Introduction
Stromal cells are a heterogenous class of connective tissue cells that are integral to the function of any organ. 37 These cells can be obtained, isolated, and cultured from adipose tissue to generate an injectable derivative with an immunophenotype consistent with that of mesenchymal stem cells (MSCs), thus opening a variety of potential applications in regenerative medicine.12,14,33,39,40,54,56,64,66,70,71 Despite a myriad of proposed benefits, specific processing and harvest protocols can lead to substantial variation in the molecular and chemical makeup of the injectate. 43 In orthopaedics specifically, these adipose-derived therapies have been used as an adjunct to arthroscopic knee surgery, 8 for the treatment of aseptic nonunion, 55 and for various forms of tendinopathy and acute tendon injury. 51
Within foot and ankle surgery, adipose-derived mesenchymal stem cell (ADSC) application is in its infancy. Proposed applications include osteochondral lesions (OCLs) of the talus, 16 diabetic foot wounds, 35 end-stage osteoarthritis (OA) of the ankle in conjunction with arthroscopic debridement, 53 and as an adjunct to improve cartilage restoration after lateral displacement calcaneal osteotomy for varus ankle OA. 28 The purpose of the current work is to provide the foot and ankle practitioner with an understanding of the collection, isolation, and preparation of ADSCs. In addition, their potential therapeutic components and reported clinical efficacy will be reviewed.
Collection, Isolation, and Preparation
The specific harvest site and technique for the collection of ADSCs varies. For example, in the Lipogems technique, a skin incision is made in the flank area after injecting a tumescent solution (25 mL of 2% lidocaine and 0.5 mL epinephrine [1:1000] in 250 mL of normal saline) using a disposable 17-gauge blunt cannula connected to a Luer-lock 60-mL syringe. The tissue is then agitated followed by a 15-minute resting period to allow for adequate emulsification of the fatty tissues. The ADSCs are then harvested using a liposuction 13-gauge blunt cannula connected to a Vaclock 20-mL syringe. After processing, the final product is transferred into 10-mL syringes and can be injected into the target site. 53 ADSC harvesting is substantially more involved than the harvest required for other orthobiologic products, such as platelet-rich plasma, which involves a simple blood draw. Thus, considerations such as the comfort of liposuction techniques of the clinician who may be using these products are relevant to their clinical practice.
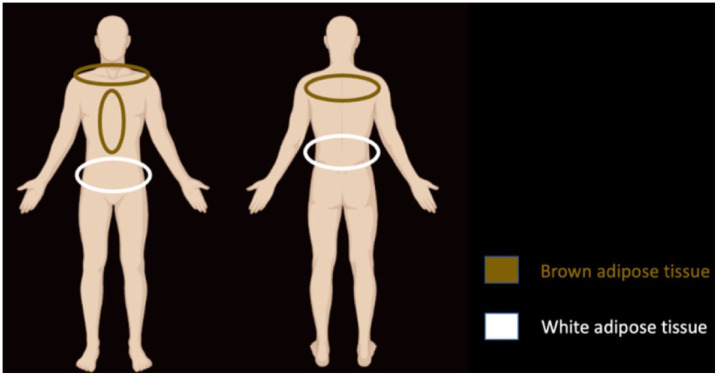
The substantial variation in the biomolecular composition of ADSC products is in large part due to the site from which they are harvested. ADSCs can be harvested from white or brown adipose tissue. 13 These sources differ both functionally and morphologically. Brown adipose tissue is far less abundant than white adipose tissue and is typically found in the neck, mediastinum, and interscapular areas (Figure 1). 17 Brown adipose tissue contains a high mitochondrial density and expresses uncoupling protein 1 (UCP1), a compound regulated by adrenergic signaling through sympathetic innervations. 15 Thus, brown adipose tissue is largely responsible for thermogenesis. Meanwhile, white adipocytes are unilocular, contain large lipid vacuoles, and function as a store of excess energy in the form of triglycerides. 57
Figure 1.
Areas of the body with a high concentration of brown adipose tissue (circled in brown) and a high concentration of white adipose tissue (circled in white).
ADSC composition can vary substantially based on brown vs white fat density in the collected sample. In murine white adipose tissue, multipotent stem cells are abundant. However, in brown adipose tissue, the stem cell number and differentiation potential are significantly reduced. 49 Less is known about ADSC differences between white adipose tissue and brown adipose tissue depots in humans. Preadipocyte and endothelial cell numbers have been found to differ between subcutaneous and omental white adipose depots in human subjects. 61 Moreover, differences in stem cell recovery have been noted based on harvest site, with greater numbers recovered from the arm when compared to the thigh, abdomen, and breast. 49
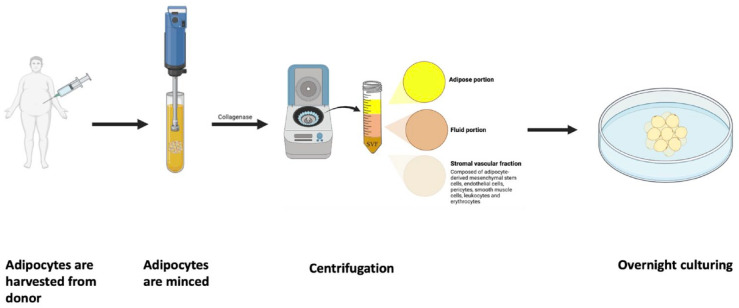
Numerous processing methodologies for ADSCs have been described, with a popular method for white adipose tissue isolation being described by Zuk et al. 74 In this protocol, human adipose tissue is obtained from elective liposuction via a hollow blunt-tipped cannula that is attached to gentle suction and moved through the adipose compartment to disrupt the fat tissue. 74 The adipose tissues are then minced, exposed to collagenase type II to undergo enzymatic digestion, and subject to centrifugation, resulting in a pellet referred to as the stromal vascular fraction (SVF). 74 The SVF contains ADSCs in addition to a variety of other components, including endothelial cells and progenitors, pericytes, smooth muscle cells, leukocytes, and erythrocytes. 72 After overnight culturing, ADSCs are isolated as the plastic-adherent population and removed from the other cellular components. Utilizing this preparation, stem cell yield is higher from adipose tissue than bone marrow: 1 g of aspirated adipose tissue yields approximately 3.5 × 105 to 1 × 106 ADSCs vs 500 to 5 × 104 of bone marrow-derived MSCs (BM-MSCs) isolated from 1 g of bone marrow aspirate. 11 These steps are outlined in Figure 2.
Figure 2.
The processing methodology for adipose-derived mesenchymal stem cells.
This process is not FDA-approved.
Of note, while the protocol described by Zuk et al is widely used in basic science investigation, this protocol and others that are used in the laboratory setting are not approved for use in humans by the Food and Drug Administration (FDA). Therefore, proprietary techniques have been developed to apply ADSCs in the clinical setting. For example, in 2010, Lipogems (Lipogems International, SpA, Milan, Italy) developed a technique to obtain microfragmented adipose tissue with an intact stromal vascular niche to isolate MSCs10,58 (Figure 3). 67 This product become clinically available in 2013, and other manufacturers subsequently developed ADSC harvesting and processing technologies, such as AutoPose Restore (Arthrex, Naples, FL).
Figure 3.
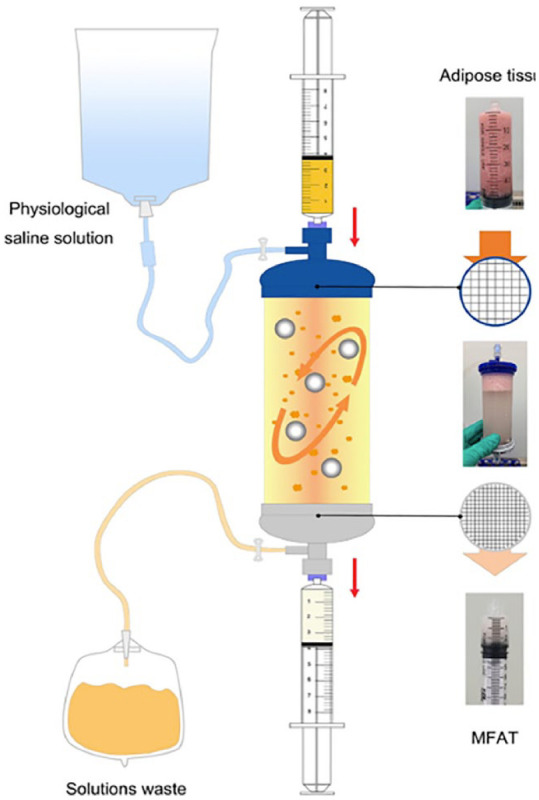
Mechanism of the FDA-approved Lipogems® device. This process involves the original adipose tissue being pushed through the blue filter for the first cluster reduction. The core device is then aggressively shaken several times until the saline solution washes off the residual blood and oil. The second cluster reduction is then performed to obtain micro-fragmented adipose tissue via the extrusion of the inner adipose clusters into a syringe. Image courtesy of Xu et al. 67 Abbreviations: FDA, Food and Drug Administration; MFAT, micro-fragmented adipose tissue.
Therapeutic Components of Adipose Tissue
Several large number of studies have been published in animal models outlining the various mechanisms by which ADSCs act on cell signaling.4,6,25,30,36,47 ADSCs exhibit multipotent differentiation, rapid proliferation, and self-renewal properties. 26 They have been found to differentiate into endothelial cells and promote vascularization through vascular endothelial growth factor (VEGF),20,73 and they exhibit potent immunomodulatory potential through both autocrine and paracrine mechanisms. 32 ADSCs induce the migration of fibroblasts and keratinocytes through Ki-67 expression and alpha-smooth muscle alpha-actin (αSMA) activation and can thus impact wound healing. 3 ADSCs have also been found to exhibit cartilage regenerative potential; specifically, the CD146+ subset has demonstrated the capacity for chondrogenic differentiation. 34 Importantly, in vitro studies have demonstrated that culture medium may impact chondrocyte differentiation of ADSCs. When cultured with growth factors such as insulin-like growth factor-1 (IGF-1), transforming growth factor-beta (TGF-β), or bone morphogenetic proteins (BMPs), ADSCs-derived chondrocytes exhibit the same type II collagen as mature chondrocytes.9,68
ADSCs also may impact bone deposition and growth. Bone modulatory potential is exhibited through the release of growth factors such as platelet-derived growth factor (PDGF), VEGF, and BMP-2. 44 ADSCs can also undergo osteogenic differentiation through major regulators such as Runx2 and Osterix (Sp7 transcription factor). 29 The Wnt signaling pathway serves as a regulatory factor between cellular lineages, guiding ADSCs away from adipogenic or chondrogenic lineage toward osteogenic lineages by increasing various signaling proteins. 50 Importantly, the mechanical environment in which ADSCs are placed will impact their osteogenic potential. For example, tensile strain through uniaxial or equiaxial stretching has been shown to increase Wnt signaling, favoring osteogenic cellular differentiation in ADSCs. 52 Thus, orthopaedists should recognize the contribution of the strain environment to the potency of ADSCs applied for various clinical use cases.
A recent study by Wang et al 65 demonstrated that adipose stem cells have a depot-dependent lineage preference. This rabbit study examined at subcutaneous adipose tissue (ScAT)– or infrapatellar fat pad (IPFP)–derived adipose stem cells. The authors concluded that ScAT- and IPFP-derived adipose stem cells of 4 donor-matched rabbits exhibited significant divergence in both proliferation and differentiation potential. Whereas the ScAT exhibited significantly higher proliferation and adipogenic capacity compared to donor-matched IPFP, stem cells from the IPFP displayed significantly higher chondrogenic potential in comparison to those from donor-matched ScAT. These results indicate that the harvest site of ADSCs is important, as different sites have different proliferation and differentiation potentials, which is an important consideration when determining the ideal harvest site.
Applications in Foot and Ankle Surgery
Despite a relative paucity of investigations related to ADSCs in the foot and ankle, several studies have been published in the last decade. Herein, we will highlight the recent literature regarding the application of ADSC injections in various pathologic conditions of the foot and ankle and provide summation statements regarding the current evidence-based recommendation for ADSCs in each of the respective pathologies. These studies are also summarized in Table 1.
Table 1.
A Summary of the Studies That Have Been Published Within the Past Decade Regarding the Use of Adipose Injections in Pathologies of the Foot and Ankle.
| Study | Pathology | Design | Number of Participants (Adipose-Derived Group) | Number of Participants (Control Group) | Outcome | Description of Results |
|---|---|---|---|---|---|---|
| Gusenoff et al 19 (2022) | PF | Prospective, randomized, crossover pilot study | 9 | 5 | Benefit | In group 1, function improved significantly on the MFPDI from 7 ± 2 at screening to 5 ± 3 at 6 mo postoperatively to 3 ± 2 at 12 mo postoperatively. Pain also improved in this group significantly from 12 ± 4 at screening to 7 ± 6 at 6 mo to 5 ± 5 at 12 mo. Both groups 1 and 2 had no significant change to the AOFAS scores at any time point. |
| Albano et al 2 (2017) | AT | Prospective, randomized, controlled trial | 21 (30 tendons) | 22 (28 tendons) | Benefit | The patients in the PRP group had a mean pretreatment VAS of 6.3 ± 1.3, which decreased to 1.9 ± 1.4 (P < .001) at the 6-mo follow-up. The patients in the adipose-derived SVF group had a mean pretreatment VAS score of 6.5 ± 1.5, which decreased to 1.6 ± 1.9 (P < .001) at the 6-mo follow-up. Furthermore, the adipose-derived SVF group had statistically significant increases in tendon thickness on both ultrasonograph and MRI scans, whereas the PRP group did not. Conversely, the PRP group had statistically significant signal intensity on MRI, whereas the adipose-derived SVF group did not. |
| Usuelli et al 60 (2018) | AT | Randomized, controlled clinical trial | 21 | 23 | Benefit | At the 15- and 30-d time points, the SVF group scored significantly better than the SVF group for the VAS, AOFAS, and VISA-A. However, there was no difference at 60, 120, and 180 d. |
| Kim et al 27 (2014) | OLT | Cohort study | 24 | 26 | Benefit | In the marrow stimulation group, the mean VAS score improved from 7.1 ± 1.2 to 3.9 ± 0.8, the mean AOFAS score improved from 68.5 ± 5.6 to 78.3 ± 4.9, and the mean Tegner score improved from 3.4 ± 0.6 to 3.5 ± 0.8. In the MSC group, the mean VAS score improved from 7.1 ± 0.8 to 3.2 ± 0.8, the mean AOFAS score improved from 67.7 ± 4.7 to 83.3 ± 7.0 and the mean Tegner score improved from 3.4 ± 0.5 to 3.9 ± 0.7. |
| Braile et al 5 (2021) | HR | Case report | 1 | 0 | Benefit | The patient’s preoperative, 6-mo, and 9-mo follow-up AOFAS pain scores were all 30. His AOFAS function score preoperatively was 35, which improved to 40 at 6-mo follow-up and was also 40 at 9-mo follow-up. His VAS score was 7 at baseline and improved to 5 at 6-mo follow-up. At 9-mo follow-up, his VAS score was 0. |
| Natali et al 41 (2021) | Ankle OA | Prospective, open-label, uncontrolled pilot study | 31 | 0 | Benefit | The AOFAS scores at preoperation, 6-mo, 12-mo, and 24-mo follow-up were 56.4 ± 17.52, 80.97 ± 9.01, 84.17 ± 12.23, and 66.5 ± 15.71, respectively. The FADI scores at preoperation, 6-mo, 12-mo, and 24-mo follow-up were 59.47 ± 16.99, 75.05 ± 8.78, 82.0 ± 11.39, and 71.83 ± 10.88, respectively. The VAS scores at preoperation, 6-mo, 12-mo, and 24-mo follow-up were 7.03 ± 0.95, 3.61 ± 0.92, 3.26 ± 0.63, and 4.35 ± 1.25, respectively. All results for AOFAS, FADI, and VAS were statistically significant aside from the AOFAS score at 24-mo follow-up. |
| Shimozono et al 53 (2021) | Ankle OA | Retrospective cohort study | 19 | 0 | Benefit | At mean follow-up of 14.3 mo, both the FAOS for pain and quality of life significantly increased from 48.8 and 20.1 preoperatively to 61.1 and 30.1, respectively. The mean VAS score also significantly improved from 6.1 at baseline to 3.8 at final follow-up. Notably, however, more than a third of patients were unsatisfied with their outcomes. |
Abbreviations: AOFAS, American Orthopaedic Foot & Ankle Society; AT, Achilles tendinopathy; FADI, Foot and Ankle Disability Index; FAOS, Foot and Ankle Outcome Score; HR, hallux rigidus; MSC, mesenchymal stem cells; MFPDI, Manchester Foot Pain and Disability Index; MRI, magnetic resonance imaging; OA, osteoarthritis; OLT, osteochondral lesions of the talus; PF, plantar fasciitis; PRP, platelet-rich plasma; SVF, stromal vascular fraction; VAS, visual analog scale; VISA-A, Victorian Institute of Sports Assessment–Achilles.
Plantar Fasciitis
One in 10 people will experience plantar fasciitis (PF) during their lifetime. 59 Risk factors for this condition include limited ankle dorsiflexion, increased body mass index, and standing for prolonged periods of time. 18 Patients commonly present with heel pain with their first steps in the morning. 7 Because of the lack of effective surgical options for this pathology, orthobiologics such as platelet-rich plasma (PRP) have been investigated for PF.23,23,69 ADSC injection has been proposed as another therapeutic option in refractory cases.
A prospective, randomized, crossover pilot study by Gusenoff et al 19 evaluated the efficacy of perforating fat injections for chronic PF. Adults with plantar fascia thickening of ≥4 mm who failed standard treatment were included in this study. The intervention group (group 1), which received a fat injection and was then followed for 12 months, was composed of 9 female patients, and was followed for 12 months. Group 2 was composed of 4 female and 1 male patient, who were observed for 6 months, injected with a perforating fat infection, and then followed for 6 months. The fat in this study was hand-harvested from an unspecified donor site and processed using the Coleman technique. A total of 2.6 ± 1.6 mL of fat was injected per foot at one of 2 sites. The authors used the Manchester Foot Pain and Disability Index (MFPDI) and American Orthopaedic Foot & Ankle Society (AOFAS) ankle-hindfoot scale scores to evaluate the clinical outcomes. In group 1, function and pain improved significantly as measured by the MFPDI from 7 ± 2 at screening to 5 ± 3 at 6 months postoperatively to 3 ± 12 at 12 months postoperatively. Pain also improved in this group significantly from 12 ± 4 at screening to 7 ± 6 at 6 months to 5 ± 5 at 12 months. Both groups 1 and 2 had no significant change to the AOFAS scores at any time point.
Although the authors concluded that perforating fat injections for chronic plantar fasciitis demonstrated significant improvement in pain, function, and plantar fascia thickness, there are several causes for concern with this study. First, the authors stated that the fat was injected in each foot at one of 2 sites; however, they did not specify these sites, which could have important implications for treatment efficacy. This lack of standardization makes the interpretation of these results difficult. Furthermore, given the lack of a control group in this study, it is difficult to appraise the true efficacy of these injections.
Achilles Tendinopathy
Achilles tendinopathy (AT) is associated with overuse and is characterized by cumulative tissue microtrauma. 63 AT can be refractory to treatment given poor blood supply to the region, 1 but there has been significant recent advancement in treatment options for this pathology. 62 Accordingly, adipose-derived SVF has become a popular area of research for the treatment of AT.
A prospective randomized controlled trial (RCT) by Albano et al 2 assessed the correlation between MRI and ultrasonography findings and clinical outcomes after intratendinous injection of leukocyte-rich PRP or adipose-derived SVF in patients with noninsertional AT. This study included 43 patients with unilateral or bilateral noninsertional AT (58 tendons overall). Of these, 22 patients (28 tendons) were assigned to the PRP group, and 21 patients (30 tendons) were assigned to the adipose-derived SVF group. The adipose tissue was manually lipoaspirated from the patient’s abdominal subcutaneous tissue by a plastic surgeon. The authors used the VAS scale as their outcome measure. The patients in the PRP group had a mean pretreatment VAS score of 6.3 ± 1.3, which decreased to 1.9 ± 1.4 (P < .001) at the 6-month follow-up. The patients in the adipose-derived SVF group had a mean pretreatment VAS of 6.5 ± 1.5, which decreased to 1.6 ± 1.9 (P < .001) at the 6-month follow-up. Furthermore, the adipose-derived SVF group had statistically significant increases in tendon thickness on both ultrasonography and MRI, whereas the PRP group did not. Despite a theoretical association between larger tendon size and worsening degeneration in AT, this larger tendon thickness correlated with improvements in VAS pain score. Conversely, the PRP group had statistically significant signal intensity on MRI, whereas the adipose-derived SVF group did not. However, it must be noted that this change in signal intensity for the PRP group was minimal. Furthermore, the data on PRP in general for AT are controversial, with many studies showing that it has no effect. 42 Thus, although the authors concluded that both PRP and adipose-derived SVF seemed to allow for clinical benefit, this finding is misleading, as the literature overwhelmingly does not support the use of PRP for the treatment of AT. Thus, the only conclusion that can be drawn is that ADSC is no more effective than PRP, which has not been shown to be superior at this point in time.
Another RCT by Usuelli et al 60 concluded that intratendinous ADSC injections provide a safe and efficacious treatment for AT. This study included 44 patients, 23 of whom were assigned to a PRP group and 21 to the ADSC group. A small volume of adipose tissue (50 mL) was manually lipoaspirated from the patient’s abdominal subcutaneous tissue by a plastic surgeon. All patients were assessed preoperatively and at 15, 30, 60, 120, and 180 days after treatment using the VAS pain scale, Victorian Institute of Sport Assessment-Achilles (VISA-A), AOFAS, and SF-36 form. At the 15- and 30-day time points, the SVF group scored significantly better than the SVF group for the VAS, AOFAS, and VISA-A. However, there was no difference at 60, 120, and 180 days. The authors concluded that both treatments allowed for a significant improvement with respect to baseline.
Osteochondral Lesions of the Talus
Because of the tenuous blood supply to the talus, some osteochondral lesions of the talus (OLTs) remain refractory to various therapeutic measures.31,48 Thus, ADSCs have been proposed as an adjunct to encourage healing in OLTs. 52
A cohort study by Kim et al 27 compared the clinical and MRI outcomes between injection of ADSC with marrow stimulation and marrow stimulation alone in patients with OLTs. A total of 49 patients (50 ankles) were included in this study. Of these ankles, 26 underwent marrow stimulation alone (conventional group), and 24 underwent marrow stimulation with an injection of SVF-containing MSCs (MSC group). Adipose tissue was harvested from the patient’s buttock through tumescent liposuction. The authors assessed outcomes with the VAS pain scale, AOFAS score, Tegner activity scale, and the Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) score.
In the conventional group, the mean VAS score improved from 7.1 ± 1.2 to 3.9 ± 0.8, the mean AOFAS score improved from 68.5 ± 5.6 to 78.3 ± 4.9, and the mean Tegner score improved from 3.4 ± 0.6 to 3.5 ± 0.8. In the MSC group, the mean VAS score improved from 7.1 ± 0.8 to 3.2 ± 0.8, the mean AOFAS score improved from 67.7 ± 4.7 to 83.3 ± 7.0, and the mean Tegner score improved from 3.4 ± 0.5 to 3.9 ± 0.7. All clinical outcomes (VAS, AOFAS, and Tegner scores) improved significantly in the MSC group compared with the conventional group (P = .003, P = .009, and P = .041, respectively).
There was also a significant difference in the mean MOCART score between the conventional and MSC groups, which is the scoring system the authors used to evaluate the MRI scans. The MOCART score is out of 100 and contains 9 variables, such as degree of repair and filling of the defect; integration to border zone; surface, structure, and signal intensity of the repair tissue; and presence of adhesions or effusion. 38 Among these 9 variables, there were significant differences between the groups in the degree of defect repair and filling of the defect and subchondral bone (P = .041 and .038, respectively). The overall MOCART score in the conventional group was 49.4 ± 16.2 compared to 62.1 ± 21.8 in the MSC group (P = .037). Furthermore, there were significant correlations of the MOCART score with all clinical outcome scores (VAS, AOFAS, and Tegner scores) in both groups (P < .05). At this point in time, the clinical and MRI outcomes of injection with SVF containing ADSCs with marrow stimulation are too limited to support their use in OLTs.
Hallux Rigidus
Hallux rigidus (HR) is a relatively common disorder that is present in 2.5% of people aged ≥50 years.21,46 To our knowledge, there is only a single case report describing the use of ADSC injections for the treatment of HR. 5 The patient in this study was treated with an injection of ADSCs, which were obtained from the abdomen, and followed up after 9 months. Although this patient’s AOFAS and VAS scores both improved at the final follow-up, no conclusions can be drawn as this is the only study to our knowledge that looks at the role of ADSC injections in the treatment of HR, and treatment efficacy cannot be determined from case reports.
Ankle Osteoarthritis
Ankle OA is a debilitating condition that affects the daily life of nearly 250 million people worldwide. 45 The disease is associated with pain, dysfunction, and is a leading cause of disability, especially among the elderly population. 24
A retrospective cohort study by Shimozono et al 53 evaluated the efficacy of intraarticular injections of autologous micronized adipose tissue with ankle arthroscopic debridement in patients with advanced-stage posttraumatic osteoarthritis (PTOA) of the ankle. This study included 19 patients (19 ankles) with Kellgren-Lawrence (KL) grades 3 and 4. Eighty to 120 mL of aspirated abdominal tissue was obtained via liposuction. This adipose tissue was then transferred from each Vaclock syringe to a single 60-mL syringe using a Luer-lock connector. The harvested fat was processed in the Lipogems processing kit according to the manufacturer’s technique guide (Lipogems International, SpA). This step decreases the size of the adipose tissue clusters with a gentle mechanical action while simultaneously removing oil and bloody residues from the mixture. The resulting microionized adipose tissue was then transferred into a few 10-mL syringes that were injected into the patient. The authors used the VAS and the Foot and Ankle Outcome Scores (FAOS) to evaluate the clinical efficacy of treatment. At a mean follow-up of 14.3 months, the mean FAOS for pain significantly increased from 48.8 preoperatively to 61.1 (P = .029) and significantly increased from 20.1 preoperatively to 30.1 (P = .048). The mean VAS score significantly improved from 6.1 to 3.8 at the final follow-up (P = .003). A total of 10.5% (2/19) of patients were very satisfied, 31.6% (6/19) satisfied, 26.3% (5/19) neutral, 21.1% (4/19) unsatisfied, and 10.5% (2/19) very unsatisfied with their outcomes.
Conclusion
ADSCs exhibit multipotent differentiation potential and are capable of the release of several growth factors and protein moieties that may impact regenerative potential in various therapeutic applications. Within foot and ankle surgery, ADSCs have been applied to PF, AT, OLT, HR, and ankle OA. Although case reports, case series, and some limited controlled trials have demonstrated the potential efficacy of ADSCs across these pathologies, further high-quality research is needed before ADSCs can be definitively recommended for foot and ankle applications.
Supplemental Material
Supplemental material, sj-pdf-1-fao-10.1177_24730114231207643 for Contemporary Review: The Use of Adipocyte-Derived Mesenchymal Stem Cells in Pathologies of the Foot and Ankle by Albert T. Anastasio, Kian Bagheri and Samuel B. Adams in Foot & Ankle Orthopaedics
Footnotes
Ethical Approval Statement: No ethics approval was needed for this study. No patients were included in this work.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Samuel B. Adams, MD, reports consulting fees from Regeneration Technologies, Inc. ICMJE forms for all authors are available online.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs: Albert T. Anastasio, MD,  https://orcid.org/0000-0001-5817-3826
https://orcid.org/0000-0001-5817-3826
Kian Bagheri, BA,  https://orcid.org/0000-0003-0303-5949
https://orcid.org/0000-0003-0303-5949
References
- 1. Ahmed IM, Lagopoulos M, McConnell P, Soames RW, Sefton GK. Blood supply of the Achilles tendon. J Orthop Res. 1998;16(5):591-596. doi: 10.1002/jor.1100160511 [DOI] [PubMed] [Google Scholar]
- 2. Albano D, Messina C, Usuelli FG, et al. Magnetic resonance and ultrasound in Achilles tendinopathy: predictive role and response assessment to platelet-rich plasma and adipose-derived stromal vascular fraction injection. Eur J Radiol. 2017;95:130-135. doi: 10.1016/j.ejrad.2017.08.006 [DOI] [PubMed] [Google Scholar]
- 3. An R, Zhang Y, Qiao Y, Song L, Wang H, Dong X. Adipose stem cells isolated from diabetic mice improve cutaneous wound healing in streptozotocin-induced diabetic mice. Stem Cell Res Ther. 2020;11(1):120. doi: 10.1186/s13287-020-01621-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Begic A, Isfoss BL, Lonnerod LK, Vigen A, Moskaug JO. Survival and inflammatory response in adipose-derived mesenchymal stem cell-enriched mouse fat grafts. Plast Reconstr Surg Glob Open. 2016;4(12):e1110. doi: 10.1097/GOX.0000000000001110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Braile A, Toro G, De Cicco A, Cecere AB, Zanchini F, Schiavone Panni A. Hallux rigidus treated with adipose-derived mesenchymal stem cells: a case report. World J Orthop. 2021;12(1):51-55. doi: 10.5312/wjo.v12.i1.51 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Cao SJ, Wang LF, Ba T, Fu X, Li F, Hao CG. Effects of allogeneic mouse adipose-derived mesenchymal stem cell-microporous sheep acellular dermal matrix on healing of wound with full-thickness skin defect in mouse and the related mechanism. Zhonghua Shao Shang Za Zhi. 2018;34(12):901-906. doi: 10.3760/cma.j.issn.1009-2587.2018.12.015 [DOI] [PubMed] [Google Scholar]
- 7. Carek PJ, Edenfield KM, Michaudet C, Nicolette GW. Foot and ankle conditions: plantar fasciitis. FP Essent. 2018;465:11-17. [PubMed] [Google Scholar]
- 8. Cattaneo G, De Caro A, Napoli F, Chiapale D, Trada P, Camera A. Micro-fragmented adipose tissue injection associated with arthroscopic procedures in patients with symptomatic knee osteoarthritis. BMC Musculoskelet Disord. 2018;19(1):176. doi: 10.1186/s12891-018-2105-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Cho H, Kim J, Kim S, et al. Dual delivery of stem cells and insulin-like growth factor-1 in coacervate-embedded composite hydrogels for enhanced cartilage regeneration in osteochondral defects. J Control Release. 2020;327:284-295. doi: 10.1016/j.jconrel.2020.08.002 [DOI] [PubMed] [Google Scholar]
- 10. Coccè V, Brini A, Giannì AB, et al. A nonenzymatic and automated closed-cycle process for the isolation of mesenchymal stromal cells in drug delivery applications. Stem Cells Int. 2018;2018:4098140. doi: 10.1155/2018/4098140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. De Ugarte DA, Morizono K, Elbarbary A, et al. Comparison of multi-lineage cells from human adipose tissue and bone marrow. Cells Tissues Organs. 2003;174(3):101-109. doi: 10.1159/000071150 [DOI] [PubMed] [Google Scholar]
- 12. Ejaz A, Epperly MW, Hou W, Greenberger JS, Rubin JP. Adipose-derived stem cell therapy ameliorates ionizing irradiation fibrosis via hepatocyte growth factor-mediated transforming growth factor-β downregulation and recruitment of bone marrow cells. Stem Cells. 2019;37(6):791-802. doi: 10.1002/stem.3000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Elabd C, Chiellini C, Carmona M, et al. Human multipotent adipose-derived stem cells differentiate into functional brown adipocytes. Stem Cells. 2009;27(11):2753-2760. doi: 10.1002/stem.200 [DOI] [PubMed] [Google Scholar]
- 14. Estrada E, Decima JL, Rodriguez M, Di Tomaso M, Roberti J. Patient-reported outcomes after platelet-rich plasma, bone marrow aspirate, and adipose-derived mesenchymal stem cell injections for symptomatic knee osteoarthritis. Clin Med Insights Arthritis Musculoskelet Disord. 2020;13:1179544120931086. doi: 10.1177/1179544120931086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Fedorenko A, Lishko PV, Kirichok Y. Mechanism of fatty-acid-dependent UCP1 uncoupling in brown fat mitochondria. Cell. 2012;151(2):400-413. doi: 10.1016/j.cell.2012.09.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Freitag J, Wickham J, Shah K, Tenen A. Effect of autologous adipose-derived mesenchymal stem cell therapy in the treatment of an osteochondral lesion of the ankle. BMJ Case Rep. 2020;13(7):e234595. doi: 10.1136/bcr-2020-234595 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Gaspar RC, Pauli JR, Shulman GI, Muñoz VR. An update on brown adipose tissue biology: a discussion of recent findings. Am J Physiol Endocrinol Metab. 2021;320(3):E488-E495. doi: 10.1152/ajpendo.00310.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Goff JD, Crawford R. Diagnosis and treatment of plantar fasciitis. Am Fam Physician. 2011;84(6):676-682. [PubMed] [Google Scholar]
- 19. Gusenoff BR, Minteer D, Gusenoff JA. Perforating fat injections for chronic plantar fasciitis: a randomized, crossover clinical trial. Plast Reconstr Surg. 2022;149(2):297e-302e. doi: 10.1097/PRS.0000000000008765 [DOI] [PubMed] [Google Scholar]
- 20. Hassan WU, Greiser U, Wang W. Role of adipose-derived stem cells in wound healing. Wound Repair Regen. 2014;22(3):313-325. doi: 10.1111/wrr.12173 [DOI] [PubMed] [Google Scholar]
- 21. Ho B, Baumhauer J. Hallux rigidus. EFORT Open Rev. 2017;2(1):13-20. doi: 10.1302/2058-5241.2.160031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Hohmann E, Tetsworth K, Glatt V. Platelet-rich plasma versus corticosteroids for the treatment of plantar fasciitis: a systematic review and meta-analysis. Am J Sports Med. 2021;49(5):1381-1393. doi: 10.1177/0363546520937293 [DOI] [PubMed] [Google Scholar]
- 23. Hurley ET, Shimozono Y, Hannon CP, Smyth NA, Murawski CD, Kennedy JG. Platelet-rich plasma versus corticosteroids for plantar fasciitis: a systematic review of randomized controlled trials. Orthop J Sports Med. 2020;8(4):2325967120915704. doi: 10.1177/2325967120915704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Jaleel A, Golightly YM, Alvarez C, Renner JB, Nelson AE. Incidence and progression of ankle osteoarthritis: the Johnston County Osteoarthritis Project. Semin Arthritis Rheum. 2021;51(1):230-235. doi: 10.1016/j.semarthrit.2020.10.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Kang KK, Lee EJ, Kim YD, et al. Vitamin C improves therapeutic effects of adipose-derived stem cell transplantation in mouse tendonitis model. In Vivo. 2017;31(3):343-348. doi: 10.21873/invivo.11065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Kim WS, Park BS, Sung JH, et al. Wound healing effect of adipose-derived stem cells: a critical role of secretory factors on human dermal fibroblasts. J Dermatol Sci. 2007;48(1):15-24. doi: 10.1016/j.jdermsci.2007.05.018 [DOI] [PubMed] [Google Scholar]
- 27. Kim YS, Lee HJ, Choi YJ, Kim YI, Koh YG. Does an injection of a stromal vascular fraction containing adipose-derived mesenchymal stem cells influence the outcomes of marrow stimulation in osteochondral lesions of the talus? A clinical and magnetic resonance imaging study. Am J Sports Med. 2014;42(10):2424-2434. doi: 10.1177/0363546514541778 [DOI] [PubMed] [Google Scholar]
- 28. Kim YS, Koh YG. Injection of mesenchymal stem cells as a supplementary strategy of marrow stimulation improves cartilage regeneration after lateral sliding calcaneal osteotomy for varus ankle osteoarthritis: clinical and second-look arthroscopic results. Arthroscopy. 2016;32(5):878-889. doi: 10.1016/j.arthro.2016.01.020 [DOI] [PubMed] [Google Scholar]
- 29. Komori T. Regulation of osteoblast differentiation by transcription factors. J Cell Biochem. 2006;99(5):1233-1239. doi: 10.1002/jcb.20958 [DOI] [PubMed] [Google Scholar]
- 30. Kotani T, Masutani R, Suzuka T, Oda K, Makino S, Ii M. Anti-inflammatory and anti-fibrotic effects of intravenous adipose-derived stem cell transplantation in a mouse model of bleomycin-induced interstitial pneumonia. Sci Rep. 2017;7(1):14608. doi: 10.1038/s41598-017-15022-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Krause F, Anwander H. Osteochondral lesion of the talus: still a problem? EFORT Open Rev. 2022;7(6):337-343. doi: 10.1530/eor-22-0024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Kuo YR, Wang CT, Cheng JT, Kao GS, Chiang YC, Wang CJ. Adipose-derived stem cells accelerate diabetic wound healing through the induction of autocrine and paracrine effects. Cell Transplant. 2016;25(1):71-81. doi: 10.3727/096368915x687921 [DOI] [PubMed] [Google Scholar]
- 33. Labusca L. Adipose tissue in bone regeneration - stem cell source and beyond. World J Stem Cells. 2022;14(6):372-392. doi: 10.4252/wjsc.v14.i6.372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Li X, Guo W, Zha K, et al. Enrichment of CD146+ adipose-derived stem cells in combination with articular cartilage extracellular matrix scaffold promotes cartilage regeneration. Theranostics. 2019;9(17):5105-5121. doi: 10.7150/thno.33904 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Liu R, Dong R, Chang M, Liang X, Wang HC. Adipose-derived stem cells for the treatment of diabetic wound: from basic study to clinical application. Front Endocrinol (Lausanne). 2022;13:882469. doi: 10.3389/fendo.2022.882469 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Luo Y, Yi X, Liang T, et al. Autograft microskin combined with adipose-derived stem cell enhances wound healing in a full-thickness skin defect mouse model. Stem Cell Res Ther. 2019;10(1):279. doi: 10.1186/s13287-019-1389-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Manetti M. Molecular morphology and function of stromal cells. Int J Mol Sci. 2021;22(24):13422. doi: 10.3390/ijms222413422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Migliorini F, Driessen A, Quack V, et al. Comparison between intra-articular infiltrations of placebo, steroids, hyaluronic and PRP for knee osteoarthritis: a Bayesian network meta-analysis. Arch Orthop Trauma Surg. 2021;141(9):1473-1490. doi: 10.1007/s00402-020-03551-y [DOI] [PubMed] [Google Scholar]
- 39. Mutschall H, Winkler S, Weisbach V, Arkudas A, Horch RE, Steiner D. Bone tissue engineering using adipose-derived stem cells and endothelial cells: effects of the cell ratio. J Cell Mol Med. 2020;24(12):7034-7043. doi: 10.1111/jcmm.15374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Nammian P, Asadi-Yousefabad SL, Daneshi S, Sheikhha MH, Tabei SMB, Razban V. Comparative analysis of mouse bone marrow and adipose tissue mesenchymal stem cells for critical limb ischemia cell therapy. Stem Cell Res Ther. 2021;12(1):58. doi: 10.1186/s13287-020-02110-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Natali S, Screpis D, Farinelli L, Iacono V, Vacca V, Gigante A, Zorzi C. The use of intra-atricular injection of autologous micro-fragmented adipose tissue as pain treatment for ankle osteoarthritis: a prospective not randomized clinical study. Int Ortho. 2021;45(9):2239-2244. doi: 10.1007/s00264-021-05093-3(Natali) [DOI] [PubMed] [Google Scholar]
- 42. Nauwelaers AK, Van Oost L, Peers K. Evidence for the use of PRP in chronic midsubstance Achilles tendinopathy: a systematic review with meta-analysis. Foot Ankle Surg. 2021;27(5):486-495. doi: 10.1016/j.fas.2020.07.009 [DOI] [PubMed] [Google Scholar]
- 43. Orbay H, Tobita M, Mizuno H. Mesenchymal stem cells isolated from adipose and other tissues: basic biological properties and clinical applications. Stem Cells Int. 2012;2012:461718. doi: 10.1155/2012/461718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Paduano F, Marrelli M, Amantea M, et al. Adipose tissue as a strategic source of mesenchymal stem cells in bone regeneration: a topical review on the most promising craniomaxillofacial applications. Int J Mol Sci. 2017;18(10):2140. doi: 10.3390/ijms18102140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Paget LDA, Reurink G, de Vos RJ, et al. Effect of platelet-rich plasma injections vs placebo on ankle symptoms and function in patients with ankle osteoarthritis: a randomized clinical trial. JAMA. 2021;326(16):1595-1605. doi: 10.1001/jama.2021.16602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Patel J, Swords M. Hallux Rigidus. StatPearls; 2022. [PubMed] [Google Scholar]
- 47. Ra K, Oh HJ, Kim EY, et al. Anti-oxidative effects of human adipose stem cell conditioned medium with different basal medium during mouse embryo in vitro culture. Animals (Basel). 2020;10(8):1414. doi: 10.3390/ani10081414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Saravi B, Lang G, Ruff R, et al. Conservative and surgical treatment of talar fractures: a systematic review and meta-analysis on clinical outcomes and complications. Int J Environ Res Public Health. 2021;18(16):8274. doi: 10.3390/ijerph18168274 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Schipper BM, Marra KG, Zhang W, Donnenberg AD, Rubin JP. Regional anatomic and age effects on cell function of human adipose-derived stem cells. Ann Plast Surg. 2008;60(5):538-544. doi: 10.1097/SAP.0b013e3181723bbe [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Senarath-Yapa K, McArdle A, Renda A, Longaker MT, Quarto N. Adipose-derived stem cells: a review of signaling networks governing cell fate and regenerative potential in the context of craniofacial and long bone skeletal repair. Int J Mol Sci. 2014;15(6):9314-9330. doi: 10.3390/ijms15069314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Senesi L, De Francesco F, Marchesini A, et al. Efficacy of adipose-derived mesenchymal stem cells and stromal vascular fraction alone and combined to biomaterials in tendinopathy or tendon injury: systematic review of current concepts. Medicina (Kaunas). 2023;59(2):273. doi: 10.3390/medicina59020273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Shafaei H, Kalarestaghi H. Adipose-derived stem cells: an appropriate selection for osteogenic differentiation. J Cell Physiol. 2020;235(11):8371-8386. doi: 10.1002/jcp.29681 [DOI] [PubMed] [Google Scholar]
- 53. Shimozono Y, Dankert JF, Kennedy JG. Arthroscopic debridement and autologous micronized adipose tissue injection in the treatment of advanced-stage posttraumatic osteoarthritis of the ankle. Cartilage. 2021;13(suppl 1):1337S-1343S. doi: 10.1177/1947603520946364 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Shin MJ, Shim IK, Kim DM, et al. Engineered cell sheets for the effective delivery of adipose-derived stem cells for tendon-to-bone healing. Am J Sports Med. 2020;48(13):3347-3358. doi: 10.1177/0363546520964445 [DOI] [PubMed] [Google Scholar]
- 55. Smakaj A, De Mauro D, Rovere G, et al. Clinical application of adipose derived stem cells for the treatment of aseptic non-unions: current stage and future perspectives-systematic review. Int J Mol Sci. 2022;23(6):3057. doi: 10.3390/ijms23063057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Tian L, Lu L, Meng Y. Bone marrow stromal stem cell fate decision: a potential mechanism for bone marrow adipose increase with aging-related osteoporosis. Curr Mol Med. 2023;23(10):1046-1057. doi: 10.2174/1566524023666221025104629 [DOI] [PubMed] [Google Scholar]
- 57. Trayhurn P, Beattie JH. Physiological role of adipose tissue: white adipose tissue as an endocrine and secretory organ. Proc Nutr Soc. 2001;60(3):329-339. doi: 10.1079/pns200194 [DOI] [PubMed] [Google Scholar]
- 58. Tremolada C, Colombo V, Ventura C. Adipose tissue and mesenchymal stem cells: state of the art and Lipogems® technology development. Curr Stem Cell Rep. 2016;2(3):304-312. doi: 10.1007/s40778-016-0053-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Trojian T, Tucker AK. Plantar fasciitis. Am Fam Physician. 2019;99(12):744-750. [PubMed] [Google Scholar]
- 60. Usuelli FG, Grassi M, Maccario C, et al. Intratendinous adipose-derived stromal vascular fraction (SVF) injection provides a safe, efficacious treatment for Achilles tendinopathy: results of a randomized controlled clinical trial at a 6-month follow-up. Knee Surg Sports Traumatol Arthrosc. 2018;26(7):2000-2010. doi: 10.1007/s00167-017-4479-9 [DOI] [PubMed] [Google Scholar]
- 61. Van Harmelen V, Röhrig K, Hauner H. Comparison of proliferation and differentiation capacity of human adipocyte precursor cells from the omental and subcutaneous adipose tissue depot of obese subjects. Metabolism. 2004;53(5):632-637. [DOI] [PubMed] [Google Scholar]
- 62. Vo TP, Ho GWK, Andrea J. Achilles tendinopathy, a brief review and update of current literature. Curr Sports Med Rep. 2021;20(9):453-461. doi: 10.1249/JSR.0000000000000884 [DOI] [PubMed] [Google Scholar]
- 63. von Rickenbach KJ, Borgstrom H, Tenforde A, Borg-Stein J, McInnis KC. Achilles tendinopathy: evaluation, rehabilitation, and prevention. Curr Sports Med Rep. 2021;20(6):327-334. doi: 10.1249/JSR.0000000000000855 [DOI] [PubMed] [Google Scholar]
- 64. Wagner JM, Conze N, Lewik G, et al. Bone allografts combined with adipose-derived stem cells in an optimized cell/volume ratio showed enhanced osteogenesis and angiogenesis in a murine femur defect model. J Mol Med (Berl). 2019;97(10):1439-1450. doi: 10.1007/s00109-019-01822-9 [DOI] [PubMed] [Google Scholar]
- 65. Wang T, Hill RC, Dzieciatkowska M, et al. Site-dependent lineage preference of adipose stem cells. Front Cell Dev Biol. 2020;8:237. doi: 10.3389/fcell.2020.00237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Winkler S, Mutschall H, Biggemann J, et al. Human umbilical vein endothelial cell support bone formation of adipose-derived stem cell-loaded and 3D-printed osteogenic matrices in the arteriovenous loop model. Tissue Eng Part A. 2021;27(5-6):413-423. doi: 10.1089/ten.TEA.2020.0087 [DOI] [PubMed] [Google Scholar]
- 67. Xu T, Yu X, Yang Q, Liu X, Fang J, Dai X. Autologous micro-fragmented adipose tissue as stem cell-based natural scaffold for cartilage defect repair. Cell Transplant. 2019;28(12):1709-1720. doi: 10.1177/0963689719880527 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Yang Q, Teng BH, Wang LN, et al. Silk fibroin/cartilage extracellular matrix scaffolds with sequential delivery of TGF-β3 for chondrogenic differentiation of adipose-derived stem cells. Int J Nanomedicine. 2017;12:6721-6733. doi: 10.2147/ijn.S141888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Yang WY, Han YH, Cao XW, et al. Platelet-rich plasma as a treatment for plantar fasciitis: a meta-analysis of randomized controlled trials. Medicine (Baltimore). 2017;96(44):e8475. doi: 10.1097/MD.0000000000008475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Yang Y, Zhang B, Yang Y, Peng B, Ye R. PLGA containing human adipose-derived stem cell-derived extracellular vesicles accelerates the repair of alveolar bone defects via transfer of CGRP. Oxid Med Cell Longev. 2022;2022:4815284. doi: 10.1155/2022/4815284 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Yoshida Y, Matsubara H, Fang X, et al. Adipose-derived stem cell sheets accelerate bone healing in rat femoral defects. PLoS One. 2019;14(3):e0214488. doi: 10.1371/journal.pone.0214488 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Yoshimura K, Shigeura T, Matsumoto D, et al. Characterization of freshly isolated and cultured cells derived from the fatty and fluid portions of liposuction aspirates. J Cell Physiol. 2006;208(1):64-76. doi: 10.1002/jcp.20636 [DOI] [PubMed] [Google Scholar]
- 73. Zhou J, Wei T, He Z. ADSCs enhance VEGFR3-mediated lymphangiogenesis via METTL3-mediated VEGF-C m6A modification to improve wound healing of diabetic foot ulcers. Mol Med. 2021;27(1):146. doi: 10.1186/s10020-021-00406-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Zuk PA, Zhu M, Mizuno H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7(2):211-228. doi: 10.1089/107632701300062859w [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-pdf-1-fao-10.1177_24730114231207643 for Contemporary Review: The Use of Adipocyte-Derived Mesenchymal Stem Cells in Pathologies of the Foot and Ankle by Albert T. Anastasio, Kian Bagheri and Samuel B. Adams in Foot & Ankle Orthopaedics