Abstract
Four marine bacteria, Alteromonas sp. strains A27, A28, A29, and A30, that lyse the diatom Skeletonema costatum NIES-324 were isolated from coastal seawater samples. They were also able to lyse the diatoms Thalassiosira sp. and Eucampia zodiacs and the raphidophycean flagellate Chattonella antiqua. Cryptic indigenous plasmids, designated pAS28 and pAS29, were detected in Alteromonas sp. strains A28 and A29, respectively. These plasmids appeared to be similar based on size and restriction site analysis. A shuttle vector that replicates in Escherichia coli and Alteromonas sp. strain A28 was constructed by fusing pAS28 and E. coli vector pCRIIc. The 16-kbp chimeric plasmid, designated pASS1, had the ability to transform strain A28 at a frequency of 106 transformants per μg of DNA. Deletion analysis of pASS1 showed that the 4.7-kb EcoRI-HindIII region of pAS28 was essential for plasmid maintenance in strain A28. This EcoRI-HindIII fragment contained an open reading frame which appeared to encode a 708-amino-acid protein.
Harmful algal blooms cause severe damage to aquacultured fishes and shellfishes (7) and are also responsible for killing other marine organisms (17). The complex interactions among algae within blooms have been discussed by previous workers (8, 11). Bacterial species which are capable of lysing harmful algae have been found in coastal environments where harmful algal blooms have often occurred (3, 5–7, 9, 12). These include Alteromonas sp. (7), Flavobacterium sp. (3), Cytophaga sp. (5, 6, 9, 12), and Saprospira sp. (12). Although the mechanism of algal bloom outbreaks is not clearly understood, the alga-lytic activities of bacteria may be a useful factor for controlling excess growth of marine algae in nature (1, 7).
Algal lysis by bacteria may be brought about by the production of extracellular products (3, 7) or cell-to-cell contact mechanisms (5, 6, 9, 12). However, virtually nothing is known about the mechanisms of algal cell lysis at the molecular level. To investigate the molecular mechanism, it is necessary to develop a plasmid transformation system for marine bacteria that are able to lyse algae. In this paper, we describe the isolation of cryptic indigenous plasmids from Alteromonas sp. strains which are able to lyse the diatom Skeletonema costatum. We also present the construction of an Escherichia coli-Alteromonas shuttle vector made by fusing the cryptic plasmid with an E. coli vector.
MATERIALS AND METHODS
Bacterial strains and plasmids.
The bacterial strains and plasmids used in the present study are shown in Table 1. Alteromonas sp. strains were grown at 28°C with shaking in ASWM medium, which was a modified SWM-III medium (2) supplemented with 0.1% Casitone (Difco) and 0.05% yeast extract (Difco). LBN medium contained 1% peptone (Difco), 0.5% yeast extract, and 2.4% NaCl. LBN medium was used for the preparation of Alteromonas cells for electroporation. E. coli MV1184 was grown at 37°C with shaking in 2× YT medium (13). Whenever necessary, ampicillin and kanamycin were added at final concentrations of 100 and 200 μg/ml, respectively.
TABLE 1.
Organisms and plasmids used in this study
| Strain or plasmid | Relevant descriptiona | Reference or sourceb |
|---|---|---|
| Algae | ||
| Skeletonema costatum NIES-324 | Diatom | NIES |
| Eucampia zodiacs | Diatom | ASRI |
| Thalassiosira sp. | Diatom | NNFRI |
| Ditylum brightwelli | Diatom | NNFRI |
| Chattonella antiqua NIES-1 | Raphidophycean flagellate | NIES |
| Gymnodinium mikimotoi G303-AX2 | Dinoflagellate | NNFRI |
| Bacteria | ||
| Alteromonas sp. strain A27 | Alga-lytic marine bacterium | This study |
| Alteromonas sp. strain A28 | Alga-lytic marine bacterium; pAS28 | This study |
| Alteromonas sp. strain A29 | Alga-lytic marine bacterium; pAS29 | This study |
| Alteromonas sp. strain A30 | Alga-lytic marine bacterium | This study |
| Escherichia coli MV1184 | ara Δ(lac-proAB) rpsL thi (φ80 lacZΔM15) Δ(srl-recA)306::Tn10 Tetr F′(traD36 proAB+ lacIqlacZΔM15) | 14 |
| Plasmids | ||
| pAS28 | Indigenous plasmid in A28 | This study |
| pCRIIc | Cloning vector; ColE1 ori Apr Kmr | Invitrogen |
Apr, ampicillin resistance; Kmr, kanamycin resistance; Tetr, tetracycline resistance.
Abbreviations: NIES, National Institute for Environmental Studies, Tsukuba, Japan; NNFRI, Nansei National Fisheries Research Institute, Hiroshima, Japan; ASRI, Ariake Sea Research Institute, Fukuoka, Japan.
Algal cultures.
The marine algae employed in this study were Thalassiosira sp., Ditylum brightwelli, and Gymnodinium mikimotoi G303-AX2 from the Nansei National Fisheries Research Institute, Hiroshima, Japan; S. costatum NIES-324 and Chattonella antiqua NIES-1 from the National Institute for Environmental Studies, Tsukuba, Japan; and Eucampia zodiacs from the Ariake Sea Research Institute, Fukuoka, Japan (Table 1). Clonal axenic cultures were routinely maintained on modified SWM-III medium made with filtered seawater as the base. The seawater used was filtered through a 0.45-μm-pore-size Nuclepore filter and stored at 4°C in darkness. The modified SWM-III medium was autoclaved to prevent contamination with bacterial spores and small bacteria. Cultures were grown at 15°C for diatoms and 25°C for C. antiqua and G. mikimotoi with illumination at 35 microeinsteins/m2/s under a 12-h light–12-h dark regimen. Modified SWM-III agar and modified SWM-III soft agar contained 1.5 and 0.8% agar (Difco), respectively. ASWM agar plates were prepared by adding 1.5% agar to ASWM medium.
Isolation of alga-lytic bacteria.
Alga-lytic bacteria were isolated by the soft-agar overlayer technique (15). Coastal seawater samples were obtained with sterilized glass bottles from the Ariake Sea, Fukuoka, Japan. Axenic cultures of S. costatum NIES-324 were grown in modified SWM-III medium for 1 week, and 1 ml of the S. costatum culture was mixed with a 0.1-ml seawater sample and 2.5 ml of molten modified SWM-III soft agar (equilibrated to 47°C). The mixture was immediately poured onto a modified SWM-III agar plate. After the agar had solidified, the plates were incubated at 15°C with illumination at 35 microeinsteins/m2/s under a 12-h light–12-h dark regimen. Bacterial colonies which produced clear zones on lawns of S. costatum were picked, purified, and maintained on ASWM agar plates.
Mixed algal-bacterial cultures.
A log-phase culture of S. costatum NIES-324 was diluted with modified SWM-III medium to a final density of 2 × 105 cells per ml. Alteromonas clones in the logarithmic phase of growth were added to the S. costatum NIES-324 culture to a final density of 104 cells per ml, and the mixed culture was further incubated at 15°C with illumination at 35 microeinsteins/m2/s under a 12-h light–12-h dark regimen for 3 weeks. Enumeration of S. costatum NIES-324 cells was accomplished with a differential interference microscope (OPTIPHOT-2 X2F-NTF-21; Nikon Co., Tokyo, Japan). Bacteria were counted with an epifluorescence microscope (OPTIPHOT-2 X2F-EFD2; Nikon) after 4′,6-diamidino-2-phenylindole (DAPI) staining.
Isolation of plasmid DNA from Alteromonas sp.
Plasmid extractions were performed essentially by the alkaline lysis method (13). When plasmid samples were turbid, proteinase K was added to the samples to a final concentration of 50 μg/ml and the mixtures were incubated for 30 min at 37°C. After incubation with proteinase K, plasmid DNA was extracted with phenol-chloroform and then precipitated with ethanol. Plasmid DNAs were electrophoresed in 1% agarose gels and detected by ethidium bromide staining.
DNA manipulation and sequencing.
Standard procedures were used for preparation of plasmid DNA from E. coli strains, restriction enzyme digestions, ligations, transformations, agarose gel electrophoresis, and Southern hybridization (13). The nucleotide sequence of both strands was determined by the dideoxynucleotide sequencing method with single-stranded DNA (14, 18). A DNA sequence similarity search of the GenBank database was done with the FASTA program (10).
Electroporation.
Alteromonas cells were transformed by electroporation. Cells grown overnight in LBN medium were inoculated into fresh LBN medium (a 1% inoculum), and after 4 h of incubation, cells were harvested by centrifugation (10,000 × g, 5 min, 4°C). Pelleted cells were resuspended in ice-cold HS buffer containing 7 mM HEPES and 252 mM sucrose (pH 7.0), washed three times with the same buffer, and resuspended in 0.1 volume of HS buffer. Electroporation was performed with an Electro Cell Manipulator 600 (BTX Inc., San Diego, Calif.) at a capacitance of 50 μF and a voltage of 1.3 kV. After electroporation, cells were allowed to grow in LBN medium overnight and spread on LBN agar plates supplemented with 200 μg of kanamycin per ml to obtain transformants.
Nucleotide sequence accession number.
The nucleotide sequence of the 4.7-kb EcoRI-HindIII region of pAS28 has been deposited in the GSDB, DDBJ, EMBL, and NCBI nucleotide sequence databases under accession no. AB009311.
RESULTS
Isolation and characterization of alga-lytic bacteria.
S. costatum is the marine diatom which occasionally produces undesirable blooms in nearshore waters (9). Marine bacteria which cause lysis of unialgal cultures of S. costatum were isolated from seawater samples collected from the Ariake Sea of Japan. The lytic activity of marine bacteria was determined by plaque formation on the soft-agar overlayer technique. Four bacterial isolates were able to form plaques of algal lysis on S. costatum NIES-324 lawns. Typical plaques of marine bacteria appeared on the lawns of S. costatum NIES-324 cells after 1 week. When plaques formed, isolated and discrete bacterial colonies appeared among the algal overlay. All of the bacteria isolated were gram-negative, short rods, motile, polarly flagellated, and DNase positive. They also oxidatively produced organic acids from glucose but did not produce diffusible pigments. On the basis of biochemical and morphological tests, the four isolates were tentatively identified as belonging to the genus Alteromonas and designated A27, A28, A29, and A30.
Mixed cultures.
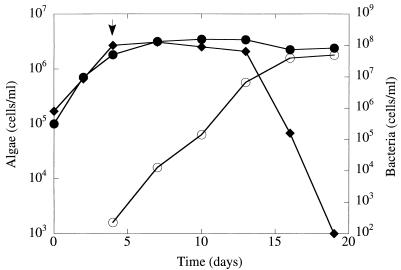
S. costatum NIES-324 demonstrated typical batch culture kinetics in modified SWM-III medium (Fig. 1). Rapid lysis of S. costatum NIES-324 occurred in the presence of Alteromonas sp. strain A28 cells (Fig. 1). When S. costatum NIES-324 and A28 were grown together, the algal population rapidly declined around 13 days after the start of cultivation. In contrast, bacteria were able to grow in the mixed culture and the bacterial population increased to up to 7 × 107 cells per ml within 16 days after the start of cultivation. Alteromonas sp. strains showed a broad host range of antialgal activity when tested for mixed cultures with several marine planktonic algae in modified SWM-III medium. Alteromonas isolates lysed the diatoms Thalassiosira sp. and E. zodiacs and the raphidophycean flagellate C. antiqua. The diatom D. brightwelli and the dinoflagellate G. mikimotoi were not lysed by Alteromonas sp. strains (data not shown).
FIG. 1.
Influence of Alteromonas sp. strain A28 on the growth of S. costatum. S. costatum was grown in modified SWM-III medium either in the presence of A28 (⧫) or in the absence of A28 (•). In the algal-bacterial mixed culture, bacterial cells (○) were added to the S. costatum culture 4 days after the start of cultivation, as indicated by the arrow.
Isolation and characterization of Alteromonas cryptic plasmids.
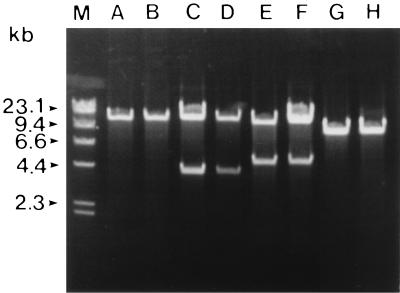
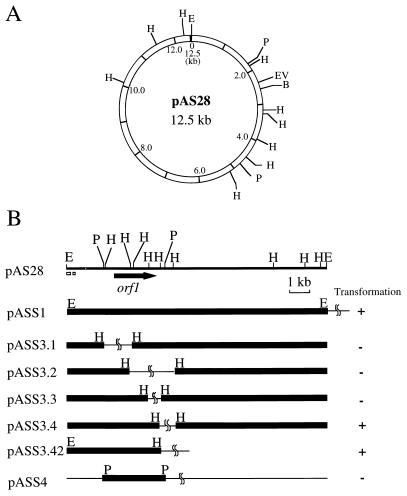
Cryptic indigenous plasmids of 12.5 kb were detected in A28 and A29 cells after alkaline lysis DNA extraction and agarose electrophoresis (Fig. 2). The plasmid in A28, designated pAS28, was purified and digested with various restriction enzymes to determine a physical map (Fig. 3A). pAS28 had unique BglII, EcoRI, and EcoRV restriction sites, two PvuII sites, and nine HindIII sites. However, no BamHI, SalI, or XbaI restriction site was detected. The physical map of the plasmid in A29 was also determined, and it was found to be identical to that of pAS28.
FIG. 2.
Agarose gel electrophoresis of EcoRI-digested plasmid DNAs isolated from Alteromonas spp. and E. coli strains. Lanes: A, A28; B, A29; C, A28(pASS1); D, E. coli MV1184(pASS1); E, A28(pASS3.4); F, E. coli MV1184(pASS3.4); G, A28(pASS3.42); H, E. coli MV1184(pASS3.42); M, HindIII-digested phage lambda DNA.
FIG. 3.
(A) Restriction map of pAS28. (B) Restriction map of pAS28 subclones. pAS28, pASS1, pASS3.1, pASS3.2, pASS3.3, pASS3.4, and pASS3.42 are shown as linearized at the unique EcoRI site. pASS4 is also shown as linearized at the EcoRI site in the pCRIIc sequence. The location and orientation of orf1 are indicated by the horizontal arrow below the restriction map of pAS28. The two small, open boxes below the restriction map of pAS28 show the locations of AT-rich regions. The abilities of recombinant plasmids to transform Alteromonas sp. strain A28 are indicated on the right. For pAS28 subclones, thin bars indicate pCRIIc DNA and thick bars represent DNA fragments subcloned from pAS28. Restriction sites: B, BglII; E, EcoRI; EV, EcoRV; H, HindIII; P, PvuII.
Construction of a shuttle vector.
Since growth of the four Alteromonas sp. strains was inhibited by kanamycin, we used a kanamycin resistance determinant for direct selection of Alteromonas transformants. E. coli plasmid pCRIIc, which carries a kanamycin resistance gene and an ampicillin resistance gene, was digested with EcoRI and ligated with EcoRI-digested pAS28 to construct pASS1 (Fig. 3B). Competent cells of E. coli MV1184 were transformed with pASS1. Plasmids were prepared from the ampicillin-resistant E. coli transformants and again used to transform A28 cells by electroporation. Kanamycin-resistant transformants of A28 were obtained at a frequency of 2 × 106/μg of DNA. To confirm that the Alteromonas transformants harbored pASS1, plasmid DNA was extracted from several kanamycin-resistant colonies and analyzed by agarose gel electrophoresis. pASS1 was detected in all of the kanamycin-resistant transformants (Fig. 2). Interestingly, pASS1 prepared from Alteromonas transformants could transform A28 at a higher frequency (107/μg of DNA) than that prepared from E. coli transformants. pASS1 was also introduced into A27, A29, and A30. In all strains, kanamycin-resistant transformants were obtained at frequencies similar to that of A28.
Derivatives of pASS1.
To determine which region is essential for plasmid maintenance in Alteromonas cells, various hybrid plasmids were constructed in E. coli MV1184. pAS28 was partially digested with HindIII and ligated with HindIII-digested pCRIIc to make pASS3.1, pASS3.2, pASS3.3, and pASS3.4 (Fig. 3B). The hybrid plasmids isolated from MV1184 were then used to transform A28. When pASC3.4 was introduced into A28, kanamycin-resistant transformants were obtained at frequencies of about 106 cells per μg of DNA. However, no transformants were obtained with pASS3.1, pASS3.2, and pASS3.3. To further determine the region necessary for plasmid maintenance, pASS3.42 and pASS4 were constructed by subcloning the 4.7-kb EcoRI-HindIII fragment and the 3.3-kb PvuI fragment of pAS28 into pCRIIc, respectively. A28 transformants were obtained with pASS3.42 but not with pASS4 (Fig. 3B). These results indicate that the 4.7-kb EcoRI-HindIII fragment of pAS28 is essential for plasmid maintenance in A28.
DNA sequence analysis.
To further analyze the region essential for plasmid maintenance, the 4.7-kb EcoRI-HindIII region of pAS28 was sequenced (data not shown). Nucleotide sequence analysis revealed the presence of one open reading frame (orf1). orf1 is preceded by a potential ribosome-binding site (16), and it was predicted that orf1 encodes a polypeptide comprising 708 amino acid residues with a molecular mass of 80,613 Da. Computer analysis of the predicted product of orf1 revealed that it is a hydrophilic and basic protein (pI 8.6). Although the Orf1 protein translation was screened for similarity to known protein sequences in the SWISSPROT and PIR databases, no homologous protein was identified. Two A+T-rich regions were found in the upstream region of the orf1 gene.
DISCUSSION
Alteromonas sp. strain A28 was isolated from a coastal seawater sample and was able to lyse various algae, including the diatoms S. costatum, Thalassiosira sp., and E. zodiacs and the raphidophycean flagellate C. antiqua. This bacterial ability to lyse algal cells is potentially useful for controlling outbreaks of harmful algal blooms, and therefore, it is important to understand the mechanism by which algae are lysed at the molecular level. Although alga-lytic bacteria have been reported previously (3, 5–7, 9, 12), virtually nothing is known about the molecular mechanism underlying the lysis of algae. This is partly because no efficient cloning system was available for alga-lytic bacteria.
In this study, indigenous plasmid pAS28 was isolated from Alteromonas sp. strain A28. Chimeric plasmid pASS3.42, which was constructed by cloning the 4.7-kb EcoRI-HindIII fragment of pAS28 into E. coli plasmid vector pCRIIc, can be used as an E. coli-Alteromonas shuttle vector. pASS3.42 carries ampicillin and kanamycin resistance determinants and contains unique EcoRI, NotI, XhoI, XbaI, NsiI, ClaI, EcoRV, PstI, and SacI restriction sites. Insertions can potentially be made into the EcoRI, XhoI, XbaI, NotI, and NsiI sites without disrupting plasmid maintenance or resistance functions. pASS3.42 can be transformed into Alteromonas sp. strain A28 by electroporation. The cloning system presented here will be used to investigate the molecular mechanism of the alga-lytic activity of Alteromonas sp. strain A28.
The efficiency with which chimeric plasmids transformed A28 was dependent upon the source of plasmid DNA. For example, when chimeric plasmid pASS1, which was constructed by fusing pAS28 and pCRIIc, was prepared from Alteromonas cells, it transformed A28 at a frequency of 107/μg of DNA. When A28 was transformed with pASS1 purified from an E. coli host, the transformation frequency decreased nearly 10-fold. pASS1 isolated from strain A28 was not digested with MboI, which recognizes the unmethylated sequence GATC. However, it was digested with DpnI, which recognizes the methylated sequence GATC (data not shown). These results suggest the presence of adenine methylation at the sequence GATC in strain A28. However, the adenine methylation is not likely to contribute to the restriction barrier of A28, because preparation of pASS1 from E. coli JM110, which carries the dam and dcm mutations (19), did not improve the transformation efficiency. Whatever system is reponsible for this phenomenon remains to be investigated. The restriction barrier is unlikely to reduce the utility of the E. coli-Alteromonas shuttle vector. One microgram of pASS1 DNA purified from E. coli could produce one million Alteromonas transformants.
DNA sequence analysis of the 4.7-kb EcoRI-HindIII region of pAS28, which is essential for plasmid maintenance in Alteromonas sp. strain A28, indicated the presence of two A+T-rich regions and one open reading frame (orf1). Computer analysis predicted that the orf1 gene encodes a hydrophilic and basic protein with a molecular mass of 81 kDa. Regardless of plasmid size, plasmid-borne information required for replication is often contained within a segment that is 3 kb or less in size (4). With the exception of ColE1-type plasmids, most of the characterized plasmids encode a specific replication (Rep) protein which is necessary for their replication. The Rep protein recognizes and binds to specific sequences at the replication origin and causes structural changes in an adjacent A+T-rich region (4). Although the predicted product of orf1 showed no significant homology to any known Rep proteins, deletion of orf1 prevented A28 replication in Alteromonas sp. A28, suggesting that its product possesses a replication (rep) function.
ACKNOWLEDGMENT
This work was supported in part by a grant from the Fisheries Agency of Japan.
REFERENCES
- 1.Burnham J C, Collart S A, Highison B W. Entrapment and lysis of the cyanobacterium Phormidium luridum by aqueous colonies of Myxococcus xanthus PCO2. Arch Microbiol. 1981;129:285–294. [Google Scholar]
- 2.Chen L C M, Edelstein T, McLachlan J. Bonnemaisonia hamifera Hariot in nature and in culture. J Phycol. 1969;5:211–220. doi: 10.1111/j.1529-8817.1969.tb02605.x. [DOI] [PubMed] [Google Scholar]
- 3.Fukami K, Yuzawa A, Nishijima T, Hata Y. Isolation and properties of a bacterium inhibiting the growth of Gymnodinium nagasakiense. Nihon Suisan Gakkai-shi. 1992;58:1073–1077. [Google Scholar]
- 4.Helinski D R, Toukdarian A E, Novick R P. Replication control and other stable maintenance mechanisms of plasmids. In: Neidhardt F C, Curtiss III R, Ingraham J L, Lin E C C, Low K B, Magasanik B, Reznikoff W S, Riley M, Schaechter M, Umbarger H E, editors. Escherichia coli and Salmonella: cellular and molecular biology. Washington, D.C: ASM Press; 1996. pp. 2295–2324. [Google Scholar]
- 5.Imai I, Ishida Y, Sawayama S, Hata Y. Isolation of a marine gliding bacterium that kills Chattonella antiqua (Raphidophyceae) Nihon Suisan Gakkai-shi. 1991;57:1409. [Google Scholar]
- 6.Imai I, Ishida Y, Hata Y. Killing of marine phytoplankton by a gliding bacterium Cytophga sp. isolated from the coastal sea of Japan. Mar Biol. 1993;116:527–532. [Google Scholar]
- 7.Imai I, Ishida Y, Sakaguchi K, Hata Y. Algicidal bacteria isolated from northern Hiroshima Bay, Japan. Fish Sci. 1995;61:628–636. [Google Scholar]
- 8.Kayser H. Growth interaction between marine dinoflagellates in multispecies culture experiments. Mar Biol. 1979;52:357–369. [Google Scholar]
- 9.Mitsutani A, Takase K, Kirita M, Ishida Y. Lysis of Skeletonema costatum by Cytophaga sp. isolated from the coastal waters of the Ariake Sea. Nihon Suisan Gakkai-shi. 1992;58:2159–2169. [Google Scholar]
- 10.Pearson W R, Lipman D J. Improved tools for biological sequence comparison. Proc Natl Acad Sci USA. 1988;85:2444–2448. doi: 10.1073/pnas.85.8.2444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Romalde J L, Toranzo A E, Barja J L. Changes in bacterial populations during red tides caused by Mesodinium rubrum and Gymnodinium catenatum in Northwest coast of Spain. J Appl Bacteriol. 1990;68:123–132. doi: 10.1111/j.1365-2672.1990.tb02556.x. [DOI] [PubMed] [Google Scholar]
- 12.Sakata T. Occurrence of marine Cytophaga sp. possessing algicidal activity for diatoms. Nihon Suisan Gakkai-shi. 1990;56:1165. [Google Scholar]
- 13.Sambrook J, Fritsch E F, Maniatis T. Molecular cloning: a laboratory manual. 2nd ed. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory Press; 1989. [Google Scholar]
- 14.Sanger F, Nicklen S, Coulson A R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA. 1977;74:5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shilo M. Lysis of blue-green algae by myxobacter. J Bacteriol. 1970;104:453–461. doi: 10.1128/jb.104.1.453-461.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shine J, Dalgarno L. The 3′ terminal sequence of Escherichia coli 16S ribosomal RNA: complementarity to nonsense triplets and ribosome binding sites. Proc Natl Acad Sci USA. 1974;71:1342–1346. doi: 10.1073/pnas.71.4.1342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sieburth J M. Sea microbes. New York, N.Y: Oxford University Press; 1979. [Google Scholar]
- 18.Vieira J, Messing J. Production of single-stranded plasmid DNA. Methods Enzymol. 1987;153:3–11. doi: 10.1016/0076-6879(87)53044-0. [DOI] [PubMed] [Google Scholar]
- 19.Yanish-Perron C, Vieira J, Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33:103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]