Abstract
Purpose
Osteoarthritis (OA) is a combination of degeneration and destruction of articular cartilage due to mechanical stress, secondary synovitis, and bone remodelling. In recent years, early knee OA, a preliminary stage of structural failure in OA, has attracted attention as a potential target for therapy to prevent the onset of OA. Intra-articular administration of monoiodoacetic acid (MIA) induces OA-like symptoms, and low doses of MIA induce early OA like symptoms. In this experiment, a low-dose of MIA was induced to early OA model mice, which were then irradiated with low-intensity pulsed ultrasound (LIPUS) to examine whether LIPUS improves symptoms of early OA.
Methods
After 4 weeks of LIPUS irradiation, articular cartilage was observed at 1 and 4 weeks. The Osteoarthritis Research Society International (OARSI) scores were calculated using Safranin-O staining results. Cartilage degeneration was detected using Denatured Collagen Detection Reagent (DCDR).
Results
We observed a significant decrease in OARSI scores in the LIPUS irradiated group at week 4. The non-LIPUS group showed widespread areas of double positivity for Type II collagen and DCDR, whereas the LIPUS group showed only a small number of DCDR-positive areas. In addition, macrophage numbers counted in the articular capsule at week 1 showed a significant decrease in the LIPUS irradiated group. Lubricin detection showed that lubricin positive cell number was significantly increased by LIPUS irradiation at week 4.
Conclusions
These results suggest that LIPUS attenuates cartilage degeneration in early OA by relieving inflammation and enhancing the inhibitory effect of lubricin on cartilage degeneration.
Keywords: Low-intensity pulsed ultrasound, Early osteoarthritis, Low-dose monoiodoacetic acid, Collagen degradation
Background
Osteoarthritis (OA) is a combination of degeneration and destruction of articular cartilage due to mechanical stress, secondary synovitis, and bone remodelling [2, 11, 21]. As the most prevalent musculoskeletal disease, OA has one of the fastest growing patient populations worldwide [1, 6]. OA currently affects more than 528 million people globally, suggesting a nearly 10% increase in the last 30 years, since 1990 [6]. OA causes chronic pain and gait disturbance, impairs daily activities, and reduces quality of life [11]. Furthermore, it is a progressive disease that cannot be reversed or stopped after onset. In recent years, early knee OA, a preliminary stage of structural failure in OA, has attracted attention as a potential target for therapy to prevent the onset of OA [20]. While an accepted definition of early-stage OA has not yet been established, it is generally described as a condition in which the symptoms of OA, mainly pain, manifest above a certain level, despite the absence of joint crevice narrowing on plain radiographs [19, 22]. Abnormal findings on magnetic resonance imaging (MRI), such as cartilage defects, meniscus injuries, and osteophytes, are found in patients over 40 years of age, even in those with no specific knee symptoms or history of trauma [7]. Cartilage degeneration, bone marrow lesions, and semilunar deviation on simple MRI were associated with knee pain in a knee joint determined to be KL grade 0 on simple radiographs [36]. It has been suggested that early initiation of treatment may increase the possibility of preventing the onset and progression of OA.
While various OA models have been developed, the monoiodoacetic acid (MIA) administration model is used as one standard OA model. Intra-articular administration of MIA induces inflammation in joints by inhibiting glycation of chondrocytes through inhibition of glyceraldehyde-3-phosphatase dehydrogenase, and induces various degrees of OA progression depending on the dose of MIA administration [28]. The conventional dosage of MIA (1–3 mg) has a strong effect on cartilage destruction [3]. However, a lower dose of MIA can induce cartilage degeneration without bone destruction, which is more similar to the joint degeneration observed in human early-OA. Rats treated with low-dose MIA are reported as animal models of early OA [41].
Low-intensity pulsed ultrasound (LIPUS) is widely used in bone fracture healing [14]. LIPUS irradiation significantly shortens treatment times in patients with bone fractures. Several studies have reported the usefulness of LIPUS for the treatment of bone fractures, delayed union, and non-union [4, 31, 42]. In addition, it affects not only osteoblasts, but also chondrocytes and inflammatory immune cells [25, 33, 44]. Furthermore, LIPUS reportedly provides pain-relieving effects and is, therefore, used to treat patients in advanced stages of OA; improvements in Visual Analogue Scale and Western Ontario and McMaster Universities Arthritis Index scores have been observed [5]. However, these results only demonstrate the analgesic effect of LIPUS in post-onset OA, not its efficacy in inhibiting the onset of OA. We hypothesised that LIPUS can inhibit the progression of early OA. Therefore, in this study, we examined whether LIPUS delays the progression of OA by attenuating cartilage degeneration and increasing lubricin expression in an early OA mouse model.
Methods
Ethics statement
All animal experiments conformed to the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health and the ARRIVE guidelines (http://www.nc3rs.org/ARRIVE). All the experiments were approved by the Institutional Committee of Laboratory Animal Experimentation of the authors’ affiliated institutions.
Animals
Female ICR mice (31–35 g, 13 weeks old) were acclimatised for 1 week before the experiments. The mice were housed under standard laboratory conditions (12-h light/dark cycle, 25 °C). They were provided ad libitum access to water and food containing 0.98% calcium, 0.80% phosphorus, and vitamin D3 (3,169 IU/kg) for pair feeding (Labo MR Stock, Nosan corp., Kanagawa, Japan).
MIA injection and LIPUS irradiation
Hair from the skin of the right hind limb was removed with depilatory cream (Reckitt Benckiser Japan Ltd., Tokyo, Japan) for LIPUS irradiation. The mice were anaesthetised with isoflurane (Mylan Inc., Pittsburgh, PA, USA) using an animal anaesthesia machine (SFSFB02, DS Pharma Biomedical Co. Ltd., Osaka, Japan) The conditions were as follows: for induction, concentration = 4%, flow rate = 2.8 L/min; for maintenance, concentration = 2%, flow rate = 2.8 L/min. The anaesthetised mice were placed on a heated pad maintained at 35 °C during the operation. The mice were administered with MIA (0.1 mg) by intra-articular injection under anaesthesia [41]. These mice were randomised by body weight into the LIPUS ( −) group (control) and LIPUS ( +) group (experimental). On the day after MIA administration, ultrasound gel was applied to an ultrasound transducer (Nippon Sigmax Co. Ltd., Tokyo, Japan), which was placed over the exposed skin of the right knee. Subsequently, LIPUS treatment was administered 5 days per week for 20 min per day. The ultrasound exposure conditions were as follows: effective transducer area, 6.85 cm2; intensity, 30 mW/cm2 with a 20% duty cycle; and pulse frequency, 2.0 MHz with a 1 kHz repeat rate. At weeks 1 and 4, mice were euthanised by posterior cervical dislocation, following sufficient isoflurane inhalation, to prepare tissue sections of the knee joint.
Histological analysis
Mouse whole knee joints were harvested at weeks 1 and 4, fixed with 4% paraformaldehyde (Fujifilm Wako Pure Chemical Corp., Osaka, Japan), demineralised with ethylenediaminetetraacetic acid (Dojindo Laboratories, Kumamoto, Japan), and embedded in paraffin to prepare 5-μm-thick sections of frontal plane tissue. Histological sections were visualised under a fluorescence microscope (BZ-9000; Keyence, Osaka, Japan).
Safranin O staining
Safranin O staining results were used to calculate the osteoarthritis research society international (OARSI) scores of all four quadrants of the joint: medial femoral condyle (MFC), medial tibial plateau (MTP), lateral femoral condyle (LFC), and lateral tibial plateau (LTP) [10]. A score of 0 represents normal cartilage, 0.5 = loss of PG with an intact surface, 1 = superficial fibrillation without loss of cartilage, 2 = vertical clefts and loss of surface lamina (any % or joint surface area).
Immunofluorescence staining
To detect degenerated collagen on the articular cartilage, the tissue sections were treated with Denatured Collagen Detection Reagent (DCDR; Funakoshi Co., Ltd., Tokyo, Japan) [39, 40]. They were then treated with anti-type II collagen antibody (Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA). The tissue sections were treated with F4/80 antibody (Bio-Rad Laboratories Inc., Hercules, CA, USA) [8] and anti-IL-6 antibody (Cell Signaling Technology, Danvers, MA, USA). The tissue sections were treated with anti-Aggrecan antibody or anti- Matrix metalloproteinase 13 (MMP-13) antibodies (Proteintech Group Inc., IL, USA). The tissue sections were treated with anti-type II collagen antibody and anti-lubricin antibody (Novus Biologicals, LLC., Central CO, CO, USA). The immunofluorescence-positive cells in the articular capsule were counted using the ImageJ software [29, 34].
Statistical analysis
The primary goal of this study was to observe the histological changes of MIA early-OA model, with and without LIPUS irradiation. Our sample size was determined by referring to a previous study that examined OARSI histological score in a mouse MIA OA model [41]. Calculations were performed according to this difference with 90% statistical power and α level of 0.05, and it was determined that at least 4 mice were needed. Parametric data are shown as mean ± standard error of the mean. P-values of < 0.05 were considered statistically significant. The significance of differences between the two groups was determined using an independent sample T test. Data were processed using the Ekuseru–Toukei 2012 software (Social Survey Research Information Co. Ltd., Tokyo, Japan).
Results
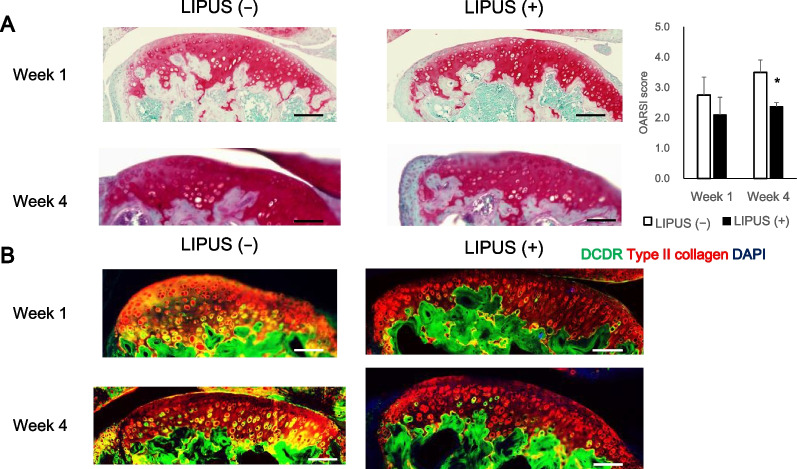
We compared cartilage degeneration in the LIPUS ( −) and LIPUS ( +) groups for Safranin O staining (Fig. 1A). At week 1, there was nearly no loss of safranin and no significant difference between groups; however, at week 4, a significant increase of OARSI score was observed in the LIPUS ( −) group. In addition, collagen denaturation of cartilage was detected by DCDR (Fig. 1B). DCDR is a reagent that binds specifically to collagen fibres that have not formed a triple helix in denaturation [39]. At both weeks 1 and 4, the LIPUS ( −) group showed widespread areas of double positivity for type II collagen and DCDR, whereas the LIPUS ( +) group showed few DCDR-positive areas. These results suggest that LIPUS irradiation inhibits low-dose MIA-induced cartilage degeneration.
Fig. 1.
LIPUS attenuates cartilage degeneration in early osteoarthritis-like model mice. A Safranin O-stained histology sections of articular cartilage after weeks 1 and 4. The results were used to calculate the osteoarthritis research society international (OARSI) scores of all four quadrants of the joint: medial femoral condyle (MFC), medial tibial plateau (MTP), lateral femoral condyle (LFC), and lateral tibial plateau (LTP). Values are expressed as mean ± standard error of the mean (SEM), n = 4; * = p < 0.05. (Scale bar: 100 μm). B Collagen degeneration on the articular cartilage was detected with Denatured Collagen Detection Reagent (green). Type II collagen was immune-stained (red). (Scale bar: 100 μm)
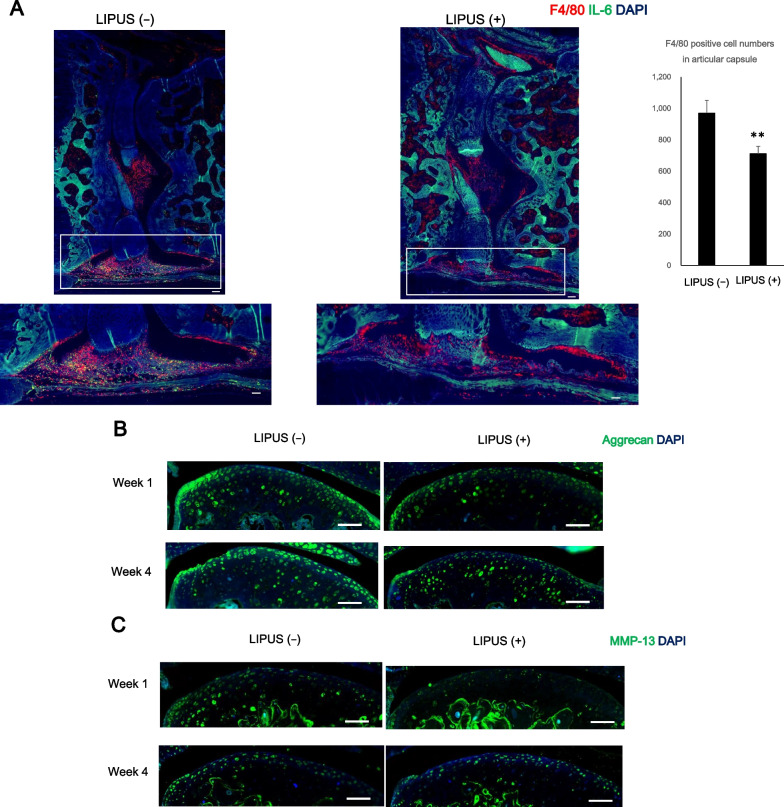
To verify whether LIPUS attenuates cartilage degeneration by suppressing inflammation, the number of F4/80-positive cells in the articular capsule was measured (Fig. 2A). Infiltration of inflammatory cells was observed by staining of the F4/80 molecule, one of the markers of mouse macrophage tissue [8]. In week 1, there was a significant decrease in the number of F4/80 positive cells in the LIPUS ( +) group compared with the LIPUS ( −) group. Aggrecan positive cells and MMP-13 positive cells were also observed (Fig. 2B and C). No significant difference was observed in the Aggrecan-positive cells with or without LIPUS irradiation; however, the MMP-13-positive cells in the LIPUS (–) group were found to be particularly abundant in the middle zone in week 1.
Fig. 2.
LIPUS attenuates infiltration of F4/80 positive macrophage in early osteoarthritis-like model mice. A Immunofluorescent staining of mouse knee arthrodesis using F4/80 antibody (red) and anti-IL-6 antibody (green). F4/80 positive cells were counted. Values are expressed as mean ± standard error of the mean (SEM), n = 4; ** = p < 0.01. (Scale bar: 100 μm). B Immunofluorescent staining of mouse articular cartilage at weeks 1 and 4, using anti-Aggrecan antibody (Green). (Scale bar: 100 μm). C Immunofluorescent staining of mouse articular cartilage at weeks 1 and 4, using anti-MMP-13 antibody (Green). (Scale bar: 100 μm)
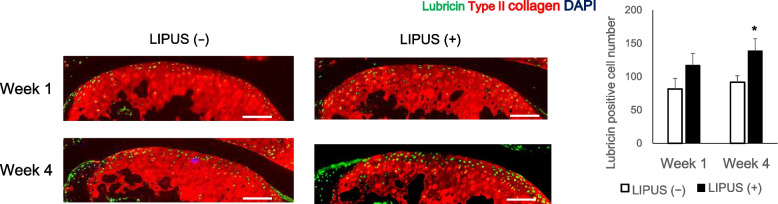
Mechanical stress on the surface layer of cartilage, such as shear stress and running, induces lubricin expression [26]. Therefore, we examined whether LIPUS irradiation increases lubricin expression. The lubricin positive cell numbers in the superficial layer of cartilage were increased in the LIPUS ( +) group (Fig. 3). These findings suggest that LIPUS suppresses inflammation and cartilage degeneration in early OA.
Fig. 3.
Immunofluorescent staining against lubricin (green) and type II collagen (red) in the articular cartilage sections. Lubricin-positive cells were counted. Values are expressed as mean ± standard error of the mean (SEM), n = 4; * = p < 0.05. (Scale bar: 100 μm)
Discussion
Matrix metalloproteinases (MMPs) and a disintegrin and metalloproteinase with thrombospondin motifs (ADAMTSs) degrade type II collagen and aggrecan, and exacerbate OA; their expression is promoted by inflammation [43]. In addition, these fragments of cartilage component proteins stimulate synovial cells and chondrocytes, inducing further inflammation and progression of OA [12, 38]. Therefore, suppression of inflammation within OA joints is considered important for maintaining joint function [38]. In this study, LIPUS irradiation reduced macrophage infiltration into the joint capsule in the acute inflammation 1 week after MIA administration, suggesting an inhibitory effect on inflammation. LIPUS promotes the secretion of extracellular vesicles and the suppression of inflammation [18].
Lubricin is a glycoprotein secreted into synovial fluid by superficial zone chondrocytes and synoviocytes [17, 32]. It reduces and protects the frictional resistance of the cartilage surface and plays an important role in maintaining cartilage homeostasis. Since TRPV2, a mechano-sensor in the superficial zone of articular cartilage, induces lubricin via cAMP response element binding protein (CREB), we postulated that LIPUS also activates this pathway [24]. Our study found that the number of lubricin-positive cells was increased in the LIPUS-irradiated cartilage surfaces. Furthermore, LIPUS suppresses MMP13 production from chondrocytes [15, 23, 27, 35]. These results suggest that LIPUS induces lubricin expression to provide further protection to cartilage integrity.
This study had some limitations. We showed that LIPUS irradiation suppressed inflammation and cartilage degeneration in early OA mice. However, deviation of the medial meniscus is frequently observed in patients with early OA [13]. Because the study was conducted only on MIA-induced OA mice, validation using surgical OA model mice, such as ligament-tearing OA models, is needed to examine the effects in human OA patients [16]. Next, although a histological evaluation was performed in this study, RNA should also be extracted from cartilage and analysed by reverse transcription-quantitative polymerase chain reaction (RT-qPCR). This is because quantitative expression analysis using RT-qPCR has become an indispensable research method in the field of orthopaedics to elucidate the molecular mechanisms of pathogenesis and disease progression.
The efficiency of early case detection and diagnosis in primary care enables health care providers to proactively and significantly reduce the burden of disease through appropriate management, such as by addressing certain lifestyle-related risk factors for disease progression [37]. One of the reasons for focusing on early-stage OA is to prevent the progression of OA by starting treatment early [9]. Although moderate exercise can increase lubricin expression [30], LIPUS irradiation may improve symptoms in patients having difficulty with physical therapy. Intervention in early OA at the onset of pain using LIPUS, a non-invasive technique, may reduce the incidence of OA. By investigating the ultrasound irradiation conditions optimised for lubricin induction and inflammation suppression, LIPUS may serve as a potential candidate for the treatment of early OA.
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
Abbreviations
- OA
Osteoarthritis
- MIA
Monoiodoacetic acid
- LIPUS
Low-intensity pulsed ultrasound
- OARSI
Osteoarthritis research society international
- DCDR
Denatured Collagen Detection Reagent
- MMP
Matrix metalloproteinase
- ADAMTSs
A disintegrin and metalloproteinase with thrombospondin motifs
- CREB
CAMP response element binding protein
Authors’ contributions
YK: Conceptualisation, Methodology, Investigation, Data Curation, Writing—Original Draft. TW: Methodology, Investigation. All authors read and approved the final manuscript.
Funding
This study was funded by Nippon Sigmax Co. Ltd. No specific grants were received for this project.
Availability of data and materials
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
All animal experiments conformed to the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health and the ARRIVE guidelines (http://www.nc3rs.org/ARRIVE). All experiments were approved by the Institutional Committee of Laboratory Animal Experimentation of the authors’ affiliated Yokohama University of Pharmacy (approval umber: 2018–004).
Consent for publication
Not applicable.
Competing interests
YK and TW are employees of Nippon Sigmax Co. Ltd.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Allen KD, Thoma LM, Golightly YM. Epidemiology of osteoarthritis. Osteoarthritis Cartilage. 2022;30:184–195. doi: 10.1016/j.joca.2021.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bijlsma JW, Berenbaum F, Lafeber FP. Osteoarthritis: an update with relevance for clinical practice. Lancet. 2011;377:2115–2126. doi: 10.1016/S0140-6736(11)60243-2. [DOI] [PubMed] [Google Scholar]
- 3.Bryk M, Chwastek J, Mlost J, Kostrzewa M, Starowicz K. Sodium monoiodoacetate dose-dependent changes in matrix metalloproteinases and inflammatory components as prognostic factors for the progression of osteoarthritis. Front Pharmacol. 2021;12:643605. doi: 10.3389/fphar.2021.643605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Busse JW, Bhandari M, Kulkarni AV, Tunks E. The effect of low-intensity pulsed ultrasound therapy on time to fracture healing: a meta-analysis. CMAJ. 2002;166:437–441. [PMC free article] [PubMed] [Google Scholar]
- 5.Chen H, Wang Z, Zhang X, Sun M. Effects of low-intensity pulsed ultrasound on knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Clin Rehabil. 2022;36:1153–1169. doi: 10.1177/02692155221097035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Collaborators GDaI, Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020;396:1204–1222. doi: 10.1016/S0140-6736(20)30925-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Culvenor AG, Øiestad BE, Hart HF, Stefanik JJ, Guermazi A, Crossley KM. Prevalence of knee osteoarthritis features on magnetic resonance imaging in asymptomatic uninjured adults: a systematic review and meta-analysis. Br J Sports Med. 2019;53:1268–1278. doi: 10.1136/bjsports-2018-099257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dos Anjos CA. F4/80 as a major macrophage marker: the case of the peritoneum and spleen. Results Probl Cell Differ. 2017;62:161–179. doi: 10.1007/978-3-319-54090-0_7. [DOI] [PubMed] [Google Scholar]
- 9.Felson DT, Hodgson R. Identifying and treating preclinical and early osteoarthritis. Rheum Dis Clin North Am. 2014;40:699–710. doi: 10.1016/j.rdc.2014.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Glasson SS, Chambers MG, Van Den Berg WB, Little CB. The OARSI histopathology initiative - recommendations for histological assessments of osteoarthritis in the mouse. Osteoarthritis Cartilage. 2010;18(Suppl 3):S17–23. doi: 10.1016/j.joca.2010.05.025. [DOI] [PubMed] [Google Scholar]
- 11.Goldring MB, Goldring SR. Articular cartilage and subchondral bone in the pathogenesis of osteoarthritis. Ann N Y Acad Sci. 2010;1192:230–237. doi: 10.1111/j.1749-6632.2009.05240.x. [DOI] [PubMed] [Google Scholar]
- 12.Grässel S, Zaucke F, Madry H. Osteoarthritis: novel molecular mechanisms increase our understanding of the disease pathology. J Clin Med. 2021;10:1938. doi: 10.3390/jcm10091938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hada S, Ishijima M, Kaneko H, Kinoshita M, Liu L, Sadatsuki R, et al. Association of medial meniscal extrusion with medial tibial osteophyte distance detected by T2 mapping MRI in patients with early-stage knee osteoarthritis. Arthritis Res Ther. 2017;19:201. doi: 10.1186/s13075-017-1411-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Harrison A, Alt V. Low-intensity pulsed ultrasound (LIPUS) for stimulation of bone healing - A narrative review. Injury. 2021;52(Suppl 2):S91–s96. doi: 10.1016/j.injury.2021.05.002. [DOI] [PubMed] [Google Scholar]
- 15.Ito A, Aoyama T, Yamaguchi S, Zhang X, Akiyama H, Kuroki H. Low-intensity pulsed ultrasound inhibits messenger RNA expression of matrix metalloproteinase-13 induced by interleukin-1β in chondrocytes in an intensity-dependent manner. Ultrasound Med Biol. 2012;38:1726–1733. doi: 10.1016/j.ultrasmedbio.2012.06.005. [DOI] [PubMed] [Google Scholar]
- 16.Kamekura S, Hoshi K, Shimoaka T, Chung U, Chikuda H, Yamada T, et al. Osteoarthritis development in novel experimental mouse models induced by knee joint instability. Osteoarthritis Cartilage. 2005;13:632–641. doi: 10.1016/j.joca.2005.03.004. [DOI] [PubMed] [Google Scholar]
- 17.Kozhemyakina E, Zhang M, Ionescu A, Ayturk UM, Ono N, Kobayashi A, et al. Identification of a Prg4-expressing articular cartilage progenitor cell population in mice. Arthritis Rheumatol. 2015;67:1261–1273. doi: 10.1002/art.39030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li X, Zhong Y, Zhou W, Song Y, Li W, Jin Q, et al. Low-intensity pulsed ultrasound (LIPUS) enhances the anti-inflammatory effects of bone marrow mesenchymal stem cells (BMSCs)-derived extracellular vesicles. Cell Mol Biol Lett. 2023;28:9. doi: 10.1186/s11658-023-00422-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Luyten FP, Bierma-Zeinstra S, Dell'Accio F, Kraus VB, Nakata K, Sekiya I, et al. Toward classification criteria for early osteoarthritis of the knee. Semin Arthritis Rheum. 2018;47:457–463. doi: 10.1016/j.semarthrit.2017.08.006. [DOI] [PubMed] [Google Scholar]
- 20.Mahmoudian A, Lohmander LS, Mobasheri A, Englund M, Luyten FP. Early-stage symptomatic osteoarthritis of the knee - time for action. Nat Rev Rheumatol. 2021;17:621–632. doi: 10.1038/s41584-021-00673-4. [DOI] [PubMed] [Google Scholar]
- 21.Martel-Pelletier J, Boileau C, Pelletier JP, Roughley PJ. Cartilage in normal and osteoarthritis conditions. Best Pract Res Clin Rheumatol. 2008;22:351–384. doi: 10.1016/j.berh.2008.02.001. [DOI] [PubMed] [Google Scholar]
- 22.Migliore A, Scirè CA, Carmona L, Herrero-Beaumont G, Bizzi E, Branco J, et al. The challenge of the definition of early symptomatic knee osteoarthritis: a proposal of criteria and red flags from an international initiative promoted by the Italian Society for Rheumatology. Rheumatol Int. 2017;37:1227–1236. doi: 10.1007/s00296-017-3700-y. [DOI] [PubMed] [Google Scholar]
- 23.Naito K, Watari T, Muta T, Furuhata A, Iwase H, Igarashi M, et al. Low-intensity pulsed ultrasound (LIPUS) increases the articular cartilage type II collagen in a rat osteoarthritis model. J Orthop Res. 2010;28:361–369. doi: 10.1002/jor.20995. [DOI] [PubMed] [Google Scholar]
- 24.Nakamoto H, Katanosaka Y, Chijimatsu R, Mori D, Xuan F, Yano F, et al. Involvement of transient receptor potential vanilloid Channel 2 in the induction of lubricin and suppression of ectopic endochondral ossification in mouse articular cartilage. Arthritis Rheumatol. 2021;73:1441–1450. doi: 10.1002/art.41684. [DOI] [PubMed] [Google Scholar]
- 25.Nishida T, Kubota S, Aoyama E, Yamanaka N, Lyons KM, Takigawa M. Low-intensity pulsed ultrasound (LIPUS) treatment of cultured chondrocytes stimulates production of CCN family protein 2 (CCN2), a protein involved in the regeneration of articular cartilage: mechanism underlying this stimulation. Osteoarthritis Cartilage. 2017;25:759–769. doi: 10.1016/j.joca.2016.10.003. [DOI] [PubMed] [Google Scholar]
- 26.Ogawa H, Kozhemyakina E, Hung HH, Grodzinsky AJ, Lassar AB. Mechanical motion promotes expression of Prg4 in articular cartilage via multiple CREB-dependent, fluid flow shear stress-induced signaling pathways. Genes Dev. 2014;28:127–139. doi: 10.1101/gad.231969.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Parvizi J, Wu CC, Lewallen DG, Greenleaf JF, Bolander ME. Low-intensity ultrasound stimulates proteoglycan synthesis in rat chondrocytes by increasing aggrecan gene expression. J Orthop Res. 1999;17:488–494. doi: 10.1002/jor.1100170405. [DOI] [PubMed] [Google Scholar]
- 28.Pitcher T, Sousa-Valente J, Malcangio M (2016) The monoiodoacetate model of osteoarthritis pain in the mouse. J Vis Exp 111:53746 [DOI] [PMC free article] [PubMed]
- 29.Rasband WS, ImageJ, U. S. National Institutes of Health, Bethesda, Maryland, USA, 1997-2018. https://imagej.nih.gov/ij/.
- 30.Roggio F, Petrigna L, Trovato B, Di Rosa M, Musumeci G. The role of lubricin, irisin and exercise in the prevention and treatment of osteoarthritis. Int J Mol Sci. 2023;24:5126. doi: 10.3390/ijms24065126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rutten S, Nolte PA, Guit GL, Bouman DE, Albers GH. Use of low-intensity pulsed ultrasound for posttraumatic nonunions of the tibia: a review of patients treated in the Netherlands. J Trauma. 2007;62:902–908. doi: 10.1097/01.ta.0000238663.33796.fb. [DOI] [PubMed] [Google Scholar]
- 32.Saito T. The superficial zone of articular cartilage. Inflamm Regen. 2022;42:14. doi: 10.1186/s41232-022-00202-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sang F, Xu J, Chen Z, Liu Q, Jiang W. Low-intensity pulsed ultrasound alleviates osteoarthritis condition through focal adhesion kinase-mediated chondrocyte proliferation and differentiation. Cartilage. 2021;13:196s–203s. doi: 10.1177/1947603520912322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012;9:671–675. doi: 10.1038/nmeth.2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sekino J, Nagao M, Kato S, Sakai M, Abe K, Nakayama E, et al. Low-intensity pulsed ultrasound induces cartilage matrix synthesis and reduced MMP13 expression in chondrocytes. Biochem Biophys Res Commun. 2018;506:290–297. doi: 10.1016/j.bbrc.2018.10.063. [DOI] [PubMed] [Google Scholar]
- 36.Sharma L, Chmiel JS, Almagor O, Dunlop D, Guermazi A, Bathon JM, et al. Significance of preradiographic magnetic resonance imaging lesions in persons at increased risk of knee osteoarthritis. Arthritis Rheumatol. 2014;66:1811–1819. doi: 10.1002/art.38611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shimizu H, Shimoura K, Iijima H, Suzuki Y, Aoyama T. Functional manifestations of early knee osteoarthritis: a systematic review and meta-analysis. Clin Rheumatol. 2022;41:2625–2634. doi: 10.1007/s10067-022-06150-x. [DOI] [PubMed] [Google Scholar]
- 38.Sokolove J, Lepus CM. Role of inflammation in the pathogenesis of osteoarthritis: latest findings and interpretations. Ther Adv Musculoskelet Dis. 2013;5:77–94. doi: 10.1177/1759720X12467868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Takita KK, Fujii KK, Ishii K, Koide T. Structural optimization of cyclic peptides that efficiently detect denatured collagen. Org Biomol Chem. 2019;17:7380–7387. doi: 10.1039/c9ob01042d. [DOI] [PubMed] [Google Scholar]
- 40.Takita KK, Fujii KK, Kadonosono T, Masuda R, Koide T. Cyclic peptides for efficient detection of collagen. ChemBioChem. 2018;19:1613–1617. doi: 10.1002/cbic.201800166. [DOI] [PubMed] [Google Scholar]
- 41.Udo M, Muneta T, Tsuji K, Ozeki N, Nakagawa Y, Ohara T, et al. Monoiodoacetic acid induces arthritis and synovitis in rats in a dose- and time-dependent manner: proposed model-specific scoring systems. Osteoarthritis Cartilage. 2016;24:1284–1291. doi: 10.1016/j.joca.2016.02.005. [DOI] [PubMed] [Google Scholar]
- 42.Warden SJ, Fuchs RK, Kessler CK, Avin KG, Cardinal RE, Stewart RL. Ultrasound produced by a conventional therapeutic ultrasound unit accelerates fracture repair. Phys Ther. 2006;86:1118–1127. [PubMed] [Google Scholar]
- 43.Woodell-May JE, Sommerfeld SD. Role of inflammation and the immune system in the progression of osteoarthritis. J Orthop Res. 2020;38:253–257. doi: 10.1002/jor.24457. [DOI] [PubMed] [Google Scholar]
- 44.Xu M, Wang L, Wu S, Dong Y, Chen X, Wang S, et al. Review on experimental study and clinical application of low-intensity pulsed ultrasound in inflammation. Quant Imaging Med Surg. 2021;11:443–462. doi: 10.21037/qims-20-680. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.