Abstract
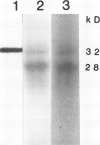
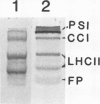
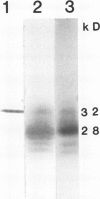
Distribution of the major light-harvesting chlorophyll a/b-protein (LHCII) and its mRNA within bundle sheath and mesophyll cells of maize (Zea mays L.) was studied using in situ immunolocalization and hybridization, respectively. In situ hybridization with specific LHCII RNA probes from maize and Lemna gibba definitively shows the presence of high levels of mRNA for LHCII in both bundle sheath cells and mesophyll cells. In situ immuno-localization studies, using an LHCII monoclonal antibody, demonstrate the presence of LHCII polypeptides in chloroplasts of both cell types. The polypeptide composition of LHCII and the amount of LHCII in bundle sheath cells are different from those in mesophyll cells. Both mesophyll and bundle sheath chloroplasts can take up, import and process the in vitro transcribed and translated LHCII precursor protein from L. gibba. Although bundle sheath chloroplasts incorporate LHCII into the pigmented light-harvesting complex, the efficiency is lower than that in mesophyll chloroplasts.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barker S. J., Harada J. J., Goldberg R. B. Cellular localization of soybean storage protein mRNA in transformed tobacco seeds. Proc Natl Acad Sci U S A. 1988 Jan;85(2):458–462. doi: 10.1073/pnas.85.2.458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burke J. J., Ditto C. L., Arntzen C. J. Involvement of the light-harvesting complex in cation regulation of excitation energy distribution in chloroplasts. Arch Biochem Biophys. 1978 Apr 15;187(1):252–263. doi: 10.1016/0003-9861(78)90031-0. [DOI] [PubMed] [Google Scholar]
- Chollet R. Photosynthetic carbon metabolism in isolated maize bundle sheath strands. Plant Physiol. 1973 Apr;51(4):787–792. doi: 10.1104/pp.51.4.787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chua N. H., Schmidt G. W. Transport of proteins into mitochondria and chloroplasts. J Cell Biol. 1979 Jun;81(3):461–483. doi: 10.1083/jcb.81.3.461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cline K. Import of proteins into chloroplasts. Membrane integration of a thylakoid precursor protein reconstituted in chloroplast lysates. J Biol Chem. 1986 Nov 5;261(31):14804–14810. [PubMed] [Google Scholar]
- Cline K., Werner-Washburne M., Lubben T. H., Keegstra K. Precursors to two nuclear-encoded chloroplast proteins bind to the outer envelope membrane before being imported into chloroplasts. J Biol Chem. 1985 Mar 25;260(6):3691–3696. [PubMed] [Google Scholar]
- Darr S. C., Somerville S. C., Arntzen C. J. Monoclonal antibodies to the light-harvesting chlorophyll a/b protein complex of photosystem II. J Cell Biol. 1986 Sep;103(3):733–740. doi: 10.1083/jcb.103.3.733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grossman A. R., Bartlett S. G., Schmidt G. W., Mullet J. E., Chua N. H. Optimal conditions for post-translational uptake of proteins by isolated chloroplasts. In vitro synthesis and transport of plastocyanin, ferredoxin-NADP+ oxidoreductase, and fructose-1,6-bisphosphatase. J Biol Chem. 1982 Feb 10;257(3):1558–1563. [PubMed] [Google Scholar]
- Kanai R., Edwards G. E. Separation of mesophyll protoplasts and bundle sheath cells from maize leaves for photosynthetic studies. Plant Physiol. 1973 Jun;51(6):1133–1137. doi: 10.1104/pp.51.6.1133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohorn B. D., Harel E., Chitnis P. R., Thornber J. P., Tobin E. M. Functional and mutational analysis of the light-harvesting chlorophyll a/b protein of thylakoid membranes. J Cell Biol. 1986 Mar;102(3):972–981. doi: 10.1083/jcb.102.3.972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langdale J. A., Metzler M. C., Nelson T. The argentia mutation delays normal development of photosynthetic cell-types in Zea mays. Dev Biol. 1987 Jul;122(1):243–255. doi: 10.1016/0012-1606(87)90349-6. [DOI] [PubMed] [Google Scholar]
- Li J. Light-harvesting chlorophyll a/b-protein: Three-dimensional structure of a reconstituted membrane lattice in negative stain. Proc Natl Acad Sci U S A. 1985 Jan;82(2):386–390. doi: 10.1073/pnas.82.2.386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martineau B., Taylor W. C. Cell-specific photosynthetic gene expression in maize determined using cell separation techniques and hybridization in situ. Plant Physiol. 1986 Oct;82(2):613–618. doi: 10.1104/pp.82.2.613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mullet J. E. The amino acid sequence of the polypeptide segment which regulates membrane adhesion (grana stacking) in chloroplasts. J Biol Chem. 1983 Aug 25;258(16):9941–9948. [PubMed] [Google Scholar]
- Schmidt G. W., Bartlett S. G., Grossman A. R., Cashmore A. R., Chua N. H. Biosynthetic pathways of two polypeptide subunits of the light-harvesting chlorophyll a/b protein complex. J Cell Biol. 1981 Nov;91(2 Pt 1):468–478. doi: 10.1083/jcb.91.2.468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuster G., Ohad I., Martineau B., Taylor W. C. Differentiation and development of bundle sheath and mesophyll thylakoids in maize. Thylakoid polypeptide composition, phosphorylation, and organization of photosystem II. J Biol Chem. 1985 Sep 25;260(21):11866–11873. [PubMed] [Google Scholar]
- Sheen J. Y., Bogorad L. Differential expression of six light-harvesting chlorophyll a/b binding protein genes in maize leaf cell types. Proc Natl Acad Sci U S A. 1986 Oct;83(20):7811–7815. doi: 10.1073/pnas.83.20.7811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smillie R. M., Andersen K. S., Bishop D. G. Plastocyanin-dependent photoreduction of NADP by agranal chloroplasts from maize. FEBS Lett. 1971 Apr 2;13(6):318–320. doi: 10.1016/0014-5793(71)80250-8. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]