Abstract
Background
Clinical utility of routinely measured serial biomarkers in predicting escalation of inpatient care intensity and mortality among hospitalized patients with COVID-19 remains unknown.
Methods
This retrospective cohort study included patients with COVID-19 who admitted to the Massachusetts General Hospital between March and June 2020 and January to March 2021. White blood cell (WBC) count, platelet count, C-reactive protein (CRP), and D-dimer values were measured on days 1, 3, and 7 of admission. Clinical outcomes include 30- and 60-day morality, ICU transfer, and overall survival (OS) over a follow-up period of 90 days. The association between serial biomarkers and outcomes were assessed using multivariable logistic regression and Cox proportional hazards models.
Measurements and main results
Of the 456 patients hospitalized with COVID-19, 199 (43.6%) were ICU, 179 (39.3%) were medical floor, and 78 (17.1%) were initially admitted to the medical floor and then transferred to the ICU. In adjusted analyses, each unit increase in the slope of CRP was associated with a 42% higher odds of ICU transfer after controlling for the initial admission level (OR = 1.42, 95% CI: 1.25–1.65, P < 0.001). Including serial change in CRP levels from initial level on admission achieved the greatest predictive accuracy for ICU transfer (AUC = 0.72, 95% CI: 0.64–0.79).
Conclusions
Serial change in CRP levels from admission is associated with escalations of inpatient care intensity and mortality among hospitalized patients with COVID-19.
Introduction
Coronavirus disease 2019 (COVID-19) is a respiratory infection that can present with a broad spectrum of clinical manifestations ranging in severity [1]. While 40–50% of COVID-19 cases are asymptomatic, a significant proportion of patients may develop rapidly progressive respiratory failure and other complications that require intensive inpatient care, such as hospitalization, supplemental oxygen therapy, and mechanical ventilation [2–4]. Patients who present to the hospital have heterogeneous disease courses and clinical outcomes that may include respiratory failure, multi-organ dysfunction, and death in the most severe cases. It is imperative for clinicians to have access to readily obtainable and clinically meaningful biomarkers to prognosticate the clinical course of disease and thereby effectively triage and optimize the therapeutic options for patients.
Given the global pandemic, there have been a number of studies evaluating the use of biomarkers for disease prognosis among hospitalized patients with COVID-19 [5–8]. Previous studies have shown that low lymphocyte, platelet counts and albumin levels, and elevated C reactive protein (CRP), white blood cell (WBC) count, blood urea nitrogen, interleukin-6 (IL-6), blood urea nitrogen, creatine kinase, procalcitonin, D-dimer, lactate dehydrogenase, alanine aminotransferase, aspartate aminotransferase and creatinine levels are all associated with poor COVID-19 outcomes [9–20]. The Sequential Organ Failure Assessment (SOFA) score is associated with worse clinical outcomes among intensive care unit (ICU) patients as well as patients with COVID-19 pneumonia requiring mechanical ventilation [21, 22].
However, there have been few studies on serial changes in laboratory biomarkers for prognosis and clinical outcomes among hospitalized patients with COVID-19. Understanding the variation and profile of obtainable clinical biomarkers since admission and how they are related to COVID-19 outcomes may facilitate the development of risk-stratified approaches and timely clinical decision-making that would benefit hospitalized patients [9]. To fill this important knowledge gap, we investigated the associations of baseline and serial changes in WBC count, platelet count, CRP, and D-dimer from whole blood samples that were consecutively measured on days 1, 3, and 7 since admission with a variety of clinical outcomes, including 30- and 60-day morality, ICU transfer, and overall survival (OS) among the 456 hospitalized patients with COVID-19 who were admitted to Massachusetts General Hospital (MGH) between March and June 2020 (surge 1) and January to March 2021 (surge 2).
Materials and methods
Patient recruitment and sample collection
We studied adult ICU patients with SARS-CoV-2 infection and acute hypoxemic respiratory failure (AHRF) and non-ICU patients with mild hypoxemia managed with 2–6 L/min supplemental oxygen who were consecutively admitted to MGH between March and June 2020 (surge 1) and January to March 2021 (surge 2) retrospectively. Informed consent was waived, and the study was approved by the institutional review board of MGH (protocol number 2015P001650). We collected baseline demographics, clinical characteristics, and outcomes from the electronic medical record. We assessed comorbidities, including body mass index (BMI), previous pulmonary disease, chronic obstructive pulmonary disease (COPD), asthma, and interstitial lung disease (ILD). We calculated modified sequential organ failure assessment (mSOFA) score on day 1 for ICU patients [23–25]. WBC count (1000/mm3), platelet count (1000/mm3), CRP (mg/L), and D-dimer (ng/ml) were consecutively measured on days 1, 3, and 7 since admission by the MGH clinical core laboratory. We report all available data without imputation, and the statistical analyses were conducted between August 2022 and February 2023
Clinical outcomes
30- and 60-day mortality were calculated, respectively, as the probabilities of death within 30 and 60 days after admission. Every patient was followed for up to 90 days after admission and OS was defined as from the date of hospital admission to death or the date of last contact, whichever happened first. ICU transfer was documented for those transferred to the ICU from medical floor after admission.
Statistical analysis
Descriptive statistics summarized the baseline demographic and clinical data. Categorical variables were reported with counts and percentages and continuous variables were reported with median and range. Kruskal-Wallis tests and chi-square tests were used for continuous and categorical variables, respectively. Logistic regression was used to evaluate the association with binary outcomes (30- and 60-day mortality and ICU transfer). Association analyses with ICU transfer were conducted in patients who were initially admitted to the medical floor. Event-time distributions were estimated using Kaplan-Meier methodology. Log-rank tests were used to test the differences in event-time distributions and Cox proportional hazards models were fitted to estimate hazard ratios (HR). Potential confounders of age, gender, smoking status, ethnicity and COVID surge were adjusted for in the multivariable analyses. The slope of serial changes in each biomarker for each patient was obtained to assess its time-varying effects with the clinical outcomes.
To address whether serial changes in biomarkers from admission provide additional predictive values on clinical outcomes, area under the curve (AUC) and C-index were calculated and compared between 1) baseline models with only biomarker level on day 1, and 2) models incorporating serial changes and levels on day 1 in combination with baseline covariates. The receiver operating characteristic curve (ROC) and area under the curve (AUC) were generated and calculated based on 5-fold cross-validation for logistic regression, while the C-index was calculated for the Cox proportional hazard model in survival analysis. 95% confidence intervals (CIs) for AUC and C-index were obtained by creating 300 bootstrap samples and taking the 2.5th and 97.5th percentiles of the re-estimates. All reported P-values are two-sided and confidence intervals are at the 95% level, with significance pre-defined to be at < 0.05.
Results
Demographic and clinical characteristics
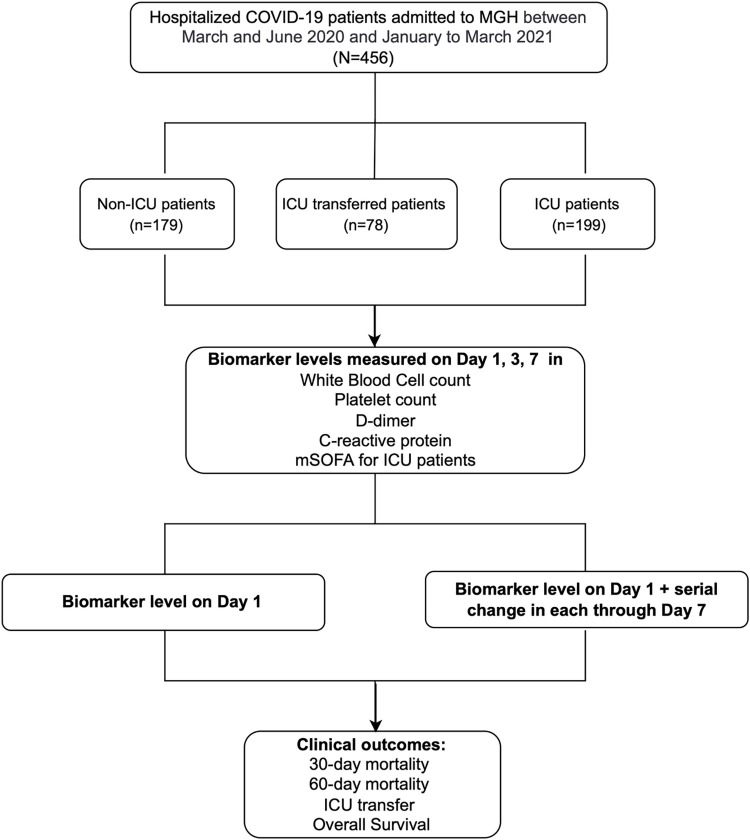
Of the 456 hospitalized COVID patients, 199 (43.6%) were admitted to ICU, 179 (39.3%) were admitted to the medical floor (non-ICU), and 78 (17.1%) were transferred to ICU after admission to the medical floor (Fig 1). Patient demographics, clinical characteristics, and clinical outcomes are summarized by COVID severity in S1 Table. A total of 182 (39.9%) patients were admitted during the second surge of COVID-19. The median age at diagnosis was 63 years (range, 51–73 yr), 282 (61.8%) patients were male, and 154 (33.8%) patients were Hispanic/Latino. 184 (40.4%) patients were former or current smokers, and 129 (28.3%) patients had preexisting pulmonary diseases, including 65 (14.3%) with asthma, 53 (11.6%) with COPD, and 7 (1.5%) with ILD.
Fig 1. Study overview.
The study was conducted in 456 hospitalized COVID-19 patients admitted to MGH between March and June 2020 (surge 1) and January to March 2021 (surge 2). Among them, 199 (43.6%) were admitted to ICU, 179 (39.3%) were non-ICU patients and 78 (17.1%) were transferred to ICU after admission, Laboratory biomarkers in white blood cell (WBC) count, platelet count, D-dimer, and C-reactive protein (CRP) were consecutively measured on day 1, 3, and 7 since admission for all while modified sequential organ failure assessment (mSOFA) at ICU admission was measured for ICU patients. We assessed the prognostic effects of each biomarker level on day 1 as well as the serial change in each controlling for their corresponding values on day 1, along with baseline covariates on 30-day, 60-day mortality, ICU transfer and overall survival with a follow-up period of 90 days.
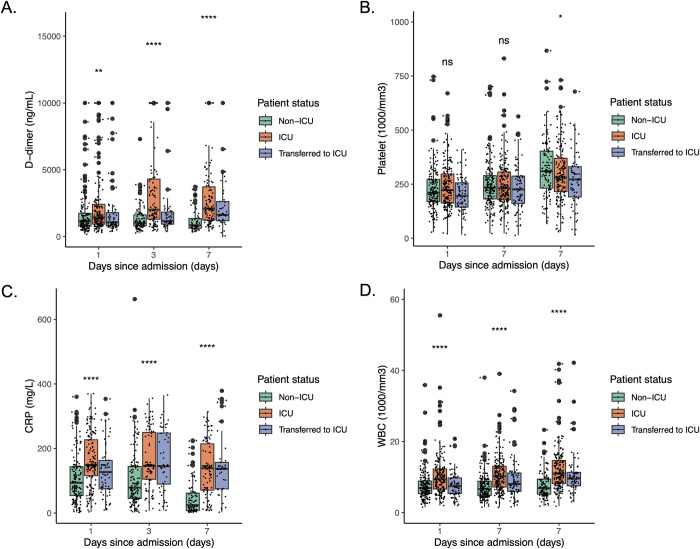
Laboratory values of WBC count (1000/mm3), platelet count (1000/mm3), CRP (mg/L) and D-dimer (ng/ml) on day 1 of admission for all patients and mSOFA score for ICU patients were summarized in S2 Table. A significantly higher WBC count, CRP and D-dimer values on day 1 of admission were observed for ICU patients (median WBC (mWBC): 9.4 (1000/mm3) range: 2.0–168 (1000/mm3); median CRP (mCRP): 148 (mg/L) range: 3.7–370 (mg/L); median D-dimer (mD-dimer): 1440 (ng/ml) range: 302–10000 (ng/ml), respectively) compared to non-ICU patients (mWBC: 6.9 (1000/mm3) range: 1.5–35.9 (1000/mm3); mCRP: 93.6 (mg/L) range: 1.4–360 (mg/L); mD-dimer: 1140 (ng/ml) range: 163–10000 (ng/ml), respectively). Serial biomarker levels on day 1, 3, 7 of admission stratified by COVID severity are shown in Fig 2. The median mSOFA was 7 (range, 1–15) for ICU patients. ICU patients had significantly higher WBC counts and CRP and D-dimer levels on day 3 and 7 of admission, compared to non-ICU patients, as described in S2 Table.
Fig 2. Serial biomarker levels on day 1, 3 and 7 since admission by COVID severity.
(A) D-dimer (ng/ml) level on day 1, 3 and 7 since admission for COVID-19 patients by severity. (B) Platelet count (1000/mm3) level on day 1, 3 and 7 since admission for COVID-19 patients by severity. (C) C-reactive protein (mg/L) level on day 1, 3 and 7 since admission for COVID-19 patients by severity. (D) White blood cell (WBC) count (1000/mm3) level on day 1, 3 and 7 since admission for COVID-19 patients by severity.
Association between biomarker levels on admission and clinical outcomes
Among ICU patients, mSOFA on admission is associated with increased 30-day mortality (OR = 1.39, 95% CI: 1.21–1.62, P < 0.001) and 60-day mortality (OR = 1.42, 95% CI: 1.24–1.66, P < 0.001), as well as increased overall mortality (HR = 1.30, 95% CI: 1.18–1.44, P < 0.001) after adjusting for age, gender, smoking status, ethnicity, and COVID surge (S3 Table). Higher CRP level on day 1 of admission is associated with increased 30- and 60-day mortality as well as increased odds of ICU transfer (for every 10 mg/L increase in CRP: 30-day mortality OR = 1.04, 95% CI: 1.01–1.08, P = 0.02; 60-day mortality OR = 1.05, 95% CI: 1.01–1.09, P = 0.01; ICU transfer OR = 1.04, 95% CI: 1.01–1.09, P = 0.02, respectively). Every 1000 ug/mL increase in D-dimer on day 1 of admission is significantly associated with a 14% increase in 30-day mortality (OR = 1.14, 95% CI: 1.02–1.28, P = 0.04) and a trend towards an increase in 60-day mortality (OR = 1.12, 95% CI: 0.99–1.25, P = 0.07). Furthermore, CRP, D-dimer, platelet count, and WBC count on day 1 of admission are all significantly associated with OS (every 10 mg/L increase in CRP: HR = 1.04, 95% CI: 1.02–1.07, P = 0.002; every 1000 ug/mL increase in D-dimer: HR = 1.10, 95% CI: 1.01–1.20, P = 0.03; every 1000/mm3 increase in platelet count: HR = 0.997, 95% CI: 0.995–1.000, P = 0.03; every 1000/mm3 increase in WBC count: HR = 1.02, 95% CI: 1.01–1.03, P = 0.001, respectively) (Table 1).
Table 1. Association between biomarkers on day 1 of admission and clinical outcomes.
| n = 381 | 30-day mortality | 60-day mortality | ICU transfer (n = 229) | Overall Survival | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OR | 95% CI | P | OR | 95% CI | P | OR | 95% CI | P | HR | 95% CI | P | |
| Every 10mg/L increase in CRP | 1.04 | 1.01–1.08 | 0.02 | 1.05 | 1.01 – 1.09 | 0.01 | 1.04 | 1.01 – 1.09 | 0.02 | 1.04 | 1.02–1.07 | 0.002 |
| Age | 1.09 | 1.06 – 1.12 | <0.001 | 1.08 | 1.06 – 1.11 | <0.001 | 1.01 | 0.99 – 1.04 | 0.24 | 1.08 | 1.06 – 1.10 | <0.001 |
| Female vs. Male | 0.58 | 0.31 – 1.07 | 0.09 | 0.56 | 0.31 – 1.02 | 0.06 | 0.79 | 0.43 – 1.45 | 0.45 | 0.72 | 0.44 – 1.17 | 0.18 |
| Ever smoker vs. never smoker | 0.68 | 0.36 – 1.27 | 0.23 | 0.74 | 0.40 – 1.34 | 0.32 | 0.49 | 0.24 – 0.97 | 0.04 | 0.75 | 0.45 – 1.23 | 0.25 |
| Surge 2 vs. 1 | 1.04 | 0.56 – 1.90 | 0.89 | 1.16 | 0.64 – 2.08 | 0.62 | 0.65 | 0.34 – 1.22 | 0.19 | 1.10 | 0.69 – 1.76 | 0.68 |
| Hispanic vs. Non-Hispanic | 1.01 | 0.48 – 2.08 | 0.98 | 0.94 | 0.46 – 1.90 | 0.87 | 0.87 | 0.42 – 1.77 | 0.71 | 0.87 | 0.48 – 1.58 | 0.65 |
| n = 377 | 30-day mortality | 60-day mortality | ICU transfer (n = 225) | Overall Survival | ||||||||
| OR | 95% CI | P | OR | 95% CI | P | OR | 95% CI | P | HR | 95% CI | P | |
| Every 1000 ug/mL increase in D-dimer | 1.14 | 1.01–1.28 | 0.04 | 1.12 | 0.99–1.25 | 0.07 | 1.07 | 0.89–1.27 | 0.45 | 1.10 | 1.01–1.20 | 0.03 |
| Age | 1.09 | 1.06 – 1.12 | <0.001 | 1.08 | 1.05 – 1.11 | <0.001 | 1.01 | 0.99 – 1.03 | 0.50 | 1.07 | 1.05 – 1.10 | <0.001 |
| Female vs. Male | 0.56 | 0.29 – 1.05 | 0.08 | 0.56 | 0.30 – 1.03 | 0.07 | 0.94 | 0.51 – 1.72 | 0.85 | 0.67 | 0.40 – 1.12 | 0.13 |
| Ever smoker vs. never smoker | 0.68 | 0.35 – 1.31 | 0.26 | 0.74 | 0.39 – 1.37 | 0.34 | 0.63 | 0.31 – 1.23 | 0.18 | 0.73 | 0.44 – 1.23 | 0.24 |
| Surge 2 vs. 1 | 0.82 | 0.43 – 1.55 | 0.55 | 1.00 | 0.54 – 1.82 | 1.00 | 0.59 | 0.30 – 1.13 | 0.12 | 0.98 | 0.60 – 1.60 | 0.94 |
| Hispanic vs. Non-Hispanic | 0.74 | 0.33 – 1.61 | 0.47 | 0.80 | 0.37 – 1.66 | 0.56 | 1.01 | 0.50 – 2.02 | 0.97 | 0.80 | 0.42 – 1.52 | 0.49 |
| n = 429 | 30-day mortality | 60-day mortality | ICU transfer (n = 248) | Overall Survival | ||||||||
| OR | 95% CI | P | OR | 95% CI | P | OR | 95% CI | P | HR | 95% CI | P | |
| Platelet count (1000/mm3) | 0.997 | 0.993–0.999 | 0.02 | 0.996 | 0.993–0.999 | 0.01 | 0.997 | 0.994–1.000 | 0.09 | 0.997 | 0.995–1.00 | 0.03 |
| Age | 1.07 | 1.05 – 1.09 | <0.001 | 1.07 | 1.04 – 1.09 | <0.001 | 1.00 | 0.98 – 1.03 | 0.65 | 1.06 | 1.04 – 1.08 | <0.001 |
| Female vs. Male | 0.54 | 0.30 – 0.93 | 0.03 | 0.56 | 0.32 – 0.95 | 0.03 | 0.82 | 0.46 – 1.44 | 0.49 | 0.72 | 0.47 – 1.12 | 0.14 |
| Ever smoker vs. never smoker | 0.82 | 0.47 – 1.43 | 0.49 | 0.86 | 0.50 – 1.47 | 0.59 | 0.59 | 0.31 – 1.11 | 0.11 | 0.81 | 0.52 – 1.25 | 0.35 |
| Surge 2 vs. 1 | 1.17 | 0.69 – 1.98 | 0.57 | 1.39 | 0.83 – 2.31 | 0.21 | 0.74 | 0.41 – 1.32 | 0.31 | 1.27 | 0.84 – 1.91 | 0.26 |
| Hispanic vs. Non-Hispanic | 1.04 | 0.53 – 1.98 | 0.91 | 1.07 | 0.56 – 2.00 | 0.83 | 1.01 | 0.51 – 1.97 | 0.98 | 0.98 | 0.58 – 1.67 | 0.94 |
| n = 431 | 30-day mortality | 60-day mortality | ICU transfer (n = 249) | Overall Survival | ||||||||
| OR | 95% CI | P | OR | 95% CI | P | OR | 95% CI | P | HR | 95% CI | P | |
| WBC count (1000/mm3) | 1.03 | 1.00 – 1.08 | 0.09 | 1.03 | 1.00 – 1.07 | 0.11 | 1.03 | 0.98 – 1.08 | 0.28 | 1.02 | 1.01 – 1.03 | 0.001 |
| Age | 1.07 | 1.05 – 1.09 | <0.001 | 1.07 | 1.04 – 1.09 | <0.001 | 1.01 | 0.99 – 1.03 | 0.54 | 1.06 | 1.04 – 1.08 | <0.001 |
| Female vs. Male | 0.51 | 0.29 – 0.89 | 0.02 | 0.52 | 0.30 – 0.88 | 0.02 | 0.77 | 0.43 – 1.36 | 0.37 | 0.69 | 0.45 – 1.07 | 0.10 |
| Ever smoker vs. never smoker | 0.77 | 0.44 – 1.34 | 0.37 | 0.84 | 0.49 – 1.42 | 0.52 | 0.61 | 0.32 – 1.14 | 0.13 | 0.83 | 0.54 – 1.28 | 0.40 |
| Surge 2 vs. 1 | 1.04 | 0.61 – 1.76 | 0.89 | 1.28 | 0.77 – 2.13 | 0.34 | 0.73 | 0.40 – 1.31 | 0.30 | 1.19 | 0.79 – 1.79 | 0.42 |
| Hispanic vs. Non-Hispanic | 1.00 | 0.51 – 1.93 | 0.99 | 1.00 | 0.53 – 1.87 | 1.00 | 1.03 | 0.52 – 2.00 | 0.93 | 0.97 | 0.56 – 1.66 | 0.90 |
Prognostic associations of serial changes in biomarker levels after admission
We also investigated the association between biomarker levels on Day 7 of admission after adjusting for levels on Day 1 and other baseline covariates, and similar findings were summarized in S4 Table. We next evaluated the prognostic associations of serial changes in biomarker levels on clinical outcomes by including in the regression models the slope for each patient along with the corresponding biomarker level on day 1 and baseline covariates (Table 2). Increasing CRP levels were associated with worse prognosis and clinical outcomes. Each unit increase in the slope of CRP was associated with a 12% increase in 30- and 60-day mortality (30-day mortality OR = 1.12, 95% CI: 1.03–1.21, P = 0.008; 60-day mortality OR = 1.12, 95% CI: 1.04–1.22, P = 0.005), a 42% increase in ICU transfer (OR = 1.42, 95% CI: 1.25–1.65, P < 0.001) and reduced OS (HR = 1.07, 95% CI: 1.01–1.14, P = 0.02). Similarly, each unit increase in the slope of WBC was associated with increased 30-day mortality (OR = 3.85, 95% CI: 1.90–8.05, P < 0.001), 60-day mortality (OR = 2.91, 95% CI: 1.47–5.87, P < 0.001), higher risk of ICU transfer (OR = 9.43, 95% CI: 2.92–34.65, P < 0.001) and increased overall mortality (HR = 1.99, 95% CI: 1.24–3.20, P = 0.004). In contrast, increasing platelet counts were associated with improved clinical outcomes; with each unit increase in the slope was associated with a 6% decrease in 30-day (OR = 0.94, 95% CI: 0.92–0.97, P < 0.001) and 60-day mortality (OR = 0.94, 95% CI: 0.91–0.96, P < 0.001), a 4% decrease in odds of ICU transfer (OR = 0.96, 95% CI: 0.94–0.99, P = 0.01) and less overall mortality (HR = 0.96, 95% CI: 0.94–0.97, P < 0.001). An increase in the slope of D-dimer was only modestly associated with an increased risk of ICU transfer after controlling for level on day 1 of admission and baseline covariates (OR = 1.01, 95% CI: 1.00–1.01, P = 0.01).
Table 2. Prognostic effects of serial changes in biomarkers since admission.
| n = 381 | 30-day mortality | 60-day mortality | ICU transfer | Overall Survival | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OR | 95% CI | P | OR | 95% CI | P | OR | 95% CI | P | HR | 95% CI | P | |
| CRP (mg/L) at Day 1 | 1.01 | 1.00 – 1.01 | 0.001 | 1.01 | 1.00 – 1.01 | <0.001 | 1.02 | 1.01 – 1.02 | <0.001 | 1.01 | 1.00 – 1.01 | <0.001 |
| Slope of CRP | 1.12 | 1.03 – 1.21 | 0.008 | 1.12 | 1.04 – 1.22 | 0.005 | 1.42 | 1.25 – 1.65 | <0.001 | 1.07 | 1.01 – 1.14 | 0.02 |
| Age | 1.09 | 1.06 – 1.12 | <0.001 | 1.08 | 1.06 – 1.11 | <0.001 | 1.01 | 0.98 – 1.03 | 0.63 | 1.08 | 1.05 – 1.10 | <0.001 |
| Female vs. Male | 0.58 | 0.30 – 1.07 | 0.09 | 0.56 | 0.30 – 1.01 | 0.06 | 0.71 | 0.36 – 1.38 | 0.32 | 0.71 | 0.43 – 1.16 | 0.17 |
| Ever smoker vs. never smoker | 0.74 | 0.39 – 1.39 | 0.36 | 0.80 | 0.43 – 1.48 | 0.48 | 0.50 | 0.23 – 1.04 | 0.07 | 0.80 | 0.49 – 1.32 | 0.39 |
| Surge 2 vs. 1 | 1.31 | 0.69 – 2.49 | 0.40 | 1.48 | 0.79 – 2.76 | 0.22 | 0.91 | 0.44 – 1.82 | 0.78 | 1.35 | 0.81 – 2.24 | 0.25 |
| Hispanic vs. Non-Hispanic | 1.04 | 0.49 – 2.17 | 0.91 | 0.97 | 0.47 – 1.98 | 0.94 | 0.96 | 0.43 – 2.10 | 0.91 | 0.90 | 0.50 – 1.62 | 0.72 |
| n = 377 | 30-day mortality | 60-day mortality | ICU transfer | Overall Survival | ||||||||
| OR | 95% CI | P | OR | 95% CI | P | OR | 95% CI | P | HR | 95% CI | P | |
| D-dimer (ug/mL) at Day 1 | 1.00 | 1.00 – 1.00 | 0.36 | 1.00 | 1.00 – 1.00 | 0.18 | 1.00 | 1.00 – 1.00 | 0.02 | 1.00 | 1.00 – 1.00 | 0.31 |
| Slope of D-dimer | 1.00 | 1.00 – 1.00 | 0.82 | 1.00 | 1.00 – 1.00 | 0.68 | 1.01 | 1.00 – 1.01 | 0.01 | 1.00 | 1.00 – 1.00 | 0.94 |
| Age | 1.09 | 1.06 – 1.12 | <0.001 | 1.08 | 1.05 – 1.11 | <0.001 | 1.01 | 0.99 – 1.03 | 0.55 | 1.07 | 1.05 – 1.10 | <0.001 |
| Female vs. Male | 0.55 | 0.29 – 1.05 | 0.07 | 0.57 | 0.30 – 1.05 | 0.07 | 0.99 | 0.53 – 1.84 | 0.98 | 0.67 | 0.40 – 1.12 | 0.13 |
| Ever smoker vs. never smoker | 0.69 | 0.35 – 1.32 | 0.26 | 0.73 | 0.39 – 1.36 | 0.33 | 0.60 | 0.29 – 1.18 | 0.15 | 0.73 | 0.44 – 1.24 | 0.25 |
| Surge 2 vs. 1 | 0.82 | 0.43 – 1.54 | 0.54 | 1.01 | 0.55 – 1.84 | 0.97 | 0.62 | 0.31 – 1.21 | 0.17 | 0.98 | 0.60 – 1.60 | 0.93 |
| Hispanic vs. Non-Hispanic | 0.74 | 0.33 – 1.61 | 0.47 | 0.80 | 0.37 – 1.66 | 0.56 | 1.07 | 0.53 – 2.15 | 0.86 | 0.80 | 0.42 – 1.52 | 0.49 |
| n = 429 | 30-day mortality | 60-day mortality | ICU transfer | Overall Survival | ||||||||
| OR | 95% CI | P | OR | 95% CI | P | OR | 95% CI | P | HR | 95% CI | P | |
| Platelet count (1000/mm3) at Day 1 | 0.99 | 0.99 – 1.00 | 0.001 | 0.99 | 0.99 – 1.00 | <0.001 | 1.00 | 0.99 – 1.00 | 0.04 | 1.00 | 0.99 – 1.00 | 0.001 |
| Slope of platelet | 0.94 | 0.92 – 0.97 | <0.001 | 0.94 | 0.91 – 0.96 | <0.001 | 0.96 | 0.94 – 0.99 | 0.01 | 0.96 | 0.94 – 0.97 | <0.001 |
| Age | 1.06 | 1.04 – 1.08 | <0.001 | 1.06 | 1.04 – 1.08 | <0.001 | 1.00 | 0.98 – 1.02 | 0.83 | 1.05 | 1.03 – 1.07 | <0.001 |
| Female vs. Male | 0.55 | 0.31 – 0.97 | 0.04 | 0.57 | 0.32 – 0.99 | 0.05 | 0.77 | 0.43 – 1.37 | 0.38 | 0.78 | 0.50 – 1.21 | 0.26 |
| Ever smoker vs. never smoker | 0.92 | 0.52 – 1.63 | 0.79 | 0.98 | 0.56 – 1.72 | 0.95 | 0.62 | 0.32 – 1.17 | 0.14 | 0.88 | 0.57 – 1.37 | 0.58 |
| Surge 2 vs. 1 | 1.04 | 0.60 – 1.79 | 0.88 | 1.23 | 0.72 – 2.09 | 0.44 | 0.72 | 0.39 – 1.29 | 0.27 | 1.20 | 0.80 – 1.80 | 0.38 |
| Hispanic vs. Non-Hispanic | 1.34 | 0.67 – 2.63 | 0.40 | 1.40 | 0.72 – 2.71 | 0.31 | 1.29 | 0.64 – 2.59 | 0.47 | 1.14 | 0.67 – 1.93 | 0.63 |
| n = 431 | 30-day mortality | 60-day mortality | ICU transfer | Overall Survival | ||||||||
| OR | 95% CI | P | OR | 95% CI | P | OR | 95% CI | P | HR | 95% CI | P | |
| WBC count (1000/mm3) at Day 1 | 1.05 | 1.01 – 1.10 | 0.01 | 1.05 | 1.01 – 1.09 | 0.02 | 1.06 | 1.01 – 1.13 | 0.03 | 1.01 | 0.99 – 1.03 | 0.25 |
| Slope of WBC | 3.85 | 1.90 – 8.05 | <0.001 | 2.91 | 1.47 – 5.87 | 0.002 | 9.43 | 2.92 – 34.65 | <0.001 | 1.99 | 1.24 – 3.20 | 0.004 |
| Age | 1.07 | 1.05 – 1.10 | <0.001 | 1.07 | 1.04 – 1.09 | <0.001 | 1.00 | 0.98 – 1.02 | 0.84 | 1.06 | 1.04 – 1.08 | <0.001 |
| Female vs. Male | 0.51 | 0.28 – 0.89 | 0.02 | 0.52 | 0.30 – 0.89 | 0.02 | 0.90 | 0.49 – 1.63 | 0.72 | 0.68 | 0.44 – 1.05 | 0.09 |
| Ever smoker vs. never smoker | 0.78 | 0.44 – 1.37 | 0.39 | 0.85 | 0.49 – 1.45 | 0.55 | 0.66 | 0.34 – 1.27 | 0.22 | 0.76 | 0.49 – 1.18 | 0.22 |
| Surge 2 vs. 1 | 0.81 | 0.46 – 1.41 | 0.46 | 1.07 | 0.63 – 1.81 | 0.81 | 0.45 | 0.23 – 0.87 | 0.02 | 1.07 | 0.70 – 1.63 | 0.75 |
| Hispanic vs. Non-Hispanic | 0.89 | 0.45 – 1.74 | 0.74 | 0.91 | 0.47 – 1.72 | 0.77 | 0.93 | 0.46 – 1.86 | 0.85 | 0.87 | 0.51 – 1.50 | 0.63 |
ROC analyses
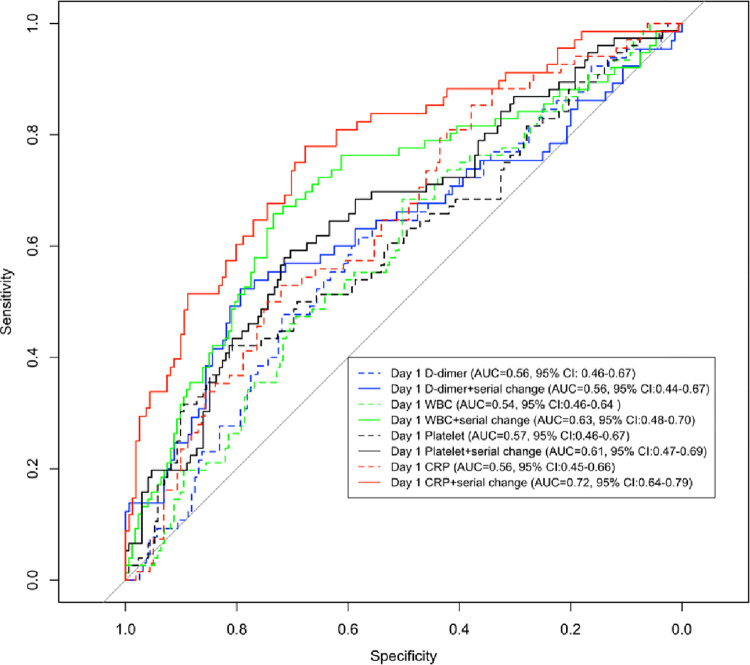
Incorporating serial changes in biomarkers provided incremental predictive values in all clinical outcomes, with the largest improvement in predicting the need for ICU transfer (Fig 3, S5 Table). Including the slope of CRP change since admission achieved the highest AUC and largest improvement in predicting ICU transfer (AUC = 0.72, 95% CI: 0.64–0.79) compared to the baseline model (AUC = 0.56, 95% CI: 0.45–0.66), suggesting potential clinical utility in triaging severe patients urgently in need of closer monitoring and more intensive care support.
Fig 3. ROC analyses for ICU transfer.
Area under the curve (AUC) for ICU transfer was calculated in each biomarker: D-dimer, WBC count, platelet count and CRP, for both models in incorporating 1) only biomarker level on day 1, and 2) serial changes over time and corresponding level on day 1, in combination with baseline covariates of age, gender, smoking status, ethnicity and COVID surge.
Discussion
While the majority of patients with COVID-19 have mild symptoms, those admitted to the hospital may experience rapid disease progression, requiring ventilatory support and ICU admission despite the increasing prevalence of vaccine administration and adjunctive therapies. Biomarkers that predict disease progression and clinical outcomes may facilitate the triage of patients with worsening disease in need of close monitoring and intensive care support, enabling efficient allocation of limited and precious healthcare resources [3]. To our knowledge, our results demonstrated for the first time that, in addition to the laboratory biomarkers measured on day 1 of admission, increases in CRP levels and WBC count, and decreases in platelet count are associated with escalation of inpatient care intensity and poor clinical outcomes among hospitalized patients with COVID-19.
The SOFA score, a mortality prediction score based on the degree of dysfunction of six organ systems, has been well adopted in COVID triage policies [26]. Consistent with prior studies, increased mSOFA score on day 1 of admission was associated with increased mortality among ICU patients [16, 21, 22]. Our study shows that elevated D-dimer, CRP, WBC count and decreased platelet count on day 1 of admission were also associated with higher risks of mortality and ICU transfer. Prior studies show that higher D-dimer level on day 1 of admission predicts higher in-hospital mortality [13, 27]. CRP, an acute inflammatory biomarker, has been widely used for assessing the severity of inflammatory and infectious conditions. Our study confirmed other small cohort studies and meta-analyses that conjectured that an elevated CRP level is associated with higher mortality and provided novel findings regarding ICU transfer [9, 28–30].
However, very few studies investigated the prognostic and predictive effects of serial biomarkers in hospitalized COVID-19 patients. Serial measurements of time-varying cardiac biomarkers, including high-sensitivity cardiac troponin (hsTnI) and NT-proBNP, predict a higher mortality rate and show an enhanced association with OS compared to the baseline level on day 1 of admission among hospitalized patients [31, 32]. Our study is innovative in characterizing the predictive value of serial measurement in D-dimer, CRP, WBC count, and platelet count on mortality, ICU transfer and OS among hospitalized COVID-19 patients. Incorporating serial changes in these biomarkers improves the model performance in all clinical outcomes, with the highest and largest increase in predicting ICU transfer using CRP. The higher mortality in patients with an increasing trend in WBC count can be due to treatment effects, generalized systemic inflammation and secondary bacterial infection, and the biological mechanisms merit further investigation. Taken together, our results suggested that consecutive measurements of these biomarkers beyond baseline concentrations are associated with COVID-19 progression course and clinical outcomes in hospitalized patients.
Our study has several limitations. First, this is a single-center retrospective study based on a sample of hospitalized COVID-19 patients admitted from March and June 2020 and January to March 2021, implying that the estimates may not be transferable with less virulent strains and in the vaccine era. Second, while there are missing data for CRP and D-dimer, it doesn’t change our main conclusion that serial change in CRP predicts ICU escalation; indeed, this only further strengthens the association as potential bias introduced could only be towards the null. Third, detailed immunosuppressive treatment information is not available in this work. However, the surge indicator may serve as a surrogate for steroid usage, and it was adjusted in the analyses. Fourth, as our study population was predominantly composed of White, non-Hispanic patients, our findings should be further tested in a larger, more diverse population in a prospective study for improved generalizability. While we do not have detailed microbiology data on the patients included in this study, it is certainly possible that some of the findings may be attributable to secondary bacterial infection. Our study was limited by its retrospective design, and some data were unavailable from the electronic medical records. Additionally, there were inconsistencies in the missingness of biomarkers at different time points, potentially reducing the accuracy of our estimates and model performances.
In conclusion, we have demonstrated that elevated mSOFA score, D-dimer, CRP, WBC count and decreased platelet count on day 1 of admission were associated with higher 30- and 60-day mortality, higher ICU transfer and more overall mortality among hospitalized COVID-19 patients. Serial CRP, WBC count, and platelet count are readily available in clinical practice and provide additional predictive value for disease progression and clinical outcomes, supporting the use of serial laboratory biomarkers in the triage and monitoring of hospitalized patients with COVID-19.
Supporting information
(PDF)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Abbreviations
- COVID-19
Coronavirus disease 2019
- ICU
Intensive Care Unit
- WBC
White blood cell
- CRP
C-reactive protein
- mSOFA
Modified sequential organ failure
- AUC
Area Under the Curve
- ROC
Receiver Operating characteristic Curve
- OR
Odds Ratio
- HR
Hazard Ratio
- CI
Confidence Interval
- OS
Overall Survival
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
XW, YL and DCC are supported by NIH grants 5U01CA209414 and P30ES000002. JWA is supported by a KL2 award (an appointed KL2 award) from Harvard Catalyst | The Harvard Clinical and Translational Science Center (National Center for Advancing Translational Sciences, National Institutes of Health Award KL2 TR002542). The content is solely the responsibility of the authors and does not necessarily represent the official views of Harvard Catalyst, Harvard University and its affiliated academic healthcare centers, or the National Institutes of Health. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Fricke-Galindo I. and Falfan-Valencia R., Genetics Insight for COVID-19 Susceptibility and Severity: A Review. Front Immunol, 2021. 12: p. 622176. doi: 10.3389/fimmu.2021.622176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Oran D.P. and Topol E.J., Prevalence of Asymptomatic SARS-CoV-2 Infection: A Narrative Review. Ann Intern Med, 2020. 173(5): p. 362–367. doi: 10.7326/M20-3012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Arnold D.T., et al., Predicting outcomes of COVID-19 from admission biomarkers: a prospective UK cohort study. Emerg Med J, 2021. 38(7): p. 543–548. doi: 10.1136/emermed-2020-210380 [DOI] [PubMed] [Google Scholar]
- 4.Wu F., et al., A new coronavirus associated with human respiratory disease in China. Nature, 2020. 579(7798): p. 265–269. doi: 10.1038/s41586-020-2008-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Battaglini D., et al., Laboratory Biomarkers for Diagnosis and Prognosis in COVID-19. Front Immunol, 2022. 13: p. 857573. doi: 10.3389/fimmu.2022.857573 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Park M., et al., Usefulness of KL-6 for Predicting Clinical Outcomes in Hospitalized COVID-19 Patients. Medicina (Kaunas), 2022. 58(10). doi: 10.3390/medicina58101317 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Park M., et al., Prognostic Utility of Procalcitonin, Presepsin, and the VACO Index for Predicting 30-day Mortality in Hospitalized COVID-19 Patients. Ann Lab Med, 2022. 42(4): p. 406–414. doi: 10.3343/alm.2022.42.4.406 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Park M., et al., Soluble ST2 as a Useful Biomarker for Predicting Clinical Outcomes in Hospitalized COVID-19 Patients. Diagnostics (Basel), 2023. 13(2). doi: 10.3390/diagnostics13020259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Malik P., et al., Biomarkers and outcomes of COVID-19 hospitalisations: systematic review and meta-analysis. BMJ Evid Based Med, 2021. 26(3): p. 107–108. doi: 10.1136/bmjebm-2020-111536 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhao Q., et al., Lymphopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A systemic review and meta-analysis. Int J Infect Dis, 2020. 96: p. 131–135. doi: 10.1016/j.ijid.2020.04.086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Huang I. and Pranata R., Lymphopenia in severe coronavirus disease-2019 (COVID-19): systematic review and meta-analysis. J Intensive Care, 2020. 8: p. 36. doi: 10.1186/s40560-020-00453-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lippi G. and Plebani M., Procalcitonin in patients with severe coronavirus disease 2019 (COVID-19): A meta-analysis. Clin Chim Acta, 2020. 505: p. 190–191. doi: 10.1016/j.cca.2020.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang L., et al., D-dimer levels on admission to predict in-hospital mortality in patients with Covid-19. J Thromb Haemost, 2020. 18(6): p. 1324–1329. doi: 10.1111/jth.14859 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yu B., et al., Evaluation of variation in D-dimer levels among COVID-19 and bacterial pneumonia: a retrospective analysis. J Thromb Thrombolysis, 2020. 50(3): p. 548–557. doi: 10.1007/s11239-020-02171-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wu C., et al., Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med, 2020. 180(7): p. 934–943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhou F., et al., Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet, 2020. 395(10229): p. 1054–1062. doi: 10.1016/S0140-6736(20)30566-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ruan Q., et al., Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med, 2020. 46(5): p. 846–848. doi: 10.1007/s00134-020-05991-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Guan W.J., et al., Comorbidity and its impact on 1590 patients with COVID-19 in China: a nationwide analysis. Eur Respir J, 2020. 55(5). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Paranjpe I., et al., Clinical Characteristics of Hospitalized Covid-19 Patients in New York City. medRxiv, 2020. [Google Scholar]
- 20.Goyal P., et al., Clinical Characteristics of Covid-19 in New York City. N Engl J Med, 2020. 382(24): p. 2372–2374. doi: 10.1056/NEJMc2010419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yang Z., et al., The prognostic value of the SOFA score in patients with COVID-19: A retrospective, observational study. Medicine (Baltimore), 2021. 100(32): p. e26900. doi: 10.1097/MD.0000000000026900 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Raschke R.A., et al., Discriminant Accuracy of the SOFA Score for Determining the Probable Mortality of Patients With COVID-19 Pneumonia Requiring Mechanical Ventilation. JAMA, 2021. 325(14): p. 1469–1470. doi: 10.1001/jama.2021.1545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Vincent J.L., et al., Use of the SOFA score to assess the incidence of organ dysfunction/failure in intensive care units: results of a multicenter, prospective study. Working group on "sepsis-related problems" of the European Society of Intensive Care Medicine. Crit Care Med, 1998. 26(11): p. 1793–800. doi: 10.1097/00003246-199811000-00016 [DOI] [PubMed] [Google Scholar]
- 24.Grissom C.K., et al., A modified sequential organ failure assessment score for critical care triage. Disaster Med Public Health Prep, 2010. 4(4): p. 277–84. doi: 10.1001/dmp.2010.40 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Alladina J., et al., Plasma Soluble Suppression of Tumorigenicity-2 Associates with Ventilator Liberation in Acute Hypoxemic Respiratory Failure. Am J Respir Crit Care Med, 2021. 203(10): p. 1257–1265. doi: 10.1164/rccm.202005-1951OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Antommaria A.H.M., et al., Ventilator Triage Policies During the COVID-19 Pandemic at U.S. Hospitals Associated With Members of the Association of Bioethics Program Directors. Ann Intern Med, 2020. 173(3): p. 188–194. doi: 10.7326/M20-1738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Milenkovic M., et al., D-dimer, CRP, PCT, and IL-6 Levels at Admission to ICU Can Predict In-Hospital Mortality in Patients with COVID-19 Pneumonia. Oxid Med Cell Longev, 2022. 2022: p. 8997709. doi: 10.1155/2022/8997709 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang L., C-reactive protein levels in the early stage of COVID-19. Med Mal Infect, 2020. 50(4): p. 332–334. doi: 10.1016/j.medmal.2020.03.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Qin C., et al., Dysregulation of Immune Response in Patients With Coronavirus 2019 (COVID-19) in Wuhan, China. Clin Infect Dis, 2020. 71(15): p. 762–768. doi: 10.1093/cid/ciaa248 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu F., et al., Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19. J Clin Virol, 2020. 127: p. 104370. doi: 10.1016/j.jcv.2020.104370 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tawiah K., et al., Serial cardiac biomarkers for risk stratification of patients with COVID-19. Clin Biochem, 2022. 107: p. 24–32. doi: 10.1016/j.clinbiochem.2022.06.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lionte C., et al., Inflammatory and Cardiac Biomarkers in Relation with Post-Acute COVID-19 and Mortality: What We Know after Successive Pandemic Waves. Diagnostics (Basel), 2022. 12(6). doi: 10.3390/diagnostics12061373 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(PDF)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.