Abstract
Purpose
This study was aimed to assess the effectiveness of Glucagon-like peptide 1 receptor agonists on pregnancy rate, menses, anthropometric and hormonal parameters in PCOS patients.
Methods
We conducted searches of the published literature in PubMed, EMBASE, Cochrane Library, Web of Science up to September 2022. Data from randomized controlled trials were obtained to assess the effects of GLP1RAs in PCOS women. Weighted mean difference, standardized mean difference, and risks ratio were employed for effect size estimation using a random-effects model.
Results
A total of 840 patients with 469 individuals in GLP1RAs group and 371 individuals in control group from 11 RCTs were included. GLP1RAs usage was associated with an improvement in natural pregnancy rate (RR: 1.72, 95% CI 1.22 to 2.43, P = 0.002, I2 = 0%) and menstrual regularity (SMD: 1.72, 95% CI 0.60 to 2.85, P < 0.001, I2 = 95.6%). There were no statistically significant differences in total pregnancy rate, IVF pregnancy rate between two groups, but total PR elevated in a short time after GLP1RAs as shown in subgroup analysis. Randomization to GLP1RAs treatment was associated with great improvement in HOMA-IR, BMI, WC, SHBG and a slight reduction in TT compared to control group. A decrease in TBF was seen in European population. GLP1RAs monotherapy was not superior to metformin when it came to fT, DHEAS, FAI.
Conclusions
Prescription of GLP1RAs improves natural pregnancy rate, menstrual cyclicity and insulin sensitivity, anthropometrics, hormonal indexes in PCOS women.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12902-023-01500-5.
Keywords: Polycystic ovary syndrome, Glucagon-like peptide 1, Pregnancy rate, Menstrual patterns, Meta-analysis
Introduction
Polycystic ovary syndrome (PCOS) is a common disorder affecting 4 to 21% reproductive-age women and also the leading cause of anovulatory infertility [1, 2]. Apart from its impact on ovulatory dysfunction, PCOS also affects overall health of women with long-lasting effects even in post-menstrual period [3]. Though first described in 1935, the etiology of the disease is still not well elucidated, which involves both genetic and environmental factors [4, 5]. Rotterdam criteria is the universally accepted diagnostic criteria and concludes the diagnosis of PCOS should be based on at least two of three major criteria, including oligo- or anovulation, clinical and/or biochemical signs of hyperandrogenism and polycystic ovaries identified by ultrasonography after excluding other androgen excess disorders [2]. Up to 80% affected women are overweight or obese, and insulin resistance (IR) occurs in most PCOS patients even in normal-weight population [6–8]. PCOS related obesity and IR also significantly increase the risk of diabetes mellitus, cardiovascular disease, and nonalcoholic steatohepatitis [9–11].
Glucagon-like peptide 1 is one of the most thoroughly studied incretins and its receptors are widely expressed in pancreas, gastrointestinal tract, heart, and even brain [12]. Agonists towards GLP1 receptors are recognized as popular anti-diabetic agents which can inhibit gastric emptying, increase measures of satiety, decrease food intake and thus cause weight reduction. GLP1RAs have recently become popular in diabetes and obese patients for its pronounced weight-losing effects, insulin-sensitizing function and additional cardiovascular protective benefits [13–16]. Furthermore, It has been discovered that GLP1R mRNA expressed in thalamus and hypothalamus, indicating potential ability of GLP1RAs to regulate GnRH from the hypothalamic neurons via a specific GLP-1R [17, 18]. Animal studies also have found GLP1 receptors expressed in ovary cells [19]. Improvement of reproductive dysfunction including follicles development, recovery of estrous and menstrual cycles and even reverse of polycystic ovary morphology were seen in several animal studies, though some studies presented with controversial results [20–24]. Given these, PCOS women particularly those obese population could benefit from GLP1RAs treatment.
A few clinical trials have shown that GLP1RAs administration improves pregnancy rate, menstrual frequency, obesity, excess of androgen and insulin levels in PCOS patients [25–28]. Former meta-analysis studies have mainly focused on anthropometric, metabolic, hormonal changes after GLP1RAs administration [29–31]. None has investigated conception rate changes or pregnancy outcomes which actually disturbed most patients. Herein, we perform this meta-analysis to evaluate the role of GLP1RAs in the management of PCOS patients especially with regard to reproductive health.
Methods
This systematic review and meta-analysis was conducted according to the guidelines for the preferred reporting items for systematic reviews and meta-analyses (PRISMA) [32].
Search strategy
A systematic search was performed in PubMed, Embase, Web of Science and Cochrane library covering the period up to September 22nd, 2022. The search terms used included the following: (“polycystic ovary syndrome” or “PCOS” or “polycystic ovary”) and (“Glucagon-Like Peptide 1” or “GLP1” or “liraglutide” or “exenatide”), and detailed search terms were given in Supplementary material. Additionally, manual checks of the reference lists within both the original literature and the reviews were also performed to identify eligible studies.
Inclusion and exclusion criteria
Inclusion criteria were as follows: (1) Confirmed diagnosis of PCOS based on Rotterdam criteria, National Institute of Health criteria or Androgen Excess Society criteria, (2) GLP1RAs prescription as intervention, (3) Either a placebo or active agents as control, (4) reporting on pregnancy rate and/or menstrual regularity changes, (5) RCTs on human.
Exclusion criteria were as follows: (1) The diagnostic criteria were not clearly stated, (2) case reports, review articles, editorials, letters, conference abstracts, non-RCTs, etc., (3) studies without outcomes of interest, (4) studies without accessible data to perform quantitative analysis, (5) studies that were republished or duplicated using same participant cohorts. Articles in languages other than English were also excluded.
Study selection and data extraction
LZ and HQ independently identified and selected relevant articles. Decisions on the inclusion of a full-text review were made and examined by both reviewers. Any discrepancies between the 2 investigators were solved by LY or discussion among all the reviewers.
Data were extracted from the studies including (1) study information: the trial name, author details, year of publication, country and diagnosis criteria for PCOS, (2) participant characteristics: age, baseline BMI, sample size and drugs(including dose usage and duration) of experimental and control groups, lifestyle instruction, (3) outcomes of interest: pregnancy rate; menstrual frequency changes; anthropometric parameters mainly BMI, waist circumference, body fat percent and HOMA-IR changes; alterations in SHBG level and androgen indexes including TT, fT, DHEAS and FAI. Similarly, the process were carried out by LZ and HQ separately. If discrepancies were found, the data in the original study were extracted again by discussion between the 2 reviewers until they reached an agreement.
Quality assessment
The Cochrane risk of bias tool with RevMan 5.4 was utilized to assess the quality of the included studies [33]. Six domains of bias (selection, performance, detection, attrition, reporting, and other biases) were assessed. Two authors (LZ and HQ) independently assessed risk of bias in each study, using ‘low risk’, ‘high risk’ and ‘unclear risk’ of bias. Any disagreements were resolved by discussion, and the ultimate decision was reached with a third author.
Statistical analysis
Statistical analysis was performed with Stata version 15.1 (Stata Corp., College Station, TX, USA) and RevMan 5.4 (The Nordic Cochrane Center, The Cochrane Collaboration). What we finally need is mean change and its corresponding standard deviation, which we can calculate based on baseline and after-treatment mean and SD [34]. One study presented with differences from baseline as mean (95% CI) [28], therefore we adopted an additional calculator tool available on Cochrane website to transform 95%CI into SD (available on: https://training.cochrane.org/resource/revman-calculator). Data of control groups were separated equally in two studies, as there were two experimental groups reported [25, 35]. Continuous data were expressed as WMD (same measure methods and units between groups) or SMD (different measure methods and units between groups), while dichotomous data were expressed as RR. All analyses were carried out using random-effects models for more conservative estimates. Heterogeneity between studies was evaluated by the X2 test and I2 statistic, and P values < 0.05 or I2 values > 50% were indicative of substantial heterogeneity. We also conducted subgroup analyses according to different drugs usage or other clinical variances such as race, medication duration etc. Furthermore, meta-regression analysis was performed to explore the potential factors for significant heterogeneity. A 2-tailed P value of less than 0.05 was considered statistically significant in all analyses. Sensitivity analyses were performed to examine the stability of pooled effect size results by serially excluding each study. Potential publication bias was evaluated by visual inspection of funnel plots as well as with Egger regression asymmetry test if the number of analyzed items was more than ten [36].
Results
Search results
As shown in Fig. 1, we identified 775 relevant studies of which 327 were duplicates, and 363 studies were excluded after screening titles and abstracts. The remaining 85 articles were further subjected to full-text review and 74 studies were subsequently excluded for reasons. Finally, a total of 11 studies were eligible for this meta-analysis [25–28, 35, 37–42].
Fig. 1.
Flowchart showing the study selection process
Basic characteristics
A pooled population of 840 patients was included, with 469 individuals in GLP1RAs arm and 371 individuals in control arm. Table 1 shows the characteristics of the included RCTs. Seven studies employed Rotterdam criteria for diagnosis and four used NIH criteria. All participants were aged 18 years older and were overweight or obese. Lifestyle modifications were promoted during medication in seven studies. Intervention groups received either GLP1RAs monotherapy or GLP1RAs plus metformin combined therapy, whilst patients in control groups used placebo or metformin alone. Exenatide was used in five studies [25–27, 38, 41] while liraglutide was adopted in others. GLP1RAs usage duration ranged from 12 to 32 weeks and dosage also varied from low dose (EXE 5ug bid) to maximum dose (LIRA 3 mg qd). In addition, since metformin is known to have beneficial effects in PCOS, any improvement in the prespecified outcome parameters or even equivalence would place GLP1RAs in a favorable position.
Table 1.
Characteristics of the included RCTs
| Authors (Publication year) | Diagnosis criteria (Country) | Interventionduration | Interventions (Sample size) | Lifestyle intervention | Trial group (Age;baseline BMI) | Control group (Age;baseline BMI) | Outcomes |
|---|---|---|---|---|---|---|---|
| Mojca Jensterle [37] (2015) | NICHD (Slovenia) | 12wk |
LIRA 1.2 mg QD (n = 17) vs MET 1000 mg BID (n = 15) |
Not actively promoted |
29.5 ± 7.7(y); 40.8 ± 6.1 kg/m2 |
25.3 ± 5.2(y); 38.2 ± 7 kg/m2 |
MF, weight, metabolic, endocrine changes, TBF |
| Karen Elkind-Hirsch [25] (2008) | Rotterdam (Louisiana) | 24wk |
COMBI (EXE 10 μg BID + MET 1000 mg BID) (n = 20) vs EXE 10 μg BID (n = 20) vs MET 1000 mg BID (n = 20) |
On unrestricted diet |
COMBI: 32.1 ± 0.7(y); 41.2 ± 1.7 kg/m2 EXE:28.2 ± 1.1(y); 39.9 ± 1.5 kg/m2 |
27.7 ± 1.3(y); 41.3 ± 1.8 kg/m2 |
MF, ovulation rate, insulin action, anthropometric measures, androgen levels, inflammatory markers |
| Renyuan Li [26] (2022) | Rotterdam (China) | 12wk |
EXE 5–10 μg BID (n = 80)*12wk + MET 1000 mg BID*52wk vs MET 1000 mg BID (n = 80)*12wk + MET 1000 mg BID*52wk |
Actively promoted |
28.19 ± 3.96(y); 29.07 ± 3.92 kg/m2 |
27.83 ± 3.52(y); 29.15 ± 4.11 kg/m2 |
Spontaneous and total PR, pregnancy outcomes, metabolic parameters |
| Juan Wang [27] (2017) | Rotterdam (China) | 3 month |
EXE 10 μg BID (n = 45) vs MET 500–2000 mg daily (n = 33) |
NM |
25.92 ± 6.75(y); 26.26 ± 5.71 kg/m2 |
25.67 ± 7.33(y); 25.74 ± 6.37 kg/m2 |
PR, sex hormone, ovulation rate, glucose metabolic index, AngII/Ang(1–7) |
| Malin Nylander [28] (2017) | Rotterdam (Denmark) | 26wk |
LIRA 1.8 mg QD (n = 48) vs Placebo 1.8 mg QD (n = 24) |
NM |
31.4(24.6–35.6)(y); 33.3 ± 5.1 kg/m2 |
26.2(24.8–31.5)(y); 33.3 ± 4.6 kg/m2 |
MF, ovarian and stromal volume, antral follicle count, sex hormone |
| Xin Liu [38] (2017) | Rotterdam (China) | 12wk |
EXE 10 μg BID*12wk + MET*12wk (n = 88) vs MET 1000 mg BID*12wk + MET*12wk (n = 88) |
Promoted |
27.93 ± 2.70(y); 29.16 ± 3.11 kg/m2 |
27.69 ± 3.80(y); 28.29 ± 1.86 kg/m2 |
Natural PR, MF, weight, BMI, TBF, metabolic and endocrine changes, inflammatory markers |
| Karen E. Elkind-Hirsch [39] (2022) | NIH (Louisiana) | 32wk |
LIRA 3 mg QD (n = 55) vs Placebo 3 mg QD (n = 27) |
Promoted |
31 ± 0.8(y); 41.6 ± 0.9 kg/m2 |
32 ± 1.1(y); 43.9 ± 1.5 kg/m2 |
MF, weight and FAI, waist circumference, BMI, sex hormone, OGTT result, insulin action, lipids profile, BP, TBF |
| Vesna Salamun [40] (2018) | Rotterdam (Slovenija) | 12wk |
COMBI (LIRA 1.2 mg QD + MET 1000 mg BID) (n = 14) vs MET 1000 mg BID (n = 14) |
Actively promoted |
30.1 ± 3.6(y); 37.8 ± 3.0 kg/m2 |
31.1 ± 4.7(y); 35.5 ± 4.9 kg/m2 |
Cumulative PR(IVF and spontaneous); oocyte and embryo quality, weight, metabolic and endocrine parameters, TBF |
| Siyuan Zheng [41] (2017) | Rotterdam (China) | 12wk |
EXE 10 μg BID (n = 41) vs MET 1000 mg BID (n = 41) |
Promoted |
27.7 ± 3.41(y); 29.18 ± 4.15 kg/m2 |
28.16 ± 3.92(y); 29 ± 4.1 kg/m2 |
MF, weight, hirsutism, metabolic and endocrine change, inflammatory marker levels |
| Mojca Jensterle [42] (2014) | NICHD (Slovenia) | 12wk |
LIRA 1.2 mg QD (n = 15) vs MET 1000 mg BID (n = 15) |
Promoted |
30.7 ± 7.9(y)a; 36.7 ± 5.6 kg/m2 |
30.7 ± 7.9(y)a; 39.4 ± 6.9 kg/m2 |
MF, weight, metabolic and endocrine change |
| Mojca Jensterle Sever [35] (2014) | NICHD (Slovenia) | 12wk |
COMBI (LIRA 1.2 mg QD + MET 1000 mg BID) (n = 13) vs LIRA 1.2 mg QD (n = 13) vs MET 1000 mg BID (n = 14) |
Not actively promoted |
COMBI:31.1 ± 5.1(y); 37.6 ± 5.1 kg/m2 LIRA:31.5 ± 6.4(y); 39.3 ± 4.2 kg/m2 |
31.3 ± 9.4(y); 36.6 ± 3.5 kg/m2 |
MF, weight, metabolic and endocrine change, TBF |
aAge were not given separately
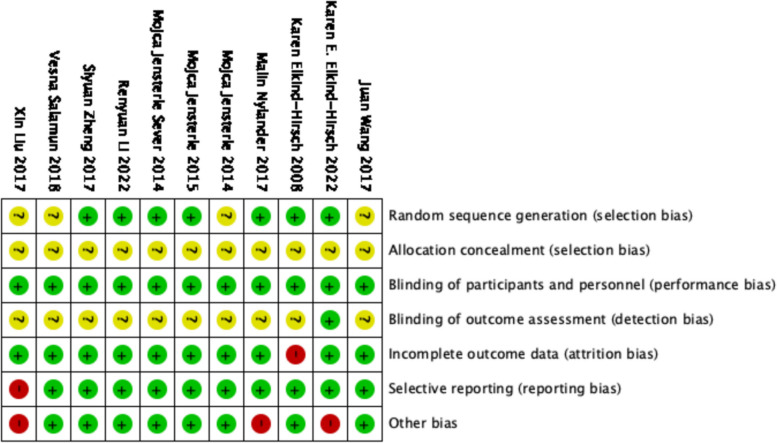
Risk of bias of included studies
As shown in Fig. 2, adequate method of random sequence generation was described in seven trials, including RAND program in Excel or computer-generated randomization list [25, 26, 28, 35, 37, 39, 41]. The other four gave unspecific randomization procedure and had an unclear risk [27, 38, 40, 42]. None provided with a detail of allocation concealment, so it was considered an uncertain risk of selection bias. Two studies [28, 39] were double-blinded, one study [38] was single-blinded, other studies were open-label, but they were all considered to be at low risk of performance bias because outcome measures were objective results. Ten studies were assessed unclear risk of detection bias except study from Elkind-Hirsch et al. [39]. One study had a quite high missing rate (more than 30%) at final assessment, but information on the number of participants who dropped out and the corresponding reasons were given [25]. Study from Liu et al. was viewed of high risks in reporting bias and other bias [38, 43]. Others were at low risk for selecting outcome reports. Two studies received funding from pharmaceutical companies and thus were deemed to be at high risk for other bias [28, 39].
Fig. 2.
Assessment of the risk of bias
Effect on pregnancy rate
Four studies reported pregnancy rate in total. Significant improvements were observed in spontaneous PR (RR: 1.72, 95% CI 1.22 to 2.43, I2 = 0%, P = 0.002) without great heterogeneity. However, there was no difference in the IVF PR (RR: 1.06, 95% CI 0.69 to 1.64, I2 = 8.3%, P = 0.791) or total PR (RR: 1.35, 95% CI 0.96 to 1.91, I2 = 40.7%, P = 0.087). Subgroup analysis showed follow-up duration might explain the heterogeneity in total PR, that is, treatment with GLP1RAs could only improve total pregnancy rate in the following time period of less than 1 year (Fig. 3). Sensitivity analysis for total PR showed substantial change in heterogeneity when excluding Li et al.’s study [26], so as the estimate of effect size (RR 1.71; 95% CI 1.17 to 2.51, I2 = 0%, P = 0.005) (sensitivity analysis results shown in Supplementary Figs. S1-S3).
Fig. 3.
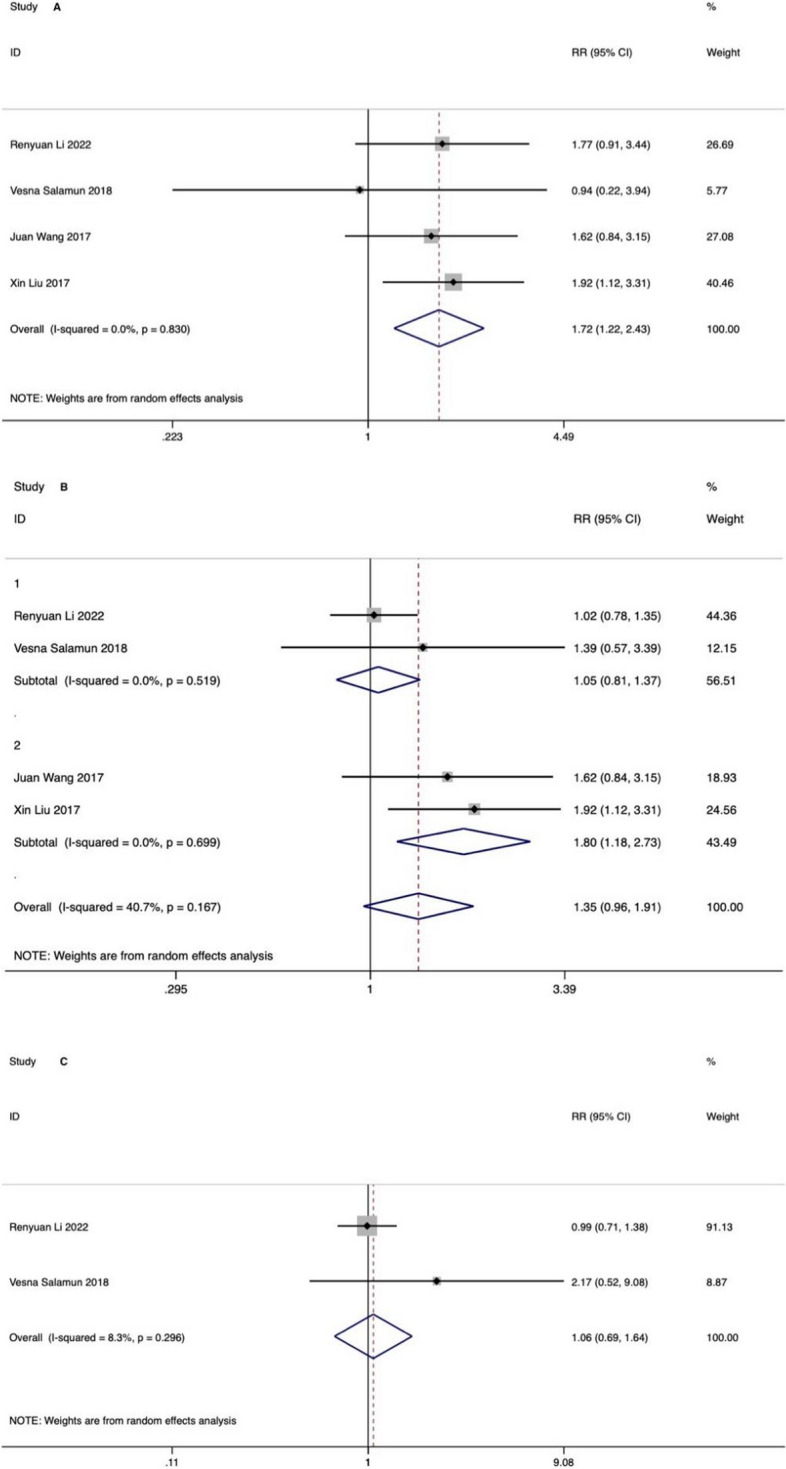
Effect on pregnancy rate: A natural pregnancy rate, B total pregnancy rate, C IVF pregnancy rate; 1 (follow-up more than 1 year), 2 (follow-up less than 1 year)
Effect on menstrual cycles
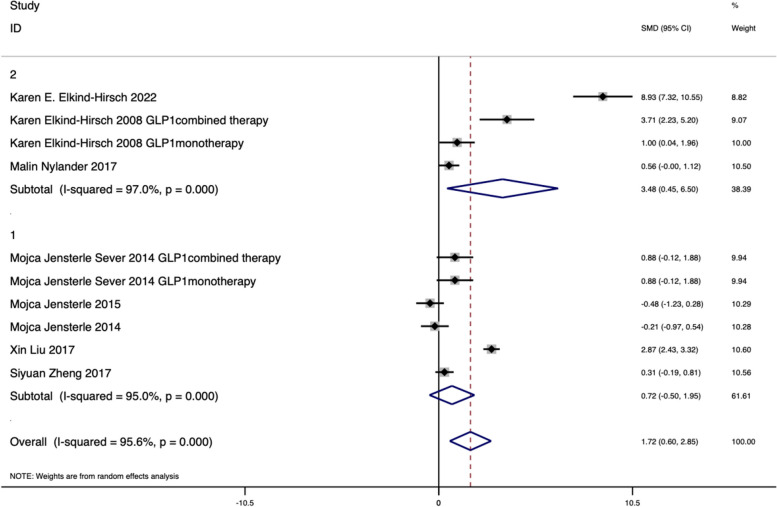
Menstrual cyclicity was documented in eight RCTs. The pooled result revealed that adding GLP1RAs treatment was more effective than metformin alone or placebo in improving menstrual frequency (SMD: 1.72, 95% CI 0.60 to 2.85, I2 = 95.6%, P < 0.001). Subgroup analysis showed longer treatment duration was associated with menstrual cyclicity improvement, that is, PCOS participants who were treated with GLP1RAs for 24, 26 and 32 weeks respectively in three trials [25, 28, 39] had the most favourable changes in menstrual pattern compared those treated for 12 weeks (shown in Fig. 4). The heterogeneities were high in both overall analysis and subgroup analysis, but further sensitivity analysis demonstrated the result robust (shown in Supplementary Fig. S4). The meta-regression analysis also indicated treatment duration was a potential influencing factor (shown in Supplementary Tab. S1). The funnel plot for the menstrual patterns was given in Supplementary Fig. S5 (Harbord-Egger test, P = 0.55).
Fig. 4.
Effect on menstrual cycles: 1 (GLP1RAs treatment duration for 12wk), 2 (GLP1RAs treatment duration for more than 12wk)
Effect on anthropometrics and HOMA-IR
Significant improvements were observed in BMI and waist circumference, as presented in Table 2. Subgroup analysis indicated significant reduction in BMI were seen for GLP1RAs versus placebo or metformin, but not in combined treatment versus metformin monotherapy. There was no difference in total body fat percentage change in two arms and ethnicity might explain the heterogeneity through subgroup analysis. HOMA-IR was improved when adding GLP1RAs treatment. The funnel plots for BMI, WC and HOMA-IR were shown in Supplementary Figs. S7, S9 and S12 (Harbord-Egger test, P = 0.97, P = 0.67, P = 0.26, respectively) (sensitivity analysis results shown in Supplementary Figs. S6, S8, S10 and S11).
Table 2.
Effect on anthropometrics and HOMA-IR
| Study Group | No. of Studies/Subgroups | Heterogeneity | Effects Estimate (95% CI) | P value |
|---|---|---|---|---|
| BMI (WMD) | 9 | P = 0.147, I2 = 31.5% | -1.21 (-1.74, -0.68) | P < 0.001 |
| GLP1RA + MET vs MET | 3 | P = 0.825, I2 = 0% | -0.71 (-2.15, 0.73) | P = 0.335 |
| GLP1RA vs MET | 7 | P = 0.218, I2 = 27.6% | -1.05 (-1.69, -0.42) | P = 0.001 |
| GLP1RA vs Placebo | 1 | Not Applicable | -2.00 (-2.79, -1.21) | P < 0.001 |
| WC (WMD) | 9 | P = 0.470, I2 = 0% | -2.90 (-3.65, -2.16) | P < 0.001 |
| TBF (WMD) | 5 | P = 0.003, I2 = 72.5% | -1.44 (-2.93, 0.05) | P = 0.058 |
| European | 5 | P = 0.772, I2 = 0% | -1.84 (-4.60, 0.92) | P < 0.001 |
| Chinese | 1 | Not Applicable | -1.30 (-1.73, -0.87) | P < 0.001 |
| HOMA-IR (WMD) | 10 | P = 0.573, I2 = 0% | -0.65 (-0.80, -0.51) | P < 0.001 |
Effect on sex hormones
A slight reduction of TT was noticed in GLP1RAs group but the effect was gone after subgroup analysis and heterogeneity was still high, as demonstrated in Table 3. The pooled results showed no difference in fT or DHEAS levels but subgroup analysis showed a significant decrease in DHEAS when adding GLP1RAs to metformin monotherapy. GLP1RAs administration elevated SHBG compared to control group. A reduction of FAI was noticed in pooled result and subgroup analysis showed GLP1RAs did equally to metformin monotherapy. The funnel plots for TT and SHBG were shown in Supplementary Figs. S14 and S18 (Harbord-Egger test, P = 0.874, P = 0.214, respectively) (sensitivity analysis results shown in Supplementary Figs. S13-S19).
Table 3.
Effect on sex hormones
| Study Group | No. of Studies/subgroups | Heterogeneity | Effects Estimate(95% CI) | P Value |
|---|---|---|---|---|
| TT (SMD) | 10 | P < 0.001, I2 = 77.3% | -0.39 (-0.78, -0.002) | P = 0.049 |
| GLP1RA + MET vs MET | 3 | P = 0.008, I2 = 79.4% | -0.63 (-1.81, 0.55) | P = 0.293 |
| GLP1RA vs MET | 7 | P = 0.009, I2 = 65% | -0.15 (-0.53, 0.24) | P = 0.465 |
| GLP1RA vs Placebo | 2 | P = 0.001, I2 = 90.2% | -1.02 (-2.27, -0.23) | P = 0.110 |
| fT(SMD) | 5 | P = 0.102, I2 = 45.5% | -0.16 (-0.59, 0.27) | P = 0.463 |
| GLP1RA + MET vs MET | 2 | P = 0.671, I2 = 0% | -0.23 (-0.83, 0.36) | P = 0.444 |
| GLP1RA vs MET | 3 | P = 0.109, I2 = 54.8% | 0.16 (-0.57, 0.89) | P = 0.665 |
| GLP1RA vs Placebo | 1 | Not Applicable | -0.66 (-1.19, -0.13) | P = 0.015 |
| DHEAS (SMD) | 7 | P = 0.002, I2 = 67.1% | -0.40 (-0.82, 0.01) | P = 0.058 |
| GLP1RA + MET vs MET | 2 | P = 0.375, I2 = 0% | -0.76 (-1.44, -0.08) | P = 0.029 |
| GLP1RA vs MET | 6 | P = 0.557, I2 = 0% | -0.08 (-0.35, 0.18) | P = 0.542 |
| GLP1RA vs Placebo | 1 | Not Applicable | -1.43 (-1.99, -0.87) | P < 0.001 |
| SHBG (WMD) | 9 | P = 0.099, I2 = 37.5% | 4.42 (2.18, 6.67) | P < 0.001 |
| GLP1RA + MET vs MET | 3 | P = 0.665, I2 = 0% | 10.19 (6.29, 14.09) | P < 0.001 |
| GLP1RA vs MET | 7 | P = 0.203, I2 = 29.5% | 3.07 (0.63, 5.51) | P = 0.014 |
| GLP1RA vs Placebo | 1 | Not Applicable | 5.40 (-0.25, 11.05) | P = 0.061 |
| FAI(WMD) | 7 | P = 0.019, I2 = 58.4% | -1.55 (-2.59, -0.51) | P = 0.004 |
| GLP1RA + MET vs MET | 1 | Not Applicable | -3.50 (-5.39, -1.61) | P < 0.001 |
| GLP1RA vs MET | 5 | P = 0.037, I2 = 60.8% | -0.04 (-2.41, 2.32) | P = 0.973 |
| GLP1RA vs Placebo | 2 | P = 0.570, I2 = 0% | -1.74 (-2.05, -1.43) | P < 0.001 |
Discussion
To our best knowledge, it is the first meta-analysis exploring the role of GLP1RAs in reproductive aspects of PCOS women. Our study indicates a significant improvement in natural pregnancy rate following GLP1RAs intervention, and total pregnancy rate also elevates though merely within a short period. However, there is no increase in the rate of IVF pregnancy. Longer duration of GLP1RAs prescription leads to a more favorable menstrual regularity. Besides, GLP1RAs treatment is associated with improvement in HOMA-IR, increment in SHBG, reduction in BMI, WC and TT but not in fT, DHEAS or FAI. A decrease in TBF is also noticed in European population subgroup.
Main alterations in HPO axis in PCOS contain an increase of LH activity, follicles resistance to FSH, hypersecretion of androgens from ovarian theca cell and thereby inhibition of dominant follicle maturation and regular ovulating. The prevalence of infertility and menstrual disorders among PCOS women ranges from approximately 40% to 75%, and almost 40% PCOS patients fail to conceive spontaneously and turn to seek help from ART [1, 2, 44]. Besides, they have a higher chance encountering complications during pregnancy or even beyond, including miscarriage, GDM, preeclampsia, premature delivery, increased perinatal mortality and a higher risk of metabolic disorders for offspring [45, 46].
Here in this study, natural pregnancy rate with GLP1RAs prescription alone or in combination with metformin was improved when compared to metformin monotherapy. And the pooled effect size was not reversed by individual studies, indicating the result robust. Given that the estimated RR was increased after exclusion of the study by Wang J et al. [27] wherein metformin was not provided in the GLP1RAs group, we assume that the combination therapy of GLP1RAs plus metformin may be more efficient than either GLP1RAs or metformin monotherapy in terms of raising natural pregnancy rate. A similar result was not found in IVF pregnancy rate, which may be partly because it also relies on the clinical experience of fertility doctors and techniques of IVF procedures. Subgroup analysis result showed that total pregnancy rate elevated during a temporary period but not in the following 1-year-long time after such a short term usage (no more than 12 weeks) of GLP1RAs. It is also worth exploring whether a extension of GLP1RAs treatment duration could impose a more long-lasting improvement in pregnancy rate among PCOS patients.
To be mentioned with, baseline characteristics and intervention in four studies focusing on pregnancy rate were quite different, though without great heterogeneity. Firstly, participants in study from Salamun et al. were poorly responsive to lifestyle modifications and resistant to the first-line reproductive treatments with CC or aromatase inhibitors previously, which might underestimate pregnancy rate after GLP1RAs. Secondly, though treatment duration of GLP1RAs were similar, the drug species, dosage, concomitant medication (metformin) usage and its corresponding continuance varied: one study compared GLP1RAs monotherapy with metformin [27], however the others also employed metformin in experimental groups which may exert an add-on effect. Thirdly, Salamun et al. adopted a drug washout period after interventions, Wang et al. combined with CC in both groups while all patients in Liu and Li et al.’s study were switched to metformin treatment for another 12 weeks and 52 weeks respectively until pregnancy confirmed. Lastly, we intended to include RCTs to reduce the likelihood of recall and selection bias, but among four studies, three of them were open-label and one was single-blind clinical trials, which to some extent increased the risk of performance and detection bias.
Menstrual frequency also increased in 273 participants treated with GLP1RAs versus 208 patients in control arms, though the heterogeneity was quite high due to different intervention protocols and the enrolling of PCOS patients of different phenotypes. Further meta regression analysis showed treatment duration might be the source of heterogeneity. Those who received 12 weeks of GLP1RAs therapy had an increase in menstrual frequency but not in a statistically significant manner. Meanwhile, patients in the longer-duration subgroup had a more preferable improvement. Accordingly, we recommend that a continuous administration of GLP1RAs for at least 24 weeks may be plausible to restore a regular menstrual cycle [25]. However, due to the high heterogeneity, inferences should be drawn cautiously and further large-scale, well-designed RCTs are warranted to determine a minimum treatment duration.
Significant enhancements in anthropometric parameters and insulin sensitivity were obtained in GLP1RAs group, which were also in line with former studies [30, 31]. Waist girth, a simple reflection of abdominal obesity, decreased profoundly but the reduction of total body fat was simply seen in European patients. The improvement in SHBG level may be attributed to increased insulin sensitivity. GLP1RAs monotherapy did equally to metformin in androgen excess, but there seemed to be an add-on effect of GLP1RAs in lowering DHEAS and FAI. Further studies can be carried out to discuss the effect of GLP1RAs on body composition, fat distribution change, hyperandrogenism in people from different ethnic backgrounds.
Of note, GLP1RAs is a category C drug for pregnancy by FDA and EMA and its administration in women preparing for pregnancy should include a washout period. There are evidences of fetus toxicity in animal studies, but a few studies have reported a normal birth and no adverse outcomes in women exposed to GLP1RAs in their first terms of pregnancy [47, 48]. Only one trial herein documented the incidence rate of gestational complications and adverse outcomes, which turned out to be similar between groups [26]. Due to the scarce of the human studies investigating the safety of GLP1RAs usage before or during gestation, the related meta-analysis result cannot be performed here.
The plausible mechanisms underlying the beneficial effect of GLP1RAs on the reproductive function in PCOS women are listed as below. Firstly, obesity and insulin resistance, as aggravating factors for infertility, are alleviated within GLP1RAs usage. It is suggested that an increase in pregnancy rate is in accordance with a reduction in HOMA-IR [49]. Higher circulating insulin level is associated with subsequent excess ovarian androgen production and reduction of SHBG. A negative feedback on HPO axis from androgens aromatization in adipose tissues worsens gonadotropin activity. GLP1RAs administration demonstrates substantial insulin sensitivity enhancement and profound weight loss effect and thus inhibits a vicious cycle among obesity, IR, hyperandrogenism, which promise its benefits in PCOS patients [15, 20, 22, 50].
Moreover, as aforementioned, GLP-1 receptors are widely distributed, including HPO axis. Preclinical researches have provided an additional perspective that GLP1RAs exerts effects directly via hypothalamus-pituitary-gonadal axis. For instance, GLP-1 and Exendin-4 can act on the gonadal axis, involving the hypothalamic kiss-1 system, to influence reproductive efficiency in female rats [51]. Wu et al. demonstrated that dulaglutide, a long-acting GLP1RA, may reduce the hyperandrogenemia of PCOS rats by regulating the expression of steroid hormone synthesis related gene proteins in the ovary and thus improving the morphology of their polycystic ovaries [52]. Granulosa cells in ovaries are essential in the follicular development and dominant follicle selection. Sun et al. reported the contribution of GLP-1 to the regulation of ovarian granulosa cells proliferation and antiapoptosis, thereby promoting oocyte maturation in PCOS rats [53]. To sum up, preclinical studies have found potential direct roles of GLP1RAs in HPO axis, but whether the conclusion could be extended to human beings needs to be verified.
In addition, low-grade chronic inflammation and adipose tissue dysfunction cannot be ignored. Higher levels of inflammatory markers including CRP, IL-18, IL-6 and TNF-α in PCOS women have been widely reported [54, 55]. Artunc-Ulkumen et al. found exenatide could protect endometrial and ovarian microenvironments against oxidative stress, fibrosis, and degeneration [56]. Previous studies also have showed a decrease in inflammatory markers in PCOS patients treated with GLP1RAs, suggesting an anti-inflammatory effect of GLP1RAs at the cellular and molecular level [21, 57].
Strengths and limitations
This meta-analysis incorporated recently published studies and addressed the potential role of GLP1RAs in reproductive aspects in PCOS patients. Since no prior meta-analysis has focused on the improvement of pregnancy rate, menstrual frequency, hyperandrogenism, HOMA-IR, and obesity-related parameters simultaneously, our study is more complete and thorough. Besides, we also performed a subgroup analysis to investigate the effects of GLP1RAs monotherapy or combination with metformin in the management of PCOS. Meanwhile, we utilized subgroup analysis, meta-regression, and sensitivity analysis to explain heterogeneities in pooled results.
However, our acceptances to both experimental and control group due to the paucity of available data, a lack of information on which disease phenotype of each participants belonged to and no uniformity of lifestyle intervention, GLP1RAs type or treatment duration, led to a moderate to high heterogeneity in some main outcomes. Secondly, participants from some studies also received metformin treatment, the results should be interpreted with caution since a true comparison between GLP1RAs and placebo was not available. Furthermore, most studies enrolled obese participants and were not specifically designed to evaluate menstrual patterns, which might cause bias. Finally, the small number of studies in subgroup and meta-regression analysis may have allowed for under powered analyses results.
Conclusions
The present study reveals that the use of GLP-1RAs contributes to a higher natural pregnancy rate and a more regular menstrual frequency, improvement in obesity, insulin resistance, gonadal parameters mainly on SHBG. It is worth noting that GLP1RAs treatment in obese PCOS patients can be a new therapeutic option beyond the goal of weight loss. Still, more long-term, large-scale, multi-ethnic, phenotype-specific, well-designed trials are warranted to confirm the efficacy and safety of GLP1RAs in preconceptional PCOS women. Besides, intensive research into the mechanisms by which GLP1RAs affect the reproductive health is still needed.
Supplementary Information
Additional file 1: Fig. S1. Sensitivity analysis of natural pregnancy rate. Fig. S2. Sensitivity analysis of total pregnancy rate. Fig. S3. Sensitivity analysis of IVF pregnancy rate. Fig. S4. Sensitivity analysis of menstrual cycles. Fig. S5. Funnel plot of menstrual cycles. Tab. S1. Summary of Meta-Regression Analyses for Menstrual Cycles. Fig. S6. Sensitivity analysis of BMI. Fig. S7. Funnel plot of BMI. Fig. S8. Sensitivity analysis of WC. Fig. S9. Funnel plot of WC. Fig. S10. Sensitivity analysis of TBF. Fig. S11. Sensitivity analysis of HOMA-IR. Fig. S12. Funnel plot of HOMA-IR. Fig. S13. Sensitivity analysis of TT. Fig. S14. Funnel plot of TT. Fig. S15. Sensitivity analysis of fT. Fig. S16. Sensitivity analysis of DHEAS. Fig. S17. Sensitivity analysis of SHBG. Fig. S18. Funnel plot of SHBG. Fig. S19. Sensitivity analysis of FAI.
Acknowledgements
We acknowledge all patients participated and staff involved in the studies.
Abbreviations
- GLP1RAs
Glucagon-like peptide 1 receptor agonists
- PCOS
Polycystic ovary syndrome
- RCT
Randomized controlled trial
- WMD
Weighted mean difference
- SMD
Standardized mean difference
- RR
Risks ratio
- CI
Confidence interval
- SD
Standard deviations
- IR
Insulin resistance
- GI
Gastrointestinal
- GnRH
Gonadotropin-releasing hormone
- LIRA
Liraglutuide
- EXE
Exenatide
- MET
Metformin
- CC
Clomifene citrate
- NM
Not mentioned
- BID
Twice a day
- QD
Once a day
- COMBI
Combined group
- MF
Menstrual frequency
- PR
Pregnancy rate
- IVF
In vitro fertilization
- TBF
Total body fat
- BMI
Body mass index
- WC
Waist circumference
- HOMA-IR
Homeostasis model assessment-insulin resistance
- TT
Total testosterone
- fT
Free testosterone
- FAI
Free androgen index
- DHEAS
Dehydroepiandrosterone sulphate
- SHBG
Sex hormone binding globulin
- OGTT
Oral glucose tolerance test
- BP
Blood pressure
- ART
Assisted reproductive technology
- GDM
Gestational diabetes mellitus
- FDA
Food and Drug Administration
- EMA
European Medicines Agency
- HPO axis
Hypothalamus pituitary ovary axis
- CRP
C-reactive protein
- IL-18
Interleukin 18
- TNF-α
Tumor necrosis factor α
- IL-6
Interleukin 6
Authors’ contributions
LZ came up with the subject. LZ, HQ and LY participated in the formulation of retrieval strategies, data acquisition, data analysis, quality assessment. LZ and HQ wrote the manuscript. LY and LS critically revised the manuscript. All authors contributed to the article and approved the submitted version.
Funding
This study was supported by grants from the Medical Science and Technology Project of Zhejiang Province (2023RC234).
Availability of data and materials
The original contributions presented in the study are included in the article or supplementary material. Further inquiries can be directed to the corresponding author.
Declarations
Ethics approval and consent to participate
The included studies’ results were published online. The relevant institutional review boards or ethics committees approved the research protocol of the individual clinical trail used in the current analysis, and all human participants gave written informed consent, which was demonstrated in the respective original papers.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Lingling Zhou and Huanjia Qu contributed equally to this work.
References
- 1.Yildiz BO, Bozdag G, Yapici Z, Esinler I, Yarali H. Prevalence, phenotype and cardiometabolic risk of polycystic ovary syndrome under different diagnostic criteria. Hum Reprod. 2012;27(10):3067–73. doi: 10.1093/humrep/des232. [DOI] [PubMed] [Google Scholar]
- 2.Lizneva D, Suturina L, Walker W, Brakta S, Gavrilova-Jordan L, Azziz R. Criteria, prevalence, and phenotypes of polycystic ovary syndrome. Fertil Steril. 2016;106(1):6–15. doi: 10.1016/j.fertnstert.2016.05.003. [DOI] [PubMed] [Google Scholar]
- 3.Cooney LG, Dokras A. Beyond fertility: polycystic ovary syndrome and long-term health. Fertil Steril. 2018;110(5):794–809. doi: 10.1016/j.fertnstert.2018.08.021. [DOI] [PubMed] [Google Scholar]
- 4.Ben-Shlomo I, Younis JS. Basic research in PCOS: are we reaching new frontiers? Reprod Biomed Online. 2014;28(6):669–83. doi: 10.1016/j.rbmo.2014.02.011. [DOI] [PubMed] [Google Scholar]
- 5.Diamanti-Kandarakis E, Piperi C, Spina J, Argyrakopoulou G, Papanastasiou L, Bergiele A, et al. Polycystic ovary syndrome: the influence of environmental and genetic factors. Hormones (Athens) 2006;5(1):17–34. doi: 10.14310/horm.2002.11165. [DOI] [PubMed] [Google Scholar]
- 6.Barber TM, Franks S. Obesity and polycystic ovary syndrome. Clin Endocrinol (Oxf) 2021;95(4):531–41. doi: 10.1111/cen.14421. [DOI] [PubMed] [Google Scholar]
- 7.Li W, Chen Q, Xie Y, Hu J, Yang S, Lin M. Prevalence and degree of insulin resistance in Chinese Han women with PCOS: results from euglycemic-hyperinsulinemic clamps. Clin Endocrinol (Oxf) 2019;90(1):138–44. doi: 10.1111/cen.13860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Stepto NK, Cassar S, Joham AE, Hutchison SK, Harrison CL, Goldstein RF, Teede HJ. Women with polycystic ovary syndrome have intrinsic insulin resistance on euglycaemic-hyperinsulaemic clamp. Hum Reprod. 2013;28(3):777–84. doi: 10.1093/humrep/des463. [DOI] [PubMed] [Google Scholar]
- 9.Osibogun O, Ogunmoroti O, Michos ED. Polycystic ovary syndrome and cardiometabolic risk: opportunities for cardiovascular disease prevention. Trends Cardiovasc Med. 2020;30(7):399–404. doi: 10.1016/j.tcm.2019.08.010. [DOI] [PubMed] [Google Scholar]
- 10.Paschou SA, Polyzos SA, Anagnostis P, Goulis DG, Kanaka-Gantenbein C, Lambrinoudaki I, Georgopoulos NA, Vryonidou A. Nonalcoholic fatty liver disease in women with polycystic ovary syndrome. Endocrine. 2020;67(1):1–8. doi: 10.1007/s12020-019-02085-7. [DOI] [PubMed] [Google Scholar]
- 11.Jellinger PS, Handelsman Y, Rosenblit PD, Bloomgarden ZT, Fonseca VA, Garber AJ, et al. American Association of Clinical Endocrinologists and American College of Endocrinology guidelines for management of dyslipidemia and prevention of cardiovascular disease. Endocr Pract. 2017;23(Suppl 2):1–87. doi: 10.4158/EP171764.APPGL. [DOI] [PubMed] [Google Scholar]
- 12.Pyke C, Heller RS, Kirk RK, Ørskov C, Reedtz-Runge S, Kaastrup P, Hvelplund A, Bardram L, Calatayud D, Knudsen LB. GLP-1 receptor localization in monkey and human tissue: novel distribution revealed with extensively validated monoclonal antibody. Endocrinology. 2014;155(4):1280–90. doi: 10.1210/en.2013-1934. [DOI] [PubMed] [Google Scholar]
- 13.Drucker DJ. Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell Metab. 2018;27(4):740–56. doi: 10.1016/j.cmet.2018.03.001. [DOI] [PubMed] [Google Scholar]
- 14.Andersen A, Lund A, Knop FK, Vilsboll T. Glucagon-like peptide 1 in health and disease. Nat Rev Endocrinol. 2018;14(7):390–403. doi: 10.1038/s41574-018-0016-2. [DOI] [PubMed] [Google Scholar]
- 15.Yaribeygi H, Sathyapalan T, Sahebkar A. Molecular mechanisms by which GLP-1 RA and DPP-4i induce insulin sensitivity. Life Sci. 2019;234:116776. doi: 10.1016/j.lfs.2019.116776. [DOI] [PubMed] [Google Scholar]
- 16.Bertoccini L, Baroni MG. GLP-1 receptor agonists and SGLT2 inhibitors for the treatment of type 2 diabetes: new insights and opportunities for cardiovascular protection. Adv Exp Med Biol. 2021;1307:193–212. doi: 10.1007/5584_2020_494. [DOI] [PubMed] [Google Scholar]
- 17.Abdalla MA, Deshmukh H, Atkin S, Sathyapalan T. The potential role of incretin-based therapies for polycystic ovary syndrome: a narrative review of the current evidence. Ther Adv Endocrinol Metab. 2021;12:2042018821989238. doi: 10.1177/2042018821989238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Beak SA, Heath MM, Small CJ, Morgan DG, Ghatei MA, Taylor AD, Buckingham JC, Bloom SR, Smith DM. Glucagon-like peptide-1 stimulates luteinizing hormone-releasing hormone secretion in a rodent hypothalamic neuronal cell line. J Clin Invest. 1998;101(6):1334–41. doi: 10.1172/JCI610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang X, Lai S, Sun Z, Shen J. Glucagon-like peptide 1 receptor localization and quantitative expression in the ovaries of polycystic ovary syndrome mice. Diabetes Metab Res Rev. 2018;34:59. doi: 10.1002/dmrr.3079. [DOI] [Google Scholar]
- 20.Singh A, Fernandes JRD, Chhabra G, Krishna A, Banerjee A. Liraglutide modulates adipokine expression during adipogenesis, ameliorating obesity, and polycystic ovary syndrome in mice. Endocrine. 2019;64(2):349–366. doi: 10.1007/s12020-019-01891-3. [DOI] [PubMed] [Google Scholar]
- 21.Sun L, Ji C, Jin L, Bi Y, Feng W, Li P, Shen S, Zhu D. Effects of exenatide on metabolic changes, sexual hormones, inflammatory cytokines, adipokines, and weight change in a DHEA-treated rat model. Reprod Sci. 2016;23(9):1242–9. doi: 10.1177/1933719116635278. [DOI] [PubMed] [Google Scholar]
- 22.Xing C, Lv B, Zhao H, Wang D, Li X, He B. Metformin and exenatide upregulate hepatocyte nuclear factor-4alpha, sex hormone binding globulin levels and improve hepatic triglyceride deposition in polycystic ovary syndrome with insulin resistance rats. J Steroid Biochem Mol Biol. 2021;214:105992. doi: 10.1016/j.jsbmb.2021.105992. [DOI] [PubMed] [Google Scholar]
- 23.Tao X, Cai L, Chen L, Ge S, Deng X. Effects of metformin and Exenatide on insulin resistance and Ampkalpha-Sirt1 molecular pathway in PCOS rats. J Ovarian Res. 2019;12(1):86. doi: 10.1186/s13048-019-0555-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hoang V, Bi J, Mohankumar SM, Vyas AK. Liraglutide improves hypertension and metabolic perturbation in a rat model of polycystic ovarian syndrome. PLoS One. 2015;10(5):e0126119. doi: 10.1371/journal.pone.0126119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Elkind-Hirsch K, Marrioneaux O, Bhushan M, Vernor D, Bhushan R. Comparison of single and combined treatment with exenatide and metformin on menstrual cyclicity in overweight women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2008;93(7):2670–8. doi: 10.1210/jc.2008-0115. [DOI] [PubMed] [Google Scholar]
- 26.Li R, Mai T, Zheng S, Zhang Y. Effect of metformin and exenatide on pregnancy rate and pregnancy outcomes in overweight or obese infertility PCOS women: long-term follow-up of an RCT. Arch Gynecol Obstet. 2022 doi: 10.1007/s00404-022-06700-3. [DOI] [PubMed] [Google Scholar]
- 27.Wang J, Ruan X, Jin F, Sun Y, Wang J. Effects of exenatide combined with clomifene citrate on insulin resistance and angiotensin II/Angiotensin-(1–7) in peripheral blood in patients with polycystic ovary syndrome. Biomed Res (India) 2017;28(19):8406–8411. [Google Scholar]
- 28.Nylander M, Frøssing S, Clausen HV, Kistorp C, Faber J, Skouby SO. Effects of liraglutide on ovarian dysfunction in polycystic ovary syndrome: a randomized clinical trial. Reprod Biomed Online. 2017;35(1):121–7. doi: 10.1016/j.rbmo.2017.03.023. [DOI] [PubMed] [Google Scholar]
- 29.Han Y, Li Y, He B. Glp-1 receptor agonists versus metformin in PCOS: a systematic review and meta-analysis. Reprod Biomed Online. 2019;39(2):332–42. doi: 10.1016/j.rbmo.2019.04.017. [DOI] [PubMed] [Google Scholar]
- 30.Ge JJ, Wang DJ, Song W, Shen SM, Ge WH. The effectiveness and safety of liraglutide in treating overweight/obese patients with polycystic ovary syndrome: a meta-analysis. J Endocrinol Invest. 2022;45(2):261–73. doi: 10.1007/s40618-021-01666-6. [DOI] [PubMed] [Google Scholar]
- 31.Guo XN, Zhou ZB, Lyu XR, Xu HY, Zhu HJ, Pan H, et al. The antiobesity effect and safety of GLP-1 receptor agonist in overweight/obese patients without diabetes: a systematic review and meta-analysis. Horm Metab Res. 2022;54(07):458–471. doi: 10.1055/a-1844-1176. [DOI] [PubMed] [Google Scholar]
- 32.Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Rev Esp Cardiol (Engl Ed) 2021;74(9):790–9. doi: 10.1016/j.rec.2021.07.010. [DOI] [PubMed] [Google Scholar]
- 33.Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Higgins JPT, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions: Version 5.0.0 [Updated February]. The Cochrane Collaboration; 2008. Available from www.training.cochrane.org/handbook.
- 35.Jensterle Sever M, Kocjan T, Pfeifer M, Kravos NA, Janez A. Short-term combined treatment with liraglutide and metformin leads to significant weight loss in obese women with polycystic ovary syndrome and previous poor response to metformin. Eur J Endocrinol. 2014;170(3):451–459. doi: 10.1530/EJE-13-0797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–34. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jensterle M, Kravos NA, Pfeifer M, Kocjan T, Janez A. A 12-week treatment with the long-acting glucagon-like peptide 1 receptor agonist liraglutide leads to significant weight loss in a subset of obese women with newly diagnosed polycystic ovary syndrome. Hormones (Athens) 2015;14(1):81–90. doi: 10.1007/BF03401383. [DOI] [PubMed] [Google Scholar]
- 38.Liu X, Zhang Y, Zheng SY, Lin R, Xie YJ, Chen H, Zheng YX, Liu E, Chen L, Yan JH, Xu W, Mai TT, Gong Y. Efficacy of exenatide on weight loss, metabolic parameters and pregnancy in overweight/obese polycystic ovary syndrome. Clin Endocrinol. 2017;87(6):767–774. doi: 10.1111/cen.13454. [DOI] [PubMed] [Google Scholar]
- 39.Elkind-Hirsch KE, Chappell N, Shaler D, Storment J, Bellanger D. Liraglutide 3 mg on weight, body composition, and hormonal and metabolic parameters in women with obesity and polycystic ovary syndrome: a randomized placebo-controlled-phase 3 study. Fertil Steril. 2022;118(2):371–81. doi: 10.1016/j.fertnstert.2022.04.027. [DOI] [PubMed] [Google Scholar]
- 40.Salamun V, Jensterle M, Janez A, Vrtacnik Bokal E. Liraglutide increases IVF pregnancy rates in obese PCOS women with poor response to first-line reproductive treatments: a pilot randomized study. Eur J Endocrinol. 2018;179(1):1–11. doi: 10.1530/eje-18-0175. [DOI] [PubMed] [Google Scholar]
- 41.Zheng S, Zhang Y, Long T, Lu J, Liu X, Yan J, Chen L, Gong Y, Wang F. Short term monotherapy with exenatide is superior to metformin in weight loss, improving insulin resistance and inflammation in Chinese overweight/obese PCOS women. Obes Med. 2017;7:15–20. doi: 10.1016/j.obmed.2017.06.003. [DOI] [Google Scholar]
- 42.Jensterle M, Salamun V, Kocjan T, VrtacnikBokal E, Janez A. Short term monotherapy with GLP-1 receptor agonist liraglutide or PDE 4 inhibitor roflumilast is superior to metformin in weight loss in obese PCOS women: a pilot randomized study. J Ovarian Res. 2015;8:32. doi: 10.1186/s13048-015-0161-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Legro RS. Letter to Liu et al.’s “Efficacy of exenatide on weight loss, metabolic parameters and pregnancy in overweight/obese polycystic ovary syndrome”. Clin Endocrinol (Oxf) 2018;88:607. doi: 10.1111/cen.13474. [DOI] [PubMed] [Google Scholar]
- 44.Teede H, Deeks A, Moran L. Polycystic ovary syndrome: a complex condition with psychological, reproductive and metabolic manifestations that impacts on health across the lifespan. BMC Med. 2010;8:41. doi: 10.1186/1741-7015-8-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Palomba S, de Wilde MA, Falbo A, Koster MP, La Sala GB, Fauser BC. Pregnancy complications in women with polycystic ovary syndrome. Hum Reprod Update. 2015;21(5):575–92. doi: 10.1093/humupd/dmv029. [DOI] [PubMed] [Google Scholar]
- 46.Shawky NM. Cardiovascular disease risk in offspring of polycystic ovary syndrome. Front Endocrinol. 2022;13:977819. doi: 10.3389/fendo.2022.977819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Williams J, Pomeroy NE, Pop-Busui R, Lash R, Douyon L, Chames M, et al. Case report exenatide use during pregnancy. Endocrinologist. 2009;19(3):119–121. doi: 10.1097/TEN.0b013e3181a5875e. [DOI] [Google Scholar]
- 48.Livadas S, Androulakis I, Angelopoulos N, Lytras A, Papagiannopoulos F, Kassi G. Liraglutide administration improves hormonal/metabolic profile and reproductive features in women with HAIR-AN syndrome. Endocrinol Diabetes Metab Case Rep. 2020;2020(1):1–4. doi: 10.1530/EDM-19-0150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Liu Y, Li J, Yan Z, Liu D, Ma J, Tong N. Improvement of insulin sensitivity increases pregnancy rate in infertile PCOS women: a systemic review. Front Endocrinol. 2021;12:657889. doi: 10.3389/fendo.2021.657889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Vatankhah A, Izadi M, Rezvani ME, Esmailidehaj M. The effect of exenatide, a glucagon like peptide, on body weight, fasting blood glucose, lipid profile and Hba1c in rats with polycystic ovarian syndrome. J Reprod Infertil. 2016;17(2):201. [Google Scholar]
- 51.Outeiriño-Iglesias V, Romaní-Pérez M, González-Matías LC, Vigo E, Mallo F. GLP-1 increases preovulatory LH source and the number of mature follicles, as well as synchronizing the onset of puberty in female rats. Endocrinology. 2015;156(11):4226–37. doi: 10.1210/en.2014-1978. [DOI] [PubMed] [Google Scholar]
- 52.Wu LM, Wang YX, Zhan Y, Liu AH, Wang YX, Shen HF, Wang YF, Wang LY, Tao ZB, Wang YQ. Dulaglutide, a long-acting GLP-1 receptor agonist, can improve hyperandrogenemia and ovarian function in DHEA-induced PCOS rats. Peptides. 2021;145:170624. doi: 10.1016/j.peptides.2021.170624. [DOI] [PubMed] [Google Scholar]
- 53.Sun Z, Li P, Wang X, Lai S, Qiu H, Chen Z, Hu S, Yao J, Shen J. GLP-1/GLP-1R signaling regulates ovarian PCOS-associated granulosa cells proliferation and antiapoptosis by modification of forkhead box protein O1 phosphorylation sites. Int J Endocrinol. 2020;2020:1484321. doi: 10.1155/2020/1484321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Alkhuriji AF, Al Omar SY, Babay ZA, El-Khadragy MF, Mansour LA, Alharbi WG, Khalil MI. Association of IL-1β, IL-6, TNF-Α, and TGFβ1 gene polymorphisms with recurrent spontaneous abortion in polycystic ovary syndrome. Dis Markers. 2020;2020:6076274. doi: 10.1155/2020/6076274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rudnicka E, Suchta K, Grymowicz M, Calik-Ksepka A, Smolarczyk K, Duszewska AM, Smolarczyk R, Meczekalski B. Chronic low grade inflammation in pathogenesis of PCOS. Int J Mol Sci. 2021;22(7):3789. doi: 10.3390/ijms22073789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Artunc-Ulkumen B, Pala HG, Pala EE, Yavasoglu A, Yigitturk G, Erbas O. Exenatide improves ovarian and endometrial injury and preserves ovarian reserve in streptozocin induced diabetic rats. Gynecol Endocrinol. 2015;31(3):196–201. doi: 10.3109/09513590.2014.975686. [DOI] [PubMed] [Google Scholar]
- 57.Chaudhuri A, Ghanim H, Vora M, Sia CL, Korzeniewski K, Dhindsa S, Makdissi A, Dandona P. Exenatide exerts a potent antiinflammatory effect. J Clin Endocrinol Metab. 2012;97(1):198–207. doi: 10.1210/jc.2011-1508. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Fig. S1. Sensitivity analysis of natural pregnancy rate. Fig. S2. Sensitivity analysis of total pregnancy rate. Fig. S3. Sensitivity analysis of IVF pregnancy rate. Fig. S4. Sensitivity analysis of menstrual cycles. Fig. S5. Funnel plot of menstrual cycles. Tab. S1. Summary of Meta-Regression Analyses for Menstrual Cycles. Fig. S6. Sensitivity analysis of BMI. Fig. S7. Funnel plot of BMI. Fig. S8. Sensitivity analysis of WC. Fig. S9. Funnel plot of WC. Fig. S10. Sensitivity analysis of TBF. Fig. S11. Sensitivity analysis of HOMA-IR. Fig. S12. Funnel plot of HOMA-IR. Fig. S13. Sensitivity analysis of TT. Fig. S14. Funnel plot of TT. Fig. S15. Sensitivity analysis of fT. Fig. S16. Sensitivity analysis of DHEAS. Fig. S17. Sensitivity analysis of SHBG. Fig. S18. Funnel plot of SHBG. Fig. S19. Sensitivity analysis of FAI.
Data Availability Statement
The original contributions presented in the study are included in the article or supplementary material. Further inquiries can be directed to the corresponding author.