Abstract
Berberine (BBR), an isoquinoline alkaloid natural product, is isolated primarily from Coptis chinensis and other Berberis plants. BBR possesses various bioactivities, including antioxidant, anti-inflammation, anticancer, immune-regulation, and antimicrobial activities. Growing scientific evidence underscores BBR’s substantial neuroprotective potential, prompting increased interest and scrutiny. In this comprehensive review, we elucidate the neuroprotective attributes of BBR, delineate the underlying molecular mechanisms, and assess its clinical safety and efficacy. The multifaceted molecular mechanisms responsible for BBR’s neuroprotection encompass the attenuation of oxidative stress, mitigation of inflammatory responses, inhibition of apoptotic pathways, facilitation of autophagic processes, and modulation of CYP450 enzyme activities, neurotransmitter levels, and gut microbiota composition. Furthermore, BBR engages numerous signaling pathways, including the PI3K/Akt, NF-κB, AMPK, CREB, Nrf2, and MAPK pathways, to confer its neuroprotective effects. This comprehensive review aims to provide a substantial knowledge base, stimulate broader scientific discourse, and facilitate advancements in the application of BBR for neuroprotection.
Keywords: berberine, neuroprotective effects, molecular mechanisms, clinical applications
1. Introduction
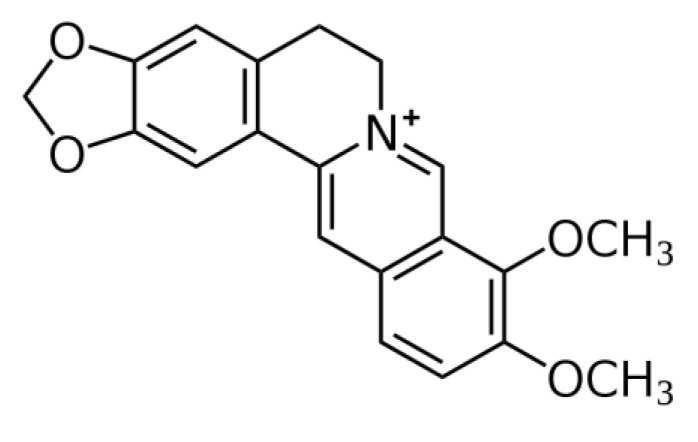
Berberine (BBR), a quaternary isoquinoline alkaloid compound, has been identified in several plants, including Ranunculus canadensis, Berberis vulgaris, Berberis aquifolium, and Coptis chinensis [1,2,3]. BBR is a yellow acicular crystal with the molecular formula of C20H18NO4, and its chemical structure is shown in Figure 1. Its current clinical use is often in the form of hydrochloride (i.e., BBR hydrochloride) or sulfate (i.e., BBR sulfate). It is well known that BBR has a series of pharmacological activities, including antimicrobial, antioxidant, anti-inflammatory, anti-heart failure, anti-arrhythmia, lowering cholesterol, anticancer, and immunomodulatory activities [4,5,6,7,8,9,10]. In China, the China Food and Drug Administration has approved BBR for treating bacterial-induced intestinal diarrhea in both humans and livestock. Despite historically limited bioavailability, ongoing preclinical trials are driving exploration into new potential applications [11,12].
Figure 1.
Chemical structure of BBR.
Recent research has revealed BBR’s ability to cross the blood–brain barrier, yielding positive impacts on brain functions [13,14,15,16,17,18,19,20]. Numerous in vitro and animal studies have showcased BBR’s capacity to offer neuroprotection against drug- and toxin-induced neurotoxicity, ischemia–reperfusion injury, and chronic neurodegenerative conditions such as Alzheimer’s, Parkinson’s, and Huntington’s diseases [13,14,15,16,17,18,19,20]. The intricate molecular mechanisms underlying BBR’s robust neuroprotective effects encompass various biological functions, including antioxidative, anti-inflammatory, and antiapoptotic actions [21]. For this review, the most important articles published from 1 January 2000 to 1 August 2023 relating to the protective effects of BBR on neurological diseases were selected from the Scopus, PubMed, and Web of Science databases. The keyword combinations used for the literature search include: ‘berberine and neuroprotection’, ‘berberine and neuroprotection’, ‘berberine and neuroprotective effect’, or ‘berberine and neurodegenerative diseases’. In addition, some important studies involving the side effects and toxic doses of BBR were also selected. In this present review, we aimed to summarize the neuroprotective effects, molecular mechanisms, side effects and toxic dosages, and clinical applications of BBR while also addressing existing challenges associated with its use as a neuroprotective agent.
2. An Overview of the Neuroprotective Effects of BBR
BBR possesses a remarkable capacity to cross the blood–brain barrier (BBB) and confer potent neuroprotection against a spectrum of neurodegenerative conditions, including Alzheimer’s disease, Parkinson’s disease, cerebral ischemia, mental depression, schizophrenia, and anxiety [5,8,19,22,23,24]. It has been reported that oral administration of BBR could effectively treat neurotoxic injury caused by drugs (e.g., doxorubicin, and 6-hydroxydopamine [6-OHDA]), environmental toxins (e.g., chlorpyrifos, mercury, aluminum, cadmium, and fluoride), aging (e.g., amyloid β-induced aging), ischemia–reperfusion, or stroke (i.e., middle cerebral artery occlusion-induced stroke) both in vivo and in vitro [15,25,26,27,28,29].
Furthermore, advancements in nanotechnology and nose-to-brain drug delivery (NBDD) techniques have enhanced BBR’s ability to penetrate the blood–brain barrier and achieve targeted brain delivery [30,31,32,33]. For instance, BBR-loaded nanostructured lipid carriers coated with chitosan (BBR-CTS-NLCs) have demonstrated improved brain targeting and enhanced therapeutic efficacy in the treatment of central nervous system diseases, such as Alzheimer’s disease, through nasal administration [34].
The molecular mechanisms of BBR’s neuroprotection are multifaceted and context-dependent. Over the past three decades, researchers have demonstrated that the neuroprotective mechanisms of BBR involve the inhibition of oxidative stress, mitochondrial dysfunction, inflammatory response, programmatic cell death (e.g., ferroptosis, necroptosis, and apoptosis), and the activation of autophagy [8,15,25,26,27,28,29,35,36]. Several signaling pathways, including phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt), mitogen-activated protein kinases (MAPKs), AMP-activated protein kinase (AMPK), hypoxia-inducible factor-1 (HIF-1), autophagy, nuclear factor kappa B (NF-κB), peroxisome proliferator-activated receptors (PPARs), cyclic AMP response element (CRE)-binding protein (CREB), p53, nuclear factor-E2-related factor 2 (Nrf2), and mitochondrial apoptotic pathways, were also involved [8,15,25,26,27,28,29,35,36]. Recent investigations have even indicated that oral BBR supplementation can elevate brain dopamine (DA) levels, ameliorating Parkinson’s disease by modulating gut microbiota, adding another layer of complexity to its mechanisms of action [37]. The detailed mechanistic aspects will be expounded upon in subsequent sections.
3. Molecular Mechanisms of BBR’s Neuroprotection
3.1. Inhibition of Oxidative Stress, Mitochondrial Dysfunction, and Apoptosis
Growing evidence underscores the pivotal role of oxidative stress in the initiation and progression of neurodegenerative disorders [38,39,40,41,42,43,44,45,46,47,48,49]. Excessive production of reactive oxygen species (ROS) and reactive nitrogen species (RNS) under oxidative stress conditions can induce lipid peroxidation, protein oxidation, protein nitration, and glycol oxidation, culminating in membrane damage, cytoskeletal abnormalities, and DNA damage within neural tissues [38,39,40,41,42,43,44,45,46,47,48,49]. Consequently, antioxidant supplementation has emerged as an effective therapeutic strategy for neurodegenerative diseases [50,51,52,53].
Numerous studies have shed light on the potent antioxidant properties of BBR, making it a promising option for treating neurodegenerative diseases [6,8,14,54,55,56,57,58,59,60,61,62,63,64,65,66,67]. In vitro investigations have demonstrated its ability to scavenge peroxynitrite (ONOO−), nitric oxide (NO), hydroxyl radical (OH•), superoxide anion (O2•−), and sodium nitroprusside, cisplatin, and Fe2+-induced lipid peroxidation (LPO) [68]. Using in vitro 2, 2-diphenyl-1-picrylhydrazyl radical (DPPH) and 2, 2-azinobis (3-ethylbenzothiazoline-6-sulfonate) radical tests showed that the half-maximal inhibitory concentrations (IC50s) of BBR are both about 0.3 mg/mL [69]. In addition, it was also found that the IC50s of BBR for NO radical scavenging, Fe2+ chelation, and OH• radical scavenging are 0.17 mg/mL, 0.12 mg/mL, and 0.11 mg/mL, respectively [69]. Notably, ONOO− is a potent oxidative and nitrating reagent capable of damaging various intracellular macromolecules, including proteins, lipids, and DNA. BBR effectively reduces superoxide levels in macrophages mediated by NADPH oxidase, restoring cellular redox balance by selectively inhibiting gp91phox expression and enhancing antioxidant enzyme activities, such as superoxide dismutase (SOD) and catalase (CAT), thereby mitigating oxidative-stress-induced cytotoxicity and brain injury [55,70]. Animal studies have corroborated these findings, showing that BBR supplementation significantly reduces malondialdehyde (MDA) levels, enhances SOD and CAT activities, and inhibits caspase activities in the hippocampus tissues of rats, thereby attenuating neurodegeneration induced by two-vessel occlusion [61]. Additionally, BBR has been shown to prevent mitochondrial ROS generation by targeting the N-methyl-D-aspartate-receptor (NMDA)R1/NADPH oxidase 3 (NOX3) pathway, protecting spiral ganglion cells from cytomegalovirus-induced apoptosis [71].
Mitochondria, being both the main targets and producers of ROS, play a central role in oxidative-stress-related neurodegeneration [72]. Studies have demonstrated that ROS production is regulated by various antioxidant defense pathways, including the PI3K/Akt pathway, Nrf2 pathway, and PPAR pathway [24,73,74]. Nrf2, a key regulator of antioxidant defense, transcriptionally controls the expression of numerous protective genes in response to oxidative stress [75]. Although direct interactions between BBR and Nrf2 require further investigation, studies have shown that BBR supplementation activates Nrf2 and its downstream target, heme oxygenase-1 (HO-1), exerting neuroprotective effects [75]. Many studies have reported that the Nrf2–antioxidant response element axis is a critical target against oxidative stress in neurodegenerative diseases [76]. Albeit it still lacks the direct evidence of BBR interaction with Nrf2, some studies have illustrated that BBR supplementation could activate the expression of Nrf2 and its downstream gene HO-1, exhibiting its neuroprotective effects [24]. Activation of the Akt signaling pathway by BBR inhibits ROS production, protecting against oxidative stress induced by various agents [77,78]. Consistently, it has been reported that BBR supplementation could alleviate rotenone-induced ROS production in human neuroblastoma cells (i.e., SH-SY5Y cells) via the inhibition of mitochondrial dysfunction through activating the PI3K/Akt signaling pathway [66]. Moreover, BBR has been reported to alleviate rotenone-induced ROS production via the activation of Nrf2 and HO-1 expression, safeguarding against high-glucose-induced cell apoptosis [79]. Hsu et al. demonstrated that BBR activates Nrf2 nuclear translocation and protects NSC34 motor neuron-like cells from oxidative damage through the activation of PI3K/Akt-dependent cytoprotective pathways [80]. Recent findings highlight BBR as a potent ligand of PPARδ, promoting Nrf2 and NQO1 expression and consequently mitigating oxidative stress and brain injury in a mouse model of middle cerebral artery occlusion (MCAO) [74]. In short, the antioxidant defense function of BBR against neurotoxic effects caused by oxidative stress mainly depends on its free radical scavenging ability and the activation of endogenous antioxidant signaling pathways, such as the Nrf2, PI3K/Akt, and PPARδ pathways.
Mitochondrial dysfunction, stemming from excessive reactive oxygen species (ROS) production, triggers a cascade of events, including ATP depletion, the opening of mitochondrial permeability transition pores, caspase activation, and cellular apoptosis [81]. The opening of mitochondrial permeability transition pores is regulated by various proteins and signaling networks, including mitochondrial membrane potential, mitochondrial Ca2+ signals, and members of the Bcl-2 family (e.g., the antiapoptotic B-cell lymphoma-extra-large [Bcl-XL] and Bcl-2; the proapoptotic members of the family: Bcl-2-associated X protein [BAX], Bcl-2 antagonist/killer 1 [BAK1], and Bcl-2-associated agonist of cell death [BAD]) [82]. BBR supplementation has been shown to upregulate mitochondrial membrane potential and ATP levels, protecting against amyloid-β-induced mitochondrial dysfunction and cell apoptosis in primary cultured hippocampal neurons [83]. BBR inhibits the release of proapoptotic factors like cytochrome c and apoptosis-inducing factors (AIFs) in response to oxygen–glucose deprivation (OGD), safeguarding against ischemic brain injury [84]. In a rat model, Singh et al. found that oral BBR supplementation at the doses of 10 or 20 mg/kg/day for 19 days could significantly improve the mitochondrial complex (I, II, and IV) activities and inhibit the activation of caspase-3 in brain tissues, followed by the amelioration of cerebral ischemia-induced brain injury [85]. The neuroprotective effect of BBR against ischemia-induced brain injury involves the activation of the Akt/GSK3β/ERK1/2 signal pathway and the inhiation of the JNK/caspase-3 pathway [18]. In addition, oral BBR supplementation could upregulate the expression of Bcl-2 protein and downregulate the expression of Bax protein in the brain tissue, inhibiting cell apoptosis and ameliorating doxorubicin-induced cognitive impairment in rats [26]. Zhang et al. found that BBR pretreatment attenuated hypoxia condition-induced neuronal cell death or brain injury through the downregulation of HIF-1α protein, inhibiting caspase-9 and caspase-3 activations and decreasing the Bcl-2/Bax ratio in PC12 cells in a rat model [86].
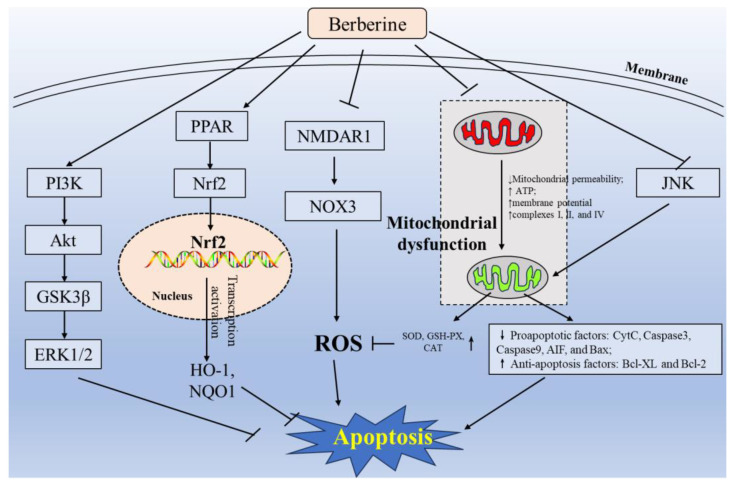
Taken together, as shown in Figure 2, BBR exerts its neuroprotective effects by inhibiting ROS production, mitochondrial apoptotic pathways, and neuronal cell apoptosis through the activation of various signaling pathways, including PI3K/Akt, PPAR/Nrf2, NMDAR1/NOX3, JNK, and the regulation of mitochondrial function.
Figure 2.
A proposed mechanism of neuroprotection by BBR against oxidative stress, mitochondrial dysfunction, and apoptosis. ↑ indicates the upregulation by BBR; ↓ indicates the downregulation by BBR. PI3K, phosphoinositide 3-kinase; Akt, protein kinase B; GSK3β, glycogen synthase kinase 3β; ERK1/2, extracellular regulated protein kinases 1/2; PPAR, peroxisome proliferators-activated receptor; Nrf2, nuclear factor-E2-related factor 2; HO-1, heme oxygenase-1; NQO1, NAD (P)H quinone oxidoreductase 1; NMDAR1, N-methyl-d-aspartate receptor 1; NOX3, NADPH oxidase 3; ROS, reactive oxygen species; JNK, c-Jun N-terminal kinase; ATP, adenosine triphosphate; SOD, superoxide dismutase; GSH-PX, glutathione peroxidase; CAT, catalase; CytC, cytochrome c; AIF, apoptosis-inducing factor; Bax, Bcl-2-associated X protein; Bcl2, B-cell lymphoma-2; Bcl-XL, B-cell lymphoma-extra-large.
3.2. Blockade of Inflammatory Response and Necroptosis
Previous studies have convincingly established that mitigating neuroinflammation could mitigate neuronal loss and decrease the morbidity associated with neurodegenerative disorders [35,87,88,89]. Neuroinflammation, characterized by the chronic activation of microglia and astrocytes, can result from various factors such as traumatic brain injury, microbial infection, drugs, neurotoxins, or toxic metabolites [35,87,88,89]. In the context of neurodegenerative diseases, the activation of microglia and astrocytes often leads to the secretion of multiple proinflammatory mediators and neurotoxic cytokines. This, in turn, fuels a detrimental cycle of neuronal damage and neuroinflammation, ultimately driving the chronic progression of neurodegenerative conditions [35,87,88,89].
Berberine (BBR) stands out as a promising agent for combating neuroinflammation, effectively regulating the inflammatory response triggered by infections, toxins, aging, or ischemia–reperfusion through multiple signaling pathways [23,90,91,92,93]. In an in vitro model simulating the pathology of Alzheimer’s disease, BBR alleviates neuroinflammatory response by reducing the production of proinflammatory cytokines in microglia [94]. Furthermore, BBR partially ameliorates cognitive dysfunction induced by lipopolysaccharide (LPS) by attenuating neuroinflammation [95]. Jia et al. found that BBR supplementation could inhibit Aβ-induced microglia inflammation by suppressing the activation of the NF-κB and MAPK signaling pathways [96]. Zhang et al. found that BBR could reduce ischemic brain injury and neuroinflammatory response via increasing the activation of the Akt/glycogen synthase kinase (GSK) signaling pathway and inhibiting the NF-κB pathway [97]. BBR supplementation could reduce TLR4/MyD88/NF-κB signaling transduction and attenuate neuronal death induced by microglial-conditioned media [92]. BBR effectively inhibits the inflammatory activation of rat brain microglia by inactivating the NF-κB/iNOS/NO pathway or activating the AMPK pathway [90,98,99]. Additionally, several studies have reported that BBR directly upregulates the expression of anti-inflammatory factors (e.g., interleukin (IL)-4 [IL-4] and IL-10) while inhibiting the expression of proinflammatory factors (e.g., COX-2, prostaglandin E2 [PGE2]), IL-1β, IL-6, and tumor necrosis factor-α (TNF-α)), displaying therapeutic effects against neuroinflammatory diseases in both in vitro and in vivo settings [99,100,101,102].
Necroptosis, a regulated form of cell death distinct from apoptosis, is known to induce marked inflammatory responses and adaptive immunity effects in the body [103]. Previous studies have indicated that BBR may act as a necroptosis inducer in cancer cells, showcasing potential anti-tumor effects [104,105,106]. BBR usually plays a neuroprotective role in the treatments for neurological diseases and neuronal cells inhibiting necroptosis [107]. Ou et al. demonstrated that BBR treatment could significantly inhibit the expression of receptor-interacting protein 1 (RIP1) and RIP3, two key regulators of necroptosis, attenuating the neuroinflammatory response and cognitive impairment in the hippocampus tissues in rats exposed to excessive L-arginine [107].
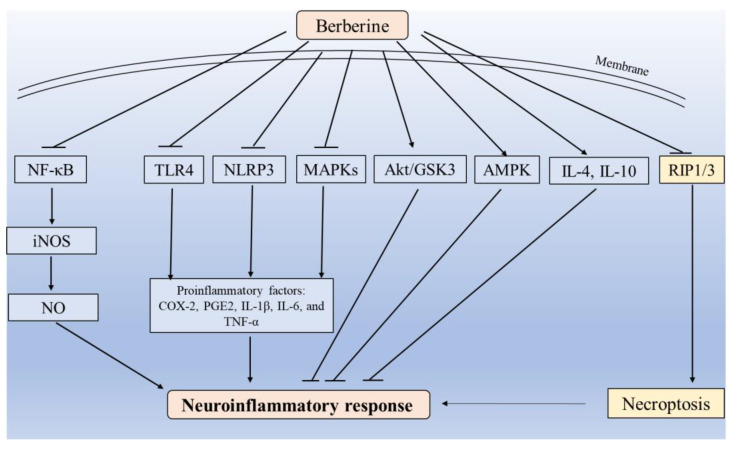
In summary, the inhibitory effects of BBR on inflammatory response and necroptosis on nerve cells or tissues partly contributed to its neuroprotective properties. These effects are mediated through the inhibition of the MAPKs, AMPK, NF-κB, TLR4, and NLRP3 pathways, as well as direct modulation of inflammatory factor secretion, as illustrated in Figure 3.
Figure 3.
Proposed regulated mechanisms for BBR’s neuroprotection against neuroinflammatory response in nerve cells. NF-κB, nuclear factor kappa B; iNOS, inducible nitric oxide synthase; NO, nitric oxide; TLR4, toll-like receptor 4; NLRP3, NOD-like receptor protein 3; MAPK, mitogen-activated protein kinase; Akt, alpha serine/threonine-protein kinase; GSK3, glycogen synthase kinase 3; AMPK, AMP-activated protein kinase; IL-4, interleukin 4; IL-10, interleukin 10; RIP1/3, receptor-interacting protein 1/3; COX-2, cyclooxygenase 2; PGE2, prostaglandin E2; IL-1β, interleukin 1β; IL-6, interleukin 6; TNF-α, tumor necrosis factor-α.
3.3. Induction of Autophagy
Berberine (BBR) exhibits neuroprotective qualities by inducing autophagy and facilitating the clearance of toxic aggregate proteins [108,109,110]. BBR has been found to trigger autophagy in various cell types, including macrophages, lymphoblastic leukemia cells, retinal cells, and neuronal cells, as well as in various tissues such as the liver, lung, kidney, stomach, breast, and myocardium [111,112,113,114,115,116,117,118,119,120,121,122,123,124,125,126,127].
One of BBR’s mechanisms of neuroprotection involves promoting autophagy to eliminate misfolded proteins. Several studies have also found that BBR supplementation could effectively improve the motor dysfunction of mice with Huntingtin’s diseases via promoting the degradation of mutant Huntingtin protein through enhancing cell autophagy [9,109]. This process ultimately alleviates motor dysfunction and extends the survival period in a mouse model of Huntington’s disease [109]. Similarly, in an APP/tau/PS1 triple-transgenic mouse model of Alzheimer’s disease, BBR treatment could significantly improve the cognitive impairment of mice by promoting autophagy while inhibiting the production of β-amyloid (Aβ) through suppressing β-site APP cleavage enzyme 1 (BACE1) expression [110]. Moreover, in another study, data showed that BBR reduced the production of Aβ and the expression of the BACE1 protein by activating AMPK in nerve cells [128]. Interestingly, a great many studies have indicated that autophagy is AMPK-dependent or triggered via the AMPK/mTOR pathway [4,129]. Consequently, BBR may activate autophagy by the AMPK signal pathway to clear wrong proteins that cause neurological diseases.
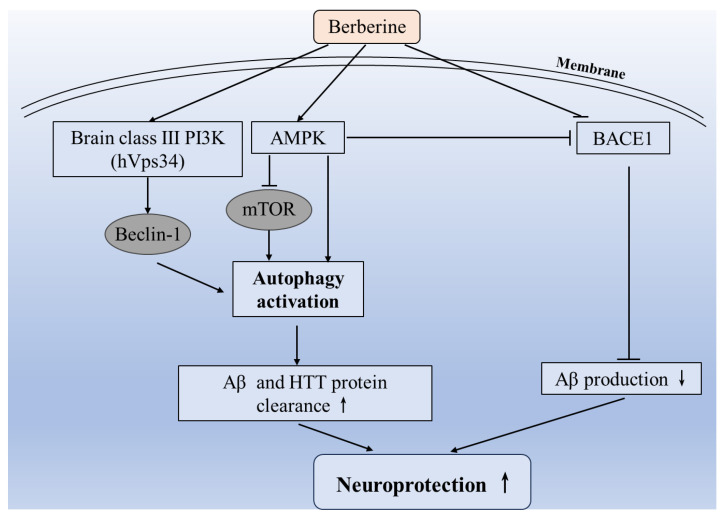
Overall, a summary conferring the molecular mechanisms of BBR’s neuroprotection via inducing autophagy is shown in Figure 4.
Figure 4.
Proposed regulated mechanisms for BBR’s neuroprotection via the activation of autophagy. ↑ indicates the upregulation by BBR; ↓ indicates the downregulation by BBR. PI3K, phosphoinositide 3-kinase; hVps34, human vacuolar protein sorting 34; AMPK, AMP-activated protein kinase; mTOR, mammalian target of rapamycin; Aβ, β-amyloid; HTT, Huntington protein; BACE1, β-site APP cleavage enzyme 1.
3.4. Modulation of Neurotransmitters
Neurotransmitters are pivotal in facilitating intraneuronal communication and neurobehavioral functions [130]. BBR has demonstrated the ability to inhibit the activity of acetylcholinesterase, butyrylcholinesterase, and monoamine oxidases (MAOs), which play vital roles in regulating the levels of neurotransmitters [131,132]. For example, MAO-A and MAO-B, two isozymes of MAOs, are principally responsible for the degradation of various amine neurotransmitters, including DA, norepinephrine (NE), serotonin (5-HT), and epinephrine, all of which are crucial in the development of neurodegenerative diseases [133,134].
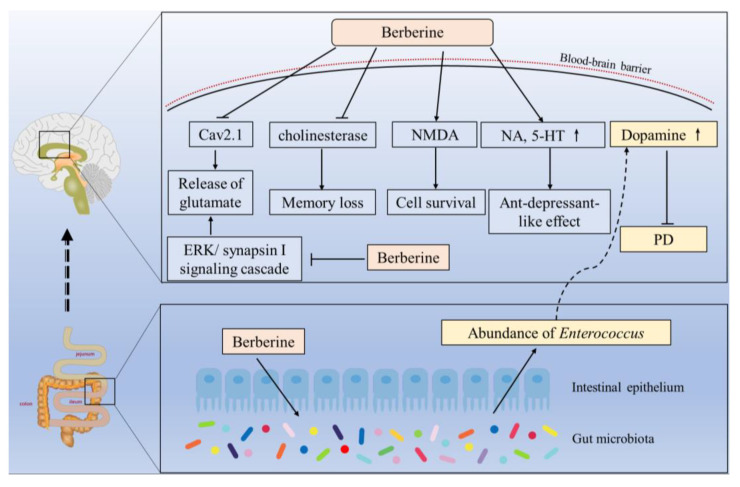
A previous study showed that oral administration of BBR at the final doses of 10 or 20 mg/kg could show a potent antidepressant-like effect in mice via upregulation of the levels of NA and 5-HT in the hippocampus and frontal cortex [131]. Additionally, BBR has been reported to significantly enhance DA levels in the brains of mice with Parkinson’s disease, attributed to increased gut Enterococcus abundance, thereby ameliorating Parkinson’s disease symptoms [37]. BBR administration has also been shown to suppress cholinesterase activity, thereby protecting the cholinergic system, and enhancing memory function in diabetic rats [67,133]. Glutamate is a primary excitatory neurotransmitter in the brains of animals and humans. N-methyl-d-aspartate (NMDA) is its receptor. Furthermore, BBR treatment has been found to protect against MK-801-induced neurodegeneration in rat brains by enhancing NMDA-mediated activity-dependent cell survival [135]. Similarly, BBR administration has been shown to significantly reduce the release of glutamate from rats’ cortical synaptosomes via inhibition of presynaptic Cav2.1 channels as well as the downregulation of the ERK/synapsin I signaling cascade [136].
Overall, a summary conferring the molecular mechanisms of BBR’s neuroprotection via regulating neurotransmitter levels in the brain is shown in Figure 5.
Figure 5.
Proposed regulated mechanisms for BBR’s neuroprotection via the regulation of neurotransmitter levels in the brain. ↑ indicates the upregulation by BBR. Solid arrows indicate the direct effects and dashed arrows indicate the indirect effects. Cav2.1, voltage-gated Ca2+ channel 2.1; ERK, extracellular regulated protein kinases; NMDA, N-methyl-d-aspartate; NA, noradrenaline; 5-HT, 5-hydroxytryptamine; PD, Parkinson’s disease.
3.5. Modulation of CYP450 Enzyme Activities
Recent studies have underscored the critical role that cytochrome P450 (CYP450) may play in the development or treatment of neurological diseases based on its effects on temperature control, maintenance of brain cholesterol homeostasis, neuropeptide release, and regulation of neurotransmitter levels [137]. It has been reported that BBR has a potent regulatory effect on CYP450 activities in liver, heart, kidney, and breast tissues [7,138,139,140,141,142,143,144,145,146,147].
Earlier investigations revealed that BBR could inhibit the activities of CYP1 enzymes, including CYP1A1, CYP1A2, and CYP1B1, with a stronger preference for CYP1B1 [148]. CYP1B1 has been reported to make an important contribution in the procession of various neurological disorders based on its regulated effects on the production of ROS and redox homeostasis [149]. It has been demonstrated that the deficiency of CYP1B1 could protect retinal astrocytes against oxidative stress and inflammation [150]. In addition, in a human clinical trial, researchers found that repeated administration of BBR (oral administration at a dose of 300 mg, three times daily, for 14 days) decreased CYP2D6, CYP2C9, and CYP3A4 activities [142,151]. Based on the inhibitory effects of BBR on the activities of the CYP3A4 enzyme, BBR significantly increases the blood concentration of cyclosporine when co-administrated [152]. In addition, p-glycoprotein (P-gp)-mediated gut efflux properties are responsible for the reduced bioavailability of BBR and verapamil, a p-gp inhibitor, and co-treatment could significantly enhance the neuroprotective effect of BBR against streptozotocin-induced cognitive dysfunction in a rat model [153].
To date, there is still limited information about the interaction between BBR and CYP450 enzymes in nerve tissue, and further investigations into the precise molecular mechanisms are warranted.
3.6. Others
Berberine (BBR) has demonstrated notable neuroprotective potential through various molecular pathways. Notably, it has been reported that BBR enhances synaptic plasticity by activating the cAMP response element-binding protein (CREB), leading to the production of brain-derived neurotrophic factor (BDNF) through the upregulation of SIRT1. This, in turn, contributes to the amelioration of cognitive impairment induced by chemotherapy drugs, such as doxorubicin [26]. In a transient middle cerebral artery occlusion rat model, BBR administration could induce gut–brain axis signal transmission and stimulate the vague nerve by inducing the production of intestinal hydrogen through the regulation of gut microbiota, finally offering neuroprotection [154]. Furthermore, BBR treatment has also been shown to reduce hippocampus neuronal damage by inhibiting the expression of the matrix metalloproteinase-9 protein and gelatinase activities and downregulating the expression of laminin and neuronal nuclei antigen (NeuN) proteins in the hippocampal CA1 and CA2 areas [155]. These functions also contribute to the therapeutic effects of BBR against multiple sclerosis disease [156]. Another study by Wu et al. highlights the neuroprotective potential of BBR by blocking neuronal ATP-sensitive K+ channels in substantia nigra pars compacta dopaminergic neurons [157]. BBR has also shown promise in enhancing nerve growth factor (NGF) expression, which promotes Nrf2- and Akt-related neurite outgrowth and differentiation, thus protecting against oxidative stress and neuroinflammatory responses [79,158,159].
4. Safety and Toxic Adverse of BBR
A series of studies has confirmed that the toxicity of BBR depends on the route and duration of administration [160,161,162,163]. Animal experiments showed that oral administration of BBR is safe and intravenous or intraperitoneal injection are toxic. For example, Kheir et al. showed that the median lethal dosages (LD50) of BBR via oral, intravenous, or intraperitoneal administration in mice are >20.8 g/kg, 9.04, and 57.6 mg/kg, respectively [160]. This difference is mainly dependent on the final concentration of BBR in the blood [160]. It also suggests that a single oral dose of BBR at 2.97 g/kg of body weight is safe according to the conversion of body surface area between humans and mice (approximately seven times between humans and mice) [164].
It has been reported that BBR exhibits potent cytotoxicity in vitro, exhibiting a potential anticancer effect [165]. BBR has been demonstrated to have potent nucleic-acid-binding activity, which is a key reason for inhibiting cell differentiation and inducing cycle arrest and DNA damage [166,167]. In addition, there are similar targets in the anticancer and neuroprotective effects of BBR, such as Akt, MAPKs, and Bcl-2 proteins, and the main difference are in the dosage [168]. Consistently, long-term administration and intraperitoneal or intravenous injection of berberine could induce multiple toxic effects, including neurotoxicity, immunotoxicity, phototoxicity, cardiotoxicity, and jaundice, and these toxic effects are in dose- and time-dependent manners [161,162,169]. This evidence also suggests that we may need safety measures with BBR, especially when administered intravenously or intraperitoneally in clinical practices. The toxic mechanisms of BBR may be related to its ability of directly interacting with DNA or inhibiting the presence of signaling pathways in cells [161,162,169,170].
In the clinic, it has been reported that oral administration of BBR at a dose of 1.5 g/day (500 mg per time for three times per day) for 13 weeks could result in potential gastrointestinal toxicity (such as diarrhea, constipation, flatulence, and abdominal complaint) in patients with type-2 diabetes, but no marked hepatotoxicity nor nephrotoxicity was detected [171].
Rad and colleagues systemically reviewed the toxic effects of BBR and Berberis vulgaris extract [161]. More information can be found in this review.
5. Clinical Trials and Therapeutic Applications
Accumulated evidence from clinical trials underscores the wide-ranging therapeutic applications of BBR. Among randomized clinical trials involving BBR and barberry (Berberis vulgaris) in the treatment of different human diseases, the effects of reducing lipids and improving insulin resistance are the most studied. Additionally, clinical investigations have delved into its potential benefits in cardiovascular, anticancer, gastrointestinal, central nervous system, and endocrine-related contexts. Importantly, oral administration of BBR has exhibited low toxicity and minimal side effects at standard doses, with occasional mild gastrointestinal reactions observed in some patients [10]. A randomized clinical trial involving 55 patients with acute ischemic stroke found that the effects of BBR combined with atorvastatin (20 mg/day) were better than that of atorvastatin alone (20 mg/day) [172]. In another clinical trial, oral administration of BBR at a dose of 1 g/day (i.e., 0.5 g per time, twice a day) for 16 weeks could effectively regulate the structure and function of the human gut microbiota, and Bifidobacterium probiotics could further enhance the hypoglycemic effect of BBR [173]. Li et al. found that oral BBR treatment could significantly reduce the serum intima–media thickness (IMT) and IL-6 levels, reducing the degree of carotid atherosclerosis to a certain extent and improving the neurological impairment and prognosis of acute cerebral ischemic stroke (AIS) patients [174]. In a recent study, oral administration of BBR at a dose of 0.5 g/day for 8 weeks significantly elevated blood DA levels in individuals with hyperlipidemia, suggesting a potential benefit in treating Parkinson’s disease [37]. While progress has been made in clinical trials exploring BBR’s potential for treating neurological diseases, further research remains imperative.
6. Conclusions and Future Directions
Currently, there are multiple studies indicating that oral BBR supplementation could provide potential neuroprotective effects against chronic neurodegenerative diseases or acute brain injury caused by ischemia–reperfusion or drugs. However, the oral bioavailability of BBR (less than 1%) is very limited [175]. It has been reported that the levels of BBR in brain tissues are lower than that in other tissues after oral administration [176]. More and more evidences suggest that gut microbiota may play an important role in various neurological diseases, and it is also the main mediator of BBR’s neuroprotective effect, but it still needs more clinical trials.
In conclusion, the published data indicate that the molecular mechanisms of BBR’s neuroprotective effects may involve multiple targets, including antioxidation, anti-inflammation, anti-apoptosis, anti-necroptosis, induction of autophagy, and modulation of CYP450 enzyme activities and gut microbiota. In addition, to overcome the poor bioavailability of BBR in clinical applications, recent advances in nanodrug delivery systems, encompassing polymeric-based, graphene-based, silver, lipid-based, dendrimer-based, magnetic mesoporous silica-based, and gold nanoparticles, have emerged as pivotal strategies. For example, nasal brain drug delivery technology, driven by nanotechnology, is gaining popularity in enhancing the efficacy of BBR in treating neurological diseases [177,178]. Future clinical studies are anticipated to elucidate the neuroprotective mechanisms of BBR further while optimizing its clinical efficacy and minimizing potential side effects. At this moment, while increasing the bioavailability of BBR, we may also need to consider its safety more. Furthermore, exploring the direct regulatory effects of BBR on CYP450 enzymes within the brain holds great clinical significance and represents an important avenue for research in uncovering the neuroprotective mechanisms of BBR.
Author Contributions
Conceptualization, C.D.; methodology, E.T.; writing—original draft preparation, E.T. and C.D.; writing—review and editing, C.D. and G.S.; supervision, C.D.; project administration, C.D.; funding acquisition, C.D. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research was supported by the National Natural Science Foundation of China (award number 32102724) and the Special Fund Management Office for Basic Research Business Expenses of China Agricultural University (2023TC028).
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Hesari A., Ghasemi F., Cicero A.F.G., Mohajeri M., Rezaei O., Hayat S.M.G., Sahebkar A. Berberine: A potential adjunct for the treatment of gastrointestinal cancers? J. Cell. Biochem. 2018;119:9655–9663. doi: 10.1002/jcb.27392. [DOI] [PubMed] [Google Scholar]
- 2.Yarla N.S., Bishayee A., Sethi G., Reddanna P., Kalle A.M., Dhananjaya B.L., Dowluru K.S., Chintala R., Duddukuri G.R. Targeting arachidonic acid pathway by natural products for cancer prevention and therapy. Semin. Cancer Biol. 2016;40–41:48–81. doi: 10.1016/j.semcancer.2016.02.001. [DOI] [PubMed] [Google Scholar]
- 3.Pirillo A., Catapano A.L. Berberine, a plant alkaloid with lipid- and glucose-lowering properties: From in vitro evidence to clinical studies. Atherosclerosis. 2015;243:449–461. doi: 10.1016/j.atherosclerosis.2015.09.032. [DOI] [PubMed] [Google Scholar]
- 4.Zhou H., Feng L., Xu F., Sun Y., Ma Y., Zhang X., Liu H., Xu G., Wu X., Shen Y., et al. Berberine inhibits palmitate-induced NLRP3 inflammasome activation by triggering autophagy in macrophages: A new mechanism linking berberine to insulin resistance improvement. Biomed. Pharmacother. 2017;89:864–874. doi: 10.1016/j.biopha.2017.03.003. [DOI] [PubMed] [Google Scholar]
- 5.Shayganfard M. Berberine: Is a Promising Agent for Mental Disorders Treatment? Curr. Mol. Pharmacol. 2022;16:307–320. doi: 10.2174/1874467215666220509213122. [DOI] [PubMed] [Google Scholar]
- 6.Li Z., Geng Y.N., Jiang J.D., Kong W.J. Antioxidant and anti-inflammatory activities of berberine in the treatment of diabetes mellitus. Evid.-Based Complement. Altern. Med. 2014;2014:289264. doi: 10.1155/2014/289264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhang P., Ma D., Wang Y., Zhang M., Qiang X., Liao M., Liu X., Wu H., Zhang Y. Berberine protects liver from ethanol-induced oxidative stress and steatosis in mice. Food Chem. Toxicol. 2014;74:225–232. doi: 10.1016/j.fct.2014.10.005. [DOI] [PubMed] [Google Scholar]
- 8.Song D., Hao J., Fan D. Biological properties and clinical applications of berberine. Front. Med. 2020;14:564–582. doi: 10.1007/s11684-019-0724-6. [DOI] [PubMed] [Google Scholar]
- 9.Cheng Z., Kang C., Che S., Su J., Sun Q., Ge T., Guo Y., Lv J., Sun Z., Yang W., et al. Berberine: A Promising Treatment for Neurodegenerative Diseases. Front. Pharmacol. 2022;13:845591. doi: 10.3389/fphar.2022.845591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Imenshahidi M., Hosseinzadeh H. Berberine and barberry (Berberis vulgaris): A clinical review. Phytother. Res. PTR. 2019;33:504–523. doi: 10.1002/ptr.6252. [DOI] [PubMed] [Google Scholar]
- 11.Habtemariam S. Berberine pharmacology and the gut microbiota: A hidden therapeutic link. Pharmacol. Res. 2020;155:104722. doi: 10.1016/j.phrs.2020.104722. [DOI] [PubMed] [Google Scholar]
- 12.Khoshandam A., Imenshahidi M., Hosseinzadeh H. Pharmacokinetic of berberine, the main constituent of Berberis vulgaris L.: A comprehensive review. Phytother. Res. PTR. 2022;36:4063–4079. doi: 10.1002/ptr.7589. [DOI] [PubMed] [Google Scholar]
- 13.Kulkarni S.K., Dhir A. Berberine: A plant alkaloid with therapeutic potential for central nervous system disorders. Phytother. Res. PTR. 2010;24:317–324. doi: 10.1002/ptr.2968. [DOI] [PubMed] [Google Scholar]
- 14.Ahmed T., Gilani A.U., Abdollahi M., Daglia M., Nabavi S.F., Nabavi S.M. Berberine and neurodegeneration: A review of literature. Pharmacol. Rep. 2015;67:970–979. doi: 10.1016/j.pharep.2015.03.002. [DOI] [PubMed] [Google Scholar]
- 15.Zhu J.R., Lu H.D., Guo C., Fang W.R., Zhao H.D., Zhou J.S., Wang F., Zhao Y.L., Li Y.M., Zhang Y.D., et al. Berberine attenuates ischemia-reperfusion injury through inhibiting HMGB1 release and NF-κB nuclear translocation. Acta Pharmacol. Sin. 2018;39:1706–1715. doi: 10.1038/s41401-018-0160-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cui H.S., Matsumoto K., Murakami Y., Hori H., Zhao Q., Obi R. Berberine exerts neuroprotective actions against in vitro ischemia-induced neuronal cell damage in organotypic hippocampal slice cultures: Involvement of B-cell lymphoma 2 phosphorylation suppression. Biol. Pharm. Bull. 2009;32:79–85. doi: 10.1248/bpb.32.79. [DOI] [PubMed] [Google Scholar]
- 17.Zhu F., Qian C. Berberine chloride can ameliorate the spatial memory impairment and increase the expression of interleukin-1beta and inducible nitric oxide synthase in the rat model of Alzheimer’s disease. BMC Neurosci. 2006;7:78. doi: 10.1186/1471-2202-7-78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Simões Pires E.N., Frozza R.L., Hoppe J.B., Menezes Bde M., Salbego C.G. Berberine was neuroprotective against an in vitro model of brain ischemia: Survival and apoptosis pathways involved. Brain Res. 2014;1557:26–33. doi: 10.1016/j.brainres.2014.02.021. [DOI] [PubMed] [Google Scholar]
- 19.Wang L., Sheng W., Tan Z., Ren Q., Wang R., Stoika R., Liu X., Liu K., Shang X., Jin M. Treatment of Parkinson’s disease in Zebrafish model with a berberine derivative capable of crossing blood brain barrier, targeting mitochondria, and convenient for bioimaging experiments. Comp. Biochem. Physiol. Toxicol. Pharmacol. CBP. 2021;249:109151. doi: 10.1016/j.cbpc.2021.109151. [DOI] [PubMed] [Google Scholar]
- 20.Sadeghnia H.R., Kolangikhah M., Asadpour E., Forouzanfar F., Hosseinzadeh H. Berberine protects against glutamate-induced oxidative stress and apoptosis in PC12 and N2a cells. Iran. J. Basic Med. Sci. 2017;20:594–603. doi: 10.22038/ijbms.2017.8847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lin X., Zhang N. Berberine: Pathways to protect neurons. Phytother. Res. PTR. 2018;32:1501–1510. doi: 10.1002/ptr.6107. [DOI] [PubMed] [Google Scholar]
- 22.Wang L., Zhou B.Q., Li Y.H., Jiang Q.Q., Cong W.H., Chen K.J., Wen X.M., Wu Z.Z. Lactoferrin modification of berberine nanoliposomes enhances the neuroprotective effects in a mouse model of Alzheimer’s disease. Neural Regen. Res. 2023;18:226–232. doi: 10.4103/1673-5374.344841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wu X., Liu X., Yang L., Wang Y. Berberine Protects against Neurological Impairments and Blood-Brain Barrier Injury in Mouse Model of Intracerebral Hemorrhage. Neuroimmunomodulation. 2021;29:317–326. doi: 10.1159/000520747. [DOI] [PubMed] [Google Scholar]
- 24.Zhang C., Li C., Chen S., Li Z., Jia X., Wang K., Bao J., Liang Y., Wang X., Chen M., et al. Berberine protects against 6-OHDA-induced neurotoxicity in PC12 cells and zebrafish through hormetic mechanisms involving PI3K/AKT/Bcl-2 and Nrf2/HO-1 pathways. Redox Biol. 2017;11:1–11. doi: 10.1016/j.redox.2016.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jiang W., Li S., Li X. Therapeutic potential of berberine against neurodegenerative diseases. Sci. China Life Sci. 2015;58:564–569. doi: 10.1007/s11427-015-4829-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shaker F.H., El-Derany M.O., Wahdan S.A., El-Demerdash E., El-Mesallamy H.O. Berberine ameliorates doxorubicin-induced cognitive impairment (chemobrain) in rats. Life Sci. 2021;269:119078. doi: 10.1016/j.lfs.2021.119078. [DOI] [PubMed] [Google Scholar]
- 27.Ye M., Fu S., Pi R., He F. Neuropharmacological and pharmacokinetic properties of berberine: A review of recent research. J. Pharm. Pharmacol. 2009;61:831–837. doi: 10.1211/jpp.61.07.0001. [DOI] [PubMed] [Google Scholar]
- 28.Abdel Moneim A.E. The neuroprotective effect of berberine in mercury-induced neurotoxicity in rats. Metab. Brain Dis. 2015;30:935–942. doi: 10.1007/s11011-015-9652-6. [DOI] [PubMed] [Google Scholar]
- 29.Hussien H.M., Abd-Elmegied A., Ghareeb D.A., Hafez H.S., Ahmed H.E.A., El-Moneam N.A. Neuroprotective effect of berberine against environmental heavy metals-induced neurotoxicity and Alzheimer’s-like disease in rats. Food Chem. Toxicol. 2018;111:432–444. doi: 10.1016/j.fct.2017.11.025. [DOI] [PubMed] [Google Scholar]
- 30.Saleh S.R., Abady M.M., Nofal M., Yassa N.W., Abdel-Latif M.S., Nounou M.I., Ghareeb D.A., Abdel-Monaem N. Berberine Nanoencapsulation Attenuates Hallmarks of Scoplomine Induced Alzheimer’s-like Disease in Rats. Curr. Rev. Clin. Exp. Pharmacol. 2021;16:139–154. doi: 10.2174/27724336MTA31NzQh0. [DOI] [PubMed] [Google Scholar]
- 31.Raju M., Kunde S.S., Auti S.T., Kulkarni Y.A., Wairkar S. Berberine loaded nanostructured lipid carrier for Alzheimer’s disease: Design, statistical optimization and enhanced in vivo performance. Life Sci. 2021;285:119990. doi: 10.1016/j.lfs.2021.119990. [DOI] [PubMed] [Google Scholar]
- 32.Long Y., Yang Q., Xiang Y., Zhang Y., Wan J., Liu S., Li N., Peng W. Nose to brain drug delivery—A promising strategy for active components from herbal medicine for treating cerebral ischemia reperfusion. Pharmacol. Res. 2020;159:104795. doi: 10.1016/j.phrs.2020.104795. [DOI] [PubMed] [Google Scholar]
- 33.Tavakkoli A., Iranshahi M., Hasheminezhad S.H., Hayes A.W., Karimi G. The neuroprotective activities of natural products through the Nrf2 upregulation. Phytother. Res. PTR. 2019;33:2256–2273. doi: 10.1002/ptr.6427. [DOI] [PubMed] [Google Scholar]
- 34.Abo El-Enin H.A., Elkomy M.H., Naguib I.A., Ahmed M.F., Alsaidan O.A., Alsalahat I., Ghoneim M.M., Eid H.M. Lipid Nanocarriers Overlaid with Chitosan for Brain Delivery of Berberine via the Nasal Route. Pharmaceuticals. 2022;15:281. doi: 10.3390/ph15030281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fan D., Liu L., Wu Z., Cao M. Combating Neurodegenerative Diseases with the Plant Alkaloid Berberine: Molecular Mechanisms and Therapeutic Potential. Curr. Neuropharmacol. 2019;17:563–579. doi: 10.2174/1570159X16666180419141613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shi X., Zhang T., Lou H., Song H., Li C., Fan P. Anticancer Effects of Honokiol via Mitochondrial Dysfunction Are Strongly Enhanced by the Mitochondria-Targeting Carrier Berberine. J. Med. Chem. 2020;63:11786–11800. doi: 10.1021/acs.jmedchem.0c00881. [DOI] [PubMed] [Google Scholar]
- 37.Wang Y., Tong Q., Ma S.R., Zhao Z.X., Pan L.B., Cong L., Han P., Peng R., Yu H., Lin Y., et al. Oral berberine improves brain dopa/dopamine levels to ameliorate Parkinson’s disease by regulating gut microbiota. Signal Transduct. Target. Ther. 2021;6:77. doi: 10.1038/s41392-020-00456-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Teleanu D.M., Niculescu A.G., Lungu I.I., Radu C.I., Vladâcenco O., Roza E., Costăchescu B., Grumezescu A.M., Teleanu R.I. An Overview of Oxidative Stress, Neuroinflammation, and Neurodegenerative Diseases. Int. J. Mol. Sci. 2022;23:5938. doi: 10.3390/ijms23115938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Vejux A. Cell Death, Inflammation and Oxidative Stress in Neurodegenerative Diseases: Mechanisms and Cytoprotective Molecules. Int. J. Mol. Sci. 2021;22:13657. doi: 10.3390/ijms222413657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Saleem U., Sabir S., Niazi S.G., Naeem M., Ahmad B. Role of Oxidative Stress and Antioxidant Defense Biomarkers in Neurodegenerative Diseases. Crit. Rev. Eukaryot. Gene Expr. 2020;30:311–322. doi: 10.1615/CritRevEukaryotGeneExpr.2020029202. [DOI] [PubMed] [Google Scholar]
- 41.Singh A., Kukreti R., Saso L., Kukreti S. Oxidative Stress: A Key Modulator in Neurodegenerative Diseases. Molecules. 2019;24:1583. doi: 10.3390/molecules24081583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Islam M.T. Oxidative stress and mitochondrial dysfunction-linked neurodegenerative disorders. Neurol. Res. 2017;39:73–82. doi: 10.1080/01616412.2016.1251711. [DOI] [PubMed] [Google Scholar]
- 43.Niedzielska E., Smaga I., Gawlik M., Moniczewski A., Stankowicz P., Pera J., Filip M. Oxidative Stress in Neurodegenerative Diseases. Mol. Neurobiol. 2016;53:4094–4125. doi: 10.1007/s12035-015-9337-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kim G.H., Kim J.E., Rhie S.J., Yoon S. The Role of Oxidative Stress in Neurodegenerative Diseases. Exp. Neurobiol. 2015;24:325–340. doi: 10.5607/en.2015.24.4.325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Thanan R., Oikawa S., Hiraku Y., Ohnishi S., Ma N., Pinlaor S., Yongvanit P., Kawanishi S., Murata M. Oxidative stress and its significant roles in neurodegenerative diseases and cancer. Int. J. Mol. Sci. 2014;16:193–217.:193. doi: 10.3390/ijms16010193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Li J., Ao W., Li W., Jiang Z.-G., Ghanbari H.A. Oxidative stress and neurodegenerative disorders. Int. J. Mol. Sci. 2013;14:24438–24475.:4438. doi: 10.3390/ijms141224438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chen X., Guo C., Kong J. Oxidative stress in neurodegenerative diseases. Neural Regen. Res. 2012;7:376–385. doi: 10.3969/j.issn.1673-5374.2012.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Uttara B., Singh A.V., Zamboni P., Mahajan R.T. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Curr. Neuropharmacol. 2009;7:65–74. doi: 10.2174/157015909787602823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lin M.T., Beal M.F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature. 2006;443:787–795. doi: 10.1038/nature05292. [DOI] [PubMed] [Google Scholar]
- 50.Fernandes C., Oliveira C., Benfeito S., Soares P., Garrido J., Borges F. Nanotechnology and antioxidant therapy: An emerging approach for neurodegenerative diseases. Curr. Med. Chem. 2014;21:4311–4327. doi: 10.2174/0929867321666140915141836. [DOI] [PubMed] [Google Scholar]
- 51.Danta C.C., Piplani P. The discovery and development of new potential antioxidant agents for the treatment of neurodegenerative diseases. Expert Opin. Drug Discov. 2014;9:1205–1222. doi: 10.1517/17460441.2014.942218. [DOI] [PubMed] [Google Scholar]
- 52.Guerra-Araiza C., Álvarez-Mejía A.L., Sánchez-Torres S., Farfan-García E., Mondragón-Lozano R., Pinto-Almazán R., Salgado-Ceballos H. Effect of natural exogenous antioxidants on aging and on neurodegenerative diseases. Free Radic. Res. 2013;47:451–462. doi: 10.3109/10715762.2013.795649. [DOI] [PubMed] [Google Scholar]
- 53.Tardiolo G., Bramanti P., Mazzon E. Overview on the Effects of N-Acetylcysteine in Neurodegenerative Diseases. Molecules. 2018;23:3305. doi: 10.3390/molecules23123305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhou J.Y., Zhou S.W. Protective effect of berberine on antioxidant enzymes and positive transcription elongation factor b expression in diabetic rat liver. Fitoterapia. 2011;82:184–189. doi: 10.1016/j.fitote.2010.08.019. [DOI] [PubMed] [Google Scholar]
- 55.Zhang Y., Wang L., Li G., Gao J. Berberine-Albumin Nanoparticles: Preparation, Thermodynamic Study and Evaluation Their Protective Effects Against Oxidative Stress in Primary Neuronal Cells as a Model of Alzheimer’s Disease. J. Biomed. Nanotechnol. 2021;17:1088–1097. doi: 10.1166/jbn.2021.2995. [DOI] [PubMed] [Google Scholar]
- 56.Yaw L.S., Lindsei S., Karmin O. Redox regulation in health and disease—Therapeutic potential of berberine. Food Res. Int. 2011;44:2409–2417. doi: 10.1016/j.foodres.2010.12.038. [DOI] [Google Scholar]
- 57.Thirupurasundari C.J., Padmini R., Devaraj S.N. Effect of berberine on the antioxidant status, ultrastructural modifications and protein bound carbohydrates in azoxymethane-induced colon cancer in rats. Chem. Biol. Interact. 2009;177:190–195. doi: 10.1016/j.cbi.2008.09.027. [DOI] [PubMed] [Google Scholar]
- 58.Tan Y., Tang Q., Hu B.R., Xiang J.Z. Antioxidant properties of berberine on cultured rabbit corpus cavernosum smooth muscle cells injured by hydrogen peroxide. Acta Pharmacol. Sin. 2007;28:1914–1918. doi: 10.1111/j.1745-7254.2007.00705.x. [DOI] [PubMed] [Google Scholar]
- 59.Singh A.K., Singh S.K., Nandi M.K., Mishra G., Maurya A., Rai A., Rai G.K., Awasthi R., Sharma B., Kulkarni G.T. Berberine: A Plant-derived Alkaloid with Therapeutic Potential to Combat Alzheimer’s disease. Cent. Nerv. Syst. Agents Med. Chem. 2019;19:154–170. doi: 10.2174/1871524919666190820160053. [DOI] [PubMed] [Google Scholar]
- 60.Shirwaikar A., Shirwaikar A., Rajendran K., Punitha I.S. In vitro antioxidant studies on the benzyl tetra isoquinoline alkaloid berberine. Biol. Pharm. Bull. 2006;29:1906–1910. doi: 10.1248/bpb.29.1906. [DOI] [PubMed] [Google Scholar]
- 61.Pirmoradi Z., Yadegari M., Moradi A., Khojasteh F., Zare Mehrjerdi F. Effect of berberine chloride on caspase-3 dependent apoptosis and antioxidant capacity in the hippocampus of the chronic cerebral hypoperfusion rat model. Iran. J. Basic Med. Sci. 2019;22:154–159. doi: 10.22038/ijbms.2018.31225.7534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Liu W.H., Hei Z.Q., Nie H., Tang F.T., Huang H.Q., Li X.J., Deng Y.H., Chen S.R., Guo F.F., Huang W.G., et al. Berberine ameliorates renal injury in streptozotocin-induced diabetic rats by suppression of both oxidative stress and aldose reductase. Chin. Med. J. 2008;121:706–712. doi: 10.1097/00029330-200804020-00009. [DOI] [PubMed] [Google Scholar]
- 63.Kazaz I.O., Mentese A., Demir S., Kerimoglu G., Colak F., Bodur A., Alver A., Kutlu O., Turedi S. Berberine inhibits the ischemia-reperfusion induced testicular injury through decreasing oxidative stress. Am. J. Emerg. Med. 2020;38:33–37. doi: 10.1016/j.ajem.2019.04.001. [DOI] [PubMed] [Google Scholar]
- 64.Hsieh Y.S., Kuo W.H., Lin T.W., Chang H.R., Lin T.H., Chen P.N., Chu S.C. Protective effects of berberine against low-density lipoprotein (LDL) oxidation and oxidized LDL-induced cytotoxicity on endothelial cells. J. Agric. Food Chem. 2007;55:10437–10445. doi: 10.1021/jf071868c. [DOI] [PubMed] [Google Scholar]
- 65.Germoush M.O., Mahmoud A.M. Berberine mitigates cyclophosphamide-induced hepatotoxicity by modulating antioxidant status and inflammatory cytokines. J. Cancer Res. Clin. Oncol. 2014;140:1103–1109. doi: 10.1007/s00432-014-1665-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Deng H., Jia Y., Pan D., Ma Z. Berberine alleviates rotenone-induced cytotoxicity by antioxidation and activation of PI3K/Akt signaling pathway in SH-SY5Y cells. Neuroreport. 2020;31:41–47. doi: 10.1097/WNR.0000000000001365. [DOI] [PubMed] [Google Scholar]
- 67.Bhutada P., Mundhada Y., Bansod K., Tawari S., Patil S., Dixit P., Umathe S., Mundhada D. Protection of cholinergic and antioxidant system contributes to the effect of berberine ameliorating memory dysfunction in rat model of streptozotocin-induced diabetes. Behav. Brain Res. 2011;220:30–41. doi: 10.1016/j.bbr.2011.01.022. [DOI] [PubMed] [Google Scholar]
- 68.Yokozawa T., Ishida A., Kashiwada Y., Cho E.J., Kim H.Y., Ikeshiro Y. Coptidis Rhizoma: Protective effects against peroxynitrite-induced oxidative damage and elucidation of its active components. J. Pharm. Pharmacol. 2004;56:547–556. doi: 10.1211/0022357023024. [DOI] [PubMed] [Google Scholar]
- 69.Adefegha S.A., Oboh G., Okeke B.M. Comparative effects of berberine and piperine on the neuroprotective potential of neostigmine. J. Complement. Integr. Med. 2021;18:491–497. doi: 10.1515/jcim-2020-0055. [DOI] [PubMed] [Google Scholar]
- 70.Sarna L.K., Wu N., Hwang S.Y., Siow Y.L., Karmin O. Berberine inhibits NADPH oxidase mediated superoxide anion production in macrophages. Can. J. Physiol. Pharmacol. 2010;88:369–378. doi: 10.1139/Y09-136. [DOI] [PubMed] [Google Scholar]
- 71.Zhuang W., Li T., Wang C., Shi X., Li Y., Zhang S., Zhao Z., Dong H., Qiao Y. Berberine exerts antioxidant effects via protection of spiral ganglion cells against cytomegalovirus-induced apoptosis. Free Radic. Biol. Med. 2018;121:127–135. doi: 10.1016/j.freeradbiomed.2018.04.575. [DOI] [PubMed] [Google Scholar]
- 72.Dai C., Tian E., Hao Z., Tang S., Wang Z., Sharma G., Jiang H., Shen J. Aflatoxin B1 Toxicity and Protective Effects of Curcumin: Molecular Mechanisms and Clinical Implications. Antioxidants. 2022;11:2031. doi: 10.3390/antiox11102031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Tian H., Kang Y.M., Gao H.L., Shi X.L., Fu L.Y., Li Y., Jia X.Y., Liu K.L., Qi J., Li H.B., et al. Chronic infusion of berberine into the hypothalamic paraventricular nucleus attenuates hypertension and sympathoexcitation via the ROS/Erk1/2/iNOS pathway. Phytomed. Int. J. Phytother. Phytopharm. 2019;52:216–224. doi: 10.1016/j.phymed.2018.09.206. [DOI] [PubMed] [Google Scholar]
- 74.Shou J.W., Li X.X., Tang Y.S., Lim-Ho Kong B., Wu H.Y., Xiao M.J., Cheung C.K., Shaw P.C. Novel mechanistic insight on the neuroprotective effect of berberine: The role of PPARδ for antioxidant action. Free Radic. Biol. Med. 2022;181:62–71. doi: 10.1016/j.freeradbiomed.2022.01.022. [DOI] [PubMed] [Google Scholar]
- 75.Tonelli C., Chio I.I.C., Tuveson D.A. Transcriptional Regulation by Nrf2. Antioxid. Redox Signal. 2018;29:1727–1745. doi: 10.1089/ars.2017.7342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Buendia I., Michalska P., Navarro E., Gameiro I., Egea J., León R. Nrf2-ARE pathway: An emerging target against oxidative stress and neuroinflammation in neurodegenerative diseases. Pharmacol. Ther. 2016;157:84–104. doi: 10.1016/j.pharmthera.2015.11.003. [DOI] [PubMed] [Google Scholar]
- 77.Li D., Dai C., Yang X., Wang F., Yu X., Xiao X., Tang S. Critical role of p21 on olaquindox-induced mitochondrial apoptosis and S-phase arrest involves activation of PI3K/AKT and inhibition of Nrf2/HO-1pathway. Food Chem. Toxicol. 2017;108:148–160. doi: 10.1016/j.fct.2017.07.054. [DOI] [PubMed] [Google Scholar]
- 78.Deng S., Tang S., Zhang S., Zhang C., Wang C., Zhou Y., Dai C., Xiao X. Furazolidone induces apoptosis through activating reactive oxygen species-dependent mitochondrial signaling pathway and suppressing PI3K/Akt signaling pathway in HepG2 cells. Food Chem. Toxicol. 2015;75:173–186. doi: 10.1016/j.fct.2014.11.019. [DOI] [PubMed] [Google Scholar]
- 79.Hsu Y.Y., Tseng Y.T., Lo Y.C. Berberine, a natural antidiabetes drug, attenuates glucose neurotoxicity and promotes Nrf2-related neurite outgrowth. Toxicol. Appl. Pharmacol. 2013;272:787–796. doi: 10.1016/j.taap.2013.08.008. [DOI] [PubMed] [Google Scholar]
- 80.Hsu Y.Y., Chen C.S., Wu S.N., Jong Y.J., Lo Y.C. Berberine activates Nrf2 nuclear translocation and protects against oxidative damage via a phosphatidylinositol 3-kinase/Akt-dependent mechanism in NSC34 motor neuron-like cells. Eur. J. Pharm. Sci. 2012;46:415–425. doi: 10.1016/j.ejps.2012.03.004. [DOI] [PubMed] [Google Scholar]
- 81.Phadwal K., Vrahnas C., Ganley I.G., MacRae V.E. Mitochondrial Dysfunction: Cause or Consequence of Vascular Calcification? Front. Cell Dev. Biol. 2021;9:611922. doi: 10.3389/fcell.2021.611922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Bonora M., Giorgi C., Pinton P. Molecular mechanisms and consequences of mitochondrial permeability transition. Nat. Rev. Mol. Cell Biol. 2022;23:266–285. doi: 10.1038/s41580-021-00433-y. [DOI] [PubMed] [Google Scholar]
- 83.Zhao C., Su P., Lv C., Guo L., Cao G., Qin C., Zhang W. Berberine Alleviates Amyloid β-Induced Mitochondrial Dysfunction and Synaptic Loss. Oxidative Med. Cell. Longev. 2019;2019:7593608. doi: 10.1155/2019/7593608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Zhou X.Q., Zeng X.N., Kong H., Sun X.L. Neuroprotective effects of berberine on stroke models in vitro and in vivo. Neurosci. Lett. 2008;447:31–36. doi: 10.1016/j.neulet.2008.09.064. [DOI] [PubMed] [Google Scholar]
- 85.Singh D.P., Chopra K. Verapamil augments the neuroprotectant action of berberine in rat model of transient global cerebral ischemia. Eur. J. Pharmacol. 2013;720:98–106. doi: 10.1016/j.ejphar.2013.10.043. [DOI] [PubMed] [Google Scholar]
- 86.Zhang Q., Qian Z., Pan L., Li H., Zhu H. Hypoxia-inducible factor 1 mediates the anti-apoptosis of berberine in neurons during hypoxia/ischemia. Acta Physiol. Hung. 2012;99:311–323. doi: 10.1556/APhysiol.99.2012.3.8. [DOI] [PubMed] [Google Scholar]
- 87.Klegeris A., McGeer P.L. Non-steroidal anti-inflammatory drugs (NSAIDs) and other anti-inflammatory agents in the treatment of neurodegenerative disease. Curr. Alzheimer Res. 2005;2:355–365. doi: 10.2174/1567205054367883. [DOI] [PubMed] [Google Scholar]
- 88.Esposito E., Di Matteo V., Benigno A., Pierucci M., Crescimanno G., Di Giovanni G. Non-steroidal anti-inflammatory drugs in Parkinson’s disease. Exp. Neurol. 2007;205:295–312. doi: 10.1016/j.expneurol.2007.02.008. [DOI] [PubMed] [Google Scholar]
- 89.Apetz N., Munch G., Govindaraghavan S., Gyengesi E. Natural compounds and plant extracts as therapeutics against chronic inflammation in Alzheimer’s disease--a translational perspective. CNS Neurol. Disord. Drug Targets. 2014;13:1175–1191. doi: 10.2174/1871527313666140917110635. [DOI] [PubMed] [Google Scholar]
- 90.Lu D.Y., Tang C.H., Chen Y.H., Wei I.H. Berberine suppresses neuroinflammatory responses through AMP-activated protein kinase activation in BV-2 microglia. J. Cell. Biochem. 2010;110:697–705. doi: 10.1002/jcb.22580. [DOI] [PubMed] [Google Scholar]
- 91.Gabet B., Kuo P.C., Fuentes S., Patel Y., Adow A., Alsakka M., Avila P., Beam T., Yen J.H., Brown D.A. Identification of N-benzyltetrahydroisoquinolines as novel anti-neuroinflammatory agents. Bioorg. Med. Chem. 2018;26:5711–5717. doi: 10.1016/j.bmc.2018.10.020. [DOI] [PubMed] [Google Scholar]
- 92.Chen C.C., Hung T.H., Lee C.Y., Wang L.F., Wu C.H., Ke C.H., Chen S.F. Berberine protects against neuronal damage via suppression of glia-mediated inflammation in traumatic brain injury. PLoS ONE. 2014;9:e115694. doi: 10.1371/journal.pone.0115694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Qin Z., Shi D.D., Li W., Cheng D., Zhang Y.D., Zhang S., Tsoi B., Zhao J., Wang Z., Zhang Z.J. Berberine ameliorates depression-like behaviors in mice via inhibiting NLRP3 inflammasome-mediated neuroinflammation and preventing neuroplasticity disruption. J. Neuroinflamm. 2023;20:54. doi: 10.1186/s12974-023-02744-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Wong L.R., Tan E.A., Lim M.E.J., Shen W., Lian X.L., Wang Y., Chen L., Ho P.C. Functional effects of berberine in modulating mitochondrial dysfunction and inflammatory response in the respective amyloidogenic cells and activated microglial cells—In vitro models simulating Alzheimer’s disease pathology. Life Sci. 2021;282:119824. doi: 10.1016/j.lfs.2021.119824. [DOI] [PubMed] [Google Scholar]
- 95.Sadraie S., Kiasalari Z., Razavian M., Azimi S., Sedighnejad L., Afshin-Majd S., Baluchnejadmojarad T., Roghani M. Berberine ameliorates lipopolysaccharide-induced learning and memory deficit in the rat: Insights into underlying molecular mechanisms. Metab. Brain Dis. 2019;34:245–255. doi: 10.1007/s11011-018-0349-5. [DOI] [PubMed] [Google Scholar]
- 96.Jia L., Liu J., Song Z., Pan X., Chen L., Cui X., Wang M. Berberine suppresses amyloid-beta-induced inflammatory response in microglia by inhibiting nuclear factor-kappaB and mitogen-activated protein kinase signalling pathways. J. Pharm. Pharmacol. 2012;64:1510–1521. doi: 10.1111/j.2042-7158.2012.01529.x. [DOI] [PubMed] [Google Scholar]
- 97.Zhang X., Zhang X., Wang C., Li Y., Dong L., Cui L., Wang L., Liu Z., Qiao H., Zhu C., et al. Neuroprotection of early and short-time applying berberine in the acute phase of cerebral ischemia: Up-regulated pAkt, pGSK and pCREB, down-regulated NF-κB expression, ameliorated BBB permeability. Brain Res. 2012;1459:61–70. doi: 10.1016/j.brainres.2012.03.065. [DOI] [PubMed] [Google Scholar]
- 98.Nam K.N., Kim J.H., Jung H.J., Park J.-M., Moon S.K., Kim Y., Kim S.Y., Lee E.H. Berberine inhibits inflammatory activation of rat brain microglia. Neural Regen. Res. 2010;5:1384–1390. doi: 10.3969/j.issn.1673-5374.2010.18.004. [DOI] [Google Scholar]
- 99.Liu Y.M., Niu L., Wang L.L., Bai L., Fang X.Y., Li Y.C., Yi L.T. Berberine attenuates depressive-like behaviors by suppressing neuro-inflammation in stressed mice. Brain Res. Bull. 2017;134:220–227. doi: 10.1016/j.brainresbull.2017.08.008. [DOI] [PubMed] [Google Scholar]
- 100.Zhang Z., Li X., Li F., An L. Berberine alleviates postoperative cognitive dysfunction by suppressing neuroinflammation in aged mice. Int. Immunopharmacol. 2016;38:426–433. doi: 10.1016/j.intimp.2016.06.031. [DOI] [PubMed] [Google Scholar]
- 101.Tian Y., Zheng Y., Wang Q., Yan F., Tao Z., Zhao F., Wang Y., Huang Y., Li F., Du Y., et al. Berberine Ameliorates Cognitive Impairment by Regulating Microglial Polarization and Increasing Expression of Anti-inflammatory Factors following Permanent Bilateral Common Carotid Artery Occlusion in Rats. CNS Neurol. Disord. Drug Targets. 2022;21:869–879. doi: 10.2174/1871527321666220124140323. [DOI] [PubMed] [Google Scholar]
- 102.Yoo K.Y., Hwang I.K., Kim J.D., Kang I.J., Park J., Yi J.S., Kim J.K., Bae Y.S., Won M.H. Antiinflammatory effect of the ethanol extract of Berberis koreana in a gerbil model of cerebral ischemia/reperfusion. Phytother. Res. PTR. 2008;22:1527–1532. doi: 10.1002/ptr.2527. [DOI] [PubMed] [Google Scholar]
- 103.Lomphithak T., Akara-Amornthum P., Murakami K., Hashimoto M., Usubuchi H., Iwabuchi E., Unno M., Cai Z., Sasano H., Jitkaew S. Tumor necroptosis is correlated with a favorable immune cell signature and programmed death-ligand 1 expression in cholangiocarcinoma. Sci. Rep. 2021;11:11743. doi: 10.1038/s41598-021-89977-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Xiong J., Wang L., Fei X.C., Jiang X.F., Zheng Z., Zhao Y., Wang C.F., Li B., Chen S.J., Janin A., et al. MYC is a positive regulator of choline metabolism and impedes mitophagy-dependent necroptosis in diffuse large B-cell lymphoma. Blood Cancer J. 2017;7:8503511. doi: 10.1038/bcj.2017.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Liu L., Fan J., Ai G., Liu J., Luo N., Li C., Cheng Z. Berberine in combination with cisplatin induces necroptosis and apoptosis in ovarian cancer cells. Biol. Res. 2019;52:37. doi: 10.1186/s40659-019-0243-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Sengupta D., Chowdhury K.D., Chatterjee S., Sarkar A., Paul S., Sur P.K., Sadhukhan G.C. Modulation of adenylate cyclase signaling in association with MKK3/6 stabilization under combination of SAC and berberine to reduce HepG2 cell survivability. Apoptosis. 2017;22:1362–1379. doi: 10.1007/s10495-017-1407-x. [DOI] [PubMed] [Google Scholar]
- 107.Ou X., Hua Y., Liao X., Gong C., Kang Y. Cognitive impairments induced by severe acute pancreatitis are attenuated by berberine treatment in rats. Mol. Med. Rep. 2018;18:3437–3444. doi: 10.3892/mmr.2018.9313. [DOI] [PubMed] [Google Scholar]
- 108.Chang C.F., Lee Y.C., Lee K.H., Lin H.C., Chen C.L., Shen C.J., Huang C.C. Therapeutic effect of berberine on TDP-43-related pathogenesis in FTLD and ALS. J. Biomed. Sci. 2016;23:72. doi: 10.1186/s12929-016-0290-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Jiang W., Wei W., Gaertig M.A., Li S., Li X.J. Therapeutic Effect of Berberine on Huntington’s Disease Transgenic Mouse Model. PLoS ONE. 2015;10:e0134142. doi: 10.1371/journal.pone.0134142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Huang M., Jiang X., Liang Y., Liu Q., Chen S., Guo Y. Berberine improves cognitive impairment by promoting autophagic clearance and inhibiting production of β-amyloid in APP/tau/PS1 mouse model of Alzheimer’s disease. Exp. Gerontol. 2017;91:25–33. doi: 10.1016/j.exger.2017.02.004. [DOI] [PubMed] [Google Scholar]
- 111.Li S., Jiang Y., Xing X., Lin R., Li Q., Zhou W., Qiu W., Zheng W. Protective Mechanism of Berberine on Human Retinal Pigment Epithelial Cells against Apoptosis Induced by Hydrogen Peroxide via the Stimulation of Autophagy. Oxidative Med. Cell. Longev. 2021;2021:7654143. doi: 10.1155/2021/7654143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Chen H., Ji Y., Yan X., Su G., Chen L., Xiao J. Berberine attenuates apoptosis in rat retinal Müller cells stimulated with high glucose via enhancing autophagy and the AMPK/mTOR signaling. Biomed. Pharmacother. 2018;108:1201–1207. doi: 10.1016/j.biopha.2018.09.140. [DOI] [PubMed] [Google Scholar]
- 113.Liu J., Liu P., Xu T., Chen Z., Kong H., Chu W., Wang Y., Liu Y. Berberine Induces Autophagic Cell Death in Acute Lymphoblastic Leukemia by Inactivating AKT/mTORC1 Signaling. Drug Des. Dev. Ther. 2020;14:1813–1823. doi: 10.2147/DDDT.S239247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Zhu N., Cao X., Hao P., Zhang Y., Chen Y., Zhang J., Li J., Gao C., Li L. Berberine attenuates mitochondrial dysfunction by inducing autophagic flux in myocardial hypoxia/reoxygenation injury. Cell Stress Chaperones. 2020;25:417–426. doi: 10.1007/s12192-020-01081-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Li M.H., Zhang Y.J., Yu Y.H., Yang S.H., Iqbal J., Mi Q.Y., Li B., Wang Z.M., Mao W.X., Xie H.G., et al. Berberine improves pressure overload-induced cardiac hypertrophy and dysfunction through enhanced autophagy. Eur. J. Pharmacol. 2014;728:67–76. doi: 10.1016/j.ejphar.2014.01.061. [DOI] [PubMed] [Google Scholar]
- 116.Zhang Y.J., Yang S.H., Li M.H., Iqbal J., Bourantas C.V., Mi Q.Y., Yu Y.H., Li J.J., Zhao S.L., Tian N.L., et al. Berberine attenuates adverse left ventricular remodeling and cardiac dysfunction after acute myocardial infarction in rats: Role of autophagy. Clin. Exp. Pharmacol. Physiol. 2014;41:995–1002. doi: 10.1111/1440-1681.12309. [DOI] [PubMed] [Google Scholar]
- 117.Wang N., Feng Y., Zhu M., Tsang C.M., Man K., Tong Y., Tsao S.W. Berberine induces autophagic cell death and mitochondrial apoptosis in liver cancer cells: The cellular mechanism. J. Cell. Biochem. 2010;111:1426–1436. doi: 10.1002/jcb.22869. [DOI] [PubMed] [Google Scholar]
- 118.Sun Y., Xia M., Yan H., Han Y., Zhang F., Hu Z., Cui A., Ma F., Liu Z., Gong Q., et al. Berberine attenuates hepatic steatosis and enhances energy expenditure in mice by inducing autophagy and fibroblast growth factor 21. Br. J. Pharmacol. 2018;175:374–387. doi: 10.1111/bph.14079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Lin Y., Sheng M., Weng Y., Xu R., Lu N., Du H., Yu W. Berberine protects against ischemia/reperfusion injury after orthotopic liver transplantation via activating Sirt1/FoxO3α induced autophagy. Biochem. Biophys. Res. Commun. 2017;483:885–891. doi: 10.1016/j.bbrc.2017.01.028. [DOI] [PubMed] [Google Scholar]
- 120.Hou Q., Tang X., Liu H., Tang J., Yang Y., Jing X., Xiao Q., Wang W., Gou X., Wang Z. Berberine induces cell death in human hepatoma cells in vitro by downregulating CD147. Cancer Sci. 2011;102:1287–1292. doi: 10.1111/j.1349-7006.2011.01933.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.He Q., Mei D., Sha S., Fan S., Wang L., Dong M. ERK-dependent mTOR pathway is involved in berberine-induced autophagy in hepatic steatosis. J. Mol. Endocrinol. 2016;57:251–260. doi: 10.1530/JME-16-0139. [DOI] [PubMed] [Google Scholar]
- 122.Peng P.L., Kuo W.H., Tseng H.C., Chou F.P. Synergistic tumor-killing effect of radiation and berberine combined treatment in lung cancer: The contribution of autophagic cell death. Int. J. Radiat. Oncol. Biol. Phys. 2008;70:529–542. doi: 10.1016/j.ijrobp.2007.08.034. [DOI] [PubMed] [Google Scholar]
- 123.Chen P., Dai C.H., Shi Z.H., Wang Y., Wu J.N., Chen K., Su J.Y., Li J. Synergistic inhibitory effect of berberine and icotinib on non-small cell lung cancer cells via inducing autophagic cell death and apoptosis. Apoptosis. 2021;26:639–656. doi: 10.1007/s10495-021-01694-w. [DOI] [PubMed] [Google Scholar]
- 124.Chitra P., Saiprasad G., Manikandan R., Sudhandiran G. Berberine inhibits Smad and non-Smad signaling cascades and enhances autophagy against pulmonary fibrosis. J. Mol. Med. 2015;93:1015–1031. doi: 10.1007/s00109-015-1283-1. [DOI] [PubMed] [Google Scholar]
- 125.Lopes T.Z., de Moraes F.R., Tedesco A.C., Arni R.K., Rahal P., Calmon M.F. Berberine associated photodynamic therapy promotes autophagy and apoptosis via ROS generation in renal carcinoma cells. Biomed. Pharmacother. 2020;123:109794. doi: 10.1016/j.biopha.2019.109794. [DOI] [PubMed] [Google Scholar]
- 126.Zhang Q., Wang X., Cao S., Sun Y., He X., Jiang B., Yu Y., Duan J., Qiu F., Kang N. Berberine represses human gastric cancer cell growth in vitro and in vivo by inducing cytostatic autophagy via inhibition of MAPK/mTOR/p70S6K and Akt signaling pathways. Biomed. Pharmacother. 2020;128:110245. doi: 10.1016/j.biopha.2020.110245. [DOI] [PubMed] [Google Scholar]
- 127.Han B., Wang K., Tu Y., Tan L., He C. Low-Dose Berberine Attenuates the Anti-Breast Cancer Activity of Chemotherapeutic Agents via Induction of Autophagy and Antioxidation. Dose-Response. 2020;18:1559325820939751. doi: 10.1177/1559325820939751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Zhang H., Zhao C., Cao G., Guo L., Zhang S., Liang Y., Qin C., Su P., Li H., Zhang W. Berberine modulates amyloid-β peptide generation by activating AMP-activated protein kinase. Neuropharmacology. 2017;125:408–417. doi: 10.1016/j.neuropharm.2017.08.013. [DOI] [PubMed] [Google Scholar]
- 129.Fan X., Wang J., Hou J., Lin C., Bensoussan A., Chang D., Liu J., Wang B. Berberine alleviates ox-LDL induced inflammatory factors by up-regulation of autophagy via AMPK/mTOR signaling pathway. J. Transl. Med. 2015;13:92. doi: 10.1186/s12967-015-0450-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Kline A.E., Leary J.B., Radabaugh H.L., Cheng J.P., Bondi C.O. Combination therapies for neurobehavioral and cognitive recovery after experimental traumatic brain injury: Is more better? Prog. Neurobiol. 2016;142:45–67. doi: 10.1016/j.pneurobio.2016.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Peng W.H., Lo K.L., Lee Y.H., Hung T.H., Lin Y.C. Berberine produces antidepressant-like effects in the forced swim test and in the tail suspension test in mice. Life Sci. 2007;81:933–938. doi: 10.1016/j.lfs.2007.08.003. [DOI] [PubMed] [Google Scholar]
- 132.Kuznetsova L.P., Sochilina E.E., Faddeeva M.D., Iagodina O.V. Effect of some isoquinoline alkaloids on enzymatic activity of acetylcholinesterase and monoamine oxidase. Ukr. Biokhim. Zh. 2005;77:147–153. [PubMed] [Google Scholar]
- 133.Ji H.F., Shen L. Molecular basis of inhibitory activities of berberine against pathogenic enzymes in Alzheimer’s disease. Sci. World J. 2012;2012:823201. doi: 10.1100/2012/823201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Ge P.Y., Qu S.Y., Ni S.J., Yao Z.Y., Qi Y.Y., Zhao X., Guo R., Yang N.Y., Zhang Q.C., Zhu H.X. Berberine ameliorates depression-like behavior in CUMS mice by activating TPH1 and inhibiting IDO1-associated with tryptophan metabolism. Phytother. Res. PTR. 2023;37:342–357. doi: 10.1002/ptr.7616. [DOI] [PubMed] [Google Scholar]
- 135.Lee T., Heo H., Kim Kwon Y. Effect of Berberine on Cell Survival in the Developing Rat Brain Damaged by MK-801. Exp. Neurobiol. 2010;19:140–145. doi: 10.5607/en.2010.19.3.140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Lin T.Y., Lin Y.W., Lu C.W., Huang S.K., Wang S.J. Berberine Inhibits the Release of Glutamate in Nerve Terminals from Rat Cerebral Cortex. PLoS ONE. 2013;8:e67215. doi: 10.1371/journal.pone.0067215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Liu M., Hurn P.D., Alkayed N.J. Cytochrome P450 in neurological disease. Curr. Drug Metab. 2004;5:225–234. doi: 10.2174/1389200043335540. [DOI] [PubMed] [Google Scholar]
- 138.Zverinsky I.V., Zverinskaya H.G., Sutsko I.P., Telegin P.G., Shlyahtun A.G. Effects of berberine on the recovery of rat liver xenobiotic-metabolizing enzymes after partial hepatectomy. Biomed. Khim. 2015;61:381–383. doi: 10.18097/PBMC20156103381. [DOI] [PubMed] [Google Scholar]
- 139.Zhao X., Zhang J.J., Wang X., Bu X.Y., Lou Y.Q., Zhang G.L. Effect of berberine on hepatocyte proliferation, inducible nitric oxide synthase expression, cytochrome P450 2E1 and 1A2 activities in diethylnitrosamine- and phenobarbital-treated rats. Biomed. Pharmacother. 2008;62:567–572. doi: 10.1016/j.biopha.2007.02.009. [DOI] [PubMed] [Google Scholar]
- 140.Lee S.Y., Jang H., Lee J.Y., Ma J.Y., Oh S.J., Kim S.K. Inhibitory effects of Hwang-Ryun-Hae-Dok-Tang on cytochrome P450 in human liver microsomes. Xenobiotica. 2015;45:131–138. doi: 10.3109/00498254.2014.953624. [DOI] [PubMed] [Google Scholar]
- 141.Guo Y., Pope C., Cheng X., Zhou H., Klaassen C.D. Dose-response of berberine on hepatic cytochromes P450 mRNA expression and activities in mice. J. Ethnopharmacol. 2011;138:111–118. doi: 10.1016/j.jep.2011.08.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Guo Y., Chen Y., Tan Z.R., Klaassen C.D., Zhou H.H. Repeated administration of berberine inhibits cytochromes P450 in humans. Eur. J. Clin. Pharmacol. 2012;68:213–217. doi: 10.1007/s00228-011-1108-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Cui H.M., Zhang Q.Y., Wang J.L., Chen J.L., Zhang Y.L., Tong X.L. In vitro studies of berberine metabolism and its effect of enzyme induction on HepG2 cells. J. Ethnopharmacol. 2014;158 Pt A:388–396. doi: 10.1016/j.jep.2014.10.018. [DOI] [PubMed] [Google Scholar]
- 144.Chatuphonprasert W., Nemoto N., Sakuma T., Jarukamjorn K. Modulations of cytochrome P450 expression in diabetic mice by berberine. Chem. Biol. Interact. 2012;196:23–29. doi: 10.1016/j.cbi.2012.01.006. [DOI] [PubMed] [Google Scholar]
- 145.McDonald M.G., Tian D.D., Thummel K.E., Paine M.F., Rettie A.E. Modulation of Major Human Liver Microsomal Cytochromes P450 by Component Alkaloids of Goldenseal: Time-Dependent Inhibition and Allosteric Effects. Drug Metab. Dispos. Biol. Fate Chem. 2020;48:1018–1027. doi: 10.1124/dmd.120.091041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Wen C.J., Wu L.X., Fu L.J., Shen D.Y., Zhang X., Zhang Y.W., Yu J., Zhou H.H. Preferential induction of CYP1A1 over CYP1B1 in human breast cancer MCF-7 cells after exposure to berberine. Asian Pac. J. Cancer Prev. 2014;15:495–499. doi: 10.7314/APJCP.2014.15.1.495. [DOI] [PubMed] [Google Scholar]
- 147.Lo S.N., Wang C.W., Chen Y.S., Huang C.C., Wu T.S., Li L.A., Lee I.J., Ueng Y.F. Berberine Activates Aryl Hydrocarbon Receptor but Suppresses CYP1A1 Induction through miR-21-3p Stimulation in MCF-7 Breast Cancer Cells. Molecules. 2017;22:1847. doi: 10.3390/molecules22111847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Lo S.N., Chang Y.P., Tsai K.C., Chang C.Y., Wu T.S., Ueng Y.F. Inhibition of CYP1 by berberine, palmatine, and jatrorrhizine: Selectivity, kinetic characterization, and molecular modeling. Toxicol. Appl. Pharmacol. 2013;272:671–680. doi: 10.1016/j.taap.2013.07.005. [DOI] [PubMed] [Google Scholar]
- 149.Falero-Perez J., Song Y.S., Sorenson C.M., Sheibani N. CYP1B1: A key regulator of redox homeostasis. Trends Cell Mol. Biol. 2018;13:27–45. [PMC free article] [PubMed] [Google Scholar]
- 150.Falero-Perez J., Sorenson C.M., Sheibani N. Cyp1b1-deficient retinal astrocytes are more proliferative and migratory and are protected from oxidative stress and inflammation. Am. J. Physiol. Cell Physiol. 2019;316:C767–C781. doi: 10.1152/ajpcell.00021.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Qiu W., Jiang X.H., Liu C.X., Ju Y., Jin J.X. Effect of berberine on the pharmacokinetics of substrates of CYP3A and P-gp. Phytother. Res. PTR. 2009;23:1553–1558. doi: 10.1002/ptr.2808. [DOI] [PubMed] [Google Scholar]
- 152.Wang M., Zhang J., Zhao H., Wan D., Jiang Z. Berberine combined with cyclosporine A alleviates acute graft-versus-host disease in murine models. Int. Immunopharmacol. 2020;81:106205. doi: 10.1016/j.intimp.2020.106205. [DOI] [PubMed] [Google Scholar]
- 153.Kumar A., Ekavali M.J., Chopra K., Dhull D.K. Possible role of P-glycoprotein in the neuroprotective mechanism of berberine in intracerebroventricular streptozotocin-induced cognitive dysfunction. Psychopharmacology. 2016;233:137–152. doi: 10.1007/s00213-015-4095-7. [DOI] [PubMed] [Google Scholar]
- 154.Ni S.J., Yao Z.Y., Wei X., Heng X., Qu S.Y., Zhao X., Qi Y.Y., Ge P.Y., Xu C.P., Yang N.Y., et al. Vagus nerve stimulated by microbiota-derived hydrogen sulfide mediates the regulation of berberine on microglia in transient middle cerebral artery occlusion rats. Phytother. Res. PTR. 2022;36:2964–2981. doi: 10.1002/ptr.7490. [DOI] [PubMed] [Google Scholar]
- 155.Hong J.S., Chu Y.K., Lee H., Ahn B.H., Park J.H., Kim M.J., Lee S., Ryoo H.S., Jang J.H., Lee S.R., et al. Effects of berberine on hippocampal neuronal damage and matrix metalloproteinase-9 activity following transient global cerebral ischemia. J. Neurosci. Res. 2012;90:489–497. doi: 10.1002/jnr.22756. [DOI] [PubMed] [Google Scholar]
- 156.Ma X., Jiang Y., Wu A., Chen X., Pi R., Liu M., Liu Y. Berberine attenuates experimental autoimmune encephalomyelitis in C57 BL/6 mice. PLoS ONE. 2010;5:e13489. doi: 10.1371/journal.pone.0013489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Wu C., Yang K., Liu Q., Wakui M., Jin G.Z., Zhen X., Wu J. Tetrahydroberberine blocks ATP-sensitive potassium channels in dopamine neurons acutely-dissociated from rat substantia nigra pars compacta. Neuropharmacology. 2010;59:567–572. doi: 10.1016/j.neuropharm.2010.08.018. [DOI] [PubMed] [Google Scholar]
- 158.Shigeta K., Ootaki K., Tatemoto H., Nakanishi T., Inada A., Muto N. Potentiation of nerve growth factor-induced neurite outgrowth in PC12 cells by a Coptidis Rhizoma extract and protoberberine alkaloids. Biosci. Biotechnol. Biochem. 2002;66:2491–2494. doi: 10.1271/bbb.66.2491. [DOI] [PubMed] [Google Scholar]
- 159.Han A.M., Heo H., Kwon Y.K. Berberine promotes axonal regeneration in injured nerves of the peripheral nervous system. J. Med. Food. 2012;15:413–417. doi: 10.1089/jmf.2011.2029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Kheir M.M., Wang Y., Hua L., Hu J., Li L., Lei F., Du L. Acute toxicity of berberine and its correlation with the blood concentration in mice. Food Chem. Toxicol. 2010;48:1105–1110. doi: 10.1016/j.fct.2010.01.033. [DOI] [PubMed] [Google Scholar]
- 161.Rad S.Z.K., Rameshrad M., Hosseinzadeh H. Toxicology effects of Berberis vulgaris (barberry) and its active constituent, berberine: A review. Iran. J. Basic Med. Sci. 2017;20:516–529. doi: 10.22038/ijbms.2017.8676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Mahmoudi M., Zamani Taghizadeh Rabe S., Balali-Mood M., Karimi G., Memar B., Rahnama M., Tabasi N., Khazaee M., Riahi-Zanjani B. Immunotoxicity induced in mice by subacute exposure to berberine. J. Immunotoxicol. 2016;13:255–262. doi: 10.3109/1547691X.2015.1058306. [DOI] [PubMed] [Google Scholar]
- 163.Küpeli E., Koşar M., Yeşilada E., Hüsnü K., Başer C. A comparative study on the anti-inflammatory, antinociceptive and antipyretic effects of isoquinoline alkaloids from the roots of Turkish Berberis species. Life Sci. 2002;72:645–657. doi: 10.1016/S0024-3205(02)02200-2. [DOI] [PubMed] [Google Scholar]
- 164.Terpstra A.H. Differences between humans and mice in efficacy of the body fat lowering effect of conjugated linoleic acid: Role of metabolic rate. J. Nutr. 2001;131:2067–2068. doi: 10.1093/jn/131.7.2067. [DOI] [PubMed] [Google Scholar]
- 165.Liu Y., Hua W., Li Y., Xian X., Zhao Z., Liu C., Zou J., Li J., Fang X., Zhu Y. Berberine suppresses colon cancer cell proliferation by inhibiting the SCAP/SREBP-1 signaling pathway-mediated lipogenesis. Biochem. Pharmacol. 2020;174:113776. doi: 10.1016/j.bcp.2019.113776. [DOI] [PubMed] [Google Scholar]
- 166.Bhadra K., Kumar G.S. Therapeutic potential of nucleic acid-binding isoquinoline alkaloids: Binding aspects and implications for drug design. Med. Res. Rev. 2011;31:821–862. doi: 10.1002/med.20202. [DOI] [PubMed] [Google Scholar]
- 167.Yadav R.C., Kumar G.S., Bhadra K., Giri P., Sinha R., Pal S., Maiti M. Berberine, a strong polyriboadenylic acid binding plant alkaloid: Spectroscopic, viscometric, and thermodynamic study. Bioorg. Med. Chem. 2005;13:165–174. doi: 10.1016/j.bmc.2004.09.045. [DOI] [PubMed] [Google Scholar]
- 168.Rauf A., Abu-Izneid T., Khalil A.A., Imran M., Shah Z.A., Emran T.B., Mitra S., Khan Z., Alhumaydhi F.A., Aljohani A.S.M., et al. Berberine as a Potential Anticancer Agent: A Comprehensive Review. Molecules. 2021;26:7368. doi: 10.3390/molecules26237368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Jahnke G.D., Price C.J., Marr M.C., Myers C.B., George J.D. Developmental toxicity evaluation of berberine in rats and mice. Birth Defects Res. Part B Dev. Reprod. Toxicol. 2006;77:195–206. doi: 10.1002/bdrb.20075. [DOI] [PubMed] [Google Scholar]
- 170.Singh N., Sharma B. Toxicological Effects of Berberine and Sanguinarine. Front. Mol. Biosci. 2018;5:21. doi: 10.3389/fmolb.2018.00021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Yin J., Xing H., Ye J. Efficacy of berberine in patients with type 2 diabetes mellitus. Metab. Clin. Exp. 2008;57:712–717. doi: 10.1016/j.metabol.2008.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Zhu F.-q., Chen L., Zhu J.-h. Influence of berberine combining with atorvastatin on serum high-sensitivity C-reactive protein and adipocyte fatty acid-binding protein in patients with acute ischemic stroke. Chin. J. Contemp. Neurol. Neurosurg. 2015;15:43–47. doi: 10.3969/j.issn.1672-6731.2015.01.010. [DOI] [Google Scholar]
- 173.Ming J., Yu X., Xu X., Wang L., Ding C., Wang Z., Xie X., Li S., Yang W., Luo S., et al. Effectiveness and safety of Bifidobacterium and berberine in human hyperglycemia and their regulatory effect on the gut microbiota: A multi-center, double-blind, randomized, parallel-controlled study. Genome Med. 2021;13:125. doi: 10.1186/s13073-021-00942-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Li Y., Wang P., Chai M.J., Yang F., Li H.S., Zhao J., Wang H., Lu D.D. Effects of berberine on serum inflammatory factors and carotid atherosclerotic plaques in patients with acute cerebral ischemic stroke. Zhongguo Zhong Yao Za Zhi. 2016;41:4066–4071. doi: 10.4268/cjcmm20162128. [DOI] [PubMed] [Google Scholar]
- 175.Liu C.S., Zheng Y.R., Zhang Y.F., Long X.Y. Research progress on berberine with a special focus on its oral bioavailability. Fitoterapia. 2016;109:274–282. doi: 10.1016/j.fitote.2016.02.001. [DOI] [PubMed] [Google Scholar]
- 176.Tan X.S., Ma J.Y., Feng R., Ma C., Chen W.J., Sun Y.P., Fu J., Huang M., He C.Y., Shou J.W., et al. Tissue distribution of berberine and its metabolites after oral administration in rats. PLoS ONE. 2013;8:e77969. doi: 10.1371/journal.pone.0077969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Wang Q.S., Li K., Gao L.N., Zhang Y., Lin K.M., Cui Y.L. Intranasal delivery of berberine via in situ thermoresponsive hydrogels with non-invasive therapy exhibits better antidepressant-like effects. Biomater. Sci. 2020;8:2853–2865. doi: 10.1039/C9BM02006C. [DOI] [PubMed] [Google Scholar]
- 178.Xu D., Qiu C., Wang Y., Qiao T., Cui Y.L. Intranasal co-delivery of berberine and evodiamine by self-assembled thermosensitive in-situ hydrogels for improving depressive disorder. Int. J. Pharm. 2021;603:120667. doi: 10.1016/j.ijpharm.2021.120667. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.