Abstract
COVID-19, caused by SARS-CoV-2, has spread globally. Coinfection with other endemic viruses is likely to complicate the clinical presentation and outcome. Information on clinical manifestations and management strategies on COVID-19 coinfection with endemic diseases in children is yet to evolve. The risk of dengue infection exists in 129 countries and it is endemic in more than 100 countries. The SARS-CoV-2 pandemic might overlap with the dengue epidemics in tropical countries. We report the first paediatric case to the best of our knowledge of COVID-19 encephalitis with dengue shock syndrome. This clinical syndrome could be attributed to serological cross-reactivity, incidental coinfection or perhaps a warning for dengue-endemic regions to face the unique challenge of differentiating and managing two disease entities together. Enhanced understanding of potential COVID-19 and dengue coinfection warrants immediate attention of researchers and international health policy makers.
Keywords: global health, influenza, tropical medicine (infectious disease), paediatric intensive care, coma and raised intracranial pressure
Background
COVID-19 continues to spread worldwide since December 2019. The symptoms of SARS-CoV-2 infection vary widely, from asymptomatic disease to multisystem organ failure.1 SARS-CoV-2 pandemic is likely to overlap with the dengue epidemics in tropical countries. Due to overlapping clinical and laboratory features, it may be difficult to distinguish dengue from COVID-19.2 One currently commonly used diagnostic test for COVID-19 is reverse transcriptase PCR (RT-PCR) based detection of nasopharyngeal SARS CoV-2 RNA. Laboratory diagnosis of dengue during the febrile phase is established directly by detection of virus expressed soluble non-structural protein 1 (NS1 antigen) or/and dengue-specific IgM antibodies by means of ELISA.3 The reported specificity of dengue NS1 antigen test is 87.4%–97.7% during the febrile phase.4 To the best of our knowledge, there is no paediatric case reported yet with these two coinfections. We describe the first paediatric case of COVID-19 with positive dengue NS1 antigen and IgM antibody who presented to our paediatric intensive care unit with features of encephalitis, raised intracranial pressure (ICP) and shock. The patient’s syndrome could be attributed to incidental coinfection or serological cross-reactivity of dengue with SARS-CoV-2.
Case presentation
A 14-year-old, previously healthy female adolescent belonging to the upper-middle socioeconomic strata was brought to paediatric emergency with high-grade fever, headache and vomiting. The patient was in her usual state of health 6 days previously when she developed low-grade fever, associated with myalgias and headache. Her fever progressively increased to 103°F, associated with a severe, throbbing frontal headache over the next 2–3 days. She also developed nausea, vomiting and generalised pain abdomen. She did not have cough, seizure, urinary symptoms or known history of dengue. Her paediatric quick Sequential Organ Failure Assessment (qSOFA) score was 3 (out of 3) on admission5; she had respiratory distress and hypotensive shock. Therefore, the septic shock management protocol was initiated in the emergency room. Our hospital is a tertiary care centre, situated in Bihar (India) and declared as a dedicated COVID-hospital during the COVID-19 pandemic.
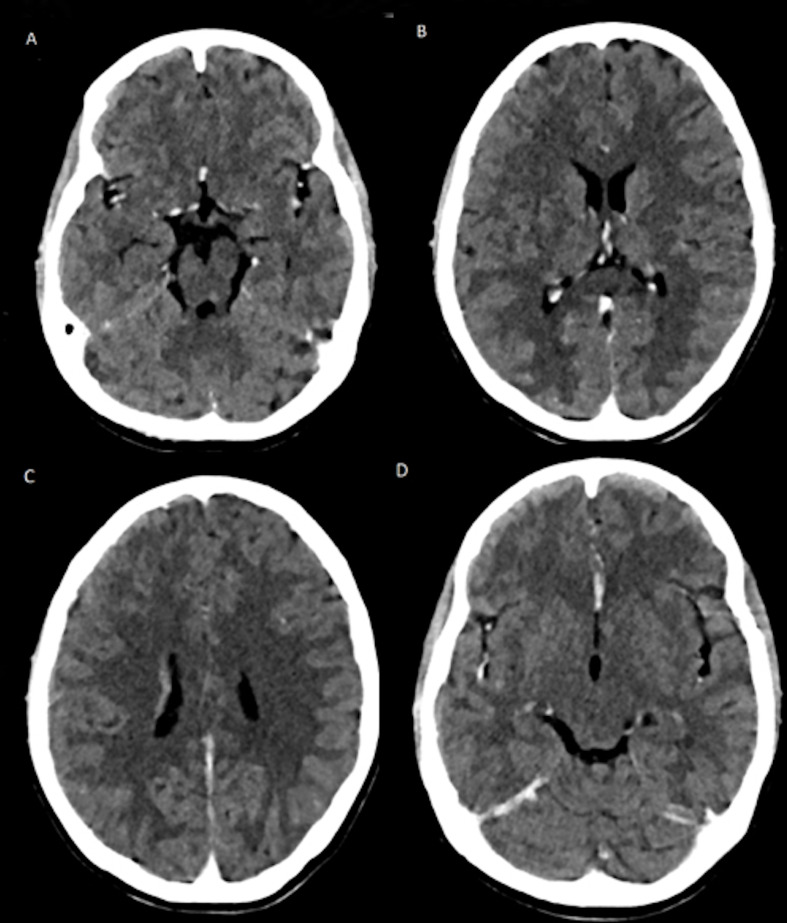
She had tachypnoea (respiratory rate 28 breaths/min), tachycardia (heart rate 120 beats/min), SpO2 of 96%–98% on room air, blood pressure of 88/52 mm Hg, which was below fifth percentile for her age, gender and height. Her Glasgow Coma Scale (GCS) score was 13 (E3V4M6) and her pupils were normal in size and reactive to light. She had brisk deep tendon reflexes and plantar response was extensor on the right side. She had no petechial rash or haemorrhagic manifestations, and her tourniquet test was negative. Her weight was 30 kg (below the third percentile for her age); her height was 150 cm (between the 11th and 25th percentiles for her age); and her body mass index was 13.33 kg/m2 (below the third percentile for her age). She had abdominal tenderness, more on the right hypochondrium. Her liver was palpable 2 cm below the right coastal margin with a span of 14 cm. She was treated for shock due to presumed COVID-19 infection. Echocardiography demonstrated normal cardiac structure and contractility without pericardial effusion, and the inferior vena cava filling was responsive to fluid therapy. She received intravenous fluids and inotropic support, as well as serial echocardiogram monitoring of her fluid status. She had clinical features of raised ICP in the form of low GCS score, headache and vomiting. Her ECG was normal. She was treated for raised ICP ensuring thermoregulation, head elevation to 30°, maintenance of euglycaemia, euvolaemia, stress ulcer prophylaxis and 3% hypertonic saline, along with other supportive care. Empirically, she was given intravenous ceftriaxone and oral azithromycin. As shown in table 1, her renal function test was unremarkable, but she had leucopenia, thrombocytopenia and mild transaminitis. Her clinical presentation with hypotensive shock, low GCS score, low platelets and low white blood cell count in the setting of the COVID-19 pandemic were strong indications towards COVID-19-associated multisystem inflammatory syndrome in children (MIS-C); however, these findings can also be consistent with dengue shock syndrome (DSS) in dengue-endemic zones. Other differential diagnoses included typhoid fever due to Salmonella typhi, leptospirosis and scrub typhus (table 1). She had a high C reactive protein, D-dimer and serum ferritin, suggestive of hyperinflammatory status as seen in COVID-19. Chest X-ray showed reticulonodular opacities in bilateral lung fields without any evidence of pleural effusion. The nasopharyngeal SARS-CoV-2 RNA was detected on PCR testing. Additionally, the dengue NS1 antigen in the serum was also positive. Additional workup to rule out potential causes of encephalitis included malaria, tuberculosis, herpes simplex virus and antibodies for immune encephalitis. Contrast-enhanced CT scan of the brain showed an ill-defined hypodensity in bilateral frontal lobes, right parietal lobe, bilateral temporal lobe, basal ganglia, corpus callosum, mid-brain and pons, suggestive of viral encephalitis (figure 1). The imaging findings supported the findings of COVID-19-associated encephalitis with DSS. For management, fluid therapy, inotropic support and measures to prevent elevated ICP were continued. She was not given any antiviral drug or investigational immunomodulator therapy for COVID-19. Her clinical status improved progressively as indicated by improving SOFA score, modified shock index, Vasoactive Inotrope Score and Modified Rankin Scale in table 2.
Table 1.
Laboratory investigations
| Investigation | Result (on admission) | Normal value |
| Haemoglobin | 113 g/L | 120–150 g/L |
| Platelets | 70×103/mm3 | 150–450×103/mm3 |
| Leucocyte count | 2620/mm3 | 4000–11 000/mm3 |
| Differential leucocyte count N/L/M/E/B (%) | 45.7/50.4/3.1/0.0/0.8 | 40–80/20–40/2–10/1–6/0–1 |
| Serum bilirubin (total) | 0.59 mg/dL | 0.3–1.2 mg/dL |
| Serum bilirubin (direct) | 0.22 mg/dL | <0.3 mg/dL |
| Aspartate aminotransferase | 124.8 IU/L | <31 IU/L |
| Alanine aminotransferase | 54.8 IU/L | 10–28 IU/L |
| Alkaline phosphatase | 125.7 IU/L | 100–290 IU/L |
| Prothrombin time | 14.20 s | <14 s |
| Serum urea | 20.7 mg/dL | 13–43 mg/dL |
| Serum creatinine | 0.47 mg/dL | 0.7–1.3 mg/dL |
| Serum uric acid | 2.13 mg/dL | 3.5–7.2 mg/dL |
| Serum sodium | 137.55 meq/L | 135–145 meq/L |
| Serum potassium | 4.16 meq/L | 3.5–5 meq/L |
| LDH | 1166.24 μ/L | 230–460 μ/L |
| CRP | 58 mg/L | 0–5 mg/L |
| Serum ammonia | 76.22 μmol/L | 10–47 μmol/l |
| D-dimer | 3.05 µg/mL | <0.2 µg/mL |
| Serum ferritin | 614.64 ng/mL | 10–291 ng/mL |
| CSF cells | 5/mm3 | |
| CSF glucose | 70 mg/dL | |
| CSF protein | 30.92 mg/dL | |
| CSF ADA | 3.52 U/L | |
| CSF LDH | 266 U/L | |
| CSF RT-PCR for COVID-19 | Negative | |
| CSF CBNAAT for Mycobacterium tuberculosis | Acid fast Bacilli not demonstrated |
|
| CSF acid-fast bacilli | Absent | |
| CSF IgM–HSV | Negative | |
| CSF bacterial culture | Sterile | |
| Gastric aspirate for AFB | Negative | |
| Blood smear for malarial parasite | Negative | |
| Dengue NS1 antigen* | Positive | |
| Dengue IgM antibody† | Positive | |
| Widal test for Salmonella typhi, rapid test for scrub typhus, Leptospira IgM, hepatitis B surface antigen, hepatitis C antibody test | Negative | |
| Antiaquaporin 4 (NMO) IgG antibodies | Negative | |
| Anti-MOG IgG antibodies | Negative |
*On day 4 and day 11 of illness.
†On day 14 of illness.
ADA, adenosine deaminase; AFB, acid-fast bacilli; B, basophil; CBNAAT, cartridge-based nucleic acid amplification test; CRP, C reactive protein; CSF, cerebrospinal fluid; E, eosinophil; HSV, Herpes simplex virus; L, lymphocyte; LDH, lactate dehydrogenase; M, monocyte; MOG, myelin oligodendrocyte glycoprotein; N, neutrophil; NMO, neuromyelitis optica; RT-PCR, reverse transcriptase PCR.
Figure 1.
Contrast-enhanced CT scan showing ill-defined marked hypodensities in (A). Mid-brain; (B) thalamus, basal ganglia and corpus callosum; (C) bilateral periventricular area; and (D) bilateral frontal lobes.
Table 2.
Clinical status of the present case during the course of PICU stay
| Clinical scores | Day 1 | Day 3 | Day 5 | Day 7 | Day 9 |
| SOFA score/risk of mortality (%) | 7/≤33.3 | 7/≤33.3 | 5/≤33.3 | 2/≤33.3 | 1/≤33.3 |
| Modified shock index | 2.01 | 1.69 | 1.24 | 1.15 | 1.04 |
| Vasoactive Inotrope Score | 30 | 30 | 10 | – | – |
| Modified Rankin Scale | 4 | 4 | 3 | 2 | 1 |
PICU, paediatric intensive care unit; SOFA, Sequential Organ Failure Assessment.
Dengue IgM antibody by ELISA was negative on the 11th day of illness but became positive on the 14th day of illness. Cerebrospinal fluid (CSF) examination, deferred initially due to overt signs of raised ICP, was done during the second week of illness. The CSF profile demonstrated no pleocytosis, normal protein and normal glucose (table 1). CSF RT-PCR for COVID-19 was negative. Neither the CSF dengue antibody nor nucleic acid amplification test could be performed due to lack of testing capacity. Neither blood nor CSF culture demonstrated growth of any organism. Workup for encephalitis, including malaria, tuberculosis, Salmonella typhi, scrub typhus, Leptospira and herpes simplex virus, was negative. CSF for immune encephalitis antibodies (anti-aquaporin 4 IgG antibodies and anti-myelin oligodendrocyte glycoprotein IgG) was also negative (table 1).
Global health problem list
The COVID-19 pandemic continues to spread worldwide, and it is likely to overlap with the dengue epidemics in tropical countries. Due to overlapping clinical and laboratory features, it may be difficult to distinguish dengue from COVID-19.2 The index case is the first paediatric case to the best of our knowledge of COVID-19 encephalitis with DSS. This clinical syndrome could be attributed to serological cross-reactivity, incidental coinfection or perhaps a warning for dengue-endemic regions to face the unique challenge of differentiating and managing two disease entities together. Understanding of potential COVID-19 and dengue coinfection warrants immediate attention of researchers and international health policy makers.
Global health problem analysis
COVID-19, caused by the highly pathogenic SARS-CoV-2, is a global health emergency.6 Most patients experience mild to moderate respiratory illness and recover without any specific treatment. Older people and those with underlying comorbidities are more likely to develop severe disease.7 Paediatric multisystem inflammatory syndrome is a rare life-threatening presentation of COVID-19 which has been recently noted.8–10 Varying presentations of this disease involving different organ systems in children are emerging as the pandemic spreads.9 10
The risk of dengue infection exists in 129 countries and it is endemic in more than 100 countries. Seventy per cent of the actual burden of disease is in Asia.11 One modelling estimate indicates 390 million global dengue virus infections per year (95% CI 284 to 528 million), of which 96 million manifest clinically.12 13 As the COVID-19 pandemic spreads in tropical countries, the risk of coinfection increases. This patient is a resident of Bihar, in the eastern part of India, which is endemic for dengue and where access to health facilities is generally scarce, particularly during the COVID-19 pandemic, due to restricted transportation. Serological cross-reactivity between dengue and Japanese encephalitis (JE) is well known and common in endemic regions, but they can be fairly differentiated on clinicolaboratory and radiological grounds. COVID-19 and dengue fever are difficult to differentiate because they share clinical and laboratory features.2 Yan et al recently described two cases which were initially diagnosed as dengue but later confirmed to be COVID-19.14 It has been recently reported in Brazil and other regions that both viruses spread simultaneously as copandemics.15 Serological cross-reactivity between SARS-CoV-2 and dengue viruses may interfere with initial clinicolaboratory diagnosis and treatment decisions and may also lead to spread of COVID-19 infections due to lack of isolation precautions.3 Southeast Asia and South America have reported cocirculation of dengue and COVID-19.15 Twenty-two per cent of COVID-19 infections may be falsely identified as dengue.3 Little is known about the causes of false-positive NS1 antigen tests, except for possible cross-reactivity with other flaviviruses and possibly cytomegalovirus. Antigenic cross-reactivity between dengue, JE and COVID-19 could be due to similarity of specific SARS-CoV-2, JE and dengue protein structures regardless of genetic distance.16
The most common haematological changes observed in patients with COVID-19 include thrombocytopenia and lymphopenia, with a smaller population of these patients having leucopenia similar to dengue fever.17 A retrospective study performed on 1099 patients with COVID-19 showed 82.1% and 36.2% of patients with lymphopenia and thrombocytopenia on admission, respectively, and 33.7% of patients with leucopenia.18 A meta-analysis of 1779 patients with COVID-19 showed that thrombocytopenia was associated with a threefold enhanced risk of severe COVID-19 and a lower platelet count correlated with mortality. Thus, platelet count may serve as a simple and inexpensive biomarker for disease severity and risk of mortality for patients in the intensive care unit.19 The pathophysiological processes include autoantibodies and immune complex-mediated direct attack on haematopoietic stem/progenitor cells and damage to the lungs. Decreased thrombopoietin production due to liver damage and dysfunctional bone marrow microenvironment, increased platelet clearance and consumption, and antiviral drugs may lead to the development of thrombocytopenia in patients with COVID-19 mimicking laboratory findings of dengue fever.17 Diagnostic criteria for dengue infection include NS1 antigen positivity in the first week and/or detection of dengue IgM antibody 7–10 days after the infection. Central nervous system (CNS) manifestations of SARS-CoV-2 are reported, though the virus yielded from CSF remains poor.9 20 Diagnostic criteria for uncommonly seen dengue encephalitis are presence of dengue CNS involvement (impaired consciousness, neck stiffness, focal neurological signs or seizure) and presence of dengue virus RNA, IgM or NS1 antigen in CSF, and CSF pleocytosis without other neuroinvasive pathogens.21 Dengue genetic material or antibody could not be demonstrated in the CSF, but dengue and SARS-CoV-2 coinfection was confirmed in serum and nasopharyngeal swab, respectively, in this patient. It is difficult to assess whether neurological symptoms were due to SARS-CoV-2, dengue virus or because of poor perfusion and hypoxaemia related to DSS.
Currently, no therapy is available beyond supportive care for COVID-19 or dengue. Low-molecular-weight heparin (LMWH) as thrombotic prophylaxis and anticoagulation therapy is often used in patients with COVID-19. Intravenous immunoglobin and steroids are commonly used anti-inflammatory drugs in MIS-C.22 Dexamethasone resulted in lower 28-day mortality among patients with COVID-19 on oxygen therapy or invasive mechanical ventilation.23 Complicated dengue, that is, dengue hemorrhagic fever and DSS are mainly caused by immunological dysfunction. Steroids are feared to increase bleeding risk in dengue, though their role as anti-inflammatory or immune modulators has not been studied well in large randomised studies. In a recent systemic review, Bandara et al concluded that viraemia or any other significant adverse effects are unlikely due to use of corticosteroids in patients with dengue.24 Response to methylprednisolone and immunoglobulin has been reported to be beneficial in cases of reactive haemophagocytosis in dengue25 that is attributed to uncontrolled inflammatory response in dengue, similar to cytokine storm in COVID-19. While treating dengue and COVID-19 coinfection, one has to carefully weigh the benefits of corticosteroid use and LMWH against the risk of bleeding on a case-to-case basis. Randomised control trials on the effectiveness of corticosteroids and/or LMWH in patients with dengue and COVID-19 coinfection are needed to guide the management, particularly in dengue-endemic regions with limited access to intensive care units and ventilatory support.
This clinical case brings forth the unique challenge of differentiating and managing two disease entities, including a COVID-19 pandemic in a dengue-endemic region. Delay in diagnosis or treatment due to poor access to healthcare facilities in dengue-endemic regions may lead to poor outcome, particularly during the COVID-19 pandemic when transportation is further restricted. As she resides in an urban area where this tertiary care hospital is situated, early access to healthcare facilities and follow-up was possible. As the child improved on supportive care, and there is no definite antiviral therapy for any of these viruses, we did not add any investigational therapy for COVID-19 though therapeutic management for COVID-19 would have been reassessed had the patient not responded. In summary, coinfection of COVID-19 and dengue virus is a unique global challenge needing immediate attention of researchers, policy makers and frontline clinicians while managing such cases, particularly in dengue-endemic regions.
Patient’s perspective.
When our child started having fever, we were concerned she may have a symptom of COVID-19, as she had come in contact with a COVID-19-positive case. When she deteriorated, she was tested and admitted to AIIMS Patna. Later on, we came to know that she had tested positive for dengue serum NS1 antigen as well. Those were indeed challenging times for us as we were aware of the severity and complexity of both diseases. Due to the untiring efforts of the managing team, our child clinically improved and she was discharged home.
Learning points.
Coinfection of COVID-19 and dengue virus is likely to pose a unique global challenge, especially in dengue-endemic countries.
This challenge warrants immediate attention of researchers and international health policy makers.
Clinicians should keep a high index of suspicion of COVID-19 even in a child with a positive dengue rapid test, given misdiagnosing COVID-19 as dengue has serious implications in outcomes for the patient and on a more global scale.
Acknowledgments
We thank Dr P K Singh, director, AIIMS Patna, for overall guidance; Dr C M Singh for facilitation of many additional services in management; Dr Arun Prasad, associate professor of paediatrics; Dr Bhabesh Kant, assistant professor of paediatrics; and Dr Amit Sinha, associate professor of paediatric surgery, All India Institute of Medical Sciences, Patna, who contributed in day-to-day clinical management of the child.
Footnotes
Contributors: LT: concept and design, supervised initial draft, review of literature, analysis and final drafting of the manuscript and treatment decisions; SS, AB and PK: involved in patient management, literature search and initial draft writing. All the authors contributed to the final manuscript. The corresponding author had full access to all of the data and the final responsibility to submit for publication.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Parental/guardian consent obtained.
Ethics approval
Ethical clearance and written consent for publishing the case was granted by the institute ethics committee and the patient’s parents, respectively.
References
- 1.World Health Organization . Coronavirus disease (COVID-19) outbreak situation. Available: Https://www.who.int/emergencies/diseases/novel-coronavirus-2019 [Accessed 13 Sep 2020].
- 2.Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 2020;395:507–13. 10.1016/S0140-6736(20)30211-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lustig Y, Keler S, Kolodny R, et al. Potential antigenic cross-reactivity between SARS-CoV-2 and Dengue viruses [published online ahead of print, 2020 Aug 14]. Clin Infect Dis 2020:ciaa1207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chong ZL, Sekaran SD, Soe HJ, et al. Diagnostic accuracy and utility of three dengue diagnostic tests for the diagnosis of acute dengue infection in Malaysia. BMC Infect Dis 2020;20:210. 10.1186/s12879-020-4911-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tiwari L, Anand C, Kumar G, et al. Abstract PCCLB-42: new predictive score for PICU: pediatric quick sofa score – AIIMS Patna model. Ped Critical Care Med 2018;19:256. [Google Scholar]
- 6.Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med 2020;382:727–33. 10.1056/NEJMoa2001017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.World Health Organization . Coronavirus disease (COVID-19) outbreak situation. Available: Https://www.who.int/emergencies/diseases/novel-coronavirus-2019 [Accessed 13 Sept 2020].
- 8.Jiang L, Tang K, Levin M, et al. COVID-19 and multisystem inflammatory syndrome in children and adolescents. Lancet Infect Dis 2020;S1473-3099:30651–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Garg RK, Paliwal VK, Gupta A. Encephalopathy in patients with COVID‐19: a review. J Med Virol 2020;0140. 10.1002/jmv.26207 [DOI] [PubMed] [Google Scholar]
- 10.Abdel-Mannan O, Eyre M, Löbel U, et al. Neurologic and radiographic findings associated with COVID-19 infection in children. JAMA Neurol 2020. doi: 10.1001/jamaneurol.2020.2687. [Epub ahead of print: 01 Jul 2020]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.World Health Organization . Dengue and severe dengue, 2020. Available: Https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue [Accessed 23 Aug 2020].
- 12.Bhatt S, Gething PW, Brady OJ, et al. The global distribution and burden of dengue. Nature 2013;496:504–7. 10.1038/nature12060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Brady OJ, Gething PW, Bhatt S, et al. Refining the global spatial limits of dengue virus transmission by evidence-based consensus. PLoS Negl Trop Dis 2012;6:e1760. 10.1371/journal.pntd.0001760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yan G, Lee CK, Lam LTM, et al. Covert COVID-19 and false-positive dengue serology in Singapore. Lancet Infect Dis 2020;20:536. 10.1016/S1473-3099(20)30158-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Magalhaes T, Chalegre KDM, Braga C, et al. The endless challenges of arboviral diseases in Brazil. TropicalMed 2020;5:75. 10.3390/tropicalmed5020075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chung SJ, Krishnan PU, Leo YS. Two cases of false-positive dengue non-structural protein 1 (NS1) antigen in patients with hematological malignancies and a review of the literature on the use of NS1 for the detection of dengue infection. Am J Trop Med Hyg 2015;92:367–9. 10.4269/ajtmh.14-0247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang Y, Zeng X, Jiao Y, et al. Mechanisms involved in the development of thrombocytopenia in patients with COVID-19. Thromb Res 2020;193:110–5. 10.1016/j.thromres.2020.06.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Guan W-J, Ni Z-Y, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020;382:1708–20. 10.1056/NEJMoa2002032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lippi G, Plebani M, Henry BM. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: a meta-analysis. Clinica Chimica Acta 2020;506:145–8. 10.1016/j.cca.2020.03.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Destras G, Bal A, Escuret V, et al. Systematic SARS-CoV-2 screening in cerebrospinal fluid during the COVID-19 pandemic. The Lancet Microbe 2020;1:e149. 10.1016/S2666-5247(20)30066-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Carod-Artal FJ, Wichmann O, Farrar J, et al. Neurological complications of dengue virus infection. Lancet Neurol 2013;12:906–19. 10.1016/S1474-4422(13)70150-9 [DOI] [PubMed] [Google Scholar]
- 22.Abrams JY, Godfred-Cato SE, Oster ME, et al. Multisystem Inflammatory Syndrome in Children Associated with Severe Acute Respiratory Syndrome Coronavirus 2: A Systematic Review. J Pediatr 2020;226:45–54. 10.1016/j.jpeds.2020.08.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Horby P, Lim WS. et al. Dexamethasone in Hospitalized Patients with Covid-19 - Preliminary Report. N Engl J Med 2020;17:NEJMoa2021436. [Google Scholar]
- 24.Bandara SMR, Herath HMMTB, Herath H. Effectiveness of corticosteroid in the treatment of dengue - A systemic review. Heliyon 2018;4:e00816. 10.1016/j.heliyon.2018.e00816 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Srichaikul T, Punyagupta S, Kanchanapoom T, et al. Hemophagocytic syndrome in dengue hemorrhagic fever with severe multiorgan complications. J Med Assoc Thai 2008;91:104–9. [PubMed] [Google Scholar]