Abstract
Purpose
To investigate the application of critical flicker fusion frequency (CFF) in demyelinating optic neuritis (DON).
Methods
A cross-sectional study. A total of 127 eyes in 69 DON patients and 63 eyes in 33 healthy control (HC) groups were included between January 2021 to September 2021 from Department of Ophthalmology, PLA General Hospital. Patients underwent best-corrected visual acuity (BCVA), visual field, optical coherence tomography (OCT), flash visual evoked potential (F-VEP), and CFF examinations. The affected eyes were divided into aquaporins 4 (AQP4-), myelin oligodendrocyte glycoprotein (MOG-), and double negative DON according to serum antibody; mild, moderate, severe degree visual impairment according to BCVA ≥ 0.5, 0.1-0.5, < 0.1; and 4 groups: < 1, 1 ∼< 3, 3 ∼ < 6 and > 6 months according to time interval from onset to CFF examination. One-way ANOVA was used to perform above subgroup analysis. The correlations between CFF and F-VEP peak time, peak value, BCVA and mean visual filed defect (MD) were analyzed in order via Pearson correlation analysis.
Results
he trichromatic values of red, green, and yellow in DON affected eyes were 21.83 ± 9.03, 23.66 ± 10.21, 24.09 ± 10.77 Hz, respectively, which was significantly reduced compared with the HC group (t = -14.82, -14.22, -14.00; P < 0.001). The subgroup analysis showed no significant difference between different antibody subtypes (P = 0.914 <0.848 <0.604), whereas, a significant decrease of CFF trichromatic value was found in severe visual acuity impairment group (P < 0.001). There was a significant difference in CFF- trichromatic values at different time points (P < 0.001), to be specific, CFF fluctuated under 20Hz within 3 months after onset and tended to be stable around 24-28Hz. Correlation analysis showed that the peak time of F-VEP (r = -0.486, -0.515, -0.526; P < 0.001), BVCA (r = -0.640, -0.659, -0.642; P < 0.001), were negatively correlated with CFF trichromatic values, MD and CFF were positively correlated (r = 0.486, 0.453, 0.476; P = 0.003, 0.006, 0.004).
Conclusions
A significant decrease of CFF value was found in DON-affected eyes, and it has a good correlation with BCVA, MD and latency of F-VEP, and can better reflect the impairment of visual function
Keywords: Critical flicker fusion frequency, Demyelinating optic neuritis
1. Introduction
Demyelinating optic neuritis (DON) is an inflammatory demyelinating lesion of the optic nerve, which can cause acute or subacute vision loss, and mainly affects young and middle-aged adults.1, 2, 3, 4, 5, 6, 7 Due to high recurrence rates, blindness, and disability, the effective evaluation of visual function has become a major priority. Currently, clinicians primarily rely on visual field, optical coherence tomography (OCT), and visual electrophysiological examinations. However, due to high costs and prolonged examination periods, timely and effective visual impairment assessments are often not available.
The critical flicker fusion frequency (CFF) detector is a small portable, age-free, and highly repetitive device, based on visual residue phenomenon principles, and reflects temporal aspect of visual function. When flickering light enters the eye, flickering sensations occur, which gradually disappear with increasing flickering frequency, to finally form a stable light called light fusion. The critical flicker fusion frequency (CFF) is the minimum intermittent frequency between flicker and stability that induces continuous fusion sensation. In recent years, studies reported a reduced CFF in patients with multiple sclerosis (MS), DON, and diabetic retinopathy, even without visual acuity and field impairment.8, 9, 10, 11, 12, 13, 14, 15 A recent study highlighted the precision of dynamic visual function assessment; it identified a demyelinating attack, and prolonged visual evoked potential (VEP) latencies closely related to CFF values in 23 DON patients.16,17 The purpose of our study was to explore the clinical application of CFF in Asian patients with DON.
2. Methods
This study was approved by the Ethics Committee of the Chinese People's Liberation Army General Hospital (PLAGH) (Grant No.: S2017-093-01) and adhered to the Declaration of Helsinki. Informed consent was obtained from patients and their parents/guardians.
2.1. Patients
Clinical data were prospectively collected from hospitalized patients diagnosed with DON at the Neuro-Ophthalmology Department of the Chinese PLAGH between January 1st and September 30th, 2021.
DON was diagnosed according to evidence-based guidelines for the diagnosis and treatment of DON in China (2021).18 The detailed inclusion criteria were as follows: (1) Acute impairment of visual acuity, with or without ocular rotation pain. (2) Complication of at least the following two abnormalities: relative afferent pupillary defect (RAPD), visual field defect, abnormal visual evoked potential, dyschromatopsia. (3) Vascular, traumatic, compressive, infiltrative, toxic, metabolic, and hereditary optic neuropathy were excluded. The diagnostic criteria for each subtype are as follows: (1) aquaporins 4 related optic neuritis (AQP4-DON): DON with positive aquaporins 4 (AQP-4) antibody; (2) myelin oligodendrocyte glycoprotein related optic neuritis (MOG-DON): DON with positive myelin oligodendrocyte glycoprotein (MOG) antibody; (3) double negative DON: AQP-4 and MOG antibody were negative, the vision continued to decline for less than 2 weeks, and the vision began to recover about 3 weeks after the onset. Other ocular and systemic diseases that caused visual impairment and visual field defects were excluded, for example, glaucoma, severe cataracts, uveitis, retinal disease, amblyopia, and keratopathy.
In total, 69 DON patients with 98 affected eyes and 29 contralateral eyes, and 33 HC cases (HC group) with 63 eyes were assessed.
2.2. CFF and associated examinations
All patients underwent best-corrected visual acuity (BCVA), color vision, relative afferent pupillary defect (RAPD), intraocular pressure assessment, and other basic ophthalmological examinations, including OCT, Humphrey visual field assessments, and flash-visual evoked potential (F-VEP) before 1 week of the CFF examination. We recorded the following information: thickness of peripheral retinal nerve fiber layer (pRNFL) and macular inner limiting membrane-retinal pigment epithelium (mILM-RPE), F-VEP peak times, VEP peak values, and visual field mean defect (MD) values.
All participants underwent CFF (handheld CFF detector type 2 instrument, NEITZ, Japan) examinations (Fig. 1). Specific inspection steps were: external light was shielded and the test conducted in a relatively quiet environment in a darkroom. All participants were trained in advance to master the operating procedure to avoid errors during the process. The visual target was placed 25 cm in front of the patient's eyes, the deviation angle was < 2°, and CFF values for monocular red, green, and yellow (rCFF, gCFF, and yCFF) light were recorded, respectively. When adjusting to a particular color, the frequency was gradually increased until the minimum fusion frequency had occurred. Data were then recorded.
Fig. 1.
Handheld CFF detector type 2 machine (NEITZ, Japan).
2.3. Statistical analysis
Data were statistically analyzed by SPSS Ver. 26 (IBM Corporation, NY, USA). If measurement data were normally distributed, they was expressed as the mean ± standard deviation, while categorical data were described as the number of cases and percentages. Based on the time interval from condition onset to CFF examination, patients were divided into four groups: < 1 m (within 1 month), 1∼< 3 m (between 1 and 3 months), 3∼< 6 m (between 3 and 6 months), and >6 m (more than 6 months). We defined BCVA ≥0.5, 0.1∼< 0.5, and <0.1 as mild, moderate, and severe visual impairment, respectively. CFF trichromatic values were compared between different antibody subtypes, different degrees of visual impairment, and different check intervals, using one way analysis of variance (ANOVA) post hoc tests. The Student-Newman-Keuls (SNK) method was used for comparisons between groups. Correlations between CFF and other ophthalmologic examination: F-VEP peak times, peak values, BCVA and MD were analyzed, respectively. The correlation coefficient was r and P < 0.05 was considered statistically significant.
3. Results
3.1. Demographics and ophthalmological examination results
In total, 69 DON patients with a mean age of 34.19 ± 12.45 years and 33 HC cases with a mean age of 35.18 ± 10.96 years were assessed, respectively. We observed a female predominance in the DON group; 51 (73.9%) were female, while only 16 (48.5%) were female in the HC group. The median logarithm of the minimum angle of resolution (logMAR) visual acuity was 0.22 (interquartile range (IQR): 0.00, 1.27) for the 98 affected eyes in the DON group. Seventy-three affected eyes in this group underwent optic disc OCT examination, and 70 eyes underwent macular OCT examination. The median thickness of pRNFL and mILM-RPE was 67.50 μm (IQR: 61.00, 96.25) and 234.00 μm (IQR: 224.00, 247.25), respectively. The mean MD value was −12.40 ± 7.79 dB in 35 affected eyes. The mean peak values and peak times for F-VEP were 15.26 ± 7.67 μV and 115.60 ± 24.60 ms, respectively, in 50 affected eyes.
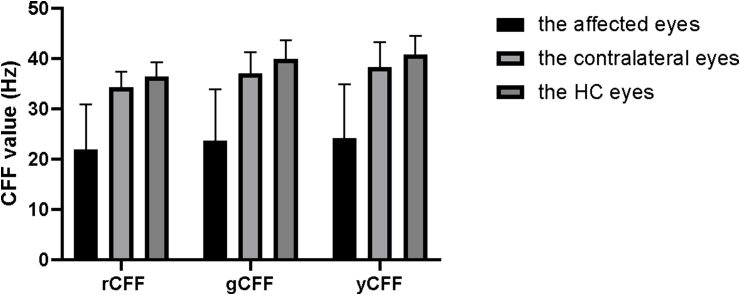
When compared with HC eyes, we observed a significant decline in DON eyes (P < 0.001), especially in affected eyes of DON patients, and the mean CFF values for red, green, and yellow were 21.83 ± 9.03, 23.66 ± 10.21, and 24.09 ± 10.77 Hz, respectively in affected eyes, and 34.21 ± 3.18, 37.03 ± 4.20, and 38.24 ± 4.93 Hz, respectively in contralateral eyes (Table 1, Fig. 2).
Table 1.
Comparison of CFF between DON and HC eyes.
| group | N | rCFF | gCFF | yCFF |
|---|---|---|---|---|
| the affected eyes | 98 | 21.83 ± 9.03 | 23.66 ± 10.21 | 24.09 ± 10.77 |
| the contralateral eyes | 29 | 34.21 ± 3.18 | 37.03 ± 4.20 | 38.24 ± 4.93 |
| the HC eyes | 63 | 36.38 ± 2.90 | 39.81 ± 3.77 | 40.71 ± 3.78 |
| ta | −14.82 | −14.22 | −14.00 | |
| tb | −11.40 | −10.34 | −9.96 | |
| P | <0.001 | <0.001 | <0.001 |
N: number; rCFF, gCFF, yCFF: critical flicker fusion frequency for red, green, yellow; HC: healthy control.
The affected eyes vs HC eyes.
The contralateral eyes vs HC eyes.
Fig. 2.
Comparison of CFF value between DON and HC eyes.
3.2. Subgroup analyses of CFF values
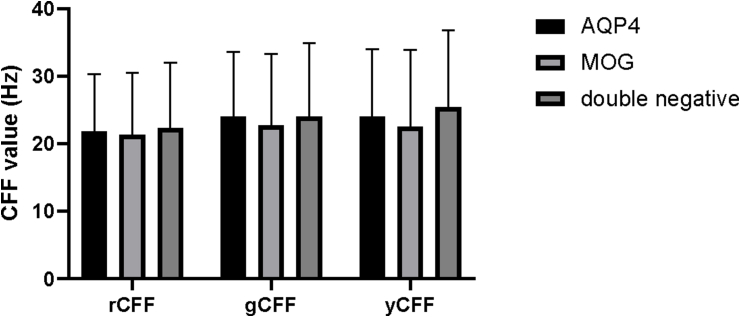
We observed no statistically significant differences in CFF tricolor values between AQP4-DON, MOG-DON, and double-negative DON (P > 0.05) (Table 2, Fig. 3).
Table 2.
Comparison of CFF at different subgroups in DON affected eyes.
| N | rCFF | P | gCFF | P | yCFF | P | |
|---|---|---|---|---|---|---|---|
| subtype | 0.914 | 0.848 | 0.604 | ||||
| AQP4 | 39 | 21.77 ± 8.51 | 24.00 ± 9.59 | 23.97 ± 9.99 | |||
| MOG | 25 | 21.28 ± 9.23 | 22.64 ± 10.60 | 22.52 ± 11.32 | |||
| Double negative | 34 | 22.29 ± 9.69 | 24.03 ± 10.86 | 25.38 ± 11.37 | |||
| BCVA | <0.001 | <0.001 | <0.001 | ||||
| mild | 48 | 27.17 ± 6.87 | 29.94 ± 7.60 | 30.69 ± 8.07 | |||
| moderate | 18 | 20.61 ± 8.07 | 22.11 ± 8.30 | 22.11 ± 8.80 | |||
| severe | 32 | 14.50 ± 6.87 | 15.13 ± 7.97 | 15.31 ± 8.51 | |||
| Check interval | <0.001 | <0.001 | <0.001 | ||||
| <1 m | 25 | 17.00 ± 7.87 | 17.56 ± 8.67 | 17.84 ± 9.27 | |||
| 1∼<3 m | 16 | 18.25 ± 8.51 | 20.06 ± 9.09 | 19.88 ± 9.16 | |||
| 3∼<6 m | 16 | 24.25 ± 6.65 | 26.44 ± 7.60 | 27.88 ± 7.50 | |||
| >6 m | 41 | 25.22 ± 9.10 | 27.71 ± 10.34 | 28.07 ± 11.11 |
N: number; rCFF, gCFF, yCFF: critical flicker fusion frequency for red, green, yellow; AQP4: aquaporins 4-antibody; MOG: myelin oligodendrocyte glycoprotein antibody; BCVA: best-corrected visual acuity; <1 m: within 1 month; 1∼<3 m: between 1 to 3 months; 3∼<6 m: between 3 and 6 months; >6 m: above 6 months.
Fig. 3.
Comparison of CFF value between different antibody subtypes in DON affected eyes.
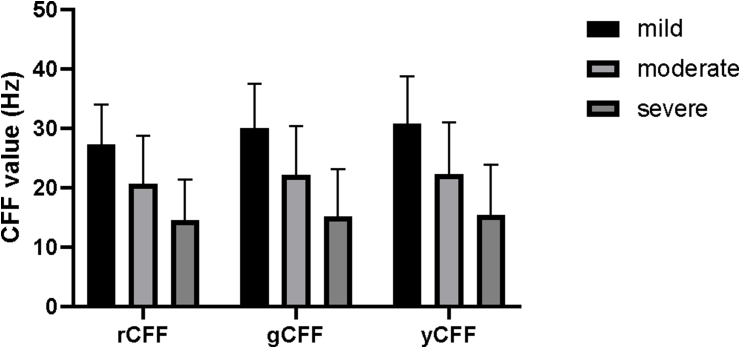
We observed that rCFF, gCFF, and yCFF values varied across different impaired vision groups (P < 0.001). Values were lower in severely impaired vision eyes; 14.50 ± 6.87, 15.13 ± 7.97, and 15.31 ± 8.51 Hz for rCFF, gCFF, and yCFF, respectively (Table 2, Fig. 4).
Fig. 4.
Comparison of CFF value between the different impaired vision groups in DON affected eyes.
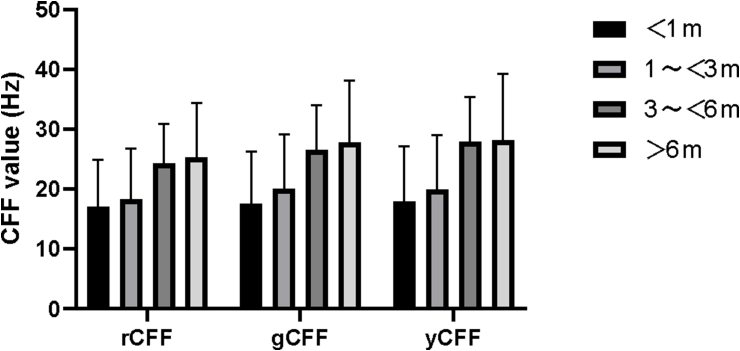
Subgroup analyses revealed a significant drop of CFF values fluctuated under 20 Hz within 3 months, but gradually increased and stabilized to 24–28 Hz after 3 months (P < 0.001) (Table 2). A statistical difference was observed before and after 3 months of onset (P < 0.05). (Table 3, Fig. 5).
Table 3.
Comparison of CFF results at different check intervals using the SNK method.
| check interval | N | rCFF (Hz) |
gCFF (Hz) |
yCFF (Hz) |
|||
|---|---|---|---|---|---|---|---|
| 1a | 1a | 2 | 1a | 2 | |||
| <1 m | 25 | 17.00 | 17.56 | 17.84 | |||
| 1∼<3 m | 16 | 18.25 | 20.06 | 19.88 | |||
| 3∼<6 m | 16 | 24.25 | 26.44 | 27.88 | |||
| >6 m | 41 | 25.22 | 27.71 | 28.07 | |||
| P | 0.628 | 0.707 | 0.784 | 0.659 | 0.503 | 0.948 | |
N: number; rCFF, gCFF, yCFF: critical flicker fusion frequency for red, green, yellow; <1 m: within 1 month; 1∼<3 m: between 1 and 3 months; 3∼<6 m: between 3 and 6 months; >6 m: above 6 months.
BetweenColumns 1 and 2 subsets of alpha = 0.05.
Fig. 5.
Comparison of CFF value between different check intervals in DON affected eyes.
3.3. Correlation analyses between F-VEP, BCVA, visual field, and CFF values
Strong negative correlations were identified between F-VEP peak times and CFF trichromatic values in affected eyes (P < 0.001); correlation coefficients were −0.486, −0.515, and −0.526, respectively. However, we observed no statistical correlations between F-VEP peak value and CFF values (P > 0.05) (Table 4).
Table 4.
Correlation analysis of BCVA, visual field, F-VEP, and CFF in DON patients.
| N | rCFF |
gCFF |
yCFF |
||||
|---|---|---|---|---|---|---|---|
| r | P | r | P | r | P | ||
| the affected eyes | |||||||
| BCVA | 98 | −0.640 | <0.001 | −0.659 | <0.001 | −0.642 | <0.001 |
| MD | 35 | 0.483 | 0.003 | 0.453 | 0.006 | 0.476 | 0.004 |
| Peak value | 50 | 0.210 | 0.142 | 0.253 | 0.077 | 0.298 | 0.036 |
| Peak time | 50 | −0.486 | <0.001 | −0.515 | <0.001 | −0.526 | <0.001 |
| the contralateral eyes | |||||||
| BCVA | 29 | −0.206 | 0.285 | −0.222 | 0.248 | −0.314 | 0.097 |
| MD | 9 | 0.393 | 0.295 | 0.285 | 0.458 | 0.269 | 0.484 |
| Peak value | 10 | 0.146 | 0.686 | −0.123 | 0.736 | −0.051 | 0.889 |
| Peak time | 10 | 0.420 | 0.226 | 0.180 | 0.619 | 0.246 | 0.494 |
N: number; rCFF, gCFF, yCFF: critical flicker fusion frequency for red, green, yellow; BCVA: best-corrected visual acuity; MD: mean defect.
We also analyzed correlations between BCVA, visual field, and CFF values. Strong negative correlations were observed between logMAR visual acuity and CFF values in affected eyes (P < 0.001); correlation coefficients were −0.640, −0.659, and −0.642, respectively. Furthermore, positive correlations were identified between the visual field and rCFF (r = 0.483), gCFF (r = 0.453), and yCFF (r = 0.476) in affected eyes (P < 0.05) (Table 4).
Correlations were not evident between F-VEP, BCVA, visual field, and CFF values in contralateral healthy eyes (P > 0.05) (Table 4).
4. Discussion
Generally, visual function refers to the ability to acquire visual information. This consists of static visual function and temporal acuity; the former includes visual acuity, visual field, color perception, brightness, contrast sensitivity, and spatial resolution, whereas the latter reflects visual system responses to variations in light over time, as measured by CFF.19 CFF is a psychophysical test of visual temporal resolution and has been widely used as an analytical tool for retinal diseases, e.g., chloroquine toxic retinopathy, diabetic retinopathy, central serous retinopathy, age-related macular degeneration, and predicting postoperative visual outcomes in subjects with cataracts.20, 21, 22 In 1957, Brenton included several optic nerve diseases, including ischemic, compression, demyelinating, and glaucoma optic neuropathy,23 and the results showed that CFF was more sensitive and specific than RAPD or color vision in the assessment of optic neuropathy. Daniel et al. reported impaired CFF in 13 recovered ON patients, and proposed the involvement of axonal projections of magnocellular retinal ganglion cells.24 Due to limited equipment, the clinical application of CFF to ON has been neglected and there are few clinical studies on CFF in optic nerve diseases.
Our study included 69 DON patients and 33 HCs and demonstrated a significant decrease in CFF values in DON eyes when compared with HC eyes, which is more noteworthy in the affected eyes. It is hypothesized that axons posterior to the lamina cribrosa are segmentally wrapped with myelin sheaths composed of oligodendrocytes, therefore, visual information is transmitted by leaps. During acute disease phases, the myelin sheath may be attacked by inflammatory cells, inducing massive disintegration and delayed visual transmission. Therefore, it is not difficult to understand prolonged F-VEP peak times and decreased CFF values in DON patients.
Subgroup analyses indicated little impact of CFF values on differentiating DON subtypes. The main differences between ON subtypes involve different antibody-mediated immune responses. Hence, CFF as a functional indicator cannot easily distinguish between DON subtypes. Additionally, we divided affected eyes into four groups according to the time interval from condition onset, we find a prominent difference of CFF exists before and after 3 months of condition onset. CFF fluctuated under 20 Hz within the first 3 months, whereas CFF trichromatic results were stable around 24–28 Hz, although they continued to improve slowly after 3 months. In accordance with these findings, Daniah et al.25 performed multifocal VEP assessments on ON evolution and observed that amplitude recovery and latency shortening were fastest within the first 3 months, and continued to improve slowly after 3 months, but changes were not significant. Latency shortening after ON is primarily due to remyelination which occurs during early post-acute stages, where cells form new myelin sheaths.26 These observations suggest that prompt and effective treatments are necessary to restore myelin integrity during the first 3 months. However, remyelination is often incomplete, as significant residual CFF even 12 months after acute ON.
The severe visual impairment group had lower CFF values. Correlation analyses demonstrated a strong negative correlation between logMAR visual acuity and CFF values, F-VEP latent times and CFF values in affected eyes; correlation coefficients were more than 0.4. Similarly, a correlation was identified between MD and CFF values in affected eyes. These results indicated a consistency in visual functional indicators in DON affected eyes. CFF was sensitive to changes in visual function and F-VEP, although these conclusions may be slightly biased due to the unequal and small cohort size.
However, despite this and absent longitudinal follow-up data, we demonstrated that CFF examinations were valuable for patients with DON. We observed significant decreases across different parameters in DON patients, especially during acute disease phases, and it will gradually recover and stabilize after 3 months. In conclusion, CFF better reflected the visual function of affected eyes, and was closely associated with BCVA, MD, and VEP peak times.
Study Approval
This study is approved by National Key Research and Development Program (2018YFE0113900)(2018YFE0113900).
This study was reviewed and approved by the Ethics Committee of PLA General Hospital (Approval No.: S2017-093-01).
Author Contributions
All authors had full access to all the data in the study and were responsible for the integrity and accuracy of data.
Study concept and design, acquisition of data, analysis and interpretation of data, drafting and revision of manuscript:JXF; Acquisition of data, analysis and interpretation of data:YPW; Critical revision of manuscript for intellectual content:SYT; Acquisition of data:GCX, HFZ; Study supervision: QGX; Study concept and design, study supervision, and critical revision of manuscript for intellectual content: SHW.
Acknowledgments
We are grateful to all participants in this study. We express our sincere gratitude to all optometrists and nurses at the Department of Ophthalmology in the Chinese People's Liberation Army General Hospital & The Chinese People's Liberation Army Medical School for their assistance in this project.
Funding
This work supported by National Key Research and Development Program (2018YFE0113900).
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Abbreviations
- CFF
critical flicker fusion frequency
- rCFF
critical flicker fusion frequency for red
- gCFF
critical flicker fusion frequency for green
- yCFF
critical flicker fusion frequency for yellow
- DON
demyelinating optic neuritis
- HC
healthy control
- BCVA
best-corrected visual acuity
- OCT
optical coherence tomography
- VEP
visual evoked potential
- F-VEP
flash visual evoked potential
- MS
multiple sclerosis
- RAPD
relative afferent pupillary defect
- AQP4
aquaporins 4
- AQP4-DON
aquaporins 4 antibody related demyelinating optic neuritis
- MOG
myelin oligodendrocyte glycoprotein
- MOG-DON
myelin oligodendrocyte glycoprotein antibody related demyelinating optic neuritis
- MD
mean defect
- mILM-RPE
macular inner limiting membrane-retinal pigment epithelium
References
- 1.Hassan M.B., Stern C., Flanagan E.P., et al. Population-based incidence of optic neuritis in the era of aquaporin-4 and myelin oligodendrocyte glycoprotein antibodies. Am J Ophthalmol. 2020;220:110–114. doi: 10.1016/j.ajo.2020.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Braithwaite T., Subramanian A., Petzold A., et al. Trends in optic neuritis incidence and prevalence in the UK and association with systemic and neurologic disease. JAMA Neurol. 2020;77(12):1514–1523. doi: 10.1001/jamaneurol.2020.3502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wingerchuk D.M., Banwell B., Bennett J.L., et al. International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology. 2015;85(2):177–189. doi: 10.1212/wnl.0000000000001729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Toosy A.T., Mason D.F., Miller D.H. Optic neuritis. Lancet Neurol. 2014;13(1):83–99. doi: 10.1016/s1474-4422(13)70259-x. [DOI] [PubMed] [Google Scholar]
- 5.Wakakura M., Ishikawa S., Oono S., et al. [Incidence of acute idiopathic optic neuritis and its therapy in Japan. Optic neuritis treatment trial multicenter cooperative research group (ONMRG)] Nippon Ganka Gakkai Zasshi. 1995;99(1):93–97. [PubMed] [Google Scholar]
- 6.Rodriguez M., Siva A., Cross S.A., et al. Optic neuritis: a population-based study in Olmsted County, Minnesota. Neurology. 1995;45(2):244–250. doi: 10.1212/wnl.45.2.244. [DOI] [PubMed] [Google Scholar]
- 7.Dorfman L. Optic neuritis. N Engl J Med. 2006;355(2):212. doi: 10.1056/NEJMc061396. author reply 212. [DOI] [PubMed] [Google Scholar]
- 8.Daley M.L., Swank R.L., Ellison C.M. Flicker fusion thresholds in multiple sclerosis. A functional measure of neurological damage. Arch Neurol. 1979;36(5):292–295. doi: 10.1001/archneur.1979.00500410070010. [DOI] [PubMed] [Google Scholar]
- 9.Miller P.N., Parsons O.A. Flicker fusion thresholds in multiple sclerosis. AMA Arch Neurol Psychiatry. 1957;77(2):134–139. doi: 10.1001/archneurpsyc.1957.02330320032004. [DOI] [PubMed] [Google Scholar]
- 10.Frank C., Harrer G., Schiner P. [Flicker fusion threshold and its diagnostic value in multiple sclerosis (author's transl)] Nervenarzt. 1980;51(3):168–175. [PubMed] [Google Scholar]
- 11.Patterson V.H., Foster D.H., Heron J., et al. Multiple sclerosis. Luminance threshold and measurements of temporal characteristics of vision. Arch Neurol. 1981;38(11):687–689. doi: 10.1001/archneur.1981.00510110047005. [DOI] [PubMed] [Google Scholar]
- 12.Dain S.J., Rammohan K.W., Benes S.C., et al. Chromatic, spatial, and temporal losses of sensitivity in multiple sclerosis. Invest Ophthalmol Vis Sci. 1990;31(3):548–558. [PubMed] [Google Scholar]
- 13.Galvin R., Regan D., JJBajon Heron. Impaired temporal resolution of vision after acute retrobulbar neuritis. Brain. 1976;99(2):255–268. doi: 10.1093/brain/99.2.255. [DOI] [PubMed] [Google Scholar]
- 14.Woung L., Wakakura M., Ishikawa SJJjoo. Critical flicker frequency in acute and recovered optic neuritis. 1993;37(2):122–129. [PubMed] [Google Scholar]
- 15.Gregori B., Papazachariadis O., Farruggia A., et al. A differential color flicker test for detecting acquired color vision impairment in multiple sclerosis and diabetic retinopathy. J Neurol Sci. 2011;300(1-2):130–134. doi: 10.1016/j.jns.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 16.Young M.T., Braich P.S., Haines S.R. Critical flicker fusion frequency in demyelinating and ischemic optic neuropathies. Int Ophthalmol. 2018;38(3):1069–1077. doi: 10.1007/s10792-017-0561-z. [DOI] [PubMed] [Google Scholar]
- 17.Raz N., Shear-Yashuv G., Backner Y., et al. Temporal aspects of visual perception in demyelinative diseases. J Neurol Sci. 2015;357(1-2):235–239. doi: 10.1016/j.jns.2015.07.037. [DOI] [PubMed] [Google Scholar]
- 18.Neuro-ophthalmology Group of Ophthalmology Branch of Chinese Medical Association, Evidence-based Medicine Center of Lanzhou University/World Health Organization Collaborating Centre for Guideline Implementation and Knowledge Translation (2021) Evidence-based guidelines for diagnosis and treatment of demyelinating optic neuritis in China (2021). Chin J Ophthalmol.3:171-186. 10.3760/cma.j.cn112142-20201124-00769. [DOI] [PubMed]
- 19.Fleishman J.A., Beck R.W., Linares O.A., et al. Deficits in visual function after resolution of optic neuritis. Ophthalmology. 1987;94(8):1029–1035. doi: 10.1016/s0161-6420(87)33349-4. [DOI] [PubMed] [Google Scholar]
- 20.Douthwaite W.A., Vianya-Estopà M., et al. Predictions of postoperative visual outcome in subjects with cataract: a preoperative and postoperative study. Br J Ophthalmol. 2007;91(5):638–643. doi: 10.1136/bjo.2006.093401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vianya-Estopà M., Douthwaite W.A., Noble B.A., et al. Capabilities of potential vision test measurements: clinical evaluation in the presence of cataract or macular disease. J Cataract Refract Surg. 2006;32(7):1151–1160. doi: 10.1016/j.jcrs.2006.01.111. [DOI] [PubMed] [Google Scholar]
- 22.del Romo G.B., Douthwaite W.A., Elliott D.B. Critical flicker frequency as a potential vision technique in the presence of cataracts. Invest Ophthalmol Vis Sci. 2005;46(3):1107–1112. doi: 10.1167/iovs.04-1138. [DOI] [PubMed] [Google Scholar]
- 23.Brenton R.S., Thompson H.S., Maxner C. 1989. Critical Flicker Frequency: A New Look at an Old Test: New Methods of Sensory Visual Testing. [Google Scholar]
- 24.Jacobson D.M., Olson K.A. Impaired critical flicker frequency in recovered optic neuritis. Ann Neurol. 1991;30(2):213–215. doi: 10.1002/ana.410300216. [DOI] [PubMed] [Google Scholar]
- 25.Alshowaeir D., Yannikas C., Garrick R., et al. Multifocal VEP assessment of optic neuritis evolution. Clin Neurophysiol. 2015;126(8):1617–1623. doi: 10.1016/j.clinph.2014.11.010. [DOI] [PubMed] [Google Scholar]
- 26.Prineas J.W., Barnard R.O., Kwon E.E., et al. Multiple sclerosis: remyelination of nascent lesions. Ann Neurol. 1993;33(2):137–151. doi: 10.1002/ana.410330203. [DOI] [PubMed] [Google Scholar]