Abstract
Two strains of obligately barophilic bacteria were isolated from a sample of the world’s deepest sediment, which was obtained by the unmanned deep-sea submersible Kaiko in the Mariana Trench, Challenger Deep, at a depth of 10,898 m. From the results of phylogenetic analysis based on 16S rRNA gene sequences, DNA-DNA relatedness study, and analysis of fatty acid composition, the first strain (DB21MT-2) appears to be most highly similar to Shewanella benthica and close relatives, and the second strain (DB21MT-5) appears to be closely related to the genus Moritella. The optimal pressure conditions for growth of these isolates were 70 MPa for strain DB21MT-2 and 80 MPa for strain DB21MT-5, and no growth was detected at pressures of less than 50 MPa with either strain. This is the first evidence of the existence of an extreme-barophile bacterium of the genus Moritella isolated from the deep-sea environment.
The Mariana Trench, Challenger Deep (11°22′N, 142°25′E), is the deepest ocean bottom in the world, and relatively few kinds of organisms have been isolated from it. Pseudomonas bathycetes was the first bacterial isolate obtained from a sediment sample collected from the Mariana Trench (13). Apparently, P. bathycetes is not an organism adapted to the deep-sea environment, since its generation time is 33 days under such conditions (2°C, 100 MPa) (18). This bacterium grows nonbarophilically at atmospheric pressure and at temperatures of as high as 25°C (15). With respect to other deep-sea organisms, in 1978 Yayanos et al. were successful in collecting an amphipod (Hirondellea gigas) from the Mariana Trench at a depth of 10,476 m by using an insulated trap (22). An obligately barophilic bacterium strain, MT41, was isolated from this deep-sea amphipod, and this bacterium could grow only under pressure conditions of greater than 518 bars, i.e., of approximately 50 MPa or more (22). DeLong et al. reported that strain MT41 was closely related to the genus Colwellia and suggested that this genus was one of the typical deep-sea psychrophilic and barophilic bacterial genera (2).
To investigate the environment and the biology of the deep-sea world, several manned and unmanned deep-sea submersibles have been constructed. The newly constructed unmanned submersible Kaiko, which means trench in Japanese and which is operated by the Japan Marine Science and Technology Center, is the most capable of all, with an ability to submerge to the world’s deepest sea bottom depth of 11,000 m (11). On 28 February 1996, we set out to obtain sediment samples from the world’s deepest point in the ocean. This was Kaiko dive number 21 during our last Mariana Trench cruise, which involved the use of a sterilized sediment sampler at a depth of 10,898 m. It seems likely that this was the first time sediment samples have been recovered from the world’s deepest point without any microbiological contamination derived from overlying waters. We have reported the molecular analysis of this sediment and the sequences of pressure-regulated genes found in DNA extracted from these samples (7). These findings suggested that deep-sea-adapted barophilic bacteria are present in the sediment.
In this paper, we describe the isolation of obligately barophilic bacteria from the Mariana sediment obtained in the Kaiko dives described above, and we show that the isolates do not belong to a single genus but to two genera, Shewanella and Moritella.
MATERIALS AND METHODS
Sample collection.
Deep-sea sediment samples were collected from the Mariana Trench, Challenger Deep (11°22.10′N, 142°25.85′E), at a depth of 10,898 m, by means of sterilized mud samplers, with the unmanned submersible Kaiko. At the bottom of the Mariana Trench, brown soft sediment was widespread and no rock was observed. Several deep-sea animals were observed, suggesting the presence of a large community of deep-sea invertebrates (6). The deepest mud samples were taken by Kaiko’s manipulator and put into the sample holder of the sterilized sampler, and then the samples were carried to the sea surface without any change in temperature but were subject to changes in pressure (7). A part of each of the samples employed for isolation of barophilic bacteria was pressurized at approximately 100 MPa in a pressure vessel which was placed in a refrigerator (2 to 4°C) on the support ship M. S. Yokosuka.
Isolation of the obligately barophilic bacteria and growth studies.
Barophilic bacteria were isolated according to the procedure reported previously (9). For single-colony isolation, the cultures were incubated under a pressure of 100 MPa in plastic bags. Growth of the isolates under conditions of 0.1 to 100 MPa at 10°C in pressure vessels was tested in Marine Broth 2216 (Difco Laboratories, Detroit, Mich.).
Electron microscopy.
The morphology of isolated cells was observed by transmission electron microscopy. For negative staining, 1 drop of culture was placed on a copper grid coated with Pioloform and carbon and stained with 1% potassium phosphotungstic acid adjusted to pH 6.5 with potassium hydroxide. The negatively stained cells were observed with a model JEM-1210 transmission electron microscope (JEOL, Tokyo, Japan) at an accelerating voltage of 80 kV.
Phylogenetic analysis and DNA studies.
Chromosomal DNAs were extracted from the isolates by the method of Saito and Miura (16), and 16S rRNA genes (16S rDNA) were amplified by PCR with the primers Eubac27F and Eubac1492R (1). Amplified DNA was sequenced by a direct sequencing procedure with an automated DNA sequencer (model 373S; Perkin Elmer/Applied Biosystems Co., Foster City, Calif.) according to the manual. Phylogenetic analysis was done with the Clustal W Multi-alignment program (19), and a phylogenetic tree was constructed by the neighbor-joining (NJ) method (17). For analysis of relatedness, DNA-DNA hybridization was carried out at 45°C for 3 h and measured fluorometrically by the method described by Ezaki et al. (5).
Fatty acid analysis.
Cells were grown in marine broth 2216 under high pressure (70 MPa for strain DB21MT-2 and 80 MPa for strain DB21MT-5) at 10°C for 3 days (early stationary phase), washed twice with 3% NaCl solution at 4°C by centrifugation at 8,000 × g, and freeze dried. The dried cells (20 mg) were placed in a Teflon-lined, screw-capped tube containing 2 ml of anhydrous methanolic HCl, and the tube was heated at 100°C for 3 h. After cooling, 1 ml of water was added, and the fatty acid methyl esters were extracted with n-hexane. The samples were analyzed for cellular fatty acids by a gas-liquid chromatography-mass spectrometer (model HP5971A-MSD; Hewlett-Packard Co., Palo Alto, Calif.) (10).
Nucleotide sequence accession numbers.
The 16S rDNA sequences of strains DB21MT-2 and DB21MT-5 determined in this study have been deposited in the DDBJ, EMBL, and GenBank nucleotide sequence databases under accession nos. AB008796 and AB008797, respectively.
RESULTS
Analysis of the Mariana isolates.
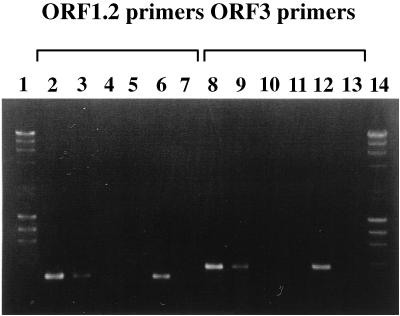
Six obligately barophilic strains which were able to grow well at 100 MPa and which were not able to grow at atmospheric pressure were isolated from the Mariana Trench sediment. As shown in Fig. 1, the pressure-regulated operons particularly identified in Shewanella barophilic strains (12) were amplified by PCR from DNAs extracted from three strains, DB21MT-2, -4, and -7, but were not amplified from DNAs from the other three strains, DB21MT-5, -6, and -9. From results for partially sequenced 16S ribosomal DNA (rDNA), we determined that two different 16S rDNA sequences were present, one in the three isolates with the pressure-regulated operons and the other in the three isolates without these operons. Therefore, two barophilic strains, DB21MT-2 and -5, were chosen for further study.
FIG. 1.
PCR amplification of pressure-regulated operons ORF1.2 and ORF3 from the DNAs of Mariana isolates by using the ORF1.2 and ORF3 primers. Lanes: 1 and 14, DNA size marker DRIgestIII (λHindIII plus φX174 HaeIII; Pharmacia Biotech, Uppsala, Sweden); 2 and 8, DB21MT-2; 3 and 9, DB21MT-4; 4 and 10, DB21MT-5; 5 and 11, DB21MT-6; 6 and 12, DB21MT-9; and 7 and 13, DB21MT-9.
Morphology and growth properties.
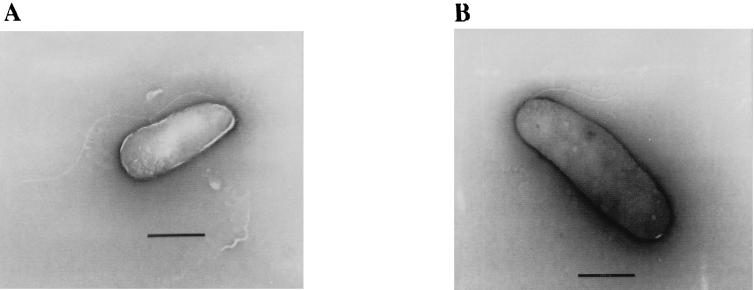
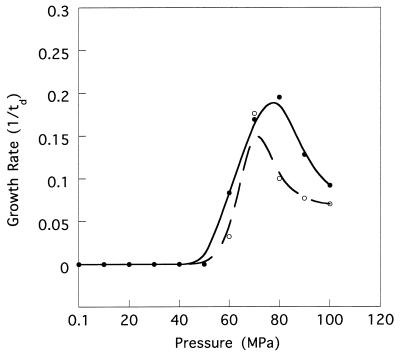
The cells of strain DB21MT-2 were 2 by 0.8 to ∼1 μm in size, and those of strain DB21MT-5 were a little bigger, 2.5 to ∼3 by 1 μm. Both of these strains were found to have a single polar flagellum (Fig. 2). As shown in Fig. 3, the optimal pressure conditions for growth of strains DB21MT-2 and -5 were 70 and 80 MPa, respectively. Neither of these strains was able to grow at pressures of less than 50 MPa, but both were able to grow well at higher pressures, even at 100 MPa.
FIG. 2.
Electron micrographs (negative staining) of extreme-barophile bacteria (strains DB21MT-2 [A] and DB21MT-5 [B]). Bar, 1 μm.
FIG. 3.
Growth properties of the extreme barophiles at elevated hydrostatic pressure. Dotted line, DB21MT-2; solid line, DB21MT-5. td, doubling time (in hours).
Phylogenetic relationships of the isolated hyperbarophiles.
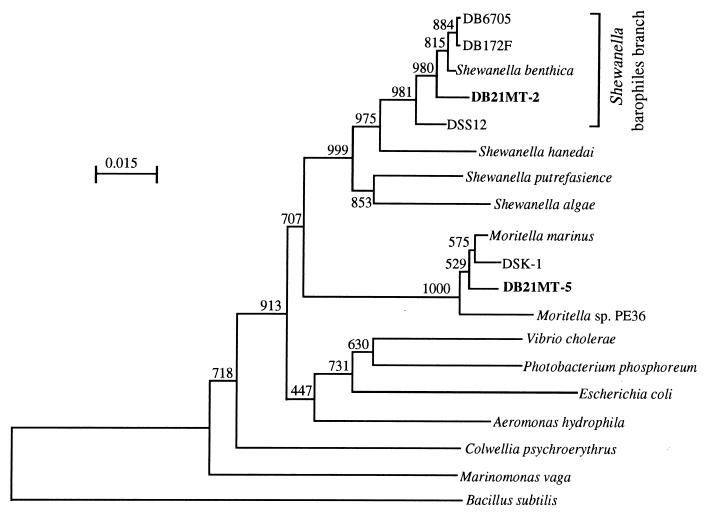
A phylogenetic tree was constructed by the NJ method based on 16S rDNA sequences, as illustrated in Fig. 4. Strain DB21MT-2 is closely related to Shewanella benthica and other barophilic strains belonging to the Shewanella barophile branch. This result corresponds well with the findings shown in Fig. 1, since this strain was found to have the pressure-regulated operon identified only in members of this branch (12). The other isolated strain, DB21MT-5, is closely related to Moritella marinus and to the barotolerant strain DSK1 reported previously (9).
FIG. 4.
Phylogenetic tree showing the relationships among the isolated bacteria within the γ subdivision of the class Proteobacteria. The scale represents the average numbers of nucleotide substitutions per site. Bootstrap values are shown for frequencies per 1,000 times.
DNA-DNA hybridization.
The results of DNA-DNA hybridization analysis comparing the isolated strains DB21MT-2 and DB21MT-5 with the related bacterial species suggested from the 16S rDNA phylogenetic tree (Fig. 4) are shown in Tables 1 and 2, respectively. A high level of DNA-DNA relatedness among strain DB21MT-2, the previously reported barophilic isolates DB6705 (9) and DB172F (8), and S. benthica was evident (77 to 84%), indicating that these strains might be members of the same species (20). The hybridization values obtained between strain DB21MT-5 and the Moritella and Shewanella reference strains were significantly less than that accepted as the phylogenetic definition of a species as described by Wayne et al. (20). In addition to the phylogenetic analysis, this suggested that strain DB21MT-5 is a new species of the genus Moritella; however, more taxonomic studies are necessary to clarify this matter.
TABLE 1.
Levels of homology for the chromosomal DNAs of strain DB21MT-2 and its closely related strainsa
| Strain | % Homology with DNA from:
|
||||||
|---|---|---|---|---|---|---|---|
| S. han | S. put | S. ben | 172F | 6705 | MT-2 | DSS12 | |
| S. han | 100 | 7 | 7 | 5 | 13 | 10 | 16 |
| S. put | 15 | 100 | 6 | 6 | 11 | 9 | 9 |
| S. ben | 12 | 7 | 100 | 79 | 88 | 80 | 47 |
| 172F | 16 | 9 | 90 | 100 | 96 | 77 | 39 |
| 6705 | 14 | 10 | 91 | 79 | 100 | 84 | 46 |
| MT-2 | 20 | 21 | 81 | 79 | 78 | 100 | 48 |
| DSS12 | 11 | 13 | 47 | 43 | 45 | 40 | 100 |
S. han, S. hanedai; S. put, S. putrefaciens; S. ben, S. benthica; 172F, strain DB172F; 6705, strain DB6705; MT-2, strain DB21MT-2; and DSS12, strain DSS12.
TABLE 2.
Levels of homology for the chromosomal DNAs of strain DB21MT-5 and its closely related strainsa
| Strain | % Homology with DNA from:
|
||||
|---|---|---|---|---|---|
| S. put | S. ben | M. mar | DSK1 | MT-5 | |
| S. put | 100 | 8 | 9 | 10 | 7 |
| S. ben | 7 | 100 | 11 | 14 | 12 |
| M. mar | 9 | 8 | 100 | 33 | 28 |
| DSK1 | 16 | 4 | 17 | 100 | 22 |
| MT-5 | 10 | 8 | 32 | 41 | 100 |
M. mar, M. marinus; DSK1, strain DSK1; MT-5, strain DB21MT-5.
Fatty acid analysis.
The whole-cell fatty acid compositions of the isolated barophilic strains DB21MT-2 and -5 and those of selected reference strains (S. benthica and M. marinus, respectively) are shown in Table 3. The dominant fatty acid in the isolated barophiles was hexadecenoic acid (16:1), and the long-chain polyunsaturated fatty acids (PUFAs) present in significant amounts were eicosapentaenoic acid (EPA) (20:5) in strain DB21MT-2 and docosahexaenoic acid (DHA) (22:6) in strain DB21MT-5. The fatty acid profiles of these isolated strains, DB21MT-2 and -5, were basically similar to those of the reference species S. benthica and M. marinus, respectively, particularly the PUFA patterns. Approximately 70% of the membrane lipids in both extreme barophiles were unsaturated fatty acids, and, compared with the reference strains, higher amounts of octadecenoic acid (18:1) in strain DB21MT-2 and tetradecenoic acid (14:1) in strain DB21MT-5 were evident.
TABLE 3.
Fatty acid compositions of the isolated strains and type strainsa
| Strain | % showing the following compositions:
|
|||||||
|---|---|---|---|---|---|---|---|---|
| 14:0 | 14:1 | 15:0 iso | 16:0 | 16:1 | 18:1 | 20:5 (EPA) | 22:6 (DHA) | |
| DB21MT-2 | 4 | 5 | 22 | 31 | 19 | 18 | ||
| S. benthica | 21 | 6 | 18 | 45 | 1 | 10 | ||
| DB21MT-5 | 25 | 12 | 9 | 45 | 1 | 8 | ||
| M. marinus | 16 | 1 | 1 | 21 | 45 | 2 | 12 | |
Type strains were grown at atmospheric pressure and at 4°C (S. benthica) or at 15°C (M. marinus).
DISCUSSION
Extremely barophilic bacteria, which we defined as bacteria that are unable to grow at pressures of less than 50 MPa but that are able to grow well at 100 MPa, were isolated from sediment obtained by means of the unmanned submersible Kaiko system from the world’s deepest ocean bottom, the Mariana Trench, Challenger Deep, at a depth of 10,898 m. One of the isolates, strain DB21MT-2, which showed optimal growth at a pressure of 70 MPa, belongs to the species S. benthica as determined based on 16S rDNA sequence comparisons and DNA-DNA hybridization analysis (Fig. 4; Table 1). This strain particularly produces EPA as a component of its membrane lipids (Table 3). All of the barophilic Shewanella strains isolated from the deep sea produce EPA. Another isolate, strain DB21MT-5, which shows optimal growth at a pressure of 80 MPa, is closely related to the genus Moritella as determined based on 16S rDNA sequence comparisons; however, DNA-DNA hybridization results suggest that this strain does not belong to the same species as known Moritella strains (Fig. 4; Table 2). This strain produces DHA as a component of its membrane lipids (Table 3). M. marinus also produces DHA (Table 3); thus, strain DB21MT-5 may share some similar properties with this species, except for its pressure requirements for growth. The Moritella strains reported are not obligately barophilic; they are barotolerant (strain DSK1 [9]) and facultatively barophilic (strain PE36 [21]) deep-sea bacteria.
We have reported that EPA production is one of the particular characteristics of deep-sea-adapted Photobacterium species, such as Photobacterium profundum (14), and the incidence of such PUFAs in barophilic genera suggests that PUFAs play an important role in maintenance of membrane fluidity at low temperature and high pressure (3).
The first obligately barophilic strain isolated from the Mariana Trench was strain MT41 (22), which was unable to grow at pressures of less than 50 MPa, and its optimal pressure for growth was approximately 100 MPa (21). Thus, this strain is also an extreme-barophile bacterium. It is not related to the genus Shewanella or Moritella but is closely related to the genus Colwellia (2). Deming et al. reported that optimal pressures for growth of the obligately barophilic bacterium strain BNL-1 were 925 atm (about 93 MPa) at 10°C and 740 atm (about 74 MPa) at 2°C (4). Strain BNL-1 was designated Colwellia hadaliensis, and this strain is very similar to extreme barophiles. Shewanella sp. strains PT99, PT48 (3), and DB172F (8) are also obligate barophiles, showing optimal growth at pressures of 69, 62, and 70 MPa, respectively. However, these Shewanella strains are able to grow at pressures of less than 50 MPa; thus, they are not extreme-barophile strains. Among the members of the genus Moritella, a facultatively barophilic strain, PE36, has been reported (21), but no obligately barophilic strains are known.
Therefore, this is the first evidence of the existence of an obligately barophilic Moritella sp. from the deep-sea environment, and it is an extreme-barophile bacterium.
ACKNOWLEDGMENTS
We are very grateful to the Kaiko operation team, I. Kawana and coworkers, and the crew of M. S. Yokosuka for helping us to collect the deep-sea samples. We also thank W. R. Bellamy for assistance in editing the manuscript.
REFERENCES
- 1.DeLong E F. Archaea in coastal marine environments. Proc Natl Acad Sci USA. 1992;89:5685–5689. doi: 10.1073/pnas.89.12.5685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.DeLong E F, Franks D G, Yayanos A A. Evolutionary relationships of cultivated psychrophilic and barophilic deep-sea bacteria. Appl Environ Microbiol. 1997;63:2105–2108. doi: 10.1128/aem.63.5.2105-2108.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.DeLong E F, Yayanos A A. Biochemical function and ecological significance of novel bacterial lipids in deep-sea prokaryotes. Appl Environ Microbiol. 1986;51:730–737. doi: 10.1128/aem.51.4.730-737.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Deming J W, Somers L K, Straube W L, Swartz D G, Macdonell M T. Isolation of an obligately barophilic bacterium and description of a new genus, Colwellia gen. nov. Syst Appl Microbiol. 1988;10:152–160. [Google Scholar]
- 5.Ezaki T, Hashimoto Y, Yabuuchi E. Fluorometric deoxyribonucleic acid-deoxyribonucleic acid hybridization in microdilution wells as an alternative to membrane filter hybridization in which radioisotopes are used to determine genetic relatedness among bacterial strains. Int J Syst Bacteriol. 1989;39:224–229. [Google Scholar]
- 6.Kato C, Li L, Nakamura Y, Nogi Y, Tamaoka J, Masui N, Horikoshi K. Biodiversity in the world’s deepest sediment of the Mariana Trench, at a depth of 11,000 m and isolation of hyperbarophilic bacteria from the same sediment. JAMSTEC J Deep Sea Res. 1997;13:119–130. [Google Scholar]
- 7.Kato C, Li L, Tamaoka J, Horikoshi K. Molecular analyses of the sediment of the 11000 m deep Mariana Trench. Extremophiles. 1997;1:117–123. doi: 10.1007/s007920050024. [DOI] [PubMed] [Google Scholar]
- 8.Kato C, Masui N, Horikoshi K. Properties of obligately barophilic bacteria isolated from a sample of deep-sea sediment from the Izu-Bonin trench. J Mar Biotechnol. 1996;4:96–99. [Google Scholar]
- 9.Kato C, Sato T, Horikoshi K. Isolation and properties of barophilic and barotolerant bacteria from deep-sea mud samples. Biodivers Conserv. 1995;4:1–9. [Google Scholar]
- 10.Komagata K, Suzuki K. Lipid and cell-wall analysis in bacterial systematics. Methods Microbiol. 1987;19:161–207. [Google Scholar]
- 11.Kyo M, Miyazaki E, Tsukioka S, Ochi H, Amitani Y, Tsuchiya T, Aoki T, Takagawa S. The sea trial of “KAIKO”, the full ocean depth research ROV. OCEANS’95. 1995;3:1991–1996. [Google Scholar]
- 12.Li L, Kato C, Nogi Y, Horikoshi K. Distribution of the pressure-regulated operons in deep-sea bacteria. FEMS Microbiol Lett. 1998;159:159–166. doi: 10.1111/j.1574-6968.1998.tb12855.x. [DOI] [PubMed] [Google Scholar]
- 13.Morita R Y. Survival of bacteria in cold and moderate hydrostatic pressure environments with special reference to psychrophilic and barophilic bacteria. In: Gray R G, Postgate J R, editors. The survival of vegetative microbes. Cambridge, United Kingdom: Cambridge University Press; 1976. pp. 279–298. [Google Scholar]
- 14.Nogi Y, Masui N, Kato C. Photobacterium profundum sp. nov., a new, moderately barophilic bacterial species isolated from a deep-sea sediment. Extremophiles. 1998;2:1–7. doi: 10.1007/s007920050036. [DOI] [PubMed] [Google Scholar]
- 15.Pope D H, Smith W P, Swartz R W, Landau J V. Role of bacterial ribosomes in barotolerance. J Bacteriol. 1975;121:664–669. doi: 10.1128/jb.121.2.664-669.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Saito H, Miura K. Preparation of transforming deoxyribonucleic acid by phenol treatment. Biochim Biophys Acta. 1963;72:612–629. [PubMed] [Google Scholar]
- 17.Saitou N, Nei M. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol. 1987;4:406–425. doi: 10.1093/oxfordjournals.molbev.a040454. [DOI] [PubMed] [Google Scholar]
- 18.Schwarz J R, Colwell R R. Abstracts of the 75th Annual Meeting of the American Society for Microbiology 1975. Washington, D.C: American Society for Microbiology; 1975. Macromolecular biosynthesis in Pseudomonas bathycetes at deep-sea pressure and temperature, session 104, abstr. K.94. [Google Scholar]
- 19.Thompson J D, Higgins D G, Gibson T J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994;22:4673–4680. doi: 10.1093/nar/22.22.4673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wayne L G, Brenner D J, Colwell R R, Grimont P A D, Kandler O, Krichevsky M I, Moore L H, Moore W E C, Murray R G E, Stackbrandt E, Starr M P, Truper H G. Report of the Ad Hoc Committee on reconciliation of approaches of bacterial systematics. Int J Syst Bacteriol. 1987;37:463–464. [Google Scholar]
- 21.Yayanos A A. Evolutional and ecological implications of the properties of deep-sea barophilic bacteria. Proc Natl Acad Sci USA. 1986;83:9542–9546. doi: 10.1073/pnas.83.24.9542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yayanos A A, Dietz A S, Boxtel R V. Obligately barophilic bacterium from the Mariana Trench. Proc Natl Acad Sci USA. 1981;78:5212–5215. doi: 10.1073/pnas.78.8.5212. [DOI] [PMC free article] [PubMed] [Google Scholar]