Abstract
A series of macrolide-lincosamide-streptogramin B (MLS)-resistant pneumococcal isolates of a variety of serotypes was examined and was found to contain Tn917-like elements by DNA-DNA hybridization. Like Tn1545, Tn917 also encodes an ermAM gene but does not mediate resistance to other antimicrobial agents. Furthermore, nucleotide sequence analyses of the DNAs flanking three of the Tn917-like elements revealed that they were inserted into orf9 of a Tn916-like element in a composite transposon-like structure (Tn3872). Other MLS-resistant strains appeared to contain Tn1545-like elements that had suffered a deletion of sequences including the aphA-3 sequences responsible for kanamycin resistance. Thus, the MLS resistance phenotype in pneumococci appears to be mediated by the ermAM present on a much wider variety of genetic elements than was previously appreciated.
Erythromycin resistance in Streptococcus pneumoniae has been ascribed in part to the presence of ermAM, which encodes an rRNA methylase similar to that encoded by ermB of staphylococci (65). Erythromycin MICs for pneumococci harboring ermAM are typically ≥64 μg/ml. A second mechanism of erythromycin resistance in pneumococci, an efflux pump encoded by mefE (57), produces MICs in the range of 1 to 32 μg/ml. In some pneumococci, ermAM has been shown to reside on Tn1545, a 25.3-kb conjugative transposon that also encodes resistance to tetracycline via tet(M) and kanamycin via aphA-3 (6, 13). Tn1545 belongs to a larger class of transposons that includes Tn916, a conjugative transposable element that is widely disseminated among a variety of bacterial species (10, 52, 53). Tn1545/Tn916-like elements have been described in a variety of gram-positive genera, including enterococci, streptococci, and staphylococci, as well as Bacteroides ureolyticus and Ureaplasma urealyticum (8–10, 14, 18, 40, 47, 48, 52).
Recently, LeBlanc and colleagues (31) used DNA-DNA hybridization to demonstrate that some erythromycin-resistant isolates of pneumococci contained Tn917-like sequences. Tn917 is a 5.257-kb transposable element that contains the ermAM gene but does not encode resistance to either tetracycline or kanamycin (54, 59). This transposon is also widely distributed in enterococci of human and animal origin (30, 49).
The goal of this study was to determine if a Tn917-like element was present in erythromycin-resistant strains of pneumococci representing a number of well-documented clones. Our results suggest that Tn1545, deleted versions of Tn1545, and a Tn917-like element that is part of a larger composite transposon-like structure (Tn3872) are all responsible for macrolide-lincosamide-streptogramin B (MLS)-type resistance in S. pneumoniae.
MATERIALS AND METHODS
Bacterial strains.
Twelve strains of S. pneumoniae were obtained from the culture collection of the Centers for Disease Control and Prevention and from the University of Texas Health Science Center at San Antonio. Isolates were selected on the basis of their antimicrobial resistance profiles, serotypes, and pulsed-field gel electrophoresis (PFGE) patterns to represent a broad array of clonal types. Pneumococcal isolate BM4200 (5), containing the conjugative transposon Tn1545 (13), was kindly provided by P. Courvalin (Institut Pasteur, Paris, France). This isolate originally tested serotype 23F, but in our hands it was nontypeable. Organisms were identified by standard methods (15, 51), and serotypes were determined by the Centers for Disease Control and Prevention.
Susceptibility testing.
The MICs of chloramphenicol, clindamycin, erythromycin, penicillin, and tetracycline were determined by the broth microdilution method with cation-adjusted Mueller-Hinton broth (Difco Laboratories, Detroit, Mich.) containing 5% lysed horse blood as described by National Committee for Clinical Laboratory Standards document M7-A4 (41) and elsewhere (25). Organisms were incubated for 18 to 20 h at 35°C in ambient air. S. pneumoniae ATCC 49619 was used for quality control. Resistance to kanamycin was presumed if the isolate grew on a sheep blood agar plate in the presence of 500 μg of kanamycin per ml.
PFGE.
Genomic DNA was prepared in situ in agarose blocks as described previously (37) and was digested with SmaI (Life Technologies, Gaithersburg, Md.). The fragments were resolved by PFGE in 1% agarose (SeaKem GTG agarose; FMC Bioproducts, Rockland, Maine) in 0.5× Tris-borate-EDTA buffer for 20 h at 14°C at 6 V/cm in a CHEF-DR II system (Bio-Rad Laboratories, Hercules, Calif.). The parameters were an initial pulse time of 1 s that was increased to 20 s.
Filter mating.
Overnight cultures of donor and recipient cells were transferred into 10 ml of prewarmed Todd-Hewitt broth containing 0.5% yeast extract and were grown for approximately 6 h to the late exponential phase (5 × 108 CFU/ml). The cells were harvested by centrifugation and were concentrated 10-fold in broth containing DNase I (50 μg/ml; Life Technologies) to give 5 × 109 CFU/ml. Donor and recipient cells were mixed in a 1:2 ratio, and 100 μl of the mating mixture was spread onto 0.22-μm-pore-size nitrocellulose membrane filters (Millipore Corp., Bedford, Mass.) that were placed on Mueller-Hinton agar plates with 5% sheep blood and DNase I. After 20 h of incubation at 35°C, the cells were washed from the filters in 500 μl of broth with DNase I, and 100 μl was spread onto selective agar plates containing either 4 μg of tetracycline or erythromycin per ml and either 1 mg of streptomycin per ml or 100 μg of rifampin per ml and 25 μg of fusidic acid per ml. Pneumococcal isolate BM4200 served as the positive control donor of Tn1545, and Enterococcus faecalis CG110, a plasmid-free JH2-2 transconjugant containing Tn916, was the positive control donor of Tn916 (20). Recipients were either streptomycin-resistant or rifampin- and fusidic acid-resistant variants of pneumococcal control isolate R6. Transconjugants were confirmed by testing for tet(M), ermAM, and aphA-3 by PCR and by PFGE profile analysis. The transfer frequency for each mating was calculated as the rate per donor CFU.
DNA isolation.
Genomic DNA was isolated from pneumococcal cultures by two methods: (i) the Puregene DNA isolation kit (Gentra Systems, Inc., Research Triangle Park, N.C.) was used according to the manufacturer’s directions, except that spheroplasting of the cells was aided by the addition of mutanolysin (10 U/ml) and lysozyme (2.5 mg/ml), and (ii) a mutanolysin extraction procedure was used followed by a modification of the salting out method described by Miller et al. (38). Plasmid DNA was isolated from streptococci by the method of Anderson and McKay (1) with modifications as described previously (32) and from Escherichia coli by the method of Birnboim and Doly (4).
PCR-based detection of ermAM, aphA-3, and tet(M).
Pneumococcal genomic DNA was assayed for the presence of ermAM, aphA-3, and tet(M) by PCR. The oligonucleotide primers used for the amplification of a 550-bp segment of erm-AM were 5′ TTG GAA CAG GTA AAG GGC ATT 3′ (forward primer) and 5′ TTT GGC GTG TTT CAT TGC TTG 3′ (reverse primer). The first 5′ bases of the primers correspond to positions 431 and 981, respectively, in the published sequence (61). The 100-μl reaction mixture contained 50 mM KCl, 10 mM Tris-HCl (pH 8.3), 1.5 mM MgCl2, 1 μM oligonucleotide primers, 200 μM each deoxyribonucleoside triphosphate, and 2.5 U of native Taq DNA polymerase (Perkin-Elmer Applied Biosystems, Foster City, Calif.). The PCR assay was performed in a GeneAmp PCR System 9600 thermal cycler (Perkin-Elmer Applied Biosystems) with the following cycling parameters: initial denaturation at 94°C for 5 min followed by 35 cycles of 94°C for 1 min, 53°C for 1 min, and 72°C for 2 min. A 291-bp segment of aphA-3 corresponding to positions 988 to 1279 in the published sequence (60) was amplified with oligonucleotide primers aphA-3-1 and aphA-3-2 as described previously (62). A 1,249-bp segment of the tet(M) gene corresponding to positions 216 to 1464 in the published sequence of the gene associated with Tn1545 (35) was amplified with the oligonucleotide primers TETM1 (forward) and TETM3 (reverse) as described by Olsvik et al. (45).
Amplification and DNA sequence analysis of an internal fragment of tet(M).
Oligonucleotide primers TETM2 and TETM3 (45) were used to amplify, from genomic DNA, a 740-bp segment of the tet(M) gene corresponding to positions 725 to 1464 in the published sequence from Tn1545 (35). PCR products were purified with QIAquick PCR purification columns (Qiagen, Inc., Chatsworth, Calif.). Additional oligonucleotide primers identified for DNA sequencing were TETM (Up/1092; 5′ TGA AGT TAA ATA GTG TTC TTG G 3′) and TETM (Dn/1113; 5′ CCA AGA ACA CTA TTT AAC TTC A 3′). The first 5′ bases of the primers correspond to positions 1092 and 1113, respectively, in the published sequence of tet(M) carried by Tn1545 (35). Cycle sequencing was performed in a GeneAmp PCR System 9600 thermal cycler with the ABI Prism dRhodamine Terminator Cycle Sequencing Ready Reaction Kit (Perkin-Elmer Applied Biosystems). These products were analyzed on an ABI Prism 377 DNA Sequencer (Perkin-Elmer Applied Biosystems). In order to eliminate errors that may have been introduced during amplification, the DNA sequences of leading and lagging strands were determined with products from independent PCRs.
Probes and Southern hybridization.
The ermAM probe consisted of a 1.7-kb SstI-HindIII fragment containing ermAM of pAMβ1 originally present in pCER110 (50). The probe for Tn917 was pAM225 (32), composed of pBR325 plus a 5.6-kb insert containing Tn917. To avoid cross hybridization of vector DNA on blots containing authentic Tn917 sequences, the latter consisted of plasmid pAM238 (32) composed of the streptococcal plasmid pVA380-1 with the same 5.6-kb Tn917-containing insert as pAM225. 32P labeling of the ermAM probe was by random priming, and that of pAM225 containing Tn917 was by nick translation; both procedures were done according to the procedures of Life Technologies, Gaithersburg, Md.
DNA probes were also prepared from fragments generated by PCR with ermAM, aphA-3, and tet(M) intragenic primers, excised from agarose gels, and purified with the Spin-X centrifuge unit (Costar, Cambridge, Mass.). The amplification products were labeled with digoxigenin by use of a randomly primed DNA labeling reaction (Boehringer Mannheim Corp., Indianapolis, Ind.). Agarose gel electrophoresis of endonuclease-cleaved DNA prepared in situ in agarose blocks, preparation of Southern blots, and hybridization under stringent conditions were all performed as described previously (32, 36).
Inverse PCR and DNA sequence analysis.
The DNA region flanking Tn917 was obtained by inverse PCR by the method of Ochman et al. (43), with the following modifications. S. pneumoniae genomic DNA was digested with HindIII (Life Technologies). Fragments were ligated under conditions favoring the production of monomeric circles. (The DNA concentration in the ligation reaction was <0.5 μg/ml). The ligated monomeric circles were used as DNA templates for PCR and were amplified with the Expand High-Fidelity PCR system (Boehringer Mannheim Corp.), as suggested by the supplier. Primers L2 (5′-CTC ACA ATA GAG AGA TGT CAC GCT C-3′) and HDL4 (5′-TCG CTT GGC TAA AAG ACA TTC CAG G-3′) and primers R2 (5′-GCC TTG AAA CAT TGG TTT AGT GGG A-3′) and HDR5 (5′-TTC CGT CTG AAG CAG TGG TT-3′) were synthesized (Center for Advanced DNA Technology, University of Texas Health Science Center at San Antonio) and were used to amplify the left and right Tn917 junction fragments, respectively. The resulting inverse PCR products were purified with the Wizard PCR Preps Purification System (Promega Corp., Madison, Wis.) and were submitted to the Center for Advanced DNA Technology (University of Texas Health Science Center at San Antonio) for nucleic acid sequencing. Primers L2 and R5 were used in the initial sequencing reactions. Primers for the reverse-strand sequencing reactions were synthesized on the basis of the results for the initial sequences that were obtained. The nucleotide sequences were analyzed with the MacDNASIS sequence analysis software package (Hitachi Software, San Bruno, Calif.). Both the nucleotide sequences and the predicted amino acid sequences were compared to sequences in the National Center for Biotechnology Information databases by using the BLAST network service.
Nucleotide sequence accession numbers.
The DNA sequences of the left and right junctions of Tn917 have been assigned GenBank accession no. AF066796 and AF066797, respectively.
RESULTS
Bacterial strains.
The organisms and their relevant characteristics are presented in Table 1. Erythromycin MICs for all isolates were ≥64 μg/ml, and all isolates were also clindamycin resistant (MICs, ≥64 μg/ml). It was not possible to determine the induction of erythromycin and clindamycin resistance because none of the isolates produced a zone of inhibition around erythromycin and clindamycin disks. All isolates with the exception of isolate E42 were resistant to tetracycline; isolate E42 was susceptible to tetracycline, yet it still had tet(M). Chloramphenicol resistance was variable.
TABLE 1.
Properties of resistant clinical isolates of S. pneumoniae
| Isolate no. | Serotype/clonea | Antibiogramb | PCR result
|
Size(s) (kb) of AvaI fragment(s) in Tn917c | ||
|---|---|---|---|---|---|---|
| ermAM | tet(M) | aphA-3 | ||||
| E1 | 23F/Spanish | PN, CM, TC, EM, CC | + | + | − | 2.3, 1.60, 1.27 |
| E2 | 23F/Spanish | PN, CM, TC, EM, CC | + | + | − | 2.3, 1.75, 1.27 |
| E16 | 6B/Spain-Iceland | PN, CM, TC, EM, CC | + | + | − | 2.3, 1.75, 1.27 |
| 1003 | Nontypeable | PN, TC, EM, CC | + | + | − | 2.3, 1.75, 1.27 |
| E41 | Nontypeable | PN, TC, EM, CC | + | + | − | 2.3, 1.75, 1.27 |
| BM4200 | Nontypeable | PN, CM, TC, EM, CC | + | + | + | 22.5 |
| E6 | 23F/Spanish | PN, CM, TC, EM, CC | + | + | − | 16 |
| E12 | 6B/Spain-Iceland | PN, CM, TC, EM, CC | + | + | − | 22 |
| E42 | 19A | PN, EM, CC | + | + | − | 22 |
| E43 | 6B/Spain-Iceland | PN, TC, EM, CC | + | + | − | 19 |
| 3543 | 6B/Spain-Iceland | PN, CM, TC, EM, CC | + | + | − | 17.5 |
| 2649 | 6B/Spain-Iceland | PN, CM, TC, EM, CC | + | + | − | 22 |
Clonal type relatedness was determined by PFGE.
The antibiogram indicates resistance to the indicated drugs. The resistance breakpoints were as follows: penicillin (PN), MIC ≥ 2 μg/ml; chloramphenicol (CM), MIC ≥ 8 μg/ml; tetracycline (TC), MIC ≥ 8 μg/ml; erythromycin (EM), MIC ≥ 1 μg/ml; clindamycin (CC), MIC ≥ 1 μg/ml.
Molecular size(s) of AvaI-digested genomic DNA fragments hybridized with digoxigenin-labeled Tn917.
SmaI digests of genomic DNA from serotype 23F isolates E1, E2, and E6 produced PFGE patterns similar to that of the multidrug-resistant, serotype 23F Spanish clone, while serotype 6B isolates E16, E12, E43, 3543, and 2649 had SmaI PFGE profiles that resembled that of the serotype 6B Spain/ Iceland clone. The PFGE banding patterns of the other isolates (isolates 1003, E41, E42, and BM4200), which were all nontypeable in our hands, were unique.
PCR studies.
All of the isolates were tested for the presence of ermAM, tet(M), and aphA-3 by PCR. Each was positive for ermAM and tet(M), including isolate E42, which was tetracycline susceptible, but only BM4200 was positive for aphA-3 (Table 1).
Hybridization studies.
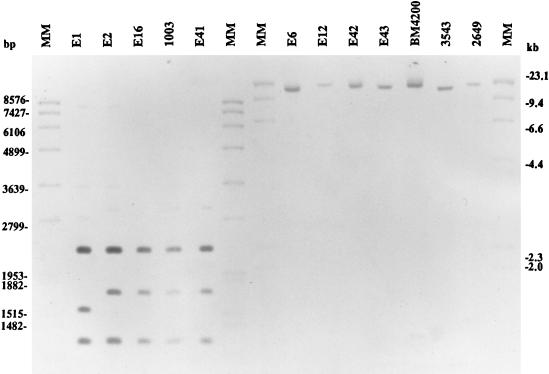
Genomic DNA was prepared from all of the isolates, digested with AvaI, separated on an agarose gel, transferred to nylon filters, and hybridized to a Tn917 probe. DNA from isolates E2, E16, E41, and 1003 contained three AvaI fragments that hybridized to the probe, and all three fragments were the same size as those predicted from the sequence of Tn917 (54) (Fig. 1). These three AvaI fragments account for 99.9% of authentic Tn917 (3, 12). For isolate E1 there was a slight variation in the size of the second fragment that hybridized to the Tn917 probe. This fragment represented the right-hand end of the transposon. Isolates BM4200, E6, E12, E42, E43, 3543, and 2649 demonstrated only a single large AvaI fragment that hybridized to the probe.
FIG. 1.
Southern blot of pneumococcal DNA digested with AvaI and hybridized with Tn917. MM, molecular mass marker.
DNA sequencing studies.
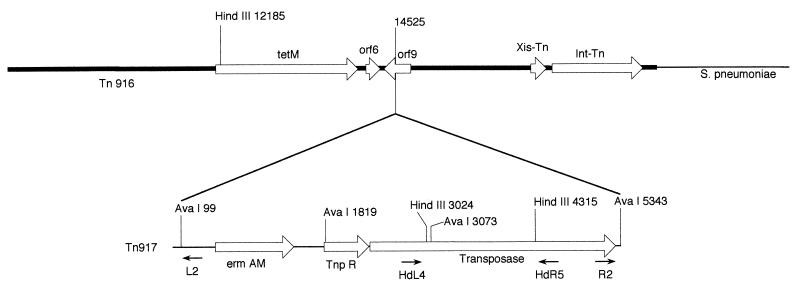
To determine whether the Tn917-like elements inserted into the same or random sites in the chromosomes of S. pneumoniae E1, E2, E16, and 1003, the right and left junctions of the putative Tn917 transposon within the chromosome were generated by inverse PCR (IPCR), and the DNA sequences were determined. After repeated attempts, we were not able to obtain the right end of the Tn917-like element of isolate E1. The Tn917-like element appeared to be inserted in a Tn916-like conjugative transposon at base 14525 (base numbers are from the sequence of Flannagan et al. [17]) and included a 5-base duplication (AGGTA) (Fig. 2 and Fig. 3A and B). The left ends of the Tn917-like elements of all four isolates was near tet(M) (orf11) of Tn916. The right ends of the Tn917-like elements of three of the isolates were near the end containing the int-Tn and xis-Tn functions. A search of the DNA data bank revealed that this Tn917-like element was inserted within a structure similar to orf9 of Tn916 (17), which is downstream of the transcription termination site of tet(M) (56). The sequences of the relevant regions of Tn916, Tn917, and the Tn917-like junction elements are shown with the IPCR fragments in Fig. 3A and B. The left ends of isolates E1, E2, E16, and 1003 yielded PCR products of similar sizes, and the sequences of these products were identical to each other; however, the right ends of isolates E2, E16, and 1003, which contained pneumococcal DNA, yielded products that varied in size, as follows: E2, 5.0 kb; E16, 4.0 kb; and 1003, 3.5 kb.
FIG. 2.
Schematic diagram showing insertion of Tn917 into Tn916 in S. pneumoniae. Relevant open reading frames (orfs) and restriction endonuclease sites are indicated. Numbers indicate nucleotide numbers based on GenBank sequences of Tn917 and Tn916. Location and the 5′ to 3′ direction of IPCR primers are indicated under the schematic for Tn917. Tn917 was inserted into orf9 at base 14525 in the published sequence of Tn916.
FIG. 3.
Nucleotide sequences of left and right IPCR fragments, Tn916 and Tn917, respectively. The beginnings and ends of orf6 and orf9 are as indicated. Uppercase letters indicate Tn916-derived sequences; lowercase letters indicate Tn917-derived sequences. Numbers at the right of the Tn916 and Tn917 sequences indicate nucleotide numbers based on GenBank sequences. The AvaI sites at the left and right termini of Tn917 are underlined. The positions of primers L2 and R2 are indicated by arrows below the sequence of Tn917. The 5-base target site duplication is boxed. (A) Sequence of the Tn917 left terminal junction fragment. (B) Sequence of the Tn917 right terminal junction.
To confirm that Tn917 was inserted into a Tn916-like element, a 740-bp segment from the middle of tet(M) from S. pneumoniae E2, E16, and 1003 was sequenced since this region shows 66-bp differences between the tet(M) carried by Tn1545 (35) and that carried by Tn916 (17). DNA sequence results were compared to those obtained for S. pneumoniae BM4200 (Tn1545) and E. faecalis CG110 (Tn916). Analysis of the tet(M) sequences obtained from isolate E2 showed that 50 of the 66 variable sites were identical to those found in Tn916, while 16 of the sites matched those found in Tn1545. E16 showed 29 Tn916-like sites and 37 Tn1545-like sites. Finally, for isolate 1003, 58 of the 66 variable sites were Tn1545-like, while the remainder matched those in the gene found in Tn916. The Tn916- and Tn1545-like segments of tet(M) found in these S. pneumoniae isolates appeared as relatively discrete blocks forming a mosaic pattern, as opposed to being scattered throughout the portion of the gene that was sequenced.
Filter matings.
We attempted to transfer the Tn916/Tn917-like element from isolates E1, E2, E16, and 1003 using S. pneumoniae R6 variants as recipients. S. pneumoniae BM4200 (Tn1545) and E. faecalis CG110 (Tn916) were used as positive mating controls. While Tn1545 transferred to R6 at a frequency of 8 × 10−8 transconjugants per donor cell and Tn916 transferred to R6 at a frequency of 5 × 10−7 transconjugants per donor cell, we did not detect the transfer of the cointegrate to strain R6 in multiple mating experiments.
DISCUSSION
In S. pneumoniae, drug resistance determinants such as those encoding erythromycin resistance usually are located on conjugative transposons that insert into the host chromosome rather than on plasmids, which are very rare in pneumococcal isolates (5). Erythromycin-resistant strains of S. pneumoniae are becoming more common in the United States and around the world (22, 26–28, 66). MLS resistance in pneumococci was assumed to be due to the presence of Tn1545, a conjugative transposon that encodes ermAM, in addition to tet(M) and aphA-3 (6, 13). Tn1545 shares a high degree of homology with Tn916, having termini identical to those of Tn916, a transposon that also encodes resistance to tetracycline but not to kanamycin or erythromycin (10, 52). Tn916 was the first conjugative transposon to be identified (10, 19) and is the prototype of a closely related family of conjugative transposons widespread in gram-positive cocci (10, 46, 53). The nucleotide base sequence of the entire transposon has been determined (17). In this study, we demonstrated the presence of the MLS resistance determinant (ermAM) on an element similar to the enterococcal transposon, Tn917, in several different strains of S. pneumoniae. In the three isolates examined, sequence data for the right and left junctions of the Tn917-like element demonstrate that this transposon inserted into the same site within the enterococcal conjugative transposon, Tn916, or a Tn916-like structure. We have designated this composite transposon Tn3872 and have selected E2 as the prototype strain.
Whether the integration of the Tn917-like element into Tn916 occurred in a strain of S. pneumoniae directly or occurred first in an enterococcal strain (or another bacterial species) and was transferred subsequently to a strain of S. pneumoniae cannot be determined from these results. However, we believe that the latter is the more likely explanation on the basis of the following observations. First, a Tn917-Tn916 composite has been generated experimentally in E. faecalis (designated Tn925::Tn917) (7) and was capable of transferring in broth matings to a Bacillus subtilis recipient, suggesting that the composite transposon could function as a conjugal delivery system for the Tn917 element. Second, it has been well established that the Tn916-Tn1545 family has a broad host range and that members of the Tn916 family of transposons can be transferred by conjugation to numerous bacterial species (11, 16, 21). In this study we demonstrated that Tn916 can be transferred from E. faecalis to the S. pneumoniae R6 recipient strain. Third, several of the other transposons in the Tn916-Tn1545 family have complex structures involving more than one transposable element in a composite arrangement (e.g., Tn5253 [2, 5, 63, 64], Tn3951 [24], and Tn3701 [23, 33, 34]). In each case, their extensive homology in the regions of DNA surrounding the tet(M) determinant suggests that Tn916 could have served as the precursor in the evolution of these larger elements (2, 7, 11, 20, 44).
Several other questions with regard to the dissemination of the transposons in pneumococci remain. The first concerns whether the three strains in this study contain the same composite transposon, Tn3872. Although the limited sequence data for the Tn917 junctions suggest this to be the case, the sequence data for tet(M) from three isolates suggest either that the origins of tet(M) may be different or that rearrangements of tet(M) occurred after the composite was introduced into its pneumococcal host. When the sequence data for nine tet(M) genes (44), including the tet(M) of Tn1545 from S. pneumoniae BM4200 and tet(M) of Tn916 from E. faecalis, were aligned with the tet(M) sequence data for the three Tn3872 isolates, a striking mosaic pattern of tet(M) sequences from Tn916 and Tn1545 was revealed. Of the 66 variable nucleotide positions observed, all but 2 of the sites for all nine organisms aligned with either tet(M) of Tn916 or tet(M) of Tn1545. While tet(M) from S. pneumoniae E2 (a member of the 23F Spanish clone) had more sites aligning with tet(M) of Tn916 (from E. faecalis CG110 [17]), the tet(M) of S. pneumoniae 1003 resembled the tet(M) of Tn1545 (from pneumococcal isolate BM4200 [35]). The variable sites present in the first half of tet(M) from E16 (6B Spain/Iceland clone) were the same as those in the tet(M) from Tn1545, whereas the variable sites of the second half were the same as those in tet(M) of Tn916. Although there are no direct data indicating whether the heterogeneity of the tet(M) locus occurred before or after integration of the element into pneumococci, we speculate that the latter is the case and that the mosaic structure of tet(M) evolved through homologous recombination in the pneumococcus after integration of Tn3872 into the host chromosome. In support of this, Norgren and Scott (42) have shown that the presence of Tn916 on the chromosome does not prevent the introduction of a second element into the cell and that up to 10% of incoming elements recombine with the resident transposon, resulting in mosaics of various transposon-encoded genes.
Has the introduction of Tn3872 into the pneumococcal chromosome occurred on multiple occasions? Since the left junction fragment of the Tn917-like insertion appeared to be identical in all three strains and was composed entirely of Tn917- and Tn916-derived sequences and the right junction fragments were all of different sizes and included pneumococcal chromosomal DNA, we speculate that Tn3872 has been transferred as a composite transposon to at least these three different strains of S. pneumoniae. However, it is possible that since the three isolates were from different pneumococcal clones, this region of the chromosome represents an area where mutational events may be more common, possibly resulting in the fragment length polymorphisms at the right junction fragment of Tn3872 that we observed. Further sequence analysis will help to differentiate between minor mutations in the same region of insertion or between totally different insertion sites in three different strains of S. pneumoniae. The possibility that Tn3872 has inserted by homologous recombination rather than by transposition into the same region of the chromosome of three different strains of S. pneumoniae must also be considered.
Although we were able to transfer Tn916 from an enterococcal donor to a pneumococcal recipient and Tn1545 from one pneumococcus to another pneumococcus in filter matings, we were unable to transfer the composite transposon Tn3872 under the same conditions. It is possible that the transfer frequency of Tn3872 was too low to be detected, or it may be that Tn3872 is incapable of transferring in pneumococci either because the insertion of Tn917 into orf9 has inactivated the mobility of Tn916 or because the inserted Tn3872 has subsequently suffered a deletion that resulted in a loss of mobility (58).
Among the isolates in this study are several that belong to the multiple-antimicrobial agent-resistant serotype 23F lineage known as the “Spanish clone” (36, 39). Two of these, E1 and E2, appear to have acquired the Tn3872 sequences. The appearance of erythromycin resistance has been noted before in this clone (29, 36); however, the genetic determinant responsible for this resistance was not reported. The Tn3872 element was also found in the 6B Spain/Iceland clone (55) and in several nontypeable isolates of pneumococci from different geographic regions. Thus, acquisition of this composite element may not be an uncommon event.
Six additional clinical isolates from the United States have what may be modified forms of Tn916 and Tn1545 on the basis of the size of the hybridization products and the lack of internal AvaI sites. They also lack the aphA-3 kanamycin resistance gene. This group of isolates also contains representatives of the Spanish 23F and Spain/Iceland serotype 6B clones. Our data suggest that the ermAM determinant has entered into these and other clones on multiple occasions on at least two different types of transposons, i.e., a composite Tn3872 transposon and a Tn916/Tn1545-like element. While composite transposons, such as Tn5253, previously known as Ω (cat tet) (2, 8), have been reported to reside in pneumococci, this is the first report of such a compound element in pneumococci that mediates erythromycin resistance associated with transposon Tn917. The ability of pneumococci to harbor and transmit composite transposons may explain, in part, the plethora of multiple-drug-resistant strains now being observed around the globe.
ACKNOWLEDGMENTS
We thank Terry Thompson and Bertha Hill for technical assistance.
REFERENCES
- 1.Anderson D G, McKay L L. Simple and rapid method for isolating large plasmid DNA from lactic streptococci. Appl Environ Microbiol. 1983;46:549–552. doi: 10.1128/aem.46.3.549-552.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ayoubi P, Kilic A O, Vijayakumar M N. Tn5253, the pneumococcal Ω (cat tet) BM6001 element, is a composite structure of two conjugative transposons, Tn5251 and Tn5252. J Bacteriol. 1991;173:1617–1622. doi: 10.1128/jb.173.5.1617-1622.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Banai M K, LeBlanc D J. Streptococcus faecalis R plasmid pJH1 contains an erythromycin resistance transposon (Tn3871) similar to transposon Tn917. J Bacteriol. 1984;158:1172–1174. doi: 10.1128/jb.158.3.1172-1174.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Birnboim H C, Doly J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 1979;7:1513–1523. doi: 10.1093/nar/7.6.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Buu-Hoi A, Horodniceanu T. Conjugative transfer of multiple antibiotic resistance markers in Streptococcus pneumoniae. J Bacteriol. 1980;143:313–320. doi: 10.1128/jb.143.1.313-320.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Caillaud F, Carlier C, Courvalin P. Physical analysis of the conjugative shuttle transposon Tn1545. Plasmid. 1987;17:58–60. doi: 10.1016/0147-619x(87)90009-6. [DOI] [PubMed] [Google Scholar]
- 7.Christie P J, Korman R Z, Zahler S A, Adsit J C, Dunny G M. Two conjugation systems associated with Streptococcus faecalis plasmid pCF10: identification of a conjugative transposon that transfers between S. faecalis and Bacillus subtilis. J Bacteriol. 1987;169:2529–2536. doi: 10.1128/jb.169.6.2529-2536.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Clermont D, Horaud T. Genetic and molecular studies of a composite chromosomal element (Tn3705) containing a Tn916-modified structure (Tn3704) in Streptococcus anginosus F22. Plasmid. 1994;31:40–48. doi: 10.1006/plas.1994.1005. [DOI] [PubMed] [Google Scholar]
- 9.Clewell D B, An F Y, White B A, Gawron-Burke C. Streptococcus faecalis sex pheromone (cAM373) also produced by Staphylococcus aureus and identification of a conjugative transposon (Tn918) J Bacteriol. 1985;162:1212–1220. doi: 10.1128/jb.162.3.1212-1220.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Clewell D B, Flannagan S E, Jaworski D D. Unconstrained bacterial promiscuity: the Tn916-Tn1545 family of conjugative transposons. Trends Microbiol. 1995;3:229–236. doi: 10.1016/s0966-842x(00)88930-1. [DOI] [PubMed] [Google Scholar]
- 11.Clewell D B, Gawron-Burke C. Conjugative transposons and the dissemination of antibiotic resistance in streptococci. Annu Rev Microbiol. 1986;40:635–659. doi: 10.1146/annurev.mi.40.100186.003223. [DOI] [PubMed] [Google Scholar]
- 12.Clewell D B, Tomich P K, Gawron-Burke M C, Franke A E, Yagi Y, An F Y. Mapping of Streptococcus faecalis plasmids pAD1 and pAD2 and studies relating to transposition of Tn917. J Bacteriol. 1982;152:1220–1230. doi: 10.1128/jb.152.3.1220-1230.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Courvalin P, Carlier C. Transposable multiple antibiotic resistance in Streptococcus pneumoniae. Mol Gen Genet. 1986;205:291–297. doi: 10.1007/BF00430441. [DOI] [PubMed] [Google Scholar]
- 14.de Barbeyrac B, Dupon M, Rodriguez P, Renaudin H, Bébéar C. A Tn1545-like transposon carries the tet(M) gene in tetracycline resistant strains of Bacteroides ureolyticus as well as Ureaplasma urealyticum but not Neisseria gonorrhoeae. J Antimicrob Chemother. 1996;37:223–232. doi: 10.1093/jac/37.2.223. [DOI] [PubMed] [Google Scholar]
- 15.Facklam R R, Carey R B. Streptococci and aerococci. In: Lennette E H, Balows A, Hausler W J Jr, Shadomy H J, editors. Manual of clinical microbiology. 4th ed. Washington, D.C: American Society for Microbiology; 1985. pp. 154–175. [Google Scholar]
- 16.Fitzgerald G F, Clewell D B. A conjugative transposon (Tn919) in Streptococcus sanguis. Infect Immun. 1985;47:415–420. doi: 10.1128/iai.47.2.415-420.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Flannagan S E, Zitzow L A, Su Y A, Clewel D B. Nucleotide sequence of the 18-kb conjugative transposon Tn916 from Enterococcus faecalis. Plasmid. 1994;32:350–354. doi: 10.1006/plas.1994.1077. [DOI] [PubMed] [Google Scholar]
- 18.Fletcher H M, Marri L, Daneo-Moore L. Transposon-916-like elements in clinical isolates of Enterococcus faecium. J Gen Microbiol. 1989;135:3067–3077. doi: 10.1099/00221287-135-11-3067. [DOI] [PubMed] [Google Scholar]
- 19.Franke A E, Clewell D B. Evidence for a chromosome-borne resistance transposon (Tn916) in Streptococcus faecalis that is capable of “conjugal” transfer in the absence of a conjugative plasmid. J Bacteriol. 1981;145:494–502. doi: 10.1128/jb.145.1.494-502.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gawron-Burke C, Clewell D B. A transposon in Streptococcus faecalis with fertility properties. Nature. 1982;300:281–284. doi: 10.1038/300281a0. [DOI] [PubMed] [Google Scholar]
- 21.Guild W R, Smith M D, Shoemaker N B. Conjugative transfer of chromosomal R determinants in Streptococcus pneumoniae. In: Schlessinger D, editor. Microbiology—1982. Washington, D.C: American Society for Microbiology; 1982. pp. 88–92. [Google Scholar]
- 22.Hofmann J, Cetron M S, Farley M M, Baughman W S, Facklam R R, Elliott J A, Deaver K A, Breiman R F. The prevalence of drug-resistant Streptococcus pneumoniae in Atlanta. N Engl J Med. 1995;333:481–486. doi: 10.1056/NEJM199508243330803. [DOI] [PubMed] [Google Scholar]
- 23.Horaud T, Delbos F, de Cespedes G. Tn3702, a conjugative transposon in Enterococcus faecalis. FEMS Microbiol Lett. 1990;72:189–194. doi: 10.1016/0378-1097(90)90370-6. [DOI] [PubMed] [Google Scholar]
- 24.Inamine J M, Burdett V. Structural organization of a 67-kilobase streptococcal conjugative element mediating multiple antibiotic resistance. J Bacteriol. 1985;161:620–626. doi: 10.1128/jb.161.2.620-626.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jorgensen J H, Swenson J M, Tenover F C, Ferraro M J, Hindler J A, Murray P R. Development of interpretive criteria and quality control limits for broth microdilution and disk diffusion antimicrobial susceptibility testing of Streptococcus pneumoniae. J Clin Microbiol. 1994;32:2448–2459. doi: 10.1128/jcm.32.10.2448-2459.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kanavaki S, Karabela S, Marinis E, Legakis N J. Antibiotic resistance of clinical isolates of Streptococcus pneumoniae in Greece. J Clin Microbiol. 1994;32:3056–3058. doi: 10.1128/jcm.32.12.3056-3058.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kell C M, Jordens J Z, Daniels M, Coffey T J, Bates J, Paul J, Gilks C, Spratt B G. Molecular epidemiology of penicillin-resistant pneumococci isolated in Nairobi, Kenya. Infect Immun. 1993;61:4382–4391. doi: 10.1128/iai.61.10.4382-4391.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Klugman K. Pneumococcal resistance to antibiotics. Clin Microbiol Rev. 1990;3:171–196. doi: 10.1128/cmr.3.2.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Klugman K P, Coffey T J, Smith A. Cluster of an erythromycin-resistant variant of the Spanish multiply resistant 23F clone of Streptococcus pneumoniae in South Africa. Eur J Clin Microbiol Infect Dis. 1994;13:171–174. doi: 10.1007/BF01982193. [DOI] [PubMed] [Google Scholar]
- 30.LeBlanc D J, Inamine J M, Lee L N. Broad geographical distribution of homologous erythromycin, kanamycin, and streptomycin resistance determinants among group D streptococci of human and animal origin. Antimicrob Agents Chemother. 1986;29:549–555. doi: 10.1128/aac.29.4.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.LeBlanc D J, Jorgensen J H, Patterson J E, Abu-Al-Jaibat A, Lee L N, McDougal L K, Tenover F C. Abstracts of the 96th General Meeting of the American Society for Microbiology 1996. Washington, D.C: American Society for Microbiology; 1996. Erythromycin-resistant Streptococcus pneumoniae isolates carrying a Tn917-like transposon, abstr. A107; p. 152. [Google Scholar]
- 32.LeBlanc D J, Lee L N. Characterization of two tetracycline resistance determinants in Streptococcus faecalis JH1. J Bacteriol. 1986;150:835–843. doi: 10.1128/jb.150.2.835-843.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Le Bouguenec C, de Cespedes G, Horaud T. Molecular analysis of a composite chromosomal conjugative element (Tn3701) of Streptococcus pyogenes. J Bacteriol. 1988;170:3930–3936. doi: 10.1128/jb.170.9.3930-3936.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Le Bouguenec C, de Cespedes G, Horaud T. Presence of chromosomal elements resembling the composite structure Tn3701 in streptococci. J Bacteriol. 1990;172:727–734. doi: 10.1128/jb.172.2.727-734.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Martin P, Trieu-Cuot P, Courvalin P. Nucleotide sequence of the tet(M) tetracycline resistance determinant of the streptococcal conjugative shuttle transposon Tn1545. Nucleic Acids Res. 1986;14:7047–7058. doi: 10.1093/nar/14.17.7047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.McDougal L K, Facklam R, Reeves M, Hunter S, Swenson J M, Hill B C, Tenover F C. Analysis of multiply antimicrobial-resistant isolates of Streptococcus pneumoniae from the United States. Antimicrob Agents Chemother. 1992;36:2176–2184. doi: 10.1128/aac.36.10.2176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.McDougal L K, Rasheed J K, Biddle J W, Tenover F C. Identification of multiple clones of extended-spectrum cephalosporin-resistant Streptococcus pneumoniae in the United States. Antimicrob Agents Chemother. 1996;39:2282–2288. doi: 10.1128/aac.39.10.2282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Miller S A, Dykes D D, Polesky H F. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1988;16:1215. doi: 10.1093/nar/16.3.1215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Muñoz R, Coffey T J, Daniels M, Dowson C G, Laible G, Casal J, Hakenbeck R, Jacobs M, Musser J M, Spratt B G, Tomasz A. Intercontinental spread of a multiresistant clone of serotype 23F Streptococcus pneumoniae. J Infect Dis. 1990;164:302–306. doi: 10.1093/infdis/164.2.302. [DOI] [PubMed] [Google Scholar]
- 40.Murray B E, An F Y, Clewell D B. Plasmids and pheromone response of the β-lactamase producer Streptococcus (Enterococcus) faecalis HH22. Antimicrob Agents Chemother. 1988;32:547–551. doi: 10.1128/aac.32.4.547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.National Committee for Clinical Laboratory Standards. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. 4th ed. 1997. Approved standard M7-A4. National Committee for Clinical Laboratory Standards, Wayne, Pa. [Google Scholar]
- 42.Norgren M, Scott J R. The presence of conjugative transposon Tn916 in the recipient strain does not impede transfer of a second copy of the element. J Bacteriol. 1991;173:319–324. doi: 10.1128/jb.173.1.319-324.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ochman H, Gerber A S, Hart D L. Genetic applications of an inverse polymerase chain reaction. Genetics. 1988;120:621–623. doi: 10.1093/genetics/120.3.621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Oggioni M R, Dowson C G, Smith J M, Provvedi R, Pozzi G. The tetracycline resistance gene tet(M) exhibits mosaic structure. Plasmid. 1996;35:156–163. doi: 10.1006/plas.1996.0018. [DOI] [PubMed] [Google Scholar]
- 45.Olsvik B, Tenover F C, Olsen I, Rasheed J K. Three subtypes of the tet(M) gene identified in bacterial isolates from periodontal pockets. Oral Microbiol Immunol. 1996;5:299–303. doi: 10.1111/j.1399-302x.1996.tb00185.x. [DOI] [PubMed] [Google Scholar]
- 46.Poyart-Salmeron P, Trieu-Cuot P, Carlier C, Courvalin P. The integration-excision system of the conjugative transposon Tn1545 is structurally and functionally related to those of lambdoid phages. Mol Microbiol. 1990;4:1513–1520. doi: 10.1111/j.1365-2958.1990.tb02062.x. [DOI] [PubMed] [Google Scholar]
- 47.Rice L B, Marshall S H, Carias L L. Tn5381, a conjugative transposon identifiable as a circular form in Enterococcus faecalis. J Bacteriol. 1992;174:7308–7315. doi: 10.1128/jb.174.22.7308-7315.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Roberts M C. Characterization of the tet(M) determinants in urogenital and respiratory bacteria. Antimicrob Agents Chemother. 1990;34:476–478. doi: 10.1128/aac.34.3.476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rollins L D, Lee L N, LeBlanc D J. Evidence for a disseminated erythromycin resistance determinant mediated by Tn917-like sequences among group D streptococci isolated from pigs, chickens, and humans. Antimicrob Agents Chemother. 1985;27:439–444. doi: 10.1128/aac.27.4.439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rubens C E, Heggen L M. Tn916ΔE: a Tn916 transposon derivative expressing erythromycin resistance. Plasmid. 1988;20:137–142. doi: 10.1016/0147-619x(88)90016-9. [DOI] [PubMed] [Google Scholar]
- 51.Ruoff K L. Streptococci. In: Murray P R, Baron E J, Pfaller M A, Tenover F C, Yolken R H, editors. Manual of clinical microbiology. 6th ed. Washington, D.C: American Society for Microbiology; 1995. pp. 299–307. [Google Scholar]
- 52.Salyers A A, Shoemaker N B, Stevens A M, Li L. Conjugative transposons: an unusual and diverse set of integrated gene transfer elements. Microbiol Rev. 1995;59:579–590. doi: 10.1128/mr.59.4.579-590.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Scott J R, Churchward G G. Conjugative transposition. Annu Rev Microbiol. 1995;49:367–397. doi: 10.1146/annurev.mi.49.100195.002055. [DOI] [PubMed] [Google Scholar]
- 54.Shaw J H, Clewell D B. Complete nucleotide sequence of macrolide-lincosamide-streptogramin B-resistance transposon Tn917 in Streptococcus faecalis. J Bacteriol. 1985;164:782–796. doi: 10.1128/jb.164.2.782-796.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Soares S, Kristinsson K G, Musser J M, Tomasz A. Evidence for the introduction of a multiresistant clone of serotype 6B Streptococcus pneumoniae from Spain to Iceland in the late 1980s. J Infect Dis. 1993;168:158–163. doi: 10.1093/infdis/168.1.158. [DOI] [PubMed] [Google Scholar]
- 56.Su Y A, Clewell D B. Characterization of the left 4 kb of conjugative transposon Tn916: determinants involved in excision. Plasmid. 1993;30:234–250. doi: 10.1006/plas.1993.1055. [DOI] [PubMed] [Google Scholar]
- 57.Sutcliffe J, Tait-Kamradt A, Wondrack L. Streptococcus pneumoniae and Streptococcus pyogenes resistant to macrolides but sensitive to clindamycin: a common resistance pattern mediated by an efflux system. Antimicrob Agents Chemother. 1996;40:1817–1824. doi: 10.1128/aac.40.8.1817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Swartley J S, McAllister C F, Hajjeh R A, Heinrich D W, Stephens D S. Deletions of Tn916-like transposons are implicated in tet(M)-mediated resistance in pathogenic Neisseria. Mol Microbiol. 1993;10:299–310. doi: 10.1111/j.1365-2958.1993.tb01956.x. [DOI] [PubMed] [Google Scholar]
- 59.Tomich P K, An F Y, Clewell D B. Properties of erythromycin-inducible transposon Tn917 in Streptococcus faecalis. J Bacteriol. 1980;141:1366–1374. doi: 10.1128/jb.141.3.1366-1374.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Trieu-Cuot R, Courvalin P. Nucleotide sequence of the Streptococcus faecalis plasmid gene encoding the 3′5′-aminoglycoside phosphotransferase type III. Gene. 1983;23:331–341. doi: 10.1016/0378-1119(83)90022-7. [DOI] [PubMed] [Google Scholar]
- 61.Trieu-Cuot R, Courvalin P. Nucleotide sequence of the erythromycin resistance gene of the conjugative transposon Tn1545. Nucleic Acids Res. 1990;18:3660. doi: 10.1093/nar/18.12.3660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Van de Klundert J A M, Vliegenthart J S. PCR detection of genes coding for aminoglycoside-modifying enzymes. In: Persing D H, Smith T F, Tenover F C, White T J, editors. Diagnostic molecular microbiology: principles and applications. Washington, D.C: American Society for Microbiology; 1993. pp. 547–552. [Google Scholar]
- 63.Vijayakumar M N, Priebe S D, Guild W R. Structure of a conjugative element in Streptococcus pneumoniae. J Bacteriol. 1986;166:978–984. doi: 10.1128/jb.166.3.978-984.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Vijayakumar M N, Priebe S D, Pozzi G, Hageman J M, Guild W R. Cloning and physical characterization of chromosomal conjugative elements in streptococci. J Bacteriol. 1986;166:972–977. doi: 10.1128/jb.166.3.972-977.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Weisblum B. Erythromycin resistance by ribosome modification. Antimicrob Agents Chemother. 1995;39:577–585. doi: 10.1128/AAC.39.3.577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Yoshida R, Kaku M, Kohno S, Ishida K, Mizukane R, Takemura H, Tanaka H, Usui T, Tomono K, Koga H, Hara K. Trends in antimicrobial resistance of Streptococcus pneumoniae in Japan. Antimicrob Agents Chemother. 1995;39:1196–1198. doi: 10.1128/aac.39.5.1196. [DOI] [PMC free article] [PubMed] [Google Scholar]