Abstract
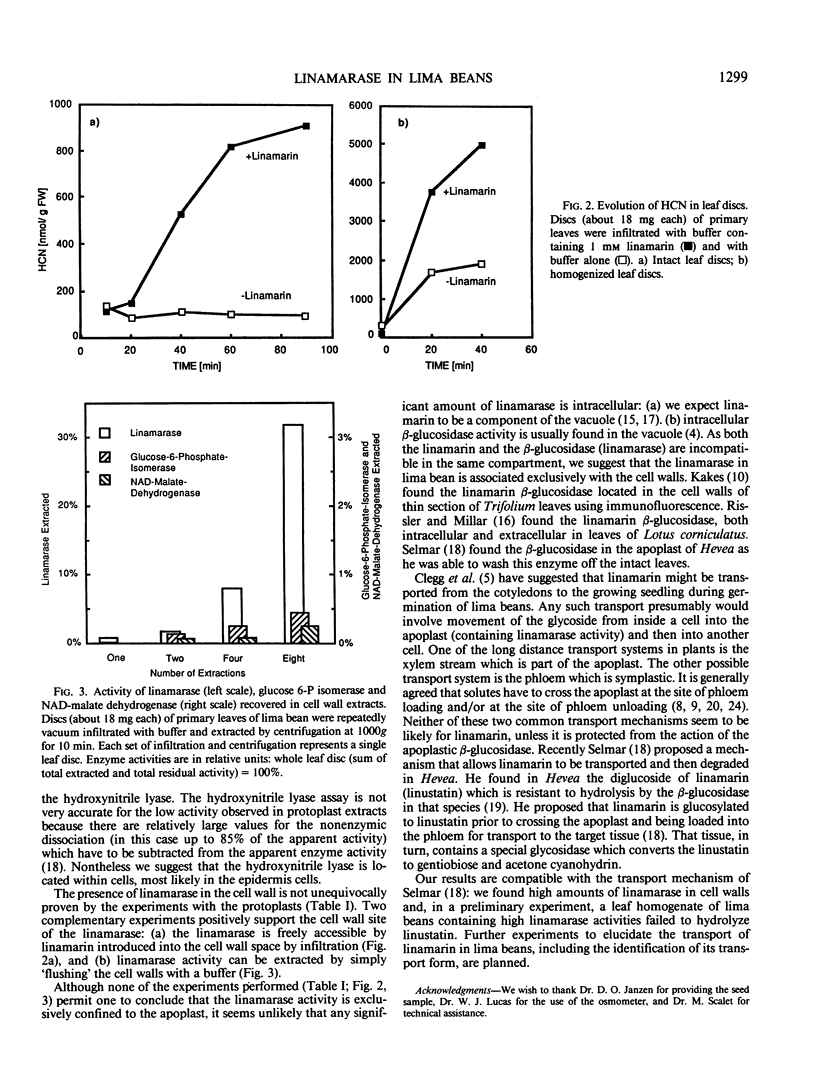
Analysis of mesophyll protoplasts and cell wall extracts of leaf discs of Costa Rican wild lima bean (Phaseolus lunatus L.) shows that the linamarase activity is confined to the apoplast. Its substrate linamarin, together with the related enzyme hydroxynitrile lyase, is found inside the cells. This compartmentation prevents cyanogenesis from occurring in intact tissue, and suggests that linamarin has to be protected during any translocation across the linamarase rich apoplast.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arnon D. I. COPPER ENZYMES IN ISOLATED CHLOROPLASTS. POLYPHENOLOXIDASE IN BETA VULGARIS. Plant Physiol. 1949 Jan;24(1):1–15. doi: 10.1104/pp.24.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beier H., Bruening G. The use of an abrasive in the isolation of cowpea leaf protoplasts which support the multiplication of cowpea mosaic virus. Virology. 1975 Mar;64(1):272–276. doi: 10.1016/0042-6822(75)90099-9. [DOI] [PubMed] [Google Scholar]
- Giaquinta R. T., Lin W., Sadler N. L., Franceschi V. R. Pathway of Phloem unloading of sucrose in corn roots. Plant Physiol. 1983 Jun;72(2):362–367. doi: 10.1104/pp.72.2.362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kojima M., Poulton J. E., Thayer S. S., Conn E. E. Tissue Distributions of Dhurrin and of Enzymes Involved in Its Metabolism in Leaves of Sorghum bicolor. Plant Physiol. 1979 Jun;63(6):1022–1028. doi: 10.1104/pp.63.6.1022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saunders J. A., Conn E. E. Presence of the cyanogenic glucoside dhurrin in isolated vacuoles from sorghum. Plant Physiol. 1978 Feb;61(2):154–157. doi: 10.1104/pp.61.2.154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selmar D., Lieberei R., Biehl B., Voigt J. Hevea Linamarase-A Nonspecific beta-Glycosidase. Plant Physiol. 1987 Mar;83(3):557–563. doi: 10.1104/pp.83.3.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Servaites J. C., Schrader L. E., Jung D. M. Energy-dependent Loading of Amino Acids and Sucrose into the Phloem of Soybean. Plant Physiol. 1979 Oct;64(4):546–550. doi: 10.1104/pp.64.4.546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terry M. E., Bonner B. A. An Examination of Centrifugation as a Method of Extracting an Extracellular Solution from Peas, and Its Use for the Study of Indoleacetic Acid-induced Growth. Plant Physiol. 1980 Aug;66(2):321–325. doi: 10.1104/pp.66.2.321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thayer S. S., Conn E. E. Subcellular Localization of Dhurrin beta-Glucosidase and Hydroxynitrile Lyase in the Mesophyll Cells of Sorghum Leaf Blades. Plant Physiol. 1981 Apr;67(4):617–622. doi: 10.1104/pp.67.4.617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolswinkel P., Ammerlaan A., Peters H. F. Phloem Unloading of Amino Acids at the Site of Attachment of Cuscuta europaea. Plant Physiol. 1984 May;75(1):13–20. doi: 10.1104/pp.75.1.13. [DOI] [PMC free article] [PubMed] [Google Scholar]


