Abstract
Background
Huntington’s disease is an autosomal dominant inherited disorder characterized by personality changes (such as irritability and restlessness) and psychotic symptoms (such as hallucinations and delusions). When the personality changes become noticeable, involuntary movements (chorea) also develop. The disease is caused by the CAG repeat expansion in the coding region of the HTT gene, and the diagnosis is based on the presence of this expansion. However, there is currently no effective treatment for the progression of Huntington’s disease and its involuntary motor symptoms. Herein, we present a case in which memantine was effective in treating the chorea movements of Huntington’s disease.
Case presentation
A 75-year-old Japanese woman presented to the hospital with involuntary movements of Huntington’s disease that began when she was 73 years old. In a cerebral blood flow test (N-isopropyl-p-iodoamphetamine–single-photon emission computed tomography), decreased blood flow was observed in the precuneus (anterior wedge) and posterior cingulate gyrus. Usually, such areas of decreased blood flow are observed in patients with Alzheimer’s-type dementia. So, we administered memantine for Alzheimer’s-type dementia, and this treatment suppressed the involuntary movements of Huntington’s disease, and the symptoms progressed slowly for 7 years after the onset of senility. In contrast, her brother died of complications of pneumonia during the course of Huntington’s disease.
Conclusions
We recorded changes in parameters such as the results of the N-isopropyl-p-iodoamphetamine–single-photon emission computed tomography and gait videos over 7 years. Treatment with memantine prevented the chorea movement and the progression of Huntington’s disease. We believe this record will provide clinicians with valuable information in diagnosing and treating Huntington’s disease.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13256-023-04161-z.
Keywords: Huntington’s disease, SPECT, Alzheimer’s disease, Memantine, Case report
Background
Huntington’s disease is an autosomal dominant inherited disorder characterized by personality changes (such as irritability and restlessness) and psychotic symptoms (such as hallucinations and delusions). When the personality changes become noticeable, involuntary movements (chorea) develop [1]. The disease is inherited through autosomal dominance and is caused by the expansion of the CAG repeat sequence in the HTT gene coding region. The diagnosis is based on the presence of this extension. The average age of onset of Huntington’s disease is 35–44 years. In approximately two-thirds of affected patients, neurological symptoms develop first; however, in others, the first symptoms are psychological changes. In the early stage after diagnosis, slight changes appear in eye movements, chorea movements, as do inconspicuous involuntary movements, positioning of the head, depressive symptoms, and irritability [2, 3]. Chorea movements can be partially suppressed by haloperidol and tetrabenazine, the monoamine-depleting agents [1]. However, to the best of our knowledge, no drug can completely treat Huntington’s disease yet.
Herein, we report the case of a woman with Huntington’s disease in whom the symptoms progressed slowly for 7 years after the onset of old age and in whom changes over time, such as those reflected in N-isopropyl-p-iodoamphetamine–single-photon emission computed tomography (IMP-SPECT), were recorded. Also, in this case, we show that memantine had an effect on chorea, swallowing symptoms, and oral symptoms and that treatment with memantine prevented the chorea movement and the progression of this disease.
Case presentation
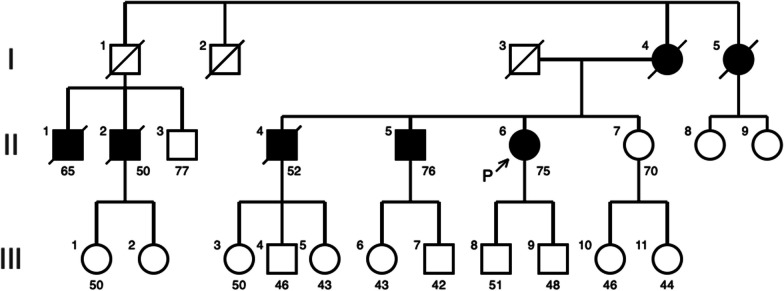
The patient was a 75-year-old Japanese woman (II-6) who was suspected to have Huntington’s disease (because she exhibited chorea in her neck and upper limbs and had a family history of the disease) on her first visit to our hospital 2 years earlier (at age 73 years). Genetic testing was performed (with the patient’s consent) and revealed a repeat abnormality (42 repeat) of the HTT gene; thus, she was diagnosed with Huntington’s disease. Psychological test results were as follows: 25 points on the Mini-Mental State Examination and 10 points on the Alzheimer’s disease Assessment Scale—Cognitive Subscale. Tetrabenazine (the only drug capable of preventing disease progression) was then administered; however, nausea and fatigue necessitated the discontinuation of oral administration. The patient’s older brother (II-5) who developed the disease at the same age as she did had rapidly-progressing chorea; 5 years later, he was almost immobile and bedridden (Fig. 1). She took memantine for 5 years but did not experience any special adverse effects.
Fig. 1.
The patient’s family tree
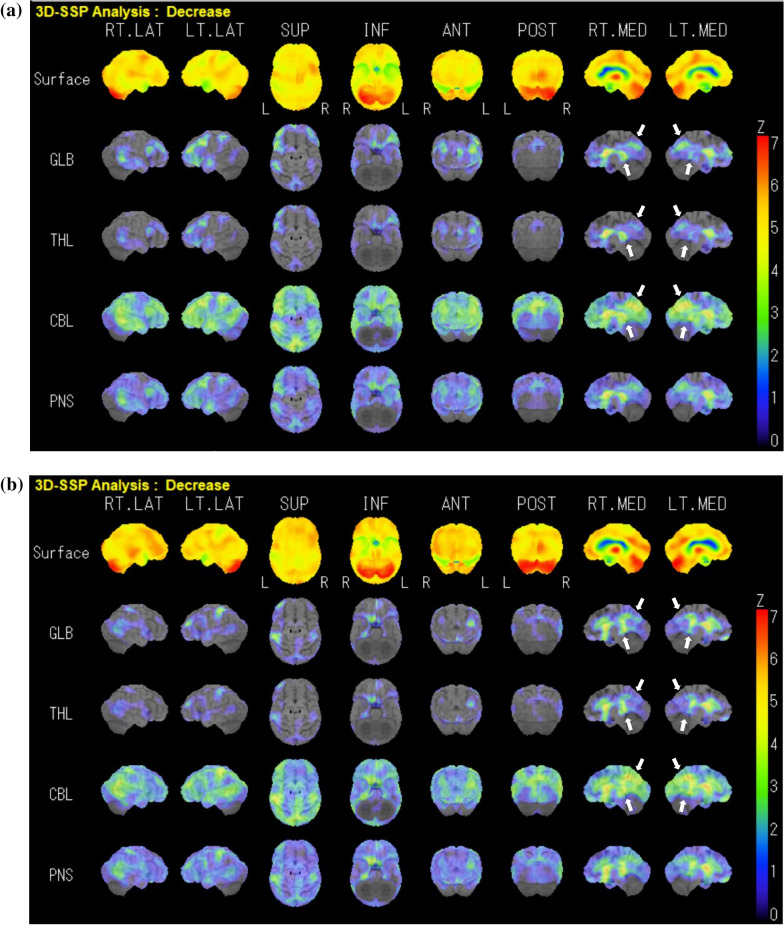
Magnetic resonance imaging of the head revealed changes in the striatum. In a cerebral blood flow test (IMP-SPECT), decreased blood flow was observed in the precuneus (anterior wedge) and posterior cingulate gyrus (Fig. 2). Usually, such areas of decreased blood flow are observed in patients with Alzheimer’s-type dementia. Therefore, we started treatment with an anti-dementia drug as would be done for a case of Huntington’s disease combined with Alzheimer’s disease.
Fig. 2.
a N-isopropyl-p-iodoamphetamine–single-photon emission computed tomography three-dimensional stereotactic surface projection showing a decrease in cerebral blood flow on the date of diagnosis (at the age of 75 years). The white arrows indicate the posterior cingulate gyrus and precuneus, where blood flow is reduced in Alzheimer's disease. b Follow-up data (at the age of 80 years). The blood flow of white arrows is decreased
Initially, donepezil administration was considered; however, because the patient was irritable, a regimen of low-dose memantine (5 mg/day) was started. Thereafter, the patient’s chorea, swallowing symptoms, and oral symptoms improved. A total of 3 months later, the dose of memantine was increased to 10 mg/day; however, the chorea worsened, so the dose was reduced to 5 mg/day. A total of 5 years since then, clinical manifestations have not worsened. The chorea movements also have not worsened substantially, and we have recorded the patient’s gait videos at the age of 75 years (Additional file 1: Video S1a) and 80 years (Additional file 2: Video S1b). The patient did not show progression in gait speed and balance. Also, after 5 years, IMP-SPECT revealed improved blood flow images in the frontotemporal lobe. Additionally, the information provided by family members and medical examinations by physicians specializing in rehabilitation did not reveal any deterioration in the patient’s swallowing function or decrease in her activities of daily living.
Discussion and conclusions
The patient in this case demonstrated improvement in the symptoms of Huntington’s disease after the administration of oral memantine according to video recordings and cerebral blood flow tests over 7 years. The cognitive and behavioral symptoms exhibited by patients with Huntington’s disease are more similar to those exhibited by patients with frontotemporal dementia than those exhibited by patients with Alzheimer’s disease [4, 5]. Therefore, in our patients, cerebral blood flow examination indicated Alzheimer’s-type dementia. In general, many patients with Huntington’s disease show cognitive decline. Over time, involuntary movements increase, and the patient’s communication ability is affected by the deterioration of voluntary cognitive writing ability and speech. Thus, although the patient may understand the content of the evaluator’s questions, involuntary movements causing dysarthria may impair expression and communication [5]. Therefore, we believe that, in patients with Huntington’s disease who are at an old, advanced age, IMP-SPECT should be performed. Because elderly people may exhibit different types of dementia (such as Alzheimer’s-type dementia, Lewy body dementia, and frontotemporal dementia). IMP-SPECT revealed that our elderly female patient with Huntington’s disease also had Alzheimer’s-type dementia.
Recently in Japan, tetrabenazine reportedly improved Huntington’s-disease-associated chorea; however, clinical manifestations other than chorea did not improve [6]. Tetrabenazine depletes monoamines (dopamine, serotonin, and noradrenaline) at nerve endings by selectively inhibiting monoamine vesicle transporter 2 at presynaptic receptors of the central nervous system [7]. The effect of tetrabenazine on chorea results mainly from dopamine depletion in the striatum, which is the main lesion site of Huntington’s disease. However, in our patient, the oral administration of this drug caused nausea and fatigue, and the drug had to be discontinued as a result. So, considering her dementia symptoms and IMP-SPCT results, we prescribed 5 mg of memantine because it has demonstrated its efficacy in Alzheimer’s-type dementia.
Memantine is an N-methyl-d-aspartate (NMDA) receptor inhibitor that is usually administered to improve cognitive function in Alzheimer’s-type dementia [8]. Recently, its additional effects such as the improvement of swallowing function, lip dyskinesia, and chorea-like symptoms have been reported [8]. Several patients with Huntington’s disease were administered 20 mg of memantine and demonstrated improvement thereafter [9]. Moreover, two memantine clinical trials are ongoing for Huntington’s disease [10, 11]. This drug has also been used in treating other neurodegenerative diseases. The NMDA receptor antagonist, amantadine, has long been used not only as an anti-parkinsonism drug but also in the treatment of dyskinesia [12].
Tetrabenazines, which are involved in the regulation of monoamine receptors, have recently been approved by the US Food and Drug Administration for treating tardive dyskinesia [13, 14] and implicate a possible mechanism for chorea in Huntington’s disease. Similarly, the administration of memantine, an NMDA receptor inhibitor, may alleviate motor symptoms because monoamines and NMDA receptor regulation are closely related in the brain.
In our patient, dysphagia and dysarthria were not exacerbated for 7 years after the oral administration of 5 mg of memantine. Most memantine clinical trials focusing on Huntington’s disease have been evaluations of cognitive improvement. To the best of our knowledge, no detailed observational study of motor symptoms has been conducted [10]. The patient’s older brother (II-5 in Fig. 1) developed the disease at about the same time as she did; however, it progressed more rapidly in her brother (over 5 years). He frequently had aspiration pneumonia and was unable to walk. Thereafter, he died of this pneumonia the third time it occurred, which was 5 years after the onset of the disease. Conversely, our patient (II-8 in Fig. 1) remained ambulatory and could maintain activities of daily living, even to the extent of taking a bath and using the toilet without assistance.
Memantine has been reported to induce clinical manifestations such as myoclonus [15]; conversely, it is effective against other motor symptoms and dystonia [16]. One of the sites targeted by memantine is thought to be the dysfunctional glutamatergic neurotransmitter system via the NMDA receptor [17, 18]. In our patient, a small amount of memantine may have been effective in slowing down the progression of Huntington’s disease.
Memantine was reportedly effective in a small number of patients with Huntington’s disease [9]; in patients with the HTT gene who are yet to develop symptoms, memantine may suppress the future worsening of symptoms [11]. Findings in our patient suggest that oral memantine has some influence on the motor symptoms of Huntington’s disease.
We recorded changes in the results of the IMP-SPECT and videos of the patient’s gait over 5 years. To the best of our knowledge, this is the first report stating that memantine has an ameliorating effect on chorea movements in Huntington’s disease. We believe this record will provide clinicians with valuable information regarding the diagnosis and treatment of this disease.
Supplementary Information
Additional file 1: Video S1. (a) Chorea symptom video on the date of diagnosis (at the age of 75 years).
Additional file 2: Video S1. (b) Chorea symptom video at the follow-up date (at the age of 80 years).
Acknowledgements
The authors thank Enago (www.enago.jp) for the English language review.
Abbreviations
- IMP-SPECT
N-isopropyl-p-iodoamphetamine–single-photon emission computed tomography
- NMDA
N-methyl-d-aspartate
Author contributions
KS performed data analyses and wrote the manuscript. MH, IO, MS, KY, AI, YM, and YY assessed patients clinically and revised the manuscript. KS, MH, MS, YM, SK, and YN reviewed and edited the manuscript for important intellectual content. KS designed the study, collected samples, assessed all patients clinically, and revised the manuscript.
Funding
The authors received a grant from The Research Foundation for Dementia of Osaka, which is a public funding agency. The funding body played a role in the collection and analysis of study data. And this work was supported by JSPS KAKENHI Grant Number JP12345678.
Availability of data and materials
Upon written request, the corresponding author will provide the data used to support the findings and conclusions.
Declarations
Ethics approval consent to participate
This study was conducted with the approval of the Ethics Committee of Kindai University, Osaka, Japan (number 16-011).
Consent for publication
Written informed consent was obtained from the patient for the publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Competing interests
The authors have no competing interests to declare.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ross CA, Aylward EH, Wild EJ, Langbehn DR, Long JD, Warner JH, et al. Huntington disease: natural history, biomarkers and prospects for therapeutics. Nat Rev Neurol. 2014;10:204–216. doi: 10.1038/nrneurol.2014.24. [DOI] [PubMed] [Google Scholar]
- 2.Liu D, Long JD, Zhang Y, Raymond LA, Marder K, Rosser A, PREDICT-HD Investigators and coordinators of the Huntington study group et al. Motor onset and diagnosis in Huntington disease using the diagnostic confidence level. J Neurol. 2015;262:2691–2698. doi: 10.1007/s00415-015-7900-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Samukawa M, Hirano M, Saigoh K, Kawai S, Hamada Y, Takahashi D, et al. PSP-phenotype in SCA8: case report and systemic review. Cerebellum. 2019;18:76–84. doi: 10.1007/s12311-018-0955-0. [DOI] [PubMed] [Google Scholar]
- 4.Peinemann A, Schuller S, Pohl C, Jahn T, Weindl A, Kassubek J. Executive dysfunction in early stages of Huntington’s disease is associated with striatal and insular atrophy: a neuropsychological and voxel-based morphometric study. J Neurol Sci. 2005;239:11–19. doi: 10.1016/j.jns.2005.07.007. [DOI] [PubMed] [Google Scholar]
- 5.Bates GP, Dorsey R, Gusella JF, Hayden MR, Kay C, Leavitt BR, et al. Huntington disease. Nat Rev Dis Primers. 2015;1:15005. doi: 10.1038/nrdp.2015.5. [DOI] [PubMed] [Google Scholar]
- 6.Huntington Study Group Tetrabenazine as antichorea therapy in Huntington disease: a randomized controlled trial. Neurology. 2006;66:366–372. doi: 10.1212/01.wnl.0000198586.85250.13. [DOI] [PubMed] [Google Scholar]
- 7.Ross CA, Tabrizi SJ. Huntington’s disease: from molecular pathogenesis to clinical treatment. Lancet Neurol. 2011;10:83–98. doi: 10.1016/S1474-4422(10)70245-3. [DOI] [PubMed] [Google Scholar]
- 8.Johanssen T, Suphantarida N, Donnelly PS, Liu XM, Petrou S, Hill AF, et al. PBT2 inhibits glutamate-induced excitotoxicity in neurons through metal-mediated preconditioning. Neurobiol Dis. 2015;81:176–185. doi: 10.1016/j.nbd.2015.02.008. [DOI] [PubMed] [Google Scholar]
- 9.Cankurtaran ES, Ozalp E, Soygur H, Cakir A. Clinical experience with risperidone and memantine in the treatment of Huntington’s disease. J Natl Med Assoc. 2006;98:1353–1355. [PMC free article] [PubMed] [Google Scholar]
- 10.Corey-Bloom J, Goldstein J. Study of memantine to treat Huntington’s Disease. ClinicalTrials.gov database Identifier: NCT00652457, 2008.
- 11.Leavitt B, Huntington Study Group. A trial of memantine as smptomatic treatment for early Huntington Disease (MITIGATE-HD). ClinicalTrials.gov database Identifier: NCT01458470, 2011.
- 12.Elkurd MT, Bahroo LB, Pahwa R. The role of extended-release amantadine for the treatment of dyskinesia in Parkinson’s disease patients. Neurodegener Dis Manag. 2018;8:73–80. doi: 10.2217/nmt-2018-0001. [DOI] [PubMed] [Google Scholar]
- 13.Aguilar L, Lorenzo C, Fernández-Ovejero R, Roncero C, Montejo AL. Tardive dyskinesia after aripiprazole treatment that improved with tetrabenazine, clozapine, and botulinum toxin. Front Pharmacol. 2019;10:281. doi: 10.3389/fphar.2019.00281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cummings MA, Proctor GJ, Stahl SM. Deuterium tetrabenazine for tardive dyskinesia. Clin Schizophr Relat Psychoses. 2018;11:214–220. doi: 10.3371/CSRP.CUPR.010318. [DOI] [PubMed] [Google Scholar]
- 15.Kitagawa N, Takeuchi A. Memantine-induced myoclonus. Neurology. 2014;83:1387. doi: 10.1212/WNL.0000000000000863. [DOI] [PubMed] [Google Scholar]
- 16.Borges LG, Bonakdarpour B. Memantine-induced chorea and dystonia. Pract Neurol. 2017;17:133–134. doi: 10.1136/practneurol-2016-001470. [DOI] [PubMed] [Google Scholar]
- 17.Dau A, Gladding CM, Sepers MD, Raymond LA. Chronic blockade of extrasynaptic NMDA receptors ameliorates synaptic dysfunction and pro-death signaling in Huntington disease transgenic mice. Neurobiol Dis. 2014;62:533–542. doi: 10.1016/j.nbd.2013.11.013. [DOI] [PubMed] [Google Scholar]
- 18.Milnerwood AJ, Gladding CM, Pouladi MA, Kaufman AM, Hines RM, Boyd JD, et al. Early increase in extrasynaptic NMDA receptor signaling and expression contributes to phenotype onset in Huntington’s disease mice. Neuron. 2010;65:178–190. doi: 10.1016/j.neuron.2010.01.008. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Video S1. (a) Chorea symptom video on the date of diagnosis (at the age of 75 years).
Additional file 2: Video S1. (b) Chorea symptom video at the follow-up date (at the age of 80 years).
Data Availability Statement
Upon written request, the corresponding author will provide the data used to support the findings and conclusions.