Abstract
Background
Health authorities stress the temperature sensitivity of human insulin, advising protection from heat and freezing, with manufacturers suggesting low‐temperature storage for intact vials, and once opened, storage at room temperature for four to six weeks, though usage time and maximum temperature recommendations vary. For human insulin, the recommendations of current shelf life in use may range from 10 to 45 days, and the maximum temperature in use varies between 25 °C and 37 °C. Optimal cold‐chain management of human insulin from manufacturing until the point of delivery to people with diabetes should always be maintained, and people with diabetes and access to reliable refrigeration should follow manufacturers' recommendations. However, a growing segment of the diabetes‐affected global population resides in challenging environments, confronting prolonged exposure to extreme heat due to the climate crisis, all while grappling with limited access to refrigeration.
Objectives
To analyse the effects of storing human insulin above or below the manufacturers' recommended insulin temperature storage range or advised usage time, or both, after dispensing human insulin to people with diabetes.
Search methods
We used standard, extensive Cochrane search methods. The latest search date was 12 July 2023.
Selection criteria
We included clinical and laboratory studies investigating the storage of human insulin above or below manufacturers' recommended temperature storage range, advised usage time, or both.
Data collection and analysis
We used standard Cochrane methods. We used GRADE to assess the certainty of evidence for the clinical study. Most information emerged from in vitro studies, mainly from pharmaceutical companies. There is no validated risk of bias and certainty of evidence rating for in vitro studies. We thus presented a narrative summary of the results.
Main results
We included 17 eligible studies (22 articles) and additional information from pharmaceutical companies.
Pilot clinical study
One pilot clinical study investigated temperature conditions for insulin stored for six weeks in an unglazed clay pot with temperatures ranging between 25 °C and 27 °C. The mean fall in plasma glucose in eight healthy volunteers after clay pot‐stored insulin injection was comparable to refrigerator‐stored insulin injection (very low‐certainty evidence).
In‐vitro studies
Nine, three and four laboratory studies investigated storage conditions for insulin vials, insulin cartridges/pens and prefilled plastic syringes, respectively. The included studies reported numerous methods, laboratory measurements and storage conditions.
Three studies on prefilled syringes investigating insulin potency at 4 °C up to 23 °C for up to 28 days showed no clinically relevant loss of insulin activity.
Nine studies examined unopened vials and cartridges. In studies with no clinically relevant loss of insulin activity for human short‐acting insulin (SAI), intermediate‐acting insulin (IAI) and mixed insulin (MI) temperatures ranged between 28.9 °C and 37 °C for up to four months. Two studies reported up to 18% loss of insulin activity after one week to 28 days at 37 °C. Four studies examined opened vials and cartridges at up to 37 °C for up to 12 weeks, indicating no clinically relevant reduction in insulin activity. Two studies analysed storage conditions for oscillating temperatures ranging between 25 °C and 37 °C for up to 12 weeks and observed no loss of insulin activity for SAI, IAI and MI. Four studies, two on vials (including one on opened vials), and two on prefilled syringes, investigated sterility and reported no microbial contamination.
Data from pharmaceutical companies
Four manufacturers (BIOTON, Eli Lilly and Company, Novo Nordisk and Sanofi) provided previously unreleased human insulin thermostability data mostly referring to unopened containers (vials, cartridges). We could not include the data from Sanofi because the company announced the permanent discontinuation of the production of human insulins Insuman Rapid, Basal and Comb 25.
BIOTON provided data on SAI after one, three and six months at 25 °C: all investigated parameters were within reference values, and, compared to baseline, loss of insulin activity was 1.1%, 1.0% and 1.7%, respectively.
Eli Lilly and Company provided summary data: at below 25 °C or 30 °C SAI/IAI/MI could be stored for up to 25 days or 12 days, respectively. Thereafter, patient in‐use was possible for up to 28 days.
Novo Nordisk provided extensive data: compared to baseline, after three and six months at 25 °C, loss of SAI activity was 1.8% and 3.2% to 3.5%, respectively. Loss of IAI activity was 1.2% to 1.9% after three months and 2.0% to 2.3% after six months. Compared to baseline, after one, two and three months at 37 °C, loss of SAI activity was 2.2% to 2.8%, 5.7% and 8.3% to 8.6%, respectively. Loss IAI activity was 1.4% to 1.8%, 3.0% to 3.8% and 4.7% to 5.3%, respectively. There was no relevant increase in insulin degradation products observed. Up to six months at 25 °C and up to two months at 37 °C high molecular weight proteins were within specifications. Appearance, visible particles or macroscopy, particulate matter, zinc, pH, metacresol and phenol complied with specifications.
There were no data for cold environmental conditions and insulin pumps.
Authors' conclusions
Under difficult living conditions, pharmaceutical companies' data indicate that it is possible to store unopened SAI and IAI vials and cartridges at up to 25 °C for a maximum of six months and at up to 37 °C for a maximum of two months without a clinically relevant loss of insulin potency. Also, oscillating temperatures between 25 °C and 37 °C for up to three months result in no loss of insulin activity for SAI, IAI and MI. In addition, ambient temperature can be lowered by use of simple cooling devices such as clay pots for insulin storage. Clinical studies on opened and unopened insulin containers should be performed to measure insulin potency and stability after varying storage conditions. Furthermore, more data are needed on MI, insulin pumps, sterility and cold climate conditions.
Keywords: Drug Stability, Drug Storage, Insulin, Temperature
Plain language summary
Temperature and storage conditions for human insulin
What is insulin?
Insulin is a hormone made by the pancreas that helps your body turn food into energy and controls your blood sugar levels. People with diabetes cannot make enough insulin or use it effectively, so some have to inject it themselves.
How should insulin be stored?
Health authorities and pharmaceutical manufacturers recommend keeping insulin away from sunlight, not freezing it and storing it in a refrigerator. Unopened human insulin vials or flasks and cartridges for insulin pens should be stored at temperatures between 2 °C and 8 °C, which requires reliable refrigeration. Once opened, vials or cartridges can be stored at 'room temperature' and used for approximately four to six weeks. However, recommendations differ regarding usage time and maximum temperature once open (in‐use insulin). Also, advice varies for the type of insulin, brand, insulin concentrations and container (vial, cartridge/pen, insulin pump). Regulatory bodies guide how to handle insulin transport and storage temperature ('cold chain') from manufacturer to delivery to people with diabetes. If reliable refrigeration is available, people with diabetes should store insulin according to official recommendations. However, there is no clear consensus on how human insulin should be stored at home if reliable refrigeration cannot be guaranteed.
What are the problems with insulin storage?
Many people with diabetes live in settings with little access to healthcare facilities and refrigeration. Challenging locations affected by natural disasters, periods of extreme heat due to the climate crisis or war‐like conditions additionally complicate appropriate insulin storage.
What did we want to find out?
We wanted to analyse the effects of storing human insulin above or below the manufacturers' recommended insulin temperature or advised usage time, or both.
What did we look for?
We searched medical databases and contacted study authors and pharmaceutical manufacturers about studies on people with diabetes. We also looked for laboratory studies investigating the effects of storing human insulin above or below the manufacturers' recommended insulin temperature storage range or advised usage time, or both.
What did we find?
We found one clinical study. Nine, three and four laboratory studies investigated storage conditions for insulin vials, insulin cartridges and prefilled plastic insulin syringes, respectively.
Key results
In the clinical study, authors investigated insulin stored for six weeks in an unglazed clay pot or in a refrigerator. Outside temperatures were between 34 °C and 43 °C, and clay pot temperatures were between 25 °C and 27 °C. The study authors only reported on the glucose level in the blood of eight healthy volunteers after injecting clay‐pot‐stored insulin or refrigerator‐stored insulin. Insulin in the clay pot had similar glucose‐lowering activity to insulin in a refrigerator.
Laboratory studies on prefilled syringes stored at 4 °C to 23 °C for up to 28 days showed no clinically relevant loss of insulin activity.
Nine laboratory studies examined unopened vials and cartridges. In studies with no clinically relevant loss of insulin activity, temperatures ranged between 28.9 °C and 37 °C for up to four months. Two studies reported a more pronounced loss of insulin activity after one week to 28 days at 37 °C. Four studies examined opened vials and cartridges at up to 37 °C for up to 12 weeks, and showed no clinically relevant reduction in insulin activity.
Two laboratory studies analysed storage conditions for shifting temperatures between 25 °C and 37 °C up to 12 weeks, resembling daytime and night‐time fluctuations in tropical countries. There was no loss of insulin activity for short‐acting, intermediate‐acting and mixed insulin (a combination of short‐acting and intermediate‐acting human insulin).
Four laboratory studies, two on vials, one of them on opened vials, and two on prefilled syringes, investigated sterility and reported no bacterial contamination.
Four manufacturers provided previously unpublished temperature and storage conditions data. If unopened human insulin vials or cartridges were stored at up to 25 °C for a maximum of six months or at up to 37 °C for a maximum of two months, only small amounts of insulin activity were lost. We could not include the provided data from the manufacturer Sanofi because the company announced the permanent discontinuation of the production of human insulins called Insuman Rapid, Basal and Comb 25.
If no reliable refrigeration is possible, room temperature can be lowered using simple cooling devices such as clay pots for insulin storage.
There were no data for cold environmental conditions or insulin pumps.
What are the limitations of the evidence?
Data on the quality of human insulin in unopened vials and cartridges appeared reliable. However, most data came from laboratory studies, and we were unable to assess the certainty of evidence because there are currently no validated methods for doing so. More studies should investigate the possible bacterial contamination risk of in‐use insulin vials and cartridges. Also, clinical studies in particular are needed to inform on temperature and storing conditions for insulin outside the official recommendations.
How up to date is this review?
This evidence is up to date as of 12 July 2023.
Summary of findings
Summary of findings 1. Clinical studies.
| Patient or population: healthy physicians Setting: outpatients, opened vials Intervention: neutral human insulin, stored in a refrigerator at 4 °C Comparison: neutral human insulin, stored in a zeera at a mean of 26.6 °C | |||
| Outcome | № of participants (studies) | Certainty of the evidence (GRADE) | Effect |
| Potency/bioactivity parameters of human insulin | — | — | Not reported |
| Diabetic ketoacidosis | — | — | Not reported |
| Hypoglycaemic episodes | — | — | Not reported |
| Adverse events | — | — | Not reported |
| Health‐related quality of life | — | — | Not reported |
| HbA1c | — | — | Not reported |
| Mean fall in plasma glucose levels (mmol/L/min) | 8 (1 RCT) | ⊕⊝⊝⊝ Very lowb | Intravenous injection of 0.1 U/kg bodyweight of human insulin with blood samples taken every 5 min up to 30 min following the injection. Refrigerator insulin (mean 4 °C, 6 weeks' storage): 0.213 mmol/L/min Zeer‐stored insulin (mean 26.6 °C, 6 weeks' storage): 0.192 mmol/L/min Quote: "The observed difference in the rate of decline of glucose following insulin stored in a zeer, and that … being stored in a refrigerator, was not statistically significant." |
| HbA1c: glycosylated haemoglobin A1c; min: minute; RCT: randomised controlled trial; U: unit. | |||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | |||
aZeer: a clay pot that was unglazed, enabling water to slowly seep through the wall and evaporate, thus lowering the temperature of the remaining water. A glass jar with partially filled desert sand was put in the centre of the zeer. A water bath was created by adding water outside the jar to the same level as that of the sand in the glass jar. Water was added to the mark (topped up) daily. The zeer was closed with a clay lid and kept in the shade. bDowngraded three levels due to the risk of bias (unclear randomisation and allocation concealment), indirectness (surrogate outcome, insufficient time frame) and imprecision (very small sample size, one study only).
Summary of findings 2. Published in‐vitro studies.
| Outcome: insulin potencya measured by HPLC (mean % change compared to baseline) | ||||
| Study ID | Short‐acting insulin (vials) | Intermediate‐acting insulin (vials) | Premixed short‐acting/NPH insulin (vials) | Container |
| Vimalavathini 2009 | 32 °C / 37 °C (28 days): −14% / −18% | — | 32 °C / 37 °C (28 days)b: −11% / −14% | Probably unopened |
| Kaufmann 2021 | 25–37 °Cc (4 / 12 weeks): +0.1% / +1.3% 37 °Cd (2 weeks): −12.5% | 25–37 °Cc (4 / 12 weeks): +1.3% / +1.6% 31 °Cd (1 / 4 / 8 weeks): +0.2% / −5.2% / −17.2% | 25–37 °Cc (4 / 12 weeks): +0.6% / +1.9% 37 °Cd (1 week): −15.4% |
Opened (some samples) |
| Pendsey 2023 | 26.3–28.9 °C (1 / 2 / 4 months): −0.7% / −2.0% / −2.4% | 26.3–28.9 °C (1 / 2 / 4 months): +2.1% / −1.7% / −0.7% | 26.3–28.9 °C (1 / 2 / 4 months): +0.8% / −1.8% / −1.4% | Unopened |
| Study ID | Short‐acting insulin (prefilled syringes) | Intermediate‐acting insulin (prefilled syringes) | Premixed short‐acting/NPH insulin (prefilled syringes) | Container |
| Tarr 1991 | 4 °C (28 days): −1.5% to + 0.6% 23 °C (28 days): −1.9% to +1.7% |
4 °C (28 days): +1.0% to +2.5% 23 °C (28 days): +0.7% to +4.7% |
4 °C (28 days): −0.2% to 2.4% 23 °C (28 days): −1.0% to −1.7% |
Prefilled |
| Outcome: insulin potencya measured by HPLC, mass spectrometry, radioimmunological activity | ||||
| Study ID | Short‐acting insulin (vials) | Intermediate‐acting insulin (vials) | Premixed short‐acting/NPH insulin (vials) | Container |
| Schrader 1985 | Shaken for 72 hours at 37 °C and at ambient temperature: same molecular insulin weight | — | — | Unopened |
| Gregory 1991 | 4 °C (26 weeks): −0.069% total protein/week 37 °C (26 weeks): –1.184% total protein/week Direct sunlight (26 weeks): −0.749% total protein/week | — | — | Opened (some samples) |
| Silva 2013 | — | 22–24 °C (42 days): 12 min peak 93% of baseline AUC reference; 21 min peak 94% of baseline AUC reference | — | Unopened |
| Moses 2019 | 30 °C (6 weeks): −3.1 IU/mL (95% CI 4.0) 30 °C (10 weeks): −4.0 IU/mL (95% CI 4.9) | 30 °C (6 weeks): −1.0 IU/mL (95% CI 1.9) 30 °C (10 weeks): −1.4 IU/mL (95% CI 2.3) | — | Unopened |
| Baechler 2020e | 4 °C (86 days): 98.6 U/mL (CV 5.5%) 22 °C (86 days): 99.4 U/mL (CV 5%) 37 °C (86 days): 98.7 U/mL (CV 4.6%) | — | — | Unopened |
| Study ID | Short‐acting insulin (cartridges/pens) | Intermediate‐acting insulin (cartridges/pens) | Premixed short‐acting/NPH insulin (cartridges/pens) | Container |
| Shnek 1998 | 25–37 °Cc and 37 °Cd (28 days): potency within specifications | 25–37 °Cc and 37 °Cd (28 days): potency within specifications; agglomerates (37 °Cd) | 25–37 °Cc and 37 °Cd (28 days): potency within specifications; agglomerates (37 °Cd) | Unopened |
| Kongmalai 2021 | — | 29.7 °C (28 days): −2.6% to +1.4% (compared to baseline AUC) 37 °C (28 days): −1.5% to +0.7% (compared to baseline AUC) | — | Opened |
| Study ID | Short‐acting insulin (prefilled syringes) | Intermediate‐acting insulin (prefilled syringes) | Premixed short‐acting/NPH insulin (prefilled syringes) | Container |
| Dunbar 1986 | 4 °C (2 weeks): no change in potency | — | — | Prefilled |
| Lull 2013 | 3–5 °C (28 days): 93.3% (SD 2.4) AUC of initial concentration | — | — | Prefilled |
| Outcome: protein structure, formation of amyloid material | ||||
| Study ID | Short‐acting insulin (vials) | Intermediate‐acting insulin (vials) | Premixed short‐acting/NPH insulin (vials) | Container |
| Kaufmann 2021 | 25–37 °Cc (4 / 12 weeks): tridimensional structure verified | 25–37 °Cc (4 / 12 weeks): tridimensional structure verified | 25–37 °Cc (4 / 12 weeks): tridimensional structure verified | Opened (some samples) |
| Silva‐Jr 2022 | 4 °C and 37 °C (5 weeks): no amyloid particles, large amorphous agglomerates | 4 °C and 37 °C (5 weeks): no amyloid particles, large amorphous agglomerates | — | Unopened |
| Pendsey 2023 | 26.3–28.9 °C (1 / 2 / 4 months): altered conformation or multimerisation in a minor fraction of the insulin molecules | 26.3–28.9 °C (1 / 2 / 4 months): altered conformation or multimerisation in a minor fraction of the insulin molecules | 26.3–28.9 °C (1 / 2 / 4 months): altered conformation or multimerisation in a minor fraction of the insulin molecules | Unopened |
| Study ID | Short‐acting insulin (cartridges/pens) | Intermediate‐acting insulin (cartridges/pens) | Premixed short‐acting/NPH insulin (cartridges/pens) | Container |
| Delbeck 2021a | — | 37 °C (10 weeks): small changes | — | Unopened |
| Outcome: sterility, microbial contamination | ||||
| Study ID | Short‐acting insulin (vials) | Intermediate‐acting insulin (vials) | Premixed short‐acting/NPH insulin (vials) | Container |
| Schrader 1985 | Shaken for 72 hours at 37 °C and at ambient temperature: no bacterial growth | — | — | Unopened |
| Kaufmann 2021 | 25–37 °Cc (4 / 12 weeks): no microbial contamination | 25–37 °Cc (4 / 12 weeks): no microbial contamination | 25–37 °Cc (4 / 12 weeks): no microbial contamination | Opened (some samples) |
| Study ID | Short‐acting insulin (prefilled syringes) | Intermediate‐acting insulin (prefilled syringes) | Premixed short‐acting/NPH insulin (prefilled syringes) | Container |
| Tarr 1991 | 4 °C (28 days): no microbial growth 23 °C (28 days): no microbial growth |
4 °C (28 days): no microbial growth 23 °C (28 days): no microbial growth |
4 °C (28 days): no microbial growth 23 °C (28 days): no microbial growth |
Prefilled |
| Koffler 1992 | — | 4 °C (10 days): no bacterial growth | — | Prefilled |
— denotes not investigated.
aUS Pharmacopeia (USP) stipulates that human insulin should have a potency of 95% to 105% of the potency stated on the label, expressed in USP insulin human units/mL, i.e. human insulin labelled '100 U/mL' has to contain 95 U/mL to 105 U/mL of insulin. bConcentration of the short‐acting insulin in the biphasic insulin formulation. cOscillating temperatures. dContinuous temperature. eMean insulin concentration (with CV) across all measurements and 86 days. AUC: area under the curve; CI: confidence interval; CV: coefficient of variation; HPLC: high‐performance liquid chromatography; NPH: neutral protamine Hagedorn; SD: standard deviation; U: unit.
Summary of findings 3. In‐vitro data from pharmaceutical manufacturers.
| Data from BIOTONa, b: human insulin potency compared to baseline (mean % change (SD) of several insulin batches) | |||||||||
| 25 °C storage | 1 month | 3 months | 6 months | ||||||
| Vials | Vials | Vials | |||||||
| Short‐acting insulin (100 IU/mL) | −1.1 | −1.0 | −1.7 | ||||||
| 30 °C storage | 28 days | 42 days | |||||||
| Vials | Vials | ||||||||
| Short‐acting insulin (40 IU/mL) | −2.4 | −2.0 | |||||||
| Data from Eli Lilly and Companyc: human insulin (100 IU/mL) potency | |||||||||
| Storage before start use | Patient in‐use | ||||||||
| < 25 °C (short‐acting, intermediate‐acting, mixed insulin) | Up to 25 days | Up to 28 days | |||||||
| < 30 °C (short‐acting, intermediate‐acting, mixed insulin) | Up to 12 days | Up to 28 days | |||||||
| Data from Sanofi | |||||||||
| Sanofi permanently discontinued the production of its human insulins Insuman rapid 100 IU/mL, Insuman rapid SoloStar 100 IU/mL, Insuman basal 100 IU/mL, Insuman basal SoloStar 100 IU/mL, Insuman comb 25 100 IU/mL and Insuman comb 25 SoloStar 100 IU/mL worldwide. Data provided by the manufacturer could not be included in this review. | |||||||||
| Data from Novo Nordiska, d: human insulin (100 IU/mL) potency compared to baseline (mean % change (SD) of several insulin batches) | |||||||||
| 25 °C storage | 1 month | 3 months | 6 months | 12 months | |||||
| Vials | Cartridges | Vials | Cartridges | Vials | Cartridges | Vials | Cartridges | ||
| Short‐acting insulin | −0.4 (0.3) | −0.5 (0.4) | −1.8 (0.6) | −1.8 (0.6) | −3.5 (0.4) | −3.2 (0.5) | −7.4 (0.6) | −6.5 (0.5) | |
| Intermediate‐acting insulin | −0.7 (0.4) | −0.5 (NA) | −1.2 (0.5) | −1.9 (NA) | −2.0 (0.4) | −2.3 (NA) | −3.4 (0.7) | −3.9 (NA) | |
| 37 °C storage | 1 month | 2 months | 3 months | ||||||
| Vials | Cartridges | Vials | Cartridges | Vials | Cartridges | ||||
| Short‐acting insulin | −2.2 (0.6) | −2.8 (0.3) | −5.7 (0.7) | −5.7 (1.5) | −8.6 (0.6) | −8.3 (0.8) | |||
| Intermediate‐acting insulin | −1.8 (0.3) | −1.4 (NA) | −3.8 (0.4) | −3.0 (NA) | −5.3 (1.0) | −4.7 (NA) | |||
aUS Pharmacopeia (USP) stipulates that human insulin should have a potency of 95% to 105% of the potency stated on the label, expressed in USP insulin human units/mL, i.e. human insulin labelled '100 U/mL' has to contain 95 U/mL to 105 U/mL of insulin. bA21 desamido insulin, impurities, total zinc, metacresol and pH all remained within reference values. cAll product quality attributes met established acceptance criteria. dUp to 6 months at 25 °C and up to 2 months at 37 °C high molecular weight proteins were within specifications. Appearance, visible particles or macroscopy, particulate matter, zinc, pH, metacresol and phenol complied with specifications. No data were available on bacterial endotoxins and sterility. IU: insulin units; NA: not applicable.
Background
There are two major forms of diabetes, type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM). T1DM results from an autoimmune process destroying the insulin‐producing beta cells (β‐cells) of the pancreas. T1DM is the major cause of diabetes in childhood and adolescence, and injections of insulin are necessary for survival. In the tenth revision of its diabetes atlas, the International Diabetes Federation (IDF) estimated that the number of people with diabetes worldwide would increase from 573 million in 2021 to 783 million in 2045, representing a 46% increase (IDF 2021). This rise is calculated to be higher in low‐ and middle‐income countries (LMIC), with projected increases of 134% in Africa (87% in the Middle East and North Africa), 68% in South‐East Asia, and 50% in South and Central America (IDF 2021). In comparison, the estimates for Europe, the Western Pacific and North America, and the Caribbean are 13%, 27% and 24%, respectively (IDF 2021). Approximately 90% of people with diabetes have T2DM, and the projected increases to 2045 are expected to be mainly due to a rising number of people with T2DM. This increase is probably caused by changes in living conditions, such as sedentary behaviour, dietary changes, obesity and other factors (Sun 2022).
Banting and Best discovered the peptide hormone insulin in 1921; it was first used in the treatment of diabetes in 1922 and has been included in the World Health Organization's (WHO's) Model List of Essential Medicines since 1977 (WHO EML 2021). T2DM accounts for approximately 90% of diabetes and is associated with insulin resistance: as a consequence of ineffective insulin action to transport glucose into cells, an increased insulin demand develops, and over time inadequate insulin production occurs due to exhaustion of pancreatic β‐cells. Several factors are related to risk of T2DM, including body composition, physical inactivity, diet, age, family history and history of gestational diabetes. It is initially treated with lifestyle modification and later on a combination of lifestyle modification and glucose‐lowering drugs (WHO 2020). At some point, insulin may be needed to improve diabetes management further, in case pancreatic β‐cells are unable to compensate for the increased insulin demand. The need for insulin worldwide to treat T2DM is expected to increase by more than 20% from 2018 to 2030 (Basu 2019). Diabetes may also occur during pregnancy (gestational diabetes) or may develop because of diseases of the pancreas, endocrine diseases, infections, drugs, immune disorders or genetic syndromes.
Even 100 years after its discovery, access to insulin still remains a challenge for many people around the globe. Barriers to access mainly relate to affordability and availability, with costs also related to associated necessary medical devices (e.g. test strips, syringes, pens), placing a large burden on people with diabetes and healthcare systems (Beran 2006; Beran 2016). For people with T1DM, the goal of insulin therapy is to provide insulin that mimics physiological insulin secretion in order to achieve near‐normal glycaemic levels. Insulin is most commonly administered by subcutaneous injection and is usually applied through insulin syringes, insulin pens or insulin pumps. Since the early 1920s, people with diabetes were treated with insulin, which was purified from bovine or porcine pancreas (animal insulin). Later, recombinant human insulin utilising recombinant DNA technology (Chance 1993), and insulin analogues (insulin‐like molecules, engineered on the basis of the molecular structure of human insulin by changing amino acid sequences and physiochemical properties) were marketed (Fullerton 2018; Hemmingsen 2021; Semlitsch 2020).
Early data indicated a loss of insulin potency depending on storage temperatures (Brange 1987). The stability of human insulin depends upon a number of environmental factors (e.g. purity, pH, humidity, changes in the primary structure of the insulin molecule, added substances for enhancing or prolonging insulin absorption) and is thought to be especially susceptible to temperature and sunlight exposure (Pingel 1972; Storvick 1968; Vimalavathini 2009). These factors may become more important because of increasingly hot weather conditions and extreme heat caused by the climate crisis (Atwoli 2021; Ebi 2021; Quijal‐Zamorano 2021). Temperature affects not only diabetes control (Westphal 2010), but also the pharmaceutical quality of various drugs, especially of protein and peptide‐based compounds such as insulin or monoclonal antibodies used to treat various inflammatory diseases. The potency of insulin is measured in units per millilitre (U/mL), indicating the blood glucose‐lowering activity of an insulin formulation per volume. High‐performance liquid chromatography (HPLC) is the gold standard for insulin concentration and insulin stability assessment (Farid 1989; Fisher 1986). HPLC quantification of the chemical potency of insulin is correlated with its biological activity. Traditionally, an in vivo rabbit biopotency assay was used to monitor blood glucose over time (Farid 1989; Fisher 1986; Smith 1985). However, the measured insulin potency might differ from the actual biological potency of insulin (Baechler 2020; Delbeck 2020a; Delbeck 2020b). Insulin potency may be impacted by high or low ambient temperature, sunlight and usage beyond the expiration date, as well as exceeding the recommended usage time once opened or in use. For insulin pumps worn close to the body, the temperature in the reservoir is even higher than room temperature, and constant movements during daily activities may increase fibril formation in the insulin reservoir (Herr 2014; Pryce 2009).
In order to prevent self‐aggregation during transport and storage, zinc ions and phenolic conservative agents are added to insulin formulations to stabilise insulin molecules as hexameric complexes (Brange 1997; Huus 2005; Huus 2006). Agitation or heating may cause the hexamers to dissociate and release monomers which are susceptible to forming fibrils (Ahmad 2003; Kurouski 2012). Formation of these insulin polymers is reported to occur up to 10 times faster at 37 °C compared to 25 °C (Weiss 2013). Amyloid fibrils may also exhibit immunogenic capabilities (Brange 1997; Mori 2021). Insulin fibrillation by means of aggregation of insulin molecules also adversely impacts potency (Groenning 2009). If insulin within fibrils is injected subcutaneously, bioavailability is reduced because stable bonds of insulin molecules within fibrils provide fewer insulin monomers available for resorption (Derewenda 1989). Fibrils may be visible in vials with the naked eye but are difficult to detect in pens or pump reservoirs. If the temperature rises, insulin degradation increases, and the combination of agitation and heat further accelerates the formation of agglomerates. Therefore, the advice is to discard insulin formations which appear visually modified (Shnek 1998). However, thermal denaturation is a complex process, correlated with both time and temperature, and it may be partially reversible (Huus 2005). The application of high temperatures for a prolonged time usually leads to denaturation due to irreversible conformational changes in insulin and the formation of insulin fibrils (Vimalavathini 2009). In contrast, fluctuating temperatures (compared to continuous heating conditions) may not cause irreversible aggregation of insulin molecules. For more information on insulin to help put data into perspective, see Appendix 1.
Recommendations by health authorities underline the fact that insulin is temperature sensitive and should be protected from heat and freezing conditions (ADA 2021; Bahendeka 2019; Danne 2018). The American Diabetes Association (ADA) published the following advice on its website (ADA 2021). "Although manufacturers recommend storing your insulin in the refrigerator, injecting cold insulin can sometimes make the injection more painful. To avoid this, many providers suggest storing the bottle of insulin you are using at room temperature. Insulin kept at room temperature will last approximately one month. Remember though, if you buy more than one bottle at a time to save money, store the extra bottles in the refrigerator. Then, take out the bottle ahead of time so it is ready for your next injection. Here are some other tips for storing insulin.
Do not store your insulin near extreme heat or extreme cold.
Never store insulin in the freezer, in direct sunlight, or in the glove compartment of a car.
Check the expiration date before using, and don't use any insulin beyond its expiration date.
Examine the bottle closely to make sure the insulin looks normal before you draw the insulin into the syringe."
Manufacturers' specifications recommend keeping insulin away from sunlight, not freezing it, and storing it in a refrigerator or at 'room temperature', usually not exceeding 25 °C or 30 °C (Lilly 2023; Novo Nordisk 2020a; Novo Nordisk 2020b). Nevertheless, room temperature may be defined differently in various parts of the world. Intact vials of insulin should be stored at low temperatures (i.e. between 2 °C and 8 °C), requiring affordable and reliable refrigeration. However, refrigeration may not be available or possible in some parts of the world (Ogle 2016). Moreover, unlike pharmaceutical refrigerators, household refrigeration may be unreliable, and temperatures can drop below the freezing point (Heinemann 2021). Once opened, a vial or cartridge can be stored at ambient temperature and used for approximately four to six weeks (Lilly 2023; Novo Nordisk 2020a; Novo Nordisk 2020b). The contents of the vial or cartridge are technically no longer sterile, and it is recommended to use the insulin in as short a time as possible to minimise concerns about potential microbiological contamination once the container has been opened or punctured. However, recommendations with regard to advised usage time once open and maximum temperature in‐use differ with type of insulin, brand, insulin concentrations and carrier (vial, pen, cartridge, pump) (table 1 in Heinemann 2021 and table S1 in Kaufmann 2021). For human insulin, the current shelf life in use may range from 10 to 45 days and the recommended maximum temperature in‐use varies between 25 °C and 37 °C. Tropical regions will often exceed these temperature thresholds and, due to the climate crisis, periods of extreme heat will occur more often in almost any part of the world. Whereas WHO (WHO 2011) and regulatory bodies provide guidance on how to handle the professional insulin cold chain, there does not seem to be a consensus on the issue of temperature and storage conditions of insulin at patients' level. Unclear or conflicting information about optimal insulin storage may not only put patients' health at risk, but may also lead to the possibility that unused insulin is unnecessarily discarded (Flood 2015; Grajower 2003; Grajower 2014).
For this review, optimal cold‐chain management from manufacturing until the point of delivery to people with diabetes, and adequate storage conditions at the professional healthcare level, are seen as a prerequisite to investigating whether it is possible for consumers to safely use insulin outside manufacturers' recommended temperature range, especially without refrigeration during the period of use in hot climate conditions. This is particularly important in tropical and low‐resource settings to remove at least one significant barrier in the treatment of people with diabetes aside from the major health equity issues of affordability and availability of human insulin (Kaufmann 2021).
Why it is important to do this review
According to Naby Baldé, Vice‐President of the IDF, "The majority of families in low‐ and middle‐income countries do not have refrigeration facilities in their homes, or electric power and, therefore, diverse methods of safely storing insulin should be devised" (Devi 2021). Around 770 million people in LMICs do not have access to electricity (Burki 2022; Pande 2022). Therefore, we believe that this Cochrane Review provides useful information for the global diabetes community residing in tropical regions and living in challenging settings such as war‐torn and natural disaster‐stricken areas or experiencing climate crisis‐related periods of extreme heat.
Objectives
To analyse the effects of storing human insulin above or below the manufacturers' recommended insulin temperature storage range or advised usage time, or both, after dispensing human insulin to people with diabetes.
Methods
Inclusion and exclusion criteria
With regard to the objectives, we established the following 'Population, Intervention, Comparator, Outcome, Timing' (PICOT) table, as follows.
| Item | Definition |
| Population | 1. Clinical studies: people with type 1 or type 2 diabetes mellitus treated with human insulin 2. Laboratory studies investigating insulin thermostability |
| Intervention | Storage of human insulin above or below manufacturers' recommended temperature storage range or advised usage time, or both |
| Comparator | Storage of human insulin according to manufacturers' recommended temperature storage range or advised usage time, or both |
| Outcome | 1. and 2. Potency and bioactivity parameters (e.g. measured by HPLC, bioassay, assessment of insulin monomer structure, insulin efficiency, structural physical damage, physical damage visible to the naked eye, antimicrobial activity) 1. Clinical parameters (e.g. HbA1c, blood sugar, hypoglycaemic episodes, diabetic ketoacidosis, adverse events including potential additive effects of temperatures and recommended storage periods, health‐related quality of life) |
| Timing | Temperature studies: any time of exposure Usage time studies: above manufacturers' recommended usage time for the analysed human insulin formulation |
| HbA1c: glycosylated haemoglobin A1c; HPLC: high‐performance liquid chromatography. | |
Types of studies
We included any clinical study fulfilling the above‐mentioned PICOT criteria. For this review, we were especially interested in studies emulating living conditions of people with diabetes, mimicking the daily use of human insulin. We also included laboratory studies investigating the storage of human insulin above or below manufacturers' recommended temperature storage range or advised usage time, or both.
Diagnostic criteria for diabetes mellitus
In order to be consistent with changes in the classification of and diagnostic criteria for diabetes mellitus over the years, the diagnosis should have been established using the standard criteria valid at the time of the study commencing (e.g. ADA 2003; ADA 2017; WHO 1999). Ideally, the diagnostic criteria should have been described. We used the study authors' definition of diabetes mellitus if necessary.
Specific exclusion criteria
We excluded the following studies.
Studies investigating animal insulin.
Studies investigating insulin analogues.
Studies investigating the immediate effects of ambient temperature on people with diabetes (i.e. no focus on storage conditions of human insulin).
In‐vitro studies on cells or receptors.
Case reports or case series.
Search methods for identification of studies
Electronic searches
One review author (MIM) developed the search strategies using analytical text‐mining of 24 relevant publications already known to a second review author (BR), using the tools PubReMiner (hgserver2.amc.nl/cgi-bin/miner/miner2.cgi) and Yale MeSH Analyzer (mesh.med.yale.edu).
We searched the following sources from inception of each database to 12 July 2023 and placed no restrictions on the language of publication:
Cochrane Central Register of Controlled Trials (CENTRAL) via the Cochrane Register of Studies Online (CRSO);
MEDLINE (Ovid MEDLINE(R) ALL 1946 to 22 July 2023);
CINAHL EBSCO (Cumulative Index to Nursing and Allied Health Literature);
Science Citation Index Expanded (Web of Science);
WHO Global Index Medicus (www.globalindexmedicus.net);
ClinicalTrials.gov (www.clinicaltrials.gov);
WHO International Clinical Trials Registry Platform (ICTRP) (www.who.int/trialsearch).
For detailed search strategies, see Appendix 2. We continuously applied an email alert service for MEDLINE via OvidSP to identify newly published studies using the search strategy detailed in Appendix 2.
Searching other resources
We contacted the three leading insulin‐producing pharmaceutical companies (Eli Lilly and Company, Novo Nordisk, Sanofi) and two pharmaceutical companies producing biosimilar insulin (BIOTON, Gan and Lee Pharmaceuticals) to obtain published and unpublished data on insulin thermostability. We attempted to identify other potentially eligible studies or ancillary publications by searching the reference lists of included studies, systematic reviews and meta‐analyses. We also contacted the authors of included studies to obtain additional information on the studies and establish whether we may have missed further studies.
Data collection and analysis
Selection of studies
Two review authors (BR, BB) independently screened the abstract, title, or both, of every record retrieved by the electronic searches. We obtained the full text of all potentially relevant records. We planned to resolve disagreements through consensus or by recourse to a third review author (MIM). If we could not resolve a disagreement, we planned to categorise the study as 'awaiting classification' and would have contacted the study authors for clarification. We presented an adapted PRISMA flow diagram to show the process of study selection (Page 2021). We listed all articles excluded after full‐text assessment in the Characteristics of excluded studies table and provided the reasons for exclusion.
Data extraction and management
For studies that fulfilled our inclusion criteria, one review author (BR) extracted the type of experimental study, study characteristics, stated aim of the study, type and brand of human insulin, use of concomitant oral antidiabetic agents, data on cold chain management, temperature (constant, oscillating or both), light exposure and usage time, container use (unopened, open, in‐use), container type (vial, cartridge, prefilled pens), sterility, injection device (syringe, pen, pump), storage conditions, potency/bioactivity parameters and clinical outcomes.
Another review author (BB) checked these data extractions, and we planned to resolve any disagreements by discussion or, if required, by consultation with a third review author (MIM). We established our own context‐specific data extraction sheets after piloting data extraction for five studies. We planned to store all extracted data and risk of bias evaluations (see below) online in an open repository (Zenodo.org).
We emailed all authors of included studies to enquire whether they would be willing to answer questions regarding their studies. We presented the results of this survey in an appendix. Thereafter, we sought relevant missing information on the study from the primary study author(s), if required.
We planned to provide information for potentially relevant ongoing trials in the Characteristics of ongoing studies table.
Dealing with duplicate and companion publications
In the event of duplicate publications, companion documents or multiple reports of a primary study, we maximised the information yield by collating all available data, and we used the most complete data set aggregated across all known publications. We listed duplicate publications, companion documents, multiple reports of a primary study, and trial documents of included trials (such as trial registry information) as secondary references under the study identifier of the included study. Furthermore, we listed duplicate publications, companion documents, multiple reports of a study and trial documents of excluded trials (such as trial registry information) as secondary references under the study identifier of the excluded study.
Data from clinical trial registers
If data from included trials were available as study results in clinical trial registers, such as ClinicalTrials.gov or similar sources, we planned to make full use of this information and extract the data. If there was also a full publication of the study, we collated and critically appraised all available data. If an included study was marked as completed in a clinical trial register but there was no additional information (study results or publication, or both) available, we planned to add this study to the Characteristics of studies awaiting classification table.
Assessment of risk of bias in included studies
Two review authors (BR, BB) planned to independently assess the risk of bias for each included study. We planned to resolve disagreements by consensus or by consulting a third review author (MIM). If adequate information was not available from the study publications, study protocols or other sources, we planned to contact the study authors to request missing data on items relating to the risk of bias.
For randomised controlled trials (RCTs), we planned to use the Cochrane RoB 2 tool (Flemyng 2023; Higgins 2022a). For non‐randomised clinical studies, we planned to use the Cochrane risk of bias in non‐randomised studies of interventions tool, ROBINS‐I (Sterne 2016). For all other types of included studies, we planned to address risk of bias in a meaningful and transparent way, if possible, using appropriate instruments described in the literature.
Dealing with missing data
We attempted to obtain missing data from the authors of included studies.
Data synthesis
We did not expect data suitable for meta‐analysis. If possible, we planned to perform statistical analyses according to the statistical guidelines presented in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2022b). In case statistical analyses were not possible or meaningful, we planned to present data in a highly structured tabulated format which we wanted to store online in an open repository (Zenodo.org).
Subgroup analysis
We expected the following characteristics to introduce heterogeneity, and we planned to investigate the following subgroups, if feasible.
Insulin type
Storage temperature (depending on data)
Usage time (depending on data)
Injection device
Sensitivity analysis
Should meta‐analysis have been possible, we planned to explore the influence of important factors on effect sizes, by performing sensitivity analyses in which we restricted the analyses to the following.
Published studies
Studies with low risk of bias
Large studies, to establish the extent to which they dominate the results
Certainty of the evidence
For clinical studies, we planned to present the overall certainty of the evidence for each outcome specified below, according to the GRADE approach, which takes into account issues related to internal validity (risk of bias, inconsistency, imprecision, publication bias) and external validity (such as directness of results). Two review authors (BR, BB) planned to independently assess the certainty of the evidence for each outcome. We wanted to resolve any differences in assessment by discussion or by consultation with a third review author (MIM). We planned to use GRADEpro GDT software and wanted to present evidence profile tables as an appendix.
If meta‐analysis was not possible, we presented the results in a narrative format in the summary of findings tables. If possible, we justified all decisions to downgrade the certainty of the evidence using footnotes, and we made comments to aid the reader's understanding of the Cochrane Review when necessary.
Summary of findings table
We presented a summary of the evidence in summary of findings tables. For clinical studies, we provided key information about the best estimate of the magnitude of effect, in relative terms and as absolute differences for each relevant comparison of alternative management strategies; the numbers of participants and studies addressing each important outcome; and a rating of overall confidence in effect estimates for each outcome. We created summary of findings tables using the methods described in the Cochrane Handbook for Systematic Reviews of Interventions (Schünemann 2022).
According to our PICOT setting, interventions presented in the summary of findings tables were planned to be the storage of human insulin above or below manufacturers' recommended temperature storage range or advised usage time, or both. The comparators were the storage of human insulin according to manufacturers' recommended temperature storage range or advised usage time, or both.
We wanted to evaluate the following outcomes reported in clinical studies, listed according to priority.
Potency/bioactivity parameters of human insulin
Diabetic ketoacidosis
Hypoglycaemic episodes
Adverse events
Health‐related quality of life
Glycosylated haemoglobin A1c (HbA1c)
Fasting blood/plasma glucose
Results
Results of the search
After removal of duplicates, our search identified 7051 records (Figure 1). We excluded most of the references on the basis of their titles and abstracts because they clearly did not meet the inclusion criteria. We evaluated 91 full‐text articles. Of these, we excluded 69 articles. The main reasons for exclusion were studies investigated insulin other than human insulin, commentaries/viewpoints providing no data and studies on storage conditions without insulin. We did not identify any ongoing trials. Finally, we included 17 studies (22 records). We supplemented our searches by screening reference lists of all eligible articles.
1.

Study flow diagram.
We contacted all study authors in case we required further information or clarification (Appendix 3). Six authors replied (Baechler 2020; Delbeck 2021a; Kaufmann 2021; Kongmalai 2021; Silva 2013; Silva‐Jr 2022), and three authors made additional data available (Delbeck 2021a; Kaufmann 2021; Kongmalai 2021).
Building on the advice from the WHO, we contacted five manufacturers of human insulin (BIOTON and Gan and Lee Pharmaceuticals (both biosimilar manufacturers), Eli Lilly and Company, Novo Nordisk, Sanofi). We requested specific data on human insulin when tested above or below the manufacturers' recommended insulin temperature storage range or advised usage time, or both and provided an Excel template detailing the sort of container, batch number, storage condition, time points of storage, investigated components, results including result unit and reference range. We received data with varying degrees of detail from BIOTON, Eli Lilly and Company, Novo Nordisk and Sanofi.
Baseline characteristics of included studies
For details on baseline characteristics of the included studies, see Table 4; Table 5.
1. Baseline characteristics (I).
| Study ID | Study type | Stated aim of study (quotes) | Country | Type and brand of human insulin |
| Al Shaibi 1999 | Pilot clinical study | "The purpose of our study was to demonstrate the possible effect on the bioactivity of insulin, following storage in a zeer, a semiporous clay pot containing water, in desert conditions." | Saudi Arabia | Neutral human insulin |
| Baechler 2020 | Lab study | "In an attempt to challenge the findings of Carter and Heinemann, we set out to develop a high‐throughput (1 min/sample) high‐resolution MS method to quantify insulin content and stability in vials." | Switzerland | Human insulin Insulin aspart Faster‐acting insulin aspart Insulin glargine Insulin lispro |
| Delbeck 2021a | Lab study | "Infrared (IR) spectroscopy has been successfully utilized for secondary structure analysis in cases of protein misfolding and fibril formation." | Germany | Insulin detemir Insulin aspart NPH insulin human Insulin lispro Insulin lispro NPH insulin Insulin glargine |
| Dunbar 1986 | Lab study | "The purpose of this study was to evaluate the immunologic and radioreceptor degradation of insulin storage in commercial plastic syringes." | USA | Human insulin Porcine insulin Lente iletin |
| Gregory 1991 | Lab study | "First, how much insulin transformation had occurred in vials by the time patients had finished with them? Next what was the importance of temperature, light, agitation, and regular withdrawal of insulin on the rates of appearance of three groups of ITP (insulin transformation product)? Finally, how did rates of transformation in vials issued to patients compare with vials from the same batch stored in various laboratory environments?" | UK | Human insulin Beef insulin |
| Kaufmann 2021 | Lab study | "In this work, therefore, the question addressed is whether commercially available insulin is stable under high oscillating ambient temperatures for a period of four weeks (28 days), which corresponds to the usual recommended period of use of most commercially available insulins upon opening of the vial or cartridge." | Switzerland | Actrapid Insulatard Mixtard 30 Novorapid Lantus Solostar Humalog KwikPen Humalog KwikPen mix 25 |
| Koffler 1992 | Lab study | "The aim of this study was to evaluate the sterility of insulin and syringes when they were kept under refrigeration for up to 10 days." | Israel | Human insulin Rapid‐acting insulin |
| Kongmalai 2021 | Lab study | "Investigates the effect of temperature on the stability of basal insulin at 28 days after opening." | Thailand | NPH insulins Insulin analogue |
| Lull 2013 | Lab study | "The objective of this study was to determine if U‐500 insulin is stable when stored in prefilled syringes at 4 °C for up to 28 days." | USA | Human insulin |
| Moses 2019 | Lab study | "On the background of the article by Carter and Heinemann and given the importance of insulin quality on patient outcomes, we evaluated insulin concentrations in vials from three different sources: (1) we analyzed insulin content and stability based on production batches covering 7 years of production, (2) we measured insulin content from the last three years of insulin samples returned to Novo Nordisk from patients in the United States because of concerns expressed by the patients, and (3) we reviewed data from 10 years of independent European Medicines Association (EMA) sampling and testing of products." | USA | Short‐acting insulin Intermediate‐acting insulin |
| Pendsey 2023 | Lab study | "In this pilot study, we aimed to identify: (1) the potency of six 100 IU/mL insulins (vials of human soluble; Eli Lilly), human isophane (Eli Lilly), and human soluble–isophane (30:70; Novo Nordisk); and 3 mL cartridges of insulin aspart (Novo Nordisk), and two preparations of insulin glargine (Sanofi and Eli Lilly) stored unopened for 1–4 months in non‐refrigerated conditions in a real‐world setting during the summer in India, all compared with control samples of each insulin, which remained refrigerated; and (2) whether there was any difference in storage temperatures and changes in potency between samples stored within and outside clay pots. These insulins were chosen to include commonly used insulins." | India | Human insulin: Humulin R Humulin N Mixtard 30/70 Analogue insulin: Novorapid Lantus/Basaglar |
| Schrader 1985 | Lab study | "In this study we investigated with High Performance Liquid Chromatography (HPLC) (reversed phase chromatography and gel‐filtration) the aggregational behaviour of pork and human soluble insulin formulations under variations of motion and temperature." | Germany | Human insulin Pork insulin |
| Shnek 1998 | Lab study | "In this study, aspects of the test development have been addressed such as the test conditions, automation of physical stress testing, and results for different formulations." | USA | Human insulin regular, NPH, biphasic (70/30) |
| Silva 2013 | Lab study | "To evaluate the stability of two long‐acting insulin formulations, Humulin 70/30 and Levemir, after 28 days." | USA | Human insulin biphasic (70/30) Insulin detemir |
| Silva‐Jr 2022 | Lab study | "In this work, we investigated the effects of temperature and intermittent handling on the formation of subvisible and submicrometer protein particles in commercial insulin formulations." | Brazil | Human insulin H – regular H Human insulin N – regular N |
| Tarr 1991 | Lab study | "This study was conducted to determine the stability of a variety of biosynthetic human insulin products, along with their preservatives, when stored in two commonly used plastic insulin syringes." | USA | Human insulin regular, NPH, biphasic (70/30) |
| Vimalavathini 2009 | Lab study | "… we undertook this study to compare the potency of human insulin from three different manufacturers after storing at different temperatures and durations." | India | Human insulin regular, biphasic (30/70) |
IU: insulin unit; lab: laboratory; MS: mass spectrometric; NPH: neutral protamine Hagedorn.
2. Baseline characteristics (II).
| Study ID | Injection and container device | Temperature and storage conditions | Measurements |
| Al Shaibi 1999 | Vials (opened) |
|
Twice‐daily sampling of insulin 10 U (simulation of usage conditions)
|
| Baechler 2020 | Vials (unopened) |
|
|
| Delbeck 2021a | Cartridges (unopened) |
|
|
| Dunbar 1986 | Prefilled syringes |
|
|
| Gregory 1991 | Vials (opened, some samples) |
|
Twice‐daily sampling (simulation of usage conditions)
|
| Kaufmann 2021 | Vials and cartridges/pens (opened, some samples) |
|
For some samples, insulin was taken out daily (simulation of usage conditions with dosages of 10 IU in the morning and 12 IU in the evening)
|
| Koffler 1992a | Prefilled syringes |
|
|
| Kongmalai 2021 | Cartridges/pens (opened) |
|
2 units of insulin were discarded every day (simulation of usage conditions)
|
| Lull 2013 | Prefilled syringes |
|
|
| Moses 2019 | Vials (unopened) |
|
|
| Pendsey 2023 | Vials (unopened) |
|
|
| Schrader 1985 | Vials (unopened) |
|
|
| Shnek 1998 | Cartridges/pens (unopened) |
|
Resuspensions were conducted twice daily on a mechanical device outside the incubator unit
|
| Silva 2013 | Vials (unopened) |
|
|
| Silva‐Jr 2022 | Vials (unopened) |
|
Protein structure:
|
| Tarr 1991 | Prefilled syringes |
|
|
| Vimalavathini 2009 | Vials (probably unopened) |
|
|
aShort‐acting insulin was withdrawn for commercial reasons by the manufacturer.
1H NMR: proton nuclear magnetic resonance spectroscopy; HMWP: high molecular weight protein; HPLC: high‐performance liquid chromatography; IU: insulin unit; (RP)‐HPLC: (reverse‐phase) high‐performance liquid chromatography; SD: standard deviation; U: unit; UV: ultraviolet.
Of the 17 included studies, there was only one pilot clinical study (Al Shaibi 1999), and the remaining 16 investigations were laboratory (in‐vitro) studies. Ten studies investigated storage conditions for insulin vials (Al Shaibi 1999; Baechler 2020; Gregory 1991; Kaufmann 2021; Moses 2019; Pendsey 2023; Schrader 1985; Silva 2013; Silva‐Jr 2022; Vimalavathini 2009). Three studies investigated storage conditions for insulin cartridges/pens (Delbeck 2021a; Kongmalai 2021; Shnek 1998). Four studies investigated storage conditions for prefilled plastic syringes (Dunbar 1986; Koffler 1992; Lull 2013; Tarr 1991).
Six studies were published in the USA; two studies each in Germany, India and Switzerland; and one study each in Brazil, Israel, Saudi Arabia, the UK and Thailand.
Data from study publications
In this review, we reported data on human insulin formulations only (for details, see Characteristics of included studies table). To put human insulin data into perspective, see Appendix 1.
Pilot clinical study
For an overview of main results, see Table 1.
Eight healthy physicians (aged 25 to 60 years) participated in the study (Al Shaibi 1999). In the open section of the study, four males used 'prestorage' insulin as reference insulin. In the double‐blind section of the study, four females used refrigerated (4 °C) insulin as reference. For these participants, the reference insulin and the insulin stored in a clay pot were available at the same time and the order of use was randomised using a coin toss. Here, both study participants and study personnel were reported to be blinded to the allocation. In addition, two people with diabetes who used regular and neutral protamine Hagedorn (NPH) insulin stored in a clay pot during a two‐month period while living in a tent in the desert participated.
Human insulin was stored in a refrigerator at 4 °C or in a zeer (a semi‐porous unglazed clay pot enabling water to slowly seep through the wall and evaporate, lowering the temperature of the remaining water). A glass jar was partially filled with desert sand and put in the centre of the zeer. A water bath was created by adding water outside the jar to the same level as that of the sand in the glass jar. Water was added to the mark (topped up) daily from a larger water storage zeer. The zeer was closed with a clay lid and kept in the shade. An exact replica, but without water (a dry zeer) was also used to store insulin and record temperatures. Additional bottles of insulin were kept outside in the shade, in the bathroom of an air‐conditioned house and in a refrigerator. Ten units (U) of insulin were removed twice daily from each insulin vial to simulate usage conditions. Temperature readings were taken at 7:00 hours or 12:00 hours during the six‐week storage period.
To investigate insulin sensitivity, baseline blood samples were taken in fasting participants and insulin 0.1 U/kg bodyweight was intravenously applied. For 30 minutes after the injection, blood samples were drawn every five minutes. Study authors measured the fall in plasma glucose levels using a computerised best fit method.
The mean temperatures were as follows: outside 38.3 °C (range 34 °C to 43 °C), insulin zeer 26.6 °C (range 25 °C to 27 °C), dry zeer 34.6 °C (range 30 °C to 39 °C), insulin vials stored in the shade 35.3 °C (range 31 °C to 39 °C), water zeer 26.7 °C (range 25 °C to 28 °C), bathroom 26.4 °C (range 25 °C to 27 °C) and refrigerator 4 °C. There were no changes in colour or appearance of the insulins. The mean fall in plasma glucose levels following reference insulin was 0.213 mmol/L/minute, whereas it was 0.192 mmol/L/minute following the zeer‐stored insulin, representing 9.9% less fall in glucose in the zeer‐stored insulin (very low‐certainty evidence). Investigators reported that the observed difference in the rate of glucose decline was not 'statistically significant'. The two participants with diabetes reported no changes in the appearance of insulin or adverse events and no changes in diabetic control.
Study authors did not report on diabetic ketoacidosis, hypoglycaemic episodes, adverse events, health‐related quality of life or HbA1c.
Certainty of the evidence was very low due to risk of bias (unclear randomisation and allocation concealment, indirectness (surrogate outcome, insufficient time frame) and imprecision (very small sample size, one study only)).
In‐vitro studies
For an overview of the main results, see Table 2.
Of note, European and US Pharmacopeia (USP) stipulate that human insulin should have a potency of 95% to 105% of the potency stated on the label, expressed in USP insulin human units/mL (i.e. human insulin labelled '100 U/mL' has to contain 95 U/mL to 105 U/mL of insulin) (European Pharmacopoeia 2023; USP 2016). No data exist clearly relating different insulin potencies to insulin efficacy (i.e. the lowering of blood glucose). However, it is evident that, for example, insulin 80 U/mL lowers blood glucose to a lesser degree than, for example, 90 U/mL. However, the clinical insulin effect depends on many individual factors (e.g. age, bodyweight, physical activity, dietary habits, comorbidities such as infections and blood glucose values before insulin injection).
Due to considerable heterogeneous study data, it was not possible to draw up a straightforward evidence synthesis of publication results. Instead, Table 6 provides an overview of data and key results from study publications. The included studies reported numerous methods, laboratory measurements and storage conditions. Study authors often applied reverse‐phase high‐performance liquid chromatography (RP‐HPLC) to measure insulin potency (Kaufmann 2021; Kongmalai 2021; Lull 2013; Moses 2019; Pendsey 2023; Schrader 1985; Shnek 1998; Silva 2013; Tarr 1991; Vimalavathini 2009). Older publications cited methods that are rarely used today (e.g. radioimmunological activity) (Dunbar 1986; Schrader 1985), degradation of protein (Gregory 1991), cultures in infusion broths (Koffler 1992), and short‐time plasma glucose levels after intravenous insulin injection (Al Shaibi 1999). Some study authors employed sophisticated methods (e.g. mass spectrometry (Baechler 2020) or proton nuclear magnetic resonance spectroscopy (1H NMR) (Pendsey 2023)) for insulin concentration and newer methods to evaluate insulin protein structural changes (Delbeck 2021a; Silva‐Jr 2022).
3. Overview of data from study publications.
| Study ID | Study type | Type of insulin | Injection and container device | Maximum temperature | Maximum storage period | Main measurement(s) | Key resultsa |
| Al Shaibi 1999 | Pilot clinical study | SAI | Vials | Insulin in clay pot 26.6 °C Insulin in refrigerator 4 °C | 6 weeks | Plasma glucose | No difference |
| Baechler 2020 | Lab study | SAI | Vials | Up to 37 °C | 85 days | Insulin potency | 98.7 U/mL (CV 4.6%) |
| Gregory 1991 | Lab study | SAI | Vials | 37 °C | 26 weeks | Insulin potency | −1.184% total protein/week |
| Kaufmann 2021 | Lab study | SAI IAI MI | Vials | 25 °C ‐ 37 °Cb 37 °Cc | 12 weeks 1 week / 2 weeks | Insulin potency Microbial contamination Protein structure | SAI: 101.3% (SD 2.3) IAI: 101.6% (SD 8.9) MI: 101.9% (SD 3.5) MI: −15.4% / SAI: −12.5% No microbial contamination No changes |
| Moses 2019 | Lab study | SAI IAI | Vials | 30 °C | 10 weeks | Insulin potency | SAI: −4.0 IU/mL (95% CI 4.9) IAI: 1.4 IU/mL (95% CI 2.3) |
| Pendsey 2023 | Lab study | SAI IAI MI | Vials | 28.9 °C | 4 months | Insulin potency Protein structure | SAI: −2.4% IAI: −0.7% MI: −1.4% Small changes |
| Schrader 1985 | Lab study | SAI | Vials | 37 °C | 72 hours | Insulin potency Bacterial growth | No changes No bacterial growth |
| Silva 2013 | Lab study | IAI | Vials | 24 °C | 42 days | Insulin potency | 93–94% AUC |
| Silva‐Jr 2022 | Lab study | SAI IAI | Vials | 37 °C | 5 weeks | Protein structure | Amorphous agglomerates (both at 4 °C and 37 °C) No changes in chemical stability |
| Vimalavathini 2009 | Lab study | SAI MI | Vials | 37 °C | 28 days | Insulin potency | SAI: −18% IAI: −14% |
| Delbeck 2021a | Lab study | IAI | Cartridges | 37 °C | 10 weeks | Protein structure | Small changes |
| Kongmalai 2021 | Lab study | IAI | Pens | 37 °C | 28 days | Insulin potency | −1.5% to +0.7% |
| Shnek 1998 | Lab study | SAI, IAI, MI | Cartridges | 25–37 °Cb 37 °Cc | 28 days | Insulin potency | Within specifications Visual changes (agglomerates) |
| Dunbar 1986 | Lab study | SAI | Prefilled syringes | 4 °C | 14 days | Insulin potency | No changes |
| Koffler 1992 | Lab study | SAI, IAI | Prefilled syringes | 4 °C | 10 days | Sterility | No signs of bacterial growth |
| Lull 2013 | Lab study | SAI | Prefilled syringes | 5 °C | 28 days | Insulin potency | 93.3% (SD 2.4) AUC |
| Tarr 1991 | Lab study | SAI IAI MI | Prefilled syringes | 23 °C | 28 days | Insulin potency Sterility |
SAI: −1.9% to +1.7%
IAI: +0.7% to +4.7%
MI: −1.0% to −1.7% No microbial growth |
aAll insulin activity data refer to baseline (time zero). According to US Pharmacopeia (USP) human insulin should have a potency of 95% to 105% of the potency stated on the label, expressed in USP insulin human units/mL, i.e. human insulin labelled '100 U/mL' has to contain 95 U/mL to 105 U/mL of insulin bOscillating temperatures cContinuous temperature
AUC: area under the curve; CI: confidence interval; CV: coefficient of variation; IAI: intermediate‐acting insulin; IU: insulin unit; MI: mixed insulin (short‐acting + intermediate‐acting insulin); SAI: short‐acting insulin; SD: standard deviation; U: unit.
Applied temperature and storage of human insulin vials and cartridges/pens also varied across studies. Study authors investigated insulin potency at 24 °C for up to 42 days (Silva 2013), at 30 °C for up to 10 weeks (Moses 2019), and at 26.3 °C to 28.9 °C for up to four months (Pendsey 2023). Many authors investigated insulin potency (Baechler 2020; Gregory 1991; Kaufmann 2021; Kongmalai 2021; Vimalavathini 2009), or structural changes of the insulin protein (Delbeck 2021a; Silva‐Jr 2022), at up to continuous 37 °C, ranging from 28 days (Vimalavathini 2009) to 26 weeks (Gregory 1991). Two publications applied fluctuating temperatures between 25 °C and 37 °C for 28 days to 12 weeks, mimicking real‐life conditions (Kaufmann 2021; Shnek 1998). Two publications used additional stress tests, such as rotation of cartridges (Shnek 1998) or shaking of insulin vials (Schrader 1985).
Four studies reported twice‐daily insulin sampling simulating usage by people with diabetes (Al Shaibi 1999; Gregory 1991; Kaufmann 2021; Kongmalai 2021). Multiple punctures of vials and cartridges as well as reduced volume of insulin in the container did not alter insulin potency.
Investigations on prefilled syringes, important for people with visual or manual impairments, included a temperature range of 4 °C to 23 °C for 10 to 28 days (Dunbar 1986; Koffler 1992; Lull 2013; Tarr 1991). There was no clinically relevant loss of insulin activity, and no signs of microbial growth.
Investigations on vials with different temperature and storage conditions showed no (Kaufmann 2021, fluctuating temperatures at 25 °C to 37 °C for up to three months) or no clinically relevant reduction in insulin concentrations for short‐acting, intermediate‐acting and mixed insulin (Al Shaibi 1999; Baechler 2020; Moses 2019; Pendsey 2023; Schrader 1985; Silva 2013; Silva‐Jr 2022). Two publications reported a more pronounced loss in insulin activity: Vimalavathini 2009 noted 18% after 28 days at 37 °C and 14% after 28 days at 37 °C for short‐acting and biphasic human insulin, respectively. Kaufmann 2021 noted a decrease of 17.2% after eight weeks at continuous 31 °C for NPH insulin. These authors also reported a decrease of 15.4% after one week at continuous 37 °C for premixed short‐acting/NPH insulin as well as a 12.5% decrease after two weeks at 37 °C for short‐acting insulin.
Investigations on cartridges/pens did not show a decrease in insulin potency outside the specification limits after 28 days of fluctuating temperatures at 25 °C to 37 °C and agitation for intermediate‐acting and mixed insulin (Shnek 1998). However, after continuous high temperatures over 28 days at 37 °C and agitation, there were visual changes (Shnek 1998). Other investigators observed no reductions of insulin activity outside the specification limits after 28 days at 37 °C for intermediate‐acting insulin (Kongmalai 2021), or insulin protein structural changes after 10 weeks at 37 °C for intermediate‐acting NPH insulin (Delbeck 2021a).
The following sections provide a detailed overview of the individual study data reported in publications.
Studies on vials
Ten studies investigated storage conditions for insulin vials (for the pilot clinical study (Al Shaibi 1999) see above).
Schrader 1985: various insulin manufacturers provided soluble neutral human insulin 40 U/mL which were investigated by HPLC, using reversed phase and gel‐filtration chromatography. Preparations were shaken for 72 hours at 37 °C and at ambient temperature. Radioimmunological activity was tested and investigators inoculated parts of the insulin sediment on culture plates to look for bacterial growth. Investigations performed at ambient temperature showed the same molecular insulin weight as investigations performed at 37 °C. The material inoculated on culture plates showed no bacterial growth.
Gregory 1991: single vials of short‐acting human insulin were stored under each of the following environmental conditions and in the laboratory: 4 °C in the dark; room temperature 19 °C to 24 °C in the dark; room temperature 18 °C to 25 °C in daylight; in direct sunlight on a window sill (8 °C to 35 °C); at 37 °C in the dark; and carried in the breast pocket of a shirt. Some vials were subjected to twice‐daily sampling (i.e. small amounts of insulin were removed to simulate injections). HPLC measurements were taken immediately after dispensing and at 2, 4, 6, 8, 10, 14, 18 and 26 weeks. The initial insulin content in human insulin vials was 98.2% (standard deviation (SD) 0.08) of total protein. None of the environmental conditions caused a significant loss of total protein during the six months of study. Human insulin kept at 4 °C and in the dark without sampling was associated with the slowest degradation (−0.069% total protein/week). The vials kept in direct sunlight and at 37 °C in the dark degraded most quickly (−0.749% total protein/week and −1.184% total protein/week, respectively). The slope estimates from the four vials kept at room temperature were similar. Repeated sampling only affected the degradation rate in the vials kept at 4 °C in the dark.
Vimalavathini 2009: two human insulin formulations, short‐acting insulin and mixed insulin (30/70) were purchased from the open market from three different manufacturers. Human insulin formulations were stored at five different temperatures (i.e. 5 °C (refrigerator), 25 °C (air‐conditioned room), 26° (mud pot), 32 °C (room temperature) and 37 °C (room temperature in summer)). Samples were drawn from these vials every seven days for 28 days and analysed by HPLC. Study authors also performed an insulin tolerance test in rabbits showing a considerable decrease in blood glucose levels with respect to basal glucose (for details, see Table 7). All three brands of insulin for both formulations did not differ considerably in their insulin content during various sampling times and temperatures. There was no difference in concentration of insulin in vials stored at 25 °C and 26 °C compared to those stored at 5 °C on all five sampling days. Insulin in vials stored in the mud pot (26 °C) retained insulin potency. There was a 14% and 18% decrease in the concentration of short‐acting insulin in vials stored at 32 °C and 37 °C on day 28. There was an 11% and 14% decrease in the concentration of short‐acting insulin in biphasic insulin formulation in vials stored at 32 °C and 37 °C on day 28.
Silva 2013: intermediate‐acting insulin in vials were stored in a refrigerator at 2 °C to 8 °C and at room temperature at 22 °C to 24 °C. A modified HPLC assay was used to quantify insulin area under the curve (AUC) profiles under different storage conditions (i.e. time 0 (opening) and days 28, 35 and 42). Investigators could not confirm which peaks corresponded to metacresol preservative or insulin nor additional procedures to detect degradants. After 35 days at room temperature, the 12‐minute and 21‐minute peak ranged between 94% and 101% and 94% and 100% of time zero AUC reference values, respectively. After 42 days, the 12‐minute and 21‐minute peak was 93% and 94% of time zero AUC reference values, respectively.
Moses 2019: short‐acting and intermediate‐acting insulins were analysed for insulin concentration and stability by means of RP‐HPLC. Following storage for 30 months at 5 °C and handling as per procedure during the cold‐chain transport and during pharmacy storage samples were then: 1. left at 30 °C for six weeks to mimic in‐use impact (mimicking as per label conditions); and 2. left for four weeks at 30 °C to mimic non‐recommended handling conditions and then left at 30 °C for six weeks to mimic in‐use impact. None of the samples were outside the US Food and Drug Administration's (FDA) specifications. After 6 and 10 weeks at 30 °C, the mean decrease in insulin concentration for short‐acting insulin was 3.1 IU/mL (95% confidence interval (CI) 4.0) and 4.0 IU/mL (95% CI 4.9), respectively. After 6 and 10 weeks at 30 °C, the mean decrease in insulin concentration for intermediate‐acting insulin was 1.0 IU/mL (95% CI 1.9; as reported in publication) and 1.4 IU/mL (95% CI 2.3; as reported in publication), respectively.
Baechler 2020: vials containing short‐acting human insulin were stored according to manufacturer specifications until measurement of insulin content and stability by mass spectrometry. Formulations were sampled on days 1, 2, 3, 4, 5, 8, 9, 10, 11, 12, 15, 16, 17, 54 and 85 upon storage at 4 °C, 22 °C and 37 °C in temperature‐controlled environments with electronic temperature monitoring (either refrigerator or incubators) to simulate various real‐life conditions. There was no systematic difference in insulin content between unopened vials (obtained from the pharmacy) and already used vials (obtained from hospital wards and people with diabetes). Screening for degradation products throughout the study was negative. The mean insulin concentrations (with coefficient of variation (CV)) over all measurements and 86 days were as follows: 4 °C: 98.6 U/mL (CV 5.5%); 22 °C: 99.4 U/mL (CV 5%); 37 °C: 98.7 U/mL (CV 4.6%).
-
Kaufmann 2021: specifically investigated the influence of fluctuating temperatures on insulin thermostability, thereby mimicking cycling temperatures which are observed in hot regions and in conflict and humanitarian emergency settings. The study authors measured temperature fluctuations in the Dagahaley refugee camp (Northern Kenya) and observed oscillating temperatures between 25 °C and 37 ˚C with a 12‐hour cycle (starting at 25 ˚C, increasing to 37 ˚C in 12 hours, then back to 25 ˚C in 12 hours) which were replicated in a laboratory in Geneva (Switzerland) for a first series of vials. For a second series of samples, insulin was taken out daily to simulate a typical daily treatment with doses of 10 IU in the morning and 12 IU in the evening. In a third series, the effect of continuous (isothermal) exposure to high temperatures of 31 °C and 37 °C was investigated to compare results with data available in the literature. The two insulin formulations considered as essential medicines by the WHO (i.e. short‐acting insulin and intermediate‐acting NPH insulin) were exposed to a continuous temperature of 37 °C. Also, a series of mixed insulin vials were stored at ambient temperature in Kenya for four weeks of use by people with diabetes and brought back to Geneva for analysis. Insulin stored at 2 °C to 8 °C was used as positive control and heat‐degraded insulin as negative control analysing degradation after heating to 80 °C and agitation for 30 minutes. Study authors investigated three parameters: insulin potency by HPLC, conservation of the tridimensional conformation of insulin by circular dichroism and residual bioactivity by measuring insulin efficiency on insulin receptors. Visual inspection was performed according to European Pharmacopeia prescriptions. Concentrations of the preservatives phenol and metacresol indicating proper antimicrobial activity in the formulations were measured. Microbial contamination was investigated by taking transmission electron microscopy (TEM) images of the samples. Insulins studied were short‐acting insulin, intermediate‐acting NPH insulin and premixed short‐acting/NPH insulin (30% short‐acting insulin/70% NPH insulin) in vials.
Oscillating temperatures (25 °C to 37 °C): after 4 and 12 weeks, short‐acting insulin potency as compared to time zero (baseline) was 100.1% (SD 0.5) and 101.3% (SD 2.3), respectively. Results for NPH insulin were 101.3% (SD 1.3) and 101.6% (SD 8.9), respectively. Results for premixed short‐acting/NPH insulin were 100.6% (SD 1.6) and 101.9% (SD 3.5), respectively. Vials of mixed insulin returned from Kenya after four weeks of storage at ambient temperature at the individual's home were above 90% compared to the same mixed insulin exposed to four weeks of temperature cycling in the laboratory. None of the examined formulations showed visual changes in terms of yellowish colour or larger aggregates. After 12 weeks, phenol and metacresol concentrations were 92.4% to 94% and 92.7% to 97.7% of claimed values, respectively. There was no microbial contamination detected. Tridimensional conformation of insulin monomers was identical in all investigated samples. Mixed insulin under storage conditions oscillating between 25 °C and 37 °C for 12 weeks maintained bioactivity similar to insulin stored at 4 °C. Mixed insulin stored for four weeks out of refrigeration in a person's home in Kenya and mixed insulin at 25 °C to 37 °C for 12 weeks in the laboratory maintained bioactivity similar to insulin stored at 4 °C. The presence of air in the vials and multiple septa punctures had no impact on stability, and there was no difference in stability between insulin conditioned in pen cartridges and 10 mL glass vials.
Isothermal temperatures (31 °C and 37 °C): after 1, 4 and 8 weeks at continuous 31 °C results for NPH insulin activity as compared to time zero were 100.2% (SD 6.3), 94.8% (SD 5.4) and 82.8% (SD 4.0), respectively. After one week at continuous 37 °C, premixed short‐acting/NPH insulin activity showed a 15.4% decrease, and after two weeks, short‐acting insulin activity showed a 12.5% decrease.
-
In an additional stability study on premixed short‐acting/NPH insulin in vials (30% short‐acting insulin/70% NPH insulin) study authors investigated the following parameters (personal communication).
Temperature cycles between 17 °C and 45 °C: insulin formulations were stable.
Proportion between 'night' and 'day' by changes in temperature cycles 25 °C to 37 °C with 14 hours of exposure to higher temperatures, defined as 'day' period: insulin formulations were stable over four weeks. Insulin concentrations were all in the range of 100% ± 10% of the value at time zero.
Residual bioactivity of insulin and molecular conformation: an excellent correlation was found between potency determination by HPLC, tridimensional structure verification and residual bioactivity in living cell lines.
Silva‐Jr 2022: vials containing short‐acting and intermediate‐acting human insulin were stored at 4 °C and 37 °C for five weeks with weekly sampling to study the formation of subvisible particles. Measurements included TEM to analyse the presence of agglomerates, thioflavin T (ThT) fluorescence to measure the formation of amyloid material and electrospray ionisation ion‐mobility spectrometry mass spectrometry (ESI‐IMS‐MS) to evaluate the formation of aggregates due to chemical degradation. Additionally, dynamic light scattering (DLS) monitored the oligomeric and particulate distribution of insulin, and size‐exclusion chromatography (SEC) was performed for additional evaluation of the products. No amyloid particles were detected after four weeks of storage. However, large amorphous agglomerates were observed at 35 days after first handling, both at 4 °C and 37 °C. The chemical stability of the investigated short‐acting human insulin was not affected. Overall, data suggested the formation of subvisible and submicrometre amorphous particulate matter in insulin shortly upon use.
Pendsey 2023: vials containing human short‐acting, intermediate‐acting and mixed 30/70 insulin were stored in water‐tight bags, in open plastic containers, placed on a cupboard or high shelf, or in water‐tight bags in clay pots with a separate water compartment and all in shaded areas. Six families with a young person with T1DM attending the Diabetes Research Education and Management (DREAM) Trust in Nagpur, India participated in the study. Control vials of each insulin were also kept refrigerated at the DREAM Trust. All samples of each type of insulin came from one batch. Insulins were stored by families for one, two and four months, and then retrieved and refrigerated at the DREAM Trust. Samples were cold‐chain couriered to the University of Florida, USA and University of Gothenburg, Sweden and kept refrigerated until analysis. All insulin vials remained unused until analysis. At the University of Florida, analyses were conducted by HPLC. The University of Gothenburg used 1H NMR. For all analyses, an insulin concentration of 100% ± 5% was acceptable according to the USP standard. In the analysis at the University of Florida, insulin activity at 26.3 °C to 28.9 °C and up to four months compared to baseline for short‐acting (Figure 2), intermediate‐acting (Figure 3), and mixed insulin (Figure 4) ranged between −0.7% and −2.4%, −0.7% and +2.1%, and −1.8% and +0.8%, respectively. In the analysis performed at the University of Gothenburg, there was a subtle line width increase and slight peak shifts in nuclear magnetic resonance spectra, which correlated with storage time for non‐refrigerated samples. According to the study authors, this was probably due to altered conformation or multimerisation in a minor fraction of the insulin molecules. There was no loss of relative concentration using nuclear magnetic resonance. There was no decrease in total concentration in any of the insulin types observed as being larger than the precision of measurements (less than 1%), regardless of storage time or type of storage. The decline in mean potency for those samples stored in clay pots across all insulins at month four (0.5%) was lower than that for those stored in open boxes (3.6%).
4. Mean blood sugar levels in rabbits after intraperitoneal administration of regular human insulin stored at different temperatures for 24 days.
| Novo Nordisk human insulin (mean mg/dL (SEM)) | |||||
| 5 °C | 25 °C | 26 °C | 32 °C | 37 °C | |
| 0 min | 104.6 (1.5) | 105.3 (1.3) | 105.1 (1.1) | 107.6 (1.2) | 105.8 (1.4) |
| 15 min | 77.6 (1.1) | 77.5 (1.0) | 77.6 (1.6) | 79.6 (1.8) | 80.8 (1.8) |
| 30 min | 54.6 (1.2) | 54.6 (1.3) | 54.8 (1.1) | 56.8 (1.5) | 59.3 (1.1) |
| 60 min | 53.5 (1.2) | 53.5 (1.2) | 53.6 (1.4) | 58.5 (0.8) | 60.8 (1.3) |
| Eli Lilly and Company human insulin (mean mg/dL (SEM)) | |||||
| 0 min | 102.6 (1.5) | 102.6 (2.3) | 103.3 (1.5) | 102.5 (1.9) | 102.1 (1.9) |
| 15 min | 78.6 (1.1) | 78.5 (1.2) | 78.6 (1.8) | 79.3 (1.1) | 80.3 (1.1) |
| 30 min | 54.6 (1.6) | 54.8 (1.6) | 54.8 (1.5) | 58.1 (1.4) | 59.8 (1.3) |
| 60 min | 55.1 (1.5) | 55.1 (1.3) | 55.0 (0.9) | 60.3 (1.2) | 62.8 (1.2) |
| BIOCON human insulin (mean mg/dL (SEM)) | |||||
| 0 min | 102.8 (1.0) | 103.3 (1.0) | 102.6 (1.9) | 104.1 (1.3) | 102.1 (1.4) |
| 15 min | 78.6 (1.2) | 78.8 (1.0) | 78.8 (1.3) | 80.5 (1.6) | 81.3 (1.9) |
| 30 min | 55.5 (1.5) | 55.5 (1.6) | 55.6 (1.2) | 57.8 (1.1) | 59.5 (1.1) |
| 60 min | 54.0 (1.4) | 54.0 (1.2) | 54.6 (1.4) | 60.3 (1.1) | 63.0 (1.1) |
Data from table in Vimalavathini 2009.
min: minutes; SEM: standard error of the mean.
2.

Short‐acting human insulin assay (mean IU/mL with standard deviation) stored at 26.3 °C to 28.9 °C between baseline (month 0) and up to 4 months (data calculated from supplementary appendix Pendsey 2023, insulin reference value 95 IU/mL to 105 IU/mL).
IU: international units.
3.

Intermediate‐acting human insulin assay (mean IU/mL with standard deviation) stored at 26.3 °C to 28.9 °C between baseline (month 0) and up to 4 months (data calculated from supplementary appendix Pendsey 2023, insulin reference value 95 IU/mL to 105 IU/mL).
IU: international units.
4.
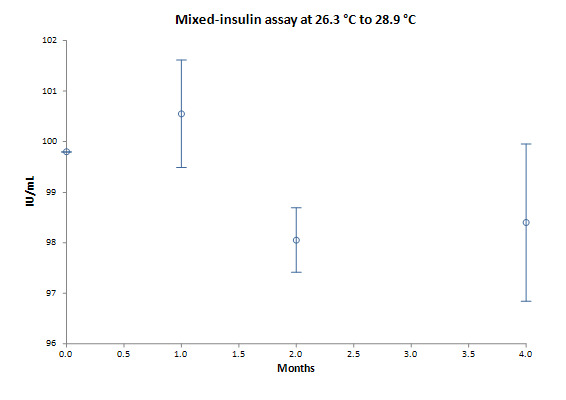
Mixed human insulin assay (mean IU/mL with standard deviation) stored at 26.3 °C to 28.9 °C between baseline (month 0) and up to 4 months (data calculated from supplementary appendix Pendsey 2023, insulin reference value 95 IU/mL to 105 IU/mL).
IU: international units.
Studies on cartridges/pens
Three studies investigated storage conditions for insulin cartridges/pens.
Shnek 1998: short‐acting, intermediate acting and mixed human insulin cartridges were investigated to evaluate the combined effect of temperature and agitation. Study authors performed temperature cycling (25 °C to 37 °C) tests in an incubator unit combined with resuspensions conducted twice daily on a mechanical device outside the incubator unit. The incubator unit was calibrated within the temperature range 25 °C to 37 °C. The temperature changed from 25 °C to 37 °C over two hours, with 10‐hour periods at each temperature. The temperature was decreased from 37 °C to 25 °C over two hours and the cycle was repeated for 28 days. Cartridges were visually assessed by trained operators after resuspension on test days 0, 7, 14, 21 and 28. Cartridges were checked for visual changes compared to quiescent refrigerated control samples. For solutions, visual changes included changes in colour and clarity from a clear solution. For suspensions, visual changes included the presence of large aggregates (clumps), material adhering to the cartridge wall (frosting), or both. RP‐HPLC was used to determine total insulin activity. Additionally, study authors performed continuous high‐temperature (37 °C) exposure tests in an incubator unit combined with periodic daily agitation by rotation devices while still inside the incubator unit. The incubator unit was calibrated to 37 °C. The cartridges were rotated at 30 rotations per minute (rpm) for four hours by custom‐made rotators. Additional agitation was imparted on the test cartridges through resuspension outside the incubator unit. The resuspension machine was used after the cartridges were removed from the incubator unit and prior to visual assessment. The resuspension machine performed three sets of 10 rolls plus 10 inversions. Cartridges were visually assessed after resuspension by trained operators on test days 0, 2, 5, 7, 9, 11, 14, 17, 21, 24 and 28. Also, microscopy, particle size determination and electrophoretic mobility measurements were carried out. For short‐acting soluble insulin, all stress testing studies showed no change in the product for all conditions tested. For intermediate‐acting and mixed human insulin suspension, HPLC assays were within specification after 28 days of temperature cycling and agitation. Continuous high temperature testing of both suspension products resulted in an HPLC potency measurement for a visually unchanged cartridge that was within specifications after one week of testing. However, after continuous high temperature over 28 days, there were visual changes (agglomerate formations) observed.
Kongmalai 2021: study authors investigated the effect of different temperatures in a tropical country on the stability of intermediate‐acting insulins after opening. Opened insulin pens were stored at room temperature, in a refrigerator (2 °C to 8 °C) or in an incubator (37 °C) for 28 days. Room temperature and humidity were recorded every hour throughout the study period. In the experimental group, all insulin pens were opened and flipped back and forth 20 times. Two units of insulin were discarded every day to simulate the daily usage by insulin‐treated people with diabetes. In the control group, an unopened insulin pen from each batch was stored in a refrigerator. All in‐use insulin pens were kept away from direct exposure to sunlight or artificial light. Insulin was analysed using ultra HPLC and the quantity of insulin was reported as mean and SD of AUC. During the study period, the mean room temperature was 29.7 °C (SD 2.8) (range 25.5 °C to 37.1 °C). The mean daytime temperature (6 a.m. to 7 p.m.) was 31.1 °C (SD 2.9) (range 27.1 °C to 37.1 °C) and the mean night‐time temperature (7 p.m. to 6 a.m.) was 28 °C (SD 1.3) (range 26 °C to 31 °C). The mean humidity in the room was 74.6% (SD 9) (range 50.0% to 89.5%). The mean humidity was 50% in the refrigerator and 38% in the incubator. The percent change in insulin quantity from baseline AUC after 28 days ranged from −2.6% to +1.4% and −1.5% to +0.7% at room temperature and incubator condition, respectively.
Delbeck 2021a: insulin specimens were studied by means of infrared (IR) attenuated total reflection (ATR) spectroscopy for comparison of protein structural changes. Insulin cartridges contained intermediate‐acting human NPH insulin with a biological activity of 100 insulin units (IU)/3 mL. Samples were stored in climatic exposure cabinets at 0 °C, 20 °C and 37 °C for different durations (up to 10 weeks). Significant changes within the insulin samples at various storage temperatures due to misfolding and subsequent fibrillation allowed the assumption of a decrease in bioactivity. Especially under storage conditions at high temperature (37 °C), there was an increased quantity of β‐sheet fractions in ultrafiltrated insulin samples (non‐stabilised hormone purified by ultrafiltration, i.e. pure insulin without excipients) detected by band deconvolution, underlining protein structural misfolding. Compared to the ultrafiltrated insulin samples, there were only small shifts detected for the other formulations, suggesting that the added stabilisers were protecting the insulin effectively from accelerated misfolding.
Studies on prefilled syringes
Four studies investigated storage conditions for prefilled plastic syringes.
Dunbar 1986: insulin syringes of three manufacturers were filled with short‐acting insulin and stored at 4 °C for two weeks. After 1, 3, 7 and 14 days, the syringes were removed, and aliquots were taken for radioimmunoassay and radioreceptor determinations. Samples of refrigerated stock insulin served as controls. The insulin content of samples stored in plastic syringes for up to two weeks was similar to that of the stock controls and comparable in their degradation. Regarding biological potency of insulin, receptor‐binding isotherms were the same for the different types of insulin as well as for the type of storage.
Tarr 1991: the stability and sterility of short‐acting, intermediate‐acting, mixed and extemporaneously prepared combination human insulin was evaluated after storage in 1 mL polypropylene syringes. For each type of insulin, 0.4 mL samples were withdrawn from multiple‐dose vials into polypropylene syringes and propylene‐ethylene copolymer syringes. Half of the syringes were placed in a plastic bag and kept at room temperature (22 °C to 23 °C), while the other half were placed in a plastic bag and kept in the refrigerator (3 °C to 5 °C). A vial of each type of insulin was maintained under the same conditions as the syringes. On days 1, 3, 7, 14, 21 and 28, three syringes stored at each temperature were removed, and the contents were analysed by HPLC for insulin potency, and metacresol and phenol concentrations. Study authors compared insulin potency data with the potency of insulin in the syringes on day 0. The concentrations of the preservatives on each test day were compared with the initial concentrations in the vials on day 0. The human insulins stored at 4 °C and 23 °C did not show a significant loss of activity in either type of syringe during the 28‐day test period (Table 8). The metacresol concentration decreased in all human insulins tested at room temperature during the 28‐day study. The percent change from baseline insulin concentration after 28 days at 4 °C for short‐acting insulin, intermediate‐acting insulin and mixed insulin ranged from −1.5% to +0.6%, +1.0% to +2.5% and −0.2% to 2.4%, respectively. The percent change from baseline concentration after 28 days at 23 °C for short‐acting insulin, intermediate‐acting insulin and mixed insulin ranged from −1.9% to +1.7%, +0.7% to +4.7% and −1.0% to −1.7%, respectively. The samples stored in polypropylene syringes tended to show a greater decrease in metacresol concentration than those stored in propylene‐ethylene copolymer syringes. Phenol concentrations were less affected than metacresol concentrations in both types of plastic syringes. Phenol concentrations decreased in the samples stored in plastic syringes at room temperature. None of the human insulin products that were drawn into the two different plastic syringes showed any microbial growth.
Koffler 1992: syringes were filled with 10 U of short‐acting insulin (later withdrawn for commercial reasons by the manufacturer) and intermediate‐acting insulin. The prefilled insulin syringes were kept in a refrigerator at 4 °C for up to 10 days. After 10 days, insulin samples from each syringe were cultured concurrently in brain–heart infusion broth and in thioglycollate brain infusion broth. The samples were incubated at a temperature of 37 °C under aerobic and anaerobic conditions for up to seven days, with a daily check for growth. There were no signs of bacterial growth evident in any of the specimens.
Lull 2013: the stability of short‐acting U‐500 insulin was evaluated after storage in polypropylene insulin syringes. Each syringe was filled with U‐500 insulin 0.25 mL (125 U). Samples were analysed immediately to establish day 0 stability values. The remaining syringes were kept capped inside an open tray in a laboratory refrigerator (3 °C to 5 °C). At days 7, 14, 21 and 28, syringes were removed for analysis by RP‐HPLC. In addition, a sample was drawn for a forced degradation sample. Study authors calculated the mean AUC for insulin peak. The percentage of the initial concentration remaining at days 7, 14, 21 and 28 was 97.8% (SD 0.6), 99.4% (SD 3.9), 99.9% (SD 2.4) and 93.3% (SD 2.4), respectively.
5. Stability of human insulin formulations in plastic syringes.
| Temperature | Initial concentration (units/mL) (mean ± SD) | Stability of human insulin in plastic syringes: % initial concentration remaining (mean ± SD) | |||||
| Day 1 | Day 3 | Day 7 | Day 14 | Day 21 | Day 28 | ||
| Short‐acting human insulin (Humulin R) | |||||||
| T (4 °C) | 100.0 ± 4.3 | 101.3 ± 0.8 | 100.3 ± 1.5 | 102.1 ± 2.8 | 100.8 ± 3.3 | 101.6 ± 1.7 | 98.5 ± 3.4 |
| P (4 °C) | 100.0 ± 0.8 | 102.3 ± 4.0 | 104.6 ± 4.3 | 101.0 ± 2.0 | 99.2 ± 0.8 | 101.9 ± 1.2 | 100.6 ± 1.1 |
| T (23 °C) | 100.0 ± 4.3 | 101.4 ± 2.1 | 103.0 ± 1.5 | 100.1 ± 1.2 | 97.4 ± 1.9 | 101.6 ± 1.7 | 101.7 ± 2.8 |
| P (23 °C) | 100.0 ± 0.8 | 99.8 ± 0.4 | 102.1 ± 3.8 | 100.0 ± 2.9 | 100.2 ± 3.0 | 100.0 ± 0.4 | 98.1 ± 5.2 |
| Intermediate‐acting isophane human insulin (Humulin N) | |||||||
| T (4 °C) | 100.0 ± 1.1 | 100.1 ± 1.4 | 102.8 ± 2.6 | 101.4 ± 2.4 | 99.4 ± 3.0 | 101.1 ± 2.0 | 101.0 ± 2.3 |
| P (4 °C) | 100.0 ± 1.7 | 100.6 ± 1.4 | 104.4 ± 2.5 | 105.0 ± 0.4 | 99.8 ± 1.9 | 101.4 ± 4.0 | 102.5 ± 3.6 |
| T (23 °C) | 100.0 ± 1.1 | 105.0 ± 3.8 | 102.3 ± 3.9 | 99.2 ± 4.0 | 102.3 ± 2.3 | 98.9 ± 3.2 | 100.7 ± 2.3 |
| P (23 °C) | 100.0 ± 1.7 | 103.4 ± 2.5 | 102.6 ± 1.3 | 102.6 ± 2.4 | 100.6 ± 2.3 | 98.2 ± 2.1 | 104.7 ± 3.6 |
| Mixed human insulin: short‐acting human insulin + intermediate‐acting human insulin (Humulin N/R 70/30) | |||||||
| T (4 °C) | 100.0 ± 3.0 | 97.7 ± 5.2 | 100.6 ± 2.8 | 100.0 ± 5.2 | 100.9 ± 7.5 | 97.1 ± 2.7 | 99.8 ± 3.1 |
| P (4 °C) | 100.0 ± 4.1 | 98.6 ± 4.0 | 102.0 ± 5 0 | 100.7 ± 3 8 | 104.8 ± 6.3 | 100.0 ± 5.8 | 102.4 ± 2.5 |
| T (23 °C) | 100.0 ± 3.0 | 102.2 ± 3.8 | 103.7 ± 3.6 | 98.3 ± 4.8 | 98 9 ± 2.8 | 98.7 ± 4.9 | 99.0 ± 1.8 |
| P (23 °C) | 100.0 ± 4.1 | 99.9 ± 2.5 | 102.6 ± 2.4 | 101.4 ± 1.1 | 101.4 ± 1.6 | 98.3 ± 3.4 | 98.3 ± 2.9 |
| Extemporaneous mixed insulin: intermediate‐acting human insulin + short‐acting human insulin (Humulin N/R 70/30) | |||||||
| T (4 °C) | 100.0 ± 4.0 | 99.5 ± 2.9 | 102.8 ± 3.8 | 101.6 ± 3.9 | 102.4 ± 5.4 | 98.6 ± 2.4 | 102.6 ± 4.4 |
| P (4 °C) | 100.0 ± 2.8 | 100.2 ± 2.3 | 100.2 ± 1.9 | 97.4 ± 4.5 | 99.3 ± 6.6 | 96.2 ± 3.8 | 99.6 ± 3.2 |
| T (23 °C) | 100.0 ± 4.0 | 102.2 ± 5.2 | 101.0 ± 4.2 | 98.3 ± 7.2 | 103.7 ± 6.6 | 96.9 ± 2.5 | 100.7 ± 2.2 |
| P (23 °C) | 100.0 ± 2.8 | 100.7 ± 2.8 | 101.5 ± 4.8 | 100.0 ± 4.0 | 102 5 ± 5.6 | 100.5 ± 6.0 | 102.8 ± 3.0 |
Data from table 1 in Tarr 1991
P: Plastipak (polypropylene); SD: standard deviation; T: Terumo (propylene‐ethylene copolymer).
Data from manufacturers
Most data below refer to unopened containers (vials, cartridges) being punctured only once for analysis, which is standard procedure throughout the pharmaceutical industry.
Four manufacturers (BIOTON (biosimilar manufacturer), Eli Lilly and Company, Novo Nordisk and Sanofi) provided data on thermostability of human insulin. We did not obtain data from Gan and Lee Pharmaceuticals (biosimilar manufacturer). For an overview of main results, see Table 3.
As Sanofi permanently discontinued the production of its human insulins Insuman rapid 100 IU/mL, Insuman rapid SoloStar 100 IU/mL, Insuman basal 100 IU/mL, Insuman basal SoloStar 100 IU/mL, Insuman comb 25 100 IU/mL and Insuman comb 25 SoloStar 100 IU/mL worldwide, data provided by the manufacturer could not be included in this review (Sanofi 2023a; Sanofi 2023b).
BIOTON provided data on short‐acting human insulin 40 IU/mL vials after 28 and 42 days at 30 °C: insulin activity, A21 desamido insulin, impurities, total zinc, metacresol and pH all remained within reference values. BIOTON also provided data on short‐acting human insulin 100 IU/mL vials after one, three and six months at 25 °C: insulin activity, A21 desamido insulin, impurities, total zinc, metacresol and pH all remained within reference values. Loss of insulin activity, calculated as percentage change compared to baseline after one, three and six months was 1.1%, 1.0% and 1.7%, respectively.
Eli Lilly and Company provided summary data: at below 25 °C short‐acting, intermediate‐acting and mixed insulin could be stored for up to 25 days before start use and then 28 days for patient in‐use. At below 30 °C, short‐acting, intermediate‐acting and mixed insulin could be stored for up to 12 days before start use and then 28 days for patient in‐use. All product quality attributes met established acceptance criteria.
The results presented below are based on data from Novo Nordisk, which provided the most detailed information. Insulin concentrations were measured by HPLC.
We used the mean concentration from refrigerated samples at time zero (baseline) as a reference value to assess changes in the following parameters, if provided (depending on insulin formulation different reference values were applicable).
Insulin activity 100 IU/mL (activity, potency or concentration; reference value: 95 IU/mL to 105 IU/mL)
A21 desamido insulin (reference value: 1.5% or less), degradation product
A21 desamido insulin + N‐oxalyl desPheB1 (desPhe) insulin (reference value: 1.54% or less), degradation product
B3 desamido insulin (reference value: 6.54% or less), degradation product
High molecular weight proteins (HMWP) (reference value: 2% or less, 3% or less)
Other related proteins (ORP) (reference value: 3.0% or less, 4.5% or less)
Particulate matter (particles 10 µm/container or less, reference value: 6000 or less; particles 25 µm/container or less, reference value: 600 or less)
Macroscopic appearance
Metacresol (reference value: 1.35 mg/mL to 1.65 mg/mL, 2.7 mg/mL to 3.3 mg/mL), preservative/stabiliser
Phenol (reference value: 0.59 mg/mL to 0.72 mg/mL), preservative/stabiliser
pH (reference value: 6.9 to 7.5, 7.0 to 7.8)
Zinc (reference value: 10.0 µg/mL to 40.0 µg/mL, 40.0 µg/mL or less), preservative/stabiliser
Sterility, bacterial endotoxins
Results for short‐acting insulins
Storage at 25 °C
Insulin potency
Figure 5 and Figure 6 show insulin assays for vials and cartridges, respectively, stored at 25 °C between 1 and 12 months.
5.

Short‐acting human insulin assay for vials stored at 25 °C between baseline and up to 12 months (data from Novo Nordisk, insulin reference value 95 IU/mL to 105 IU/mL).
CI: confidence interval; IU: international units.
6.

Short‐acting human insulin assay for cartridges stored at 25 °C between baseline and up to 12 months (data from Novo Nordisk, insulin reference value 95 IU/mL to 105 IU/mL)
CI: confidence interval; IU: international units.
Loss of insulin activity (percentage change compared to baseline)
After one month, mean loss for vials was 0.4% (SD 0.3) and for cartridges 0.5% (SD 0.4).
After three months, mean loss for vials was 1.8% (SD 0.6) and for cartridges 1.8% (SD 0.6).
After six months, mean loss for vials was 3.5% (SD 0.4) and for cartridges 3.2% (SD 0.5).
After 12 months, mean loss for vials was 7.4% (SD 0.6) and for cartridges 6.5% (SD 0.5).
A21 desamido insulin, A21 desamido insulin plus desPhe, B3 desamido insulin
Mean values for A21 desamido insulin, A21 desamido insulin plus desPhe and B3 desamido insulin in vials or cartridges were in range for up to 12 months, except for B3 desamido insulin at month 12 (6.63%).
High molecular weight proteins, other related proteins
At time zero, mean value for HMWP was 0.1% for vials. After 1, 3, 6 and 12 months, HMWP increased to 0.3%, 0.6%, 1.1% and 2.2%, respectively.
At time zero, mean value for HMWP was 0.2% for cartridges. After 1, 3, 6 and 12 months, HMWP increased to 0.3%, 0.6%, 1.0% and 1.8%, respectively.
ORP were in range up to six months. After 12 months, the mean ORP increased to 7.2% (vials) and 6.2% (cartridges).
Particulate matter
Macroscopy and particles per container complied with specifications.
Bacterial endotoxins, sterility
Except for time zero, there were no data available.
Metacresol
Mean metacresol values remained within reference range.
Zinc
Mean zinc values remained within reference range.
pH
Mean pH values remained within reference range.
Storage at 37 °C
Insulin potency
Figure 7 and Figure 8 show insulin assays for vials and cartridges stored at 37 °C between one and three months. After one month, insulin content either remained within specifications for both vials and cartridges or showed a small decline below the lower limit, which was comparable to results after two months.
7.

Short‐acting human insulin assay for vials stored at 37 °C between baseline and up to 3 months (data from Novo Nordisk, insulin reference value 95 IU/mL to 105 IU/mL).
CI: confidence interval; IU: international units.
8.

Short‐acting human insulin assay for cartridges stored at 37 °C between baseline and up to 3 months (data from Novo Nordisk, insulin reference value 95 IU/mL to 105 IU/mL).
CI: confidence interval; IU: international units.
Loss of insulin activity (percentage change compared to baseline)
After one month, mean loss for vials was 2.2% (SD 0.6) and for cartridges 2.8% (SD 0.3).
After two months, mean loss for vials was 5.7% (SD 0.7) and for cartridges 5.7% (SD 1.5).
After three months, mean loss for vials was 8.6% (SD 0.6) and for cartridges 8.3% (SD 0.8).
A21 desamido insulin, A21 desamido insulin plus desPhe, B3 desamido insulin
Mean values for A21 desamido insulin, A21 desamido insulin plus desPhe and B3 desamido insulin in vials and cartridges were in range for up to three months.
High molecular weight proteins, other related proteins
At time zero, mean value for HMWP was 0.1% for vials. After one, two and three months, mean HMWP values increased to 1.0%, 2.0% and 3.0%, respectively. At time zero, mean value for HMWP was 0.2% for cartridges. After one, two and three months, mean HMWP values increased to 1.0%, 2.0% and 2.9%, respectively.
ORP were in range after one month. After two and three months, mean ORP in vials increased to 5.1% and 7.8%, respectively. After two and three months, mean ORP in cartridges increased to 5.5% and 7.9%, respectively.
Particulate matter
Macroscopy and particles per container complied with specifications.
Bacterial endotoxins, sterility
Except for baseline, there were no data available.
Metacresol
Mean metacresol values remained within reference range.
Zinc
Mean zinc values remained within reference range.
pH
Mean pH values remained within reference range.
Storage at 5 °C
For reasons of comparison, we only report insulin potency data at optimal storage conditions (refrigerator).
Insulin potency
Figure 9 and Figure 10 show insulin assays for vials and cartridges stored at 5 °C between 12 and 24 months. Up to 30 months, insulin content remained within specifications for both vials and cartridges.
9.

Short‐acting human insulin assay for vials stored at 5 °C between baseline and up to 24 months (data from Novo Nordisk, insulin reference value 95 IU/mL to 105 IU/mL).
CI: confidence interval; IU: international units.
10.

Short‐acting human insulin assay for cartridges stored at 5 °C between baseline and up to 24 months (data from Novo Nordisk, insulin reference value 95 IU/mL to 105 IU/mL).
CI: confidence interval; IU: international units.
If refrigerated (5 °C) there was a minimal loss of insulin, for example, after 24 months of storage, mean loss for vials was 0.7% (SD 0.4) and for cartridges 1.2% (SD 0.6).
Results for intermediate‐acting insulins
Storage at 25 °C
Insulin potency
Figure 11 and Figure 12 show insulin assays for vials and cartridges stored at 25 °C between 1 and 12 months.
11.

Intermediate‐acting human insulin assay for vials stored at 25 °C between baseline and up to 12 months (data from Novo Nordisk, insulin reference value 95 IU/mL to 105 IU/mL).
CI: confidence interval; IU: international units.
12.

Intermediate‐acting human insulin assay for cartridges stored at 25 °C between baseline and up to 12 months (one batch data from Novo Nordisk, insulin reference value 95 IU/mL to 105 IU/mL).
IU: international units.
Loss of insulin activity (percentage change compared to baseline)
After one month, mean loss for vials was 0.7% (SD 0.4) and for cartridges 0.5%.
After three months, mean loss for vials was 1.2% (SD 0.5) and for cartridges 1.9%.
After six months, mean loss for vials was 2.0% (SD 0.4) and for cartridges 2.3%.
After 12 months, mean loss for vials was 3.4% (SD 0.7) and for cartridges 3.9%.
High molecular weight proteins, other related proteins
At time zero, mean value for HMWP was 0.2% for vials. After 1, 3, 6 and 12 months, HMWP increased to 0.3%, 0.6%, 1.0% and 1.8%, respectively. At time zero, mean value for HMWP was 0.2% for cartridges. After 1, 3, 6 and 12 months, HMWP increased to 0.4%, 0.7%, 1.0% and 1.5%, respectively.
ORP were in range for all measurements, except after 12 months of storage for cartridges where ORP increased to 3.1%.
Particulate matter
There were no data available.
Bacterial endotoxins, sterility
There were no data available.
Metacresol
Mean metacresol values remained within reference range.
Phenol
Mean phenol values remained within reference range.
Zinc
Mean zinc values remained within reference range.
pH
Mean pH values remained within reference range.
Storage at 37 °C
Insulin potency
Figure 13 and Figure 14 show insulin assays for vials and cartridges stored at 37 °C between one and three months.
13.

Intermediate‐acting human insulin assay for vials stored at 37 °C between baseline and up to 3 months (data from Novo Nordisk, insulin reference value 95 IU/mL to 105 IU/mL).
CI: confidence interval; IU: international units.
14.

Intermediate‐acting human insulin assay for cartridges stored at 37 °C between baseline and up to 3 months (one batch data from Novo Nordisk, insulin reference value 95 IU/mL to 105 IU/mL).
IU: international units.
Loss of insulin activity (percentage change compared to baseline)
After one month, mean loss for vials was 1.8% (SD 0.3) and for cartridges 1.4%.
After two months, mean loss for vials was 3.8% (SD 0.4) and for cartridges 3.0%.
After three months, mean loss for vials was 5.3% (SD 1.0) and for cartridges 4.7%.
High molecular weight proteins, other related proteins
At time zero, mean value for HMWP was 0.2% for vials. After one, two and three months, mean HMWP values increased 0.9%, 1.9% and 2.5%, respectively. At time zero, mean value for HMWP was 0.2% for cartridges. After one, two and three months, mean HMWP values increased to 0.8%, 1.5% and 2.1%, respectively.
ORP were in range after one and two months. After three months, ORP increased to 4.0% (vials) and 3.3% (cartridges).
Particulate matter
There were no data available.
Bacterial endotoxins, sterility
There were no data available.
Metacresol
Mean metacresol values remained within reference range.
Phenol
Mean phenol values remained within reference range.
Zinc
Mean zinc values remained within reference range.
pH
Mean pH values remained within reference range.
Storage at 5 °C
For reasons of comparison, we only reported insulin potency data at optimal storage conditions (refrigerator).
Insulin potency
Figure 15 and Figure 16 show insulin assays for vials and cartridges stored at 5 °C between 12 and 24 months. Up to 24 months, insulin content remained within specifications for both vials and cartridges.
15.

Intermediate‐acting human insulin assay for vials stored at 5 °C between baseline and up to 24 months (data from Novo Nordisk, insulin reference value 95 IU/mL to 105 IU/mL).
CI: confidence interval; IU: international units.
16.

Intermediate‐acting human insulin assay for cartridges stored at 5 °C between baseline and up to 24 months (data from Novo Nordisk, insulin reference value 95 IU/mL to 105 IU/mL).
CI: confidence interval; IU: international units.
If refrigerated (5 °C), there was a minimal loss of insulin, for example, after 24 months of storage, mean loss for vials was 0.6% (SD 0.6) and for cartridges 0.1% (0.6).
Discussion
Summary of main results
We utilised two information sources, that is, study publications and previously unpublished data from pharmaceutical manufacturers.
One pilot clinical study reporting on plasma glucose after intravenous insulin injection showed no difference between clay pot‐stored insulin and insulin stored in a refrigerator. Apart from two studies reporting a more pronounced loss in insulin activity, the majority of studies on vials and cartridges/pens using different temperature and storage conditions showed no clinically relevant decreases in insulin concentrations. Also, oscillating temperatures between 25 °C and 37 °C for up to three months resulted in no loss of insulin activity. Multiple punctures of vials and cartridges imitating in‐use periods by people with diabetes did not result in clinically relevant decreases in insulin activity. Investigations on prefilled syringes showed no decrease in insulin potency or signs of microbial growth. Thermostability studies from pharmaceutical companies manufacturing human insulin investigating storage conditions at up to 25 °C for a maximum of six months and at up to 37 °C for a maximum of two months did not show a clinically relevant change in insulin activity.
Overall completeness and applicability of evidence
We identified one pilot clinical study (Al Shaibi 1999). Only a few studies evaluated mixed human insulin (Kaufmann 2021; Pendsey 2023; Shnek 1998; Tarr 1991; Vimalavathini 2009). We found no data on continuous subcutaneous insulin infusion (CSII) pumps, which would be important due to the impact of elevated temperature exposure and mechanical stress on the integrity of human insulin, as CSII pumps are worn close to the body.
Unfortunately, data on human insulin storage conditions for people living in cold climates are scarce (Khurana 2019). People living in cold climates have to find innovative ways to protect insulin: during the winter in the Himalayan areas, some people wear warm clothes made up of sheep and yolk wool to protect them from severe cold. One such cloth is an abdominal binder which may be used to wrap insulin vials and pens in multiple layers in the abdominal binder where temperatures range between 5 °C and 10 °C (Khurana 2019).
There are concerns that the measured and actual biological potency of insulin might differ due to misfolding of the insulin molecules and subsequent fibrillation if subjected to higher temperatures (Baechler 2020; Delbeck 2020a; Delbeck 2020b). Delbeck 2021a reported findings in ultrafiltrated insulin samples (i.e. non‐stabilised pure insulin without excipients) of protein structural misfolding, possibly reducing insulin potency. However, added stabilisers protected intermediate‐acting human insulin effectively from accelerated misfolding. There are also in‐vitro study reports showing that metacresol had an inhibitory effect on amyloid fibrillation, which is thought to be immunogenic, potentially reducing insulin efficacy and causing allergic reactions (Ohno 2019). Another report indicated excellent correlation between potency determination by HPLC, tridimensional insulin structure verification and residual bioactivity in living cell lines (Kaufmann 2021). Finally, one study investigating the formation of subvisible particles in short‐acting human insulin showed that amorphous aggregates developed within weeks of handling, regardless of storage temperature at 4 °C or 37 °C (Silva‐Jr 2022).
Even without access to reliable refrigeration, there are simple ways to prevent insulin from overheating. Several studies demonstrated the efficacy of various cooling devices, such as clay pots filled with water or wet sand and the placement of insulin inside (Al Shaibi 1999; Gill 2002; Gilligan 2021; Khurana 2019; Ogle 2016; Pendsey 2006; Taerahkun 2022).
More studies are needed on opened or in‐use vials and cartridges to investigate potential microbial ingress risk after multiple punctures of the container.
Certainty of the evidence
Extensive data from manufacturers of human insulin appeared reliable. Due to the objective measurements of insulin activity and other factors in the manufacturing process, we have confidence in the results provided. These investigations also appeared to be part of the approval process for the respective insulins, with drug regulatory authorities' overview of quality assurance. However, manufacturers' data on various storage conditions took place under optimal laboratory conditions (i.e. incubators assured continuous temperature conditions).
Agreements and disagreements with other reviews
We found no other reviews on this topic, apart from one narrative review addressing general aspects of insulin storage during the supply chain, highlighting the need for practical storage recommendations especially in homes of people with diabetes (Heinemann 2021).
Contrary to other studies and data from manufacturers of human insulin, two studies described a pronounced loss in insulin activity at continuous temperatures of 37 °C after one or two weeks (Kaufmann 2021) and after 28 days (Vimalavathini 2009). However, insulin in vials stored in a mud pot that reduced outside temperatures to 26 °C retained insulin potency (Vimalavathini 2009).
Strengths and weaknesses of the review
Strengths of our review include our extensive search of the literature for relevant studies, with no language or date restrictions, and our detailed scrutiny of all references listed in all included studies and (systematic) reviews. We also established an email alert service for MEDLINE via OvidSP that was run on a weekly basis to identify newly published studies. In addition, we contacted pharmaceutical manufacturers of human insulin and received previously unpublished thermostability data.
A potential weakness of our review is its body of evidence (i.e. mainly laboratory studies). For laboratory studies investigating loss of insulin potency at varying temperatures over time, there are no published validated methods to investigate risk of bias. However, shared data from pharmaceutical manufacturers of human insulin probably have a low risk of bias due to the objective measurements which were used in the thermostability investigations. Moreover, these analyses apparently formed part of the drug approval process under scrutiny from drug regulatory authorities in order to establish safety margins of insulin storage.
Due to distinct variation in methods, laboratory measurements and storage conditions reported in the included studies, it was not possible to establish a straightforward evidence synthesis of publication results. Instead, we tried to provide a transparent overview of data and key results from study publications.
Authors' conclusions
Implications for practice
An increasing portion of the global diabetes community resides in settings with poor or no access to healthcare facilities and refrigeration. In these circumstances, pharmaceutical companies' data indicate that it is possible to store unopened short‐acting and intermediate‐acting human insulin vials or cartridges at up to 25 °C for a maximum of six months and at up to 37 °C for a maximum of two months without a clinically relevant loss of insulin potency. Also, oscillating temperatures between 25 °C and 37 °C, resembling daytime and night‐time fluctuations in tropical countries, for up to three months do not result in a clinically relevant loss of insulin activity of short‐acting, intermediate‐acting and mixed human insulin. With the help of simple cooling devices for insulin storage such as clay pots, it is possible to effectively reduce high outside temperatures in many high‐temperature regions of the world.
Implications for research
Since there are few clinical studies on the thermostability of human insulin to date, clamp studies and other clinical studies allowing the investigation of insulin pharmacodynamics should be performed to measure insulin potency and safety after varying storage conditions in human volunteers and people with diabetes. More studies are needed on mixed insulin, as well as on opened vials and cartridges to investigate potential microbial ingress risk after multiple punctures of the container. Finally, studies on cold environmental conditions and insulin pumps are required.
On 22 April 2022, Novo Nordisk announced that the European Medicines Agency (EMA) has issued the company a positive scientific opinion for a proposed update of the storage conditions of two human insulins: "EMA's positive scientific opinion will now be used to support approval of the more flexible storage conditions by national health authorities in a number of relevant low‐ and middle‐income countries. This is to allow Actrapid (short‐acting insulin), and Insulatard (intermediate‐acting insulin) can be stored for four weeks outside a refrigerator (if kept below 30 °C) prior to use if there are six months or more to the expiry date. Once in use, the insulin should be stored without refrigeration." Other manufacturers of human insulin should also contact drug approval authorities and make thermostability data available to establish better real‐life updates on storage recommendations of human insulin.
History
Protocol first published: Issue 1, 2022
Acknowledgements
We thank the pharmaceutical companies BIOTON, Eli Lilly and Company, Novo Nordisk and Sanofi for providing previously unpublished data.
We thank Dr Yuan Chi for translating the Chinese publication Zhao 2015.
We thank study authors Baechler 2020, Delbeck 2021a, Kaufmann 2021, Kongmalai 2021, and Silva 2013 for replying to our survey of study investigators, and study authors Kaufmann 2021, Kongmalai 2021, and Silva 2013 for providing additional data.
Editorial and peer‐reviewer contributions
Cochrane Metabolic and Endocrine Disorders Group supported the authors in the development of this review.
The following people conducted the editorial process for this article.
Sign‐off Editor (final editorial decision): Mauricio Didac
Managing Editor (selected peer reviewers, collated peer‐reviewer comments, provided editorial guidance to authors, edited the article): Juan Victor Ariel Franco, Institute of General Practice, Medical Faculty of the Heinrich‐Heine‐University Düsseldorf, Düsseldorf, Germany
Methods Editor: Rachel Richardson, Methods Support Unit (Cochrane)
Copy Editor (copy‐editing and production): Anne Lawson, Cochrane Central Production Service
-
Peer‐reviewers:
Morten Hach, Scientific Director, Novo Nordisk A/S
Mats Rasmussen, Novo Nordisk
Dr Beatrice Kaufmann, Institute of Pharmaceutical Sciences of Western Switzerland (ISPSO), University of Geneva, Switzerland
Professor Stephen Colagiuri, Faculty of Medicine and Health and Charles Perkins Centre, University of Sydney
Appendices
Appendix 1. Additional information on insulin
Human insulin is a two‐chain (chain A with 21 amino acids, chain B with 30 amino acids) small hormone or polypeptide with a complex tridimensional shape generating so‐called secondary and tertiary structures of insulin. Insulin potency is expressed as units of biological activity such as US Pharmacopeia (USP) units, insulin or international units (IU), μg of activity/g or mg. By convention, one (USP) human insulin unit is equivalent to 0.0347 mg of pure human insulin. The potency of insulin in units (U) per millilitre (U/mL) describes the quantified blood glucose lowering of insulin per volume. Insulin preparations are standardised and are listed as concentrations, e.g. 40, 100 or 500 insulin units/mL of solution or suspension, denoted as U‐40, U‐100 and U‐500, respectively. It is important to note that syringes have to correspond to insulin concentrations: e.g. one cannot use U‐100 syringes when using a U‐500 insulin product. The injection of 50 units U‐100 means a volume of 0.5 mL, and injection of 50 units U‐500 means a volume of 0.1 mL.
NPH (neutral protamine Hagedorn) insulin or isophane insulin is an intermediate‐acting insulin. Mixtures of human insulin contain both short‐acting (regular) and intermediate‐acting insulin, defining a ratio of the two components. In Europe, regular insulin is listed first, e.g. a 30/70 mix would indicate 30% short‐acting insulin and 70% intermediate‐acting insulin. In the US, the ratio is defined as the intermediate‐acting component to the short‐acting component (e.g. a 75/25 mix would indicate 75% intermediate‐acting insulin and 25% short‐acting insulin). According to pharmacopoeias (a country's legally binding collection of standards and quality specifications for medicines used), the isotonic, sterile solution of human insulin in water for injection has a potency of 95.0% to 105.0% of the potency stated on the label, expressed in USP insulin human units/mL (USP 2016). Therefore, an insulin vial labelled '100 U/mL' has to contain 95 U/mL to 105 U/mL of insulin.
Human insulin, if manufactured by recombinant DNA (rDNA) technology in living cells, inevitably contains host cell DNA which needs to be cleared as much as possible. Also, microbial contamination must be minimised. With reference to bacterial endotoxins there should be no more than 80 USP endotoxin units per 100 USP human insulin units. Purity of human insulin is evaluated by (reversed‐phase) high‐performance liquid chromatography ((RP‐) HPLC). There are several degradation products of insulin, e.g. bioactive A‐21 desamido insulin and B‐3 desamido insulin. According to 2021 USP criteria, the upper limit of A‐21 desamido insulin is 2% and that of total impurities no more than 2% excluding A‐21 desamido insulin. During storage, high‐molecular weight proteins (HMWP) may form inducing dimers between two insulin molecules. At higher temperatures, higher order insulin oligomers develop. The biopotency of HMWP is significantly less than monomeric insulin. The upper limit for HMWP formation is 1%.
Adding zinc cations, phenol or its derivatives such as metacresol reinforces the hexameric form to increase insulin stability and ensure sterility. In insulin injections (solutions or suspensions) the total amount of zinc should be no more than 40.0 µg per 100 IU of human insulin. However, a transition from the hexamer to the monomer form in the body is necessary to exert the physiologically fast effect of insulin monomers. Therefore, it is difficult to achieve optimal insulin stability and efficacy at the same time.
Insulin aggregation may lead to a loss in insulin potency and pronounced aggregation may induce insulin fibrils. Fibrillar insulin consists of partially unfolded monomers but is relatively thermoresistant and does not precipitate even at 100 °C (Arora 2004). However, shear stress (shaking or stirring an insulin suspension) causes rapid fibrillogenesis (Huus 2005). The physical stability of human insulin is assessed by visual inspection for macroscopic changes, microscopy and various particle‐sizing techniques. Physical changes in soluble insulin are detected by changes in colour or clarity or formations of precipitates. Insulin suspensions are more susceptible to changes in physical stability and aggregates in suspensions may cause visible clumping.
With the help of in‐vitro cellular assays such as insulin receptor‐expressing hepatic HepG2 cells, it is nowadays possible to investigate the physiological activity or bioactivity of insulin (Calo 2016; Kaufmann 2021).
Appendix 2. Search strategies
PMIDs (PubMed‐IDs) of 24 relevant references used to develop the search strategies (Ovid syntax) (5039832 or 5668013 or 9811506 or 11044282 or 15111554 or 16101301 or 16969698 or 18463345 or 19217734 or 19797814 or 20687866 or 20920435 or 22538135 or 24246357 or 24876429 or 27472257 or 30815830 or 31009254 or 31994414 or 32281880 or 33259116 or 33534816 or 12941735 or 29114035).ui.
Cochrane Central Register of Controlled Trials (Cochrane Register of Studies Online)
1. MESH DESCRIPTOR Drug Stability
2. MESH DESCRIPTOR Drug Storage
3. MESH DESCRIPTOR Tropical Climate
4. MESH DESCRIPTOR Thermodynamics
5. #1 OR #2 OR #3 OR #4
6. MESH DESCRIPTOR Insulin
7. insulin*:TI,AB,KY
8. #6 OR #7
9. #5 AND #8
10. ((temperature* OR stability OR storage OR thermostab* OR heat*) ADJ12 insulin*):TI,AB,KY
11. #9 OR #10
MEDLINE (Ovid)
1. (Drug Stability/ or Drug Storage/ or Tropical Climate/ or Thermodynamics/) and insulin*.mp.
2. ((temperature* or stability or storage or thermostab* or heat*) adj12 insulin*).tw.
3. 1 or 2
CINAHL (EbscoHost)
1. MH "Drug Stability"
2. MH "Drug Storage"
3. S1 OR S2
4. MH "Insulin"
5. TI (insulin*) OR AB (insulin*)
6. S4 OR S5
7. S3 AND S6
8. TI ((temperature* OR stability OR storage OR thermostab* OR heat* OR thermodynamic* OR climate*) N12 insulin*) OR AB ((temperature* OR stability OR storage OR thermostab* OR heat* OR thermodynamic* OR climate*) N12 insulin*)
Science Citation Index Expanded (Web of Science)
AB=(("tropical climate" OR thermodynamic* OR temperature* OR stability OR storage OR thermostab* OR heat*) NEAR/12 insulin*) OR TI=(("tropical climate" OR thermodynamic* OR temperature* OR stability OR storage OR thermostab* OR heat*) NEAR/12 insulin*)
WHO Global Index Medicus
Title: (insulin* AND (tropic* OR thermodynamic* OR temperature* OR stability OR storage OR thermostab* OR heat* OR hot OR thermal OR environment* OR climate*)) WHO ICTRP Search Portal (Standard search)
insulin* AND (tropic* OR thermodynamic* OR temperature* OR stability* OR storage* OR thermostab* OR heat* OR climate* OR thermal*)
ClinicalTrials.gov (Advanced search)
Title/Acronym: insulin AND (tropical OR tropics OR thermodynamic OR thermodynamics OR temperature OR temperatures OR stability OR storage OR thermostability OR heat OR hot OR thermal OR environment OR environmental OR climate OR climates)
Appendix 3. Survey of study investigators providing information on included studies
| Study ID | Author(s) confirmed willingness to answer questions, if necessary | Author(s) provided additional data |
| Al Shaibi 1999 | No reply | — |
| Baechler 2020 | Yes | No supplementary information necessary |
| Delbeck 2021a | Yes | Yes |
| Dunbar 1986 | No reply | — |
| Gregory 1991 | No contact possible | — |
| Kaufmann 2021 | Yes | Yes |
| Koffler 1992 | No contact possible | — |
| Kongmalai 2021 | Yes | Yes |
| Lull 2013 | No reply | — |
| Moses 2019 | No reply | — |
| Pendsey 2023 | Not contacted | — |
| Schrader 1985 | No contact possible | — |
| Shnek 1998 | No reply | — |
| Silva 2013 | Yes | No supplementary information necessary |
| Silva‐Jr 2022 | Yes | No supplementary information necessary |
| Tarr 1991 | No contact possible | — |
| Vimalavathini 2009 | No reply | — |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Al Shaibi 1999.
| Study characteristics | ||
| Study design | 8 healthy physicians (mean age 45 years) participated Allocation by block design (4 females, 4 males) allowing for "partial double‐blinding". For block 1, prestorage insulin was used as the reference; for block 2, the reference insulin was stored in a refrigerator at 4 °C. For block 2, both the reference and clay pot insulins were available at the same time. The order of use was randomised using a coin toss (participants and investigators were blinded to the allocation). Storage: neutral human insulin stored in a refrigerator at 4 °C or in a zeer (unglazed clay pot with an internal diameter of 30 cm and a height of 30 cm enabling water to slowly seep through the wall and evaporate, lowering the temperature of the remaining water). A glass jar was partially filled with desert sand and put in the centre of the zeer. A water bath was created by adding water outside the jar to the same level as that of the sand in the glass jar. Water was added to the mark (topped up) daily from a larger water storage zeer. The zeer was closed with a clay lid and kept in the shade. An exact replica, but without water (a dry zeer) was also used to store insulin and record temperatures. Additional bottles of insulin were kept outside in the shade, in the bathroom of an air‐conditioned house and in a refrigerator. Insulin 10 U was removed twice daily from each insulin vial to simulate usage conditions. Temperature readings were taken on 12 separate occasions (18 June 1998 to 20 July 1998 at 7:00 hours or 12:00 hours) during the 6‐week storage period. |
|
| Measurements | Insulin sensitivity: baseline blood sample in fasting participants and IV injection of insulin 0.1 U/kg bodyweight. Blood samples every 5 min for 30 min after the injection. Determination of plasma glucose levels (CV ranging from 2.8% at 1.9 mmol/L and 2.4% at 5.1 mmol/L). Determination of the fall in plasma glucose levels by a computerised best fit method. Calculation of the differences in the slopes for each individual as well as the group's mean and SEM. Also, 2 participants with diabetes using regular and NPH/isophane insulins, mixed just prior to use, stored the insulin they were using during a 2‐month period in an insulin zeer while living in tent in the desert during July and August. |
|
| Results | Mean temperature (range): Insulin zeer: 26.6 °C (25–27 °C) Outside mean temperature: 38.3 °C (34–43 °C) Zeer without water: 34.6 °C (30–39 °C) Insulin vials stored in the shade: 35.3 °C (31–39 °C) Water zeer: 26.7 °C (25–28 °C) Bathroom: 26.4 °C (25–27 °C) Refrigerator: 4 °C No change in colour or appearance of the insulins was noted. The mean fall in plasma glucose levels following reference insulin was 0.213 mmol/L/min, whereas it was 0.192 mmol/L/min following the zeer‐stored insulin, representing 9.9% less fall in glucose in the zeer‐stored insulin (difference not statistically significant). 2 participants with diabetes: no changes in the appearance of insulin or adverse events; no changes in diabetic control. |
|
| Funding sources/conflict of interest | Not mentioned. Probably investigator‐initiated study. | |
| Notes | Small pilot clinical study | |
| Item | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | No | Quote: "They were allocated to the trial using a block design, which allowed partial double‐blinding. Block 1 consisted of the four males and block 2 of the four females. For block 1, pre‐storage insulin was used as the standard (reference) at the start of the experiment, thereby precluding blinding of both subjects and experimentors. For block 2, the reference insulin was that stored in a refrigerator at 4°C. For block 2, both the reference and zeer insulins were available at the same time, and their order of use was randomized using a coin toss. Both subjects and experimentors were blinded to the allocation in block 2." Comment: randomisation on female participants only. |
| Allocation concealment (selection bias) | No | Quote: "They were allocated to the trial using a block design, which allowed partial double‐blinding. Block 1 consisted of the four males and block 2 of the four females. For block 1, pre‐storage insulin was used as the standard (reference) at the start of the experiment, thereby precluding blinding of both subjects and experimentors. For block 2, the reference insulin was that stored in a refrigerator at 4°C. For block 2, both the reference and zeer insulins were available at the same time, and their order of use was randomized using a coin toss. Both subjects and experimentors were blinded to the allocation in block 2." Comment: unclear allocation on female participants only. |
| Blinding of participants and personnel (performance bias) Plasma glucose | Yes | Quote: "Blood samples every 5 min for 30 min after the injection." Comment: objective measurement unlikely to have been influenced by (partial) blinding. |
| Blinding of outcome assessment (detection bias) Plasma glucose | Yes | Quote: "Blood samples every 5 min for 30 min after the injection." Comment: objective measurement unlikely to have been influenced by (partial) blinding. |
| Incomplete outcome data (attrition bias) Plasma glucose | Yes | Comment: plasma glucose values available for all 8 healthy physicians. |
| Selective reporting (reporting bias) Plasma glucose | Yes | Comment: none detected. |
Baechler 2020.
| Study characteristics | ||
| Study design | U‐100 vials comprising insulin aspart (Novorapid), faster‐acting insulin aspart (Fiasp), short‐acting human insulin (Actrapid) (all Novo Nordisk), insulin glargine (Lantus) and insulin lispro (Humalog, Eli Lilly, Indianapolis, US) were stored according to the specifications of the manufacturer until measurement. To assess potential changes in insulin content and stability over time, formulations were sampled from vials and cartridges on days 1, 2, 3, 4, 5, 8, 9, 10, 11, 12, 15, 16, 17, 54 and 85 after receipt from the hospital pharmacy upon storage at 4 °C, 22 °C and 37 °C in temperature‐controlled environments with electronic temperature monitoring (either refrigerator or incubators) to simulate real‐life conditions. |
|
| Measurements | Mass spectrometric measurements were performed. Vial contents were directly analysed by mass spectrometry. Change in insulin content (quantification of monomeric insulin) was determined using linear regression. A significant decline in concentration was considered a slope of the regression line significantly different from zero, evidenced by P < 0.05. | |
| Results | No systematic difference between unopened vials (obtained from the pharmacy) and already used vials (obtained from hospital wards and people with diabetes) was observed. The time‐course experiment during which the insulin content of the different formulations stored at different temperatures (4 °C, 22 °C or 37 °C) was investigated yielded results according to specification. Screening for degradation products throughout the study turned out negative. According to FDA requirements, each manufacturer has to guarantee a concentration of 95–105 IU/mL of intact insulin in U‐100 formulations. Human insulin (Actrapid) in original glass vials: mean was average concentration over all measurements (15 time points over 86 days). 4 °C: 98.6 IU/mL (CV 5.5%) 22 °C: 99.4 IU/mL (CV 5%) 37 °C: 98.7 IU/mL (CV 4.6%) |
|
| Funding sources/conflict of interest | University Institute of Clinical Chemistry/Center of Laboratory Medicine, Clinical Metabolomics Facility, Inselspital, Bern, Switzerland, University Hospital: support in logistics and instrumentation. UDEM Scientific Fund and Foundation "Fonds pour la Recherche Thérapeutique", Puilly, Switzerland. |
|
| Notes | ||
Delbeck 2021a.
| Study characteristics | ||
| Study design | Examples of formulated insulin specimens, and the United States Pharmacopeia (USP) human insulin standard were studied for comparison of protein structural changes. Insulin cartridges included insulin detemir (Levemir), insulin aspart (NovoRapid), NPH insulin human (Protaphane) (all produced by Novo Nordisk), insulin lispro (Humalog), NPH insulin lispro (Humalog Mix50), insulin glargine (Abasaglar) (all produced by Eli Lilly), and insulin lispro (Insulin lispro Sanofi, Sanofi‐Aventis Groupe). All insulins were available as formulations in 3 mL cartridges with a biological activity of 100 IU. In addition to the commercial insulin samples, ultrafiltrates of the original formulations were prepared. The insulin formulations, aqueous solutions of insulins (purified by ultrafiltration) and the human insulin USP standard were kept in 500‐μL sterile sealed plastic tubes when stored in climatic exposure test cabinets at 0 °C, 20 °C and 37 °C, respectively, for different durations (up to 10 weeks). |
|
| Measurements | Infrared attenuated total reflection spectroscopy was performed. For analysing the insulin secondary structure alteration, the analytical evaluation tools were either based on the monitoring of specific band shifts or a band deconvolution and band area integration providing quantitative figures about degradation. Significant changes within the insulin samples at various storage temperatures due to misfolding and subsequent fibrillation allowed the assumption of a decrease in bioactivity. The data evaluation was carried out using the QUANT2 analysis module of the OPUS software for PCA calculations of spectral data sets, comprised of multiple‐repeat spectra of different insulin pharmaceuticals. Another PCA analysis was performed using 4 different formulated insulin samples (insulin lispro, insulin aspart, human NPH insulin, insulin detemir) as stored for 10 weeks at 37 °C. |
|
| Results | Especially under storage conditions at high temperature (37 °C), an increased amount of β‐sheet fractions could be detected by band deconvolution, underlining the protein structural misfolding. Compared to the ultrafiltrated insulin samples, only small shifts were detected for the insulin formulations, suggesting that the added stabilisers were protecting the insulin effectively from accelerated misfolding. | |
| Funding sources/conflict of interest | Quotes: "The author(s) received no financial support for the research, authorship, and/or publication of this article." "The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article." |
|
| Notes | ||
Dunbar 1986.
| Study characteristics | ||
| Study design | Insulin syringes of 3 manufacturers were filled with insulin (regular Iletin I, Humuline R, Lente Iletin I, Eli Lilly) and stored at 4 °C for 2 weeks. After 1, 3, 7 and 14 days, the syringes were removed, and aliquots were taken for radioimmunoassay and radioreceptor determinations. Samples of refrigerated stock insulin served as controls. 6 syringes per syringe type and insulin type were evaluated for each time period. | |
| Measurements | Insulin radioimmunoassay: highly purified crystalline porcine insulin (gift of Eli Lilly and Company) was used as the insulin standard. Radioreceptor assay: the biological potential of insulin during storage was evaluated by binding to hepatic insulin receptors. Hepatocytes were isolated from Wistar Furth rats and standard insulin‐binding isotherms were established. Samples of stock and syringe‐stored insulin were evaluated based on their capacity to bind and displace standard insulin preparations. |
|
| Results | The insulin content of samples stored in plastic syringes for up to 2 weeks was not significantly different from that of the stock controls. The 3 types of insulin tested were comparable in their degradation. Insulin Lente gave more assay variability on storage, possibly due to settling of suspension in the syringe. Receptor‐binding isotherms were the same for the different types of insulin as well as for the type of syringe storage. Insulin Lente was shown to be less effective in displacing labelled insulin when compared with regular or Humulin insulin. |
|
| Funding sources/conflict of interest | Funding not mentioned. Quote: "Highly purified crystalline porcine insulin (Lot No. 615‐2H2‐300, gift of Eli Lilly) was used as the insulin standard." |
|
| Notes | ||
Gregory 1991.
| Study characteristics | ||
| Study design | Single vials of short‐acting human (human Actrapid) and beef insulin (Quicksol, The Boots Company PLC) were stored under each of the following environmental conditions in the laboratory: 1. 4 °C in the dark, 2. room temperature 19–24 °C in the dark, 3. room temperature 18–25 °C in daylight, 4. in direct sunlight on a window sill (8–35 °C), 5. at 37 °C in the dark, and 6. carried in the breast pocket of a shirt. Single vials of each insulin were also stored under conditions 1, 2, 3, and 6 and subjected to twice‐daily sampling, i.e. the removal of a small amount of insulin to simulate injections. 4 measurements were taken from each vial, i.e. the amount of insulin and the 3 ITP groups. These were obtained from HPLC traces taken immediately after dispensing and at 2, 4, 6, 8, 10, 14, 18 and 26 weeks. Thereby, the degradation of both human and beef insulin was studied and the concurrent changes in ITP over 6 months to assess the effects of different environmental conditions and sampling on the process of insulin transformation. | |
| Measurements | The area under the trace was measured by HPLC with an integration programme. Only the area from the start of the insulin peak was included to eliminate the solvent front and preservative (phenol and metacresol) peaks. Results were expressed as a percentage of the total area. Baseline measurements were approximately equal within each data set. The relationship between each variable and time was approximately linear. The computer package GLIM was used to fit a series of linear models to each data set. | |
| Results | The initial insulin content in human vials was 98.2 ± 0.08% of total protein. None of the environmental conditions caused a significant loss of total protein during the 6 months of study. The best‐fitting model included both environment and sampling as significant factors. Human insulin kept at 4 °C and in the dark without sampling was associated with the slowest degradation (−0.069% total protein/week). The vials kept in direct sunlight and at 37 °C in the dark degraded most quickly (−0.749% total protein/week and –1.184% total protein/week, respectively). The slope estimates from the 4 vials kept at room temperature were similar. Exposure to daylight did not accelerate insulin degradation in vials stored at room temperature. Repeated sampling only affected the degradation rate in the vials kept at 4 °C in the dark. |
|
| Funding sources/conflict of interest | Grant from the British Diabetic Association. Study author RG was a Clinical Research Fellow sponsored by Novo Laboratories, Basingstoke, UK. |
|
| Notes | ||
Kaufmann 2021.
| Study characteristics | ||
| Study design | Human insulin formulations (rapid, NPH/isophane and premixed rapid/NPH insulins) and 4 analogue insulin formulations commercially available in Switzerland and France were bought in a local pharmacy and tested, with proper storage conditions ensured. The cold chain was respected and monitored throughout the supply process until reception in the refugee camp pharmacy and throughout its storage there. All vials at T = 0 were assumed to be amyloid seed‐free. Temperature fluctuations were measured in Dagahaley refugee camp (northern Kenya) using log tag recorders. Ambient temperature oscillated between 25 °C and 37 °C with a 12‐hour cycle, exceeding usual recommendations for insulin use out of refrigeration. In order to assess insulin stability, temperature oscillations that were observed in the field were replicated in the laboratory, and insulin formulations were exposed to these fluctuating temperatures in the laboratory. A series of mixed insulin vials were stored at ambient temperature in Kenya during a period of 4 weeks of use by people with diabetes and brought back to Geneva for analysis. Insulin stored at 2–8 °C was used as positive control and heat‐degraded insulin as negative control. Furthermore, the effect of continuous exposure to high temperatures of 31 °C and 37 °C was also investigated, as controls comparative with data available in the literature (potency determination). |
|
| Measurements | Reference samples: a complete series of vials or cartridges of the insulin formulations were stored at 4 °C, and used as references for all 3 analytical methods (HPLC calibration curves preparation at each time point of the stability study, circular dichroism measurements, biological activity assessment). Samples: 3 independent series of vials or cartridges were placed in an incubator. Temperature cycles were programmed as follows: start at 25 °C, increase to 37 °C in 12 hours, then back to 25 °C in 12 hours, in continuous loop mode. These conditions were intended to mimic the field oscillating temperature cycling both in terms of frequency and amplitude. Temperature variation over time in the incubator was monitored. 3 aliquots were withdrawn at the defined time points throughout the study and analysed in triplicate. A second series of samples including vials or cartridges of each tested formulation was submitted to temperature cycles, and insulin was taken out daily to simulate a typical daily treatment with dosages of 10 IU in the morning and 12 IU in the evening. A total of 220 μL per day of insulin was withdrawn from the vials or cartridges with 1 IU per 10 μL. This was intended to assess whether the variation of air volume present in the vials had any influence on the overall stability, since the interface between air and insulin solution has been reported to trigger aggregation. For this series, 6 injections at each time point were performed. A third series of samples was exposed to standard isothermal conditions to allow comparison with previously released studies. Since the median value of temperature during the cycling process remains above the temperature of 31 °C indicated by the manufacturers, a series of vials and cartridges was exposed continuously to this temperature, and insulin quantification was performed after 1, 4 and 8 weeks of this treatment. Finally, rapid and NPH/isophane insulins were exposed to a continuous temperature of 37 °C. For each condition, 6 injections were performed. Insulin quantification and potency determination was performed by RP‐HPLC coupled with ultraviolet detection. All the measured values for time = 0 were considered 100% and were used as references for the whole stability study. Circular dichroism: near‐ and far‐ultraviolet spectra were obtained on a Jasco J‐815 circular dichroism spectrophotometer. To produce negative control for the bioactivity and circular dichroism assays, and based on the 80 °C results, forced degradation of the samples was achieved by heating them at 115 °C for 30 min. These samples were also analysed in terms of insulin quantity. Bioactivity testing: the bioactivity of the stored insulin samples was assessed using 2 hepatocytes cellular models, in which insulin efficiency on insulin receptor (IR) and akt kinase activation (Akt) phosphorylation was detected and quantified. Visual inspection: visual inspection was performed according to European Pharmacopoeia prescriptions. For solutions, a visual detection of any colour change or variation in clarity was performed in front of a white and of a black surface. For suspensions, the visual inspection included monitoring of appearance of large aggregates (clumps) or material adhering to the cartridge/vial walls (frosting). |
|
| Results | Insulin instability: the analysis of the samples in which instability was forced by high temperature (80 °C) showed a decrease in insulin content of 9–14% compared to corresponding reference formulations stored at 2–8 °C. The quantity of insulin detected in the samples heated at 115 °C for 30 min for bioactivity and circular dichroism measurements, was 85.5%. The sample also appeared yellowish and contained visible structured aggregates. Insulin quantification by HPLC stored under oscillating conditions:
Isothermal studies at 31 °C and 37 °C: more classical isothermal storage experiments were performed with the same insulin formulations and compared with data from the literature as well as from the studied oscillating temperatures. Since the median value of temperature during the cycling process (31 °C) remained above the temperature specified by manufacturers, a series of vials was exposed continuously to this temperature, and insulin quantification was performed at 1, 4 and 8 weeks. All insulin formulations started to degrade after 4 weeks (degradation proportion between 5% and 13.5%, compared to values obtained for the corresponding formulations at time = 0). There was further degradation after 8 weeks (12–19%), with all formulations out of the range accepted by the pharmacopeia (100 ± 10%). NPH/isophane human insulin and short‐acting human insulin were exposed to a continuous temperature of 37 °C. Both were determined to be out of range after 1 week for the suspension (15.4% decrease) and 2 weeks for the solution (12.5% decrease). Antimicrobial protection: in order to confirm antimicrobial protection, quantification of the preservatives present in the formulations was performed by HPLC at time = 0 and after the temperature cycling period to determine whether degradation and/or decrease in concentration and therefore in efficacy occurred. After 12 weeks of continuous temperature cycling, concentrations of the preservatives phenol and metacresol were 92.4–94% and 92.7–97.7% of claimed values, respectively. No microbial contamination was detected while taking transmission electron microscope images of the same samples. 3‐dimensional conformation of insulin under oscillating storage conditions: tridimensional conformation of insulin monomers was identical in all the investigated samples, except in denatured mixed insulin. Insulin bioactivity under oscillating storage temperatures: residual bioactivity of mixed insulin was verified on 2 hepatic cell lines for insulin samples that were exposed to fluctuating temperatures (25–37 °C) for 12 weeks and for a reference formulation stored at 2–8 °C (mean ± SEM of 4 different experiments). Insulin‐induced phosphorylation of the insulin receptor (IR) and Akt (key intracellular signalling pathways activated by insulin to control glucose homeostasis) was assessed in 2 classical cell models of hepatocytes, i.e. HepG2 and Huh‐7 cells. After overnight serum starvation, cells were stimulated for 15 min at 37 °C with 10−8 M of each different insulin. Mixed insulin exposed to 4 weeks out of refrigeration in an individual's home in Kenya and mixed insulin undergoing variations between 25 °C and 37 °C for 12 weeks maintained a bioactivity similar to insulin kept at 4 °C. Insulin samples heated at 115 °C for 30 min (mixed insulin degraded) showed a strong decrease in their bioactivity. The presence of air in the vials and multiple septa punctures had no impact on stability, and no significant difference in stability was found between insulin conditioned in pen cartridges and 10 mL glass vials. |
|
| Funding sources/conflict of interest | Funded by Médecins Sans Frontières (MSF) Switzerland. | |
| Notes | HepG2/Huh‐7 cells: immortalised cell line consisting of human liver carcinoma cells. | |
Koffler 1992.
| Study characteristics | ||
| Study design | Each day 4 syringes were filled with insulin 10 U, 2 syringes with short‐acting insulin (Velosulin human, Nordiska) and 2 syringes with intermediate‐acting insulin (Insulatard human, Nordisk) by a registered nurse from a Diabetes Unit. The prefilled insulin syringes were kept in a refrigerator (4 °C) for up to 10 days. | |
| Measurements | After 10 days, the insulin samples from each syringe were cultured concurrently in a brain–heart infusion broth and in a thioglycollate brain infusion broth. The samples were incubated at 37 °C under aerobic and anaerobic conditions for up to 7 days, with a daily check for growth. | |
| Results | No signs of bacterial growth were evident in any of the specimens. | |
| Funding sources/conflict of interest | Not mentioned. Probably investigator‐initiated study. | |
| Notes | aShort‐acting insulin was withdrawn for commercial reasons by the manufacturer. | |
Kongmalai 2021.
| Study characteristics | ||
| Study design | 4 different commercial basal insulins were evaluated, including "long‐acting insulin, NPH‐1, NPH‐2 and NPH‐3." To investigate the effect of different temperatures on the stability of insulin, opened insulin pens were stored at room temperature, in a refrigerator (2–8 °C) or in an incubator (37 °C) for 28 days. For each type of insulin, 2 batches of insulin were used to minimise the variation of the manufacturer's production. An unopened insulin pen from each batch was stored in a refrigerator as a control. Room temperature and humidity were recorded every hour throughout the study period. 2 batches of commercial insulin pens were divided into experimental groups and controlled groups. In the experimental groups, all insulin pens were opened and flipped back and forth 20 times. 2 units of insulin were discarded every day to simulate the daily usage of insulin‐treated people with diabetes. Unopened insulin pens in the control group were stored in a refrigerator. All in‐use insulin pens were kept away from direct exposure to sunlight or artificial light. |
|
| Measurements | Insulin was analysed using the ultra HPLC technique. The temperatures were reported as a mean ± SD during daytime (6 a.m. to 7 p.m.), night‐time (7 p.m. to 6 a.m.) and all day. The quantity of insulin was reported as mean ± SD (AUC). The difference between insulin potency in each storage condition was analysed using an independent t‐test. | |
| Results | During the study period from 3 July to 31 July 2017, the mean room temperature was 29.7 °C (SD 2.8) (range 25.5–37.1 °C). The mean daytime temperature (6 a.m. to 7 p.m.) was 31.1 °C (SD 2.9) (range 27.1–37.1 °C) and the mean night‐time temperature (7 p.m. to 6 a.m.) was 28.0 °C (SD 1.3) (range 26–31 °C). The mean humidity in the air‐conditioned room was 74.6% (SD 9) (range 50.0–89.5%). The mean humidity was 50% in the refrigerator and 38% in the incubator. The percent change from baseline AUC ranged from −2.8% to +1.5% in the intermediate‐acting insulins: NPH1 −2.6% (room temperature), −2.8% (refrigerator), −1.0% (incubator); NPH2 +1.4% (room temperature), +1.5% (refrigerator), +0.7% (incubator); NPH3 −1.3% (room temperature), −2.2% (refrigerator), −1.5% (incubator). | |
| Funding sources/conflict of interest | Supported by Faculty of Medicine Siriraj Hospital, Mahidol University, Grant Number R016032029. Quote: "The authors had no commercial associations that might create a conflict of interest in connection with this article." |
|
| Notes | We contacted the study authors to clarify the exact nature of the analysed NPH insulins. NPH‐1 was Humulin N, NPH‐2 was Insulatard and NPH‐3 was Gensulin N. | |
Lull 2013.
| Study characteristics | ||
| Study design | The stability U‐500 insulin (Humulin R U‐500 regular insulin) was evaluated after storage in 0.3 mL polypropylene insulin syringes. 0.25 mL (125 units) of U‐500 insulin were drawn into each syringe. Samples were analysed immediately to establish baseline (day 0) stability values. The remaining syringes were kept capped inside an open tray in a laboratory refrigerator (3–5 °C). At days 7, 14, 21, and 28, 3 of the stored syringes were removed for analysis. A sample also was drawn for a forced degradation sample. | |
| Measurements | The RP‐HPLC method was adapted for use with modifications according to standard insulin HPLC protocols used by the column manufacturer. A standard curve of concentrations ranging from 0 U/mL to 100 U/mL was prepared to establish the linearity of the peak area versus concentration. Stability‐indicating ability of the method was determined through the assay of a forced degradation sample. The mean (SD) AUC for insulin peak 1 was calculated. Mean values for days 7, 14, 21 and 28 were adjusted to a percent of the mean day 0 value (measured at 500 U/mL; diluted sample measured at 40 U/mL). | |
| Results | Results were given as percent initial concentration of 500 U/mL remaining (mean of 3 biological replicates).
|
|
| Funding sources/conflict of interest | Quote: "The authors declare no conflicts of interest or financial interests in any product or service mentioned in this article, including grants, employment, gifts, stock holdings, or honoraria." | |
| Notes | ||
Moses 2019.
| Study characteristics | ||
| Study design | Authors reported an analysis of insulin concentration and stability of production batches covering 7 years of production of NPH insulin: Insulatard (Novolin N, Novo Nordisk A/S) and regular human insulin Actrapid (Novolin R, Novo Nordisk A/S). Actrapid and Insulatard were analysed for impact on insulin concentration under the following 2 conditions.
Additionally, an analysis of previous 3 years of returned insulin samples from US people with diabetes was performed. |
|
| Measurements | Insulin concentrations were measured using RP‐HPLC. | |
| Results | None of the samples were outside FDA specifications. The analysis was based on stability data from ≥ 85 insulin production batches covering 7 years of production. | |
| Funding sources/conflict of interest | Quotes: "Alan Moses, Jesper Bjerrum, Morten Hach, Lars Holm Wæhrens, Anders Dyhr Toft are full‐time employees and minor stockholders of Novo Nordisk." "This study was funded by Novo Nordisk A/S." |
|
| Notes | ||
Pendsey 2023.
| Study characteristics | ||
| Study design | 6 insulins (100 IU/mL; purchased from local pharmacies) were studied: 10 mL vials of human soluble (Eli Lilly), human isophane (Eli Lilly) and human soluble/isophane 30/70 (Novo Nordisk) vials; and 3 mL cartridges of insulin aspart (Novo Nordisk) and 2 preparations of insulin glargine (Sanofi and Eli Lilly). 6 consenting families with a young person with insulin‐requiring diabetes, attending the Diabetes Research Education and Management (DREAM) Trust in Nagpur, India participated in the study. All families lived in Nagpur city within 5 km of the DREAM Trust clinic. Each received 2 different types of insulin which were stored either in water‐tight bags, in open plastic containers, placed on a cupboard or high shelf, or in water‐tight bags in clay pots with a separate water compartment and all in shaded areas. 12 control 10 mL vials or 3 mL cartridges of each insulin were also kept refrigerated at the DREAM Trust (except for human isophane and human 30/70 insulins where only 6 vials/cartridges were stored). All samples of each type of insulin came from the 1 batch. | |
| Measurements | Each family was provided with Microlite USB temperature and humidity data loggers for temperature measurements (Fourtec). Temperatures were recorded in all storage containers every 15 min and during shipment for analyses at 2 analytical centres (University of Florida, USA and University of Gothenburg, Sweden). The temperature sensitive phase of the study was undertaken March to June 2021. Insulins were stored by families for 1, 2 and 4 months, and then retrieved and refrigerated at the DREAM Trust. Samples were cold‐chain couriered to University of Florida and University of Gothenburg in July 2021, and kept refrigerated until analysis, with temperature data loggers used during transit as well. All insulin vials and cartridges remained unused until analysis. Excluding Isophane‐containing insulins which were solely transported to the University of Florida, insulins collected were equally distributed between the 2 institutions. Samples received were analysed based on insulin type using 2 different methods, randomised in terms of analysis sequence within each type of insulin. Both approaches used the mean concentration from refrigerated control samples as reference to assess changes over the study period. At the University of Florida, analyses were conducted using the respective USP monographs for each insulin type. Chromatographic separation and detection were conducted with a Vanquish ultra HPLC variable wavelength detector (ThermoScientific). All data were processed and analysed with Chromeleon 7. At the University of Gothenburg, 1H NMR was used to evaluate 4 insulin types using a sample preparation protocol ensuring minimal perturbation of the native formulations. 1H NMR was not used to assess the isophane and mixed insulin because the high viscosity of isophane results in too broad spectral lines in solution NMR. An insulin concentration of 100% ± 5% was acceptable according to the USP standard. |
|
| Results | In the analysis at the University of Florida, all human insulin samples maintained ≥ 95% of the refrigerated potency except for 1 vial each of human soluble, human soluble–isophane (30:70), and human isophane (range 92.4–94.1%), all at 4 months. In the analysis done at the University of Gothenburg, there was an apparent subtle line width increase and slight peak shifts in NMR spectra, which correlated with storage time for non‐refrigerated samples, probably due to altered conformation or multimerisation in a minor fraction of the insulin molecules. No loss of relative concentration was found using NMR. A decrease in total concentration in any of the insulin types was not observed as being larger than the precision of measurements (less than 1%), regardless of storage time or type of storage. When the insulin potency difference across all investigated insulins between refrigerated samples and samples stored in clay pots was compared to the difference between refrigerated samples and samples stored in open boxes, the decline in mean potency for those samples stored in clay pots at month 4 (0.5%) was significantly lower than that for those stored in open boxes (3.6%, P = 0.001). The potency difference between refrigerator and clay pot samples was −2.8% to 2.0%, while the potency difference between refrigerator and open boxes was −7.9% to −0.1%. The overall mean ± SD and median (IQR) were −0.5% (SD 1.7) and −0.3% (IQR −2.1% to 1.0%) for clay pot; −3.6% (SD 2.3) and −3.7% (IQR −5.7% to −1.8%) for open box. |
|
| Funding sources/conflict of interest | Quote: "JM and GDO work for the Life for a Child programme at Diabetes NSW & ACT, which does receive non‐salary support from Eli Lilly. MAA reports cooperation (consultative, educational, and research) with companies and entities (CodeBio, Diamyd Medical, Endsulin, IM therapeutics, and Repitoire) interested in type 1 diabetes prevention and reversal (not directly insulin thermostability), as well as receiving an independent award from the Novo Nordisk Foundation. MAA is also President of Insulin for Life USA. All other authors declare no competing interests." | |
| Notes | ||
Schrader 1985.
| Study characteristics | ||
| Study design | Lilly human insulin(Eli Lilly GmbH), Velasulina Nordisk (Novo), Hoechst human insulin 31H (0‐40) (Hoechst AG), Actrapid human insulin (Novo) and Actrapid MC insulin (Novo) were investigated 10 months after production. Between the experiments, the insulin preparations were stored at 4 °C. Investigators received from Hoechst an insulin formulation which was stored for 3 years at 37 °C. This preparation contained aggregates and smaller parts of the insulin molecule. Eli Lilly and Company and Nordisk provided the date of production of the tested insulin. All these preparations were soluble neutral insulins, 40 U/mL. 2 preparations each from the insulins were shaken for 72 hours at 37 °C and at ambient temperature for 72 hours. Investigators collected samples of about 1 mL of insulin during the separations before and after shaking. Then, radioimmunological activity was tested with an insulin kit. Investigators inoculated parts of the insulin sediment on culture plates to look for bacterial growth. |
|
| Measurements | Insulin potency was investigated by RP‐HPLC and gel‐filtration chromatography. Authors collected samples of about 1 mL at the retention time of insulin during the separations before and after shaking. Then, radioimmunological activity was tested with an insulin kit from Sorin Biomedica (Saluggia, Italy). Furthermore, study authors measured bacterial growth after inoculation. | |
| Results | There were no differences between the insulin formulations separated at ambient temperature as well as at 37 °C. Investigations performed at ambient temperature showed the same molecular weight range for insulin as investigations performed at 37 °C. After shaking at 37 °C for 72 hours, the Hoechst human insulin and Actrapid MC showed a milky suspension. Under the same conditions, the human insulin of Eli Lilly remained a clear solution. After shaking at ambient temperature and at 2 different velocities, each of the 2 formulations remained clear. The immediately performed gel‐filtration showed in no case higher molecular aggregates but the formulations showing milky suspensions contained insulin in an obviously reduced quantity. The material, inoculated on culture plates, showed no bacterial growth. The sample of insulin received during separations before and after shaking showed radioimmunological activity. For the insulin stored for 3 years at 37 °C separations of insulin aggregates were visible. |
|
| Funding sources/conflict of interest | Quotes: "We received from the company of Hoechst an insulin formulation which was stored during three years at 37°C." "We appreciate the fact that the companies of Eli Lilly and Nordisk have been so kind as to give us the date of production of the tested insulin. All these preparations were soluble neutral insulins, 40 E/ml." "Supported by Foundation Herbert‐Weishaupt e.V." |
|
| Notes | aShort‐acting insulin was withdrawn for commercial reasons by the manufacturer. | |
Shnek 1998.
| Study characteristics | ||
| Study design | Study authors investigated short‐acting, intermediate‐acting and mixed human insulin (Humulin Regular, Humulin NPH, and Humulin 70/30 [N/R]). | |
| Measurements | Combined effects of temperature and agitation on insulin pen cartridge products were investigated. Physical stress test, TCRT included temperature cycling (25–37 °C) in an incubator unit combined with resuspension conducted twice daily on a mechanical device outside the incubator unit. Temperature changed from 25 °C to 37 °C over 2 hours, with 10‐hour periods at each temperature. The temperature was decreased from 37 °C to 25 °C over 2 hours and the cycle resumed for 28 days. Cartridges were placed on a specially constructed resuspension machine which mechanically mimicked the resuspension instructions provided in the literature for people with diabetes. The resuspension machine performed the designated number of rolls and inversions on the cartridges, consisting of 3 sets of 10 rolls plus 10 inversions twice daily. Visual assessment: cartridges were visually assessed by trained operators after resuspension on test days 0, 7, 14, 21, and 28. Cartridges were checked for visual changes compared to quiescent refrigerated control samples. For solutions, visual changes included changes in colour and clarity from a clear solution. For suspensions, visual changes included the presence of large aggregates (clumps) or material adhered to the cartridge wall (frosting), or both. Insulin potency: RP‐HPLC was used to determine total insulin potency. Physical stress test, HTEAT included continuous high temperature (37 °C) exposure in an incubator unit combined with periodic daily agitation by specially constructed rotation devices while still inside the incubator unit. The incubator unit was calibrated to 37 °C. The cartridges were rotated at 30 rotations per min for 4 hours by custom‐made rotators. Additional agitation was imparted on the test cartridges through resuspension outside the incubator unit. The resuspension machine was used after the cartridges were removed from the incubator unit and prior to visual assessment. The resuspension machine performed 3 sets of 10 rolls plus 10 inversions. Visual assessment: cartridges were visually assessed after resuspension by trained operators on test days 0, 2, 5, 7, 9, 11, 14, 17, 21, 24 and 28. Cartridges were checked for visual changes compared to quiescent refrigerated control samples. For solutions, visual changes included changes in colour and clarity from a clear solution. For suspensions, visual changes included the presence of large aggregates (clumps) or material adhered to the cartridge wall (frosting), or both Insulin potency: dose potency was defined as the quantification of IUs recovered from a specific dose dispensed from a cartridge. RP‐HPLC was used to determine the dose potency. Microscopy was performed and photographs were taken at 1000 × magnification. Particle size measurements were performed and the volume percent distribution mean was reported for insulin crystals. Electrophoretic mobility measurements were performed on insulin suspensions. |
|
| Results | For soluble short‐acting formulations (Humulin R), all stress testing studies showed no change in the product for all conditions tested over 28 days. Neither the potency nor visual assessments of intermediate‐acting insulin suspensions (Humulin NPH [N] and Humulin 70/30 [N/R]) were negatively impacted when subjected to TCRT conditions. HTEAT testing of both suspension products resulted in an HPLC potency measurement for a visually unchanged cartridge that was within specifications after 1 week on the test. For suspensions, the HTEAT stress conditions caused visual changes (agglomerates) over 28 days. |
|
| Funding sources/conflict of interest | Quote: "Contribution from Lilly Research Laboratories, Eli Lilly and Company, Indianapolis, Indiana 46285." | |
| Notes | ||
Silva 2013.
| Study characteristics | ||
| Study design | Intermediate‐acting mixed human insulin (Humulin 70/30) and insulin detemir (Levemir) vials were opened, time 0 samples were taken, and the vials were randomly assigned to room temperature or refrigerated storage. Vials stored in refrigerated conditions were kept at 2–8 °C and those stored at room temperature were stored at 22–24 °C. | |
| Measurements | A modified HPLC assay was used to quantify insulin AUC profiles under different storage conditions. AUC was quantified at opening (time 0) and at days 28, 35 and 42. 2 batches of insulin were stored in refrigerated conditions and 2 batches at room temperature conditions, each run at different times. Batch 1 ran for 35 days and batch 2 ran for 42 days. Investigators could not confirm which peaks corresponded to metacresol preservative or insulin nor additional procedures to detect degradants. |
|
| Results | AUC for refrigerated samples days 28, 35 and 42 were compared to time 0 samples. After 35 days for the intermediate‐acting insulin, the 12‐min peak ranged between 99% and 101% of time 0 AUC reference values. The 21‐min peak ranged between 99.6% and 100% of time 0 AUC reference values. After 42 days, the 12‐min peak was 93% of time 0 AUC reference values. The 21‐min peak was 94% of time 0 AUC reference values. |
|
| Funding sources/conflict of interest | Quotes: "Competing interests: none declared". "None of this work was funded, supplied or supported by any pharmaceutical company." |
|
| Notes | ||
Silva‐Jr 2022.
| Study characteristics | ||
| Study design | Commercial insulin (100 U/mL, equivalent to 600 μM according to WHO), short‐acting human insulin (Humulin R), intermediate‐acting insulin (Humulin N) and rapid‐acting insulin analogues (Lispro and Aspart), in 3 mL ampoules, were purchased from local suppliers, transported in ice and stored at 4 °C until use. All experiments were performed in replicates from independent insulin vials. All analyses were performed by incubating at 4 °C and 37 °C for 5 weeks, with weekly sampling of about 300 μL aliquots using new sterile 1 mL insulin syringes, transferring these to low protein binding polypropylene tubes, while being kept in ice during analysis and discarded afterwards. | |
| Measurements | TEM: evaluation for the presence of agglomerates. ThT fluorescence: analysis of the formation of amyloid material. Electrospray ionisation – ion mobility spectrometry‐mass spectrometry (ESI‐IMS‐MS): evaluation of the formation of aggregates due to chemical degradation. DLS to monitor the oligomeric and particulate distribution of insulin. SEC: additional evaluation of the products. |
|
| Results | TEM: authors found no agglomerates in the freshly opened products. Large amorphous agglomerates were observed at 35 days after first handling, both at 4 °C and 37 °C. No evidence for other morphologies were found in replicates of these material at 3000 × magnification, suggesting that the material consisted mainly of amorphous aggregates. ThT fluorescence: compared with baseline measurements, authors observed that ThT fluorescence remained largely stable for up to 28 days, during which the samples were routinely handled and stored at 4 °C and 37 °C. After 35 days, a slight increase in ThT fluorescence was found, although no other evidence of amyloid was found in TEM and DLS. These data suggested that amyloid material was not dominant as indicated by ThT, and actually formed during handling of insulin products, as identified by DLS, and was likely amorphous as shown by TEM. Electrospray ionisation – ion mobility spectrometry‐mass spectrometry: the ion mobility spectra revealed multiple conformers for charged states of insulin monomers, that remained similar after 35 days of handling and storage at 4 °C and 37 °C. The ion mobility spectra did not show stable high‐order oligomers as observed in DLS and TEM. The insulin formulations revealed multiple charged state as expected for their respective molecular weight, without evidence of detectable oxidation, deamidation or other degradation. Deconvoluted spectra confirmed their molecular weight and the lack of detectable degradation products. These data suggested the chemical stability of the investigated insulin formulations. DLS: there was a main peak with a hydrodynamic radius (Rh) of about 2 nm for all insulin products. A smaller second population with higher Rh was found for regular and Lispro insulins. From day 7 up to day 35 of evaluation, all formulations presented populations of increasing Rh at both incubation temperatures. Authors observed a progressive decrease in the relative amount of the lower Rh population over time after 7 days of use. The decrease was more pronounced at 4 °C for both the regular insulin products and the insulin analogue Aspart, whereas a reverse pattern was observed for Lispro insulin. Overall, results indicated that at both storage temperatures, there was a progressive reduction in the relative amount of the low Rh species after 7 days from first use of the insulin products. SEC: the chromatogram of the insulin formulations after 35 days of weekly handling and storage at 4 °C and 37 °C and freshly opened products revealed 1 major peak at retention time of about 10.0 min, corresponding to the major insulin oligomer and others corresponding to phenolic compounds accordingly at about 17.5 min and 21.5 min. No other major peak was observed in the SEC analysis. |
|
| Funding sources/conflict of interest | Quotes: "This work was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, Finance Code #001), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq; PQ/311582/2017‐6; PQ/313179/2020‐4, to LMTRL), Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro Carlos Chagas Filho (FAPERJ; grants E‐26/202.998/2017‐BOLSA, E‐26/200.833/2021‐BOLSA, E‐26/010.001434/2019‐Tematico and E‐26/210.195/2020 to LMTRL) and Programa Nacional de Apoio ao Desenvolvimento da Metrologia, Qualidade e Tecnologia (PRONAMETRO, to LMTRL; 01/2018‐BOLSA) from the Instituto Nacional de Metrologia, Qualidade e Tecnologia (INMETRO). The funding agencies had no role in the study design, data collection and analysis, or decision to publish or prepare of the manuscript." "The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Luis Mauricio T. R. Lima reports financial support was provided by Conselho Nacional de Desenvolvimento Científico e Tecnológico. Luis Mauricio T. R. Lima reports financial support was provided by Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro Carlos Chagas Filho. Luis Mauricio T. R. Lima reports financial support was provided by Instituto Nacional de Metrologia, Qualidade e Tecnologia. Talita Stelling de Araujo reports financial support was provided by Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro Carlos Chagas Filho. Hamilton da Silva Jr reports a relationship with Novageia Biotechnology that includes: board membership and equity or stocks. Marcius da Slva Almeida reports a relationship with Biozeus Biotechnology that includes: consulting or advisory. none." "Prof. Hamilton da Silva Junior is a Collaborator Professor at UFRJ and has job appointment at Novageia Biotechnology S/A. MSA has served as consultant and received financial support from BioZeus Biotechnology S/A. The other declare no financial or intellectual conflicts of interest with the contents of this article." |
|
| Notes | ||
Tarr 1991.
| Study characteristics | ||
| Study design | The stability of regular (Humulin R, 100 units/mL), isophane (Humulin 100 units/mL), combination (Humulin N/R (70/30)), and extemporaneously prepared combination human insulin (Humulin 7R (70/30) and Humulin) was evaluated after storage in 1 mL polypropylene syringes. Extemporaneously prepared combination human insulin was made by withdrawing 30% of the volume from isophane insulin vials and replacing that volume with regular insulin. For each type of insulin, 0.4 mL samples were withdrawn from multiple‐dose vials into polypropylene syringes and propylene‐ethylene copolymer syringes. The contents of 3 syringes of each type were analysed immediately to establish baseline (time 0) values. Of the remaining syringes, half were placed in a plastic bag and kept at room temperature (22–23 °C), while the other half were placed in a plastic bag and kept in the refrigerator (3–5 °C). A vial of each type of insulin was also maintained under the same conditions as the syringes. At days 1, 3, 7, 14, 21 and 28, 3 syringes stored at each temperature were removed, and the contents were analysed for insulin potency, metacresol and phenol concentrations. | |
| Measurements | HPLC was used as a measure of insulin potency. The insulin products were also tested in triplicate for sterility after 1, 2, and 4 weeks of storage at 4 °C and 23 °C in the 2 plastic syringes. Before the direct inoculation of the insulin products into the sterility test media, the same preservatives used in these products were added to the media for evaluation of the bacteriostatic or fungistatic activity produced by the preservatives in the media. Because no bacteriostasis or fungistasis occurred, the direct inoculation procedure was deemed appropriate. In all sterility studies, the media were concurrently challenged for growth potential. The presence or absence of microbial growth after the incubation period was used to determine whether sterility was maintained. Insulin potency data and concentrations of the preservatives at each test day were compared with the potency of insulin in the syringes at day 0 and initial concentrations, respectively. |
|
| Results | Human insulins stored at 4 °C and 23 °C did not show a significant loss of potency in either type of syringe during the 28‐day test period. The metacresol concentration decreased in all human insulins tested at room temperature during the 28‐day study. The decrease was smaller at 4 °C than at 23 °C. The samples stored in polypropylene syringes tended to show a greater decrease in metacresol concentration than those stored in propylene‐ethylene copolymer syringes. Phenol concentrations were less affected than metacresol concentrations in both types of plastic syringe. However, phenol concentrations were decreased in the samples stored in plastic syringes at room temperature. None of the human insulins that were drawn into the 2 different plastic syringes had any microbial growth. |
|
| Funding sources/conflict of interest | Quote: "Supported by a grant from Eli Lilly and Company." | |
| Notes | ||
Vimalavathini 2009.
| Study characteristics | ||
| Study design | 2 human insulin formulations, regular and biphasic insulin (30/70) were purchased from the open market from 3 different manufacturers (Novo Nordisk, Eli Lily and Company, and BIOCON). Pure insulin powder which was used as reference standard was procured from Sigma‐Aldrich Chemicals, USA. Human insulin formulations were stored at 5 different temperatures based on manufacturers recommendations and the way people with diabetes were storing insulin in their homes, i.e. 5 °C (refrigerator), 25 °C (air‐conditioned room), 26 °C (mud pot), 32 °C (room temperature) and 37 °C (room temperature in summer). Samples were withdrawn from these vials every 7 days for 28 days and analysed by HPLC. Study duration was 28 days. |
|
| Measurements | HPLC analysis showed intraday and interday coefficients of variation < 3.8% and 8.7%, respectively. Potency of biphasic insulin was reported in terms of regular insulin. The concentration of insulin by the HPLC method was reported as mean. Indian pharmacopoeia recommended a deviation of up to ± 10% of the stated label claim of insulin and this was followed. Data were expressed as mean ± SEM. Insulin tolerance test in rabbits: New Zealand white rabbits of either sex weighing approximately 2 kg were used. The rabbits were divided into 5 groups with 6 or 7 animals in each group. Each group received regular human insulin of 3 different brands stored at 5 different temperatures for 25 days. The vial was opened on the 25th day. Rabbits were deprived of food but not of water for 2 hours before the start of the experiment. Insulin was injected at 0.75 U/kg bodyweight intraperitoneally. Blood sugar was obtained at 0, 15, 30 and 60 min from the ear vein using a glucometer. |
|
| Results | All 3 brands of insulin for both the formulations did not differ considerably in their insulin content during various sampling time and temperatures. There was no difference in concentration of insulin in vials stored at 25 °C and 26 °C compared to those stored at 5 °C on all 5 sampling days. There was a 14% and 18% decrease in the concentration of regular insulin in vials stored at 32 °C and 37 °C on day 28 for all the 3 brands. There was an 11% and 14% decrease in the concentration of regular insulin in biphasic insulin formulation in vials stored at 32 °C and 37 °C on day 28. Insulin injection showed a considerable decrease in blood glucose levels in rabbits with respect to basal glucose at 15, 30 and 60 min (for details see Table 7). |
|
| Funding sources/conflict of interest | Funding not mentioned. Quote: "Conflict of interest: none." |
|
| Notes | ||
1H NMR: proton nuclear magnetic resonance spectroscopy; AUC: area under the curve; CV: coefficient of variations; DLS: dynamic light scattering; FDA: Food and Drug Administration; HPLC: high‐performance liquid chromatography; HTEAT: high temperature and extreme agitation test; IQR: interquartile range; ITP: insulin transformation product; IU: insulin unit; min: minute; nm: nanometre (10−9 m); NMR: nuclear magnetic resonance; NPH: neutral protamine Hagedorn; PCA: principal component analysis; RP‐HPLC: reverse phase high‐performance liquid chromatography; SD: standard deviation; SEC: size‐exclusion chromatography; SEM; standard error of the mean; TCRT: temperature cycling and resuspension test; TEM: transmission electron microscopy; ThT: thioflavin T; U: unit; USB: universal serial bus; USP: US Pharmacopeia.
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Albergo 1992 | Opinion letter, no data |
| Allen 1982 | Not human insulin |
| Bishop 1970 | Letter, no data |
| Braune 2019 | Investigation of storage conditions of insulin in domestic refrigerators and when carried by patients |
| Casey 1983 | Viewpoint, no data |
| Davis 2010 | Description of a temperature stability database for refrigerated medications, no data |
| Delbeck 2021b | Investigation of insulin analogues |
| Dokken 2017 | Letter, no data |
| Fisch 1987 | Not human insulin |
| Gill 2000 | Viewpoint, no data |
| Gill 2002 | Temperature measurements, insulin quantity not measured |
| Gilligan 2021 | Temperature measurements, insulin quantity not measured |
| Grajower 2003 | Commentary, no data |
| Grajower 2014 | Commentary, no data |
| Grant 1946 | Not human insulin |
| Grau 1985 | In‐vitro investigation of several insulin pump devices (delivery system environment test) |
| Havlova 1966 | Not human insulin |
| Hearnshaw 2009 | Letter, no data |
| Herr 2014 | Intervention not outside recommended temperature or storage; unclear type of insulin |
| Hildebrandt 1986 | Not human insulin |
| Hildebrandt 1987 | Not human insulin |
| Kalra 2012 | Letter, no data |
| Kasmer 1986 | Not human insulin |
| Keller 2003 | General information on insulin storage, no data |
| Khurana 2019 | Review, no additional data |
| Kircher 1990 | Narrative review, no data |
| Kongmalai 2022 | Investigation of insulin analogue |
| Kristensen 2011 | Stress tests of insulin pen, not of insulin |
| Krogh 1928 | Heating experiment, destructive action of heat (buffered solution of insulin, unclear type of insulin) |
| Levine 1970 | Instruction course, no data |
| Lopez 2014 | Investigation of bubble formation in insulin pumps filled with an insulin analogue |
| Minuto 2010 | Case report |
| Mitchell 2012 | Cross‐sectional study on administration technique and storage of insulin pens reported by people with diabetes |
| Mokta 2014 | Letter, no data |
| Nassar 2010 | Cross‐sectional study on self‐management strategies used by people with diabetes in hot weather |
| Ogle 2016 | Temperature measurements, insulin quantity not measured |
| Oliva 2009 | Arrhenius plot of isothermal and non‐isothermal methods to estimate the shelf‐life of human insulin |
| Pendsey 2006 | Temperature measurements, insulin quantity not measured |
| Perriello 1988 | Short‐term pharmacokinetic/pharmacodynamic study under regular storage conditions |
| Peters 1987 | Clamp study on mixing neutral protamine Hagedorn and regular insulin |
| Pfützner 2016 | Proof‐of‐concept device test, no insulin stability data |
| Pfützner 2017 | Device test, no insulin stability data |
| Pfützner 2018a | Investigation of an insulin analogue |
| Pfützner 2018b | Investigation of an insulin analogue |
| Pfützner 2018c | Investigation of an insulin analogue |
| Pingel 1972 | Not human insulin |
| Plager 2017 | Investigation of withdrawing insulin from a vial with/without returning air |
| Pryce 2009 | Case report |
| Puepet 2007 | Survey of Nigerian patients' insulin storage at home |
| Rathod 1985 | Not human insulin |
| Richards 2013 | Narrative review, no data |
| Santiago 2002 | Investigation of an insulin pen injection device |
| Seyoum 1997 | Cross‐sectional study on how patients in Ethiopia store their insulin at home |
| Steil 1988 | Narrative review, no data |
| Stephenson 1960 | Not human insulin |
| Stephenson 2018 | Opinion letter, no data |
| Storvick 1968 | Not human insulin |
| TCTR20210611002 | Investigation of an insulin analogue |
| Thirunavukkarasu 1968 | Case series |
| Vereshchetin 2016 | Investigation of an insulin analogue and 2 different batteries for insulin pumps |
| Virmani 2020 | Viewpoint, no data |
| Westphal 2010 | Narrative review, no data |
| Yun‐Chu 2017 | Investigation of sol‐gel tailored silica to protect insulin during transport and distribution |
| Zell 1983 | Not human insulin |
| Zhao 2015 | Review, no additional data |
Differences between protocol and review
We made the following changes from the protocol (Richter 2022).
We planned to store all extracted data and risk of bias evaluations online in an open repository. Due to the limited amount of information, all relevant data are presented in this Cochrane Review.
We could not perform subgroup or sensitivity analyses due to a lack of data.
Contributions of authors
BR: protocol draft, search strategy development, acquisition of data from manufacturers, study selection, data extraction, data interpretation, review of the draft and review updates
BB: protocol draft, study selection, data extraction, data analysis, data interpretation, review of the draft and review updates
MIM: protocol draft, search strategy development, data interpretation, review of the draft and review updates
Sources of support
Internal sources
No sources of support provided
External sources
-
World Health Organization (WHO), Other
This review was funded by WHO.
Declarations of interest
BR: none.
BB: none.
MIM: none.
The authors are editors for the Cochrane Metabolic and Endocrine Disorders group, but they had no role in the editorial processing of this review.
New
References
References to studies included in this review
Al Shaibi 1999 {published data only}
- Al Shaibi K, Falata W, Sayes N, Al Shareef M, Al Taweel M, Abozenadah A, et al. Storing insulin in a clay pot in the desert causes no loss of activity: a preliminary report. Annals of Saudi Medicine 1999;19(6):547-9. [DOI] [PubMed] [Google Scholar]
- Arya SC. Insulin storage in a clay pot. Annals of Saudi Medicine 2000;20(5-6):491. [DOI: 10.5144/0256-4947.2000.491] [DOI] [PubMed] [Google Scholar]
- Tevaarwerk GJ. Reply to "Insulin storage in a clay pot". Annals of Saudi Medicine 2000;20(5-6):491-2. [DOI: 10.5144/0256-4947.2000.491a] [DOI] [PubMed] [Google Scholar]
Baechler 2020 {published data only}
- Baechler F, Stettler C, Vogt B, Bally L, Groessl M. Concentration and chemical stability of commercially available insulins: a high-resolution mass spectrometry study. Diabetes Technology & Therapeutics 2020;22(4):326-9. [DOI: 10.1089/dia.2019.0412] [DOI] [PubMed] [Google Scholar]
Delbeck 2021a {published data only}
- Delbeck S, Heise HM. Quality assurance of commercial insulin formulations: novel assay using infrared spectroscopy. Journal of Diabetes Science and Technology 2021;15(4):865-73. [DOI: 10.1177/1932296820913874] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delbeck S, Heise HM. Systematic stability testing of insulins as representative biopharmaceuticals using ATR FTIR-spectroscopy with focus on quality assurance. Journal of Biomedical Optics 2021;26(4):043001-16. [DOI: 10.1117/1.JBO.26.4.043007] [DOI] [PMC free article] [PubMed] [Google Scholar]
Dunbar 1986 {published data only}
- Dunbar JC, Simon FA. Immunologic and biologic evaluation of insulin degradation following prolonged storage in plastic syringes. Pancreas 1986;1(4):317-9. [DOI: 10.1097/00006676-198607000-00004] [DOI] [PubMed] [Google Scholar]
Gregory 1991 {published data only}
- Gregory R, Edwards S, Yateman NA. Demonstration of insulin transformation products in insulin vials by high-performance liquid chromatography. Diabetes Care 1991;14(1):42-8. [DOI: 10.2337/diacare.14.1.42] [DOI] [PubMed] [Google Scholar]
Kaufmann 2021 {published data only}
- Kaufmann B, Boulle P, Berthou F, Fournier M, Beran D, Ciglenecki I, et al. Heat-stability study of various insulin types in tropical temperature conditions: new insights towards improving diabetes care. PLOS One 2021;16:1-17. [DOI: 10.1371/journal.pone.0245372] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makoni M. Insulin storage without refrigeration. Lancet Diabetes Endocrinology 2021;9(4):202. [DOI: 10.1016/S2213-8587(21)00057-7] [DOI] [PubMed] [Google Scholar]
Koffler 1992 {published data only}
- Koffler M, Orni-Wasserlauf R, Golani N, Berger SA. Prefilled insulin syringes can be kept sterile for at least 10 days when kept under refrigeration. Journal of Diabetic Complications 1992;6(3):207-8. [DOI: 10.1016/1056-8727(92)90038-m] [DOI] [PubMed] [Google Scholar]
Kongmalai 2021 {published data only}
- Kongmalai T, Preechasuk L, Junnu S, Manocheewa S, Srisawat C, Sriwijitkamol A. The effect of temperature on the stability of in-use insulin pens. Diabetes 2018;67(Suppl 1):2. [DOI: 10.2337/db18-1008-P] [DOI] [PubMed] [Google Scholar]
- Kongmalai T, Preechasuk L, Junnu S, Manocheewa S, Srisawat C, Sriwijitkamol A. The effect of temperature on the stability of in-use insulin pens. Experimental and Clinical Endocrinology & Diabetes 2021;129(9):683-8. [DOI: 10.1055/a-1010-5466] [DOI] [PubMed] [Google Scholar]
Lull 2013 {published data only}
- Lull ME, Piacentino JJ, Traina AN. Stability of U-500 regular insulin in prefilled syringes. Journal of the American Pharmaceutical Association 2013;53(3):304-6. [DOI: 10.1331/JAPhA.2013.12164] [DOI] [PubMed] [Google Scholar]
Moses 2019 {published data only}
- Moses A, Bjerrum J, Hach M, Waehrens LH, Toft AD. Concentrations of intact insulin concurs with FDA and EMA standards when measured by HPLC in different parts of the distribution cold chain. Journal of Diabetes Science and Technology 2019;13(1):55-9. [DOI: 10.1177/1932296818783783] [DOI] [PMC free article] [PubMed] [Google Scholar]
Pendsey 2023 {published data only}
- Pendsey S, James S, Garrett TJ, Nord AB, Pendsey S, Malmodin D, et al. Insulin thermostability in a real-world setting. Lancet Diabetes Endocrinology 2023;11(5):310-2. [DOI: 10.1016/S2213-8587(23)00028-1] [DOI] [PubMed] [Google Scholar]
Schrader 1985 {published data only}
- Schrader E, Pfeiffer EF. The influence of motion and temperature upon the aggregational behavior of soluble insulin formulations investigated by high-performance liquid-chromatography. Journal of Liquid Chromatography 1985;8(6):1139-57. [DOI: 10.1080/01483918508067133] [DOI] [Google Scholar]
Shnek 1998 {published data only}
- Shnek DR, Hostettler DL, Bell MA, Olinger JM, Frank BH. Physical stress testing of insulin suspensions and solutions. Journal of Pharmaceutical Sciences 1998;87(11):1459-65. [DOI: 10.1021/js980074x] [DOI] [PubMed] [Google Scholar]
Silva 2013 {published data only}
- Silva MA, Chuong M, Kerr S, Cabrera A. Stability of two long-acting insulin formulations after 28 days. Journal of Pharmacy Practice & Research 2013;43(1):37-40. [DOI: 10.1002/j.2055-2335.2013.tb00213.x] [DOI] [Google Scholar]
Silva‐Jr 2022 {published data only}
- Silva-Jr H, Araújo TS, da Silva Almeida M, Scapin SM, Lima LM. Formation of subvisible particles in commercial insulin formulations. Colloids and Surfaces B: Biointerfaces 2022;216:112566. [DOI: 10.1016/j.colsurfb.2022.112566] [DOI] [PubMed] [Google Scholar]
Tarr 1991 {published data only}
- Tarr BD, Campbell RK, Workman TM. Stability and sterility of biosynthetic human insulin stored in plastic insulin syringes for 28 days. American Journal of Hospital Pharmacy 1991;48(12):2631-4. [PubMed] [Google Scholar]
Vimalavathini 2009 {published data only}
- Vimalavathini R, Gitanjali B. Effect of temperature on the potency & pharmacological action of insulin. Indian Journal of Medical Research 2009;130(2):166-9. [PubMed] [Google Scholar]
References to studies excluded from this review
Albergo 1992 {published data only}
- Albergo R, Bunting J, Belsky MK, Fallon JJ Jr. Insulin storage and injection recommendation. Diabetes Care 1992;15(8):1113-4. [DOI: 10.2337/diacare.15.8.1113] [DOI] [PubMed] [Google Scholar]
Allen 1982 {published data only}
- Allen SC. A cool storage pot for insulin in rural Africa. Medical Journal of Zambia 1982;16(4):83-4. [PubMed] [Google Scholar]
Bishop 1970 {published data only}
- Bishop J. Proper storage of insulin preparations. Canadian Medical Association Journal 1970;102(1):104. [PMC free article] [PubMed] [Google Scholar]
Braune 2019 {published data only}
- Braune K, Kraemer LA, Weinstein J, Zayani A, Heinemann L. Storage conditions of insulin in domestic refrigerators and when carried by patients: often outside recommended temperature range. Diabetes Technology & Therapeutics 2019;21(5):238-44. [DOI: 10.1089/dia.2019.0046] [DOI] [PubMed] [Google Scholar]
Casey 1983 {published data only}
- Casey CA. Storage of insulin stirs controversy. Rn National Magazine for Nurses 1983;46(7):73. [Google Scholar]
Davis 2010 {published data only}
- Davis SR, Anderson EA. Creation of a temperature stability database for refrigerated medications. Journal of Pharmacy Practice and Research 2010;40(1):31-5. [DOI: 10.1002/j.2055-2335.2010.tb00722.x] [DOI] [Google Scholar]
Delbeck 2021b {published data only}
- Delbeck S, Heise HM. Quality assurance of commercial insulin formulations: novel assay using infrared spectroscopy. Journal of Diabetes Science and Technology 2021;15(4):865-73. [DOI: 10.1177/1932296820913874] [DOI] [PMC free article] [PubMed] [Google Scholar]
Dokken 2017 {published data only}
- Dokken BB, Rosinko M. Reported insulin pump temperature fluctuations lack clinical relevance. Medical Devices: Evidence and Research 2017;10:43-6. [DOI: 10.2147/MDER.S128526] [DOI] [PMC free article] [PubMed] [Google Scholar]
Fisch 1987 {published data only}
- Fisch A, Leblanc H, Cisse H, Pichard E, Zirinis P. Stability of ordinary insulins under African climatic conditions. Medecine d'Afrique Noire 1987;34(12):1043-8. [Google Scholar]
Gill 2000 {published data only}
- Gill GV. Viewpoint: stability of insulin in tropical countries. Tropical Medicine & International Health 2000;5(9):666-7. [DOI: 10.1046/j.1365-3156.2000.00617.x] [DOI] [PubMed] [Google Scholar]
Gill 2002 {published data only}
- Gill G, Price C, English P, Eriksson-Lee J. Traditional clay pots as storage containers for insulin in hot climates. Tropical Doctor 2002;32(4):237-8. [DOI: 10.1177/004947550203200421] [DOI] [PubMed] [Google Scholar]
Gilligan 2021 {published data only}
- Gilligan MM, Linnes JC, Oettingen JE, Altenor K. From toy to tool: using water beads for insulin storage in Haiti. Pediatric Diabetes 2021;22(5):729-33. [DOI: 10.1111/pedi.13167] [DOI] [PubMed] [Google Scholar]
Grajower 2003 {published data only}
- Gallo M, Comoglio M, De Micheli A, Monge L, Vespasiani G. Insulin storage in Europe: a comment to Grajower et al., Eli Lilly, and Novo Nordisk. Diabetes Care 2004;27(5):1225-6. [DOI: 10.2337/diacare.27.5.1225] [DOI] [PubMed] [Google Scholar]
- Grajower MM, Fraser CG, Holcombe JH, Daugherty ML, Harris WC, De Felippis MR, et al. How long should insulin be used once a vial is started? Diabetes Care 2003;26(9):2665-6; discussion 266-9. [DOI: 10.2337/diacare.26.9.2665] [DOI] [PubMed] [Google Scholar]
- Holcombe JH, Daugherty ML, De Felippis MR. How long should insulin be used once a vial is started? Diabetes Care 2004;27(5):1241-2. [DOI: 10.2337/diacare.27.5.1241] [DOI] [PubMed] [Google Scholar]
- Molitch ME. How long should insulin be used once a vial is started? Diabetes Care 2004;27(5):1240-1; author reply 1241-2. [DOI: 10.2337/diacare.27.5.1240-a] [DOI] [PubMed] [Google Scholar]
Grajower 2014 {published data only}
- Flood TM. In response: how long should insulin be used once a vial is opened? Endocrine Practice 2015;21(2):213. [PubMed] [Google Scholar]
- Grajower MM. How long can a vial of insulin be used after it is started: where are we 10 years later? Endocrine Practice 2014;20(2):188-90. [DOI: 10.4158/EP13427.CO] [DOI] [PubMed] [Google Scholar]
Grant 1946 {published data only}
- Grant RL. Loss of potency of commercial insulin stored at room temperature. Federation Proceedings 1946;5(1 Pt 2):181. [PubMed] [Google Scholar]
Grau 1985 {published data only}
- Grau U. Chemical stability of insulin in a delivery system environment. Diabetologia 1985;28(7):458-63. [DOI] [PubMed] [Google Scholar]
Havlova 1966 {published data only}
- Havlova M, Nobilis M. Undesirable factors influencing the stability of insulin preparations [NEŽÁDOUCÍ VLIVY PŮSOBÍCÍ NA STABILITU INSULINOVÝCH PREPARÁTŮ]. Ceskoslovenska Farmacie 1966;15(9):504-5. [PubMed] [Google Scholar]
Hearnshaw 2009 {published data only}
- Hearnshaw HM. Insulin pumps exposed to heat. Put an indicator on the bottle. BMJ 2009;338:b2009. [DOI: 10.1136/bmj.b2009] [DOI] [PubMed] [Google Scholar]
Herr 2014 {published data only}
- Herr JK, Keith S, Klug R, Pettis RJ. Characterizing normal-use temperature conditions of pumped insulin. Journal of Diabetes Science and Technology 2014;8(4):850-4. [DOI: 10.1177/1932296814532327] [DOI] [PMC free article] [PubMed] [Google Scholar]
Hildebrandt 1986 {published data only}
- Hildebrandt R, Jannasch R, Lotz U. On the stability of insulin solutions in syringes. Zeitschrift für Klinische Medizin 1986;41(1):51-3. [Google Scholar]
Hildebrandt 1987 {published data only}
- Hildebrandt R, Rafalski I. Stability and sterility of neutral insulin after opening. Zeitschrift für Klinische Medizin 1987;42(14):1279-81. [Google Scholar]
Kalra 2012 {published data only}
- Kalra S, Kalra B. Storage of insulin in rural areas. Journal of Academy of Medical Sciences 2012;2(2):88-9. [DOI: 10.4103/2249-4855.118669] [DOI] [Google Scholar]
Kasmer 1986 {published data only}
- Kasmer RJ, Walker N, Hurlburt F, Rotter G, Shlaes DM, Hostetler C. Sterility of preloaded insulin syringes. American Journal of Infectious Diseases 1986;14(4):180-3. [DOI: 10.1016/0196-6553(86)90098-2] [DOI] [PubMed] [Google Scholar]
Keller 2003 {published data only}
- Keller KJ. Insulin storage and stability update. South Dakota Journal of Medicine 2003;56(8):303-4. [PubMed] [Google Scholar]
Khurana 2019 {published data only}
- Khurana G, Gupta VK. Effect on insulin upon storage in extreme climatic conditions (temperature and pressure) and their preventive measures. Journal of Social Health and Diabetes 2019;7(1):6-10. [DOI: 10.1055/s-0039-1692371] [DOI] [Google Scholar]
Kircher 1990 {published data only}
- Kircher W. Drug information. Instructions for storage and administration. Insulin preparations [Arzneimittelanwendung. Aufbewahrungs- und Abgabehinweise. Insulinpräparate]. Medizinische Monatsschrift für Pharmazeuten 1990;13(10):318-20. [PubMed] [Google Scholar]
Kongmalai 2022 {published data only}
- Kongmalai T, Orarachin P, Dechates B, Chanphibun P, Junnu S, Srisawat C, et al. The effect of high temperature on the stability of basal insulin in a pen: a randomized controlled, crossover, equivalence trial. BMJ Open Diabetes Research & Care 2022;10(6):1-8. [DOI: 10.1136/bmjdrc-2022003105] [DOI] [PMC free article] [PubMed] [Google Scholar]
Kristensen 2011 {published data only}
- Kristensen CM, Lilleore SK. Dose accuracy and durability of a durable insulin pen before and after simulated lifetime use. Current Medical Research and Opinion 2011;27(10):1877-83. [DOI: 10.1185/03007995.2011.609885] [DOI] [PubMed] [Google Scholar]
Krogh 1928 {published data only}
- Krogh A, Hemmingsen AM. The destructive action of heat on insulin solutions. Biochemical Journal 1928;22(5):1231-8. [DOI: 10.1042/bj0221231] [DOI] [PMC free article] [PubMed] [Google Scholar]
Levine 1970 {published data only}
- Levine R, Mahler R, Moss JM. The management of diabetes. 7. Characteristics of the available insulins. Problems in insulin storage. American Family Physician/GP 1970;2(5):151-63. [PubMed] [Google Scholar]
Lopez 2014 {published data only}
- Lopez PE, King BR, Goss PW, Chockalingam G. Bubble formation occurs in insulin pumps in response to changes in ambient temperature and atmospheric pressure but not as a result of vibration. BMJ Open Diabetes Research and Care 2014;2(1):1-4. [DOI: 10.1136/bmjdrc-2014-000036] [DOI] [PMC free article] [PubMed] [Google Scholar]
Minuto 2010 {published data only}
- Minuto N, Tambroni B, Vannati M, Emmanuele V, Russo C, Lorini R, et al. Diabetic ketoacidosis caused by exposure of insulin to low temperature. Diabetes Technology & Therapeutics 2010;12(9):745-6. [DOI: 10.1089/dia.2010.0055] [DOI] [PubMed] [Google Scholar]
Mitchell 2012 {published data only}
- Mitchell VD, Porter K, Beatty SJ. Administration technique and storage of disposable insulin pens reported by patients with diabetes. Diabetes Educator 2012;38(5):651-8. [DOI: 10.1177/0145721712450921] [DOI] [PubMed] [Google Scholar]
Mokta 2014 {published data only}
- Mokta JK, Kalra S. Insulin storage in the Upper Himalayas. Rural and Remote Health 2014;14(2983):1-2. [PubMed] [Google Scholar]
Nassar 2010 {published data only}
- Nassar AA, Childs RD, Boyle ME, Jameson KA, Fowke M, Waters KR, et al. Diabetes in the desert: what do patients know about the heat? Journal of Diabetes Science and Technology 2010;4(5):1156-63. [DOI: 10.1177/193229681000400514] [DOI] [PMC free article] [PubMed] [Google Scholar]
Ogle 2016 {published data only}
- Ogle GD, Abdullah M, Mason D, Januszewski AS, Besancon S. Insulin storage in hot climates without refrigeration: temperature reduction efficacy of clay pots and other techniques. Diabetic Medicine 2016;33(11):1544-53. [DOI: 10.1111/dme.13194] [DOI] [PubMed] [Google Scholar]
Oliva 2009 {published data only}
- Oliva A, Suarez M, Hernandez JR, Llabres M, Farina JB. Evaluation of non-isothermal methods in stability studies of human insulin pharmaceutical preparations. Journal of Pharmaceutical and Biomedical Analysis 2009;49(4):916-22. [DOI: 10.1016/j.jpba.2009.01.024] [DOI] [PubMed] [Google Scholar]
Pendsey 2006 {published data only}
- Pendsey S. Keeping insulin cool naturally – the DREAM Trust storage system. Diabetes Voice 2006;51(Special Issue):19. [Google Scholar]
Perriello 1988 {published data only}
- Perriello G, Torlone E, Di Santo S, Fanelli C, De Feo P, Santeusanio F, et al. Effect of storage temperature of insulin on pharmacokinetics and pharmacodynamics of insulin mixtures injected subcutaneously in subjects with type 1 (insulin-dependent) diabetes mellitus. Diabetologia 1988;31(11):811-5. [DOI: 10.1007/BF00277482] [DOI] [PubMed] [Google Scholar]
Peters 1987 {published data only}
- Peters AL, Davidson MB. Effect of storage on action of NPH and regular insulin mixtures. Diabetes Care 1987;10(6):799-800. [DOI] [PubMed] [Google Scholar]
Pfützner 2016 {published data only}
- Pfützner A, Nagar R. Technology solutions to Increase insulin stability in case of extreme weather conditions. Diabetes Technology & Therapeutics 2016;18:A122-3. [Google Scholar]
Pfützner 2017 {published data only}
- Pfützner A, Pesach G, Nagar R. Technology-derived storage solutions for stabilizing insulin in extreme weather conditions I: the ViViCap-1 device. Expert Opinion on Drug Delivery 2017;14(6):709-14. [DOI: 10.1080/17425247.2017.1316261] [DOI] [PubMed] [Google Scholar]
Pfützner 2018a {published data only}
- Pfützner A, Reeh W, Nagar R, Rose D. The Vivi-Cap stabilizes insulin in extreme temperature conditions. Diabetes Technology & Therapeutics 2018;20:A140-1. [Google Scholar]
Pfützner 2018b {published data only}
- Pfützner A, Reeh W, Strobl S, Rose D. ViViCap – reliable, reusable protection against insulin degradation in high and low ambient temperatures. Diabetes Stoffwechsel und Herz 2018;27(6):307-11. [Google Scholar]
Pfützner 2018c {published data only}
- Pfützner A, Nagar R, Reeh W. A reusable cap for disposable insulin pens protects insulin against temperature-induced degradation. Diabetes 2018;67(Suppl 1):2. [DOI: 10.2337/db18-2281-PUB] [DOI] [Google Scholar]
Pingel 1972 {published data only}
- Pingel M, Volund A. Stability of insulin preparations. Diabetes 1972;21(7):805-13. [DOI: 10.2337/diab.21.7.805] [DOI] [PubMed] [Google Scholar]
Plager 2017 {published data only}
- Plager P, Nurie K, Omann T, Moran A, Piloya T, Bahendeka S, et al. Is insulin diluted when stored in water? Pediatric Diabetes 2017;18(3):237-40. [DOI: 10.1111/pedi.12367] [DOI] [PubMed] [Google Scholar]
Pryce 2009 {published data only}
- Pryce R. Diabetic ketoacidosis caused by exposure of insulin pump to heat and sunlight. BMJ 2009;338:a2218. [DOI: 10.1136/bmj.a2218] [DOI] [PubMed] [Google Scholar]
Puepet 2007 {published data only}
- Puepet FH, Mijinyawa BB, Akogu IA. Insulin storage by patients with diabetes in Jos, Nigeria. Journal of Medical Tropics 2007;9:37-40. [Google Scholar]
Rathod 1985 {published data only}
- Rathod M, Saravolatz L, Pohlod D, Whitehouse F, Goldman J. Evaluation of the sterility and stability of insulin from multidose vials used for prolonged periods. Infection Control 1985;6(12):491-4. [DOI: 10.1017/s019594170006361x] [DOI] [PubMed] [Google Scholar]
Richards 2013 {published data only}
- Richards P, Hillebrandt D. The practical aspects of insulin at high altitude. High Altitude Medicine & Biology 2013;14(3):197-204. [DOI: 10.1089/ham.2013.1020] [DOI] [PubMed] [Google Scholar]
Santiago 2002 {published data only}
- Santiago OM, Khutoryansky NM, Bilbo CM, Lawton SA, Kristensen CM. Accuracy and precision of the NovoPen 3 insulin delivery device after mechanical and temperature stresses. Endocrine Practice 2002;8(5):351-5. [DOI: 10.4158/EP.8.5.351] [DOI] [PubMed] [Google Scholar]
Seyoum 1997 {published data only}
- Seyoum B, Abdulkadir J, Feleke Y, Worku Y. Insulin storage by diabetic patients in Ethiopia. Diabetologia 1997;40:2568. [Google Scholar]
Steil 1988 {published data only}
- Steil CF. Insulin storage. Diabetes Educator 1988;14(6):564-5. [DOI: 10.1177/014572178801400624] [DOI] [PubMed] [Google Scholar]
Stephenson 1960 {published data only}
- Stephenson NR, Romans RG. Thermal stability of insulin made from zinc insulin crystals. Journal of Pharmacy and Pharmacology 1960;12:372-6. [DOI: 10.1111/j.2042-7158.1960.tb12677.x] [DOI] [PubMed] [Google Scholar]
Stephenson 2018 {published data only}
- Stephenson J. Domestic refrigerators 'may pose risk to insulin quality'. Nursing Times 2018;114(10):46. [Google Scholar]
Storvick 1968 {published data only}
- Storvick WO, Henry HJ. Effect of storage temperature on stability of commercial insulin preparations. Diabetes 1968;17(8):499-502. [DOI: 10.2337/diab.17.8.499] [DOI] [PubMed] [Google Scholar]
TCTR20210611002 {published data only}
- TCTR20210611002. Efficacy of in-used insulin pen kept at 37 Celsius for 21 days on mean daily glucose measured by continuous glucose monitoring compare with the effect of newly used insulin pen, the equivalence trial. www.who.int/trialsearch/Trial2.aspx?TrialID=TCTR20210611002 (accessed 3 January 2022).
Thirunavukkarasu 1968 {published data only}
- Thirunavukkarasu K. Soluble insulin in the tropics. Ceylon Medical Journal 1968;13(3):150-3. [PubMed] [Google Scholar]
Vereshchetin 2016 {published data only}
- Vereshchetin P, McCann TW Jr, Ojha N, Venugopalan R, Levy BL. Comparison of rechargeable versus battery-operated insulin pumps: temperature fluctuations. Medical Devices: Evidence and Research 2016;9:371-6. [DOI: 10.2147/MDER.S116666] [DOI] [PMC free article] [PubMed] [Google Scholar]
Virmani 2020 {published data only}
- Virmani A, Avni TC. A case for expanding thermochromic vial monitor technology to insulin and other biologics. Indian Pediatrics 2020;57(1):17-9. [PubMed] [Google Scholar]
Westphal 2010 {published data only}
- Westphal SA, Childs RD, Seifert KM, Boyle ME, Fowke M, Iniguez P, et al. Managing diabetes in the heat: potential issues and concerns. Endocrine Practice 2010;16(3):506-11. [DOI: 10.4158/EP09344.RA] [DOI] [PubMed] [Google Scholar]
Yun‐Chu 2017 {published data only}
- Yun-Chu C, Francoise K, Karen EJ, Asel S. Thermal stability, storage, release and delivery of insulin – a protection through sol-gel tailored silica. European Biophysics Journal 2017;46(Suppl 1):S320. [Google Scholar]
Zell 1983 {published data only}
- Zell M, Paone RP. Stability of insulin in plastic syringes. American Journal of Hospital Pharmacy 1983;40(4):637-8. [PubMed] [Google Scholar]
Zhao 2015 {published data only}
- Zhao Xiaolei, Zheng Silin. Research progress on period of validity and storage conditions of opened insulin [胰岛素开启后有效期和贮藏条件的研究进展]. Chinese Nursing Research 2015;29(11A):3848-50. [DOI: 10.3969/j.issn.1009-6493.2015.31.003] [DOI] [Google Scholar]
Additional references
ADA 2003
- Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care 2003;26(Suppl 1):S5-20. [DOI] [PubMed] [Google Scholar]
ADA 2017
- American Diabetes Association. Standards of medical care in diabetes – 2017. Diabetes Care 2017;40(Suppl 1):S11-33. [Google Scholar]
ADA 2021
- American Diabetes Association. Insulin storage and syringe safety. www.diabetes.org/healthy-living/medication-treatments/insulin-other-injectables/insulin-storage-and-syringe-safety (accessed 15 October 2021).
Ahmad 2003
- Ahmad A, Millett IS, Doniach S, Uversky VN, Fink AL. Partially folded intermediates in insulin fibrillation. Biochemistry 2003;42(39):11404-16. [DOI: 10.1016/j.actbio.2013.03.002] [DOI] [PubMed] [Google Scholar]
Arora 2004
- Arora A, Ha C, Park CB. Insulin amyloid fibrillation at above 100 degrees C: new insights into protein folding under extreme temperatures. Protein Science 2004;13(9):2429-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
Atwoli 2021
- Atwoli L, Baqui AH, Benfield T, Bosurgi R, Godlee F, Hancocks S, et al. Call for emergency action to limit global temperature Increases, restore biodiversity, and protect health. New England Journal of Medicine 2021;385(12):1134-7. [DOI: 10.1056/NEJMe2113200] [DOI] [PubMed] [Google Scholar]
Bahendeka 2019
- Bahendeka S, Kaushik R, Swai AB, Otieno F, Bajaj S, Kalra S, et al. EADSG guidelines: insulin storage and optimisation of injection technique in diabetes management. Diabetes Therapy 2019;10(2):341-66. [DOI: 10.1007/s13300-019-0574-x] [DOI] [PMC free article] [PubMed] [Google Scholar]
Basu 2019
- Basu S, Yudkin JS, Kehlenbrink S, Davies JI, Wild SH, Lipska KJ, et al. Estimation of global insulin use for type 2 diabetes, 2018-30: a microsimulation analysis. Lancet Diabetes Endocrinology 2019;7(1):25-33. [DOI: 10.1016/S2213-8587(18)30303-6] [DOI] [PubMed] [Google Scholar]
Beran 2006
- Beran D, Yudkin JS. Diabetes care in sub-Saharan Africa. Lancet 2006;368(9548):1689-95. [DOI: 10.1016/S0140-6736(06)69704-3] [DOI] [PubMed] [Google Scholar]
Beran 2016
- Beran D, Ewen M, Laing R. Constraints and challenges in access to insulin: a global perspective. Lancet Diabetes Endocrinology 2016;4(3):275-85. [DOI: 10.1016/S2213-8587(15)00521-5] [DOI] [PubMed] [Google Scholar]
Brange 1987
- Brange J. Galenics of Insulin. Heidelberg: Springer-Verlag, 1987. [Google Scholar]
Brange 1997
- Brange J, Andersen L, Laursen ED, Meyn G, Rasmussen E. Toward understanding insulin fibrillation. Journal of Pharmaceutical Sciences 1997;86(5):517-25. [DOI: 10.1021/js960297s] [DOI] [PubMed] [Google Scholar]
Burki 2022
- Burki T. Hot topics on insulin access: the issue of thermostability. Lancet. Diabetes & Endocrinology 2022;10(6):389-90. [DOI: 10.1016/ S2213-8587(22)00140-1] [DOI] [PubMed] [Google Scholar]
Calo 2016
- Calo N, Ramadori P, Sobolewski C, Romero Y, Maeder C, Fournier M, et al. Stress-activated miR-21/miR-21* in hepatocytes promotes lipid and glucose metabolic disorders associated with high-fat diet consumption. Gut 2016;65(11):1871-81. [DOI: 10.1136/gutjnl-2015-310822] [DOI] [PubMed] [Google Scholar]
Chance 1993
- Chance RE, Frank BH. Research, development, production, and safety of biosynthetic human insulin. Diabetes Care 1993;16(Suppl 3):133-42. [DOI: 10.2337/diacare.16.3.133] [DOI] [PubMed] [Google Scholar]
Danne 2018
- Danne T, Phillip M, Buckingham BA, Jarosz-Chobot P, Saboo B, Urakami T, et al. ISPAD clinical practice consensus guidelines 2018: insulin treatment in children and adolescents with diabetes. Pediatric Diabetes 2018;19(Suppl 27):115-35. [DOI: 10.1111/pedi.12718] [DOI] [PubMed] [Google Scholar]
Delbeck 2020a
- Delbeck S, Krolinski A, Dederich Y, Stoppelkamp S, Heise HM. A novel approach for the assessment of insulin potency by glycemic monitoring of an insulin-dependent human cell line. Journal of Diabetes Science and Technology 2020;14(3):691-2. [DOI: 10.1177/1932296819895541] [DOI] [PMC free article] [PubMed] [Google Scholar]
Delbeck 2020b
- Delbeck S, Heise HM. FT-IR versus EC-QCL spectroscopy for biopharmaceutical quality assessment with focus on insulin-total protein assay and secondary structure analysis using attenuated total reflection. Analytical and Bioanalytical Chemistry 2020;412(19):4647-58. [DOI: 10.1007/s00216-020-02718-1] [DOI] [PMC free article] [PubMed] [Google Scholar]
Derewenda 1989
- Derewenda U, Derewenda Z, Dodson GG, Hubbard RE, Korber F. Molecular structure of insulin: the insulin monomer and its assembly. British Medical Bulletin 1989;45(1):4-18. [DOI: 10.1093/oxfordjournals.bmb.a072320] [DOI] [PubMed] [Google Scholar]
Devi 2021
- Devi S. Cold comfort: getting insulin to those who need it. Lancet 2021;398(10313):1791. [DOI] [PubMed] [Google Scholar]
Ebi 2021
- Ebi KL, Capon A, Berry P, Broderick C, Dear R, Havenith G, et al. Hot weather and heat extremes: health risks. Lancet 2021;398(10301):698-708. [DOI: 10.1016/S0140-6736(21)01208-3] [DOI] [PubMed] [Google Scholar]
European Pharmacopoeia 2023
- European Pharmacopoeia (Ph. Eur), 11th Edition. Strasbourg: European Pharmacopoeia Commission, European Directorate for the Quality of Medicines (EDQM); 2023.
Farid 1989
- Farid NA, Atkins LM, Becker GW, Dinner A, Heiney RE, Miner DJ, et al. Liquid chromatographic control of the identity, purity and "potency" of biomolecules used as drugs. Journal of Pharmaceutical and Biomedical Analysis 1989;7(2):185-8. [DOI: 10.1016/0731-7085(89)80082-2] [DOI] [PubMed] [Google Scholar]
Fisher 1986
- Fisher BV, Smith D. HPLC as a replacement for the animal response assays for insulin. Journal of Pharmaceutical and Biomedical Analysis 1986;4(3):377-87. [DOI: 10.1016/0731-7085(86)80059-0] [DOI] [PubMed] [Google Scholar]
Flemyng 2023
- Flemyng E, Moore TH, Boutron I, Higgins JP, Hróbjartsson A, Nejstgaard CH, et al. Using Risk of Bias 2 to assess results from randomised controlled trials: guidance from Cochrane. BMJ Evidence-Based Medicine 2023;28:260-6. [DOI: 10.1136/bmjebm-2022-112102] [DOI] [PubMed] [Google Scholar]
Flood 2015
- Flood TM. In response: how long should insulin be used once a vial is opened? Endocrine Practice 2015;21(2):213. [PubMed] [Google Scholar]
Fullerton 2018
- Fullerton B, Siebenhofer A, Jeitler K, Horvath K, Semlitsch T, Berghold A, et al. Short-acting insulin analogues versus regular human insulin for adult, non-pregnant persons with type 2 diabetes mellitus. Cochrane Database of Systematic Reviews 2018, Issue 12. Art. No: CD013228. [DOI: 10.1002/14651858.CD013228] [DOI] [PMC free article] [PubMed] [Google Scholar]
GRADEpro GDT [Computer program]
- GRADEpro GDT. Version accessed 29 June 2023. Hamilton (ON): McMaster University (developed by Evidence Prime). Available at gradepro.org.
Grajower 2003
- Grajower MM, Fraser CG, Holcombe JH, Daugherty ML, Harris WC, De-Felippis MR, et al. How long should insulin be used once a vial is started? Diabetes Care 2003;26(9):2665-9. [DOI: 10.2337/diacare.26.9.2665] [DOI] [PubMed] [Google Scholar]
Grajower 2014
- Grajower M. How long can a vial of insulin be used after it is started: where are we 10 years later? Endocrine Practice 2014;20(2):188-90. [DOI: 10.4158/EP13427.CO] [DOI] [PubMed] [Google Scholar]
Groenning 2009
- Groenning M, Frokjaer S, Vestergaard B. Formation mechanism of insulin fibrils and structural aspects of the insulin fibrillation process. Current Protein & Peptide Science 2009;10(5):509-28. [DOI: 10.2174/138920309789352038] [DOI] [PubMed] [Google Scholar]
Heinemann 2021
- Heinemann L, Braune K, Carter A, Zayani A, Krämer LA. Insulin storage: a critical reappraisal. Journal of Diabetes Science and Technology 2021;15(1):147-59. [DOI: 10.1177/1932296819900258] [DOI] [PMC free article] [PubMed] [Google Scholar]
Hemmingsen 2021
- Hemmingsen B, Metzendorf MI, Richter B. (Ultra-)long-acting insulin analogues for people with type 1 diabetes mellitus. Cochrane Database of Systematic Reviews 2021, Issue 3. Art. No: CD013498. [DOI: 10.1002/14651858.CD013498.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Herr 2014
- Herr JK, Keith S, Klug R, Pettis RJ. Characterizing normal-use temperature conditions of pumped insulin. Journal of Diabetes Science and Technology 2014;8(4):850-4. [DOI: 10.1177/1932296814532327] [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2022a
- Higgins JP, Savović J, Page MJ, Elbers RG, Sterne JA. Chapter 8: Assessing risk of bias in a randomized trial. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.3 (updated February 2022). Cochrane, 2022. Available from training.cochrane.org/handbook/archive/v6.3.
Higgins 2022b
- Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.3 (updated February 2022). Cochrane, 2022. Available from training.cochrane.org/handbook/archive/v6.3.
Huus 2005
- Huus K, Havelund S, Olsen HB, de Weert M, Frokjaer S. Thermal dissociation and unfolding of insulin. Biochemistry 2005;44(33):11171-7. [DOI: 10.1021/bi0507940] [DOI] [PubMed] [Google Scholar]
Huus 2006
- Huus K, Havelund S, Olsen HB, de Weert M, Frokjaer S. Chemical and thermal stability of insulin: effects of zinc and ligand binding to the insulin zinc-hexamer. Pharmaceutical Research 2006;23(11):2611-20. [DOI: 10.1007/s11095-006-9098-y] [DOI] [PubMed] [Google Scholar]
IDF 2021
- International Diabetes Federation. IDF Diabetes Atlas 2021 (IDF Atlas 10th edition). diabetesatlas.org/en/resources (accessed 18 December 2021).
Kaufmann 2021
- Kaufmann B, Boulle P, Berthou F, Fournier M, Beran D, Ciglenecki I, et al. Heat-stability study of various insulin types in tropical temperature conditions: new insights towards improving diabetes care. PLOS One 2021;16(2):e0245372. [DOI: 10.1371/journal.pone.0245372] [DOI] [PMC free article] [PubMed] [Google Scholar]
Khurana 2019
- Khurana G, Gupta VK. Effect on insulin upon storage in extreme climatic conditions (temperature and pressure) and their preventive measures. Journal of Social Health and Diabetes 2019;7(1):6-10. [DOI: 10.1055/s-0039-1692371] [DOI] [Google Scholar]
Kurouski 2012
- Kurouski D, Washington J, Ozbil M, Prabhakar R, Shekhtman A, Lednev IK. Disulfide bridges remain intact while native insulin converts into amyloid fibrils. PLOS One 2012;7(6):e36989. [DOI: 10.1371/journal.pone.0036989] [DOI] [PMC free article] [PubMed] [Google Scholar]
Lilly 2023
- Humulin R – insulin human injection, solution. uspl.lilly.com/humulinru100/humulinru100.html#ug0 (assessed 25 June 2023).
Mori 2021
- Mori W, Kaneko N, Nakanishi A, Zako T, Masumoto J. Insulin amyloid fibrils interact directly with the NLRP3, resulting in inflammasome activation and pyroptotic cell death. International Journal of Immunopathology and Pharmacology 2021;35:1-7. [DOI: 10.1177/20587384211038357] [DOI] [PMC free article] [PubMed] [Google Scholar]
Novo Nordisk 2020a
- Insulatard: EPAR – product information. www.ema.europa.eu/documents/product-information/insulatard-epar-product-information_en.pdf (assessed 29 June 2023).
Novo Nordisk 2020b
- Actrapid: EPAR – product information. www.ema.europa.eu/documents/product-information/actrapid-epar-product-information_en.pdf (assessed 29 June 2023).
Ogle 2016
- Ogle GD, Abdullah M, Mason D, Januszewski AS, Besançon S. Insulin storage in hot climates without refrigeration: temperature reduction efficacy of clay pots and other techniques. Diabetic Medicine 2016;33(11):1544-53. [DOI: 10.1111/dme.13194] [DOI] [PubMed] [Google Scholar]
Ohno 2019
- Ohno Y, Seki T, Kojima Y, Miki R, Egawa Y, Hosoya O, et al. Investigation of factors that cause insulin precipitation and/or amyloid formation in insulin formulations. Journal of Pharmaceutical Health Care and Sciences 2019;5:22. [DOI: 10.1186/s40780-019-0151-5] [DOI] [PMC free article] [PubMed] [Google Scholar]
Page 2021
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. [DOI: 10.1136/bmj.n71] [DOI] [PMC free article] [PubMed] [Google Scholar]
Pande 2022
- Pande AR, Thakur AK. Perceptions of alternative methods of insulin storage: an ethical dilemma. Lancet. Diabetes & Endocrinology 2022;10(8):558. [DOI] [PubMed] [Google Scholar]
Pingel 1972
- Pingel M, Volund A. Stability of insulin preparations. Diabetes 1972;21(7):805-13. [DOI: 10.2337/diab.21.7.805] [DOI] [PubMed] [Google Scholar]
Pryce 2009
- Pryce R. Diabetic ketoacidosis caused by exposure of insulin pump to heat and sunlight. BMJ 2009;338:a2218. [DOI: 10.1136/bmj.a2218] [DOI] [PubMed] [Google Scholar]
Quijal‐Zamorano 2021
- Quijal-Zamorano M, Martinez-Solanas E, Achebak H, Petrova D, Robine JM, Herrmann FR, et al. Seasonality reversal of temperature attributable mortality projections due to previously unobserved extreme heat in Europe. Lancet Planet Health 2021;5(9):e573. [DOI: 10.1016/S2542-5196(21)00211-4] [DOI] [PubMed] [Google Scholar]
Sanofi 2023a
- Editor. Sanofi to discontinue Insuman® insulins over upcoming months. diabetestimes.co.uk/sanofi-to-discontinue-insuman-insulins-over-upcoming-months (assessed 11 June 2023).
Sanofi 2023b
- Information letter from Sanofi-Aventis Deutschland GmbH on the worldwide discontinuation of the production of the medicinal products Insuman basal, Insuman rapid and Insuman comb25 [Informationsschreiben der Sanofi-Aventis Deutschland GmbH zur weltweiten Einstellung der Produktion der Arzneimittel INSUMAN BASAL, INSUMAN RAPID und INSUMAN COMB25]. www.bfarm.de/SharedDocs/Arzneimittelzulassung/Lieferengpaesse/DE/2023/info_insulin_human_20230525.html?nn=471334 (assessed 29 June 2023).
Schünemann 2022
- Schünemann HJ, Higgins JP, Vist GE, Glasziou P, Akl EA, Skoetz N, et al. Chapter 14: Completing 'Summary of findings' tables and grading the certainty of the evidence. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s), Cochrane Handbook for Systematic Reviews of Interventions Version 6.3 (updated February 2022). Cochrane, 2022. Available from training.cochrane.org/handbook/archive/v6.3.
Semlitsch 2020
- Semlitsch T, Engler J, Siebenhofer A, Jeitler K, Berghold A, Horvath K. (Ultra-)long-acting insulin analogues versus NPH insulin (human isophane insulin) for adults with type 2 diabetes mellitus. Cochrane Database of Systematic Reviews 2020, Issue 11. Art. No: CD005613. [DOI: 10.1002/14651858.CD005613.pub4] [DOI] [PMC free article] [PubMed] [Google Scholar]
Shnek 1998
- Shnek DR, Hostettler DL, Bell MA, Olinger JM, Frank BH. Physical stress testing of insulin suspensions and solutions. Journal of Pharmaceutical Sciences 1998;87(11):1459-65. [DOI: 10.1021/js980074x] [DOI] [PubMed] [Google Scholar]
Smith 1985
- Smith HW Jr, Atkins LM, Binkley DA, Richardson WG, Miner DJ. A universal HPLC determination of insulin potency. Journal of Liquid Chromatography 1985;8(3):419-39. [Google Scholar]
Sterne 2016
- Sterne JA, Hernan MA, Reeves BC, Savovic J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016;355:i4919. [DOI: 10.1136/bmj.i4919] [DOI] [PMC free article] [PubMed] [Google Scholar]
Storvick 1968
- Storvick WO, Henry HJ. Effect of storage temperature on stability of commercial insulin preparations. Diabetes 1968;17(8):499-502. [DOI: 10.2337/diab.17.8.499] [DOI] [PubMed] [Google Scholar]
Sun 2022
- Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, et al. IDF Diabetes Atlas: global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Research and Clinical Practice 2022;183:109119. [DOI: 10.1016/j.diabres.2021.109119] [DOI] [PMC free article] [PubMed] [Google Scholar]
Taerahkun 2022
- Taerahkun S, Sriphrapradang C. Efficacy of alternative cooling devices used for insulin storage without refrigeration under hot-humid environment. Annals of Medicine 2022;54(1):1118-25. [DOI: 10.1080/07853890.2022.2067355] [DOI] [PMC free article] [PubMed] [Google Scholar]
USP 2016
- US Pharmacopeia. Insulin Human Drug Products 100 IU/ml. Rockville, MD: United States Pharmacopeial Convention; 2016.
Vimalavathini 2009
- Vimalavathini R, Gitanjali B. Effect of temperature on the potency & pharmacological action of insulin. Indian Journal of Medical Research 2009;130(2):166-9. [PubMed] [Google Scholar]
Weiss 2013
- Weiss M. Design of ultra-stable insulin analogues for the developing world. Journal of Health Specialities 2013;1(2):59-70. [Google Scholar]
Westphal 2010
- Westphal SA, Childs RD, Seifert KM, Boyle ME, Fowke M, Iniguez P, et al. Managing diabetes in the heat: potential issues and concerns. Endocrine Practice 2010;16(3):506-11. [DOI: 10.4158/EP09344.RA] [DOI] [PubMed] [Google Scholar]
WHO 1999
- World Health Organization. Definition, diagnosis and classification of diabetes mellitus and its complications. In: Report of a WHO Consultation. Part 1: Diagnosis and Classification of Diabetes Mellitus. Geneva (Switzerland): World Health Organization, 1999:1-59. [Google Scholar]
WHO 2011
- WHO Technical Report Series, No 961. Model guidance for the storage and transport of time- and temperature-sensitive pharmaceutical products. www.who.int/medicines/areas/quality_safety/quality_assurance/ModelGuidanceForStorageTransportTRS961Annex9.pdf?ua=1 (accessed 15 October 2021).
WHO 2020
- World Health Organization. Diagnosis and management of type 2 diabetes. apps.who.int/iris/rest/bitstreams/1274478/retrieve (accessed 11 August 2023).
WHO EML 2021
- World Health Organization. World Health Organization Model List of Essential Medicines – 22nd List (2021). apps.who.int/iris/rest/bitstreams/1374779/retrieve (accessed 15 October 2021).
References to other published versions of this review
Richter 2022
- Richter B, Bongaerts B, Metzendorf M-I. Thermal stability and storage of human insulin. Cochrane Database of Systematic Reviews 2022, Issue 1. Art. No: CD015385. [DOI: 10.1002/14651858.CD015385] [DOI] [PMC free article] [PubMed] [Google Scholar]


