Abstract
Background
Psoriasis has been linked to dyslipidemia. However, the magnitude of the association between psoriasis and serum apolipoproteins A1 and B remains unclear.
Methods
We systematically searched PubMed, Embase, and Cochrane Library databases for eligible studies published before August 10, 2023. Data were pooled using Stata software. We adopted a random-effects model for the meta-analysis. Additionally, we conducted subgroup analyses of the studies according to the psoriasis type and matched body mass index (BMI).
Results
Seventeen studies involving 2467 participants were included. Psoriasis was associated with decreased serum apolipoprotein A1 (weighted mean difference [WMD] = −9.05, P < 0.001) and increased serum apolipoprotein B (WMD = 11.68, P < 0.001). In subgroup analysis after matching BMI, the findings showing an association of psoriasis with serum apolipoprotein A1 (WMD = −14.07, P < 0.001) and serum apolipoprotein B (WMD = 13.07, P < 0.001) were consistent with the overall results. The subgroup analysis for the presence or absence of psoriatic arthritis showed that serum apolipoprotein A1 was significantly decreased in psoriasis with (WMD = −11.29, P < 0.001) and without arthritis (WMD = −8.69, P = 0.039); whereas serum apolipoprotein B was significantly increased in psoriasis with (WMD = 13.57, P < 0.001) and without arthritis (WMD = 9.21, P < 0.001).
Conclusions
Our study revealed that psoriasis is associated with decreased serum apolipoprotein A1 and increased serum apolipoprotein B levels compared with healthy controls.
Keywords: Psoriasis, Lipid metabolism, Apolipoprotein A1, Apolipoprotein B, Meta-analysis
1. Introduction
Psoriasis is a chronic immune-mediated disease characterized primarily by scaly and erythematous plaque [1,2]. The prevalence of psoriasis among adults varies worldwide, ranging from 0.4 % to 8.5 %. In the United States, more than 7.5 million adults are estimated to have psoriasis [3]. Psoriasis is gradually being recognized as a systemic inflammatory disease linked with multiple comorbidities, including psychiatric disturbances, dyslipidemia, and metabolic syndrome, leading to impaired quality of life and reduced life expectancy [[4], [5], [6]].
Over the past several decades, the knowledge of the cellular and molecular mechanisms of psoriasis has been expanded, and emerging evidence indicates that immune-mediated inflammatory responses in psoriasis, resulting in endothelial cell dysfunction, dyslipidemia, and atherosclerotic processes, might represent key pathological mechanisms underlying the complications associated with psoriasis [4,7]. Lipid metabolism in psoriasis attracted attention as early as the beginning of the 20th century, and it has been reported that the continuous detachment of psoriatic scales leads to permanent loss of lipids, which may result in abnormal serum lipids [8]. A previous meta-analysis has reported that psoriasis is associated with higher total cholesterol, low-density lipoprotein cholesterol, and triglyceride levels, suggesting that patients with psoriasis are at risk for dyslipidemia [9]. Apolipoproteins are structural protein components of lipoproteins that play a critical role in lipid transportation and metabolism [10]. There are six types of apolipoproteins, among which apolipoprotein A1 and B have received widespread attention in the context of psoriasis [8,10]. However, no consensus on the association between psoriasis and apolipoprotein A1 and B levels was exist [11,12]. Notably, obesity is an independent risk factor for both psoriasis and dyslipidemia [4,13]. Thus, the association between psoriasis and apolipoprotein A1 and B levels might be obscured by obesity.
This study aimed to examine the association between psoriasis and serum apolipoprotein A1 and B levels. Additionally, we conducted subgroup analyses of the studies according to the psoriasis type and matched body mass index (BMI).
2. Materials and methods
Our results were reported in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement (ID: CRD42022352229) [14].
2.1. Search strategy
We systematically searched PubMed, Embase, and Cochrane Library databases for eligible studies published before August 10, 2023. The literature search combined the medical subject headings terms and free words related to psoriasis and apolipoproteins. We restricted the search to human studies and English articles (Supplemental Appendix 1). FW and YW independently screened the search results and assessed their eligibility for inclusion. Disagreements were resolved by consulting X–S Y.
2.2. Inclusion and exclusion criteria
The included studies must fulfill the following criteria: 1) adult patients diagnosed with psoriasis; 2) a control group composed of individuals who reported being healthy; and 3) the study included data on serum apolipoprotein A1 and B levels. Letters, editorials, books, or studies that did not provide sufficient data were excluded.
2.3. Data extraction and quality assessment
We extracted the following data from included studies: first author, location, publication year, study type, sample size, BMI, age, psoriasis type, psoriasis area and severity index (PASI), and serum apolipoprotein A1 and B levels. The Newcastle-Ottawa Scale (NOS) was utilized for the evaluation of case-control and cohort studies, which consist of eight items [15,16]. The Agency for Healthcare Research and Quality (AHRQ) was utilized for the evaluation of cross-sectional studies, which include 11 items [17]. Two reviewers (FW and YW) independently performed the data extraction and risk of bias assessment. Disagreements were resolved by consulting a third reviewer (X–S Y).
2.4. Statistical analysis
To assess the differences in serum apolipoprotein A1 and B levels between patients with psoriasis and healthy controls, we reported the results as weighted mean differences (WMDs) with 95 % confidence intervals (CIs). The threshold for statistical significance was defined as P < 0.05. Heterogeneity was assessed using Cochran's Q and I2 statistics. In Cochran's Q statistic, statistical significance was set at P < 0.1. For the I2 statistic, we considered I2 > 50 % to represent substantial heterogeneity [5]. We adopted a random-effects model for the meta-analyses because this model provides more conservative results [18]. We evaluated publication bias using funnel plots and Egger's test [19]. Additionally, we conducted a sensitivity analysis by sequentially removing each study [20]. We conducted subgroup analyses of the studies according to matched BMI and the psoriasis type. All analyses were conducted using Stata 15.
3. Results
3.1. Search results
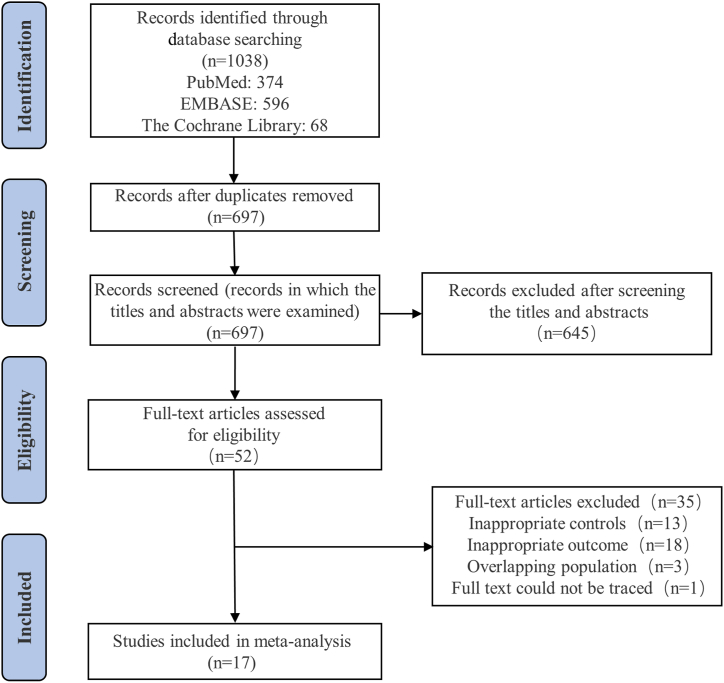
Our literature search identified 697 records after removing duplicates, of which 645 were excluded after screening the title and abstract. After examining the full text, 17 studies [[21], [22], [23], [24], [25], [26], [27], [28], [29], [30], [31], [32], [33], [34], [35], [36], [37]] with a total of 2467 participants were included. Fig. 1 and Supplemental Appendix 2 illustrate the detailed information of the literature selection procedure.
Fig. 1.
Flow diagram for the study selection process.
3.2. Study characteristics and quality
Among the 2467 participants, 1346 had psoriasis and 1121 were healthy controls. The mean age of the participants was 33–50.45 years, mean BMI was 20.88–29.78 kg/m2, and mean PASI of the patients with psoriasis was 3.04–28.07. Fourteen studies reported on serum apolipoprotein A1 levels, while 15 reported on serum apolipoprotein B levels. Twelve case-control and five cross-sectional studies were included. The characteristics of the included studies and the RoB assessed by NOS or AHRQ are summarized in Table 1.
Table 1.
Baseline characteristics of the included studies.
| Study | Design | Location | Pso Group |
Control Group |
Outcome | Quality | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N (F/M) | Age (year) | BMI (kg/m2) | Pso type | PASI | N (F/M) | Age (year) | BMI (kg/m2) | |||||
| Aguilar Martinez et al., 198917 | CCS | Spain | 61 (28/33) | 47.8 | NR | Pso | NR | 34 (19/15) | 44 | NR | Apo A1, Apo B | 4 |
| Seçkin et al., 199418 | CCS | Turkey | 32 (0/32) | 35.53 | NR | Vulgaris pso | NR | 13 (0/13) | 35.69 | NR | Apo A1, Apo B | 5 |
| Rocha-Pereira et al., 200119 | CCS | Portugal | 48 (18/30) | 47 | NR | Pso | NR | 40 (18/22) | 47 | NR | Apo A1, Apo B | 4 |
| Uyanik et al., 200220 * | CCS | Turkey | 72 (43/29) | 38.4 | NR | Pso | NR | 30 (NR) | NR | NR | Apo A1, Apo B | 4 |
| Vanizor Kural et al., 200321 | CCS | Turkey | 30 (17/13) | 34.2 | NR | Pso | 5.52 | 30 (15/15) | 36.7 | NR | Apo A1, Apo B | 5 |
| Tam et al., 200822 | CCS | Hong Kong | 102 (54/48) | 48.7 | 25.4 | PsA | NR | 82 (48/34) | 48.1 | 23.7 | Apo B | 6 |
| Nemati et al., 201323 * | CSS | Iran | 90 (41/49) | 34.7 | 25.5 | Pso without PsA | NR | 90 (41/49) | 33 | 25.3 | Apo A1, Apo B | 7 |
| Asefi et al., 201424 * | CCS | Iran | 100 (57/43) | 35.3 | 25.53 | Pso | 16.7 | 100 (56/44) | 35.7 | 24.66 | Apo A1, Apo B | 5 |
| Asha et al., 201425 * | CCS | India | 80 (20/60) | 40.58 | 24.76 | Pso | 15.6 | 80 (20/60) | 41.23 | 24.18 | Apo A1, Apo B | 4 |
| He et al., 201426 * | CSS | China | 25 (13/12) | 42.52 | 20.9 | Vulgaris pso | 15.24 | 25 (14/11) | 43.04 | 20.88 | Apo A1, Apo B | 6 |
| Pang et al., 201527 * | CCS | China | 86 (22/64) | 38.53 | 24.82 | Vulgaris and other | NR | 84 (14/70) | 41.33 | 23.87 | Apo A1 | 5 |
| Dinić et al., 201628 | CCS | Serbia | 66 (21/45) | 36.77 | 26.87 | Pso | NR | 20 (10/10) | 37.7 | 24.41 | Apo A1, Apo B | 3 |
| Kolliker Frers et al., 2018A29 * | CCS | Argentina | 9 (3/6) | 50.3 | 27 | Vulgaris pso | 5.86 | 9 (1/8) | 45.3 | 25.9 | Apo A1 | 5 |
| Kolliker Frers et al., 2018B29 | CCS | Argentina | 14 (3/11) | 49.4 | 29.1 | PsA | 3.04 | 9 (1/8) | 45.3 | 25.9 | Apo A1 | 5 |
| Colak et al., 201930 | CSS | Turkey | 50 (15/35) | 44.58 | 29.78 | PsA | 7.91 | 36 (11/25) | 43.08 | 26.31 | Apo A1, Apo B | 6 |
| Pietrzak et al., 2019A31 * | CCS | Poland | 31 (NR) | 40.68 | 27.52 | PsA | 28.07 | 60 (NR) | 41.02 | 26.31 | Apo B | 4 |
| Pietrzak et al., 2019B31 * | CCS | Poland | 62 (NR) | 40.87 | 26.48 | Vulgaris pso | 26 | 60 (NR) | 41.02 | 26.31 | Apo B | 4 |
| Arias de la Rosa et al., 202232 | CSS | Spain | 100 (60/40) | 46.75 | 28.51 | PsA | NR | 100 (56/44) | 44.59 | 25.07 | Apo B | 5 |
| Wang et al., 2022A33 | CSS | China | 136 (50/86) | 50.45 | 25.01 | PsA | 14.04 | 136(NR) | NR | NR | Apo A1, Apo B | 6 |
| Wang et al., 2022B33 | CSS | China | 152 (49/103) | 42.87 | 24.48 | Pso without PsA | 13.78 | 152(NR) | NR | NR | Apo A1, Apo B | 6 |
Apo, apolipoprotein; BMI, body mass index; CG, control group; CSS cross sectional study; CCS, case–control study; F, female; M, male; N, number; NR, not report; PASI, Psoriasis Area and Severity Index; Pso, psoriasis; PsA, psoriatic arthritis.
*The study matched BMI.
3.3. Association between psoriasis and serum apolipoprotein A1
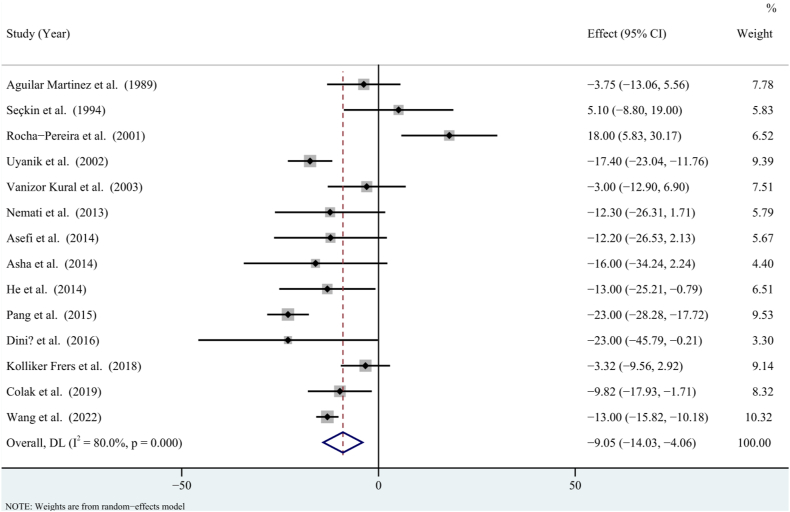
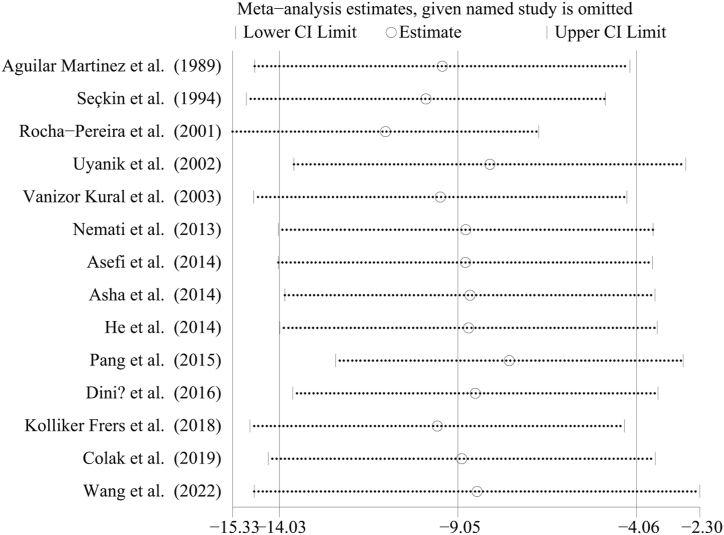
A total of 1930 participants were selected from fourteen studies which compared the levels of serum apolipoprotein A1 in patients with psoriasis and healthy controls. The pooled WMD was −9.05 (95 % CI = −14.03, −4.06, P < 0.001) (Fig. 2), with statistical heterogeneity among the studies (P < 0.001; I2 = 80 %). The sensitivity analysis revealed that removing any individual study did not significantly change the overall result (Fig. 3).
Fig. 2.
Forest plot for meta-analysis on the association between psoriasis and serum apolipoprotein A1 (weighted mean difference). CI: confidence interval; I2: inconsistency.
Fig. 3.
Sensitivity analysis of the association between psoriasis and serum apolipoprotein A1. CI: confidence interval.
3.4. Association between psoriasis and serum apolipoprotein B
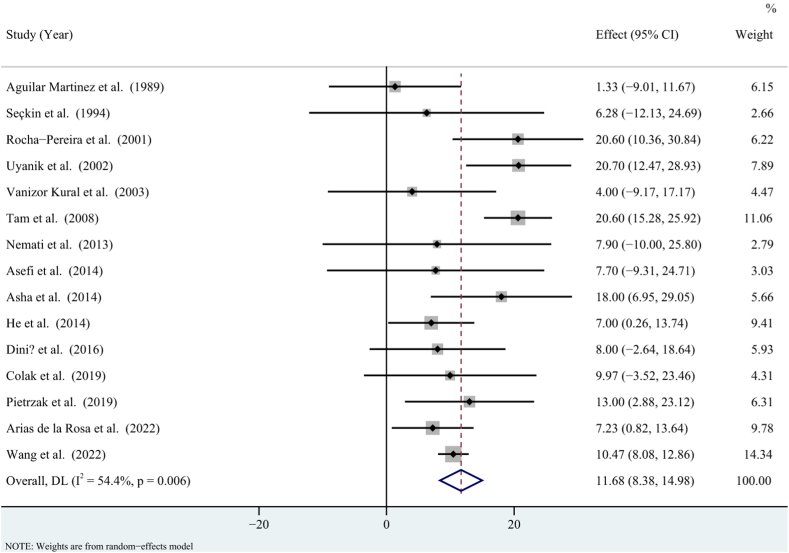
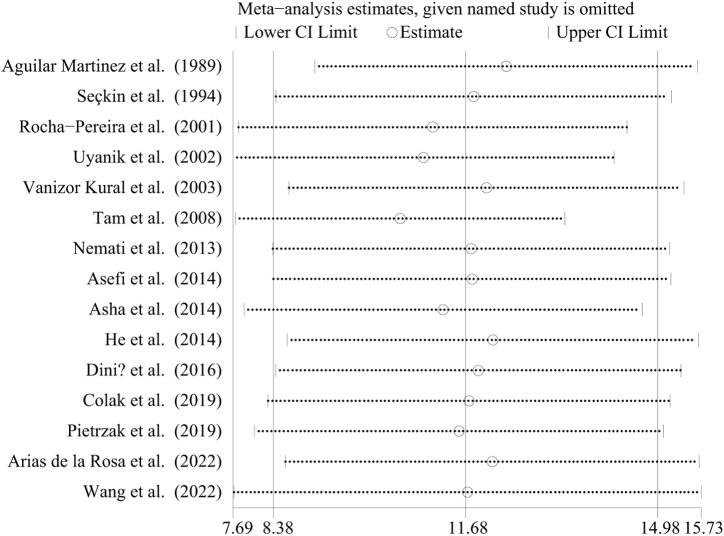
Fifteen studies involving 2265 participants reported the levels of serum apolipoprotein B in patients with psoriasis and healthy controls. The pooled WMD was 11.68 (95 % CI = 8.38, 14.98, P < 0.001) (Fig. 4), with statistical heterogeneity among the studies (P = 0.006; I2 = 54.4 %). The sensitivity analysis revealed that removing any individual study did not significantly change the overall result (Fig. 5).
Fig. 4.
Forest plot for meta-analysis on the association between psoriasis and serum apolipoprotein B (weighted mean difference). CI: confidence interval; I2: inconsistency.
Fig. 5.
Sensitivity analysis of the association between psoriasis and serum apolipoprotein B. CI: confidence interval.
3.5. Subgroup analyses
In the subgroup analysis after matching BMI, the findings regarding the association of psoriasis with serum apolipoprotein A1 (WMD = −14.07; 95 % CI = −20.75, −7.39, P < 0.001; I2 = 74.3 %) (Supplementary Fig. 1) and B (WMD = 13.07; 95 % CI = 7.66, 18.48, P < 0.001; I2 = 36.5 %) (Supplementary Fig. 2) were consistent with the overall results. The subgroup analysis according to the presence or absence of psoriatic arthritis showed that serum apolipoprotein A1 levels were significantly decreased in psoriasis with (WMD = −11.29; 95 % CI = −14.43, −8.05, P < 0.001; I2 = 0 %) and without arthritis (WMD = −8.69; 95 % CI = −16.97, −0.42, P = 0.039; I2 = 85.8 %) (Supplementary Fig. 3); whereas the serum apolipoprotein B levels were significantly increased in psoriasis with (WMD = 13.57; 95 % CI = 7.74, 19.4; P < 0.001; I2 = 71.4 %) and without arthritis (WMD = 9.21; 95 % CI = 6.42, 12; P < 0.001; I2 = 0 %) (Supplementary Fig. 4).
3.6. Publication bias
For serum apolipoprotein A1 levels, the funnel plot (Supplementary Fig. 5) showed no obvious asymmetry. Furthermore, the Egger's test results were not statistically significant (P = 0.219; Supplementary Fig. 6). For serum apolipoprotein B, an asymmetric funnel plot (Supplementary Fig. 7) showed that publication bias might exist, although the Egger's test did not reach statistical evidence (P = 0.966; Supplementary Fig. 8).
4. Discussion
This study found that patients with psoriasis showed lower serum apolipoprotein A1 and higher serum apolipoprotein B levels than healthy controls. Meanwhile, two subgroup analyses supported this result. Specifically, a significant association between psoriasis and serum apolipoprotein A1 and B levels remained in subgroup analyses according to matched BMI and presence or absence of psoriatic arthritis. Our results revealed that patients with psoriasis may be at risk for dyslipidemia, which emphasizes the need for lipid metabolism screening in patients with psoriasis.
Psoriasis is correlated with multiple comorbidities, and early identification and management of these comorbidities may be a crucial step in the holistic approach to patient health [4]. The correlation between psoriasis and metabolic diseases has been shown in many epidemiological studies and has also been found in pediatric psoriasis [9,38]. High-density lipoprotein contains the highest proportion of apolipoproteins to lipids and is widely known for its potential anti-atherosclerotic, anti-inflammatory, immunomodulatory, and antioxidant functions [[39], [40], [41]]. Apolipoprotein A1 is the principal protein component of high-density lipoprotein and can stabilize all high-density lipoprotein subclasses due to its conformational adaptability [42]. Therefore, the functions of high-density lipoprotein are closely linked to apolipoprotein A1 [40]. Lipid function is reported to depend more on structural and functional alterations than the level of lipid in psoriasis [43]. Our results revealed that psoriasis was associated with decreased serum apolipoprotein A1 levels, suggesting that psoriasis may be associated with impaired high-density lipoprotein functions. Considering the growing evidence suggesting that the simple concentration of high-density lipoprotein may not always reflect its functions, our results may partially explain the presence of multiple comorbidities in psoriasis [44]. Similarly, apolipoprotein B is a critical structural protein of atherogenic lipoproteins, and evidence shows that apolipoprotein B is a more reliable indicator of cardiovascular risk than either low-density lipoprotein cholesterol or total cholesterol [45]. Our results revealed an association between psoriasis and increased serum apolipoprotein B levels, suggesting the importance of screening lipid metabolism in patients with psoriasis. A previously published meta-analysis investigated similar topics, and revealed no significant correlation between psoriasis and serum apolipoprotein A1 levels [46]. Our study included more studies with approximately double the sample size, and revealed that psoriasis was associated with decreased serum apolipoprotein A1 levels. Meanwhile, we added sensitivity analyses to demonstrate the robustness of our results and we added subgroup analyses, which were not conducted in the published meta-analysis. Obesity is an independent risk factor for psoriasis, which leads to worse clinical outcomes [47,48]. It is well known that obesity can impair lipid metabolism; therefore, we conducted a subgroup analysis of studies for matched BMI [49]. The findings were consistent with the overall result. Our study adds to the accumulating evidence that patients with psoriasis may be at risk for dyslipidemia.
While the exact pathogenesis of psoriasis remains unclear, it is largely attributed to chronic inflammation. To some extent, psoriasis is also regarded as the initiator of the systemic inflammation [50]. Specifically, T cells and their secretion of inflammatory cytokines, such as TNF-α, largely mediate the development of psoriasis [51,52]. Some studies have revealed a significant negative correlation between serum apolipoprotein A1 levels and some systemic markers of inflammation [53,54]. TNF-α is reported to affect the lipid profile by decreasing the concentration of apolipoproteins in chronic inflammatory states, and TNF-α inhibitors may improve dyslipidemia in patients with psoriasis [55,56]. Other inflammatory cytokines, such as IL-1 and IL-6, have also been reported to inhibit apolipoprotein A1 synthesis by upregulating pentraxin expression [57]. Hence, it is acceptable to speculate that psoriasis and dyslipidemia may share common inflammatory pathways, which may underlie the etiology of psoriasis affecting dyslipidemia [58]. Moreover, psoriasis, as a driver of systemic inflammation, contributes to increased lipoprotein oxidation, manifesting as oxidatively modified lipoproteins. This phenomenon can lead to cholesterol accumulation and impaired lipid metabolism [59]. Notably, dyslipidemia may also play an important role in the pathogenesis of psoriasis, and high-cholesterol levels may exacerbate the inflammatory state of psoriasis [60]. Meanwhile, apolipoprotein B was reported to exacerbate inflammation by inducing TNF-α and IL-6 production through the p38 mitogen-activated protein kinase and NF-κB pathways [53]. Apolipoprotein B may be an important independent immunomodulator linking lipid metabolism to local and systemic inflammatory responses [53,61]. Thus, there may be a bidirectional relationship between psoriasis and abnormal lipid metabolism. Considering the beneficial effects of early identification and management of comorbidities, screening for lipid metabolism may have positive clinical implications for psoriatic patients, regardless of the bidirectional relationship between psoriasis and dyslipidemia.
To our knowledge, this study is the largest meta-analysis to investigate the relationship between psoriasis and serum apolipoprotein A1 and B levels. This meta-analysis followed the PRISMA statement and revealed that patients with psoriasis may be at risk for dyslipidemia. However, our study had some limitations. First, the majority of the included studies is a case-control design. Consequently, establishing the temporal or causal relationships between psoriasis and serum apolipoprotein A1 and B levels is difficult. Second, the included studies showed evidence of heterogeneity, which may be linked to the quality and clinical characteristics of the included studies, as well as other factors. Third, although we performed subgroup analyses of the studies according to the psoriasis type and matched BMI, we failed to match other possible influences owing to limited data availability. Similarly, limited data restricted our ability to investigate sex differences in the relationship between psoriasis and serum apolipoprotein A1 and B levels. In addition, among the included studies, only eight studies reported PASI scores, half of which were considered moderate psoriasis, as assessed by the mean PASI score. Our study failed to investigate the effect of psoriasis severity on serum apolipoprotein A1 and B levels. To overcome these limitations, further studies are required to clarify our findings.
5. Conclusions
Our study revealed that psoriasis is associated with lower serum apolipoprotein A1 and higher serum apolipoprotein B levels than healthy controls. Our results also show that patients with psoriasis may be at risk for dyslipidemia, which emphasizes the need for lipid metabolism screening in psoriatic patients. Further well-designed controlled studies are warranted to extend our findings.
Funding source
This work was supported by Scientific Research Fund project of the First Clinical Medical College of Yunnan University of Chinese Medicine (2022YB05), Yunnan Provincial Science and Technology Department-Applied Basic Research Joint Special Funds of Chinese Medicine (202101AZ070001-168), and Health Commission of Yunnan Province-Scientific and Technological Talents and Platform Program Academician (Expert) Workstation Project (202005AF150075).
Availability of data and materials
Data will be made available on request.
CRediT authorship contribution statement
Fang Wang: Conceptualization, Data curation, Formal analysis, Project administration, Writing – original draft, Writing – review & editing. Yuan Wang: Conceptualization, Data curation, Formal analysis, Project administration, Writing – original draft, Writing – review & editing. Xiangyi Kong: Formal analysis, Writing – original draft, Writing – review & editing. Jing Mu: Formal analysis, Project administration, Writing – original draft. Zhimin Wang: Writing – original draft. Xuesong Yang: Conceptualization, Data curation, Formal analysis, Project administration, Writing – original draft, Writing – review & editing. Jianzhou Ye: Conceptualization, Project administration, Supervision, Writing – review & editing.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
We would like to deliver our thanks to Project of first-class discipline construction in Yunnan Province for the support of this article.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.heliyon.2023.e21168.
Contributor Information
Xuesong Yang, Email: xuesongyang123@163.com.
Jianzhou Ye, Email: jianzhouye123@163.com.
Appendix A. Supplementary data
The following is the supplementary data to this article:
References
- 1.Moini J.A., Ayati M.H., Nadiri A.A., Nasimi D.A.R. Efficacy of hydrotherapy, spa therapy, and balneotherapy for psoriasis and atopic dermatitis: a systematic review. Int. J. Dermatol. 2022 doi: 10.1111/ijd.16080. [DOI] [PubMed] [Google Scholar]
- 2.Wang Z., Zhang G., Zhang H., Li L. Xiaoyin jiedu granules may alleviate psoriasis-like skin diseases in mice by regulating sphingosine 1-phosphate receptor expression and reducing th17 cells. Heliyon. 2023;9 doi: 10.1016/j.heliyon.2023.e19109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Armstrong A.W., Mehta M.D., Schupp C.W., Gondo G.C., Bell S.J., Griffiths C. Psoriasis prevalence in adults in the United States. Jama Dermatol. 2021;157:940–946. doi: 10.1001/jamadermatol.2021.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Takeshita J., Grewal S., Langan S.M., Mehta N.N., Ogdie A., Van Voorhees A.S. Gelfand JM: psoriasis and comorbid diseases: epidemiology. J. Am. Acad. Dermatol. 2017;76:377–390. doi: 10.1016/j.jaad.2016.07.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fu Y., Lee C.H., Chi C.C. Association of psoriasis with inflammatory bowel disease: a systematic review and meta-analysis. Jama Dermatol. 2018;154:1417–1423. doi: 10.1001/jamadermatol.2018.3631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Paksoy T., Ustaoglu G., Yaman D., Arioz O., Demirci M., Unlu O., Avci E., Polat M. The link between total antioxidant status, total oxidant status, arylesterase activity, and subgingival microbiota in psoriasis patients. Int. J. Dermatol. 2022 doi: 10.1111/ijd.16353. [DOI] [PubMed] [Google Scholar]
- 7.Armstrong E.J., Krueger J.G. Lipoprotein metabolism and inflammation in patients with psoriasis. Am. J. Cardiol. 2016;118:603–609. doi: 10.1016/j.amjcard.2016.05.060. [DOI] [PubMed] [Google Scholar]
- 8.Pietrzak A., Michalak-Stoma A., Chodorowska G., Szepietowski J.C. Lipid disturbances in psoriasis: an update. Mediat. Inflamm. 2010;2010 doi: 10.1155/2010/535612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Miller I.M., Skaaby T., Ellervik C., Jemec G.B. Quantifying cardiovascular disease risk factors in patients with psoriasis: a meta-analysis. Brit J Dermatol. 2013;169:1180–1187. doi: 10.1111/bjd.12490. [DOI] [PubMed] [Google Scholar]
- 10.Li L., Liu M.S., Li G.Q., Tang J., Liao Y., Zheng Y., Guo T.L., Kang X., Yuan M.T. Relationship between apolipoprotein superfamily and Parkinson's disease. Chinese Med J-Peking. 2017;130:2616–2623. doi: 10.4103/0366-6999.217092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ma C., Schupp C.W., Armstrong E.J., Armstrong A.W. Psoriasis and dyslipidemia: a population-based study analyzing the national health and nutrition examination survey (nhanes) J Eur Acad Dermatol. 2014;28:1109–1112. doi: 10.1111/jdv.12232. [DOI] [PubMed] [Google Scholar]
- 12.Ma L., Li M., Wang H., Li Y., Bai B. High prevalence of cardiovascular risk factors in patients with moderate or severe psoriasis in northern China. Arch. Dermatol. Res. 2014;306:247–251. doi: 10.1007/s00403-013-1437-3. [DOI] [PubMed] [Google Scholar]
- 13.Kyriakou A., Patsatsi A., Sotiriadis D., Goulis D.G. Serum leptin, resistin, and adiponectin concentrations in psoriasis: a meta-analysis of observational studies. Dermatology. 2017;233:378–389. doi: 10.1159/000481882. [DOI] [PubMed] [Google Scholar]
- 14.Moher D., Liberati A., Tetzlaff J., Altman D.G. Preferred reporting items for systematic reviews and meta-analyses: the prisma statement. Ann. Intern. Med. 2009;151:264–269. doi: 10.7326/0003-4819-151-4-200908180-00135. W64. [DOI] [PubMed] [Google Scholar]
- 15.Lo C.K., Mertz D., Loeb M. Newcastle-ottawa scale: comparing reviewers' to authors' assessments. BMC Med. Res. Methodol. 2014;14:45. doi: 10.1186/1471-2288-14-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Su L., Meng Y.H., Zhang S.Z., Cao Y., Zhu J., Qu H., Jiao Y.Z. Association between obstructive sleep apnea and male serum testosterone: a systematic review and meta-analysis. Andrology-Us. 2022;10:223–231. doi: 10.1111/andr.13111. [DOI] [PubMed] [Google Scholar]
- 17.Zeng X., Zhang Y., Kwong J.S., Zhang C., Li S., Sun F., Niu Y., Du L. The methodological quality assessment tools for preclinical and clinical studies, systematic review and meta-analysis, and clinical practice guideline: a systematic review. J. Evid. Base Med. 2015;8:2–10. doi: 10.1111/jebm.12141. [DOI] [PubMed] [Google Scholar]
- 18.Laird N.M., Mosteller F. Some statistical methods for combining experimental results. Int J Technol Assess. 1990;6:5–30. doi: 10.1017/s0266462300008916. [DOI] [PubMed] [Google Scholar]
- 19.Langhorne P. Bias in meta-analysis detected by a simple, graphical test. Prospectively identified trials could be used for comparison with meta-analyses. BMJ Br. Med. J. (Clin. Res. Ed.) 1998;316:471. [PMC free article] [PubMed] [Google Scholar]
- 20.Tian D., Zhao X., Ning Z., Gong Z., Wu J., Wang X. Migraine and risk of rheumatoid arthritis: a systematic review and meta-analysis of observational studies. Heliyon. 2023;9 doi: 10.1016/j.heliyon.2023.e18430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Martinéz A.A., Rodriguez P.G., Antunez P.A., Gil M.C.C., Gonzalez F.U., Pérez A.G. Serum levels of apolipoproteins ai, ah and b in psoriasis. Dermatol. 1989;179:200–201. doi: 10.1159/000248360. [DOI] [PubMed] [Google Scholar]
- 22.Seckin D., Tokgozoglu L., Akkaya S. Are lipoprotein profile and lipoprotein (a) levels altered in men with psoriasis? J. Am. Acad. Dermatol. 1994;31:445–449. doi: 10.1016/s0190-9622(94)70208-x. [DOI] [PubMed] [Google Scholar]
- 23.Rocha-Pereira P., Santos-Silva A., Rebelo I., Figueiredo A., Quintanilha A., Teixeira F. Dislipidemia and oxidative stress in mild and in severe psoriasis as a risk for cardiovascular disease. Clin. Chim. Acta. 2001;303:33–39. doi: 10.1016/s0009-8981(00)00358-2. [DOI] [PubMed] [Google Scholar]
- 24.Uyanik B.S., Ari Z., Onur E., Gunduz K., Tanulku S., Durkan K. Serum lipids and apolipoproteins in patients with psoriasis. Clin. Chem. Lab. Med. 2002;40:65–68. doi: 10.1515/CCLM.2002.013. [DOI] [PubMed] [Google Scholar]
- 25.Vanizor K.B., Orem A., Cimsit G., Uydu H.A., Yandi Y.E., Alver A. Plasma homocysteine and its relationships with atherothrombotic markers in psoriatic patients. Clin. Chim. Acta. 2003;332:23–30. doi: 10.1016/s0009-8981(03)00082-2. [DOI] [PubMed] [Google Scholar]
- 26.Tam L.S., Tomlinson B., Chu T.T., Li M., Leung Y.Y., Kwok L.W., Li T.K., Yu T., Zhu Y.E., Wong K.C., Kun E.W., Li E.K. Cardiovascular risk profile of patients with psoriatic arthritis compared to controls--the role of inflammation. Rheumatology. 2008;47:718–723. doi: 10.1093/rheumatology/ken090. [DOI] [PubMed] [Google Scholar]
- 27.Nemati H., Khodarahmi R., Rahmani A., Ebrahimi A., Amani M., Eftekhari K. Serum lipid profile in psoriatic patients: correlation between vascular adhesion protein 1 and lipoprotein (a) Cell Biochem. Funct. 2013;31:36–40. doi: 10.1002/cbf.2857. [DOI] [PubMed] [Google Scholar]
- 28.Asefi M., Vaisi-Raygani A., Khodarahmi R., Nemati H., Rahimi Z., Vaisi-Raygani H., Tavilani H., Pourmotabbed T., Methylentetrahydrofolatereductase rs1801133) polymorphism and psoriasis: contribution to oxidative stress, lipid peroxidation and correlation with vascular adhesion protein 1, preliminary report. J Eur Acad Dermatol. 2014;28:1192–1198. doi: 10.1111/jdv.12262. [DOI] [PubMed] [Google Scholar]
- 29.Asha K., Sharma S.B., Singal A., Aggarwal A. Association of carotid intima-media thickness with leptin and apoliprotein b/apoliprotein a-i ratio reveals imminent predictors of subclinical atherosclerosis in psoriasis patients. Acta Med. 2014;57:21–27. doi: 10.14712/18059694.2014.4. [DOI] [PubMed] [Google Scholar]
- 30.He L., Qin S., Dang L., Song G., Yao S., Yang N., Li Y. Psoriasis decreases the anti-oxidation and anti-inflammation properties of high-density lipoprotein. Biochim. Biophys. Acta. 2014;1841:1709–1715. doi: 10.1016/j.bbalip.2014.09.008. [DOI] [PubMed] [Google Scholar]
- 31.Pang X., Lin K., Liu W., Zhang P., Zhu S. Characterization of the abnormal lipid profile in Chinese patients with psoriasis. Int. J. Clin. Exp. Pathol. 2015;8:15280–15284. [PMC free article] [PubMed] [Google Scholar]
- 32.Dinic M., Zecevic R.D., Hajdukovic Z., Mijuskovic M., Duric P., Jovic Z., Grdinic A., Petrovic M., Terzic B., Pejovic J., Kandolf-Sekulovic L. Psoriasis is the independent factor for early atherosclerosis: a prospective study of cardiometabolic risk profile. Vojnosanit. Pregl. 2016;73:1094–1101. doi: 10.2298/VSP150510134D. [DOI] [PubMed] [Google Scholar]
- 33.Kolliker F.R., Cosentino V., Tau J., Kerzberg E.M., Urdapilleta A., Chiocconi M., Kogan N., Otero-Losada M., Capani F. Immune-mediated inflammation promotes subclinical atherosclerosis in recent-onset psoriatic arthritis patients without conventional cardiovascular risk factors. Front. Immunol. 2018;9:139. doi: 10.3389/fimmu.2018.00139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Colak S., Omma A., Sandikci S.C., Yucel C., Omma T., Turhan T. Vaspin, neutrophil gelatinase-associated lipocalin and apolipoprotein levels in patients with psoriatic arthritis. Bratisl Med J. 2019;120:65–69. doi: 10.4149/BLL_2019_010. [DOI] [PubMed] [Google Scholar]
- 35.Pietrzak A., Chabros P., Grywalska E., Kicinski P., Pietrzak-Franciszkiewicz K., Krasowska D., Kandzierski G. Serum lipid metabolism in psoriasis and psoriatic arthritis - an update. Arch. Med. Sci. 2019;15:369–375. doi: 10.5114/aoms.2018.74021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Arias D.L.R.I., Lopez-Montilla M.D., Roman-Rodriguez C., Perez-Sanchez C., Gomez-Garcia I., Lopez-Medina C., Ladehesa-Pineda M.L., Abalos-Aguilera M., Ruiz D., Patino-Trives A.M., Luque-Tevar M., Anon-Onate I., Perez-Galan M.J., Guzman-Ruiz R., Malagon M.M., Lopez-Pedrera C., Escudero-Contreras A., Collantes-Estevez E., Barbarroja N. The clinical and molecular cardiometabolic fingerprint of an exploratory psoriatic arthritis cohort is associated with the disease activity and differentially modulated by methotrexate and apremilast. J. Intern. Med. 2022;291:676–693. doi: 10.1111/joim.13447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang B., Deng H., Hu Y., Han L., Huang Q., Fang X., Yang K., Wu S., Zheng Z., Yawalkar N., Zhang Z., Yan K. The difference of lipid profiles between psoriasis with arthritis and psoriasis without arthritis and sex-specific downregulation of methotrexate on the apolipoprotein b/apolipoprotein a-1 ratio. Arthritis Res. Ther. 2022;24:17. doi: 10.1186/s13075-021-02715-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cho S.I., Kim Y.E., Jo S.J. Association of metabolic comorbidities with pediatric psoriasis: a systematic review and meta-analysis. Ann. Dermatol. 2021;33:203–213. doi: 10.5021/ad.2021.33.3.203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Birner-Gruenberger R., Schittmayer M., Holzer M., Marsche G. Understanding high-density lipoprotein function in disease: recent advances in proteomics unravel the complexity of its composition and biology. Prog. Lipid Res. 2014;56:36–46. doi: 10.1016/j.plipres.2014.07.003. [DOI] [PubMed] [Google Scholar]
- 40.Paiva-Lopes M.J., Delgado A.J. Psoriasis-associated vascular disease: the role of hdl. J. Biomed. Sci. 2017;24:73. doi: 10.1186/s12929-017-0382-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wang H.H., Garruti G., Liu M., Portincasa P., Wang D.Q. Cholesterol and lipoprotein metabolism and atherosclerosis: recent advances in reverse cholesterol transport. Ann. Hepatol. 2017;16:s27–s42. doi: 10.5604/01.3001.0010.5495. [DOI] [PubMed] [Google Scholar]
- 42.Phillips M.C. New insights into the determination of hdl structure by apolipoproteins: thematic review series: high density lipoprotein structure, function, and metabolism. J. Lipid Res. 2013;54:2034–2048. doi: 10.1194/jlr.R034025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhang Z.Y., Jian Z.Y., Tang Y., Li W. The relationship between blood lipid and risk of psoriasis: univariable and multivariable mendelian randomization analysis. Front. Immunol. 2023;14 doi: 10.3389/fimmu.2023.1174998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Rosenson R.S., Brewer H.J., Ansell B., Barter P., Chapman M.J., Heinecke J.W., Kontush A., Tall A.R., Webb N.R. Translation of high-density lipoprotein function into clinical practice: current prospects and future challenges. Circulation. 2013;128:1256–1267. doi: 10.1161/CIRCULATIONAHA.113.000962. [DOI] [PubMed] [Google Scholar]
- 45.Behbodikhah J., Ahmed S., Elyasi A., Kasselman L.J., De Leon J., Glass A.D., Reiss A.B. Apolipoprotein b and cardiovascular disease: biomarker and potential therapeutic target. Metabolites. 2021:11. doi: 10.3390/metabo11100690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ramezani M., Zavattaro E., Sadeghi M. Evaluation of serum lipid, lipoprotein, and apolipoprotein levels in psoriatic patients: a systematic review and meta-analysis of case-control studies. Postep Derm Alergol. 2019;36:692–702. doi: 10.5114/ada.2019.91420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Katsimbri P., Korakas E., Kountouri A., Ikonomidis I., Tsougos E., Vlachos D., Papadavid E., Raptis A., Lambadiari V. The effect of antioxidant and anti-inflammatory capacity of diet on psoriasis and psoriatic arthritis phenotype: nutrition as therapeutic tool? antioxidants-basel. 2021:10. doi: 10.3390/antiox10020157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Takeshita J. Psoriasis and comorbid diseases: epidemiology. J. Am. Acad. Dermatol. 2017;76:377–390. doi: 10.1016/j.jaad.2016.07.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Stafeev I., Sorkina E., Koksharova E., Tumanyan T., Sklyanik I., Menshikov M., Mayorov A., Parfyonova Y., Shestakova M. The effects of glucagon-like peptide type 1 (glp-1) and its analogues in adipose tissue: is there a way to thermogenesis? Curr. Mol. Med. 2021;21:527–538. doi: 10.2174/1566524020666201201095029. [DOI] [PubMed] [Google Scholar]
- 50.Hu S.C., Lan C.E. Psoriasis and cardiovascular comorbidities: focusing on severe vascular events, cardiovascular risk factors and implications for treatment. Int. J. Mol. Sci. 2017;18 doi: 10.3390/ijms18102211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Xie X., Zhang L., Li X., Liu W., Wang P., Lin Y., Han X., Li P. Liangxue jiedu formula improves psoriasis and dyslipidemia comorbidity via pi3k/akt/mtor pathway. Front. Pharmacol. 2021;12 doi: 10.3389/fphar.2021.591608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sadafi S., Ebrahimi A., Sadeghi M., Emami A.O. Association between tumor necrosis factor-alpha polymorphisms (rs361525, rs1800629, rs1799724, 1800630, and rs1799964) and risk of psoriasis in studies following hardy-weinberg equilibrium: a systematic review and meta-analysis. Heliyon. 2023;9 doi: 10.1016/j.heliyon.2023.e17552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wang B., Deng H., Hu Y., Han L., Huang Q., Fang X., Yang K., Wu S., Zheng Z., Yawalkar N., Zhang Z., Yan K. The difference of lipid profiles between psoriasis with arthritis and psoriasis without arthritis and sex-specific downregulation of methotrexate on the apolipoprotein b/apolipoprotein a-1 ratio. Arthritis Res. Ther. 2022;24:17. doi: 10.1186/s13075-021-02715-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sirnio P., Vayrynen J.P., Klintrup K., Makela J., Makinen M.J., Karttunen T.J., Tuomisto A. Decreased serum apolipoprotein a1 levels are associated with poor survival and systemic inflammatory response in colorectal cancer. SCI REP-UK. 2017;7:5374. doi: 10.1038/s41598-017-05415-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Shih C.M., Chen C.C., Chu C.K., Wang K.H., Huang C.Y., Lee A.W. The roles of lipoprotein in psoriasis. Int. J. Mol. Sci. 2020:21. doi: 10.3390/ijms21030859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hagino T., Saeki H., Fujimoto E., Kanda N. Effects of biologic therapy on laboratory indicators of cardiometabolic diseases in patients with psoriasis. J. Clin. Med. 2023:12. doi: 10.3390/jcm12051934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kravitz M.S., Pitashny M., Shoenfeld Y. Protective molecules--c-reactive protein (crp), serum amyloid p (sap), pentraxin3 (ptx3), mannose-binding lectin (mbl), and apolipoprotein a1 (apo a1), and their autoantibodies: prevalence and clinical significance in autoimmunity. J. Clin. Immunol. 2005;25:582–591. doi: 10.1007/s10875-005-7828-2. [DOI] [PubMed] [Google Scholar]
- 58.Xiao Y., Jing D., Tang Z., Peng C., Yin M., Liu H., Chen X., Shen M. Serum lipids and risk of incident psoriasis: a prospective cohort study from the UK biobank study and mendelian randomization analysis. J. Invest. Dermatol. 2022 doi: 10.1016/j.jid.2022.06.015. [DOI] [PubMed] [Google Scholar]
- 59.Nowowiejska J., Baran A., Flisiak I. Aberrations in lipid expression and metabolism in psoriasis. Int. J. Mol. Sci. 2021:22. doi: 10.3390/ijms22126561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Shih C.M., Huang C.Y., Wang K.H., Huang C.Y., Wei P.L., Chang Y.J., Hsieh C.K., Liu K.T., Lee A.W. Oxidized low-density lipoprotein-deteriorated psoriasis is associated with the upregulation of lox-1 receptor and il-23 expression in vivo and in vitro. Int. J. Mol. Sci. 2018;19 doi: 10.3390/ijms19092610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lee J.Y., Kang M.J., Choi J.Y., Park J.S., Park J.K., Lee E.Y., Lee E.B., Pap T., Yi E.C., Song Y.W. Apolipoprotein b binds to enolase-1 and aggravates inflammation in rheumatoid arthritis. Ann. Rheum. Dis. 2018;77:1480–1489. doi: 10.1136/annrheumdis-2018-213444. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data will be made available on request.