Gallium-68-labelled somatostatin analogues are a gold standard of neuroendocrine tumors (NETs) PET imaging being a tool for personalized treatment. However, in some cases of low somatostatin receptor (SSTR) expressing tumors, its clinical value can be limited. SSTR antagonists in comparison to currently used agonistic analogues recognize more binding sites on NET cells [1], which may improve the diagnostic efficacy enabling more precise staging leading to the better outcome.
SPECT radiopharmaceuticals represent the cornerstone of molecular imaging due to their wide availability [2] and the development of a radiopharmaceutical based on technetium-99 m-labeled SSTR antagonist would improve access to such clinically feasible diagnostic tool.
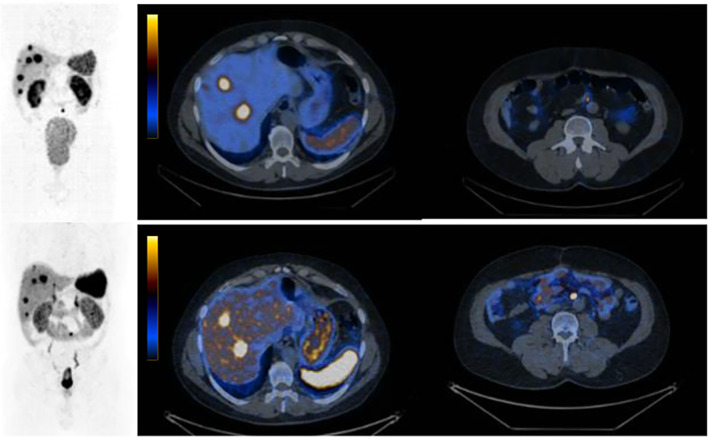
Below (Fig. 1), we present a comparison of SPECT/CT with a SSTR antagonist [99mTc]Tc-TECANT1 (N4-p-Cl-Phe-cyclo(D-Cys-Tyr-D-Aph(Cbm)-Lys-Thr-Cys)-D-Tyr-NH2, where D-Aph(Cbm): D-4-amino-carbamoyl-phenylalanine) [3] and [68 Ga]Ga-DOTA-TATE PET/CT (EudraCT no 2019–003379-20). Please note better visualization of the lesions seen in the study with [99mTc]Tc-TECANT1 (in 6 out of 7 lesions) as well as significantly higher tumor-to-background ratio (in primary and metastatic lesions) obtained with the novel tracer (measured as an absolute number of counts in lesion to background (TBR)) in comparison to [68 Ga]Ga-DOTA-TATE PET/CT (4,07 (range 1,36–7,58) vs 2,26 (range 1,15–3,39)).
Fig. 1 Maximum intensity projection (MIP) and axial fused images: upper line -[99mTc]Tc-TECANT1 SPECT/CT (4 h post injection, injected activity 785 MBq, 120 frames, 20 s per frame); bottom line — [68 Ga]Ga-DOTA-TATE PET/CT (1 h post injection, injected activity 146 MBq; 3 min per bed). Scans were obtained within 13 days. Long-acting somatostatin analogue was withdrawn 4 weeks before imaging
Although PET is considered more sensitive than SPECT [4], the presented new radiopharmaceutical holds promise to provide higher TBR values compared to the current gold standard 68 Ga-DOTA-TATE PET/CT. In combination with the development of quantitative SPECT imaging of NETs, the use of 99mTc-labelled SSTR antagonists could provide a widely available, clinically significant step in the personalized management of NETs.
Funding
This research is a part of the project “Novel 99mTc-labelled somatostatin receptor antagonists in the diagnostic algorithm of neuroendocrine neoplasms — a feasibility study” (TECANT), funded by ERA PerMed (ERAPERMED2018-125), including Austrian Science Fund FWF (project no I 4220-B) to Medical University Innsbruck, National Centre for Research and Development (Poland) contracts no ERAPerMed/01/2019 to Jagiellonian University Medical College and no ERA-PerMed/02/2019 to National Centre for Nuclear Research, MIZS (Ministry of Education, Science and Sport) project no C3330- 19–522011 to University Ljubliana and University Medical Centre Ljubljana.
Data Availability
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding author.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Fani M, Nicolas GP, Wild D. Somatostatin receptor antagonists for imaging and therapy. J Nucl Med. 2017;58(2):61S–66S. doi: 10.2967/jnumed.116.186783. [DOI] [PubMed] [Google Scholar]
- 2.OECD/NEA The supply of medical isotopes: an economic diagnosis and possible solutions. OECD Publishing, Paris, 2019 doi: 10.1787/9b326195-en. [DOI] [Google Scholar]
- 3.Novak D, Janota B, Hörmann AA, Sawicka A, Kroselj M, Hubalewska-Dydejczyk A, Fani M, Mikolajczak R, Kolenc P, Decristoforo C, Garnuszek P. Development of the 99mTc-labelled SST2 antagonist TECANT-1 for a first-in-man multicentre clinical study. Pharmaceutics. 2023;15(3):885. doi: 10.3390/pharmaceutics15030885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alqahtani FF. SPECT/CT and PET/CT, related radiopharmaceuticals, and areas of application and comparison. Saudi Pharm J. 2023;31(2):312–328. doi: 10.1016/j.jsps.2022.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding author.