Abstract
The intricate nature of Alzheimer's disease (AD) pathogenesis poses a persistent obstacle to drug development. In recent times, neuroinflammation has emerged as a crucial pathogenic mechanism of AD, and the targeting of inflammation has become a viable approach for the prevention and management of AD. The present study conducted a comprehensive review of the literature between October 2012 and October 2022, identifying a total of 96 references, encompassing 91 distinct pharmaceuticals that have been investigated for their potential impact on AD by inhibiting neuroinflammation. Research has shown that pharmaceuticals have the potential to ameliorate AD by reducing neuroinflammation mainly through regulating inflammatory signaling pathways such as NF-κB, MAPK, NLRP3, PPARs, STAT3, CREB, PI3K/Akt, Nrf2 and their respective signaling pathways. Among them, tanshinone IIA has been extensively studied for its anti-inflammatory effects, which have shown significant pharmacological properties and can be applied clinically. Thus, it may hold promise as an effective drug for the treatment of AD. The present review elucidated the inflammatory signaling pathways of pharmaceuticals that have been investigated for their therapeutic efficacy in AD and elucidates their underlying mechanisms. This underscores the auspicious potential of pharmaceuticals in ameliorating AD by impeding neuroinflammation.
Keywords: Alzheimer's disease, neuroinflammation, inhibitor, inflammatory signaling pathways, treatment
1. Introduction
AD is a common neurodegenerative disorder characterized by gradual cognitive decline, memory loss, and behavioral changes (1). AD is mainly characterized by the accumulation of extracellular amyloid β (Aβ), which forms senile plaques, and intracellular hyperphosphorylated tau, which binds to microtubules and leads to the development of neurofibrillary tangles (2). The disease is becoming increasingly prevalent, with projections estimating a global population of 115 million patients with AD by 2050 (3). With a growing aging population, the management of AD is becoming increasingly critical.
The pathogenesis of AD is multifactorial and involves a number of hypotheses, including the cholinergic theory, the amyloid cascade theory, the oxidative stress theory, the tau protein hypothesis and the neuroinflammation hypothesis (4). Evidence supports the neuroinflammation as a crucial factor in the development of AD (5,6). Neuroinflammation (7-9) is present in the majority of patients with AD (10) and animal models (11), particularly in the cerebral cortex and hippocampus (12,13). Elevated levels of inflammatory factors and increased activation of microglia around senile plaques observed in patients with AD further support this hypothesis (14). In addition, whole-genome studies of post-mortem brain samples from patients with AD have shown upregulation of inflammation-related genes and significant downregulation of anti-inflammatory molecules (15). Activated microglia, responding to Aβ (16), demonstrate a significant inflammatory response highly correlated with the severity of AD (17). Taken together, these findings suggest that neuroinflammatory responses mediated by microglial cell activation may play a central role in the pathogenesis of AD.
Under normal circumstances, highly active microglia cells efficiently monitor the entire brain in real time (18), detecting abnormalities such as pathogens and cellular debris (19,20) and providing essential support to maintain optimal brain function (21). However, when the brain is exposed to abnormal conditions, microglia become activated and switch to a transforming state, migrating towards the site of injury to remove pathogens, cellular debris and degenerated cells (22). Depending on their activation state and environmental stimuli, microglia cells can be classified as either the pro-inflammatory M1 type or the anti-inflammatory M2 type (23). In the early stages of AD, microglia play a crucial role in maintaining a dynamic balance of amyloid protein in the brain by engulfing and clearing excess Aβ, thereby helping to delay disease progression. However, as the disease progresses, excessive accumulation of Aβ can lead to overactivation of microglia cells, causing them to adopt a pro-inflammatory M1 type (24). In the central nervous system (CNS), activated microglia are the primary source of inflammatory molecules, such as cytokines, chemokines, neurotransmitters, reactive oxygen species (ROS) and nitric oxide (NO) (25). Inflammatory molecules trigger a positive feedback mechanism that activates more microglia and thus further exacerbating the neuroinflammatory response (25,26). As a result, secreted inflammatory mediators facilitate the migration of monocytes and lymphocytes to the site of inflammation, where they penetrate the blood-brain barrier (BBB), exacerbating CNS inflammation and leading to sustained neuronal damage (27), ultimately culminating in cognitive decline. Several inhibitors, drugs and their active ingredients can exert an anti-neuroinflammatory effects, with different drugs acting via single or multiple signaling pathways. Therefore, it is essential to consolidate research findings to identify potential drug candidates for the prevention and treatment of AD.
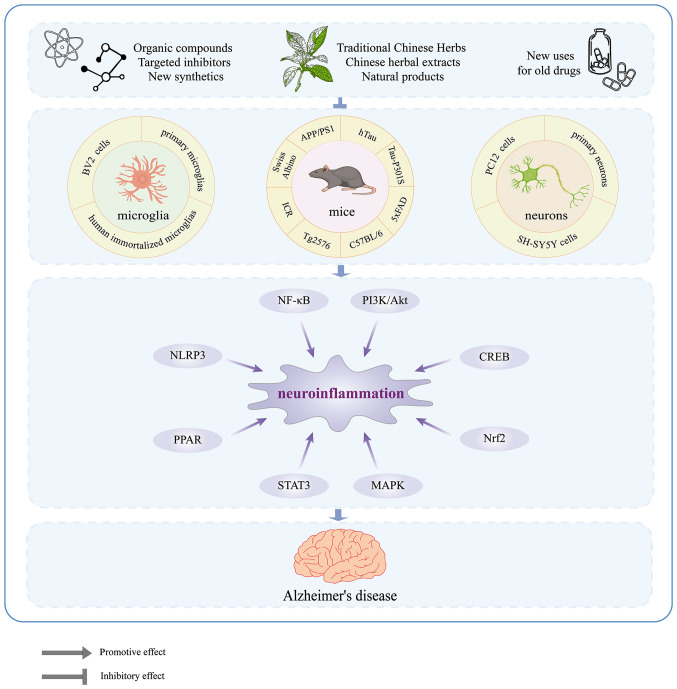
Neuroinflammation is a critical factor and even a core event in the pathogenesis of AD (17,28). Microglia, as the primary immune cells in brain tissue, play an essential role in neuroinflammation through multiple targets and signaling pathways. Therefore, the development of drugs or inhibitors that target microglia could alleviate neuroinflammation, which could have a positive effect on both the prevention and treatment of AD. The present study conducted a literature search using keywords the 'inhibitors', 'microglia', 'inflammation' and 'Alzheimer's disease' in PubMed between 2012 and 2022 to comprehensively review the major signaling pathways involved in microglia activation and the ways in which drugs exert anti-neuroinflammatory effects by targeting these pathways. Out of the 327 articles retrieved, 35 were excluded, including reviews, commentaries, retractions, or unavailability online. Also excluded were 201 articles that did not involve signaling pathways. Finally, 96 references were included. In addition, 'medicine' and 'drugs' were added as keywords to the search to further identify promising drug candidates for AD prevention.
2. NF-κB and MAPK signaling pathways
NF-κB (nuclear factor-kappa-B)
The NF-κB signaling pathway is a complex protein interaction network (29) that plays a critical role in regulating gene expression in response to various stimuli, including pro-inflammatory signals (30). In most cell types, NF-κB is activated by the classical pathway, which involves a dimer composed of p50 and p65 subunits (31). In the inactive state, the NF-κB/IκB dimer is inhibited by IκB and remains sequestered in the cytoplasm (32,33). Upon activation of the NF-κB/IκB dimer by pro-inflammatory signals, IκB kinase (IKK) phosphorylates IκB, leading to its degradation. This allows NF-κB to dissociate from the complex, enter the nucleus, and activate the transcription of cytokines and adhesion molecules (34-36), contributing to the pathogenesis of neuroinflammatory diseases such as AD.
NF-κB is widely expressed in brain tissue and plays a critical regulatory role in various target genes within the CNS. Its regulatory scope encompasses oxidative stress, neuroinflammation and microglia activation (31). In particular, excessive activation of NF-κB has been implicated in the neuropathological features of AD. Multiple studies have identified increased activation of NF-κB in the brains of patients with AD (31,37), particularly in the most affected brain regions (38-40). Additionally, the activation of NF-κB by Aβ leads to further production of Aβ, exacerbating the pathology of AD (41,42). Moreover, NF-κB not only acts downstream of tau but also seems to directly mediate its cognitive toxicity (43). This increased DNA-binding activity of NF-κB leads to aggravated oxidative stress, which exacerbates neurotoxicity. In addition, downstream pro-inflammatory mediators are activated, thereby affecting neuronal function (44,45). Above all, activation of glial cells via the NF-κB pathway serves as a critical link in the neuroinflammatory response (46), further amplifying neuroinflammation and worsening AD pathology (47,48). As such, modulation of the NF-κB signaling pathway in microglia may represent a promising new approach to the prevention and treatment of AD.
Studies have shown that certain compounds found in traditional Chinese herbal medicine possess the capacity to inhibit NF-κB activation and exert anti-inflammatory effects. Rutin, a natural flavonoid glycoside with anti-inflammatory and antioxidant properties (49), is a promising neuroprotective agent for neurodegenerative diseases (50). A recent study has revealed that treatment with Rutin can reduce NF-κB activation in the Tau-P301S mouse, resulting in lower levels of IL-1 and TNF-α in brain tissue, thereby counteracting neuroinflammation (51). Results consistent with in vivo findings were also observed in microglia induced with tau oligomers (51). Similarly, piperlongumine, an alkaloid amide from Piper longum, was found to be neuroprotective effects (52) against lipopolysaccharide (LPS)-induced neuroinflammation by inhibiting the NF-κB pathway and reducing the expression of key pro-inflammatory mediators such as cyclooxygenase-2 (COX-2), inducible nitric oxide synthase (iNOS), TNF-α, IL-1β, and IL-6. Thus, these compounds show therapeutic potential for the treatment of neuroinflammatory disorders by modulating the NF-κB signaling pathway in microglia (53). Bee venom, which contains various peptides, enzymes, and biogenic amines, has been shown to be effective in the treatment of diseases such as arthritis, rheumatism and cancer (54). A study has highlighted its potential for treating AD by inhibiting the expression of neuroinflammatory proteins such as β-site amyloid precursor protein cleaving enzyme 1 (BACE1), COX-2, iNOS, glial fibrillary acidic protein (GFAP), and ionized calcium binding adaptor molecule 1, in vitro and in vivo, through inactivation of the NF-κB pathway, resulting in a reduction in LPS-induced memory impairment (55). Punicalagin, a polyphenol sourced from pomegranate fruit, has antioxidant, anti-proliferative and anti-inflammatory properties (56). It has been shown to bind directly to NF-κB, impede IκB degradation and prevent the nuclear translocation of p50 and p65, thereby inhibiting the production of ROS, NO, TNF-α and IL-1β in LPS-induced BV-2 microglia (57). Similarly, tenuifolin, a valuable neuroprotective compound extracted from Polygala tenuifolia Willd, can block the activation of the NF-κB pathway and subsequently improve cognitive impairment symptoms in AD (58). Piperine, a crystalline alkaloid extracted from pepper, has several properties such as anticarcinogenic, stimulatory, anti-inflammatory and antiulcer activities (59). Furthermore, piperine derivatives, such as (2E,4E)-5-(benzo[d][1,3]dioxo l-5-yl)-N-[4-(hydroxymethyl) phenyl] penta-2,4-dienamide (D4) have demonstrated anti-neuroinflammatory effects (60) by inhibiting the translocation of NF-κB and suppressing the expression of iNOS and the secretion of NO, TNF-α, and IL-1β in LPS-induced human microglia clone 3. In addition, an in silico study showed excellent D4 bioavailability after oral administration (61). Bupleurum falcatum L. (BF) is a traditional oriental medicine commonly used in the treatment of chronic hepatitis and autoimmune diseases (62). It has been demonstrated that the ethanol extract of BF (BFE) can inhibit the expression of pro-inflammatory genes and NF-κB p65/RELA mRNA in BV2 microglia that have been activated with LPS. This suggests that NF-κB is a molecular target of BFE (63). In addition, BFE has been shown to inhibit the activation of microglia in the hippocampus and substantia nigra of LPS-treated mice (63), suggesting its potential as a treatment for AD. Similarly, macasiamenene F (MF), a compound extracted from Macaranga siamensis S. J. Davies (Euphorbiaceae), has also been shown to have promising potential in the treatment of neuroinflammatory responses. MF treatment significantly suppresses NF-κB activity and TNF-α expression in LPS-induced human monocytes (64), and similar responses may occur in microglia of brain given their phenotypic similarity. Miconazole (MCZ) is an azole drug commonly used as an antifungal agent that can cross the BBB and exhibits neuroprotective effects (64,65). MCZ can reduce the expression of ionized calcium binding adaptor molecule 1 (Iba-1) reactive cells and downregulate the expression of GFAP, Iba-1, and COX-2 in the hippocampus by inhibiting the NF-κB signaling pathway in a mouse model of Aβ1-42-induced memory impairment. This anti-inflammatory effect of MCZ was further confirmed in an LPS-induced BV2 microglia model (66).
Several drugs have been developed to target specific components of the body and exert anti-neuroinflammatory effects by inhibiting NF-κB (67-70). Among these, LD55, a resveratrol analogue, is widely used as a novel inhibitor of NF-κB activation (71). A study has shown that dietary supplementation with LD55 can effectively suppress the activation of microglia in transgenic amyloid-β protein/presenilin-1 (APP/PS1) mice, diminish the density of Aβ plaques in the brain and notably reduce them by 2-15 times in the hippocampal region. These findings suggest that LD55 may provide some relief from the burden of Aβ plaques and neuroinflammation in AD models (67). Additionally, glucocorticoid-induced leucine zipper (GILZ), which functions as a transcriptional regulatory protein, has the ability to impede the activity of NF-κB (72,73). A small molecule GILZ analogue, GA, was found to inhibit the levels of NF-κB p65 in the brains of 5XFAD (familial Alzheimer's disease) mice. Furthermore, GA can downregulate the expression of inflammatory factors while hindering the proliferation and activation of hippocampal microglia (68). Consequently, this leads to the suppression of neuroinflammation. Chitinase-3 like-protein-1 (CHI3L1) is a secreted, inflammatory glycoprotein that is expressed in a number of chronic neuroinflammatory diseases including AD, making it a potential biomarker for AD diagnosis (74). Conversely, CHI3L1 deficiency has been shown to attenuate microglia-mediated inflammation and inhibit the progression of AD (75,76). Study has shown that the CHI3L1 inhibitor, K284-6111, can suppress NF-κB activation and the expression of related inflammatory factors in AD animal models following intracerebroventricular infusion of Aβ1-42 and in LPS-induced BV-2 microglia cells (69). Furthermore, the anti-neuroinflammatory effects of K284-6111 are also observed in a Tg2576 mouse model and in Aβ-induced BV2 microglia, implicating the extracellular signal-regulated kinases (ERK)-mediated pentraxin 3 and NF-κB pathways (16). DL0410, an acetylcholinesterase (AChE) inhibitor, has been shown to suppress the receptor for advanced glycation end products (RAGE)/NF-κB signaling pathway, resulting in inhibition of D-galactose-induced microglia activation. This results in the downregulation of COX2 and iNOS expression, ultimately suppressing inflammation in the cortex and hippocampus of the brain (70).
Toll-like receptors (TLRs) are essential pattern recognition receptors in the immune and inflammatory responses, with TLR4 being highly expressed on microglia (77). However, excessive activation of TLRs can initiate a cascade of events, leading to activation of NF-κB in the brain, resulting in the synthesis and release of various inflammatory mediators that contribute to neuronal damage (78,79). Therefore, targeting the TLR/NF-κB pathway may prove beneficial in the treatment of AD. Several studies have illustrated that natural compounds can reduce neuroinflammation by inhibiting the TLR4/NF-κB pathway (80-82). One such compound is epigallocatechin-3-gallate (EGCG), a polyphenol found in green tea that has been extensively studied for its neuroprotective effects (83). EGCG is known to suppress the activation of both classical NOD-like receptor thermal protein domain associated protein 3 (NLRP3) inflammasomes and caspase-11-mediated non-classical inflammasomes via the TLR4/NF-κB pathway, thereby effectively exerting its anti-inflammatory properties (80). Genistein (Gen), a compound derived from Soybean isoflavone (SIF) (84), has been shown to improve memory abilities in patients with AD and to attenuate inflammation in Aβ25-35-induced BV-2 microglia through inhibition of the TLR4/NF-κB signaling pathway. These findings suggest that a diet rich in plant-derived Gen may be beneficial in reducing the risk of AD by alleviating inflammation (81). In addition, oxysophoridine extracted from Sophora alopecuroides L. seeds (85,86) was found to downregulate the expression of TNF-α and IL-1β in Aβ-induced BV-2 cells, with therapeutic effects comparable to those of the TLR4 inhibitor TAK-242. These results demonstrate the promising anti-neuroinflammatory properties of oxysophoridine (82). The initial interaction between CD14 and TLR4 is a crucial step in the activation of neuroinflammatory signals induced by LPS (87). A study has identified a novel biphenyl compound, called Protosappanin A (PTA), derived from Caesalpinia sappan L., which effectively inhibits neuroinflammation in vitro (88). PTA achieves this by disrupting the CD14-TLR4 interaction in BV-2 microglia that are stimulated by LPS, thereby inhibiting the NF-κB signaling pathway (88). Similarly, resveratrol, a natural neuroprotectant agent, has been shown to significantly reduce microglia-mediated neuroinflammation (89). Oral administration of resveratrol to APP/PS1 mice significantly reduced the number of activated microglia around amyloid plaques (90). Further in vitro research revealed that resveratrol's mechanism of action involves disruption of TLR4 oligomerization to attenuate the TLR4/NF-κB/STAT signaling pathway, ultimately leading to a reduction in TNF-α and IL-6 production (90).
Upon activation, TLR4 recruits the adaptor myeloid differentiation factor 88 (MyD88), which initiates downstream activation of the transcription factor NF-κB (91). Certain active compounds in some traditional Chinese medicines have been found to interfere with this pathway and exert anti-neuroinflammatory effects. For example, Icariside II (ICS II), an active component of Epimedium, has been shown to have multiple pharmacological activities, including anti-inflammatory, anticancer and anti-aging (92,93). In an LPS-induced SD rat model of neuroinflammation, ICS II demonstrated potent anti-inflammatory effects by reducing the expression of the microglia marker Iba-1 and downregulating related pro-inflammatory cytokine proteins by intervening in the TLR4/MyD88/NF-κB pathway (94). Similarly, DL0410 is a dual inhibitor of both AChE and butyrylcholinesterase with a unique structural scaffold (95). This compound has been shown to improve memory when administered with Aβ1-42 and scopolamine administration (96), as well as cognitive impairment when administered with D-galactose. It holds significant potential as a therapeutic agent for AD by inhibiting the TLR4-mediated/MyD88/NF-κB signaling pathway and reducing pro-inflammatory cytokines (such as TNF, IL-1 and IL-6), while increasing the anti-inflammatory cytokine IL-10 to combat neuroinflammation (97). ATP50-3 is a purified product that is extracted from crude polysaccharides obtained from the traditional Chinese medicine Acorus tatarinowii (98,99). In vitro study has shown that it effectively inhibits the activation of NF-κB and the expression of TLR4, MyD88, phosphorylated (p)-PI3K (phosphoinositide 3-kinase), p-Akt (p-, phosphorylated), and inflammatory mediators in LPS-induced BV2 cells (100). Moreover, its anti-inflammatory efficacy is further enhanced by the TLR4 inhibitor TAK242 and the PI3K inhibitor LY294002, suggesting that its neuroprotective effects against neuroinflammation are due to the regulation of the TLR4/MyD88/NF-κB and PI3K/Akt signaling pathways (100). Another natural compound, dihydromyricetin (DHM) from Ampelopsis grossedentata, has also been found to exhibit promising anti-inflammatory effects (101) and is being considered as a potential treatment for AD. In an LPS-induced inflammation model of BV-2 microglia, DHM was found to downregulate pro-inflammatory cytokine mRNA expression by inhibiting TLR4 and MyD88 expression, and activation of the NF-кB pathway induced by LPS (102). These results strongly suggest that DHM exerts anti-inflammatory effects through inhibition of the TLR4/MyD88/NF-кB signaling pathway (102). GX-50, a compound derived from Sichuan pepper, exhibits promising anti-inflammatory and AD therapeutic effects (103). Research has shown that GX-50 effectively inhibits Aβ-induced TLR4 activation, preventing the recruitment of MyD88 and TNF receptor associated factor 6. This ultimately suppresses the NF-κB and MAPK signaling pathways, demonstrating potent anti-inflammatory activity (104). WD repeat and FYVE domain-containing 1 (WDFY1), a pivotal adaptor molecule in the TLR3/TLR4 signaling pathway, facilitates the recruitment of the downstream molecule TRIF found on intracellular vesicles, leading to a pro-inflammatory effect (105,106). Forsythoside B (FTS-B), a phenylethanoid glycoside derived from Forsythiae fructus, has been found to possess significant anti-inflammatory properties and exhibit neuroprotective benefits in AD (107). In vivo study has revealed that FTS-B can ameliorate cognitive impairment, mitigate pathological changes and decrease the production of pro-inflammatory cytokines in mice with AD (108). Consistent with these findings, FTS-B has been shown to suppress the inflammatory response of LPS-induced BV-2 microglia and hippocampal HT22 cells in vitro by blocking the WDFY1/TLR3/NF-κB signaling pathway (108).
Tanshinone IIA (Tan IIA) is a lipophilic diterpenoid compound derived from Salvia miltiorrhiza Bunge with significant anti-inflammatory and antioxidant properties (109), making it beneficial in attenuating the progression of AD. Research has demonstrated that Tan IIA can effectively intervene in AD mouse models induced by the injection of Aβ1-42 into the hippocampal region. It inhibits the expression of pro-inflammatory cytokines such as IL-1β and IL-6, reduces the number of microglia, lowers levels of complement molecules and improves local brain tissue inflammation (110). Similar findings were observed in AD models induced by Aβ, where Tan IIA was found to downregulate NF-κB p65 levels, thus inhibiting neuroinflammation (111,112). RAGE is implicated in Aβ-induced neuroinflammation and Tan IIA was also found to improve cognitive impairment and neuroinflammation by inhibiting RAGE/NF-κB signaling pathway, which is known to be involved in Aβ-induced neuroinflammation (113). Evidence suggests that Tan IIA provides significant anti-inflammatory benefits, leading to cognitive improvement and neuroprotection in the presence of AD. However, the clinical application of Tan IIA is limited due to its poor water solubility and short half-life (114,115). To address this issue, scientists have discovered that using chitosan as a carrier for loading Tan IIA (CS@TanIIA) can protect Caenorhabditis elegans from AD damage (116). The chitosan coating effectively enhances the protective effect of Tan IIA against AD by increasing its solubility. As a result of this improvement, Tan IIA has great potential for clinical application.
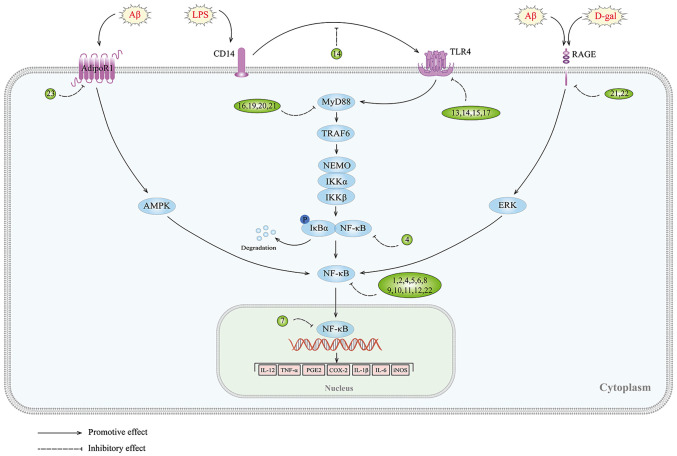
Adiponectin (APN) is an adipokine that is produced by adipocytes that binds to the AdipoR1 and AdipoR2 receptors (117). In aged mice, chronic deficiency of APN has been associated with cognitive impairment and the development of AD-like symptoms (118). It has also been revealed that APN deficiency exacerbates microglia activation and neuroinflammation in APN 5XFAD mice (119). Pre-treatment with APN can inhibit the release of TNFα and IL-1β in AβO-induced BV2 cells by activating the AdipoR1/Adenosine 5′-monophosphate (AMP)-activated protein kinase (AMPK)/NF-κB signaling pathway, thereby ameliorating neuroinflammation (120). This research highlights the potential therapeutic benefits of APN in the prevention and treatment of AD (Fig. 1 and Table I).
Figure 1.
NF-κB signaling pathway and targets of inhibitors against neuroinflammation in AD. 1, rutin; 2, piperlongumine; 3, bee venom; 4, punicalagin; 5, tenuifolin; 6, D4 (a novel piperine derivative); 7, ethanol extract of Bupleurum falcatum; 8, macasiamenene F; 9, miconazole; 10, LD55; 11, the p65 binding domain of glucocorticoid-induced leucine zipper; 12, K284-6111; 13, epigallocatechin-3-gallate; 14, genistein; 15, oxysophoridine; 16, dihydromyricetin; 17, Gx-50; 18, protosappanin A; 19, icariside II; 20, ATP50-3; 21, DL0410; 22, tanshinone IIA; 23, APN; Aβ, amyloid β; LPS, lipopolysaccharide; D-gal, D-galactose; RAGE, receptor for advanced glycation end products; TLRs, Toll-like receptors; AMPK, adenosine 5′-monophosphate-activated protein kinase; MyD88, myeloid differentiation factor 88; TRAF6, TNF receptor associated factor 6; NEMO, NF-κB essential modulator; IKK, IκB kinase.
Table I.
Drugs that prevent and treat Alzheimer's disease through the NF-κB signaling pathway.
| First author, year | Compound and original source | In vivo model | In vitro model | Dose and drug administration time | Targets | Signaling pathways | (Refs.) |
|---|---|---|---|---|---|---|---|
| Sun et al, 2021 | Rutin/- | Tau-P301S mice | Tau oligomers-induced primary microglia |
In vivo: 100 mg/kg-30 days In vitro: 8 μM-24 h |
↓: Tau aggregation, tau-mediated cytotoxicity, IL-1β, TNF-α, tau oligomer-induced toxicity, tau pathology, GFAP, Iba-1, IKK-β, p-P65/P65, synapse loss, microglial synapse engulfment ↑: microglial engulfment of extracellular tau, PP2A |
NF-κB | (51) |
| Gu et al, 2018 | PL/Piper longum | LPS-induced ICR mice | LPS-induced BV2 cells |
In vivo: 1.5,3 mg/kg; 7 days In vitro: 0.5, 1, 2.5 μM; 18 h |
↓: Aβ1-42, activities of β-secretase and γ-secretase, APP, BACE1, COX-2, iNOS, GFAP, Iba-1, NF-κB translocation, phosphorylated-IκB, TNF-α, IL-1β, IL-6 ↑: neuronal survival |
NF-κB | (53) |
| Gu et al, 2015 | BV/Bee | LPS-induced ICR mice | LPS-induced BV2 cells |
In vivo: 0.8, 1.6 μg/kg; 7 days In vitro: 0.5, 1, 2 μg/ml; 48 h |
↓: Aβ1-42, β-secretase and γ-secretase, APP, BACE1, COX-2, iNOS, GFAP, Iba-1, neuronal death, NF-κB translocation, p-IκB | NF-κB | (55) |
| Kim et al, 2017 | PUN/pomegranate | LPS-induced ICR mice | LPS-induced BV2 cells |
In vivo: 1.5 mg/kg; 7 days In vitro: 10, 20, 50 μM; 24 h |
↓: Aβ1-42, BACE1, GFAP, Iba-1, TNF-α, IL-1β, IL-6, MDA, ROS (H2O2), COX-2, iNOS, NF-κB translocation, p-IκB, NF-κB DNA binding activity ↑: GSH/GSSG |
NF-κB | (57) |
| Chen et al, 2020 | TEN/Polygala tenuifolia Willd | - | Aβ42-induced BV2 cells | In vitro: 1, 5, 10 μM; 24 h | ↓: TNF-α, IL-1β, IL-6, COX-2, iNOS, NF-κB translocation | NF-κB | (58) |
| Shahbazi et al, D 2020 | 4/black and white pepper | - | LPS-induced human microglia clone 3 | In vitro: 0.86 μM; 24 h | ↓: NO, iNOS, TNF-α, IL-1β, PPAR-γ, IKK-α, IkB-α, NF-kB p65 | NF-κB | (61) |
| Park et al, 2015 | BFE/BF | LPS-induced C57BL/6 mice | LPS-induced BV2 cells |
In vivo: 30 mg/kg; 3 days In vitro 10 μg/ml; 4 h |
↓: NO, iNOS, TNF-α, IL-1β, IL-6, NF-κB p65/RELA, GFAP, Iba-1 | NF-κB | (63) |
| Leláková et al, 2020 | MF/Macaranga siamensis | - - |
LPS-induced THP-1 and THP-1-XBlue™ -MD2-CD14 human monocytes, BV2 mouse microglia, and an ex vivo model of brain-sorted mouse microglia | In vitro: 1 μmol/l; 18 h | ↓: TNF-α, IL-1β, NF-кB, AP-1, degradation of IкBα | NF-κB | (64) |
| Yeo et al, 2020 | miconazole (MCZ)/- | LPS-induced C57BL 6/N mice Aβ1–42-induced mice with AD | LPS-induced BV2 cells |
In vivo: 40 mg/kg; 7/14 days In vitro: 1.25, 2.5, 5, 10 μM; 24 h |
↓: TNF-α, IL-1β, IL-6, COX-2, iNOS, GFAP, Iba-1, NO, p-IκB, NF-κB translocation | NF-κB | (66) |
| Solberg et al, 2014 | LD55/- | AβPP/PS-1 transgenic mice with AD | - | In vitro: a diet containing 100 ppm LD55; 12 months | ↓: Aβ plaques, activated microglia | NF-κB | (67) |
| Lindsay et al, 2021 | GA/GILZ | 5XFAD mice | - | In vivo: 100 μl GA; alternate days for 6 weeks | ↓: Aβ plaque burden, NF-κB p65, IL-1β, IL-12, IL-6, IFN-γ, GFAP, Iba-1, CD14, TLR-2, TLR-4 | NF-κB | (68) |
| Choi et al, 2018 | K284-6111/- | Aβ1-42-induced mice with AD | LPS-induced BV2 cells |
In vivo: 3 mg/kg; 4 weeks In vitro: 0.5, 1, 2 μM; 24 h |
↓: CHI3L1, iNOS, GFAP, Iba-1, TNF-α, IL-1β, IL-6, Aβ1-42, APP, BACE1, C99, p-IκB, NF-κB translocation | inactivation of NF-κB-mediated CHI3L1 | (69) |
| Ham et al, 2020 | Tg2576 mice | Aβ-induced BV2 cells |
In vivo: 3 mg/kg; 4 weeks In vitro: 0.5, 1, 2 μM; 24 h |
↓: Aβ1-42, Aβ1-40, APP, BACE1, β-secretase, COX-2, iNOS, GFAP, Iba-1, Cd86, p-IκBα, p-ERK1/2, p-JNK, CHI3L1, PTX3 | ERK-mediated PTX3 and NF-κB | (16) | |
| Lian et al, 2017 | DL0410/- | D-gal-induced ICR mice | - | In vivo: 1, 3, 10 mg/kg; 4 weeks | ↓: AChE activity, AGEs, MDA, mitochondria structure, Iba-1, GFAP, RAGE, p-P65, COX2, iNOS, p-JNK, cleaved caspase 3, cleaved PARP ↑: ACh level, TEACl, activities of catalase, GPx, SOD, OPR, the number of synapses |
RAGE/NF-κB | (70) |
| Zhong et al, 2019 | EGCG/green tea | APP/PS1 double transgenic mice with AD | LPS-induced BV2 cells Aβ1-42-induced primary microglia |
In vivo: 2 mg/kg; 4 weeks In vitro: 10 μM; 1 h |
↓: caspase-1 p20, NLRP3, caspase-11 p26, TLR4, p-IKK/IKK, p-NF-κB/NF-κB, Iba-1, IL-1β, IL-18 | TLR4/NF-κB | (80) |
| Zhou et al, 2014 | Gen/SIF | - | Aβ25–35-induced BV2 cells | In vitro: 12.5, 25, 50, 100, 200 μM; 26 h | ↓: IL-1β, iNOS, TLR4, NF-κB p65, NF-κB p50, DNA-binding activity of NF-κB ↑: cell viability, IL-10 |
TLR4/NF-κB | (81) |
| Chen et al, 2021 | Oxysophoridine/Sophora alopecuroides L. seeds | - | Aβ1-42-induced BV2 cells | In vitro: 0, 2.5, 5, 10, 20, 40 μM; 48 h | ↓: MDA, TNF-α, IL-1β, TLR4, MyD88, NF-κB p65 ↑: activities of GPx, CAT, and SOD |
TLR4/NF-κB | (82) |
| Zeng et al, 2012 | PTA/Caesalpinia sappan L. | - | LPS-induced BV2 cells | In vitro: 5, 10, 25, 50 μM; 10 min | ↓: Total ROS, gp91 phox, MDA, iNOS, NO, Nitrotyrosine, Iba-1, p-NF-κB p65 on serine-536, p65 and p50 translocations, IKKα/β, p-IκB, the interaction of TLR4 with MyD88, IRAK1 and TRAF6, interaction of LPS with TLR4 ↑: synapse remodeling |
CD14/TL R4-dependent NF-κB | (88) |
| Capiralla et al, 2012 | Resveratrol/red wines | APP/PS1 double transgenic mice with AD | LPS-induced BV2 cells |
In vivo: 350 mg/kg; 15 weeks In vitro: 100 mM; 30 min |
↓: IL-6, M-CSF, MCP-1, MCP-5, CD54, IL-1ra, IL-27, TNF-α, p-Akt, COX-2, iNOS, STAT1, STAT3, TLR4, Iba-1 | TLR4/NF-κB/STAT | (90) |
| Zhou et al, 2019 | ICS II/Epimedium brevicornum Maxim | LPS-induced SD rats | - | In vivo: 3, 10 mg/kg; 7 days | ↓: neuronal changes, neuronal degeneration, GFAP, Iba-1, COX-2, IL-1β, TNF-α, TLR4, MyD88, TRAF6, p-NF-κB ↑: IκB-α degradation |
TLR4/MyD88/NF-κB | (94) |
| Zhang et al, 2021 | DL0410/- | D-gal-induced SD rats | LPS-induced BV2 cells |
In vivo: 1,3, 10 mg/kg-8 weeks In vitro: 1-30 μM-2 h |
↓: MDA,AGEs, SOD1, SOD2, Iba-1, GFAP, TNF-α, IL-1β, IL-6, COX2, iNOS, TLR, MyD88, p-IκBα and NF-κB p65, NF-κB translocationp65, NO, TRAF6, p-IKKα/β, p-IκBα ↑: PSD95, IL-10, claudin-1, claudin-5, occludin, CX43, ZO-1 |
TLR4/MyD88/NF-κB | (97) |
| Zhong et al, 2020 | ATP50-3/Acorus tatarinowii | - | LPS-induced BV2 cells | In vitro: 2.5,5, 10 μM-2 h | ↓: TNF-α, IL-1β, IL-6, COX-2, iNOS, CD11b, TLR4, MyD88, IKKα/β, IκBα, NF-κB p65, PI3K, Akt | TLR4-mediated MyD88/NF-κB and PI3K/Akt | (100) |
| Jing et al, 2019 | DHM/Ampelopsis grossedentata | - | LPS-induced BV2 cells | In vitro: 20, 40, 80, 100 mg/l; 48 h | ↓: TNF-α, IL-1β, IL-6, COX-2, iNOS, p-p65, p-IкBα, TLR4, MyD88 ↑: BV-2 microglia viability |
TLR4/MyD88/NF-κB | (102) |
| Shi et al, 2016 | Gx-50/Sichuan pepper | APP-Tg mice | Aβ42-induced BV2 cells and primary microglia |
In vivo: 1 mg/kg; 2 months In vitro: 1 μM; 30 min |
↓: TNF-α, IL-1β, NO, PGE2, iNOS, COX2, p-IκB, NF-κB translocation, p-ERK1/2, p-p38, p-JNK, TLR4, MyD88, TRAF6 | TLR4-mediated NF-κB and MAPK | (104) |
| Kong et al, 2020 | FTS-B/Forsythiae fructus | APP/PS1 double transgenic mice with AD | LPS-induced BV2 cells |
In vivo: 10, 40 mg/kg; 36 days In vitro: 1, 2.5 μM; 3 h |
↓: Aβ deposition, JIP3, p-JNK/JNK, p-APP/APP, Aβ, TNF-α, IL-1β, IL-6, IL-8, IL-12, ELKS, p-IKK (α+β), p-IκBα, p-NF-κB (Ser536), Iba1, GFAP, NO, iNOS, apoptosis rate of the HT22 cells ↑: TLR3, p-IRF3/IRF3, IFN-β, WDFY1, p-IRF3, cell viability |
WDFY1/TLR3/NF-κB | (108) |
| Lu et al, 2016 | Tan IIA/Salvia miltiorrhiza | Aβ1-42-induced AD rats | - | In vivo: 8 mg/kg; 30 days | ↓: A β, IL-1β, IL-6, GFAP, CD11b, C1q, C3c, C3d | - | (110) |
| Li et al, 2015 | Aβ-induced AD rats | - | In vivo: 50 mg/kg; 15 days | ↓: iNOS, MMP-2, NF-κB p65 | NF-κB | (351) | |
| Maione et al, 2018 | Aβ1-42-induced mice with AD | - | In vivo: 1, 3, 10 mg/kg; 21 days | ↓: GFAP, S100β, COX-2, NF-κB p65 | NF-κB | (112) | |
| Ding et al, 2020 | APP/PS1 double transgenic mice with AD | Aβ1-42-induced BV2 cells |
In vivo: 5, 20 mg/kg; 30 days In vitro: 1, 10 μM; 30 min |
↓: Loss of Syn and PSD-95, Aβ1-40, Aβ1-42, the number of activated microglia, Iba-1, GFAP, TNF-α, IL-6, IL-1β, expression of RAGE, p-IκBα, NF-κB p65 | RAGE/NF-κB | (113) | |
| Jian et al, 2019 | APN/- | 5XFAD mice APN-/-5XFAD mice | AβO-induced BV2 cells | In vitro: 10 μg/ml-2 h | ↓: TNF-α, IL-1β, p-NF-κB p65S536, NF-κB p65, Aβ plaques ↑: p-AMPKT172, GFAP, Iba1 |
AdipoR1-AMPK-NF-κB | (120) |
PL, piperlongumine; BV, bee venom; PUN, punicalagin; TEN, Tenuifolin; D4, a novel piperine derivative; BFE, ethanol extract of BF; BF, Bupleurum falcatum L.; MF, macasiamenene F; MCZ, miconazole; GA, the p65 binding domain of GILZ; GILZ, glucocorticoid induced leucine zipper; K284-6111, 2-({3-[2-(1-cyclohexen-1-yl)ethyl]-6,7-dimethoxy-4-oxo-3,4-dihydro-2-quinazolinyl}sulfanyl)-N-(4-ethylphenyl) butanamide; EGCG, epigallocatechin-3-gallate; Gen, genistein; SIF, Soybean isoflavone; PTA, protosappanin A; ICS II, icariside II; DHM, dihydromyricetin; FTS-B, forsythoside B; Tan IIA, tanshinone IIA; APN, Adiponectin; p- phosphorylated; AD, Alzheimer's disease; Aβ, amyloid β; APP/PS1, amyloid-β protein/presenilin-1; 5XFAD, 5X familial Alzheimer's disease; AChE, acetylcholinesterase; APN, Adiponectin; AMPK, Adenosine 5′-monophosphate (AMP)-activated protein kinase; BACE1, β-site amyloid precursor protein cleaving enzyme 1; COX-2, cyclooxygenase-2; CHI3L1, chitinase-3 like-protein-1; ERK, extracellular signal-regulated kinases; GFAP, glial fibrillary acidic protein; IKK, IκB kinase; IL, interleukin; iNOS, inducible nitric oxide synthase; IFN-γ, interferon-γ; JNK, c-Jun NH2-terminal kinases; LPS, lipopolysaccha- ride; MyD88, myeloid differentiation factor 88; MAPK, mitogen-activated protein kinase; NLRP3, NOD-like receptor thermal protein domain associated protein 3; NO, nitric oxide; PTA, protosappanin A; PI3K, phosphoinositide 3-kinase; PPARs, peroxisome proliferator-activated receptors; RAGE, receptor for advanced glycation end products; ROS, reactive oxygen species; TLRs, Toll-like receptors; WDFY1, WD repeat and FYVE domain-containing 1; NF-κB, nuclear factor-kappa-B; Iba-1, ionized calcium binding adaptor molecule 1; STAT3, signal transducers and activators of transcription 3; AP-1, activator protein 1; PGE2, prostaglandin E2.
Mitogen-activated protein kinase (MAPK)
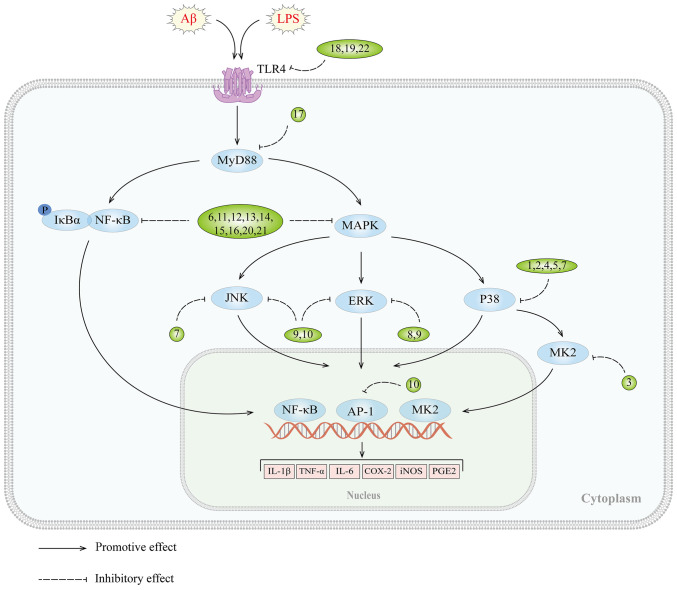
It is widely recognized that MAPKs, which include p38 MAPK, ERK, and c-Jun NH2-terminal kinases (JNK), as well as their isoforms (121), play a critical role in the regulation of various biological processes, including proliferation, differentiation, apoptosis and inflammation in mammalian cells (122). The MAPK signaling cascade comprises a MAPKK kinase, a MAPK kinase, and a MAP kinase (123) that respond to both internal and external stimuli, such as growth factors, cytokines, oxidation, and endoplasmic reticulum stress. Activation of the MAPK signaling pathway has been observed in the brains of patients with AD (124,125) and animal models (126). In vitro studies have shown that stimulation of Aβ induces the activation of this pathway in glial cell cultures, indicating its involvement in the development of AD (127-129). Inhibition of tau kinases, such as p38 MAPK, has been shown to improve cognitive deficits and reduce tau pathology in AD (130). Furthermore, blocking the ERK pathway can reverse mitochondrial dysfunction in AD (131,132), while specific JNK inhibitors can enhance synaptic function (133). Of note, the MAPK signaling pathway can also regulate the neuroinflammatory response of microglia. Aβ-induced production of inflammatory cytokines and ROS can activate this pathway, leading to more severe inflammation. A number of in vitro experiments have demonstrated that inhibition of the MAPK signaling pathway can suppress neuroinflammation in BV2 microglia (134,135), highlighting its potential as an effective strategy for treating AD (Fig. 2 and Table II).
Figure 2.
MAPK signaling pathway and targets of inhibitors against neuroinflammation in AD. AD, Alzheimer's disease; 1, MW01-2-069A-SRM; 2, MW181; 3, MMI-0100; 4, methanol extracts of Piper sarmentosum roots; 5, EGB761; 6, cryptolepine; 7, VB-037; 8, dexmedetomidine; 9, Hominis placenta; 10. BJe; 11, diammonium glycyrrhizinate; 12, tripterygium glycosides; 13, LX007; 14, pseudane-VII; 15, sorbinil and zopolrestat; 16, Artemisiae Iwayomogii Herba; 17, tectorigenin; 18, circumdatin D; 19, 1-O-acetylbritannilactone; 20, Ganoderma lucidum extract GLE; 21, Atractylodis Rhizoma Alba ethanolic extract; 22, ulmoidol; Aβ, amyloid β; LPS, lipopolysaccharide; TLRs, Toll-like receptors; MyD88, myeloid differentiation factor 88; IKK, IκB kinase; JNK, c-Jun NH2-terminal kinases; MK2, MAPK-activated protein kinase II; AP-1, activator protein 1.
Table II.
Drugs that prevent and treat Alzheimer's disease through the MAPK signaling pathway.
| First author, year | Compound and original source | In vivo model | In vitro model | Dose and drug administration time | Targets | Signaling pathways | (Refs.) |
|---|---|---|---|---|---|---|---|
| Munoz et al, 2007 | MW01-2-069A-SRM/- | Aβ1-42-induced mice with AD | - | In vivo: 2.5 mg/kg; two weeks | ↓: IL-1β, TNFα, S100B | p38 MAPK | (140) |
| Maphis et al, 2016 | MW181/- | hTau mice LPS-induced MK2-/-mice | Cx3cr1−/−CM-induced primary neurons and microglia |
In vivo: 1 mg/kg; 14 days In vitro: 2 μM; 30 min |
↓: p-tau, p-p38α MAPK (T180/Y182), tau (AT8 site), p-pATF2(T71), pATF2, pMK2, IFNγ, IL-1β, IL-6, TNFα, p38α MAPK ↑:synaptophysin, YM1,ARG1 |
p38 MAPK | (141) |
| Alam et al, 2015 | VX-745/- | Tg2576 mice | - | In vivo: 0.5, 1.5, 4.5 mg/kg; 2 weeks | ↓: amyloid plaque, IL-1β ↑: PSD95 |
p38 MAPK | (143) |
| Jiang et al, 2019 | MMI-0100/- | Aβ1-42-induced mice with AD | LPS-induced BV2 cells |
In vivo: 2 μl; the lateral ventricle; 0.5 μl/CA1 side; 15 min 25 nmol; 10 μl; intranasal infusion; 1 min In vitro:10−5-10−8 M; 24 h |
↓: CD11b, GFAP, IL-6, IL-1β, TNF-α, iNOS, p-MK2 | p38 MAPK/MK2 | (147) |
| Chan et al, 2019 | RMEOH/PS | - | Aβ-induced BV2 cells | In vitro: 6.25 μg/ml; 4 h | ↓: IL-1β, IL-6, TNF-α, NO, p38α MAPK | p38 MAPK | (151) |
| Meng et al, 2019 | EGB 761/Ginkgo biloba | - | Aβ1-42-induced BV2 cells | In vitro: 10, 90 μg/ml; 12 h | ↓: NF-κB translocation, IL-1β, TNF-α, p38 MAPK | p38 MAPK | (153) |
| Olajide et al, 2013 | Cryptolepine/Cryptolepis sanguinolenta | - | LPS-induced primary microglia and BV2 cells | In vitro: 2.5, 5 μM; 30 min | ↓: TNF-α, IL-6, IL-1β, PGE2, COX2, mPGES-1, iNOS, NO, p-p38 MAPK, MAPKAPK2, NF-κB p65 translocation | NF-κB and p38 MAPK | (157) |
| Chiu et al, 2019 | VB-037/quinoline compounds | - | LPS/IFN-γ-induced BV2 cells Aβ-GFP SH-SY5Y cells | In vitro: 10 μM; 8 h | ↓: Aβ aggregation, ROS, NO, Iba1, AChE, caspase1, IL-1β, JNK, Jun proto-oncogene, AP-1 transcription, JUN ↑: HSP27, cell viability |
P38, JNK | (155) |
| Ho et al, 2020 | AZD6244/- | - | acrolein-induced BV2 cells | In vitro: 10 μM; 16 h | ↓: p-ERK, TNF-α, COX-II, HO-1 | MEK-ERK | (163) |
| Qiu et al, 2020 | dexmedetomidine/- | - | LPS-induced BV2 cells | In vitro: 1, 5, 10 μM; 0, 6, 12, 24 h | ↓: NO, morphological changes in BV2 cells, TNF-α, iNOS, p-ERK1/2 ↑: IL-10, CD206, microglial M2 polarization |
ERK | (164) |
| Lee et al, 2013 | HP/placenta | - | LPS-induced BV2 cells | In vitro: 50 μM; 2 h | ↓: p-JNK, p-ERK, p-AKT, iNOS, NO, COX2 | JNK and ERK | (166) |
| Currò et al, 2016 | BJe/Bergamot juice | - | Aβ1–42-induced THP-1 cells | In vitro: 0.05, 0.1 mg/ml; 16 h | ↓: IL-6, IL-1β, p-p54, ERK 1/2, p46 JNK, AP-1 DNA binding activity | MAPK/AP-1 | (168) |
| Tang et al, 2021 | TGs/- | Aβ25-35-induced mice with AD | Aβ25-35-induced PC12 cells |
In vivo: 0.25 mg/10 g.d; 28 days In vitro: 25 μg/l; 24 h |
↓: Aβ25-35, p-Tau, CD11b, p-IκBα, p-P38, caspase-1, COX2, iNOS, IL-1β, TNF-α, NO ↑: The neuron number |
NF-κB and MAPK | (172) |
| Cao et al, 2018 | LX007/- | - | LPS-induced primary microglia | In vitro: 10, 20, 30 μM; 1 h | ↓: NO, iNOS, PGE2, COX-2, IL-1β, IL-6, TNF-α, p-ERK1/2, JNK, p38, p-IκBα, IκBα degradation, p65 | NF-κB and MAPK | (173) |
| Kim et al, 2018 | Pseudane-VII/Pseudoalteromonas sp.M2 | - | LPS-induced BV2 cells | In vitro: 0.5, 1, 2.5, 5 μM; 2 h | ↓: iNOS, COX-2, IL-1β, p-p65, ERK, p38 MAPK, JNK1/2, Iba-1 | NF-κB and MAPK | (174) |
| Zhao et al, 2013 | DG/GA | Aβ1-42-induced mice with AD | Aβ1-42-induced BV2 cells |
In vivo: 10 mg/kg; 14 days In vitro: 0.001 mg/ml; 1 h |
↓: TNF-α, COX-2, iNOS, IL-1β, COX-2, iNOS, GFAP, Iba-1, p65 translocation, p-ERK, JNK, p38 ↑: IL-10 |
MAPK and NF-κB | (175) |
| Song et al, 2017 | Sor and Zol/- | - | Aβ1-42-induced BV2 cells | In vitro: 20 μM; 1 h | ↓: TNF-α, IL-1β, IL-6, NF-κB p65 translocation, p-IKKβ, p-IκB, p-NF-κB, p-JNK, p-p38, p-ERK, ROS, p-PKCα/β, PKCδ, PKCζ/λ, PKCmu subtypes | ROS/PKC-dependent NF-κB and MAPK | (177) |
| Ju et al, 2021 | AIH/- | LPS-induced C57BL 6J mice | LPS-induced BV2 cells |
In vivo: 10, 30, 100 mg/kg; 14 days In vitro: 1, 10, 100 μg/ml; 24 h |
↓: NO, iNOS, COX2, TNF-α, IL-6, p-p65, p-p38, p-JNK, NLRP3, Iba-1 | NF-κB and MAPK | (180) |
| Hilliard et al, 2020 | GLE/Ganoderma lucidum | - | LPS-induced BV2 cells | In vitro: 0.5 mg/ml; 1 h | ↓: G-CSF, IL1α, MCP-5, MIP3, RANTES, CHUK, NFκB1/p50, IKBKE | NF-κB and MAPK | (181) |
| Jeong et al, 2019 | ARAE/ARA | - | LPS-induced BV2 cells | In vitro: 10, 50, 100 μg/ml; 18 h | ↓: NO, TNF-α, IL-6, IL-1β, iNOS, COX-2, HO-1, NF-κB p65 translocation, p-IκBα, IκBα degradation, p-ERK, p-p38, p-JNK | NF-κB and MAPK | (134) |
| Tang et al, 2021 | ABL/Inula britannica L. | - | LPS-induced BV2 cells | In vitro: 1, 3, 10 μM; 24 h | ↓: NO, TNF-α, PGE2, iNOS, CD14, NF-κB p65, IRAK1, p-p38 ↑: HO-1, Arg-1, IL-10 |
TLR4-mediated NF-κB and MAPK | (185) |
| Han et al, 2021 | ULM/Eucommia ulmoides Oliv | - | LPS-induced BV2 cells | In vitro: 1, 3, 10 μM; 24 h | ↓: TNF-α, IL-1β, IL-6, PGE2, COX-2, iNOS, p-IκBα, p-p38, p-ERK, p-JNK, CD14, TLR4, MD2, MyD88, TRAF6, TAK1 | TLR4-mediated NF-κB and MAPK | (188) |
| Zhang et al, 2020 | Circumdatin D/Aspergillus ochraceus | C4176 nematodes | LPS-induced BV2 cells and primary microglia |
In vivo: 50, 100, 200 μM; 16 h In vitro: 10, 20, 50 μM; 6 h |
↓: NO, AChE, TNF-α, IL-1β, iNOS, COX-2, neuron death, TLR4, MyD88, NF-κB p65, p-IKK, p-IκB, p-MAPK, p-STAT3, STAT3 translocation, p-JAK2 | TLR4-mediated NF-κB, MAPK andJAK/STAT | (189) |
| Lim et al, 2018 | TEC/Pueraria thunbergiana Benth | LPS-induced ICR mice | LPS-induced BV2 cells |
In vivo: 5, 10 mg/kg; 5 days In vitro: 12.5, 25, 50, 100 μM; 24 h |
↓: NO, PGE2, iNOS, COX-2, TNF-α, IL-6, NF-κB p65, p-ERK, p-JNK, Iba-1, TLR4, MyD88 | TLR4-MyD88-mediated inhibition of ERK/JNK and NF-κB | (192) |
RMEOH, methanol extracts of PS roots; PS, Piper sarmentosum Roxb; HP, Hominis placenta; TGs, Tripterygium glycosides; LX007, 4-[(5-bromo-3-chloro-2-hydroxybenzyl) amino]-2-hydroxybenzoic acid; DG, diammonium glycyrrhizinate; GA, glycyrrhizin acid; Sor, sorbinil; Zol, zopolrestat; AIH, Artemisiae Iwayomogii Herba; GLE, Ganoderma lucidum extract; ARAE, ARA ethanolic extract; ARA, Atractylodis Rhizoma Alba; ABL, 1-O-acetylbritannilactone; ULM, ulmoidol; TEC, tectorigenin; AD, Alzheimer's disease; Aβ, amyloid β; AChE, acetylcholinesterase; COX-2, cyclooxygenase-2; ERK, extracellular signal-regulated kinases; GFAP, glial fibrillary acidic protein; HO-1, Haem oxygenase-1; IKK, IκB kinase; IL, interleukin; iNOS, inducible nitric oxide synthase; IFN-γ, interferon-γ; JNK, c-Jun NH2-terminal kinases; JAK2, Janus kinase 2; LPS, lipopolysaccharide; MyD88, myeloid differentiation factor 88; MAPK, mitogen-activated protein kinase; MAPKKK, MAPKK kinase; MK2, MAPK-activated protein kinase II; MEK, mitogen-activated extracellular signal-regulated kinase; NLRP3, NOD-like receptor thermal protein domain associated protein 3; NO, nitric oxide; PKC, protein kinase C; PKA, protein kinase A; ROS, reactive oxygen species; TLRs, Toll-like receptors; p-, phosphorylated; NF-κB, nuclear factor-kappa-B; Iba-1, ionized calcium binding adaptor molecule 1; STAT3, signal transducers and activators of transcription 3; AP-1, activator protein 1; PGE2, prostaglandin E2.
P38 MAPK
P38, a member of the P38 MAPK subfamily, has been found to be activated in both AD brain tissue samples (136) and animal models (126) of AD. Additionally, study has shown that the absence of P38 MAPK attenuates amyloid-like pathology in AD models (137). Specifically, P38α MAPK is thought to play a crucial role in the dysregulation of microglia and neuroinflammation during AD progression, making it a recognized target for AD treatment (130,138,139). Thus, targeting P38α MAPK may offer a promising therapeutic strategy to address the underlying neuroinflammatory processes in AD.
Several inhibitors of the p38α MAPK signaling pathway, including natural product extracts, and organic compounds, have shown promise in reducing neuroinflammation and treating AD. Both preclinical and clinical trials have evaluated the pharmacological effects of these inhibitors in the brain. Selective p38α MAPK inhibitors, such as MW01-2-069A-SRM (140) and MW181 (141), which are able to penetrate the BBB, have demonstrated potent inhibitory effects on neuroinflammation. Additionally, VX-745, a small molecule inhibitor of p38α MAPK, has emerged as a promising candidate for anti-inflammatory therapy and is currently undergoing pilot trials for the treatment of rheumatoid arthritis (142). Notably, preclinical studies have revealed that VX-745 exerts its anti-neuroinflammatory effects by selectively targeting of p38α MAPK, resulting in a reduction of IL-1β levels in the hippocampus of aged rats (143). This finding highlights the potential utility of VX-745 as a therapeutic strategy for neurological disorders characterized by neuroinflammation (143). MAPK-activated protein kinase II (MK2), a downstream kinase of p38 MAPK (144), is activated and upregulated in AD mouse models and is associated with Aβ deposition, microglia activation, and the upregulation of pro-inflammatory cytokines (145). Targeting MK2 may be a promising therapeutic strategy for AD. MMI-0100, a cell-penetrating peptide inhibitor of MK2 with anti-inflammatory activity (146), has been shown to inhibit LPS-induced microglia activation and significantly reduce pro-inflammatory cytokine production in mice by inhibiting MK2 phosphorylation (147). Furthermore, intranasal administration of MMI-0100 can overcome the challenge of failed AD treatments with large molecule protein or peptide drugs due to its ability to penetrate the BBB (148,149).
Piper sarmentosum Roxb
(PS) is a medicinal plant (150) that has been the subject of recent research investigating potential therapeutic applications in neuroinflammatory diseases. In vitro experiments have demonstrated that pre-treatment of BV2 microglia with methanol extracts of PS roots results in a significant reduction in Aβ-induced expression of proinflammatory cytokine mRNA and protein, thereby exerting neuroprotective effects, which is associated with the regulation of phosphorylation of p38α MAPK in microglia (151). These findings suggest that PS represents a promising option for the management of neuroinflammatory conditions. Derived from Ginkgo biloba leaves, EGb761 has been extensively studied for its potential in ameliorating cognitive impairment and AD (152). In a cellular model of BV-2 microglia stimulated by Aβ1-42, EGb761 intervention effectively attenuated the concentration-dependent production of TNF-α and IL-1β and simultaneously downregulated their respective mRNA expressions (153). Additionally, the inhibition of p38 MAPK phosphorylation induced by Aβ was found to be selectively achieved by EGb761, while it had no significant impact on the expression of ERK and JNK. These results suggest that the anti-inflammatory mechanism of EGb761 may be due to the selective modulation of the p38 MAPK signaling pathway (153).
Quinoline, a heterocyclic aromatic organic compound, has attracted considerable attention for its antibacterial properties (154) and its ability to inhibit amyloid aggregation (155,156). Consequently, this framework is widely utilized in the research and design of innovative anti-inflammatory drugs. Cryptolepine, an indoloquinoline alkaloid isolated from Cryptolepis sanguinolenta, has demonstrated the ability to suppress LPS-induced microglia inflammation by selectively targeting the NF-κB and p38 MAPK signaling pathways (157). Similarly, VB-037 (155), a quinoline compound, has been shown to effectively mitigate BV-2 microglial activation induced by LPS/interferon-γ (IFN-γ). This attenuation is achieved by inhibiting caspase 1 activation, IL-1β expression and P38 phosphorylation, as well as by affecting the JNK, Jun oncogene and Jun signaling pathways. These findings substantiate that VB-037 selectively regulates the P38 and JNK/MAPK signaling pathways, ameliorating neuronal damage and neuroinflammation and thereby altering the progression of AD. The multifaceted mechanism of quinoline derivatives offers several opportunities for the development of AD therapeutics (155,157).
ERK and JNK
The ERK/MAPK pathway and the JNK/MAPK pathway (155), which utilize ERK and JNK as their final kinases, respectively, are essential subsets of the MAPK signaling cascade that regulate neuronal development (158,159). However, dysregulation of these pathways can lead to developmental abnormalities and behavioral deficits (158,160). Notably, chronic activation of these pathways has been observed in the hippocampus of transgenic AD mouse models overexpressing Aβ (161) and in patients with AD, where elevated brain levels of p-ERK have been positively correlated with disease progression (162). This underscores its importance in AD pathogenesis and supports the idea that drugs targeting ERK represent a promising therapeutic approach for managing AD.
Numerous inhibitors of the MEK (mitogen-activated extracellular signal-regulated kinase)/ERK signaling pathway, both natural product extracts, and organic compounds, have displayed potential in reducing neuroinflammation and treating AD. Recent study has highlighted the efficacy of AZD6244 (163), an oral MEK1/2 inhibitor, in suppressing acrolein-induced neuroinflammation by modulating of the MEK/ERK signaling pathway in BV-2 cells, leading to its neuroprotective effects (163). Similarly, Dexmedetomidine (164), an α2 adrenergic receptor agonist with sedative, analgesic and anxiolytic properties, was found to upregulate anti-inflammatory cytokines and M2 phenotype markers, while downregulating pro-inflammatory cytokines, M1 phenotype markers, and p-ERK1/2 in LPS-stimulated BV2 microglia. This effect has been shown to be reversed by LM22B-10, an ERK agonist, supporting the notion that Dexmedetomidine promotes M2 polarization in microglia through modulation of the ERK signaling pathway, ultimately exerting its anti-inflammatory properties (164). Hominis placenta (HP) is a dried placental extract from pregnant women after delivery that has been shown to promote neural regeneration (165). Lee et al (166) demonstrated that pre-treatment with HP significantly inhibited the expression of iNOS and COX2 in LPS-induced BV2 cells. This anti-inflammatory effect was achieved, at least in part, through the inhibition of the ERK pathway and the phosphorylation of JNK and ERK. In addition, Bergamot juice (BJ) was found to have antibacterial properties and to exert anti-inflammatory effects (167) through its flavonoid component (BJe) (168), which was shown to partly affect the ERK signaling pathway. The critical role of monocytic cells in neuroinflammation has been underlined by their ability to cross the BBB and differentiate into microglia in the brain parenchyma (169,170). In this context, a research team found that pretreatment with BJe resulted in a concentration-dependent reduction in the upregulation of pro-inflammatory cytokine expression and a decrease in the phosphorylation levels of JNK and ERK1/2 in Aβ1-42-induced THP-1 monocytic cells. This effect was associated with the disruption of DNA-binding activity of AP-1 (activator protein 1) and the MAPK/AP-1 pathway, thereby counteracting the pro-inflammatory activation of monocytic/microglia induced by Aβ and exerting an anti-neuroinflammatory effect (168).
NF-κB and MAPK
The NF-κB and MAPK signaling pathways have emerged as key regulators of pro-inflammatory mediator expression and NLRP3 inflammasome formation, both of which play a role in neuroinflammation. Therefore, targeting these signaling pathways represents a potential therapeutic approach to alleviate neuroinflammation. Notably, specific inhibitors or drugs have been found to exhibit dual targeting of both NF-κB and MAPK signaling pathways, which may provide a more robust anti-neuroinflammatory effect. This highlights the possibility of developing a combination therapy targeting multiple pathways for the treatment of neuroinflammation.
Several synthetic drugs or inhibitors have been discovered that have anti-neuroinflammatory effects by targeting the signaling pathways of NF-κB and MAPK. For example, Tripterygium (TG), a non-steroidal immunosuppressant, has been shown to have anti-inflammatory, anti-tumor and immunosuppressive properties (171). Research suggests that TG can alleviate neuroinflammation by inhibiting the NF-κB and MAPK signaling pathways, thereby reducing the expression of Aβ25-35, p-Tau, CD11b and various pro-inflammatory cytokines in an AD model. This implies the feasibility of TG intervention in AD pathology (172). A compound called 4-[(5-bromo-3-chloro-2-hydroxybenzyl) amino]-2-hydroxybenzoic acid (LX007) (173) has been identified as a potent mitigator of microglia-induced inflammatory responses. LX007 has demonstrated a significant anti-inflammatory activity in LPS-stimulated primary microglia inflammation models by inhibiting the phosphorylation of MAPK and NF-κB p65 nuclear translocation, effectively inhibiting NO and prostaglandin E2 (PGE2) production and reducing pro-inflammatory cytokine gene and protein expression (173). These findings imply that LX007 may be a potential drug for treating inflammatory reactions. Pseudane-VII, a secondary metabolite derived from Pseudoalteromonas sp. M2, has been shown to possess anti-inflammatory activity (173) by inhibiting the phosphorylation of p38, ERK1/2, JNK1/2 and NF-κB. Similarly, diammonium glycyrrhizinate (DG), the salt form of glycyrrhizin acid (174), has been found to play a critical role in inhibiting Aβ1-42-induced neuroinflammation by regulating the MAPK and NF-κB pathways (174). An in vivo study has revealed that DG can alleviate memory impairment in mice, inhibit activation of microglia in the hippocampus and reduce the expression and production of pro-inflammatory mediators (175). Further investigation has revealed that the anti-inflammatory effect of DG involves inhibiting the translocation of NF-κB p65 to the nucleus, as well as reducing the phosphorylation levels of ERK, JNK and p38 MAPK (175). It is notably that aldose reductase inhibitors (ARIs) exert their effects by regulating the ROS/protein kinase C (PKC)-dependent NF-κB and MAPK signaling pathways. Aldose reductase (AR), a rate-limiting enzyme in the polyol pathway of glucose metabolism, is a molecular target in various inflammatory diseases (176). An in vitro study was conducted to investigate the effects of typical ARIs, sorbinil (Sor) and zopolrestat (Zol) (177), on Aβ1-42-induced BV-2 microglia. The results demonstrated that both Sor and Zol significantly inhibited TNF-α secretion, downregulated the expression of pro-inflammatory genes and proteins via interference with the NF-κB and MAPK pathways, in addition to inhibiting the phosphorylation of several PKC subtypes (177). Notably, this inhibition of PKC was demonstrated to be mediated by reducing intracellular ROS generation (178). Taken together, these findings suggest that the anti-neuroinflammatory effects of ARIs are, at least in part, ROS/PKC dependent (177). However, further in vivo studies are necessary to confirm the efficacy and safety of ARIs, as well as to explore their potential for treating neurodegenerative diseases.
Traditional medicines, natural products, and their derivatives have demonstrated promising therapeutic properties for the treatment of neuroinflammation. Artemisiae Iwayomogii Herba (AIH), a traditional herb (179) utilized for the treatment of inflammatory conditions, was found to inhibit LPS-induced neuroinflammation in BV-2 microglia and mice brains (180). This effect was achieved by reducing NO production and the expression of pro-inflammatory mediators, as well preventing the formation of the NLRP3 inflammasome (180). The anti-inflammatory effect of AIH is associated with the regulation of the NF-κB and MAPK signaling pathways (180). Similarly, Ganoderma lucidum extract (GLE) (181) has been shown to possess neuroprotective properties (182) and has exhibited efficacy in the treatment of inflammatory diseases (183). Pretreatment with GLE downregulates the expression of pro-inflammatory genes in LPS-stimulated BV-2 microglia by modulating NF-kB and MAPK signaling pathways, thereby exerting an anti-neuroinflammatory effect (181). Atractylodis Rhizoma Alba (ARA) ethanolic extract (ARAE) (134) was also found to have anti-neuroinflammatory effects in an in vitro inflammatory model, associated with the inhibition of the NF-κB and MAPK signaling pathways (184). ARAE significantly decreased the production of NO and inflammatory cytokines and inhibited the expression of iNOS and COX-2. Further analysis indicated that the anti-inflammatory effects of ARAE were mainly due to inhibition of IκBα degradation, phosphorylation, and NF-κB p65 nuclear translocation, suggesting a multi-pathway approach to reducing neuroinflammation (134). Similarly, 1-O-acetylbritannilactone (also termed Inulicin; ABL), a natural product derived from Inula britannica L. (185) and its derivative 'compound 15' were found to inhibit neuroinflammation in LPS-induced BV-2 microglia. Compound 15 was found to block NF-κB translocation, reduce CD14 generation by TLR4 in a dose-dependent manner, and significantly inhibit p38 MAPK phosphorylation, thereby downregulating the p38 MAPK inflammatory signaling pathway. Moreover, compound 15 was found to convert BV-2 microglia from M1 to M2 phenotypes, further enhancing its ability to inhibit neuroinflammation (185). Eucommia ulmoides Oliver (Du Zhong) is a renowned traditional Chinese medicine containing therapeutic chemical compounds for a variety of diseases (186,187). Its active compounds possess anti-neuroinflammatory properties, with ulmoidol (ULM) (188) exhibiting the most potent anti-inflammatory activity. By interfering with TLR4 signaling, ULM inhibits downstream NF-κB and MAPK pathways, downregulates pro-inflammatory cytokine expression and production in LPS-induced BV-2 cells, thereby exerting its anti-neuroinflammatory effects (188). Another active compound, circumdatin D, extracted from Aspergillus ochraceus, possesses dual activity in inhibiting AChE and promoting anti-inflammatory reactions (189). It significantly inhibits NO production, TNF-α, and IL-1β release, and reduces iNOS and COX-2 expression in LPS-induced BV-2 cells by inhibiting TLR4-mediated NF-κB, MAPK, and JAK/STAT inflammatory signaling pathways. Tectorigenin (TEC), an active ingredient in a number of traditional medicines with anti-tumor (190) and antibacterial effects (191), can also be used to treat neuroinflammation. In in vitro experiments, TEC not only reduces NF-κB p65 subunit levels but also inhibits ERK and JNK phosphorylation (192). Notably, TEC pre-treatment inhibited TLR4, MyD88, and LPS-induced pro-inflammatory cytokine expression both in vivo and in vitro, indicating that its anti-inflammatory mechanisms are closely related to TLR4-MyD88-mediated inhibition of MAPK and NF-κB (192). These findings suggest that traditional Chinese herbal ingredients may be effective in treating neuroinflammatory diseases by inhibiting TLR4 signaling and downstream inflammatory pathways. Further studies are needed to explore their potential clinical applications and mechanisms of action in vivo. In summary, traditional medicines, natural products, and their derivatives have shown promise in targeting both NF-κB and MAPK signaling pathways and represent a promising therapeutic approach for managing AD.
3. NLRP3 inflammasome
The NLRP3 inflammasome is a multi-protein complex consisting of the regulatory subunit NLRP3, the adaptor protein apoptosis-associated speck-like protein (ASC) and the effector cysteine protease caspase-1 (192) that plays a central role in sterile inflammatory diseases by regulating the cleavage of IL-1β precursor (193). The inflammasome requires two signals for activation: The first signal triggers the synthesis of IL-1β precursor and other inflammasome components such as NLRP3 and caspase-1; the second signal leads to the assembly of the NLRP3 inflammasome, activation of caspase-1, and secretion of IL-1β (194). However, dysregulated signal transduction or excessive activation of the NLRP3 inflammasome can lead to a chronic inflammatory environment that promotes the pathogenesis and progression of various diseases, including AD (195). Activated NLRP3 inflammasomes have been observed in the brains of patients with AD and are closely associated with microglia. Study has shown that NLRP3 inflammasomes affect Aβ pathology and behavioral deficits in animal models of AD by modulating the phenotype and function of microglia (196). Notably, Aβ can also activate the NLRP3 inflammasome, leading to the release of proinflammatory cytokines such as IL-1β by microglia, contributing to neuroinflammation in AD (197). Thus, the NLRP3 inflammasome is a crucial target in AD and drugs that inhibit its activation through the inhibition of molecule formation, silencing of upstream signals, or direct/indirect inhibition of inflammasome complex formation may prove beneficial.
Inhibitors targeting the NLRP3 inflammasome have shown efficacy in suppressing neuroinflammation and hold promise as potential candidates for the prevention and treatment of AD. Among these inhibitors, dapansutrile (OLT1177) (198), a novel oral agent that selectively targets the NLRP3 inflammasome, has demonstrated the ability to block caspase-1 activation and IL-1β maturation and release. OLT1177 is currently in clinical trials for inflammatory diseases and has been shown to be well tolerated in humans (199,200). A study using a APP/PS1 mouse model demonstrated that OLT1177 treatment can reduce microglia activation and the number of Aβ plaques in the cortex (198). An in vitro study also suggested that OLT1177 treatment can significantly reduce the release of pro-inflammatory cytokines and improve the inflammatory status of microglia (198). Similarly, MCC950 (201), a small molecule inhibitor specific for the NLRP3 inflammasome that contains a diarylsulfonylurea structure, has shown promise as a potential treatment for AD. MCC950 has been found to improve cognitive impairment and reduce Aβ accumulation and microglia activation in the APP/PS1 mouse model (201). An in vitro study has shown that MCC950 can inhibit NLRP3 inflammasome activation and IL-1β release while promoting the phagocytic effect of Aβ in microglia (201). Similar results were observed in middle-aged APPNL-F/NL-F mice, where MCC950 blocked the NLRP3 inflammasome and attenuated the reactive response of microglia induced by AβO, leading to improvements in memory impairment (202). Additionally, a lead compound, JC124 (203), based on sulfonamide-type NLRP3 inhibitors, has recently shown beneficial effects in the prevention of AD. JC124 has been found to reduce Aβ plaques and microglia activation in the brains of APP/PS1 mice and has demonstrated certain anti-inflammatory properties (203).
In addition to specific inhibitors that target the NLRP3 inflammasome, certain herbal extracts exhibit anti-inflammatory effects on this pathway. Ginkgolide B (GB) (204), a plant ester derived from Ginkgo biloba, has been shown to possess anti-inflammatory, antioxidant and anti-apoptotic properties, as well as potent neuroprotective effects (205,206). In an in vitro study, GB treatment prevented AD pathological processes and suppressed neuroinflammation in Aβ1-42-induced BV2 microglia by inhibiting NLRP3 inflammasome activation and promoting M2 polarization (204). Paeoniflorin (PF) (207), a natural neuroprotectant from Paeonia lactiflora Pall, has shown significant therapeutic effects in experimental models of Parkinson's disease (208) and stroke (209). Research has shown that PF significantly reduces the protein levels of the pro-inflammatory cytokines TNF-α and IL-1β in APP/PS1 mice while increasing the anti-inflammatory cytokines IL-10 and IL-4. Its pharmacological effects are achieved by enhancing the activity of AKT, inhibiting the activation of glycogen synthase kinase-3β (GSK-3β) and NF-κB p65, and thereby reducing the NLRP3 expression levels (207).
Controlling the activity of various kinases that regulate NLRP3 inflammasome activity is another promising way to suppress neuroinflammation by inhibiting NLRP3 inflammasome activation. One such enzyme is hematopoietic cell kinase (HCK), which is involved in a number of inflammatory responses (210). It is suggested that HCK is an upstream regulator of the NLRP3 inflammasome and that the use of an HCK inhibitor [A419259 (211), a Src family kinase-specific inhibitor] can reduce NLRP3 inflammasome-mediated inflammation in microglia. Further mechanistic studies have shown that the absence of HCK and inhibition of HCK kinase activity directly affects NLRP3 function by inhibiting ASC oligomerization and inflammasome assembly. In vivo experiments confirm that A419259 intervention can alleviate inflammation in a mouse model of LPS-induced inflammation (211). Therefore, A419259 may therefore be a promising drug candidate for the treatment of diseases associated with NLRP3 inflammasome activation, such as AD.
Targeting the initial signal for NLRP3 inflammasome activation has emerged as an effective strategy for the treatment of neuroinflammation. TAK-242 (212), a cyclohexene derivative, is a specific small molecule inhibitor of TLR4 that is capable of crossing the BBB and exerting neuroprotective effects (213). This effect may be mediated through the modulation of the TLR4/MyD88/NF-κB/NLRP3 signaling pathway. TAK-242 can reduce TLR4 expression and attenuate inflammatory cytokine production in microglia from mice with AD carrying APP/PS1 mutations (212). As a result, there is a significant decrease in pro-inflammatory M1-type markers, such as iNOS and TNFα, while M2-type markers, including Trem-2 and Arg-1 are increased (212). Further investigation has also demonstrated that TAK-242 treatment can improve the upregulation of inflammatory cytokines, as well as MyD88, NF-κB p65 and NLRP3 (212). Similarly, the TLR4-specific inhibitor, CLI-095 (214), exerts similar anti-inflammatory effects on LPS/Aβ1-42-induced BV-2 cells and primary microglia by ameliorating neuroinflammation through the TLR4/NLRP3 pathway (214).
Activation of the second signal of the inflammasome is a mechanism by which certain drugs, such as Pterostilbene and lignin-amides Datura metel seeds (LDS), can inhibit neuroinflammation. Pterostilbene, a natural compound with neuroprotective properties (215), has been found to inhibit Aβ1-42-induced NO production, iNOS mRNA and protein expression in BV-2 cells, while also reducing the expression and secretion of inflammatory factors (216). Moreover, pterostilbene can deactivate the NLRP3/caspase-1 inflammasome activated by Aβ1-42, demonstrating its anti-inflammatory effects. The caspase-1 inhibitor, Z-YVAD-FMK, effectively reduces Aβ1-42-induced neuroinflammation in BV-2 cells, providing further support for this hypothesis (216). In addition to pterostilbene, LDS is also able to ameliorate neuroinflammation through the NLRP3/caspase-1 pathway. Wang et al (217) found that LDS had anti-inflammatory activity in LPS-induced BV2 cells. Additionally, PPSR (PEG-PEI/siROCK2), a synthetic molecule used in gene therapy for AD, was found to inhibit the increase in IL-1β induced by LPS/Aβ in primary microglia through the NLRP3/caspase-1 pathway, thus exhibiting anti-inflammatory effects (218). However, the specific mechanism through which PPSR regulates the NLRP3/caspase-1 pathway remains to be elucidated and requires further investigation (219).
Gasdermin D (GSDMD) plays a crucial role in pyroptosis, whereby intracellular inflammasomes trigger caspase-1-mediated cleavage of the effector protein GSDMD to form p30-GSDMD, resulting in the formation of cell membrane pores and release the inflammatory factors (220). Recently, two novel GSDMD cleavage inhibitors, Sulfa-4 and Sulfa-22 (221), were shown to effectively attenuate neuroinflammation and prevent AD by disrupting the NLRP3/caspase-1/GSDMD classical pyroptosis pathway. The investigation demonstrated that the administration of Sulfa-4 and Sulfa-22 inhibited the activation of microglia in the brains of APP/PS1 mice, reduced the expression of inflammatory factors and suppressed the production of p30-GSDMD and upstream NLRP3 inflammasome and caspase-1 proteins. Furthermore, the study revealed the specific binding relationship between Sulfa-4 and Sulfa-22 and the GSDMD protein, establishing a valuable basis for the development of drugs to target neuroinflammation in AD (221).
Donepezil is a commonly used AChE inhibitor for the treatment of AD (222). There is evidence that cognitive function, activities of daily living and overall clinical status, as assessed by healthcare professionals, improve slightly in individuals with AD who are treated with donepezil. In addition, the use of donepezil does not appear to significantly increase or decrease healthcare costs compared with placebo. However, it is important to note that withdrawal rates and adverse events tended to be higher at higher doses (223,224). Recent study has demonstrated that donepezil can effectively inhibit LPS-induced neuroinflammation by downregulating the mRNA levels of proinflammatory cytokines in BV2 cells (225). This effect can be attributed to the intervention of the MAPK/NLRP3/STAT3 pathway. Furthermore, in LPS-treated wild-type mice, treatment with donepezil effectively reduced the activation and quantity of microglia, as well as the levels of proinflammatory cytokines (225). In addition, donepezil was also found to improve the neuroinflammation induced by Aβ stimulation in 5XFAD mice (225). These findings are supported by the study by Kim et al (226), which demonstrated that Donepezil directly inhibits AβO-induced microglia activation by blocking the MAPK and NF-κB signals, thereby improving neuroinflammation and mitigating memory impairment.
AMPK is a vital molecule that plays a critical role in regulating energy metabolism and mitochondrial function (226). Mitochondrial dynamics are primarily controlled by mitosis (227), which promotes the expression of phosphate and tensin homolog deleted on chromosome 10 (PTEN)-induced kinase 1 (PINK1) on the damaged outer mitochondrial membrane. This, in turn, elevates the activity of the E3 ubiquitin ligase Parkin, modulating the autophagic process (228). Tetrahydroxy stilbene glycoside (TSG) (229), the major bioactive component of traditional Chinese medicine Polygoni multiflori Radix, exhibits potent antioxidant and anti-atherosclerotic properties (230) and has demonstrated a neuroprotective in repairing brain injury (231). A recent study has found that TSG can attenuate the LPS-induced inflammatory response in microglia by inhibiting the NLRP3 signaling pathway while promoting the autophagic process mediated by the AMPK/PINK1/Parkin pathway (229). Notably, the neuroprotective effect of TSG is abolished in PINK1 or Parkin knockout models, underscoring the critical role of inhibition of NLRP3 activation through the AMPK/PINK1/Parkin signaling pathway for TSG to exert its neuroprotective effects (229).
In addition, RhoA, a member of the Rho family of GTPases, forms the RhoA/ROCK signaling pathway with the downstream effector Rho-dependent coiled-coil kinase (ROCK) (232). Activation of this pathway can further activate NLRP3, leading to neuroinflammation (233) and increased Aβ production (234) through APP cleavage-dependent secretion, contributing to Aβ-induced neurotoxicity. The RhoA/ROCK signaling pathway also affects the phagocytic function (235) of microglia and neuroinflammatory responses (236), as well as interactions with Aβ and microglia (237). A recent study has demonstrated that small molecule inhibitors, such as Fasudil and Y27632, can alleviate AD pathogenesis by suppressing the RhoA/ROCK/NLRP3 signaling pathway, thereby reducing LPS-induced inflammatory responses (238).
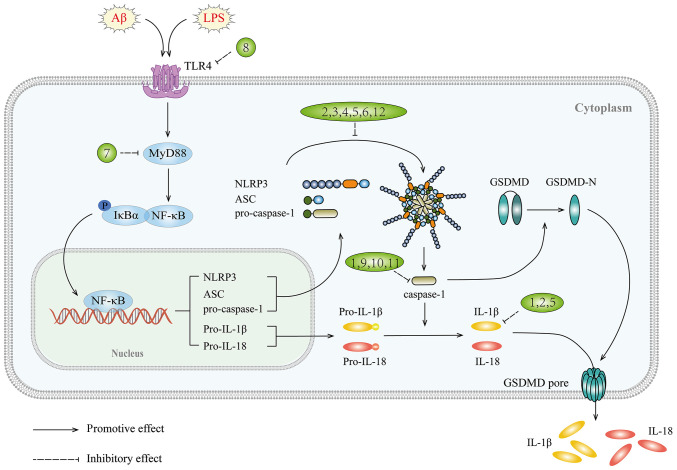
In summary, targeting the NLRP3 inflammasome has the potential to provide a multitude of effective therapeutic avenues for managing neuroinflammation in AD (Fig. 3 and Table III).
Figure 3.
NLRP3 signaling pathway and targets of inhibitors against neuroinflammation in AD. NLRP3, NOD-like receptor thermal protein domain associated protein 3; AD, Alzheimer's disease; 1, dapansutrile; 2, JC124; 3, MCC950; 4, ginkgolide B; 5, paeoniflorin; 6, A419259; 7, TAK-242; 8, CLI-095; 9, PEG-PEI/short interfering ROCK2; 10, pterostilbene; 11, LDS; 12, sulfa-4 and sulfa-22; Aβ, amyloid β; LPS, lipopolysaccharide; TLRs, Toll-like receptors; MyD88, myeloid differentiation factor 88.
Table III.
Drugs that prevent and treat AD through the NLRP3 signaling pathway.
| First author, year | Compound and original source | In vivo model | In vitro model | Dose and drug administration time | Targets | Signaling pathways | (Refs.) |
|---|---|---|---|---|---|---|---|
| Lonnemann et al, 2020 | OLT1177/- | APP/PS1Δ E9 mice | LPS-induced primary microglia |
In vivo: 3.75, 7.5 g/kg; 3 months In vitro: 5, 10 μM; 24 h |
↓: CD68, TNF-α, IL-1β, IL-6, NLRP3, Iba-1, the number of plaques | NLRP3 | (198) |
| Dempsey et al, 2017 | MCC950/- | APP/PS1 double transgenic AD mice | LPS+Aβ-induced primary microglia |
In vivo: 10 mg/kg; 3 months In vitro: 100 nM; 5 h |
↓: IL-1β, LDH, caspase 1, inflammasome assembly, Aβ, CD11b, CD68 | NLRP3 | (201) |
| Fekete et al, 2019 | AβO-induced AD rats APPNL-F/NL-F mice | - | In vivo: 1 μg; 4 weeks | ↓:Iba1, Cd11b, Cd68, Cd80, Cd86, RT1-EC2, Ccl2, Cxcl10, C3, Cfb, NLR3, Il1b, Tnf, Il12b, Nos2, Cx3cl1, Cd200, Cd22, Cx3cr1, Cd200r, Cd45 ↑: Scn1,IL-10 |
- | (202) | |
| Kuwar et al, 2021 | JC124/- | APP/PS1 double transgenic mice with AD | - | In vivo: 50, 100 mg/kg; 3 months | ↓: Aβ, Iba1, HMGB1, GFAP, D1 ↑: generation and survival of new neurons, pre-synaptic proteins, synapsin-1, synaptophysin |
NLRP3 | (203) |
| Zhang et al, 2021 | GB/Ginkgo biloba | - | Aβ1-42-induced BV2 cells | In vitro: 100 μM; 2 h | ↓: Cytotoxic, NLRP3, caspase-1, IL-1β, Aβ, CD16/32, iNOS ↑:CD206, Arg-1, CD206 |
NLRP3 | (204) |
| Zhang et al, 2015 | PF/Paeonia lactiflora Pall | APP/PS1 double transgenic mice with AD | - | In vivo: 5 mg/kg; 4 weeks | ↓: Aβ, GFAP, CD11b, TNF-α, IL-1β, p-NF-κB p65, p-I-κBa, NLRP3, caspase-1 p20 ↑: IL-10, IL-4, p-AKT, p-GSK3β-pSer9 |
NLRP3 | (207) |
| Kong et al, 2020 | A419259/- | LPS-induced C57BL/6J mice | LPS-induced primary microglia |
In vivo: 30 mg/kg; 3 h In vitro: 1 μM; 1 h |
↓: Caspase 1, IL-1β, ASC, the interaction between HCK and NLRP3, IL-6, IL-10 | NLRP3 | (211) |
| Cui et al, 2020 | TAK-242/- | APP/PS1 double transgenic mice with AD | Aβ-induced BV2 cells |
In vivo: 2 mg/kg; 28 days In vitro: 100 nM; 8 h |
↓: TLR4, CD11b, amoeboid microglial cells, iNOS, TNFα, MyD88, NF-κB p65, NLRP3, Bax, iNOS ↑: TREM-2,Arg-1 |
TLR4/MyD88/NF-κB/NLRP3 | (212) |
| Liu et al, 2020 | CLI-095/- | - | LPS + Aβ1−42-induced BV2 cells and primary microglia | In vitro: 1 μM; 2 h | ↓: NLRP3, ASC, caspase1 p10, IL-1β, Iba-1, IL-1β, TNF-α, iNOS, Cox-2 | TLR4/NLRP3 | (214) |
| Li et al, 2018 | Pterostilbene/- | - | Aβ1–42-induced BV2 cells | In vitro: 5, 10 μM; 24 h | ↓: NO, iNOS, IL-6, IL-1β, TNF-α, NLRP3, caspase1 | NLRP3/caspase1 | (216) |
| Wang et al, 2021 | LDS/- | - | LPS-induced BV2 cells | In vitro: 400, 200, 100 μg/ml; 12 h | ↓: iNOS, COX-2, NO, IL-1β, TNFα, IL-6, NLRP3, TLR4, MyD88, caspase1, Iba1, Tau | NLRP3/caspase1 | (217) |
| Liu et al, 2022 | PPSR/- | - | LPS+Aβ42-induced primary microglia | In vitro: transfection; 6 h | ↓: ROCK2, IL-1β, NLRP3, procaspase-1, caspase-1 | NLRP3/caspase1 | (219) |
| Han et al, 2021 | Sulfa-4 and sulfa-22/- | APP/PS1 double transgenic mice with AD | LPS+nigericin-induced BV2 cells |
In vivo: 5 mg/kg; 14 days In vitro: Sulfa-4 (IC50 of 3 μM); 4 h Sulfa-22 (IC50 of 5 μM); 4 h |
↓: LDH, PI uptake rate, p30-GSDMD, IL-18, IL-1β, TNF-α, NLRP3, Caspase-1, IBA-1, CD11c | NLRP3/caspase1/GSDMD | (221) |
| Kim et al, 2021 | Donepezil/- | LPS-induced C57BL6/J mice 5XFAD mice APP/PS1 double transgenic mice with AD | LPS-induced BV2 cells |
In vivo: 1 mpk; 3 days/2 weeks In vitro: 50 μM; 23.5 h |
↓: COX-2, IL-1β, IL-6, iNOS, ROS, p-AKTser473, p-AKTT308, p-ERK, p-P38 T180/Y18, p-NF-kBSer536, p-STAT3 Ser727, NLRP3, pro-IL-1β, IL-1β, Iba-1,GFAP | MAPK/NLRP3/STAT3 | (225) |
| Kim et al, 2014 | AβO-induced mice with AD | AβO1-42-induced BV2 cells, rat primary microglia and primary hippocampal cells |
In vivo: 2 mg/kg; 5 days In vitro: 0.1, 1 μM; 24 h |
↓: NO, TNF-α, IL-1β, PGE2, iNOS, COX-2, p38 MAPK, NF-κB p65 translocation to nucleus, Mac-1, GFAP ↑:cell viability |
MAPK and NF-κB signaling | (226) | |
| Gao et al, 2020 | TSG/Polygoni multiflori Radix | - | LPS-induced BV2 cells | In vitro: 1, 10 μM; 10, 100 nM; 24 h | ↓: TNF-α, IL-1β, IL-18, iNOS, COX-2, P62, p-Drp1(S637), MFF, NLRP3, procaspase-1, cleaved caspase-1, IL-1β/IL-1F2, IL-3, G-CSF, GM-CSF, IL-5, CCL5/RANTES, CCL4/MIP1β, IL-2, IL-4, IL-10, IFN-γ, CCL5/RANTES, CCL4/MIP1β, IL-2 ↑: LC3-II/LC3-I, Parkin, PINK1, Beclin1, Drp1, Mfn2 |
AMPK related PINK1/Parkin/NLRP3 | (229) |
| Zhang et al, 2019 | Fasudil and Y27632/- | - | LPS-induced BV2 cells | In vitro: Fasudil (50 μM), Y27632 (10 μM); 24 h | ↓: Cell migration, NLRP3, pro-CASP1, pro-IL-1β,IL-1β | RhoA/ROCK/NLRP3 | (238) |
AD, Alzheimer's disease; OLT1177, dapansutrile; GB, ginkgolide B; PF, paeoniflorin; TAK-242, ethyl (6R)-6-[N-(2-chloro-4-fluorophenyl) sulfamoyl] cyclohex-1-ene-1-carboxylate); LDS, lignin-amides from Datura metel seeds; PPSR, PEG-PEI/siROCK2; TSG, tetrahydroxy stilbene glycoside; Aβ, amyloid β; APP/PS1, amyloid-β protein/presenilin-1; 5XFAD, 5X familial Alzheimer's disease; AMPK, Adenosine 5′-monophosphate (AMP)-activated protein kinase; ASC, apoptosis-associated speck-like protein; COX-2, cyclooxygenase-2; ERK, extracellular signal-regulated kinases; GFAP, glial fibrillary acidic protein; GSDMD, gasdermin D; HCK, Hematopoietic cell kinase; IL, interleukin; iNOS, inducible nitric oxide synthase; IFN-γ, interferon-γ; LPS, lipopolysaccharide; MyD88, myeloid differentiation factor 88; MAPK, mitogen-activated protein kinase; NLRP3, NOD-like receptor thermal protein domain associated protein 3; NO, nitric oxide; PINK1, PTEN-induced kinase 1; ROS, reactive oxygen species; ROCK, Rho-dependent coiled-coil kinase; TLRs, Toll-like receptors; p-phosphorylated; NF-κB, nuclear factor-kappa-B; Iba-1, ionized calcium binding adaptor molecule 1; STAT3, signal transducers and activators of transcription 3; AP-1, activator protein 1; PGE2, prostaglandin E2.
4. PPAR
Peroxisome proliferator-activated receptors (PPARs) comprise three distinct forms, including PPARα, PPARβ/δ and PPARγ (239), with a large body of literature focusing on PPARγ (240-243). These receptors play a significant role in regulating energy homeostasis and metabolism (244) throughout the body (245). In the brain, PPARs are widely distributed in cognitive centers such as the prefrontal cortex and hippocampus, which are vulnerable to neurodegeneration in AD (246). Despite low baseline expression of PPARγ in the brain, it has been observed to increase in response to AD pathology (247). Studies have shown that PPARγ agonists not only improve cognitive function in patients with AD and animal models (248,249), but also reduce Aβ levels (250). Furthermore, PPARγ is highly expressed in microglia (251) and its activation induces microglia to adopt an anti-inflammatory phenotype, thereby suppressing neuroinflammatory responses (252,253). These findings highlight PPARγ as an attractive therapeutic target for the treatment of AD, with the potential to ameliorate disease pathology.
Current research has demonstrated the anti-inflammatory effects of PPARγ agonists, particularly pioglitazone (PIO), in various mouse models of AD. Berberine (BBR), an alkaloid extracted from Coptidis Rhizoma (254) with similar binding affinity to the PPARγ protein as PIO, has potentially overlapping effects (255). BBR has been found to partially improve neuroinflammation by reducing IL-6 and TNF-α levels in LPS-induced BV-2 cells, indicating a potential preventive or delayed onset of early AD (255). Rice bran extract (RBE), a novel PPARγ regulator that enhances cognitive function in rats (256), also exerts anti-inflammatory effects by regulating microglia phenotype in LPS-induced mice (257). RBE and PIO can both regulate microglia M1 to M2 phenotype, significantly reducing the expression of NF-κB and pro-inflammatory microglia markers (CD45), while increasing the expression of anti-inflammatory microglia markers and PPARγ (257). Additionally, RBE can reduce Aβ42 deposition and p-tau protein levels, thereby effectively ameliorating AD pathology (257).
AD is known to be closely associated with the activation of inflammation, which can be exacerbated by obesity and exacerbate cognitive impairment (258). Malva parviflora extract (MpHE), with its hypoglycemic, anti-inflammatory and antioxidant properties (258,259), has demonstrated the ability to improve the adverse effects of a high-fat diet in an AD mouse model through a PPARγ-dependent mechanism. MpHE not only improved spatial learning deficits and reduced insoluble Aβ peptides in the hippocampus of lean and obese 5XFAD mice but also inhibited the accumulation of small glial cells around Aβ plaques and the conversion to a pro-inflammatory M1 phenotype while promoting phagocytic capacity (260). Rescue of the phagocytic capacity of microglial cells by MpHE was achieved through a PPARγ/CD36-dependent mechanism (260). Angiotensin II receptor blockers (ARBs), used to treat metabolic disorders, have been found to ameliorate inflammation in several brain disorders by blocking angiotensin II type 1 receptors and activating PPARγ, thus exerting a neuroprotective effect (261,262). Clinical trials have shown that ARBs have a positive effect on cognitive decline (263). In addition, it has been shown that telmisartan, a typical ARB, can ameliorate AβO-induced inflammation in microglia (264). Telmisartan (264) has been shown to decrease the expression of the pro-inflammatory cytokine IL-1β, while increasing the expression of PTEN, a key lipid and protein phosphatase, and the anti-inflammatory cytokine IL-10. Furthermore, Telmisartan has also been shown to inhibit the activity of NF-κB, a key transcription factor involved in inflammation and its upstream regulators Akt and ERK (264). These anti-inflammatory effects of telmisartan have been found to be PPARγ dependent, with the PPARγ inhibitor GW9662 blocking the expression of PTEN (264). Taken together, telmisartan ameliorates AβO-induced microglial inflammation via the PPARγ/PTEN pathways. Other compounds with potential therapeutic benefit in AD through anti-inflammatory mechanisms include Bis (ethylmaltolato) oxidovanadium (BEOV) and platycodigenin. BEOV (265) has demonstrated the ability to reduce levels of pro-inflammatory cytokines and interfere with NF-κB signaling in Aβ-stimulated BV2 microglia and the hippocampus of APP/PS1 mice, and its effects have been found to be PPARγ dependent. Platycodigenin (266), a triterpenoid compound found mainly in Platycodon grandifloras, demonstrates neuroprotective and anti-inflammatory activity. Study reveals that platycodigenin can inhibit the secretion of pro-inflammatory cytokines in Aβ-stimulated BV-2 microglia and induce M1-type microglia to polarize towards the M2 type (266). Their anti-neuroinflammatory effects have been attributed to the inhibition of p38 MAPK and NF-κB p65 signaling while activating PPARγ. Although PPAR-γ agonists have shown promising anti-inflammatory activity and their potential use in the treatment of AD, long-term use of these drugs often results in serious side effects, including congestive heart failure, oedema, and weight gain (267,268). Therefore, there is an urgent need to develop PPARγ-targeting drugs with improved tolerability (Fig. 4 and Table IV).
Figure 4.
PPAR, STAT3 and PI3K/Akt signaling pathways and targets of inhibitors against neuroinflammation in AD. PPAR, peroxisome proliferator-activated receptor; AD, Alzheimer's disease; 1, berberine; 2, rice bran extract; 3, telmisartan; 4, bis(ethylmaltolato)oxidovanadium; 5, platycodigenin; 6, (E)-2, 4-bis(p-hydroxyphenyl)-2-butenal; 7, (E)-2-methoxy-4-(3-(4-methoxyphenyl) prop-1-en-1-yl) phenol; 8, astaxanthin; 9, stattic; 10, protosappanin A; 11, curcumin; 12, Ent-Sauchinone; 13, sorafenib; 14, DHCR24 (3-β-hydroxysteroid-Δ-24-reductase); 15, sulforaphene; Aβ, amyloid β; LPS, lipopolysaccharide; IKK, IκB kinase; JAK2, Janus kinase; GSK-3β, glycogen synthase kinase-3β; PTEN, phosphate and tensin homolog deleted on chromosome 10.
Table IV.
Drugs that prevent and treat Alzheimer's disease through other signaling pathways.
| First author, year | Compound and original source | In vivo model | In vitro model | Dose and drug administration time | Targets | Signaling pathways | (Refs.) |
|---|---|---|---|---|---|---|---|
| Wong et al, 2021 | Berberine/Coptidis Rhizoma | - | LPS-induced BV2 cells | In vitro: 0.3-10 μM; 2 h | ↓: Basal respiration, TNF-α, IL-6 | PPARγ | (255) |
| El-Din et al, 2021 | Rice bran extract/rice bran | LPS-induced Swiss Albino mice | - | In vivo: 100 mg/kg; 3 weeks | ↓: CD45, Aβ-42, p-Tau, NF-κB, neuron ↑: arginase1, CD36, CD163 |
PPARγ | (257) |
| Medrano-Jiménez et al, 2019 | Malva parviflora hydroalcoholic leaf extract/Malva parviflora | 5XFAD mice LPS-induced CD1 mice | - | In vivo: 50 mg/kg; 8 months 25, 50, 100 mg/kg; 7 days | ↓: Aβ, CD86, TNF, IL-6 ↑: phagocytic activity, microglia accumulation around the Aβ plaques, Mgl1, TREM-2, PPAR-γ, CD36 |
PPARγ/CD36 | (260) |
| Wang et al, 2020 | telmisartan/- | - | AβO-induced BV2 cells | In vitro: 5 μM-2 h | ↓: IL-1β, TNF-α, NF-κB activation, p-Akt, p-ERK ↑: IL-10, PPARγ, PTEN |
PPARγ/PTEN pathway | (264) |
| He et al, 2021 | Bis (ethylmaltolato) oxidovanadium | APP/PS1 double transgenic mice with AD | Aβ-induced BV2 cells |
In vivo: 0.2, 1 mM; 3 months In vitro: 5, 10, 20 μmol/l; 2 h |
↓: NO, PGE2, iNOS, COX-2, TNF-α, I L-6, IL-1β, p-IκB-α, NF-κB/p65 translocation, Iba1, iNOS, COX-2 ↑: PPARγ |
PPARγ/NF-κB | (265) |
| Yang et al, 2019 | Platycodigenin/Platycodon grandifloras | - | Aβ-induced BV2 cells | In vitro: 0.1, 1, 10 μM; 12 h | ↓: TNF-α, IL-1β, IL-6, NO, iNOS, Cox2, p- p65, p38, neuronal death, neuritic atrophy ↑: IL-10, IL4, CD206, Arg1, TGFβ, Ym1/2, PPARγ |
PPARγ | (266) |
| Jin et al, 2013 | (E)-2, 4-bis (p-hydroxyphenyl)-2-butenal | Tg2576 mice | - | In vivo: 5 mg/kg; 1 month | ↓: Aβ plaques, Aβ1-42, β-secretase, APP, C99, BACE1, GFAP, Iba1, iNOS, COX-2, NF-κB translocation, DNA binding activity of NF-κB, p-IκB, p-STAT1, p-STAT3 | STAT3 | (276) |
| Choi et al, 2017 | (E)-2-methoxy-4-(3-(4-methoxyphenyl) prop-1-en-1-yl) phenol | LPS-induced ICR mice | LPS-induced BV2 cells |
In vivo: 5 mg/kg; 4 weeks In vitro: 1, 5, 10 μg/ml; 24 h |
↓: Aβ, β-secretase, APP, BACE1, C99, GFAP, the DNA binding activity of STAT3, p-STAT3, Iba-1, iNOS, COX-2, IL-6, IL-10 | STAT3 | (277) |
| Han et al, 2019 | Astaxanthin/marine environment | LPS-induced ICR mice | LPS-induced BV2 cells |
In vivo: 30, 50 mg/kg; 4 weeks In vitro: 5, 10, 20 μM; 24 h |
↓: Aβ, β-secretase, APP, BACE1, GFAP, IBA-1, iNOS, COX-2, MCP-1, MIP-1α, MIP-1β, GSH/GSSG, total GSH, NO, TBARS, STAT3 activation | STAT3 | (279) |
| Millot et al, 2020 | Stattic/- | LPS-induced C57BL/6 mice | - | In vivo: 20 mg/kg; 3 days | ↓: PhosphoSTAT3Tyr 705/STAT3 ratio, IBA-1, MAC-1, IL-1β, TNF-α, IL-6, IFN-γ, BACE1 | STAT3 | (282) |
| Wang et al, 2017 | Protosappanin A/Caesalpinia sappan L. | - | LPS-induced BV2 cells | In vitro: 12.5, 25, 50 μmol·l−1; 24 h | ↓: NO, TNF-α, IL-1β, IL-6, MCP-1, p-JAK2, p-STAT3, STAT3 translocation | JAK2/STAT3 | (285) |
| Porro et al, 2019 | Curcumin/Curcuma longa (turmeric) | - | LPS-induced BV2 cells | In vitro: 10, 30, 50 μM; 1 h | ↓: p-JAK2, p-STAT3, iNOS ↑: IL-4, IL-10, SOCS-1, ARG-1 |
JAK/STAT3/SOCS | (286) |
| Song et al, 2014 | Ent-Sauchinone/plants | - | LPS-induced BV2 cells | In vitro: 1, 5, 10 μM; 24 h | ↓: iNOS,COX-2, ROS, NF-κB binding activity, p-IκB, NF-κB translocation, BACE1, C99, I ba1, Aβ accumulation, DNA binding activity of STAT3, STAT3 activity | STAT3/NF-κB | (290) |
| Kim et al, 2021 | Sorafenib/- | LPS induced C57BL 6/J mice 5XFAD mice | LPS-induced BV2 cells |
In vivo: 10 mg/kg; 3 times at 2-h intervals 10 mg/kg; 3 days In vitro: 5 μM; 30 min |
↓: COX-2, IL-1β, p-AKTS473, p-P38T 180/Y182, p-STAT 3S727, p-NF-kB S536, caspase-3, Iba-1, GFAP, p-AKT, shank-1 | AKT/P38-linked STAT3/NF-κB | (291) |
| Zu et al, 2020 | DHCR24 (3-β-hydroxysteroid-Δ-24-reductase)/- | - | Aβ25–35-induced BV2 cells | In vitro: lentiviral transfection-72 h | ↓: iNOS, iNOSCD 11b, IL-1β, TNF-α ↑: Arg-1CD11b, IL-4, TGF-β, p-Akt, p-GSK3β(S9), p-Akt/Akt, p-GSK3β/GSK 3β |
Akt/GSK -3β | (298) |
| Yang et al, 2020 | Sulforaphene/Raphani Semen | STZ-induced SD rats | LPS-induced BV2 cells |
In vivo: 25, 50 mg/kg; 6 weeks In vitro: 0.5-32 μM; 1 h |
↓: TNF-α, IL-6, Iba-1, GFAP, p-tau (Thr205), p-tau (Ser396), p-tau (Ser404), NO, IL-1β, NF-κB p65 translocation ↑: IL-10, p-Akt/Akt, p-GSK-3β (S9)/GSK-3β,PI3K p110α |
PI3K/Akt/GSK-3 β | (300) |
| Zhang et al, 2013 | Sildenafil/- | APP/PS1 double transgenic mice with AD | - | In vivo: 10 mg/kg; 10 days | ↓: IL-1β, IL-6, TNF-α, Aβ1-40, Aβ1-42 ↑: pCREB |
PKG/CREB | (313) |
| Wang et al, 2022 | Thioperamide/- | LPS-induced C57BL/6 mice | LPS-induced BV2 cells |
In vivo: 5 mg/kg; 7 days In vitro: 1 μM; 30 min |
↓: Iba-1, IL-1β, IL-6, TNF-α, NF-κB/CBP ↑: BrdU, DCX, BrdU/DCX, BrdU/NeuN, p-CREB, p-PKA, CREB/CBP, IL-4, IL-10, BDNF, total dendritic length |
histamine-dependent H2R/cAMP/PKA/CREB | (317) |
| Fragoulis et al, 2017 | Methysticin/kava | APP/PS1 double transgenic mice with AD | - | In vivo: 6 mg/kg; 3 weeks | ↓: Iba1, GFAP, TNF-α, IL-17A ↑: Nrf2/ARE, HO-1, Gclc, Nrf2 |
Nrf2 | (323) |
| Mattioli et al, 2019 | Polyphenol extract/Arabidopsis thaliana | transgenic AD flies | Aβ25-35-induced BV2 cells |
In vivo: 40 μl/ml; 3-5 days, 10-12 days In vitro: 20 μl/ml; 24 h |
↓: IL-6, IL-1β, TNF-α, p65 ↑: IL-4, IL-10, IL-13, Nrf2, HO-1, NQO1 |
Nrf2 | (325) |
| Alvariño et al, 2019 | Gracilin A/Spongionella gracilis | - | LPS-induced BV2 cells | In vitro: 0.01-1 μM; 1 h | ↓: IL-1β, IL-6, TNF-α, GM-CSF, ROS, NO, iNOS, p38 MAPK kinase, p-p38, p65 ↑: Nrf2 |
Nrf2 | (327) |
| Huang et al, 2020 | Engeletin/Engelhardia roxburghiana | - | Aβ1–42-induced BV2 cells | In vitro: 20, 40 μM; 24 h | ↓: ROS, MDA, LDH, NO, iNOS, TNF-α, IL-1β, IL-6, Keap1 cell viability ↑: GSH-Px, SOD, Nrf2 |
Keap1/Nrf2 | (331) |
| Eom et al, 2012 | Bambusae Caulis in Taeniam ethyl acetate fraction/Phyllostachys nigra var. henonis | - | LPS-induced BV2 cells | In vitro: 10, 20, 40, 60, 80 μg/ml; C 1 h | ↓: NO, TNF-α, IL-1β, IL-6, iNOS, OX-2 ↑: HO-1, Nrf2 |
Nrf2/HO-1 | (334) |
| Chen et al, 2017 | L-F001/- | LPS- induced C57BL/6 mice | LPS-induced BV2 cells |
In vivo: 35 mg/kg; 24 h In vitro: 0-10 μM; 24 h |
↓: Reactive oxygen, NO, IL-6, TNF-α, CD16/32, iNOS, COX-2, NF-κB p65, degradation of IκB ↑: CD206,Nrf2 |
Nrf2 and NF-κB | (335) |
| Gao et al, 2020 | Beta-naphthoflavone | - | LPS-induced BV2 cells | In vitro: 2.5, 5, 10, 20 μM; 0, 0.5, 1, 3, 6 h | ↓: IL-6, TNF-α, iNOS, COX-2, deterioration of IκBα, p-IκB, p-p65, NF-κB p65 translocation ↑: activation of AKT, Nrf2 translocation, HO-1 |
Akt/Nrf-2/HO-1 signaling axis | (338) |
| Jin et al, 2021 | Cangrelor/- | Aβ1-42-induced mice with AD | - | In vivo: 2, 4 μg/mouse; 6 days | ↓: GPR17, BACE1, Aβ1-42, MDA, TNF-α, IL-1β, Iba1, NF-κB p65 ↑: GSH, SOD, CAT, Nrf2, HO-1, PSD-95, SYN |
Nrf2/HO-1 and NF-κB | (337) |
| Park et al, 2018 | Bakkenolide B/Petasites japonicus | - | LPS-induced BV2 cells | In vitro: 40 μM-4,8, 12,16,24 h | ↓: IL-1β, IL-6, IL-12, TNF-α, ROS ↑: Nrf2, HO-1, NQO1, ARE-promoter activity, p-AMPK |
AMPK/Nrf2 | (349) |
AD, Alzheimer's disease; Aβ, amyloid β; APP/PS1, amyloid-β protein/presenilin-1; 5XFAD, 5X familial Alzheimer's disease; AMPK, Adenosine 5′-monophosphate (AMP)-activated protein kinase; BACE1, β-site amyloid precursor protein cleaving enzyme 1; BDNF, brain-derived neurotrophic factor; COX-2, cyclooxygenase-2; CREB, cyclic AMP response element binding; ERK, extracellular signal-regulated kinases; GFAP, glial fibrillary acidic protein; GSK-3β, glycogen synthase kinase-3β; HO-1, haem oxygenase-1; IKK, IκB kinase; IL, interleukin; iNOS, inducible nitric oxide synthase; IBA-1, ionized calcium binding adaptor molecule 1; interferon-γ, IFN-γ; JAK2, Janus kinase 2; Keap1, Kelch-like ECH-associated protein 1; LPS, lipopolysaccharide; NLRP3, NOD-like receptor thermal protein domain associated protein 3; NO, nitric oxide; NF-κB, nuclear factor-kappa-B; Nrf2, Nuclear factor erythroid 2-related factor 2; PI3K, phosphoinositide 3-kinase; PGE2, prostaglandin E2; PPARs, Peroxisome proliferator-activated receptors; PTEN, phosphate and tensin homolog deleted on chromosome 10; PKA, protein kinase A; PKG, cGMP-dependent protein kinase; ROS, reactive oxygen species; STAT3, signal transducers and activators of transcription 3; SOCS, suppressor of cytokine signaling; TLRs, Toll-like receptors; WDFY1, WD repeat and FYVE domain-containing 1; p-phosphorylated; AP-1, activator protein 1.
5. STAT3
STATs are a group of potential transcription factors that are activated by cytokines and growth factors. When stimulated by LPS, IFN-γ, and other cytokines, they can trigger inflammatory signals that translocate STATs from the cytoplasm to the nucleus and activate the expression of a number of pro-inflammatory genes (269). Of the seven types of STAT proteins found in humans, STAT3 has been extensively studied for its involvement in acute stress responses, cell growth, differentiation, and immune reactions (270). Previous studies have demonstrated elevated activation of STAT3 in hippocampal slices in patients with AD (271) and mouse models (272). Furthermore, STAT3 plays a crucial role in regulating the reactivity of microglia and in mediating pro-inflammatory responses, indicating a close functional interplay with microglia (273). Given the dependence of neuronal differentiation and cytokine signaling on STAT3, STAT3 phosphorylation is closely linked to cytokine secretion (274). Therefore, targeting the signal network that activates STAT3 may be an effective therapeutic strategy for the treatment of AD (275).
It has been reported that (E)-2, 4-b is (p-hydroxyphenyl)-2-butenal (HPB242) (276) exhibits significant anti-inflammatory effects by inhibiting STAT3 activation in AD. Another structurally similar compound, (E)-2-methoxy-4-(3-(4-methoxyphenyl) prop-1-en-1-yl) phenol (MMPP) (277), has been found to inhibit LPS-induced neuroinflammation and memory impairment. In a mouse model of neuroinflammation induced by intraperitoneal injection of LPS, MMPP significantly reduced Aβ deposition in the brain and improved cognitive dysfunction by inhibiting COX-2 and iNOS expression, as well as the activation of microglia in the brain. Furthermore, MMPP treatment reduces the expression of inflammatory protein and APP in LPS-induced BV-2 microglia. In both in vivo and in vitro experiments, MMPP can inhibit the DNA binding activity of STAT3 activation (277). Additionally, axstaxanthin (AXT), a naturally occurring carotenoid compound with anti-inflammatory, antioxidant and neuroprotective properties (278), has exhibited effects on MMPP. In vivo and in vitro experiments have shown that AXT reduces the expression of inflammatory proteins induced by LPS and improves LPS-induced memory impairment by directly binding to the DNA binding domain (DBD) and linker domain (LD) of STAT3, resulting in an anti-neuroinflammatory response and inhibiting APP formation (279).
BACE1 plays a critical role in in the generation of Aβ (280), a major component of AD pathology. The transcriptional regulation of BACE1 by STAT3 (271) is strongly implicated in AD pathology as it can elevate Aβ production (281). Notably, STAT3 is activated in response to LPS-induced neuroinflammation, which in turn increases BACE1 levels in the brain (275). Treatment with stattic, a selective inhibitor of STAT3 activation, has been shown to prevent neuroinflammation and abnormal BACE1 regulation (282). In an LPS-induced mouse model of neuroinflammation, static could inhibit STAT3 phosphorylation and microglia activation in the hippocampus, consequently reducing levels of inflammatory factors in the brain. Notably, treatment with both LPS and Stattic significantly reduced hippocampal BACE1 levels in the hippocampus compared to LPS alone (282). These findings suggest that Stattic may address two pathological aspects of AD in the hippocampus, making it a promising candidate for the treatment of AD.
Janus kinase 2 (JAK2) is a non-receptor protein tyrosine kinase that plays a critical role in the JAK2/STAT3 signaling pathway in the CNS (283). Activation of the JAK2/STAT3 pathway leads to the transcription and expression of inflammatory genes, resulting in an excessive accumulation of inflammatory mediators and subsequent inflammation (284). Therefore, inhibition of the JAK2/STAT3 pathway may be a potential therapeutic approach for neuroinflammatory injury. A promising compound, protosappanin A (PTA), which is a major bioactive component isolated from Caesalpinia sappan L., was found to regulate LPS-induced neuroinflammation by inhibiting the JAK2/STAT3 pathway (285). In the LPS-induced BV2 cell model, PTA treatment reduced the production of TNF-α, IL-1β and NO in microglia, while also dose-dependently decreasing IL-6 and IL-1β mRNA expression (285). A further study demonstrated that PTA inhibited JAK2/STAT3-dependent inflammatory pathways by downregulating JAK2 and STAT3 phosphorylation as well as STAT3 nuclear translocation (285). Furthermore, the Porro et al (286) found that curcumin, a pigment isolated from Curcuma longa (turmeric) with anti-inflammatory, antioxidant, and anticancer activities (287), regulates neuroinflammation by inducing an anti-inflammatory response against microglia through the JAK2/STAT/SOCS (suppressor of cytokine signaling) signaling pathway. Curcumin treatment increased the production of the anti-inflammatory cytokines IL-4 and IL-10, upregulated the expression of the cytokine signaling suppressor SOCS-1, blocked JAK2 and STAT3 phosphorylation and reduced the M1/M2 ratio of microglia phenotype in the same LPS-induced BV2 cell model, thereby ameliorating neuroinflammation from multiple perspectives (286).
NF-κB and STAT3 are two key regulators of cytokine production that can reciprocally modulate each other (288,289). Inhibition of STAT3 activation has been shown to reduce NF-κB activation, thereby attenuating amyloidogenesis and neuroinflammation (271,272). Ent-Sauchinone, a polyphenolic compound from the lignan family, exerts inhibitory effects on neuroinflammation and amyloidogenesis by blocking the STAT3/NF-κB pathway (271). In LPS-stimulated BV-2 microglia, ent-Sauchinone dose-dependently reduces the production of ROS and NO, as well as the expression of iNOS and COX-2, while inhibiting NF-κB activation and the elevated DNA-binding activity of STAT3 induced by LPS. Inhibition of neuroinflammation and prevention of neuroinflammation-induced Aβ production were further confirmed using short interfering RNA and pharmacological inhibitors of STAT3 (290). Sorafenib (291), an anti-cancer drug, also exerts anti-neuroinflammatory effects by modulating the AKT/P38-linked STAT3/NF-κB signaling pathway. It reduces the mRNA expression of pro-inflammatory cytokines in LPS-induced BV-2 microglia and inhibits the increase in STAT3 and NF-κB phosphorylation levels by inhibiting AKT and P38 signaling. An in vivo study further confirmed the anti-inflammatory effects of Sorafenib, suggesting its potential as a therapeutic agent to inhibit neuroinflammatory responses in the brain (291) (Fig. 4 and Table IV).
6. PI3K/Akt
The PI3K/Akt pathway is a vital signaling pathway that regulates a variety of transcription factors and cellular functions (292). Its association with various pathogenic factors of AD, including aging, Aβ and synaptic loss, has been uncovered (293). There are reports of reduced expression of the PI3K/Akt pathway in the brains of patients with AD, while upregulation of this pathway can alleviate tau-induced neurotoxicity and Aβ deposition (294), improve learning and memory capacity and reduce brain damage, and reduce inflammation and oxidative stress in mice with AD (295). Therefore, the role of the PI3K/Akt pathway in microglia has received increasing attention. Studies indicate that PI3K/Akt phosphorylation directly regulates NF-κB in microglia, suggesting a strong link between PI3K/Akt and neuroinflammation (296). GSK-3β signaling, which is involved in inflammation, oxidative stress, and apoptosis, can be activated by Akt phosphorylation upstream (297). Therefore, the Akt/GSK-3β signaling pathway, an important mediator of the inflammatory response, is closely linked to the PI3K/Akt pathway and the role of microglia.
DHCR24 (298), also known as 3-β-hydroxysteroid-Δ24-reductase (seladin-1), exerts neuroprotective effects by participating in the degradation of amyloid precursor proteins, thereby preventing Aβ toxicity, endoplasmic reticulum stress and cellular oxidative damage, which are beneficial in both the prevention and treatment of AD (298-299). A recent study has revealed that DHCR24 can also exert anti-inflammatory effects by activating the Akt/GSK-3β signaling pathway (298). Lentivirus was used to overexpress DHCR24 in BV-2 cells and the results demonstrated that DHCR24 has the ability to attenuate the inflammatory response induced by Aβ25-35 by altering the polarization phenotype of microglia (298). Further mechanistic analysis revealed that DHCR24 affected the protein expression levels of P-Akt and P-GSK-3β. Furthermore, the Akt inhibitor MK2206 attenuated this effect, thus demonstrating the neuroprotective function of DHCR24 in AD-associated inflammatory injury (298). Sulforaphene (SF) (300) is an isothiocyanate derived from Raphani Semen (301). SF inhibits neuroinflammation by modulating the PI3K/Akt/GSK-3β pathway (300). In rats treated with intravenous streptozotocin (STZ), SF treatment significantly improved STZ-induced cognitive impairment, inhibited the production of pro-inflammatory factors and promoted the release of anti-inflammatory factors. Additionally, SF increased the ratio of p-Akt/Akt and p-GSK-3β/GSK-3β in the rat hippocampus (300). In LPS-stimulated BV-2 cells, SF exerted significant inhibitory effects on the release of NO, TNF-α, and IL-6, while also affecting the nuclear translocation of p-NF-κB p65 and the p-GSK-3β (Ser9)/GSK-3β ratio (300). Therefore, SF shows promising potential as a neuroprotective agent and could be further developed as a therapeutic treatment for AD (Fig. 4 and Table IV).
7. CREB
The cyclic AMP response element binding (CREB) is a stimulus-inducible transcription factor that dimers with the conserved cyclic AMP response element (CRE) (302) to activate CRE-responsive genes in response to extracellular stimuli (303). In the CNS, CREB regulates various protein kinases, including protein kinase A (PKA) and MAPKs, which are involved in neuronal development, synaptic plasticity, short-term to long-term memory conversion and neuroprotection in the CNS (304,305). Furthermore, dysregulated CREB phosphorylation has been identified in AD mouse models (306) and patients with AD (307), demonstrating the important role of CREB in the pathogenesis of AD (308). Notably, CREB has been found to be associated with neuroinflammation and may be an effective therapeutic target for the treatment of AD (309). Phosphorylation of CREB has been shown to reduce neuroinflammation by regulating NF-κB to block the transcription of inflammatory mediators (310). Moreover, phosphorylation of CREB promotes the production of anti-inflammatory cytokines in activated microglia that induce microglia inactivation or polarization to the M2 phenotype (311), thus modulating neuroinflammation for neuroprotection in AD.
The cGMP-dependent protein kinase (PKG) plays an important role in mediating the transcriptional regulation of CREB by phosphorylating CREB and activating different downstream genes (312). In an aged Tg APP/PS1 mouse model, sildenafil was found to be effective in reducing neuroinflammation and Aβ levels in the brain. Specifically, sildenafil suppressed Aβ-induced pro-inflammatory factors in the hippocampus, and this effect was mediated through the PKG/CREB signaling pathway (313). Inhibition of PKG in the hippocampus prior to sildenafil injection resulted in blocked CREB phosphorylation, resulting in a reduced production of inflammatory factors and ultimately produced anti-inflammatory effects. Furthermore, there is ample evidence in the literature to support the crucial role of the PKA/CREB pathway as a drug target in AD (314), particularly in the context of downregulation of the transcriptional cascade that contributes to the disease. Additionally, inhibition of histamine H3 receptors (H3R) has been shown to improve cognitive deficits in AD (315,316). The histamine H3R antagonist, thioperamide (317), can effectively inhibit inflammatory cell recruitment (318), further highlighting the importance of the PKA/CREB pathway in modulating neuroinflammation as a therapeutic target for AD. It has been found that thioperamide exerts its effects on the PKA/CREB signaling pathway, suppressing microglia activity and promoting their conversion from M1 to M2 phenotype, ultimately impeding LPS-induced neuroinflammation and restoring cognitive function in mice (317). Mechanistically, the downstream PKA/CREB pathway activated by H2R stimulation triggers CBP (CREB-CREB binding protein) interactions that facilitated the release of anti-inflammatory factors and brain-derived neurotrophic factor, while simultaneously attenuating NF-κB-CBP interactions to reduce the secretion of pro-inflammatory factors. These effects were found to be reversible by cimetidine (H2R antagonist) but not by piramine (H1R antagonist), indicating a novel H2R-dependent histamine-mediated mechanism underlying the therapeutic effects of thioperamide on neuroinflammation (317) (Table IV).
8. Nrf2
Nuclear factor erythroid 2-related factor 2 (Nrf2) is a transcription factor that plays a crucial role in regulating oxidative stress in various cell types, including glial cells and neurons (319). Notably, a reduction in Nrf2 expression has been detected in the brains of patients with AD (320). Moreover, a growing body of research indicates that augmenting Nrf2 signaling has the potential to improve Aβ-induced neurodegeneration and oxidative stress in in vitro and in vivo models of AD (321). Such investigations have also revealed that enhancing Nrf2 signaling can alleviate microglia-mediated inflammation in the brain (322), highlighting the potential for therapeutic intervention targeting Nrf2 in the development of drugs for the treatment of AD.
Studies have shown that certain herbs and natural products contain active ingredients that can interfere with Nrf2, thereby inhibiting neuroinflammation. For example, Methysticin (323), a kavalactones derived from the Piperaceae plant kava (324), has been demonstrated to inhibit neuroinflammation and oxidative damage and to attenuate long-term memory loss in APP/PS1 mice. These effects are attributed to its ability to significantly reduce microglia activation and the secretion of pro-inflammatory factors in the hippocampus and cortex, possibly mediated by Nrf2. Similarly, a polyphenol extract derived from Arabidopsis thaliana was found to have anti-inflammatory activity in transgenic AD flies and Aβ25-35-induced BV2 cells by influencing the nuclear translocation of Nrf2 and NF-κB (325). Gracilin A, a natural product isolated from the marine sponge Spongionella gracilis (326), has been associated with Nrf2-involved inflammation. An in vitro study has shown that Gracilin A reduces the release of pro-inflammatory factors from BV2 cells induced by LPS by inhibiting the expression of iNOS and the activation of p38 MAPK, which affects the translocation of NF-κB p65 and Nrf2 (327).
Kelch-like ECH-associated protein 1 (Keap1), as an adapter protein, inhibits the function of Nrf2 by degrading it in the normal state of the cell (328). However, when cells are exposed to external stimuli, the degradation of Keap1 (dependent on Nrf2), is inhibited, leading to the accumulation of Nrf2 in the nucleus and its regulatory role in the expression of various antioxidant genes (329). Engeletin, a flavonol glycoside derived from the leaves of Engelhardia roxburghiana (330), has demonstrated anti-inflammatory properties. Specifically, it has been shown to inhibit the expression and secretion of Aβ1-42-induced pro-inflammatory factors and to enhance the activation of the Keap1/Nrf2 pathway in BV-2 cells. However, when Nrf2 was knocked down, the inhibitory effect of Engeletin was reversed. These findings further underscore the potential of pharmacological intervention targeting the Keap1/Nrf2 pathway in anti-AD therapy (331).
Haem oxygenase-1 (HO-1), a stress-inducible protein, exerts a protective effect against inflammatory and oxidative stress and has been shown to be beneficial in neurodegenerative diseases including AD (332). The promotion of HO-1 expression is mediated by Nrf2 (333). Study has shown that Bambusae Caulis in Taeniam ethyl acetate fraction (BCE) (334) as a modulator of Nrf2 signaling, regulates the neuroprotective and anti-neuroinflammatory effects of microglia BV2 by modulating the expression of HO-1. BCE was shown to inhibit the production of pro-inflammatory mediators and cytokines in LPS-induced BV2 cells, while upregulating the mRNA and protein expression levels of HO-1, and influencing the accumulation and transactivation of Nrf2 in the cells (334). Further evidence for the involvement of HO-1 in the observed anti-inflammatory effects of BCE was obtained by using the selective HO-1 inhibitor, SnPP, which reversed these effects (334).
Researchers have identified multiple inhibitors that act on multiple pathways to inhibit neuroinflammation by targeting Nrf2. Among these inhibitors, L-F001 (335), a newly developed ROCK inhibitor, has shown promise in the treatment of AD by inhibiting NF-κB and activating Nrf2. An in vitro study has demonstrated that L-F001 significantly inhibits the expression of iNOS and COX-2 as well as the secretion of pro-inflammatory mediators in BV-2 cells following LPS induction (335). This is accompanied by inhibition of NF-κB signaling and upregulation the expression of HO-1 and glutamate cysteine ligase modifier subunit, downstream effectors of Nrf2 (335). Similarly in vivo experiments, on mice have confirmed that L-F001 significantly reduces the levels of pro-inflammatory mediators induced by LPS, in line with the in vitro findings (335). In addition, the researchers have found that G protein-coupled receptor 17 was expressed in neurons and microglia (336) and that its antagonist, cangrelor (337), had an inhibitory effect on neuroinflammation. In a mouse model of AD with intracerebroventricular injection of Aβ1-42, cangrelor reduced BACE1 activity as well as Aβ1-42 levels in the hippocampus and frontal cortex of mice, while inhibiting microglia activation and levels of pro-inflammatory factors through a mechanism involving Nrf2/HO-1 and NF-κB signaling (337). Another inhibitor, β-naphthoflavone (BNF) (338), a derivative of a natural flavonoid widely used in the pharmaceutical industry, has antioxidant and anti-inflammatory effects. Pretreatment with BNF was found to inhibit activation of the NF-κB pathway in LPS-treated BV-2 cells, promote AKT activation, enhance the nuclear translocation of Nrf2, lead to an upregulation of the HO-1 protein levels, and significantly reduce the expression of pro-inflammatory mediators (338). The use of MK2206 (an AKT inhibitor), RA (an Nrf2 inhibitor) and SnPP IX (an HO-1 inhibitor) further confirms that BNF inhibits the production of pro-inflammatory mediators by activating this pathway (338).
AMPK is an important cellular metabolic sensor and regulator (339) that is expressed in peripheral tissues and particularly in neuronal cells in the brain (340). Reports show that AMPK is hyperactivated in neurons from patients with AD (341) and impaired in the hippocampus of APP/PS1 mice (342), and is involved in Aβ clearance (343) and tau phosphorylation (344). AMPK has also been associated with neuroinflammation (345) and is dependent on microglia regulation (346). A link between AMPK and the Nrf2/ARE pathway has been suggested (347,348) and Bakkenolide B (349), the major constituent of Petasites japonicus leaves (350), has been found to activate the AMPK/Nrf2 signaling pathway. An in vitro study demonstrated that Bakkenolide B significantly reduces LPS-mediated production of pro-inflammatory factors in microglia and upregulates the expression of Nrf2-associated downstream effectors, such as NQO-1 and HO-1 (349). Knockdown of Nrf2, HO-1, and NQO-1 attenuated the anti-inflammatory effects of Bakkenolide B, whereas AMPK inhibitors reversed these effects. These findings indicate that Bakkenolide B induces the AMPK/Nrf2 signaling pathway to reduce neuroinflammation (349) (Table IV).
9. Conclusion and future perspectives
The pathogenesis of AD is a multifaceted process, but studies ranging from cellular and animal models, as well as studies involving patients with AD, have unequivocally established the pivotal role of neuroinflammation. Excessive activation of microglia releases inflammatory mediators that contribute to the pathological features of AD. Thus, inhibiting microglia-mediated inflammation is a promising approach to combat this disease.
Intracellular signaling pathways play a crucial role in maintaining cellular function and metabolism and are intricately associated with the pathogenesis of AD, including neuroinflammation. It is worth noting that these signaling pathways are complex, interconnected and capable of interacting with each other. By intervening in the pertinent signaling pathways through the use of drugs or inhibitors, it is possible to inhibit neuroinflammation and exert an effect on AD. Therefore, this review focused on neuroinflammation in AD and presented a comprehensive synthesis and summary of the mechanisms of action and potential signaling proteins linked with inhibitors, herbal medicines, and their active ingredients and metabolites, from the standpoint of signaling pathways (Fig. 5).
Figure 5.
Targeting inflammatory signaling pathways with drugs shows promise in AD treatment. By modulating various signaling pathways, drugs and inhibitors can effectively target microglia, neurons, and mice with AD. This targeted approach helps regulate inflammatory responses, promote neuronal survival, and restore functional recovery, ultimately alleviating symptoms of AD. AD, Alzheimer's disease; NLRP3, NOD-like receptor thermal protein domain associated protein 3; CREB, cyclic AMP response element binding; PPAR, Peroxisome proliferator-activated receptor; Nrf2, Nuclear factor erythroid 2-related factor 2.
Various drugs or inhibitors can regulate various signaling pathways, and multiple drugs can also target the same pathway. It is worth noting that NF-κB, MAPK, and NLRP3 are key signaling molecules targeted in neuroinflammation and have been extensively studied in drug discovery. By interfering with one or more of these signaling pathways, drugs can synergistically modulate multiple targets, achieve a balance between antioxidant and pro-inflammatory effects, and ultimately improve cognitive impairment in patients with AD (refer to Fig. 1 for details). Given the complexity of AD pathogenesis, drugs or inhibitors with multi-level and multi-target potential hold promise as a breakthrough in AD drug development. Exploring the anti-inflammatory effects of commonly used clinical drugs may broaden their potential application. However, current research is still primarily focused on animal and cellular experiments, with a focus on LPSor Aβ-stimulated BV2 microglia. Although a number of problems have prevented a number of drugs from entering clinical trials, inhibitors, that target neuroinflammation remain a potentially promising therapeutic option for AD.
In summary, the present review highlighted the prominent role of neuroinflammation in AD pathology and reviewed various anti-inflammatory inhibitors targeting molecular targets and signaling pathways. These inhibitors have shown significant potential as drug treatments for AD and have provided a foundation for the further development of novel AD therapeutics. However, the specificity, efficacy, safety, and availability of these inhibitors, natural ingredients and metabolites, are critical considerations for their clinical application. Moreover, further studies on their pharmacokinetic profiles and underlying mechanisms are necessary for the development of novel AD therapeutics. Despite these challenges, the potential benefits of these drugs underscore the need for continued research into their efficacy as treatments for AD.
Acknowledgments
Not applicable.
Abbreviations
- AD
Alzheimer's disease
- Aβ
amyloid β
- APP/PS1
amyloid-β protein/presenilin-1
- 5XFAD
5X familial Alzheimer's disease
- p-
phosphorylated
- AChE
acetylcholinesterase
- APN
Adiponectin
- AMPK
Adenosine 5′-monophosphate (AMP)-activated protein kinase
- ASC
apoptosis-associated speck-like protein
- BBB
blood-brain barrier
- BACE1
β-site amyloid precursor protein cleaving enzyme 1
- NF-κB
nuclear factor-kappa-B
- CNS
central nervous system
- COX-2
cyclooxygenase-2
- CHI3L1
chitinase-3 like-protein-1
- CREB
cyclic AMP response element binding
- ERK
extracellular signal-regulated kinases
- GFAP
glial fibrillary acidic protein
- Iba-1
ionized calcium binding adaptor molecule 1
- GSK-3β
glycogen synthase kinase-3β
- STAT3
signal transducers and activators of transcription 3
- GSDMD
gasdermin D
- HCK
Hematopoietic cell kinase
- HO-1
Haem oxygenase-1
- IKK
IκB kinase
- IL
interleukin
- iNOS
inducible nitric oxide synthase
- IFN-γ
interferon-γ
- AP-1
activator protein 1
- JNK
c-Jun NH2-terminal kinases
- JAK2
Janus kinase 2
- Keap1
Kelch-like ECH-associated protein 1
- LPS
lipopolysaccharide
- MyD88
myeloid differentiation factor 88
- MAPK
mitogen-activated protein kinase
- MK2
MAPK-activated protein kinase II
- MEK
mitogen-activated extracellular signal-regulated kinase
- PGE2
prostaglandin E2
- NLRP3
NOD-like receptor thermal protein domain associated protein 3
- NO
nitric oxide
- Nrf2
nuclear kinase II
- MEK
mitogen-activated extracellular signal-regulated kinase
- PGE2
prostaglandin E2
- NLRP3
NOD-like receptor thermal protein domain associated protein 3
- NO
nitric oxide
- Nrf2
nuclear factor erythroid 2-related factor 2
- PI3K
phosphoinositide 3-kinase
- PKC
protein kinase C
- PINK1
PTEN-induced kinase 1
- PPARs
peroxisome proliferator-activated receptors
- PTEN
phosphate and tensin homolog deleted on chromosome 10
- PKA
protein kinase A
- PKG
cGMP-dependent protein kinase
- RAGE
receptor for advanced glycation end products
- ROS
reactive oxygen species
- ROCK
Rho-dependent coiled-coil kinase
- SOCS
suppressor of cytokine signaling
- TLRs
Toll-like receptors
- WDFY1
WD repeat and FYVE domain-containing 1
Funding Statement
The present study was supported by the National Natural Science Foundation of China (grant nos. 82174470 and 82274318) and Natural Science Foundation of Tianjin City, China (grant no. 21JCQNJC01170).
Availability of data and materials
Data sharing is not applicable to this article, as no data sets were generated or analyzed during the current study.
Authors' contributions
YZ wrote the original draft of the manuscript. ZW, RZ and XZ reviewed and edited the manuscript. QG, JG and PX produced the diagrams and charts. XJ and LY contributed to the conception, design and drafting of the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Wang X, Iyaswamy A, Xu D, Krishnamoorthi S, Sreenivasmurthy SG, Yang Y, Li Y, Chen C, Li M, Li HW, Wong MS. Real-time detection and visualization of amyloid-β aggregates induced by hydrogen peroxide in cell and mouse models of Alzheimer's disease. ACS Appl Mater Interfaces. 2023;15:39–47. doi: 10.1021/acsami.2c07859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Shih YH, Tu LH, Chang TY, Ganesan K, Chang WW, Chang PS, Fang YS, Lin YT, Jin LW, Chen YR. TDP-43 interacts with amyloid-β, inhibits fibrillization, and worsens pathology in a model of Alzheimer's disease. Nat Commun. 2020;11:5950. doi: 10.1038/s41467-020-19786-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nasaruddin ML, Pan X, McGuinness B, Passmore P, Kehoe PG, Holscher C, Graham SF, Green BD. Evidence that parietal lobe fatty acids may be more profoundly affected in moderate Alzheimer's disease (AD) pathology than in severe AD pathology. Metabolites. 2018;8:69. doi: 10.3390/metabo8040069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chen YG. Research progress in the pathogenesis of Alzheimer's disease. Chin Med J (Engl) 2018;131:1618–1624. doi: 10.4103/0366-6999.235112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ardura-Fabregat A, Boddeke EWGM, Boza-Serrano A, Brioschi S, Castro-Gomez S, Ceyzériat K, Dansokho C, Dierkes T, Gelders G, Heneka MT, et al. Targeting neuroinflammation to treat Alzheimer's disease. CNS Drugs. 2017;31:1057–1082. doi: 10.1007/s40263-017-0483-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Finneran DJ, Nash KR. Neuroinflammation and fractalkine signaling in Alzheimer's disease. J Neuroinflammation. 2019;16:30. doi: 10.1186/s12974-019-1412-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Souza LC, Filho CB, Goes AT, Fabbro LD, de Gomes MG, Savegnago L, Oliveira MS, Jesse CR. Neuroprotective effect of physical exercise in a mouse model of Alzheimer's disease induced by β-amyloid 2013. 1-40 peptide. Neurotox Res. 2013;24:148–163. doi: 10.1007/s12640-012-9373-0. [DOI] [PubMed] [Google Scholar]
- 8.Janelsins MC, Mastrangelo MA, Oddo S, LaFerla FM, Federoff HJ, Bowers WJ. Early correlation of microglial activation with enhanced tumor necrosis factor-alpha and monocyte chemoattractant protein-1 expression specifically within the entorhinal cortex of triple transgenic Alzheimer's disease mice. J Neuroinflammation. 2005;2:23. doi: 10.1186/1742-2094-2-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ma K, Mount HTJ, McLaurin J. Region-specific distribution of β-amyloid peptide and cytokine expression in TgCRND8 mice. Neurosci Lett. 2011;492:5–10. doi: 10.1016/j.neulet.2011.01.035. [DOI] [PubMed] [Google Scholar]
- 10.Wu YY, Hsu JL, Wang HC, Wu SJ, Hong CJ, Cheng IHJ. Alterations of the neuroinflammatory markers IL-6 and TRAIL in Alzheimer's disease. Dement Geriatr Cogn Dis Extra. 2015;5:424–434. doi: 10.1159/000439214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Takeda S, Sato N, Ikimura K, Nishino H, Rakugi H, Morishita R. Increased blood-brain barrier vulnerability to systemic inflammation in an Alzheimer disease mouse model. Neurobiol Aging. 2013;34:2064–2070. doi: 10.1016/j.neurobiolaging.2013.02.010. [DOI] [PubMed] [Google Scholar]
- 12.Mann DM, Tucker CM, Yates PO. Alzheimer's disease: An olfactory connection? Mech Ageing Dev. 1988;42:1–15. doi: 10.1016/0047-6374(88)90058-9. [DOI] [PubMed] [Google Scholar]
- 13.Christen-Zaech S, Kraftsik R, Pillevuit O, Kiraly M, Martins R, Khalili K, Miklossy J. Early olfactory involvement in Alzheimer's disease. Can J Neurol Sci. 2003;30:20–25. doi: 10.1017/S0317167100002389. [DOI] [PubMed] [Google Scholar]
- 14.Abe Y, Ikegawa N, Yoshida K, Muramatsu K, Hattori S, Kawai K, Murakami M, Tanaka T, Goda W, Goto M, et al. Behavioral and electrophysiological evidence for a neuroprotective role of aquaporin-4 in the 5xFAD transgenic mice model. Acta Neuropathol Commun. 2020;8:67. doi: 10.1186/s40478-020-00936-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Walker DG, Dalsing-Hernandez JE, Campbell NA, Lue LF. Decreased expression of CD200 and CD200 receptor in Alzheimer's disease: A potential mechanism leading to chronic inflammation. Exp Neurol. 2009;215:5–19. doi: 10.1016/j.expneurol.2008.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ham HJ, Lee YS, Yun J, Son DJ, Lee HP, Han SB, Hong JT. K284-6111 alleviates memory impairment and neuroinflammation in Tg2576 mice by inhibition of chitinase-3-like 1 regulating ERK-dependent PTX3 pathway. J Neuroinflammation. 2020;17:350. doi: 10.1186/s12974-020-02022-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cullen NC, Malarstig AN, Stomrud E, Hansson O, Mattsson-Carlgren N. Accelerated inflammatory aging in Alzheimer's disease and its relation to amyloid, tau, and cognition. Sci Rep. 2021;11:1965. doi: 10.1038/s41598-021-81705-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Voet S, Mc Guire C, Hagemeyer N, Martens A, Schroeder A, Wieghofer P, Daems C, Staszewski O, Vande Walle L, Jordao MJC, et al. A20 critically controls microglia activation and inhibits inflammasome-dependent neuroinflammation. Nat Commun. 2018;9:2036. doi: 10.1038/s41467-018-04376-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Erny D, Hrabě de Angelis AL, Jaitin D, Wieghofer P, Staszewski O, David E, Keren-Shaul H, Mahlakoiv T, Jakobshagen K, Buch T, et al. Host microbiota constantly control maturation and function of microglia in the CNS. Nat Neurosci. 2015;18:965–977. doi: 10.1038/nn.4030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sierra A, Beccari S, Diaz-Aparicio I, Encinas JM, Comeau S, Tremblay MÈ. Surveillance, phagocytosis, and inflammation: How never-resting microglia influence adult hippocampal neurogenesis. Neural Plast. 2014;2014:610343. doi: 10.1155/2014/610343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ben-Yehuda H, Matcovitch-Natan O, Kertser A, Spinrad A, Prinz M, Amit I, Schwartz M. Maternal Type-I interferon signaling adversely affects the microglia and the behavior of the offspring accompanied by increased sensitivity to stress. Mol Psychiatr. 2020;25:1050–1067. doi: 10.1038/s41380-019-0604-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Town T, Nikolic V, Tan J. The microglial 'activation' continuum: From innate to adaptive responses. J Neuroinflammation. 2005;2:24. doi: 10.1186/1742-2094-2-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Leng F, Edison P. Neuroinflammation and microglial activation in Alzheimer disease: Where do we go from here? Nat Rev Neurol. 2021;17:157–172. doi: 10.1038/s41582-020-00435-y. [DOI] [PubMed] [Google Scholar]
- 24.Song WM, Joshita S, Zhou Y, Ulland TK, Gilfillan S, Colonna M. Humanized TREM2 mice reveal microglia-intrinsic and -extrinsic effects of R47H polymorphism. J Exp Med. 2018;215:745–760. doi: 10.1084/jem.20171529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tansey MG, McCoy MK, Frank-Cannon TC. Neuroinflammatory mechanisms in Parkinson's disease: Potential environmental triggers, pathways, and targets for early therapeutic intervention. Exp Neurol. 2007;208:1–25. doi: 10.1016/j.expneurol.2007.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jankowsky JL, Patterson PH. Cytokine and growth factor involvement in long-term potentiation. Mol Cell Neurosci. 1999;14:273–286. doi: 10.1006/mcne.1999.0792. [DOI] [PubMed] [Google Scholar]
- 27.Das S, Basu A. Inflammation: A new candidate in modulating adult neurogenesis. J Neurosci Res. 2008;86:1199–1208. doi: 10.1002/jnr.21585. [DOI] [PubMed] [Google Scholar]
- 28.Heneka MT, Carson MJ, El KJ, Landreth GE, Brosseron F, Feinstein DL, Jacobs AH, Wyss-Coray T, Vitorica J, Ransohoff RM, et al. Neuroinflammation in Alzheimer's disease. Lancet Neurol. 2015;14:388–405. doi: 10.1016/S1474-4422(15)70016-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Giridharan S, Srinivasan M. Mechanisms of NF-κB p65 and strategies for therapeutic manipulation. J Inflamm Res. 2018;11:407–419. doi: 10.2147/JIR.S140188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Noort AR, Tak PP, Tas SW. Non-canonical NF-κB signaling in rheumatoid arthritis: Dr Jekyll and Mr Hyde? Arthritis Res Ther. 2015;17:15. doi: 10.1186/s13075-015-0527-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mattson MP, Meffert MK. Roles for NF-kappaB in nerve cell survival, plasticity, and disease. Cell Death Differ. 2006;13:852–860. doi: 10.1038/sj.cdd.4401837. [DOI] [PubMed] [Google Scholar]
- 32.Miyauchi T, Uchida Y, Kadono K, Hirao H, Kawasoe J, Watanabe T, Ueda S, Okajima H, Terajima H, Uemoto S. Up-regulation of FOXO1 and reduced inflammation by β-hydroxybutyric acid are essential diet restriction benefits against liver injury. Proc Natl Acad Sci USA. 2019;116:13533–13542. doi: 10.1073/pnas.1820282116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Liu Y, Li D, Jiang Q, Zhang Q, Liu P, Wang L, Zong M, Zhang Q, Li H, An Y, et al. (3R, 7R)-7-Acetoxyl-9-Oxo-de-O-methyllasiodiplodin, a secondary metabolite of penicillium Sp., inhibits LPS-mediated inflammation in RAW 264.7 macrophages through blocking ERK/MAPKs and NF-κB signaling pathways. Inflammation. 2019;42:1463–1473. doi: 10.1007/s10753-019-01009-x. [DOI] [PubMed] [Google Scholar]
- 34.Christian F, Smith EL, Carmody RJ. The regulation of NF-κB subunits by phosphorylation. Cells. 2016;5:12. doi: 10.3390/cells5010012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Oeckinghaus A, Ghosh S. The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb Perspect Biol. 2009;1:a000034. doi: 10.1101/cshperspect.a000034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yano H, Sakai M, Matsukawa T, Yagi T, Naganuma T, Mitsushima M, Iida S, Inaba Y, Inoue H, Unoki-Kubota H, et al. PHD3 regulates glucose metabolism by suppressing stress-induced signalling and optimising gluconeogenesis and insulin signalling in hepatocytes. Sci Rep. 2018;8:14290. doi: 10.1038/s41598-018-32575-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Terai K, Matsuo A, McGeer PL. Enhancement of immunoreactivity for NF-kappa B in the hippocampal formation and cerebral cortex of Alzheimer's disease. Brain Res. 1996;735:159–168. doi: 10.1016/0006-8993(96)00310-1. [DOI] [PubMed] [Google Scholar]
- 38.Kotilinek LA, Westerman MA, Wang Q, Panizzon K, Lim GP, Simonyi A, Lesne S, Falinska A, Younkin LH, Younkin SG, et al. Cyclooxygenase-2 inhibition improves amyloid-beta-mediated suppression of memory and synaptic plasticity. Brain. 2008;131:651–664. doi: 10.1093/brain/awn008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kolesnick R, Golde DW. The sphingomyelin pathway in tumor necrosis factor and interleukin-1 signaling. Cell. 1994;77:325–328. doi: 10.1016/0092-8674(94)90147-3. [DOI] [PubMed] [Google Scholar]
- 40.Kitamura Y, Shimohama S, Ota T, Matsuoka Y, Nomura Y, Taniguchi T. Alteration of transcription factors NF-kappaB and STAT1 in Alzheimer's disease brains. Neurosci Lett. 1997;237:17–20. doi: 10.1016/S0304-3940(97)00797-0. [DOI] [PubMed] [Google Scholar]
- 41.Chen CH, Zhou W, Liu S, Deng Y, Cai F, Tone M, Tone Y, Tong Y, Song W. Increased NF-κB signalling up-regulates BACE1 expression and its therapeutic potential in Alzheimer's disease. Int J Neuropsychopharmacol. 2012;15:77–90. doi: 10.1017/S1461145711000149. [DOI] [PubMed] [Google Scholar]
- 42.Snow WM, Albensi BC. Neuronal gene targets of NF-κB and their dysregulation in Alzheimer's disease. Front Mol Neurosci. 2016;9:118. doi: 10.3389/fnmol.2016.00118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wang C, Fan L, Khawaja RR, Liu B, Zhan L, Kodama L, Chin M, Li Y, Le D, Zhou Y, et al. Microglial NF-κB drives tau spreading and toxicity in a mouse model of tauopathy. Nat Commun. 2022;13:1969. doi: 10.1038/s41467-022-29552-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.López N, Tormo C, De Blas I, Llinares I, Alom J. Oxidative stress in Alzheimer's disease and mild cognitive impairment with high sensitivity and specificity. J Alzheimers Dis. 2013;33:823–829. doi: 10.3233/JAD-2012-121528. [DOI] [PubMed] [Google Scholar]
- 45.Wang SW, Yang SG, Liu W, Zhang YX, Xu PX, Wang T, Ling TJ, Liu RT. Alpha-tocopherol quinine ameliorates spatial memory deficits by reducing beta-amyloid oligomers, neuroinflammation and oxidative stress in transgenic mice with Alzheimer's disease. Behav Brain Res. 2016;296:109–117. doi: 10.1016/j.bbr.2015.09.003. [DOI] [PubMed] [Google Scholar]
- 46.Sun HJ, Xue DD, Lu BZ, Li Y, Sheng LX, Zhu Z, Zhou YW, Zhang JX, Lin GJ, Lin SZ, et al. A novel synthetic steroid of 2β,3α,5α-trihydroxy-androst-6-one alleviates the loss of rat retinal ganglion cells caused by acute intraocular hypertension via inhibiting the inflammatory activation of microglia. Molecules. 2019;24:252. doi: 10.3390/molecules24020252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Belkhelfa M, Rafa H, Medjeber O, Arroul-Lammali A, Behairi N, Abada-Bendib M, Makrelouf M, Belarbi S, Masmoudi AN, Tazir M, Touil-Boukoffa C. IFN-γ and TNF-α are involved during Alzheimer disease progression and correlate with nitric oxide production: a study in Algerian patients. J Interferon Cytokine Res. 2014;34:839–847. doi: 10.1089/jir.2013.0085. [DOI] [PubMed] [Google Scholar]
- 48.Sui Y, Stehno-Bittel L, Li S, Loganathan R, Dhillon NK, Pinson D, Nath A, Kolson D, Narayan O, Buch S. CXCL10-induced cell death in neurons: Role of calcium dysregulation. Eur J Neurosci. 2006;23:957–964. doi: 10.1111/j.1460-9568.2006.04631.x. [DOI] [PubMed] [Google Scholar]
- 49.Xu PX, Wang SW, Yu XL, Su YJ, Wang T, Zhou WW, Zhang H, Wang YJ, Liu RT. Rutin improves spatial memory in Alzheimer's disease transgenic mice by reducing Aβ oligomer level and attenuating oxidative stress and neuroinflammation. Behav Brain Res. 2014;264:173–180. doi: 10.1016/j.bbr.2014.02.002. [DOI] [PubMed] [Google Scholar]
- 50.Budzynska B, Faggio C, Kruk-Slomka M, Samec D, Nabavi SF, Sureda A, Devi KP, Nabavi SM. Rutin as neuroprotective agent: From bench to bedside. Curr Med Chem. 2019;26:5152–5164. doi: 10.2174/0929867324666171003114154. [DOI] [PubMed] [Google Scholar]
- 51.Sun XY, Li LJ, Dong QX, Zhu J, Huang YR, Hou SJ, Yu XL, Liu RT. Rutin prevents tau pathology and neuroinflammation in a mouse model of Alzheimer's disease. J Neuroinflammation. 2021;18:131. doi: 10.1186/s12974-021-02182-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bernard CB, Krishanmurty HG, Chauret D, Durst T, Philogène BJ, Sánchez-Vindas P, Hasbun C, Poveda L, San Román L, Arnason JT. Insecticidal defenses of piperaceae from the neotropics. J Chem Ecol. 1995;21:801–814. doi: 10.1007/BF02033462. [DOI] [PubMed] [Google Scholar]
- 53.Gu SM, Lee HP, Ham YW, Son DJ, Kim HY, Oh KW, Han SB, Yun J, Hong JT. Piperlongumine improves lipopolysaccharide-induced amyloidogenesis by suppressing NF-KappaB pathway. Neuromolecular Med. 2018;20:312–327. doi: 10.1007/s12017-018-8495-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Son DJ, Lee JW, Lee YH, Song HS, Lee CK, Hong JT. Therapeutic application of anti-arthritis, pain-releasing, and anti-cancer effects of bee venom and its constituent compounds. Pharmacol Ther. 2007;115:246–270. doi: 10.1016/j.pharmthera.2007.04.004. [DOI] [PubMed] [Google Scholar]
- 55.Gu SM, Park MH, Hwang CJ, Song HS, Lee US, Han SB, Oh KW, Ham YW, Song MJ, Son DJ, Hong JT. Bee venom ameliorates lipopolysaccharide-induced memory loss by preventing NF-kappaB pathway. J Neuroinflammation. 2015;12:124. doi: 10.1186/s12974-015-0344-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lin CC, Hsu YF, Lin TC. Effects of punicalagin and punicalin on carrageenan-induced inflammation in rats. Am J Chin Med. 1999;27:371–376. doi: 10.1142/S0192415X99000422. [DOI] [PubMed] [Google Scholar]
- 57.Kim YE, Hwang CJ, Lee HP, Kim CS, Son DJ, Ham YW, Hellström M, Han SB, Kim HS, Park EK, Hong JT. Inhibitory effect of punicalagin on lipopolysaccharide-induced neuroinflammation, oxidative stress and memory impairment via inhibition of nuclear factor-kappaB. Neuropharmacology. 2017;117:21–32. doi: 10.1016/j.neuropharm.2017.01.025. [DOI] [PubMed] [Google Scholar]
- 58.Chen S, Jia J. Tenuifolin attenuates amyloid-β42-induced neuroinflammation in microglia through the NF-κB signaling pathway. J Alzheimers Dis. 2020;76:195–205. doi: 10.3233/JAD-200077. [DOI] [PubMed] [Google Scholar]
- 59.Hammad AS, Ravindran S, Khalil A, Munusamy S. Structure-activity relationship of piperine and its synthetic amide analogs for therapeutic potential to prevent experimentally induced ER stress in vitro. Cell Stress Chaperones. 2017;22:417–428. doi: 10.1007/s12192-017-0786-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Shahbazi S, Zakerali T, Frycz B, Kaur J. Impact of novel N-aryl substituted piperamide on NF-kappa B translocation as a potent anti-neuroinflammatory agent. Biomed Pharmacother. 2020;127:110199. doi: 10.1016/j.biopha.2020.110199. [DOI] [PubMed] [Google Scholar]
- 61.Shahbazi S, Zakerali T, Frycz BA, Kaur J. The critical role of piperamide derivative D4 in the regulation of inflammatory response by the microglia and astrocytic glial cells. Biomed Pharmacother. 2020;132:110895. doi: 10.1016/j.biopha.2020.110895. [DOI] [PubMed] [Google Scholar]
- 62.Hsu YL, Kuo PL, Chiang LC, Lin CC. Involvement of p53, nuclear factor kappaB and Fas/Fas ligand in induction of apoptosis and cell cycle arrest by saikosaponin d in human hepatoma cell lines. Cancer Lett. 2004;213:213–221. doi: 10.1016/j.canlet.2004.03.044. [DOI] [PubMed] [Google Scholar]
- 63.Park WH, Kang S, Piao Y, Pak CJ, Oh MS, Kim J, Kang MS, Pak YK. Ethanol extract of Bupleurum falcatum and saikosaponins inhibit neuroinflammation via inhibition of NF-κB. J Ethnopharmacol. 2015;174:37–44. doi: 10.1016/j.jep.2015.07.039. [DOI] [PubMed] [Google Scholar]
- 64.Leláková V, Béraud-Dufour S, Hošek J, Šmejkal K, Prachyawarakorn V, Pailee P, Widmann C, Václavík J, Coppola T, Mazella J, et al. Therapeutic potential of prenylated stilbenoid macasiamenene F through its anti-inflammatory and cytoprotective effects on LPS-challenged monocytes and microglia. J Ethnopharmacol. 2020;263:113147. doi: 10.1016/j.jep.2020.113147. [DOI] [PubMed] [Google Scholar]
- 65.Najm FJ, Madhavan M, Zaremba A, Shick E, Karl RT, Factor DC, Miller TE, Nevin ZS, Kantor C, Sargent A, et al. Drug-based modulation of endogenous stem cells promotes functional remyelination in vivo. Nature. 2015;522:216–220. doi: 10.1038/nature14335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Yeo IJ, Yun J, Son DJ, Han SB, Hong JT. Antifungal drug miconazole ameliorated memory deficits in a mouse model of LPS-induced memory loss through targeting iNOS. Cell Death Dis. 2020;11:623. doi: 10.1038/s41419-020-2619-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Solberg NO, Chamberlin R, Vigil JR, Deck LM, Heidrich JE, Brown DC, Brady CI, Vander Jagt TA, Garwood M, Bisoffi M, et al. Optical and SPION-enhanced MR imaging shows that trans-stilbene inhibitors of NF-κB concomitantly lower Alzheimer's disease plaque formation and microglial activation in AβPP/PS-1 transgenic mouse brain. J Alzheimers Dis. 2014;40:191–212. doi: 10.3233/JAD-131031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lindsay A, Hickman D, Srinivasan M. A nuclear factor-kappa B inhibiting peptide suppresses innate immune receptors and gliosis in a transgenic mouse model of Alzheimer's disease. Biomed Pharmacother. 2021;138:111405. doi: 10.1016/j.biopha.2021.111405. [DOI] [PubMed] [Google Scholar]
- 69.Choi JY, Yeo IJ, Kim KC, Choi WR, Jung JK, Han SB, Hong JT. K284-6111 prevents the amyloid beta-induced neuroinflammation and impairment of recognition memory through inhibition of NF-κB-mediated CHI3L1 expression. J Neuroinflammation. 2018;15:224. doi: 10.1186/s12974-018-1269-3. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 70.Lian W, Jia H, Xu L, Zhou W, Kang D, Liu A, Du G. Multi-protection of DL0410 in ameliorating cognitive defects in D-galactose induced aging mice. Front Aging Neurosci. 2017;9:409. doi: 10.3389/fnagi.2017.00409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Heynekamp JJ, Weber WM, Hunsaker LA, Gonzales AM, Orlando RA, Deck LM, Jagt DL. Substituted trans-stilbenes, including analogues of the natural product resveratrol, inhibit the human tumor necrosis factor alpha-induced activation of transcription factor nuclear factor kappaB. J Med Chem. 2006;49:7182–7189. doi: 10.1021/jm060630x. [DOI] [PubMed] [Google Scholar]
- 72.Ayroldi E, Riccardi C. Glucocorticoid-induced leucine zipper (GILZ): A new important mediator of glucocorticoid action. FASEB J. 2009;23:3649–3658. doi: 10.1096/fj.09-134684. [DOI] [PubMed] [Google Scholar]
- 73.Cannarile L, Zollo O, D'Adamio F, Ayroldi E, Marchetti C, Tabilio A, Bruscoli S, Riccardi C. Cloning, chromosomal assignment and tissue distribution of human GILZ, a glucocorticoid hormone-induced gene. Cell Death Differ. 2001;8:201–203. doi: 10.1038/sj.cdd.4400798. [DOI] [PubMed] [Google Scholar]
- 74.Bonneh-Barkay D, Wang G, Starkey A, Hamilton RL, Wiley CA. In vivo CHI3L1 (YKL-40) expression in astrocytes in acute and chronic neurological diseases. J Neuroinflammation. 2010;7:34. doi: 10.1186/1742-2094-7-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Muszyński P, Groblewska M, Kulczyńska-Przybik A, Kułakowska A, Mroczko B. YKL-40 as a potential biomarker and a possible target in therapeutic strategies of Alzheimer's disease. Curr Neuropharmacol. 2017;15:906–917. doi: 10.2174/1570159X15666170208124324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Di Rosa M, Malaguarnera L. Chitinase 3 like-1: An emerging molecule involved in diabetes and diabetic complications. Pathobiology. 2016;83:228–242. doi: 10.1159/000444855. [DOI] [PubMed] [Google Scholar]
- 77.Billod JM, Lacetera A, Guzmán-Caldentey J, Martín-Santamaría S. Computational approaches to Toll-like receptor 4 modulation. Molecules. 2016;21:994. doi: 10.3390/molecules21080994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Mertowski S, Grywalska E, Gosik K, Smarz-Widelska I, Hymos A, Dworacki G, Niedźwiedzka-Rystwej P, Drop B, Roliński J, Załuska W. TLR2 expression on select lymphocyte subsets as a new marker in glomerulonephritis. J Clin Med. 2020;9:541. doi: 10.3390/jcm9020541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Elmaidomy AH, Alhadrami HA, Amin E, Aly HF, Othman AM, Rateb ME, Hetta MH, Abdelmohsen UR, M Hassan H. Anti-inflammatory and antioxidant activities of terpene- and polyphenol-rich premna odorata leaves on alcohol-inflamed female wistar albino rat liver. Molecules. 2020;25:3116. doi: 10.3390/molecules25143116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Zhong X, Liu M, Yao W, Du K, He M, Jin X, Jiao L, Ma G, Wei B, Wei M. Epigallocatechin-3-gallate attenuates microglial inflammation and neurotoxicity by suppressing the activation of canonical and noncanonical inflammasome via TLR4/NF-κB pathway. Mol Nutr Food Res. 2019;63:e1801230. doi: 10.1002/mnfr.201801230. [DOI] [PubMed] [Google Scholar]
- 81.Zhou X, Yuan L, Zhao X, Hou C, Ma W, Yu H, Xiao R. Genistein antagonizes inflammatory damage induced by β-amyloid peptide in microglia through TLR4 and NF-κB. Nutrition. 2014;30:90–95. doi: 10.1016/j.nut.2013.06.006. [DOI] [PubMed] [Google Scholar]
- 82.Chen R, Wang Z, Zhi Z, Tian J, Zhao Y, Sun J. Targeting the TLR4/NF-κB pathway in β-amyloid-stimulated microglial cells: A possible mechanism that oxysophoridine exerts anti-oxidative and anti-inflammatory effects in an in vitro model of Alzheimer's disease. Brain Res Bull. 2021;175:150–157. doi: 10.1016/j.brainresbull.2021.07.019. [DOI] [PubMed] [Google Scholar]
- 83.Onishi S, Meguro S, Pervin M, Kitazawa H, Yoto A, Ishino M, Shimba Y, Mochizuki Y, Miura S, Tokimitsu I, Unno K. Green tea extracts attenuate brain dysfunction in high-fat-diet-fed SAMP8 mice. Nutrients. 2019;11:821. doi: 10.3390/nu11040821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Balázs A, Faisal Z, Csepregi R, Kőszegi T, Kriszt B, Szabó I, Poór M. In Vitro evaluation of the individual and combined cytotoxic and estrogenic effects of zearalenone, its reduced metabolites, alternariol, and genistein. Int J Mol Sci. 2021;22:6281. doi: 10.3390/ijms22126281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wang R, Deng X, Gao Q, Wu X, Han L, Gao X, Zhao S, Chen W, Zhou R, Li Z, Bai C. Sophora alopecuroides L.: An ethnopharmacological, phytochemical, and pharmacological review. J Ethnopharmacol. 2020;248:112172. doi: 10.1016/j.jep.2019.112172. [DOI] [PubMed] [Google Scholar]
- 86.Rui C, Yuxiang L, Ning J, Ningtian M, Qingluan Z, Yinju H, Ru Z, Lin M, Tao S, Jianqiang Y. Anti-apoptotic and neuroprotective effects of oxysophoridine on cerebral ischemia both in vivo and in vitro. Planta Med. 2013;79:916–923. doi: 10.1055/s-0032-1328705. [DOI] [PubMed] [Google Scholar]
- 87.Wang Y, Wang Y, Jia S, Dong Q, Chen Y, Lu S, Hou L. Effect of lipid-bound apolipoprotein A-I cysteine mutant on ATF3 in RAW264.7 cells. Biosci Rep. 2017;37:BSR20160398. doi: 10.1042/BSR20160398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zeng KW, Zhao MB, Ma ZZ, Jiang Y, Tu PF. Protosappanin A inhibits oxidative and nitrative stress via interfering the interaction of transmembrane protein CD14 with Toll-like receptor-4 in lipopolysaccharide-induced BV-2 microglia. Int Immunopharmacol. 2012;14:558–569. doi: 10.1016/j.intimp.2012.09.004. [DOI] [PubMed] [Google Scholar]
- 89.Mo Y, Sun YY, Liu KY. Autophagy and inflammation in ischemic stroke. Neural Regen Res. 2020;15:1388–1396. doi: 10.4103/1673-5374.274331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Capiralla H, Vingtdeux V, Zhao H, Sankowski R, Al-Abed Y, Davies P, Marambaud P. Resveratrol mitigates lipopolysaccharide- and Aβ-mediated microglial inflammation by inhibiting the TLR4/NF-κB/STAT signaling cascade. J Neurochem. 2012;120:461–472. doi: 10.1111/j.1471-4159.2011.07594.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Xu Y, Huang X, Huangfu B, Hu Y, Xu J, Gao R, Huang K, He X. Sulforaphane ameliorates nonalcoholic fatty liver disease induced by high-fat and high-fructose diet via LPS/TLR4 in the gut-liver axis. Nutrients. 2023;15:743. doi: 10.3390/nu15030743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Song J, Feng L, Zhong R, Xia Z, Zhang L, Cui L, Yan H, Jia X, Zhang Z. Icariside II inhibits the EMT of NSCLC cells in inflammatory microenvironment via down-regulation of Akt/NF-κB signaling pathway. Mol Carcinogen. 2017;56:36–48. doi: 10.1002/mc.22471. [DOI] [PubMed] [Google Scholar]
- 93.Lee KS, Lee HJ, Ahn KS, Kim SH, Nam D, Kim DK, Choi DY, Ahn KS, Lu J, Kim SH. Cyclooxygenase-2/prostaglandin E2 pathway mediates icariside II induced apoptosis in human PC-3 prostate cancer cells. Cancer Lett. 2009;280:93–100. doi: 10.1016/j.canlet.2009.02.024. [DOI] [PubMed] [Google Scholar]
- 94.Zhou J, Deng Y, Li F, Yin C, Shi J, Gong Q. Icariside II attenuates lipopolysaccharide-induced neuroinflammation through inhibiting TLR4/MyD88/NF-κB pathway in rats. Biomed Pharmacother. 2019;111:315–324. doi: 10.1016/j.biopha.2018.10.201. [DOI] [PubMed] [Google Scholar]
- 95.Zhou D, Zhou W, Song JK, Feng ZY, Yang RY, Wu S, Wang L, Liu AL, Du GH. DL0410, a novel dual cholinesterase inhibitor, protects mouse brains against Aβ-induced neuronal damage via the Akt/JNK signaling pathway. Acta Pharmacol Sin. 2016;37:1401–1412. doi: 10.1038/aps.2016.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Yang RY, Zhao G, Wang DM, Pang XC, Wang SB, Fang JS, Li C, Liu AL, Wu S, Du GH. DL0410 can reverse cognitive impairment, synaptic loss and reduce plaque load in APP/PS1 transgenic mice. Pharmacol Biochem Behav. 2015;139:15–26. doi: 10.1016/j.pbb.2015.10.009. [DOI] [PubMed] [Google Scholar]
- 97.Zhang B, Lian W, Zhao J, Wang Z, Liu A, Du G. DL0410 Alleviates memory impairment in D-galactose-induced aging rats by suppressing neuroinflammation via the TLR4/MyD88/NF-κB pathway. Oxid Med Cell Longev. 2021;2021:6521146. doi: 10.1155/2021/6521146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Mao J, Huang S, Liu S, Feng XL, Yu M, Liu J, Sun YE, Chen G, Yu Y, Zhao J, Pei G. A herbal medicine for Alzheimer's disease and its active constituents promote neural progenitor proliferation. Aging Cell. 2015;14:784–796. doi: 10.1111/acel.12356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Geng Y, Li C, Liu J, Xing G, Zhou L, Dong M, Li X, Niu Y. Beta-asarone improves cognitive function by suppressing neuronal apoptosis in the beta-amyloid hippocampus injection rats. Biol Pharm Bull. 2010;33:836–843. doi: 10.1248/bpb.33.836. [DOI] [PubMed] [Google Scholar]
- 100.Zhong J, Qiu X, Yu Q, Chen H, Yan C. A novel polysaccharide from Acorus tatarinowii protects against LPS-induced neuroinflammation and neurotoxicity by inhibiting TLR4-mediated MyD88/NF-κB and PI3K/Akt signaling pathways. Int J Biol Macromol. 2020;163:464–475. doi: 10.1016/j.ijbiomac.2020.06.266. [DOI] [PubMed] [Google Scholar]
- 101.Liu M, Guo H, Li Z, Zhang C, Zhang X, Cui Q, Tian J. Molecular level insight into the benefit of myricetin and dihydromyricetin uptake in patients with Alzheimer's diseases. Front Aging Neurosci. 2020;12:601603. doi: 10.3389/fnagi.2020.601603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Jing N, Li X. Dihydromyricetin attenuates inflammation through TLR4/NF-kappaB pathway. Open Med (Wars) 2019;14:719–725. doi: 10.1515/med-2019-0083. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 103.Gu RX, Gu H, Xie ZY, Wang JF, Arias HR, Wei DQ, Chou KC. Possible drug candidates for Alzheimer's disease deduced from studying their binding interactions with alpha7 nicotinic acetylcholine receptor. Med Chem. 2009;5:250–262. doi: 10.2174/157340609788185909. [DOI] [PubMed] [Google Scholar]
- 104.Shi S, Liang D, Chen Y, Xie Y, Wang Y, Wang L, Wang Z, Qiao Z. Gx-50 reduces β-amyloid-induced TNF-α, IL-1β, NO, and PGE2 expression and inhibits NF-κB signaling in a mouse model of Alzheimer's disease. Eur J Immunol. 2016;46:665–676. doi: 10.1002/eji.201545855. [DOI] [PubMed] [Google Scholar]
- 105.Zheng P, Huang C, Leng D, Sun B, Zhang XD. Transcriptome analysis of peripheral whole blood identifies crucial lncRNAs implicated in childhood asthma. Bmc Med Genomics. 2020;13:136. doi: 10.1186/s12920-020-00785-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Zhong X, Feng L, Xu WH, Wu X, Ding YD, Zhou Y, Lei CQ, Shu HB. The zinc-finger protein ZFYVE1 modulates TLR3-mediated signaling by facilitating TLR3 ligand binding. Cell Mol Immunol. 2020;17:741–752. doi: 10.1038/s41423-019-0265-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Wang Z, Xia Q, Liu X, Liu W, Huang W, Mei X, Luo J, Shan M, Lin R, Zou D, Ma Z. Phytochemistry, pharmacology, quality control and future research of Forsythia suspensa (Thunb.) Vahl: A review. J Ethnopharmacol. 2018;210:318–339. doi: 10.1016/j.jep.2017.08.040. [DOI] [PubMed] [Google Scholar]
- 108.Kong F, Jiang X, Wang R, Zhai S, Zhang Y, Wang D. Forsythoside B attenuates memory impairment and neuroinflammation via inhibition on NF-κB signaling in Alzheimer's disease. J Neuroinflammation. 2020;17:305. doi: 10.1186/s12974-020-01967-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Lu TC, Wu YH, Chen WY, Hung YC. Targeting oxidative stress and endothelial dysfunction using tanshinone IIA for the treatment of tissue inflammation and fibrosis. Oxid Med Cell Longev. 2022;2022:2811789. doi: 10.1155/2022/2811789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Lu BL, Li J, Zhou J, Li WW, Wu HF. Tanshinone IIA decreases the levels of inflammation induced by Aβ1-42 in brain tissues of Alzheimer's disease model rats. Neuroreport. 2016;27:883–893. doi: 10.1097/WNR.0000000000000618. [DOI] [PubMed] [Google Scholar]
- 111.Jiang P, Li C, Xiang Z, Jiao B. Tanshinone IIA reduces the risk of Alzheimer's disease by inhibiting iNOS, MMP-2 and NF-κBp65 transcription and translation in the temporal lobes of rat models of Alzheimer's disease. Mol Med Rep. 2014;10:689–694. doi: 10.3892/mmr.2014.2254. [DOI] [PubMed] [Google Scholar]
- 112.Maione F, Piccolo M, De Vita S, Chini MG, Cristiano C, De Caro C, Lippiello P, Miniaci MC, Santamaria R, Irace C, et al. Down regulation of pro-inflammatory pathways by tanshinone IIA and cryptotanshinone in a non-genetic mouse model of Alzheimer's disease. Pharmacol Res. 2018;129:482–490. doi: 10.1016/j.phrs.2017.11.018. [DOI] [PubMed] [Google Scholar]
- 113.Ding B, Lin C, Liu Q, He Y, Ruganzu JB, Jin H, Peng X, Ji S, Ma Y, Yang W. Tanshinone IIA attenuates neuroinflammation via inhibiting RAGE/NF-κB signaling pathway in vivo and in vitro. J Neuroinflammation. 2020;17:302. doi: 10.1186/s12974-020-01981-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Xing L, Tan ZR, Cheng JL, Huang WH, Zhang W, Deng W, Yuan CS, Zhou HH. Bioavailability and pharmacokinetic comparison of tanshinones between two formulations of Salvia miltiorrhiza in healthy volunteers. Sci Rep. 2017;7:4709. doi: 10.1038/s41598-017-02747-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Hu X, Zhang J, Deng L, Hu H, Hu J, Zheng G. Galactose-modified PH-sensitive niosomes for controlled release and hepatocellular carcinoma target delivery of tanshinone IIA. AAPS PharmSciTech. 2021;22:96. doi: 10.1208/s12249-021-01973-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Zhang X, Kang X, Du L, Zhang L, Huang Y, Wang J, Wang S, Chang Y, Liu Y, Zhao Y. Tanshinone IIA loaded chitosan nanoparticles decrease toxicity of β-amyloid peptide in a Caenorhabditis elegans model of disease. Free Radical Bio Med. 2022;193:81–94. doi: 10.1016/j.freeradbiomed.2022.09.030. [DOI] [PubMed] [Google Scholar]
- 117.Thundyil J, Pavlovski D, Sobey CG, Arumugam TV. Adiponectin receptor signalling in the brain. Brit J Pharmacol. 2012;165:313–327. doi: 10.1111/j.1476-5381.2011.01560.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Ng RC, Cheng OY, Jian M, Kwan JS, Ho PW, Cheng KK, Yeung PK, Zhou LL, Hoo RL, Chung SK, et al. Chronic adiponectin deficiency leads to Alzheimer's disease-like cognitive impairments and pathologies through AMPK inactivation and cerebral insulin resistance in aged mice. Mol Neurodegener. 2016;11:71. doi: 10.1186/s13024-016-0136-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Boza-Serrano A, Yang Y, Paulus A, Deierborg T. Innate immune alterations are elicited in microglial cells before plaque deposition in the Alzheimer's disease mouse model 5xFAD. Sci Rep. 2018;8:1550. doi: 10.1038/s41598-018-19699-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Jian M, Kwan JSC, Bunting M, Ng RCL, Chan KH. Adiponectin suppresses amyloid-β oligomer (AβO)-induced inflammatory response of microglia via AdipoR1-AMPK-NF-κB signaling pathway. J Neuroinflammation. 2019;16:110. doi: 10.1186/s12974-019-1492-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Chen X, Su J, Wang R, Hao R, Fu C, Chen J, Li J, Wang X. Structural optimization of cannabidiol as multifunctional cosmetic raw materials. Antioxidants (Basel) 2023;12:314. doi: 10.3390/antiox12020314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Malakoti F, Targhazeh N, Abadifard E, Zarezadeh R, Samemaleki S, Asemi Z, Younesi S, Mohammadnejad R, Hadi Hossini S, Karimian A, et al. DNA repair and damage pathways in mesothelioma development and therapy. Cancer Cell Int. 2022;22:176. doi: 10.1186/s12935-022-02597-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Schiffmann SN, Vanderhaeghen JJ. Distribution of cells containing mRNA encoding cholecystokinin in the rat central nervous system. J Comp Neurol. 1991;304:219–233. doi: 10.1002/cne.903040206. [DOI] [PubMed] [Google Scholar]
- 124.Hensley K, Floyd RA, Zheng NY, Nael R, Robinson KA, Nguyen X, Pye QN, Stewart CA, Geddes J, Markesbery WR, et al. p38 kinase is activated in the Alzheimer's disease brain. J Neurochem. 1999;72:2053–2058. doi: 10.1046/j.1471-4159.1999.0722053.x. [DOI] [PubMed] [Google Scholar]
- 125.Zhu X, Raina AK, Rottkamp CA, Aliev G, Perry G, Boux H, Smith MA. Activation and redistribution of c-jun N-terminal kinase/stress activated protein kinase in degenerating neurons in Alzheimer's disease. J Neurochem. 2001;76:435–441. doi: 10.1046/j.1471-4159.2001.00046.x. [DOI] [PubMed] [Google Scholar]
- 126.Jin Y, Yan EZ, Fan Y, Zong ZH, Qi ZM, Li Z. Sodium ferulate prevents amyloid-beta-induced neurotoxicity through suppression of p38 MAPK and upregulation of ERK-1/2 and Akt/protein kinase B in rat hippocampus. Acta Pharmacol Sin. 2005;26:943–951. doi: 10.1111/j.1745-7254.2005.00158.x. [DOI] [PubMed] [Google Scholar]
- 127.McDonald DR, Bamberger ME, Combs CK, Landreth GE. beta-Amyloid fibrils activate parallel mitogen-activated protein kinase pathways in microglia and THP1 monocytes. J Neurosci. 1998;18:4451–4460. doi: 10.1523/JNEUROSCI.18-12-04451.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Koistinaho M, Koistinaho J. Role of p38 and p44/42 mitogen-activated protein kinases in microglia. Glia. 2002;40:175–183. doi: 10.1002/glia.10151. [DOI] [PubMed] [Google Scholar]
- 129.Kim SH, Smith CJ, Van Eldik LJ. Importance of MAPK pathways for microglial pro-inflammatory cytokine IL-1 beta production. Neurobiol Aging. 2004;25:431–439. doi: 10.1016/S0197-4580(03)00126-X. [DOI] [PubMed] [Google Scholar]
- 130.Liang Z, Zhang B, Xu M, Morisseau C, Hwang SH, Hammock BD, Li QX. 1-Trifluoromethoxyphenyl-3-(1-propionylpiperidin-4-yl) urea, a selective and potent dual inhibitor of soluble epoxide hydrolase and p38 kinase intervenes in Alzheimer's signaling in human nerve cells. ACS Chem Neurosci. 2019;10:4018–4030. doi: 10.1021/acschemneuro.9b00271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Chang D, Li H, Qian C, Wang Y. DiOHF protects against doxorubicin-induced cardiotoxicity through ERK1 signaling pathway. Front Pharmacol. 2019;10:1081. doi: 10.3389/fphar.2019.01081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Lee HC, Yu HP, Liao CC, Chou AH, Liu FC. Escin protects against acetaminophen-induced liver injury in mice via attenuating inflammatory response and inhibiting ERK signaling pathway. Am J Transl Res. 2019;11:5170–5182. [PMC free article] [PubMed] [Google Scholar]
- 133.Sclip A, Tozzi A, Abaza A, Cardinetti D, Colombo I, Calabresi P, Salmona M, Welker E, Borsello T. c-Jun N-terminal kinase has a key role in Alzheimer disease synaptic dysfunction in vivo. Cell Death Dis. 2014;5:e1019. doi: 10.1038/cddis.2013.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Jeong YH, Li W, Go Y, Oh YC. Atractylodis rhizoma alba attenuates neuroinflammation in BV2 microglia upon LPS stimulation by inducing HO-1 activity and inhibiting NF-κB and MAPK. Int J Mol Sci. 2019;20:4015. doi: 10.3390/ijms20164015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Dang TK, Hong SM, Dao VT, Tran PTT, Tran HT, Do GH, Hai TN, Nguyet Pham HT, Kim SY. Anti-neuroinflammatory effects of alkaloid-enriched extract from Huperzia serrata on lipopolysaccharide-stimulated BV-2 microglial cells. Pharm Biol. 2023;61:135–143. doi: 10.1080/13880209.2022.2159450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Sun A, Liu M, Nguyen XV, Bing G. P38 MAP kinase is activated at early stages in Alzheimer's disease brain. Exp Neurol. 2003;183:394–405. doi: 10.1016/S0014-4886(03)00180-8. [DOI] [PubMed] [Google Scholar]
- 137.Schnöder L, Hao W, Qin Y, Liu S, Tomic I, Liu X, Fassbender K, Liu Y. Deficiency of neuronal p38α MAPK attenuates amyloid pathology in Alzheimer disease mouse and cell models through facilitating lysosomal degradation of BACE1. J Biol Chem. 2016;291:2067–2079. doi: 10.1074/jbc.M115.695916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Wu H, Hu B, Zhou X, Zhou C, Meng J, Yang Y, Zhao X, Shi Z, Yan S. Artemether attenuates LPS-induced inflammatory bone loss by inhibiting osteoclastogenesis and bone resorption via suppression of MAPK signaling pathway. Cell Death Dis. 2018;9:498. doi: 10.1038/s41419-018-0540-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Xing B, Bachstetter AD, Van Eldik LJ. Microglial p38α MAPK is critical for LPS-induced neuron degeneration, through a mechanism involving TNFα. Mol Neurodegener. 2011;6:84. doi: 10.1186/1750-1326-6-84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Munoz L, Ralay Ranaivo H, Roy SM, Hu W, Craft JM, McNamara LK, Chico LW, Van Eldik LJ, Watterson DM. A novel p38 alpha MAPK inhibitor suppresses brain proinflammatory cytokine up-regulation and attenuates synaptic dysfunction and behavioral deficits in an Alzheimer's disease mouse model. J Neuroinflammation. 2007;4:21. doi: 10.1186/1742-2094-4-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Maphis N, Jiang S, Xu G, Kokiko-Cochran ON, Roy SM, Van Eldik LJ, Watterson DM, Lamb BT, Bhaskar K. Selective suppression of the α isoform of p38 MAPK rescues late-stage tau pathology. Alzheimers Res Ther. 2016;8:54. doi: 10.1186/s13195-016-0221-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Haddad JJ. VX-745. Vertex pharmaceuticals. Curr Opin Investig Drugs. 2001;2:1070–1076. [PubMed] [Google Scholar]
- 143.Alam JJ. Selective brain-targeted antagonism of p38 MAPKα reduces hippocampal IL-1β levels and improves morris water maze performance in aged rats. J Alzheimers Dis. 2015;48:219–227. doi: 10.3233/JAD-150277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Hitti E, Iakovleva T, Brook M, Deppenmeier S, Gruber AD, Radzioch D, Clark AR, Blackshear PJ, Kotlyarov A, Gaestel M. Mitogen-activated protein kinase-activated protein kinase 2 regulates tumor necrosis factor mRNA stability and translation mainly by altering tristetraprolin expression, stability, and binding to adenine/uridine-rich element. Mol Cell Biol. 2006;26:2399–2407. doi: 10.1128/MCB.26.6.2399-2407.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Culbert AA, Skaper SD, Howlett DR, Evans NA, Facci L, Soden PE, Seymour ZM, Guillot F, Gaestel M, Richardson JC. MAPK-activated protein kinase 2 deficiency in microglia inhibits pro-inflammatory mediator release and resultant neurotoxicity. Relevance to neuroinflammation in a transgenic mouse model of Alzheimer disease. J Biol Chem. 2006;281:23658–23667. doi: 10.1074/jbc.M513646200. [DOI] [PubMed] [Google Scholar]
- 146.Brugnano JL, Chan BK, Seal BL, Panitch A. Cell-penetrating peptides can confer biological function: regulation of inflammatory cytokines in human monocytes by MK2 inhibitor peptides. J Control Release. 2011;155:128–133. doi: 10.1016/j.jconrel.2011.05.007. [DOI] [PubMed] [Google Scholar]
- 147.Jiang J, Wang Z, Liang X, Nie Y, Chang X, Xue H, Li S, Min C. Intranasal MMI-0100 attenuates Aβ1-42 - and LPS-induced neuroinflammation and memory impairments via the MK2 signaling pathway. Front Immunol. 2019;10:2707. doi: 10.3389/fimmu.2019.02707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Mittal D, Ali A, Md S, Baboota S, Sahni JK, Ali J. Insights into direct nose to brain delivery: current status and future perspective. Drug Deliv. 2014;21:75–86. doi: 10.3109/10717544.2013.838713. [DOI] [PubMed] [Google Scholar]
- 149.Mistry A, Stolnik S, Illum L. Nose-to-brain delivery: Investigation of the transport of nanoparticles with different surface characteristics and sizes in excised porcine olfactory epithelium. Mol Pharm. 2015;12:2755–2766. doi: 10.1021/acs.molpharmaceut.5b00088. [DOI] [PubMed] [Google Scholar]
- 150.Zakaria ZA, Patahuddin H, Mohamad AS, Israf DA, Sulaiman MR. In vivo anti-nociceptive and anti-inflammatory activities of the aqueous extract of the leaves of Piper sarmentosum. J Ethnopharmacol. 2010;128:42–48. doi: 10.1016/j.jep.2009.12.021. [DOI] [PubMed] [Google Scholar]
- 151.Chan EWL, Yeo ETY, Wong KWL, See ML, Wong KY, Gan SY. Piper sarmentosum Roxb. Root extracts confer neuroprotection by attenuating beta amyloid-induced pro-inflammatory cytokines released from microglial cells. Curr Alzheimer Res. 2019;16:251–260. doi: 10.2174/1567205016666190228124630. [DOI] [PubMed] [Google Scholar]
- 152.Wang X, Zhao X, Tang S. Inhibitory effects of EGb761 on the expression of matrix metalloproteinases (MMPs) and cartilage matrix destruction. Cell Stress Chaperon. 2015;20:781–786. doi: 10.1007/s12192-015-0600-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Meng M, Ai D, Sun L, Xu X, Cao X. EGb 761 inhibits Aβ1-42-induced neuroinflammatory response by suppressing P38 MAPK signaling pathway in BV-2 microglial cells. Neuroreport. 2019;30:434–440. doi: 10.1097/WNR.0000000000001223. [DOI] [PubMed] [Google Scholar]
- 154.Marchand G, Wambang N, Pellegrini S, Molinaro C, Martoriati A, Bousquet T, Markey A, Lescuyer-Rousseau A, Bodart JF, Cailliau K, et al. Effects of ferrocenyl 4-(Imino)-1,4-dihydro-quinolines on xenopus laevis prophase I-arrested oocytes: Survival and hormonal-induced M-phase entry. Int J Mol Sci. 2020;21:3049. doi: 10.3390/ijms21093049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Chiu YJ, Hsieh YH, Lin TH, Lee GC, Hsieh-Li HM, Sun YC, Chen CM, Chang KH, Lee-Chen GJ. Novel compound VB-037 inhibits Aβ aggregation and promotes neurite outgrowth through enhancement of HSP27 and reduction of P38 and JNK-mediated inflammation in cell models for Alzheimer's disease. Neurochem Int. 2019;125:175–186. doi: 10.1016/j.neuint.2019.01.021. [DOI] [PubMed] [Google Scholar]
- 156.Czarnecka K, Girek M, Maciejewska K, Skibiński R, Jończyk J, Bajda M, Kabziński J, Sołowiej P, Majsterek I, Szymański P. New cyclopentaquinoline hybrids with multifunctional capacities for the treatment of Alzheimer's disease. J Enzym Inhib Med Chem. 2017;33:158–170. doi: 10.1080/14756366.2017.1406485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Olajide OA, Bhatia HS, de Oliveira AC, Wright CW, Fiebich BL. Inhibition of neuroinflammation in LPS-activated microglia by cryptolepine. Evid Based Complement Alternat Med. 2013;2013:459723. doi: 10.1155/2013/459723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Castro-Torres RD, Busquets O, Parcerisas A, Verdaguer E, Olloquequi J, Ettcheto M, Beas-Zarate C, Folch J, Camins A, Auladell C. Involvement of JNK1 in neuronal polarization during brain development. Cells. 2020;9:1897. doi: 10.3390/cells9081897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Ma Z, Zang T, Birnbaum SG, Wang Z, Johnson JE, Zhang CL, Parada LF. TrkB dependent adult hippocampal progenitor differentiation mediates sustained ketamine antidepressant response. Nat Commun. 2017;8:1668. doi: 10.1038/s41467-017-01709-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Guo YJ, Pan WW, Liu SB, Shen ZF, Xu Y, Hu LL. ERK/MAPK signalling pathway and tumorigenesis. Exp Ther Med. 2020;19:1997–2007. doi: 10.3892/etm.2020.8454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Paquet C, Nicoll JA, Love S, Mouton-Liger F, Holmes C, Hugon J, Boche D. Downregulated apoptosis and autophagy after anti-Aβ immunotherapy in Alzheimer's disease. Brain Pathol. 2018;28:603–610. doi: 10.1111/bpa.12567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Yuan Y, Chen J, Ge X, Deng J, Xu X, Zhao Y, Wang H. Activation of ERK-Drp1 signaling promotes hypoxia-induced Aβ accumulation by upregulating mitochondrial fission and BACE1 activity. Febs Open Bio. 2021;11:2740–2755. doi: 10.1002/2211-5463.13273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Ho WC, Hsu CC, Huang HJ, Wang HT, Lin AMY. Anti-inflammatory effect of AZD6244 on acrolein-induced neuroinflammation. Mol Neurobiol. 2020;57:88–95. doi: 10.1007/s12035-019-01758-8. [DOI] [PubMed] [Google Scholar]
- 164.Qiu Z, Lu P, Wang K, Zhao X, Li Q, Wen J, Zhang H, Li R, Wei H, Lv Y, et al. Dexmedetomidine inhibits neuroinflammation by altering microglial M1/M2 polarization through MAPK/ERK pathway. Neurochem Res. 2020;45:345–353. doi: 10.1007/s11064-019-02922-1. [DOI] [PubMed] [Google Scholar]
- 165.Goldfarb G, Doan Ba Tri, Duran A. Human placenta for chronic leg ulcers. Lancet. 1980;2:40. doi: 10.1016/S0140-6736(80)92921-9. [DOI] [PubMed] [Google Scholar]
- 166.Lee KW, Ji HM, Kim DW, Choi SM, Kim S, Yang EJ. Effects of Hominis placenta on LPS-induced cell toxicity in BV2 microglial cells. J Ethnopharmacol. 2013;147:286–292. doi: 10.1016/j.jep.2013.02.033. [DOI] [PubMed] [Google Scholar]
- 167.Filocamo A, Bisignano C, Ferlazzo N, Cirmi S, Mandalari G, Navarra M. In vitro effect of bergamot (Citrus bergamia) juice against cagA-positive and-negative clinical isolates of Helicobacter pylori. BMC Complement Altern Med. 2015;15:256. doi: 10.1186/s12906-015-0769-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Currò M, Risitano R, Ferlazzo N, Cirmi S, Gangemi C, Caccamo D, Ientile R, Navarra M. Citrus bergamia juice extract attenuates β-amyloid-induced pro-inflammatory activation of THP-1 cells through MAPK and AP-1 pathways. Sci Rep. 2016;6:20809. doi: 10.1038/srep20809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Eglitis MA, Mezey E. Hematopoietic cells differentiate into both microglia and macroglia in the brains of adult mice. Proc Natl Acad Sci USA. 1997;94:4080–4085. doi: 10.1073/pnas.94.8.4080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Hickey WF, Kimura H. Perivascular microglial cells of the CNS are bone marrow-derived and present antigen in vivo. Science. 1988;239:290–292. doi: 10.1126/science.3276004. [DOI] [PubMed] [Google Scholar]
- 171.Tao M, Zheng D, Liang X, Wu D, Hu K, Jin J, He Q. Tripterygium glycoside suppresses epithelial-to-mesenchymal transition of diabetic kidney disease podocytes by targeting autophagy through the mTOR/Twist1 pathway. Mol Med Rep. 2021;24:592. doi: 10.3892/mmr.2021.12231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Tang L, Xiang Q, Xiang J, Zhang Y, Li J. Tripterygium glycoside ameliorates neuroinflammation in a mouse model of Aβ25-35-induced Alzheimer's disease by inhibiting the phosphorylation of IκBα and p38. Bioengineered. 2021;12:8540–8554. doi: 10.1080/21655979.2021.1987082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Cao X, Jin Y, Zhang H, Yu L, Bao X, Li F, Xu Y. The antiinflammatory effects of 4-[(5-Bromo-3-chloro-2-hydroxybenzyl) amino]-2-hydroxybenzoic acid in lipopolysaccharide-activated primary microglial cells. Inflammation. 2018;41:530–540. doi: 10.1007/s10753-017-0709-z. [DOI] [PubMed] [Google Scholar]
- 174.Kim ME, Jung I, Na JY, Lee Y, Lee J, Lee JS, Lee JS. Pseudane-VII regulates LPS-induced neuroinflammation in brain microglia cells through the inhibition of iNOS expression. Molecules. 2018;23:3196. doi: 10.3390/molecules23123196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Zhao H, Wang SL, Qian L, Jin JL, Li H, Xu Y, Zhu XL. Diammonium glycyrrhizinate attenuates Aβ(1-42)-induced neuroinflammation and regulates MAPK and NF-κB pathways in vitro and in vivo. CNS Neurosci Ther. 2013;19:117–124. doi: 10.1111/cns.12043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Ramana KV, Srivastava SK. Aldose reductase: A novel therapeutic target for inflammatory pathologies. Int J Biochem Cell Biol. 2010;42:17–20. doi: 10.1016/j.biocel.2009.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Song XM, Yu Q, Dong X, Yang HO, Zeng KW, Li J, Tu PF. Aldose reductase inhibitors attenuate β-amyloid-induced TNF-α production in microlgia via ROS-PKC-mediated NF-κB and MAPK pathways. Int Immunopharmacol. 2017;50:30–37. doi: 10.1016/j.intimp.2017.06.005. [DOI] [PubMed] [Google Scholar]
- 178.Meza CA, La Favor JD, Kim DH, Hickner RC. Endothelial dysfunction: Is there a hyperglycemia-induced imbalance of NOX and NOS? Int J Mol Sci. 2019;20:3775. doi: 10.3390/ijms20153775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Lee J, Narayan VP, Hong EY, Whang WK, Park T. Artemisia iwayomogi extract attenuates high-fat diet-induced hypertriglyceridemia in mice: Potential involvement of the adiponectin-AMPK pathway and very low density lipoprotein assembly in the liver. Int J Mol Sci. 2017;18:1762. doi: 10.3390/ijms18081762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Ju IG, Huh E, Kim N, Lee S, Choi JG, Hong J, Oh MS. Artemisiae iwayomogii herba inhibits lipopolysaccharide-induced neuroinflammation by regulating NF-κB and MAPK signaling pathways. Phytomedicine. 2021;84:153501. doi: 10.1016/j.phymed.2021.153501. [DOI] [PubMed] [Google Scholar]
- 181.Hilliard A, Mendonca P, Soliman KFA. Involvement of NFkB and MAPK signaling pathways in the preventive effects of Ganoderma lucidum on the inflammation of BV-2 microglial cells induced by LPS. J Neuroimmunol. 2020;345:577269. doi: 10.1016/j.jneuroim.2020.577269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Cheung WM, Hui WS, Chu PW, Chiu SW, Ip NY. Ganoderma extract activates MAP kinases and induces the neuronal differentiation of rat pheochromocytoma PC12 cells. FEBS Lett. 2000;486:291–296. doi: 10.1016/S0014-5793(00)02317-6. [DOI] [PubMed] [Google Scholar]
- 183.Geng X, Zhong D, Su L, Yang B. Preventive and therapeutic effect of ganoderma (Lingzhi) on renal diseases and clinical applications. Adv Exp Med Biol. 2019;1182:243–262. doi: 10.1007/978-981-32-9421-9_10. [DOI] [PubMed] [Google Scholar]
- 184.Chen LG, Jan YS, Tsai PW, Norimoto H, Michihara S, Murayama C, Wang CC. Anti-inflammatory and antinociceptive constituents of atractylodes japonica koidzumi. J Agric Food Chem. 2016;64:2254–2262. doi: 10.1021/acs.jafc.5b05841. [DOI] [PubMed] [Google Scholar]
- 185.Tang JJ, Wang MR, Dong S, Huang LF, He QR, Gao JM. 1,10-Seco-Eudesmane sesquiterpenoids as a new type of anti-neuroinflammatory agents by suppressing TLR4/NF-κB/MAPK pathways. Eur J Med Chem. 2021;224:113713. doi: 10.1016/j.ejmech.2021.113713. [DOI] [PubMed] [Google Scholar]
- 186.Choi MS, Jung UJ, Kim HJ, Do GM, Jeon SM, Kim MJ, Lee MK. Du-zhong (Eucommia ulmoides Oliver) leaf extract mediates hypolipidemic action in hamsters fed a high-fat diet. Am J Chin Med. 2008;36:81–93. doi: 10.1142/S0192415X08005606. [DOI] [PubMed] [Google Scholar]
- 187.He X, Wang J, Li M, Hao D, Yang Y, Zhang C, He R, Tao R. Eucommia ulmoides Oliv.: Ethnopharmacology, phytochemistry and pharmacology of an important traditional Chinese medicine. J Ethnopharmacol. 2014;151:78–92. doi: 10.1016/j.jep.2013.11.023. [DOI] [PubMed] [Google Scholar]
- 188.Han R, Yuan T, Yang Z, Zhang Q, Wang WW, Lin LB, Zhu MQ, Gao JM. Ulmoidol, an unusual nortriterpenoid from Eucommia ulmoides Oliv. Leaves prevents neuroinflammation by targeting the PU.1 transcriptional signaling pathway. Bioorg Chem. 2021;116:105345. doi: 10.1016/j.bioorg.2021.105345. [DOI] [PubMed] [Google Scholar]
- 189.Zhang C, Hu L, Liu D, Huang J, Lin W. Circumdatin D exerts neuroprotective effects by attenuating LPS-induced pro-inflammatory responses and downregulating acetylcholinesterase activity in vitro and in vivo. Front Pharmacol. 2020;11:760. doi: 10.3389/fphar.2020.00760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Gong P, Deng F, Zhang W, Ji J, Liu J, Sun Y, Hu J. Tectorigenin attenuates the MPP+-induced SH-SY5Y cell damage, indicating a potential beneficial role in Parkinson's disease by oxidative stress inhibition. Exp Ther Med. 2017;14:4431–4437. doi: 10.3892/etm.2017.5049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Oh KB, Kang H, Matsuoka H. Detection of antifungal activity in Belamcanda chinensis by a single-cell bioassay method and isolation of its active compound, tectorigenin. Biosci Biotechnol Biochem. 2001;65:939–942. doi: 10.1271/bbb.65.939. [DOI] [PubMed] [Google Scholar]
- 192.Lim HS, Kim YJ, Kim BY, Park G, Jeong SJ. The Anti-neuroinflammatory activity of tectorigenin pretreatment via downregulated NF-κB and ERK/JNK pathways in BV-2 microglial and microglia inactivation in mice with lipopolysaccharide. Front Pharmacol. 2018;9:462. doi: 10.3389/fphar.2018.00462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Wang BR, Shi JQ, Ge NN, Ou Z, Tian YY, Jiang T, Zhou JS, Xu J, Zhang YD. PM2.5 exposure aggravates oligomeric amyloid beta-induced neuronal injury and promotes NLRP3 inflammasome activation in an in vitro model of Alzheimer's disease. J Neuroinflammation. 2018;15:132. doi: 10.1186/s12974-018-1178-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Shao BZ, Xu ZQ, Han BZ, Su DF, Liu C. NLRP3 inflammasome and its inhibitors: A review. Front Pharmacol. 2015;6:262. doi: 10.3389/fphar.2015.00262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Feng YS, Tan ZX, Wu LY, Dong F, Zhang F. The involvement of NLRP3 inflammasome in the treatment of Alzheimer's disease. Ageing Res Rev. 2020;64:101192. doi: 10.1016/j.arr.2020.101192. [DOI] [PubMed] [Google Scholar]
- 196.Heneka MT, Kummer MP, Stutz A, Delekate A, Schwartz S, Vieira-Saecker A, Griep A, Axt D, Remus A, Tzeng TC, et al. NLRP3 is activated in Alzheimer's disease and contributes to pathology in APP/PS1 mice. Nature. 2013;493:674–678. doi: 10.1038/nature11729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Halle A, Hornung V, Petzold GC, Stewart CR, Monks BG, Reinheckel T, Fitzgerald KA, Latz E, Moore KJ, Golenbock DT. The NALP3 inflammasome is involved in the innate immune response to amyloid-beta. Nat Immunol. 2008;9:857–865. doi: 10.1038/ni.1636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Lonnemann N, Hosseini S, Marchetti C, Skouras DB, Stefanoni D, D'Alessandro A, Dinarello CA, Korte M. The NLRP3 inflammasome inhibitor OLT1177 rescues cognitive impairment in a mouse model of Alzheimer's disease. Proc Natl Acad Sci USA. 2020;117:32145–32154. doi: 10.1073/pnas.2009680117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Marchetti C, Swartzwelter B, Gamboni F, Neff CP, Richter K, Azam T, Carta S, Tengesdal I, Nemkov T, D'Alessandro A, et al. OLT1177, a β-sulfonyl nitrile compound, safe in humans, inhibits the NLRP3 inflammasome and reverses the metabolic cost of inflammation. Proc Natl Acad Sci USA. 2018;115:E1530–E1539. doi: 10.1073/pnas.1716095115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Klück V, Jansen TLTA, Janssen M, Comarniceanu A, Efdé M, Tengesdal IW, Schraa K, Cleophas MCP, Scribner CL, Skouras DB, et al. Dapansutrile, an oral selective NLRP3 inflammasome inhibitor, for treatment of gout flares: An open-label, dose-adaptive, proof-of-concept, phase 2a trial. Lancet Rheumatol. 2020;2:e270–e280. doi: 10.1016/S2665-9913(20)30065-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Dempsey C, Rubio Araiz A, Bryson KJ, Finucane O, Larkin C, Mills EL, Robertson AAB, Cooper MA, O'Neill LAJ, Lynch MA. Inhibiting the NLRP3 inflammasome with MCC950 promotes non-phlogistic clearance of amyloid-β and cognitive function in APP/PS1 mice. Brain Behav Immun. 2017;61:306–316. doi: 10.1016/j.bbi.2016.12.014. [DOI] [PubMed] [Google Scholar]
- 202.Fekete C, Vastagh C, Dénes Á, Hrabovszky E, Nyiri G, Kalló I, Liposits Z, Sárvári M. Chronic amyloid β oligomer infusion evokes sustained inflammation and microglial changes in the rat hippocampus via NLRP3. Neuroscience. 2019;405:35–46. doi: 10.1016/j.neuroscience.2018.02.046. [DOI] [PubMed] [Google Scholar]
- 203.Kuwar R, Rolfe A, Di L, Blevins H, Xu Y, Sun X, Bloom GS, Zhang S, Sun D. A novel inhibitor targeting NLRP3 inflammasome reduces neuropathology and improves cognitive function in Alzheimer's disease transgenic mice. J Alzheimers Dis. 2021;82:1769–1783. doi: 10.3233/JAD-210400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Zhang Y, Zhao Y, Zhang J, Gao Y, Li S, Chang C, Yu D, Yang G. Ginkgolide B inhibits NLRP3 inflammasome activation and promotes microglial M2 polarization in Aβ1-42 -induced microglia cells. Neurosci Lett. 2021;764:136206. doi: 10.1016/j.neulet.2021.136206. [DOI] [PubMed] [Google Scholar]
- 205.Gu JH, Ge JB, Li M, Wu F, Zhang W, Qin ZH. Inhibition of NF-κB activation is associated with anti-inflammatory and anti-apoptotic effects of ginkgolide B in a mouse model of cerebral ischemia/reperfusion injury. Eur J Pharm Sci. 2012;47:652–660. doi: 10.1016/j.ejps.2012.07.016. [DOI] [PubMed] [Google Scholar]
- 206.Kaur N, Dhiman M, Perez-Polo JR, Mantha AK. Ginkgolide B revamps neuroprotective role of apurinic/apyrimidinic endonuclease 1 and mitochondrial oxidative phosphorylation against Aβ25-35-induced neurotoxicity in human neuroblastoma cells. J Neurosci Res. 2015;93:938–947. doi: 10.1002/jnr.23565. [DOI] [PubMed] [Google Scholar]
- 207.Zhang HR, Peng JH, Cheng XB, Shi BZ, Zhang MY, Xu RX. Paeoniflorin atttenuates amyloidogenesis and the inflammatory responses in a transgenic mouse model of Alzheimer's disease. Neurochem Res. 2015;40:1583–1592. doi: 10.1007/s11064-015-1632-z. [DOI] [PubMed] [Google Scholar]
- 208.Liu HQ, Zhang WY, Luo XT, Ye Y, Zhu XZ. Paeoniflorin attenuates neuroinflammation and dopaminergic neurodegeneration in the MPTP model of Parkinson's disease by activation of adenosine A1 receptor. Brit J Pharmacol. 2006;148:314–325. doi: 10.1038/sj.bjp.0706732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Liu DZ, Xie KQ, Ji XQ, Ye Y, Jiang CL, Zhu XZ. Neuroprotective effect of paeoniflorin on cerebral ischemic rat by activating adenosine A1 receptor in a manner different from its classical agonists. Br J Pharmacol. 2005;146:604–611. doi: 10.1038/sj.bjp.0706335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.English BK, Ihle JN, Myracle A, Yi T. Hck tyrosine kinase activity modulates tumor necrosis factor production by murine macrophages. J Exp Med. 1993;178:1017–1022. doi: 10.1084/jem.178.3.1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Kong X, Liao Y, Zhou L, Zhang Y, Cheng J, Yuan Z, Wang S. Hematopoietic cell kinase (HCK) is essential for NLRP3 inflammasome activation and lipopolysaccharide-induced inflammatory response in vivo. Front Pharmacol. 2020;11:581011. doi: 10.3389/fphar.2020.581011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Cui W, Sun C, Ma Y, Wang S, Wang X, Zhang Y. Inhibition of TLR4 induces M2 microglial polarization and provides neuroprotection via the NLRP3 inflammasome in Alzheimer's disease. Front Neurosci. 2020;14:444. doi: 10.3389/fnins.2020.00444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Hua F, Tang H, Wang J, Prunty MC, Hua X, Sayeed I, Stein DG. TAK-242, an antagonist for Toll-like receptor 4, protects against acute cerebral ischemia/reperfusion injury in mice. J Cereb Blood Flow Metab. 2015;35:536–542. doi: 10.1038/jcbfm.2014.240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Liu Y, Dai Y, Li Q, Chen C, Chen H, Song Y, Hua F, Zhang Z. Beta-amyloid activates NLRP3 inflammasome via TLR4 in mouse microglia. Neurosci Lett. 2020;736:135279. doi: 10.1016/j.neulet.2020.135279. [DOI] [PubMed] [Google Scholar]
- 215.Poulose SM, Thangthaeng N, Miller MG, Shukitt-Hale B. Effects of pterostilbene and resveratrol on brain and behavior. Neurochem Int. 2015;89:227–233. doi: 10.1016/j.neuint.2015.07.017. [DOI] [PubMed] [Google Scholar]
- 216.Li Q, Chen L, Liu X, Li X, Cao Y, Bai Y, Qi F. Pterostilbene inhibits amyloid-β-induced neuroinflammation in a microglia cell line by inactivating the NLRP3/caspase-1 inflammasome pathway. J Cell Biochem. 2018;119:7053–7062. doi: 10.1002/jcb.27023. [DOI] [PubMed] [Google Scholar]
- 217.Wang SY, Liu Y, Li XM, Algradi AM, Jiang H, Sun YP, Guan W, Pan J, Kuang HX, Yang BY. Discovery of active ingredients targeted TREM2 by SPR biosensor-UPLC/MS recognition system, and investigating the mechanism of anti-neuroinflammatory activity on the lignin-amides from Datura metel seeds. Molecules. 2021;26:5946. doi: 10.3390/molecules26195946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Liu Y, Yang X, Lei Q, Li Z, Hu J, Wen X, Wang H, Liu Z. PEG-PEI/siROCK2 protects against Aβ42-induced neurotoxicity in primary neuron cells for Alzheimer disease. Cell Mol Neurobiol. 2015;35:841–848. doi: 10.1007/s10571-015-0178-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Liu Y, Zhang H, Peng A, Cai X, Wang Y, Tang K, Wu X, Liang Y, Wang L, Li Z. PEG-PEI/siROCK2 inhibits Aβ42-induced microglial inflammation via NLRP3/caspase 1 pathway. Neuroreport. 2022;33:26–32. doi: 10.1097/WNR.0000000000001752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Schneider KS, Groß CJ, Dreier RF, Saller BS, Mishra R, Gorka O, Heilig R, Meunier E, Dick MS, Ćiković T, et al. The inflammasome drives GSDMD-independent secondary pyroptosis and IL-1 release in the absence of caspase-1 protease activity. Cell Rep. 2017;21:3846–3859. doi: 10.1016/j.celrep.2017.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Han C, Hu Q, Yu A, Jiao Q, Yang Y. Mafenide derivatives inhibit neuroinflammation in Alzheimer's disease by regulating pyroptosis. J Cell Mol Med. 2021;25:10534–10542. doi: 10.1111/jcmm.16984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Wang CZ, Du GJ, Zhen Z, Calway T, Yuan CS. Significant dose differences in donepezil purchased from the United States and Canada. Ann Intern Med. 2011;155:279–280. doi: 10.7326/0003-4819-155-4-201108160-00026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Birks JS, Harvey RJ. Donepezil for dementia due to Alzheimer's disease. Cochrane Database Syst Rev. 2018;6:CD001190. doi: 10.1002/14651858.CD001190.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Wang H, Zong Y, Han Y, Zhao J, Liu H, Liu Y. Compared of efficacy and safety of high-dose donepezil vs standard-dose donepezil among elderly patients with Alzheimer's disease: A systematic review and meta-analysis. Expert Opin Drug Saf. 2022;21:407–415. doi: 10.1080/14740338.2022.2027905. [DOI] [PubMed] [Google Scholar]
- 225.Kim J, Lee HJ, Park SK, Park JH, Jeong HR, Lee S, Lee H, Seol E, Hoe HS. Donepezil regulates LPS and Aβ-stimulated neuroinflammation through MAPK/NLRP3 inflammasome/STAT3 signaling. Int J Mol Sci. 2021;22:10637. doi: 10.3390/ijms221910637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Kim HG, Moon M, Choi JG, Park G, Kim AJ, Hur J, Lee KT, Oh MS. Donepezil inhibits the amyloid-beta oligomer-induced microglial activation in vitro and in vivo. Neurotoxicology. 2014;40:23–32. doi: 10.1016/j.neuro.2013.10.004. [DOI] [PubMed] [Google Scholar]
- 227.Chen M, Chen Z, Wang Y, Tan Z, Zhu C, Li Y, Han Z, Chen L, Gao R, Liu L, Chen Q. Mitophagy receptor FUNDC1 regulates mitochondrial dynamics and mitophagy. Autophagy. 2016;12:689–702. doi: 10.1080/15548627.2016.1151580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Devi TS, Somayajulu M, Kowluru RA, Singh LP. TXNIP regulates mitophagy in retinal Müller cells under high-glucose conditions: Implications for diabetic retinopathy. Cell Death Dis. 2017;8:e2777. doi: 10.1038/cddis.2017.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Gao Y, Li J, Li J, Hu C, Zhang L, Yan J, Li L, Zhang L. Tetrahydroxy stilbene glycoside alleviated inflammatory damage by mitophagy via AMPK related PINK1/Parkin signaling pathway. Biochem Pharmacol. 2020;177:113997. doi: 10.1016/j.bcp.2020.113997. [DOI] [PubMed] [Google Scholar]
- 230.Li F, Zhang T, He Y, Gu W, Yang X, Zhao R, Yu J. Inflammation inhibition and gut microbiota regulation by TSG to combat atherosclerosis in ApoE−/− mice. J Ethnopharmacol. 2020;247:112232. doi: 10.1016/j.jep.2019.112232. [DOI] [PubMed] [Google Scholar]
- 231.Mu Y, Xu Z, Zhou X, Zhang H, Yang Q, Zhang Y, Xie Y, Kang J, Li F, Wang S. 2,3,5,4'-Tetrahydroxystilbene-2-O-β-D-gluc oside attenuates ischemia/reperfusion-induced brain injury in rats by promoting angiogenesis. Planta Med. 2017;83:676–683. doi: 10.1055/s-0042-120544. [DOI] [PubMed] [Google Scholar]
- 232.Zhou W, Yang Y, Mei C, Dong P, Mu S, Wu H, Zhou Y, Zheng Y, Guo F, Yang JQ. Inhibition of rho-kinase downregulates Th17 cells and ameliorates hepatic fibrosis by schistosoma japonicum infection. Cells. 2019;8:1262. doi: 10.3390/cells8101262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Kimura T, Horikoshi Y, Kuriyagawa C, Niiyama Y. Rho/ROCK pathway and noncoding RNAs: Implications in ischemic stroke and spinal cord injury. Int J Mol Sci. 2021;22:11573. doi: 10.3390/ijms222111573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Zhou Y, Su Y, Li B, Liu F, Ryder JW, Wu X, Gonzalez-DeWhitt PA, Gelfanova V, Hale JE, May PC, et al. Nonsteroidal anti-inflammatory drugs can lower amyloidogenic Abeta42 by inhibiting Rho. Science. 2003;302:1215–1217. doi: 10.1126/science.1090154. [DOI] [PubMed] [Google Scholar]
- 235.Scheiblich H, Bicker G. Regulation of microglial phagocytosis by RhoA/ROCK-inhibiting drugs. Cell Mol Neurobiol. 2017;37:461–473. doi: 10.1007/s10571-016-0379-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Alokam R, Singhal S, Srivathsav GS, Garigipati S, Puppala S, Sriram D, Perumal Y. Design of dual inhibitors of ROCK-I and NOX2 as potential leads for the treatment of neuroinflammation associated with various neurological diseases including autism spectrum disorder. Mol Biosyst. 2015;11:607–617. doi: 10.1039/C4MB00570H. [DOI] [PubMed] [Google Scholar]
- 237.Moon MY, Kim HJ, Li Y, Kim JG, Jeon YJ, Won HY, Kim JS, Kwon HY, Choi IG, Ro E, et al. Involvement of small GTPase RhoA in the regulation of superoxide production in BV2 cells in response to fibrillar Aβ peptides. Cell Signal. 2013;25:1861–1869. doi: 10.1016/j.cellsig.2013.05.023. [DOI] [PubMed] [Google Scholar]
- 238.Zhang X, Ye P, Wang D, Liu Y, Cao L, Wang Y, Xu Y, Zhu C. Involvement of RhoA/ROCK signaling in Aβ-induced chemotaxis, cytotoxicity and inflammatory response of microglial BV2 cells. Cell Mol Neurobiol. 2019;39:637–650. doi: 10.1007/s10571-019-00668-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.van der Meer DLM, Degenhardt T, Väisänen S, de Groot PJ, Heinäniemi M, de Vries SC, Müller M, Carlberg C, Kersten S. Profiling of promoter occupancy by PPARalpha in human hepatoma cells via ChIP-chip analysis. Nucleic Acids Res. 2010;38:2839–2850. doi: 10.1093/nar/gkq012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Xia P, Pan Y, Zhang F, Wang N, Wang E, Guo Q, Ye Z. Pioglitazone confers neuroprotection against ischemia-induced pyroptosis due to its inhibitory effects on HMGB-1/RAGE and Rac1/ROS pathway by activating PPAR-ɤ. Cell Physiol Biochem. 2018;45:2351–2368. doi: 10.1159/000488183. [DOI] [PubMed] [Google Scholar]
- 241.Janani C, Ranjitha Kumari BD. PPAR gamma gene-a review. Diabetes Metab Syndr. 2015;9:46–50. doi: 10.1016/j.dsx.2014.09.015. [DOI] [PubMed] [Google Scholar]
- 242.Stark JM, Coquet JM, Tibbitt CA. The role of PPAR-γ in allergic disease. Curr Allergy Asthma Rep. 2021;21:45. doi: 10.1007/s11882-021-01022-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Kumar AP, P P, Kumar BRP, Jeyarani V, Dhanabal SP, Justin A. Glitazones, PPAR-γ and neuroprotection. Mini Rev Med Chem. 2021;21:1457–1464. doi: 10.2174/1389557521666210304112403. [DOI] [PubMed] [Google Scholar]
- 244.Furth N, Pateras IS, Rotkopf R, Vlachou V, Rivkin I, Schmitt I, Bakaev D, Gershoni A, Ainbinder E, Leshkowitz D, et al. LATS1 and LATS2 suppress breast cancer progression by maintaining cell identity and metabolic state. Life Sci Alliance. 2018;1:e201800171. doi: 10.26508/lsa.201800171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Villapol S. Roles of peroxisome proliferator-activated receptor gamma on brain and peripheral inflammation. Cell Mol Neurobiol. 2018;38:121–132. doi: 10.1007/s10571-017-0554-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Moosecker S, Gomes P, Dioli C, Yu S, Sotiropoulos I, Almeida OFX. Activated PPARγ abrogates misprocessing of amyloid precursor protein, tau missorting and synaptotoxicity. Front Cell Neurosci. 2019;13:239. doi: 10.3389/fncel.2019.00239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.de la Monte SM, Wands JR. Molecular indices of oxidative stress and mitochondrial dysfunction occur early and often progress with severity of Alzheimer's disease. J Alzheimers Dis. 2006;9:167–181. doi: 10.3233/JAD-2006-9209. [DOI] [PubMed] [Google Scholar]
- 248.Watson GS, Cholerton BA, Reger MA, Baker LD, Plymate SR, Asthana S, Fishel MA, Kulstad JJ, Green PS, Cook DG, et al. Preserved cognition in patients with early Alzheimer disease and amnestic mild cognitive impairment during treatment with rosiglitazone: A preliminary study. Am J Geriatr Psychiatry. 2005;13:950–958. doi: 10.1176/appi.ajgp.13.11.950. [DOI] [PubMed] [Google Scholar]
- 249.Risner ME, Saunders AM, Altman JF, Ormandy GC, Craft S, Foley IM, Zvartau-Hind ME, Hosford DA, Roses AD, Rosiglitazone in Alzheimer's Disease Study Group Efficacy of rosiglitazone in a genetically defined population with mild-to-moderate Alzheimer's disease. Pharmacogenomics J. 2006;6:246–254. doi: 10.1038/sj.tpj.6500369. [DOI] [PubMed] [Google Scholar]
- 250.Mandrekar-Colucci S, Karlo JC, Landreth GE. Mechanisms underlying the rapid peroxisome proliferator-activated receptor-γ-mediated amyloid clearance and reversal of cognitive deficits in a murine model of Alzheimer's disease. J Neurosci. 2012;32:10117–10128. doi: 10.1523/JNEUROSCI.5268-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Moreno S, Farioli-Vecchioli S, Cerù MP. Immunolocalization of peroxisome proliferator-activated receptors and retinoid X receptors in the adult rat CNS. Neuroscience. 2004;123:131–145. doi: 10.1016/j.neuroscience.2003.08.064. [DOI] [PubMed] [Google Scholar]
- 252.Liu ZJ, Liu W, Liu L, Xiao C, Wang Y, Jiao JS. Curcumin protects neuron against cerebral ischemia-induced inflammation through improving PPAR-gamma function. Evid Based Complement Alternat Med. 2013;2013:470975. doi: 10.1155/2013/470975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Song GJ, Nam Y, Jo M, Jung M, Koo JY, Cho W, Koh M, Park SB, Suk K. A novel small-molecule agonist of PPAR-γ potentiates an anti-inflammatory M2 glial phenotype. Neuropharmacology. 2016;109:159–169. doi: 10.1016/j.neuropharm.2016.06.009. [DOI] [PubMed] [Google Scholar]
- 254.Qi Y, Zhang Q, Zhu H. Huang-Lian Jie-Du decoction: A review on phytochemical, pharmacological and pharmacokinetic investigations. Chin Med. 2019;14:57. doi: 10.1186/s13020-019-0277-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Wong LR, Tan EA, Lim MEJ, Shen W, Lian XL, Wang Y, Chen L, Ho PCL. Functional effects of berberine in modulating mitochondrial dysfunction and inflammatory response in the respective amyloidogenic cells and activated microglial cells-in vitro models simulating Alzheimer's disease pathology. Life Sci. 2012;282:119824. doi: 10.1016/j.lfs.2021.119824. [DOI] [PubMed] [Google Scholar]
- 256.Hagl S, Asseburg H, Heinrich M, Sus N, Blumrich EM, Dringen R, Frank J, Eckert GP. Effects of long-term rice bran extract supplementation on survival, cognition and brain mitochondrial function in aged NMRI mice. Neuromolecular Med. 2016;18:347–363. doi: 10.1007/s12017-016-8420-z. [DOI] [PubMed] [Google Scholar]
- 257.El-Din SS, Abd ES, Rashed L, Fayez S, Aboulhoda BE, Heikal OA, Galal AF, Nour ZA. Possible role of rice bran extract in microglial modulation through PPAR-gamma receptors in alzheimer's disease mice model. Metab Brain Dis. 2021;36:1903–1915. doi: 10.1007/s11011-021-00741-4. [DOI] [PubMed] [Google Scholar]
- 258.De Felice FG, Ferreira ST. Inflammation, defective insulin signaling, and mitochondrial dysfunction as common molecular denominators connecting type 2 diabetes to Alzheimer disease. Diabetes. 2014;63:2262–2272. doi: 10.2337/db13-1954. [DOI] [PubMed] [Google Scholar]
- 259.Bouriche H, Meziti H, Senator A, Arnhold J. Anti-inflammatory, free radical-scavenging, and metal-chelating activities of Malva parviflora. Pharm Biol. 2011;49:942–946. doi: 10.3109/13880209.2011.558102. [DOI] [PubMed] [Google Scholar]
- 260.Medrano-Jiménez E, Jiménez-Ferrer Carrillo I, Pedraza-Escalona M, Ramírez-Serrano CE, Álvarez-Arellano L, Cortés-Mendoza J, Herrera-Ruiz M, Jiménez-Ferrer E, Zamilpa A, Tortoriello J, et al. Malva parviflora extract ameliorates the deleterious effects of a high fat diet on the cognitive deficit in a mouse model of Alzheimer's disease by restoring microglial function via a PPAR-γ-dependent mechanism. J Neuroinflammation. 2019;16:143. doi: 10.1186/s12974-019-1515-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Villapol S, Yaszemski AK, Logan TT, Sanchez-Lemus E, Saavedra JM, Symes AJ. Candesartan, an angiotensin II AT1-receptor blocker and PPAR-γ agonist, reduces lesion volume and improves motor and memory function after traumatic brain injury in mice. Neuropsychopharmacology. 2012;37:2817–2829. doi: 10.1038/npp.2012.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Shindo T, Takasaki K, Uchida K, Onimura R, Kubota K, Uchida N, Irie K, Katsurabayashi S, Mishima K, Nishimura R, et al. Ameliorative effects of telmisartan on the inflammatory response and impaired spatial memory in a rat model of Alzheimer's disease incorporating additional cerebrovascular disease factors. Biol Pharm Bull. 2012;35:2141–2147. doi: 10.1248/bpb.b12-00387. [DOI] [PubMed] [Google Scholar]
- 263.Li NC, Lee A, Whitmer RA, Kivipelto M, Lawler E, Kazis LE, Wolozin B. Use of angiotensin receptor blockers and risk of dementia in a predominantly male population: Prospective cohort analysis. BMJ. 2010;340:b5465. doi: 10.1136/bmj.b5465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Wang ZF, Li J, Ma C, Huang C, Li ZQ. Telmisartan ameliorates Aβ oligomer-induced inflammation via PPARγ/PTEN pathway in BV2 microglial cells. Biochem Pharmacol. 2020;171:113674. doi: 10.1016/j.bcp.2019.113674. [DOI] [PubMed] [Google Scholar]
- 265.He Z, Li X, Han S, Ren B, Hu X, Li N, Du X, Ni J, Yang X, Liu Q. Bis(ethylmaltolato)oxidovanadium (IV) attenuates amyloid-beta-mediated neuroinflammation by inhibiting NF-κB signaling pathway via a PPARγ-dependent mechanism. Metallomics. 2021;13:mfab036. doi: 10.1093/mtomcs/mfab036. [DOI] [PubMed] [Google Scholar]
- 266.Yang Z, Liu B, Yang LE, Zhang C. Platycodigenin as potential drug candidate for Alzheimer's disease via modulating microglial polarization and neurite regeneration. Molecules. 2019;24:3207. doi: 10.3390/molecules24183207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Pan D, Xu Y, Zhang L, Su Q, Chen M, Li B, Xiao Q, Gao Q, Peng X, Jiang B, et al. Gene expression profile in peripheral blood mononuclear cells of postpartum depression patients. Sci Rep. 2018;8:10139. doi: 10.1038/s41598-018-28509-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Ahn S, Jang DM, Park SC, An S, Shin J, Han BW, Noh M. Cyclin-dependent kinase 5 inhibitor butyrolactone I elicits a partial agonist activity of peroxisome proliferator-activated receptor γ. Biomolecules. 2020;10:275. doi: 10.3390/biom10020275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Wang J, Zhu G, Sun C, Xiong K, Yao T, Su Y, Fang H. TAK-242 ameliorates DSS-induced colitis by regulating the gut microbiota and the JAK2/STAT3 signaling pathway. Microb Cell Fact. 2020;19:158. doi: 10.1186/s12934-020-01417-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Kulesza DW, Ramji K, Maleszewska M, Mieczkowski J, Dabrowski M, Chouaib S, Kaminska B. Search for novel STAT3-dependent genes reveals SERPINA3 as a new STAT3 target that regulates invasion of human melanoma cells. Lab Invest. 2019;99:1607–1621. doi: 10.1038/s41374-019-0288-8. [DOI] [PubMed] [Google Scholar]
- 271.Lee DY, Hwang CJ, Choi JY, Park MH, Song MJ, Oh KW, Han SB, Park WK, Cho HY, Cho SY, et al. KRICT-9 inhibits neuroinflammation, amyloidogenesis and memory loss in Alzheimer's disease models. Oncotarget. 2017;8:68654–68667. doi: 10.18632/oncotarget.19818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Zhang ZH, Yu LJ, Hui XC, Wu ZZ, Yin KL, Yang H, Xu Y. Hydroxy-safflor yellow A attenuates Aβ1-42-induced inflammation by modulating the JAK2/STAT3/NF-κB pathway. Brain Res. 2014;1563:72–80. doi: 10.1016/j.brainres.2014.03.036. [DOI] [PubMed] [Google Scholar]
- 273.Zheng ZV, Chen J, Lyu H, Lam S, Lu G, Chan WY, Wong GKC. Novel role of STAT3 in microglia-dependent neuroinflammation after experimental subarachnoid haemorrhage. Stroke Vasc Neurol. 2022;7:62–70. doi: 10.1136/svn-2021-001028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Wan J, Fu AK, Ip FC, Ng HK, Hugon J, Page G, Wang JH, Lai KO, Wu Z, Ip NY. Tyk2/STAT3 signaling mediates beta-amyloid-induced neuronal cell death: Implications in Alzheimer's disease. J Neurosci. 2010;30:6873–6881. doi: 10.1523/JNEUROSCI.0519-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Carret-Rebillat AS, Pace C, Gourmaud S, Ravasi L, Montagne-Stora S, Longueville S, Tible M, Sudol E, Chang RC, Paquet C, et al. Neuroinflammation and Aβ accumulation linked to systemic inflammation are decreased by genetic PKR down-regulation. Sci Rep. 2015;5:8489. doi: 10.1038/srep08489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Jin P, Kim JA, Choi DY, Lee YJ, Jung HS, Hong JT. Anti-inflammatory and anti-amyloidogenic effects of a small molecule, 2,4-bis(p-hydroxyphenyl)-2-butenal in Tg2576 Alzheimer's disease mice model. J Neuroinflammation. 2013;10:2. doi: 10.1186/1742-2094-10-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Choi JY, Hwang CJ, Lee DY, Gu SM, Lee HP, Choi DY, Oh KW, Han SB, Hong JT. (E)-2-methoxy-4-(3-(4-methoxyphenyl) prop-1-en-1-yl) phenol ameliorates LPS-mediated memory impairment by inhibition of STAT3 pathway. Neuromolecular Med. 2017;19:555–570. doi: 10.1007/s12017-017-8469-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Hussein G, Sankawa U, Goto H, Matsumoto K, Watanabe H. Astaxanthin, a carotenoid with potential in human health and nutrition. J Nat Prod. 2006;69:443–449. doi: 10.1021/np050354+. [DOI] [PubMed] [Google Scholar]
- 279.Han JH, Lee YS, Im JH, Ham YW, Lee HP, Han SB, Hong JT. Astaxanthin ameliorates lipopolysaccharide-induced neuroinflammation, oxidative stress and memory dysfunction through inactivation of the signal transducer and activator of transcription 3 pathway. Mar Drugs. 2019;17:123. doi: 10.3390/md17020123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Safar MM, Shahin NN, Mohamed AF, Abdelkader NF. Suppression of BACE1 and amyloidogenic/RAGE axis by sitagliptin ameliorates PTZ kindling-induced cognitive deficits in rats. Chem Biol Interact. 2020;328:109144. doi: 10.1016/j.cbi.2020.109144. [DOI] [PubMed] [Google Scholar]
- 281.Wen Y, Yu WH, Maloney B, Bailey J, Ma J, Marié I, Maurin T, Wang L, Figueroa H, Herman M, et al. Transcriptional regulation of beta-secretase by p25/cdk5 leads to enhanced amyloidogenic processing. Neuron. 2008;57:680–690. doi: 10.1016/j.neuron.2008.02.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Millot P, San C, Bennana E, Porte B, Vignal N, Hugon J, Paquet C, Hosten B, Mouton-Liger F. STAT3 inhibition protects against neuroinflammation and BACE1 upregulation induced by systemic inflammation. Immunol Lett. 2020;228:129–134. doi: 10.1016/j.imlet.2020.10.004. [DOI] [PubMed] [Google Scholar]
- 283.Long QH, Wu YG, He LL, Ding L, Tan AH, Shi HY, Wang P. Suan-Zao-Ren decoction ameliorates synaptic plasticity through inhibition of the Aβ deposition and JAK2/STAT3 signaling pathway in AD model of APP/PS1 transgenic mice. Chin Med. 2021;16:14. doi: 10.1186/s13020-021-00425-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Jia T, Xing Z, Wang And H, Li G. Protective effect of dexmedetomidine on intestinal mucosal barrier function in rats after cardiopulmonary bypass. Exp Biol Med (Maywood) 2022;247:498–508. doi: 10.1177/15353702211062509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Wang LC, Liao LX, Zhao MB, Dong X, Zeng KW, Tu PF. Protosappanin A exerts anti-neuroinflammatory effect by inhibiting JAK2-STAT3 pathway in lipopolysaccharide-induced BV2 microglia. Chin J Nat Med. 2017;15:674–679. doi: 10.1016/S1875-5364(17)30096-1. [DOI] [PubMed] [Google Scholar]
- 286.Porro C, Cianciulli A, Trotta T, Lofrumento DD, Panaro MA. Curcumin regulates anti-inflammatory responses by JAK/STAT/SOCS signaling pathway in BV-2 microglial cells. Biology (Basel) 2019;8:51. doi: 10.3390/biology8030051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Amalraj A, Pius A, Gopi S, Gopi S. Biological activities of curcuminoids, other biomolecules from turmeric and their derivatives-a review. J Tradit Complement Med. 2016;7:205–233. doi: 10.1016/j.jtcme.2016.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.He G, Karin M. NF-κB and STAT3-key players in liver inflammation and cancer. Cell Res. 2011;21:159–168. doi: 10.1038/cr.2010.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Sim DY, Lee HJ, Jung JH, Im E, Hwang J, Kim DS, Kim SH. Suppression of STAT3 phosphorylation and RelA/p65 acetylation mediated by MicroRNA134 plays a pivotal role in the apoptotic effect of lambertianic acid. Int J Mol Sci. 2019;20:2993. doi: 10.3390/ijms20122993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Song SY, Jung YY, Hwang CJ, Lee HP, Sok CH, Kim JH, Lee SM, Seo HO, Hyun BK, Choi DY, et al. Inhibitory effect of ent-Sauchinone on amyloidogenesis via inhibition of STAT3-mediated NF-κB activation in cultured astrocytes and microglial BV-2 cells. J Neuroinflammation. 2014;11:118. doi: 10.1186/1742-2094-11-118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Kim J, Park JH, Park SK, Hoe HS. Sorafenib modulates the LPS- and Aβ-induced neuroinflammatory response in cells, wild-type mice, and 5xFAD mice. Front Immunol. 2021;12:684344. doi: 10.3389/fimmu.2021.684344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Hei YY, Xin M, Zhang H, Xie XX, Mao S, Zhang SQ. Synthesis and antitumor activity evaluation of 4,6-disubstituted quinazoline derivatives as novel PI3K inhibitors. Bioorg Med Chem Lett. 2016;26:4408–4413. doi: 10.1016/j.bmcl.2016.08.015. [DOI] [PubMed] [Google Scholar]
- 293.You JS, Li CY, Chen W, Wu XL, Huang LJ, Li RK, Gao F, Zhang MY, Liu HL, Qu WL. A network pharmacology-based study on Alzheimer disease prevention and treatment of Qiong Yu Gao. BioData Min. 2020;13:2. doi: 10.1186/s13040-020-00212-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Yang S, Chen Z, Cao M, Li R, Wang Z, Zhang M. Pioglitazone ameliorates Aβ42 deposition in rats with diet-induced insulin resistance associated with AKT/GSK3β activation. Mol Med Rep. 2017;15:2588–2594. doi: 10.3892/mmr.2017.6342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Liu X, Wang H, Bei J, Zhao J, Jiang G, Liu X. The protective role of miR-132 targeting HMGA2 through the PI3K/AKT pathway in mice with Alzheimer's disease. Am J Transl Res. 2021;13:4632–4643. [PMC free article] [PubMed] [Google Scholar]
- 296.Hwang SY, Jung JS, Lim SJ, Kim JY, Kim TH, Cho KH, Han IO. LY294002 inhibits interferon-gamma-stimulated inducible nitric oxide synthase expression in BV2 microglial cells. Biochem Biophys Res Commun. 2004;318:691–697. doi: 10.1016/j.bbrc.2004.04.082. [DOI] [PubMed] [Google Scholar]
- 297.He Y, Zhou A, Jiang W. Toll-like receptor 4-mediated signaling participates in apoptosis of hippocampal neurons. Neural Regen Res. 2013;8:2744–2753. doi: 10.3969/j.issn.1673-5374.2013.29.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Zu HB, Liu XY, Yao K. DHCR24 overexpression modulates microglia polarization and inflammatory response via Akt/GSK3β signaling in Aβ25-35 treated BV-2 cells. Life Sci. 2020;260:118470. doi: 10.1016/j.lfs.2020.118470. [DOI] [PubMed] [Google Scholar]
- 299.Sarajärvi T, Haapasalo A, Viswanathan J, Mäkinen P, Laitinen M, Soininen H, Hiltunen M. Down-regulation of seladin-1 increases BACE1 levels and activity through enhanced GGA3 depletion during apoptosis. J Biol Chem. 2009;284:34433–34443. doi: 10.1074/jbc.M109.036202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Yang W, Liu Y, Xu QQ, Xian YF, Lin ZX. Sulforaphene ameliorates neuroinflammation and hyperphosphorylated tau protein via regulating the PI3K/Akt/GSK-3 β pathway in experimental models of Alzheimer's disease. Oxid Med Cell Longev. 2020;2020:4754195. doi: 10.1155/2020/4754195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Ivanovics G, Horvath S. Raphanin, an antibacterial principle of the radish (Raphanus sativus) Nature. 1947;160:297. doi: 10.1038/160297a0. [DOI] [PubMed] [Google Scholar]
- 302.Lonze BE, Ginty DD. Function and regulation of CREB family transcription factors in the nervous system. Neuron. 2002;35:605–623. doi: 10.1016/S0896-6273(02)00828-0. [DOI] [PubMed] [Google Scholar]
- 303.Ahn SH, Suh JS, Jang YK, Kim HS, Choi GH, Kim E, Kim TJ. Rhynchosia volubilis promotes cell survival via cAMP-PKA/ERK-CREB pathway. Pharmaceuticals (Basel) 2022;15:73. doi: 10.3390/ph15010073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Hu YJ, Sun Q, Zhang WH, Huo YJ, Xu CJ, Liu JF. Specific activation of mGlu2 induced IGF-1R transactivation in vitro through FAK phosphorylation. Acta Pharmacol Sin. 2019;40:460–467. doi: 10.1038/s41401-018-0033-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Viola H, Furman M, Izquierdo LA, Alonso M, Barros DM, de Souza MM, Izquierdo I, Medina JH. Phosphorylated cAMP response element-binding protein as a molecular marker of memory processing in rat hippocampus: effect of novelty. J Neurosci. 2000;20:RC112. doi: 10.1523/JNEUROSCI.20-23-j0002.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Dineley KT, Westerman M, Bui D, Bell K, Ashe KH, Sweatt JD. Beta-amyloid activates the mitogen-activated protein kinase cascade via hippocampal alpha7 nicotinic acetylcholine receptors: In vitro and in vivo mechanisms related to Alzheimer's disease. J Neurosci. 2001;21:4125–4133. doi: 10.1523/JNEUROSCI.21-12-04125.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Yamamoto-Sasaki M, Ozawa H, Saito T, Rösler M, Riederer P. Impaired phosphorylation of cyclic AMP response element binding protein in the hippocampus of dementia of the Alzheimer type. Brain Res. 1999;824:300–303. doi: 10.1016/S0006-8993(99)01220-2. [DOI] [PubMed] [Google Scholar]
- 308.Mizuno M, Yamada K, Maekawa N, Saito K, Seishima M, Nabeshima T. CREB phosphorylation as a molecular marker of memory processing in the hippocampus for spatial learning. Behav Brain Res. 2002;133:135–141. doi: 10.1016/S0166-4328(01)00470-3. [DOI] [PubMed] [Google Scholar]
- 309.Sharma VK, Singh TG. CREB: A multifaceted target for Alzheimer's disease. Curr Alzheimer Res. 2020;17:1280–1293. doi: 10.2174/1567205018666210218152253. [DOI] [PubMed] [Google Scholar]
- 310.Li C, Chen T, Zhou H, Feng Y, Hoi MPM, Ma D, Zhao C, Zheng Y, Lee SMY. BHDPC is a novel neuroprotectant that provides anti-neuroinflammatory and neuroprotective effects by inactivating NF-κB and activating PKA/CREB. Front Pharmacol. 2018;9:614. doi: 10.3389/fphar.2018.00614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Ghosh M, Xu Y, Pearse DD. Cyclic AMP is a key regulator of M1 to M2a phenotypic conversion of microglia in the presence of Th2 cytokines. J Neuroinflammation. 2016;13:9. doi: 10.1186/s12974-015-0463-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Tripathi MK, Kartawy M, Amal H. The role of nitric oxide in brain disorders: Autism spectrum disorder and other psychiatric, neurological, and neurodegenerative disorders. Redox Biol. 2020;34:101567. doi: 10.1016/j.redox.2020.101567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Zhang J, Guo J, Zhao X, Chen Z, Wang G, Liu A, Wang Q, Zhou W, Xu Y, Wang C. Phosphodiesterase-5 inhibitor sildenafil prevents neuroinflammation, lowers beta-amyloid levels and improves cognitive performance in APP/PS1 transgenic mice. Behav Brain Res. 2013;250:230–237. doi: 10.1016/j.bbr.2013.05.017. [DOI] [PubMed] [Google Scholar]
- 314.Zeitlin R, Patel S, Burgess S, Arendash GW, Echeverria V. Caffeine induces beneficial changes in PKA signaling and JNK and ERK activities in the striatum and cortex of Alzheimer's transgenic mice. Brain Res. 2011;1417:127–136. doi: 10.1016/j.brainres.2011.08.036. [DOI] [PubMed] [Google Scholar]
- 315.Bitner RS, Markosyan S, Nikkel AL, Brioni JD. In-vivo histamine H3 receptor antagonism activates cellular signaling suggestive of symptomatic and disease modifying efficacy in Alzheimer's disease. Neuropharmacology. 2011;60:460–466. doi: 10.1016/j.neuropharm.2010.10.026. [DOI] [PubMed] [Google Scholar]
- 316.Medhurst AD, Atkins AR, Beresford IJ, Brackenborough K, Briggs MA, Calver AR, Cilia J, Cluderay JE, Crook B, Davis JB, et al. GSK189254, a novel H3 receptor antagonist that binds to histamine H3 receptors in Alzheimer's disease brain and improves cognitive performance in preclinical models. J Pharmacol Exp Ther. 2007;321:1032–1045. doi: 10.1124/jpet.107.120311. [DOI] [PubMed] [Google Scholar]
- 317.Wang J, Liu B, Sun F, Xu Y, Luan H, Yang M, Wang C, Zhang T, Zhou Z, Yan H. Histamine H3R antagonist counteracts the impaired hippocampal neurogenesis in lipopolysaccharide-induced neuroinflammation. Int Immunopharmacol. 2022;110:109045. doi: 10.1016/j.intimp.2022.109045. [DOI] [PubMed] [Google Scholar]
- 318.Hiraga N, Adachi N, Liu K, Nagaro T, Arai T. Suppression of inflammatory cell recruitment by histamine receptor stimulation in ischemic rat brains. Eur J Pharmacol. 2007;557:236–244. doi: 10.1016/j.ejphar.2006.11.020. [DOI] [PubMed] [Google Scholar]
- 319.Amin FU, Shah SA, Badshah H, Khan M, Kim MO. Anthocyanins encapsulated by PLGA@PEG nanoparticles potentially improved its free radical scavenging capabilities via p38/JNK pathway against Aβ1-42-induced oxidative stress. J Nanobiotechnology. 2017;15:12. doi: 10.1186/s12951-016-0227-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 320.von Otter M, Landgren S, Nilsson S, Zetterberg M, Celojevic D, Bergström P, Minthon L, Bogdanovic N, Andreasen N, Gustafson DR, et al. Nrf2-encoding NFE2L2 haplotypes influence disease progression but not risk in Alzheimer's disease and age-related cataract. Mech Ageing Dev. 2010;131:105–110. doi: 10.1016/j.mad.2009.12.007. [DOI] [PubMed] [Google Scholar]
- 321.Kanninen K, Malm TM, Jyrkkänen HK, Goldsteins G, Keksa-Goldsteine V, Tanila H, Yamamoto M, Ylä-Herttuala S, Levonen AL, Koistinaho J. Nuclear factor erythroid 2-related factor 2 protects against beta amyloid. Mol Cell Neurosci. 2008;39:302–313. doi: 10.1016/j.mcn.2008.07.010. [DOI] [PubMed] [Google Scholar]
- 322.Li XX, Zheng X, Liu Z, Xu Q, Tang H, Feng J, Yang S, Vong CT, Gao H, Wang Y. Cryptotanshinone from Salvia miltiorrhiza Bunge (Danshen) inhibited inflammatory responses via TLR4/MyD88 signaling pathway. Chin Med. 2020;15:20. doi: 10.1186/s13020-020-00303-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Fragoulis A, Siegl S, Fendt M, Jansen S, Soppa U, Brandenburg LO, Pufe T, Weis J, Wruck CJ. Oral administration of methysticin improves cognitive deficits in a mouse model of Alzheimer's disease. Redox Biol. 2017;12:843–853. doi: 10.1016/j.redox.2017.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 324.Bilia AR, Scalise L, Bergonzi MC, Vincieri FF. Analysis of kavalactones from Piper methysticum (kava-kava) J Chromatogr B Analyt Technol Biomed Life Sci. 2004;812:203–214. doi: 10.1016/S1570-0232(04)00644-0. [DOI] [PubMed] [Google Scholar]
- 325.Mattioli R, Francioso A, d'Erme M, Trovato M, Mancini P, Piacentini L, Casale AM, Wessjohann L, Gazzino R, Costantino P, Mosca L. Anti-inflammatory activity of a polyphenolic extract from Arabidopsis thaliana in in vitro and in vivo models of Alzheimer's disease. Int J Mol Sci. 2019;20:708. doi: 10.3390/ijms20030708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Rateb ME, Houssen WE, Schumacher M, Harrison WT, Diederich M, Ebel R, Jaspars M. Bioactive diterpene derivatives from the marine sponge Spongionella sp. J Nat Prod. 2009;72:1471–1476. doi: 10.1021/np900233c. [DOI] [PubMed] [Google Scholar]
- 327.Alvariño R, Alonso E, Abbasov ME, Chaheine CM, Conner ML, Romo D, Alfonso A, Botana LM. Gracilin A derivatives target early events in Alzheimer's disease: In vitro effects on neuroinflammation and oxidative stress. ACS Chem Neurosci. 2019;10:4102–4111. doi: 10.1021/acschemneuro.9b00329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Wang X, Asghar M. Protein disulfide isomerase regulates renal AT1 receptor function and blood pressure in rats. Am J Physiol Renal Physiol. 2017;313:F461–F466. doi: 10.1152/ajprenal.00580.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 329.Itoh K, Mimura J, Yamamoto M. Discovery of the negative regulator of Nrf2, Keap1: A historical overview. Antioxid Redox Signal. 2010;13:1665–1678. doi: 10.1089/ars.2010.3222. [DOI] [PubMed] [Google Scholar]
- 330.Wu H, Zhao G, Jiang K, Li C, Qiu C, Deng G. Engeletin alleviates lipopolysaccharide-induced endometritis in mice by inhibiting TLR4-mediated NF-κB activation. J Agric Food Chem. 2016;64:6171–6178. doi: 10.1021/acs.jafc.6b02304. [DOI] [PubMed] [Google Scholar]
- 331.Huang Z, Ji H, Shi J, Zhu X, Zhi Z. Engeletin attenuates Aβ1-42-induced oxidative stress and neuroinflammation by Keap1/Nrf2 pathway. Inflammation. 2020;43:1759–1771. doi: 10.1007/s10753-020-01250-9. [DOI] [PubMed] [Google Scholar]
- 332.Barone E, Di Domenico F, Sultana R, Coccia R, Mancuso C, Perluigi M, Butterfield DA. Heme oxygenase-1 posttranslational modifications in the brain of subjects with Alzheimer disease and mild cognitive impairment. Free Radic Biol Med. 2012;52:2292–2301. doi: 10.1016/j.freeradbiomed.2012.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 333.Zou Y, Hong B, Fan L, Zhou L, Liu Y, Wu Q, Zhang X, Dong M. Protective effect of puerarin against beta-amyloid-induced oxidative stress in neuronal cultures from rat hippocampus: Involvement of the GSK-3β/Nrf2 signaling pathway. Free Radic Res. 2013;47:55–63. doi: 10.3109/10715762.2012.742518. [DOI] [PubMed] [Google Scholar]
- 334.Eom HW, Park SY, Kim YH, Seong SJ, Jin ML, Ryu EY, Kim MJ, Lee SJ. Bambusae caulis in taeniam modulates neuroprotective and anti-neuroinflammatory effects in hippocampal and microglial cells via HO-1- and Nrf-2-mediated pathways. Int J Mol Med. 2012;30:1512–1520. doi: 10.3892/ijmm.2012.1128. [DOI] [PubMed] [Google Scholar]
- 335.Chen J, Yin W, Tu Y, Wang S, Yang X, Chen Q, Zhang X, Han Y, Pi R. L-F001, a novel multifunctional ROCK inhibitor, suppresses neuroinflammation in vitro and in vivo: Involvement of NF-κB inhibition and Nrf2 pathway activation. Eur J Pharmacol. 2017;806:1–9. doi: 10.1016/j.ejphar.2017.03.025. [DOI] [PubMed] [Google Scholar]
- 336.Zhan TW, Tian YX, Wang Q, Wu ZX, Zhang WP, Lu YB, Wu M. Cangrelor alleviates pulmonary fibrosis by inhibiting GPR17-mediated inflammation in mice. Int Immunopharmacol. 2018;62:261–269. doi: 10.1016/j.intimp.2018.06.006. [DOI] [PubMed] [Google Scholar]
- 337.Jin S, Wang X, Xiang X, Wu Y, Hu J, Li Y, Lin DY, Tan Y, Wu X. Inhibition of GPR17 with cangrelor improves cognitive impairment and synaptic deficits induced by Aβ1-42 through Nrf2/HO-1 and NF-κB signaling pathway in mice. Int Immunopharmacol. 2021;101:108335. doi: 10.1016/j.intimp.2021.108335. [DOI] [PubMed] [Google Scholar]
- 338.Gao X, He D, Liu D, Hu G, Zhang Y, Meng T, Su Y, Zhou A, Huang B, Du J, Fu S. Beta-naphthoflavone inhibits LPS-induced inflammation in BV-2 cells via AKT/Nrf-2/HO-1-NF-kappaB signaling axis. Immunobiology. 2020;225:151965. doi: 10.1016/j.imbio.2020.151965. [DOI] [PubMed] [Google Scholar]
- 339.Just PA, Charawi S, Denis RGP, Savall M, Traore M, Foretz M, Bastu S, Magassa S, Senni N, Sohier P, et al. Lkb1 suppresses amino acid-driven gluconeogenesis in the liver. Nat Commun. 2020;11:6127. doi: 10.1038/s41467-020-19490-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Marinangeli C, Didier S, Ahmed T, Caillerez R, Domise M, Laloux C, Bégard S, Carrier S, Colin M, Marchetti P, et al. AMP-activated protein kinase is essential for the maintenance of energy levels during synaptic activation. iScience. 2018;9:1–13. doi: 10.1016/j.isci.2018.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 341.Vingtdeux V, Davies P, Dickson DW, Marambaud P. AMPK is abnormally activated in tangle- and pre-tangle-bearing neurons in Alzheimer's disease and other tauopathies. Acta Neuropathol. 2011;121:337–349. doi: 10.1007/s00401-010-0759-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 342.Shah SA, Yoon GH, Chung SS, Abid MN, Kim TH, Lee HY, Kim MO. Novel osmotin inhibits SREBP2 via the AdipoR1/AMPK/SIRT1 pathway to improve Alzheimer's disease neuropathological deficits. Mol Psychiatry. 2017;22:407–416. doi: 10.1038/mp.2016.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 343.Vingtdeux V, Chandakkar P, Zhao H, d'Abramo C, Davies P, Marambaud P. Novel synthetic small-molecule activators of AMPK as enhancers of autophagy and amyloid-β peptide degradation. FASEB J. 2011;25:219–231. doi: 10.1096/fj.10-167361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 344.Domise M, Didier S, Marinangeli C, Zhao H, Chandakkar P, Buée L, Viollet B, Davies P, Marambaud P, Vingtdeux V. AMP-activated protein kinase modulates tau phosphorylation and tau pathology in vivo. Sci Rep. 2016;6:26758. doi: 10.1038/srep26758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Li C, Zhang C, Zhou H, Feng Y, Tang F, Hoi M, He C, Ma D, Zhao C, Lee S. Inhibitory effects of betulinic acid on LPS-induced neuroinflammation involve M2 microglial polarization via CaMKKβ-Dependent AMPK activation. Front Mol Neurosci. 2018;11:98. doi: 10.3389/fnmol.2018.00098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 346.Voss U, Ekblad E. Lipopolysaccharide-induced loss of cultured rat myenteric neurons-role of AMP-activated protein kinase. PLoS One. 2014;9:e114044. doi: 10.1371/journal.pone.0114044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 347.Zhou Z, Zhang L, Liu Y, Huang C, Xia W, Zhou H, Zhou Z, Zhou X. Luteolin protects chondrocytes from H O oxidative injury and attenuates osteoarthritis progression by 2 2-induced activating AMPK-Nrf2 signaling. Oxid Med Cell Longev. 2022;2022:5635797. doi: 10.1155/2022/5635797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 348.Zhou F, Wang M, Ju J, Wang Y, Liu Z, Zhao X, Yan Y, Yan S, Luo X, Fang Y. Schizandrin A protects against cerebral ischemia-reperfusion injury by suppressing inflammation and oxidative stress and regulating the AMPK/Nrf2 pathway regulation. Am J Transl Res. 2019;11:199–209. [PMC free article] [PubMed] [Google Scholar]
- 349.Park SY, Choi MH, Park G, Choi YW. Petasites japonicus bakkenolide B inhibits lipopolysaccharide-induced pro-inflammatory cytokines via AMPK/Nrf2 induction in microglia. Int J Mol Med. 2018;41:1683–1692. doi: 10.3892/ijmm.2017.3350. [DOI] [PubMed] [Google Scholar]
- 350.Lee KP, Kang S, Park SJ, Choi YW, Lee YG, Im DS. Anti-allergic and anti-inflammatory effects of bakkenolide B isolated from Petasites japonicus leaves. J Ethnopharmacol. 2013;148:890–894. doi: 10.1016/j.jep.2013.05.037. [DOI] [PubMed] [Google Scholar]
- 351.Li J, Wen PY, Li WW, Zhou J. Upregulation effects of tanshinone IIA on the expressions of NeuN, Nissl body, and IκB and downregulation effects on the expressions of GFAP and NF-κB in the brain tissues of rat models of Alzheimer's disease. Neuroreport. 2015;26:758–766. doi: 10.1097/WNR.0000000000000419. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing is not applicable to this article, as no data sets were generated or analyzed during the current study.