Abstract
The diversity of pathology of severe paediatric asthma demonstrates that the one-size-fits-all approach characterising many guidelines is inappropriate. The term “asthma” is best used to describe a clinical syndrome of wheeze, chest tightness, breathlessness, and sometimes cough, making no assumptions about underlying pathology. Before personalising treatment, it is essential to make the diagnosis correctly and optimise basic management. Clinicians must determine exactly what type of asthma each child has. We are moving from describing symptom patterns in preschool wheeze to describing multiple underlying phenotypes with implications for targeting treatment. Many new treatment options are available for school age asthma, including biological medicines targeting type 2 inflammation, but a paucity of options are available for non-type 2 disease. The traditional reliever treatment, shortacting β2 agonists, is being replaced by combination inhalers containing inhaled corticosteroids and fast, longacting β2 agonists to treat the underlying inflammation in even mild asthma and reduce the risk of asthma attacks. However, much decision making is still based on adult data extrapolated to children. Better inclusion of children in future research studies is essential, if children are to benefit from these new advances in asthma treatment.
Keywords: asthma, pediatrics
Introduction
Paediatric asthma is a heterogenous disease, often but not invariably characterised by eosinophilic lower airway inflammation and reversible airway obstruction. Advances in the understanding of the underlying pathogenesis and the identification of clinical phenotypes and molecular endotypes has prompted a shift towards personalised treatment for children and young people with asthma. However, it is increasingly becoming apparent that personalised medicine is not being underpinned by correct diagnosis and high quality basic management. For example, the UK National Review of Asthma Deaths (NRAD)1 highlighted the urgent need to improve basic asthma care received by children and young people. Healthcare professionals caring for children and young people with asthma should be aware of how to use objective measurements to make the diagnosis, and the advances in understanding of the pathogenesis of paediatric asthma that are substantially changing the management of asthma. This review will use evidence from clinical studies in children and young people to outline a structured objective approach for diagnosing and managing asthma. This approach forms the basis of reviewing the efficacy and usefulness of new treatment options including biological medicines targeting type 2 inflammation, the change in the paradigm for reliever treatment, and the heightened awareness of the importance of asthma attacks and the concept of risk.
Sources and selection criteria
We reviewed articles on paediatric asthma published from 1 January 1992 to 31 February 2023. We searched the PubMed database for articles on paediatric asthma management, pathogenesis, and diagnosis. We used specific search terms (Asthma OR wheez* AND Child OR infant AND management OR treatment OR biologic), and limited the search to clinical trials, randomised controlled trials, meta-analyses, and systematic reviews. We also manually searched the reference lists of relevant articles for additional references. Manuscripts were reviewed by the authors and selected on the basis of relevance to the review. Only full manuscripts published in English in peer reviewed journals were included.
Epidemiology of severe asthma
One in 11 children in UK have asthma,2 which is the most common chronic disease during childhood. The vast majority of cases of asthma can be controlled with low to moderate doses of inhaled corticosteroids (ICS). About 3-5% of children have severe asthma (box 1).3
Box 1. Key definitions.
Type 2 inflammation:
Inflammation mediated by T helper 2 lymphocytes and type 2 innate lymphoid cells via the typical signature cytokines interleukin (IL) 4, IL5, and IL13, which drive eosinophilic airway inflammation.
Asthma:
Defined clinically as wheeze, chest tightness, breathlessness, and sometimes cough.6
Severe asthma:
Asthma that requires treatment with high dose inhaled corticosteroids plus a second controller or systemic corticosteroids to prevent it from becoming uncontrolled, or asthma that remains uncontrolled despite this treatment.63
Personalised treatment:
Treatment that is targeted and individualised to a patient.
Treatable traits:
A precision medicine strategy for patients with airway disease that is label-free and based on the identification of treatable traits in each patient.7 An approach based on treatable traits enables an individualised and personalised approach that is not disease specific and is not limited by the diagnosis. A treatable trait is a treatment target identified through the identification of phenotypes or examination of underlying causal pathways.
Paediatric asthma across the life course
Personalised treatment is impossible without first appreciating the developmental changes in the different types of asthma as the child matures. The use of a single word (ie, asthma) to describe many different airway diseases has hindered the perception that schoolchildren are not little adults, and toddlers and preschoolers are not small schoolchildren. Mixing pathology and clinical features in past definitions of asthma has led to wheeze being considered as an indication for ICS prescription without considering whether eosinophilic inflammation is actually present.4 However, we now appreciate that "asthma" is an umbrella term comprising many different phenotypes and endotypes,5 and so The Lancet Commission6 defined asthma in purely clinical terms (box 1) with no assumptions about the underlying pathology. Just as the diagnosis of arthritis should automatically lead to further exploration into determining the underlying type of arthritis, the same applies to asthma; the asthma label is the start and not the end of the diagnostic journey. The next step is to deconstruct and examine in detail the airway, in order to especially delineate treatable (and untreatable) traits at all ages as far as is possible (box 1 and table 1). The in-depth examination of the airway and identification of traits enables a better understanding of airway inflammation in asthma and identification of coexisting and alternative airway pathologies.7 The methods used will depend on the age and developmental maturity of the child.
Table 1.
Detailed examination of the airway in children with suspected asthma to identify coexisting and alternative airway pathologies and provide targeted treatment
| Airway component | Investigative method | Underlying cause | Treatment recommendations | Treatment outcomes |
| Fixed airflow obstruction | FEV1 <1.96 SD scores despite prednisolone and acute administration of SABA | Congenital/environmental (eg, cigarette smoke exposure); acquired remodelling | Not treatable, aim to prevent further deterioration, but do not overtreat | Deterioration in airway growth halted |
| Variable airflow obstruction | Variation in spirometry or peak flow by an arbitrary threshold (usually 15%) over time or with treatment | Bronchoconstriction; airway malacia118; intraluminal mucus/inflammatory debris | SABA and LABA; airway clearance, mucoactive agents | Bronchodilatation; reduction in airflow obstruction |
| Airway inflammation | Induced sputum; FeNO; peripheral blood eosinophil counts | Eosinophilic; neutrophilic; pauci-inflammatory | ICS; neutrophilic asthma is rare in children, no evidence base for treatment in children,63 determine underlying cause of neutrophilia | Better asthma control, fewer attacks; improved outcomes, especially symptom control; reduction in courses of systemic steroids; avoidance of inappropriate treatments |
| Airway infection | Cough swab; induced sputum; bronchoscopy | Bacterial, viral, or fungal cause related to systemic or mucosal immune paresis | Targeted antibiotics for bacterial infection; consideration of antifungal treatment for severe asthma with fungal sensitisation119 | Improved outcomes, especially symptom control |
| Heightened cough reflex | Cough challenge (rarely performed in children) | Mechanisms not understood in children | No licensed treatment in children | Nothing to change outcomes |
FeNO=fractional exhaled nitric oxide; FEV1=forced expiratory volume in 1 second; ICS=inhaled corticosteroids; LABA=longacting β2 agonist; SABA=shortacting β2 agonist; SD=standard deviation.
Deconstructing the airway is particularly important in preschool asthma. Unlike school age children with asthma, many have no evidence of type 2 inflammation, and if ICS is prescribed indiscriminately to all preschool children with wheeze, no benefit is seen.8 However, use of two biomarkers, peripheral blood eosinophil count, and aeroallergen sensitisation allows delineation of a steroid responsive group.9 10 Building on this finding to delineate other phenotypes should be a consideration for the future.
Personalised treatment: getting the core basics of diagnosis and management right
Frequent misdiagnosis of asthma in children
Asthma, although common, is a complex disease; however, diagnoses in children and young people are often based on reported symptoms that have low specificity and sensitivity in adult studies,11 and it is unlikely that the findings would be different in children. Furthermore, in children with suspected asthma, objective testing to confirm the asthma diagnosis is done infrequently.12 13 Overdiagnosis is estimated at between 30% and 50% of the general population with paediatric asthma14 15; many of these diagnoses are made for children who are otherwise healthy but with recurrent coughs and colds. Underdiagnosis also occurs14 and the consequences of misdiagnosis are important.16 An erroneous diagnosis of asthma can lead to healthy children being given unnecessary drug treatments, or more rarely, an important diagnosis might be missed in children whose symptoms are caused by another treatable condition.
Both misdiagnosis groups might then be exposed to the risks of side effects of unnecessary asthma treatments, particularly as ICS doses might be increased when symptoms do not respond. Furthermore, if inhalers are given to children with recurrent coughs and colds who do not need them, the diagnosis of asthma might be trivialised and the seriousness of the condition underestimated.16 Children with asthma who do not have a diagnosis are also untreated and are at risk of asthma attacks and persistent symptoms, leading to time off school, reduced exercise tolerance, and affected quality of life. An asthma diagnosis is often made by primary care and secondary care clinicians who have little or no paediatric respiratory experience, and who do not perform objective testing as recommended by the National Institute for Health and Care Excellence (NICE). This lack of experience commonly results in incorrect diagnoses and management.15 NRAD highlighted that the potential for adverse outcomes, including death, in children and young people is poorly recognised among healthcare professionals.1 The large number of children with a wrong label of asthma makes it more difficult to identify those with poorly controlled disease or at high risk of an attack, leading to inadequate management.17 Furthermore, a diagnosis based on clinical history without objective measurement results in a limited understanding of the type of asthma in the individual and likely implementation of inappropriate management strategies. Hence, recent NICE guidance stresses the importance of objective tests being performed before making a diagnosis of asthma.11
Ensuring the correct basic management steps before use of biological medicines
A crucial initial step is ensuring the accuracy of the diagnostic label of asthma. A key message11 18 is that objective testing should be used to diagnose asthma in accord with NICE guidelines.15 Even quite young preschool children can perform spirometry and bronchodilator reversibility in a clinical setting,19 and techniques such as offline measurement of fractional exhaled nitric oxide (FeNO), multiple breath washout, and impulse oscillometry—which require no more than passive cooperation—are increasingly used clinically. The peripheral blood eosinophil count is now a validated point-of-care test,20 which could make it easier to measure a blood eosinophil count in children. A blood eosinophil count can be used across the developmental spectrum as a biomarker of airway eosinophilia, and therefore can be predictive of a response to ICS and biological medicines such as mepolizumab (see below).
One caveat is that some children with asthma could have non-obstructive spirometry when well. There is no one asthma diagnostic test; table 2 lists supportive tests, and the more tests that are negative, the less likely the diagnosis. NICE has stressed the need for objective testing to support a diagnosis of asthma.11 The tests that are available will depend on local resources; in high income settings, spirometry should be performed as a minimum, and FeNO is increasingly available in primary care. In accordance with NICE guidance, we advise that testing should be done before a diagnosis is made and especially if the prescription of ICS is being contemplated. Children with suspected asthma or preschool wheeze should be referred to a paediatric respiratory specialist if the diagnosis is uncertain, the response to treatment is poor, and severe attacks are ongoing, or if primary care or parents request a second opinion. Often the first step is re-evaluation of the diagnosis and further tests, rather than escalating treatment that might not be appropriate.
Table 2.
Objective tests for asthma diagnosis and management
| What to measure | When to measure | Considerations |
| Spirometry (FEV1, FVC) | Before commencement of asthma treatment if possible, or withhold LABA for 12 hours and SABA for 4 hours. | Airflow obstruction is variable and spirometry can be normal, even in severe asthma; testing when symptomatic can be very informative. |
| Peak flow | Pre-treatment if possible, for 2 weeks, after which data collection is less accurate and complete; might also be useful to guide management in those individuals with poor symptom perception. | Electronic peak flow metres are likely to be much more useful and reliable. Clinicians should be aware of the possibility of fabricated measurements with standard peak flow metres.120 |
| FeNO | Pre-treatment if possible; before other forced expiratory manoeuvres.121 | FeNO is a useful measure of adherence to treatment with ICS. If FeNO levels fall after a period of directly observed treatment with ICS, then previous adherence to ICS treatment was poor. FeNO will remain elevated despite good adherence to treatment if the child does not have steroid responsive asthma. FeNO is also raised in individuals who do not have asthma but have atopic disease (ie, those with allergic rhinitis, eczema) |
| Tests of airway hyper-responsiveness (AHR) | For those individuals aged over 4 years with consistently normal spirometry and FeNO; LABA must be withheld for 12 hours and SABA for 6 hours. | AHR testing for children not widely available; field tests, such as running up and down the stairs in clinic, might be helpful for confirming exercise induced bronchoconstriction, but has poor sensitivity. |
| Blood eosinophils | Consider pre-treatment with ICS for preschool children; an important test for the investigation of severe asthma. | Children of preschool age with a raised blood eosinophil count are more likely to benefit from ICS. In children with severe asthma and a low blood eosinophil count, the blood eosinophil count should be checked at least three times. In children on maintenance OCS, the blood eosinophil count should be checked on the lowest possible dose of OCS to maintain asthma control. The highest blood eosinophil count within a 12 month period should be used to determine non-type 2 airway inflammation.122 If the blood eosinophil count is high in a person with asthma, it does not need to be repeated. In low to middle income countries, blood eosinophilia could reflect helminth infection. |
| Skin prick testing | Consider testing in preschool children; useful to identify aeroallergen triggers in children with poorly controlled asthma. | Preschool children with aeroallergen sensitisation might be more likely to respond to ICS; useful to identify aeroallergen triggers and guide avoidance strategies. |
AHR=airway hyper-responsiveness; FeNO=fractional exhaled nitric oxide; FEV1=forced expiratory volume in 1 second; FVC=forced vital capacity; ICS=inhaled corticosteroids; LABA=longacting β2 agonist; OCS=oral corticosteroids; ppb=parts per billion; SABA=shortacting β2 agonist; pre-treatment if possible=ideally before prescribing and starting regular maintenance asthma treatment.
The treatment approaches to asthma vary in different childhood age groups: young children (age ≤5 years), school age (6-11 years), and adolescence (age 12-18 years), because what works for one age group might not be the best approach for another. Most school age children with asthma achieve good symptom control when treated with low to moderate dose ICS with or without a longacting β agonist, but a minority (3-5% of children with asthma)3 have severe disease and poor symptom control despite maximal treatment, and these children need further escalation in support and intervention. Not all these children with symptoms have severe, treatment resistant asthma, which is rare.
The World Health Organization categorised severe asthma into three groups21: untreated severe asthma resulting from a failure of diagnosis or lack of availability of treatment (which is not confined to low and middle income countries); severe asthma that is difficult to treat, whereby social and environmental factors or comorbidities hinder the response to treatment; and severe, treatment resistant asthma that requires high level treatment with ICS and other controllers to achieve control or that remains uncontrolled despite high level treatment (figure 1).
Figure 1.
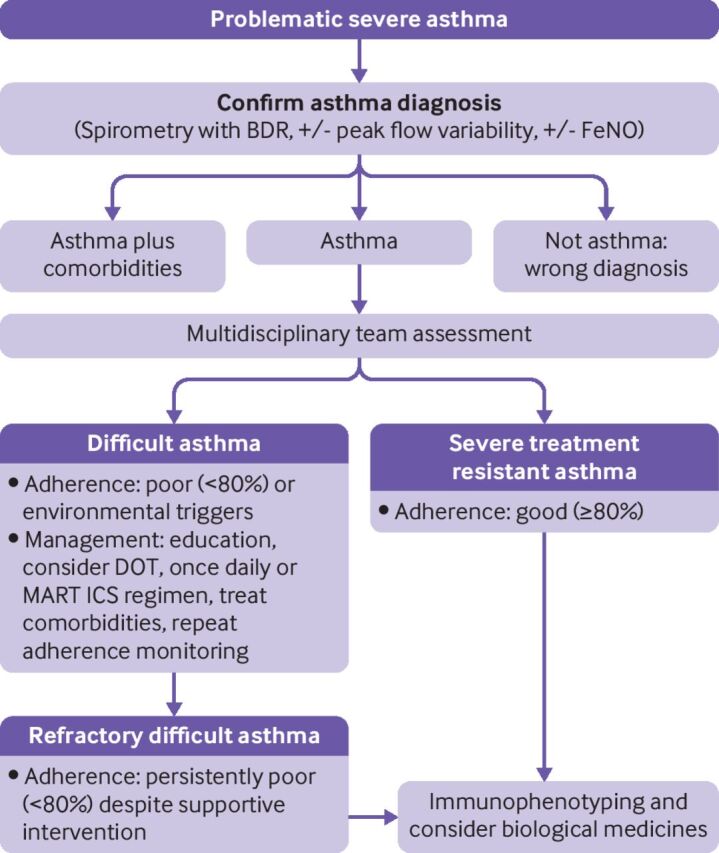
Stepwise protocol for assessment and management of children with severe asthma. BDR=bronchodilator reversibility; DOT=directly observed treatment; FeNO=fractional exhaled nitric oxide; ICS=inhaled corticosteroids; MART=maintenance and reliever treatment
A multidisciplinary approach is paramount in children with severe asthma.22 Severe asthma necessitates a broader approach that recognises extrapulmonary influences such as adherence, family values, comorbidities such as exposure to tobacco smoke, ongoing exposure to aeroallergens to which the child is sensitised, obesity, breathing pattern disorders, and poor symptom perception. Ancillary testing to exclude alternative diagnoses might be indicated.22 This team approach is essential to ensure that the right children are prescribed expensive and invasive biological medicines.
New concepts in preschool wheeze: from symptom based treatments to personalised treatment
The prevalence of wheezing in preschool age children has tripled in the past 30 years.23 24 In the UK, the number of children aged 1-5 years presenting to primary care with acute wheezing has steadily risen between 2007 and 2017,25 by contrast with acute presentations at school age.26 Management of recurrent preschool wheezing is suboptimal because it has been driven by symptom based labels (episodic viral wheeze, multiple trigger wheeze,27 persistent wheeze28). However, preschool wheezing is heterogeneous with multiple underlying pathologies, all presenting with similar symptoms, which bear little relation to the underlying phenotype.29 30
Most preschool wheeze is managed in primary care, and if simple intermittent treatment suffices to control attacks of wheeze and any interval symptoms, further tests might be unnecessary. The main goals of therapeutic intervention are to control symptoms and prevent or reduce the severity of attacks. Importantly, and unlike in older children, about 60% of preschool age children do not develop recurrent wheezing after their first episode, so clinical guidelines31 recommend maintenance treatment should only be considered for those with recurrent wheezing (four or more episodes of wheeze per year). However, if the initial episodes are so severe that, for example, treatment in a high dependency unit or paediatric intensive care unit is necessary, preventive treatment can be initiated earlier.
Management of eosinophilic, allergic, preschool wheeze
Twenty five per cent of children with preschool wheeze have eosinophilic allergic preschool wheeze, which is associated with blood eosinophilia and aeroallergen sensitisation. Use of maintenance ICS for preschool age children with recurrent wheezing associated with aeroallergen sensitisation and blood eosinophilia is often very effective; but in the absence of blood eosinophilia and aeroallergen sensitisation, a response is unlikely. Studies of lower airway inflammation have shown tissue and airway luminal eosinophilia in a subgroup of people with severe, recurrent wheeze.32–34 The INFANT trial (n=300) was among the first to show benefit with ICS in preschool children with wheeze who had aeroallergen sensitisation or blood eosinophilia, with the optimal response in children positive for both.9 However, eosinophilia might be present even in those without aeroallergen sensitivity,34 so these children might respond to ICS, and elevated blood eosinophils could be a biomarker for clinical response to ICS. A recent clinical trial (n=1074) explored the optimal blood eosinophil cut-off point for predicting a wheeze exacerbation, by combining the results of three phase 3 studies of recurrent wheezing in preschool children.10 The risk of a wheeze exacerbation was highest in those children with higher cut-off points of blood eosinophils. Adding allergen sensitisation to the blood eosinophil count improved the sensitivity of exacerbation detection. In children who were prescribed daily ICS, a significant reduction in the number of episodes of wheeze was seen in children with a baseline eosinophil count higher than 200 cells/µL and with aeroallergen sensitisation. Exploratory analysis showed a reduction in FeNO in children on daily ICS with a blood eosinophil count higher than 150 cells/µL. Further studies are needed to validate blood eosinophil cut-off points for predicting wheeze exacerbation and explore the usefulness of elevated FeNO in preschool age children as a non-invasive biomarker of response to ICS.
Management of non-allergic wheeze associated with infection
About 75% of preschool age children with wheeze have non-allergic wheeze associated with infection that does not respond to ICS. Recurrent wheezing in preschool children with non-allergic wheeze is most commonly triggered by viral upper respiratory infections, with many children being completely well between episodes. Parents of these children are often therefore reluctant to use daily preventive treatment, which in any case is unlikely to be effective.35
Unbiased assessment of lower airway inflammation in preschool children with wheeze has shown a cluster who have predominant neutrophilia, are steroid refractory, and are distinct from those with aeroallergen sensitisation. They also have lower airway viral infection (predominantly rhinovirus) and bacterial infection associated with neutrophilia, even when they are clinically stable.29 This group might benefit from a prolonged course (2-6 weeks) of targeted antibiotics.36 The antibiotic azithromycin also has anti-inflammatory and immunomodulatory effects.
The Early Administration of Azithromycin and Prevention of Severe Lower Respiratory Tract Illnesses (APRIL) trial (n=607) showed a significant reduction in number of upper respiratory infections that progressed to acute wheeze with the early use of azithromycin compared with placebo.37 However, time to next episode was unaffected and bacterial macrolide resistance was concerning. A second trial (n=72) in children aged 1-3 years showed a significant shortening of acute episodes, especially with early initiation of treatment.38 However, a third trial (n=300) that assessed the usefulness of azithromycin on presentation to the emergency department with an acute wheeze attack showed no benefit of azithromycin over placebo.39 All these studies, however, relied only on clinical phenotyping, so whether a particular cluster would benefit cannot be determined. The evidence suggests that early azithromycin might prevent progression to a severe attack, but clinical benefits must be balanced against concerns for antimicrobial resistance.
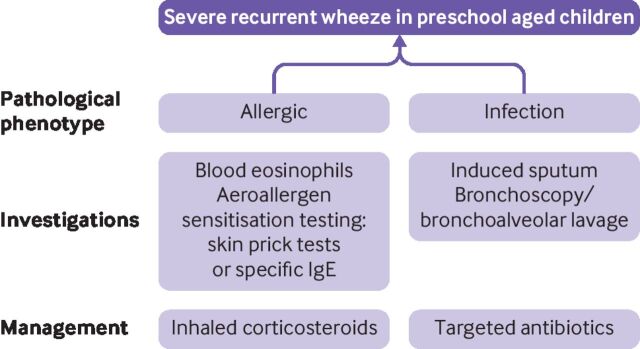
Before deciding how preschool children with more than trivial recurrent wheezing should be treated, the type of airways disease should be defined, particularly in those with severe episodes of recurrent wheeze requiring recurrent admission to hospital and intensive treatment. Skin prick testing for aeroallergen sensitisation and measuring blood eosinophil count for evidence of airway eosinophilia are both useful initial measurements that can be done before initiating ICS treatment in preschool children with recurrent wheeze (figure 2). In children with severe episodes of recurrent wheeze that require hospital admission or emergency care attendances, a referral to a specialist paediatric respiratory centre is essential to enable further investigation of the underlying disease process and separate children according to allergen sensitisation, evidence of eosinophilia, evidence of neutrophilia or infection, or evidence of airway obstruction. This approach might require additional testing (eg, bronchoscopy and induced sputum) to investigate the presence and nature of any underlying airway inflammation (figure 3). Although biomarkers for patients with type 2 inflammation are known, there is currently an urgent need for non-invasive biomarkers that can identify preschool age children with wheeze who have airway neutrophilia with bacterial dysbiosis, and interventional studies that focus on non-allergic wheeze in preschool age children.
Figure 2.
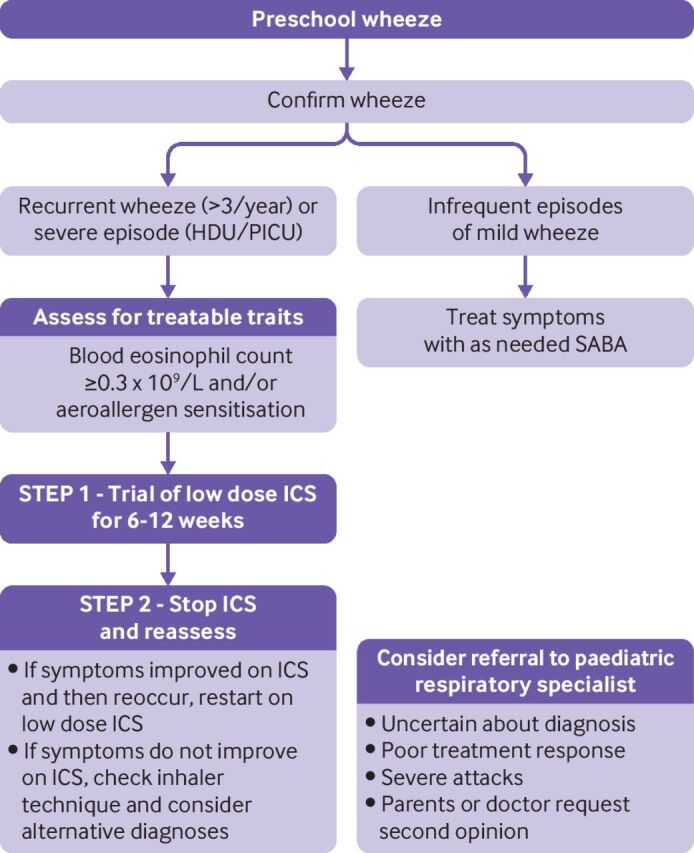
Overview of a proposed approach for the non-acute assessment and management of wheeze in children of preschool age. HDU=high dependency unit; ICS=inhaled corticosteroids; PICU=paediatric intensive care unit; SABA=shortacting β2 agonist
Figure 3.
Phenotype guided assessment and management of severe recurrent wheeze in children of preschool age. Overview of proposed pathological phenotype approach using objective measurements to guide directed management
When to reduce the use of SABA
Asthma attacks are red flags because they point to a high risk of future attacks and asthma deaths.40 41 A systematic review of factors associated with future asthma attacks in children observed that previous attacks were associated with a greatly increased risk of future attacks.41 Additionally, asthma attacks are associated with a less favourable evolution of lung function and prognosis.40 42 Most patients at risk can be readily identified in primary care. Alerts should include those individuals prescribed more than six SABA canisters/year and those not accessing ICS prescriptions43 44; recent discharge from hospital after a severe asthma attack; those who have attended emergency departments in the previous year; and those who repeatedly do not attend follow-up consultations.45 High FeNO and blood eosinophil count predicts risk of asthma attacks at least in adults.46
At least two major asthma paradoxes exist. Firstly, we prescribe SABA at step 1 of asthma management guidelines,11 with no anti-inflammatory treatment, but not LABA as solo agents at any stage, despite the belief that type 2 inflammation underlies even mild asthma during school age. The second paradox is that the morbidity of so-called mild asthma is considerable,47 and indeed about half of all deaths from asthma are not in those individuals with severe asthma. Overuse of SABA and underuse of ICS are risk factors for serious asthma attacks.1 However, despite asthma being driven by type 2 inflammation and strong evidence for harm, SABA alone still continues to be widely prescribed.48
Salmeterol is a partial β agonist with a slow onset of bronchodilatation and thus is unsuitable as a reliever. Formoterol has as rapid an onset as salbutamol and is a full agonist. Combined budesonide/formoterol as regular and reliever treatment in young people aged ≥12 years49 (SMART regimen) is well established; but with only one study in 6-11 year olds.50 Budesonide/formoterol (used as needed) results in important reductions in severe asthma attacks even in patients with mild asthma aged ≥12 years.51–55 The dose of ICS with this approach is substantially less than regular low dose ICS in step 2 asthma management.11
The Global Intiative for Asthma (GINA) now recommend formoterol/budesonide as reliever treatment at all levels of asthma severity,56 which we consider to be correct for children and young people aged ≥12 years; there is no biological reason why SABA alone is a good option in school age asthma. However, there are two problems. Firstly, the European Medicines Agency has not approved this indication because it has slightly less symptom control than SABA alone, although by less than the minimally clinically important difference. However, asthma attacks are the cause of asthma mortality, not day-to-day symptoms. Secondly, no data are available for 4-11 year olds, and fewer randomised controlled trials have been conducted in younger children with asthma, owing to a preference from the pharmaceutical industry to fund studies in adults.57 58
Research in children of all ages is challenging for multiple reasons.59–61 These include ethical considerations; for example, neither a child nor parent can give consent to a procedure of more than minimal risk purely for research purposes, limiting the use of, for example, CT scanning or bronchoscopy. Physiological testing (eg, spirometry) might not be able to be performed by young children. Research visits might need to be scheduled after school, during antisocial hours. Whatever the reasons, many more randomised controlled trials are performed in adults than children. The ethical and logistical challenges of delivering research studies in children can be overcome, for example, by opportunistically recruiting children having a clinically indicated bronchoscopy to mechanistic research studies; by arranging research study visits around clinic consultations; and by involving children and their parents early in the research study design to ensure that the study is acceptable to children and their family.
Assessment of asthma severity
Currently, many guidelines classify school age asthma severity predominantly on symptoms and lung function and do not include underlying markers of airway inflammation in the definition of severity.62 Asthma severity is mostly determined in retrospect, based on doses of prescribed drugs.47 62 63 NRAD reported that more than two thirds of asthma deaths occurred in patients classed as having mild to moderate asthma, based on the drug treatments prescribed.1 Also, the paediatric U-BIOPRED cohort study (n=282) showed that severe and mild/moderate asthma cohorts according to standard classifications were very similar in terms of morbidity, atopy, lung function, and bronchodilator reversibility at baseline.47 Hence, a definition of asthma must incorporate components of future risk, and assessment of risk should be part of asthma care at all levels.
Pathology of severe asthma: new insights
Airway inflammation can persist in the absence of symptoms, and the extent of pathophysiological abnormalities is variable.64–67 Several clinical and inflammatory phenotypes are described based on analysis of induced sputum, bronchoalveolar lavage (BAL), and endobronchial biopsies.68–70 Most school age children with mild or moderate asthma have steroid-sensitive allergic, eosinophilic airways disease associated with type 2 immune responses, including elevated interleukin (IL) 4, IL5, and IL13.71 72 Severe asthma in children is often but not invariably associated with persistent eosinophilic airway inflammation, which might be relatively resistant to treatment with ICS73 and persist despite reduced levels of type 2 cytokines. The absence of these cytokines with persistent eosinophilia in severe, treatment resistant asthma has led to the hypothesis that innate mediators such as IL33, which appear to be relatively steroid resistant, could dominate the immune response in severe, treatment resistant asthma.73 The Severe Asthma Research Program network reported that severe asthma in children was neither type 1 nor type 2 dominated, but observed elevated growth related oncogene (GRO, CXCL1); elevated regulated on activation; normal T cell expressed and secreted (RANTES, CCL5); and elevated IL10, IL12, and interferon gamma in these children.74
An observational exploratory study of cytokines in BAL from (n=91) children with severe asthma has shown a variable picture of type 1, type 2, and T helper (Th) 17 sometimes associated with BAL neutrophilia.75 In a prosective observational study of 350 children with moderate/severe asthma, researchers observed that children with neutrophilic inflammation were younger than those with eosinophilic or non-neutrophilic inflammation (median age 6, 11, and 10 years, respectively), suggesting that neutrophilic disease is more common in preschool asthma.33 In a further observational study76 of lower airway inflammation in 52 children with asthma, researchers observed a mixture of Th1, Th2, and Th17 proinflammatory cytokines; frequent viruses and bacteria in BAL; and elevated type 2 cytokines that correlated with total serum immunoglobulin (Ig) E. They also observed increased levels of IL5, IL33, and IL28A/interferon lambda 2 in children sensitised to multiple allergens. In addition, BAL IL5 levels increased with age, and correlated with peripheral blood eosinophil count. These findings could have important implications for studies of anti-IL5 strategies in children (see below).
In summary, not all paediatric severe asthma is driven by type 2 inflammation or will respond to anti-IL5 strategies. Critically determining what is driving the airway pathology is key to personalising treatment.
Beyond long term oral steroids—novel biological medicines
Biological medicine agents are monoclonal antibodies that currently can only target specific molecules that drive allergic, eosinophilic asthma (figure 4). They are an important step change because they result in a substantial reduction in asthma attacks and an improved quality of life, while enabling oral steroid sparing.
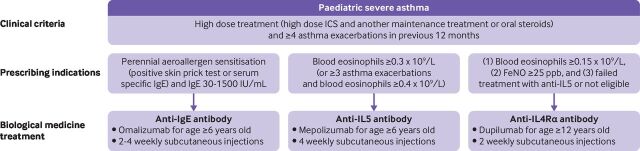
Figure 4.
Licensed biological medicine treatments for severe asthma in children. Summary of prescribing indications for biological medicine treatments licensed in the UK as an additional treatment for children with severe treatment resistant asthma. No evidence or guidance currently exists on which biological medicine should be prescribed if a child is eligible for treatment with more than one biological medicine treatment. IgE=immunoglobulin E; FeNO=fractional exhaled nitric oxide; IL4Rα=interleukin 4 receptor; IL5=interleukin 5; ppb=parts per billion; SPT=skin prick test
Omalizumab
The anti-IgE humanised monoclonal antibody omalizumab was the first biological medicine to be licensed for children and has been studied extensively in the paediatric population.77 78 Omalizumab antagonises IgE by binding to free circulating IgE, hence preventing allergen specific IgE binding to high affinity IgE receptors (FcεRI).79 In addition, the reduction in free circulating IgE results in reduced expression of FcεRI on antigen presenting cells, mast cells, and basophils.79 Omalizumab is licensed for use in severe allergic asthma from age 6 years and above. Omalizumab can be prescribed if both perennial aeroallergen sensitisation (positive skin prick test or serum specific IgE) and a total serum IgE count of 30-1500 IU/mL are present.80 The limited IgE range for prescription of omalizumab prevents the use of omalizumab in a third of children who would have otherwise been eligible.81 A meta-analysis of three randomised controlled trials including 1380 children showed a 49% decrease in exacerbation rate (odds ratio 0.51, 95% confidence interval 0.44 to 0.58, P<0.001) and a reduction in hospital admissions, despite a reduction in the dose of ICS by omalizumab compared with placebo.77 82 Given that there is most paediatric experience with omalizumab, most doctors would use this agent first in eligible patients rather than anti-IL5 and anti-IL4/IL13 biological medicines.
Mepolizumab
Mepolizumab is an anti-IL5 humanised monoclonal antibody. Mepolizumab inhibits the pleiotropic effects of IL5 on eosinophils, including proliferation, differentiation, activation, survival, and migration.83 Mepolizumab is licensed for use in children aged ≥6 years with severe eosinophilic asthma. Mepolizumab is recommended for those children with blood eosinophil counts ≥0.3×109/L and four asthma attacks requiring high dose steroids (or ≥3 asthma exacerbations and blood eosinophil counts of ≥0.4×109 /L).84 Large randomised controlled trials of mepolizumab in adults and adolescents with severe eosinophilic asthma have shown a significant reduction in asthma attacks, hospital admissions, and maintenance oral steroids, and an improved quality of life.85–88 Elevated blood eosinophil count before commencing mepolizumab treatment is strongly associated with a clinical response to treatment. Secondary analysis of phase 3 studies showed a 52% reduction in asthma exacerbations (rate ratio 0.48, 95% confidence interval 0.39 to 0.58) if the baseline blood eosinophil count was ≥150 cells/µL, and a 70% reduction in asthma exacerbations (0.30, 0.23 to 0.40) if the blood eosinophil count was ≥500 cells/µL. Mepolizumab did not reduce asthma exacerbations in those with a blood eosinophil count of ≤150 cells/µL.89 A post hoc meta-analysis of 34 adolescents with eosinophilic severe asthma who were included in phase 3 studies of mepolizumab suggested similar effects in this group and in the overall population studied, but the small sample precluded conclusions. The safety profile among adolescents appeared similar to adults, although the number of patients who could be evaluated was small.
The MUPPITS-2 study90 is the first randomised controlled trial of mepolizumab in children aged 6-17 years. The study recruited children from socioeconomically disadvantaged areas in the US with ≥2 asthma attacks in the previous year and blood eosinophil counts of ≥150 cells/µL. Researchers randomised 146 children to mepolizumab and 144 to placebo, and observed a 27% reduction in attacks in the mepolizumab group (rate ratio 0.73, 95% confidence interval 0.56 to 0.96; P=0.027). All children underwent nasal lavage and RNA sequencing at baseline and at week 52. A considerable downregulation in the type 2 pathway after treatment was seen with mepolizumab. However, eosinophil activation and hypersecretion were unchanged, as were neutrophil chemotaxis and type 1 interferon regulation associated with lower attack rates. Benefit from mepolizumab was seen in children using a lower blood eosinophil cut-off point than used in adult trials, but the effect size was also less than seen in adult studies, both factors highlighting the need for trials in children.
Currently, the main biomarker used to decide eligibility for mepolizumab is a blood eosinophil count of >300 cells/µL, extrapolated from adults. However, healthy children younger than 18 years have significantly higher blood eosinophils than adults.91 The optimal cut-off point for blood eosinophils as a biomarker for children is unknown (data from the MUPPITS-2 study suggest a lower cut-off point might be optimal). Eosinophils also have pleiotropic functions and are involved in immune homeostasis as well as disease,92 and are active against many pathogens, including viruses and parasites.93 Thus, complete eosinophilic ablation might result in unwanted effects, including excessive infections.
Dupilumab
Dupilumab is a humanised anti-IL4Rα monoclonal antibody, blocking IL4 and IL13 signalling. Dupilumab was recently licensed in the UK for use in children with severe asthma aged ≥12 years. Dupilumab is recommended by NICE for those with four severe asthma attacks in the previous year, and failed treatment or ineligibility with anti-IL5 treatment, and a blood eosinophil count of ≥0.15×109/L, and FeNO ≥25 parts per billion. The Liberty Asthma QUEST phase 3 multicentre study of dupilumab in 1902 adults and adolescents with moderate to severe asthma recruited patients without any restriction on baseline type 2 inflammatory markers. Dupilumab significantly reduced asthma attacks and improved lung function. A greater clinical response was seen in those with high baseline eosinophil count and FeNO.94 Recently, the VOYAGER trial demonstrated efficacy of dupilumab in children. In a randomised controlled trial of 408 children (aged 6-11 years) with uncontrolled moderate to severe asthma, dupilumab significantly reduced severe attacks and improved lung function and asthma related quality of life. Dupilumab had the greatest clinical effect in children with biomarkers of type 2 (inflammation) high asthma (ie, elevated baseline blood eosinophil counts >150 cells/µL or FeNO >20 parts per billion).95
Limitations of paediatric data
Despite the promise of these new approaches, several limitations remain in their application to childhood severe asthma. As mentioned above, childhood severe asthma is heterogeneous, and evidence of type 2 inflammation is less likely in school age children than in adults. Therefore, although much but not all childhood severe asthma is characterised by multiple and severe aeroallergen sensitisation, with airway eosinophilia96 and the recently licensed biological medicines for children target eosinophils, we need more evidence of efficacy in young children and this is an important evidence gap. In addition, the absence of head-to-head clinical trials of biological medicines in children is a considerable research gap to inform the optimal choice for each child with type 2 high severe asthma.
A Cochrane review of anti-IL13, anti-IL4, anti-IL5, and anti-IgE treatments in severe asthma has confirmed that of 41 randomised controlled trials, only five included children and adolescents, accounting for less than 5% of all data available on efficacy.97 The VOYAGER trial of dupilumab95 and MUPPITS-2 trial of mepolizumab90 are the two largest trials of biological medicines in children with severe asthma. Before the MUPPITS-2 study, only 34 children (aged ≥12 years) were included in randomised controlled trials of mepolizumab. More clinical trials of biological medicines in children are essential to accurately determine efficacy in children, as well as enable investigation of mechanisms of action in airway samples in children and identify biomarkers of treatment success. This need is underscored by the very different results between the MUPPITS-2 randomised controlled trial of mepolizumab in children aged 6-17 years and the DREAM study, a multicentre, double blind, placebo controlled, randomised trial of mepolizumab in 621 adults and adolescents aged ≥12 years and over.98
Type 2 low asthma
For children with poorly controlled asthma who do not have evidence of type 2 inflammation (ie, type 2 low asthma), the options for add-on treatments are limited. For any add-on treatment, consideration should be given to increased complexity of the regimen, and objective evidence of benefit should be measured and treatment stopped if there is none. Some evidence suggests that use of a combined ICS/formoterol reliever inhaler is effective irrespective of the presence of type 2 biomarkers.52 Tiotropium, a longacting muscarinic antagonist (LAMA), can be considered particularly for those children with persistently obstructed spirometry.99 The evidence in children is limited. Two phase 2 studies reported mixed results; in the adolescent study (n=392), the primary and important secondary endpoints were not met; in the trial of younger children (n=401), 5 µg but not 2.5 µg of tiotropium improved lung function.100 101 Evidence from clinical trials to support the long term use of azithromycin in children is very limited,38 102 103 and its use should probably only be considered for those children with a pattern of severe, life threatening attacks or in whom bacterial infections are an important driver of symptoms. Likewise, the European Respiratory Society/American Thoracic Society task force on severe asthma management found insufficient evidence supporting the use of macrolides in type 2 low asthma in children.63
Tezepelumab has some promise for those individuals with type 2 low asthma, although evidence of benefit in children and young people is awaited. It inhibits thymic stromal lymphopoietin, a bronchial epithelial cell derived alarmin implicated in multiple downstream processes including the regulation of type 2 immunity. However, tezepelumab has also been shown to mediate interactions between airway structural cells and immune cells, which are not exclusively driven by type 2 inflammation.104 In the recently published phase 3 study (NAVIGATOR) of 1061 patients (age ≥12 years), researchers saw a significant reduction in attacks compared with placebo even in those without evidence of type 2 inflammation (blood eosinophil count <150 cells/µL or FeNO <25 parts per billion).105
Guidelines
Table 3 lists key recommendations from three international guidelines for the diagnosis and management of asthma in children.11 31 106 Overall, they are similar in their recommendations for the use of objectives measurements to confirm the diagnosis of asthma. The guidelines, however, differ in the age brackets used to classify management recommendations, which slightly limits comparisons. A key fundamental difference is that the recent GINA guideline does not recommend the use of SABA alone for the management of asthma in children and adolescents aged 6 years and above.31 For mild asthma (step 1 treatment), GINA advise using a combination of low dose ICS and formoterol inhaler as needed. In children aged 6-11 years, owing to device limitations, SABA as needed via a metered dose inhaler plus a dose of ICS at the same time is recommended as step 1 treatment. GINA further recommend that a combination of low dose ICS and formoterol inhaler is used as a reliever for children aged 12 years and over. The GINA recommendations are a major change to the management of mild asthma and reliever treatment in asthma.
Table 3.
Clinical guidelines and recommendations on key issues relating to diagnosis and management of asthma in childhood
| Key issue | Global Initiative for Asthma guidelines31 | National Institute for Health and Care Excellence guidelines11 | British Thoracic Society/Scottish Intercollegiate Guidelines Network106 |
| Is objective evidence needed to confirm the diagnosis of asthma in children aged over 6 years old? | Yes. Strong certainty recommendation. | Yes. Strong certainty recommendation. | Yes. Low certainty recommendation. |
| Should SABA be used alone to treat asthma in children aged over 6 years old with mild asthma (step 1)? | No. Use as-needed low dose ICS-formoterol (preferred option) or low dose ICS whenever SABA taken. | Yes, if infrequent short episodes of wheeze and normal lung function. | Yes, as initial treatment. Moderate certainty recommendation. |
| Should low dose ICS-formoterol be used as a reliever in children? | Yes, this is the preferred option. The alternative, less desirable approach is SABA reliever for those children on daily maintenance ICS treatment. Strong certainty recommendation. | Yes, if using ICS-formoterol as maintenance and reliever treatment. Low certainty recommendation. | Yes, if using ICS-formoterol as maintenance and reliever treatment. |
| What are the treatment options for children with severe asthma? | The additional treatment options that might be considered by a respiratory specialist, after optimising existing treatment, include add-on LAMA, biological medicines. | Not specified. | Not specified. |
| How is asthma diagnosed in children younger than 5 years? | Clinical diagnosis, using a probability based approach that is based on recurrent symptom pattern trend, family history, and physical examination. A therapeutic trial with daily low dose ICS is a useful test to consider. Low certainty recommendation. |
Difficult to diagnose in children younger than 5 years. Advise to treat symptoms based on clinical judgment and confirm diagnosis when individual is able to undergo objective tests. | Clinical diagnosis. Assess probability of asthma. If individual has intermediate probability of asthma and symptom pattern suggests asthma, offer a trial of treatment. |
| Is maintenance ICS recommended in children younger than 5 years with wheeze? | Yes, consider a three month trial of daily low dose ICS if symptom pattern suggests asthma and persistent symptoms, and recurrent episodes or severe episodes of wheeze. | Yes, consider an eight week trial of daily moderate dose ICS if symptoms are frequent or if symptoms not controlled with SABA alone. | Yes, consider a monitored 6-8 week trial if individuals is showing symptoms. |
ICS=inhaled corticosteroids; LAMA=longacting muscarinic antagonists; SABA=shortacting β2 agonist.
Future strategies
We suggest that future strategies will include moving away from symptom management to primary or secondary prevention of eosinophilic asthma; better inclusion of children in research projects; improving subtyping of severe asthma and developing treatments for type 2 low asthma in children; and the development of biomarkers, especially for risk prediction.
Can asthma be prevented?
ICS can treat symptoms, but three large randomised controlled trials show that ICS is not disease modifying107–109—that is, early or prolonged use does not permanently switch off the pathways driving type 2 inflammation (but this has been shown by studying two genetically almost identical populations, the Amish and Hutterites). Differences in environment driven by different farming methods (traditional and modern) are associated with substantial differences in atopic disease. These beneficial results have been attributed to increased environmental bacterial and fungal diversity and lipopolysaccharide content,110 and have led to an interest in the use of oral bacterial lysates as disease modifying treatment.111 The lack of knowledge of the disease pathways could mean that a potentially muddled approach will flounder until we understand the basic biology.
Participation of young children in research
The evidence gaps in young children has been highlighted above, and should not continue, for example, as demonstrated by those affected by cystic fibrosis. Knowledge of genetic subtypes of cystic fibrosis led to the development of molecular treatments that are specific to different gene subclasses.112 This was followed by definitive clinical trials in people aged ≥12 years. When these were successful, trials were performed successively in younger age groups, down to 0-1 years. Since the younger children did not show visible respiratory symptoms, a surrogate of efficacy had to be found, namely, sweat chloride that falls substantially at all ages.113 Furthermore, in vitro models were developed to demonstrate efficacy, which correlated with in vivo response.114 115 Of course, a polygenic, environmentally modulated disease such as asthma is far more complex, but the example of cystic fibrosis should encourage researchers to discover more about basic biology and spread the benefits across the developmental course of the disease.
Treatments for type 2 low asthma
Several biological medicines are licensed, and in development, for the treatment of severe type 2 high asthma. In patients with severe asthma aged ≥12 years old with type 2 low biomarkers, tezepelumab (an inhibitor of thymic stromal lymphopoietin) showed a significant reduction in asthma exacerbations, but this observation was not significant in the adolescent group.105 Currently, no licensed immunomodulating treatments are available for children with type 2 low asthma. Future research and clinical practice needs to resolve this knowledge gap by improving the understanding of underlying pathogenesis and the subtyping of severe asthma in children.
Biomarker development
We highlighted above the need to move from total reliance on history and examination to a measurement culture. FeNO and peripheral blood eosinophil count might be useful risk predictors in adults,46 but they are unvalidated in children. Biomarkers must be used in a non-invasive and practical test at the point of care. The use of exhaled breath with sensing devices such as the e-nose116 are obvious areas to explore. Evidence gaps such as these and other areas are highlighted in the box of future research questions.
Conclusions
For a long time, asthma treatment comprised "a blue and a brown inhaler (… usually left to gather dust in the bathroom cabinet), measuring urinary cotinine and looking menacingly at the pet cat."117 We are beginning to better understand the diversities of pathology underlying the stereotypic symptoms of airway disease. If children are to benefit, the diagnosis must be correctly made, based on objective measurements, and standard protocolised management correctly implemented. For those individuals who do not respond or are unable to implement the treatment, the next step is to understand the underlying pathology.
Cluster analysis of preschool wheeze has shown at least four phenotypes of the disease, and we should explore the treatment options revealed by this approach. New options for severe allergic asthma at school age include omalizumab, mepolizumab, and dupilumab, but paediatric data to predict successful treatment are still needed. Options are currently scarce for non-type 2 asthma. Asthma attacks are a serious red flag event, and overuse of shortacting β2 agonists and underuse of ICS is an important factor. The use of ICS longacting β2 agonists in combination as rescue treatment offers superior outcomes and is more biologically rational, but data in children aged ≤11 years are lacking. Current asthma research has not focused enough on children, and obtaining evidence to inform treatment across the developmental spectrum, rather than relying on data extrapolated from adults, should be a priority. Children deserve evidence based and personalised treatment, which must be delivered.
Questions for future research.
What are the pathways driving the development of asthma, and can they be blocked?
How effective is mepolizumab at treating severe asthma in young people younger than 12 years?
How effective is combined rescue formoterol and budesonide compared with salbutamol in children aged 4-11 years?
What are the molecular pathways and therapeutic options driving non-eosinophilic asthma?
What are the optimal biomarkers that predict response to inhaled corticosteroids and biological medicines in children?
What is the role of bacteria in pathogenesis of preschool wheeze, and is treatment with targeted antibiotics useful?
Patient involvement.
We spoke with a group of children with severe asthma and their parents to gain their perspective on asthma management advances, in particular personalised treatment. This discussion helped to inform the areas covered in this review, and highlighted the importance of talking to both the child and their family about available treatment options in detail, because this makes an important positive difference to patient experience, engagement with treatment, and feeling of empowerment.
Children and their parents were very supportive of research in children with asthma, and emphasised the need to personalise care, consider each child as individual, and not provide blanket treatment.
Acknowledgments
We thank the Asthma UK Centre for Applied Research, for their funding contribution to AB and LF.
Footnotes
Contributors: ES, AB, LF, SSa, and SSo wrote the initial draft of the manuscript. AB is guarantor. The manuscript was reviewed and edited by ES and AB; all authors agreed the final version.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: We have read and understood the BMJ policy on declaration of interests and declare the following interests: none.
Provenance and peer review: Commissioned; externally peer reviewed.
References
- 1. Royal College of Physicians . Why asthma still kills: the National Review of Asthma Deaths (NRAD) confidential enquiry Report.2014. Available: https://rcplondon.ac.uk/projects/outputs/why-asthma-still-kills [Accessed 16 Nov 2022].
- 2. Asthma UK . Asthma facts and statistics.2018. Available: https://asthma.org.uk/about/media/facts-and-statistics [Accessed 2 Dec 2022].
- 3. Lang A, Carlsen KH, Haaland G, et al. Severe asthma in childhood: assessed in 10 year olds in a birth cohort study. Allergy 2008;63:1054–60. 10.1111/j.1398-9995.2008.01672.x [DOI] [PubMed] [Google Scholar]
- 4. Drew R. Richard Asher talking sense. Proceedings of the Royal Society of Medicine 1973;66:488. 10.1177/003591577306600536 [DOI] [Google Scholar]
- 5. Wenzel SE. Asthma: defining of the persistent adult phenotypes. Lancet 2006;368:804–13. 10.1016/S0140-6736(06)69290-8 [DOI] [PubMed] [Google Scholar]
- 6. Pavord ID, Beasley R, Agusti A, et al. After asthma: redefining airways diseases. Lancet 2018;391:350–400. 10.1016/S0140-6736(17)30879-6 [DOI] [PubMed] [Google Scholar]
- 7. Agusti A, Bel E, Thomas M, et al. Treatable traits: toward precision medicine of chronic airway diseases. Eur Respir J 2016;47:410–9. 10.1183/13993003.01359-2015 [DOI] [PubMed] [Google Scholar]
- 8. Grigg J, Nibber A, Paton JY, et al. Matched cohort study of therapeutic strategies to prevent preschool wheezing/asthma attacks. J Asthma Allergy 2018;11:309–21. 10.2147/JAA.S178531 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Fitzpatrick AM, Jackson DJ, Mauger DT, et al. Individualized therapy for persistent asthma in young children. J Allergy Clin Immunol 2016;138:1608–1618. 10.1016/j.jaci.2016.09.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Fitzpatrick AM, Grunwell JR, Cottrill KA, et al. Blood Eosinophils for prediction of exacerbation in preschool children with recurrent wheezing. J Allergy Clin Immunol Pract 2023;11:1485–93. 10.1016/j.jaip.2023.01.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. National Institute for Health and Care Excellence . Asthma: diagnosis and monitoring of asthma in adults, children and young people (NICE guideline Ng80).2017. 2021. Available: https://nice.org.uk/guidance/ng80 [Accessed 5 Jan 2023]. [PubMed]
- 12. Lo DK, Beardsmore CS, Roland D, et al. Lung function and asthma control in school-age children managed in UK primary care: a cohort study. Thorax 2020;75:101–7. 10.1136/thoraxjnl-2019-213068 [DOI] [PubMed] [Google Scholar]
- 13. Jonsson M, Egmar A-C, Kiessling A, et al. Adherence to national guidelines for children with asthma at primary health centres in Sweden: potential for improvement. Prim Care Respir J 2012;21:276–82. 10.4104/pcrj.2012.00051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Yang CL, Simons E, Foty RG, et al. Misdiagnosis of asthma in schoolchildren. Pediatr Pulmonol 2017;52:293–302. 10.1002/ppul.23541 [DOI] [PubMed] [Google Scholar]
- 15. Looijmans-van den Akker I, van Luijn K, Verheij T. Overdiagnosis of asthma in children in primary care: a retrospective analysis. Br J Gen Pract 2016;66:e152–7. 10.3399/bjgp16X683965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Bush A, Fleming L. Is asthma overdiagnosed Arch Dis Child 2016;101:688–9. 10.1136/archdischild-2015-309053 [DOI] [PubMed] [Google Scholar]
- 17. Levy ML, Fleming L, Warner JO, et al. Paediatric asthma care in the UK: fragmented and fatally fallible. Br J Gen Pract 2019;69:405–6. 10.3399/bjgp19X704933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Gaillard EA, Kuehni CE, Turner S, et al. European respiratory society clinical practice guidelines for the diagnosis of asthma in children aged 5-16 years. Eur Respir J 2021;58:2004173. 10.1183/13993003.04173-2020 [DOI] [PubMed] [Google Scholar]
- 19. Busi LE, Restuccia S, Tourres R, et al. Assessing bronchodilator response in preschool children using spirometry. Thorax 2017;72:367–72. 10.1136/thoraxjnl-2015-207961 [DOI] [PubMed] [Google Scholar]
- 20. Heffler E, Terranova G, Chessari C, et al. Point-of-care blood eosinophil count in a severe asthma clinic setting. Ann Allergy Asthma Immunol 2017;119:16–20. 10.1016/j.anai.2017.05.016 [DOI] [PubMed] [Google Scholar]
- 21. Bousquet J, Mantzouranis E, Cruz AA, et al. Uniform definition of asthma severity, control, and exacerbations: document presented for the World Health Organization consultation on severe asthma. J Allergy Clin Immunol 2010;126:926–38. 10.1016/j.jaci.2010.07.019 [DOI] [PubMed] [Google Scholar]
- 22. Bush A. This child’s asthma appears to be severe: but where actually is the severe problem? Acta Med Acad 2020;49:103–16. 10.5644/ama2006-124.290 [DOI] [PubMed] [Google Scholar]
- 23. Akinbami LJ, Schoendorf KC. Trends in childhood asthma: prevalence, health care utilization, and mortality. Pediatrics 2002;110:315–22. 10.1542/peds.110.2.315 [DOI] [PubMed] [Google Scholar]
- 24. Akinbami LJ, Moorman JE, Bailey C, et al. Trends in asthma prevalence, health care use, and mortality in the United States, 2001-2010. NCHS Data Brief 2012;2012:1–8. [PubMed] [Google Scholar]
- 25. Bloom CI, Franklin C, Bush A, et al. Burden of preschool wheeze and progression to asthma in the UK: population-based cohort 2007 to 2017. J Allergy Clin Immunol 2021;147:1949–58. 10.1016/j.jaci.2020.12.643 [DOI] [PubMed] [Google Scholar]
- 26. Bloom CI, Saglani S, Feary J, et al. Changing prevalence of current asthma and Inhaled corticosteroid treatment in the UK: population-based cohort 2006-2016. Eur Respir J 2019;53:1802130. 10.1183/13993003.02130-2018 [DOI] [PubMed] [Google Scholar]
- 27. Brand PLP, Baraldi E, Bisgaard H, et al. Definition, assessment and treatment of wheezing disorders in preschool children: an evidence-based approach. Eur Respir J 2008;32:1096–110. 10.1183/09031936.00002108 [DOI] [PubMed] [Google Scholar]
- 28. Castro-Rodriguez JA, Cifuentes L, Martinez FD. Predicting asthma using clinical indexes. Front Pediatr 2019;7:320. 10.3389/fped.2019.00320 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Robinson PFM, Fontanella S, Ananth S, et al. Recurrent severe preschool wheeze: from prespecified diagnostic labels to underlying endotypes. Am J Respir Crit Care Med 2021;204:523–35. 10.1164/rccm.202009-3696OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Teague WG, Lawrence MG, Williams S, et al. Novel treatment-refractory preschool wheeze phenotypes identified by cluster analysis of lung Lavage constituents. J Allergy Clin Immunol Pract 2021;9:2792–801. 10.1016/j.jaip.2021.03.059 [DOI] [PubMed] [Google Scholar]
- 31. Global Initiative for Asthma . Global strategy for asthma management and prevention. 2019. Available: http://ginasthma.org/reports [Accessed 1 Feb 2023].
- 32. Saglani S, Payne DN, Zhu J, et al. Early detection of airway wall remodeling and eosinophilic inflammation in preschool wheezers. Am J Respir Crit Care Med 2007;176:858–64. 10.1164/rccm.200702-212OC [DOI] [PubMed] [Google Scholar]
- 33. Guiddir T, Saint-Pierre P, Purenne-Denis E, et al. Neutrophilic steroid-refractory recurrent wheeze and eosinophilic steroid-refractory asthma in children. J Allergy Clin Immunol Pract 2017;5:1351–61. 10.1016/j.jaip.2017.02.003 [DOI] [PubMed] [Google Scholar]
- 34. Turato G, Barbato A, Baraldo S, et al. Nonatopic children with multitrigger wheezing have airway pathology comparable to atopic asthma. Am J Respir Crit Care Med 2008;178:476–82. 10.1164/rccm.200712-1818OC [DOI] [PubMed] [Google Scholar]
- 35. Wilson N, Sloper K, Silverman M. Effect of continuous treatment with topical corticosteroid on episodic viral wheeze in preschool children. Arch Dis Child 1995;72:317–20. 10.1136/adc.72.4.317 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Schwerk N, Brinkmann F, Soudah B, et al. Wheeze in preschool age is associated with pulmonary bacterial infection and resolves after antibiotic therapy. PLoS One 2011;6:e27913. 10.1371/journal.pone.0027913 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Bacharier LB, Guilbert TW, Mauger DT, et al. Early administration of azithromycin and prevention of severe lower respiratory tract illnesses in preschool children with a history of such illnesses: a randomized clinical trial. JAMA 2015;314:2034–44. 10.1001/jama.2015.13896 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Stokholm J, Chawes BL, Vissing NH, et al. Azithromycin for episodes with asthma-like symptoms in young children aged 1-3 years: a randomised, double-blind, placebo-controlled trial. Lancet Respir Med 2016;4:19–26. 10.1016/S2213-2600(15)00500-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Mandhane PJ, Paredes Zambrano de Silbernagel P, Aung YN, et al. Treatment of preschool children presenting to the emergency department with wheeze with azithromycin: a placebo-controlled randomized trial. PLoS One 2017;12:e0182411. 10.1371/journal.pone.0182411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. O’Byrne PM, Pedersen S, Lamm CJ, et al. Severe exacerbations and decline in lung function in asthma. Am J Respir Crit Care Med 2009;179:19–24. 10.1164/rccm.200807-1126OC [DOI] [PubMed] [Google Scholar]
- 41. Buelo A, McLean S, Julious S, et al. At-risk children with asthma (ARC): a systematic review. Thorax 2018;73:813–24. 10.1136/thoraxjnl-2017-210939 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Belgrave DCM, Buchan I, Bishop C, et al. Trajectories of lung function during childhood. Am J Respir Crit Care Med 2014;189:1101–9. 10.1164/rccm.201309-1700OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Spitzer WO, Suissa S, Ernst P, et al. The use of Β-agonists and the risk of death and near death from asthma. N Engl J Med 1992;326:501–6. 10.1056/NEJM199202203260801 [DOI] [PubMed] [Google Scholar]
- 44. Suissa S, Ernst P, Boivin JF, et al. A cohort analysis of excess mortality in asthma and the use of Inhaled beta-agonists. Am J Respir Crit Care Med 1994;149:604–10. 10.1164/ajrccm.149.3.8118625 [DOI] [PubMed] [Google Scholar]
- 45. Bush A. Asthma: What’s new, and what should be old but is not Pediatr Respirol Crit Care Med 2017;1:2. 10.4103/prcm.prcm_11_16 [DOI] [Google Scholar]
- 46. Couillard S, Laugerud A, Jabeen M, et al. Derivation of a prototype asthma attack risk scale centred on blood eosinophils and exhaled nitric oxide. Thorax 2022;77:199–202. 10.1136/thoraxjnl-2021-217325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Fleming L, Murray C, Bansal AT, et al. The burden of severe asthma in childhood and adolescence: results from the Paediatric U-BIOPRED cohorts. Eur Respir J 2015;46:1322–33. 10.1183/13993003.00780-2015 [DOI] [PubMed] [Google Scholar]
- 48. Bush A, Dalziel SR, Byrnes CA, et al. Has the time come to end use of the blue inhaler? Lancet Respir Med 2021;9:e51. 10.1016/S2213-2600(21)00185-5 [DOI] [PubMed] [Google Scholar]
- 49. Crossingham I, Turner S, Ramakrishnan S, et al. Combination fixed-dose beta agonist and steroid Inhaler as required for adults or children with mild asthma: a Cochrane systematic review. BMJ Evid Based Med 2022;27:178–84. 10.1136/bmjebm-2021-111764 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Bisgaard H, Le Roux P, Bjåmer D, et al. Budesonide/formoterol maintenance plus reliever therapy: a new strategy in pediatric asthma. Chest 2006;130:1733–43. 10.1378/chest.130.6.1733 [DOI] [PubMed] [Google Scholar]
- 51. O’Byrne PM, FitzGerald JM, Bateman ED, et al. Inhaled combined budesonide-formoterol as needed in mild asthma. N Engl J Med 2018;378:1865–76. 10.1056/NEJMoa1715274 [DOI] [PubMed] [Google Scholar]
- 52. Beasley R, Holliday M, Reddel HK, et al. Controlled trial of budesonide-formoterol as needed for mild asthma. N Engl J Med 2019;380:2020–30. 10.1056/NEJMoa1901963 [DOI] [PubMed] [Google Scholar]
- 53. Bateman ED, Reddel HK, O’Byrne PM, et al. As-needed budesonide-formoterol versus maintenance Budesonide in mild asthma. N Engl J Med 2018;378:1877–87. 10.1056/NEJMoa1715275 [DOI] [PubMed] [Google Scholar]
- 54. Hardy J, Baggott C, Fingleton J, et al. Budesonide-formoterol reliever therapy versus maintenance budesonide plus terbutaline reliever therapy in adults with mild to moderate asthma (PRACTICAL): a 52-week, open-label, Multicentre, superiority, randomised controlled trial. Lancet 2019;394:919–28. 10.1016/S0140-6736(19)31948-8 [DOI] [PubMed] [Google Scholar]
- 55. Hatter L, Bruce P, Braithwaite I, et al. ICS-Formoterol reliever versus ICS and short-acting beta2-agonist reliever in asthma: a systematic review and meta-analysis. ERJ Open Res 2021;7. 10.1183/23120541.00701-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Reddel HK, FitzGerald JM, Bateman ED, et al. GINA 2019: a fundamental change in asthma management: treatment of asthma with short-acting bronchodilators alone is no longer recommended for adults and adolescents. Eur Respir J 2019;53:1901046. 10.1183/13993003.01046-2019 [DOI] [PubMed] [Google Scholar]
- 57. Speer EM, Lee LK, Bourgeois FT, et al. The state and future of pediatric research-an introductory overview: the state and future of pediatric research series. Pediatr Res 2023:1–5. 10.1038/s41390-022-02439-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Bourgeois FT, Murthy S, Pinto C, et al. Pediatric versus adult drug trials for conditions with high pediatric disease burden. Pediatrics 2012;130:285–92. 10.1542/peds.2012-0139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Dobra R, Bentley S, Edmondson C, et al. Going the extra mile: why clinical research in cystic fibrosis must include children. Children (Basel) 2022;9:1080. 10.3390/children9071080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Modi N, Vohra J, Preston J, et al. Guidance on clinical research involving infants, children and young people: an update for researchers and research ethics committees. Arch Dis Child 2014;99:887–91. 10.1136/archdischild-2014-306444 [DOI] [PubMed] [Google Scholar]
- 61. Nordenmalm S, Kimland E, Ligas F, et al. Children’s views on taking medicines and participating in clinical trials. Arch Dis Child 2019;104:900–5. 10.1136/archdischild-2018-316511 [DOI] [PubMed] [Google Scholar]
- 62. Jaffé A, Bush A. Anti-inflammatory effects of macrolides in lung disease. Pediatr Pulmonol 2001;31:464–73. 10.1002/ppul.1076 [DOI] [PubMed] [Google Scholar]
- 63. Chung KF, Wenzel SE, Brozek JL, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J 2014;43:343–73. 10.1183/09031936.00202013 [DOI] [PubMed] [Google Scholar]
- 64. Bui DS, Walters HE, Burgess JA, et al. Childhood respiratory risk factor profiles and middle-age lung function: a prospective cohort study from the first to sixth decade. Annals ATS 2018;15:1057–66. 10.1513/AnnalsATS.201806-374OC [DOI] [PubMed] [Google Scholar]
- 65. van den Toorn LM, Overbeek SE, de Jongste JC, et al. Airway inflammation is present during clinical remission of Atopic asthma. Am J Respir Crit Care Med 2001;164:2107–13. 10.1164/ajrccm.164.11.2006165 [DOI] [PubMed] [Google Scholar]
- 66. Laprise C, Laviolette M, Boutet M, et al. Asymptomatic airway hyperresponsiveness:relationships with airway inflammation and remodelling. Eur Respir J 1999;14:63–73. 10.1034/j.1399-3003.1999.14a12.x [DOI] [PubMed] [Google Scholar]
- 67. Covar RA, Strunk R, Zeiger RS, et al. Predictors of remitting, periodic, and persistent childhood asthma. J Allergy Clin Immunol 2010;125:359–66. 10.1016/j.jaci.2009.10.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Wenzel SE, Schwartz LB, Langmack EL, et al. Evidence that severe asthma can be divided pathologically into two inflammatory subtypes with distinct physiologic and clinical characteristics. Am J Respir Crit Care Med 1999;160:1001–8. 10.1164/ajrccm.160.3.9812110 [DOI] [PubMed] [Google Scholar]
- 69. Gibson PG, Henry RL, Thomas P. Noninvasive assessment of airway inflammation in children: induced sputum, exhaled nitric oxide, and breath condensate. Eur Respir J 2000;16:1008–15. [PubMed] [Google Scholar]
- 70. Gibson PG, Simpson JL, Hankin R, et al. Relationship between induced sputum eosinophils and the clinical pattern of childhood asthma. Thorax 2003;58:116–21. 10.1136/thorax.58.2.116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Fahy JV. Type 2 inflammation in asthma--present in most, absent in many. Nat Rev Immunol 2015;15:57–65. 10.1038/nri3786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Covar RA, Szefler SJ, Martin RJ, et al. Relations between exhaled nitric oxide and measures of disease activity among children with mild-to-moderate asthma. J Pediatr 2003;142:469–75. 10.1067/mpd.2003.187 [DOI] [PubMed] [Google Scholar]
- 73. Pijnenburg MW, Fleming L. Advances in understanding and reducing the burden of severe asthma in children. Lancet Respir Med 2020;8:1032–44. 10.1016/S2213-2600(20)30399-4 [DOI] [PubMed] [Google Scholar]
- 74. Fitzpatrick AM, Higgins M, Holguin F, et al. The molecular phenotype of severe asthma in children. J Allergy Clin Immunol 2010;125:851–7. 10.1016/j.jaci.2010.01.048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Steinke JW, Lawrence MG, Teague WG, et al. Bronchoalveolar lavage cytokine patterns in children with severe neutrophilic and paucigranulocytic asthma. J Allergy Clin Immunol 2021;147:686–93. 10.1016/j.jaci.2020.05.039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Wisniewski JA, Muehling LM, Eccles JD, et al. TH1 signatures are present in the lower airways of children with severe asthma, regardless of allergic status. J Allergy Clin Immunol 2018;141:2048–60. 10.1016/j.jaci.2017.08.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Deschildre A, Marguet C, Salleron J, et al. Add-on omalizumab in children with severe allergic asthma: a 1-year real life survey. Eur Respir J 2013;42:1224–33. 10.1183/09031936.00149812 [DOI] [PubMed] [Google Scholar]
- 78. Deschildre A, Marguet C, Langlois C, et al. Real-life long-term omalizumab therapy in children with severe allergic asthma. Eur Respir J 2015;46:856–9. 10.1183/09031936.00008115 [DOI] [PubMed] [Google Scholar]
- 79. MacGlashan DW, Bochner BS, Adelman DC, et al. Down-regulation of Fc(Epsilon)RI expression on human basophils during in vivo treatment of Atopic patients with anti-IGE antibody. J Immunol 1997;158:1438–45. 10.4049/jimmunol.158.3.1438 [DOI] [PubMed] [Google Scholar]
- 80. National Institute for Health and Care Excellence . Omalizumab for treating severe persistent allergic asthma (NICE technology appraisal guidance Ta278). 2013. Available: https://www.nice.org.uk/guidance/ta278 [Accessed 12 Dec 2022].
- 81. Bossley CJ, Fleming L, Ullmann N, et al. Assessment of corticosteroid response in pediatric patients with severe asthma by using a multidomain approach. J Allergy Clin Immunol 2016;138:413–20. 10.1016/j.jaci.2015.12.1347 [DOI] [PubMed] [Google Scholar]
- 82. Fu Z, Xu Y, Cai C. Efficacy and safety of omalizumab in children with moderate-to-severe asthma: a meta-analysis. J Asthma 2021;58:1350–8. 10.1080/02770903.2020.1789875 [DOI] [PubMed] [Google Scholar]
- 83. Roufosse F. Targeting the interleukin-5 pathway for treatment of eosinophilic conditions other than asthma. Front Med 2018;5:89. 10.3389/fmed.2018.00049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. National Institute for Health and Care Excellence . Mepolizumab for treating severe eosinophilic asthma (NICE technology appraisal guidance Ta671). 2021. Available: https://www.nice.org.uk/guidance/ta671
- 85. Ortega HG, Liu MC, Pavord ID, et al. Mepolizumab treatment in patients with severe eosinophilic asthma. N Engl J Med 2014;371:1198–207. 10.1056/NEJMoa1403290 [DOI] [PubMed] [Google Scholar]
- 86. Bel EH, Wenzel SE, Thompson PJ, et al. Oral glucocorticoid-sparing effect of mepolizumab in eosinophilic asthma. N Engl J Med 2014;371:1189–97. 10.1056/NEJMoa1403291 [DOI] [PubMed] [Google Scholar]
- 87. Chupp GL, Bradford ES, Albers FC, et al. Efficacy of mepolizumab add-on therapy on health-related quality of life and markers of asthma control in severe eosinophilic asthma (MUSCA): a randomised, double-blind, placebo-controlled, parallel-group, multicentre, phase 3B trial. The Lancet Respiratory Medicine 2017;5:390–400. 10.1016/S2213-2600(17)30125-X [DOI] [PubMed] [Google Scholar]
- 88. Pavord ID, Korn S, Howarth P, et al. Mepolizumab for severe eosinophilic asthma (DREAM): a multicentre, double-blind, placebo-controlled trial. Lancet 2012;380:651–9. 10.1016/S0140-6736(12)60988-X [DOI] [PubMed] [Google Scholar]
- 89. Ortega HG, Yancey SW, Mayer B, et al. Severe eosinophilic asthma treated with mepolizumab stratified by baseline eosinophil thresholds: a secondary analysis of the DREAM and MENSA studies. Lancet Respir Med 2016;4:549–56. 10.1016/S2213-2600(16)30031-5 [DOI] [PubMed] [Google Scholar]
- 90. Jackson DJ, Bacharier LB, Gergen PJ, et al. Mepolizumab for urban children with exacerbation-prone eosinophilic asthma in the USA (MUPPITS-2): a randomised, double-blind, placebo-controlled, parallel-group trial. Lancet 2022;400:502–11. 10.1016/S0140-6736(22)01198-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Hartl S, Breyer M-K, Burghuber OC, et al. Blood eosinophil count in the general population: typical values and potential confounders. Eur Respir J 2020;55:1901874. 10.1183/13993003.01874-2019 [DOI] [PubMed] [Google Scholar]
- 92. Rodrigo-Muñoz JM, Gil-Martínez M, Sastre B, et al. Emerging evidence for pleiotropism of eosinophils. IJMS 2021;22:7075. 10.3390/ijms22137075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Percopo CM, Dyer KD, Ochkur SI, et al. Activated mouse eosinophils protect against lethal respiratory virus infection. Blood 2014;123:743–52. 10.1182/blood-2013-05-502443 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Castro M, Corren J, Pavord ID, et al. Dupilumab efficacy and safety in moderate-to-severe uncontrolled asthma. N Engl J Med 2018;378:2486–96. 10.1056/NEJMoa1804092 [DOI] [PubMed] [Google Scholar]
- 95. Bacharier LB, Maspero JF, Katelaris CH, et al. Dupilumab in children with uncontrolled moderate-to-severe asthma. N Engl J Med 2021;385:2230–40. 10.1056/NEJMoa2106567 [DOI] [PubMed] [Google Scholar]
- 96. Bossley CJ, Fleming L, Gupta A, et al. Pediatric severe asthma is characterized by eosinophilia and remodeling without T(H)2 Cytokines. J Allergy Clin Immunol 2012;129:974–82. 10.1016/j.jaci.2012.01.059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Gallagher A, Edwards M, Nair P, et al. Anti-interleukin-13 and anti-interleukin-4 agents versus placebo, anti-Interleukin-5 or anti-immunoglobulin-E agents, for people with asthma. Cochrane Database Syst Rev 2021;10:CD012929. 10.1002/14651858.CD012929.pub2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Bush A. Differing effects of mepolizumab across the life course. Lancet Respir Med 2023;11:123–5. 10.1016/S2213-2600(23)00004-8 [DOI] [PubMed] [Google Scholar]
- 99. Holguin F, Cardet JC, Chung KF, et al. Management of severe asthma: a European Respiratory Society/American Thoracic Society guideline. Eur Respir J 2020;55:1900588. 10.1183/13993003.00588-2019 [DOI] [PubMed] [Google Scholar]
- 100. Hamelmann E, Bernstein JA, Vandewalker M, et al. A randomised controlled trial of tiotropium in adolescents with severe symptomatic asthma. Eur Respir J 2017;49:1601100. 10.1183/13993003.01100-2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Szefler SJ, Murphy K, Harper T, et al. A phase III randomized controlled trial of tiotropium add-on therapy in children with severe symptomatic asthma. J Allergy Clin Immunol 2017;140:1277–87. 10.1016/j.jaci.2017.01.014 [DOI] [PubMed] [Google Scholar]
- 102. Bacharier LB, Guilbert TW, Martinez FD. Early azithromycin treatment to prevent severe lower respiratory tract illnesses in children--reply. JAMA 2016;315:2122. 10.1001/jama.2016.0921 [DOI] [PubMed] [Google Scholar]
- 103. Strunk RC, Bacharier LB, Phillips BR, et al. Azithromycin or montelukast as inhaled corticosteroid-sparing agents in moderate-to-severe childhood asthma study. J Allergy Clin Immunol 2008;122:1138–44. 10.1016/j.jaci.2008.09.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Corren J, Parnes JR, Wang L, et al. Tezepelumab in adults with uncontrolled asthma. N Engl J Med 2017;377:936–46. 10.1056/NEJMoa1704064 [DOI] [PubMed] [Google Scholar]
- 105. Menzies-Gow A, Corren J, Bourdin A, et al. Tezepelumab in adults and adolescents with severe, uncontrolled asthma. N Engl J Med 2021;384:1800–9. 10.1056/NEJMoa2034975 [DOI] [PubMed] [Google Scholar]
- 106. British Thoracic Society/Scottish Intercollegiate Guidelines Network . British guideline on the management of asthma. 2019. Available: https://%20https://www.brit-thoracic.org.uk/quality-improvement/guidelines/asthma/ [Accessed 2 Apr 2023].
- 107. Bisgaard H, Hermansen MN, Loland L, et al. Intermittent Inhaled corticosteroids in infants with episodic wheezing. N Engl J Med 2006;354:1998–2005. 10.1056/NEJMoa054692 [DOI] [PubMed] [Google Scholar]
- 108. Guilbert TW, Morgan WJ, Zeiger RS, et al. Long-term inhaled corticosteroids in preschool children at high risk for asthma. N Engl J Med 2006;354:1985–97. 10.1056/NEJMoa051378 [DOI] [PubMed] [Google Scholar]
- 109. Murray CS, Woodcock A, Langley SJ, et al. Secondary prevention of asthma by the use of inhaled fluticasone propionate in wheezy infants (IFWIN): double-blind, randomised, controlled study. Lancet 2006;368:754–62. 10.1016/S0140-6736(06)69285-4 [DOI] [PubMed] [Google Scholar]
- 110. Stein MM, Hrusch CL, Gozdz J, et al. Innate immunity and asthma risk in Amish and Hutterite farm children. N Engl J Med 2016;375:411–21. 10.1056/NEJMoa1508749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Martinez FD, Guerra S. Early origins of asthma. role of microbial dysbiosis and metabolic dysfunction. Am J Respir Crit Care Med 2018;197:573–9. 10.1164/rccm.201706-1091PP [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. De Boeck K, Amaral MD. Progress in therapies for cystic fibrosis. Lancet Respir Med 2016;4:662–74. 10.1016/S2213-2600(16)00023-0 [DOI] [PubMed] [Google Scholar]
- 113. Shteinberg M, Haq IJ, Polineni D, et al. Cystic fibrosis. Lancet 2021;397:2195–211. 10.1016/S0140-6736(20)32542-3 [DOI] [PubMed] [Google Scholar]
- 114. Ramalho AS, Fürstová E, Vonk AM, et al. Correction of CFTR function in intestinal organoids to guide treatment of cystic fibrosis. Eur Respir J 2021;57:1902426. 10.1183/13993003.02426-2019 [DOI] [PubMed] [Google Scholar]
- 115. Sette G, Lo Cicero S, Blaconà G, et al. Theratyping cystic fibrosis in vitro in ALI culture and organoid models generated from patient-derived nasal epithelial conditionally Reprogrammed stem cells. Eur Respir J 2021;58:2100908. 10.1183/13993003.00908-2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Dragonieri S, Pennazza G, Carratu P, et al. Electronic nose technology in respiratory diseases. Lung 2017;195:157–65. 10.1007/s00408-017-9987-3 [DOI] [PubMed] [Google Scholar]
- 117. Rosenthal M. CON: encouraging resistance to rule-based medicine is essential to improving outcomes. Thorax 2015;70:112–4. 10.1136/thoraxjnl-2014-206010 [DOI] [PubMed] [Google Scholar]
- 118. Wallis C, Priftis K, Chang A, et al. ERS statement on tracheomalacia and bronchomalacia in children. Eur Respir J 2019;54:1902271. 10.1183/13993003.02271-2019 [DOI] [PubMed] [Google Scholar]
- 119. Bush A. Difficult asthma and fungus. JoF 2020;6:55. 10.3390/jof6020055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Redline S, Wright EC, Kattan M, et al. Short-term compliance with peak flow monitoring: results from a study of inner city children with asthma. Pediatr Pulmonol 1996;21:203–10. 10.1002/(SICI)1099-0496(199604)21:4<203::AID-PPUL1>3.0.CO;2-P [DOI] [PubMed] [Google Scholar]
- 121. Silkoff PE, Stevens A, Pak J, et al. A method for the standardized offline collection of exhaled nitric oxide. Chest 1999;116:754–9. 10.1378/chest.116.3.754 [DOI] [PubMed] [Google Scholar]
- 122. Global Initiative for Asthma . Difficult-to-treat & severe asthma in adolescent and adult patients.A GINA pocket guide for health professionals. 2019. Available: https://ginasthma.org/wp-content/uploads/2019/04/GINA-Severe-asthma-Pocket-Guide-v2.0-wms-1.pdf [Accessed 3 Feb 2023].