Abstract
Cancer stem-like cells (CSCs), a subpopulation of cancer cells, possess remarkable capability in proliferation, self-renewal, and differentiation. Their presence is recognized as a crucial factor contributing to tumor progression and metastasis. CSCs have garnered significant attention as a therapeutic focus and an etiologic root of treatment-resistant cells. Increasing evidence indicated that specific biomarkers, aberrant activated pathways, immunosuppressive tumor microenvironment (TME), and immunoevasion are considered the culprits in the occurrence of CSCs and the maintenance of CSCs properties including multi-directional differentiation. Targeting CSC biomarkers, stemness-associated pathways, TME, immunoevasion and inducing CSCs differentiation improve CSCs eradication and, therefore, cancer treatment. This review comprehensively summarized these targeted therapies, along with their current status in clinical trials. By exploring and implementing strategies aimed at eradicating CSCs, researchers aim to improve cancer treatment outcomes and overcome the challenges posed by CSC-mediated therapy resistance.
Keywords: Cancer stem cells, Tumor microenvironment, Immune evasion, Targeted therapies
Introduction
CSCs were firstly identified in acute myeloid leukemia (AML), where they exhibited stem cell-like and cancer cell-like properties and were found to be the sole cause of the initiation and progression of the corresponding cancer [1]. The tumorigenicity and self-renewal ability are indispensable properties of CSCs. To identify potential populations of CSCs, an important test was developed to determine the ability of CSCs to form tumors at low cell densities. This test, known as Extreme Limiting Dilution Assays (ELDA) became widely used as a gold standard to estimate active CSCs frequencies [2]. Studies have shown that as few as 100 cells exhibiting the CSCs phenotype were capable of forming tumors in mice, while tens of thousands of cells with an alternative phenotype were unable to do so [3]. Furthermore, CSCs must have the ability to sustain themselves and continue to generate cells with the same tumorigenicity and primitive tumor-forming capabilities [4]. A study provided evidence by using human cells with CD45 marker from the bone marrow of AML transplant recipients. These cells were found to have the same capacity to induce most subtypes of AML in secondary recipients, highlighting the self-renewal property of CSCs [5]. Unfortunately, CSCs are highly resistant to systemic anti-cancer therapies due to their complicated drug resistance mechanisms and active DNA repair capacity. Even after successfully resecting the primary tumor, the dormant disseminated CSCs can contribute tumor relapse and metastasis due to their long-term capacity for self-renewal [6].
Based on CSC theory, only a small subset of cells sustain tumorigenesis and contribute to cellular heterogeneity in primary tumors. These CSCs share certain properties of stem cells although they are not necessarily derived from stem cells found in normal tissues [7]. This suggests CSCs are a particular cell state that could initiate tumor growth. One widely accepted hypothesis is that cells can be transformed from more specialized, non-stem cells into stem-like cells through a process called epithelial-mesenchymal transition (EMT) [8]. During EMT, cancer cells undergo heritable phenotypic changes that are brought about by epigenetic modifications, rather than the introduction of new genetic alterations. As a result, the cancer cells lose their epithelial characteristics, such as cell–cell junctions and apical-basal polarity and gain mesenchymal features such as elongated, fibroblast-like morphology. These EMT-activated cancer cells exhibit CSC-like characteristics, including the expression of cell markers associated with stemness, as well as the ability to form tumors [9, 10]. The coexistence of both epithelial and mesenchymal characteristics allows cancer cells to survive, metastasize and colonize distal organs. Current research on CSCs focuses on understanding how these cells interact with the surrounding TME. This interaction involves various bidirectional cellular mechanisms, such as direct cell-to-cell contact, ligand-receptor interactions, and interactions with non-tumor cells that are present in the TME [11]. These interactions play a role in driving tumor progression. The chronic inflammatory and immunosuppressive TME is thought to be the primary factor contributing to EMT and the high stemness of CSCs [12]. Signals from the TME activate intracellular signaling pathways, leading to changes in biomarker expression, and promote immune evasion, which ultimately converge to maintain CSCs properties [13, 14].
Evidence suggests that conventional cancer therapies often fail to completely eliminate cancer cells that have undergone a switch to the CSC state. This switch is made possible through the activation of the EMT program, which can lead to CSC-related clinical recurrence. In light of these findings, eradicating or differentiating the CSC subpopulation appears to be a potential strategy for cancer treatment. However, despite these promising prospects, there is still a long way to go in comprehensively demonstrating the formation and development of CSCs, as well as in developing targeted therapies to counter them. This review focuses on current status of research and development in eradicating CSCs. The strategies discussed include cell biomarker-/pathway-targeting strategies, TME-targeting strategies, immune modification strategies, and agent-induced differentiation strategies. By examining these different approaches, this review aims to provide insight into the progress made thus far in the field of CSC-targeted therapies and to highlight the potential for future advancements in this area.
CSC biomarkers, stemness-associated pathways targeting therapies
Cell markers that distinguish CSCs from normal cells have potential applications in the diagnosis, treatment, and prognosis of cancer.
CSCs biomarkers
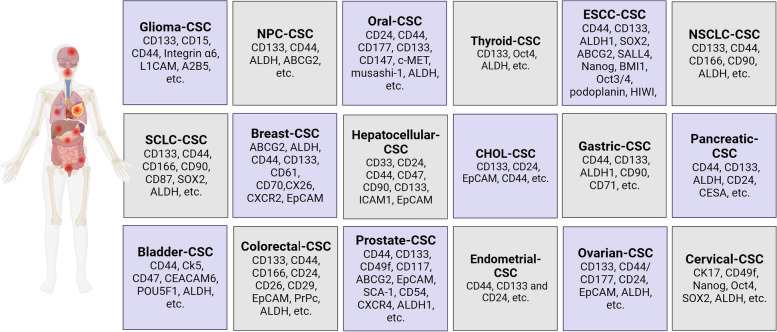
Different biomarkers have been utilized to identify CSCs, with cancer type-specific biomarkers being well-reviewed and showed in Fig. 1 [15–33]. This revealed common biomarkers for CSCs including CD44, CD133, aldehyde dehydrogenase (ALDH), and epithelial cell adhesion molecule (EpCAM).
Fig. 1.
Specific biomarkers for CSCs in different types of cancer
CD44 expression is upregulated in cancer cell subpopulations and serves as a molecular marker of CSCs. CD44+ cells isolated from cancer patients were capable of initiating tumor growth when transplanted into immunocompromised mice and showed increased resistance to radiochemotherapy [34]. CD44 is a crucial receptor for hyaluronic acid (HA) and extracellular matrix (ECM). Binding of HA or osteopontin to CD44 results in the activation of the STAT3, Oct4-Sox2-Nanog or c-Src kinase signaling pathways, which are known to promote anti-apoptosis and chemoresistance in CSCs [35]. In addition, CD44 also acts as a receptor for growth factors and cytokines such as transforming growth factor (TGF)-β, vascular endothelial growth factor (VEGF), and matrix metalloproteinase (MMP). Through these interactions, CD44 facilitates intercellular communication and mediates process such as EMT. This suggests that CD44 plays a key role in coordinating the cellular responses to various environmental signals to contribute to the self-renewal and invasiveness of CSCs [36]. Clinical studies have highlighted the clinicopathological role of CD44 in promoting tumorigenesis and have identified CD44 as a potential therapeutic target.
CD133, a pentosan membrane glycoprotein, is one of the most well-characterized biomarkers used for isolation of CSCs. It is found to promote tumorigenicity, spheroid formation ability, and the EMT program [37]. One significant finding is that cancer cells from patients with lung cancer that express CD133 could be maintained in specific media indefinitely. In contrast, cancer cells lacking CD133 died within a few weeks of culture [38]. Moreover, the CD133+ population consistently generated tumors in immunocompromised mice, but not CD133− cells [38]. These observations indicate the crucial role of CD133 in the maintenance and growth of cancer cells. Additionally, CD133 has been found to recruit histone deacetylase 6 (HDAC6) to deacetylate β-catenin, thereby causing the stabilization and nuclear localization of β-catenin. This, in turn, promotes the interaction between β-catenin and T-cell factors (TCF), resulting in the acceleration of cancer cell growth [39]. CD133 also interacted with p58 to activate the PI3K/Akt pathway, which further regulated CSC proliferation and tumorigenesis [40]. The combined expression of CD133 and CD44 in colorectal cancer has been shown a sevenfold increase in tumorigenicity. However, CD133 alone showed a change of more than 1.45 times, and CD44 alone led to a twofold increase in tumorigenicity [41]. Overall, these findings highlight the critical role of CD133 in the maintenance, growth, and tumorigenicity of cancer cells.
The EpCAM is considered as a multi-functional transmembrane protein associated with the regulation of cell adhesion, proliferation, migration, stemness, and EMT of cancer cells [42]. It served as an oncogenic signal transducer that is more accessible to be targeted by antibodies than those on normal cells [43]. The expression of EpCAM has been found to be tumorigenic, meaning that it is involved in the formation and growth of tumors. Transplantation of EpCAM+CD45− cells isolated from patients with hepatocellular carcinoma (HCC) into NOD/SCID mice triggered tumor formation, while EpCAM−CD45− cells did not have the same effect [44]. EpCAM controls cell cycle progression and differentiation via regulated intramembrane proteolysis (RIP). Following RIP-mediated EpCAM cleavage, the EpCAM intracellular domain (EpICD) is released and translocated into nucleus, where it binds to transcription factors and adaptor molecules such as four-and-a-half LIM domains protein 2 (FHL2), β-catenin, and TCF to initiate the stemness-related gene expression and cell reprogramming [45]. The binding of TCF/β-catenin complex, in turn, could bind to EpCAM promoter and regulate EpCAM gene expression [46]. The interaction of EpCAM and β-catenin signaling creates a positive feedback loop to promote the enrichment of the EpCAM+ cell population, which contributes to the promotion of cell self-renewal, differentiation, and invasiveness [47]. In addition, the overexpression of EpCAM has been found to promote EMT and upregulate cellular transcriptional factors such as Nanog, Oct4 and SOX2 [48]. The high expression of EpCAM indicates poor differentiation grade that directly associated with poor survival rate [49]. The above findings suggest that the measurement of EpCAM expression could serve as a prognostic marker for cancer patients and its antibody-targeted therapy could have potential application.
ALDHs are essential regulators of aldehyde metabolism in human body that protect organisms from active aldehydes-induced damage. ALDHs deficiency and polymorphisms in organisms are associated with diseases such as Parkinson’s disease, Type 2 hyperprolinaemia, hypertension and Sjögren-Larsson syndrome [50], while upregulated ALDH expression indicates a high degree of tumor malignancy [51, 52]. However, the exact mechanism of the effect by which ALDHs contribute to the maintenance of CSCs has not been clarified. Current research suggests that the interactions of ALDHs with retinoic acid (RA), reactive oxygen species (ROS), and reactive aldehydes may contribute to their functional roles [50]. ALDHs suppress the transformation of retinal to RA, which in turn reduce the ability of RA to inhibit clonogenic and tumorigenic potential [53]. The administration of all-trans RA (ATRA) inhibited the expression of ALDH1 and the activation of nuclear factor erythroid 2-like 2 (NRF2), resulting in the attenuation of CSC-like properties [54]. In addition, ALDHs regulate the expression of hypoxia-inducible transcription factors 2 (TIF-2) and decrease the ROS accumulation in tumor, thereby preventing CSCs apoptosis [55]. ALDHs can detoxify and metabolize cytotoxic chemotherapeutic drugs by oxidizing the aldehyde groups of chemotherapeutic drugs into non-toxic carboxylic acids. This is the main cause for CSCs maintaining drug resistance [56]. On the other hand, ALDH is associated with Nanog expression. It’s reported that the activity of ALDH is regulated by Nanog through the Notch1/Akt signaling pathway to induce CSC stemness and cellular radioresistance [57]. SOX2/Oct4 interacts with the Wnt/β-catenin signaling pathway to maintain CSC stemness and therapy resistance [58, 59], and as an upstream regulator of Nanog, SOX2/Oct4 is thought to be involved in modulating ALDH activity to control CSC properties. Suppression of ALDH activity can induce apoptosis and alleviate drug resistance [60].
Although the number of CSC markers identified is growing, it has been reported that not all of these markers are equally effective in identifying CSCs. On this case, investigation that focuses on stemness-associated CSCs signaling networks is necessary to comprehensively understand the role of CSCs in tumor progression.
Stemness-associated pathways
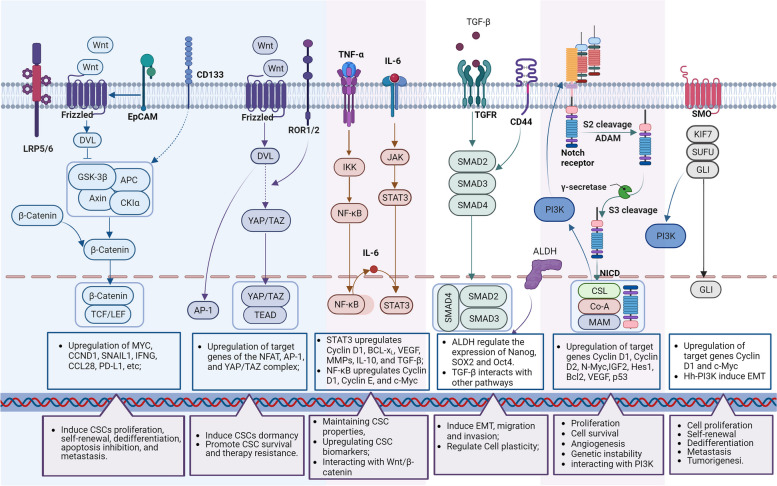
Deregulated signaling pathways controlling self-renewal and differentiation of CSCs are identified during CSC-induced tumor initiation, including Wnt/β-catenin, Notch, Hedgehog (Hh), and Hippo signaling pathways. In addition, these self-renewal related pathways interact with other oncogenic signaling pathways, including the nuclear factor-κB (NF-κB), signal transducer and activator of transcription 3 (STAT3), and PI3K/Akt signaling pathways, to inhibit apoptosis and promote cell proliferation [61] (Fig. 2).
Fig. 2.
Contribution of aberrantly activated pathways in CSCs. Dysregulated pathways activate the transcriptional activity of target genes, thereby contributing to the maintenance of the proliferation/dormancy, survival, self-renewal, migration, and evasion properties of CSCs
Wnt signaling pathway
The canonical Wnt signaling pathway, also known as Wnt/β-catenin signaling, is activated when Wnt ligands bind to Frizzled (FZD) family receptors and LRP5/6 coreceptors. This binding then transduces signals into the β-catenin signaling cascade. In the absence of canonical Wnt ligands, the β-catenin complexes are phosphorylated and subsequently degraded in the cytoplasm [62]. However, in the presence of Wnt ligands, the binding of Wnt ligands and receptors induces the accumulation of β-catenin in cytoplasm, which is then phosphorylated by glycogen synthase kinase 3β (GSK3β). The phosphorylated β-catenin translocates to the nucleus to activate the transcription of CCND1 and MYC directly, followed by upregulation of SNAIL1, IFNG, CCL28, PD-L1, and others, to initiate the EMT process and regulate cell progression and differentiation [63]. Canonical Wnt signaling is also reported to interact with canonical TGF-β pathway to induce the development of EMT [64]. The accumulated β-catenin in the nucleus reinforces canonical Wnt signaling in response to TGF-β reactions [65]. Both nuclear β-catenin and TGF-β repress the expression of E-cadherin gene. Loss of E-cadherin expression during EMT is believed to promote metastasis by allowing cancer cells to dissociate and invade [66, 67].
In contrast, the non-canonical Wnt signaling cascade induces CSC dormancy [68]. In the process of Wnt/PCP signal transduction, ROR1 receptors induce RhoA-mediated YAP stabilization and nuclear translocation, reprogramming LGR5 + proliferating CSCs into LGR5-dormant CSCs [69]. The ROR2 receptor induces CSC dormancy by inhibiting canonical Wnt signaling especially through promoting the degradation of β-catenin [70]. It is worth noting that proliferative CSCs are more vulnerable to DNA damage caused by chemotherapy, while non-proliferative CSCs are more resistant to such treatments [71]. Both canonical and non-canonical Wnt signaling pathways contribute to therapeutic resistance and subsequent relapse occurs due to the expansion of proliferative CSCs and the survival of dormant CSCs.
Notch signaling pathway
The canonical Notch cascade operates by direct cell-to-cell contact. This contact is initiated when transmembrane ligands expressed by signal-sending cells ligate receptors expressed on signal-receiving cells [72]. The ligand-mediated activation induces a series of proteolytic cleavages in Notch family receptors and releases the receptors’ NICD [73]. The NICD then translocates to the nucleus to initiate the Notch target genes transcription [74].
It is important to note that abnormal Notch signaling has been found to facilitate self-renewal and metastasis of CSCs [75]. Additionally, there is a crosstalk between Notch signaling and other pathways, such as TGF-β and EGFR. Activated Notch pathway leads to an expansion of TGF-β-induced smad2/3 signaling and EGFR inhibitors resistance [14, 76]. Notably, the pathogenic role of the Notch pathway appears to be tumor type dependent. Aberrant activation of Notch signaling promotes the development of most solid tumors but suppresses myeloid malignancies [77, 78]. Therefore, a comprehensive demonstration of the mechanisms of Notch signaling in specific cancers is essential for the successful development of therapeutics targeting this pathway.
Hh signaling pathway
Accumulating evidence suggests that aberrant Hh activation leads to tumor transformation, progression, and therapeutic resistance in a variety of cancers. The Hh signaling drives cancer progression by regulating cancer cell proliferation, malignant transformation, metastasis, and the CSC expansion [79]. Upon binding of the Hh ligands, such as Sonic (SHh), India (IHh), or Desert (DHh) to their receptors, the Hh signaling pathway is activated to initiate GLI transcription factors. The nucleus localization of GLI drives the expression of Hh target genes, most of which are associated with proliferation, cell survival, angiogenesis, and genetic instability [80]. The relatively high expression of GLI1, GLI2, PTCH1, and Hedgehog interacting protein (Hip) in CSCs indicates that the Hh signaling is preferentially activated in this cellular compartment [81].
The Hh signaling pathway has been shown to crosstalk with other oncogenic pathways in many cancer types, such as RAS/RAF/MEK/ERK, PI3K/Akt, EGFR, and Notch [82]. KRAS has been reported to activate Hh signaling by modulating the expression, phosphorylation, and degradation of GLI1. Simultaneous activation of RAS/RAF/MEK/ERK and Hh pathway increases the proliferation of CSCs and their potential for tumor formation. Activation of the PI3K/Akt pathway promotes GLI1 phosphorylation and nuclear translocation, which in turn increases the expression of GLI1 target genes and its oncogenic function. EGFR cooperates with Hh signaling to enhance CSCs attributes. The combined activation of EGFR and Hh pathway synergistically promote tumor formation, in part due to EGFR-GLI regulated RAF/MEK/ERK target JUN/AP-1 expression. JUN/AP-1 interacts with GLI protein to activate downstream GLI/EGF target genes.
Hippo signaling pathway
Hippo pathway is a conserved signaling that modulates cell proliferation, differentiation, and survival. The dysregulation of the Hippo pathway can cause a variety of diseases including cancer [83]. The Hippo pathway status controls the dynamic localization of YAP/TAZ between nucleus and cytoplasm. Once activated, the Hippo pathway limits cell proliferation through phosphorylation and degradation of YAP/TAZ in cytoplasm. In contract, when the Hippo pathway is off, YAP/TAZ translocates into nucleus and binds with TEAD to induce cell proliferation, survival and migration [84]. As highly tumor-associated transcriptional regulators, YAP/TAZ are pervasively activated in human malignancies and initiates the growth of most solid tumors [85]. In addition, Hippo signaling and subsequent activation of YAP/TAZ is a major mechanism involved in CSC therapy resistance and therefore a potential target for CSC eradication [86].
Interactions between self-renewal pathways and oncogenic pathways
Self-renewal signaling pathways contribute to cell proliferation, survival, and differentiation properties in CSCs. In the meanwhile, oncogenic pathways including NF-κB, STAT3, and PI3K/Ak signaling pathways, participate cell stemness regulation by controlling their downstream gene expression, such as cytokines, growth factors, apoptosis genes in CSCs [61].
The NF-κB signaling pathway (including canonical and alternative NF-κB signaling) plays a crucial role in the regulation of inflammation and immune system, and its overactivation is associated with cancer progression by promoting cell proliferation, survival, angiogenesis and invasion [87]. Researches demonstrated that both canonical and alternative NF-κB and their targeted genes are upregulated in the majority cancers [88, 89]. The increased transcriptional activity of NF-κB-targeted genes leads to the upregulated expression of Cyclin D1, Cyclin E, and c-Myc, which are responsible for cell proliferation [90]. What’s more, NF-κB participates in the regulation of a variety of carcinogenic mechanisms, including enhancing inflammatory responses through secretion of tumor necrosis factor (TNF)-α, IL-1β, IL-6, MCP1, COX2, and iNOS; promotion of EMT by the expression of vimentin and Twist; remodeling the extracellular matrix through induction of angiogenesis factors (e.g., IL-8 and VEGF); and facilitating invasion and metastasis through MMPs [91].
STAT3 signaling has been shown to have central role in CSCs in promoting cell proliferation, survival, tumor invasion, angiogenesis, and immunosuppression [92]. IL-6 acts directly on cancer cells to induce the expression of STAT3 target genes, including Cyclin D1, BCL2-like protein 1 (BCL-xL), VEGF, MMPs, IL-10, and TGF-β, thereby contributing to the maintenance of CSC properties [93]. Activated STAT3 is essential for maintaining the expression of CSC biomarkers such as CD24, CD34, CD38,CD44, CD90, CD133, and ALDH [94]. Additionally, induction of EMT and expansion of the CSC population were observed followed by STAT3 activation [95]. NF-κB and STAT3 are major factors regulating CSC angiogenesis and invasiveness during cancer progression, and their activation and interaction play an important role in controlling communication between cancer cells and inflammatory cells [96]. NF-κB is a key transcription factor that drives the expression of IL-6 [97]. In particular, NF-κB is overexpressed in multiple human cancers and activated STAT3 in tumor also induce IL-6. This creates a positive-feedback loop and suggests that IL-6 may be the basis for NF-κB and STAT3 interaction [98]. Wnt/β-catenin is involved in the IL-6 feedback loop to provides an important basis for inflammation-induced tumorigenesis [99]. The Wnt ligands are upregulated in inflammatory tissues and produce IL-1β and IL-6 by acting on the transcriptional factors NF-κB and STAT3. These cytokines activate STAT3, which facilitates Wnt ligands production and to Wnt/β-catenin pathway activation [68].
PI3K/Akt pathway is a highly conserved major transduction network in cells that promotes cell survival, growth and proliferation [100]. Dysregulation of PI3K/Akt pathway is achieved by variety of mechanisms, including loss or inactivation of the tumor suppressor PTEN, mutation or amplification of PI3K, and activation of tyrosine kinase growth factor receptors or oncogenes upstream of PI3K [101]. Increasing evidence indicated that activation of PI3K in cancers is associated with cancer progression by enhancing CSC phenotype, EMT, and therapy resistance [102]. Downregulation of PTEN induces PI3K activation to promote cell survival, maintenance of stemness, and tumorigenicity of prostate cancer stem-like cell population [103]. Indeed, important crosstalk and interactions between Notch signaling and PI3K/Akt pathway has been observed. Overactivation of PI3K/Akt pathway can upregulate the expression of Notch 1 ligand via NF-κB. Notch 1 in turn supports PI3K/Akt activity and prevents their dephosphorylation by inhibiting protein phosphatase 2 (PP2A) and PTEN activation, thereby supporting and promoting cancer progression [104]. Additionally, membrane-tethered Notch may activate the PI3K/Akt pathway to promote the transcription of IL-10 and IL-12 [105]. The cross talk between Hh and PI3K/Akt is a crucial regulator of EMT, Hh-GLI induces EMT and invasion and metastasis by activation of PI3K/Akt [106]. Furthermore, the mechanistic Target of Rapamycin (mTOR) is a downstream component of the KI3K/Akt pathway and has been reported to be involved in programmed cell death protein 1 (PD-1) expression through Hh signaling cascade, independent of SMO [107, 108].
Abnormal activation of Wnt, Notch, Hh, and Hippo pathways has been shown to facilitate the maintenance of CSC properties and promote tumor progression. Moreover, these pathways interact with other oncogenic cascades, such as RAF/MEK/ERK, PI3K/Akt, TGF-β, EGFR, STAT3, and NF-κB to further enhance the tumorigenicity of CSCs. Therefore, targeting the WNT, Notch, Hh, and Hippo pathways and related oncogenic pathways is crucial for the eradication of CSCs and the prevention of tumor progression.
Biomarker- and pathway-targeting strategies
Biomarker-targeting strategies
Selective targeting of CSCs is an effective strategy to inhibit cancer progression and reduce risk of tumor relapse. Clinical trials have shown that suppressing the expression of CSCs biomarkers can reduce CSCs stemness [109–118] (Table 1). The interaction between CD44 and HA is associated with poor prognosis, and CD44 has been shown to be an important biomarker for CSCs. RG7356, for example, is a recombinant anti-CD44 immunoglobulin G1 humanized monoclonal antibody. It binds specifically to the HA-binding region of the extracellular domain of all CD44 heterodimers, to inhibit the interaction between HA and CD44, and has exhibited growth inhibition effect on several tumors in vitro [109]. A randomized phase II trial of HA-irinotecan on CD44-expressing SCLC showed significant clinical benefit in patients, suggesting that delivery of chemotherapeutic agents to activated CD44, thereby abrogating the interaction of CD44 with HA, is a compelling approach to cancer treatment [110]. CD133 has been reported to play a role in tumor spread and is considered a good candidate for targeting CSCs. A phase II clinical study provides preliminary evidence that CART-133 cells have antitumor activity with a manageable safety profile in advanced HCC [118]. High expression of EpCAM has been observed on CSCs and targeting EpCAM is an effective strategy for cancer treatment. A phase I clinical trial indicated that VB4-845 successful blocks tumor growth in patients with high EpCAM expressing non-muscle-invasive bladder cancer [115]. However, although in vitro experiments have shown significant anticancer effects, clinical trials of adecatumumab, huKS-IL-2, and catumaxomab targeting EpCAM exhibited limited clinical benefits for cancer patients [114, 116, 117]. ALDHs have been evaluated as potential prognostic markers of cancer. In a clinical trial of curcumin and curcumin combined with 5-fluorouracil/oxaliplatin in patients with colorectal liver metastases (CRLM), curcumin alone and in combination significantly reduced the number of spheroids and ALDH-active cells. Curcumin enhanced anti-proliferation and apoptosis effects of 5-fluorouracil/oxaliplatin and reduced the expression of stem cell associated markers ALDH and CD133 [111]. Nevertheless, no clinical benefits were observed in phase II trials of paclitaxel (in combination with reparixin) and disulfiram [112, 113]. In summary, targeting CSC biomarkers to eradicate CSCs selectively is expected to be an effective strategy to inhibit cancer progression and reduce the risk of tumor relapse.
Table 1.
Clinical trials of different strategies for eradicating CSCs
| Therapies | Agents | Targets | Diseases | Clinical trial ID | Study phase | Ref. |
|---|---|---|---|---|---|---|
| Biomarker-targeting strategy | RG7356 | CD44 | Solid tumors | NCT01358903 | Phase I | [109] |
| HA-irinotecan | CD44 | Extensive-stage SCLC | / | Phase IIa | [110] | |
| CART-CD133 | CD133 | HCC | NCT02541370 | phase II | [118] | |
| Curcumin | ALDH | CRC | NCT01490996 | Phase I | [111] | |
|
Paclitaxel (with reparixin) |
ALDH | TNBC | NCT01861054 | Phase II | [112] | |
| Disulfiram | ALDH | Germ cell tumor | NCT03950830 | Phase II | [113] | |
| Adecatumumab | EpCAM | Hormone refractory prostate cancer | / | Phase I | [114] | |
| VB4-845 | EpCAM | Nonmuscle-invasive bladder cancer | / | Phase I | [115] | |
| huKS-IL2 | EpCAM | Advanced solid tumors | NCT00132522 | Phase Ib | [116] | |
| Catumaxomab | EpCAM | Epithelial cancer | NCT01320020 | Phase I | [117] | |
| Pathway-targeting strategy | WNT974 | Porcupine | CRC | NCT01351103 | Phase I | [119] |
|
Ipafricept (IPA) with nabpaclitaxel/ gemcitabine |
FZD | Pancreatic cancer | NCT01351103 | Phase Ib | [120] | |
|
Vantictumab (OMP‑18R5) with paclitaxel |
FZD | HER2-nagetive BC | NCT01973309 | Phase Ib | [121] | |
| E7449 | Tankyrase | Advanced solid tumor | NCT01618136 | Phase I | [122] | |
| CWP232291 | β-catenin complex | AML | NCT01398462 | Phase I | [123] | |
| Acylhydrazones | β-catenin complex | AML | NCT00990587 | Phase I | [124] | |
| Cirmtuzumab | ROR1 | Chronic lymphocytic leukemia | NCT02222688 | Phase I | [125] | |
| Collagenase (CHH) | YAP | Uterine fibroids | NCT02889848 | Phase I | [126] | |
| PF-03084014 | GSIs | Desmoid fibromatosis | NCT00878189 | Phase I | [127] | |
| OMP-59R5 | GSIs | Solid tumors | NCT01277146 | Phase I | [128] | |
| BMS-986115 | GSIs | Advanced solid tumor | NCT01986218 | Phase I | [129] | |
| RO2929097 | GSIs | High grade gliomas | NCT01119599 | Phase 0/I | [130] | |
| MK-0752 | GSIs | Pancreatic ductal adenocarcinoma | NCT01098344 | Phase I | [131] | |
|
RO4929097 (with Vismodegib) |
GSIs | Advanced sarcoma | NCT01154452 | Phase Ib/II | [132] | |
| LY900009 | GSIs | Advanced-stage cancer | NCT01158404 | Phase I | [133] | |
|
Demcizumab (with pemetrexed/ carboplatin) |
Notch ligand | NSCLC | NCT01189968 | Phase IB | [134] | |
| Rovalpituzumab tesirine | Notch ligand | Extensive-stage-SCLC | NCT03033511 | Phase III | [135] | |
| Enoticumab | Notch ligand | Advanced-stage solid tumors | NCT00871559 | Phase I | [136] | |
| Brontictuzumab | Notch receptor | Solid tumors | NCT01778439 | Phase I | [137] | |
| Tarextumab (with etoposide/cisplatin) | Notch receptor | Extensive-stage SCLC | NCT01859741 | Phase II | [138] | |
|
Vismodegib (GDC-0449) |
SMO | Advanced Basal-Cell Carcinoma | NCT00607724 | Phase I | [139] | |
| IPI-926 | SMO | Solid tumors | / | Phase I | [140] | |
| TAK-441 | SMO | Advanced solid tumors | NCT01204073 | Phase I | [141] | |
| PF-04449913 | SMO | Advanced solid tumors | NCT01286467 | Phase I | [142] | |
| LDE225 | SMO | Extensive stage SCLC | 01579929 | Phase I | [143] | |
| LY2940680 | SMO | Advanced/metastatic cancer | NCT01226485 | Phase I | [144] | |
|
Sarigegib (with cetuximab) |
GLI | Recurrent/metastatic head and neck squamous cell carcinoma | NCT01255800 | Phase I | [145] | |
| Itraconazole | GLI |
Biochemically relapsed prostate cancer |
NCT01787331 | Phase II | [146] | |
| AZD9150 | STAT3 | Lymphoma/lung cancer | / | Phase I | [147] | |
| OPB-111077 | STAT3 | Advanced Cancers | NCT01711034 | Phase I | [148] | |
| Alpelisib | PI3K | Epithelial ovarian cancer | NCT01623349 | Phase Ib | [149] | |
| Duvelisib | PI3K | T-cell lymphoma | NCT01476657 | Phase I | [150] | |
| TME-targeting strategy | Bintrafusp Alfa |
TGF-β/ PD-L |
NSCLC | NCT02517398 | Phase I | [151] |
| M7824 | TGF-β | Advanced solid tumors | NCT02517398 | Phase I | [152] | |
| Galunisertib | TGF-β | Advanced rectal cancer | NCT02688712 | Phase II | [153] | |
| FIGHT-101 |
FGF/ FGFR |
Advanced malignancies | NCT02393248 | Phase I/II | [154] | |
| Fruquintinib |
VEGF/ VEGFR |
metastasis CRC | NCT02314819 | Phase III | [155] | |
| Ramucirumab /Pembrolizumab versus Standard of care (SOC) |
VEGF/ VEGFR |
NSCLC | NCT03971474 | phase II | [156] | |
|
Rilotumumab (AMG 102) |
HGF | NSCLC | NCT02318368 | Phase I/II | [157] | |
| Siltuximab | IL-6 | Advanced solid tumors | / | Phase I/II | [158] | |
| Immune-targeting strategy | Neo-DCVac | DC | NSCLC | NCT02956551 | Phase I | [159] |
| DCs in combination of NAC-AC | DC | Breast cancer | NCT03450044 | Phase I/II | [160] | |
| DCs in combination of poly-ICLC | DC | Pancreatic cancer | NCT01410968 | Phase I | [161] | |
| DCVax-L plus SOC | DC | Glioblastoma | NCT00045968 | Phase III | [162] | |
|
Decitabine (with talazoparib) |
DNMT | AML | NCT02878785 | Phase I | [163] | |
|
Azacitidine (with pembrolizumab) |
DNMT | Colorectal cancer | NCT02260440 | Phase II | [164] | |
|
Guadecitabine (with GM-CSF) |
DNMT | Advanced colorectal cancer | NCT01966089 | Phase I | [165] | |
| Abexinostat | HDAC | Solid tumor malignancies | EudraCT-2009–013691-47 | phase II | [166] | |
| Panobinostat | HDAC | AML | / | Phase I | [167] | |
| Entinostat | HDAC | HER2 + metastatic BC | NCT02833155 | Phase I | [168] | |
| Tazemetostat | EZH2 | B-cell non-Hodgkin lymphoma | / | Phase II | [169] | |
| GSK2816126 | EZH2 | solid tumors or B-cell lymphomas | NCT02082977 | Phase I | [170] | |
| SHR2554 | EZH2 | Mature lymphoid neoplasms | NCT03603951 | Phase I | [171] | |
| Nivolumab (with chemotherapy) | PD-1 | Oesophageal adenocarcinoma | NCT02872116 | Phase III | [172] | |
| Pembrolizumab | PD-1 |
Advanced Colorectal Cancer |
NCT02563002 | Phase III | [173] | |
| Balstilimab/Zalifrelimab |
PD-1/ CTLA-4 |
Advanced cervical cancer | NCT03495882 | Phase II | [174] | |
| Tremelimumab | CTLA-4 | HCC | NCT01853618 | Phase I/II | [175] | |
| LY3415244 |
PD-L1/ TIM-3 |
Advanced solid tumors | NCT03752177 | Phase I | [176] | |
| Atezolizumab | PD-L1 |
Non-squamous NSCLC |
JapicCTI-184038 | Phase II | [177] | |
| Pembrolizumab | PD-L1 | NSCLC | NCT02142738 | Phase III | [178] | |
| Tiragolumab | PD-L1 | NSCLC | NCT03563716 | Phase III | [179] | |
| Induced differentiation strategy |
ATRA (with apatinib) |
Differentiation | Head and neck adenoid cystic carcinoma | NCT02775370 | phase II | [180] |
| ATRA | Differentiation | Adenoid cystic carcinoma | / | phase II | [181] | |
|
ATRA (with belinostat) |
Differentiation | Pancreatic cancer | NCT03307148 | Phase I | [182] | |
|
ATRA (with paclitaxel and interferon α2b) |
Differentiation | Cervical cancer | / | phase II | [183] | |
| IDH1-vac | IDH1 |
Newly diagnosed glioma |
NCT02454634 | phase I | [184] | |
|
Olutasidenib (FT-2102) |
IDH1 | Relapsed or refractory IDH1-mutant glioma | NCT03684811 | Phase Ib/II | [185] | |
| Ivosidenib | IDH1 |
Chemotherapy-refractory cholangiocarcinoma |
NCT02989857 | phase III | [186] | |
| Vorasidenib | IDH1/2 | Recurrent or progressive glioma | NCT02481154 | Phase I | [187] | |
| Temozolomide | IDH1/2 |
1p/19q non-co-deleted anaplastic glioma |
NCT00626990 | phase III | [188] | |
| Olaparib | IDH1/2 |
IDH1/IDH2-Mutant Mesenchymal Sarcomas |
NCT02576444 | phase II | [189] |
Pathway-targeting strategies
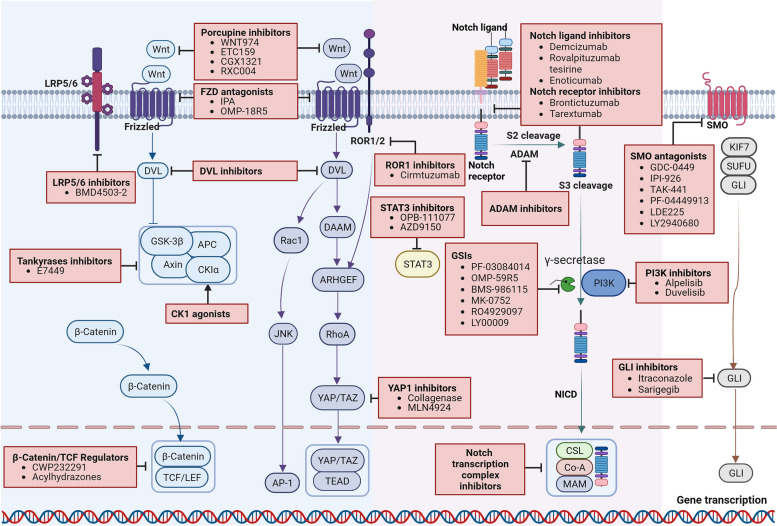
Developmental signaling pathways that regulate the maintenance and survival of CSCs are potential targets to eradicate CSCs, such as Wnt, Notch, Hh, and Hippo (Table 1, Fig. 3).
Fig. 3.
Illustration of abnormal pathways and potential targets in CSCs. FZD antagonists target either the Wnt proteins or FZD receptor complexes to inhibit the ligand-receptor interactions in both canonical and non-canonical Wnt pathway. DVL inhibitors block the DVL-PDZ interaction, resulting in subsequently inhibition of the signal transduction pathway. Tankyrase inhibitors stabilize Axin via inhibition of its proteasomal degradation, conversely resulting in increased degradation of β-catenin. CK1 agonists selectively potentiate CK1 kinase activity and stabilize the β-catenin destruction complex that decreasing Wnt signaling. β-catenin/TCF regulators inhibit Wnt-mediated transcriptional activity. LRP5/6 inhibitors competitively bind to the LRP5/6-sclerostin complex thus reverse the activation of Wnt/β-catenin signaling. ROR1-inhibitors ameliorate the access activated Wnt-signaling-mediated cancer cell proliferation, invasion, and therapy resistance. Potential anticancer therapeutic agents targeting the Notch pathway include targeting Notch ligands or receptors, inhibitors of the γ-secretase complex, and inhibitors of NICD-interacting transcriptional complex. SMO inhibitors block the Hh signaling by cyclopamine-competitively binding to SMO. GLI inhibitors prevent the transportation of GLI protein to nucleus thus decreased tumorigenesis gene expression. Inhibition of STAT3 and PI3K blocks their interactions with self-renewal pathways to facilitate CSCs eradication
Inhibition of Wnt signaling involves several critical steps, including blocking the secretion of Wnt ligands, interference with ligand-receptor binding, and modulation of intracellular signal transduction. Targeting these steps is crucial to inhibit Wnt signaling and its effects on CSCs. Porcupine inhibitors, which block the secretion of Wnt ligands, hold promise as ideal drugs for eliminating proliferative CSCs induced by canonical Wnt signaling and dormant CSCs induced by non-canonical Wnt signaling. Clinically, porcupine inhibitors have shown their therapeutic potential in various tumors. Currently, only 4 molecules, namely LGK974, ETC159, CGX1321, and RXC004, are undergoing Phase I clinical trials [119, 190]. However, the clinical use of these inhibitors may have a relatively narrow therapeutic window due to the involvement of the Wnt signaling cascade in the homeostasis of key organs and tissue [191]. Interfering with the binding of Wnt ligands to their receptors is another effective strategy to inhibit Wnt signaling. Wnt/FZD antagonists such as ipafricept (IPA; OMP54F28) and vantictumab (OMP-18R5) can directly bind to Wnt ligands or FZD receptors, competing with Wnt ligands for binding to FZD receptors [120, 121]. This competition inhibits Wnt regulatory processes in both the canonical and non-canonical Wnt pathways [192, 193]. Alternatively, blocking intracellular signal transduction may help to inhibit Wnt signaling. DVL inhibitors disrupt the interaction between DVL and PDZ, resulting in the subsequent inhibition of the signaling pathway [194, 195]. In canonical Wnt signaling, the LRP5/6 inhibitor BMD4503-2 competitively binds to the LRP5/6-sclerostin complex, thereby reversing Wnt/β-catenin pathway activation [196]. Stabilization of the β-catenin structural complex prevents the localization of β-catenin in the nucleus, making it an attractive therapeutic target. Tankyrases inhibitor, E7449, regulates the stability of AXIN by directing its ubiquitylation and proteasomal degradation, thereby increasing the activity of the destruction complex and reducing free β-catenin [122, 197]. CK1 agonists selectively potentiate CK1 kinase activity and stabilize the β-catenin destruction complex that decreasing Wnt signaling [198]. Finally, targeting the downstream effectors, such as β-catenin/TCF and β-catenin-dependent transcriptional activators, is another feasible strategy to inhibit Wnt-mediated transcriptional activity [199, 200]. Previous studies have suggested that compounds like CWP232291 and acylhydrazones block or disrupt the interaction between β-catenin and the TCF complex, suppressing Wnt target genes [123, 124]. In non-canonical Wnt signaling, ROR1-inhibitors ameliorate the access of a ctivated Wnt-signaling-mediated cancer cell proliferation, invasion, and therapy resistance [201]. Cirmtuzumab has shown obvious inhibitory effects on ROR1 expression and tumor progression in chronic lymphocytic leukemia patients [125].
Various potential anticancer therapeutics targeting the Notch pathway have been explored to eliminate CSCs including inhibiting the release of Notch ligands, blocking the proteolytic cleavage of Notch receptors, interrupting the Notch signaling transduction and inhibiting the expression of target genes associated with Notch signaling. Targeting Notch ligands or receptors can inhibit aberrant signaling initiation and decrease tumorigenesis in CSCs [135–138, 202]. Phase I Clinical trials have demonstrated the antitumor activity of DLL4-targeted agents, such as demcizumab and enotizumab, either as monotherapy or in combination therapy [134, 136]. Brontictuzumab, a Notch 1 inhibitor, has shown efficacy signal in patients with Notch 1 activation and combination therapy with other anticancer agents has improved clinical benefits [137]. However, Notch 2/3 receptor inhibitor tarextumab had limited additional effects when combined with gemcitabine and nab-paclitaxel in untreated advanced pancreatic adenocarcinoma [138]. ADAMs-catalyzed S2 cleavage occurs in the ligand-receptor binding domain, which mediates the release of the ectodermal structural domain and regulates the rate of Notch signaling [203]. Notch signaling from extracellular to intracellular relies heavily on the γ-secretase complex-mediated final cleavage. γ-secretase inhibitors (GSIs) block the S3 cleavage of Notch receptors, preventing the release of NICO and subsequent activation of Notch signaling [204]. Several GSIs including PF-03084014, OMP-59R5, BMS-986115, RO2929097, MK-0752, RO4929097, and LY900009, have been investigated in clinical trials [127–133]. However, dose-limiting toxicities have been observed with most GSIs. A potentially superior option to GSIs is the use of γ-secretase modulators (GSMs). GSMs modify the catalytic activity of γ-secretase rather than non-selectively inhibiting it, thereby preserving some Notch signaling function and theoretically reducing side effects [205]. Activating target gene transcription is the final step in Notch signaling transduction. Disrupting the Notch transcriptional complex downstream of abnormal Notch activation has advantages in repressing the expression of Notch targeted genes [206]. Although inhibition of the Notch pathway has shown significant anti-tumor efficacy in preclinical research, these results have not been consistently identified in clinical trials.
Inhibiting the expression of Hh ligands, SMO and GLI transcription factors is thought to be an effective way to suppress the over activation of the Hh signaling in CSCs. Clinical trials are currently underway for agents targeting these components [207]. Intracellular cyclopamine is known to inhibit the activity of the Hh pathway through directly binding and inactivating SMO. Cyclopamine derivatives also show SMO inhibitory effect by competitively binding to SMO, thereby blocking Hh signaling [208]. Several SMO inhibitors, including GDC-0449, IPI-926, TAK-441, PF-04449913, LDE225, and LY2940690 were under clinical trials, all of which showed anticancer effects through interrupting Hh signaling [139–144]. However, tumor cells frequently acquire resistance to SMO inhibitors through SMO mutation [209]. Furthermore, SMO antagonists have limited efficacy when aberrant Hh activation occurs due to genetic alterations downstream of SMO, or SMO-independent activation of GLI transcription factors. In this context, the development of GLI inhibitors is considered an alternative strategy [210]. In preclinical studies, GLI inhibitors have shown promising results. Encouraging anticancer effects was observed in Sarigerib treated recurrent/metastatic head and neck squamous cell carcinoma patients. Mechanical research indicated that the clinical response was associated with the regulation of Hh signaling pathway [145]. Importantly, targeting both SMO and GLI, in a synergistic inhibition of the Hh pathway, provides stronger efficacy than monotherapy [211]. Endoplasmic reticulum aminopeptidase 1 (ERAP1) plays a positive role in regulating Hh signaling. Inhibition of ERAP1 controls Hh-induced tumor growth, suggesting that ERAP1 is a promising therapeutic target in Hh over-activated tumors [212]. Continued research and clinical trials are necessary to further explore and optimize strategies targeting the above signaling pathway for the effective treatment of CSCs and related cancers.
Inhibition of YAP/TAZ activity is a potential strategy to eliminate CSCs, while the direct inhibitors of YAP/TAZ are still under development [86]. A clinical trial of collagenase on patients with uterine fibroids showed that collagenase decreased the expression of cell proliferation marker and phosphorylated YAP [126]. Inhibition of upstream Hippo kinases can be an attractive strategy to suppress tumor progression. Loss of LATS1/2 in tumor cells inhibits tumor growth and enhances anti-tumor immune response [213].
On the other hand, interfering the interaction between pathways might help to eradicate CSCs. STAT3 inhibitor, AZD9150, has showed preclinical activity in patients with highly treatment-refractory lymphoma and NSCLC in a phase I dose-escalation study [147]. OPB-111077, a novel inhibitor of STAT3 and mitochondrial oxidative phosphorylation, showed notable clinical activity in a subject with diffuse large B-cell lymphoma [148]. PI3K inhibitor, alpelisib, provided preliminary clinical activity in epithelial ovarian cancer [149]. Duvelisib (IPI-145) is an oral inhibitor, which demonstrated promising clinical activity and an acceptable safety profile in relapsed/refractory T-cell lymphoma [150].
TME and TME-targeting strategies
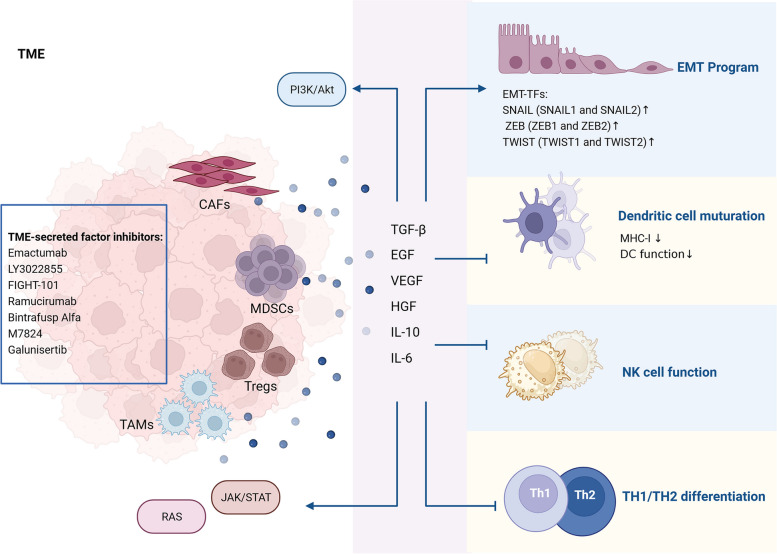
The TME play a crucial role in maintaining CSC properties and creating an inflammatory and immunosuppressive niche [214]. TME-induced chronic inflammation and immunosuppressive niche are closely related to the high cancer incidence. A prolonged and unresolved inflammatory response leads to the aberrant activation and accumulation of various stromal cells, which disrupts the normal function of stromal cells in maintaining homeostasis and promotes EMT activation and the formation of a tumorigenic environment. Furthermore, exosomes secreted from CSCs and stromal cells contribute to drug resistance and tumor immunosuppression environment formation, thus promoting CSC stemness [215, 216]. A properly functioning host immune system is essential to prevent inflammation-induced dysregulation of TME homeostasis. However, the recruitment and activation of stromal cells can suppress the host immune system [217]. On this basis, various strategies targeting EMT have been employed to eliminate CSCs (Table 1, Fig. 4).
Fig. 4.
TME-induced immunosuppressive environment for the maintenance of CSC properties. Cytokines and growth factors from CAFs, Tregs, MDSCs and TAMs activate cancer-related EMT and suppress DC cell maturation, NK cell function, and TH1/TH2 differentiation. Additionally, TME-derived factors activate cancer-associated pathways including PI3K/Akt, JAK/STAT, and RAS
The role of TME in CSC formation
Cancer associated fibroblasts (CAFs) within the TME play an integral role in maintaining CSCs and promoting drug resistance. Therapeutic agents have been developed to directly target surface markers on CAFs, such as PAF, S1004A, and TEM8, in order to control tumor growth [218]. Moreover, cytokines and growth factors secreted by CAFs, including TGF-β, IL-6, EGF, VEGF, and HGF, have been reported to facilitate tumor progression [219]. Within the TME, several types of immune cells contribute to chronic inflammation and promote an immunosuppressive environment. These include regulatory T cells (Tregs), myeloid-derived suppressor cells (MDSCs), and inflammatory tumor-associated macrophages (TAMs). Tregs are a subset of CD4+ T cells characterized by the expression of FoxP3 in the nucleus and CD25 and CTLA-4 on the cell surface. They exert significant immunosuppressive effects by producing immune inhibitory cytokines such as TGF-β and IL-10 as well as expressing immune checkpoint molecules like PD-1, CTLA4, LAG3, TIM3, and TIGIT [220]. TAMs are one of the most abundant infiltrating immune cells in TME [221]. Immunosuppressive factors from TAMs, such as TGF-β and IL-10, can stimulate the activity of Tregs and suppress the immune activity of T-cells, leading to immune escape. TAMs also secreted pro-inflammatory cytokines, such as IL-6, which activate signaling pathways like STAT3, PI3K/Akt, cyclooxygenase 2 (COX2), prostaglandin E2 (PGE2), β-catenin, and Ras-MARK pathways. Activating of these pathways alters the expression patterns of genes associated with proliferation, survival, and cell cycle regulation, thereby promoting cancer cell invasion and drug resistance [222, 223]. MDSCs support tumor progression through promoting cancer cell survival, angiogenesis, invasion, and metastasis. Expansion and recruitment of MDSCs driven by TME and their secreted inflammatory factors (e.g., TGF-β, VEGF, IL-10, IL-12, IL-13, etc.) mediate chronic inflammatory and immunosuppressive activity [224]. Elevated TGF-β has been proved to suppress the effector function of nature killer (NK) cell, hinder dendritic cell (DCs) function, and prevent TH1 and TH2 cell differentiation, while promoting TH17 and Treg cell programme [225]. Modulation of CAFs, Tregs, TAMs, and MDSCs may alleviate immunosuppressive and inflammatory TME, thus increase sensitivity of CSCs to treatment.
TME-targeting strategies
Regulation of the secreted growth factors and cytokines is one of the main strategies to modulate TME. Among these growth factors, TGF-β is one of the most extensively studied cytokines derived from TME. TGF-β signaling in stromal cells contributes to cancer progression by suppressing T-cell responses and regulating immune escape [225]. In addition, Non-canonical TGF-β has been reported to activate non-Smad pathways, including MAPK, YAP/TAZ, PI3K/Akt, and AMPK signaling, leading to fibrosis, immune evasion, and EMT, ultimately promoting cancer progression [226]. At present, TGF-β-targeting agents have been designed and have achieved satisfactory clinical activity. Clinical trials of M7824 and galunisertib in cancer patients have shown encouraging clinical efficacy by targeting TGF-β [152, 153]. EGF/EGFR interact with the MEK-ERK and AKT-PI3K signaling pathways, which leads to cancer cell proliferation [227]. Afatinib or olmutinib in combination with conventional chemotherapeutic agents improve CSC eradicating efficacy by inhibiting EGFR tyrosine kinase and ATP-binding cassette subfamily G member 2 (ABCG2) [228, 229]. VEGF/VEGFR interaction involves the activation of downstream pathways, including Ras-Raf-MAPK, AKT-mTOR, and Scr-FAK. Activation of these pathways promotes cell survival, proliferation, migration, and differentiation [227]. A randomized clinical trial indicated that fruquintinib, a VEGFR inhibitor, increased overall survival (OS) in patients with metastatic CRC [155]. Ramucirumab increased the sensitivity of cancer cells to immune checkpoint inhibitors (ICIs) and improved OS by inhibiting VEGF/VEGFR [156]. HGF from TME has been shown to regulate the innate resistance of BRAF-mutant cancer cells to RAF inhibitors by activating the MAPK and PI3K/Akt signaling pathways in cancer cells [230]. A phase I/II clinical trial evaluating rilotumumab, an anti-HGF antibody, in combination with erlotinib in patients with metastatic NSCLC showed that the combination of rilotumumab and erlotinib was more effective than erlotinib alone [157]. Suppressing TME-induced cytokines production can help restore antitumor immunity and sensitize tumors to immunotherapy. IL-6 plays a critical role in inflammation and cancer development. It has been reported that high levels of IL-6 confer resistance to cisplatin in patients with non-small-cell lung cancer (NSCLC) [231]. However, siltuximab, an anti-IL-6 monoclonal antibody, showed limited efficacy in patients with advanced solid tumors [158].
CSCs and immunoevasion
Tumor immunosurveillance is orchestrated by tumor immunogenicity and immunoevasion, immune cell infiltration, and T cell checkpoints [232]. Stromal cells and recruited immune cells in TME collectively formed an immunosuppressive niche that facilitates the maintenance and proliferation of CSCs. Furthermore, dysregulated cellular antigen processing and presentation machinery, as well as upregulated expression of immune checkpoint molecules allow CSCs to escape from immune surveillance (Fig. 5).
Fig. 5.
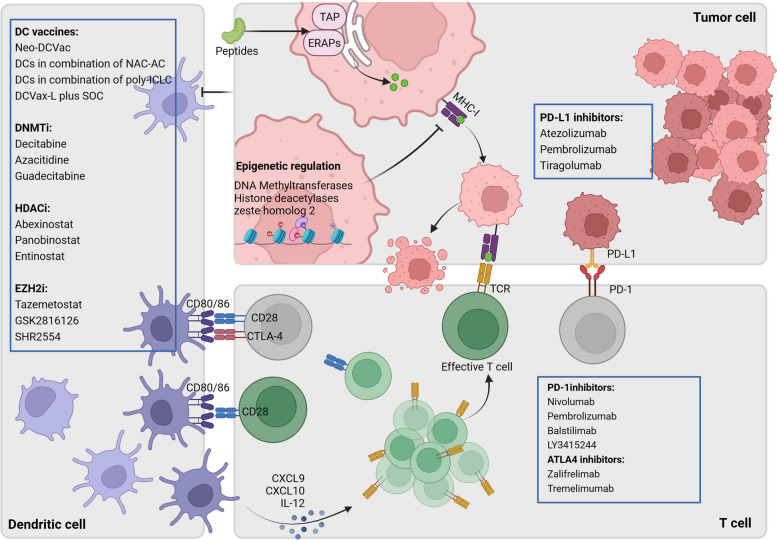
DC and T cell mediated tumor immunology and immunotherapy. In MHC-I antigen presentation pathway, oligopeptides degraded from cytosolic and nuclear protein are taken up and translocated into ER by TAP and further trimmed by ERAPs. The modified peptides bind to MHC-I and are transported to the cell surface for exposure to CD8 + T cells [233]. DC produce CXCL9, CXCL10 and IL-2 to recruit effective T cells therefore increase immune response. Activation of CTLA-4 and PD1/PDL1 reprogrammed immune homeostasis and induced cancer cells to eliminate T cell function. Immunotherapies that inhibit CTLA-4 and PD1/PDL1 and enhance MHC-I expression can effectively modulate DC function and improve cancer immunotherapy
CD8+ T lymphocytes play an indispensable role in regulating adaptive immune responses. They detect antigenic peptides bound to MHC-I perceptively and efficiently eliminate abnormal cells, thereby preventing cancer cell colonization. Stimulation with interferon-gamma (IFN-γ) can upregulate the expression of MHC-I antigen presentation components [233]. While these compositions (TAP, ERAPs, IFN-γ) are not strictly required for cell proliferation, their loss results in a reduction of pathway function and decreased cell surface levels of MHC-I molecules [234]. The presentation of cancer-associated peptide antigen by MHC-I is an important step for antitumor CD8 + T cell responses. However, CSCs have mapped out strategies to reduce antigen presentation thus to escape immune recognition, including inhibition of DC function and downregulation of MHC-I expression [235]. A growing number of studies demonstrated that CSCs alter DC phenotypes and impair their recruitment to limit them to activate T cells [236, 237]. On the other hand, activation of immune checkpoint pathways in the TME, including CTLA-4 and PD1/PDL1, reprograms immune homeostasis and enables cancer cells to evade immune attack [238]. ICIs that block CTLA-4 and PD1/PDL1 have demonstrated promising therapeutic efficacy in various human cancers. However, the effective antitumor responses of ICIs often require the secretion of IL-12 by DCs [239]. Additionally, ICIs induce, instead of relieve, T cell dysfunction in situation of low MHC-I levels [240]. In this basis, enhancement of antigen processing and presentation machinery and/or combination with ICIs may be an attractive strategy for CSCs eradication.
Type 1 conventional DCs (cDC1s) are a major subset of DCs that respond to invasive pathogens and antitumor immunity via antigen presentation to cytotoxic CD8+ T cells [241]. The cDC1s facilitate the differentiation and recruitment of tissue-resident memory CD8+ T cells within the TME by producing CXCL9 and CXCL10 to activate STING pathway. In addition, cDC1s-derived IL-12 increases the sensitivity of cancer cells to ICIs by augmenting CD8+ T cell activation. Adequate CD8+ T cell activation, in turn, induces the maturation and migration of cDC1s to the draining lymph nodes [235]. Therefore, DCs serve as a potent tool for motivating antitumor responses to eradicate CSCs effectively. DC vaccines are currently under clinical investigation and have shown promising therapeutic modality. DC vaccines, including Neo-DCVac, autologous DCs in combination with doxorubicin and cyclophosphamide (NAC-AC) or toll-like receptor agonist have demonstrated efficacy in restraining tumor progression by promoting T cell mediated immunity and sensitizing cancer cells to ICIs (Table 1) [159–162].
The binding of MHC-I with specific peptides and their presentation on the cell surface is an indispensable step in antitumor immunity. Loss expression of MHC-I renders CSCs invisible to the immune system. Clinical evidence has confirmed that advanced melanoma patients with low levels of MHC-I on the cancer cell surface derived limited benefit from ICIs therapy [242]. The expression level of MHC-I is dependent on NOD-like receptor family CARD domain containing 5 (NLRC5), which is regulated by IFNγ-activated STAT1 signaling [243]. Yet no clinical trial records regarding the regulation of MHC-I by NLRC5 in the PubMed database. The downregulated expression of MHC-I is associated with repressive histone modifications of Lys-27 in histone 3 (H3K27m3), including hypermethylation, histone deacetylation and trimethylation. These modifications are partly regulated by enhancer of zeste homolog 2 (EZH2). Accordingly, inhibition of DNA Methyltransferases (DNMTi), Histone deacetylases (HDACi), and EZH2 (EZH2i) theoretically has the potential to increase the expression of MHC-I [244]. The efficacy of these drugs, either as monotherapy or in combination with other treatments, has been evaluated in clinical trials. Some of these trials have demonstrated improved sensitivity of cancer cells to drug administration (Table 1) [163–169]. For instance, SHR2554, an EZH2 inhibitor, has shown promising antitumor activity in patients with relapsed or refractory follicular lymphoma, peripheral T-cell lymphoma, and classical Hodgkin lymphoma [171]. EZH2 inhibitor tazemetostat showed encouraging efficacy in patients with R/R EZH2 mutation-positive follicular lymphoma with a manageable safety profile in the overall population [169]. HDAC inhibitors including abexinostat, panobinostat, and entinostat have shown antitumor efficacy in clinical trials [166–168]. However, DNMT inhibitors, such as decitabine, azacitidine, and guadecitabine, have shown limited clinical activities on tested cancers [163–165].
T cells are activated through a complex interplay involving antigen specific T cell receptor recognition of peptides presented by MHC molecules and interactions between membrane proteins on antigen-presenting cells (APCs, including CD80 and CD86) and CD28 on T cells [245]. CTLA-4 inhibits the activation of T cells by competing with CD28 for binding to CD80 and CD86 on APCs, thereby attenuating CD8+ T cell responses. Another important immune checkpoint pathway involves the PD-1 and its ligand PD-L1. The expression of PD-1 on T cells and PD-L1 on tumor cells or other immune cells is considered a hallmark of T cell dysfunction and promotes T cell exhaustion [246]. Blockade of the expression of CTLA-4 and the interaction between PD-1 and PD-L1, has been shown to restore T cell function and enhance antitumor responses during chronic infections and in the TME [247]. ICIs specifically target these immune checkpoint molecules and modify the immune environment, have demonstrated the effectiveness of ICIs in various cancers (Table 1) [172–179]. Nivolumab and pembrolizumab, the PD1 inhibitors, have shown longer OS/FPS in multiple cancers compared to chemotherapy in clinical trials [172, 173]. Balstilimab (PD-1 inhibitor) and zalifrelimab (CTLA-4 inhibitor) are checkpoint inhibitors emerging as promising investigational agents for the treatment of advanced cervical cancer. A phase II clinical trial indicated that the combination of balstilimab and zalifrelimab had a high proportion of complete responses and efficacy in patients with recurrent and/or metastatic cervical cancer [174]. Tremelimumab is a fully monoclonal antibody that binds to CTLA-4 on the surface of activated T cells, which triggered the accumulation of intratumoral CD8 + cells in patients with advanced HCC [175]. PD-L1 inhibitors, such as atezolizumab, pembrolizumab, tiragolumab, improve objective response rates and are associated with significantly longer PFS and OS [177–179]. However, LY3415244, a TIM-3/PD-L1 inhibitor designed to overcome primary and acquired anti-PD-(L)1 resistance, was terminated early due to unexpected immunogenicity [176].
Agent-induced CSC differentiation
CSC-induced poorly differentiated cancers are more malignant, differentiation therapy is therefore a strategy to inhibit tumorigenesis by inducing the conversion of highly malignant undifferentiated cancer cells into benign differentiated cells. Several agents, including retinoic acid (RA), cAMP, sodium butyrate and cytokines, have been proved to induce cell differentiation in specific types of cancer. ATRA induces terminal differentiation and exhibits significant anticancer effect in patients with AML and acute promyelocytic leukemia (APL) [248]. Ongoing research is exploring the potential of ATRA and its derivatives for differentiation therapy in non-AML/APL [180, 181, 183]. New differentiation-inducing drugs that inhibit mutant isocitrate dehydrogenase (IDH) 1 and IDH2 have shown differentiating potential clinically, which have been approved for AML therapy. Mutations in IDH1 and IDH2 render their functional activity in differentiation regulation, particularly by increasing histone and DNA methylation [249]. Furthermore, epigenetic regulatory inhibitors such as DNMTi and HDACi, which promote MHC-I expression, have also demonstrated their involvement in cell differentiation. IDH1/2 mutations have been observed in solid tumors, and clinical trials are underway to evaluate the effectiveness of IDH1/2 inhibitors in inducing CSC differentiation in solid tumors. Continued research in differentiation therapy holds promise for expanding its application across various cancer types (Table 1) [184–189]. IDH1-Vaccine or IDH1-targeting agents including ivosidenib and olutasidenib showed clinical benefit in cancer patients in terms of reduced tumor burden and increased PFS [184–186]. Vorasidenib, a dual IDH1/2 inhibitor, showed preliminary antitumor efficacy in patients with recurrent or progressive nonenhancing mIDH lower grade gliomas [187].
Other strategies of target CSCs
Cancer is a complex disease that affects the health of people around the world. Chemotherapy remains an important treatment for cancer, despite advances in surgery and radiotherapy. Current treatments are expensive and are associated with many adverse events. In addition, cancer cells become resistant to chemotherapy as treatment progresses, making it difficult for patients to benefit from unmodified chemotherapy. The development of new drugs remains a top priority, but with the financial burden of drug research, drug repurposing is an innovative way to update the chemotherapy arsenal [250]. Metformin is the first choice for treating type 2 diabetes because of its robust glucose-lowering effects, well-established safety profile and relatively low cost [251]. However, metformin is repurposed as an anti-cancer agent. A clinical trial indicated that metformin has showed anticancer efficacy by inhibiting CD133 [252]. In addition, a phase II clinical trial showed that metformin treatment resulted in a significant reduction in the CSC population and alteration of DNA methylation of chemoresistance carcinoma-associated mesenchymal stem cells (CA-MSCs), which eliminated CA-MSC–driven increases in chemoresistance [253]. The drug repurposing strategy provides an alternative for cancer treatment.
Conclusion and future perspectives
Despite ongoing debates regarding the origin and specific characteristics of CSCs, it is widely acknowledged that these cells have exhibited stemness properties such as self-renewal, proliferation, differentiation, and therapy resistance. Therefore, targeting CSCs with different therapeutic agents holds great promise for future antitumor treatments. Currently, most cancer therapies only control the growth and proliferation of CSCs instead of completely eradicating the tumor bulk. In this context, researchers are exploring the modulation of abnormal signaling pathways, inhibition of CSC specific proteins, and regulation of the immune environment to gain new insights into cancer treatment strategies. However, several challenges remain a significant hurdle, as it is crucial to specifically target CSCs while minimizing damage to normal cells. Based on the plasticity and heterogeneity of CSCs, precision oncology will be the future trend of tumor therapy. Selecting the right combination of drugs for each patient and using them at the right stage of the disease require a comprehensive understanding of the biomarkers, stemness-associated pathways, TME, and immune mechanisms in CSCs. Additionally, efforts are ongoing to mitigate adverse effects associated with treatment, and explore innovative approaches for delivering therapeutic agents and maintaining effective drug concentrations. Continued research and development in these areas hold the potential to revolutionize cancer treatment by specifically targeting CSCs, overcoming therapy resistance, and achieving more comprehensive and durable therapeutic outcomes.
Acknowledgements
We use BioRender (https://biorender.com/) to create figures. The work is supported by the National Natural Science Foundation of China (U21A20421).
Abbreviations
- CSCs
Cancer stem cells
- TME
Tumor microenvironment
- AML
Acute myeloid leukemia
- ELDA
Extreme Limiting Dilution Assays
- EMT
Epithelial-mesenchymal transition
- ALDH
Aldehyde dehydrogenase
- EpCAM
Epithelial cell adhesion molecule
- HA
Hyaluronic acid
- ECM
Extracellular matrix
- TGF-β
Transforming growth factor-beta
- VEGF
Vascular endothelial growth factor
- MMPs
Matrix metalloproteinase
- HDAC6
Histone deacetylase 6
- TCF
T-cell factors
- RIP
Intramembrane proteolysis
- FHL2
Four-and-a-half LIM domains protein 2
- ROS
Reactive oxygen species
- RA
Retinoic acid
- TIF-2
Hypoxia-inducible transcription factors 2
- ATRA
All-trans retinoic acid
- NRF2
Nuclear factor erythroid 2-like 2
- FZD
Frizzled
- GSK3β
Glycogen synthase kinase 3β
- DVL
Disheveled
- ADAM
Disintegrin and metallo-proteinase
- NICD
Notch intracellular domain
- SHh
Sonic hedgehog
- IHh
India hedgehog
- DHh
Desert hedgehog
- SMO
Smoothened
- PTCH
Patch
- Hip
Hedgehog interacting protein
- GSIs
γ-Secretase inhibitors
- GSMs
γ-Secretase modulators
- ERAP1
Endoplasmic reticulum aminopeptidase 1
- CAFs
Cancer associated fibroblasts
- NSCLC
Non-small-cell lung cancer
- ABCG2
ATP-binding cassette subfamily G member 2
- TAMs
Tumor-associated macrophages
- Treg cells
Regulatory T cells
- NK cells
Nature killer cells
- DCs
Dendritic cells
- COX2
Cyclooxygenase 2
- PGE2
Prostaglandin E2
- G-CSF
Granulocyte colony-stimulating factor
- TAP
Transporter associated with antigen processing
- IFN-γ
Interferon-gamma
- ICIs
Immune checkpoint inhibitors
- IL-12
Interleukin-12
- NLRC5
NOD-like receptor family CARD domain containing 5
- H3K27m3
Histone modifications of Lys-27 in histone 3
- DNMTi
Methyltransferases inhibitor
- HDACi
Histone deacetylases inhibitor
- EZH2i
Enhancer of zeste homolog 2 inhibitor
- APL
Acute promyelocytic leukemia
- IDH
Isocitrate dehydrogenase
Authors’ contributions
YS M wrote the main manuscript text. JY S, C Y, CL X and LW F revised the manuscript. LW F and CL X supervised the work. The authors read and approved the final manuscript.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Chenglai Xia, Email: xiachenglai@smu.edu.cn.
Liwu Fu, Email: Fulw@mail.sysu.edu.cn.
References
- 1.Eppert K, Takenaka K, Lechman ER, Waldron L, Nilsson B, van Galen P, Metzeler KH, Poeppl A, Ling V, Beyene J, et al. Stem cell gene expression programs influence clinical outcome in human leukemia. Nat Med. 2011;17:1086–1093. doi: 10.1038/nm.2415. [DOI] [PubMed] [Google Scholar]
- 2.Hu Y, Smyth GK. ELDA: extreme limiting dilution analysis for comparing depleted and enriched populations in stem cell and other assays. J Immunol Methods. 2009;347:70–78. doi: 10.1016/j.jim.2009.06.008. [DOI] [PubMed] [Google Scholar]
- 3.Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci U S A. 2003;100:3983–3988. doi: 10.1073/pnas.0530291100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Abbaszadegan MR, Bagheri V, Razavi MS, Momtazi AA, Sahebkar A, Gholamin M. Isolation, identification, and characterization of cancer stem cells: a review. J Cell Physiol. 2017;232:2008–2018. doi: 10.1002/jcp.25759. [DOI] [PubMed] [Google Scholar]
- 5.Huntly BJ, Gilliland DG. Leukaemia stem cells and the evolution of cancer-stem-cell research. Nat Rev Cancer. 2005;5:311–321. doi: 10.1038/nrc1592. [DOI] [PubMed] [Google Scholar]
- 6.Borst P. Cancer drug pan-resistance: pumps, cancer stem cells, quiescence, epithelial to mesenchymal transition, blocked cell death pathways, persisters or what? Open Biol. 2012;2:120066. doi: 10.1098/rsob.120066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Visvader JE, Lindeman GJ. Cancer stem cells: current status and evolving complexities. Cell Stem Cell. 2012;10:717–728. doi: 10.1016/j.stem.2012.05.007. [DOI] [PubMed] [Google Scholar]
- 8.Espinoza I, Miele L. Deadly crosstalk: Notch signaling at the intersection of EMT and cancer stem cells. Cancer Lett. 2013;341:41–45. doi: 10.1016/j.canlet.2013.08.027. [DOI] [PubMed] [Google Scholar]
- 9.Chaffer CL, Brueckmann I, Scheel C, Kaestli AJ, Wiggins PA, Rodrigues LO, Brooks M, Reinhardt F, Su Y, Polyak K, et al. Normal and neoplastic nonstem cells can spontaneously convert to a stem-like state. Proc Natl Acad Sci U S A. 2011;108:7950–7955. doi: 10.1073/pnas.1102454108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yilmaz M, Christofori G. EMT, the cytoskeleton, and cancer cell invasion. Cancer Metastasis Rev. 2009;28:15–33. doi: 10.1007/s10555-008-9169-0. [DOI] [PubMed] [Google Scholar]
- 11.Bayik D, Lathia JD. Cancer stem cell-immune cell crosstalk in tumour progression. Nat Rev Cancer. 2021;21:526–536. doi: 10.1038/s41568-021-00366-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Erin N, Grahovac J, Brozovic A, Efferth T. Tumor microenvironment and epithelial mesenchymal transition as targets to overcome tumor multidrug resistance. Drug Resist Updat. 2020;53:100715. doi: 10.1016/j.drup.2020.100715. [DOI] [PubMed] [Google Scholar]
- 13.Patel S, Alam A, Pant R, Chattopadhyay S. Wnt signaling and its significance within the tumor microenvironment: novel therapeutic insights. Front Immunol. 2019;10:2872. doi: 10.3389/fimmu.2019.02872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Meurette O, Mehlen P. Notch signaling in the tumor microenvironment. Cancer Cell. 2018;34:536–548. doi: 10.1016/j.ccell.2018.07.009. [DOI] [PubMed] [Google Scholar]
- 15.Islam F, Gopalan V, Wahab R, Smith RA, Lam AK. Cancer stem cells in oesophageal squamous cell carcinoma: identification, prognostic and treatment perspectives. Crit Rev Oncol Hematol. 2015;96:9–19. doi: 10.1016/j.critrevonc.2015.04.007. [DOI] [PubMed] [Google Scholar]
- 16.Brungs D, Aghmesheh M, Vine KL, Becker TM, Carolan MG, Ranson M. Gastric cancer stem cells: evidence, potential markers, and clinical implications. J Gastroenterol. 2016;51:313–326. doi: 10.1007/s00535-015-1125-5. [DOI] [PubMed] [Google Scholar]
- 17.Mihanfar A, Aghazadeh Attari J, Mohebbi I, Majidinia M, Kaviani M, Yousefi M, Yousefi B. Ovarian cancer stem cell: a potential therapeutic target for overcoming multidrug resistance. J Cell Physiol. 2019;234:3238–3253. doi: 10.1002/jcp.26768. [DOI] [PubMed] [Google Scholar]
- 18.Du L, Cheng Q, Zheng H, Liu J, Liu L, Chen Q. Targeting stemness of cancer stem cells to fight colorectal cancers. Semin Cancer Biol. 2022;82:150–161. doi: 10.1016/j.semcancer.2021.02.012. [DOI] [PubMed] [Google Scholar]
- 19.Lee TK, Guan XY, Ma S. Cancer stem cells in hepatocellular carcinoma - from origin to clinical implications. Nat Rev Gastroenterol Hepatol. 2022;19:26–44. doi: 10.1038/s41575-021-00508-3. [DOI] [PubMed] [Google Scholar]
- 20.Guo W, Qiao T, Li T. The role of stem cells in small-cell lung cancer: evidence from chemoresistance to immunotherapy. Semin Cancer Biol. 2022;87:160–169. doi: 10.1016/j.semcancer.2022.11.006. [DOI] [PubMed] [Google Scholar]
- 21.Bai X, Ni J, Beretov J, Graham P, Li Y. Cancer stem cell in breast cancer therapeutic resistance. Cancer Treat Rev. 2018;69:152–163. doi: 10.1016/j.ctrv.2018.07.004. [DOI] [PubMed] [Google Scholar]
- 22.Verma P, Shukla N, Kumari S, Ansari MS, Gautam NK, Patel GK. Cancer stem cell in prostate cancer progression, metastasis and therapy resistance. Biochim Biophys Acta Rev Cancer. 2023;1878:188887. doi: 10.1016/j.bbcan.2023.188887. [DOI] [PubMed] [Google Scholar]
- 23.Patil K, Khan FB, Akhtar S, Ahmad A, Uddin S. The plasticity of pancreatic cancer stem cells: implications in therapeutic resistance. Cancer Metastasis Rev. 2021;40:691–720. doi: 10.1007/s10555-021-09979-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tahmasebi E, Alikhani M, Yazdanian A, Yazdanian M, Tebyanian H, Seifalian A. The current markers of cancer stem cell in oral cancers. Life Sci. 2020;249:117483. doi: 10.1016/j.lfs.2020.117483. [DOI] [PubMed] [Google Scholar]
- 25.Ortiz-Sánchez Elizabeth, Santiago-Lopez Luz, Cruz-Domínguez Verónica B, Toledo-Guzmán Mariel E, Hernández-Cueto Daniel, Muñiz-Hernández Saé, Garrido Efraín, León David Cantú De, García-Carrancá Alejandro. Characterization of cervical cancer stem cell-like cells phenotyping, stemness, and human papilloma virus co-receptor expression. Oncotarget. 2016;7:31943–31954. doi: 10.18632/oncotarget.8218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Biserova K, Jakovlevs A, Uljanovs R, Strumfa I. Cancer stem cells: significance in origin pathogenesis and treatment of glioblastoma. Cells. 2021;10:621. doi: 10.3390/cells10030621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Aghaalikhani N, Rashtchizadeh N, Shadpour P, Allameh A, Mahmoodi M. Cancer stem cells as a therapeutic target in bladder cancer. J Cell Physiol. 2019;234:3197–3206. doi: 10.1002/jcp.26916. [DOI] [PubMed] [Google Scholar]
- 28.Hua Z, White J, Zhou J. Cancer stem cells in TNBC. Semin Cancer Biol. 2022;82:26–34. doi: 10.1016/j.semcancer.2021.06.015. [DOI] [PubMed] [Google Scholar]
- 29.Leon G, MacDonagh L, Finn SP, Cuffe S, Barr MP. Cancer stem cells in drug resistant lung cancer: targeting cell surface markers and signaling pathways. Pharmacol Ther. 2016;158:71–90. doi: 10.1016/j.pharmthera.2015.12.001. [DOI] [PubMed] [Google Scholar]
- 30.Kokuryo T, Yokoyama Y, Nagino M. Recent advances in cancer stem cell research for cholangiocarcinoma. J Hepatobiliary Pancreat Sci. 2012;19:606–613. doi: 10.1007/s00534-012-0542-6. [DOI] [PubMed] [Google Scholar]
- 31.ConstanzeBanz-Jansen LPH. Barbara Kaltschmidt endometrial cancer stem cells where do we stand and where should we go. Int J Mol Sci. 2022;23:3412. doi: 10.3390/ijms23063412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lin R-Y. Thyroid cancer stem cells. Nat Rev Endocrinol. 2011;7:609–616. doi: 10.1038/nrendo.2011.127. [DOI] [PubMed] [Google Scholar]
- 33.Wei Pingpin, Niu Man, Pan Suming, Zhou Yanhong, Shuai Cijun, Wang Jing, Peng Shuping, Li Guiyuan. Cancer stem-like cell a novel target for nasopharyngeal carcinoma therapy. Stem Cell Res Ther. 2014;5:44. doi: 10.1186/scrt433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Takaishi S, Okumura T, Tu S, Wang SS, Shibata W, Vigneshwaran R, Gordon SA, Shimada Y, Wang TC. Identification of gastric cancer stem cells using the cell surface marker CD44. Stem Cells. 2009;27:1006–1020. doi: 10.1002/stem.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bourguignon LY, Spevak CC, Wong G, Xia W, Gilad E. Hyaluronan-CD44 interaction with protein kinase C(epsilon) promotes oncogenic signaling by the stem cell marker Nanog and the Production of microRNA-21, leading to down-regulation of the tumor suppressor protein PDCD4, anti-apoptosis, and chemotherapy resistance in breast tumor cells. J Biol Chem. 2009;284:26533–26546. doi: 10.1074/jbc.M109.027466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ponta H, Sherman L, Herrlich PA. CD44: from adhesion molecules to signalling regulators. Nat Rev Mol Cell Biol. 2003;4:33–45. doi: 10.1038/nrm1004. [DOI] [PubMed] [Google Scholar]
- 37.Barzegar Behrooz A, Syahir A, Ahmad S. CD133: beyond a cancer stem cell biomarker. J Drug Target. 2019;27:257–269. doi: 10.1080/1061186X.2018.1479756. [DOI] [PubMed] [Google Scholar]
- 38.O'Flaherty JD, Barr M, Fennell D, Richard D, Reynolds J, O'Leary J, O'Byrne K. The cancer stem-cell hypothesis: its emerging role in lung cancer biology and its relevance for future therapy. J Thorac Oncol. 2012;7:1880–1890. doi: 10.1097/JTO.0b013e31826bfbc6. [DOI] [PubMed] [Google Scholar]
- 39.Mak AB, Nixon AM, Kittanakom S, Stewart JM, Chen GI, Curak J, Gingras AC, Mazitschek R, Neel BG, Stagljar I, Moffat J. Regulation of CD133 by HDAC6 promotes beta-catenin signaling to suppress cancer cell differentiation. Cell Rep. 2012;2:951–963. doi: 10.1016/j.celrep.2012.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jang JW, Song Y, Kim SH, Kim J, Seo HR. Potential mechanisms of CD133 in cancer stem cells. Life Sci. 2017;184:25–29. doi: 10.1016/j.lfs.2017.07.008. [DOI] [PubMed] [Google Scholar]
- 41.Abbasian M, Mousavi E, Arab-Bafrani Z, Sahebkar A. The most reliable surface marker for the identification of colorectal cancer stem-like cells: A systematic review and meta-analysis. J Cell Physiol. 2019;234:8192–8202. doi: 10.1002/jcp.27619. [DOI] [PubMed] [Google Scholar]
- 42.Mohtar MA, Syafruddin SE, Nasir SN, Low TY. Revisiting the roles of pro-metastatic EpCAM in cancer. Biomolecules. 2020;10:255. doi: 10.3390/biom10020255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Munz M, Baeuerle PA, Gires O. The emerging role of EpCAM in cancer and stem cell signaling. Cancer Res. 2009;69:5627–5629. doi: 10.1158/0008-5472.CAN-09-0654. [DOI] [PubMed] [Google Scholar]
- 44.Sun YF, Xu Y, Yang XR, Guo W, Zhang X, Qiu SJ, Shi RY, Hu B, Zhou J, Fan J. Circulating stem cell-like epithelial cell adhesion molecule-positive tumor cells indicate poor prognosis of hepatocellular carcinoma after curative resection. Hepatology. 2013;57:1458–1468. doi: 10.1002/hep.26151. [DOI] [PubMed] [Google Scholar]
- 45.Gires O, Pan M, Schinke H, Canis M, Baeuerle PA. Expression and function of epithelial cell adhesion molecule EpCAM: where are we after 40 years? Cancer Metastasis Rev. 2020;39:969–987. doi: 10.1007/s10555-020-09898-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yu T, Ma Y, Wang H. EpCAM intracellular domain promotes porcine cell reprogramming by upregulation of pluripotent gene expression via beta-catenin signaling. Sci Rep. 2017;7:46315. doi: 10.1038/srep46315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Huang L, Yang Y, Yang F, Liu S, Zhu Z, Lei Z, Guo J. Functions of EpCAM in physiological processes and diseases (Review) Int J Mol Med. 2018;42:1771–1785. doi: 10.3892/ijmm.2018.3764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Brown TC, Sankpal NV, Gillanders WE. Functional implications of the dynamic regulation of EpCAM during epithelial-to-mesenchymal transition. Biomolecules. 2021;11:956. doi: 10.3390/biom11070956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ko CJ, Li CJ, Wu MY, Chu PY. Overexpression of epithelial cell adhesion molecule as a predictor of poor outcome in patients with hepatocellular carcinoma. Exp Ther Med. 2018;16:4810–4816. doi: 10.3892/etm.2018.6794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Xu X, Chai S, Wang P, Zhang C, Yang Y, Yang Y, Wang K. Aldehyde dehydrogenases and cancer stem cells. Cancer Lett. 2015;369:50–57. doi: 10.1016/j.canlet.2015.08.018. [DOI] [PubMed] [Google Scholar]
- 51.Croker AK, Goodale D, Chu J, Postenka C, Hedley BD, Hess DA, Allan AL. High aldehyde dehydrogenase and expression of cancer stem cell markers selects for breast cancer cells with enhanced malignant and metastatic ability. J Cell Mol Med. 2009;13:2236–2252. doi: 10.1111/j.1582-4934.2008.00455.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhang H, Fu L. The role of ALDH2 in tumorigenesis and tumor progression: Targeting ALDH2 as a potential cancer treatment. Acta Pharmaceutica Sinica B. 2021;11:1400–1411. doi: 10.1016/j.apsb.2021.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ying M, Wang S, Sang Y, Sun P, Lal B, Goodwin CR, Guerrero-Cazares H, Quinones-Hinojosa A, Laterra J, Xia S. Regulation of glioblastoma stem cells by retinoic acid: role for Notch pathway inhibition. Oncogene. 2011;30:3454–3467. doi: 10.1038/onc.2011.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kim D, Choi BH, Ryoo IG, Kwak MK. High NRF2 level mediates cancer stem cell-like properties of aldehyde dehydrogenase (ALDH)-high ovarian cancer cells: inhibitory role of all-trans retinoic acid in ALDH/NRF2 signaling. Cell Death Dis. 2018;9:896. doi: 10.1038/s41419-018-0903-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kim RJ, Park JR, Roh KJ, Choi AR, Kim SR, Kim PH, Yu JH, Lee JW, Ahn SH, Gong G, et al. High aldehyde dehydrogenase activity enhances stem cell features in breast cancer cells by activating hypoxia-inducible factor-2alpha. Cancer Lett. 2013;333:18–31. doi: 10.1016/j.canlet.2012.11.026. [DOI] [PubMed] [Google Scholar]
- 56.Muzio G, Maggiora M, Paiuzzi E, Oraldi M, Canuto RA. Aldehyde dehydrogenases and cell proliferation. Free Radic Biol Med. 2012;52:735–746. doi: 10.1016/j.freeradbiomed.2011.11.033. [DOI] [PubMed] [Google Scholar]
- 57.Harati M, Rodemann H, Toulany M. Nanog signaling mediates radioresistance in ALDH-positive breast cancer cells. Int J Mol Sci. 2019;20:1151. doi: 10.3390/ijms20051151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Piva M, Domenici G, Iriondo O, Rabano M, Simoes BM, Comaills V, Barredo I, Lopez-Ruiz JA, Zabalza I, Kypta R, Vivanco M. Sox2 promotes tamoxifen resistance in breast cancer cells. EMBO Mol Med. 2014;6:66–79. doi: 10.1002/emmm.201303411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhang Q, Han Z, Zhu Y, Chen J, Li W. The role and specific mechanism of OCT4 in cancer stem cells: a review. Int J Stem Cells. 2020;13:312–325. doi: 10.15283/ijsc20097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Guo F, Yang Z, Kulbe H, Albers AE, Sehouli J, Kaufmann AM. Inhibitory effect on ovarian cancer ALDH+ stem-like cells by Disulfiram and Copper treatment through ALDH and ROS modulation. Biomed Pharmacother. 2019;118:109371. doi: 10.1016/j.biopha.2019.109371. [DOI] [PubMed] [Google Scholar]
- 61.Yang L, Shi P, Zhao G, Xu J, Peng W, Zhang J, Zhang G, Wang X, Dong Z, Chen F, Cui H. Targeting cancer stem cell pathways for cancer therapy. Signal Transduct Target Ther. 2020;5:8. doi: 10.1038/s41392-020-0110-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Liu Chunming, Li Yiming, Baeg Gyeong-Hun, Zhang Zhuohua, Lin Xinhua. Control of β-Catenin Phosphorylation/Degradation by a dual-kinase mechanism. Cell. 2002;108:837–847. doi: 10.1016/s0092-8674(02)00685-2. [DOI] [PubMed] [Google Scholar]
- 63.Hernandez AR, Klein AM, Kirschner MW. Kinetic responses of beta-catenin specify the sites of Wnt control. Science. 2012;338:1337–1340. doi: 10.1126/science.1228734. [DOI] [PubMed] [Google Scholar]
- 64.Xu J, Lamouille S, Derynck R. TGF-beta-induced epithelial to mesenchymal transition. Cell Res. 2009;19:156–172. doi: 10.1038/cr.2009.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Thiery JP, Sleeman JP. Complex networks orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell Biol. 2006;7:131–142. doi: 10.1038/nrm1835. [DOI] [PubMed] [Google Scholar]
- 66.Gonzalez DM, Medici D. Signaling mechanisms of the epithelial-mesenchymal transition. Sci Signal. 2014;7:re8. doi: 10.1126/scisignal.2005189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Na TY, Schecterson L, Mendonsa AM, Gumbiner BM. The functional activity of E-cadherin controls tumor cell metastasis at multiple steps. Proc Natl Acad Sci U S A. 2020;117:5931–5937. doi: 10.1073/pnas.1918167117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Katoh M, Katoh M. WNT signaling and cancer stemness. Essays Biochem. 2022;66:319–331. doi: 10.1042/EBC20220016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Cheung P, Xiol J, Dill MT, Yuan WC, Panero R, Roper J, Osorio FG, Maglic D, Li Q, Gurung B, et al. Regenerative reprogramming of the intestinal stem cell state via hippo signaling suppresses metastatic colorectal cancer. Cell Stem Cell. 2020;27(590–604):e599. doi: 10.1016/j.stem.2020.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ren D, Dai Y, Yang Q, Zhang X, Guo W, Ye L, Huang S, Chen X, Lai Y, Du H, et al. Wnt5a induces and maintains prostate cancer cells dormancy in bone. J Exp Med. 2019;216:428–449. doi: 10.1084/jem.20180661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Phan TG, Croucher PI. The dormant cancer cell life cycle. Nat Rev Cancer. 2020;20:398–411. doi: 10.1038/s41568-020-0263-0. [DOI] [PubMed] [Google Scholar]
- 72.Brendan D'Souza LM-K, Weinmaster G. Canonical and Non-Canonical Notch Ligands. Curr Top Dev Biol. 2010;92:73–129. [DOI] [PMC free article] [PubMed]
- 73.Bray SJ. Notch signalling in context. Nat Rev Mol Cell Biol. 2016;17:722–735. doi: 10.1038/nrm.2016.94. [DOI] [PubMed] [Google Scholar]
- 74.Andersson ER, Sandberg R, Lendahl U. Notch signaling: simplicity in design, versatility in function. Development. 2011;138:3593–3612. doi: 10.1242/dev.063610. [DOI] [PubMed] [Google Scholar]
- 75.Pannuti A, Foreman K, Rizzo P, Osipo C, Golde T, Osborne B, Miele L. Targeting Notch to target cancer stem cells. Clin Cancer Res. 2010;16:3141–3152. doi: 10.1158/1078-0432.CCR-09-2823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wang Y, Chen P, Zhao M, Cao H, Zhao Y, Ji M, Hou P, Chen M. EGFL7 drives the evolution of resistance to EGFR inhibitors in lung cancer by activating NOTCH signaling. Cell Death Dis. 2022;13:910. doi: 10.1038/s41419-022-05354-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kannan S, Sutphin RM, Hall MG, Golfman LS, Fang W, Nolo RM, Akers LJ, Hammitt RA, McMurray JS, Kornblau SM, et al. Notch activation inhibits AML growth and survival: a potential therapeutic approach. J Exp Med. 2013;210:321–337. doi: 10.1084/jem.20121527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Tyagi A, Sharma AK, Damodaran C. A review on notch signaling and colorectal cancer. Cells. 2020;9:1549. doi: 10.3390/cells9061549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Sari IN, Phi LTH, Jun N, Wijaya YT, Lee S, Kwon HY. Hedgehog signaling in cancer: a prospective therapeutic target for eradicating cancer stem cells. Cells. 2018;7:208. doi: 10.3390/cells7110208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Merchant AA, Matsui W. Targeting Hedgehog–a cancer stem cell pathway. Clin Cancer Res. 2010;16:3130–3140. doi: 10.1158/1078-0432.CCR-09-2846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Varnat F, Duquet A, Malerba M, Zbinden M, Mas C, Gervaz P. Ruiz i Altaba A: Human colon cancer epithelial cells harbour active HEDGEHOG-GLI signalling that is essential for tumour growth, recurrence, metastasis and stem cell survival and expansion. EMBO Mol Med. 2009;1:338–351. doi: 10.1002/emmm.200900039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Patni AP, Harishankar MK, Joseph JP, Sreeshma B, Jayaraj R, Devi A. Comprehending the crosstalk between Notch, Wnt and Hedgehog signaling pathways in oral squamous cell carcinoma - clinical implications. Cell Oncol (Dordr) 2021;44:473–494. doi: 10.1007/s13402-021-00591-3. [DOI] [PubMed] [Google Scholar]
- 83.Fu M, Hu Y, Lan T, Guan K-L, Luo T, Luo M. The Hippo signalling pathway and its implications in human health and diseases. Signal Transduct Target Ther. 2022;7:376. doi: 10.1038/s41392-022-01191-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Park Jae Hyung, Shin Ji Eun, Park Hyun Woo. The Role of Hippo Pathway in Cancer Stem Cell Biology. Mol Cells. 2018;41:83–92. doi: 10.14348/molcells.2018.2242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Zanconato F, Cordenonsi M, Piccolo S. YAP/TAZ at the Roots of Cancer. Cancer Cell. 2016;29:783–803. doi: 10.1016/j.ccell.2016.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Nguyen CDK, Yi C. YAP/TAZ Signaling and Resistance to Cancer Therapy. Trends in Cancer. 2019;5:283–296. doi: 10.1016/j.trecan.2019.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Rinkenbaugh A, Baldwin A. The NF-κB pathway and cancer stem cells. Cells. 2016;5:16. doi: 10.3390/cells5020016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Nakagawa MM, Rathinam CV. Constitutive activation of the canonical NF-κB pathway leads to bone marrow failure and induction of erythroid signature in hematopoietic stem cells. Cell Rep. 2018;25:2094–2109.e2094. doi: 10.1016/j.celrep.2018.10.071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Liu W, Lu X, Shi P, Yang G, Zhou Z, Li W, Mao X, Jiang D, Chen C. TNF-α increases breast cancer stem-like cells through up-regulating TAZ expression via the non-canonical NF-κB pathway. Sci Rep. 2020;10:1804. doi: 10.1038/s41598-020-58642-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Baldwin AS. Regulation of cell death and autophagy by IKK and NF-κB critical mechanisms in immune function and cancer. Immunol Rev. 2012;246:327–345. doi: 10.1111/j.1600-065X.2012.01095.x. [DOI] [PubMed] [Google Scholar]
- 91.Basseres DS, Baldwin AS. Nuclear factor-kappaB and inhibitor of kappaB kinase pathways in oncogenic initiation and progression. Oncogene. 2006;25:6817–6830. doi: 10.1038/sj.onc.1209942. [DOI] [PubMed] [Google Scholar]
- 92.Schroeder A, Herrmann A, Cherryholmes G, Kowolik C, Buettner R, Pal S, Yu H, Muller-Newen G, Jove R. Loss of androgen receptor expression promotes a stem-like cell phenotype in prostate cancer through STAT3 signaling. Cancer Res. 2014;74:1227–1237. doi: 10.1158/0008-5472.CAN-13-0594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Bournazou E, Bromberg J. Targeting the tumor microenvironment: JAK-STAT3 signaling. Jak-Stat. 2014;2:e23828. doi: 10.4161/jkst.23828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Pattabiraman DR, Weinberg RA. Tackling the cancer stem cells - what challenges do they pose? Nat Rev Drug Discov. 2014;13:497–512. doi: 10.1038/nrd4253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Galoczova M, Coates P, Vojtesek B. STAT3, stem cells, cancer stem cells and p63. Cellular & Molecular Biology Letters. 2018;23:12. doi: 10.1186/s11658-018-0078-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Fan Y, Mao R, Yang J. NF-kappaB and STAT3 signaling pathways collaboratively link inflammation to cancer. Protein Cell. 2013;4:176–185. doi: 10.1007/s13238-013-2084-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Yoon S, Woo SU, Kang JH, Kim K, Shin HJ, Gwak HS, Park S, Chwae YJ. NF-kappaB and STAT3 cooperatively induce IL6 in starved cancer cells. Oncogene. 2012;31:3467–3481. doi: 10.1038/onc.2011.517. [DOI] [PubMed] [Google Scholar]
- 98.Johnson DE, O'Keefe RA, Grandis JR. Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat Rev Clin Oncol. 2018;15:234–248. doi: 10.1038/nrclinonc.2018.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Ma B, Hottiger MO. Crosstalk between Wnt/β-Catenin and NF-κB Signaling Pathway during Inflammation. Front Immunol. 2016;7:378. doi: 10.3389/fimmu.2016.00378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Glaviano A, Foo ASC, Lam HY, Yap KCH, Jacot W, Jones RH, Eng H, Nair MG, Makvandi P, Geoerger B, et al. PI3K/AKT/mTOR signaling transduction pathway and targeted therapies in cancer. Mol Cancer. 2023;22:138. doi: 10.1186/s12943-023-01827-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Yang J, Nie J, Ma X, Wei Y, Peng Y, Wei X. Targeting PI3K in cancer: mechanisms and advances in clinical trials. Mol Cancer. 2019;18:26. doi: 10.1186/s12943-019-0954-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Chang L, Graham PH, Hao J, Ni J, Bucci J, Cozzi PJ, Kearsley JH, Li Y. Acquisition of epithelial-mesenchymal transition and cancer stem cell phenotypes is associated with activation of the PI3K/Akt/mTOR pathway in prostate cancer radioresistance. Cell Death Dis. 2013;4:e875. doi: 10.1038/cddis.2013.407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Dubrovska A, Kim S, Salamone RJ, Walker JR, Maira SM, Garcia-Echeverria C, Schultz PG, Reddy VA. The role of PTEN/Akt/PI3K signaling in the maintenance and viability of prostate cancer stem-like cell populations. Proc Natl Acad Sci U S A. 2009;106:268–273. doi: 10.1073/pnas.0810956106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Hales EC, Orr SM, Larson Gedman A, Taub JW, Matherly LH. Notch1 receptor regulates AKT protein activation loop (Thr308) dephosphorylation through modulation of the PP2A phosphatase in phosphatase and tensin homolog (PTEN)-null T-cell acute lymphoblastic leukemia cells. J Biol Chem. 2013;288:22836–22848. doi: 10.1074/jbc.M113.451625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Gentle ME, Rose A, Bugeon L, Dallman MJ. Noncanonical Notch signaling modulates cytokine responses of dendritic cells to inflammatory stimuli. J Immunol. 2012;189:1274–1284. doi: 10.4049/jimmunol.1103102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Ke Z, Caiping S, Qing Z, Xiaojing W. Sonic hedgehog-Gli1 signals promote epithelial-mesenchymal transition in ovarian cancer by mediating PI3K/AKT pathway. Med Oncol. 2015;32:368. doi: 10.1007/s12032-014-0368-y. [DOI] [PubMed] [Google Scholar]
- 107.Lastwika KJ, Wilson W, 3rd, Li QK, Norris J, Xu H, Ghazarian SR, Kitagawa H, Kawabata S, Taube JM, Yao S, et al. Control of PD-L1 expression by oncogenic activation of the AKT-mTOR pathway in non-small cell lung cancer. Cancer Res. 2016;76:227–238. doi: 10.1158/0008-5472.CAN-14-3362. [DOI] [PubMed] [Google Scholar]
- 108.Koh V, Chakrabarti J, Torvund M, Steele N, Hawkins JA, Ito Y, Wang J, Helmrath MA, Merchant JL, Ahmed SA, et al. Hedgehog transcriptional effector GLI mediates mTOR-Induced PD-L1 expression in gastric cancer organoids. Cancer Lett. 2021;518:59–71. doi: 10.1016/j.canlet.2021.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Menke-van der Houven CW, Gomez-Roca C, van Herpen C, Coveler AL, Mahalingam D, Verheul HM, van der Graaf WT, Christen R, Rüttinger D, Weigand S, et al. First-in-human phase I clinical trial of RG7356, an anti-CD44 humanized antibody, in patients with advanced, CD44-expressing solid tumors. Oncotarget. 2016;7:80046–80058. doi: 10.18632/oncotarget.11098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Alamgeer M, Neil Watkins D, Banakh I, Kumar B, Gough DJ, Markman B, Ganju V. A phase IIa study of HA-irinotecan, formulation of hyaluronic acid and irinotecan targeting CD44 in extensive-stage small cell lung cancer. Invest New Drugs. 2018;36:288–298. doi: 10.1007/s10637-017-0555-8. [DOI] [PubMed] [Google Scholar]
- 111.James MI, Iwuji C, Irving G, Karmokar A, Higgins JA, Griffin-Teal N, Thomas A, Greaves P, Cai H, Patel SR, et al. Curcumin inhibits cancer stem cell phenotypes in ex vivo models of colorectal liver metastases, and is clinically safe and tolerable in combination with FOLFOX chemotherapy. Cancer Lett. 2015;364:135–141. doi: 10.1016/j.canlet.2015.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Goldstein LJ, Mansutti M, Levy C, Chang JC, Henry S, Fernandez-Perez I, Prausova J, Staroslawska E, Viale G, Butler B, et al. A randomized, placebo-controlled phase 2 study of paclitaxel in combination with reparixin compared to paclitaxel alone as front-line therapy for metastatic triple-negative breast cancer (fRida) Breast Cancer Res Treat. 2021;190:265–275. doi: 10.1007/s10549-021-06367-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Mego M, Svetlovska D, Angelis VD, Kalavska K, Lesko P, Makovnik M, Obertova J, Orszaghova Z, Palacka P, Reckova M, et al. Phase II study of Disulfiram and Cisplatin in refractory germ cell tumors the GCT-SK-006 phase II trial. Invest New Drugs. 2022;40:1080–1086. doi: 10.1007/s10637-022-01271-1. [DOI] [PubMed] [Google Scholar]
- 114.Oberneder R, Weckermann D, Ebner B, Quadt C, Kirchinger P, Raum T, Locher M, Prang N, Baeuerle PA, Leo E. A phase I study with adecatumumab, a human antibody directed against epithelial cell adhesion molecule, in hormone refractory prostate cancer patients. Eur J Cancer. 2006;42:2530–2538. doi: 10.1016/j.ejca.2006.05.029. [DOI] [PubMed] [Google Scholar]
- 115.Kowalski M, Entwistle J, Cizeau J, Niforos D, Loewen S, Chapman W, MacDonald GC. A phase I study of an intravesically administered immunotoxin targeting EpCAM for the treatment of nonmuscle-invasive bladder cancer in BCGrefractory and BCG-intolerant patients. Drug Des Devel Ther. 2010;4:313–320. doi: 10.2147/DDDT.S14071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Connor JP, Cristea MC, Lewis NL, Lewis LD, Komarnitsky PB, Mattiacci MR, Felder M, Stewart S, Harter J, Henslee-Downey J, et al. A phase 1b study of humanized KS-interleukin-2 (huKS-IL2) immunocytokine with cyclophosphamide in patients with EpCAM-positive advanced solid tumors. BMC Cancer. 2013;13:20. doi: 10.1186/1471-2407-13-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Mau-Sorensen M, Dittrich C, Dienstmann R, Lassen U, Buchler W, Martinius H, Tabernero J. A phase I trial of intravenous catumaxomab: a bispecific monoclonal antibody targeting EpCAM and the T cell coreceptor CD3. Cancer Chemother Pharmacol. 2015;75:1065–1073. doi: 10.1007/s00280-015-2728-5. [DOI] [PubMed] [Google Scholar]
- 118.Dai H, Tong C, Shi D, Chen M, Guo Y, Chen D, Han X, Wang H, Wang Y, Shen P. Efficacy and biomarker analysis of CD133-directed CAR T cells in advanced hepatocellular carcinoma: a single-arm, open-label, phase II trial. OncoImmunology. 2020;9:1846926. doi: 10.1080/2162402X.2020.1846926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Rodon J, Argiles G, Connolly RM, Vaishampayan U, de Jonge M, Garralda E, Giannakis M, Smith DC, Dobson JR, McLaughlin ME, et al. Phase 1 study of single-agent WNT974, a first-in-class Porcupine inhibitor, in patients with advanced solid tumours. Br J Cancer. 2021;125:28–37. doi: 10.1038/s41416-021-01389-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Dotan E, Cardin DB, Lenz HJ, Messersmith W, O'Neil B, Cohen SJ, Denlinger CS, Shahda S, Astsaturov I, Kapoun AM, et al. Phase Ib study of Wnt inhibitor ipafricept with gemcitabine and nab-paclitaxel in patients with previously untreated stage IV pancreatic cancer. Clin Cancer Res. 2020;26:5348–5357. doi: 10.1158/1078-0432.CCR-20-0489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Diamond JR, Becerra C, Richards D, Mita A, Osborne C, O'Shaughnessy J, Zhang C, Henner R, Kapoun AM, Xu L, et al. Phase Ib clinical trial of the anti-frizzled antibody vantictumab (OMP-18R5) plus paclitaxel in patients with locally advanced or metastatic HER2-negative breast cancer. Breast Cancer Res Treat. 2020;184:53–62. doi: 10.1007/s10549-020-05817-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Plummer R, Dua D, Cresti N, Drew Y, Stephens P, Foegh M, Knudsen S, Sachdev P, Mistry BM, Dixit V, et al. First-in-human study of the PARP/tankyrase inhibitor E7449 in patients with advanced solid tumours and evaluation of a novel drug-response predictor. Br J Cancer. 2020;123:525–533. doi: 10.1038/s41416-020-0916-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lee JH, Faderl S, Pagel JM, Jung CW, Yoon SS, Pardanani AD, Becker PS, Lee H, Choi J, Lee K, et al. Phase 1 study of CWP232291 in patients with relapsed or refractory acute myeloid leukemia and myelodysplastic syndrome. Blood Adv. 2020;4:2032–2043. doi: 10.1182/bloodadvances.2019000757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Song S, Christova T, Perusini S, Alizadeh S, Bao RY, Miller BW, Hurren R, Jitkova Y, Gronda M, Isaac M, et al. Wnt inhibitor screen reveals iron dependence of beta-catenin signaling in cancers. Cancer Res. 2011;71:7628–7639. doi: 10.1158/0008-5472.CAN-11-2745. [DOI] [PubMed] [Google Scholar]
- 125.Choi MY, Widhopf GF, 2nd, Ghia EM, Kidwell RL, Hasan MK, Yu J, Rassenti LZ, Chen L, Chen Y, Pittman E, et al. Phase I trial: cirmtuzumab inhibits ROR1 signaling and stemness signatures in patients with chronic lymphocytic leukemia. Cell Stem Cell. 2018;22(951–959):e953. doi: 10.1016/j.stem.2018.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Islam MS, Afrin S, Singh B, Jayes FL, Brennan JT, Borahay MA, Leppert PC, Segars JH. Extracellular matrix and Hippo signaling as therapeutic targets of antifibrotic compounds for uterine fibroids. Clin Transl Med. 2021;11:e475. doi: 10.1002/ctm2.475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Villalobos VM, Hall F, Jimeno A, Gore L, Kern K, Cesari R, Huang B, Schowinsky JT, Blatchford PJ, Hoffner B, et al. Long-term follow-up of Desmoid Fibromatosis treated with PF-03084014, an oral gamma secretase inhibitor. Ann Surg Oncol. 2018;25:768–775. doi: 10.1245/s10434-017-6082-1. [DOI] [PubMed] [Google Scholar]
- 128.Smith DC, Chugh R, Patnaik A, Papadopoulos KP, Wang M, Kapoun AM, Xu L, Dupont J, Stagg RJ, Tolcher A. A phase 1 dose escalation and expansion study of Tarextumab (OMP-59R5) in patients with solid tumors. Invest New Drugs. 2019;37:722–730. doi: 10.1007/s10637-018-0714-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Aung KL, El-Khoueiry AB, Gelmon K, Tran B, Bajaj G, He B, Chen T, Zhu L, Poojary S, Basak S, et al. A multi-arm phase I dose escalating study of an oral NOTCH inhibitor BMS-986115 in patients with advanced solid tumours. Invest New Drugs. 2018;36:1026–1036. doi: 10.1007/s10637-018-0597-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Xu R, Shimizu F, Hovinga K, Beal K, Karimi S, Droms L, Peck KK, Gutin P, Iorgulescu JB, Kaley T, et al. Molecular and clinical effects of notch inhibition in glioma patients: a phase 0/I trial. Clin Cancer Res. 2016;22:4786–4796. doi: 10.1158/1078-0432.CCR-16-0048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Cook N, Basu B, Smith DM, Gopinathan A, Evans J, Steward WP, Palmer D, Propper D, Venugopal B, Hategan M, et al. A phase I trial of the gamma-secretase inhibitor MK-0752 in combination with gemcitabine in patients with pancreatic ductal adenocarcinoma. Br J Cancer. 2018;118:793–801. doi: 10.1038/bjc.2017.495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Gounder MM, Rosenbaum E, Wu N, Dickson MA, Sheikh TN, D'Angelo SP, Chi P, Keohan ML, Erinjeri JP, Antonescu CR, et al. A Phase Ib/II randomized study of RO4929097, a gamma-secretase or notch inhibitor with or without vismodegib, a hedgehog inhibitor, in advanced sarcoma. Clin Cancer Res. 2022;28:1586–1594. doi: 10.1158/1078-0432.CCR-21-3874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Pant S, Jones SF, Kurkjian CD, Infante JR, Moore KN, Burris HA, McMeekin DS, Benhadji KA, Patel BKR, Frenzel MJ, et al. A first-in-human phase I study of the oral Notch inhibitor, LY900009, in patients with advanced cancer. Eur J Cancer. 2016;56:1–9. doi: 10.1016/j.ejca.2015.11.021. [DOI] [PubMed] [Google Scholar]
- 134.McKeage MJ, Kotasek D, Markman B, Hidalgo M, Millward MJ, Jameson MB, Harris DL, Stagg RJ, Kapoun AM, Xu L, Hughes BGM. Phase IB trial of the anti-cancer stem cell DLL4-binding agent demcizumab with pemetrexed and carboplatin as first-line treatment of metastatic non-squamous NSCLC. Target Oncol. 2018;13:89–98. doi: 10.1007/s11523-017-0543-0. [DOI] [PubMed] [Google Scholar]
- 135.Johnson ML, Zvirbule Z, Laktionov K, Helland A, Cho BC, Gutierrez V, Colinet B, Lena H, Wolf M, Gottfried M, et al. Rovalpituzumab tesirine as a maintenance therapy after first-line platinum-based chemotherapy in patients with extensive-stage-SCLC: results from the phase 3 MERU study. J Thorac Oncol. 2021;16:1570–1581. doi: 10.1016/j.jtho.2021.03.012. [DOI] [PubMed] [Google Scholar]
- 136.Chiorean EG, LoRusso P, Strother RM, Diamond JR, Younger A, Messersmith WA, Adriaens L, Liu L, Kao RJ, DiCioccio AT, et al. A phase I first-in-human study of Enoticumab (REGN421), a fully human delta-like ligand 4 (Dll4) monoclonal antibody in patients with advanced solid tumors. Clin Cancer Res. 2015;21:2695–2703. doi: 10.1158/1078-0432.CCR-14-2797. [DOI] [PubMed] [Google Scholar]
- 137.Ferrarotto R, Eckhardt G, Patnaik A, LoRusso P, Faoro L, Heymach JV, Kapoun AM, Xu L, Munster P. A phase I dose-escalation and dose-expansion study of brontictuzumab in subjects with selected solid tumors. Ann Oncol. 2018;29:1561–1568. doi: 10.1093/annonc/mdy171. [DOI] [PubMed] [Google Scholar]
- 138.Hu ZI, Bendell JC, Bullock A, LoConte NK, Hatoum H, Ritch P, Hool H, Leach JW, Sanchez J, Sohal DPS, et al. A randomized phase II trial of nab-paclitaxel and gemcitabine with tarextumab or placebo in patients with untreated metastatic pancreatic cancer. Cancer Med. 2019;8:5148–5157. doi: 10.1002/cam4.2425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Von Hoff Daniel D. , Lorusso Patricia M. , Rudin Charles M., Reddy Josina C., Yauch Robert L., Tibes Raoul, Weiss Glen J., Borad Mitesh J., Hann Christine L., Brahmer Julie R., Mackey Howard M., Lum Bertram L., Darbonne Walter C., Marsters James C., Jr., de Sauvage Frederic J., Low Jennifer A. Inhibition of the hedgehog pathway in advanced basal-cell carcinoma. N Engl J Med. 2009;361(12):1164–72. doi: 10.1056/NEJMoa0905360. [DOI] [PubMed] [Google Scholar]
- 140.Jimeno A, Weiss GJ, Miller WH, Jr, Gettinger S, Eigl BJ, Chang AL, Dunbar J, Devens S, Faia K, Skliris G, et al. Phase I study of the Hedgehog pathway inhibitor IPI-926 in adult patients with solid tumors. Clin Cancer Res. 2013;19:2766–2774. doi: 10.1158/1078-0432.CCR-12-3654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Goldman J, Eckhardt SG, Borad MJ, Curtis KK, Hidalgo M, Calvo E, Ryan DP, Wirth LJ, Parikh A, Partyka J, et al. Phase I dose-escalation trial of the oral investigational Hedgehog signaling pathway inhibitor TAK-441 in patients with advanced solid tumors. Clin Cancer Res. 2015;21:1002–1009. doi: 10.1158/1078-0432.CCR-14-1234. [DOI] [PubMed] [Google Scholar]
- 142.Wagner AJ, Messersmith WA, Shaik MN, Li S, Zheng X, McLachlan KR, Cesari R, Courtney R, Levin WJ, El-Khoueiry AB. A phase I study of PF-04449913, an oral hedgehog inhibitor, in patients with advanced solid tumors. Clin Cancer Res. 2015;21:1044–1051. doi: 10.1158/1078-0432.CCR-14-1116. [DOI] [PubMed] [Google Scholar]
- 143.Pietanza MC, Litvak AM, Varghese AM, Krug LM, Fleisher M, Teitcher JB, Holodny AI, Sima CS, Woo KM, Ng KK, et al. A phase I trial of the Hedgehog inhibitor, sonidegib (LDE225), in combination with etoposide and cisplatin for the initial treatment of extensive stage small cell lung cancer. Lung Cancer. 2016;99:23–30. doi: 10.1016/j.lungcan.2016.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Bendell J, Andre V, Ho A, Kudchadkar R, Migden M, Infante J, Tiu RV, Pitou C, Tucker T, Brail L, Von Hoff D. Phase I study of LY2940680, a Smo antagonist, in patients with advanced cancer including treatment-naive and previously treated basal cell carcinoma. Clin Cancer Res. 2018;24:2082–2091. doi: 10.1158/1078-0432.CCR-17-0723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Bowles DW, Keysar SB, Eagles JR, Wang G, Glogowska MJ, McDermott JD, Le PN, Gao D, Ray CE, Rochon PJ, et al. A pilot study of cetuximab and the hedgehog inhibitor IPI-926 in recurrent/metastatic head and neck squamous cell carcinoma. Oral Oncol. 2016;53:74–79. doi: 10.1016/j.oraloncology.2015.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Lee M, Hong H, Kim W, Zhang L, Friedlander TW, Fong L, Lin AM, Small EJ, Wei XX, Rodvelt TJ, et al. Itraconazole as a noncastrating treatment for biochemically recurrent prostate cancer: a phase 2 study. Clin Genitourin Cancer. 2019;17:e92–e96. doi: 10.1016/j.clgc.2018.09.013. [DOI] [PubMed] [Google Scholar]
- 147.Hong D, Kurzrock R, Kim Y, Woessner R, Younes A, Nemunaitis J, Fowler N, Zhou T, Schmidt J, Jo M, et al. AZD9150, a next-generation antisense oligonucleotide inhibitor of STAT3 with early evidence of clinical activity in lymphoma and lung cancer. Sci Transl Med. 2015;7:314ra185. doi: 10.1126/scitranslmed.aac5272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Tolcher A, Flaherty K, Shapiro GI, Berlin J, Witzig T, Habermann T, Bullock A, Rock E, Elekes A, Lin C, et al. A first-in-human phase I study of OPB-111077, a small-molecule STAT3 and oxidative phosphorylation inhibitor, in patients with advanced cancers. Oncologist. 2018;23:658–e672. doi: 10.1634/theoncologist.2017-0325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Konstantinopoulos PA, Barry WT, Birrer M, Westin SN, Cadoo KA, Shapiro GI, Mayer EL, O'Cearbhaill RE, Coleman RL, Kochupurakkal B, et al. Olaparib and alpha-specific PI3K inhibitor alpelisib for patients with epithelial ovarian cancer: a dose-escalation and dose-expansion phase 1b trial. Lancet Oncol. 2019;20:570–580. doi: 10.1016/S1470-2045(18)30905-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Horwitz SM, Koch R, Porcu P, Oki Y, Moskowitz A, Perez M, Myskowski P, Officer A, Jaffe JD, Morrow SN, et al. Activity of the PI3K-delta, gamma inhibitor duvelisib in a phase 1 trial and preclinical models of T-cell lymphoma. Blood. 2018;131:888–898. doi: 10.1182/blood-2017-08-802470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Paz-Ares L, Kim TM, Vicente D, Felip E, Lee DH, Lee KH, Lin CC, Flor MJ, Di Nicola M, Alvarez RM, et al. Bintrafusp alfa, a bifunctional fusion protein targeting TGF-beta and PD-L1, in second-line treatment of patients with NSCLC: results from an expansion cohort of a phase 1 trial. J Thorac Oncol. 2020;15:1210–1222. doi: 10.1016/j.jtho.2020.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Strauss J, Heery CR, Schlom J, Madan RA, Cao L, Kang Z, Lamping E, Marte JL, Donahue RN, Grenga I, et al. Phase I trial of M7824 (MSB0011359C), a bifunctional fusion protein targeting PD-L1 and TGFbeta, in advanced solid tumors. Clin Cancer Res. 2018;24:1287–1295. doi: 10.1158/1078-0432.CCR-17-2653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Yamazaki T, Gunderson AJ, Gilchrist M, Whiteford M, Kiely MX, Hayman A, O'Brien D, Ahmad R, Manchio JV, Fox N, et al. Galunisertib plus neoadjuvant chemoradiotherapy in patients with locally advanced rectal cancer: a single-arm, phase 2 trial. Lancet Oncol. 2022;23:1189–1200. doi: 10.1016/S1470-2045(22)00446-6. [DOI] [PubMed] [Google Scholar]
- 154.Subbiah V, Iannotti NO, Gutierrez M, Smith DC, Feliz L, Lihou CF, Tian C, Silverman IM, Ji T, Saleh M. FIGHT-101, a first-in-human study of potent and selective FGFR 1–3 inhibitor pemigatinib in pan-cancer patients with FGF/FGFR alterations and advanced malignancies. Ann Oncol. 2022;33:522–533. doi: 10.1016/j.annonc.2022.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Li J, Qin S, Xu RH, Shen L, Xu J, Bai Y, Yang L, Deng Y, Chen ZD, Zhong H, et al. Effect of fruquintinib vs placebo on overall survival in patients with previously treated metastatic colorectal cancer. JAMA. 2018;319:2486–2496. doi: 10.1001/jama.2018.7855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Reckamp KL, Redman MW, Dragnev KH, Minichiello K, Villaruz LC, Faller B, Al Baghdadi T, Hines S, Everhart L, Highleyman L, et al. Phase II randomized study of ramucirumab and pembrolizumab versus standard of care in advanced non-small-cell lung cancer previously treated with immunotherapy-lung-MAP S1800A. J Clin Oncol. 2022;40:2295–2306. doi: 10.1200/JCO.22.00912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Tarhini AA, Rafique I, Floros T, Tran P, Gooding WE, Villaruz LC, Burns TF, Friedland DM, Petro DP, Farooqui M, et al. Phase 1/2 study of rilotumumab (AMG 102), a hepatocyte growth factor inhibitor, and erlotinib in patients with advanced non-small cell lung cancer. Cancer. 2017;123:2936–2944. doi: 10.1002/cncr.30717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Angevin E, Tabernero J, Elez E, Cohen SJ, Bahleda R, van Laethem JL, Ottensmeier C, Lopez-Martin JA, Clive S, Joly F, et al. A phase I/II, multiple-dose, dose-escalation study of siltuximab, an anti-interleukin-6 monoclonal antibody, in patients with advanced solid tumors. Clin Cancer Res. 2014;20:2192–2204. doi: 10.1158/1078-0432.CCR-13-2200. [DOI] [PubMed] [Google Scholar]
- 159.Ding Z, Li Q, Zhang R, Xie L, Shu Y, Gao S, Wang P, Su X, Qin Y, Wang Y, et al. Personalized neoantigen pulsed dendritic cell vaccine for advanced lung cancer. Signal Transduct Target Ther. 2021;6:26. doi: 10.1038/s41392-020-00448-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Bernal-Estevez DA, Ortiz Barbosa MA, Ortiz-Montero P, Cifuentes C, Sanchez R, Parra-Lopez CA. Autologous dendritic cells in combination with chemotherapy restore responsiveness of T cells in breast cancer patients: a single-arm phase I/II trial. Front Immunol. 2021;12:669965. doi: 10.3389/fimmu.2021.669965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Mehrotra S, Britten CD, Chin S, Garrett-Mayer E, Cloud CA, Li M, Scurti G, Salem ML, Nelson MH, Thomas MB, et al. Vaccination with poly(IC:LC) and peptide-pulsed autologous dendritic cells in patients with pancreatic cancer. J Hematol Oncol. 2017;10:82. doi: 10.1186/s13045-017-0459-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Liau LM, Ashkan K, Brem S, Campian JL, Trusheim JE, Iwamoto FM, Tran DD, Ansstas G, Cobbs CS, Heth JA, et al. Association of autologous tumor lysate-loaded dendritic cell vaccination with extension of survival among patients with newly diagnosed and recurrent glioblastoma: a phase 3 prospective externally controlled cohort trial. JAMA Oncol. 2023;9:112–121. doi: 10.1001/jamaoncol.2022.5370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Baer MR, Kogan AA, Bentzen SM, Mi T, Lapidus RG, Duong VH, Emadi A, Niyongere S, O'Connell CL, Youngblood BA, et al. Phase I clinical trial of DNA methyltransferase inhibitor decitabine and PARP inhibitor talazoparib combination therapy in relapsed/refractory acute myeloid leukemia. Clin Cancer Res. 2022;28:1313–1322. doi: 10.1158/1078-0432.CCR-21-3729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Kuang C, Park Y, Augustin RC, Lin Y, Hartman DJ, Seigh L, Pai RK, Sun W, Bahary N, Ohr J, et al. Pembrolizumab plus azacitidine in patients with chemotherapy refractory metastatic colorectal cancer: a single-arm phase 2 trial and correlative biomarker analysis. Clin Epigenetics. 2022;14:3. doi: 10.1186/s13148-021-01226-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Bever KM, Thomas DL, 2nd, Zhang J, Diaz Rivera EA, Rosner GL, Zhu Q, Nauroth JM, Christmas B, Thompson ED, Anders RA, et al. A feasibility study of combined epigenetic and vaccine therapy in advanced colorectal cancer with pharmacodynamic endpoint. Clin Epigenetics. 2021;13:25. doi: 10.1186/s13148-021-01014-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Ribrag V, Kim WS, Bouabdallah R, Lim ST, Coiffier B, Illes A, Lemieux B, Dyer MJS, Offner F, Felloussi Z, et al. Safety and efficacy of abexinostat, a pan-histone deacetylase inhibitor, in non-Hodgkin lymphoma and chronic lymphocytic leukemia: results of a phase II study. Haematologica. 2017;102:903–909. doi: 10.3324/haematol.2016.154377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Wieduwilt MJ, Pawlowska N, Thomas S, Olin R, Logan AC, Damon LE, Martin T, Kang M, Sayre PH, Boyer W, et al. Histone deacetylase inhibition with panobinostat combined with intensive induction chemotherapy in older patients with acute myeloid leukemia: phase I study results. Clin Cancer Res. 2019;25:4917–4923. doi: 10.1158/1078-0432.CCR-19-0171. [DOI] [PubMed] [Google Scholar]
- 168.Wang J, Zhang Q, Li Q, Mu Y, Jing J, Li H, Li W, Wang J, Yu G, Wang X, et al. Phase I study and pilot efficacy analysis of entinostat, a novel histone deacetylase inhibitor, in Chinese postmenopausal women with hormone receptor-positive metastatic breast cancer. Target Oncol. 2021;16:591–599. doi: 10.1007/s11523-021-00823-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Izutsu K, Ando K, Nishikori M, Shibayama H, Teshima T, Kuroda J, Kato K, Imaizumi Y, Nosaka K, Sakai R, et al. Phase II study of tazemetostat for relapsed or refractory B-cell non-Hodgkin lymphoma with EZH2 mutation in Japan. Cancer Sci. 2021;112:3627–3635. doi: 10.1111/cas.15040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Yap TA, Winter JN, Giulino-Roth L, Longley J, Lopez J, Michot JM, Leonard JP, Ribrag V, McCabe MT, Creasy CL, et al. Phase I study of the novel enhancer of Zeste Homolog 2 (EZH2) inhibitor GSK2816126 in patients with advanced hematologic and solid tumors. Clin Cancer Res. 2019;25:7331–7339. doi: 10.1158/1078-0432.CCR-18-4121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Song Y, Liu Y, Li ZM, Li L, Su H, Jin Z, Zuo X, Wu J, Zhou H, Li K, et al. SHR2554, an EZH2 inhibitor, in relapsed or refractory mature lymphoid neoplasms: a first-in-human, dose-escalation, dose-expansion, and clinical expansion phase 1 trial. Lancet Haematol. 2022;9:e493–e503. doi: 10.1016/S2352-3026(22)00134-X. [DOI] [PubMed] [Google Scholar]
- 172.Janjigian YY, Shitara K, Moehler M, Garrido M, Salman P, Shen L, Wyrwicz L, Yamaguchi K, Skoczylas T, Campos Bragagnoli A, et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet. 2021;398:27–40. doi: 10.1016/S0140-6736(21)00797-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Andre T, Shiu KK, Kim TW, Jensen BV, Jensen LH, Punt C, Smith D, Garcia-Carbonero R, Benavides M, Gibbs P, et al. Pembrolizumab in microsatellite-instability-high advanced colorectal cancer. N Engl J Med. 2020;383:2207–2218. doi: 10.1056/NEJMoa2017699. [DOI] [PubMed] [Google Scholar]
- 174.O’Malley David M, Neffa Maryna, de Speville Uribe Bernard Doger, Lugowska Iwona. Dual PD-1 and CTLA-4 checkpoint blockade using Balstilimab and Zalifrelimab combination as second-line treatment for advanced cervical cancer an open-label phase II study. J Clin Oncol. 2021;40:762–771. doi: 10.1200/JCO.21.02067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Duffy AG, Ulahannan SV, Makorova-Rusher O, Rahma O, Wedemeyer H, Pratt D, Davis JL, Hughes MS, Heller T, ElGindi M, et al. Tremelimumab in combination with ablation in patients with advanced hepatocellular carcinoma. J Hepatol. 2017;66:545–551. doi: 10.1016/j.jhep.2016.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Hellmann MD, Bivi N, Calderon B, Shimizu T, Delafontaine B, Liu ZT, Szpurka AM, Copeland V, Hodi FS, Rottey S, et al. Safety and immunogenicity of LY3415244, a bispecific antibody against TIM-3 and PD-L1, in patients with advanced solid tumors. Clin Cancer Res. 2021;27:2773–2781. doi: 10.1158/1078-0432.CCR-20-3716. [DOI] [PubMed] [Google Scholar]
- 177.Seto T, Nosaki K, Shimokawa M, Toyozawa R, Sugawara S, Hayashi H, Murakami H, Kato T, Niho S, Saka H, et al. Phase II study of atezolizumab with bevacizumab for non-squamous non-small cell lung cancer with high PD-L1 expression (@Be Study) J Immunother Cancer. 2022;10:e004025. doi: 10.1136/jitc-2021-004025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Reck M, Rodriguez-Abreu D, Robinson AG, Hui R, Csoszi T, Fulop A, Gottfried M, Peled N, Tafreshi A, Cuffe S, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. 2016;375:1823–1833. doi: 10.1056/NEJMoa1606774. [DOI] [PubMed] [Google Scholar]
- 179.Cho BC, Abreu DR, Hussein M, Cobo M, Patel AJ, Secen N, Lee KH, Massuti B, Hiret S, Yang JCH. Tiragolumab plus atezolizumab versus placebo plus atezolizumab as a first-line treatment for PD-L1-selected non-small-cell lung cancer (CITYSCAPE): primary and follow-up analyses of a randomised, double-blind, phase 2 study. Lancet Oncol. 2022;23:781–792. doi: 10.1016/S1470-2045(22)00226-1. [DOI] [PubMed] [Google Scholar]
- 180.Ye L, Zhang L, Li R, Pan X, Li J, Dou S, Jiang W, Wang C, Chen W, Zhu G. Combined all-trans retinoic acid with low-dose apatinib in treatment of recurrent/metastatic head and neck adenoid cystic carcinoma: A single-center, secondary analysis of a phase II study. Cancer Med. 2023;12:9144–9155. doi: 10.1002/cam4.5653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Hanna GJ, Oneil A, Cutler JM, Flynn M, Vijaykumar T, Clark JR, Wirth LJ, Lorch JH, Park JC, Mito JK, et al. A phase II trial of all-trans retinoic acid (ATRA) in advanced adenoid cystic carcinoma. Oral Oncol. 2021;119:105366. doi: 10.1016/j.oraloncology.2021.105366. [DOI] [PubMed] [Google Scholar]
- 182.Luu T, Frankel P, Beumer JH, Lim D, Cristea M, Appleman LJ, Lenz HJ, Gandara DR, Kiesel BF, Piekarz RL, Newman EM. Phase I trial of belinostat in combination with 13-cis-retinoic acid in advanced solid tumor malignancies: a California Cancer Consortium NCI/CTEP sponsored trial. Cancer Chemother Pharmacol. 2019;84:1201–1208. doi: 10.1007/s00280-019-03955-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Song M, DiPaola RS, Cracchiolo BM, Gibbon DG, Hellmann M, Nieves-Neira W, Vaidya A, Wagreich AR, Shih WJ, Rodriguez-Rodriguez L. Phase 2 trial of paclitaxel, 13-cis retinoic acid, and interferon alfa-2b in the treatment of advanced stage or recurrent cervical cancer. Int J Gynecol Cancer. 2014;24:1636–1641. doi: 10.1097/IGC.0000000000000258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Platten M, Bunse L, Wick A, Bunse T, Le Cornet L, Harting I, Sahm F, Sanghvi K, Tan CL, Poschke I, et al. A vaccine targeting mutant IDH1 in newly diagnosed glioma. Nature. 2021;592:463–468. doi: 10.1038/s41586-021-03363-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.de la Fuente MI, Colman H, Rosenthal M, Van Tine BA, Levacic D, Walbert T, Gan HK, Vieito M, Milhem MM, Lipford K, et al. Olutasidenib (FT-2102) in patients with relapsed or refractory IDH1-mutant glioma: a multicenter, open-label, phase Ib/II trial. Neuro Oncol. 2023;25:146–156. doi: 10.1093/neuonc/noac139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Abou-Alfa GK, Macarulla T, Javle MM, Kelley RK, Lubner SJ, Adeva J, Cleary JM, Catenacci DV, Borad MJ, Bridgewater J, et al. Ivosidenib in IDH1-mutant, chemotherapy-refractory cholangiocarcinoma (ClarIDHy): a multicentre, randomised, double-blind, placebo-controlled, phase 3 study. Lancet Oncol. 2020;21:796–807. doi: 10.1016/S1470-2045(20)30157-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Mellinghoff IK, Penas-Prado M, Peters KB, Burris HA, 3rd, Maher EA, Janku F, Cote GM, de la Fuente MI, Clarke JL, Ellingson BM, et al. Vorasidenib, a dual inhibitor of mutant IDH1/2, in recurrent or progressive glioma; results of a first-in-human phase I trial. Clin Cancer Res. 2021;27:4491–4499. doi: 10.1158/1078-0432.CCR-21-0611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.van den Bent MJ, Tesileanu CMS, Wick W, Sanson M, Brandes AA, Clement PM, Erridge S, Vogelbaum MA, Nowak AK, Baurain JF, et al. Adjuvant and concurrent temozolomide for 1p/19q non-co-deleted anaplastic glioma (CATNON; EORTC study 26053–22054): second interim analysis of a randomised, open-label, phase 3 study. Lancet Oncol. 2021;22:813–823. doi: 10.1016/S1470-2045(21)00090-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Eder JP, Doroshow DB, Do KT, Keedy VL, Sklar JS, Glazer P, Bindra R, Shapiro GI. Clinical Efficacy of Olaparib in IDH1/IDH2-Mutant Mesenchymal Sarcomas. JCO Precis Oncol. 2021;5:466–472. doi: 10.1200/PO.20.00247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Shah K, Panchal S, Patel B. Porcupine inhibitors: Novel and emerging anti-cancer therapeutics targeting the Wnt signaling pathway. Pharmacol Res. 2021;167:105532. doi: 10.1016/j.phrs.2021.105532. [DOI] [PubMed] [Google Scholar]
- 191.Katoh M, Katoh M. Grand Challenges in Molecular Medicine for Disease Prevention and Treatment Through Cyclical Innovation. Front Mol Med. 2021;1:720577. doi: 10.3389/fmmed.2021.720577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Gurney A, Axelrod F, Bond CJ, Cain J, Chartier C, Donigan L, Fischer M, Chaudhari A, Ji M, Kapoun AM, et al. Wnt pathway inhibition via the targeting of Frizzled receptors results in decreased growth and tumorigenicity of human tumors. Proc Natl Acad Sci U S A. 2012;109:11717–11722. doi: 10.1073/pnas.1120068109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Zhang Y, Wang X. Targeting the Wnt/beta-catenin signaling pathway in cancer. J Hematol Oncol. 2020;13:165. doi: 10.1186/s13045-020-00990-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Coluccia A, Bufano M, La Regina G, Puxeddu M, Toto A, Paone A, Bouzidi A, Musto G, Badolati N, Orlando V, et al. Anticancer Activity of (S)-5-Chloro-3-((3,5-dimethylphenyl)sulfonyl)-N-(1-oxo-1-((pyridin-4-ylmethyl)amino)propan-2-yl)-1H-indole-2-carboxamide (RS4690), a New Dishevelled 1 Inhibitor. Cancers (Basel) 2022;14:1358. doi: 10.3390/cancers14051358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Fujii N, You L, Xu Z, Uematsu K, Shan J, He B, Mikami I, Edmondson LR, Neale G, Zheng J, et al. An antagonist of dishevelled protein-protein interaction suppresses beta-catenin-dependent tumor cell growth. Cancer Res. 2007;67:573–579. doi: 10.1158/0008-5472.CAN-06-2726. [DOI] [PubMed] [Google Scholar]
- 196.Fenderico N, van Scherpenzeel RC, Goldflam M, Proverbio D, Jordens I, Kralj T, Stryeck S, Bass TZ, Hermans G, Ullman C, et al. Anti-LRP5/6 VHHs promote differentiation of Wnt-hypersensitive intestinal stem cells. Nat Commun. 2019;10:365. doi: 10.1038/s41467-018-08172-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Arques O, Chicote I, Puig I, Tenbaum SP, Argiles G, Dienstmann R, Fernandez N, Caratu G, Matito J, Silberschmidt D, et al. Tankyrase inhibition blocks Wnt/beta-Catenin pathway and reverts resistance to PI3K and AKT inhibitors in the treatment of colorectal cancer. Clin Cancer Res. 2016;22:644–656. doi: 10.1158/1078-0432.CCR-14-3081. [DOI] [PubMed] [Google Scholar]
- 198.Thorne CA, Hanson AJ, Schneider J, Tahinci E, Orton D, Cselenyi CS, Jernigan KK, Meyers KC, Hang BI, Waterson AG, et al. Small-molecule inhibition of Wnt signaling through activation of casein kinase 1alpha. Nat Chem Biol. 2010;6:829–836. doi: 10.1038/nchembio.453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Fang L, Zhu Q, Neuenschwander M, Specker E, Wulf-Goldenberg A, Weis WI, von Kries JP, Birchmeier W. A small-molecule antagonist of the beta-Catenin/TCF4 interaction blocks the self-renewal of cancer stem cells and suppresses tumorigenesis. Cancer Res. 2016;76:891–901. doi: 10.1158/0008-5472.CAN-15-1519. [DOI] [PubMed] [Google Scholar]
- 200.Che M, Kweon SM, Teo JL, Yuan YC, Melstrom LG, Waldron RT, Lugea A, Urrutia RA, Pandol SJ, Lai KKY. Targeting the CBP/beta-Catenin interaction to suppress activation of cancer-promoting pancreatic stellate cells. Cancers (Basel) 2020;12:1476. doi: 10.3390/cancers12061476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Menck K, Heinrichs S, Baden C, Bleckmann A. The WNT/ROR pathway in cancer: from signaling to therapeutic intervention. Cells. 2021;10:142. doi: 10.3390/cells10010142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Gu JW, Rizzo P, Pannuti A, Golde T, Osborne B, Miele L. Notch signals in the endothelium and cancer “stem-like” cells: opportunities for cancer therapy. Vasc Cell. 2012;4:7. doi: 10.1186/2045-824X-4-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Saha N, Robev D, Himanen JP, Nikolov DB. ADAM proteases: Emerging role and targeting of the non-catalytic domains. Cancer Lett. 2019;467:50–57. doi: 10.1016/j.canlet.2019.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.McCaw TR, Inga E, Chen H, Jaskula-Sztul R, Dudeja V, Bibb JA, Ren B, Rose JB. Gamma secretase inhibitors in cancer: a current perspective on clinical performance. Oncologist. 2021;26:e608–e621. doi: 10.1002/onco.13627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Wolfe MS. γ-Secretase modulators. Curr Alzheimer Res. 2007;4:571–573. doi: 10.2174/156720507783018299. [DOI] [PubMed] [Google Scholar]
- 206.Clara JA, Monge C, Yang Y, Takebe N. Targeting signalling pathways and the immune microenvironment of cancer stem cells - a clinical update. Nat Rev Clin Oncol. 2020;17:204–232. doi: 10.1038/s41571-019-0293-2. [DOI] [PubMed] [Google Scholar]
- 207.Yang L, Xie G, Fan Q, Xie J. Activation of the hedgehog-signaling pathway in human cancer and the clinical implications. Oncogene. 2010;29:469–481. doi: 10.1038/onc.2009.392. [DOI] [PubMed] [Google Scholar]
- 208.Zhang H, Sun Z, Liu Z, Song C. Overcoming the emerging drug resistance of smoothened: an overview of small-molecule SMO antagonists with antiresistance activity. Future Med Chem. 2018;10:2855–2875. doi: 10.4155/fmc-2018-0200. [DOI] [PubMed] [Google Scholar]
- 209.Pricl S, Cortelazzi B, Dal Col V, Marson D, Laurini E, Fermeglia M, Licitra L, Pilotti S, Bossi P, Perrone F. Smoothened (SMO) receptor mutations dictate resistance to vismodegib in basal cell carcinoma. Mol Oncol. 2015;9:389–397. doi: 10.1016/j.molonc.2014.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Quaglio D, Infante P, Di Marcotullio L, Botta B, Mori M. Hedgehog signaling pathway inhibitors: an updated patent review (2015-present) Expert Opin Ther Pat. 2020;30:235–250. doi: 10.1080/13543776.2020.1730327. [DOI] [PubMed] [Google Scholar]
- 211.Berardozzi S, Bernardi F, Infante P, Ingallina C, Toscano S, De Paolis E, Alfonsi R, Caimano M, Botta B, Mori M, et al. Synergistic inhibition of the Hedgehog pathway by newly designed Smo and Gli antagonists bearing the isoflavone scaffold. Eur J Med Chem. 2018;156:554–562. doi: 10.1016/j.ejmech.2018.07.017. [DOI] [PubMed] [Google Scholar]
- 212.Bufalieri F, Infante P, Bernardi F, Caimano M, Romania P, Moretti M, Lospinoso Severini L, Talbot J, Melaiu O, Tanori M, et al. ERAP1 promotes Hedgehog-dependent tumorigenesis by controlling USP47-mediated degradation of betaTrCP. Nat Commun. 2019;10:3304. doi: 10.1038/s41467-019-11093-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Moroishi T, Hayashi T, Pan WW, Fujita Y, Holt MV, Qin J, Carson DA, Guan KL. The hippo pathway kinases LATS1/2 suppress cancer immunity. Cell. 2016;167(1525–1539):e1517. doi: 10.1016/j.cell.2016.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Suarez-Carmona M, Lesage J, Cataldo D, Gilles C. EMT and inflammation: inseparable actors of cancer progression. Mol Oncol. 2017;11:805–823. doi: 10.1002/1878-0261.12095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Su C, Zhang J, Yarden Y, Fu L. The key roles of cancer stem cell-derived extracellular vesicles. Signal Transduct Target Ther. 2021;6:109. doi: 10.1038/s41392-021-00499-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Huang L, Wang F, Wang X, Su C, Wu S, Yang C, Luo M, Zhang J, Fu L. M2-like macrophage-derived exosomes facilitate metastasis in non-small-cell lung cancer by delivering integrin alphaVbeta3. MedComm. 2020;2023(4):e191. doi: 10.1002/mco2.191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19:1423–1437. doi: 10.1038/nm.3394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Sun HR, Wang S, Yan SC, Zhang Y, Nelson PJ, Jia HL, Qin LX, Dong QZ. Therapeutic strategies targeting cancer stem cells and their microenvironment. Front Oncol. 2019;9:1104. doi: 10.3389/fonc.2019.01104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Dongre A, Weinberg RA. New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat Rev Mol Cell Biol. 2019;20:69–84. doi: 10.1038/s41580-018-0080-4. [DOI] [PubMed] [Google Scholar]
- 220.Sakaguchi S, Mikami N, Wing JB, Tanaka A, Ichiyama K, Ohkura N. Regulatory T cells and human disease. Annu Rev Immunol. 2020;38:541–566. doi: 10.1146/annurev-immunol-042718-041717. [DOI] [PubMed] [Google Scholar]
- 221.Cheng K, Cai N, Zhu J, Yang X, Liang H, Zhang W. Tumor-associated macrophages in liver cancer: from mechanisms to therapy. Cancer Commun. 2022;42:1112–1140. doi: 10.1002/cac2.12345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Ghandadi M, Sahebkar A. Interleukin-6: a critical cytokine in cancer multidrug resistance. Curr Pharm Des. 2016;22:518–526. doi: 10.2174/1381612822666151124234417. [DOI] [PubMed] [Google Scholar]
- 223.Che D, Zhang S, Jing Z, Shang L, Jin S, Liu F, Shen J, Li Y, Hu J, Meng Q, Yu Y. Macrophages induce EMT to promote invasion of lung cancer cells through the IL-6-mediated COX-2/PGE2/beta-catenin signalling pathway. Mol Immunol. 2017;90:197–210. doi: 10.1016/j.molimm.2017.06.018. [DOI] [PubMed] [Google Scholar]
- 224.Condamine T, Ramachandran I, Youn J-I, Gabrilovich DI. Regulation of tumor metastasis by myeloid-derived suppressor cells. Annu Rev Med. 2015;66:97–110. doi: 10.1146/annurev-med-051013-052304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Tauriello DVF, Sancho E, Batlle E. Overcoming TGFbeta-mediated immune evasion in cancer. Nat Rev Cancer. 2022;22:25–44. doi: 10.1038/s41568-021-00413-6. [DOI] [PubMed] [Google Scholar]
- 226.Shi X, Yang J, Deng S, Xu H, Wu D, Zeng Q, Wang S, Hu T, Wu F, Zhou H. TGF-beta signaling in the tumor metabolic microenvironment and targeted therapies. J Hematol Oncol. 2022;15:135. doi: 10.1186/s13045-022-01349-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Wee P, Wang Z. Epidermal growth factor receptor cell proliferation signaling pathways. Cancers. 2017;9:52. doi: 10.3390/cancers9050052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Wang XK, He JH, Xu JH, Ye S, Wang F, Zhang H, Huang ZC, To KK, Fu LW. Afatinib enhances the efficacy of conventional chemotherapeutic agents by eradicating cancer stem-like cells. Cancer Res. 2014;74:4431–4445. doi: 10.1158/0008-5472.CAN-13-3553. [DOI] [PubMed] [Google Scholar]
- 229.Zhang Z, Guo X, To KKW, Chen Z, Fang X, Luo M, Ma C, Xu J, Yan S, Fu L. Olmutinib (HM61713) reversed multidrug resistance by inhibiting the activity of ATP-binding cassette subfamily G member 2 in vitro and in vivo. Acta Pharm Sin B. 2018;8:563–574. doi: 10.1016/j.apsb.2018.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Straussman R, Morikawa T, Shee K, Barzily-Rokni M, Qian ZR, Du J, Davis A, Mongare MM, Gould J, Frederick DT, et al. Tumour micro-environment elicits innate resistance to RAF inhibitors through HGF secretion. Nature. 2012;487:500–504. doi: 10.1038/nature11183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Shintani Y, Fujiwara A, Kimura T, Kawamura T, Funaki S, Minami M, Okumura M. IL-6 secreted from cancer-associated fibroblasts mediates chemoresistance in NSCLC by increasing epithelial-mesenchymal transition signaling. J Thorac Oncol. 2016;11:1482–1492. doi: 10.1016/j.jtho.2016.05.025. [DOI] [PubMed] [Google Scholar]
- 232.Dersh D, Hollý J, Yewdell JW. A few good peptides: MHC class I-based cancer immunosurveillance and immunoevasion. Nat Rev Immunol. 2020;21:116–128. doi: 10.1038/s41577-020-0390-6. [DOI] [PubMed] [Google Scholar]
- 233.Zhou F. Molecular mechanisms of IFN-gamma to up-regulate MHC class I antigen processing and presentation. Int Rev Immunol. 2009;28:239–260. doi: 10.1080/08830180902978120. [DOI] [PubMed] [Google Scholar]
- 234.Rock KL, Reits E, Neefjes J. Present yourself! By MHC class I and MHC class II molecules. Trends Immunol. 2016;37:724–737. doi: 10.1016/j.it.2016.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Jhunjhunwala S, Hammer C, Delamarre L. Antigen presentation in cancer: insights into tumour immunogenicity and immune evasion. Nat Rev Cancer. 2021;21:298–312. doi: 10.1038/s41568-021-00339-z. [DOI] [PubMed] [Google Scholar]
- 236.Jachetti E, Caputo S, Mazzoleni S, Brambillasca CS, Parigi SM, Grioni M, Piras IS, Restuccia U, Calcinotto A, Freschi M, et al. Tenascin-C protects cancer stem–like cells from immune surveillance by arresting T-cell activation. Can Res. 2015;75:2095–2108. doi: 10.1158/0008-5472.CAN-14-2346. [DOI] [PubMed] [Google Scholar]
- 237.Zhong M, Zhong C, Cui W, Wang G, Zheng G, Li L, Zhang J, Ren R, Gao H, Wang T, et al. Induction of tolerogenic dendritic cells by activated TGF-β/Akt/Smad2 signaling in RIG-I-deficient stemness-high human liver cancer cells. BMC Cancer. 2019;19:439. doi: 10.1186/s12885-019-5670-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Topalian SL, Taube JM, Anders RA, Pardoll DM. Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat Rev Cancer. 2016;16:275–287. doi: 10.1038/nrc.2016.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Garris CS, Arlauckas SP, Kohler RH, Trefny MP, Garren S, Piot C, Engblom C, Pfirschke C, Siwicki M, Gungabeesoon J, et al. Successful Anti-PD-1 cancer immunotherapy requires T cell-dendritic cell crosstalk involving the Cytokines IFN-gamma and IL-12. Immunity. 2018;49(1148–1161):e1147. doi: 10.1016/j.immuni.2018.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Verma V, Shrimali RK, Ahmad S, Dai W, Wang H, Lu S, Nandre R, Gaur P, Lopez J, Sade-Feldman M, et al. PD-1 blockade in subprimed CD8 cells induces dysfunctional PD-1(+)CD38(hi) cells and anti-PD-1 resistance. Nat Immunol. 2019;20:1231–1243. doi: 10.1038/s41590-019-0441-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Kvedaraite Egle, Ginhoux Florent. Human dendritic cells in cancer. Sci Immunol. 2022;7:eabm9409. doi: 10.1126/sciimmunol.abm9409. [DOI] [PubMed] [Google Scholar]
- 242.Shklovskaya E, Lee JH, Lim SY, Stewart A, Pedersen B, Ferguson P, Saw RP, Thompson JF, Shivalingam B, Carlino MS, et al. Tumor MHC expression guides first-line immunotherapy selection in Melanoma. Cancers (Basel) 2020;12:3374. doi: 10.3390/cancers12113374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Kobayashi KS, van den Elsen PJ. NLRC5: a key regulator of MHC class I-dependent immune responses. Nat Rev Immunol. 2012;12:813–820. doi: 10.1038/nri3339. [DOI] [PubMed] [Google Scholar]
- 244.Shklovskaya E, Rizos H. MHC class I deficiency in solid tumors and therapeutic strategies to overcome it. Int J Mol Sci. 2021;22:6741. doi: 10.3390/ijms22136741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Chapman NM, Boothby MR, Chi H. Metabolic coordination of T cell quiescence and activation. Nat Rev Immunol. 2020;20:55–70. doi: 10.1038/s41577-019-0203-y. [DOI] [PubMed] [Google Scholar]
- 246.Xu W, Atkins MB, McDermott DF. Checkpoint inhibitor immunotherapy in kidney cancer. Nat Rev Urol. 2020;17:137–150. doi: 10.1038/s41585-020-0282-3. [DOI] [PubMed] [Google Scholar]
- 247.Budimir N, Thomas GD, Dolina JS, Salek-Ardakani S. Reversing T-cell exhaustion in cancer: lessons learned from PD-1/PD-L1 immune checkpoint blockade. Cancer Immunol Res. 2022;10:146–153. doi: 10.1158/2326-6066.CIR-21-0515. [DOI] [PubMed] [Google Scholar]
- 248.Madan V, Koeffler HP. Differentiation therapy of myeloid leukemia: four decades of development. Haematologica. 2021;106:26–38. doi: 10.3324/haematol.2020.262121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.de The H. Differentiation therapy revisited. Nat Rev Cancer. 2018;18:117–127. doi: 10.1038/nrc.2017.103. [DOI] [PubMed] [Google Scholar]
- 250.Turabi KS, Deshmukh A, Paul S, Swami D, Siddiqui S, Kumar U, Naikar S, Devarajan S, Basu S, Paul MK, Aich J. Drug repurposing—an emerging strategy in cancer therapeutics. Naunyn Schmiedebergs Arch Pharmacol. 2022;395:1139–1158. doi: 10.1007/s00210-022-02263-x. [DOI] [PubMed] [Google Scholar]
- 251.Traci E. LaMoia GIS: cellular and molecular mechanisms of metformin action. Endocr Rev. 2021;42:77–96. doi: 10.1210/endrev/bnaa023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Maehara O, Ohnishi S, Asano A, Suda G, Natsuizaka M, Nakagawa K, Kobayashi M, Sakamoto N, Takeda H. Metformin regulates the expression of CD133 through the AMPK-CEBPbeta pathway in hepatocellular carcinoma cell lines. Neoplasia. 2019;21:545–556. doi: 10.1016/j.neo.2019.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Brown JR, Chan DK, Shank JJ, Griffith KA, Fan H, Szulawski R, Yang K, Reynolds RK, Johnston C, McLean K, et al. Phase II clinical trial of metformin as a cancer stem cell-targeting agent in ovarian cancer. JCI Insight. 2020;5:e133247. doi: 10.1172/jci.insight.133247. [DOI] [PMC free article] [PubMed] [Google Scholar]