.1093/oxfordjournals.pcp.a076958. [DOI] [PubMed] [Google Scholar]
- Serafini-Fracassini D., Del Duca S., D'Orazi D. First evidence for polyamine conjugation mediated by an enzymic activity in plants. Plant Physiol. 1988 Jul;87(3):757–761. doi: 10.1104/pp.87.3.757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slocum R. D., Kaur-Sawhney R., Galston A. W. The physiology and biochemistry of polyamines in plants. Arch Biochem Biophys. 1984 Dec;235(2):283–303. doi: 10.1016/0003-9861(84)90201-7. [DOI] [PubMed] [Google Scholar]
- Tabor C. W., Tabor H. Polyamines in microorganisms. Microbiol Rev. 1985 Mar;49(1):81–99. doi: 10.1128/mr.49.1.81-99.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams-Ashman H. G., Canellakis Z. N. Transglutaminase-mediated covalent attachment of polyamines to proteins: mechanisms and potential physiological significance. Physiol Chem Phys. 1980;12(5):457–472. [PubMed] [Google Scholar]


