Abstract
The thermal inactivation kinetics of Salmonella enteritidis PT4 between 49 and 60°C were investigated. Using procedures designed to eliminate methodological artifacts, we found that the death kinetics deviated from the accepted model of first-order inactivation. When we used high-density stationary-phase populations and sensitive enumeration, the survivor curves at 60°C were reproducibly biphasic. The decimal reduction time at 60°C (D60°C) of the tail subpopulation was more than four times that of the majority population. This difference decreased with decreasing temperature; i.e., the survivor curves became more linear, but the proportion of tail cells remained a constant proportion of the initial population, about 1 in 104 to 105. Z plots (log D versus temperature) for the two populations showed that the D values coincided at 51°C, indicating that the survivor curves should be linear at this temperature, and this was confirmed experimentally. Investigations into the nature of the tails ruled out genotypic differences between the populations and protection due to leakage from early heat casualties. Heating of cells at 59°C in the presence of 5 or 100 μg of chloramphenicol per ml resulted in reductions in the levels of tailing. These reductions were greatest at the higher chloramphenicol concentration. Our results indicate that de novo protein synthesis of heat shock proteins is responsible for the observed tailing. Chemostat-cultured cells heated at 60°C also produced biphasic survivor curves in all but one instance. Cells with higher growth rates were more heat sensitive, but tailing was comparable with batch cultures. Starved cells (no dilution input) displayed linear inactivation kinetics, suggesting that during starvation a rapid heat shock response cannot be initiated.
The heat resistance of a microorganism can be quantified by determining its time to decimal reduction (D value), the time that it takes to inactivate 90% of the population at a given temperature. This value can be derived from a plot of log10 survivors versus elapsed time (20), which is assumed to result in a straight line. The D value is equal to the reciprocal of the slope.
Explanations for first-order death remain contentious (16). Two theoretical approaches were originally described by Madsen and Nyman (25). In the mechanistic concept of these authors, microbial inactivation is treated as a monomolecular reaction. Later, it was suggested that within bacteria, one or a few key target volumes exist, which when subjected to heat energy render the cells nonviable if sufficient energy is transferred from surrounding water molecules. At any one instant there are a certain number of neighboring water molecules possessing sufficient energy to damage a target molecule (7, 16). Much work has focused on the nature of the potential intracellular targets; proteins, membranes, ribosomes, and nucleic acid material have all been shown to be adversely affected by heat (2, 3, 24). Alternatively, the vitalistic concept proposes that in a genetically homogeneous population, phenotypic variation exists such that resistance to a lethal agent is not uniform. Withell (31) described how a log-normal distribution of resistance could describe both log-linear death kinetics and observed deviations from linearity, depending on the standard deviation.
Successful use of D values for defining food-processing requirements relies on the inactivation rate being log linear. Previously published data, however, have frequently suggested that this is not the case (8). The reported deviations from first-order inactivation have included shouldered death curves in which the initial death rate is slower and, of greater concern for the food industry, tailing in which a subpopulation displays greater heat resistance than the majority population (6).
Tailing deviations are often viewed as a result of experimental difficulties when thermal inactivation assessments are made. Tailing has been found in a range of microorganisms by using a variety of experimental techniques (8, 9, 15, 28). In this study we used a simple experimental approach to determine the death kinetics of Salmonella enteritidis PT4 over a range of lethal temperatures. Our objectives were to eliminate artifactual causes of tailing and to investigate the origins of any remaining deviation from log-linear death.
MATERIALS AND METHODS
Organism.
S. enteritidis phage type 4 strain P167807, supplied by the Division of Enteric Pathogens, Central Public Health Laboratory, London, United Kingdom, was stored frozen in bead vials (Protect; Technical Service Consultants Ltd., Heywood, Lancashire, United Kingdom) at −70°C and was resuscitated by growing it to a concentration of 109 CFU ml−1 in 10 ml of nutrient broth (NB) (Unipath UK Ltd., Basingstoke, Hampshire, United Kingdom) at 37°C for 24 h.
Heat challenge protocol. (i) Preparation of cells.
Resuscitated cultures were diluted 10-fold in maximum recovery diluent (MRD) (Unipath UK Ltd.) for inoculation into prewarmed (to 37°C) NB (100 ml, 500 ml, or 1.5 liters) to provide initial suspensions containing approximately 1 CFU ml−1. All broth media were incubated statically at 37°C for 18 ± 1 h and immediately centrifuged (6,500 × g for 15 min at 20°C). A previously determined growth curve obtained under these conditions had shown that a culture was 4 h into the stationary phase at this stage.
(ii) Heat challenge.
Centrifuged cell pellets were resuspended in NB to give a volume of 1.5 ml and allowed to stand for 15 min at 37°C. One milliliter of the resulting concentrate was added to 40 ml of NB kept at the investigation temperature (by using a water bath) in a plugged 100-ml flask. The flask contents were stirred with a magnetic flea propelled by a custom-made 12-V direct-current submersible stirrer operating at 60 rpm to minimize vortex formation. The external water bath level was maintained at the neck of the flask, and polypropylene spheres were used to reduce evaporation. The temperature was regulated by a Haake model DC-1 circulator-heater (Fisons Scientific Equipment, Loughborough, Leicestershire, United Kingdom). The heating menstruum temperature was measured by using a National Accreditation of Measurement and Sampling (NAMAS)-certified probe and digital indicator (model Pt 100 probe and series 268 indicator; Anville Instruments, Camberley, Surrey, United Kingdom), and the circulator correction factor was set accordingly to give an accuracy of ±0.05°C.
Heat challenge experiments. (i) Cell density variation and death kinetics.
Resuspended pellets obtained from 1.5-liter cultures were diluted 10-fold in NB to give concentrations of 1011 to 107 CFU ml−1. One milliliter of each dilution was heat challenged at 60°C for 5 min. This procedure was replicated five times.
(ii) D and z value assessment over a temperature range.
Cells recovered from 1.5-liter cultures were heat challenged triplicate at temperatures ranging from 60 to 55°C (in 1°C intervals), as well as at 53, 51, and 49°C. D values were calculated for linear regions of the mean survivor curve for the data sets obtained at 60 to 55°C, which allowed us to calculate z (the temperature change that produces 1 log change in D value).
(iii) Regrowth of late survivors and subsequent heat challenge.
Cells were heat challenged at 60°C, and undiluted samples were collected during the later stages of heating (corresponding to a survivor level of <102 CFU ml−1). These samples were pooled, and 1 ml was used to inoculate 1.5 liters of prewarmed (to 37°C) NB. The broth was incubated for 18 h prior to harvesting and heat challenge at 60°C.
(iv) Reinoculation of a heating menstruum.
Cells were collected from a 100-ml NB culture and heat challenged at 55°C. Once this was done, a second similarly prepared cell concentrate was introduced into the same heating menstruum, and the preparation was sampled like the first culture.
(v) Protein synthesis inhibition during heat challenge.
Cells harvested from 1.5-liter 18-h NB cultures were resuspended in NB, treated with chloramphenicol (Sigma Chemical Company Ltd., Poole, Dorset, United Kingdom) at concentrations of 5 μg ml−1 (determined to be the MIC) and 100 μg ml−1, and heat challenged at 59°C in 50 ml of NB containing chloramphenicol at the same concentrations. At a chloramphenicol concentration of 100 μg ml−1, the low-level enumeration method was modified to avoid carryover of antibiotic. Low-level enumeration was accomplished by adding 1 ml of sampled menstruum to 50 ml of MRD, allowing 30 min for equilibration, and then filtering the preparation through 0.2-μm-pore-size WCN cellulose nitrate membrane filters (Whatman International Ltd., Maidstone, Kent, United Kingdom). The membrane filters were placed on nutrient agar (NA) (Unipath UK Ltd.) for incubation.
To ensure that chloramphenicol remained active at the investigation temperature, 10-ml portions of NB containing chloramphenicol (5 and 100 μg ml−1) were heated at 59°C for 1 h, cooled, and inoculated with 107 CFU of salmonella per ml prior to incubation at 37°C for 18 h.
Heat challenge of chemostat-cultivated cells.
Fermentations were performed in a 1.5-liter vessel with pH control (pH 6.5 ± 0.25), temperature control, (37 ± 1°C), an airflow rate of approximately 1.5 liters min−1, and a 30-liter growth medium reservoir (LH Engineering Co. Ltd., Stoke Poges, Buckinghamshire, United Kingdom). The vessel contents were stirred at 800 rpm, and the medium input was controlled by a calibrated peristaltic pump (Watson-Marlow, Falmouth, Cornwall, United Kingdom).
Carbon was the limiting nutrient in a synthetic basal medium prepared by using the salts solutions described by Evans et al. (13); no specified trace elements were included. A filter-sterilized glucose solution was added to the basal medium after autoclaving to give a final glucose concentration of 0.5 g liter−1.
The fermentor was inoculated to give an initial Salmonella suspension containing approximately 104 CFU ml−1, and the culture was incubated until the late log phase (as determined by measuring the A540). At this time, medium was added at a dilution rate of 0.15 h−1 for 24 h, and steady-state growth was verified by measuring the A540. Approximately 100-ml samples were removed in triplicate for heat challenge. The heat resistance of cells at 60°C was determined for the following dilution rates: 0.15, 0.35, 0.6, and 0 h−1. The medium input was increased by 0.5 h−1 between dilution rates to maintain a smooth transition in the growth rate. At a dilution rate of 0.6 h−1 the absorbance decreased marginally over a 3-h period, and so this dilution rate was considered the approximate μmax. Following this final dilution rate experiment, the medium input was stopped, and the culture was incubated for 12 h prior to the removal of zero-dilution samples.
Chemostat samples were centrifuged like the NB batch cultures, and the pellets were resuspended in NB prior to heat challenge.
Recovery and enumeration procedure.
Heating menstruum samples were pipetted from the heat challenge flask periodically. When necessary, 1-ml volumes were added directly to MRD and further diluted 10-fold before 10 μl was spread plated onto NA. Neat samples were dispensed into glass universals (at room temperature) to cool. Depending on the sensitivity required, samples were surface plated onto NA (10- or 100-μl spread plates) or used to produce 1- or 2.5-ml pour plates. All plates were prepared in duplicate and incubated aerobically at 37°C for 48 h.
To enumerate time-zero populations, representative samples of concentrates were diluted and plated appropriately prior to heating.
Statistical analysis.
Replicate data sets were plotted to obtain mean survivor curves together with standard deviations. Linear regression lines were fitted together with correlation coefficients (r2) to gauge degrees of linearity in survivor curves.
Inhibition of protein synthesis by chloramphenicol.
A radiolabeled solution of l-leucine was prepared by using 5 mM unlabeled leucine and 90 μCi of [3,4,5-3H(N)]leucine (Sigma Chemical Company Ltd.). A 100-ml 18-h salmonella culture was centrifuged, and the cells were resuspended in 100 ml of fresh medium, from which three 10-ml volumes were removed. Two of these samples were treated with chloramphenicol concentrations of 5 and 100 μg ml−1. All of the cultures were incubated for an additional 15 min before 100 μl of the leucine solution was added. After this addition, 1-ml portions were immediately removed from each culture and added to 5 ml of iced 10% (wt/vol) trichloroacetic acid (TCA) (Sigma Chemical Company Ltd.). Additional 1-ml samples were removed at 10, 20, 30, and 60 min. After 30 min of chilling in TCA, each sample was filtered through 0.2-μm-pore-size WCN cellulose nitrate membrane filters (Whatman International Ltd.) and washed three times with 5 ml of 5% (wt/vol) TCA. The membrane filters were subsequently dried and placed in plastic scintillation vials. Five milliliters of OptiPhase Safe scintillation fluid (Wallac Scintillation Products, Wallac UK, Milton Kenyes, United Kingdom) was added to each vial, and the radioactivity levels were recorded for 60 s by using a model 1410 scintillation counter (Wallac UK).
RESULTS
Heating system.
Our initial investigations were centered on reproducible techniques to achieve survivor curves that were, as far as could be determined, free from methodological artifacts that influenced their shape. Mixing of the inoculated heating menstruum was considered necessary to limit temperature differences throughout the liquid. An initial setup in which a single-plane shaking bath was used was shown to produce tailing at 55°C with negligible death at 2 to 3 log10 survivors. This tailing was attributed to the heating menstruum traveling up the sides of the vessels to a lower-temperature region and depositing cells. This allowed the cells to survive and thus allowed recontamination. To avoid this, a low-speed, submersible, magnetic stirrer that did not produce a visible vortex was used. The use of this stirrer also eliminated the need for complicated top drive stirrer systems, which hindered the removal of samples.
Cooling and resuscitation of sampled cells were also investigated. Ice cooling is often employed (4, 11, 27), as is incorporation of a resuscitation stage prior to dilution and plating (8). Death may be enhanced by cold shock when heat-injured cells are artificially cooled by chilling. A comparison of survivor curves produced by this method and one curve obtained when samples were dispensed into vessels at room temperature revealed no differences in the survival rate. Similarly, the addition of a resuscitation step in the heating procedure had no effect on the outcome of survivor curves compared to direct dilution and plating. For reasons of simplicity, in the heating protocol used in this study we employed room temperature cooling and direct dilution and plating.
Cell density variation and death kinetics.
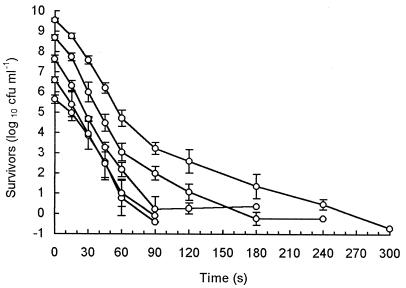
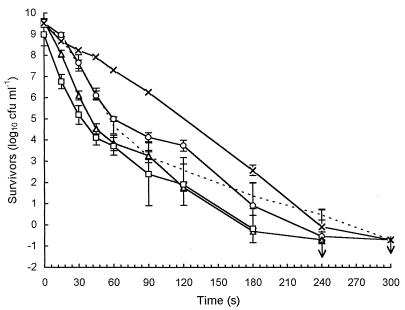
Cell populations of less than 107 CFU ml−1 resulted in virtually linear survivor curves (Fig. 1). Larger populations showed biphasic inactivation, with two distinguishable linear regions. Extrapolation of the secondary death rate, or tail region, back to time zero indicated that the more heat-resistant individuals represented 1 in 104 to 105 cells in the total initial population. High initial cell numbers and sensitive detection methods were therefore required before significant reproducible tailing was seen (Fig. 1).
FIG. 1.
Log-linear plot of survivors at 60°C for 10-fold dilutions of a prepared population. Each curve represents the mean from five replicates, and standard deviations are indicated.
D and z value assessment over a temperature range.
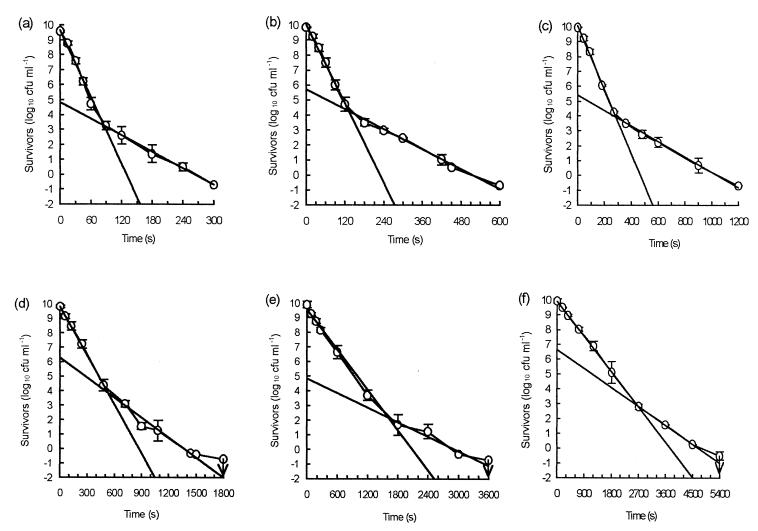
Measured inactivation of high-density populations at temperatures between 60 and 55°C at 1°C intervals gave rise to tailed survivor curves in all cases (Fig. 2). Over the whole temperature range, two distinct linear inactivation rates were detected, and D values were assigned to these rates (Table 1). In each case, the more resistant survivors were a relatively constant proportion of the total population, approximately 1 in 104 to 105 cells. When analyzed by using complete survivor curves, the death kinetics revealed a trend toward greater linearity as the temperature decreased; the correlation coefficients ranged from 0.8815 at 60°C to 0.9743 at 55°C (Table 1).
FIG. 2.
Mean numbers of survivors and standard deviations for 60°C (a), 59°C (b), 58°C (c), 57°C (d), 56°C (e), and 55°C (f), showing the best-fit lines for initial and tailing populations. The arrows indicate the limits of detection.
TABLE 1.
Calculated D values for linear and tail regions of the survivor curves shown in Fig. 2 and the corresponding correlation coefficients for best fits of linear portions, tail portions, and complete death kinetics
| Temp (°C)a | Linear D value (min)b | Linear D r2 value | Tail D value (min)c | Tail D r2 value | Complete death kinetics r2 value |
|---|---|---|---|---|---|
| 60 | 0.22 | 0.9863 | 0.91 | 0.9970 | 0.8815 |
| 59 | 0.38 | 0.9951 | 1.49 | 0.9870 | 0.8990 |
| 58 | 0.77 | 0.9966 | 3.18 | 0.9935 | 0.8690 |
| 57 | 1.47 | 0.9994 | 3.51 | 0.9715 | 0.9306 |
| 56 | 3.62 | 0.9920 | 9.14 | 0.9083 | 0.9418 |
| 55 | 6.28 | 0.9994 | 11.76 | 0.9995 | 0.9743 |
Temperature ± 0.05°C.
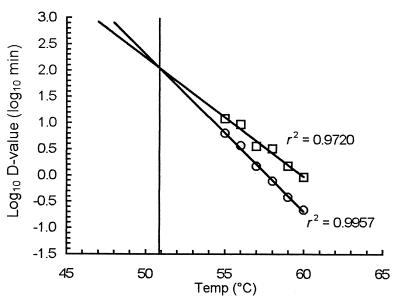
The z value for the linear D data is 3.32 °C (r2 = 0.9957).
The z value for the tail D data is 4.27 °C (r2 = 0.9720).
z values for the two death rates were derived from plots of the log10 D values versus temperature (Fig. 3). The initial linear inactivation gave a z of 3.32°C, and the tailing kinetics produced a z of 4.27°C (Table 1). Extrapolation of the regression fits to lower temperatures revealed convergence at approximately 51°C, indicating that survivor curves at temperatures above 51°C should be tailed and survivor curves at temperatures below 51°C should be linear. Survivor plots for 53, 51, and 49°C confirmed this. Survival at 53°C displayed tailing, although at a reduced level compared with higher temperatures; inactivation at 51 and 49°C resulted in an initial shoulder, which was longer at the lower temperature, followed by linear death without tailing (Fig. 4).
FIG. 3.
Z plots for initial linear inactivation (○) and tailing inactivation (□) over a 5°C temperature range at 1°C intervals. The lines of best fit converge at approximately 51°C; survivors at this temperature should exhibit no tailing.
FIG. 4.
Survivor curves for cultures grown at 53°C (a), 51°C (b), and 49°C (c), showing that there was no tailing at or below 51°C, the temperature at which Z plots for the tailing and nontailing populations converge. The arrow indicates the limit of detection.
Investigations into the causes of tailing.
Cells recovered from a 60°C heat-challenged preparation, cultured, and subjected to a second heating displayed no significant difference in death kinetics compared with the mean data obtained at 60°C. Freshly prepared cells introduced into a previously inoculated and enumerated heating menstruum kept at 55°C gave almost identical death kinetics, and similar levels of tailing were observed. The inclusion of chloramphenicol in the heating menstruum prior to and during heating at 59°C resulted in virtually identical initial linear death rates for treated and untreated cultures at both chloramphenicol concentrations used, 5 and 100 μg ml−1. Tailing in chloramphenicol-treated populations occurred at a lower level. In the culture treated with 5 μg ml−1, the tail survivors accounted for about 1 in 105 cells in the initial population, a value which was approximately 90% less than the value obtained with untreated cells. In cultures treated with 100 μg ml−1, the level was reduced 10-fold further, but tailing was still present. The calculated tail D values for each data set revealed an increase in heat sensitivity with increasing chloramphenicol concentration (Table 2). In untreated cultures, the tailing cells were nearly four times as resistant as the majority of the cells; in the presence of 5 and 100 μg of chloramphenicol per ml this value decreased to 3 and 2.5, respectively. Chloramphenicol at either concentration was shown to be inhibitory to the growth of salmonellae after heating at 59°C.
TABLE 2.
D values determined at 59°C in the presence and absence of chloramphenicol: comparison of initial and tail inactivation rates
| Chloramphenicol concn (μg ml−1) | Initial D59°C (s)a | Initial D59°Cr2 value | Tail D59°C (s) | Tail D59°Cr2 value |
|---|---|---|---|---|
| 0 | 22.80 | 0.9951 | 89.13 | 0.9870 |
| 5b | 25.47 | 0.9948 | 77.78 | 0.9333 |
| 100 | 25.30 | 0.9933 | 63.76 | 0.9300 |
D59°C, D value at 59°C.
Determined to be the MIC.
Assessment of the efficiency of chloramphenicol to inhibit protein synthesis.
The ability of chloramphenicol to inhibit protein synthesis was calculated as a percentage of the radiolabel incorporated into untreated cells. In the presence of 5 μg of chloramphenicol per ml protein synthesis was reduced by more than 60%; at 100 μg ml−1, protein synthesis was reduced by nearly 80%. Despite a concentration that was 20 times the MIC, protein synthesis could not be completely inhibited.
Heat challenge of chemostat-grown cells.
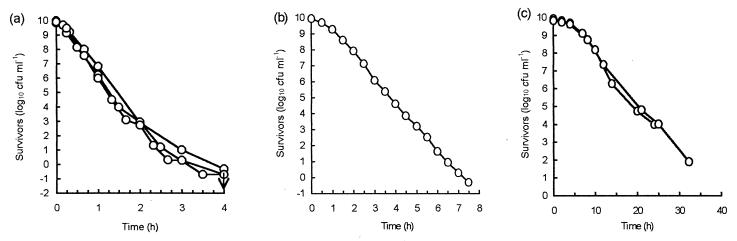
Actively growing chemostat populations growing at a uniform growth rate also produced biphasic death curves, although the heat resistance of the linear majority decreased as the dilution rate (growth rate) increased. The initial linear death rate for cultures with a growth rate of 0.15 h−1 was similar to that of cells cultured batchwise in NB. The tailing proportion was the same for each dilution rate, 1 in 104 to 105 cells of the initial population. Cells allowed to starve (D = 0) exhibited completely linear death with greatly increased heat tolerance (Fig. 5).
FIG. 5.
Survivor curves at 60°C for cells cultured in a chemostat (——) at dilution rates of 0.15 h−1 (○), 0.35 h−1 (▵), and 0.6 h−1 (□). ×, zero dilution; – – –, 18-h NB batch-cultured cells. Standard deviations are indicated for the chemostat culture data. The arrows indicate the limits of detection.
DISCUSSION
At temperatures at which tailing was apparent, the biphasic nature of the survivor curves suggests that two discrete populations were present. A relatively low initial cell density and high limit of detection can hide a tail subpopulation, as demonstrated in Fig. 1. Stumbo (28) highlighted cell density as a potential source of experimental artifacts. Combinations of certain heating media with high cell densities could lead to flocculation during the course of heating which could lead to bias in colony counts and reveal tailing in survivor curves. This was not a factor in this study since wet microscopic mounts of cell inocula and samples collected from heat challenges over the range of temperatures showed that no aggregation occurred.
Survivor curves for salmonellae depicting death over 4 to 5 log10 cycles would, according to data presented here, not display tailing or show sporadic deviations only at the very ends of the survivor curves (10, 17). Minor deviations such as these have previously been attributed to errors associated with low-level survivor enumeration. For this reason, Cerf (6) concluded that workers should have no confidence in data corresponding to <102 survivors ml−1 unless experiments were replicated repeatedly. Replication and the development of a simple low-level enumeration procedure were considered essential elements of the approach adopted in this study.
Methods for determining thermal inactivation kinetics can be divided into two categories, each having its relative merits (28). Closed sample methods require periodic sampling of one or several sample vessels, so each sample must be considered a separate population. Small volumes have to be employed to minimize problems with heating and cooling lags, which creates difficulties when low-level enumeration is desirable. With open systems, these restrictions are eliminated, but new ones are introduced, the most crucial of which is inoculation of the heating menstruum and successive sampling.
The flask protocol employed here cannot be performed with a completely submerged unit. Consequently, there is a possibility that contamination of the flask neck and plug may occur during inoculation and sampling. These regions are at a significantly lower temperature than the heating menstruum and could serve as a possible source for underheated contaminants to reenter the system. Great care therefore must be taken to minimize the risk of neck contamination via aerosol creation. Survival of contaminants in this manner should be greatest at lower temperatures when heat transfer through the neck glass is at its slowest. Tailing was, however, most pronounced at the higher temperatures, indicating that this was not a problem with the method which we employed.
The tailing reported here was quantifiable by D value analysis. This contrasts with some previously published observations that there was little or no change in tail survival levels after extended periods of heating (15, 26), which were probably indicative of artifactual effects. Donnelly et al. (11) demonstrated the possibility that these effects occurred with Listeria-inoculated milk heated in incompletely submerged capped tubes. Survivor curves for a range of temperatures showed initial linear survivor reductions of 3 to 4 log10, followed by prolonged erratic tailing in which the numbers seldom decreased further and in some cases increased. A second method in which fully submerged vials were used produced linear death. Clearly, the method of heat resistance assessment can have a major influence on the shape of survivor curves obtained, so it is not surprising that the existence of tailing has not been fully accepted yet.
Investigations into biological reasons for the reported tailing demonstrated that a simple one-step mutation event occurring in a small number of cells during growth could not account for the different heat sensitivities of the two populations. This observation agrees with several other previously published reports which suggested that tail survivors are not genetically distinct (5, 26, 29).
One consequence of thermal injury is a loss of cell membrane integrity and leakage of a variety of intracellular constituents (18). The presence of external solutes can protect against this outcome and thus possibly delay the onset of injury and death (2, 19). It follows that initial survivors of heating may with time gain additional protection from cellular material released by early casualties, leading to tailing. If this were so, reinoculation of a previously heated culture ought to give rise to greater survival in the second batch. This was not the case, which confirmed the results of Moats et al. (26), who also observed no difference in survival curves for a single heating menstruum inoculated twice.
Sublethal habituation induces the synthesis of heat shock proteins (HSPs) prior to heating and can lead to significantly increased resistance to temperature and many other adverse agents (23). Allan et al. (1) were able to demonstrate HSP induction in Pseudomonas aeruginosa following a temperature shift from 30 to 45°C that led to the synthesis of at least 17 proteins within approximately 1 min. Thus, it is possible that during lethal heating, HSP production occurs in a small proportion of cells and results in tailing of survivor curves. This possibility is supported by the observation that inhibition of de novo protein synthesis during heating dramatically reduced the levels of tailing. The fact that some tailing was still evident is probably due to the failure of chloramphenicol to inhibit protein synthesis completely.
The fact that the tails represented a constant proportion of the initial population suggests that only this proportion was already protected by HSPs or capable of exhibiting a rapid heat shock response (HSR). The requirement for de novo protein synthesis indicates that if cells were already protected by HSPs, these proteins were subject to rapid turnover. As the temperature decreased, the heat resistance of the bulk of the population approached that of the tail, suggesting that the majority of the cells were able to mount an increasingly effective HSR at a reduced temperature, ultimately resulting in completely log-linear death kinetics at 51°C. The introduction of a shoulder deviation at and below this temperature suggests that the inactivation of HSP-protected cells may be the result of injury accumulation.
Culture age has been reported to affect both heat resistance and death kinetics (12, 22, 30). Survivor curves obtained for chemostat-cultured cells confirmed the effect of growth rate on heat sensitivity, but tailing similar to that observed in batch cultures was still apparent at all growth rates. This observation precludes the possibility that differential growth rates in batch cultures could account for the tailing observed. Starved cells produced by halting the medium flow displayed linearity and increased heat resistance at 60°C. This can be explained by the induction of the starvation stress response (SSR), which allows bacteria to remain viable during conditions unsatisfactory for growth (21). However, at 60°C the D value of starved cells was one-half that of tailing cells (at 0.42 min), whether the cells were batch or chemostat derived. Clearly, the induced SSR of such cells, while providing elevated resistance to a variety of stresses, does not lead to a state of great thermotolerance. This is not surprising because the HSR and SSR rely on different sigma factors for induction (ςs and ς32, respectively [14, 21]) and SSR cells should be incapable of initiating rapid HSP synthesis due to their state of “hibernation.”
Our results indicate that the assumption that log-linear thermal death kinetics occur, which generally is made in food processing, is not always valid and that the deviations sometimes observed in practice should not always be dismissed as experimental artifacts. In current practice, margins of safety are generally sufficient for low-level tailing and there is not a problem, but this could change with the increasing trend toward minimal processing.
ACKNOWLEDGMENT
We gratefully acknowledge the support of a United Kingdom Ministry of Agriculture, Fisheries and Food cooperative studentship to L.H.
REFERENCES
- 1.Allan B, Linseman M, MacDonald L A, Lam J S, Kropinski A M. Heat shock response of Pseudomonas aeruginosa. J Bacteriol. 1988;170:3668–3674. doi: 10.1128/jb.170.8.3668-3674.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Allwood M C, Russell A D. Mechanism of thermal injury in Staphylococcus aureus. I. Relationship between viability and leakage. Appl Microbiol. 1967;15:1266–1269. doi: 10.1128/am.15.6.1266-1269.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Anderson W A, McClure P J, Baird-Parker A C, Cole M B. Thermal inactivation of Listeria monocytogenes studied by differential scanning calorimetry. J Gen Microbiol. 1991;137:1419–1424. doi: 10.1099/00221287-137-6-1419. [DOI] [PubMed] [Google Scholar]
- 4.Baird-Parker A C, Boothroyd M, Jones E. The effect of water activity on the heat resistance of heat sensitive and heat resistant strains of salmonellae. J Appl Bacteriol. 1970;33:515–522. doi: 10.1111/j.1365-2672.1970.tb02228.x. [DOI] [PubMed] [Google Scholar]
- 5.Buchanan R L, Golden M H, Whiting R C, Phillips J G, Smith J L. Non-thermal inactivation models for Listeria monocytogenes. J Food Sci. 1994;59:179–188. [Google Scholar]
- 6.Cerf O. Tailing of survival curves in bacterial spores. J Appl Bacteriol. 1977;42:1–19. doi: 10.1111/j.1365-2672.1977.tb00665.x. [DOI] [PubMed] [Google Scholar]
- 7.Charm S E. The kinetics of bacterial inactivation by heat. Food Technol. 1958;12:4–8. [Google Scholar]
- 8.Cole M B, Davies K W, Munro G, Holyoak C D, Kilsby D C. A vitalistic model to describe the thermal inactivation of Listeria monocytogenes. J Ind Microbiol. 1993;12:232–239. [Google Scholar]
- 9.Condón S, Garcia M L, Sala F J. Effect of culture age pre-incubation at low temperature and pH on the thermal resistance of Aeromonas hydrophila. J Appl Bacteriol. 1992;72:322–326. doi: 10.1111/j.1365-2672.1992.tb01842.x. [DOI] [PubMed] [Google Scholar]
- 10.Corry J E L, Barnes E M. The heat resistance of salmonellae in egg albumen. Br Poult Sci. 1968;9:253–260. doi: 10.1080/00071666808415716. [DOI] [PubMed] [Google Scholar]
- 11.Donnelly C W, Briggs E H, Donnelly L S. Comparison of heat resistance of Listeria monocytogenes in milk as determined by two methods. J Food Prot. 1987;50:14–17. doi: 10.4315/0362-028X-50.1.14. [DOI] [PubMed] [Google Scholar]
- 12.Elliker P R, Frazier W C. Influence of time and temperature of incubation on the heat resistance of Escherichia coli. J Bacteriol. 1938;36:83–98. doi: 10.1128/jb.36.1.83-98.1938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Evans C G T, Herbert D, Tempest D W. The continuous cultivation of micro-organisms. 2. Construction of a chemostat. Methods Microbiol. 1970;2:277–327. [Google Scholar]
- 14.Foster J W, Spector M P. How Salmonella survive against the odds. Annu Rev Microbiol. 1995;49:145–175. doi: 10.1146/annurev.mi.49.100195.001045. [DOI] [PubMed] [Google Scholar]
- 15.Fujikawa H, Itoh T. Tailing of thermal inactivation curves of Aspergillus niger spores. Appl Environ Microbiol. 1996;62:3745–3749. doi: 10.1128/aem.62.10.3745-3749.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gould G W. Heat-induced injury and inactivation. In: Gould G W, editor. Mechanisms of action of food preservation procedures. London, United Kingdom: Elsevier Applied Science; 1989. pp. 11–42. [Google Scholar]
- 17.Humphrey T J, Richardson N P, Gawler A H L, Allen M J. Heat resistance of Salmonella enteritidis PT4: the influence of prior exposure to alkaline conditions. Lett Appl Microbiol. 1991;12:258–260. [Google Scholar]
- 18.Hurst A. Bacterial injury: a review. Can J Microbiol. 1977;23:935–944. doi: 10.1139/m77-139. [DOI] [PubMed] [Google Scholar]
- 19.Hurst A, Hughes A. Stability of ribosomes of Staphylococcus aureus S6 sublethally heated in different buffers. J Bacteriol. 1978;133:564–568. doi: 10.1128/jb.133.2.564-568.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Katzin L I, Sandholzer L A, Strong M E. Application of the decimal reduction time principle to a study of the resistance of coliform bacteria to pasteurization. J Bacteriol. 1943;45:265–272. doi: 10.1128/jb.45.3.265-272.1943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kolter R, Siegele D A, Tormo A. The stationary phase of the bacterial life cycle. Annu Rev Microbiol. 1993;47:855–874. doi: 10.1146/annurev.mi.47.100193.004231. [DOI] [PubMed] [Google Scholar]
- 22.Lemcke R M, White H R. The heat resistance of Escherichia coli cells from cultures of different ages. J Appl Bacteriol. 1959;22:193–201. [Google Scholar]
- 23.Lindquist S. The heat-shock response. Annu Rev Biochem. 1986;55:1151–1191. doi: 10.1146/annurev.bi.55.070186.005443. [DOI] [PubMed] [Google Scholar]
- 24.Mackey B M, Miles C A, Seymour D A, Parsons S E. Thermal denaturation and loss of viability in Escherichia coli and Bacillus stearothermophilus. Lett Appl Microbiol. 1991;16:56–58. [Google Scholar]
- 25.Madsen T, Nyman M. Zur theorie der desinfektion. Z Hyg Infektionskr. 1907;57:388–404. [Google Scholar]
- 26.Moats W A, Dabbah R, Edwards V M. Interpretation of nonlogarithmic survivor curves of heated bacteria. J Food Sci. 1971;36:523–526. [Google Scholar]
- 27.Ng H, Bayne H G, Garibaldi J A. Heat resistance of Salmonella: the uniqueness of Salmonella senftenberg 775W. Appl Microbiol. 1969;17:78–82. doi: 10.1128/am.17.1.78-82.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Stumbo C R. Thermobacteriology in food processing. 2nd ed. New York, N.Y: Academic Press; 1973. [Google Scholar]
- 29.Vas K, Proszt G. Observations on the heat destruction of spores of Bacillus cereus. J Appl Bacteriol. 1957;21:431–441. [Google Scholar]
- 30.White H R. The heat resistance of Streptococcus faecalis. J Gen Microbiol. 1953;8:27–37. doi: 10.1099/00221287-8-1-27. [DOI] [PubMed] [Google Scholar]
- 31.Withell E R. The significance of the variation in shape of the time-survivor curves. J Hyg. 1942;42:124–132. doi: 10.1017/s0022172400035361. [DOI] [PMC free article] [PubMed] [Google Scholar]