Abstract
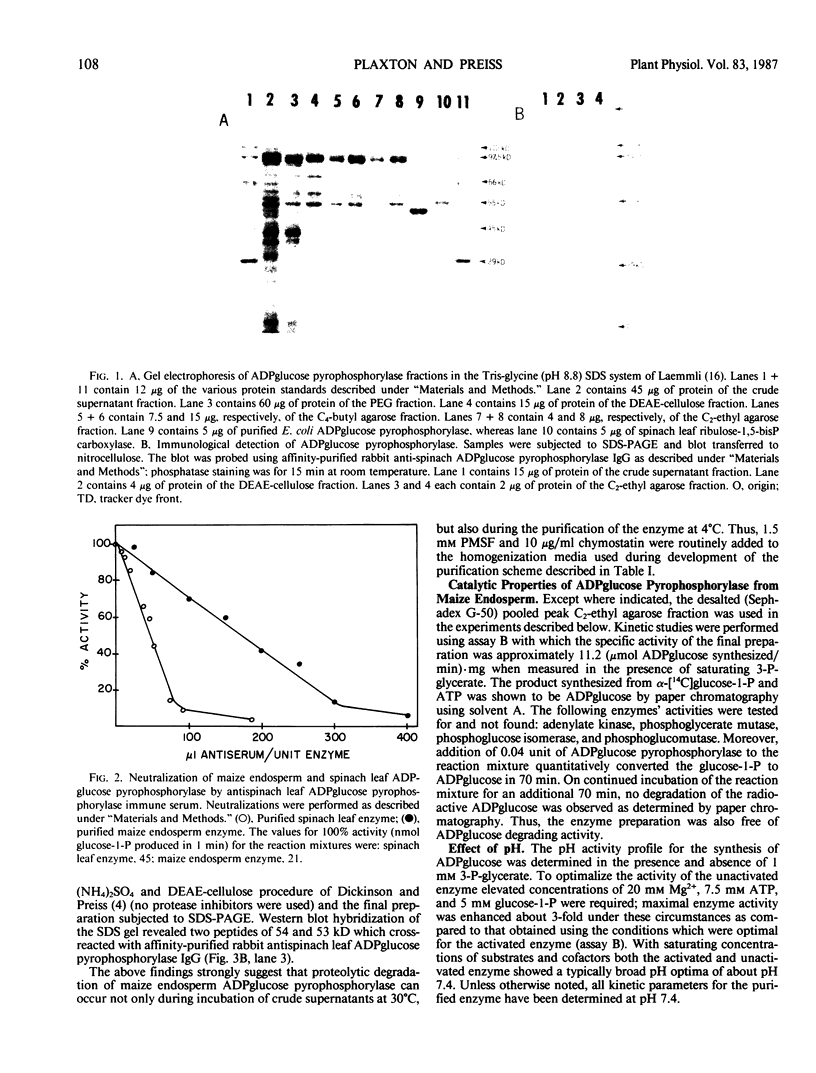
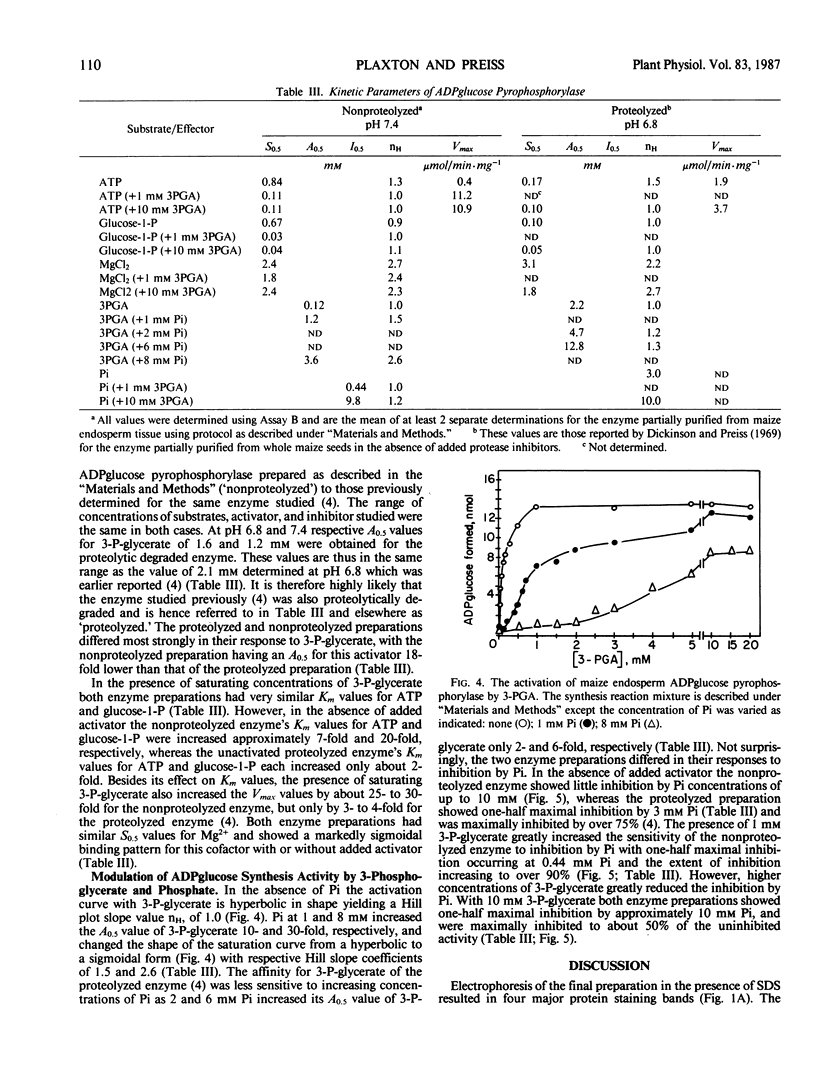
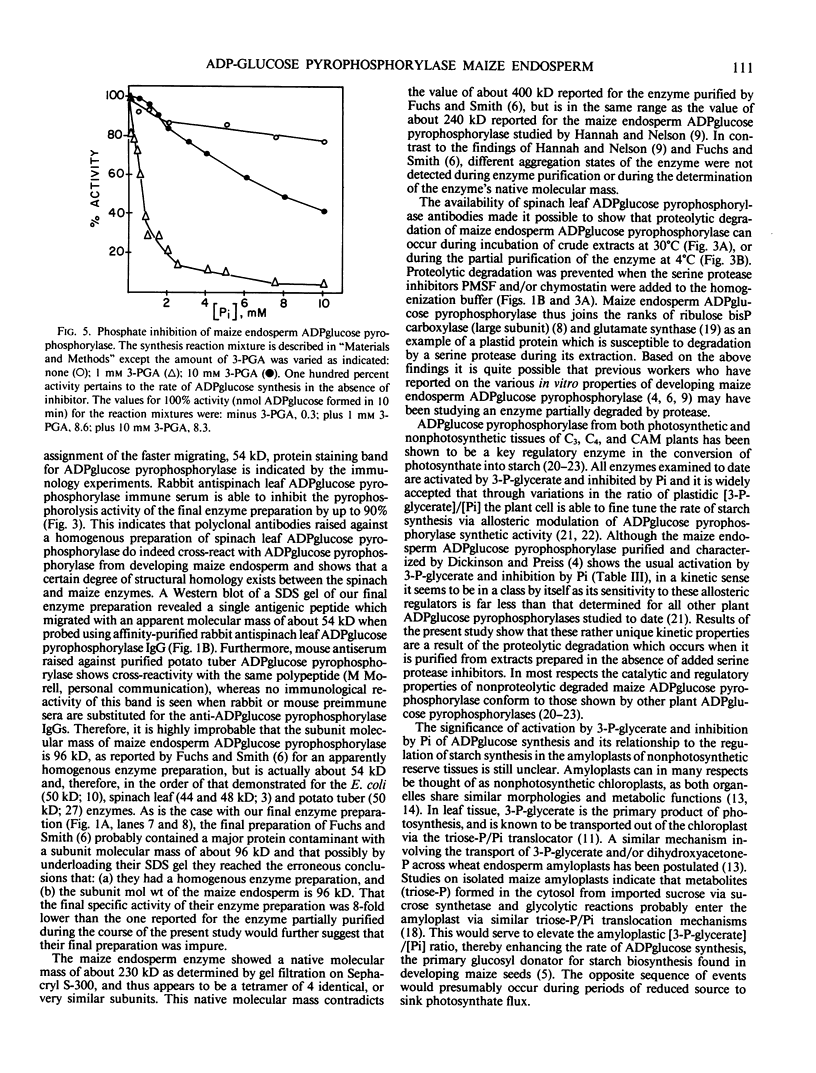
ADPglucose pyrophosphorylase from developing endosperm tissue of starchy maize (Zea mays) was purified 88-fold to a specific activity of 34 micromoles α-glucose-1-P produced per minute per milligram protein. Rabbit antiserum to purified spinach leaf ADPglucose pyrophosphorylase was able to inhibit pyrophosphorolysis activity of the purified enzyme by up to 90%. The final preparation yielded four major protein staining bands following sodium dodecyl sulfate polyacrylamide gel electrophoresis. When analyzed by Western blot hybridization only the fastest migrating, 54 kilodaltons, protein staining band cross-reacted with affinity purified rabbit antispinach leaf ADPglucose pyrophosphorylase immunoglobulin. The molecular mass of the native enzyme was estimated to be 230 kilodaltons. Thus, maize endosperm ADPglucose pyrophosphorylase appears to be comprised of four subunits. This is in contrast to the respective subunit and native molecular masses of 96 and 400 kilodaltons reported for a preparation of maize endosperm ADPglucose pyrophosphorylase (Fuchs RL and JO Smith 1979 Biochim Biophys Acta 556: 40-48). Proteolytic degradation of maize endosperm ADPglucose pyrophosphorylase appears to occur during incubation of crude extracts at 30°C or during the partial purification of the enzyme according to a previously reported procedure (DB Dickinson, J Preiss 1969 Arch Biochem Biophys 130: 119-128). The progressive appearance of a 53 kilodalton antigenic peptide suggested the loss of a 1 kilodalton proteolytic fragment from the 54 kilodalton subunit. The complete conservation of the 54 kilodalton subunit structure following extraction of the enzyme in the presence of phenylmethylsulfonyl fluoride and/or chymostain was observed. The allosteric and catalytic properties of the partially purified proteolytic degraded versus nondegraded enzyme were compared. The major effect of proteolysis was to enhance enzyme activity in the absence of added activator while greatly decreasing its sensitivity to the allosteric effectors 3-P-glycerate and inorganic phosphate.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Copeland L., Preiss J. Purification of Spinach Leaf ADPglucose Pyrophosphorylase. Plant Physiol. 1981 Nov;68(5):996–1001. doi: 10.1104/pp.68.5.996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickinson D. B., Preiss J. ADP glucose pyrophosphorylase from maize endosperm. Arch Biochem Biophys. 1969 Mar;130(1):119–128. doi: 10.1016/0003-9861(69)90017-4. [DOI] [PubMed] [Google Scholar]
- FRYDMAN R. B. STARCH SYNTHETASE OF POTATOES AND WAXY MAIZE. Arch Biochem Biophys. 1963 Aug;102:242–248. doi: 10.1016/0003-9861(63)90177-2. [DOI] [PubMed] [Google Scholar]
- Fuchs R. L., Smith J. D. The purification and characterization of ADP-glucose pyrophosphorylase A from developing maize seeds. Biochim Biophys Acta. 1979 Jan 12;566(1):40–48. doi: 10.1016/0005-2744(79)90246-8. [DOI] [PubMed] [Google Scholar]
- Ghosh H. P., Preiss J. Adenosine diphosphate glucose pyrophosphorylase. A regulatory enzyme in the biosynthesis of starch in spinach leaf chloroplasts. J Biol Chem. 1966 Oct 10;241(19):4491–4504. [PubMed] [Google Scholar]
- Gray J. C., Kerwick R. G. An immunological investigation of the structure and function of ribulose 1,5-bisphosphate carboxylase. Eur J Biochem. 1974 May 15;44(2):481–489. doi: 10.1111/j.1432-1033.1974.tb03506.x. [DOI] [PubMed] [Google Scholar]
- Haugen T. H., Ishaque A., Preiss J. Biosynthesis of bacterial glycogen. Characterization of the subunit structure of Escherichia coli B glucose-1-phosphate adenylyltransferase (EC 2.7.7.27). J Biol Chem. 1976 Dec 25;251(24):7880–7885. [PubMed] [Google Scholar]
- Heldt H. W., Rapley L. Specific transport of inorganic phosphate, 3-phosphoglycerate and dihydroxyacetonephosphate, and of dicarboxylates across the inner membrane of spinach chloroplasts. FEBS Lett. 1970 Oct 5;10(3):143–148. doi: 10.1016/0014-5793(70)80438-0. [DOI] [PubMed] [Google Scholar]
- Koshland D. E., Jr, Némethy G., Filmer D. Comparison of experimental binding data and theoretical models in proteins containing subunits. Biochemistry. 1966 Jan;5(1):365–385. doi: 10.1021/bi00865a047. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lehmann M., Preiss J. Biosynthesis of bacterial glycogen: purification and properties of Salmonella typhimurium LT-2 adenosine diphosphate glucose pyrophosphorylase. J Bacteriol. 1980 Jul;143(1):120–127. doi: 10.1128/jb.143.1.120-127.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu T. T., Shannon J. C. Measurement of Metabolites Associated with Nonaqueously Isolated Starch Granules from Immature Zea mays L. Endosperm. Plant Physiol. 1981 Mar;67(3):525–529. doi: 10.1104/pp.67.3.525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh B. K., Greenberg E., Preiss J. ADPglucose Pyrophosphorylase from the CAM Plants Hoya carnosa and Xerosicyos danguyi. Plant Physiol. 1984 Mar;74(3):711–716. doi: 10.1104/pp.74.3.711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith D. E., Fisher P. A. Identification, developmental regulation, and response to heat shock of two antigenically related forms of a major nuclear envelope protein in Drosophila embryos: application of an improved method for affinity purification of antibodies using polypeptides immobilized on nitrocellulose blots. J Cell Biol. 1984 Jul;99(1 Pt 1):20–28. doi: 10.1083/jcb.99.1.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith P. K., Krohn R. I., Hermanson G. T., Mallia A. K., Gartner F. H., Provenzano M. D., Fujimoto E. K., Goeke N. M., Olson B. J., Klenk D. C. Measurement of protein using bicinchoninic acid. Anal Biochem. 1985 Oct;150(1):76–85. doi: 10.1016/0003-2697(85)90442-7. [DOI] [PubMed] [Google Scholar]
- Sowokinos J. R., Preiss J. Pyrophosphorylases in Solanum tuberosum: III. PURIFICATION, PHYSICAL, AND CATALYTIC PROPERTIES OF ADPGLUCOSE PYROPHOSPHORYLASE IN POTATOES. Plant Physiol. 1982 Jun;69(6):1459–1466. doi: 10.1104/pp.69.6.1459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sowokinos J. R. Pyrophosphorylases in Solanum tuberosum: I. Changes in ADP-Glucose and UDP-Glucose Pyrophosphorylase Activities Associated with Starch Biosynthesis during Tuberization, Maturation, and Storage of Potatoes. Plant Physiol. 1976 Jan;57(1):63–68. doi: 10.1104/pp.57.1.63. [DOI] [PMC free article] [PubMed] [Google Scholar]