Key Points
Question
Does transfusion of early and empirical high-dose cryoprecipitate in addition to standard care improve survival in bleeding patients with trauma who require activation of a major hemorrhage protocol?
Findings
In this multicenter, international, randomized clinical trial of 1604 patients with trauma comparing standard care vs usual care plus 3 early pools of cryoprecipitate, there was no difference in all-cause 28-day mortality (26.1% vs 25.3%). There was no difference in safety outcomes, transfusion requirements, or incidence of thrombotic events across study groups.
Meaning
The addition of early and empirical high-dose cryoprecipitate to usual care did not improve clinical outcomes in patients with trauma and bleeding.
Abstract
Importance
Critical bleeding is associated with a high mortality rate in patients with trauma. Hemorrhage is exacerbated by a complex derangement of coagulation, including an acute fibrinogen deficiency. Management is fibrinogen replacement with cryoprecipitate transfusions or fibrinogen concentrate, usually administered relatively late during hemorrhage.
Objective
To assess whether survival could be improved by administering an early and empirical high dose of cryoprecipitate to all patients with trauma and bleeding that required activation of a major hemorrhage protocol.
Design, Setting, and Participants
CRYOSTAT-2 was an interventional, randomized, open-label, parallel-group controlled, international, multicenter study. Patients were enrolled at 26 UK and US major trauma centers from August 2017 to November 2021. Eligible patients were injured adults requiring activation of the hospital’s major hemorrhage protocol with evidence of active hemorrhage, systolic blood pressure less than 90 mm Hg at any time, and receiving at least 1 U of a blood component transfusion.
Intervention
Patients were randomly assigned (in a 1:1 ratio) to receive standard care, which was the local major hemorrhage protocol (reviewed for guideline adherence), or cryoprecipitate, in which 3 pools of cryoprecipitate (6-g fibrinogen equivalent) were to be administered in addition to standard care within 90 minutes of randomization and 3 hours of injury.
Main Outcomes and Measures
The primary outcome was all-cause mortality at 28 days in the intention-to-treat population.
Results
Among 1604 eligible patients, 799 were randomized to the cryoprecipitate group and 805 to the standard care group. Missing primary outcome data occurred in 73 patients (principally due to withdrawal of consent) and 1531 (95%) were included in the primary analysis population. The median (IQR) age of participants was 39 (26-55) years, 1251 (79%) were men, median (IQR) Injury Severity Score was 29 (18-43), 36% had penetrating injury, and 33% had systolic blood pressure less than 90 mm Hg at hospital arrival. All-cause 28-day mortality in the intention-to-treat population was 26.1% in the standard care group vs 25.3% in the cryoprecipitate group (odds ratio, 0.96 [95% CI, 0.75-1.23]; P = .74). There was no difference in safety outcomes or incidence of thrombotic events in the standard care vs cryoprecipitate group (12.9% vs 12.7%).
Conclusions and Relevance
Among patients with trauma and bleeding who required activation of a major hemorrhage protocol, the addition of early and empirical high-dose cryoprecipitate to standard care did not improve all cause 28-day mortality.
Trial Registration
ClinicalTrials.gov Identifier: NCT04704869; ISRCTN Identifier: ISRCTN14998314
This clinical trial examines whether early, empirical, high-dose cryoprecipitate, in addition to standard major hemorrhage protocols, would improve survival in the first 28 days after hospital admission following injury.
Introduction
Hemorrhage contributes to more than half of the annual 4.4 million trauma deaths worldwide, and 1 in 4 severely injured patients with major blood loss will die. Trauma causes a multifactorial clotting disorder that exacerbates bleeding and confounds surgical and/or resuscitative attempts at hemostasis.1
Fibrinogen is the precursor of fibrin and the primary substrate for stable clot formation. It is depleted after major trauma hemorrhage due to a combination of fibrinogen consumption, breakdown (fibrinolysis), and dilution.1,2 Observational studies have shown that in severely injured patients with trauma, low fibrinogen levels are strongly associated with mortality.3,4,5,6 Many patients with trauma have fibrinogen levels on admission below replacement thresholds in major bleeding treatment guidelines.7 Major hemorrhage protocols (MHPs) guide the delivery of blood components during resuscitation and aim to deliver a balanced transfusion of red blood cell, plasma, and platelet components in ratios approaching the concentrations found in whole blood.7,8,9 However, concentrated fibrinogen products are required to raise levels toward normal range and support coagulation in patients with bleeding.6,10,11,12,13 Cryoprecipitate is a whole blood–derived, concentrated component that is standard care for fibrinogen replacement in the US and the UK.
A key question in contemporary trauma resuscitation is whether fibrinogen treatment should be given empirically and in high doses to rapidly correct levels or later in the course of bleeding as is current practice.14 The CRYOSTAT-2 trial was designed and powered by the results of the pilot randomized controlled trial (CRYOSTAT),10 which found that early transfusion of cryoprecipitate within an MHP was feasible and restored fibrinogen levels. The primary hypothesis was that early, empirical, high-dose cryoprecipitate, in addition to a standard MHP, would improve survival in the first 28 days after hospital admission following injury.
Methods
Study Design and Patients
CRYOSTAT-2 was a multicenter, phase 3, interventional, randomized, open-label, parallel-group trial conducted at 26 major trauma centers in the UK and US. The trial protocol has been previously published in full15 and is available in Supplement 1. Patients were assessed for eligibility by the receiving trauma team leader at each study site. Patients were eligible for the trial if judged to be an adult at the time of screening (if identity was not known), 16 years or older, and have sustained severe injury. Inclusion criteria were evidence of active hemorrhage requiring activation of the local MHP and to have started or received at least 1 U of any blood component. MHP activation criteria at all centers included systolic blood pressure less than 90 mm Hg at any point. Exclusion criteria were the patient being transferred from another hospital, injuries incompatible with life as assessed by the trauma team leader, or more than 3 hours elapsing from the time of injury. A waiver of consent was utilized to enroll patients, with written informed consent sought to continue data collection as soon as appropriate after randomization from the participant or a personal or professional consultee (known as a legally authorized representative in the US). The trial was approved in the UK (17/SC/0164) and granted Section 251 support (19/CAG/0161). In the US, the trial was conducted according to the US Food and Drug Administration Investigational New Drug regulations (18859) under the Exception from Informed Consent for Emergency Research, 21 CFR 50.24. Community consultation and public disclosure processes were performed before beginning the study. Mortality data at 6 months and 1 year were obtained in the UK via a data access request to NHS Digital (at 6 months only in US patients via site data collection). This study followed the Consolidated Standards of Reporting Trials reporting guideline.
Trial Oversight
An independent data monitoring committee reviewed the interim data analysis and monitored patient safety. A trial steering committee provided oversight for the study. The study was performed in accordance with the principles of the Declaration of Helsinki.
Randomization
Randomization was performed using opaque sealed envelopes in sequence at each site and conducted by study research personnel. The randomization sequence was computer-generated, had a varying undisclosed block size, and was stratified by center. Envelopes were securely stored in the emergency department or transfusion laboratory at each site.
Intervention
Patients in both groups received standard treatment according to the local MHP with a balanced, empirical ratio of red blood cells (RBCs) and fresh frozen plasma (FFP). MHPs at participating sites were reviewed by lead trial investigators14 to ensure consistency.8,16 Typically, standard MHPs delivered RBC and FFP in 4 + 4 unit packs, with platelet pools transfused with the second and subsequent packs to achieve a 1:1:1 ratio of RBC, FFP, and platelets. Standard protocols also typically include 2 pools of cryoprecipitate (4-g fibrinogen equivalent), added again to the second and subsequent packs. One prehospital helicopter medical service in the UK utilized a combined “RBC and plasma” product and whole blood was available in the US. When transfused, both were recorded as 1 U of RBCs and 1 U of FFP. In the intervention group, patients were to be administered an additional 3 pools of cryoprecipitate (6-g fibrinogen equivalent) as early as possible, with the aim to start within 90 minutes of admission.
Outcomes
The primary outcome was all-cause mortality at 28 days. Twenty-five prespecified secondary outcomes included all-cause mortality (including death from bleeding) at 6 hours, 24 hours, 6 months, and 12 months from admission; transfusion requirements (RBC, FFP, platelets, cryoprecipitate) at 24 hours; critical care and hospital stay outcomes; destination at discharge; and quality of life measures 5-level EQ-5D version (EQ-5D-5L)17 and Glasgow Outcome Scale scores18 at discharge or day 28 (whichever came first) and 6 months (eTable 1 in Supplement 3). Safety outcome measures included symptomatic venous or arterial thrombotic events up to day 28 or discharge (whichever came first). A post hoc secondary outcome of massive transfusion was added (≥10 U of RBCs from injury to 24 h after admission).
Statistical Analysis
The CRYOSTAT-2 trial was designed to detect an absolute mortality difference of 7% from a baseline mortality of 26% with 90% power using a 5% level of significance and a 2-tailed test. The baseline mortality and expected difference between treatment groups chosen for CRYOSTAT-2 was justified by the 28% mortality rate in the feasibility study, CRYOSTAT,10 and was consistent with other recent studies.19,20,21 A group sequential design utilizing O’Brien-Fleming stopping guidelines22 required 1530 patients, including interim analyses by the data monitoring committee to assess for evidence of harm or benefit after 500 and 1000 patients had been followed up for 28 days. The design meant the significance level to be applied at the final analysis was P < .0453 to preserve an overall type I error throughout the trial of 5%. Allowing for a 2.5% dropout rate, the initial estimated sample size was 1568 patients. The dropout rate was higher than anticipated, so it was increased to 4.4%, requiring 1600 patients. The statistical analysis plans are available in Supplement 2. All data were analyzed using SAS version 9.4 (SAS Institute).
Analyses were performed on an intention-to-treat (ITT) basis and included all randomized patients. Patients discharged prior to 28 days were inferred to be alive at 28 days. The primary outcome analysis was a mixed logistic regression model adjusted for center. This was supplemented by a risk-adjusted mixed logistic regression analysis to account for other factors associated with the outcome. Candidate risk factors were age, sex, type of injury (blunt or penetrating), Injury Severity Score, systolic blood pressure at admission, Glasgow Coma Scale score, and tranexamic acid administration. An unadjusted analysis (without adjustment for center) was conducted as a sensitivity analysis.
Multiple imputation based on full conditional specification was used to impute values of potential risk-adjustment variables. Primary and secondary outcome measures were not imputed and these were treated as missing data. A sensitivity analysis assessed the impact of inferring patients who were discharged to be alive by randomly changing the outcome to deceased for 2% of inferred cases (a UK national audit of major hemorrhage in trauma showed that 2.2% of patients died between hospital discharge and 1 year19).
Survival rates after 28 days were estimated using the Kaplan-Meier method and compared using Cox proportional hazards regression. Transfusion requirements were summarized as the median number of units (RBC and FFP), cryoprecipitate pools (5 single donations per pool), or platelet pools (4 whole-blood donations per pool/a single donor concentrate) administered from injury to 24 hours after admission and included the cryoprecipitate transfusion per the study intervention. Hospital stay, critical care stay, and ventilator days were estimated using a competing risks analysis with discharge/extubation as the event and death as the competing risk. A competing risks framework was also used to estimate the cumulative incidence of thrombotic events at day 28. Median EQ-5D-5L values were compared between groups using the Mann-Whitney test. Glasgow Outcome Scale score was analyzed using ordinal regression adjusted for center. No corrections for multiple comparisons were made in any of the outcome analyses, and all hypothesis testing was prespecified in the statistical analysis plan (Supplement 2).
Mortality according to timing of cryoprecipitate administration was analyzed using prespecified categories of 45 minutes from admission or less, 46 to 60 minutes from admission, 61 to 90 minutes from admission, and more than 90 minutes from admission. Further predefined subgroup analyses of the primary outcome were performed for the following variables: head injury (Abbreviated Injury Scale score <4 and ≥4), participant sex, age (<70 and ≥70 years), injury type (blunt and penetrating), receipt of cryoprecipitate (or not) in the cryoprecipitate group, and location (UK and US). A secondary outcome analysis of 6- and 24-hour mortality was also performed for head injury subgroups. A post hoc subgroup analysis for systolic blood pressure less than 90 and greater than or equal to 90 mm Hg at hospital arrival was conducted at the request of a reviewer.
A planned per-protocol analysis focused on the patients who could have benefitted from the intervention, excluding patients randomized in error, who died within 90 minutes of admission, who did not require blood components after hospital arrival, or with protocol deviations unrelated to cryoprecipitate administration. This analysis did not exclude those patients only due to nonadherence to the intervention itself, due to potential bias in such exclusions from the lack of a placebo group.
Results
A total of 1604 patients were enrolled from 25 major trauma centers in the UK (n = 1555) and 1 in the US (n = 49) between August 2017 and November 2021. On hospital arrival, 805 patients were randomized to receive the standard MHP (standard care) and 799 were randomized to receive an additional 3 pools of cryoprecipitate. Primary outcome data were missing for 73 patients (most commonly due to withdrawal of consent), leaving 1531 in the ITT analysis (Figure 1). The median time from admission to randomization was 15 minutes. Standard care and cryoprecipitate groups were well matched in baseline clinical characteristics (Table 1) (median age, 39 years; 1251 [79%] men). Median Injury Severity Score was 29 among all participants, 36% sustained a penetrating injury, and 26% had a severe head injury (Abbreviated Injury Scale score ≥4). On admission, 33% had systolic blood pressure less than 90 mm Hg. Prior to hospital arrival, 43% of participants received a blood component transfusion and 79% received tranexamic acid (96% of patients received tranexamic acid either before or in the hospital).
Figure 1. Flow of Participants in CRYOSTAT-2.
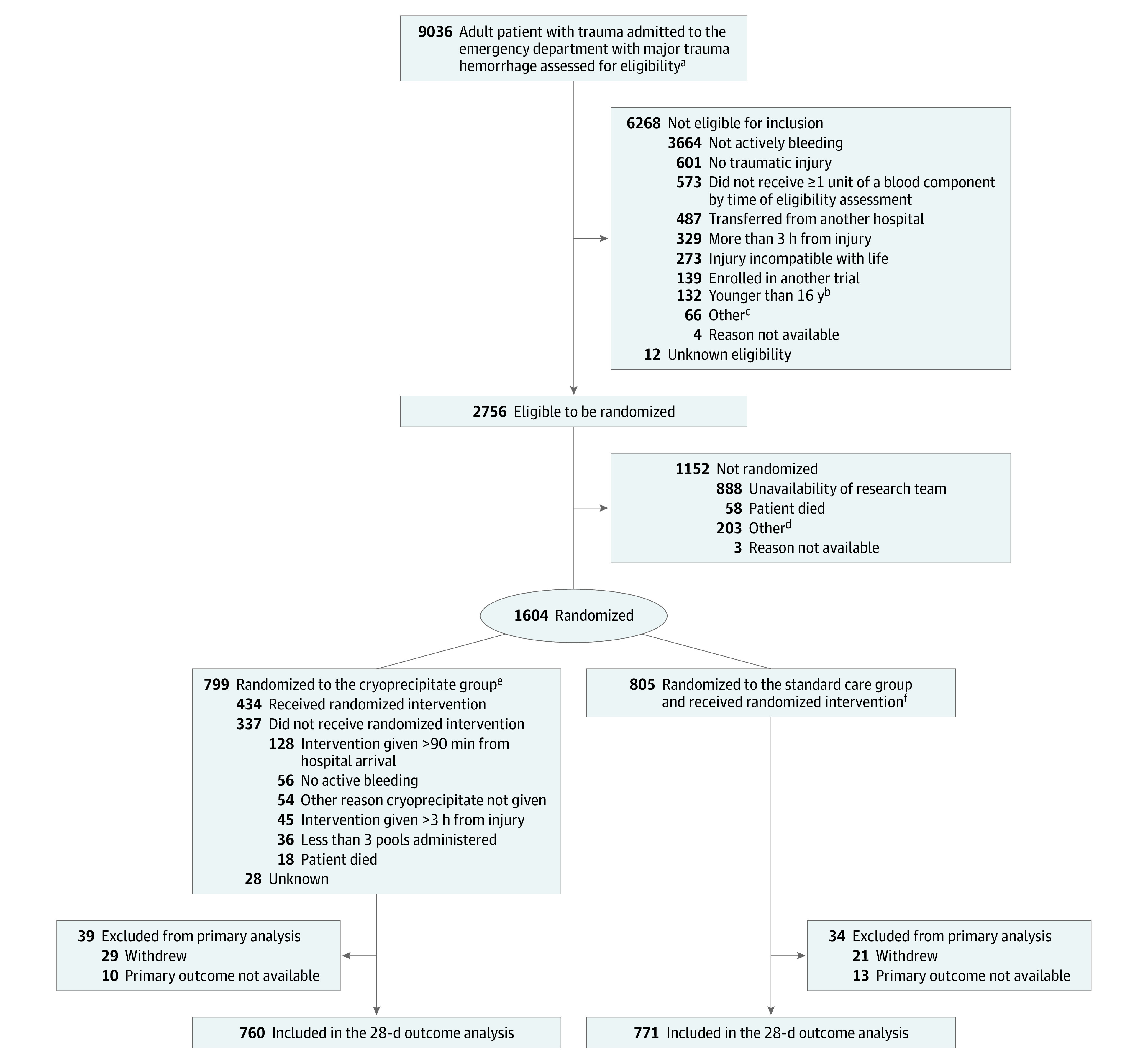
aThe trauma team leader assessed the patient to confirm whether the participant was 16 years or older either by information known to the team or by visual inspection; sustained severe injury; was bleeding (using a combination of vital signs/clinical inspection and examination) and needed transfusion therapy according to the major hemorrhage protocol; and had/or was receiving 1 U of any blood component.
bIf the age of a participant was not known at randomization, eligibility was fulfilled if the trauma team leader judged the participant to be 16 years or older.
cIncluded pregnancy, local policy exclusions, and patient in police custody.
dIncluded COVID-19, out of hours, research team not contacted, study closed, and rushed to surgery.
eAn additional 3 pools of cryoprecipitate delivered as early as possible and starting within 90 minutes of admission were added to the standard care.
fStandard major hemorrhage protocols delivered red blood cells and fresh frozen plasma in 4×4 unit packs, with a pool of platelets and 2 pools of cryoprecipitate in the second and subsequent packs.
Table 1. Demographic and Injury Characteristics.
| Characteristic | No./total No. (%) | |
|---|---|---|
| Cryoprecipitate group (n = 799) | Standard care group (n = 805) | |
| Participants | ||
| Men | 618/785 (79) | 633/796 (80) |
| Women | 167/785 (21) | 163/796 (20) |
| Age, median (IQR), y | 38 (25-55) | 40 (26-55) |
| Age ≥70 y | 71/781 (9) | 86/790 (11) |
| Time from injury to ED arrival, median (IQR), min | 75 (55-99) | 77 (55-100) |
| Injuries and physiology on ED arrival | ||
| Blunt injury | 495/785 (63) | 519/796 (65) |
| Injury Severity Score, median (IQR)a | 29 (17-43) | 29 (18-43) |
| Head AIS ≥4b | 157/665 (24) | 191/664 (29) |
| Systolic blood pressure, median (IQR), mm Hg | 102 (84-124) | 103 (83-126) |
| Systolic blood pressure <90 mm Hg | 230/724 (32) | 250/738 (34) |
| Heart rate/min, median (IQR) | 108 (88-126) | 108 (88-127) |
| In cardiac arrest | 12/717 (2) | 17/735 (2) |
| Glasgow Coma Score, median (IQR)c | 14 (3-15) | 13 (3-15) |
| Prehospital interventions administered | ||
| Red blood cell, median (IQR), U | 0 (0-2) | 0 (0-2) |
| Fresh frozen plasma, median (IQR), U | 0 (0-1) | 0 (0-1) |
| Crystalloids, median (IQR), mL | 0 (0-250) | 0 (0-250) |
| Colloids, median (IQR), mL | 0 (0-0) | 0 (0-0) |
| Tranexamic acid | 615/783 (79) | 639/796 (80) |
Abbreviations: AIS, Abbreviated Injury Scale; ED, emergency department.
The score range was 0 to 75. A score greater than 15 indicates major trauma.
The score range was 0 to 6. A score of 4 or more indicates severe head injury.
The score range was 3 to 15. In patients with trauma with no other reason for altered mental status, a score of 8 or less indicates a severe injury that has significantly affected an individual’s consciousness level.
Cryoprecipitate was administered to 665 of 785 patients (85%) in the cryoprecipitate group and 256 of 795 (32%) in the standard care group within 24 hours of hospital admission. The main reasons for study cryoprecipitate not being transfused were no evidence of active bleeding (n = 56), hemostasis achieved (n = 21), or the patient died (n = 18). Median time from admission to first administration of cryoprecipitate was 68 minutes in the cryoprecipitate group vs 120 minutes in the standard care group (P < .001; eFigure 1 in Supplement 3). In the cryoprecipitate group, 68% of patients received their first dose of cryoprecipitate within the study goal of 90 minutes after admission compared with 9% in the standard care group.
All-cause 28-day mortality for 1531 patients in the ITT analysis was 25.3% (192 of 760) in the cryoprecipitate group vs 26.1% (201 of 771) in the standard care group (odds ratio [OR], 0.96 [95% CI, 0.75-1.23]; P = .74; Figure 2 and Figure 3; relative risk [RR], 0.97 [95% CI, 0.81-1.17]). Mortality was similar between the cryoprecipitate and standard care groups at 6 hours (7.1% vs 8.6%; OR, 0.82 [95% CI, 0.58-1.17]; P = .26), 24 hours (11.2% vs 12.2%; OR, 0.91 [95% CI, 0.63-1.31]; P = .61), and 6 and 12 months (Table 2). The proportion of deaths from bleeding in the first 6 and 24 hours was not different between the 2 groups. Median time to death from hemorrhage was 191 minutes in the cryoprecipitate group vs 86 minutes in the standard care group (for those patients who died from this cause). There was no difference in the causes of death between study groups (eTable 2 in Supplement 3).
Figure 2. Mortality Overall and by Injury Type.
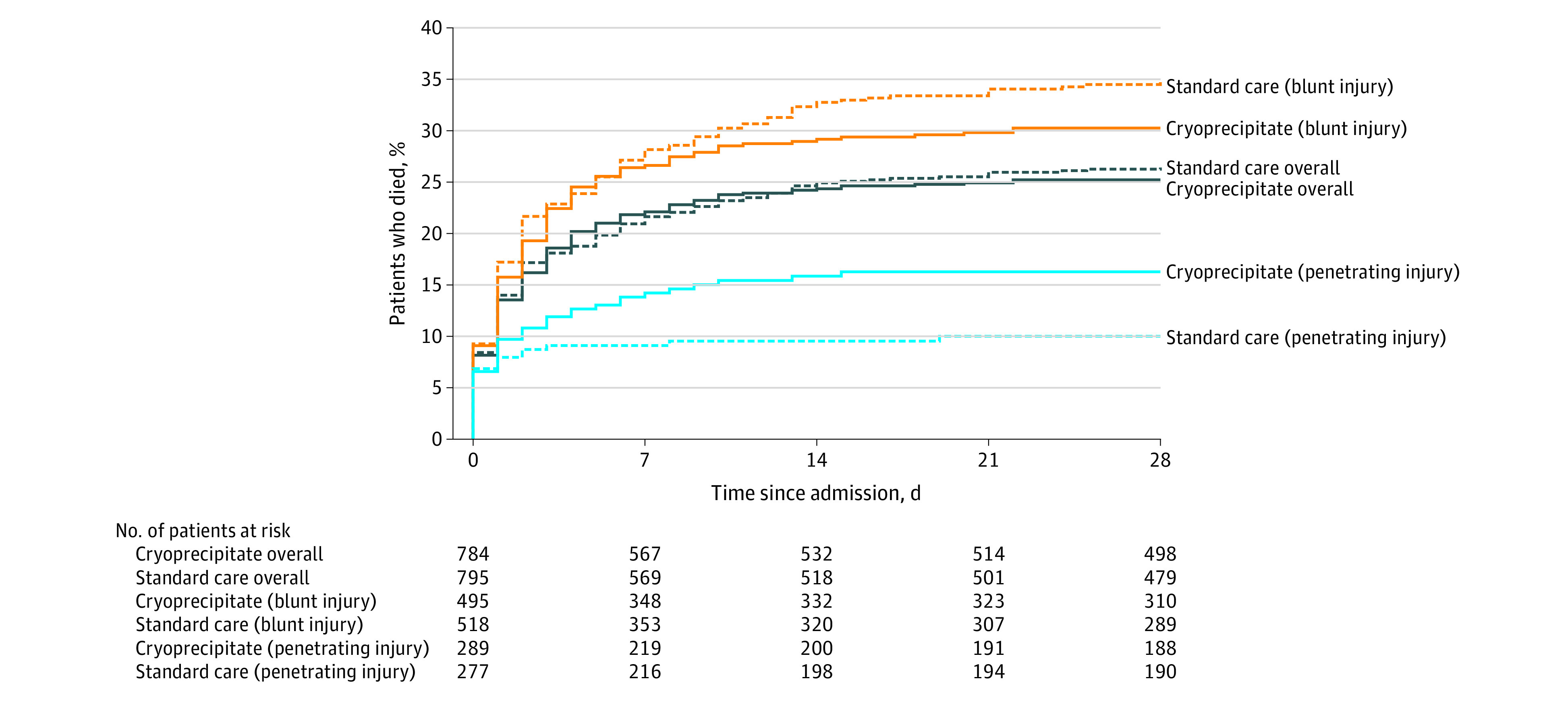
The median number of days observed was 28 days for all groups. Mortality at day 28 was analyzed as a binary outcome with odds ratios, 95% CIs, and P values reported in the results and in Figure 3.
Figure 3. Primary Outcome Analyses of All-Cause Mortality by 28 Days.

aP value for treatment group term in mixed logistic regression model, adjusted for center.
bP value for interaction term between subgroup and treatment group in a mixed logistic regression model, adjusted for center, subgroup and treatment group.
cThe Abbreviated Injury Scale (AIS) is a severity scoring system that categorizes injury by body region using a 6-point score. A score of 1 describes minor injury and a score of 6 maximal injury. An AIS score of 4 or more defines severe injury.
dP value for treatment group term in an unadjusted logistic regression model.
Table 2. Secondary Outcomes.
| Outcome | Cryoprecipitate group (n = 799) | Standard care group (n = 805) | Absolute difference (95% CI) | Odds or hazard ratio (95% CI) | P value |
|---|---|---|---|---|---|
| Mortality at 6 h from admission, No./total No. (%) | 56/784 (7.1) | 68/795 (8.6) | −1.4% (−4.1 to 1.2) | OR: 0.82 (0.58 to 1.17) | .26a |
| Mortality at 24 h from admission, No./total No. (%) | 88/783 (11.2) | 97/794 (12.2) | −1.0% (−4.2 to 2.2) | OR: 0.91 (0.63 to 1.31) | .61a |
| Kaplan-Meier estimated mortality rate at 6 mo from admission (95% CI), % | 26.1 (23.2 to 29.4) | 27.3 (24.3 to 30.7) | −1.2% (−5.7 to 3.3) | HR: 0.96 (0.79 to 1.17) | .67b |
| Kaplan-Meier estimated mortality rate at 12 mo from admission (95% CI), % | 26.6 (23.6 to 30.0) | 27.7 (24.6 to 31.1) | −1.0% (−5.5 to 3.5) | HR: 0.96 (0.79 to 1.17) | .71b |
| Components transfused over the first 24 h, median (IQR) | |||||
| Red blood cell, U | 5 (3 to 9) | 5 (3 to 8) | 0 (−0.6 to 0.6) | ||
| Fresh frozen plasma, U | 4 (2 to 8) | 4 (2 to 8) | 0 (−0.5 to 0.5) | ||
| Platelets, poolsc | 0 (0 to 1) | 0 (0 to 1) | 0 (0 to 0) | ||
| Cryoprecipitate, poolsd | 3 (3 to 3) | 0 (0 to 2) | 3 (3 to 3) | ||
| Total blood products, U | 12 (7 to 21) | 10 (5 to 18) | 2 (0.8 to 3.2) | ||
| Massive transfusion, No./total No. (%) | 179/785 (23) | 169/796 (21) | 1.6% (−2.5 to 5.7) | ||
| Crystalloids, mL | 2000 (700 to 3500) | 1600 (250 to 3200) | 400 (131.5 to 668.5) | ||
| Colloids, mL | 0 (0 to 0) | 0 (0 to 0) | 0 (0 to 0) | ||
| Safety outcomes | |||||
| Venous thromboembolism events, No./total No. (%)e | 55/799 (6.9) | 57/805 (7.1) | −0.2% (−2.7 to 2.3) | ||
| Deep venous thrombosis | 20 | 23 | |||
| Pulmonary embolism | 38 | 36 | |||
| Arterial thrombotic events, No./total No. (%)e | 26/799 (3.3) | 26/805 (3.2) | 0.0% (−1.7 to 1.8) | ||
| Stroke | 11 | 11 | |||
| Myocardial infarction | 4 | 4 | |||
| Occlusion of other artery | 12 | 11 | |||
| Cumulative incidence of all thrombotic events at day 28 (95% CI), % | 12.7 (10.1 to 15.6) | 12.9 (10.2 to 15.8) | −0.2% (−4.1 to 3.7) | .89f | |
| Critical care outcomes | |||||
| Ventilator days | 1 (0 to 6) | 1 (0 to 7) | 0 (−0.3 to 0.3) | .90f | |
| Critical care days (first episode) | 4 (1 to 12) | 4 (1 to 13) | 0 (−0.9 to 0.9) | .85f | |
| Hospital outcomes | |||||
| Length of stay | 11 (3 to 27) | 11 (3 to 27) | 0 (−2.4 to 2.4) | .88f | |
| Destination at discharge | |||||
| Home | 280/375 (75) | 278/374 (74) | |||
| Nursing home/rehabilitation facility | 9/375 (2) | 8/374 (2) | |||
| Other hospital | 63/375 (17) | 72/374 (19) | |||
| Otherg | 23/375 (6) | 16/374 (4) | |||
| Quality of life | |||||
| Median EQ-5D-5L index value at dischargeh | 0.51 (0.26 to 0.72) | 0.50 (0.20 to 0.73) | 0 (−0.1 to 0.1) | .80i | |
| GOS score at discharge or day 28 (whichever was first)j | .55k | ||||
| Low disability | 226/705 (32) | 221/712 (31) | |||
| Moderate disability | 129/705 (18) | 129/712 (18) | |||
| Severe disability | 155/705 (22) | 153/712 (21) | |||
| Severe disability | 155/705 (22) | 153/712 (21) | |||
| Persistent vegetative state | 21/705 (3) | 27/712 (4) | |||
| Death | 174/705 (25) | 182/712 (26) |
Abbreviations: GOS, Glasgow Outcome Scale; HR, hazard ratio; OR, odds ratio.
P value for treatment group term in mixed logistic regression model, adjusted for center.
P value for treatment group term in Cox regression model with frailty for center.
Defined as 1 pooled platelet concentrate derived from 4 donations of whole blood or as a single-donor apheresis platelet concentrate (an equivalent product).
Defined as the cryoprecipitate pool derived from 5 single cryoprecipitate donations.
The number of patients affected is reported. Patients may have experienced more than 1 event and all events are reported.
P value for treatment group term in Fine and Gray model.
The most common “Other” destinations at discharge were mental health facility and police custody.
The 5-level EQ-5D version (EQ-5D-5L) score range was −0.148 (worst health state) to 0.949 (best health state).
P value from Mann-Whitney test.
The score used 5 descriptive terms defining the global recovery of a participant from injury: low, moderate, or severe disability; persistent vegetative state; or death. One of these descriptive terms was assigned to each participant at hospital discharge.
P value when comparing −log2 of model with and without treatment group term in mixed ordinal regression model, adjusted for center.
There were no observed differences for any secondary outcomes between study groups for 24-hour transfusion requirements (other than cryoprecipitate units), critical care and hospital stays, destination at discharge, EQ-5D-5L score, or Glasgow Outcome Scale score (Table 2). There was no difference in any safety outcomes with similar cumulative incidence of thrombotic events between cryoprecipitate and standard care (12.7% vs 12.9%; P = .89).
The prespecified analysis of time to cryoprecipitate administration showed that 28-day mortality in the intervention group had an OR of 1.45 (95% CI, 0.91-2.31) for cryoprecipitate transfusion within 45 minutes of arrival (between 46-60 minutes: OR, 1.16 [95% CI, 0.78-1.73]; between 61-90 minutes: OR, 0.57 [95% CI, 0.38-0.87]; and more than 90 minutes: OR, 1.00 [95% CI, 0.62-1.60]) (eFigure 2 in Supplement 3). Lack of a placebo group precluded splitting the standard care group by time, and comparison was with whole-cohort mortality. Patients in the cryoprecipitate group who received cryoprecipitate very early had more severe injury and shock on admission, with a trend to higher transfusion requirements (eTables 3 and 4 in Supplement 3). The other prespecified analysis separated patients in the cryoprecipitate group into those who did and did not receive study cryoprecipitate, with 28-day mortality rates of 24.0% vs 31.7%. There was no statistically significant difference compared with the standard care group (received cryoprecipitate: OR, 0.90 [95% CI, 0.72-1.13]; P = .36; did not receive cryoprecipitate: OR, 1.28 [95% CI, 0.70-2.34]; P = .41).
In the prespecified subgroup analyses, for the 36% of patients with penetrating trauma, 28-day mortality was significantly higher in the cryoprecipitate group than the standard care group (16.2% vs 10.0%; OR, 1.74 [95% CI, 1.20-2.51]; P = .006; Figure 2 and Figure 3; RR, 1.62 [95% CI, 1.17-2.23]). There were no observed differences in clinical characteristics between the study groups with this mechanism of injury (eTable 5 in Supplement 3). In contrast, for the 64% of patients with blunt injury, respective mortality rates in the cryoprecipitate and standard care groups were 30.4% vs 34.8% (OR, 0.82 [95% CI, 0.62-1.09]; P = .16; Figure 2 and Figure 3; RR, 0.88 [95% CI, 0.72-1.06]). There were no significant differences in the other subgroup analyses (Figure 3; eFigure 2 and eTable 6 in Supplement 3).
In the per-protocol analysis, 170 patients were excluded from the full cohort, principally due to no longer requiring blood transfusion, dying within 90 minutes of arrival, or being randomized in error. Clinical characteristics were similar to the ITT cohort (eTable 7 in Supplement 3), with no difference in all-cause 28-day mortality in the cryoprecipitate group vs the standard care group (23.1% vs 22.5%; OR, 1.03 [95% CI, 0.77-1.37]; P = .83; RR, 1.02 [95% CI, 0.82-1.27]). Secondary outcomes, including transfusion requirements, complications, and causes of death, showed similar patterns to the ITT cohort results with no observed differences other than the expected differences in the amount of cryoprecipitate transfusion. Massive transfusion (RBC >10 U) was similar between the cryoprecipitate and standard care groups in both ITT (179/785 [23%] vs 169/796 [21%]) and per protocol (163/727 [22%] vs 146/707 [21%]) analyses. In ITT sensitivity analyses, the risk-adjusted analysis of 28-day mortality included adjustment for Glasgow Coma Scale score, Injury Severity Score, age, and systolic blood pressure and gave a risk-adjusted OR of 1.15 (95% CI, 0.93-1.42; P = .20). Using an unadjusted model and assessing the impact of inferring vital status based on discharge gave consistent results to the primary ITT analysis (Figure 3).
Discussion
In this multicenter, international, parallel group randomized phase 3 trial at 26 major trauma centers, the addition of early and empirical high-dose cryoprecipitate to standard care did not improve clinical outcomes in patients who were severely injured and bleeding. There were no differences in mortality at any time or in secondary or safety outcomes.
The CRYOSTAT-2 clinical trial enrolled patients who were severely injured, hypotensive, and received substantial blood component transfusions. This was the study population of interest, and the 26% mortality rate in the standard care group was as predicted in the power analysis. The findings of the CRYOSTAT-2 study are not consistent with the biological rationale for fibrinogen supplementation in trauma hemorrhage, the results of observational studies,5,6,23,24 or previous pilot or feasibility randomized controlled trials.10,11,12,13,25 It is possible that some patients could have benefited from but did not receive cryoprecipitate in time, or at a sufficient dose, to restore functional fibrinogen levels. Although the study aimed for early cryoprecipitate administration, the median time to first transfusion was more than an hour after arrival, reflecting the logistical challenge of preparing and administering a frozen blood component stored in a blood laboratory remote from the patient.
Empirical transfusion of cryoprecipitate could have resulted in a substantial proportion of patients receiving fibrinogen who did not, and would never, develop hypofibrinogenemia. Fibrinogen has a fundamental importance to clot formation with well-described derangements in trauma-induced coagulopathy, but empirical treatment has similarly not been found to be of benefit in other bleeding conditions (eg, cardiac surgery,26 postpartum hemorrhage27). High levels of fibrinogen are known to have proinflammatory and procoagulant effects,28 which could lead to increased extent and severity of complications such as thrombosis, infarction, and organ dysfunction. There was no overall increase in thrombotic events in this study. However, the observation of increased mortality in those with penetrating injury and trends in patients who received cryoprecipitate early indicate the need for research to fully characterize the safety of fibrinogen in all patients including those who may not have developed low fibrinogen levels. There has not been a specific trauma trial of diagnostic-guided fibrinogen therapy powered for mortality, but clinical trials of precision-guided coagulation therapy support a targeted approach to administration. Any potential benefit of early supplementation may only be seen in those who are bleeding quickly and the most coagulopathic.11,29,30
Limitations
This study has many limitations. First, the multiple challenges in rapidly delivering the intervention led to variability of timing of cryoprecipitate administration and an overlap with patients in the standard care group receiving it as part of their usual MHP treatment. It was not possible to specifically examine the effect of timing of cryoprecipitate administration on outcomes because of the lack of a placebo group. For patients who died of bleeding, the time to death was prolonged in the cryoprecipitate group by a mean of more than 100 minutes. The clinical effectiveness of cryoprecipitate in these patients may also have been missed due to not all patients being actively bleeding at the time of intervention and the comparatively long time to administering the intervention. Second, a research blood sample was not required to determine a fibrinogen level immediately before cryoprecipitate administration. This was to reduce delays to the intervention and maintain the pragmatic design of the trial to be deliverable at all study sites. From previous studies it was expected that approximately one-third of all patients with trauma have a fibrinogen concentration less than 2 g/L on admission and in up to 75% with trauma-induced coagulopathy.3,4,10,11,12,29 However, the prehospital administration of tranexamic acid and blood components was high and may reduce the incidence and severity of admission coagulopathy, with relative preservation of fibrinogen levels. In contrast, the longer prehospital times, albeit consistent with other UK trauma hemorrhage trials,20,31 might potentially result in more bleeding, greater consumption of fibrinogen, and therefore a greater expected effect of early replacement therapy. Third, more US sites were unable to be opened due to regulatory delays in getting trial approvals, and therefore the study results may not fully reflect US or other international practices and outcomes.
Conclusions
Among patients with bleeding and trauma who required activation of an MHP, the addition of early and empirical high-dose cryoprecipitate to standard care did not improve all cause 28-day mortality.
Section Editor: Christopher Seymour, MD, Associate Editor, JAMA (christopher.seymour@jamanetwork.org).
Trial Protocol
Statistical Analysis Plan
eFigures and eTables
Nonauthor Collaborators
Data Sharing Statement
References
- 1.Moore EE, Moore HB, Kornblith LZ, et al. Trauma-induced coagulopathy. Nat Rev Dis Primers. 2021;7(1):30. doi: 10.1038/s41572-021-00264-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fries D, Martini WZ. Role of fibrinogen in trauma-induced coagulopathy. Br J Anaesth. 2010;105(2):116-121. doi: 10.1093/bja/aeq161 [DOI] [PubMed] [Google Scholar]
- 3.Hagemo JS, Stanworth S, Juffermans NP, et al. Prevalence, predictors and outcome of hypofibrinogenaemia in trauma: a multicentre observational study. Crit Care. 2014;18(2):R52. doi: 10.1186/cc13798 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.McQuilten ZK, Wood EM, Bailey M, Cameron PA, Cooper DJ. Fibrinogen is an independent predictor of mortality in major trauma patients: a five-year statewide cohort study. Injury. 2017;48(5):1074-1081. doi: 10.1016/j.injury.2016.11.021 [DOI] [PubMed] [Google Scholar]
- 5.Inaba K, Karamanos E, Lustenberger T, et al. Impact of fibrinogen levels on outcomes after acute injury in patients requiring a massive transfusion. J Am Coll Surg. 2013;216(2):290-297. doi: 10.1016/j.jamcollsurg.2012.10.017 [DOI] [PubMed] [Google Scholar]
- 6.Rourke C, Curry N, Khan S, et al. Fibrinogen levels during trauma hemorrhage, response to replacement therapy, and association with patient outcomes. J Thromb Haemost. 2012;10(7):1342-1351. doi: 10.1111/j.1538-7836.2012.04752.x [DOI] [PubMed] [Google Scholar]
- 7.Rossaint R, Afshari A, Bouillon B, et al. The European guideline on management of major bleeding and coagulopathy following trauma: sixth edition. Crit Care. 2023;27(1):80. doi: 10.1186/s13054-023-04327-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cannon JW, Khan MA, Raja AS, et al. Damage control resuscitation in patients with severe traumatic hemorrhage: a practice management guideline from the Eastern Association for the Surgery of Trauma. J Trauma Acute Care Surg. 2017;82(3):605-617. doi: 10.1097/TA.0000000000001333 [DOI] [PubMed] [Google Scholar]
- 9.Stanworth SJ, Dowling K, Curry N, et al. ; Transfusion Task Force of the British Society for Haematology . Haematological management of major haemorrhage: a British Society for Haematology Guideline. Br J Haematol. 2022;198(4):654-667. doi: 10.1111/bjh.18275 [DOI] [PubMed] [Google Scholar]
- 10.Curry N, Rourke C, Davenport R, et al. Early cryoprecipitate for major haemorrhage in trauma: a randomized controlled feasibility trial. Br J Anaesth. 2015;115(1):76-83. doi: 10.1093/bja/aev134 [DOI] [PubMed] [Google Scholar]
- 11.Innerhofer P, Fries D, Mittermayr M, et al. Reversal of trauma-induced coagulopathy using first-line coagulation factor concentrates or fresh frozen plasma (RETIC): a single-centre, parallel-group, open-label, randomized trial. Lancet Haematol. 2017;4(6):e258-e271. doi: 10.1016/S2352-3026(17)30077-7 [DOI] [PubMed] [Google Scholar]
- 12.Curry N, Foley C, Wong H, et al. Early fibrinogen concentrate therapy for major haemorrhage in trauma (E-FIT 1): results from a UK multi-centre, randomized, double blind, placebo-controlled pilot trial. Crit Care. 2018;22(1):164. doi: 10.1186/s13054-018-2086-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Winearls J, Wullschleger M, Wake E, et al. Fibrinogen Early In Severe Trauma Study (FEISTY): study protocol for a randomized controlled trial. Trials. 2017;18(1):241. doi: 10.1186/s13063-017-1980-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Marsden M, Benger J, Brohi K, et al. ; CRYOSTAT-2 investigators . Coagulopathy, cryoprecipitate and CRYOSTAT-2: randomize the potential of a nationwide trauma system for a national clinical trial. Br J Anaesth. 2019;122(2):164-169. doi: 10.1016/j.bja.2018.10.055 [DOI] [PubMed] [Google Scholar]
- 15.Curry N, Davenport R, Lucas J, et al. The CRYOSTAT2 trial: the rationale and study protocol for a multi-centre, randomized, controlled trial evaluating the effects of early high-dose cryoprecipitate in adult patients with major trauma haemorrhage requiring major haemorrhage protocol activation. Transfus Med. 2022. [DOI] [PubMed] [Google Scholar]
- 16.Glen J, Constanti M, Brohi K; Guideline Development Group . Assessment and initial management of major trauma: summary of NICE guidance. BMJ. 2016;353:i3051. doi: 10.1136/bmj.i3051 [DOI] [PubMed] [Google Scholar]
- 17.Herdman M, Gudex C, Lloyd A, et al. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res. 2011;20(10):1727-1736. doi: 10.1007/s11136-011-9903-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jennett B, Bond M. Assessment of outcome after severe brain damage. Lancet. 1975;1(7905):480-484. doi: 10.1016/S0140-6736(75)92830-5 [DOI] [PubMed] [Google Scholar]
- 19.Stanworth SJ, Davenport R, Curry N, et al. Mortality from trauma haemorrhage and opportunities for improvement in transfusion practice. Br J Surg. 2016;103(4):357-365. doi: 10.1002/bjs.10052 [DOI] [PubMed] [Google Scholar]
- 20.Holcomb JB, Tilley BC, Baraniuk S, et al. ; PROPPR Study Group . Transfusion of plasma, platelets, and red blood cells in a 1:1:1 vs a 1:1:2 ratio and mortality in patients with severe trauma: the PROPPR randomized clinical trial. JAMA. 2015;313(5):471-482. doi: 10.1001/jama.2015.12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cole E, Weaver A, Gall L, et al. A decade of damage control resuscitation: new transfusion practice, new survivors, new directions. Ann Surg. 2021;273(6):1215-1220. doi: 10.1097/SLA.0000000000003657 [DOI] [PubMed] [Google Scholar]
- 22.O’Brien PC, Fleming TR. A multiple testing procedure for clinical trials. Biometrics. 1979;35(3):549-556. doi: 10.2307/2530245 [DOI] [PubMed] [Google Scholar]
- 23.Stinger HK, Spinella PC, Perkins JG, et al. The ratio of fibrinogen to red cells transfused affects survival in casualties receiving massive transfusions at an army combat support hospital. J Trauma. 2008;64(2)(suppl):S79-S85. doi: 10.1097/TA.0b013e318160a57b [DOI] [PubMed] [Google Scholar]
- 24.Morrison JJ, Ross JD, Dubose JJ, Jansen JO, Midwinter MJ, Rasmussen TE. Association of cryoprecipitate and tranexamic acid with improved survival following wartime injury: findings from the MATTERs II study. JAMA Surg. 2013;148(3):218-225. doi: 10.1001/jamasurg.2013.764 [DOI] [PubMed] [Google Scholar]
- 25.Ziegler B, Bachler M, Haberfellner H, et al. ; FIinTIC study group . Efficacy of prehospital administration of fibrinogen concentrate in trauma patients bleeding or presumed to bleed (FIinTIC): a multicentre, double-blind, placebo-controlled, randomized pilot study. Eur J Anaesthesiol. 2021;38(4):348-357. doi: 10.1097/EJA.0000000000001366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bilecen S, de Groot JA, Kalkman CJ, et al. Effect of fibrinogen concentrate on intraoperative blood loss among patients with intraoperative bleeding during high-risk cardiac surgery: a randomized clinical trial. JAMA. 2017;317(7):738-747. doi: 10.1001/jama.2016.21037 [DOI] [PubMed] [Google Scholar]
- 27.Wikkelsø AJ, Edwards HM, Afshari A, et al. ; FIB-PPH trial group . Pre-emptive treatment with fibrinogen concentrate for postpartum haemorrhage: randomized controlled trial. Br J Anaesth. 2015;114(4):623-633. doi: 10.1093/bja/aeu444 [DOI] [PubMed] [Google Scholar]
- 28.Peng S, Lv K. The role of fibrinogen in traumatic brain injury: from molecular pathological mechanisms to clinical management. Eur J Trauma Emerg Surg. 2022. [DOI] [PubMed] [Google Scholar]
- 29.Baksaas-Aasen K, Gall LS, Stensballe J, et al. Viscoelastic haemostatic assay augmented protocols for major trauma haemorrhage (ITACTIC): a randomized, controlled trial. Intensive Care Med. 2021;47(1):49-59. doi: 10.1007/s00134-020-06266-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gonzalez E, Moore EE, Moore HB, et al. Goal-directed hemostatic resuscitation of trauma-induced coagulopathy: a pragmatic randomized clinical trial comparing a viscoelastic assay to conventional coagulation assays. Ann Surg. 2016;263(6):1051-1059. doi: 10.1097/SLA.0000000000001608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sperry JL, Guyette FX, Adams PW. Prehospital plasma during air medical transport in trauma patients. N Engl J Med. 2018;379(18):1783. doi: 10.1056/NEJMc1811315 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Trial Protocol
Statistical Analysis Plan
eFigures and eTables
Nonauthor Collaborators
Data Sharing Statement


