Abstract
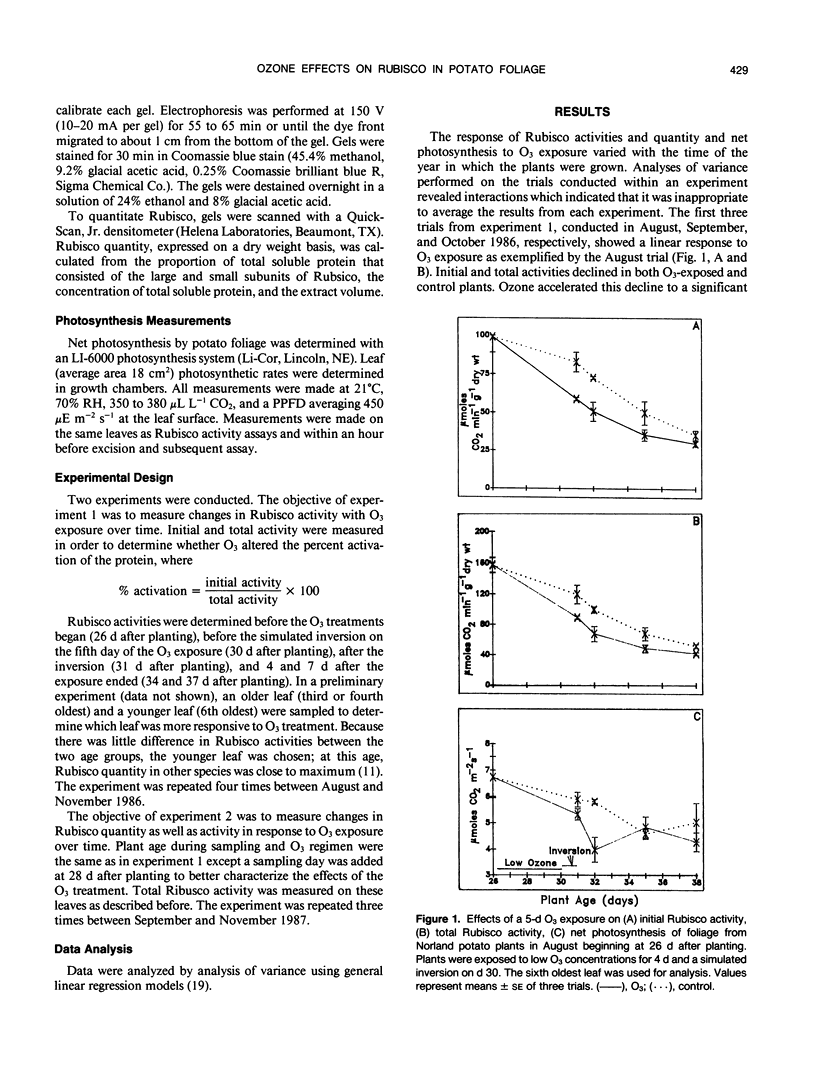
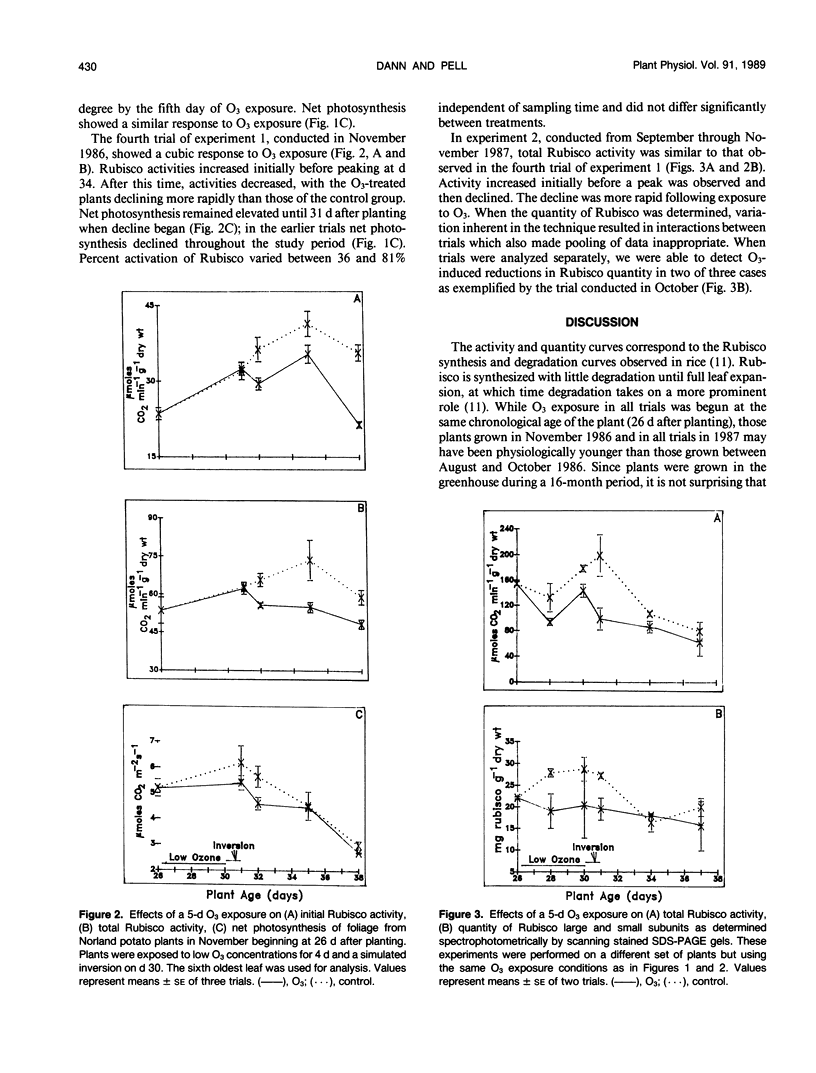
The effect of ozone (O3) on ribulose bisphosphate carboxylase/oxygenase (Rubisco) activity and quantity and net photosynthesis in greenhouse-grown Solanum tuberosum L. cv `Norland' foliage was studied in relation to oxidant-induced premature senescence. Plants, 26 days old, were exposed to 0.06 to 0.08 microliters per liter O3 from 1000 to 1600 hours for 4 days in a controlled environment chamber. On day 5, plants were exposed to a 6-hour simulated inversion in which O3 peaked at 0.12 microliters per liter. Net photosynthesis declined in response to O3 but recovered to near control levels 3 days after the exposure ended. Rubisco activity and quantity in control potato foliage increased and then decreased during the 12-day interval of the study. In some experiments foliage studied was physiologically mature and Rubisco activity had peaked when O3 exposure commenced. In those cases, O3 accelerated the decline in Rubisco activity. When less mature foliage was treated with O3, the leaves never achieved the maximal level of Rubisco activity observed in control foliage and also exhibited more rapid decline in initial and total activity. Percent activation of Rubisco (initial/total activity) was not affected significantly by treatment. Quantity of Rubisco decreased in concert with activity. The decrease in activities is most likely due to a decrease in available protein rather than a decrease in the percentage of Rubisco activated in vivo. The reduction in the quantity of Rubisco, an important foliar storage protein, could contribute to premature senescence associated with toxicity of this air pollutant.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Miller B. L., Huffaker R. C. Hydrolysis of Ribulose-1,5-bisphosphate Carboxylase by Endoproteinases from Senescing Barley Leaves. Plant Physiol. 1982 Jan;69(1):58–62. doi: 10.1104/pp.69.1.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miziorko H. M., Lorimer G. H. Ribulose-1,5-bisphosphate carboxylase-oxygenase. Annu Rev Biochem. 1983;52:507–535. doi: 10.1146/annurev.bi.52.070183.002451. [DOI] [PubMed] [Google Scholar]
- Pell E. J., Pearson N. S. Ozone-Induced Reduction in Quantity of Ribulose-1,5-bisphosphate Carboxylase in Alfalfa Foliage. Plant Physiol. 1983 Sep;73(1):185–187. doi: 10.1104/pp.73.1.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reich P. B., Amundson R. G. Ambient levels of ozone reduce net photosynthesis in tree and crop species. Science. 1985 Nov 1;230(4725):566–570. doi: 10.1126/science.230.4725.566. [DOI] [PubMed] [Google Scholar]
- Seemann J. R., Berry J. A., Freas S. M., Krump M. A. Regulation of ribulose bisphosphate carboxylase activity in vivo by a light-modulated inhibitor of catalysis. Proc Natl Acad Sci U S A. 1985 Dec;82(23):8024–8028. doi: 10.1073/pnas.82.23.8024. [DOI] [PMC free article] [PubMed] [Google Scholar]


