Abstract
Dilated cardiomyopathy (DCM) is a cardiac condition with structural and functional impairment, where either the left ventricle or both ventricular chambers are enlarged, coinciding with reduced systolic pump function (reduced ejection fraction, rEF). The prevalence of DCM is more than 1:250 individuals, and mortality largely due to heart failure in two-third of cases, and sudden cardiac death in one-third of patients. Damage to the myocardium, whether from a genetic or environmental cause such as viruses, triggers inflammation and recruits immune cells to the heart to repair the myocardium. Examination of myocardial biopsy tissue often reveals an inflammatory cell infiltrate, T lymphocyte (T cell) infiltration, or other activated immune cells. Despite medical therapy, adverse outcomes for DCM remain. The evidence base and existing literature suggest that upregulation of CX3CR1, migration of immune cells, together with cytomegalovirus (CMV) seropositivity is associated with worse outcomes in patients with dilated cardiomyopathy. We hypothesise that this potentially occurs through cardiac inflammation and fibrosis, resulting in adverse remodelling. Immune modulators to target this pathway may potentially improve outcomes above and beyond current guideline-recommended therapy.
Keywords: dilated cardiomyopathy, CX3CR1, fractalkine, T lymphocytes, cytomegalovirus
1. Introduction
A reduced ejection fraction, or HFrEF, and enlargement of either the left ventricle or both ventricular chambers are hallmarks of dilated cardiomyopathy (DCM), a disorder that affects both the structure and function of the heart. Clinically, patients display heart failure symptoms in the absence of congenital, valvular, or coronary artery disease. DCM is categorised as either inherited (familial) or acquired (non familial) by the European Society of Cardiology (ESC). Male patients experience DCM more frequently (2:1 to 3:1) and with worse outcomes than female patients [1]. More than 1 in 250 people have DCM; progressive heart failure (70%) and sudden cardiac death (30%) account for the majority of deaths [2]. There is still a significant mortality rate even with recent improvements in the guideline-recommended heart failure treatment. Remodelling and fibrosis leading to the development of scar tissue promote the distortion of the ventricle into a spherical shape, causing the chambers to dilate, and as a result, the stroke volume, cardiac output, ventricular filling, and left ventricular end diastolic pressure (LVEDP) are all affected negatively. Early intervention is difficult before permanent damage has taken place since heart failure symptoms frequently appear gradually after the beginning of cardiac damage.
Damage to the myocardium, whether caused by genetic or environmental factors, induces inflammation and recruits immune cells to repair the myocardium; the most common causes of inflammatory DCM are infections and autoimmunity [3]. Examining myocardial biopsy tissue typically reveals an inflammatory cell infiltrate, such as CD3 T lymphocyte infiltration, or other activated immune cells, such as M2 macrophages and B lymphocytes, in autoimmune diseases such as rheumatoid arthritis, systemic sclerosis, systemic lupus erythematosus, and Dresslers syndrome to name a few. All of these are capable of releasing cytokines, including transforming growth factor-1 (TGF1), interleukin-4 (IL-4), IL-1, IL-17A, IL-33, and tumour necrosis factor (TNF), which can promote remodelling, collagen deposition, and fibrosis [4]. Fibrosis results from inflammation at the site of tissue injury and is the distinguishing pathological feature of DCM, in addition to dilatation [5] (see Figure 1).
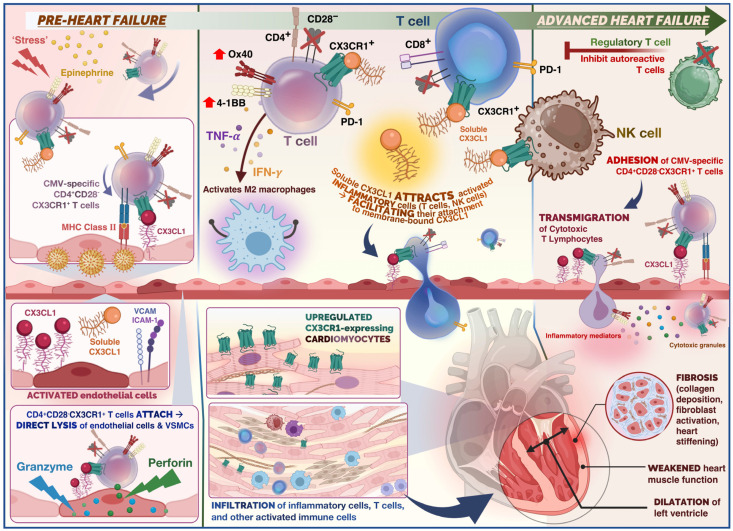
Figure 1.
Summary of the role of immune cells in the cardiovascular disease continuum from the pre-heart failure to advanced heart failure stage in dilated cardiomyopathy (DCM). Physiological stress and release of epinephrine in the acute phase (pre-heart failure), triggers release of CX3CR1+ T cells. In the case of stress-induced viral reactivation, rapid mobilisation for CMV-specific T cells occurs. CX3CR1+ CMV-specific CD4+ T cells would adhere to endothelium, and undergo antigen recognition, leading to activation of cytotoxic effector cells. Direct lysis of endothelial cells and vascular smooth muscle cells (VSMCs) occurs (mediated by granzymes and perforin). Release of cytokines such as TNF-α and IFN-γ as well as chemokines from CMV-specific CD4+ T cells potentially attracts further immune cells such as polarised macrophages and NK cells through endothelial induction of fractalkine, which may enhance endothelial injury. The CX3CR1– regulatory T cells (Tregs) are capable of inhibiting autoreactive T cells. Thus, administration of Tregs serves as a potential therapeutic target to promote immune tolerance. Soluble fractalkine binds to its chemokine receptor on cytotoxic T cells and NK cells which facilitates adhesion and transmigration across the endothelium. Infiltrating T cells and inflammatory cells release inflammatory mediators and cytotoxic granules which promote remodelling, collagen deposition, and fibrosis. This ultimately results in weakening of the ventricular heart muscle and ventricular dilatation.
Similar to healing post-myocardial infarction, fibrotic scar tissue ultimately replaces damaged tissue, stiffening the heart through fibrosis, and eventually exacerbating the progression to dilation and heart failure. The absence of treatment guidelines for inflammatory DCM causes necessitates further clinical investigation. Infections caused by viruses are a common cause of myocarditis, which can contribute to inflammatory DCM in certain individuals [6].
While DCM accounts for half of the cases of non-ischemic cardiomyopathies, this review will focus primarily on patients with idiopathic DCM (30–40%) and auto-immune disease background (5–10%) who do not respond well to standard treatment (see Table 1 for causes) [7]. Both are believed to have a significant inflammatory component that could be targeted by specific immune modulators. A worse prognosis is associated with an increased myocardial immune response in patients with DCM [8]. The association between M2 macrophages and collagen production suggests that ventricular remodelling in DCM may be associated with the phenotypic polarisation of macrophages towards M2.
Table 1.
Causes of Dilated Cardiomyopathy.
| Genetic Causes | LMNA44, MYH6, MYH7, MYBPC3, TNNT2, TTN46, RBM20, SCN5A, BAG3, PSEN3 | |
|---|---|---|
| Infections | Viruses |
Adenovirus spp., Coronavirus spp., Coxsackievirus spp. (groups A and B), Cytomegalovirus spp., Dengue virus, Echovirus spp., Epstein-Barr virus, Hepatitis B virus, Hepatitis C virus, Herpes Simplex Virus, Human Herpesvirus 6, HIV, Influenza A and Influenza B viruses, Mumps rubulavirus, parvovirus (B19), poliovirus, Rabies virus, Respiratory syncytial virus, Rubella virus, Measles virus, and Varicella- zoster virus |
| Bacteria | ß-haemolytic streptococci, Borrelia burgdorferi, Brucella spp., Campylobacter jejuni, Chlamydia spp., Clostridium spp., Corynebacterium diphtheriae, Neisseria spp., Haemophilus influenza, Legionella pneumophila, Listeria monocytogenes, Mycoplasma pneumonia, Neisseria meningitidis, Salmonella (Berta and Typhi), Streptococcus pneumonia, Staphylococcus spp., and Treponema pallidum | |
| Protozoa | Entamoeba histolytica, Leishmania spp., Plasmodium vivax, Plasmodium falciparum, Toxoplasma gondii, and Trypanosoma cruzi | |
| Helminths | Taenia spp., Echinococcus spp., Schistosoma spp., Toxocara spp., and Trichinella spp. | |
| Fungi | Actinomyces spp., Aspergilus spp., Coccidioides immitis, and Cryptococcus neoformans | |
| Autoimmunity | Systemic sclerosis, rheumatoid arthritis, systemic lupus erythematosus, dermatomyositis, sarcoidosis, Dressler syndrome, post-cardiotomy syndrome, post-infectious autoimmune disease, and post-radiation autoimmune disease | |
| Toxin exposure | Alcohol, amphetamines, anthracyclines, cannabis, catecholamines, cocaine, 5-fluorouracil, lithium, heavy metals (cobalt, lead, and mercury), and carbon monoxide |
|
| Metabolic or Endocrine | Cushing disease, hypothyroidism, hyperthyroidism, phaeochromocytoma, chronic hypocalcaemia, hypophosphataemia, and inborn errors of metabolism such as mitochondrial diseases and nutritional deficiency (carnitine, thiamine, and selenium) | |
| Pregnancy | Peripartum cardiomyopathy | |
2. Phenotypic Clustering of Dilated Cardiomyopathy Patients
Recent research from the Maastricht Cardiomyopathy Registry included 795 consecutive DCM patients who underwent extensive phenotyping, including extensive clinical data on aetiology and comorbidities, imaging, and endomyocardial biopsies [9]. On the basis of unsupervised hierarchical clustering of principal components, the authors found four mutually exclusive and clinically distinct phenogroups (PG):
PG1 included 331 patients with mild systolic dysfunction
(Mean ejection fraction: 43% ± 9%),
PG2 included 83 patients with auto-immune disease background
PG3 included 165 patients with cardiac arrhythmias (mainly atrial fibrillation and ventricular tachycardias), including patients with genetic causes (Familial cardiomyopathy)
PG4 included 216 patients with severe systolic dysfunction (Mean ejection fraction: 23% ± 8%)
RNA-sequencing of cardiac biopsy samples (n = 91) revealed a unique molecular profile per PG: pro-inflammatory (PG2, auto-immune), pro-fibrotic (PG3, arrhythmia), and metabolic (PG4, low EF) gene expression. In addition, event-free survival varied between the four phenogroups, even after adjustment for well-known clinical predictors. Using decision tree modelling, four clinical parameters (autoimmune disease, ejection fraction, atrial fibrillation, and renal function) were identified by which every DCM patient from two independent DCM cohorts could be assigned to one of the four phenogroups with corresponding outcomes.
3. Aetiologies of Dilated Cardiomyopathy
3.1. Familial Cardiomyopathy
Although a genetic basis for familial DCM is well established, the majority of cases of DCM appear to be sporadic; that is, even when the family members of patients newly diagnosed with idiopathic DCM are clinically screened, the majority of family members have no evidence of DCM; thus, patients are ultimately diagnosed with nonfamilial (sporadic) DCM. No large, multicentre study of families whose members have been systematically clinically screened for DCM and also undergone exome or genome sequencing to identify a potential genetic cause has been published to date. A total of 15–30% of patients with DCM may be diagnosed with familial DCM if their family members undertake clinical screening, according to family-based studies [3]. Historically, large multigenerational familial DCM pedigrees have served as the foundation for the majority of DCM-associated gene discovery. These multigenerational genealogies have provided statistically sound genetic evidence for the causality of variants in DCM-associated genes. The LMNA44, MYH6, MYH7, MYBPC3, TNNT2, TTN46, RBM20, SCN5A, BAG3, PSEN3 and other common DCM-causing genes were initially identified in large DCM lineage charts, along with other genes. TTN truncating mutations are a frequent cause of DCM, affecting 25% of familial cases and 18% of sporadic cases [10]. Studies suggest there is a correlation between certain DCM genotypes and left ventricular reverse remodelling (LVRR). Verdonshot and colleagues demonstrated that the TTN variant is strongly associated with higher chances of LVRR, whereas the opposite is true for the LMNA pathogenic variant [9].
3.2. Autoimmune Myocarditis
Autoimmune myocarditis is a condition in which cardiac muscle is damaged by self-reactive immune cells. This condition is typically associated with numerous genetic and environmental risk factors. Various systemic autoimmune diseases, such as sarcoidosis or SLE (lupus), can compromise cardiac function by a variety of mechanisms [11]. The prognosis for patients with either of these conditions is typically dismal. However, it has been demonstrated that early administration of glucocorticosteroids and immunosuppressive agents improves patients’ conditions [12]. Restoring the equilibrium between autoimmunity and immune tolerance, which is regulated by Th17 and regulatory T cells (Tregs), is one of the primary therapeutic approaches for autoimmune disease [13]. Treg cells are capable of inhibiting autoreactive T cells, and multiple studies have repeatedly identified a deficiency in the quantity or function of Treg cells in autoimmune disorders [13]. Therefore, Treg administration has been demonstrated to be a promising treatment for a variety of autoimmune disorders. Additionally, one study revealed that a modest dose of interleukin-2 can expand Tregs, thereby promoting immune tolerance [14]. Downregulating Th-17 is another promising therapeutic target for autoimmune myocarditis. Based on an experimental model of autoimmune myocarditis, Th17 was responsible for the progression of acute myocarditis with an excessive immune response [13].
3.3. Post-Viral Myocarditis
Myocarditis is frequently preceded by infection, with viruses being the most prevalent cause of this condition [15,16]. There are typically three phases of viral myocarditis. During the first phase, which lasts several days, the virus obtains entry and actively replicates. Therefore, the innate immune response will be triggered against these exogenous substances. During this phase, direct viral burden contributes significantly to myocardial damage. The subsequent phase revealed that an excessive immune response is the leading cause of cardiac damage. During this phase, T cell infiltration has been reported alongside an increase in fibrosis and calcification of the myocardium. In the final phase, patients may experience either remission or progression to dilated cardiomyopathy (DCM), depending on the heart’s capacity to recover from previous insults of direct viral injury and immune persistence [15,16]. By intervening in each phase of disease progression, various therapeutic targets for post-viral myocarditis can be achieved. During the viral replication phase, for instance, a potential immunotherapy for monoclonal antibodies entails shifting the immune response from Th1 to Th2 to alleviate the severity. This could be achieved by inhibiting the IP-10 signalling pathway to reduce Th1 induction and tissue recruitment [17,18].
3.4. Immune Checkpoint Inhibitor-Related Myocarditis
Immune checkpoint inhibitors (ICIs) are a novel type of cancer treatment that is being applied to a growing number of cancer types for example malignant melanoma, Hodgkin’s lymphoma and non small cell lung cancer to name a few. The immune modulators CTLA-4, PD-1, and PD-L1 are the targets of ICIs [19]. ICIs may, however, stimulate T cell activity against host tissues, leading to immune-related adverse events (irAEs). Myocarditis is a rare adverse reaction associated with ICIs, with incidence rates ranging from 0.1% to 2% [20]. With ICI myocarditis, there is a reduction in absolute lymphocyte count and an increase in neutrophils, both of which are associated with subsequent significant adverse cardiac events [20]. Cardiovascular magnetic resonance (CMR) utilising tissue characterisation techniques such as late gadolinium enhancement (LGE) and the presence of myocardial oedema, is the gold standard non-invasive imaging test for diagnosis and risk prediction in myocarditis of other aetiologies. LGE is present in >80% of patients with non-ICI-associated myocarditis, but in 50% of patients with ICI-associated myocarditis. The longer the interval between clinical presentation and CMR, the greater the likelihood of LGE detection [21].
CTLA-4 is a crucial regulator of T cell inhibition via multiple mechanisms, including negative signalling of B7-CD28, inhibition of IL-2 mRNA expression, and interaction with the TCR-CD3 pathway to inhibit T cell activation [22,23]. After IFN-g exposure, PD-1 and PD-L1 were highly expressed to prevent T-cell-mediated signalling on host cells. The FDA has currently approved seven ICI medications for cancer therapy. Although ICI-associated myocarditis is uncommon, its mortality rate can reach 25 to 50 percent [23]. In addition, this adverse event was reported to occur within 1 to 2 months of the initial ICI dose. The underlying pathogenesis of ICI-associated myocarditis remains unknown. As these cells shared similar antigens, one study suggested a cross-reaction of T cells towards tumour and cardiac muscle [24]. Myocarditis caused by ICI presents a spectrum of moderate to severe symptoms. The final stage of this disease, if left untreated, can be fatal, manifesting as cardiogenic shock and severe arrhythmia. Therefore, additional diagnostic procedures, such as CMR, cardiac biomarkers, and echocardiography, are advised for ICI-treated patients suspected of having myocarditis. Patients with suspected ICI-associated myocarditis should generally discontinue ICI therapy to prevent further cardiac damage. The current recommendation for ICI-induced myocarditis is the administration of glucocorticoids with continuous troponin monitoring [23]. If the patient does not respond adequately to glucocorticoids, additional treatments such as intravenous immunoglobulin, plasmapheresis, infliximab, abatacept, alemtuzumab, anti-thymocyte globulin, and mycophenolate can be considered.
4. Role of Fractalkine Signalling in Cardiovascular Disease
4.1. Fractalkine (CX3CL1) and Its Receptor CX3CR1 in Atherogenesis
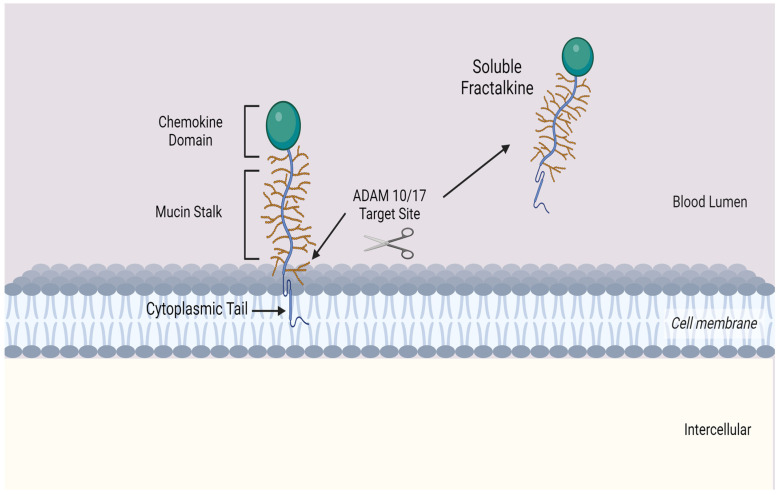
Fractalkine consists of 373 amino acids and is the only member of the CX3C chemokine subfamily with both membrane-bound and soluble variants [25,26,27,28]. The former is an adhesion molecule with four sections: an extracellular N-terminal domain, a mucin-like stalk, a transmembrane alpha helix, and a brief cytoplasmic tail. Fractalkine can be cleaved at the juncture of its stalk and transmembrane helix my metalloproteinases ADAM 10 or ADAM 17 to produce its soluble form, which functions as a chemoattractant (see Figure 2) for monocytes, NK cells, and T cells. Fractalkine is predominantly expressed on endothelial cells, and its expression is stimulated by IFN-g and TNF-α [29]. Its G-protein-coupled receptor, CX3CR1, is found on inflammatory cells including NK cells, cytotoxic T cells, and monocytes [30,31], as well as on cardiomyocytes [32]. These cells are attracted along a gradient by soluble fractalkine, and endothelial cell membrane-bound fractalkine attaches to CX3CR1 on the surface of inflammatory cells to internalise them.
Figure 2.
Transmembrane chemokine Fractalkine (CX3CL1). Graphic illustration of the transmembrane chemokine. It contains an extracellular N-domain, mucin stalk and a short cytoplasmic trail. The soluble form is generated by shedding and cleavage by metalloproteinases ADAM 10 and ADAM 17. The resulting release of the mucin-like stalk and chemokine domain acts as a chemoattractant for inflammatory cells.
Atherogenesis begins with the sequestration of lipid-laden macrophages in the intima, a process in which fractalkine is almost certainly involved. Indeed, CX3CR1− rodents develop fewer atherosclerotic plaques, and atherosclerotic human coronary arteries are stained with fractalkine, whereas healthy human coronaries are not [33,34]. Two polymorphisms in the CX3CR1 locus (V249I and T280M) have been linked to a decreased risk of atherosclerotic disease in humans [35,36,37,38,39,40,41,42,43]. Patients with the V249I polymorphism exhibited enhanced endothelium-dependent vasodilation compared to V249 homozygous patients, but comparable endothelium-independent vasodilation. It has been demonstrated that the T280M polymorphism reduces fractalkine-dependent cell–cell adhesion and induces less migration of CX3CR1-expressing inflammatory cells under shear stress.
Our previous research partially characterised the dynamics of CX3CR1 following MI and reperfusion with PCI [44]. Among CD4+ and CD8+ T cells, subdivided into CCR7− and CCR7+ subpopulations, pre-reperfusion surface expression of CX3CR1 correlated strongly with the decline of that cell type at 90 min post-reperfusion, indicating that CX3CR1 is involved in the mechanism by which T-cells are eliminated from the blood after reperfusion. CX3CR1 expression is a cytotoxicity marker in both CD4+ and CD8+ T cells, and it is frequently co-expressed with perforin and granzyme B [30,45]. CMV-specific T cells (which constitute a substantial proportion of total T cells) [46,47] express a cytotoxic phenotype that includes CX3CR1 [48], prompting our hypothesis that CX3CR1-expressing T cells contribute to worse cardiovascular outcomes in CMV seropositive patients. Moreover, a study in kidney transplant recipients revealed that cytotoxic CD4+ T cells proliferate in response to non-specific inflammation only in CMV-seropositive patients [49,50,51], which may be explained by another group’s discovery that CMV-specific T cells express two adrenergic receptors and are therefore more sensitive to systemic inflammation [48].
4.2. CX3CR1 and Myocardial Infarction
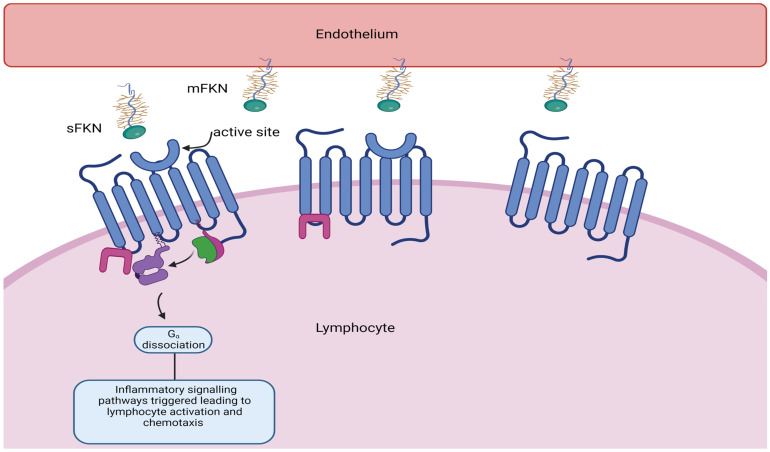
According to our own investigations, the CX3CR1 receptor is essential for the recruitment of lymphocytes that contribute to cardiac ischaemia/reperfusion injury and subsequent adverse remodelling. A subset of lymphocytes with cytotoxic properties adhere and are marginalised upon interaction with the fractalkine receptor CX3CR1 expressed on circulating lymphocytes (see Figure 3). It is hypothesised that lymphocyte marginalisation, myocardial damage, and myocardial inflammation can be mitigated by targeting the chemokine receptor CX3CR1 with inhibitors such as KAND567. Our FRACTAL phase IIa trial (ISRCTN 18402242) is presently investigating the safety and tolerability of intravenous infusion followed by oral administration of KAND567 versus placebo in 70 patients with early presenting anterior ST-elevation myocardial infarction (STEMI). To support further development in coronary heart disease and heart failure, the effectiveness of anti-inflammatory and cardioprotective effects will be examined in the same patients.
Figure 3.
Soluble fractalkine (CX3CL1) binds to CX3CR1, its G-coupled receptor, resulting in lymphocyte activation adhesion and migration across the endothelium, resulting in tissue damage.
It is believed that inflammation is not only a significant factor in the development of injury, but also in the healing process and the extent of myocardial damage and function loss. Chemokines and their receptors rigorously regulate the activation and migration of immune blood cells following injury. Within 120 min of reperfusion by primary percutaneous coronary intervention (PPCI) in STEMI patients, the chemokine ligand fractalkine is activated on the endothelium and the circulating levels of subsets of T cells and monocytes expressing the fractalkine receptor (CX3CR1) are significantly decreased [44]. Observation of cellular retention across the myocardium (as determined by transcoronary gradients) suggests evidence for infiltration of these cells into cardiovascular tissue [44]. This immune-cell infiltration is associated with a higher risk of complications (microvascular obstruction) and a shorter three-year survival rate [44].
As it is not yet possible to track lymphocyte fate during acute myocardial infarction (MI) in patients, clinical studies have focused on the analysis of lymphocyte numbers in peripheral blood and identification of transmyocardial gradients during the acute phase of reperfusion following PPCI. A temporary decrease in lymphocytes has been identified following reperfusion [44,52]. We measured T cell gradients between the infarct-related artery and the coronary sinus, which drains venous blood from the anterior myocardium, to determine whether T cells were sequestered in the infarcted myocardium of patients [44]. A notable drop in total T cells (−3.8% ± 0.8%, p = 0.028) was noted in cases of anterior STEMI across this gradient when samples were collected within 45 min but not beyond this point, surmising that T cell sequestration occurs early following reperfusion.
4.3. Fractalkine Signalling in Heart Failure
In a study conducted by Nakayama and colleagues, higher myocardial immune activation was found to be associated with a poor prognosis for DCM [8]. Patients with higher counts of CD3−, CD68−, and CD163-positive infiltrating cells had substantially worse outcomes (p = 0.007, p = 0.011, and p = 0.022, respectively). The association between M2 macrophages and collagen synthesis suggests that ventricular remodelling in DCM may be associated with the phenotypic polarisation of macrophages towards M2. CX3CR1 and CX3CL1 expression is consistently upregulated in cardiac tissue from heart failure patients with various aetiologies, including end-stage DCM, according to our unpublished data. CX3CR1 and CX3CL1 gene expression has been found to be elevated in both DCM and ICM patient cardiac tissue [32]. Richter and coworkers measured the plasma levels of CX3CL1 in 349 patients with advanced systolic heart failure [53]. During a median of 5 years of follow-up, 56% of patients died. Fractalkine was a significant predictor of overall mortality (p < 0.001), with a hazard ratio of 2.8 (95% confidence interval: 1.95–3.95) for the third tertile relative to the first. This association remained significant after multivariable adjustment for demographics, clinical predictive factors, and N-terminal pro-B-type natriuretic peptide (NT-proBNP, p = 0.008). The predictive value of fractalkine did not differ significantly (p = 0.79) between patients with ischemic and non-ischemic causes of HF. Moreover, fractalkine was an independent indicator of cardiovascular mortality. Fractalkine levels in patients treated with an angiotensin-converting enzyme inhibitor were significantly reduced. The authors conclude that pro-inflammatory and immunomodulatory circulating fractalkine is an independent predictor of mortality in patients with advanced heart failure. In addition to NT-proBNP, fractalkine improves risk prediction, which may aid in the identification of high-risk patients requiring specialised care [53].
5. Link between CX3CR1 and Cytomegalovirus
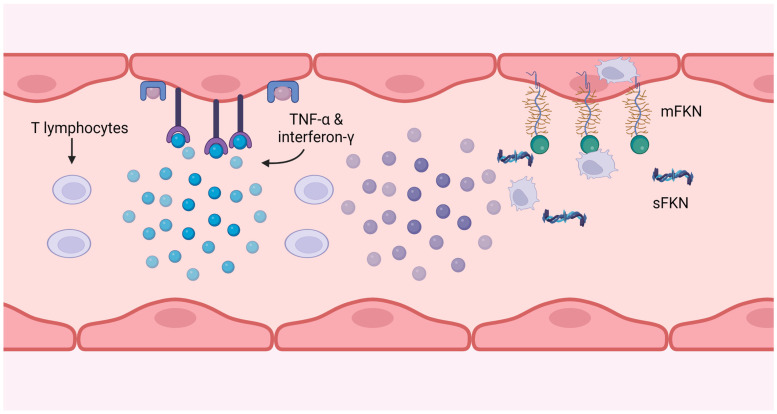
Cytomegalovirus (CMV) is a pervasive herpes virus with a seroprevalence of greater than 60% in individuals aged 50 and older in the majority of studies [54] and 85% in those aged 80 and older [46]. CMV causes an asymptomatic or moderate initial response in immunocompetent hosts, but it is never eliminated from the body, resulting in lifelong latent infection with the possibility of reactivation [54]. Seropositivity for CMV induces significant changes in the T cell composition of the host. CMV-specific CD8+ and, to a lesser extent, CD4+ memory T cells, particularly TEMRA cells, account for a disproportionately high proportion of total T cells; this phenomenon is known as memory inflation [55,56,57]. There is evidence that memory expansion in the CD8 compartment is greater in older patients, indicating that the CMV-specific memory population continues to expand with age, but this is less evident and has been less thoroughly studied in CD4 T cells [48]. Memory inflation may be an adaptive response to suppress reactivations [58], and whether this unbalanced expansion impedes the immune system’s function remains debatable, as some contend that the CD8 compartment is sufficiently plastic to permit expansion of one phenotype without disruption of others [59]. In the presence of viral antigen, CMV-specific CD4+ T cells have been shown to cause endothelial injury, with more damage occurring in donors with higher frequencies of CMV-specific CD4+ T cells [60,61,62]. This injury is caused by the release of IFN-g and TNF-α by T cells at sufficient levels to induce endothelial cell induction of fractalkine, thereby attracting natural killer (NK) cells and monocyte-macrophages, as depicted in Figure 4. The same group demonstrated that inhibiting the fractalkine receptor, CX3CR1, mitigated this endothelial injury.
Figure 4.
CMV-specific T cells produce interferon-g and TNF-α, which induce fractalkine expression on the endothelium. The soluble form attracts monocytes and NK cells, and the membrane-bound form catches and internalises them, removing them from the circulation.
CMV infection transforms the T cell population towards a cytotoxic phenotype, as evidenced by the upregulation of transcription factors such as Hobit and the expression of granzymes A and B, perforin, and CX3CR1 [63]. Granzymes and perforin are used to lyse target cells, while fractalkine, a chemoattractant and adhesion molecule, is the CX3CR1 ligand. These cells were found to strongly express perforin, granzyme B, IFN-γ and TNF-α [44]. In CMV-seropositive patients, the proportion of T cells that are CD27− is also considerably increased, and these cells were found to strongly express perforin, granzyme B, IFN-γ and TNF-α. This cytotoxic T cells population expands once viral load declines substantially and the infection becomes latent; it proliferates significantly more in response to CMV antigen than to other recall antigens; thus, it is CMV specific [45]. While the preponderance of CD4+CD27− T cells in CMV-seropositive donors were CMV specific, a significantly higher proportion of CD4+CD27− T cells were specific to other herpes virus than CMV-seronegative patients [46]. This suggests that CMV seropositivity influence extends beyond CMV-specific T cells.
CMV infection has been linked to cardiovascular disease in numerous studies, as Ji and colleagues reviewed in detail in 2012 [64]. More recently, a prospective epidemiological study in octogenarians conducted by our group revealed that CMV seropositivity and higher levels of senescent T cells such as TEMRA cells predicted worse cardiovascular outcomes [46]. Together, these factors contribute to a potential pathway in which CMV-seropositive individuals have large numbers of cytotoxic CMV-specific T cells, disproportionately TEMRA cells, and these cells respond to occasional CMV antigen presentation by inducing endothelial inflammation, thereby predisposing patients to cardiovascular disease. Additionally, relevant, animal models have demonstrated that monocytes migrate in significant numbers from the spleen to the myocardium following MI [65,66]. Monocytes are a major reservoir of CMV during latent infection [67,68], and their presence in the myocardium after an infarct could provide CMV antigen to initiate a similar pathway to that proposed above, which prolongs the inflammatory response after MI in CMV-seropositive patients, resulting in adverse remodelling.
The cardiovascular hazards associated with CMV seropositivity remain debatable. Case–control studies, such as that conducted by Siscovick [69], have failed to identify a correlation between CMV IgG and the onset of cardiovascular disease. As CMV and cardiovascular disease are so prevalent, its study design likely lacked the ability to identify this association, as acknowledged by the authors. Due to the high prevalence of CMV, a 2017 meta-analysis of prospective epidemiological studies estimated a 7–38% increased relative risk of cardiovascular disease in seropositive patients, which the authors estimated would account for 13% of the total burden of cardiovascular disease [70]. However, the effect of CMV on ventricular remodelling after STEMI has not been thoroughly studied. In a small pilot study, we demonstrated that CMV-seropositive patients exhibit worse remodelling on CMR, with ventricular dilatation 12 weeks after pPCI. This is evidence that latent CMV infection promotes a clinically significant adverse response to STEMI and pPCI, although the observed effect was small and it is imperative that this association be investigated in larger groups of patients followed for extended periods of time. A significant association after only 12 weeks may portend a significant decline in function over the course of years. Evidence from mouse models of myocardial infarction and reperfusion suggests that an abnormal inflammatory response results in detrimental remodelling. The Frangiogiannis group [71] demonstrated that CCR5 knockout mice are incapable of recruiting regulatory T cells to the infarcted myocardium and exhibit poorer remodelling, whereas Cochain and colleagues demonstrated that the decoy receptor D6, which internalises and deactivates pro-inflammatory chemokines, is required for healthy remodelling in the infarcted murine heart [72]. FoxP3−CD4+ T cells substantially contribute to age-related myocardial inflammation in mice, as demonstrated by our collaborators [73]. Further research revealed that CMV-seropositive patients exhibit evidence of accelerated immune ageing following myocardial infarction, which appear to be related to impaired myocardial healing [74,75].
6. Conclusions
The evidence base and existing literature suggest that upregulation of fractalkine/CX3CR1, together with CMV seropositivity and consequent migration of immune and inflammatory cells, is associated with worse outcomes in patients with inflammatory conditions including dilated cardiomyopathy. We hypothesise that dilated cardiomyopathy, whether acquired secondary to infection, autoimmunity, drugs and toxins or familial in origin, may lead to cardiac inflammation and fibrosis, resulting in adverse remodelling through the interplay of these factors. Robust observational studies are required to identify a subgroup of at-risk patients with dilated cardiomyopathy through in-depth phenomapping, genotyping, evaluation of immune status, and viral presence, which in turn may inform future drug trials utilising immune modulators to target this pathway.
Author Contributions
Conceptualisation, I.S.; writing—original draft preparation, V.J., D.A. and I.S.; writing—review and editing, V.J., D.A., S.X.L., V.K.W. and I.S. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data sharing not applicable.
Conflicts of Interest
I.S. and D.A. are supported by research grants from Astra Zeneca (Cambridge, UK), Kancera (Stockholm, Sweden) and previously received funding from TA Sciences (New York, NY, USA).
Funding Statement
I.S. is supported by research grants from the British Heart Foundation and Heart Research UK. The BHF Grant details are PG/23/11093 and PG/22/10788. The HRUK grant number is COV04/21.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Elliott P., Andersson B., Arbustini E., Bilinska Z., Cecchi F., Charron P., Dubourg O., Kuhl U., Maisch B., McKenna W.J., et al. Classification of the cardiomyopathies: A position statement from the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 2008;29:270–276. doi: 10.1093/eurheartj/ehm342. [DOI] [PubMed] [Google Scholar]
- 2.Lozano R., Naghavi M., Foreman K., Lim S., Shibuya K., Aboyans V., Abraham J., Adair T., Aggarwal R., Ahn S.Y., et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2095–2128. doi: 10.1016/S0140-6736(12)61728-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schultheiss H.P., Fairweather D., Caforio A.L.P., Escher F., Hershberger R.E., Lipshultz S.E., Liu P.P., Matsumori A., Mazzanti A., McMurray J., et al. Dilated cardiomyopathy. Nat. Rev. Dis. Primers. 2019;5:32. doi: 10.1038/s41572-019-0084-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Epelman S., Liu P.P., Mann D.L. Role of innate and adaptive immune mechanisms in cardiac injury and repair. Nat. Rev. Immunol. 2015;15:117–129. doi: 10.1038/nri3800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Caforio A.L., Pankuweit S., Arbustini E., Basso C., Gimeno-Blanes J., Felix S.B., Fu M., Helio T., Heymans S., Jahns R., et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: A position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 2013;34:2636–2648. doi: 10.1093/eurheartj/eht210. [DOI] [PubMed] [Google Scholar]
- 6.Kuhl U., Pauschinger M., Seeberg B., Lassner D., Noutsias M., Poller W., Schultheiss H.P. Viral persistence in the myocardium is associated with progressive cardiac dysfunction. Circulation. 2005;112:1965–1970. doi: 10.1161/CIRCULATIONAHA.105.548156. [DOI] [PubMed] [Google Scholar]
- 7.Kuhl U., Pauschinger M., Noutsias M., Seeberg B., Bock T., Lassner D., Poller W., Kandolf R., Schultheiss H.P. High prevalence of viral genomes and multiple viral infections in the myocardium of adults with “idiopathic” left ventricular dysfunction. Circulation. 2005;111:887–893. doi: 10.1161/01.CIR.0000155616.07901.35. [DOI] [PubMed] [Google Scholar]
- 8.Nakayama T., Sugano Y., Yokokawa T., Nagai T., Matsuyama T.A., Ohta-Ogo K., Ikeda Y., Ishibashi-Ueda H., Nakatani T., Ohte N., et al. Clinical impact of the presence of macrophages in endomyocardial biopsies of patients with dilated cardiomyopathy. Eur. J. Heart Fail. 2017;19:490–498. doi: 10.1002/ejhf.767. [DOI] [PubMed] [Google Scholar]
- 9.Verdonschot J.A.J., Merlo M., Dominguez F., Wang P., Henkens M., Adriaens M.E., Hazebroek M.R., Mase M., Escobar L.E., Cobas-Paz R., et al. Phenotypic clustering of dilated cardiomyopathy patients highlights important pathophysiological differences. Eur. Heart J. 2021;42:162–174. doi: 10.1093/eurheartj/ehaa841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Herman D.S., Lam L., Taylor M.R., Wang L., Teekakirikul P., Christodoulou D., Conner L., DePalma S.R., McDonough B., Sparks E., et al. Truncations of titin causing dilated cardiomyopathy. N. Engl. J. Med. 2012;366:619–628. doi: 10.1056/NEJMoa1110186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Comarmond C., Cacoub P. Myocarditis in auto-immune or auto-inflammatory diseases. Autoimmun. Rev. 2017;16:811–816. doi: 10.1016/j.autrev.2017.05.021. [DOI] [PubMed] [Google Scholar]
- 12.Merken J., Hazebroek M., Van Paassen P., Verdonschot J., Van Empel V., Knackstedt C., Abdul Hamid M., Seiler M., Kolb J., Hoermann P., et al. Immunosuppressive Therapy Improves Both Short- and Long-Term Prognosis in Patients with Virus-Negative Nonfulminant Inflammatory Cardiomyopathy. Circ. Heart Fail. 2018;11:e004228. doi: 10.1161/CIRCHEARTFAILURE.117.004228. [DOI] [PubMed] [Google Scholar]
- 13.Scheinecker C., Goschl L., Bonelli M. Treg cells in health and autoimmune diseases: New insights from single cell analysis. J. Autoimmun. 2020;110:102376. doi: 10.1016/j.jaut.2019.102376. [DOI] [PubMed] [Google Scholar]
- 14.Eggenhuizen P.J., Ng B.H., Ooi J.D. Treg Enhancing Therapies to Treat Autoimmune Diseases. Int. J. Mol. Sci. 2020;21:7015. doi: 10.3390/ijms21197015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Olejniczak M., Schwartz M., Webber E., Shaffer A., Perry T.E. Viral Myocarditis-Incidence, Diagnosis and Management. J. Cardiothorac. Vasc. Anesth. 2020;34:1591–1601. doi: 10.1053/j.jvca.2019.12.052. [DOI] [PubMed] [Google Scholar]
- 16.Pollack A., Kontorovich A.R., Fuster V., Dec G.W. Viral myocarditis–diagnosis, treatment options, and current controversies. Nat. Rev. Cardiol. 2015;12:670–680. doi: 10.1038/nrcardio.2015.108. [DOI] [PubMed] [Google Scholar]
- 17.Han L.N., He S., Wang Y.T., Yang L.M., Liu S.Y., Zhang T. Advances in monoclonal antibody application in myocarditis. J. Zhejiang Univ. Sci. B. 2013;14:676–687. doi: 10.1631/jzus.BQICC711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jensen L.D., Marchant D.J. Emerging pharmacologic targets and treatments for myocarditis. Pharmacol. Ther. 2016;161:40–51. doi: 10.1016/j.pharmthera.2016.03.006. [DOI] [PubMed] [Google Scholar]
- 19.Curnock A.P., Bossi G., Kumaran J., Bawden L.J., Figueiredo R., Tawar R., Wiseman K., Henderson E., Hoong S.J., Gonzalez V., et al. Cell-targeted PD-1 agonists that mimic PD-L1 are potent T cell inhibitors. JCI Insight. 2021;6:e152468. doi: 10.1172/jci.insight.152468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Drobni Z.D., Zafar A., Zubiri L., Zlotoff D.A., Alvi R.M., Lee C., Hartmann S., Gilman H.K., Villani A.C., Nohria A., et al. Decreased Absolute Lymphocyte Count and Increased Neutrophil/Lymphocyte Ratio with Immune Checkpoint Inhibitor-Associated Myocarditis. J. Am. Heart Assoc. 2020;9:e018306. doi: 10.1161/JAHA.120.018306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhang L., Awadalla M., Mahmood S.S., Nohria A., Hassan M.Z.O., Thuny F., Zlotoff D.A., Murphy S.P., Stone J.R., Golden D.L.A., et al. Cardiovascular magnetic resonance in immune checkpoint inhibitor-associated myocarditis. Eur. Heart J. 2020;41:1733–1743. doi: 10.1093/eurheartj/ehaa051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ganatra S., Neilan T.G. Immune Checkpoint Inhibitor-Associated Myocarditis. Oncologist. 2018;23:879–886. doi: 10.1634/theoncologist.2018-0130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Palaskas N., Lopez-Mattei J., Durand J.B., Iliescu C., Deswal A. Immune Checkpoint Inhibitor Myocarditis: Pathophysiological Characteristics, Diagnosis, and Treatment. J. Am. Heart Assoc. 2020;9:e013757. doi: 10.1161/JAHA.119.013757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Johnson D.B., Balko J.M., Compton M.L., Chalkias S., Gorham J., Xu Y., Hicks M., Puzanov I., Alexander M.R., Bloomer T.L., et al. Fulminant Myocarditis with Combination Immune Checkpoint Blockade. N. Engl. J. Med. 2016;375:1749–1755. doi: 10.1056/NEJMoa1609214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Garton K.J., Gough P.J., Blobel C.P., Murphy G., Greaves D.R., Dempsey P.J., Raines E.W. Tumor necrosis factor-alpha-converting enzyme (ADAM17) mediates the cleavage and shedding of fractalkine (CX3CL1) J. Biol. Chem. 2001;276:37993–38001. doi: 10.1074/jbc.M106434200. [DOI] [PubMed] [Google Scholar]
- 26.Goda S., Imai T., Yoshie O., Yoneda O., Inoue H., Nagano Y., Okazaki T., Imai H., Bloom E.T., Domae N., et al. CX3C-chemokine, fractalkine-enhanced adhesion of THP-1 cells to endothelial cells through integrin-dependent and -independent mechanisms. J. Immunol. 2000;164:4313–4320. doi: 10.4049/jimmunol.164.8.4313. [DOI] [PubMed] [Google Scholar]
- 27.Imai T., Hieshima K., Haskell C., Baba M., Nagira M., Nishimura M., Kakizaki M., Takagi S., Nomiyama H., Schall T.J., et al. Identification and molecular characterization of fractalkine receptor CX3CR1, which mediates both leukocyte migration and adhesion. Cell. 1997;91:521–530. doi: 10.1016/S0092-8674(00)80438-9. [DOI] [PubMed] [Google Scholar]
- 28.Kanazawa N., Nakamura T., Tashiro K., Muramatsu M., Morita K., Yoneda K., Inaba K., Imamura S., Honjo T. Fractalkine and macrophage-derived chemokine: T cell-attracting chemokines expressed in T cell area dendritic cells. Eur. J. Immunol. 1999;29:1925–1932. doi: 10.1002/(SICI)1521-4141(199906)29:06<1925::AID-IMMU1925>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- 29.Patel A., Jagadesham V.P., Porter K.E., Scott D.J., Carding S.R. Characterisation of fractalkine/CX3CL1 and fractalkine receptor (CX3CR1) expression in abdominal aortic aneurysm disease. Eur. J. Vasc. Endovasc. Surg. 2008;36:20–27. doi: 10.1016/j.ejvs.2008.01.014. [DOI] [PubMed] [Google Scholar]
- 30.Nishimura M., Umehara H., Nakayama T., Yoneda O., Hieshima K., Kakizaki M., Dohmae N., Yoshie O., Imai T. Dual functions of fractalkine/CX3C ligand 1 in trafficking of perforin+/granzyme B+ cytotoxic effector lymphocytes that are defined by CX3CR1 expression. J. Immunol. 2002;168:6173–6180. doi: 10.4049/jimmunol.168.12.6173. [DOI] [PubMed] [Google Scholar]
- 31.Yoneda O., Imai T., Goda S., Inoue H., Yamauchi A., Okazaki T., Imai H., Yoshie O., Bloom E.T., Domae N., et al. Fractalkine-mediated endothelial cell injury by NK cells. J. Immunol. 2000;164:4055–4062. doi: 10.4049/jimmunol.164.8.4055. [DOI] [PubMed] [Google Scholar]
- 32.Husberg C., Nygard S., Finsen A.V., Damas J.K., Frigessi A., Oie E., Waehre A., Gullestad L., Aukrust P., Yndestad A., et al. Cytokine expression profiling of the myocardium reveals a role for CX3CL1 (fractalkine) in heart failure. J. Mol. Cell Cardiol. 2008;45:261–269. doi: 10.1016/j.yjmcc.2008.05.009. [DOI] [PubMed] [Google Scholar]
- 33.Combadière C., Potteaux S., Gao J.L., Esposito B., Casanova S., Lee E.J., Debré P., Tedgui A., Murphy P.M., Mallat Z. Decreased atherosclerotic lesion formation in CX3CR1/apolipoprotein E double knockout mice. Circulation. 2003;107:1009–1016. doi: 10.1161/01.CIR.0000057548.68243.42. [DOI] [PubMed] [Google Scholar]
- 34.Lesnik P., Haskell C.A., Charo I.F. Decreased atherosclerosis in CX3CR1-/- mice reveals a role for fractalkine in atherogenesis. J. Clin. Investig. 2003;111:333–340. doi: 10.1172/JCI15555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Apostolakis S., Amanatidou V., Papadakis E.G., Spandidos D.A. Genetic diversity of CX3CR1 gene and coronary artery disease: New insights through a meta-analysis. Atherosclerosis. 2009;207:8–15. doi: 10.1016/j.atherosclerosis.2009.03.044. [DOI] [PubMed] [Google Scholar]
- 36.Apostolakis S., Baritaki S., Kochiadakis G.E., Igoumenidis N.E., Panutsopulos D., Spandidos D.A. Effects of polymorphisms in chemokine ligands and receptors on susceptibility to coronary artery disease. Thromb. Res. 2007;119:63–71. doi: 10.1016/j.thromres.2005.12.016. [DOI] [PubMed] [Google Scholar]
- 37.Chan C.C., Tuo J., Bojanowski C.M., Csaky K.G., Green W.R. Detection of CX3CR1 single nucleotide polymorphism and expression on archived eyes with age-related macular degeneration. Histol. Histopathol. 2005;20:857–863. doi: 10.14670/HH-20.857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ghilardi G., Biondi M.L., Turri O., Guagnellini E., Scorza R. Internal carotid artery occlusive disease and polymorphisms of fractalkine receptor CX3CR1: A genetic risk factor. Stroke. 2004;35:1276–1279. doi: 10.1161/01.STR.0000128528.56009.d4. [DOI] [PubMed] [Google Scholar]
- 39.Hattori H., Ito D., Tanahashi N., Murata M., Saito I., Watanabe K., Suzuki N. T280M and V249I polymorphisms of fractalkine receptor CX3CR1 and ischemic cerebrovascular disease. Neurosci. Lett. 2005;374:132–135. doi: 10.1016/j.neulet.2004.10.042. [DOI] [PubMed] [Google Scholar]
- 40.Kimouli M., Miyakis S., Georgakopoulos P., Neofytou E., Achimastos A.D., Spandidos D.A. Polymorphisms of fractalkine receptor CX3CR1 gene in patients with symptomatic and asymptomatic carotid artery stenosis. J. Atheroscler. Thromb. 2009;16:604–610. doi: 10.5551/jat.1107. [DOI] [PubMed] [Google Scholar]
- 41.McDermott D.H., Halcox J.P., Schenke W.H., Waclawiw M.A., Merrell M.N., Epstein N., Quyyumi A.A., Murphy P.M. Association between polymorphism in the chemokine receptor CX3CR1 and coronary vascular endothelial dysfunction and atherosclerosis. Circ. Res. 2001;89:401–407. doi: 10.1161/hh1701.095642. [DOI] [PubMed] [Google Scholar]
- 42.Norata G.D., Garlaschelli K., Ongari M., Raselli S., Grigore L., Catapano A.L. Effects of fractalkine receptor variants on common carotid artery intima-media thickness. Stroke. 2006;37:1558–1561. doi: 10.1161/01.STR.0000221803.16897.22. [DOI] [PubMed] [Google Scholar]
- 43.Wu J., Yin R.X., Lin Q.Z., Guo T., Shi G.Y., Sun J.Q., Shen S.W., Li Q. Two polymorphisms in the Fractalkine receptor CX3CR1 gene influence the development of atherosclerosis: A meta-analysis. Dis. Markers. 2014;2014:913678. doi: 10.1155/2014/913678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Boag S.E., Das R., Shmeleva E.V., Bagnall A., Egred M., Howard N., Bennaceur K., Zaman A., Keavney B., Spyridopoulos I. T lymphocytes and fractalkine contribute to myocardial ischemia/reperfusion injury in patients. J. Clin. Investig. 2015;125:3063–3076. doi: 10.1172/JCI80055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Umehara H., Bloom E.T., Okazaki T., Nagano Y., Yoshie O., Imai T. Fractalkine in vascular biology: From basic research to clinical disease. Arterioscler. Thromb. Vasc. Biol. 2004;24:34–40. doi: 10.1161/01.ATV.0000095360.62479.1F. [DOI] [PubMed] [Google Scholar]
- 46.Spyridopoulos I., Martin-Ruiz C., Hilkens C., Yadegarfar M.E., Isaacs J., Jagger C., Kirkwood T., von Zglinicki T. CMV seropositivity and T-cell senescence predict increased cardiovascular mortality in octogenarians: Results from the Newcastle 85+ study. Aging Cell. 2016;15:389–392. doi: 10.1111/acel.12430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Strindhall J., Skog M., Ernerudh J., Bengner M., Lofgren S., Matussek A., Nilsson B.O., Wikby A. The inverted CD4/CD8 ratio and associated parameters in 66-year-old individuals: The Swedish HEXA immune study. Age. 2013;35:985–991. doi: 10.1007/s11357-012-9400-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Pachnio A., Ciaurriz M., Begum J., Lal N., Zuo J., Beggs A., Moss P. Cytomegalovirus Infection Leads to Development of High Frequencies of Cytotoxic Virus-Specific CD4+ T Cells Targeted to Vascular Endothelium. PLoS Pathog. 2016;12:e1005832. doi: 10.1371/journal.ppat.1005832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kuijpers T.W., Vossen M.T., Gent M.R., Davin J.C., Roos M.T., Wertheim-van Dillen P.M., Weel J.F., Baars P.A., van Lier R.A. Frequencies of circulating cytolytic, CD45RA+CD27−, CD8+ T lymphocytes depend on infection with CMV. J. Immunol. 2003;170:4342–4348. doi: 10.4049/jimmunol.170.8.4342. [DOI] [PubMed] [Google Scholar]
- 50.Pekalski M., Jenkinson S.E., Willet J.D., Poyner E.F., Alhamidi A.H., Robertson H., Ali S., Kirby J.A. Renal allograft rejection: Examination of delayed differentiation of Treg and Th17 effector T cells. Immunobiology. 2013;218:303–310. doi: 10.1016/j.imbio.2012.05.014. [DOI] [PubMed] [Google Scholar]
- 51.Willet J.D., Pichitsiri W., Jenkinson S.E., Brain J.G., Wood K., Alhasan A.A., Spielhofer J., Robertson H., Ali S., Kirby J.A. Kidney transplantation: Analysis of the expression and T cell-mediated activation of latent TGF-beta. J. Leukoc. Biol. 2013;93:471–478. doi: 10.1189/jlb.0712324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.de Dios E., Rios-Navarro C., Perez-Sole N., Gavara J., Marcos-Garces V., Rodriguez E., Carratala A., Forner M.J., Navarro J., Blasco M.L., et al. Similar Clinical Course and Significance of Circulating Innate and Adaptive Immune Cell Counts in STEMI and COVID-19. J. Clin. Med. 2020;9:3484. doi: 10.3390/jcm9113484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Richter B., Koller L., Hohensinner P.J., Rychli K., Zorn G., Goliasch G., Berger R., Mortl D., Maurer G., Huber K., et al. Fractalkine is an independent predictor of mortality in patients with advanced heart failure. Thromb. Haemost. 2012;108:1220–1227. doi: 10.1160/TH12-03-0195. [DOI] [PubMed] [Google Scholar]
- 54.Weltevrede M., Eilers R., de Melker H.E., van Baarle D. Cytomegalovirus persistence and T-cell immunosenescence in people aged fifty and older: A systematic review. Exp. Gerontol. 2016;77:87–95. doi: 10.1016/j.exger.2016.02.005. [DOI] [PubMed] [Google Scholar]
- 55.Waller E.C., Day E., Sissons J.G., Wills M.R. Dynamics of T cell memory in human cytomegalovirus infection. Med. Microbiol. Immunol. 2008;197:83–96. doi: 10.1007/s00430-008-0082-5. [DOI] [PubMed] [Google Scholar]
- 56.Pourgheysari B., Khan N., Best D., Bruton R., Nayak L., Moss P.A. The cytomegalovirus-specific CD4+ T-cell response expands with age and markedly alters the CD4+ T-cell repertoire. J. Virol. 2007;81:7759–7765. doi: 10.1128/JVI.01262-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Munks M.W., Cho K.S., Pinto A.K., Sierro S., Klenerman P., Hill A.B. Four distinct patterns of memory CD8 T cell responses to chronic murine cytomegalovirus infection. J. Immunol. 2006;177:450–458. doi: 10.4049/jimmunol.177.1.450. [DOI] [PubMed] [Google Scholar]
- 58.Khan N., Hislop A., Gudgeon N., Cobbold M., Khanna R., Nayak L., Rickinson A.B., Moss P.A. Herpesvirus-specific CD8 T cell immunity in old age: Cytomegalovirus impairs the response to a coresident EBV infection. J. Immunol. 2004;173:7481–7489. doi: 10.4049/jimmunol.173.12.7481. [DOI] [PubMed] [Google Scholar]
- 59.Goronzy J.J., Weyand C.M. Successful and Maladaptive T Cell Aging. Immunity. 2017;46:364–378. doi: 10.1016/j.immuni.2017.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Bolovan-Fritts C.A., Trout R.N., Spector S.A. High T-cell response to human cytomegalovirus induces chemokine-mediated endothelial cell damage. Blood. 2007;110:1857–1863. doi: 10.1182/blood-2007-03-078881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bolovan-Fritts C.A., Trout R.N., Spector S.A. Human cytomegalovirus-specific CD4+-T-cell cytokine response induces fractalkine in endothelial cells. J. Virol. 2004;78:13173–13181. doi: 10.1128/JVI.78.23.13173-13181.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Bolovan-Fritts C.A., Spector S.A. Endothelial damage from cytomegalovirus-specific host immune response can be prevented by targeted disruption of fractalkine-CX3CR1 interaction. Blood. 2008;111:175–182. doi: 10.1182/blood-2007-08-107730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hertoghs K.M., Moerland P.D., van Stijn A., Remmerswaal E.B., Yong S.L., van de Berg P.J., van Ham S.M., Baas F., ten Berge I.J., van Lier R.A. Molecular profiling of cytomegalovirus-induced human CD8+ T cell differentiation. J. Clin. Investig. 2010;120:4077–4090. doi: 10.1172/JCI42758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ji Y.N., An L., Zhan P., Chen X.H. Cytomegalovirus infection and coronary heart disease risk: A meta-analysis. Mol. Biol. Rep. 2012;39:6537–6546. doi: 10.1007/s11033-012-1482-6. [DOI] [PubMed] [Google Scholar]
- 65.Marinkovic G., Koenis D.S., de Camp L., Jablonowski R., Graber N., de Waard V., de Vries C.J., Goncalves I., Nilsson J., Jovinge S., et al. S100A9 Links Inflammation and Repair in Myocardial Infarction. Circ. Res. 2020;127:664–676. doi: 10.1161/CIRCRESAHA.120.315865. [DOI] [PubMed] [Google Scholar]
- 66.Sun Y., Pinto C., Camus S., Duval V., Alayrac P., Zlatanova I., Loyer X., Vilar J., Lemitre M., Levoye A., et al. Splenic Marginal Zone B Lymphocytes Regulate Cardiac Remodeling After Acute Myocardial Infarction in Mice. J. Am. Coll. Cardiol. 2022;79:632–647. doi: 10.1016/j.jacc.2021.11.051. [DOI] [PubMed] [Google Scholar]
- 67.Stanier P., Taylor D.L., Kitchen A.D., Wales N., Tryhorn Y., Tyms A.S. Persistence of cytomegalovirus in mononuclear cells in peripheral blood from blood donors. BMJ. 1989;299:897–898. doi: 10.1136/bmj.299.6704.897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Nikitina E., Larionova I., Choinzonov E., Kzhyshkowska J. Monocytes and Macrophages as Viral Targets and Reservoirs. Int. J. Mol. Sci. 2018;19:2821. doi: 10.3390/ijms19092821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Siscovick D.S., Schwartz S.M., Corey L., Grayston J.T., Ashley R., Wang S.P., Psaty B.M., Tracy R.P., Kuller L.H., Kronmal R.A. Chlamydia pneumoniae, herpes simplex virus type 1, and cytomegalovirus and incident myocardial infarction and coronary heart disease death in older adults: The Cardiovascular Health Study. Circulation. 2000;102:2335–2340. doi: 10.1161/01.CIR.102.19.2335. [DOI] [PubMed] [Google Scholar]
- 70.Wang H., Peng G., Bai J., He B., Huang K., Hu X., Liu D. Cytomegalovirus Infection and Relative Risk of Cardiovascular Disease (Ischemic Heart Disease, Stroke, and Cardiovascular Death): A Meta-Analysis of Prospective Studies Up to 2016. J. Am. Heart Assoc. 2017;6:e005025. doi: 10.1161/JAHA.116.005025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Dobaczewski M., Xia Y., Bujak M., Gonzalez-Quesada C., Frangogiannis N.G. CCR5 signaling suppresses inflammation and reduces adverse remodeling of the infarcted heart, mediating recruitment of regulatory T cells. Am. J. Pathol. 2010;176:2177–2187. doi: 10.2353/ajpath.2010.090759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Cochain C., Auvynet C., Poupel L., Vilar J., Dumeau E., Richart A., Recalde A., Zouggari Y., Yin K.Y., Bruneval P., et al. The chemokine decoy receptor D6 prevents excessive inflammation and adverse ventricular remodeling after myocardial infarction. Arterioscler. Thromb. Vasc. Biol. 2012;32:2206–2213. doi: 10.1161/ATVBAHA.112.254409. [DOI] [PubMed] [Google Scholar]
- 73.Ramos G.C., van den Berg A., Nunes-Silva V., Weirather J., Peters L., Burkard M., Friedrich M., Pinnecker J., Abesser M., Heinze K.G., et al. Myocardial aging as a T-cell-mediated phenomenon. Proc. Natl. Acad. Sci. USA. 2017;114:E2420–E2429. doi: 10.1073/pnas.1621047114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Hoffmann J., Shmeleva E.V., Boag S.E., Fiser K., Bagnall A., Murali S., Dimmick I., Pircher H., Martin-Ruiz C., Egred M., et al. Myocardial ischemia and reperfusion leads to transient CD8 immune deficiency and accelerated immunosenescence in CMV-seropositive patients. Circ. Res. 2015;116:87–98. doi: 10.1161/CIRCRESAHA.116.304393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Spyridopoulos I., Hoffmann J., Aicher A., Brummendorf T.H., Doerr H.W., Zeiher A.M., Dimmeler S. Accelerated telomere shortening in leukocyte subpopulations of patients with coronary heart disease: Role of cytomegalovirus seropositivity. Circulation. 2009;120:1364–1372. doi: 10.1161/CIRCULATIONAHA.109.854299. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing not applicable.