Abstract
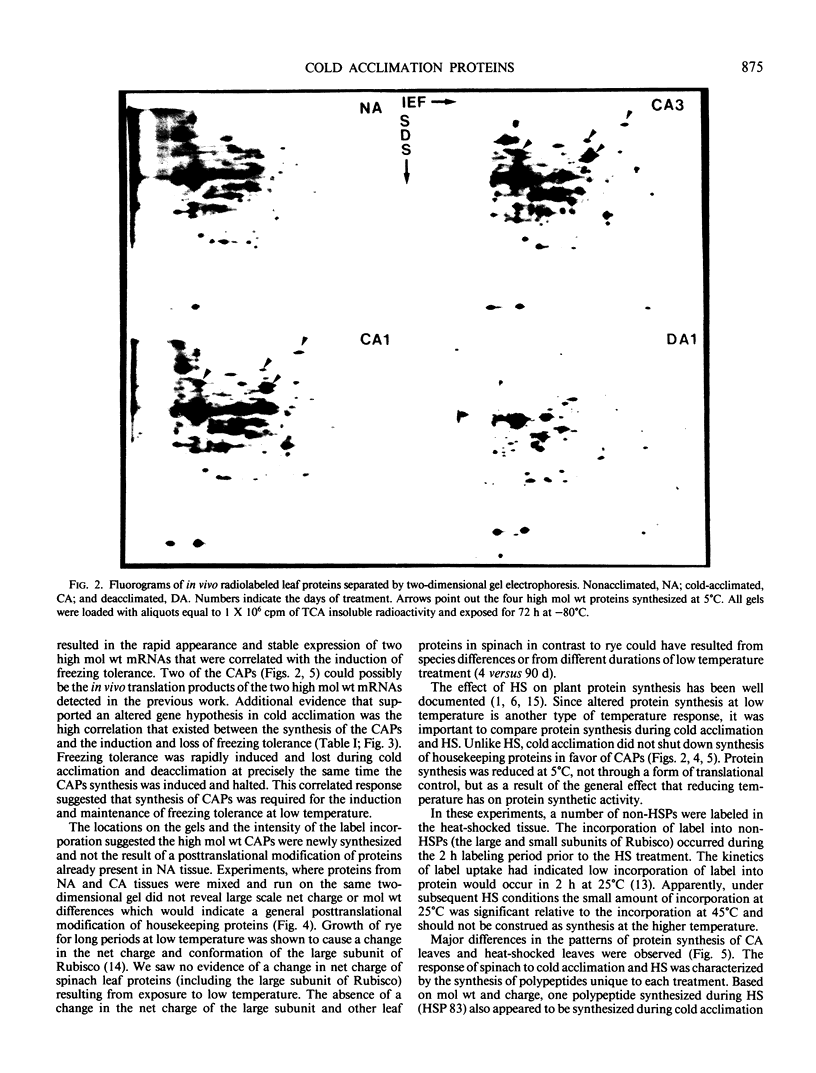
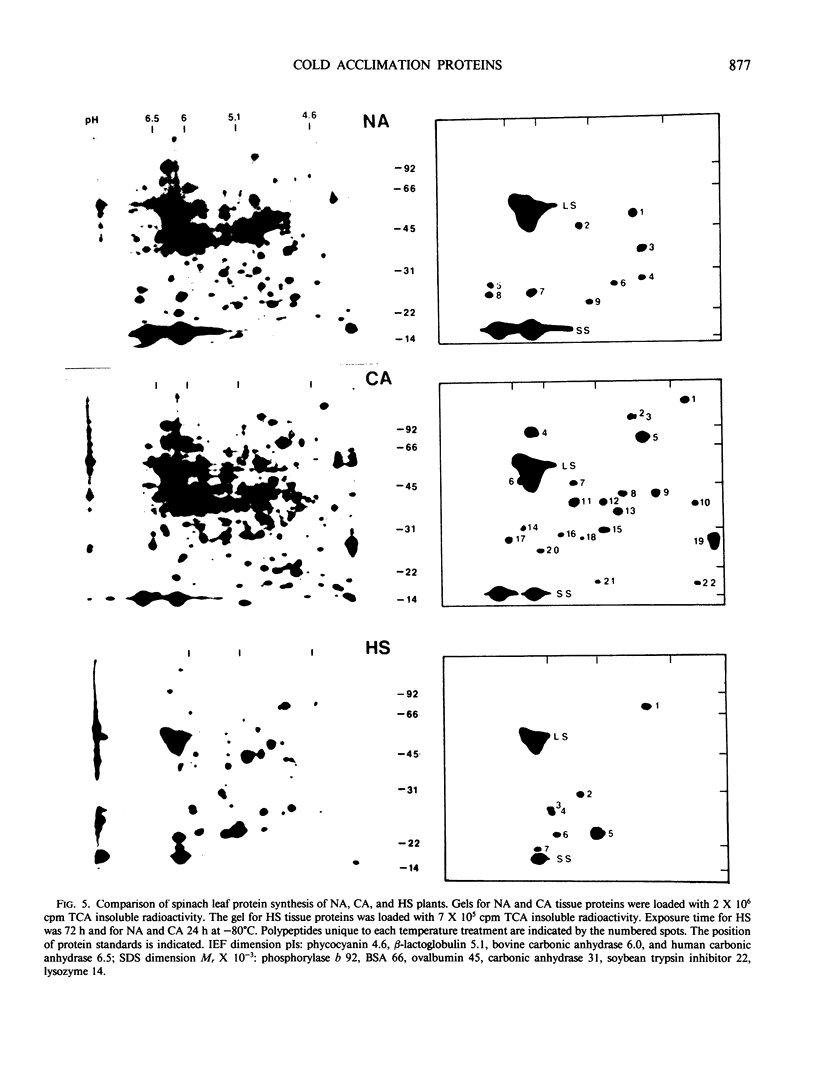
Spinach (Spinacia oleracea L. cv Bloomsdale) seedlings cultured in vitro were used to study changes in protein synthesis during cold acclimation. Seedlings grown for 3 weeks postsowing on an inorganic-nutrient-agar medium were able to increase their freezing tolerance when grown at 5°C. During cold acclimation at 5°C and deacclimation at 25°C, the kinetics of freezing tolerance induction and loss were similar to that of soil-grown plants. Freezing tolerance increased after 1 day of cold acclimation and reached a maximum within 7 days. Upon deacclimation at 25°C, freezing tolerance declined within 1 day and was largely lost by the 7th day. Leaf proteins of intact plants grown at 5 and 25°C were in vivo radiolabeled, without wounding or injury, to high specific activities with [35S]methionine. Leaf proteins were radiolabeled at 0, 1, 2, 3, 4, 7, and 14 days of cold acclimation and at 1, 3, and 7 days of deacclimation. Up to 500 labeled proteins were separated by two-dimensional gel electrophoresis and visualized by fluorography. A rapid and stable change in the protein synthesis pattern was observed when seedlings were transferred to the low temperature environment. Cold-acclimated leaves contained 22 polypeptides not found in nonacclimated leaves. Exposure to 5°C induced the synthesis of three high molecular weight cold acclimation proteins (CAPs) (Mr of about 160,000, 117,000, and 85,000) and greatly increased the synthesis of a fourth high molecular weight protein (Mr 79,000). These proteins were synthesized during day 1 and throughout the 14 day exposure to 5°C. During deacclimation, the synthesis of CAPs 160, 117, and 85 was greatly reduced by the first day of exposure to 25°C. However, CAP 79 was synthesized throughout the 7 day deacclimation treatment. Thus, the induction at low temperature and termination at warm temperature of the synthesis of CAPs 160, 117, and 85 was highly correlated with the induction and loss of freezing tolerance. Cold acclimation did not result in a general posttranslational modification of leaf proteins. Most of the observed changes in the two-dimensional gel patterns could be attributed to the de novo synthesis of proteins induced by low temperature. In spinach leaf tissue, heat shock altered the pattern of protein synthesis and induced the synthesis of several heat shock proteins (HSPs). One polypeptide synthesized in cold-acclimated leaves had a molecular weight and net charge (Mr 79,000, pI 4.8) similar to that of a HSP (Mr 83,000, pI 4.8). However, heat shock did not increase the freezing tolerance, and cold acclimation did not increase heat tolerance over that of nonacclimated plants, but heat-shocked leaf tissue was more tolerant to high temperatures than nonacclimated or cold-acclimated leaf tissue. When protein extracts from heat-shocked and cold-acclimated leaves were mixed and separated in the same two-dimensional gel, the CAP and HSP were shown to be two separate polypeptides with slightly different isoelectric points and molecular weights.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chen H. H., Li P. H., Brenner M. L. Involvement of abscisic Acid in potato cold acclimation. Plant Physiol. 1983 Feb;71(2):362–365. doi: 10.1104/pp.71.2.362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan R., Hershey J. W. Evaluation of isoelectric focusing running conditions during two-dimensional isoelectric focusing/sodium dodecyl sulfate-polyacrylamide gel electrophoresis: variation of gel patterns with changing conditions and optimized isoelectric focusing conditions. Anal Biochem. 1984 Apr;138(1):144–155. doi: 10.1016/0003-2697(84)90783-8. [DOI] [PubMed] [Google Scholar]
- Guy C. L., Hummel R. L., Haskell D. Induction of Freezing Tolerance in Spinach during Cold Acclimation. Plant Physiol. 1987 Jul;84(3):868–871. doi: 10.1104/pp.84.3.868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guy C. L., Niemi K. J., Brambl R. Altered gene expression during cold acclimation of spinach. Proc Natl Acad Sci U S A. 1985 Jun;82(11):3673–3677. doi: 10.1073/pnas.82.11.3673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guy C. L., Plesofsky-Vig N., Brambl R. Heat shock protects germinating conidiospores of Neurospora crassa against freezing injury. J Bacteriol. 1986 Jul;167(1):124–129. doi: 10.1128/jb.167.1.124-129.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huner N. P., Macdowall F. D. Changes in the net charge and subunit properties of ribulose bisphosphate carboxylase--oxygenase during cold hardening of Puma rye. Can J Biochem. 1979 Feb;57(2):155–164. doi: 10.1139/o79-019. [DOI] [PubMed] [Google Scholar]
- Key J. L., Lin C. Y., Chen Y. M. Heat shock proteins of higher plants. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3526–3530. doi: 10.1073/pnas.78.6.3526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krasnuk M., Jung G. A., Witham F. H. Electrophoretic studies of several dehydrogenases in relation to the cold tolerance of alfalfa. Cryobiology. 1976 Jun;13(3):375–393. doi: 10.1016/0011-2240(76)90121-8. [DOI] [PubMed] [Google Scholar]
- Meza-Basso L., Alberdi M., Raynal M., Ferrero-Cadinanos M. L., Delseny M. Changes in Protein Synthesis in Rapeseed (Brassica napus) Seedlings during a Low Temperature Treatment. Plant Physiol. 1986 Nov;82(3):733–738. doi: 10.1104/pp.82.3.733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Farrell P. H. High resolution two-dimensional electrophoresis of proteins. J Biol Chem. 1975 May 25;250(10):4007–4021. [PMC free article] [PubMed] [Google Scholar]
- Schuster A. M., Davies E. Ribonucleic Acid and protein metabolism in pea epicotyls : I. The aging process. Plant Physiol. 1983 Nov;73(3):809–816. doi: 10.1104/pp.73.3.809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Etten J. L., Freer S. N., McCune B. K. Presence of a major (storage?) protein in dormant spores of the fungus Botryodiplodia theobromae. J Bacteriol. 1979 May;138(2):650–652. doi: 10.1128/jb.138.2.650-652.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Volger H. G., Heber U. Cryoprotective leaf proteins. Biochim Biophys Acta. 1975 Dec 15;412(2):335–349. doi: 10.1016/0005-2795(75)90048-3. [DOI] [PubMed] [Google Scholar]
- Weiser C. J. Cold Resistance and Injury in Woody Plants: Knowledge of hardy plant adaptations to freezing stress may help us to reduce winter damage. Science. 1970 Sep 25;169(3952):1269–1278. doi: 10.1126/science.169.3952.1269. [DOI] [PubMed] [Google Scholar]
- Yoshida S., Uemura M. Protein and Lipid Compositions of Isolated Plasma Membranes from Orchard Grass (Dactylis glomerata L.) and Changes during Cold Acclimation. Plant Physiol. 1984 May;75(1):31–37. doi: 10.1104/pp.75.1.31. [DOI] [PMC free article] [PubMed] [Google Scholar]