Abstract
Introduction
Psoriasis is a systemic immune-mediated disease primarily manifesting as skin redness and inflammation. Balneotherapy proved to be a successful non-pharmacological option to reduce the skin areas affected by the disease, but the specific mechanisms underlying this effect have not been elucidated yet. Here we test the hypothesis that the effect of thermal treatments on psoriatic lesions could be partially mediated by changes in the resident microbial population, i.e., the microbiome.
Methods
In this study, we enrolled patients with psoriasis and monitored changes in their skin and gut microbiome after a 12-bath balneotherapy course with a combination of 16S rRNA amplicon sequencing and metagenomics. Changes in the resident microbiome were then correlated with thermal therapy outcomes evaluated as changes in Psoriasis Area and Severity Index (PASI) and Body Surface Area index (BSA).
Results
The amplicon sequencing analysis of the skin microbiome showed that after thermal treatment the microbiome composition of affected areas improved to approach that typical of unaffected skin. We moreover identified some low-abundance bacterial biomarkers indicative of disease status and treatment efficacy, and we showed via metagenomic sequencing that thermal treatments and thermal water drinking affect the fecal microbiome to host more species associated with favorable metabolic health.
Conclusions
Changes in lower-abundance microbial taxa presence and abundance could be the basis for the positive effect of thermal water treatment and drinking on the cutaneous and systemic symptomatology of psoriasis.
Supplementary Information
The online version contains supplementary material available at 10.1007/s13555-023-01036-5.
Keywords: Psoriasis, Thermal therapy, Balneotherapy, Skin microbiome, Gut microbiome, Bacterial biomarkers, Thermal water drinking
Plain Language Summary
Psoriasis is an immune-mediated disease primarily manifesting as skin redness and inflammation that affects 2–3% of the world’s population. No cure is currently available for this condition, and patients are offered pharmacological and non-pharmacological options to alleviate the discomfort. Previous studies and clinical practice have shown that thermal water treatment can be a non-pharmacological option to reduce the areas affected by the disease. However, the specific mechanisms causing this reduction have not been clarified yet. Given that neither the chemical nor the physical composition of thermal water can explain this beneficial effect, recent studies have suggested that it might be due to the effect of thermal water on the microbial communities living on the skin (i.e., the skin microbiome).
In this work carried out at Terme di Comano, Northern Italy, we describe the effect of thermal water treatment on the skin microbiome of patients with psoriasis and we highlight the potentially beneficial effect of thermal water drinking on the microbial communities living in the gut, namely the gut microbiome. Specifically, we show that after balneotherapy the areas affected by psoriasis have a higher diversity of microbes usually present on healthy skin, potentially explaining the reduction in disease severity after treatment, and we describe how the gut microbiome of patients who drank thermal water changes to host more species linked with favorable metabolic health. These findings highlight that thermal water treatment and drinking could reduce both the skin and systemic symptomatology of psoriasis by affecting the skin and gut microbiome.
Supplementary Information
The online version contains supplementary material available at 10.1007/s13555-023-01036-5.
Key Summary Points
| Psoriasis cutaneous manifestations lack a definitive cure but can be effectively reduced by balneotherapy, a non-pharmacological treatment whose mode of action has not been elucidated yet. |
| As thermal water hosts unique microbial communities whose lysates exert a positive effect on human skin fibroblast proliferation in vitro, here we postulated that this effect might be mediated by the changes induced by balneotherapy in the composition and structure of resident microbial communities, i.e., the microbiome. |
| Here we show that balneotherapy changes the affected skin microbiome composition to host more species associated with healthy skin, and that after thermal treatment the fecal microbiome of patients with psoriasis hosts more microbial species previously associated with favorable metabolic health. |
| Changes in lower-abundance microbial taxa presence and abundance could be the basis for the positive effect of thermal water treatment and drinking on the cutaneous and systemic symptomatology of psoriasis. |
| More mechanistic approaches are needed to understand how changes in the resident skin and gut microbiome affect psoriatic symptoms after thermal water treatment and drinking. |
Introduction
Psoriasis is a chronic and systemic immune-mediated condition primarily affecting the skin [1]. It presents with red, itchy, and dry skin patches mostly located on the elbows, knees, and scalp and can cover even the full body of the affected person [1–4]. Psoriasis affects 2–3% of the world’s population, with typical onset during adulthood [2–5]. As for other immune-mediated diseases, the onset of psoriasis is thought to be due to a combination of both genetic and environmental factors, but also to predisposing comorbidities (i.e., metabolic syndrome) [5, 6]. To date, there is no definitive cure available [6]. Current treatment options include ointments and moisturizing creams to prevent itchiness, topical corticosteroids, vitamin D analogues, calcineurin inhibitors, phototherapy, keratolytics, and biological monoclonal antibodies (especially in the most severe cases of psoriatic arthritis) [3, 7–17]. Short-term balneotherapy with thermal spring water constitutes a valid and proven adjuvant treatment of psoriatic lesions [18]. However, the mechanisms underlying the beneficial effect of thermal water on psoriatic lesions are not fully elucidated.
Previous works investigated how tap and thermal water differ at the chemical and physical levels, but could not find any difference that could explain the effect of thermal water on psoriatic skin [19]. Nevertheless, tap water is treated to reduce the microbial load [20], whereas spring water is not. A previous study showed that Comano spring water harbors a distinct environmental microbiota [21], and lysates obtained from these communities exert positive effects when tested in vitro [22]. This might suggest that the reduction in psoriasis severity after balneotherapy could be due to cross talk between microbes or microbial molecules present in the water used for the thermal treatments and the skin microbiome (i.e., the bacteria, fungi, and viruses living on the human skin [23, 24]) of patients with psoriasis. It is indeed also known that psoriasis has a microbial component and that infections may cause disease exacerbation [6]. The skin microbiome also plays a role in educating the immune responses of the host and restricting or inhibiting pathogen colonization [25–30], which are both parts of the etiology of psoriasis [31–34]. Additionally, the composition of the skin microbiome and the presence and abundance of a number of microbial species is tightly linked with some inflammatory and immune-mediated conditions that affect the skin, including psoriasis [35–37].
In this study, we aimed to elucidate the effect of thermal water treatments on psoriatic skin by highlighting changes in the skin microbiome associated with a 12-bath treatment and changes in disease severity as assessed by the Psoriasis Area and Severity Index (PASI) and Body Surface Area index (BSA) [38]. Additionally, we tested the effect of the combination of thermal treatment and thermal water drinking on the fecal microbiome composition and more specifically with the dynamics of species previously associated with metabolic health [39].
Methods
Patient Cohort Recruitment
This study was performed under the approval of the Ethical Committee of the University of Trento and all 57 participants gave their informed consent before clinical assessment by a clinically accredited dermatologist. The study was performed in accordance with the Helsinki Declaration of 1964 and its later amendments. Female and male subjects affected by psoriasis were enrolled at Terme di Comano, in Trentino (Italy), provided a medical history, and were assessed for psoriasis severity according to PASI and BSA [38] (Table 1 and Supplementary Material S1). Patients had different manifestations of psoriasis, including plaque psoriasis (N = 34), guttate psoriasis (N = 1), or combinations of plaque and guttate (N = 3), plaque and inverse (N = 2), or plaque, guttate, and inverse psoriasis (N = 3) (Supplementary Material S1). Twelve patients were also affected by psoriatic arthritis (Table 1 and Supplementary Material S1). PASI ranged between 2 and 30 before treatment, and BSA ranged between 1 and 50; after treatment, 67.4% of patients reported an improvement in at least one of the two indexes. The thermal treatment course consisted of 12 baths in thermal water at Terme di Comano. Patients were also recommended to drink approximately 1 L of thermal water per day on an empty stomach.
Table 1.
Clinical and anthropometric characteristics of the selected patient cohort (N = 43)
| Cohort description (N = 43) | Before thermal treatment | After thermal treatment |
|---|---|---|
| Age | 58 ± 11 (30–77)a | |
| BMI | 28 ± 5 (19.4–52.1) | |
| Gender (F/M), n | 17/26 | |
| Psoriatic arthritis (yes/no), n | 12/31 | |
| PASI | 6.5 ± 5 (2–30) | 5 ± 5 (1–30)a |
| BSA | 18.6 ± 14.6 (1–50) | 16.6 ± 13.3 (1–50)a |
Continuous variables are presented as mean ± standard deviation (range)
aMissing data (N = 2). More details including therapy are available in Supplementary Material S1
Skin Swabs and Fecal Sample Collection, DNA Extraction, and Sequencing
For each patient, we sampled the skin microbiome non-invasively with sterile swabs on areas with evident psoriatic lesions on the retroauricular area (behind the ear) and on the antecubital fossa (elbow pit). Two other swab samples were collected in matching areas considered healthy or peri-lesion. The same set of samples was collected before and after treatment (12 baths, one per day, 20 min each) at Terme di Comano. A total of eight swabs were therefore obtained for each patient. To minimize collection bias, samples were collected by the same clinically accredited dermatologist as previously validated by the Human Microbiome Project Consortium [40] in a temperature-controlled room after clinical assessment. Samples were collected in different seasons to have a more realistic and representative cohort. Sterile cotton tip swabs (VWR, Milan, Italy) moistened in SCF-1 buffer (50 mM Tris–HCl, pH 7.5; 1 mM EDTA, pH 8.0; 0.5% Tween-20) were swabbed on the skin applying pressure for approximately 30 s. The swab was then scrubbed against the side of 15-ml sterile tubes containing SCF-1 buffer and frozen at − 20 °C.
For DNA extraction, the Qiagen DNeasy PowerSoil Pro kit was used with an initial modification. Briefly, samples were thawed and centrifuged to collect the buffer at the bottom of the 15-ml tube. Each swab was transferred to a clean PowerBead tube and the head of the swab was cut with sterile scissors. Buffer was transferred to sterile 2-ml tubes and centrifuged for 5 min at 10,000g, before discarding half of the supernatant. The pellet was resuspended and transferred to the matching PowerBead tube. Samples were gently mixed before adding 800 μl of CD1 solution and vortexing at maximum speed for 10 min on the dedicated adapter. The swab head was then removed with sterile tweezers and the next DNA extraction steps were performed according to Qiagen DNeasy PowerSoil Pro kit instructions. Extracted DNA was quantified with the Qubit fluorometer.
V1–V3 variable regions of the bacterial 16S rRNA gene were amplified using the 27F-534R primers (27F, AGAGTTTGATCCTGGCTCAG; 534R, ATTACCGCGGCTGCTGG [41, 42]) and sequenced on the Illumina MiSeq platform.
Each patient also provided a fecal sample before and after balneotherapy. Fecal samples were collected in provided sterile containers and immediately frozen at − 20 °C. The fecal DNA was extracted using the Qiagen DNeasy PowerSoil Pro kit according to manufacturer’s instructions. Sequencing libraries were constructed using the NexteraXT DNA Library Preparation Kit (Illumina) and sequenced on the Illumina HiSeq2500 platform with a target depth of 5 Gb/sample.
Taxonomic and Functional Profiling and Statistical Analysis
For skin microbiome samples, the taxonomic profile and relative abundance of all bacterial species of each sample were obtained using QIIME1 [43]. Open reference OTU picking was performed against the SILVA database (version 132, [44]) using UCLUST [45]. Alpha and beta diversity indexes were calculated using the adiv_boxplot and bdiv_boxplot functions from rbiom [46]. Differential taxonomic analysis was performed with MaAsLin2 [47] using default parameters. Genus-level relative abundances were normalized using TSS and used as input for a compound Poisson linear model (CPLM) in which patient IDs were treated as random effects and disease status or timepoints were used as fixed effects.
For fecal samples, we estimated the microbial species profile using the MetaPhlAn 3 software with default parameters [48]. Potential functional profiling of the stool samples was performed using HUMAnN 3 [48]. UniRef90 gene families were collapsed into MetaCyc pathways and abundances normalized using counts per millions (CPM). Statistical analysis was performed using MaAsLin2 using the CPLM with default parameters on both the species and pathway abundance profiles. Patient IDs were used as random effect and timepoint or improvement status (for taxonomic and functional profiles, respectively) as fixed effects. Coefficient estimates obtained by each CPLM were used as effect size and then converted to fold change (FC) with the formula FC = log2(exp(coeff)). Fold change absolute values larger than 1 were considered relevant. P values obtained by MaAsLin2 were subjected to multiple hypothesis testing using the Benjamini–Hochberg procedure with a threshold on q values of 0.1.
Results
To evaluate the impact of thermal treatment on the skin microbiome of patients with psoriasis, we enrolled 57 patients affected by psoriasis at Terme di Comano, in Trentino, Italy. We collected four swab samples from patients affected by psoriatic lesions in at least one of the two retroauricular crease areas (herein the ear) or on one of the two antecubital fossae (herein the elbow). We preferentially selected patients who were discordant for the presence of the psoriatic lesions on the two elbows and ears and collected samples from the peri-lesion when this was not possible (see “Methods” and Supplementary Material S1). We collected paired samples affected/unaffected by the disease from the same patients as skin microbiomes differ greatly across unrelated individuals [24, 49], and comparing same-patient affected/unaffected samples can overcome this limitation. In addition to skin swabs, enrolled patients provided stool samples to test whether balneotherapy and thermal water drinking could affect the gut microbiome as well. Skin swabs and fecal samples were collected both at baseline and after a 12-bath treatment at Terme di Comano. Two patients did not provide samples after treatment and were therefore excluded from further analyses (Supplementary Material S1). In the present study, we analyzed a total of 300 skin samples obtained from 43 patients for whom at least one of the two body sites had all four skin samples (i.e., affected at T1 and T2, unaffected at T1 and T2) retained after DNA extraction, sequencing, and quality control (see “Methods”). We moreover analyzed 90 paired fecal samples obtained from 45 patients who provided them both before and after treatment. Patients considered in this study (17 female and 26 male) had a mean age of 58 ± 11 years and a mean BMI of 28 ± 5 (Table 1 and Supplementary Material S1). Following treatment at Terme di Comano, most of the patients (29/43) reported an improvement in PASI, BSA, or both, while a minority (12/43, missing data N = 2) reported no improvement (P < 0.05, Fisher’s exact test, Supplementary Material S1).
Overall Microbial Diversity is Conserved Between Affected and Unaffected Skin
To assess the complexity of the microbial communities inhabiting the skin areas affected by psoriasis and to compare it with one of the unaffected areas, we considered the within-sample bacterial diversity (i.e., alpha diversity) computed as species richness (number of observed operational taxonomic units, i.e., OTUs) as well as species diversity (Shannon diversity index). Although alpha diversity is frequently lower in chronic and inflammatory conditions [50], we did not find any consistent statistically significant reduction in bacterial richness and diversity in areas affected by psoriasis (Supplementary Fig. S1A), in line with previous studies on psoriasis [37, 51]. Alpha diversity of diseased skin was similarly not influenced by the 12-bath balneotherapy course (Supplementary Fig. S1A). Likewise, the microbiome composition of the affected and unaffected skin was relatively similar (PERMANOVA P = 0.063, Fig. 1a, Supplementary Fig. S1B, Supplementary Material S2), although the effect size of the differences was limited and the distributions highly overlapping.
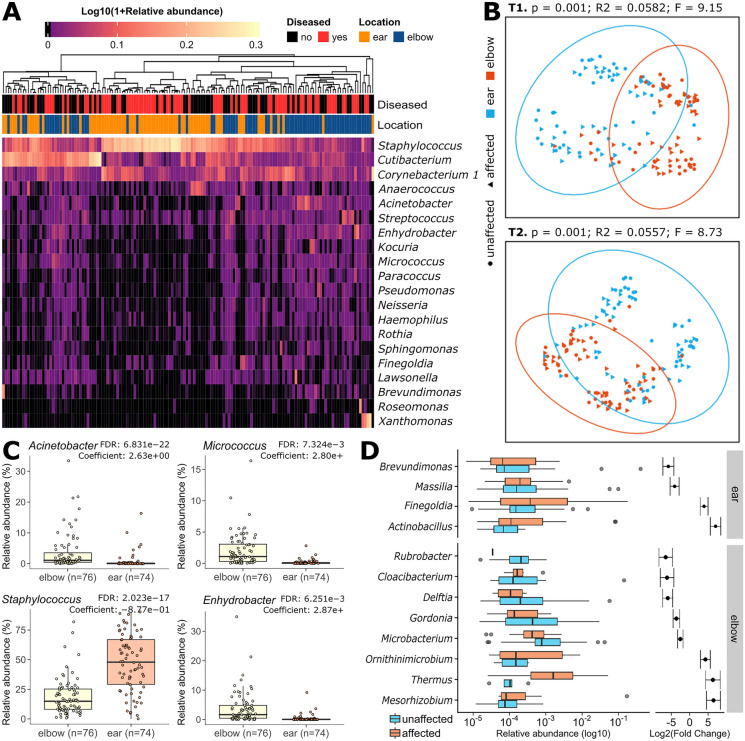
Fig. 1.
Skin microbiome composition according to body site and disease status. a The 20 most abundant bacterial genera on the affected and unaffected skin of patients with psoriasis before balneotherapy. Retroauricular crease (ear) samples are dominated by Staphylococcus, whereas antecubital fossa (elbow) samples are dominated by Corynebacterium 1, Cutibacterium, and Staphylococcus genera. A heatmap with the most abundant OTUs is available in Supplementary Fig. S1B. b Principal component analysis (PCoA) of pairwise Bray–Curtis dissimilarities on taxonomic composition shows a separation between ear and elbow samples both at T1 and T2, but no clear separation between affected and unaffected areas at the same location. PCoAs with all available samples and by disease status are reported in Supplementary Fig. S1C, D. Statistics were performed with Adonis (1000 permutations). c Genera differentially abundant on the ear and elbow skin. d Differentially abundant genera between affected and unaffected sites on the elbows and behind the ears. Differentially abundant genera between before and after balneotherapy are available in Supplementary Fig. S2B
Psoriatic Lesions and Unaffected Skin Harbor the Same Dominant Taxa and Few Lowly Abundant Site-Specific Differential Genera
The most abundant bacterial species found on the skin belonged to the genera Staphylococcus, Cutibacterium, and Corynebacterium (Fig. 1a), which were present at an average relative abundance of 34.1%, 18.2%, and 13.5%, respectively. These three genera were the dominant ones both on the affected and unaffected skin, followed by Anaerococcus, Acinetobacter, Enhydrobacter, and Streptococcus (Fig. 1a and Supplementary Material S2). These genera’s similar prevalence and abundance on affected and unaffected skin are confirmed by the absence of relevant clustering between the two conditions (Supplementary Fig. S1C, D). Microbiome compositional stratification was instead observed by location, with lower abundance taxa differing between ear and elbow skin (Fig. 1b, c and Supplementary Material S3).
More specifically, before balneotherapy, Staphylococcus was significantly enriched on the retroarticular crease (37.5% vs 26.5% on the elbow, FDR-corrected P value = 2.07E−08). Yet, Staphylococcus was consistently higher both on affected and unaffected ear samples in our cohort. Conversely, the genera Micrococcus, Enhydrobacter, Acinetobacter, and many others were associated with the antecubital fossa skin microbiome (FDR-corrected P values < 9.53E−12, Fig. 1c and Supplementary Material S3). Given this difference in the microbial composition of the two skin areas, we performed all further analyses by stratifying for sampling location.
Balneotherapy Restores a Healthy-Like Abundance of Bacterial Taxa Normally Impaired on Psoriatic Lesions on the Elbows
We then looked for differentially abundant taxa between psoriatic and unaffected skin by applying MaAsLin2 [47], a tool used to determine multivariable associations between taxonomic features and phenotypes, environments, exposures, and covariates.
In baseline pre-treatment samples, we identified two genera that were enriched on the diseased skin behind the ear (Finegoldia and Actinobacillus) and three that were enriched on the affected elbow skin (Ornithinimicrobium, Mesorhizobium, and Thermus, Fig. 1d). Two other genera were instead significantly reduced behind the ears affected by psoriasis (Brevundimonas and Massilia) and five were reduced on the affected skin on the elbows (Rubrobacter, Cloacibacterium, Delftia, Gordonia, and Microbacterium, Fig. 1d). Notably, the mean relative abundance of each of these differentially abundant taxa was lower than 1% (Supplementary Material S2).
After a treatment of 12 baths, 10/11 of the genera associated with disease or lack thereof and significantly changing after balneotherapy showed variations toward the abundances of the non-disease condition (Supplementary Fig. S2A). For instance, after balneotherapy, we observed a significant decrease in Ornithinimicrobium, Mesorhizobium, and Thermus, which are the three taxa that were previously associated with disease on the elbows. On the contrary, Delftia, which was previously reduced in areas affected by psoriasis, increased after thermal treatments (Supplementary Fig. S2A and Supplementary Material S3). Other genera previously reduced in psoriatic skin samples, such as Gordonia and Cloacibacterium, increased after balneotherapy, while Microbacterium, previously associated with the diseased skin, decreased, but their trends toward a healthy-like abundance were not significant (Supplementary Fig. S2A and Supplementary Material S3). No genera were significantly differentially abundant before and after balneotherapy on the psoriatic skin behind the ear.
Some Microbial Genera are Associated with Psoriasis Improvement or Lack of Improvement After Balneotherapy
To identify biomarkers of thermal treatment efficacy, we tested whether changes in the relative abundance of some taxa might be associated with an improvement in the condition of patients with psoriasis by correlating them with patients’ outcomes after treatment. Overall, 67.4% of the patients treated with balneotherapy at Terme di Comano reported an improvement in the PASI and/or BSA (i.e., responders to treatment), whereas 27.9% did not report an improvement (i.e., non-responders, missing data N = 2, Supplementary Material S1).
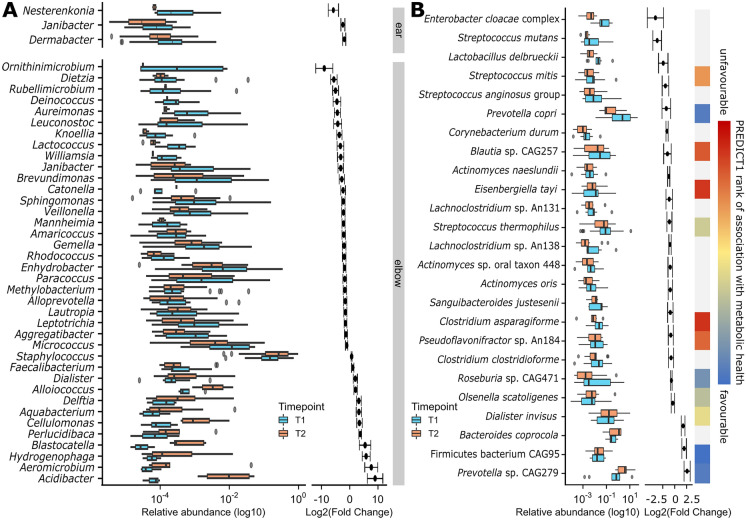
By applying MaAsLin2 [47] (see “Methods”), we found that an increase in Massilia, Parvimonas, Limnobacter, and Klebsiella, and a decrease in Anaerococcus, Micrococcus, Haemophilus, Finegoldia, Aggregatibacter, and other rarer genera was associated with improvement of psoriatic disease behind the ears (Fig. 2a). On the elbows, an increase in Acidibacter, Ezakiella, Erwinia, Delftia, and Staphylococcus and a decrease in Micrococcus, Ornithinimicrobium, Streptococcus, Aggregatibacter, and Enhydrobacter was associated with improvement (Fig. 2a). Other lower abundance taxa were also associated with positive or negative responses to thermal treatments (Supplementary Material S3). Notably, 83.87% of the differential genera were not concordant between responders and non-responders (Supplementary Material S3).
Fig. 2.
The skin and gut microbiomes are positively affected by thermal therapy. a Differentially abundant genera on the skin affected by psoriasis between before (T1) and after (T2) thermal treatment in patients that reported an improvement after balneotherapy. Differentially abundant genera on the unaffected skin of the same patients are available in Supplementary Fig. S2C. b Differentially abundant fecal taxa before (T1) and after (T2) balneotherapy and thermal water drinking. After balneotherapy and thermal water drinking, there is an increase in species associated with favorable metabolic health according to [39] and a decrease in species associated with bad metabolic health. Species lacking a rank of association in the original study are here reported in gray
Further investigation of the correlation between disease amelioration and an increase in Staphylococcus species highlighted an increase in the relative abundance of Staphylococcus aureus on the elbows that was associated with a decreased PASI and/or BSA (coefficient = 0.926, FDR-corrected P value = 7.39E−05, Supplementary Material S3). Conversely, psoriatic skin manifestations improvements were associated with the decrease of Staphylococcus saprophyticus (coefficient = − 1.34, FDR-corrected P value = 0.0017, Supplementary Material S3).
Fecal Microbiome Changes with Balneotherapy to Host More Species Associated with Healthy Metabolism
To better understand the overall effect of balneotherapy and thermal water drinking (see “Methods”) on the host microbiome, we compared the microbial composition of the stool samples obtained before and after treatment at Terme di Comano SpA. Despite no significant change in alpha diversity, we observed a significant reduction in the intra-subject beta diversity after treatment (Supplementary Fig. S2D, E). The analysis of 90 paired fecal samples (Supplementary Material S4) highlighted 25 differentially abundant species in patients with psoriasis before and after balneotherapy. Of these, four of the five species that increased after balneotherapy were previously described as being among the species most associated with favorable metabolic health and healthy diets [39] (Fig. 2b). On the contrary, species with a significantly reduced abundance after treatment included a number of taxa previously associated with bad metabolic health [39] (Fig. 2b). Functional analyses did not highlight significant differences by time point, but overall functional differences were observed by treatment response, with responders having more vitamin B6 and fewer l-methionine biosynthesis pathways than non-responders (Supplementary Material S4).
Discussion
The findings presented in this study shed light on how the skin microbiome may be mediating the effect of thermal therapy on psoriatic skin and systemic manifestations, with particular emphasis on the impact of balneotherapy as a potential therapeutic intervention.
Our study revealed that, despite high interindividual differences in the baseline skin microbiome [24, 49], there is a remarkable conservation of microbial diversity and structure between affected and unaffected sites within the same individual, as the dominance of Staphylococcus, Cutibacterium, and Corynebacterium genera aligns with the usual composition of a healthy human skin microbiome. These genera indeed include the most abundant species usually found on healthy human skin, namely Staphylococcus epidermidis, Cutibacterium acnes, and Corynebacterium tuberculostearicum [52]. Nonetheless, our data showed also an increased relative abundance of Staphylococcus on the retroarticular crease both on affected and unaffected skin, in contrast with previous studies that associated this bacterial genus with moist skin areas such as the elbow [24, 52], suggesting that increased abundance of Staphylococcus could be a characteristic of either the psoriatic disease or of our cohort. Furthermore, the increase in S. aureus and the decrease in S. saprophyticus, both known to cause a number of skin and soft tissue diseases [53–57], being associated with psoriasis improvement raises questions about the potential roles of different Staphylococcus species in skin health and disease.
Furthermore, the observed stratification of microbiome composition by location in line with previous studies [23, 58] underscores the importance of considering regional variations when studying the skin microbiome and its role in cutaneous diseases. Indeed, after stratifying the analysis of the skin microbiome following treatment based on body site, we observed that the abundance of genera associated with disease or lack thereof and significantly altered after balneotherapy showed a trend towards the non-disease condition on the elbows, as for the decreasing Ornithinimicrobium and Mesorhizobium. The former was previously associated with the lesional skin of patients with leprosy in India [46] and the latter with the skin microbiome of older women [47], while both genera are normally present in aquatic and soil environments [48–53] consistent with prolonged exposure to balneotherapy water. The genus Delftia, increased after balneotherapy, is instead known to degrade organic pollutants and includes both environmental and opportunistic pathogenic strains [59–63]. Interestingly, Delftia was previously associated with a reduction of S. epidermidis and a consequent increase in S. aureus on the skin of patients with atopic dermatitis [64], a trend also observed in our cohort. These findings suggest that balneotherapy influences and possibly shifts skin microbiome relative abundances toward a more healthy-like composition.
By further stratifying for therapy response, we moreover identified shifts in microbial composition associated with disease amelioration. Our study revealed that certain genera, such as Finegoldia and Ornithinimicrobium, were enriched on diseased skin at baseline but significantly decreased after balneotherapy in patients who reported psoriasis improvement. Conversely, others like Massilia and Delftia showed potential as “good taxa,” as they were negatively associated with affected skin before treatment and increased after successful balneotherapy. Notably, the consistent negative association of Micrococcus and Aggregatibacter with psoriasis improvement regardless of body site suggests that these genera might serve as biomarkers of a lack of clinical response to treatment. Both genera are known to cause infections, particularly in immunocompromised individuals [65–70], suggesting a potential link between microbiome composition and disease exacerbation. These findings highlight the potential role of specific microbial taxa in psoriasis and suggest that shifts in microbiome composition may contribute to changes in disease severity.
Moreover, our study shed light on the intriguing impact of balneotherapy and thermal water consumption on the gut microbiome in individuals with psoriasis, for which we observed noteworthy trends. While there was no significant alteration in alpha diversity, a significant reduction in intra-subject beta diversity was evident post-treatment, indicating a convergence towards a more stable microbial community structure. Further examination revealed an increase in species associated with favorable metabolic health and healthy diets and a decrease in those associated with poor metabolic health [39]. Particularly striking was the substantial increase in Firmicutes bacterium CAG95, which was previously identified as the species with the highest association with metabolic health [39]. These findings collectively suggest that in addition to its established efficacy in improving psoriatic skin manifestations, balneotherapy merits investigation for its potential to positively influence the gut microbiome and associated systemic inflammation levels, which are intimately connected with cutaneous psoriasis [71].
In conclusion, our study underscores the potential impact of balneotherapy and thermal water drinking on the psoriatic skin and gut microbiome, hinting at the possibility of restoring a healthier microbial composition associated with lesion reduction. The connections between microbiome changes and clinical improvements raise intriguing questions about the role of the skin and gut microbiome in psoriasis pathogenesis and treatment.
Despite most of the differential skin genera not being concordant between responders and non-responders and thus supporting a connection between changes in microbiome composition and changes in psoriasis severity, it is crucial to acknowledge that our study design does not establish a causal relationship between alterations in microbiome composition and changes in psoriasis clinical severity following balneotherapy. However, our findings provide a solid foundation for future, more targeted investigations to elucidate the mechanistic underpinnings of these observations.
Conclusions
In this work, we showed that the perturbation of the cutaneous microbiome of psoriatic lesions compared with that of unaffected or peri-lesion skin of the same patient can be ameliorated by balneotherapy. In our observational study, the 12-bath treatment led to a shift both in the microbial community complexity and taxonomic composition to resemble those of unaffected skin. These changes in the skin microbiome structure and complexity were associated with improvements in the PASI and BSA, leading to the identification of bacterial biomarkers of treatment effectiveness. Additionally, we showed that the gut microbiome composition of treated patients harbors more bacteria associated with favorable metabolic health and less of those associated with unfavorable metabolic health of the host, suggesting that balneotherapy could also ameliorate systemic health parameters associated with psoriasis. Taken together, these results call for further targeted investigations into the effects of thermal treatments on the cutaneous and systemic manifestations of psoriasis, ideally considering less targeted approaches like shotgun metagenomics to explore also the role of and the effect on the cutaneous fungal communities.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
The authors would like to thank all patients enrolled in this work.
Author Contributions
Serena Manara: Data curation (Equal), Formal analysis (Supporting), Investigation (Equal), Methodology (Equal), Project administration (Equal), Writing – original draft (Lead), Writing – review & editing (Equal). Francesco Beghini: Data curation (Equal), Formal analysis (Lead), Methodology (Equal), Project administration (Equal), Visualization (Equal), Writing – original draft (Supporting), Writing – review & editing (Equal). Giulia Masetti: Data curation (Lead), Formal analysis (Supporting), Project administration (Equal), Writing – review & editing (Supporting). Federica Armanini: Data curation (Equal), Project administration (Equal). Davide Geat: Resources (Equal). Giulia Galligioni: Resources (Equal). Nicola Segata: Conceptualization (Lead), Funding acquisition (Equal), Project administration (Equal), Supervision (Lead), Writing – review & editing (Lead). Stefania Farina: Conceptualization (Equal), Resources (Lead), Writing – review & editing (Supporting). Mario Cristofolini: Conceptualization (Equal), Funding acquisition (Lead), Project administration (Equal), Supervision (Equal), Writing – review & editing (Supporting).
Funding
This study was funded by Terme di Comano, TN, Italy, Istituto G. B. Mattei, Stenico, Italy, and the Tourism and Sport Service of the Autonomous Province of Trento (ref. protocol: RT 1_2018), Italy. The Rapid Service Fee was funded by the authors.
Data Availability
Raw sequences were deposited in the NCBI Sequence Read Archive (SRA) under the accession number PRJNA909312.
Declarations
Conflict of Interest
Stefania Farina and Mario Cristofolini are board members of Istituto G.B. Mattei without any Financial interest. Nicola Segata received funding from Istituto G.B. Mattei to conduct the present research. All other authors declare no competing interests.
Ethical Approval
This study was performed under the approval of the Ethical Committee of the University of Trento and all 57 participants gave their informed consent before clinical assessment by a clinically accredited dermatologist. The study was performed in accordance with the Helsinki Declaration of 1964 and its later amendments.
Footnotes
Serena Manara, Francesco Beghini, and Giulia Masetti contributed equally to this work.
Nicola Segata, Stefania Farina, and Mario Cristofolini are co-last authors.
References
- 1.Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020;182:840–848. doi: 10.1111/bjd.18245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lebwohl M. Psoriasis. Lancet. 2003 doi: 10.1016/s0140-6736(03)12954-6. [DOI] [PubMed] [Google Scholar]
- 3.Armstrong AW, Read C. Pathophysiology, clinical presentation, and treatment of psoriasis: a review. JAMA. 2020;323:1945–1960. doi: 10.1001/jama.2020.4006. [DOI] [PubMed] [Google Scholar]
- 4.Walter K. Psoriasis. JAMA. 2022;327:1936. doi: 10.1001/jama.2022.5270. [DOI] [PubMed] [Google Scholar]
- 5.Greb JE, Goldminz AM, Elder JT, et al. Psoriasis. Nat Rev Dis Primers. 2016;2:16082. doi: 10.1038/nrdp.2016.82. [DOI] [PubMed] [Google Scholar]
- 6.Boehncke W-H, Schön MP. Psoriasis. Lancet. 2015;386:983–994. doi: 10.1016/S0140-6736(14)61909-7. [DOI] [PubMed] [Google Scholar]
- 7.Menter A, Korman NJ, Elmets CA, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis. Guidelines of care for the management and treatment of psoriasis with topical therapies. J Am Acad Dermatol. 2009;60:643–659. doi: 10.1016/j.jaad.2008.12.032. [DOI] [PubMed] [Google Scholar]
- 8.Soleymani T, Hung T, Soung J. The role of vitamin D in psoriasis: a review. Int J Dermatol. 2015;54:383–392. doi: 10.1111/ijd.12790. [DOI] [PubMed] [Google Scholar]
- 9.Lebwohl M, Menter A, Weiss J, et al. Calcitriol 3 microg/g ointment in the management of mild to moderate plaque type psoriasis: results from 2 placebo-controlled, multicenter, randomized double-blind, clinical studies. J Drugs Dermatol. 2007;6:428–435. [PubMed] [Google Scholar]
- 10.Darley CR, Cunliffe WJ, Green CM, Hutchinson PE, Klaber MR, Downes N. Safety and efficacy of calcipotriol ointment (Dovonex) in treating children with psoriasis vulgaris. Br J Dermatol. 1996;135:390–393. doi: 10.1046/j.1365-2133.1996.d01-1010.x. [DOI] [PubMed] [Google Scholar]
- 11.Liao YH, Chiu HC, Tseng YS, Tsai TF. Comparison of cutaneous tolerance and efficacy of calcitriol 3 microg g(−1) ointment and tacrolimus 0.3 mg g(−1) ointment in chronic plaque psoriasis involving facial or genitofemoral areas: a double-blind, randomized controlled trial. Br J Dermatol. 2007;157:1005–1012. doi: 10.1111/j.1365-2133.2007.08201.x. [DOI] [PubMed] [Google Scholar]
- 12.Weinstein GD, Krueger GG, Lowe NJ, et al. Tazarotene gel, a new retinoid, for topical therapy of psoriasis: vehicle-controlled study of safety, efficacy, and duration of therapeutic effect. J Am Acad Dermatol. 1997;37:85–92. doi: 10.1016/S0190-9622(97)70216-0. [DOI] [PubMed] [Google Scholar]
- 13.Leonardi C, Bagel J, Yamauchi P, et al. Efficacy and safety of calcipotriene plus betamethasone dipropionate aerosol foam in patients with psoriasis vulgaris–a randomized phase III study (PSO-FAST) J Drugs Dermatol. 2015;14:1468–1477. [PubMed] [Google Scholar]
- 14.Griffiths CEM, Reich K, Lebwohl M, et al. Comparison of ixekizumab with etanercept or placebo in moderate-to-severe psoriasis (UNCOVER-2 and UNCOVER-3): results from two phase 3 randomised trials. Lancet. 2015;386:541–551. doi: 10.1016/S0140-6736(15)60125-8. [DOI] [PubMed] [Google Scholar]
- 15.Lebwohl MG, Sugarman JL, Gold LS, et al. Long-term safety results from a phase 3 open-label study of a fixed combination halobetasol propionate 0.01% and tazarotene 0.045% lotion in moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2019;80:282–285. doi: 10.1016/j.jaad.2018.09.002. [DOI] [PubMed] [Google Scholar]
- 16.Armstrong AW, Puig L, Joshi A, et al. Comparison of biologics and oral treatments for plaque psoriasis: a meta-analysis. JAMA Dermatol. 2020;156:258–269. doi: 10.1001/jamadermatol.2019.4029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ellis CN, Fradin MS, Messana JM, et al. Cyclosporine for plaque-type psoriasis. Results of a multidose, double-blind trial. N Engl J Med. 1991;324:277–284. doi: 10.1056/NEJM199101313240501. [DOI] [PubMed] [Google Scholar]
- 18.Moini-Jazani A, Ayati MH, Nadiri AA, Nasimi-Doost-Azgomi R. Efficacy of hydrotherapy, spa therapy, and balneotherapy for psoriasis and atopic dermatitis: a systematic review. Int J Dermatol. 2022 doi: 10.1111/ijd.16080. [DOI] [PubMed] [Google Scholar]
- 19.Zumiani G, Zanoni M, Agostini G. Valutazione dell’efficacia dell’acqua della fonte termale di Comano versus acqua di acquedotto nella cura della psoriasi. G Ital Dermatol Venereol. 2000;135:1–5. [Google Scholar]
- 20.Dinelli E, Lima A, Albanese S, et al. Comparative study between bottled mineral and tap water in Italy. J Geochem Explor. 2012;112:368–389. doi: 10.1016/j.gexplo.2011.11.002. [DOI] [Google Scholar]
- 21.Pedron R, Esposito A, Bianconi I, et al. Genomic and metagenomic insights into the microbial community of a thermal spring. Microbiome. 2019;7:8. doi: 10.1186/s40168-019-0625-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nicoletti G, Saler M, Tresoldi MM, Faga A, Benedet M, Cristofolini M. Regenerative effects of spring water-derived bacterial lysates on human skin fibroblast in in vitro culture: preliminary results. J Int Med Res. 2019;47:5777–5786. doi: 10.1177/0300060519880371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Grice EA, Segre JA. The skin microbiome. Nat Rev Microbiol. 2011;9:244–253. doi: 10.1038/nrmicro2537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Oh J, Byrd AL, Deming C, et al. Biogeography and individuality shape function in the human skin metagenome. Nature. 2014;514:59–64. doi: 10.1038/nature13786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chehoud C, Rafail S, Tyldsley AS, Seykora JT, Lambris JD, Grice EA. Complement modulates the cutaneous microbiome and inflammatory milieu. Proc Natl Acad Sci U S A. 2013;110:15061–15066. doi: 10.1073/pnas.1307855110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Naik S, Bouladoux N, Wilhelm C, et al. Compartmentalized control of skin immunity by resident commensals. Science. 2012;337:1115–1119. doi: 10.1126/science.1225152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lai Y, Cogen AL, Radek KA, et al. Activation of TLR2 by a small molecule produced by Staphylococcus epidermidis increases antimicrobial defense against bacterial skin infections. J Invest Dermatol. 2010;130:2211–2221. doi: 10.1038/jid.2010.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Naik S, Bouladoux N, Linehan JL, et al. Commensal–dendritic-cell interaction specifies a unique protective skin immune signature. Nature. 2015 doi: 10.1038/nature14052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Belkaid Y, Segre JA. Dialogue between skin microbiota and immunity. Science. 2014;346:954–959. doi: 10.1126/science.1260144. [DOI] [PubMed] [Google Scholar]
- 30.Scharschmidt TC, Fischbach MA. What lives on our skin: ecology, genomics and therapeutic opportunities of the skin microbiome. Drug Discov Today Dis Mech. 2013 doi: 10.1016/j.ddmec.2012.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tomi NS, Kränke B, Aberer E. Staphylococcal toxins in patients with psoriasis, atopic dermatitis, and erythroderma, and in healthy control subjects. J Am Acad Dermatol. 2005;53:67–72. doi: 10.1016/j.jaad.2005.02.034. [DOI] [PubMed] [Google Scholar]
- 32.Raza N, Usman M, Hameed A. Chronic plaque psoriasis: Streptococcus pyogenes throat carriage rate and therapeutic response to oral antibiotics in comparison with oral methotrexate. J Coll Physicians Surg Pak. 2007;17:717–720. [PubMed] [Google Scholar]
- 33.Rudramurthy SM, Honnavar P, Chakrabarti A, Dogra S, Singh P, Handa S. Association of Malassezia species with psoriatic lesions. Mycoses. 2014;57:483–488. doi: 10.1111/myc.12186. [DOI] [PubMed] [Google Scholar]
- 34.Fry L, Baker BS, Powles AV, Engstrand L. Psoriasis is not an autoimmune disease? Exp Dermatol. 2015;24:241–244. doi: 10.1111/exd.12572. [DOI] [PubMed] [Google Scholar]
- 35.Langan EA, Künstner A, Miodovnik M, et al. Combined culture and metagenomic analyses reveal significant shifts in the composition of the cutaneous microbiome in psoriasis. Br J Dermatol. 2019;181:1254–1264. doi: 10.1111/bjd.17989. [DOI] [PubMed] [Google Scholar]
- 36.Alekseyenko AV, Perez-Perez GI, De Souza A, et al. Community differentiation of the cutaneous microbiota in psoriasis. Microbiome. 2013;1:31. doi: 10.1186/2049-2618-1-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tett A, Pasolli E, Farina S, et al. Unexplored diversity and strain-level structure of the skin microbiome associated with psoriasis. NPJ Biofilms Microbiomes. 2017;3:14. doi: 10.1038/s41522-017-0022-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ashcroft DM, Wan Po AL, Williams HC, Griffiths CE. Clinical measures of disease severity and outcome in psoriasis: a critical appraisal of their quality. Br J Dermatol. 1999;141:185–191. doi: 10.1046/j.1365-2133.1999.02963.x. [DOI] [PubMed] [Google Scholar]
- 39.Asnicar F, Berry SE, Valdes AM, et al. Microbiome connections with host metabolism and habitual diet from 1098 deeply phenotyped individuals. Nat Med. 2021;27:321–332. doi: 10.1038/s41591-020-01183-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Human Microbiome Project Consortium Structure, function and diversity of the healthy human microbiome. Nature. 2012;486:207–214. doi: 10.1038/nature11234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Muyzer G, de Waal EC, Uitterlinden AG. Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol. 1993;59:695–700. doi: 10.1128/aem.59.3.695-700.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jiang H, Dong H, Zhang G, Yu B, Chapman LR, Fields MW. Microbial diversity in water and sediment of lake Chaka, an Athalassohaline Lake in Northwestern China. Appl Environ Microbiol. 2006;72:3832–3845. doi: 10.1128/AEM.02869-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Caporaso JG, Kuczynski J, Stombaugh J, et al. QIIME allows analysis of high-throughput community sequencing data. Nat Methods. 2010;7:335–336. doi: 10.1038/nmeth.f.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pruesse E, Quast C, Knittel K, et al. SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res. 2007;35:7188–7196. doi: 10.1093/nar/gkm864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Edgar RC. Search and clustering orders of magnitude faster than BLAST. Bioinformatics. 2010;26:2460–2461. doi: 10.1093/bioinformatics/btq461. [DOI] [PubMed] [Google Scholar]
- 46.rbiom: Interact with Biological Observation Matrix files. Github. https://github.com/cmmr/rbiom. Accessed 29 Nov 2022.
- 47.Mallick H, Rahnavard A, McIver LJ, et al. Multivariable association discovery in population-scale meta-omics studies. PLoS Comput Biol. 2021;17:e1009442. doi: 10.1371/journal.pcbi.1009442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Beghini F, McIver LJ, Blanco-Míguez A, et al. Integrating taxonomic, functional, and strain-level profiling of diverse microbial communities with bioBakery 3. Elife. 2021 doi: 10.7554/eLife.65088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Costello EK, Lauber CL, Hamady M, Fierer N, Gordon JI, Knight R. Bacterial community variation in human body habitats across space and time. Science. 2009;326:1694–1697. doi: 10.1126/science.1177486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Pickard JM, Zeng MY, Caruso R, Núñez G. Gut microbiota: role in pathogen colonization, immune responses, and inflammatory disease. Immunol Rev. 2017;279:70–89. doi: 10.1111/imr.12567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Martin R, Henley JB, Sarrazin P, Seité S. Skin microbiome in patients with psoriasis before and after balneotherapy at the Thermal Care Center of La Roche-Posay. J Drugs Dermatol. 2015;14:1400–1405. [PubMed] [Google Scholar]
- 52.Byrd AL, Belkaid Y, Segre JA. The human skin microbiome. Nat Rev Microbiol. 2018;16:143–155. doi: 10.1038/nrmicro.2017.157. [DOI] [PubMed] [Google Scholar]
- 53.Tong SYC, Davis JS, Eichenberger E, Holland TL, Fowler VG., Jr Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management. Clin Microbiol Rev. 2015;28:603–661. doi: 10.1128/CMR.00134-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Esposito S, Noviello S, Leone S. Epidemiology and microbiology of skin and soft tissue infections. Curr Opin Infect Dis. 2016;29:109–115. doi: 10.1097/QCO.0000000000000239. [DOI] [PubMed] [Google Scholar]
- 55.Akinduti PA, Obafemi YD, Ugboko H, et al. Emerging vancomycin-non susceptible coagulase negative Staphylococci associated with skin and soft tissue infections. Ann Clin Microbiol Antimicrob. 2022;21:31. doi: 10.1186/s12941-022-00516-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Natsis NE, Cohen PR. Coagulase-negative Staphylococcus skin and soft tissue infections. Am J Clin Dermatol. 2018;19:671–677. doi: 10.1007/s40257-018-0362-9. [DOI] [PubMed] [Google Scholar]
- 57.Akiyama H, Kanzaki H, Tada J, Arata J. Coagulase-negative staphylococci isolated from various skin lesions. J Dermatol. 1998;25:563–568. doi: 10.1111/j.1346-8138.1998.tb02459.x. [DOI] [PubMed] [Google Scholar]
- 58.Grice EA, Kong HH, Conlan S, et al. Topographical and temporal diversity of the human skin microbiome. Science. 2009;324:1190–1192. doi: 10.1126/science.1171700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Han J, Sun L, Dong X, et al. Characterization of a novel plant growth-promoting bacteria strain Delftia tsuruhatensis HR4 both as a diazotroph and a potential biocontrol agent against various plant pathogens. Syst Appl Microbiol. 2005 doi: 10.1016/j.syapm.2004.09.003. [DOI] [PubMed] [Google Scholar]
- 60.Zhang L-L, He D, Chen J-M, Liu Y. Biodegradation of 2-chloroaniline, 3-chloroaniline, and 4-chloroaniline by a novel strain Delftia tsuruhatensis H1. J Hazard Mater. 2010;179:875–882. doi: 10.1016/j.jhazmat.2010.03.086. [DOI] [PubMed] [Google Scholar]
- 61.De Gusseme B, Vanhaecke L, Verstraete W, Boon N. Degradation of acetaminophen by Delftia tsuruhatensis and Pseudomonas aeruginosa in a membrane bioreactor. Water Res. 2011;45:1829–1837. doi: 10.1016/j.watres.2010.11.040. [DOI] [PubMed] [Google Scholar]
- 62.Preiswerk B, Ullrich S, Speich R, Bloemberg GV, Hombach M. Human infection with Delftia tsuruhatensis isolated from a central venous catheter. J Med Microbiol. 2011;60:246–248. doi: 10.1099/jmm.0.021238-0. [DOI] [PubMed] [Google Scholar]
- 63.Keusch S, Speich R, Treder U, Ulrich SS. Central venous catheter infections in outpatients with pulmonary hypertension treated with continuous iloprost. Respiration. 2013;86:402–406. doi: 10.1159/000350441. [DOI] [PubMed] [Google Scholar]
- 64.Ohkubo T, Matsumoto Y, Cho O, Ogasawara Y, Sugita T. Delftia acidovorans secretes substances that inhibit the growth of Staphylococcus epidermidis through TCA cycle-triggered ROS production. PLoS ONE. 2021;16:e0253618. doi: 10.1371/journal.pone.0253618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Rubin LG. Principles and practice of pediatric infectious diseases. Elsevier; 2018. Other gram-negative coccobacilli; pp. 967–9.e1. [Google Scholar]
- 66.Wang C-Y, Wang H-C, Li J-M, et al. Invasive infections of Aggregatibacter (Actinobacillus) actinomycetemcomitans. J Microbiol Immunol Infect. 2010;43:491–497. doi: 10.1016/S1684-1182(10)60076-X. [DOI] [PubMed] [Google Scholar]
- 67.Loomis KH, Wu SK, Ernlund A, et al. A mixed community of skin microbiome representatives influences cutaneous processes more than individual members. Microbiome. 2021;9:22. doi: 10.1186/s40168-020-00963-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Nørskov-Lauritsen N. Classification, identification, and clinical significance of Haemophilus and Aggregatibacter species with host specificity for humans. Clin Microbiol Rev. 2014;27:214–240. doi: 10.1128/CMR.00103-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Smith KJ, Neafie R, Yeager J, Skelton HG. Micrococcus folliculitis in HIV-1 disease. Br J Dermatol. 1999;141:558–561. doi: 10.1046/j.1365-2133.1999.03060.x. [DOI] [PubMed] [Google Scholar]
- 70.Nuñez M. Encyclopaedia of food microbiology. Elsevier; 2014. Micrococcus; pp. 627–633. [Google Scholar]
- 71.Salem I, Ramser A, Isham N, Ghannoum MA. The gut microbiome as a major regulator of the gut-skin axis. Front Microbiol. 2018;9:1459. doi: 10.3389/fmicb.2018.01459. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Raw sequences were deposited in the NCBI Sequence Read Archive (SRA) under the accession number PRJNA909312.