Abstract
Background
Visceral hypersensitivity in functional dyspepsia can be localized or widespread, and there is no simple method of assessment. Measuring interoceptive accuracy at different sites provides an assessment of perceptual hypersensitivity to specific ecological phenomena. The purpose of this study was to characterize visceral hypersensitivity by comparing gastric sensory and cardiac perceptual tests in patients with postprandial distress syndrome and in healthy volunteers.
Methods
Sixteen patients with postprandial distress syndrome (age = 47.5 ± 17.4, all female) and 16 healthy volunteers (age = 43.3 ± 16.1, all female) participated in the study after a six-hour fast. Each participant answered questionnaires about physical and mental quality of life, depression and anxiety, tendency of alexithymia, and somatosensory amplification. After completing the questionnaire, the participants took the heartbeat tracking task and the five-minute water load test. We performed statistical analysis using the Mann–Whitney U test and Spearman’s rank correlation coefficient.
Results
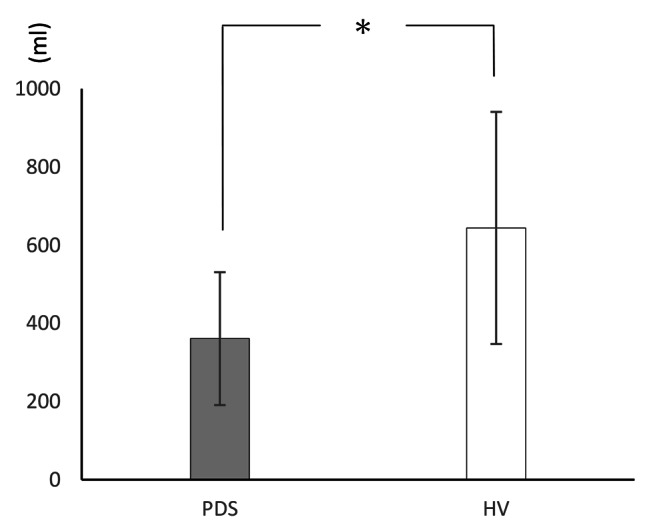
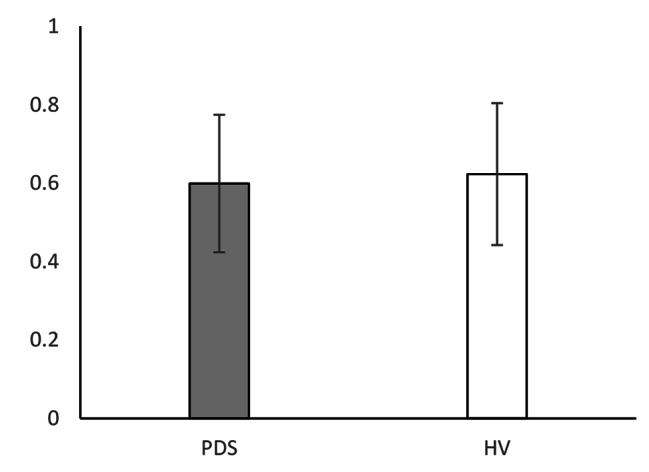
Subjects with postprandial distress syndrome had a lower drinking capacity than healthy volunteers (postprandial distress syndrome = 360.9 ± 170.0 mL, healthy volunteers = 644.1 ± 297 mL, P = 0.009), but there was no significant difference in the heartbeat perception score (postprandial distress syndrome = 0.599 ± 0.175, healthy volunteers = 0.623 ± 0.181, P = 0.647). There was a negative correlation (r = − 0.509, P < 0.05) between drinking capacity and the heartbeat perception score in healthy volunteers, but no correlation in postprandial distress syndrome (r = − 0.156, P = 0.564). Heartbeat perception score did not correlate with psychological measures.
Conclusions
Compared with healthy volunteers, only the five-minute water load test values were reduced in patients with postprandial distress syndrome, and no difference was observed in the heartbeat tracking task. Combining the 5-minute water load test and the heart rate tracking task revealed a lost cardiac-gastric perceptual relationship in patients with postprandial distress syndrome that was not observed in healthy volunteers, suggesting that there is hypersensitivity in gastric interoceptive perceptual function. Performing sensory examinations at two different sites may be useful in clarifying whether visceral hypersensitivity is localized.
Trial registration
UMIN000057586. Registered11 March 2023(retrospectively registered).
Keywords: Postprandial distress syndrome, Interoceptive accuracy, Heartbeat tracking task, Five-minute water load test, Visceral hypersensitivity
Background
Interoception is internal body representation, which includes perceiving, interpreting, integrating, and regulating information about physiological states throughout the body [1, 2]. Interoceptive signals provide vital information to central control programs and contribute to the maintenance of homeostasis [3]. Interoception is composed of three dimensions: objective, subjective, and metacognitive aspects [4, 5]. Interoceptive accuracy (IAc) is the objective aspect of interoception and is the core component among its three dimensions [5].
The various interoceptive signals are delivered from the peripheral nerves to the central nervous system and integrated in the anterior insular cortex (AIC) [2, 6]. This process is separable by bodily axes, a set of single organs and their peripheral nerves and central controls [7], and the IAc dimension of each organ or peripheral site is fractionated across each bodily axis [7–9]. The gastric and cardiac axes are correlated, but the cardiac and respiratory axes are not related in the IAc dimension [8, 10, 11]. Measuring interoception across multiple bodily axes is an indicator of whether biological phenomena of one organ system are perceived more accurately than those of other organ systems and helps to capture the characteristics of an individual’s interoceptive process [7].
Visceral hypersensitivity (VH) is an important factor in symptom generation as part of functional gastrointestinal disorder (FGID) [12, 13]. In functional dyspepsia (FD), VH is present when there is hypersensitivity to gastric stretch stimuli, to capsaicin, and to acid [14–17]. VH is one of the explanatory variables of symptoms of postprandial distress syndrome (PDS) [18]. Several studies have reported conflicting results, with sensory hyperalgesia in FGID being limited to the symptom site [19–21] or occurring across other organs and body parts [17, 22]. It has been suggested that this difference may be caused by differences in whether central or peripheral factors are more pronounced within an individual [20]. When comparing the central disturbances of the subgroups in epigastric pain syndrome (EPS), increased central activity at rest is important in the pathogenesis [23]; PDS has been suggested to induce symptoms via central mechanisms in the stimulation of gastric advancement by drinking water, but central activity at rest is not important [23].
In FD, interoceptive disturbances in brain–gut signaling are thought to lead to hypersensitivity and visceral pain and discomfort [24]. However, VH and other symptoms in FD can be localized or widespread and vary from individual to individual. Although visceral perception has been the focus of much attention, studies of interoception in FGID have focused specifically on the site of symptoms, and there is a lack of research on how other organs are perceived. Previous studies have shown that VH is an explanatory variable for PDS symptoms, that PDS is induced by a central response to gastric stretch, and that there is a difference in central activity during rest between EPS and PDS. Based on these findings, we hypothesize that PDS is a problem of gastric axis interoceptive processes and involves only abnormal sensory processing in the stomach, not hypersensitivity across organs. The aim of this study was to characterize VH in PDS by examining the relationship between the cardiac axis IAc and gastric sensory perception, which is negatively correlated in healthy women with PDS.
Methods
Study design
This case control study was done from November 2018 through December 2019. Participants enrolled in the study without any compensation. The study was approved by the Ethics Review Board of the University Hospital (2,017,316).
Participants
The patient group consisted of 16 women diagnosed with PDS and treated at the Department of Psychosomatic Medicine at a university hospital from November 2018 to December 2019. The diagnosis was made by a psychosomatic physician who treated the patient according to the Rome IV criteria. The control group consisted of 19 healthy women from the university who responded to recruitment posters. The exclusion criteria were age under 20 years, mental illness, and arrhythmia. The diagnosis of a psychiatric disorder was assessed by a psychosomatic physician who treated the patient according to DSM-V. In accordance with the study protocol approved by the Institutional Review Board, written informed consent was obtained from all participants. In the control group, one participant who could not do a task and two others for whom we were unable to measure the heart rate owing to technical difficulties were excluded, leaving the data of 16 healthy volunteers (HV) available for analysis.
Procedure
Before the study, the participants stopped taking medications or alcohol for 24 h and had no caffeine for 12 h. After at least 6 h of fasting, they completed a self-administered questionnaire survey in a quiet room with the room temperature maintained at 25 °C in the morning. They then took the heartbeat tracking task (HTT) and the five-minute water load test (WL5) in that order.
Measures
Heartbeat tracking task
The HTT is a highly reliable method of measuring IAc [5]. A wearable electrocardiograph device (myBeat heart rate sensor, WHS-2 Union Tool Co., Ltd., Tokyo, Japan) was used, and the Ag/AgCl adhesive disposable gel electrodes were affixed to the participants’ chests. Following Schandry’s mental tracking method, the participants were asked to close their eyes and count their heartbeats in random order for four intervals (each lasting 25, 35, 45, and 55 s) divided by 30-second breaks [10, 25]. Participants were instructed to place their hands on their thighs when counting heartbeats and were asked not to perform any actions that would facilitate heartbeat detection. Electrocardiograms were recorded during all the counting periods. The heartbeat perception score was calculated across all four intervals using the following conversion formula:
HTT score = 1/4Σ [1– (|recorded heartbeats – counted heartbeats|)/recorded heartbeats].
This score varies between 0 and 1, with 1 indicating a perfect match between the counted and recorded heartbeats.
Five-minute water load test
Drinking capacity in the WL5 test is used as a scale of gastric sensation [15]. This test has high reproducibility as an index of VH in patients with FD, and it also correlates positively with barostat test results [15]. While barostat testing is expensive and invasive [26], the WL5 is noninvasive because it uses natural expansion stimuli [26, 27], making it easy for clinical use. The experimental method followed that of Herbert et al. [10]. The participants were asked to consume water at a temperature of 25 °C ad libitum from a paper cup at a constant rate for at least 5 min until they reached the point of subjective fullness. The participants were instructed to stop drinking at the first signs of fullness. Behind a screen, the investigator kept refilling the cup out of sight of the participant. Therefore, the cup was “bottomless,” and each participant was blinded as to the amount of water they had consumed. The amount consumed was recorded by the end of the test.
Health-related quality of life (HRQoL)
HRQoL was measured with the Japanese version of the SF-8™ [28]. This item is scored in eight dimensions, comprising general health, physical functioning, role functioning—physical, body pain, vitality, social functioning, mental health, and role functioning—emotional. The physical component summary (PCS) and mental component summary (MCS) were calculated from these scores. Scores range from 62.6 to 19.6 for PCS and 62.7–14.8 for MCS. Higher PCS scores are assessed as higher physical health and higher MCS scores as higher mental health. The mean scores and standard deviations for the Japanese are as follows; PCS mean 49.84 ± 5.99 (Female mean 49.72 ± 6.09), MCS mean 50.09 ± 6.04 (Female mean 49.78 ± 6.05).
Anxiety and depression
Anxiety and depression were measured using the Hospital Anxiety and Depression Scale (HADS) [29]. This consists of seven items about anxiety (HADS-anxiety) and seven items about depression (HADS-depression). Each item is rated on a 0–3-point scale, and the total score range is 0–21-points. The cut-off point for clinical levels was set at a score > 11.
Subjective somatic sensitivity
Psychological sensitivity to somatic symptoms was assessed using the Somatosensory Amplification Scale (SSAS) [30], in which 10 items were rated on a 1–5-point scale. A higher total score indicates a greater tendency to feel somatic sensations as uncomfortable and unpleasant.
Alexithymia
Alexithymia was measured using the Toronto Alexithymia Scale of 20 items (TAS-20) [31]. These items are measured on a scale of 1–5 points, with the total score ranging from 20 to 100. A higher total score indicates a stronger tendency toward alexithymia.
Data analysis
Data are presented as mean and standard deviation (SD). A Mann–Whitney U test was used to compare each score between the two groups. Spearman’s rank correlation coefficient was used to analyze the correlations between the scales in each group. P < 0.05 was regarded as significant in all analyses. The collected data were analyzed using PASW Statistics 18.0 for Windows TM (SPSS Inc., Chicago, IL).
Results
Characteristics of the participants
Table 1 shows the mean and SD of the participants’ age, height, weight, and body mass index. There were no statistically significant differences in any of these characteristics between the PDS and HV groups (P > 0.05).
Table 1.
Participant demographics
| PDS | HV | P a | |||||
|---|---|---|---|---|---|---|---|
| N = 16 | N = 16 | ||||||
| Mean | (SD) | Mean | (SD) | ||||
| Age | 47.5 | (17.4) | 43.3 | (16.1) | 0.584 | ||
| Height (cm) | 155.7 | (6.1) | 158.3 | (5.0) | 0.416 | ||
| Weight (kg) | 45.4 | (9.5) | 50.4 | (5.8) | 0.131 | ||
| Body mass index | 18.8 | (4.0) | 20.2 | (2.6) | 0.546 | ||
a Mann–Whitney U test
PDS, postprandial distress syndrome; HV, healthy volunteers; SD, standard deviation
Differences between PDS and HV groups in all measures
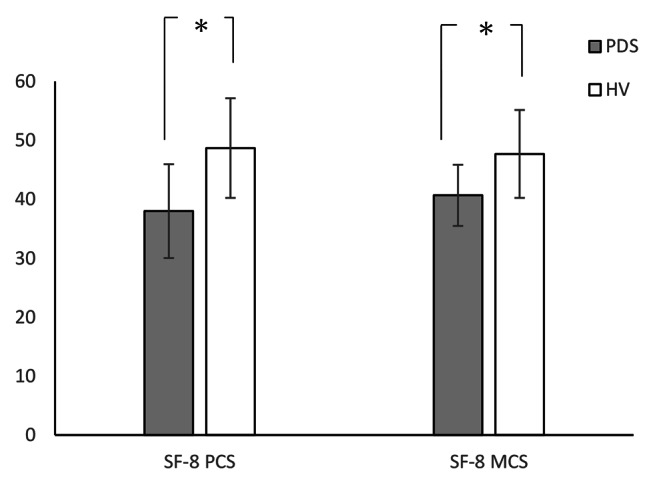
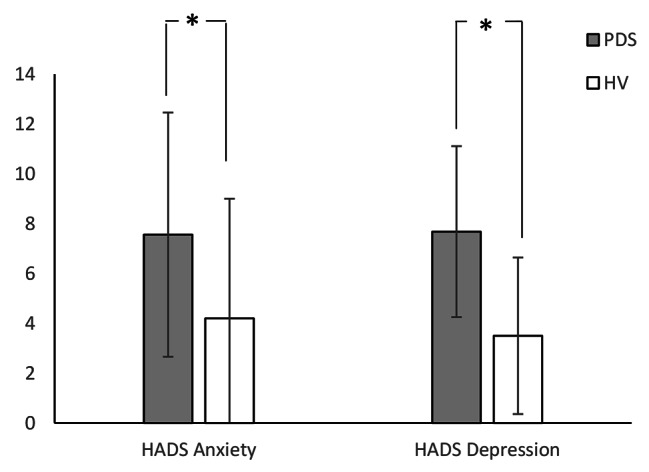
Table 2 shows the means, SDs, and differences of all measures. The WL5 values were significantly lower in the PDS group (PDS: mean 360.9 ± 170 mL; HV: mean 644.1 ± 297 mL; P = 0.009; Fig. 1). However, there was no difference in HTT scores between the PDS and HV groups (PDS: mean 0.599 ± 0.175; HV: 0.623 ± 0.181; P = 0.647; Fig. 2). With respect to HRQoL, both PCS (PDS: 38.0 ± 8.0; HV: 48.7 ± 5.2; P < 0.001) and MCS (PDS: 40.7 ± 8.5; HV: 47.7 ± 7.5; P = 0.017) were significantly lower in the PDS group compared with the HV group (Fig. 3). While the HADS-depression score was significantly higher in the PDS group than in the HV group (PDS: 7.7 ± 4.8; HV: 3.5 ± 3.1; P = 0.007; Fig. 4), HADS-depression and HADS-anxiety scores were within normal limits in both groups (cut off point: <11). SSAS and TAS-20 showed no significant differences between the groups (Figs. 5 and 6).
Table 2.
WL5, HTT scores, and other measures: differences between PDS and HV
| Variable | PDS | HV | P a | ||||
|---|---|---|---|---|---|---|---|
| (N = 16) | (N = 16) | ||||||
| Mean | (SD) | Mean | (SD) | ||||
| WL5 (ml) | 360.9 | (170.0) | 644.1 | (297.0) | 0.009* | ||
| HTT | 0.599 | (0.175) | 0.623 | (0.181) | 0.647 | ||
| PCS | 38.0 | (8.0) | 48.7 | (5.2) | 0.000* | ||
| MCS | 40.7 | (8.5) | 47.7 | (7.5) | 0.017* | ||
| HADS-anxiety | 7.6 | (4.9) | 4.2 | (3.4) | 0.067 | ||
| HADS-depression | 7.7 | (4.8) | 3.5 | (3.1) | 0.007* | ||
| SSAS | 33.1 | (4.9) | 29.8 | (5.4) | 0.094 | ||
| TAS-20 | 55.3 | (12.0) | 48.2 | (13.0) | 0.094 | ||
a Mann–Whitney U test; * P < 0.05
PDS, postprandial distress syndrome; HV, healthy volunteers; SD, standard deviation; WL5, five-minute water load test; HTT, heartbeat tracking task; PCS, physical component summary; MCS, mental component summary; HADS, Hospital Anxiety and Depression Scale; SSAS, Somatosensory Amplification Scale; TAS-20, Toronto Alexithymia Scale of 20 items
Fig. 1.
Drinking capacity (WL5)
Fig. 2.
HTT score
Fig. 3.
SF-8 score
Fig. 4.
HADS score
Fig. 5.
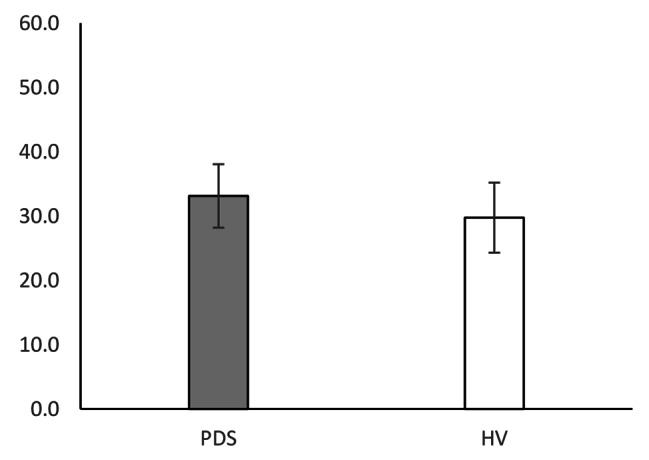
SSAS score
Fig. 6.
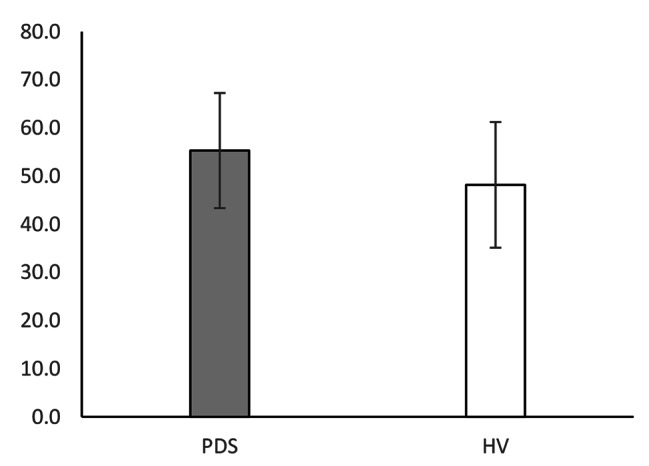
TAS-20 score
Correlation between WL5, HTT, and other measures in patients with PDS
Table 3 shows the correlation coefficients for each of the measured items within the PDS group. The WL5 and HTT scores were not correlated (r = − 0.156, P > 0.05). However, there was a strong positive significant correlation between WL5 and MCS scores (r = 0.762, P < 0.001), and a moderate negative significant correlation with the HADS-depression score (r = − 0.555, P < 0.05). The HTT was not correlated with any other measurement, including HADS-anxiety, in the PDS group.
Table 3.
Spearman’s rank correlation between WL5, the HTT score, and other measures in patients with PDS
| WL5 | HTT | |||||||
|---|---|---|---|---|---|---|---|---|
| r | P b | r | P b | |||||
| HTT | − 0.156 | 0.564 | ||||||
| PCS | 0.426 | 0.099 | − 0.176 | 0.513 | ||||
| MCS | 0.762 | 0.001* | 0.000 | 1.000 | ||||
| HADS-anxiety | − 0.378 | 0.149 | 0.094 | 0.728 | ||||
| HADS-depression | − 0.555 | 0.026* | 0.201 | 0.456 | ||||
| SSAS | 0.209 | 0.437 | − 0.060 | 0.826 | ||||
| TAS-20 | 0.214 | 0.427 | − 0.108 | 0.692 | ||||
b Spearman’s rank correlation
WL5, five-minute water load test; HTT, heartbeat tracking task; PDS, postprandial distress syndrome; PCS, physical component summary; MCS, mental component summary; HADS, Hospital Anxiety and Depression Scale; SSAS, Somatosensory Amplification Scale; TAS-20, Toronto Alexithymia Scale of 20 items
Correlation between WL5, HTT, and other measures in the HV group
Table 4 shows the correlation coefficients for each of the measured items within the HV group. There was a moderate negative significant correlation between WL5 and HTT scores (r = − 0.509, P < 0.05) and a moderate negative significant correlation between WL5 and HADS-depression scores (r = − 0.524, P < 0.05). The HTT was not correlated with any other measurement, including HADS-anxiety, in the HV group.
Table 4.
Spearman’s rank correlation between WL5, the HTT score, and other measures in HV
| WL5 | HTT | |||||||
|---|---|---|---|---|---|---|---|---|
| r | Pb | r | Pb | |||||
| HTT | − 0.509 | 0.044* | ||||||
| PCS | 0.024 | 0.931 | − 0.178 | 0.509 | ||||
| MCS | 0.106 | 0.696 | 0.434 | 0.093 | ||||
| HADS-anxiety | − 0.460 | 0.073 | − 0.061 | 0.823 | ||||
| HADS-depression | − 0.524 | 0.037* | − 0.078 | 0.775 | ||||
| SSAS | − 0.340 | 0.197 | − 0.033 | 0.905 | ||||
| TAS-20 | − 0.103 | 0.704 | − 0.362 | 0.168 | ||||
b Spearman’s rank correlation
WL5, five-minute water load test; HTT, heartbeat tracking task; HV, healthy volunteers; PCS, physical component summary; MCS, mental component summary; HADS, Hospital Anxiety and Depression Scale; SSAS, Somatosensory Amplification Scale; TAS-20, Toronto Alexithymia Scale of 20 items
Discussion
Our aim was to determine the relationship between WL5 drinking volume and HTT in PDS and HV groups. The results showed no significant difference in HTT performance between the groups; only the drinking water volume was significantly lower in the PDS group. In addition, the perceptual relationship between organs observed in HV was lost in patients with PDS.
In PDS, the cardiac-gastric perceptual relationship is lost and the interoceptive perceptual function of the stomach is abnormal. The relationship between the WL5 drinking volume and HTT score in HV was similar to that in previous studies [10, 11]. Patients with PDS showed hypersensitivity limited to the stomach, and the perceptual relationship between bodily axes that is present in HV was lost. Compared with patients with EPS, those with PDS have comparable brain activity during water loading, but resting activity is not a problem [23]. Irritable bowel syndrome (IBS), which is included in FGID, is also a syndrome of organ-specific hypersensitivity such as FD [19, 20], while IBS and healthy individuals show no difference in cardiac IAc [32]. Furthermore, it has been reported that heartbeat perception does not differ between healthy persons and those with fibromyalgia [33]. The findings of these previous studies were consistent with our results in that no hypersensitivity was found in the cardiac tissue, which is not a symptomatic site. Moreover, the results of the lost cardiac-gastric perceptual relationship observed in HV indicated that the interoceptive perceptual function of PDS patients may be altered. Although our study was unable to determine why this change in interoceptive perceptual function occurred, we consider several possibilities. Fibromyalgia is a disease that is thought to involve hypersensitivity; however, the hypersensitivity is limited to the threat modality and not to the innocuous organ of the heart [33]. It has also been reported that the satiety and bloating in FD patients are induced by cognitive influences [34]. As in fibromyalgia, changes in gastric interoceptive perception may occur in PDS due to a specific awareness regarding gastric sensations and eating.
The SSAS, a measure of severity of subjective visceral sensory hypersensitivity, did not differ between PDS and HV; previous studies have shown that FD tends to have mildly higher SSAS [35, 36], and our results differ from the SSAS characteristics assumed in FD patients. It is possible that this is due to the fact that PDS symptoms appear mainly as postprandial symptoms. Postprandial symptoms in FD are influenced by hypersensitivity to peripheral stretch rather than central influences such as subjective hypersensitivity [37]. The result that subjective sensitivity in PDS is not related to the volume of water loading is similar to that reported by Steinsvik. The loss of the cardiac-gastric perceptual relationship, the lack of a SSAS relationship, and the lack of any difference from HV indicates that the VH of PDS affects only the stomach, not all organs.
Testing WL5 and HTT simultaneously can be helpful in determining whether the VH is localized or widespread. Although testing across bodily axes has been examined in relation to gastric and rectal barostats, VH, and somatic pain thresholds, there has been no simple method of testing [17, 20]. We have identified different sensory perception relationships between bodily axes in patients with PDS and HV. The combination of two tests that can look at the relationship between perception of the stomach and different bodily axes provides clues to understanding whether VH with FD is localized or widespread.
Considering the negative correlation between drinking volume and depression in both PDS and HV, it is possible that gastric perception is influenced by specific emotional experiences in depressive mood states. The anterior cingulate cortex (ACC) is an important site in visceral sensory processing associated with emotional experiences [38]. The ACC is responsible for the processing of negative emotions and is involved in the activity patterns seen in depression [39, 40]. The ACC is activated by gastric balloon stimulation [38] and is involved in the emotionally motivated response to visceral pain in negative emotional states [41]. FD is associated with activation of the ACC, failure of AIC activation, and failure to integrate gastric sensation, and accompanying psychological states may affect gastric sensory processing [24, 42]. Our results showed a similar negative relationship between the stomach and heart in HV who exhibited significantly lower HADS-depression scores than patients with PDS. This suggests that depressive symptoms as well as negative emotional states may affect gastric sensations, especially satiety and thresholds for extensor stimulation.
Future directions
Patients with EPS may show a different perceptual relationship between bodily axes than those with PDS; EPS has a stronger central hyperactivity at rest than PDS, which is involved in its pathogenesis [23]. It has been suggested that the difference between localized or widespread VH may be a difference in the intensity of central versus peripheral pathology [20]. In the future, it may be possible to clarify the difference in pathophysiology between EPS and PDS by examining the relationship between cardiac IAc and gastric perception in patients with EPS as well. Second, it may be necessary to examine the involvement of comorbid psychiatric symptoms in patients with FD as well as their multifaceted emotional state in hypersensitivity. Because the present study used the HADS, which focuses on symptoms, it was not possible to determine whether specific emotions are involved in stomach perception. Compared with anxiety and depression, the association between negative emotional states that are not symptoms and sensory perception has not been the topic of much research.
Limitations
First, the method for measuring IAc in the gastric region is underdeveloped, and the water loading test may be influenced by gastric accommodation, adaptive relaxation, and gastric emptying. Second, we did not measure sensory perception in other bodily axes, such as the respiratory system. Finally, our study has a small sample size and a single-center design, in addition to a sample with low depression and anxiety scores and no comorbid psychiatric disorders, which are common characteristics of patients with FD. Therefore, we must be cautious about generalizing our results.
Conclusion
Patients with PDS showed visceral hypersensitivity limited to the stomach, and the perceptual relationships between organs observed in HV were lost. PDS may cause abnormal interoceptive perceptual function of sensory stimuli in the stomach; the combination of the WL5 and HTT, tests that examine sensation in different bodily axes, is useful in assessing whether VH is localized or widespread.
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
Abbreviations
- ACC
anterior cingulate cortex
- AIC
anterior insular cortex
- EPS
epigastric pain syndrome
- FD
functional dyspepsia
- FGID
functional gastrointestinal disorder
- HADS
Hospital Anxiety and Depression Scale
- HRQoL
Health-related quality of life
- HTT
heartbeat tracking task
- HV
healthy volunteers
- IAc
interoceptive accuracy
- IBS
irritable bowel syndrome
- MCS
mental component summary
- PCS
physical component summary
- PDS
postprandial distress syndrome
- SD
standard deviation
- SSAS
Somatosensory Amplification Scale
- TAS-20
Toronto Alexithymia Scale of 20 items
- VH
visceral hypersensitivity
- WL5
five-minute water load test
Authors’ contributions
KY and TA designed the study and wrote the manuscript. KY and TA collected and entered data. All authors read and approved the final manuscript.
Funding
This study was not supported by any external funding.
Data Availability
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Declarations
Ethics approval and consent to participate
In accordance with the study protocol approved by the institutional ethics committee of Kansai Medical University Hospital, written informed consent was obtained from all participants.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Craig AD. How do you feel? Interoception: the sense of the physiological condition of the body. Nat Rev Neurosci. 2002;3:655–66. doi: 10.1038/nrn894. [DOI] [PubMed] [Google Scholar]
- 2.Chen WG, Schloesser D, Arensdorf AM, Simmons JM, Cui C, Valentino R, et al. The Emerging Science of Interoception: sensing, integrating, interpreting, and regulating signals within the self. Trends Neurosci. Volume 44. Elsevier Ltd; 2021. pp. 3–16. [DOI] [PMC free article] [PubMed]
- 3.Critchley HD, Harrison NA. Visceral influences on Brain and Behavior. Neuron. Volume 77. Elsevier Inc.; 2013. pp. 624–38. [DOI] [PubMed]
- 4.Garfinkel SN, Critchley HD et al. Interoception, emotion and brain: new insights link internal physiology to social behaviour. Commentary on:: “Anterior insular cortex mediates bodily sensibility and social anxiety” by Terasawa. (2012). Soc Cogn Affect Neurosci. 2013;8:231–4. [DOI] [PMC free article] [PubMed]
- 5.Garfinkel SN, Seth AK, Barrett AB, Suzuki K, Critchley HD. Knowing your own heart: distinguishing interoceptive accuracy from interoceptive awareness. Biol Psychol Elsevier B V. 2015;104:65–74. doi: 10.1016/j.biopsycho.2014.11.004. [DOI] [PubMed] [Google Scholar]
- 6.Gogolla N. The insular cortex. Curr Biol Elsevier. 2017;27:R580–6. doi: 10.1016/j.cub.2017.05.010. [DOI] [PubMed] [Google Scholar]
- 7.Suksasilp C, Garfinkel SN. Towards a comprehensive assessment of interoception in a multi-dimensional framework. Biol Psychol Elsevier B V. 2022;168:108262. doi: 10.1016/j.biopsycho.2022.108262. [DOI] [PubMed] [Google Scholar]
- 8.Garfinkel SN, Manassei MF, Hamilton-Fletcher G, den Bosch YI, Critchley HD, Engels M. Interoceptive dimensions across cardiac and respiratory axes. Philos Trans R Soc B Biol Sci. 2016;371. [DOI] [PMC free article] [PubMed]
- 9.Ferentzi E, Bogdány T, Szabolcs Z, Csala B, Horváth Á, Köteles F. Multichannel investigation of interoception: sensitivity is not a generalizable feature. Front Hum Neurosci. 2018;12:1–9. doi: 10.3389/fnhum.2018.00223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Herbert BM, Muth ER, Pollatos O, Herbert C. Interoception across modalities: on the relationship between cardiac awareness and the sensitivity for gastric functions. Tsakiris M, editor. PLoS One. 2012;7:e36646. [DOI] [PMC free article] [PubMed]
- 11.Van Dyck Z, Vögele C, Blechert J, Lutz APC, Schulz A, Herbert BM. The water load test as a measure of gastric interoception: development of a two-stage protocol and application to a healthy female population. PLoS ONE. 2016;11:1–14. doi: 10.1371/journal.pone.0163574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tack J, Bisschops R, Sarnelli G. Pathophysiology and treatment of functional dyspepsia. Gastroenterology. 2004;127:1239–55. doi: 10.1053/j.gastro.2004.05.030. [DOI] [PubMed] [Google Scholar]
- 13.Simrén M, Törnblom H, Palsson OS, Van Tilburg MAL, Van Oudenhove L, Tack J, et al. Visceral hypersensitivity is associated with GI symptom severity in functional GI disorders: consistent findings from five different patient cohorts. Gut. 2018;67:255–62. doi: 10.1136/gutjnl-2016-312361. [DOI] [PubMed] [Google Scholar]
- 14.Tack J, Caenepeel P, Fischler B, Piessevaux H, Janssens J. Symptoms associated with hypersensitivity to gastric distention in functional dyspepsia. Gastroenterology. 2001;121:526–35. doi: 10.1053/gast.2001.27180. [DOI] [PubMed] [Google Scholar]
- 15.Jones MP, Hoffman S, Shah D, Patel K, Ebert CC. The water load test: observations from healthy controls and patients with functional dyspepsia. Am J Physiol - Gastrointest Liver Physiol. 2003;284:896–904. doi: 10.1152/ajpgi.00361.2002. [DOI] [PubMed] [Google Scholar]
- 16.Samsom M, Verhagen MAMT, Henegouwen GPV, Smout AJPM. Abnormal clearance of exogenous acid and increased acid sensitivity of the proximal duodenum in dyspeptic patients. Gastroenterology. 1999;116:515–20. doi: 10.1016/S0016-5085(99)70171-X. [DOI] [PubMed] [Google Scholar]
- 17.Li X, Cao Y, Wong RKM, Ho KY, Wilder-Smith CH. Visceral and somatic sensory function in functional dyspepsia. Neurogastroenterol Motil. 2013;25:246–54. doi: 10.1111/nmo.12044. [DOI] [PubMed] [Google Scholar]
- 18.Clauwaert N, Jones MP, Holvoet L, Vandenberghe J, Vos R, Tack J et al. Associations between gastric sensorimotor function, depression, somatization, and symptom-based subgroups in functional gastroduodenal disorders: are all symptoms equal? Neurogastroenterol Motil. 2012;24. [DOI] [PubMed]
- 19.Bouin M, Plourde V, Boivin M, Riberdy M, Lupien F, Laganière M, et al. Rectal distention testing in patients with irritable bowel syndrome: sensitivity, specificity, and predictive values of pain sensory thresholds. Gastroenterology. 2002;122:1771–7. doi: 10.1053/gast.2002.33601. [DOI] [PubMed] [Google Scholar]
- 20.Bouin M, Lupien F, Riberdy M, Boivin M, Plourde V, Poitras P. Intolerance to visceral distension in functional dyspepsia or irritable bowel syndrome: an organ specific defect or a pan intestinal dysregulation? Neurogastroenterol Motil. 2004;16:311–4. doi: 10.1111/j.1365-2982.2004.00511.x. [DOI] [PubMed] [Google Scholar]
- 21.Faure C, Wieckowska A. Somatic referral of visceral sensations and rectal sensory threshold for Pain in Children with Functional Gastrointestinal Disorders. J Pediatr. 2007;150:66–71. doi: 10.1016/j.jpeds.2006.08.072. [DOI] [PubMed] [Google Scholar]
- 22.Bouin M, Meunier P, Riberdy-Poitras M, Poitras P. Pain hypersensitivity in patients with functional gastrointestinal disorders: a gastrointestinal-specific defect or a general systemic condition? Dig Dis Sci. 2001;46:2542–8. doi: 10.1023/A:1012356827026. [DOI] [PubMed] [Google Scholar]
- 23.Chen Y, Wang R, Hou B, Feng F, Fang X, Zhu L, et al. Regional brain activity during rest and gastric water load in subtypes of functional dyspepsia: a preliminary brain functional magnetic resonance imaging study. J Neurogastroenterol Motil. 2018;24:268–79. doi: 10.5056/jnm17076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Van Oudenhove L, Aziz Q. The role of psychosocial factors and psychiatric disorders in functional dyspepsia. Nat Rev Gastroenterol Hepatol Nature Publishing Group. 2013;10:158–67. doi: 10.1038/nrgastro.2013.10. [DOI] [PubMed] [Google Scholar]
- 25.Schandry R. Heart beat perception and emotional experience. Psychophysiology. 1981;18:483–8. doi: 10.1111/j.1469-8986.1981.tb02486.x. [DOI] [PubMed] [Google Scholar]
- 26.Kim DY, Myung SJ, Camilleri M. Novel testing of human gastric motor and sensory functions: Rationale, methods, and potential applications in clinical practice. Am J Gastroenterol. 2000;95:3365–73. doi: 10.1111/j.1572-0241.2000.03346.x. [DOI] [PubMed] [Google Scholar]
- 27.Andresen V. Visceral sensitivity testing. Best pract res clin gastroenterol. Volume 23. Elsevier Ltd; 2009. pp. 313–24. [DOI] [PubMed]
- 28.Fukuhara S, Suzukamo Y. Manual of the SF-8 japanese version. Kyoto: Institute for Health Outcomes & Process Evaluation Research; 2004. [Google Scholar]
- 29.Zigmond AS, Snaith RP. The Hospital anxiety and Depression Scale. Acta Psychiatr Scand. 1983;67:361–70. doi: 10.1111/j.1600-0447.1983.tb09716.x. [DOI] [PubMed] [Google Scholar]
- 30.Barsky AJ, Wyshak G, Klerman GL. The somatosensory amplification scale and its relationship to hypochondriasis. J Psychiatr Res. 1990;24:323–34. doi: 10.1016/0022-3956(90)90004-A. [DOI] [PubMed] [Google Scholar]
- 31.Taylor GJ, Bagby RM, Parker JDA. The 20-Item Toronto Alexithymia Scale: IV. Reliability and factorial validity in different languages and cultures. J Psychosom Res. 2003;55:277–83. doi: 10.1016/S0022-3999(02)00601-3. [DOI] [PubMed] [Google Scholar]
- 32.Gajdos P, Chrisztó Z, Rigó A. The association of different interoceptive dimensions with functional gastrointestinal symptoms. J Health Psychol. 2020;135910532092942. [DOI] [PubMed]
- 33.Rost S, Van Ryckeghem DML, Schulz A, Crombez G, Vögele C. Generalized hypervigilance in fibromyalgia: normal interoceptive accuracy, but reduced self-regulatory capacity. J Psychosom Res. 2017;93:48–54. doi: 10.1016/j.jpsychores.2016.12.003. [DOI] [PubMed] [Google Scholar]
- 34.Feinle-Bisset C, Meier B, Fried M. Role of cognitive factors in symptom induction following high and low fat meals in patients with functional dyspepsia. Gut. 2003;52:1414–8. doi: 10.1136/gut.52.10.1414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jones MP, Ann Schettler RN, Olden K, Crowell MD. Alexithymia and Somatosensory amplification in functional dyspepsia. Psychosomatics. 2004;45:508–16. doi: 10.1176/appi.psy.45.6.508. [DOI] [PubMed] [Google Scholar]
- 36.Jones MP, Roth LM, Crowell MD. Symptom reporting by functional dyspeptics during the water load test. Am J Gastroenterol. 2005;100:1334–9. doi: 10.1111/j.1572-0241.2005.40802.x. [DOI] [PubMed] [Google Scholar]
- 37.Steinsvik EK, Valeur J, Hausken T, Gilja OH. Postprandial symptoms in patients with functional dyspepsia and irritable bowel syndrome: relations to ultrasound measurements and psychological factors. J Neurogastroenterol Motil. 2020;26:96–105. doi: 10.5056/jnm19072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Stephan E. Functional neuroimaging of gastric distention. J Gastrointest Surg. 2003;7:740–9. doi: 10.1016/S1091-255X(03)00071-4. [DOI] [PubMed] [Google Scholar]
- 39.Mayberg HS, Liotti M, Brannan SK, McGinnis S, Mahurin RK, Jerabek PA, et al. Reciprocal limbic-cortical function and negative Mood: converging PET findings in Depression and normal sadness. Am J Psychiatry. 1999;156:675–82. doi: 10.1176/ajp.156.5.675. [DOI] [PubMed] [Google Scholar]
- 40.Drevets WC, Öngür D, Price JL. Neuroimaging abnormalities in the subgenual prefrontal cortex: implications for the pathophysiology of familial mood disorders. Mol Psychiatry. 1998;3:220–6. doi: 10.1038/sj.mp.4000370. [DOI] [PubMed] [Google Scholar]
- 41.Coen SJ, Yágüez L, Aziz Q, Mitterschiffthaler MT, Brammer M, Williams SCR, et al. Negative Mood affects Brain Processing of visceral sensation. Gastroenterology. Volume 137. AGA Institute American Gastroenterological Association; 2009. pp. 253–261e2. [DOI] [PubMed]
- 42.Farmer AD, Aziz Q, Tack J, Van Oudenhove L. The future of neuroscientific research in functional gastrointestinal disorders: integration towards multidimensional (visceral) pain endophenotypes? J Psychosom Res Elsevier Inc. 2010;68:475–81. doi: 10.1016/j.jpsychores.2009.12.006. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.


