Abstract
Background
We sought to predict clinically meaningful changes in physical, sexual, and psychosocial well-being for women undergoing cancer-related mastectomy and breast reconstruction 2 years after surgery using machine learning (ML) algorithms trained on clinical and patient-reported outcomes data.
Patients and Methods
We used data from women undergoing mastectomy and reconstruction at 11 study sites in North America to develop three distinct ML models. We used data of ten sites to predict clinically meaningful improvement or worsening by comparing pre-surgical scores with 2 year follow-up data measured by validated Breast-Q domains. We employed ten-fold cross-validation to train and test the algorithms, and then externally validated them using the 11th site’s data. We considered area-under-the-receiver-operating-characteristics-curve (AUC) as the primary metric to evaluate performance.
Results
Overall, between 1454 and 1538 patients completed 2 year follow-up with data for physical, sexual, and psychosocial well-being. In the hold-out validation set, our ML algorithms were able to predict clinically significant changes in physical well-being (chest and upper body) (worsened: AUC range 0.69–0.70; improved: AUC range 0.81–0.82), sexual well-being (worsened: AUC range 0.76–0.77; improved: AUC range 0.74–0.76), and psychosocial well-being (worsened: AUC range 0.64–0.66; improved: AUC range 0.66–0.66). Baseline patient-reported outcome (PRO) variables showed the largest influence on model predictions.
Conclusions
Machine learning can predict long-term individual PROs of patients undergoing postmastectomy breast reconstruction with acceptable accuracy. This may better help patients and clinicians make informed decisions regarding expected long-term effect of treatment, facilitate patient-centered care, and ultimately improve postoperative health-related quality of life.
Supplementary Information
The online version contains supplementary material available at 10.1245/s10434-023-13971-w.
Keywords: Machine learning, Postmastectomy breast reconstruction, PRO, QOL
Postmastectomy breast reconstruction (PMBR) has important long-term effects on quality of life (QOL).1 With advances in reconstructive techniques and an increasing number of women undergoing risk reducing mastectomy, there is a trend toward a rising demand of PMBR.2 PBMR is beneficial for improving body image and minimizing the negative impact of mastectomy on QOL.3 However, facing different breast reconstruction treatment options (e.g., implant-based versus autologous), many women have difficulties making high-quality decisions due to anecdotal methods used for patient education.4
Clinical studies have been conducted to compare different options and evaluate the outcome of PMBR, to provide insights into treatment options, and to inform patients’ decision-making.5–7 For example, a previous prospective cohort study concluded that autologous reconstruction offers benefits over implant-based reconstruction in terms of QOL.1 However, recommendations and conclusions derived from group-level studies are not suitable for a specific individual’s situation. Tailoring individual care to match each patient’s expected QOL after reconstruction is necessary and warrants further investigation. Fortunately, the emergence of cutting-edge computational techniques—machine learning (ML)—accompanied by the usage of individual patient-reported outcome (PRO) data provides the potential to address this knowledge gap and to help patients and clinicians make informed decisions before the initiation of breast reconstruction procedures to facilitate patient-centered care.
As a branch of artificial intelligence, ML involves training algorithms to identify intricate patterns within data and make precise predictions.8 By learning patterns from data, ML has the unique capability to predict future outcomes at the individual level. This ability to provide personalized predictions and recommendations tailored to individual patients has the potential to greatly enhance patient care, leading to growing enthusiasm for the application of ML techniques in addressing clinical problems. Trained ML models using supervised learning techniques have consistently demonstrated exceptional performance across a range of challenging prediction tasks in the medical field. These tasks include, but are not limited to, prediction of mortality in cancer patients,9 natural language processing,10 prediction of financial toxicity caused by cancer treatment,11 and classification of benign or malignant tumor.12 The success of ML in these predictive tasks can be attributed to its strong capability to identify subtle nonlinear interactions between events and outcomes within multidimensional data.13 This ability allows ML models to uncover complex relationships that may not be discernible through traditional methods, resulting in more accurate predictions and improved decision-making across various healthcare domains.
Machine learning algorithms have previously achieved excellent performance in predicting breast satisfaction, one of the key outcomes for women undergoing PMBR, both at 1 and 2 year follow-up.14,15 In this comprehensive study, we aimed to develop and validate ML algorithms to accurately predict clinically meaningful, long-term changes in physical, sexual, and psychosocial well-being for women undergoing PMBR at 2 year follow-up to enhance decision-making in this area, shifting a focus from satisfaction to the critical areas of health-related QOL, using the same study population as those prior two studies, and affording unique insights into the PRO on the health-related QOL prediction for women with breast cancer.
Patients and Methods
Study Participants
This study cohort was a subgroup of the international Mastectomy Reconstruction Outcomes Consortium (MROC, NCT01723423) study that was conducted at 11 study sites in both Canada and the USA between 2012 and 2017. A total of 3058 women undergoing PMBR were recruited as described in detail elsewhere.1,14
Inclusion criteria were women aged 18 years or older, undergoing first time bilateral or unilateral, immediate or delayed PMBR for risk reducing or therapeutic purposes. These patients could have undergone implant-based and/or autologous reconstruction, based on the surgeon’s recommendation or their preferences. Exclusion criteria were patients with previous failed breast reconstruction. For the present analysis, patients with unreported PROs at baseline or 2 year follow-up were also excluded.
All included study sites received ethical approval from the respective institutional review board.
Study Design
Patient-reported physical, sexual, and psychosocial well-being were evaluated before the initiation of the reconstruction procedure and at 2 year follow up by the validated and reliable BREAST-Q.16 Cronbach’s alpha coefficients are reportedly greater than 0.8 and the score of each scale ranges from 0 (worst well-being) to 100 (best well-being).17
Minimal clinically important difference (MCID) estimates have previously been reported: MCID in physical well-being (chest and upper body) is a score difference of at least 3, and a score difference of at least 4 in both sexual and psychosocial well-being.18 We defined three types of outcomes for each domain when comparing baseline PROs with those at 2 year follow-up: outcomes of health-related QOL were (1) worsened if the 2-year follow-up score was reduced at least by the respective MCIDs compared to baseline, (2) improved if the 2-year follow-up score was increased at least by the respective MCIDs compared with baseline, or (3) otherwise stable.
To facilitate the construction of ML predictive models, we recoded the outcome into binary (i.e., improved versus not improved, worsened versus not worsened).
Algorithm Selection
We trained three ML algorithms with varying levels of complexity for each domain given their demonstrated promising performance in published similar medical studies conducted by our team,14,15,19 and reported findings following relevant guidelines (TRIPOD).20
We briefly describe each algorithm below. A detailed description can be found in online supplemental documents of our previously published study.14
-
Logistic regression (LR) with elastic net penalty.
The LR with penalized magnitudes of coefficients is known for its easy-to-interpret prediction process, ability to avoid overfitting, and enhanced generalizability on new datasets.13
-
Extreme gradient boosting (XGBoost) tree.
The XGboost tree, as an ensemble-learning algorithm of several built models, is suitable for complex classification tasks due to its enhanced capability in identifying complex relationships among predictors.21
-
Neural network.
A neural network has a unique network structure consisting of connected units that is inspired by the structure of the human cortex. This enables identification of complex patterns within the dataset and capturing nonlinear relations among the input and output variables.
Data Preparation
We split the 11-site data into a development set of 10 sites and a validation set of 1 site. The validation site with initials of “BW” was chosen based on the number of events, as reported in our previous research on breast satisfaction prediction.14 We included four patients, five preoperative PRO, and seven clinical variables as predictive factors (Table 1 in Supplement 1).
For data preparation, we imputed missing values using the K-nearest neighbors algorithm (K = 5), removed zero variance variables, centered and scaled all numerical variables, and dummied all categorical variables with one hot encoding. Variables having an absolute correlation with other variables over a threshold of 0.9 were removed, to address the multicollinearity issues.
For ML algorithm training and internal testing on the development set, we adopted ten-fold cross-validation with three repetitions and a hypergrid search to train the models and tune hyperparameters. We computed sensitivity, specificity, the area-under-the-receiver-operating-characteristics-curve (AUC), precision, and recall, to assess model performance in each fold. We embraced the “Kappa” metric to evaluate final model performance in the test fold because of the possible class-imbalance effect. We chose the simplest model that was within a 3% tolerance of the empirically optimal model as the final model to reduce overfitting and improve generalizability to new datasets.22
Based on our previous research, we excluded five socioeconomic and racial variables to avoid racial bias.23 We compared model performance among each racial group to evaluate the fairness of ML algorithms.24
Analysis Strategies
The predictive performance of the ML algorithms were measured via accuracy and AUC. Point estimates along with a 95% confidence interval (CI) are reported. To provide insights into model predictions and improve transparency and interpretability, we reported regularized coefficients for the LR with elastic net penalty, Shapley Additive explanations (SHAP) values for XGBoost tree,25 and local interpretable model-agnostic explanations (LIME) for the neural network.26 For comparison, traditional binary logistic regression models are provided as well. To assess the fairness of model performance, we compared the predictive performance of the models across all racial groups in the validation set. To assess algorithm calibration in the validation set, we plot calibration plots with predicted versus observed rates of outcome.27 We conducted the Spiegelhalter Z test for calibration accuracy assessment,28 with a p-value greater than 0.05 indicating the model was well calibrated. We calculated the scaled Brier score with a range between 0 (perfect predictive performance) and 1 (poor predictive performance).29
Lastly, we conduct receiver operating characteristic curve comparisons among ML model performance in both development and validation sets for each scale to assess their statistical significance. We plotted AUC of models to predict improved and worsened health-related QOL outcomes together to get the full picture of the performance of the trained ML model at each scale.
We carried out all analyses within the “R” programming environment with version 4.2.1. and developed ML models using the “caret” package.
Results
Clinical and Demographic Characteristics
The analysis set comprised 1538 participants for physical well-being (1320 development and 218 validation), 1454 for sexual well-being (1247 development and 207 validation), and 1538 for psychosocial well-being (1319 development and 219 validation) to train and validate ML models as shown in Fig. 1. The baseline demographic and clinical characteristics for all three BREAST-Q scales are presented in Table 1 (Table 1 with details in Supplement 2). Two years after breast reconstruction, 563 (36.6%) patients experienced improved physical well-being, 592 (40.7%) improved sexual well-being, and 769 (50.0%) improved psychosocial well-being, whereas 737 (47.9%), 647 (44.5%), and 453 (29.5%) patients experienced worsening in physical, sexual, and psychosocial well-being 2 years after surgery, respectively.
Fig. 1.
Study design and flow of participants
Table 1.
Participant baseline characteristics and health outcomes
| Physical well-being (chest and upper body) | Sexual well-being | Psychosocial well-being | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Development set | Validation set | p-Valuea | Development set | Validation set | p-Valuea | Development set | Validation set | p-Valuea | |
| (n = 1320) | (n = 218) | (n =1247) | (n = 207) | (n =1319) | (n =219) | ||||
| Patient variables | |||||||||
| BMId, mean (SD) (kg/m2) | 26.63 (5.48) | 25.72 (4.78) | 0.011b | 26.54 (5.43) | 25.53 (4.76) | 0.006b | 26.61 (5.48) | 25.70 (4.78) | 0.011b |
| Preoperative patient-reported outcome data | |||||||||
| BREAST-Q physical well-being chest and upper bodyd, mean (SD), 0–100 | 78.46 (14.55) | 81.22 (14.32) | 0.009b | 78.62 (14.37) | 81.21 (14.09) | 0.015b | 78.45 (14.54) | 81.24 (14.29) | 0.008b |
| BREAST-Q physical well-being abdomend, mean (SD), 0–100 | 89.26 (13.79) | 91.07 (11.41) | 0.038b | 89.35 (13.55) | 91.14 (11.48) | 0.046b | 89.27 (13.79) | 91.03 (11.39) | 0.042b |
| Clinical variables | |||||||||
| Reconstruction techniqued | |||||||||
| Tissue expander (TE), no. (%) | 691 (52.3) | 129 (59.2) | 0.061c | 654 (52.4) | 124 (59.9) | 0.046c | 690 (52.3) | 130 (59.4) | 0.053c |
| Superficial inferior epigastric artery (SIEA) flap, no. (%) | 48 (3.6) | 0 (0.00) | 0.004c | 42 (3.4) | 0 (0.0) | 0.007c | 48 (3.6) | 0 (0.0) | 0.004c |
| Axillary interventiond | |||||||||
| Axillary lymph node dissection (ALND), no. (%) | 354 (26.8) | 43 (19.7) | 0.027c | 339 (27.2) | 41 (19.8) | 0.025c | 352 (26.7) | 43 (19.6) | 0.027c |
| Sentinel lymph node biopsy (SLNB), no. (%) | 579 (43.9) | 112 (51.4) | 0.039c | 545 (43.7) | 108 (52.2) | 0.023c | 579 (43.9) | 113 (51.6) | 0.034c |
| Socioeconomic and racial data | |||||||||
| Education level | |||||||||
| High school degree, no. (%) | 113 (8.6) | 9 (4.1) | 0.024c | 97 (7.8) | 8 (3.9) | 0.043c | 112 (8.5) | 9 (4.1) | 0.025c |
| Masters/doctoral degree, no. (%) | 382 (29.0) | 88 (40.4) | 0.001c | 368 (29.6) | 86 (41.5) | 0.001c | 387 (29.4) | 88 (40.2) | 0.001c |
| Working status | |||||||||
| Retired, no. (%) | 128 (9.8) | 11 (5.1) | 0.024c | 108 (8.8) | 9 (4.4) | 0.033c | 129 (9.9) | 11 (5.0) | 0.022c |
| Part time employed, no. (%) | 175 (13.4) | 41 (18.9) | 0.033c | 164 (13.3) | 40 (19.4) | 0.020c | 174 (13.3) | 41 (18.8) | 0.032c |
| Household income per year | |||||||||
| $25,000–49,999, no. (%) | 147 (11.6) | 14 (6.5) | 0.028c | 133 (11.0) | 12 (5.9) | 0.025c | 146 (11.5) | 14 (6.5) | 0.029c |
| >$100,000, no. (%) | 611 (48.1) | 131 (61.2) | 0.0004c | 601 (49.9) | 128 (62.7) | 0.001c | 617 (48.6) | 132 (61.4) | 0.001c |
| Outcome—patient-reported well-being at 2 year follow-up compared with baselined | |||||||||
| Stable, no. (%) | 199 (15.1) | 39 (17.9) | 0.287c | 175 (14.0) | 40 (19.3) | 0.047c | 265 (20.1) | 51 (23.3) | 0.278c |
Table contains only significant comparison results between development and validation sets (see eTable 1 in Supplement 2 for more details). p-Values < 0.05 highlighted in bold.
ap-Values refer to differences in the development and validation set.
bp-Values refer to t-tests to evaluate mean differences of continuous data.
cp-Values refer to Chi-square tests for binary feature evaluation (feature true versus feature not true).
d Variable included in the predictive models.
When comparing development and validation datasets, we observed significant differences in body mass index (BMI), baseline physical well-being, baseline physical well-being abdomen, superficial inferior epigastric artery (SIEA) flap, axillary lymph node dissection (ALND), sentinel lymph node biopsy (SLNB), high school degree, masters/doctoral degree, retired working status, part-time employed working status, $25,000–49,999 household income per year, and greater than $100,000 household income per year (all p < 0.05).
The correlation between 2 year psychosocial well-being and 2 year sexual well-being was highest (r = 0.72), followed by PRO scores at baseline (r = 0.63). The lowest level of correlation was observed between baseline physical well-being and 2 year sexual well-being (r = 0.14) (Table 2 in Supplement 1).
Algorithm Performance
Table 2 displays the performance of ML models with tuned optimal hyperparameters (Table 3 in Supplement 1) in both test and validation sets for each subscale. In the validation set, AUC to predict worsened physical well-being was 0.69 (95%CI, 0.62–0.76) for the LR with elastic net penalty, 0.69 (95%CI, 0.62–0.76) for the XGBoost tree, and 0.70 (95%CI, 0.63–0.77) for the neural network. When predicting worsened sexual well-being, AUC of the three algorithms was 0.76 (95%CI, 0.70–0.82), 0.77 (95% CI, 0.70–0.83), and 0.77(95% CI, 0.70–0.83), respectively. When predicting worsened psychosocial well-being, AUCs were 0.66 (95% CI, 0.58–0.73), 0.66 (95% CI, 0.58–0.74), and 0.64 (95%CI, 0.55–0.72), respectively.
Table 2.
Evaluation of algorithms trained to predict physical, sexual, psychosocial well-beings at 2 year follow-up
| 2 Year follow-up score lower than baseline | 2 Year follow-up score higher than baseline | |||
|---|---|---|---|---|
| Accuracy (95% CI) | AUC (95% CI) | Accuracy (95% CI) | AUC (95% CI) | |
| Physical well-being | ||||
| Logistic regression with elastic net penalty | ||||
| Test set (n = 1320) | 0.67(0.66–0.68) | 0.71(0.70–0.72) | 0.70(0.69–0.71) | 0.77(0.75–0.78) |
| Additional validation set (n = 218) | 0.63(0.56–0.69) | 0.69(0.62–0.76) | 0.76(0.69–0.81) | 0.82(0.76–0.87) |
| XGBoost tree | ||||
| Test set (n = 1320) | 0.66(0.64–0.67) | 0.70(0.69–0.72) | 0.70(0.69–0.71) | 0.77(0.76–0.78) |
| Additional validation set (n = 218) | 0.64(0.57–0.71) | 0.69(0.62–0.76) | 0.75(0.69–0.81) | 0.81(0.75–0.86) |
| Neural network | ||||
| Test set (n = 1320) | 0.65(0.64–0.67) | 0.70(0.68–0.71) | 0.69(0.69–0.71) | 0.76(0.75–0.77) |
| Additional validation set (n = 218) | 0.64 (0.58–0.71) | 0.70(0.63–0.77) | 0.75(0.69–0.81) | 0.81(0.75–0.86) |
| Sexual well-being | ||||
| Logistic regression with elastic net penalty | ||||
| Test set (n = 1247) | 0.69(0.68–0.70) | 0.75(0.74–0.77) | 0.72(0.71–0.74) | 0.77(0.76–0.79) |
| Additional validation set (n = 207) | 0.69(0.62–0.75) | 0.76(0.70–0.82) | 0.69(0.62–0.75) | 0.76(0.69–0.82) |
| XGBoost tree | ||||
| Test set (n = 1247) | 0.69(0.68–0.70) | 0.75(0.74–0.76) | 0.72(0.70–0.73) | 0.76(0.74–0.77) |
| Additional validation set (n = 207) | 0.70(0.63–0.76) | 0.77(0.70–0.83) | 0.69(0.62–0.75) | 0.76(0.70–0.83) |
| Neural network | ||||
| Test set (n = 1247) | 0.67(0.66–0.69) | 0.75(0.73–0.76) | 0.71(0.69–0.72) | 0.77(0.76–0.79) |
| Additional validation set (n = 207) | 0.70(0.63–0.76) | 0.77(0.70–0.83) | 0.67(0.60–0.73) | 0.74(0.67–0.81) |
| Psychosocial well-being | ||||
| Logistic regression with elastic net penalty | ||||
| Test set (n = 1319) | 0.73(0.71–0.74) | 0.72(0.70–0.74) | 0.69(0.68–0.70) | 0.76(0.75–0.77) |
| Additional validation set (n = 219) | 0.71(0.65–0.77) | 0.66(0.58–0.73) | 0.60(0.53–0.66) | 0.66(0.59–0.73) |
| XGBoost tree | ||||
| Test set (n = 1319) | 0.70(0.69–0.71) | 0.68(0.66–0.70) | 0.70(0.69–0.72) | 0.77(0.76–0.79) |
| Additional validation set (n = 219) | 0.70(0.63–0.76) | 0.66(0.58–0.74) | 0.62(0.55–0.68) | 0.66(0.59–0.74) |
| Neural network | ||||
| Test set (n = 1319) | 0.71(0.70–0.73) | 0.72(0.70–0.73) | 0.70(0.69–0.71) | 0.76(0.75–0.78) |
| Additional validation set (n = 219) | 0.71(0.64–0.77) | 0.64(0.55–0.72) | 0.60(0.53–0.66) | 0.66(0.58–0.73) |
AUC Area-under-the-receiver-operating-characteristic-curve
All three models were more proficient at predicting improved rather than worsened physical well-being, with AUCs of 0.82 (95% CI, 0.76–0.87), 0.81 (95% CI, 0.75–0.86), and 0.81 (95%CI, 0.75–0.86), respectively. In predicting improved sexual well-being, the AUC was 0.76, (95% CI, 0.69–0.82), 0.76 (95% CI, 0.70–0.83), and 0.74 (95% CI, 0.67–0.81), respectively. For improved psychosocial well-being, AUCs were 0.66 (95% CI, 0.59–0.73), 0.66 (95% CI, 0.59–0.74), and 0.66 (95% CI, 0.58–0.73).
Figure 2 indicates that performance differences among the algorithms were not statistically significant (p > 0.05) except for the models predicting improved compared with worsened physical well-being (p < 0.05).
Fig. 2.
Performance comparison between machine learning (ml) models to predict improved and worsened physical, sexual, psychosocial well-beings with reconstructed breasts at 2 year follow-up. A ML models to predict physical well-being change. B ML models to predict sexual well-being change. C ML models to predict psychosocial well-being change
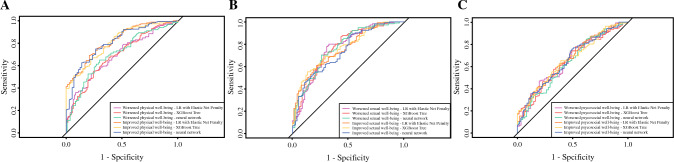
An array of AUC curves of the models for each scale are displayed in Fig. 3. Calibration plots of all the models for each scale are presented in Figs. 1–3 of Supplement 3. Spiegelhalter’s Z test results (Table 4 Supplement 1) indicate that most of the models were well calibrated except for neural network in predicting worsened physical well-being (p = 0.01) and improved psychosocial well-being (p = 0.02), LR with elastic net penalty in predicting worsened sexual well-being (p = 0.003), and XGBoost tree in predicting worsened psychosocial well-being (p = 0.01) and improved psychosocial well-being (p = 0.0001).
Fig. 3.
Receiver operating characteristic curves of machine learning models to predict improved and worsened physical, sexual, psychosocial well-beings with reconstructed breasts at 2 year follow-up. A Physical well-being with reconstructed breasts. B Sexual well-being with reconstructed breasts. C Psychosocial well-being with reconstructed breasts
Predictive Coefficients and Variable Importance
The results (Table 3) indicate that preoperative physical well-being (βregularized, −1.08) was most strongly negatively correlated with improved physical well-being at 2 year follow-up.
Table 3.
Regularized coefficients from the logistic regression with elastic net penalty
| Physical well-being (chest and upper body) | Sexual well-being | Psychosocial well-being | ||||
|---|---|---|---|---|---|---|
| Regularized coefficient for worsened physical well-being at 2 year follow-up | Regularized coefficient for improved physical well-being at 2 year follow-up | Regularized coefficient for worsened sexual well-being at 2 year follow-up | Regularized coefficient for improved sexual well-being at 2 year follow-up | Regularized coefficient for worsened psychosocial well-being at 2 year follow-up | Regularized coefficient for improved psychosocial well-being at 2 year follow-up | |
| Patient variables | ||||||
| Age | − 0.03 | − 0.06 | 0.15 | − 0.18 | 0.06 | |
| BMI | 0.01 | − 0.10 | − 0.01 | 0.09 | ||
| Diabetes | ||||||
| Yes | − 0.14 | − 0.38 | 0.23 | |||
| No | ||||||
| Smoker | ||||||
| Never | ||||||
| Previous | − 0.19 | − 0.05 | 0.24 | − 0.03 | ||
| Current | − 0.14 | − 0.12 | 0.07 | − 0.03 | ||
| Patient-reported outcomes at baseline | ||||||
| Satisfaction with breasts | 0.02 | 0.15 | − 0.23 | 0.10 | − 0.16 | |
| Psychosocial well-being | 0.04 | 0.03 | 0.02 | 0.77 | − 0.40 | |
| Physical well-being chest and upper body | 0.43 | − 1.08 | 0.02 | − 0.05 | − 0.01 | |
| Physical well-being abdomen | 0.17 | − 0.10 | ||||
| Sexual well-being | − 0.07 | 0.32 | − 0.78 | − 0.08 | − 0.08 | |
| Clinical variables | ||||||
| Radiation | ||||||
| After reconstruction | 0.40 | − 0.47 | 0.04 | − 0.44 | 0.24 | − 0.04 |
| Before reconstruction | 0.23 | − 0.20 | 0.14 | − 0.07 | 0.09 | |
| None | 0.17 | − 0.11 | ||||
| Mastectomy | ||||||
| Nipple sparing | − 0.02 | 0.14 | − 0.25 | |||
| Simple | ||||||
| Other | 0.65 | − 0.65 | ||||
| Reconstruction | ||||||
| Tissue expander (TE) | − 0.06 | 0.27 | − 0.51 | 0.43 | − 0.10 | |
| Direct-to-implant (DTI) | 0.07 | − 0.29 | 0.00 | |||
| Transverse rectus abdominis (TRAM) flap | 0.03 | − 0.06 | 0.09 | − 0.49 | 0.06 | |
| Deep inferior epigastric perforator (DIEP) flap | − 0.07 | 0.19 | − 0.11 | 0.23 | − 0.07 | 0.07 |
| Latissimus dorsi (LD) flap | 0.06 | 0.48 | − 0.63 | 0.24 | ||
| Gluteal artery perforator (GAP) flap | − 0.52 | − 0.80 | ||||
| Superficial inferior epigastric artery (SIEA) flap | − 0.15 | − 1.04 | 0.10 | |||
| Crossover flap | − 0.39 | − 0.17 | 0.42 | − 0.10 | ||
| Mixed flaps | − 0.24 | − 0.13 | 0.74 | − 0.06 | ||
| Mixed implants and autologous | 0.36 | − 0.88 | 0.12 | |||
| Chemotherapy | ||||||
| Received | 0.06 | 0.15 | − 0.21 | 0.26 | − 0.19 | |
| Not received | ||||||
| Laterality | ||||||
| Unilateral reconstruction | ||||||
| Bilateral reconstruction | − 0.01 | 0.07 | ||||
| Mastectomy indication | ||||||
| Therapeutic | ||||||
| Prophylactic | 0.17 | − 0.36 | ||||
| Axillary intervention | ||||||
| Axillary lymph node dissection (ALND) | 0.03 | − 0.17 | 0.14 | − 0.03 | − 0.03 | − 0.07 |
| Sentinel lymph node biopsy (SLNB) | 0.18 | 0.11 | ||||
| None | − 0.04 | − 0.11 | 0.05 | − 0.08 | 0.04 | |
Positive values indicate a positive correlation with the corresponding well-being, while negative values indicate a negative correlation with the corresponding well-being.
In predicting sexual well-being change at 2 year follow-up, baseline sexual well-being (βregularized, − 0.78), tissue expander (TE) reconstruction (βregularized, − 0.51), and radiation after reconstruction (βregularized, − 0.44), were most strongly negatively correlated with improved changes, whereas latissimus dorsi (LD) flap reconstruction (βregularized, 0.48) was positively correlated.
Additionally, superficial inferior epigastric artery (SIEA) flap reconstruction(βregularized, − 1.04), mixed implants and autologous reconstruction (βregularized, −0.88), GAP flap (βregularized, − 0.80), other types of mastectomy (βregularized, − 0.65), and latissimus dorsi (LD) flap reconstruction (βregularized, − 0.63) were most strongly negatively correlated with worsened psychosocial well-being at 2 year follow-up, whereas baseline psychosocial well-being (βregularized, 0.77) and mixed flaps reconstruction (βregularized, 0.74) were positively correlated with worsened outcomes. Similar variable importance, as well as its contribution to prediction, were also observed from XGBoost SHAP values and neural network LIME plots (Figs. 4 and 5 in Supplement 3), respectively.
The results of binary logistic regression identified key predictors and revealed their statistical significance in predicting changes in health-related QOL after surgery at 2 year follow-up (Table 5 in Supplement 1). When comparing the logistic regression with the coefficients of the ML models, generally the same direction and magnitude of associations could be observed with few exceptions. This gives credibility into the outcome predictions made by the ML model.
Racial Bias Evaluation
The performance of all ML models in predicting both improved and worsened physical well-being statistically differed between the Caucasian and Asian groups (p < 0.05, higher scores for Asian subgroup). Neural networks performed statistically different between groups of Caucasian versus African American (p < 0.05, higher scores for African American subgroup), and African American versus Asian in predicting worsened sexual well-being (p < 0.05, higher scores for African American subgroup). All trained ML models showed statistically better performance for the African American group compared with the Caucasian group in both improved and worsened psychosocial well-being prediction (all p < 0.05) (Tables 6 and 7 in Supplement 1).
Discussion
In this study, we developed and validated three ML algorithms to predict clinically meaningful, long-term changes in health-related QOL for women undergoing PMBR with acceptable accuracy. Our results indicate that baseline PRO data of physical, sexual, and psychosocial well-being had a much greater impact on long-term reported changes in QOL than clinical variables, revealing key predictors to consider when discussing expected QOL for patients undergoing cancer-related mastectomy.
We excluded study site as a variable in the machine learning model training for several reasons. First, neither the original study nor the present analysis considered the surgeon or study site as independent variables. Procedures were performed by 57 surgeons at 11 institutions, effectively balancing their influence despite potential skill variations. Second, adding study site as a variable would create a nonscalable algorithm that optimizes around potentially changing site characteristics. Therefore, excluding study site allows for better generalizability and scalability of the algorithm.
Compared with the sexual and psychosocial well-being of patients at 2 year follow-up, our findings show physical health improved in some patients but worsened in many more patients. This confirms previous findings that the physical well-being of the chest and upper body will not be fully restored, regardless of whether patients undergo implant-based or autologous reconstruction.1 The significant difference in performance between the prediction models for worsened and improved physical well-being suggests that the machine learning models encountered difficulties in accurately predicting worsened well-being. This could be attributed to (so far) unpredictable complications, such as infection, implant-related issues, or poor wound healing, which can arise during the postoperative period and result in suboptimal physical outcomes.
Previous studies using traditional statistical methods asserted that patients with autologous reconstruction tended to have a higher health-related QOL compared to those with implant-based reconstruction.1,30 Specifically, autologous reconstruction outperforms implants in tolerance of radiotherapy and improving QOL.31 Integrating radiotherapy with breast reconstruction results in a complex impact across multiple dimensions of a patients’ life.31 Patients with nipple-sparing mastectomy have significantly higher psychosocial and sexual well-being compared with patients with total mastectomy.32 However, inferences drawn from these group-level studies cannot infer specific treatment outcomes for individuals,33 as the relationships between variables of interest and outcomes are usually estimated after controlling for relevant co-variables, which does not reflect the real situation of each patient. Machine learning may overcome this limitation and help tailoring outcome predictions to the individual patient.13
Our results also indicate that patients with higher baseline physical, sexual, and psychosocial well-being were more likely to have worsened PROs in these three domains after breast reconstruction. Whether implant-based or autologous procedures were associated with improved or worsened QOL depended on the specific type of reconstruction, which was also seen in the binary logistic regression: taking direct-to-implant (DTI) reconstruction as a reference, TE reconstruction was associated with worsened sexual well-being and deep inferior epigastric perforator (DIEP) flap reconstruction was associated with a decreased risk of worsened physical well-being (chest and upper body). These individual-level outcomes predicted by ML models aim to better guide and optimize patient decision-making process to achieve expected postoperative outcomes when determining preferences for the exact reconstruction procedure. Nevertheless, a comparison of the performance of traditional statistical models with ML algorithms in this field appears highly warranted to help more clearly distinguish and highlight advantages of these developed intelligent decision-making tools.
Our team has previously published the development and validation of accurate ML algorithms to predict clinically meaningful changes in breast satisfaction with reconstructed breasts at 2 year follow-up in this cohort.14 AUC of the same three ML models to predict changes in satisfaction with reconstructed breasts study (improved: AUC range 0.86–0.87; decreased: AUC range 0.84–0.85) was higher compared with the performance in predicting changes in physical, sexual, and psychosocial well-being in the present analysis. Insights into predictors also underscored the importance of baseline PRO variables over clinical variables, similar to what we observed in the present analysis. Moreover, age was associated with worsened breast satisfaction (decreased: βregularized, 0.01) and physical well-being (improved: βregularized, − 0.03) but improved sexual well-being (improved: βregularized, 0.15) and psychosocial well-being (improved: βregularized, 0.06). This observation is underpinned by previous studies concluding that not all women necessarily experience worsening sexual function with higher age34 and that some older women have higher levels of sexual satisfaction,35 where psychosocial factors appear to play a crucial role.36
This study comes with several limitations. First, general guidelines for multivariate models recommend having at least 100 events for validation.37 However, none of the 11 study sites in this study met this requirement for all three scales simultaneously. Future, prospective validation with larger sample sizes seems warranted. Second, due to small samples or no samples in some racial groups (e.g., Hispanics), the ML performance assessment in these racial groups was not feasible. Some ML algorithms performed statistically significantly better in certain racial groups. We acknowledge that achieving equal performance across races becomes more challenging when sample sizes are limited, which aligns with findings of previous studies in this regard.14 Future studies may validate our findings and mitigate potential racial bias in a more diverse setting, not just academic institutions included here only. Third, although we achieved similar completion rates to similar PRO studies in literature,1 one cannot ignore that around half of the initially enrolled patients were lost to follow-up after 2 years. The results (Table 1 in Supplement 4) indicate that participants who were lost to follow-up were more likely to be younger, single, have a higher BMI, undergo TE reconstruction technique, and less likely to be married or have undergone DIEP reconstruction techniques. Future studies may consider using advanced PRO assessment approaches such as computer adaptive testing to reduce patients’ assessment burden and improve their engagement.38,39 Fourth, this study may establish a benchmark for interested researchers studying ML models on classification tasks for breast cancer patients. However, before implementing these algorithms in clinical practice to predict individual outcomes, necessary steps such as prospective clinical trials are needed to confirm their validity and reliability in real clinical settings.33 Fifth, the ML approach used in this study required dichotomization of outcomes. We chose this approach due to the recent upcoming concept of “clinically-important differences” in PROM research.40,41 However, some clinicians and patients might be more concerned about the degree or magnitude of change in their well-being due to the reconstruction procedure. This issue might be addressed in future research via modeling techniques that allow for continuous outcomes (e.g., linear modeling). Sixth, clinical implementation of digital health tools has proved to be a challenging task with many barriers including, for example, lack of transdisciplinary knowledge.42 Future research seems warranted to investigate the development and clinical feasibility of a digital tool that assists patients and clinicians in clinical decision-making process.
Conclusions
In this study, we developed and validated ML algorithms to predict clinically meaningful, long-term changes in physical, sexual, and psychosocial well-being for women undergoing cancer-related mastectomy and breast reconstruction at 2 year follow-up with acceptable accuracy. These algorithms may function as a data-driven decision-making tool to assist in making informed treatment decisions for women undergoing breast reconstruction, and further facilitate patient-centered care by tailoring individualized treatment in clinical practice.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgment
We would like to thank Dr. Edwin G. Wilkins of the Department of Surgery at the University of Michigan for all his assistance in the data collection process and for his valuable feedback on this improved manuscript. This manuscript has been authored in part by UT-Battelle, LLC, under contract DE-AC05-00OR22725 with the US Department of Energy (DOE). The US government retains and the publisher, by accepting the article for publication, acknowledges that the US government retains a nonexclusive, paid-up, irrevocable, worldwide license to publish or reproduce the published form of this manuscript, or allow others to do so, for US government purposes. DOE will provide public access to these results of federally sponsored research in accordance with the DOE Public Access Plan (http://energy.gov/downloads/doe-public-access-plan).
Author Contributors
Conception and design: CX, AP, BJM, PY, JAN, ALP, CSG. Collection and assembly of data: AP, ALP, CSG. Data analysis and interpretation: CX, AP, CSG. Manuscript writing: All authors. Final approval of manuscript: All authors. Accountable for all aspects of the work: All authors.
Funding
Supported by National Cancer Institute grant no. R01 CA152192 and in part by National Cancer Institute Support grant no. P30 CA008748.
Data Availability
Individual participant data that underlie the results reported in this article, after deidentification (text, tables, figures, and appendices) be available (including data dictionaries) on reasonable request from the corresponding author.
Disclosure
Cai Xu: No relationship to disclose. André Pfob: No relationship to disclose. Babak J. Mehrara: No relationship to disclose. Peimeng Yin: No relationship to disclose. Jonas A. Nelson: No relationship to disclose. Andrea L. Pusic: Patents, royalties, other intellectual property: I am codeveloper of BREAST-Q and receive royalty payments when it is used in for-profit industry-sponsored trials. Chris Sidey-Gibbons: No relationship to disclose.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Cai Xu and André Pfob are Joint first authors.
References
- 1.Pusic AL, Matros E, Fine N, et al. Patient-reported outcomes 1 year after immediate breast reconstruction: results of the mastectomy reconstruction outcomes consortium study. J Clin Oncol. 2017;35(22):2499–2506. doi: 10.1200/JCO.2016.69.9561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bhat S, Orucevic A, Woody C, Heidel RE, Bell JL. Evolving trends and influencing factors in mastectomy decisions. Am Surg. 2017;83(3):233–238. doi: 10.1177/000313481708300317. [DOI] [PubMed] [Google Scholar]
- 3.Metcalfe KA, Zhong T, Narod SA, et al. A prospective study of mastectomy patients with and without delayed breast reconstruction: long-term psychosocial functioning in the breast cancer survivorship period. J Surg Oncol. 2015;111(3):258–264. doi: 10.1002/jso.23829. [DOI] [PubMed] [Google Scholar]
- 4.Lee CN, Belkora J, Chang Y, Moy B, Partridge A, Sepucha K. Are patients making high-quality decisions about breast reconstruction after mastectomy? Plast Reconstr Surg. 2011;127(1):18–26. doi: 10.1097/PRS.0b013e3181f958de. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee CNH, Deal AM, Huh R, et al. Quality of patient decisions about breast reconstruction after mastectomy. JAMA Surg. 2017;152(8):741–748. doi: 10.1001/jamasurg.2017.0977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tanos G, Prousskaia E, Chow W, et al. Locally advanced breast cancer: autologous versus implant-based reconstruction. Plast Reconstr Surg–Glob Open. 2016 doi: 10.1097/GOX.0000000000000598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Voineskos SH, Frank SG, Cordeiro PG. Breast reconstruction following conservative mastectomies: predictors of complications and outcomes. Gland Surg. 2015;4(6):484–496. doi: 10.3978/j.issn.2227-684X.2015.04.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sidey-Gibbons JAM, Sidey-Gibbons CJ. Machine learning in medicine: a practical introduction. BMC Med Res Methodol. 2019;19(1):1–18. doi: 10.1186/s12874-019-0681-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Xu C, Subbiah IM, Lu SC, Pfob A, Sidey-Gibbons C. Machine learning models for 180-day mortality prediction of patients with advanced cancer using patient-reported symptom data. Qual Life Res. 2023;32(3):713–727. doi: 10.1007/s11136-022-03284-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gibbons C, Richards S, Valderas JM, Campbell J. Supervised machine learning algorithms can classify open-text feedback of doctor performance with human-level accuracy. J Med Internet Res. 2017;19(3):e6533. doi: 10.2196/jmir.6533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sidey-Gibbons C, Pfob A, Asaad M, et al. Development of machine learning algorithms for the prediction of financial toxicity in localized breast cancer following surgical treatment. JCO Clin Cancer Informatics. 2021;5:338–347. doi: 10.1200/cci.20.00088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kalaiyarasi M, Dhanasekar R, Sakthiya Ram S, Vaishnavi P. Classification of benign or malignant tumor using machine learning. IOP Conf Ser Mater Sci Eng. 2020 doi: 10.1088/1757-899X/995/1/012028. [DOI] [Google Scholar]
- 13.Lu SC, Xu C, Nguyen CH, Geng Y, Pfob A, Sidey-Gibbons C. Machine learning-based short-term mortality prediction models for patients with cancer using electronic health record data: systematic review and critical appraisal. JMIR Med Inform. 2022;10(3):e33182. doi: 10.2196/33182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pfob A, Mehrara BJ, Nelson JA, Wilkins EG, Pusic AL, Sidey-Gibbons C. Machine learning to predict individual patient-reported outcomes at 2-year follow-up for women undergoing cancer-related mastectomy and breast reconstruction (INSPiRED-001) Breast. 2021;60(520):111–122. doi: 10.1016/j.breast.2021.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pfob A, Mehrara BJ, Nelson JA, Wilkins EG, Pusic AL, Sidey-Gibbons C. Towards patient-centered decision-making in breast cancer surgery. Ann Surg. 2021 doi: 10.1097/sla.0000000000004862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pusic AL, Klassen AF, Scott AM, Klok JA, Cordeiro PG, Cano SJ. Development of a new patient-reported outcome measure for breast surgery: the BREAST-Q. Plast Reconstr Surg. 2009;124(2):345–353. doi: 10.1097/PRS.0b013e3181aee807. [DOI] [PubMed] [Google Scholar]
- 17.Cano SJ, Klassen AF, Scott AM, Cordeiro PG, Pusic AL. The BREAST-Q: further validation in independent clinical samples. Plast Reconstr Surg. 2012;129(2):293–302. doi: 10.1097/PRS.0b013e31823aec6b. [DOI] [PubMed] [Google Scholar]
- 18.Voineskos SH, Klassen AF, Cano SJ, Pusic AL, Gibbons CJ. Giving meaning to differences in BREAST-Q scores: minimal important difference for breast reconstruction patients. Plast Reconstr Surg. 2020;145(1):11e–20e. doi: 10.1097/PRS.0000000000006317. [DOI] [PubMed] [Google Scholar]
- 19.Pfob A, Sidey-Gibbons C, Rauch G, et al. Intelligent vacuum-assisted biopsy to identify breast cancer patients with pathologic complete response (ypT0 and ypN0) after neoadjuvant systemic treatment for omission of breast and axillary surgery. J Clin Oncol. 2022;40(17):1903–1915. doi: 10.1200/JCO.21.02439. [DOI] [PubMed] [Google Scholar]
- 20.Collins GS, Reitsma JB, Altman DG, Moons KGM. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement. Ann Intern Med. 2015;162(1):55–63. doi: 10.7326/M14-0697. [DOI] [PubMed] [Google Scholar]
- 21.Friedman JH. Greedy function approximation: a gradient boosting machine. Ann Stat. 2001;29(5):1189–1232. doi: 10.1214/aos/1013203451. [DOI] [Google Scholar]
- 22.Kuhn M, Wickham H, RStudio. Package “recipes.” 2022;(1):1–242. https://recipes.tidymodels.org/
- 23.Subbiah IM, Xu C, Lu S-C, et al. Development of patient reported outcomes-based machine learning algorithm for the six-month mortality prediction in patients with advanced cancer. J Clin Oncol. 2021;39(28_suppl):273–273. doi: 10.1200/jco.2020.39.28_suppl.273. [DOI] [Google Scholar]
- 24.Pfob A, Sidey-Gibbons C. Systematic bias in medical algorithms: to include or not include discriminatory demographic information? JCO Clin Cancer Informatics. 2022 doi: 10.1200/cci.21.00146. [DOI] [PubMed] [Google Scholar]
- 25.Lundberg SM, Lee SI. A unified approach to interpreting model predictions. In: Advances in Neural Information Processing Systems. Vol 2017-Decem. Neural information processing systems foundation; 2017:4766–4775. doi:10.48550/arxiv.1705.07874
- 26.Ribeiro MT, Singh S, Guestrin C. “Why should I trust you?” explaining the predictions of any classifier. In: NAACL-HLT 2016 - 2016 Conference of the North American Chapter of the Association for Computational Linguistics: Human Language Technologies, Proceedings of the Demonstrations Session. Association for Computational Linguistics (ACL); 2016:97–101. doi:10.18653/v1/n16-3020
- 27.Harrell FE, Lee KL, Mark DB. Multivariable prognostic models: issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat Med. 1996;15(4):361–387. doi: 10.1002/(SICI)1097-0258(19960229)15:4<361::AID-SIM168>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 28.Spiegelhalter D. Probabilistic prediction in patient management and clinical trials. Stat Med. 1986;5(5):421–433. doi: 10.1002/sim.4780050506. [DOI] [PubMed] [Google Scholar]
- 29.Wu YC, Lee WC. Alternative performance measures for prediction models. PLoS One. 2014;9(3):e91249. doi: 10.1371/journal.pone.0091249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nelson JA, Allen RJ, Polanco T, et al. Long-term patient-reported outcomes following postmastectomy breast reconstruction: an 8-year examination of 3268 patients. Ann Surg. 2019;270(3):473–483. doi: 10.1097/SLA.0000000000003467. [DOI] [PubMed] [Google Scholar]
- 31.Ho AY, Hu ZI, Mehrara BJ, Wilkins EG. Radiotherapy in the setting of breast reconstruction: types, techniques, and timing. Lancet Oncol. 2017;18(12):e742–e753. doi: 10.1016/S1470-2045(17)30617-4. [DOI] [PubMed] [Google Scholar]
- 32.Romanoff A, Zabor EC, Stempel M, Sacchini V, Pusic A, Morrow M. A comparison of patient-reported outcomes after nipple-sparing mastectomy and conventional mastectomy with reconstruction. Ann Surg Oncol. 2018;25(10):2909–2916. doi: 10.1245/s10434-018-6585-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pfob A, Sidey-Gibbons C, Heil J. Response prediction to neoadjuvant systemic treatment in breast cancer—yet another algorithm? JCO Clin Cancer Inform. 2021;5:654–655. doi: 10.1200/cci.21.00033. [DOI] [PubMed] [Google Scholar]
- 34.Lonnèe-Hoffmann RAM, Dennerstein L, Lehert P, Szoeke C. Sexual function in the late postmenopause: a decade of follow-up in a population-based cohort of Australian women. J Sex Med. 2014;11(8):2029–2038. doi: 10.1111/jsm.12590. [DOI] [PubMed] [Google Scholar]
- 35.Trompeter SE, Bettencourt R, Barrett-Connor E. Sexual activity and satisfaction in healthy community-dwelling older women. Am J Med. 2012;125(1):37–43. doi: 10.1016/j.amjmed.2011.07.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Thomas HN, Thurston RC. A biopsychosocial approach to women’s sexual function and dysfunction at midlife: a narrative review. Maturitas. 2016;87:49–60. doi: 10.1016/j.maturitas.2016.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Collins GS, Ogundimu EO, Cook JA, Le MY, Altman DG. Quantifying the impact of different approaches for handling continuous predictors on the performance of a prognostic model. Stat Med. 2016;35(23):4124–4135. doi: 10.1002/sim.6986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Xu C, Schaverien MV, Christensen JM, Sidey-Gibbons CJ. Efficient and precise Ultra-QuickDASH scale measuring lymphedema impact developed using computerized adaptive testing. Qual Life Res. 2022;31(3):917–925. doi: 10.1007/s11136-021-02979-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Xu C, Smith GL, Id Y-SC, et al. Short-form adaptive measure of financial toxicity from the Economic Strain and Resilience in Cancer (ENRICh) study: derivation using modern psychometric techniques. PLoS One. 2022;17(8):e0272804. doi: 10.1371/JOURNAL.PONE.0272804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Brozek JL, Guyatt GH, Schünemann HJ. How a well-grounded minimal important difference can enhance transparency of labelling claims and improve interpretation of a patient reported outcome measure. Health Qual Life Outcom. 2006;4(1):1–7. doi: 10.1186/1477-7525-4-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Revicki D, Hays RD, Cella D, Sloan J. Recommended methods for determining responsiveness and minimally important differences for patient-reported outcomes. J Clin Epidemiol. 2008;61(2):102–109. doi: 10.1016/j.jclinepi.2007.03.012. [DOI] [PubMed] [Google Scholar]
- 42.Pfob A, Sidey-Gibbons C, Schuessler M, et al. Contrast of digital and health literacy between IT and health care specialists highlights the importance of multidisciplinary teams for digital health—a pilot study. JCO Clin Cancer Inform. 2021;5:734–745. doi: 10.1200/cci.21.00032. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Individual participant data that underlie the results reported in this article, after deidentification (text, tables, figures, and appendices) be available (including data dictionaries) on reasonable request from the corresponding author.