Abstract
Male infertility is common and complex, presenting a wide range of heterogeneous phenotypes. Although about 50% of cases are estimated to have a genetic component, the underlying cause often remains undetermined. Here, from whole-exome sequencing on samples from 168 infertile men with asthenoteratozoospermia due to severe sperm flagellum, we identified homozygous ZMYND12 variants in four unrelated patients. In sperm cells from these individuals, immunofluorescence revealed altered localization of DNAH1, DNALI1, WDR66, and TTC29. Axonemal localization of ZMYND12 ortholog TbTAX-1 was confirmed using the Trypanosoma brucei model. RNAi knock-down of TbTAX-1 dramatically affected flagellar motility, with a phenotype similar to the sperm from men bearing homozygous ZMYND12 variants. Co-immunoprecipitation and ultrastructure expansion microscopy in T. brucei revealed TbTAX-1 to form a complex with TTC29. Comparative proteomics with samples from Trypanosoma and Ttc29 KO mice identified a third member of this complex: DNAH1. The data presented revealed that ZMYND12 is part of the same axonemal complex as TTC29 and DNAH1, which is critical for flagellum function and assembly in humans, and Trypanosoma. ZMYND12 is thus a new asthenoteratozoospermia-associated gene, bi-allelic variants of which cause severe flagellum malformations and primary male infertility.
Research organism: Human
Introduction
Male infertility is one of the most challenging health issues that will face us in the near future. Recent decades have been marked by a significant and constant deterioration of quantitative and qualitative sperm parameters in both humans and animals, demonstrating the detrimental impact of recent environmental changes (Rolland et al., 2013). However, environmental factors are not the sole cause of male infertility, which is often multifactorial. Among the various possible causes, it is estimated that a genetic component, alone or in association, can be found in about half of cases (Oud et al., 2022).
Spermatozoa are the most highly differentiated and specialized human cells, as reflected by the 2300 genes necessary for normal spermatogenesis (Lu et al., 2019). The complex process of spermatogenesis is divided into three phases: proliferation and differentiation of spermatogonia, meiosis, and the final critical step, spermiogenesis. During spermiogenesis, haploid round spermatids undergo an extraordinary series of transformations to acquire the specific morphological features necessary for them to competently fertilize an egg. The stages in spermiogenesis include nuclear compaction, acrosome formation, and flagellum biogenesis (O’Donnell, 2014). Any defects in genes driving these differentiation steps can lead to severe functional or morphological sperm defects, and thus male infertility.
Interestingly, the prevalence of genetic defects is higher in conditions with severe sperm phenotypes (Coutton et al., 2015). Multiple Morphological Abnormalities of the sperm Flagellum (MMAF) is a condition associated with extreme morphological sperm defects characterized by a mosaic of phenotypes. Thus, MMAF sperm cells may lack a flagellum or bear a short, irregular, or coiled flagellum. Because of these characteristics, MMAF causes total asthenozoospermia with almost no progressive sperm (Touré et al., 2021). At the ultrastructural level, MMAF is associated with severe disorganization of both axonemal and peri-axonemal structures (Nsota Mbango et al., 2019). Approximately 40 genes have been strongly or definitively associated with the MMAF phenotype so far (Wang et al., 2020). However, this high genetic heterogeneity means the prevalence of individual variants of each gene remains low, and the underlying genetic component remains unknown for about half of affected individuals. It is therefore necessary to pursue the identification of new candidate MMAF genes to increase the diagnostic yield and improve our understanding of the pathophysiological mechanisms inducing this sperm phenotype. In addition, the identification of MMAF genes provides invaluable clues to the molecular mechanisms underlying sperm flagellum biogenesis. Therefore, efforts are continuously being deployed to decipher the precise organization, localization, and interactions of each protein encoded by these genes within the flagellum.
Here, following analysis of an initial cohort of 167 MMAF individuals by whole-exome sequencing (WES), we identified truncating homozygous variants in ZMYND12 in three unrelated individuals. A fourth individual with a similar mutation was subsequently identified in a Chinese cohort. In this paper, we present a thorough investigation of the role of this new gene by studying its ortholog, TbTAX-1, in Trypanosoma, an evolutionarily distant model sharing a highly conserved flagellar structure. We first showed the importance of ZMYND12 for the axonemal structure and function of the flagellum. Subsequently, we shed light on ZMYND12 function in the flagellum using the TbTAX-1 protist model and Tttc29 KO mice. Results formally demonstrated that ZMYND12 interacts directly with DNAH1 and TTC29 (two known MMAF-related proteins) within the same axonemal complex.
Results
WES identified homozygous truncating variants in ZMYND12 in MMAF individuals
In our cohort of 167 MMAF individuals, we previously identified 83 individuals (49.7%) with harmful gene variants in known MMAF-related genes. Reanalysis of the remaining negative exomes identified three unrelated MMAF individuals (ZMYND12_1–3) (Figure 1A, Table 1) carrying homozygous variants in ZMYND12 (Zinc Finger MYND-Type Containing 12, NM_032257.5). This gene was not previously associated with any pathology. Two individuals (ZMYND12_1 and ZMYND12_3) originated from Tunisia, and one individual (ZMYND12_2) was of Iranian origin.
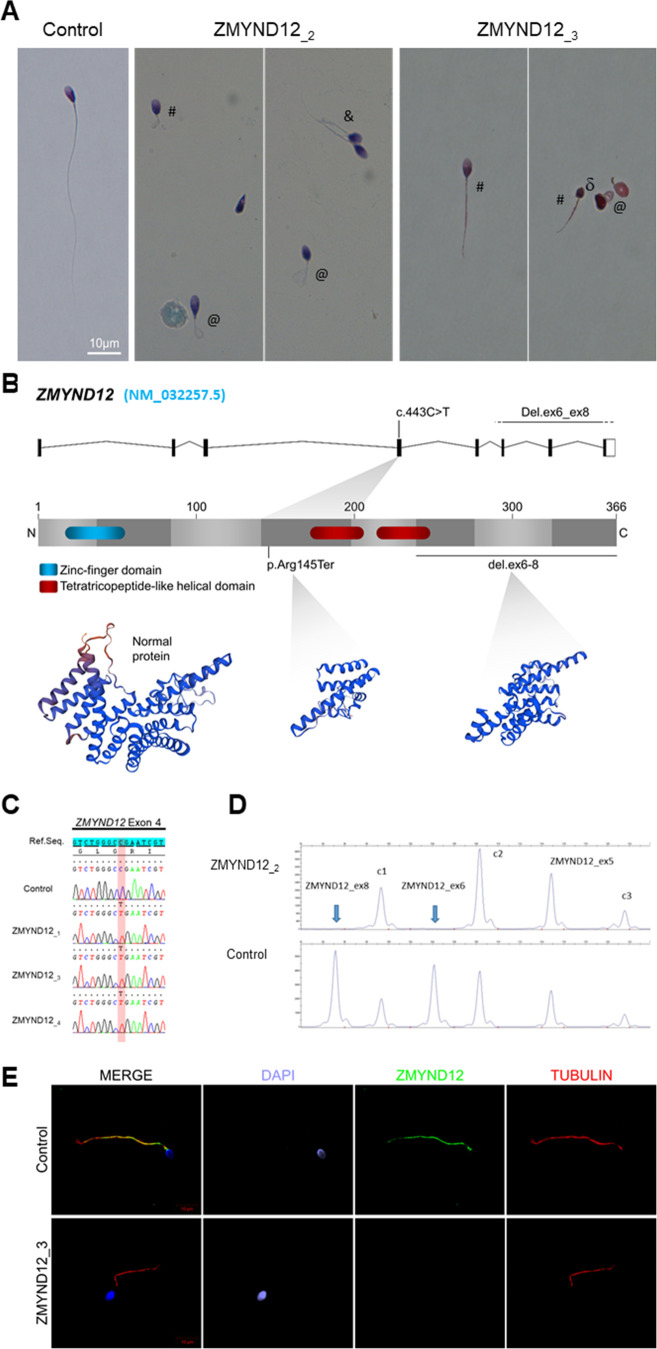
Figure 1. Morphology of normal and ZMYND12 mutant spermatozoa and variants identified in ZMYND12 individuals.
(A) Light microscopy analysis of spermatozoa from fertile control individuals and from individuals ZMYND12_2 and ZMYND12_3. Most spermatozoa from ZMYND12 individuals had short (#), coiled (@), or irregular caliber (&) flagella. Head malformations were also observed (δ). (B) Location of the variants identified within the ZMYND12 gene (NM_032257.5) and protein according to UniprotKB (Q9H0C1). The blue box represents a Zinc Finger domain and the orange boxes represent TPR domains (tetratricopeptide repeat). (C) Sanger sequencing electropherograms indicating the homozygous state of the c.433C>T variant located in exon 4 in the three individuals ZMYND12_1,3 and 4. The substituted nucleotide is highlighted in red. Variants are annotated in line with HGVS recommendations. (D) Confirmation of ZMYND12 exon deletion by MLPA. MLPA profiles obtained for control and ZMYND12_2 individuals. Each set contains three control probes for normalization purposes (c1–c3) and three ZMYND12-specific probes. Blue arrows indicate homozygous deletion of exons 6 and 8 in the ZMYND12 gene. ZMYND12 exon 5 is not included in the deletion. (E) Sperm cells from a fertile control individual and from individual ZMYND12_3 – carrier of the c.433C>T variant – were stained with anti-ZMYND12 (green) and anti-acetylated tubulin (red) antibodies. Sperm nuclear DNA was counterstained with DAPI (4′,6-diamidino-2-phenylindole) (blue). In control sperm, ZMYND12 immunostaining was present along the whole length of the flagellum; no staining was observed in sperm cells from individual ZMYND12_3. Scale bars: 10 μm.
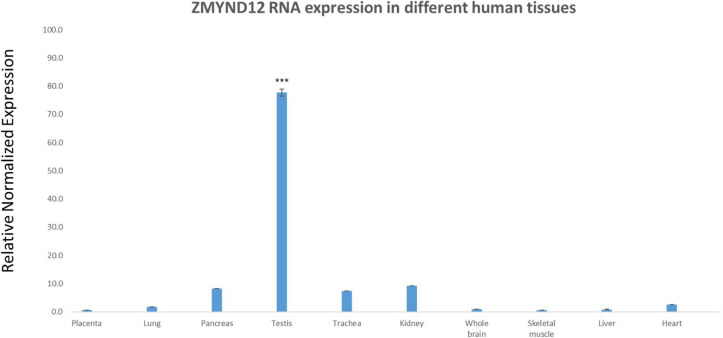
Figure 1—figure supplement 1. Relative mRNA expression of human ZMYND12 transcripts.
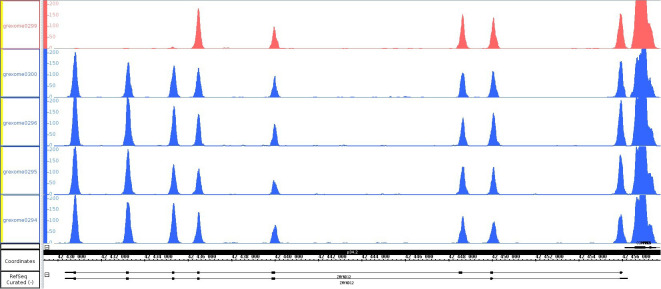
Figure 1—figure supplement 2. Depth of coverage at the ZMYND12 locus for the ZMYND12_2 individual, carrier of a homozygous deletion of exons 6–8, and four other unrelated Multiple Morphological Abnormalities of the sperm Flagellum (MMAF) individuals.
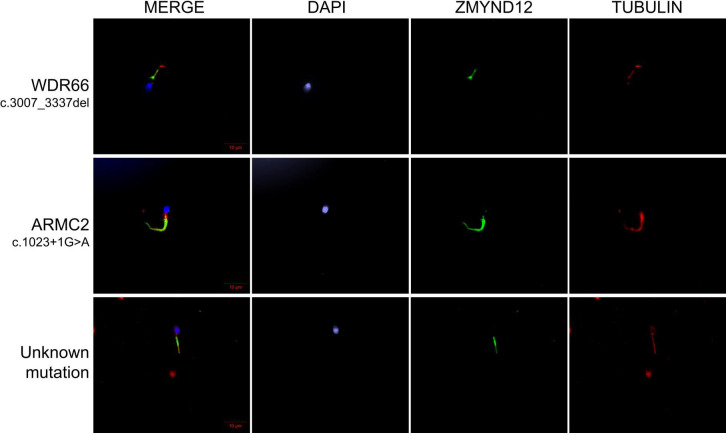
Figure 1—figure supplement 3. ZMYND12 immunostaining in human spermatozoa from Multiple Morphological Abnormalities of the sperm Flagellum (MMAF) individuals carriers of pathogenic variants in ARMC2 and WDR66 genes, or with unknwon genetic cause.
Table 1. Detailed semen parameters in the four Multiple Morphological Abnormalities of the sperm Flagellum (MMAF) individuals harboring a ZMYND12 variant.
| ZMYND12 mutated individuals | Semen parameters | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Individuals | Homozygous ZMYND12 variants |
Sperm volume (ml) | Sperm concentration (106/ml) | Total motility 1 hr | Vitality | Normal spermatozoa | Absent flagella | Short flagella | Coiled flagella | Bent flagella | Flagella of irregular caliber | Tapered head | Thin head | Micro-cephalic | Macro-cephalic | Multiple heads | Abnormal base | Abnormal acrosomal region |
| ZMYND12_1 | c.433C>T | 2 | 6.5 | 4 | 70 | 0 | 35 | 48 | 10 | 0 | 34 | 22 | 2 | 2 | 4 | 6 | 20 | 80 |
| ZMYND12_2 | Del ex6-8 | 2.5 | 4.25 | 2 | NA | 0 | NA | 72 | NA | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| ZMYND12_3 | c.433C>T | 3 | 34 | 2 | 50 | 0 | 20 | 20 | 28 | 0 | 46 | 22 | 6 | 8 | 2 | 2 | 24 | 88 |
| ZMYND12_4 | c.433C>T | 2.6 | 0.1 | 0 | NA | NA | NA | 70 | NA | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| Reference limits * | 1.5 (1.4–1.7) | 15 (12–16) | 40 (38–42) | 58 (55–63) | 23 (20–26) | 5 (4–6) | 1 (0–2) | 17 (15–19) | 13 (11–15) | 2 (1–3) | 3 (2–4) | 14 (12–16) | 7 (5–9) | 1 (0–2) | 2 (1–3) | 42 (39–45) | 60 (57–63) | |
Values are percentages unless specified otherwise. NA: not available.
Reference limits (5th centiles and their 95% confidence intervals) according to World Health Organization (WHO) standards (Cooper et al., 2010) and the distribution range of morphologically normal spermatozoa observed in 926 fertile individuals (Auger et al., 2016).
ZMYND12 is located on chromosome 1 and contains eight exons encoding the Zinc Finger MYND domain-containing protein 12, a predicted 365-amino acid protein (Q9H0C1). According to data from GTEx, ZMYND12 is strongly expressed in the testis and is associated with cilia and flagella (Yamamoto et al., 2021; Yamamoto et al., 2006). Moreover, quantitative single-cell RNA sequencing datasets from human adult testis (ReproGenomics Viewer) (Darde et al., 2019) indicate abundant expression in germ cells from zygotene spermatocyte to late spermatid stages. This expression pattern suggests a role in sperm cell differentiation and/or function. ZMYND12 protein was also detected at significant levels in the human sperm proteome (Wang et al., 2013), but at very low levels in human airway cilia (Blackburn et al., 2017). We performed quantitative real-time RT-PCR (RT-qPCR) experiments on human tissue panels (Figure 1—figure supplement 1), and results confirmed that expression of ZMYND12 mRNA is predominant in testis as compared with the other tissues tested.
A stop-gain variant c.433C>T; p.Arg145Ter (NM_032257.5, GRCh38 g.chr1:42440017G>A, rs766588568), located in ZMYND12 exon 4 (Figure 1B), was identified in individuals ZMYND12_1 and ZMYND12_3. Subsequently, a Chinese MMAF subject harboring the same homozygous stop-gain variant in ZMYND12 (ZMYND12_4) was identified in the cohort collected by the Human Sperm Bank of West China Second University Hospital of Sichuan University hospital (Figure 1, Table 1). This c.433C>T variant is present in the Genome Aggregation Database (gnomAD v2.1.1, http://gnomad.broadinstitute.org/variant/1-42905688-G-A) database with a minor allele frequency of 3.391e−05. This nonsense variant, identified by exome sequencing, was confirmed by Sanger sequencing in all four individuals (Figure 1C).
Using the ExomeDepth software package (Plagnol et al., 2012), individual ZMYND12_2, was identified based on a homozygous deletion removing ZMYND12 exons 6–8 (Figure 1—figure supplement 2). We confirmed the presence of the homozygous deletion in the affected individual by custom MLPA (Figure 1D). The deletion identified was not listed in public databases, including DECIPHER (https://www.deciphergenomics.org/) and the Database of Genomic Variants (http://dgv.tcag.ca/dgv/app/home).
For the individuals carrying ZMYND12 mutations, analysis of control databases revealed no low-frequency variants in other genes reported to be associated with cilia, flagella, or male fertility. We therefore focused our study on ZMYND12, which appeared as the best MMAF candidate gene. All of the ZMYND12 variants identified here have been deposited in ClinVar under reference SUB12540308.
ZMYND12 is located in the sperm flagellum, its variants cause severe axonemal disorganization
To further investigate the pathogenicity of the ZMYND12 variants identified, we examined the distribution of the ZMYND12 protein in control and mutated sperm cells by immunofluorescence (IF). Due to sample availability, these analyses were only carried out for individual ZMYND12_3 – carrier of the recurrent stop-gain variant c.433C>T. In sperm from control individuals, ZMYND12 antisera decorated the full length of the sperm flagellum (Figure 1E). Conversely, and in line with a truncation or degradation of the mutated protein, ZMYND12 was totally absent from all sperm cells from individual ZMYND12_3 (Figure 1E). Importantly, ZMYND12 was detected in sperm cells from MMAF individuals bearing mutations in other genes (ARMC2 and WDR66) or with unknown genetic causes (Figure 1—figure supplement 3). Thus, the absence of ZMYND12 is not a common feature of the MMAF phenotype, and is specifically associated with ZMYND12 variants.
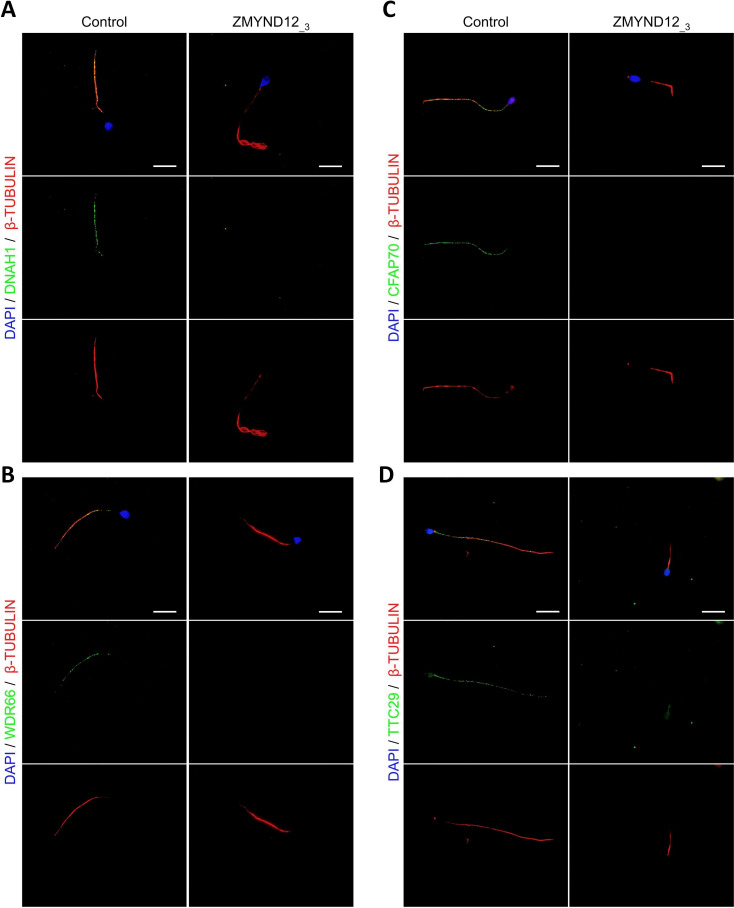
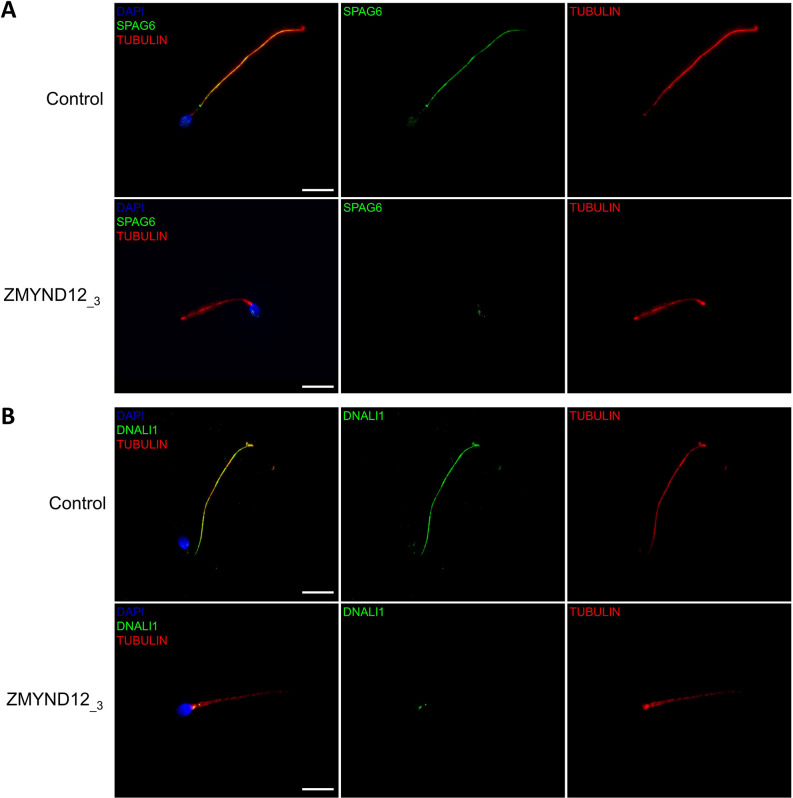
To explore the ultrastructural defects induced by ZMYND12 variants in human sperm cells, we next studied the presence of proteins present in various axonemal and peri-axonemal substructures by IF. The following proteins were investigated: AKAP4 (fibrous sheath, FS), CFAP70 (axoneme-binding protein with unknown precise location), DNAI1 (outer arm intermediate chain), DNALI1 (inner arm light intermediate chain), DNAH1 (inner arm heavy chain), DNAH8 (outer arm heavy chain), DNAH17 (outer arm heavy chain), GAS8 (Nexin-Dynein Regulatory Complex, N-DRC), RSPH1 (radial spoke, RS), SPAG6 (central pair complex, CPC), WDR66/CFAP251 (calmodulin- and spoke-associated complex, CSC), and TTC29 (axoneme-binding protein with unknown precise location). In sperm cells from ZMYND12_3, we first observed a total lack of SPAG6 staining (Figure 2—figure supplement 1A), suggesting severe CPC defects. This type of defect is a common feature of MMAF spermatozoa (Coutton et al., 2015). In addition, no immunostaining for DNAH1, DNALI1, or WDR66 was detected in sperm from individual ZMYND12_3 (Figure 2A, B and Figure 2—figure supplement 1B). These results suggested a strong disorganization of the IDAs (Inner dynein arms) and the CSC. ZMYND12 could therefore be a key axonemal component responsible for stability of these structures.
Figure 2. Altered immunostaining for DNAH1, WDR66, CFAP70, and TTC29 in the presence of ZMYND12 variants.
Immunofluorescence experiments were performed using sperm cells from control individuals and from individual ZMYND12_3 carrying the nonsense variant c.433C>T. Although tubulin staining remained detectable, sperm cells from individual ZMYND12_3 showed no immunostaining for (A) DNAH1, (B) WDR66, (C) CFAP70, and (D) TTC29. Scale bars: 10 µm.
Figure 2—figure supplement 1. Altered immunostaining for SPAG6 and DNALI1 in the presence of ZMYND12 variants.
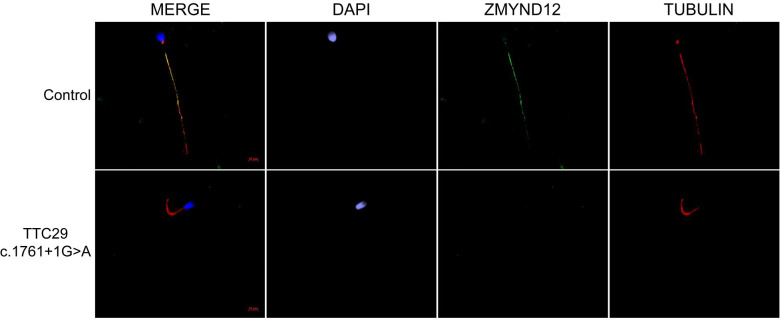
Figure 2—figure supplement 2. ZMYND12 is absent from sperm cells from TTC29 individuals.
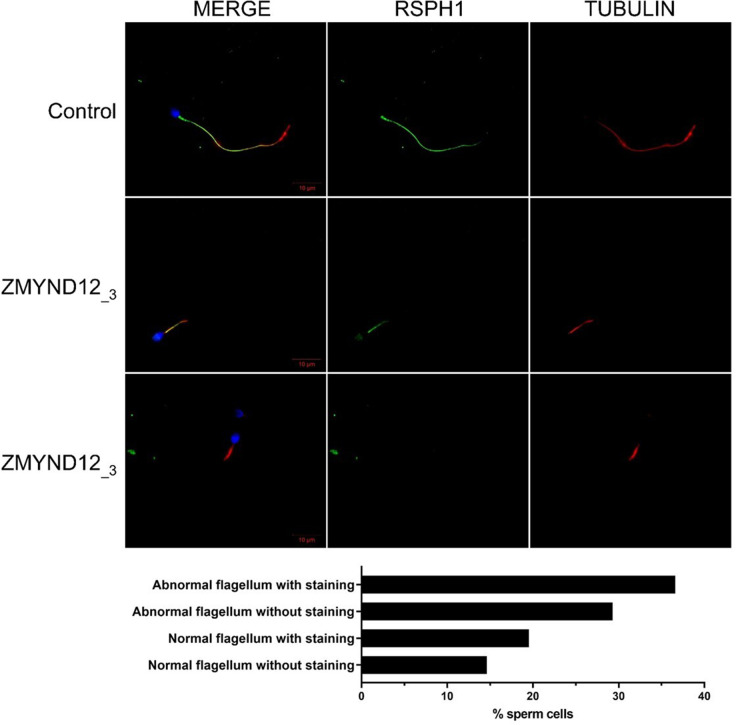
Figure 2—figure supplement 3. Radial spokes are inconstantly affected in ZMYND12 individuals.
Figure 2—figure supplement 4. Immunostaining for AKAP4, DNAH2, DNAH8, DNAH17, DNAI1, and GAS8 is not affected by ZMYND12 mutation.
We subsequently examined the presence of TTC29 and CFAP70, two proteins with as yet incompletely characterized function and localization. In control sperm, TTC29 and CFAP70 immunostaining decorated the full length of the flagellum, but neither protein was detected in cells from individual ZMYND12_3 (Figure 2C, D). Intriguingly, ZMYND12 was undetectable in the sperm flagellum from an MMAF individual carrying a previously reported TTC29 splicing variant c.1761+1G>A (Lorès et al., 2019; Figure 2—figure supplement 2). Unfortunately, no sample from an individual carrying a bi-allelic CFAP70 variant could be analyzed. Nevertheless, the available data suggest that TTC29, CFAP70, and ZMYND12 may be physically or functionally associated within the same axonemal complex.
We also observed heterogeneous RSPH1 staining with all flagellar morphologies (Figure 2—figure supplement 3), indicating that RSs are inconsistently present in sperm cells from individual ZMYND12_3. In contrast, the immunostaining patterns for AKAP4, DNAH8, DNAH17, DNAI1, and GAS8 were comparable to those observed in control sperm cells. This result indicates that the FS, the outer dynein arms (ODAs), and the N-DRC were not directly affected by ZMYND12 variants (Figure 2—figure supplement 4). Transmission electron microscopy (TEM), which could provide direct evidence of ultrastructural defects, could not be performed due to the very low number of sperm cells available from ZMYND12 individuals.
TbTAX-1, the Trypanosoma brucei ortholog of ZMYND12, is an axoneme-associated protein involved in flagellar motility
To validate our candidate gene and better characterize ZMYND12 localization and function, we used the Trypanosoma model. Forward and reverse genetic approaches are well established for this model, and have largely contributed to the characterization of the molecular pathogenesis of a number of human diseases caused by defective cilia and/or flagella (Vincensini et al., 2011). BlastP analysis of the T. brucei genome database (Aslett et al., 2010) identified the putative ortholog of ZMYND12 as Tb927.9.10370. The T. brucei gene encodes TbTAX-1, a 363 amino acid protein previously identified by proteomics analyses as a flagellar protein, annotated as the inner dynein arm protein/p38 (FAP146) (Broadhead et al., 2006; Ralston et al., 2009). Human ZMYND12 (Q9H0C1) and TbTAX-1 share 29% protein identity and 48% similarity. Both proteins contain a TPR motif domain potentially involved in protein–protein interaction (D’Andrea and Regan, 2003; Figure 3—figure supplement 1).
RNAi knock-down of TbTAX-1 in the bloodstream form (BSF) of T. brucei is reported to lead to multi-flagellated cells, suggesting incomplete cell division (Broadhead et al., 2006). This phenotype is described when proteins directly or indirectly involved in flagellar motility are knocked down in the BSF (Coutton et al., 2018; Ralston et al., 2011). In contrast, in the procyclic form, knock-down induces cell sedimentation due to motility defects (Baron et al., 2007).
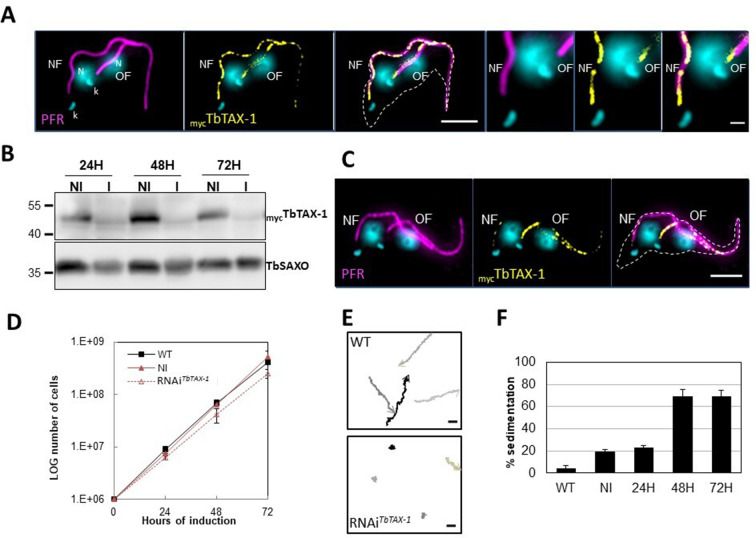
Here, to analyze the protein’s localization by IF on detergent-extracted cells (cytoskeletons, CSK), we generated a procyclic T. brucei cell line endogenously expressing myc-tagged TbTAX-1 (mycTbTAX-1). MycTbTAX-1 specifically localized to the flagellum, as shown by co-staining with the Paraflagellar Rod-2 protein (PFR2), a marker of the para-axonemal and axoneme-associated structure (PFR) (Figure 3A). Importantly, MycTbTAX-1 was found to associate with the axoneme fraction of the flagellum, with staining observed in the distal part of the flagellum, beyond the limit of PFR labeling (Figure 3A). Furthermore, mycTbTAX-1 labeling was observed in both the old flagellum (OF) and the new flagellum (NF), indicating that the protein is present at all stages of the cell cycle, in line with proteomics data reported for the Trypanosoma cell cycle (Crozier et al., 2018).
Figure 3. TbTAX-1 is an axoneme-associated protein; its knock-down leads to flagellar motility defects.
(A) Representative image of a post-mitotic detergent-extracted cell immunolabelled with anti-PFR2 (magenta) staining the paraflagellar rod (a para-axonemal structure) and anti-myc (yellow) to reveal mycTbTAX-1. The protein was detected on the axoneme in both the old flagellum (OF) and the new flagellum (NF). Immunolabeling indicated that the axonemal localization of mycTbTAX-1 extended distally along the flagellum beyond the region labelled by PFR2 (see zoom). The mitochondrial genome (kinetoplast, k) and nuclei (N) were stained with DAPI. The outline of the cell body is indicated by the dashed white line. Scale bars: 5 μm in whole-cell images; 1 μm in zoomed images. (B) Anti-myc western blot analysis of RNAiTbTAX-1 knock-down 24-, 48-, or 72-hr post-induction. mycTbTAX-1 protein levels were reduced in induced cells (I) compared to non-induced cells (NI); TbSAXO, a flagellum-specific protein, was used as loading control. (C) Representative image of a detergent-extracted post-mitotic cell after RNAiTbTAX-1 induction, showing a strong decrease in mycTbTAX-1 labeling in the NF compared to the OF. (D) Comparative T. brucei growth curves for wild-type (WT), non-induced (NI), and tetracycline-induced RNAiTbTAX-1 cell lines at 0-, 24-, 48-, and 72-hr post-induction. (E) Mobility tracking from video microscopy recordings of live cells: WT and RNAiTbTAX-1 after induction for 72 hr. The positions of individual cells are plotted at 0.28 s intervals; circles indicate start positions, arrowheads indicate end positions. A dramatic loss of progressive mobility was observed after TbTAX1 protein knock-down. Scale bars: 20 μm. (F) Sedimentation assays for T. brucei WT, non-induced (NI), and tetracycline-induced RNAiTbTAX-1 at 24-, 48-, and 72-hr post-induction. The percentage of cells sedimenting increased from 20 to 70% after depletion of the TbTAX-1 protein.
Figure 3—figure supplement 1. TbTAX-1 is the T. brucei ortholog of ZMYND12.
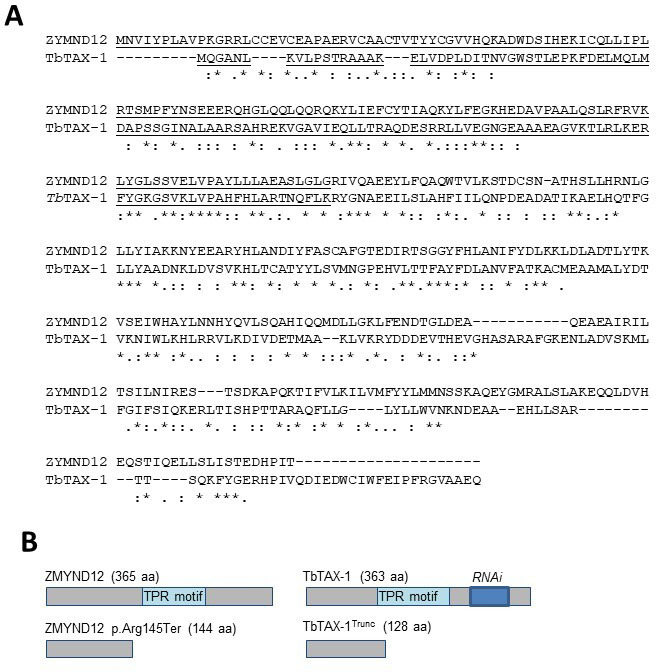
Figure 3—figure supplement 2. TbTAX-1 RNAi knock-down.
T. brucei TAX-1 is involved in flagellum motility
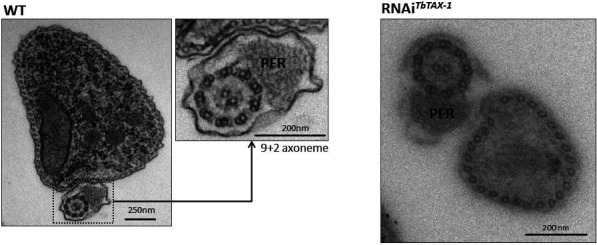
To assess the functional role of TbTAX-1 in the procyclic parasite model, we used a tetracycline-inducible RNA interference (RNAi) system (Wirtz et al., 1999) to knock-down TbTAX-1 expression. Using the mycTbTAX-1/TbTAX-1RNAi T. brucei cell line (Figure 3), we observed the effects of TbTAX-1 knock-down following the addition of tetracycline. Western blot analysis confirmed successful RNA interference, leading to a significant reduction in TbTAX-1 protein levels (Figure 3B). These results were confirmed by IF, where expression of MycTbTAX-1 was strongly reduced in the NF compared to the OF (Figure 3C). Comparison of TEM images of thin sections from wild-type (WT) and RNAi-induced cells after 7 days revealed no obvious differences in flagellum ultrastructure (Figure 3—figure supplement 2), and TbTAX-1 knock-down had no effect on cell growth (Figure 3D). However, video microscopy evidence pointed to a strong mobility defect (Videos 1 and 2). Tracking analysis (Figure 3E) indicated that reduced mobility was linked to problems with flagellar beating. In line with these observations, sedimentation assays showed that after induction for 48 hr, 70% of RNAi-induced cells had sedimented (Figure 3F).
Video 1. Video microscopy of wild-type (WT) cells.
Video 2. Video microscopy of RNAiTbTAX-1 cells 7 days post-induction.
The truncated TbTAX-1 protein is not associated with the flagellum and cannot rescue the motility defect induced by TbTAX-1 RNAi
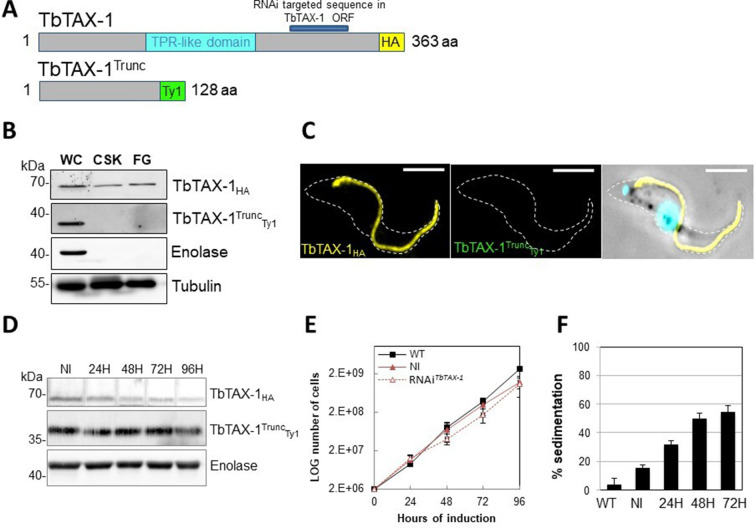
The p.Arg145Ter mutation of ZYMND12 identified in MMAF individuals could theoretically produce a truncated form of the protein (aa 1–144). To assess where a similar truncated protein – TbTAX-1 (aa 1–128) – would localize, we generated a TAX-1HA Trypanosoma cell line expressing the truncated form TbTAX-1TruncTy1 alongside the tagged full-length form. The relative sizes of the mutated, truncated ZMYND12 protein potentially encoded in human patients and the equivalent modified, truncated protein – TbTAX1Trunc – are presented in Figure 3—figure supplement 1B.
In this cell line, expression of TbTAX-1RNAi – specifically targeting the full-length protein and not the truncated form – was inducible (Figure 4A). Western blot analysis of fractionated samples (whole cells (WC), detergent-extracted cells or CSK, and the flagellum fraction (FG)), we localized the truncated TbTAX-1TruncTy1 protein (Figure 4B). The full-length TbTAX-1 protein was strongly associated with the axoneme, and was detected in the salt-extracted flagellum fraction. In contrast, the truncated TbTAX-1TruncTy1 form was detected neither in the flagellum nor in the CSK-associated fractions (Figure 4B). These results were confirmed by IF on CSK (Figure 4C). As expected, induction of TbTAX-1RNAi led to decreased TbTAX-1 levels, but had no effect on TbTAX-1TruncTy1 (Figure 4D). Cell growth was not affected (Figure 4E), and the sedimentation phenotype was still observed (Figure 4F). Thus, it appears that the N-terminal domain of TbTAX-1 is not involved in protein targeting and function, and cannot complement TbTAX-1 RNAi knock-down.
Figure 4. The truncated form of TbTAX-1 is not functional.
(A) Schematic representation of full-length TbTAX-1 and its truncated form. The TPR-like domain is illustrated in light blue, and the targeted RNA interference sequence in dark blue. (B) Western blot analysis of subcellular fractions to determine localization of TbTAX-1HA and its truncated form TbTAX-1TruncTy1. Enolase (cytoplasm) and tubulin (cytoskeleton) were used as loading controls. (C) Immunofluorescence labeling of detergent-extracted cytoskeletons to detect TbTAX-1HA (yellow) and TbTAX-1TruncTy1 (stained green, but not visible as the protein was eliminated during detergent extraction). Scale bars: 5 μm. (D) Western blot analysis of the impact of RNAiTbTAX-1 expression on TbTAX-1TruncTy1 levels. Enolase was used as a loading control. (E) Growth curves of wild-type (WT), non-induced (NI), and RNAiTbTAX-1-induced cells expressing TbTAX-1HA and TbTAX-1TruncTy1. (F) Sedimentation assays for T. brucei WT, non-induced (NI), and RNAiTbTAX-1-induced cells at 24, 48, and 72 hr.
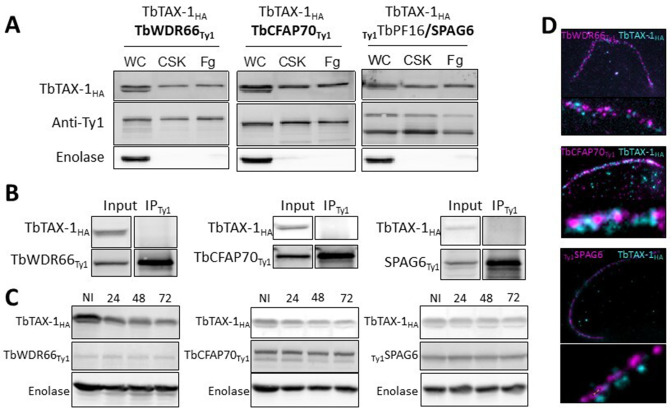
Figure 4—figure supplement 1. TbTAX-1 knock-down does not affect TbWDR66, TbCFAP70, or TbSPAG6.
TbTTC29 and TAX-1 belong to the same protein complex; TbTTC29 expression depends on TbTAX-1
As indicated above, in IF analyses of spermatozoa from individuals bearing a mutated ZYMND12, proteins CFAP70, WDR66, TTC29, and SPAG6 were undetected. To further study associations between these proteins, we generated T. brucei TbTAX-1HA/TbTAX-1RNAi background cell lines expressing their respective orthologs, that is, TbCFAP70Ty1 (Tb927.10.2380), TbWDR66Ty1 (Tb927.3.1670), TbTTC29Ty1 (Tb927.3.1990), and Ty1TbPF16/SPAG6 (Tb927.1.2670).
Western blotting revealed that CFAP70, WDR66, and SPAG6 were tightly associated with the flagellum fraction (Figure 4—figure supplement 1A). However, none of these proteins was pulled down with Tb-TAX1 upon co-IP with the anti-Ty1 antibody (Figure 4—figure supplement 1B). Furthermore, RNAi knock-down of TbTAX-1 expression did not alter their overall levels (Figure 4—figure supplement 1C). The lack of direct interaction between TbTAX-1 and these three proteins was further confirmed by ultrastructure expansion microscopy (U-ExM) IF (Gambarotto et al., 2019; Kalichava and Ochsenreiter, 2021), which provided no evidence of colocalization (Figure 4—figure supplement 1D).
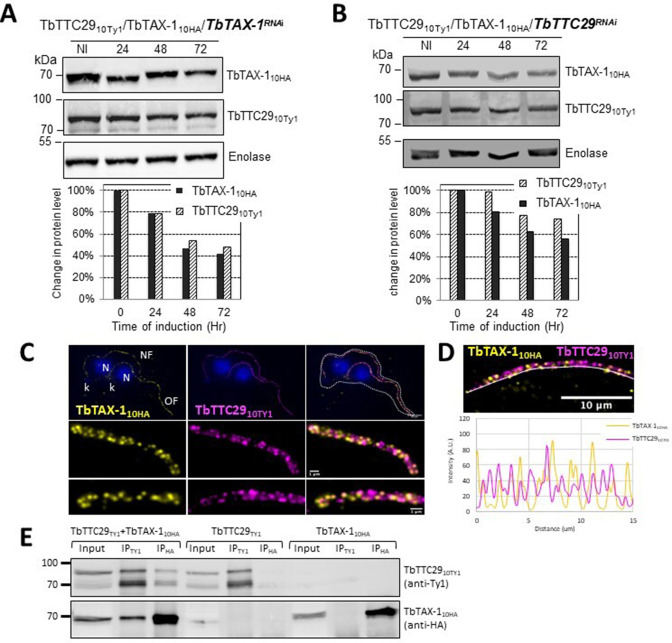
In contrast, decreased TbTAX-1 levels were associated with decreased TbTTC29 expression, and vice versa according to western blot quantification (Figure 5A and B). By IF, TbTAX-1 was also observed to co-localize with TbTTC29 on the axoneme (Figure 5C). However, analysis of U-ExM and confocal microscopy intensity peaks revealed the colocalization to be incomplete (Figure 5D). Nevertheless, IP of TbTTC29Ty1 from a TbTTC29Ty1/TbTAX-1HA cell line pulled down TbTAX-1HA and vice versa (Figure 5E). These results suggest that TbTAX-1 and TbTTC29 are part of the same protein complex.
Figure 5. TbTAX-1 and TbTTC29 are part of the same protein complex.
(A) Analysis of the impact of TbTAX-1 RNAi on TbTTC29Ty1 protein levels. Western blot (upper panel) and quantification (lower panel). (B) Analysis of the impact of TbTTC29 RNAi induction on TbTAX-1HA protein levels. Western blot (upper panel) and quantification (lower panel). (C) Ultrastructure expansion microscopy (U-ExM) analysis of TbTAX-1HA (yellow) and TbTTC29TY1 (purple) colocalization in a dividing cell with an old flagellum (OF) and a new flagellum (NF). Kinetoplast (k) and nuclei (N) were counterstained with Hoechst (blue). (D) Confocal image of U-ExM labeling of TbTAX-1HA (yellow) and TbTTC29TY1 (magenta) (upper panel). Lower panel: profile plot along the white line showing overlapping intensity peaks for TbTAX-1 and TbTTC29, confirming true colocalization. (E) Anti-Ty1 (IPTy1) or anti-HA (IPHA) co-immunoprecipitation assays in cell lines expressing both TbTAX-1HA and TbTTC29Ty1, or TbTTC29Ty1 alone or TbTAX-1HA alone to verify IP specificity. Proteins were detected simultaneously by western blotting using anti-Ty1 and anti-HA antibodies and fluorescent anti-mouse (detecting mouse anti-Ty1) and anti-rabbit (detecting rabbit anti-HA) secondary antibodies. IP assays on cell lines expressing only one or the other tagged protein did not pull down the other tagged protein.
The co-immunoprecipitated proteins were further analyzed by mass spectrometry. Comparative analysis of the significant hits in each co-IP (anti-Ty1 and anti-HA) (enrichment fold ≥2) identified two additional proteins enriched in both IPs: Tb927.11.3880 – a flagellar actin-like protein (Ersfeld and Gull, 2001) – and Tb927.11.8160 – a putative inner arm dynein heavy chain protein belonging to the IAD-4 protein family (data summarized in Supplementary file 1). Tb927.11.8160 is potentially involved in flagellum motility (Wickstead and Gull, 2007), it localizes along the T. brucei axoneme according to the TrypTag genome-wide protein localization resource (http://tryptag.org/; Dean et al., 2017). Significantly, BlastP searches of mammalian genomes for Tb927.11.8160 orthologs identified DNAH1 (Protein ID: XP_828890.1).
DNAH1 and DNALI1 both interact with TTC29 in mouse
In a recent human study, we showed that the presence of bi-allelic TTC29 variants induces an MMAF phenotype and male infertility (Lorès et al., 2019). By CRISPR/Cas9 mutagenesis and RNAi silencing, we demonstrated that the orthologous TTC29 proteins in mouse and in T. brucei, respectively, also constitute flagellar proteins that are required for proper flagellum beating, indicating an evolutionarily conserved function for TTC29 (Lorès et al., 2019).
Here, we took advantage of the availability of the mutant mouse model to further decipher the protein complexes associated with TTC29 function within the flagella. We performed comparative analyses of co-immunoprecipitations of testis protein extracts from WT mice using TTC29-specific antibodies, and from two previously generated TTC29 KO mouse lines lacking the TTC29 protein (Ttc29−/− L5 and L7) (Lorès et al., 2019). Mass spectrometry analyses of four independent experimental replicates for each mouse line identified a set of 124 proteins specifically immunoprecipitated in WT testes.
These proteins were further examined to identify TTC29-binding proteins (Supplementary file 2). Filtering of immunoprecipitated proteins based on peptide number and replicate constancy identified DNAH1 and DNALI1, in addition to TTC29, as the only proteins that were co-immunoprecipitated with a significant number of peptides in all four experimental replicates. In our previous study, DNAH1 and DNALI1 were detected at equal levels in flagella purified from WT and Ttc29−/− KO mice (Lorès et al., 2019). Therefore, we considered DNAH1 and DNALI1 to be authentic protein partners of TTC29 in sperm flagella, in line with data obtained with T. brucei. Thus, our data indicate that ZMYND12 interacts with DNAH1 and DNALI1, two established components of the axonemal IDA.
Discussion
The results presented here show that the presence of bi-allelic ZMYND12 variants induces a typical MMAF phenotype in humans, suggesting that this gene is necessary for sperm flagellum structure and function. ZMYND12 is a previously uncharacterized MYND-domain-containing zinc finger protein, we therefore explored its localization and function using a recognized model: the flagellated protozoan Trypanosoma. This model is widely used to investigate flagellum biogenesis and the function of flagellar proteins. In addition to having a very similar axonemal organization and composition to human flagellum, Trypanosoma has numerous advantages for functional studies, and is very useful for reverse genetics and biochemical or proteomics studies (Vincensini et al., 2011).
We first confirmed that ZMYND12 is an axonemal protein, and then used RNAi experiments to confirm its critical role in flagellum function. ZMYND12 knock-down led to dramatic flagellar motility defects like those observed in human sperm (Figure 3). In addition, a truncated form of TAX-1 protein lacking the TPR domain – mimicking the expected truncated ZMYND12 protein in MMAF individuals bearing the p.Arg145Ter variant – was not associated with the flagellum, and failed to rescue TAX-1 RNAi-induced motility defects (Figure 4). These results clearly indicate that the TPR motif of ZMYND12 is critical for protein targeting and function in both human sperm and Trypanosoma. Other groups also previously reported a significant role for TPR proteins in sperm flagellar biogenesis and male infertility (Liu et al., 2019b; Lorès et al., 2019). Indeed, TPR domains are known to be involved in protein-protein interaction and the assembly of multiprotein complexes (D’Andrea and Regan, 2003). We therefore decided to further explore the potential interactions between ZMYND12 and other axonemal proteins.
IF experiments in sperm cells from ZMYND12 individuals revealed severe ultrastructural defects dramatically affecting several components of the axoneme. More specifically, IDAs and the CSC were severely disorganized, as shown by the total absence of DNAH1, WDR66, and DNALI1 immunostaining (Figure 2A, B and Figure 2—figure supplement 1). Intriguingly, TTC29 and CFAP70 were also absent from sperm cells from individuals bearing ZMYND12 mutations (Figure 2C, D). These observations suggest some functional or physical interaction between these proteins. However, the exact function and location of TTC29 and CFAP70 remain unclear. In Chlamydomonas, TTC29 has been suggested to be a component of IDAs (Yamamoto et al., 2008), but this protein was also reported to possibly contribute to the IFT machinery (Brooks, 2014; Lorès et al., 2019). For CFAP70, functional studies in Chlamydomonas demonstrated that the protein localized at the base of the ODA, where it could regulate ODA activity (Shamoto et al., 2018). In T. brucei, we screened the axonemal proteins WDR66, CFAP70, SPAG6, and TTC29 to test their interaction with the ZMYND12 homolog. Our results indicated that TbTAX-1 only interacts with TbTTC29, and that the two proteins are part of the same axonemal complex (Figure 5A, B). This interaction was confirmed by U-ExM and confocal microscopy (Figure 5C, D). A third member of the complex – Tb927.11.8160, the ortholog of human DNAH1 – was subsequently identified by comparative proteomics analysis of co-immunoprecipitated proteins (Figure 5E and Supplementary file 1). These results were further confirmed using the Ttc29-null mouse model previously generated in our laboratory (Supplementary file 2). All of these results are consistent with previous studies in Chlamydomonas suggesting that p38 (ZMYND12 ortholog) and p44 (TTC29 ortholog) interact and play a role in IDAd docking on the axoneme (Yamamoto et al., 2008). Interestingly, the axonemal IDAd in Chlamydomonas is composed of the DHC2 heavy chain and the p28 light chain, orthologs of human DNAH1 and DNALI1, respectively. Moreover, the recent human reference interactome map obtained by high-throughput two-hybrid screening (Luck et al., 2020) also supports direct protein–protein interaction between ZMYND12 and TTC29. Our results thus formally demonstrate that ZMYND12 interacts with TTC29 within the axonemal complex, in association with DNAH1 and inner dynein arms. DNAH1 is directly connected through an arc-like structure (King, 2013) to the base of radial spoke 3 (RS3), which is anchored to the axoneme by the CSC (Dymek et al., 2011). In humans and Trypanosoma, the absence of ZMYND12 in the flagellum probably destabilizes this axonemal complex, and consequently alters the stability of the RS3 base-docked IDAs including DNAH1, the CSC, and RS. Each of these defects leads to severe axonemal disorganization, and an MMAF sperm phenotype. Our team previously reported deleterious variants of TTC29 and DNAH1 associated with an MMAF phenotype. The flagellar defects caused by these variants are similar to those observed in sperm from individuals with ZMYND12 defects (Ben Khelifa et al., 2014; Liu et al., 2019a; Lorès et al., 2019). The results presented here thus contribute to elucidating the physical and functional associations between ZMYND12, TTC29, and DNAH1 in the axoneme, and emphasize the key role played by this triumvirate in maintaining the structure and function of the sperm flagellum.
Adding the three ZMYND12-mutated individuals to the 83 individuals previously identified with variants in known MMAF-related genes in our cohort of 167 individuals, we now have a diagnostic efficiency of 51.4% (86/167). This figure demonstrates the efficiency and the clinical utility of WES for the investigation of the genetic causes and pathophysiology of MMAF syndrome. Among the variants identified, we found the same ZMYND12 nonsense variant c.433C>T; p.(Arg145Ter) in three unrelated individuals from diverse geographical origins (i.e., Tunisia, China), suggesting that the Arg145 residue may constitute a mutational hotspot. Importantly, as previously reported (Kherraf et al., 2018), we confirmed that WES can detect small pathogenic exon deletions. These events may constitute a recurrent disease mechanism in MMAF, and should therefore be systematically explored. Despite these significant advances, the genetic cause of MMAF remains unknown for about half the individuals affected, due to the high genetic heterogeneity of the disorder. To improve the diagnostic rate, more powerful techniques such as whole-genome sequencing may now be envisaged for MMAF patients for whom WES fails to provide results (Meienberg et al., 2016).
The results presented here demonstrate that the combined use of exome sequencing and the Trypanosoma model can efficiently reveal new genes responsible for an MMAF phenotype in human and decipher how they affect sperm flagellum architecture and function. Overall, our strategy identified bi-allelic variants in ZMYND12 associated with severe flagellum malformations resulting in asthenoteratozoospermia and primary male infertility. In conclusion, our work with human sperm and the flagellated protist T. brucei demonstrated that ZMYND12 is part of the same axonemal complex as TTC29 and DNAH1, and plays a critical role in flagellum function and assembly.
Materials and methods
Study participants
WES data from a previously established cohort of 167 MMAF individuals were analyzed (Coutton et al., 2019). All individuals presented a typical MMAF phenotype characterized by severe asthenozoospermia (total sperm motility below 10%), with >5% of the spermatozoa displaying at least three of the following flagellar abnormalities: short, absent, coiled, bent, or irregular flagella (Coutton et al., 2019). All individuals had a normal somatic karyotype (46,XY) with normal bilateral testicular size, hormone levels, and secondary sexual characteristics. Sperm were analyzed in the source laboratories during routine biological examination of the individuals according to World Health Organization (WHO) guidelines (Wang et al., 2014). An additional Chinese individual was included in the study from a familial case. This individual was enrolled from the Human Sperm Bank of West China Second University Hospital of Sichuan University; he presented with severe asthenoteratozoospermia and a typical MMAF phenotype. WES analysis for this subject was performed as described in the previous report (Liu et al., 2021). Sperm morphology was assessed following Papanicolaou staining (Figure 1A). Detailed semen parameters of the four individuals carrying ZMYND12 mutations (ZMYND12_1–4) are presented in Table 1. Sperm samples for additional phenotypic characterization were obtained from ZMYND12_3. Written informed consent was obtained from all individuals participating in the study, and institutional approval was given by the local medical ethics committee (CHU Grenoble Alpes institutional review board #6705). Samples were stored in the Fertithèque collection registered with the French Ministry of Health (DC-2015-2580) and the French Data Protection Authority (DR-2016-392).
WES and bioinformatics analysis
Genomic DNA was isolated from EDTA blood using DNeasy Blood & Tissue Kits (QIAGEN SA). Genetic data were obtained through several sequencing centers, in particular Novogene, Genoscope, and Integragen. Coding regions and intron/exon boundaries were sequenced after enrichment using Agilent SureSelect Human All Exon V5, V6, or Clinical Research capture kits. An alignment-ready GRCh38 reference genome (including ALT, decoy, and HLA) was produced using ‘run-gen-ref hs38DH’ from Heng Li’s bwakit package (https://github.com/lh3/bwa; Li, 2017). Exomes were analyzed using a bioinformatics pipeline developed in-house. The pipeline consists of two modules, both distributed under the GNU General Public License v3.0 and available on github (grexome-TIMC-Primary TIMC primary, https://github.com/ntm/grexome-TIMC-Primary; copy archived at Thierry-Mieg, 2023a, TIMC secondary, https://github.com/ntm/grexome-TIMC-Secondary; copy archived at Thierry-Mieg, 2023b). The first module takes FASTQ files as input and produces a single merged GVCF file per variant-caller, as follows: trim adaptors and filter low-quality reads with fastp 0.23.2, align reads with BWA-MEM 0.7.17, mark duplicates using samblaster 0.1.26, and sort and index BAM files with samtools 1.15.1. SNVs and short indels are called from each BAM file using strelka 2.9.10 (Kim et al., 2018) and GATK 4.1.8.1 to produce individual GVCF files from each variant-caller. These files are finally merged using mergeGVCFs.pl to obtain a single multi-sample GVCF per caller. Using several variant-callers helps to compensate for each caller’s flaws. The second module takes each merged GVCF as input and produces annotated analysis-ready TSV files. Annotation involved 17 streamlined tasks: Low-quality variant calls (DP <10, GQ <20, or less than 15% of reads supporting the ALT allele) were discarded. Variant Effect Predictor v108 (McLaren et al., 2016) was used to annotate variants and predict their impact, in order to filter out low-impact (MODIFIER) variants and/or prioritize high-impact ones (e.g., stop-gain or frameshift variants). Variants with a minor allele frequency greater than 1% in gnomAD v.2.1 or 3% in 1000 Genomes Project phase 3 were excluded. The pipeline performs an integrative analysis of all available exomes, both at the individual variant level and at the gene level. Thus, recurring genomic variants are readily identified alongside genes that are severely impacted in several patients but not in control subjects. Additional information can be found in Arafah et al., 2021. Copy number variants were searched using the ExomeDepth software package, as previously reported (Kherraf et al., 2018; Plagnol et al., 2012).
Sanger sequencing
ZMYND12 single-nucleotide variants identified by exome sequencing were validated by Sanger sequencing as previously described (Coutton et al., 2019). PCR primers and protocols used for each individual are listed in Supplementary file 3.
MLPA analysis
Deletion of exons 6–8 identified in individual ZMYND12_2 by exome sequencing was confirmed by custom MLPA analysis, according to our previously described method (Coutton et al., 2012). The MLPA probes were designed, and the MLPA reaction was performed according to the recommendations set out in the MRC-Holland synthetic protocol (https://mlpa.com/). Data analysis was also performed in line with this protocol. For this study, three synthetic MLPA probes specific for exons 5, 6, and 8 of ZMYND12 were designed. MLPA probes are listed in Supplementary file 4.
RT-qPCR analysis
RT-qPCR experiments were performed as previously described (Coutton et al., 2019) with cDNAs extracted from various human tissues purchased from Life Technologies. A panel of 10 organs was used for experiments: testis, brain, trachea, lung, kidney, liver, skeletal muscle, pancreas, placenta, and heart. Human RT-qPCR data were normalized relative to the two reference housekeeping genes RPL6 and RPL27 by the −ΔΔCt method (Livak and Schmittgen, 2001). Primer sequences and RT-qPCR conditions are indicated in Supplementary file 5. Relative expression of ZMYND12 transcripts was compared in several organs, and statistical analysis involved a two-tailed t-test (Prism 4.0 software; GraphPad, San Diego, CA). A p-value ≤0.05 was considered significant.
Immunostaining in human sperm cells
IF experiments were performed on sperm cells from control individuals, from individual ZMYND12_3 – carrier of the nonsense variant c.103C>T – and from a previously reported MMAF individual – carrier of the TTC29 splicing variant c.1761+1G>A (Lorès et al., 2019). For each MMAF individual, 200 sperm cells were analyzed by two experienced operators, and the IF staining intensity and patterns were compared to those obtained from fertile control individuals. Sperm cells were fixed in phosphate-buffered saline (PBS)/4% paraformaldehyde for 1 min at room temperature. After washing in 1 ml PBS, the sperm suspension was spotted onto 0.1% poly L-lysine pre-coated slides (Thermo Scientific). After attachment, sperm were permeabilized with 0.1% (vol/vol) Triton X-100–DPBS (Dulbecco's Phosphate-buffered saline) (Triton X-100; Sigma-Aldrich) for 5 min at RT. Slides were then blocked by applying 5% normal serum–DPBS (normal goat or donkey serum; Gibco, Invitrogen) before incubating overnight at 4°C with the primary antibody. The following primary antibodies were used: ZMYND12, DNAI1, DNALI1, DNAH1, DNAH8, DNAH17, RSPH1, SPAG6, GAS8, CFAP70, WDR66 (also known as CFAP251), TTC29, AKAP4, and acetylated-α-tubulin. The primary antibody references and dilutions are fully detailed in Supplementary file 6. Slides were washed with 0.1% (vol/vol) Tween 20–DPBS before incubating for 1 hr at room temperature with secondary antibodies. Highly cross-adsorbed secondary antibodies (Dylight 488 and Dylight 549, 1:1000) were purchased from Jackson Immunoresearch. Experimental controls were treated similarly, but omitting the primary antibodies. Samples were counterstained with DAPI and mounted with DAKO mounting media (Life Technology). Fluorescence images were captured with a confocal microscope (Zeiss LSM 710).
T. brucei cell lines, culture, and transfection
The trypanosome cell lines used in this study are derived from the procyclic parental form, T. brucei SmOxP427 (Sunter, 2016). The absence of contamination of the cell line by Mycoplasma was verified by PCR. Our PCR and sequencing data also confirmed that all cell lines were T. brucei brucei Lister 427 and carried in situ or episomal the designated sequence. These cells co-express the T7 RNA polymerase and the tetracycline repressor and were grown and transfected as previously described (Kherraf et al., 2018). When required, the culture medium was supplemented with puromycin (1 μg/ml), neomycin (10 μg/ml), phleomycin (5 μg/ml), or hygromycin (25 μg/ml). Cells were transfected using an AMAXA electroporator (Lonza) as previously described (Schumann Burkard et al., 2011). TbTAX-1 RNA interference was induced with tetracycline (10 μg/ml).
Tagged proteins were obtained from SmOxP427 cells, by inserting epitope-tagged transgenes at the endogenous locus. Transgenes were N- or C-terminally tagged with 10myc, 10Ty1, or 10HA tags using the pPOTv7 vector series, as previously described (Dean et al., 2015; Kherraf et al., 2018). For RNAiTbTAX-1 knock-down, bp 219–667 were cloned between the XhoI and XbaI sites of p2T7tiB (LaCount et al., 2002) and the linearized vector was transfected into endogenously tagged TbTAX-1-background cell lines. To target the full-length TbTAX-1 and not the shorter form, similar cloning was performed with bp 493–917. The TbTTC29Ty1 RNAi cell line is described elsewhere (Lorès et al., 2019).
Co-immunoprecipitation and mass spectrometry on Trypanosoma cells
In a 15-ml polycarbonate tube, 2 × 108 cells expressing the different tagged proteins were washed in PBS and incubated for 5 min at RT in 1 ml of immunoprecipitation buffer for cell lysis (IP buffer; 25 mM Tris–HCl pH 7.4, 100 mM NaCl, 0.25% Nonidet P-40 (Igepal), 1 mM DTT(Dithiothréitol), protease inhibitor cocktail, and 1 unit of nuclease) (Pierce). Lysis was completed by sonication (5 × 30 s ON/30 s OFF) in a Bioruptor sonication system. Cell lysates were clarified by centrifugation (10 min, 16,000 × g, 4°C) and split into two samples each. One was incubated with 3 μl of anti-Ty1 and the other with 2 μl of mouse anti-HA for 1 hr at 4°C on a rotating wheel. Protein-G Dynabead (Invitrogen 10003D) slurry (40 µl) was washed in IP buffer and incubated with the samples for 1 hr at 4. After washing three times in IP buffer, beads were resuspended in 40 μl sample buffer and boiled before loading 20 μl on sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE).
For mass spectrometry analyses, 1.8 × 109 cells were washed in PBS, resuspended in 2 ml of immunoprecipitation buffer without DTT and processed as described above. Antibodies used were: mouse anti-HA (10 μl) and anti-TY1 (15 μl). Complex precipitation was achieved by adding 200 μl of Protein-G Dynabead slurry. After washing three times in IP buffer, beads were transferred to new tubes and washed 3× in PBS before storing dry at −20°C.
Protein-G Dynabeads were resuspended in Laemmli buffer and boiled. Supernatants were loaded on a 10% acrylamide SDS–PAGE gel, and proteins were revealed by Colloidal Blue staining. Sample preparation and protein digestion by trypsin were performed as previously described (Allmann et al., 2014). NanoLC-tandem mass spectrometry (MS/MS) analyses were performed using an Ultimate 3000 RSLC Nano-UPHLC system (Thermo Scientific, USA) coupled to a nanospray Orbitrap Fusion Lumos Tribrid Mass Spectrometer (Thermo Fisher Scientific, California, USA). Each peptide extract was loaded on a 300 µm ID × 5 mm PepMap C18 pre-column (Thermo Scientific, USA) at a flow rate of 10 µl/min. After a 3-min desalting step, peptides were separated on a 50-cm EasySpray column (75 µm ID, 2 µm C18 beads, 100 Å pore size; ES903, Thermo Fisher Scientific) by applying a 4–40% linear gradient of solvent B (0.1% formic acid in 80% acetonitrile [ACN]) over 48 min. The flow rate for separation was set to 300 nl/min. The mass spectrometer was operated in positive ion mode at a 2.0 kV needle voltage. Data were acquired using Xcalibur 4.4 software in data-dependent mode. MS scans (m/z 375–1500) were recorded at a resolution of R = 120,000 (@ m/z 200) and an Automated Gain Control (AGC) target of 4 × 105 ions collected within 50 ms was applied along with a top speed duty cycle of up to 3 s for MS/MS acquisition. Precursor ions (2–7 charge states) were isolated in the quadrupole within a mass window of 1.6 Th, and fragmented by Higher-energy Collisional Dissociation (HCD), applying 28% normalized collision energy. MS/MS data were acquired in the Orbitrap cell at a resolution of R = 30,000 (m/z 200), a standard AGC target and a maximum injection time in automatic mode. A 60-s exclusion window was applied to previously selected precursors. Proteome Discoverer 2.5 was used to identify proteins and perform label-free quantification (LFQ). Proteins were identified in batch mode using MS Amanda 2.0, Sequest HT, and Mascot 2.5 algorithms, with searches performed against a T. brucei brucei TREU927 protein database (9788 entries, release 57, https://tritrypdb.org/ website). Up to two missed trypsin cleavages were allowed. Mass tolerances in MS and MS/MS were set to 10 ppm and 0.02 Da, respectively. Oxidation (M) and acetylation (K) were included as dynamic modifications, and carbamidomethylation (C) as a static modification. Peptides were validated using the Percolator algorithm (Käll et al., 2007), retaining only ‘high confidence’ peptides, corresponding to a 1% false discovery rate at the peptide level. The minora feature-detector node (LFQ) was used along with the feature-mapper and precursor ion quantifier. The following quantification parameters were applied: (1) unique peptides; (2) precursor abundance based on intensity; (3) no normalization; (4) protein abundance calculation: summed abundances; (5) protein ratio calculation: pairwise ratios; (6) imputation mode: low abundance resampling; and (7) hypothesis test: t-test (background based). For master proteins, quantitative data were considered based on a minimum of two unique peptides, a fold-change greater than 2 and a Benjamini–Hochberg-corrected p-value for the FDR of less than 0.05. Mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository (Adhikari et al., 2020), under dataset identifier PXD039470.
IF on Trypanosoma cells
Cells were harvested, washed, and processed for immunolabeling on methanol-fixed detergent-extracted cells (CSK) as described in Albisetti et al., 2017. The antibodies used and their dilutions are listed in Supplementary file 7. Images were acquired on a Zeiss Imager Z1 microscope, using a Photometrics Coolsnap HQ2 camera, with a ×100 Zeiss objective (NA 1.4) using Metamorph software (Molecular Devices), and processed with ImageJ (NIH).
U-ExM on Trypanosoma cells
The U-ExM protocol has been adapted and optimized for use on trypanosomes, as described (Gambarotto et al., 2021). Protocol details can be found online at https://doi.org/10.17504/protocols.io.bvwqn7dw (Casas et al., 2022). Images were acquired on a Zeiss AxioImager Z1 using a ×63 (1.4 NA) objective, or at the Bordeaux Imaging Center with a Leica TCS SP5 on an upright stand DM6000 (Leica Microsystems, Mannheim, Germany), fitted with a HCX Plan Apo CS ×63 oil NA 1.40 objective. Images were processed and analyzed using ImageJ and Fiji software (Schindelin et al., 2012; Schneider et al., 2012).
Trypanosoma cells: sedimentation assays and video microscopy
Sedimentation assays were performed as described (Dacheux et al., 2012). Briefly, cells were placed in cuvettes and incubated for 24 hr without shaking. Optical density (OD600 nm) was measured before mixing (ODb, to detect ‘swimming’ cells) and after mixing (ODa, to detect ‘swimming’ plus ‘sedimenting’ cells). The percentage of sedimented cells was calculated as 100 − (ODb/ODa) × 100 for the WT and RNAi cells. Sedimentation of RNAi cells was normalized relative to the tagged TbTAX-1 parental cell line. Video microscopy was carried out as described (Lorès et al., 2019). Briefly, PCF(Procyclic Form) cells (WT cell and RNAiTbTAX-1 cells 72 hr after induction) were washed in PBS, and cell mobility was recorded by phase-contrast microscopy on a Zeiss AxioImager Z1, ×40 lens (NA 1.3). A 28-s sequence of digital video was captured from separate regions and analyzed using Metamorph software (Molecular Devices) (speed x8). The positions of individual cells were plotted over 0.28-s intervals. The start and end positions of each cell were marked.
TEM on Trypanosoma cells
TEM blocks of Trypanosoma cells and thin sections were prepared as previously described (Albisetti et al., 2017). Thin sections were visualized on an FEI Tecnai 12 electron microscope. Images were captured on an ORIUS 1000 11M Pixel camera (resolution 3 ± 5 nm) controlled by Digitalmicrograph, and processed using ImageJ (NIH).
Western blot analyses of Trypanosoma cells
Proteins from WCs, detergent-extracted CSK and flagella, or co-IPs were separated on SDS–PAGE gels. Separated proteins were transferred by semi-dry transfer to PVDF(polyvinylidene difluoride) and low-fluorescence PVDF membranes for further processing, as described (Albisetti et al., 2017). The antibodies used and their dilutions are listed in Supplementary file 7. For IP analysis, primary antibodies – anti-TY1 and rabbit anti-HA (GTX115044) – and fluorescent secondary antibodies – anti-mouse Starbright Blue 520 (Bio-Rad 12005867) and anti-rabbit Starbright Blue 700 (Bio-Rad 12004162) – were used.
Co-immunoprecipitation and mass spectrometry analysis on Ttc29−/− mouse testes
Testes from WT and Ttc29−/− L5 and L7 mice (Lorès et al., 2019) were dissected in cold PBS; the albuginea was removed and the seminiferous tubules were dissociated and pelleted by brief centrifugation. Proteins from the pellets were extracted in 1 ml lysis buffer: 50 mM Tris–HCl, pH 8, 150 mM NaCl, 10 mM MgCl2, 0.5% NP40, and Complete protease inhibitor cocktail (Roche Applied science). Immunoprecipitation was performed on total-protein lysate using 1 μg of rabbit polyclonal TTC29 antibody (HPA061473, Sigma-Aldrich) and 15 μl of Protein A/G magnetics beads (Bio-Ademtec) according to the manufacturer’s instructions. The experiment was replicated with four mice for each genotype.
Total immunoprecipitated proteins were recovered in 25 µl 100 mM Tris/HCl pH 8.5 containing 10 mM TCEP(tris(2-carboxyéthyl)phosphine), 40 mM chloroacetamide, and 1% sodium deoxycholate, by sonicating three times, and incubating for 5 min at 95°C. An aliquot (20 µg) of protein lysate was diluted (1:1) in Tris 25 mM pH 8.5 in 10% ACN and subjected to overnight trypsin digestion with 0.4 µg of sequencing-grade bovine trypsin (Promega) at 37°C. Deoxycholate was removed using liquid–liquid phase extraction after adding 50 µl of 1% trifluoroacetic acid (TFA) in ethyl-acetate. A six-layer home-made Stagetip with Empore Styrene- divinylbenzene – Reversed Phase Sulfonate (SDB RPS; 3M) disks was used. Eluted peptides were dried and re-solubilized in 2% TFA before preparing five fractions on hand-made Strong Cation eXchange (SCX) StageTips (Kulak et al., 2014). Fractions were analyzed on a system combining an U3000 RSLC nanochromatograph eluting into an Orbitrap Fusion mass spectrometer (both Thermo Scientific). Briefly, peptides from each SCX fraction were separated on a C18 reverse phase column (2 μm particle size, 100 Å pore size, 75 µm inner diameter, 25 cm length) with a 145-min linear gradient starting from 99% solvent A (0.1% formic acid in H2O) and ending with 40% of solvent B (80% ACN and 0.085% formic acid in H2O). The MS1 scans spanned 350–1500 m/z with an AGC target of 1 × 106 within a maximum ion injection time (MIIT) of 60 ms, at a resolution of 60,000. The precursor selection window was set to 1.6 m/z with quadrupole filtering. HCD Normalized Collision Energy was set to 30%, and the ion trap scan rate was set to ‘rapid’ mode with AGC target 1 × 105 and 60 ms MIIT. Mass spectrometry data were analyzed using Maxquant (v.1.6.1.0) (Cox et al., 2014). The database used was a concatenation of human sequences from the Uniprot-Swissprot database (release 2017-05) and the Maxquant list of contaminant sequences. Cysteine carbamidomethylation was set as fixed modification, and acetylation of protein N-terminus and oxidation of methionine were set as variable modifications. Second peptide search and ‘match between runs’ (MBR) options were allowed. Label-free protein quantification (LFQ) was performed using both unique and razor peptides, requiring at least two peptides per protein. The quality of raw quantitative data was evaluated using PTXQC software v. 0.92.3 (Bielow et al., 2016). Perseus software, version 1.6.2.3 (Tyanova et al., 2016) was used for statistical analysis and data comparison. Proteomics data obtained by mass spectrometry analysis were deposited to the ProteomeXchange Consortium via the PRIDE partner repository (Adhikari et al., 2020) under dataset identifier PXD039854.
Acknowledgements
We thank all individuals for their participation. We thank Samuel Dean (Warwick Medical School) and Jack Sunter (Oxford Brookes University) for the pPOT plasmids and SmOx T brucei cell lines, F Bringaud (University of Bordeaux) for the anti-enolase antibody, and P Bastin (Institut Pasteur) for the anti-Ty1 antibody. We thank G Cougnet-Houlery, S Guit, and J Marcos for ongoing access to the MFP lab infrastructure. This work was supported by the Institut National de la Santé et de la Recherche Médicale (INSERM), the Centre National de la Recherche Scientifique (CNRS), University Grenoble Alpes, and French National Research Agency funding for specific projects: MASFLAGELLA (ANR-14-CE15-0002), FLAGEL-OME (ANR-19-CE17-0014), and OLIGO-SPERM (ANR-21-CE17-0007). Mass spectrometry experiments were performed at the Centre de Génomique Fonctionnelle facility (CGFB, Bordeaux) for Trypanosoma studies and at the Proteom’IC core-facility (Institut Cochin, Paris – François Guillonneau and Johanna Bruce) for mouse studies. The Orbitrap Fusion mass spectrometer at the Proteom’IC core-facility was acquired with funds from the FEDER through the « Operational Program for Competitiveness Factors and employment 2007–2013 » and from the « Cancéropôle Ile-de-France ». The Bordeaux Imaging Center is a service unit of the CNRS INSERM and Bordeaux University, a member of the French BioImaging national infrastructure funded through the French National Research Agency [ANR-10-INBS-04].
Funding Statement
The funders had no role in study design, data collection, and interpretation, or the decision to submit the work for publication.
Contributor Information
Charles Coutton, Email: ccoutton@chu-grenoble.fr.
Wei Yan, University of California, Los Angeles, United States.
Wei Yan, University of California, Los Angeles, United States.
Funding Information
This paper was supported by the following grants:
Agence Nationale de la Recherche ANR-19-CE17-0014 to Pierre F Ray.
Agence Nationale de la Recherche ANR-21-CE17-0007 to Charles Coutton.
Agence Nationale de la Recherche ANR-10-INBS-04 to Mélanie Bonhivers.
Additional information
Competing interests
No competing interests declared.
Author contributions
Resources, Data curation, Formal analysis, Funding acquisition, Validation, Methodology, Writing – original draft, Writing – review and editing.
Conceptualization, Data curation, Formal analysis, Methodology, Writing – original draft, Writing – review and editing.
Formal analysis, Methodology, Writing – original draft.
Resources, Formal analysis, Investigation, Writing – original draft.
Data curation, Formal analysis, Writing – original draft, Writing – review and editing.
Resources, Formal analysis, Validation, Investigation, Methodology, Writing – original draft.
Data curation, Formal analysis.
Conceptualization, Formal analysis, Methodology, Writing – original draft, Writing – review and editing.
Resources, Formal analysis, Methodology.
Formal analysis, Methodology.
Resources, Formal analysis.
Resources, Data curation, Formal analysis, Funding acquisition.
Resources, Formal analysis, Investigation, Project administration.
Formal analysis, Supervision, Investigation, Methodology.
Formal analysis.
Formal analysis, Funding acquisition, Investigation, Methodology.
Formal analysis, Investigation.
Formal analysis, Investigation, Methodology.
Formal analysis, Funding acquisition, Investigation, Methodology.
Formal analysis, Investigation, Methodology, Writing – original draft.
Formal analysis, Investigation, Methodology, Writing – original draft, Project administration.
Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing – original draft, Project administration.
Software, Formal analysis.
Formal analysis, Funding acquisition, Methodology, Writing – original draft.
Data curation, Formal analysis, Funding acquisition, Validation, Methodology, Writing – original draft.
Resources, Data curation, Formal analysis, Methodology, Writing – original draft.
Data curation, Formal analysis, Validation, Investigation, Methodology, Writing – original draft, Project administration.
Conceptualization, Data curation, Supervision, Funding acquisition, Validation, Investigation, Methodology, Writing – original draft, Project administration.
Conceptualization, Data curation, Formal analysis, Supervision, Funding acquisition, Validation, Investigation, Visualization, Methodology, Writing – original draft, Project administration.
Conceptualization, Data curation, Formal analysis, Funding acquisition, Validation, Investigation, Methodology, Writing – original draft, Project administration, Writing – review and editing.
Conceptualization, Data curation, Formal analysis, Funding acquisition, Validation, Investigation, Methodology, Writing – original draft, Project administration.
Conceptualization, Resources, Data curation, Formal analysis, Supervision, Funding acquisition, Validation, Investigation, Visualization, Methodology, Writing – original draft, Project administration, Writing – review and editing.
Ethics
Written informed consent was obtained from all individuals participating in the study, and institutional approval was given by the local medical ethics committee (CHU Grenoble Alpes institutional review board #6705). Samples were stored in the Fertithéque collection registered with the French Ministry of Health (DC-2015-2580) and the French Data Protection Authority (DR-2016-392).
Additional files
Data availability
All data generated or analyzed during this study are included in the manuscript and supporting file; Source Data files have been provided for Figures 3–5 and Figure 4-figure Supplement 1. All of the ZMYND12 variants identified here have been deposited in ClinVar under reference SUB12540308. Mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository, under dataset identifier PXD039470 and PXD039854.
The following datasets were generated:
Dupuy JW, Coutton C. 2023. Enrichment and mass spectrometry analysis of TbTAX-1-interacting or TbTTC29-interacting proteins by co-immunoprecipitation. ProteomeXchange. PXD039470
Touré A. 2023. Identification of TTC29 protein partners in mouse testis. ProteomeXchange. PXD039854
Ray PF, Thierry Mieg N, Kherraf Z-E, Coutton C. 2023. ZMYND12 mutations in MMAF patients. ClinVar. SUB12540308
References
- Adhikari S, Nice EC, Deutsch EW, Lane L, Omenn GS, Pennington SR, Paik Y-K, Overall CM, Corrales FJ, Cristea IM, Van Eyk JE, Uhlén M, Lindskog C, Chan DW, Bairoch A, Waddington JC, Justice JL, LaBaer J, Rodriguez H, He F, Kostrzewa M, Ping P, Gundry RL, Stewart P, Srivastava S, Srivastava S, Nogueira FCS, Domont GB, Vandenbrouck Y, Lam MPY, Wennersten S, Vizcaino JA, Wilkins M, Schwenk JM, Lundberg E, Bandeira N, Marko-Varga G, Weintraub ST, Pineau C, Kusebauch U, Moritz RL, Ahn SB, Palmblad M, Snyder MP, Aebersold R, Baker MS. A high-stringency blueprint of the human proteome. Nature Communications. 2020;11:5301. doi: 10.1038/s41467-020-19045-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albisetti A, Florimond C, Landrein N, Vidilaseris K, Eggenspieler M, Lesigang J, Dong G, Robinson DR, Bonhivers M, Matthews KR. Interaction between the flagellar pocket collar and the hook complex via a novel microtubule-binding protein in Trypanosoma brucei. PLOS Pathogens. 2017;13:e1006710. doi: 10.1371/journal.ppat.1006710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allmann S, Mazet M, Ziebart N, Bouyssou G, Fouillen L, Dupuy JW, Bonneu M, Moreau P, Bringaud F, Boshart M. Triacylglycerol storage in lipid droplets in procyclic Trypanosoma brucei. PLOS ONE. 2014;9:e114628. doi: 10.1371/journal.pone.0114628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arafah K, Lopez F, Cazin C, Kherraf ZE, Tassistro V, Loundou A, Arnoult C, Thierry-Mieg N, Bulet P, Guichaoua MR, Ray PF. Defect in the nuclear pore membrane glycoprotein 210-like gene is associated with extreme uncondensed sperm nuclear chromatin and male infertility: a case report. Human Reproduction. 2021;36:693–701. doi: 10.1093/humrep/deaa329. [DOI] [PubMed] [Google Scholar]
- Aslett M, Aurrecoechea C, Berriman M, Brestelli J, Brunk BP, Carrington M, Depledge DP, Fischer S, Gajria B, Gao X, Gardner MJ, Gingle A, Grant G, Harb OS, Heiges M, Hertz-Fowler C, Houston R, Innamorato F, Iodice J, Kissinger JC, Kraemer E, Li W, Logan FJ, Miller JA, Mitra S, Myler PJ, Nayak V, Pennington C, Phan I, Pinney DF, Ramasamy G, Rogers MB, Roos DS, Ross C, Sivam D, Smith DF, Srinivasamoorthy G, Stoeckert CJ, Subramanian S, Thibodeau R, Tivey A, Treatman C, Velarde G, Wang H. TriTrypDB: a functional genomic resource for the Trypanosomatidae. Nucleic Acids Research. 2010;38:D457–D462. doi: 10.1093/nar/gkp851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Auger J, Jouannet P, Eustache F. Another look at human sperm morphology. Human Reproduction. 2016;31:10–23. doi: 10.1093/humrep/dev251. [DOI] [PubMed] [Google Scholar]
- Baron DM, Ralston KS, Kabututu ZP, Hill KL. Functional genomics in Trypanosoma brucei identifies evolutionarily conserved components of motile flagella. Journal of Cell Science. 2007;120:478–491. doi: 10.1242/jcs.03352. [DOI] [PubMed] [Google Scholar]
- Ben Khelifa M, Coutton C, Zouari R, Karaouzène T, Rendu J, Bidart M, Yassine S, Pierre V, Delaroche J, Hennebicq S, Grunwald D, Escalier D, Pernet-Gallay K, Jouk PS, Thierry-Mieg N, Touré A, Arnoult C, Ray PF. Mutations in DNAH1, which encodes an inner arm heavy chain dynein, lead to male infertility from multiple morphological abnormalities of the sperm flagella. American Journal of Human Genetics. 2014;94:95–104. doi: 10.1016/j.ajhg.2013.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bielow C, Mastrobuoni G, Kempa S. Proteomics quality control: Quality control software for maxquant results. Journal of Proteome Research. 2016;15:777–787. doi: 10.1021/acs.jproteome.5b00780. [DOI] [PubMed] [Google Scholar]
- Blackburn K, Bustamante-Marin X, Yin W, Goshe MB, Ostrowski LE. Quantitative proteomic analysis of human airway cilia identifies previously uncharacterized proteins of high abundance. Journal of Proteome Research. 2017;16:1579–1592. doi: 10.1021/acs.jproteome.6b00972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broadhead R, Dawe HR, Farr H, Griffiths S, Hart SR, Portman N, Shaw MK, Ginger ML, Gaskell SJ, McKean PG, Gull K. Flagellar motility is required for the viability of the bloodstream trypanosome. Nature. 2006;440:224–227. doi: 10.1038/nature04541. [DOI] [PubMed] [Google Scholar]
- Brooks ER. The University of Texas at Austin; 2014. Control of intraflagellar transport: studies of the planar cell polarity effector Fuz, the small GTPase Rsg1, and the novel protein TTC29. [Google Scholar]
- Casas E, Landrein N, Bonhivers M. Ultra expansion microscopy protocol with improved setup for upright and inverted microscopes. Protocols. 2022 https://www.protocols.io/view/ultra-expansion-microscopy-protocol-with-improved-bvwqn7dw
- Cooper TG, Noonan E, von Eckardstein S, Auger J, Baker HWG, Behre HM, Haugen TB, Kruger T, Wang C, Mbizvo MT, Vogelsong KM. World Health Organization reference values for human semen characteristics. Human Reproduction Update. 2010;16:231–245. doi: 10.1093/humupd/dmp048. [DOI] [PubMed] [Google Scholar]
- Coutton C, Zouari R, Abada F, Ben Khelifa M, Merdassi G, Triki C, Escalier D, Hesters L, Mitchell V, Levy R, Sermondade N, Boitrelle F, Vialard F, Satre V, Hennebicq S, Jouk PS, Arnoult C, Lunardi J, Ray PF. MLPA and sequence analysis of DPY19L2 reveals point mutations causing globozoospermia. Human Reproduction. 2012;27:2549–2558. doi: 10.1093/humrep/des160. [DOI] [PubMed] [Google Scholar]
- Coutton C, Escoffier J, Martinez G, Arnoult C, Ray PF. Teratozoospermia: spotlight on the main genetic actors in the human. Human Reproduction Update. 2015;21:455–485. doi: 10.1093/humupd/dmv020. [DOI] [PubMed] [Google Scholar]
- Coutton C, Vargas AS, Amiri-Yekta A, Kherraf ZE, Ben Mustapha SF, Le Tanno P, Wambergue-Legrand C, Karaouzène T, Martinez G, Crouzy S, Daneshipour A, Hosseini SH, Mitchell V, Halouani L, Marrakchi O, Makni M, Latrous H, Kharouf M, Deleuze JF, Boland A, Hennebicq S, Satre V, Jouk PS, Thierry-Mieg N, Conne B, Dacheux D, Landrein N, Schmitt A, Stouvenel L, Lorès P, El Khouri E, Bottari SP, Fauré J, Wolf JP, Pernet-Gallay K, Escoffier J, Gourabi H, Robinson DR, Nef S, Dulioust E, Zouari R, Bonhivers M, Touré A, Arnoult C, Ray PF. Mutations in CFAP43 and CFAP44 cause male infertility and flagellum defects in Trypanosoma and human. Nature Communications. 2018;9:686. doi: 10.1038/s41467-017-02792-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coutton C, Martinez G, Kherraf ZE, Amiri-Yekta A, Boguenet M, Saut A, He X, Zhang F, Cristou-Kent M, Escoffier J, Bidart M, Satre V, Conne B, Fourati Ben Mustapha S, Halouani L, Marrakchi O, Makni M, Latrous H, Kharouf M, Pernet-Gallay K, Bonhivers M, Hennebicq S, Rives N, Dulioust E, Touré A, Gourabi H, Cao Y, Zouari R, Hosseini SH, Nef S, Thierry-Mieg N, Arnoult C, Ray PF. Bi-allelic Mutations in ARMC2 Lead to Severe Astheno-Teratozoospermia Due to Sperm Flagellum Malformations in Humans and Mice. American Journal of Human Genetics. 2019;104:331–340. doi: 10.1016/j.ajhg.2018.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox J, Hein MY, Luber CA, Paron I, Nagaraj N, Mann M. Accurate Proteome-wide Label-free Quantification by Delayed Normalization and Maximal Peptide Ratio Extraction, Termed MaxLFQ. Molecular & Cellular Proteomics. 2014;13:2513–2526. doi: 10.1074/mcp.M113.031591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crozier TWM, Tinti M, Wheeler RJ, Ly T, Ferguson MAJ, Lamond AI. Proteomic analysis of the cell cycle of procylic form Trypanosoma brucei. Molecular & Cellular Proteomics. 2018;17:1184–1195. doi: 10.1074/mcp.RA118.000650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dacheux D, Landrein N, Thonnus M, Gilbert G, Sahin A, Wodrich H, Robinson DR, Bonhivers M. A MAP6-related protein is present in protozoa and is involved in flagellum motility. PLOS ONE. 2012;7:e31344. doi: 10.1371/journal.pone.0031344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D’Andrea LD, Regan L. TPR proteins: the versatile helix. Trends in Biochemical Sciences. 2003;28:655–662. doi: 10.1016/j.tibs.2003.10.007. [DOI] [PubMed] [Google Scholar]
- Darde TA, Lecluze E, Lardenois A, Stévant I, Alary N, Tüttelmann F, Collin O, Nef S, Jégou B, Rolland AD, Chalmel F, Kelso J. The ReproGenomics Viewer: a multi-omics and cross-species resource compatible with single-cell studies for the reproductive science community. Bioinformatics. 2019;35:3133–3139. doi: 10.1093/bioinformatics/btz047. [DOI] [PubMed] [Google Scholar]
- Dean S, Sunter J, Wheeler RJ, Hodkinson I, Gluenz E, Gull K. A toolkit enabling efficient, scalable and reproducible gene tagging in trypanosomatids. Open Biology. 2015;5:140197. doi: 10.1098/rsob.140197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dean S, Sunter JD, Wheeler RJ. TrypTag.org: A Trypanosome Genome-wide Protein Localisation Resource. Trends in Parasitology. 2017;33:80–82. doi: 10.1016/j.pt.2016.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dymek EE, Heuser T, Nicastro D, Smith EF. The CSC is required for complete radial spoke assembly and wild-type ciliary motility. Molecular Biology of the Cell. 2011;22:2520–2531. doi: 10.1091/mbc.E11-03-0271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ersfeld K, Gull K. Targeting of cytoskeletal proteins to the flagellum of Trypanosoma brucei. Journal of Cell Science. 2001;114:141–148. doi: 10.1242/jcs.114.1.141. [DOI] [PubMed] [Google Scholar]
- Gambarotto D, Zwettler FU, Le Guennec M, Schmidt-Cernohorska M, Fortun D, Borgers S, Heine J, Schloetel J-G, Reuss M, Unser M, Boyden ES, Sauer M, Hamel V, Guichard P. Imaging cellular ultrastructures using expansion microscopy (U-ExM) Nature Methods. 2019;16:71–74. doi: 10.1038/s41592-018-0238-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gambarotto D, Hamel V, Guichard P. In: Methods in Cell Biology, Expansion Microscopy for Cell Biology. Guichard P, Hamel V, editors. Academic Press; 2021. Chapter 4 - Ultrastructure expansion microscopy (U-Exm) pp. 57–81. [DOI] [PubMed] [Google Scholar]
- Kalichava A, Ochsenreiter T. Ultrastructure expansion microscopy in Trypanosoma brucei. Open Biology. 2021;11:210132. doi: 10.1098/rsob.210132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Käll L, Canterbury JD, Weston J, Noble WS, MacCoss MJ. Semi-supervised learning for peptide identification from shotgun proteomics datasets. Nature Methods. 2007;4:923–925. doi: 10.1038/nmeth1113. [DOI] [PubMed] [Google Scholar]
- Kherraf ZE, Amiri-Yekta A, Dacheux D, Karaouzène T, Coutton C, Christou-Kent M, Martinez G, Landrein N, Le Tanno P, Fourati Ben Mustapha S, Halouani L, Marrakchi O, Makni M, Latrous H, Kharouf M, Pernet-Gallay K, Gourabi H, Robinson DR, Crouzy S, Blum M, Thierry-Mieg N, Touré A, Zouari R, Arnoult C, Bonhivers M, Ray PF. A Homozygous Ancestral SVA-Insertion-Mediated Deletion in WDR66 Induces Multiple Morphological Abnormalities of the Sperm Flagellum and Male Infertility. American Journal of Human Genetics. 2018;103:400–412. doi: 10.1016/j.ajhg.2018.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim S, Scheffler K, Halpern AL, Bekritsky MA, Noh E, Källberg M, Chen X, Kim Y, Beyter D, Krusche P, Saunders CT. Strelka2: fast and accurate calling of germline and somatic variants. Nature Methods. 2018;15:591–594. doi: 10.1038/s41592-018-0051-x. [DOI] [PubMed] [Google Scholar]
- King SM. A solid-state control system for dynein-based ciliary/flagellar motility. The Journal of Cell Biology. 2013;201:173–175. doi: 10.1083/jcb.201302077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kulak NA, Pichler G, Paron I, Nagaraj N, Mann M. Minimal, encapsulated proteomic-sample processing applied to copy-number estimation in eukaryotic cells. Nature Methods. 2014;11:319–324. doi: 10.1038/nmeth.2834. [DOI] [PubMed] [Google Scholar]
- LaCount DJ, Barrett B, Donelson JE. Trypanosoma brucei FLA1 is required for flagellum attachment and cytokinesis. The Journal of Biological Chemistry. 2002;277:17580–17588. doi: 10.1074/jbc.M200873200. [DOI] [PubMed] [Google Scholar]
- Li H. Bwa-0.7.17. r1188Github. 2017 https://github.com/lh3/bwa
- Liu C, He X, Liu W, Yang S, Wang L, Li W, Wu H, Tang S, Ni X, Wang J, Gao Y, Tian S, Zhang L, Cong J, Zhang Z, Tan Q, Zhang J, Li H, Zhong Y, Lv M, Li J, Jin L, Cao Y, Zhang F. Bi-allelic Mutations in TTC29 Cause Male Subfertility with Asthenoteratospermia in Humans and Mice. American Journal of Human Genetics. 2019a;105:1168–1181. doi: 10.1016/j.ajhg.2019.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu W, He X, Yang S, Zouari R, Wang J, Wu H, Kherraf ZE, Liu C, Coutton C, Zhao R, Tang D, Tang S, Lv M, Fang Y, Li W, Li H, Zhao J, Wang X, Zhao S, Zhang J, Arnoult C, Jin L, Zhang Z, Ray PF, Cao Y, Zhang F. Bi-allelic Mutations in TTC21A Induce Asthenoteratospermia in Humans and Mice. American Journal of Human Genetics. 2019b;104:738–748. doi: 10.1016/j.ajhg.2019.02.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu C, Tu C, Wang L, Wu H, Houston BJ, Mastrorosa FK, Zhang W, Shen Y, Wang J, Tian S, Meng L, Cong J, Yang S, Jiang Y, Tang S, Zeng Y, Lv M, Lin G, Li J, Saiyin H, He X, Jin L, Touré A, Ray PF, Veltman JA, Shi Q, O’Bryan MK, Cao Y, Tan Y-Q, Zhang F. Deleterious variants in X-linked CFAP47 induce asthenoteratozoospermia and primary male infertility. American Journal of Human Genetics. 2021;108:309–323. doi: 10.1016/j.ajhg.2021.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- Lorès P, Dacheux D, Kherraf ZE, Nsota Mbango JF, Coutton C, Stouvenel L, Ialy-Radio C, Amiri-Yekta A, Whitfield M, Schmitt A, Cazin C, Givelet M, Ferreux L, Fourati Ben Mustapha S, Halouani L, Marrakchi O, Daneshipour A, El Khouri E, Do Cruzeiro M, Favier M, Guillonneau F, Chaudhry M, Sakheli Z, Wolf JP, Patrat C, Gacon G, Savinov SN, Hosseini SH, Robinson DR, Zouari R, Ziyyat A, Arnoult C, Dulioust E, Bonhivers M, Ray PF, Touré A. Mutations in TTC29, Encoding an Evolutionarily Conserved Axonemal Protein, Result in Asthenozoospermia and Male Infertility. American Journal of Human Genetics. 2019;105:1148–1167. doi: 10.1016/j.ajhg.2019.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu Y, Oura S, Matsumura T, Oji A, Sakurai N, Fujihara Y, Shimada K, Miyata H, Tobita T, Noda T, Castaneda JM, Kiyozumi D, Zhang Q, Larasati T, Young SAM, Kodani M, Huddleston CA, Robertson MJ, Coarfa C, Isotani A, Aitken RJ, Okabe M, Matzuk MM, Garcia TX, Ikawa M. CRISPR/Cas9-mediated genome editing reveals 30 testis-enriched genes dispensable for male fertility in mice†. Biology of Reproduction. 2019;101:501–511. doi: 10.1093/biolre/ioz103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luck K, Kim D-K, Lambourne L, Spirohn K, Begg BE, Bian W, Brignall R, Cafarelli T, Campos-Laborie FJ, Charloteaux B, Choi D, Coté AG, Daley M, Deimling S, Desbuleux A, Dricot A, Gebbia M, Hardy MF, Kishore N, Knapp JJ, Kovács IA, Lemmens I, Mee MW, Mellor JC, Pollis C, Pons C, Richardson AD, Schlabach S, Teeking B, Yadav A, Babor M, Balcha D, Basha O, Bowman-Colin C, Chin S-F, Choi SG, Colabella C, Coppin G, D’Amata C, De Ridder D, De Rouck S, Duran-Frigola M, Ennajdaoui H, Goebels F, Goehring L, Gopal A, Haddad G, Hatchi E, Helmy M, Jacob Y, Kassa Y, Landini S, Li R, van Lieshout N, MacWilliams A, Markey D, Paulson JN, Rangarajan S, Rasla J, Rayhan A, Rolland T, San-Miguel A, Shen Y, Sheykhkarimli D, Sheynkman GM, Simonovsky E, Taşan M, Tejeda A, Tropepe V, Twizere J-C, Wang Y, Weatheritt RJ, Weile J, Xia Y, Yang X, Yeger-Lotem E, Zhong Q, Aloy P, Bader GD, De Las Rivas J, Gaudet S, Hao T, Rak J, Tavernier J, Hill DE, Vidal M, Roth FP, Calderwood MA. A reference map of the human binary protein interactome. Nature. 2020;580:402–408. doi: 10.1038/s41586-020-2188-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaren W, Gil L, Hunt SE, Riat HS, Ritchie GRS, Thormann A, Flicek P, Cunningham F. The ensembl variant effect predictor. Genome Biology. 2016;17:122. doi: 10.1186/s13059-016-0974-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meienberg J, Bruggmann R, Oexle K, Matyas G. Clinical sequencing: is WGS the better WES? Human Genetics. 2016;135:359–362. doi: 10.1007/s00439-015-1631-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nsota Mbango JF, Coutton C, Arnoult C, Ray PF, Touré A. Genetic causes of male infertility: snapshot on morphological abnormalities of the sperm flagellum. Basic and Clinical Andrology. 2019;29:2. doi: 10.1186/s12610-019-0083-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Donnell L. Mechanisms of spermiogenesis and spermiation and how they are disturbed. Spermatogenesis. 2014;4:e979623. doi: 10.4161/21565562.2014.979623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oud MS, Smits RM, Smith HE, Mastrorosa FK, Holt GS, Houston BJ, de Vries PF, Alobaidi BKS, Batty LE, Ismail H, Greenwood J, Sheth H, Mikulasova A, Astuti GDN, Gilissen C, McEleny K, Turner H, Coxhead J, Cockell S, Braat DDM, Fleischer K, D’Hauwers KWM, Schaafsma E, Nagirnaja L, Conrad DF, Friedrich C, Kliesch S, Aston KI, Riera-Escamilla A, Krausz C, Gonzaga-Jauregui C, Santibanez-Koref M, Elliott DJ, Vissers LELM, Tüttelmann F, O’Bryan MK, Ramos L, Xavier MJ, van der Heijden GW, Veltman JA, Genetics of Male Infertility Initiative (GEMINI) consortium A de novo paradigm for male infertility. Nature Communications. 2022;13:154. doi: 10.1038/s41467-021-27132-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plagnol V, Curtis J, Epstein M, Mok KY, Stebbings E, Grigoriadou S, Wood NW, Hambleton S, Burns SO, Thrasher AJ, Kumararatne D, Doffinger R, Nejentsev S. A robust model for read count data in exome sequencing experiments and implications for copy number variant calling. Bioinformatics. 2012;28:2747–2754. doi: 10.1093/bioinformatics/bts526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ralston KS, Kabututu ZP, Melehani JH, Oberholzer M, Hill KL. The Trypanosoma brucei flagellum: moving parasites in new directions. Annual Review of Microbiology. 2009;63:335–362. doi: 10.1146/annurev.micro.091208.073353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ralston KS, Kisalu NK, Hill KL. Structure-function analysis of dynein light chain 1 identifies viable motility mutants in bloodstream-form Trypanosoma brucei. Eukaryotic Cell. 2011;10:884–894. doi: 10.1128/EC.00298-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolland M, Le Moal J, Wagner V, Royère D, De Mouzon J. Decline in semen concentration and morphology in a sample of 26 609 men close to general population between 1989 and 2005 in France. Human Reproduction. 2013;28:462–470. doi: 10.1093/humrep/des415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez JY, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A. Fiji: an open-source platform for biological-image analysis. Nature Methods. 2012;9:676–682. doi: 10.1038/nmeth.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nature Methods. 2012;9:671–675. doi: 10.1038/nmeth.2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schumann Burkard G, Jutzi P, Roditi I. Genome-wide RNAi screens in bloodstream form trypanosomes identify drug transporters. Molecular and Biochemical Parasitology. 2011;175:91–94. doi: 10.1016/j.molbiopara.2010.09.002. [DOI] [PubMed] [Google Scholar]
- Shamoto N, Narita K, Kubo T, Oda T, Takeda S. CFAP70 Is a Novel Axoneme-Binding Protein That Localizes at the Base of the Outer Dynein Arm and Regulates Ciliary Motility. Cells. 2018;7:124. doi: 10.3390/cells7090124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sunter JD. A vanillic acid inducible expression system for Trypanosoma brucei. Molecular and Biochemical Parasitology. 2016;207:45–48. doi: 10.1016/j.molbiopara.2016.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thierry-Mieg N. Grexome-TIMC-primary TIMC primary. swh:1:rev:6313685f72a875cfe99afd14aa70845710c87b0fSoftware Heritage. 2023a https://archive.softwareheritage.org/swh:1:dir:02ba691d357bb0a6ee6cf1e179452f76f558a15b;origin=https://github.com/ntm/grexome-TIMC-Primary;visit=swh:1:snp:842436a9effd973e5faf993dda0e38dee037b85b;anchor=swh:1:rev:6313685f72a875cfe99afd14aa70845710c87b0f
- Thierry-Mieg N. Grexome-TIMC-secondary. swh:1:rev:0f454b4ea9bf98a2531afae269d92225213b37d3Software Heritage. 2023b https://archive.softwareheritage.org/swh:1:dir:9f599d31a987f0042dbce9056002f969412e2591;origin=https://github.com/ntm/grexome-TIMC-Secondary;visit=swh:1:snp:d51c2d44d22bb01d4b1b9028d1ea5b15028537b4;anchor=swh:1:rev:0f454b4ea9bf98a2531afae269d92225213b37d3
- Touré A, Martinez G, Kherraf ZE, Cazin C, Beurois J, Arnoult C, Ray PF, Coutton C. The genetic architecture of morphological abnormalities of the sperm tail. Human Genetics. 2021;140:21–42. doi: 10.1007/s00439-020-02113-x. [DOI] [PubMed] [Google Scholar]
- Tyanova S, Temu T, Sinitcyn P, Carlson A, Hein MY, Geiger T, Mann M, Cox J. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nature Methods. 2016;13:731–740. doi: 10.1038/nmeth.3901. [DOI] [PubMed] [Google Scholar]
- Vincensini L, Blisnick T, Bastin P. 1001 model organisms to study cilia and flagella. Biology of the Cell. 2011;103:109–130. doi: 10.1042/BC20100104. [DOI] [PubMed] [Google Scholar]
- Wang G, Guo Y, Zhou T, Shi X, Yu J, Yang Y, Wu Y, Wang J, Liu M, Chen X, Tu W, Zeng Y, Jiang M, Li S, Zhang P, Zhou Q, Zheng B, Yu C, Zhou Z, Guo X, Sha J. In-depth proteomic analysis of the human sperm reveals complex protein compositions. Journal of Proteomics. 2013;79:114–122. doi: 10.1016/j.jprot.2012.12.008. [DOI] [PubMed] [Google Scholar]
- Wang Y, Yang J, Jia Y, Xiong C, Meng T, Guan H, Xia W, Ding M, Yuchi M. Variability in the morphologic assessment of human sperm: use of the strict criteria recommended by the World Health Organization in 2010. Fertility and Sterility. 2014;101:945–949. doi: 10.1016/j.fertnstert.2013.12.047. [DOI] [PubMed] [Google Scholar]
- Wang WL, Tu CF, Tan YQ. Insight on multiple morphological abnormalities of sperm flagella in male infertility: what is new? Asian Journal of Andrology. 2020;22:236–245. doi: 10.4103/aja.aja_53_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wickstead B, Gull K. Dyneins across eukaryotes: A comparative genomic analysis. Traffic. 2007;8:1708–1721. doi: 10.1111/j.1600-0854.2007.00646.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wirtz E, Leal S, Ochatt C, Cross GA. A tightly regulated inducible expression system for conditional gene knock-outs and dominant-negative genetics in Trypanosoma brucei. Molecular and Biochemical Parasitology. 1999;99:89–101. doi: 10.1016/s0166-6851(99)00002-x. [DOI] [PubMed] [Google Scholar]
- Yamamoto R, Yagi T, Kamiya R. Functional binding of inner-arm dyneins with demembranated flagella of Chlamydomonas mutants. Cell Motility and the Cytoskeleton. 2006;63:258–265. doi: 10.1002/cm.20121. [DOI] [PubMed] [Google Scholar]
- Yamamoto R, Yanagisawa H-A, Yagi T, Kamiya R. Novel 44-kilodalton subunit of axonemal Dynein conserved from chlamydomonas to mammals. Eukaryotic Cell. 2008;7:154–161. doi: 10.1128/EC.00341-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto R, Hwang J, Ishikawa T, Kon T, Sale WS. Composition and function of ciliary inner-dynein-arm subunits studied in Chlamydomonas reinhardtii. Cytoskeleton. 2021;78:77–96. doi: 10.1002/cm.21662. [DOI] [PMC free article] [PubMed] [Google Scholar]