Abstract
Growing use of carbon nanotubes (CNTs) have garnered concerns regarding their association with adverse health effects. Few studies have probed how CNTs affect a host’s susceptibility to pathogens, particularly respiratory viruses. We reported that exposure of lung cells and mice to pristine single-walled CNTs (SWCNTs) leads to significantly increased influenza virus H1N1 strain A/Mexico/4108/2009 (IAV) titers in concert with repressed antiviral immune responses. In the present study, we investigated if hydroxylated multi-walled CNTs (MWCNTs), would result in similar outcomes. C57BL/6 mice were exposed to 20 μg MWCNTs on day 0 and IAV on day 3 and samples were collected on day 7. We investigated pathological changes, viral titers, immune-related gene expression in lung tissue, and quantified differential cell counts and cytokine and chemokine levels in bronchoalveolar lavage fluid. MWCNTs alone caused mild inflammation with no apparent changes in immune markers whereas IAV alone presented typical infection-associated inflammation, pathology, and titers. The co-exposure (MWCNTs + IAV) did not alter titers or immune cell profiles compared to the IAV only but increased concentrations of IL-1β, TNFα, GM-CSF, KC, MIPs, and RANTES and inhibited mRNA expression of Tlr3, Rig-i, Mda5, and Ifit2. Our findings suggest MWCNTs modulate immune responses to IAV with no effect on the viral titer and modest pulmonary injury, a result different from those reported for SWCNT exposures. This is the first study to show that MWCNTs modify cytokine and chemokine responses that control aspects of host defenses which may play a greater role in mitigating IAV infections.
Keywords: multi-walled carbon nanotubes, influenza A H1N1 virus, C57BL/6 mouse, viral titers, innate immune responses
1. Introduction
Carbon nanotubes (CNTs) are a type of carbon allotrope with single or concentric cylinders of carbon sheets, held together by van der Waals forces, to yield a tubular structure with a diameter ranging from a few nanometers to 10’s of nanometers (Guzman et al., 2006). Due to their outstanding physicochemical properties, such as high tensile strength and thermal and electrical conductivity, CNTs are among the most popular nanomaterials (NMs) that are currently utilized in various commercial and industrial sectors (De Volder et al., 2013). Worldwide production of CNTs has increased more than 10-fold since 2006 (De Volder et al., 2013) and the global CNT market is projected to reach 3.42 billion by 2022(Grand View Research, 2015). Multiwalled carbon nanotubes (MWCNTs) account for the largest fraction of the global market volume of CNTs (Grand View Research, 2015). With such an increase in their manufacture and the development of CNT-containing products, concerns with regard to safety issues related to human exposure to them are rising, particularly in occupational settings (Dong and Ma, 2015), and this necessitates systematic evaluation.
The human respiratory tract occupies a relatively large surface area that traverses areas with diverse architecture, functions, and cells. Inhaled particles larger than about 10 microns in diameter get trapped in the upper respiratory tract, whereas those smaller than about 2.5 microns in diameter, such as CNTs, can be inhaled into the deeper airways (Fujita et al., 2015). Data from epidemiological studies have found significant quantities of inhalable CNTs in the ambient environment, especially in occupational settings, CNT manufacturing factories, and research & development laboratories (Erdely et al., 2013; Liao et al., 2014; Liou et al., 2015). The fibrous nature of CNTs often initiates comparisons to notorious mineral fibers like asbestos, which are known to cause pulmonary fibrosis, mesothelioma and increased risks of lung infection (Aschberger et al., 2010; Lange et al., 2012; Donaldson et al., 2013). However, recent studies have pointed out the varied immune responses between particle and fiber types, such as asbestos and CNTs, which supports the need to better define immune and related toxicological endpoints (Park et al., 2018).
The pulmonary toxicity of CNTs has been the focus of intense research efforts. Current literature supports that CNTs, both single-walled carbon nanotubes (SWCNTs) and MWCNTs, can induce varied biological responses including lung clearance, oxidative stress, inflammation, fibrosis, and granuloma formation (Shvedova et al., 2012; Dong and Ma, 2015). Yet few studies have considered the impacts of exposure to CNTs on respiratory pathogens and in fact, our group was the first to discover that pristine SWCNTs increase H1N1 strain A/Mexico/4108/2009 (IAV) titers in human lung cells in vitro and in a mouse model (Sanpui et al., 2014; Chen et al., 2017; Chen et al., 2019). Pandemics like influenza constitute a major public health issue and can pose considerable economic and social burdens; for example, there are about 3–5 million severe infections annually caused by influenza virus and an estimated 290,000 to 650,000 deaths worldwide (World Health Organization, 2018). Exposure to small particulates, including NMs, can increase susceptibility to respiratory infections, and its understanding can inform the public health community about such disease contraction in the presence of NMs. In addition, it is also important to understand which NMs pose the greatest threat (i.e, SWCNTs or MWCNTs) and what physicochemical properties, such as surface functionalization, are driving their toxicological responses.
Due to inherent physiochemical differences, MWCNTs may lead to toxic effects that vary in type and severity from those of SWCNTs (Dong and Ma, 2015; Oner et al., 2018). Compared to the persistent pulmonary inflammation caused by SWCNTs across a 90-day study, MWCNT exposure markedly increased the levels of pulmonary inflammation immediately that became mitigated over time. In the same study, MWCNT instillation induced greater levels of pleural inflammation than SWCNTs (Fujita et al., 2016). With respect to the effects of CNTs on pathogenic infections, only a handful of studies have revealed that exposure to MWCNTs can lead to reduced bacterial clearance and exacerbated pulmonary inflammation in mice (Shvedova et al., 2005; Shvedova et al., 2008; Walling et al., 2013). One study found that MWCNT exposure aggravated pneumonia and immune cell infiltration in response to respiratory syncytial virus (RSV) in mouse lungs but did not influence the viral titers when compared with RSV treatment only (Hashiguchi et al., 2015). A recent study indicated that exposure to double-walled carbon nanotubes (DWCNTs) can reactivate the molecular features found in acute herpesvirus (murine gamma- herpesvirus 68, MHV-68) infection in mice and human cells (Sattler et al., 2017).
Pristine CNTs are inert and insoluble in aqueous solutions. To improve dispersion in solvents or polymers and to provide reactive sites for further chemical modifications, pristine CNTs can be oxidized to acquire functional groups (Ling et al., 2013; Hoa, 2018). Among various functionalization techniques, the wet chemical oxidation of MWCNTs is recognized as one of the most widely used methods, which typically forms hydroxyl (and other) surface functional groups (Hoa, 2018). Studies have shown that the hydroxyl functionalized MWCNTs confer toxicity that is inherently different than their pristine counterparts (Ursini et al., 2012a; Ursini et al., 2012b). Compared to the pristine MWCNTs, hydroxyl functionalized MWCNTs have been shown to induce less cell death but are more potent in inducing interleukin 8 (IL-8) release (Long et al., 2019), apoptosis, and DNA oxidation (Ursini et al., 2012a; Zhou et al., 2017). While we have shown that sequential exposure to pristine SWCNTs and IAV increases viral titers in mice, no other studies to date have investigated whether other types of CNTs produce a similar response with respect to viral titers, pulmonary injury, and molecular mechanisms. In this study, we investigated how the exposure of MWCNTs that are hydroxyl functionalized can impact immune responses without concomitant increase in viral titers to IAV infection in a well-established mouse model.
2. Materials and methods
2.1. MWCNT preparation and characterization
A batch of dry powder preparation of surface functionalized (~10%) MWCNTs (O.D. 8–15 nm, Cheap Tubes Inc., Grafton, VT) was acid-etched in a mixture of concentrated nitric and sulfuric acid (1:1 v/v) to introduce hydrophilic polar moieties on the MWCNT surfaces (Saleh et al., 2008; Khan et al., 2013a; Khan et al., 2013b; Khan et al., 2013c; Khan et al., 2015; Das et al., 2016; Das et al., 2018; Plazas-Tuttle et al., 2018). After refluxing the mixture at 100 °C for 3 h, functionalized MWCNTs were filtered using a 0.22 μm filter (Durapore hydrophilic PVDF, Merck KGaA, Darmstadt, Germany) until the pH of the filtrate was >5 and dried over 48 h in a desiccator. MWCNTs were weighed in a glass test tube and placed in 1% pluronic F68 in Milli-Q water (w/v) to create a working stock of 500 μg/mL. The pluronic-MWCNT mix was sonicated (Sonifier™ S-450 Digital Ultrasonic Cell Disruptor/Homogenizer, Branson Ultrasonics, Danbury, CT) at 30–50-watt power for 20 min while submerged in an ice bath. Before use, the MWCNT suspension was sonicated at 30 watts for 1 min.
The average hydrodynamic radius (HDR) of MWCNTs in 1% pluronic was measured over 24-h with an ALV/CGS-3 compact goniometer system (ALV-Laser GmbH, Langen/Hessen, Germany), equipped with a 22 mW HeNe 632.8 nm laser. A detailed method has been described elsewhere (Afrooz et al., 2013a; Khan et al., 2013a). To detect metals, MWCNTs were acidified with 5% nitric acid, chilled and filtered before analyzing for total ionic concentrations using a Varian 720 Inductively Coupled Plasma Optical Emission Spectrometer. The electrophoretic mobility (EPM) of the materials was measured with a Malvern Zetasizer (Malvern Instruments Ltd., Worcestershire, UK) at 37 °C following a well-established protocol (Saleh et al., 2008; Afrooz et al., 2013b; Khan et al., 2013b; Khan et al., 2013c; Aich et al., 2016). The physical morphology of the materials was characterized with a JEOL 2010F high-resolution transmission electron microscope (HR-TEM) operating at an acceleration voltage of 200 kV. See supplemental section for details. All samples were measured in triplicate.
2.2. Animals and study design
Male C57BL/6 mice were obtained from the Jackson Laboratory (Bar Harbor, ME) and housed in groups of five per plastic cage. Water and standard mouse diet were provided ad libitum. The environmental conditions were 25 °C, 50% humidity, and 12/12-h light/dark cycle. All mice were acclimated in the housing environment for a week prior to any exposure or treatment. The experimental protocols were approved by the Institutional Animal Care and Use Committee (IACUC #: 201408654) at the University of Florida and all animals were treated humanely according to the NIH Guide for the Care and Use of Laboratory Animals.
A total of 44 12-week old, male C57BL/6 mice were randomly assigned into four groups (N=11); the groups include control, MWCNTs only, IAV (strain: H1N1 A/Mexico/4108/2009. See supplemental for influenza virus preparation) only, and MWCNTs+IAV. On day 0, animals were anesthetized with sterile ketamine (100 mg/kg) and xylazine (5 mg/kg) (Patterson Veterinaries Inc., Charlotte, NC) through intraperitoneal injection. After anesthesia, the mice were positioned on a rodent-tilting workstation and inoculated with 40 μL of either MWCNT working suspension (20 μg per animal) or control vehicle (1% pluronic) at the entrance of the trachea. On day 3, the mice received 50 μL of either IAV (3.2 × 104 TCID50 total virus particles in two nostrils per animal) or PBS through intranasal instillation under anesthesia. On day 7, all animals were euthanized with 0.2 mL fatal-plus (pentobarbital) solution (Patterson Veterinaries Inc., Charlotte, NC). During the experiments, animals were weighed and observed for any clinical signs (e.g., weight loss, labored breathing, or lethargy) on a daily basis.
2.3. Identification and quantification of immune cells in bronchoalveolar lavage fluid (BALF)
After euthanasia, mouse lungs were lavaged with 1 mL ice-cold PBS and the recovered fluid was immediately placed on ice. The volume and cell count of each BALF sample was recorded. The volume of BALF required for 25,000 total cells per sample was calculated and spun onto glass slides using Cytospin (ThermoFisher, Waltham, MA). All slides were stained with HEMA III kit (ThermoFisher, Waltham, MA) and 200 cells on each slide were counted and identified as macrophages, neutrophils, eosinophils, basophils, and lymphocytes using light microscopy. All slides were blinded and counted by two individuals and the average values were calculated.
2.4. Titration of IAV
To quantify the number of virus particles, part of the right lung was weighed and homogenized using disposable tissue grinders (VWR, Radnor, PA) in advanced DMEM media, containing 1% penicillin-streptomycin-neomycin (PSN) antibiotic mixture, 2.5 μg/mL fungizone, and 1% L-Glutamax (ThermoFisher, Waltham, MA). MDCK cells were plated in 96-well plates and allowed to adhere overnight in advanced DMEM media with 1% PSN, 10% fetal bovine serum (FBS), and 1% L-Glutamax. Titration was performed in serum-free advanced DMEM media supplemented with 1% PSN, 1% L-Glutamax, and 1 μg/mL TPCK-trypsin. Serial dilutions (from 10−1 to 10−9 for each log-dilution) of virus stock and lung homogenates were added to cell monolayers and incubated at 33 °C for 5d. Cytopathic effects (CPE) were then observed using light microscopy; the wells that displayed CPE were marked as positive. The TCID50 was calculated as the dilution of virus at which 50% of the MDCK cultures were infected following a previously described protocol (Reed and Muench, 1938).
2.5. Histology and IAV immunohistochemistry
After lavage, the left lung was perfused with fresh 4% paraformaldehyde (PFA) in PBS at pH 7.4, excised, and stored in PFA solution. Fixed lung tissues were embedded in paraffin blocks and cut to approximately 5 μm sections, which were then deparaffinized and stained with hematoxylin and eosin (H&E). Bronchitis, bronchiolitis, and interstitial pneumonia were assessed. In addition to qualitative histopathology assessment, volume density of pneumonia was calculated by digitizing morphometry as described earlier (Li and Castleman, 1991).
2.6. Quantitation of cytokines and chemokines in BALF
The MILLIPLEX mouse cytokine/chemokine magnetic bead panel (Millipore, Burlington, MA) was used to quantify the protein levels of 25 cytokines and chemokines in BALF following the manufacturer’s instructions. Positive controls were provided in the kit and serial dilutions of the positive controls were made to create a standard curve. For each experimental group of BALF specimens, nine samples were measured on a Luminex 200 system (Luminex, Austin, TX) and the data (ng/mL) analyzed using xPONENT software (Luminex, Austin, TX).
2.7. Gene Expression
A portion of the middle quadrant of the right lung was homogenized in STAT-60 RNA Extraction Reagent (Tel Test, Friendswood, TX). Details regarding the methods used for RNA extraction and cDNA synthesis were previously described (Chen et al. 2017). The cDNA product was amplified in a Bio-Rad CFX connect detection system (Hercules, CA) using validated primers and probes (Table S1) specific to each gene target; i.e., influenza matrix protein 2 (M2), toll-like receptor 3 (Tlr3), retinoic acid-induced gene I (Rig-i), melanoma differentiation-associated protein 5 (Mda5), mitochondrial antiviral signaling (Mavs), interferon beta 1 (Ifnβ1), Chemokine (C-C motif) ligand 5 (Ccl5), interferon-induced protein with tetratricopeptide repeats 2 (Ifit2), Ifit3, interleukin 8 (Il-8), apolipoprotein E (Apoe), superoxide dismutase 2 (Sod2), glutathione peroxidase 1 (Gpx1), heme oxygenase 1 (Hmox1), surfactant protein D (Sfpd), NADPH oxidase 4 protein (Nox4), nitric oxide synthase 2 (Nos2), mitofusin 1 (Mfn1), and Mfn2. Expression of Gapdh was utilized as a standard reference gene and all data are presented as normalized fold change expression compared to controls using the ΔΔCt method (Livak and Schmittgen, 2001).
2.8. Statistical analysis
Continuous data are presented as mean ± SEM. If the data were normally distributed, one-way ANOVA was used to compare the average mean of aforementioned parameters with pairwise comparisons using Tukey’s test for equal variances and Dunnett’s test for unequal variances. Data with non-normal distribution was logarithm (Log10) transformed to achieve normal distribution or the analysis was conducted with Kruskal Wallis rank-sum test and Dunn’s pairwise comparison. Statistical differences were identified at P < 0.05.
3. Results
3.1. Characterization of MWCNTs
TEM imaging revealed that MWCNTs were long and spindle-like tubular structures with an average diameter of 22.2 ± 5.3 nm (Fig. 1A). The average HDR of MWCNTs in 1% pluronic was 58.3 ± 0.9 nm (Fig. 1B) and this size remained relatively stable over 24 h in 1% pluronic solution. The zeta potential and electrophoretic mobility measured at −29.9 ± 0.7 mV and −2.9 ± 0.1 × 10−8m2V−1s−1, respectively (Fig. 1C). Trace metal analysis of MWCNTs in 1% pluronic was performed after 24-h incubation in cell media. Besides leached zinc levels which were higher in MWCNT-containing media (64 ppm vs. 7 ppm from 1% pluronic), only a few other metals showed small but insignificant differences compared to control media (Fig. 1D). No other metals appeared in significant levels in the MWCNT suspension compared to 1% pluronic solution alone (Table S2).
Figure 1.
Characterization of MWCNTs in 1% pluronic. (A) Transmission electron micrographs of MWCNTs (Scale: 100 nm (top) and 20 nm (bottom)). (B) Average hydrodynamic radius (HDR) of MWCNTs at 37 °C for 24 hours. (C) Zeta potential and electrophoretic mobility (EPM) of MWCNTs at 37°C for 24 hours (Mean ± SD). (D) Leached metal concentrations (parts per million, ppm) from MWCNTs in 1% pluronic over 24 hours (Mean ± SD).
3.2. Viral titers are not increased by exposure to MWCNTs
At the end of the 7-day experiment, mice infected with IAV alone showed anticipated clinical symptoms including moderate labored breathing and minimal but statistically significant loss in body weight (2%). Mice exposed to MWCNTs alone did not experience any observable clinical signs or weight loss compared to controls, whereas the combined (MWCNTs+IAV) group showed clinical symptoms and weight loss (2%) that was comparable to the IAV only group and was significantly different from the control group (Fig. 2A, B). Viral titers in the IAV only group reached an average of 107 TCID50/g, consistent with the titer levels previously published for this mouse model (Chen et al., 2017). Titers for the MWCNTs+IAV combined group did not significantly differ from IAV alone (Fig. 2C). The expression of IAV M2 was also quantified as a measure of viral replication and levels were equivalent between IAV and MWCNTs+IAV groups (Fig. 2D).
Figure 2.
Changes in body weight, virus titers, and viral-specific gene expression by IAV and MWCNTs+IAV. Mice were exposed to either MWCNTs (20 μg per animal) or 1% pluronic on day 0 and then to IAV (3.2 × 104 TCID50 per animal) on day 3. Animals were euthanized on day 7. (A) Body weight and (B) body weight loss were recorded daily. Part of the right lung lobe of was used for quantification of (C) virus titers and (D) mRNA expression of M2 protein (N=11). Viral titer and M2 expression data were logarithm transformed and all data presented as Mean ± SEM. Asterisks (*) indicate statistically significant differences compared to control and different letters above each group indicate statistically significant differences between groups (P<0.05).
3.3. Immune cell profiles in BALF
Total cell numbers and differential cell counts in BALF samples were quantified. The recovered total cell numbers in the MWCNTs+IAV group exhibited a higher number of cells compared to the other groups, although this observation was not statistically significant (Fig. 3A). In both control and MWCNT exposed mice, macrophages predominated accounting for >90% of the cells while the IAV only exposed animals showed a shift to a neutrophilic profile. MWCNTs alone did not significantly alter the profile of immune cells from those of control mice. Mice from the MWCNTs+IAV co-exposure group showed a profile, consistent with the IAV only group represented by an increase in the percentage of neutrophils (Fig. 3B). The increased percentages of neutrophils in IAV alone and MWCNTs+IAV groups were also reflected by the significantly increased number of neutrophils in the BALF (Fig. 3C). There were also more lymphocytes noted in BALF of IAV only treated mice and in the MWCNTs+IAV group (Fig. 3D). Macrophages were abundantly present in all 4 groups and there was no significant difference between treatments (Fig. 3E).
Figure 3.
Changes in immune cell profiles in mouse BALF following IAV and MWCNTs+IAV exposure. Mice were exposed to either MWCNTs (20 μg per animal) or 1% pluronic on day 0 and then to IAV (3.2 × 104 TCID50 per animal) on day 3. Animals were euthanized on day 7. BALF fluid was collected to quantify (A) total immune cell number, (B) immune cell percentage, (C) total neutrophils, (D) total lymphocytes, and (E) total macrophages (N=11). Data are presented as Mean ± SEM for cell counts. Different letters above each group indicate statistically significant differences (P<0.05).
3.4. Increased pulmonary inflammation and injury by MWCNTs and IAV
Compared with control animals (Fig. 4A), mice exposed to MWCNTs alone showed very mild inflammatory reactions around bronchioles and in proximal acinar areas of alveoli (Fig. 4B). MWCNTs could be identified in the slightly increased number of alveolar macrophages that were widely distributed in the lung (Fig. 4C). The IAV only exposure group showed widespread mild bronchiolitis and multifocal interstitial pneumonia (Fig. 4D). Combined MWCNTs+IAV exposure caused lesions with slightly more severe bronchiolitis and pneumonia compared to IAV only group (Fig. 4E). Increased volume density of pneumonia was observed in the MWCNTs alone group while both IAV and MWCNTs+IAV groups presented with a statistically significant increase of pneumonia comparing with that of control mice (Fig. 4F).
Figure 4.
Histopathological changes in lung tissues by IAV and MWCNTs+IAV. Mice were exposed to either MWCNTs (20 μg per animal) or 1% pluronic on day 0 and then to IAV (3.2 × 104 TCID50 per animal) on day 3. Animals were euthanized on day 7. Left lung lobes were perfused with 4% paraformaldehyde and prepared for histopathological analysis in (A) control (100X), (B) MWCNTs alone (100X), (C) MWCNTs alone (400X), (D) IAV alone (100X), and (E) MWCNTs+IAV (100X). “Red arrow” indicates MWCNTs found in macrophages. Six samples per group were measured for volume density of pneumonia (F) and presented as Mean ± SEM. Different letters above each group indicate statistically significant differences (P<0.05).
3.5. MWCNTs modulate cytokine and chemokine profiles in IAV infected mice
MWCNTs alone did not significantly change any of the cytokines and chemokines measured, although there were some interesting observed decreases in IL-1α, IL-2, IL-5, IL-9, IL-10, IL-12(P40), and IL-13 and increases in G-CSF and KC, although none fo these values were significantly different from controls (Fig. 5,6, and S1). IAV alone significantly induced significant changes for most of the cytokines and chemokines (decreasing IL-1α, IL-2, and IL-10; increasing IL-1β, IL-5, IL-6, TNFα, G-CSF, GM-CSF, IP-10, KC, MCP-1, MIP-1α, MIP-1β, MIP-2, and RANTES). The sequential exposure of MWCNTs and IAV, significantly increased the levels of several immune targets (IL-1β, TNFα, GM-CSF, KC, MIP-1α, MIP-1β, MIP-2, and RANTES) compared with those in the IAV only group (Fig. 5,6, and S1).
Figure 5.
Changes in cytokine levels by IAV and MWCNTs+IAV in BALF. Mice were exposed to either MWCNTs (20 μg per animal) or 1% pluronic on day 0 and then to IAV (3.2 × 104 TCID50 per animal) on day 3. Animals were euthanized on day 7 (N=9). Data are presented as Mean ± SEM. Different letters above each group indicate statistically significant differences (P<0.05).
Figure 6.
Changes in chemokine levels by IAV and MWCNTs+IAV in BALF. Mice were exposed to either MWCNTs (20 μg per animal) or 1% pluronic on day 0 and then to IAV (3.2 × 104 TCID50 per animal) on day 3. Animals were euthanized on day 7 (N=9). Data are presented as Mean ± SEM. Different letters above each group indicate statistically significant differences (P<0.05).
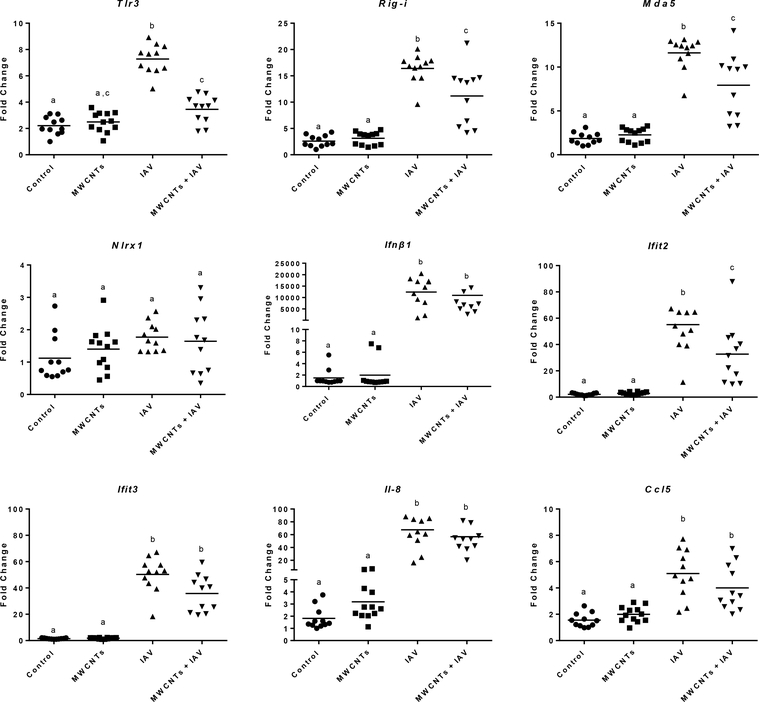
3.6. MWCNTs and IAV induced changes in antiviral and pro-inflammatory gene expression
To further investigate how the innate immune system was affected by MWCNTs and IAV exposure, mRNA expression levels of several antiviral and pro-inflammatory genes were measured. These are commonly induced by respiratory viral infections (Gerlach et al., 2013; Sanpui et al., 2014; Chen et al., 2017), and many of these have been shown to be significantly repressed by SWCNTs in our previous studies (Sanpui et al., 2014; Chen et al., 2017). In the current study, MWCNTs alone did not significantly change the expression of any of these immune genes in mouse lung tissues, while IAV exposure alone significantly induced expression of Tlr3, Rig-i, Mda5, Ifnβ1, Ifit2, Ifit3, Il-8, and Ccl5, which is in line with a typical immune response to IAV challenge. The level of Nlrx1 expression wasn’t significantly different among the four groups. Exposure of MWCNTs followed by IAV infection significantly inhibited mRNA expression of Tlr3, Rig-i, Mda5, and Ifit2 (Fig. 7).
Figure 7.
Changes in antiviral and pro-inflammatory gene expression by IAV and MWCNTs+IAV in lung tissue. Mice were exposed to either MWCNTs (20 μg per animal) or 1% pluronic on day 0 and then to IAV (3.2 × 104 TCID50 per animal) on day 3. Animals were euthanized on day 7. Part of the right lung lobes were extracted for RNA isolation. Ten to eleven samples per group were measured for the mRNA expression levels by qPCR and data presented as Mean ± SEM. Different letters above each group indicate statistically significant differences (P<0.05).
3.7. Oxidative stress-related gene expression changed by MWCNTs and IAV in mice
MWCNTs produced cellular ROS in lung cells in vitro (Fig. S2) but these did not significantly change the expression of any oxidative stress-related genes in mouse lung tissue. IAV alone significantly increased the expression of Sod2, Nos2, Hmox1, Sfpd and Mfn2, while decreasing the expression of Apoe. MWCNTs+IAV showed no statistical difference in expression for any of the genes compared to IAV alone (Fig. 8).
Figure 8.
Changes in stress-related gene expression by IAV and MWCNTs+IAV in lung tissue. Mice were exposed to either MWCNTs (20 μg per animal) or 1% pluronic on day 0 and then to IAV (3.2 × 104 TCID50 per animal) on day 3. Animals were euthanized on day 7. Part of the right lung lobes of mice were extracted for RNA isolation. Ten to eleven samples per group were measured for the mRNA expression levels by qPCR and data presented as Mean ± SEM. Different letters above each group indicate statistically significant differences (P<0.05).
4. Discussion
This study is the first to report that exposure of hydroxyl functionalized MWCNTs can impact immune responses to IAV infection without a concomitant increase in viral titers in a well-established mouse model. The observation of unchanged viral titers by MWCNTs compared to IAV only, is drastically different from our previous report showing that exposure to pristine SWCNTs led to a 63-fold increase of virus titers over IAV only (Sanpui et al., 2014; Chen et al., 2017).
Previous reports indicated that exposure doses of 10 to 31 μg MWCNTs in mice (0.4 – 1.2 mg/kg) approximated human alveolar deposition for a person performing light work in occupational settings for 2 months - 7.5 years based on alveolar epithelium surface area, mass median aerodynamic diameter of MWCNTs, and peak MWCNT-airborne dust levels (Porter et al., 2010; Porter et al., 2013). Based on this estimate, the dose used in the present study (20 ug/mouse or 0.8 mg/kg) falls within this range, thus representing an estimated occupational exposure, a value also consistently used (or less than) other rodent studies (Erdely et al., 2013). MWCNT exposure alone did not cause any significant pulmonary injury nor did it produce severe inflammatory responses, which was evidenced from the lack of notable changes in lung tissue architecture and no significant increases in immune responses (immune cell infiltration, cytokine and chemokine levels, and expression of pro-inflammatory genes) in both BALF and lung tissues. These results are consistent with other studies, where mice exposed to MWCNTs for 7 days through pharyngeal aspiration showed a mild increase in pulmonary tissue damage, with more robust pulmonary injury at time-points beyond 7 days such as 28 (Girtsman et al., 2014) and 56 (Mercer et al., 2011) days. Another study reported worsened inflammation, increased collagen deposition and granuloma formation 30 days after exposure to MWCNTs in a mouse model (Wang et al., 2011). It is possible that more severe lung tissue damage would have been observed with longer exposure time to MWCNTs beyond the 7-day time-point implemented here. It is also important to note that different types of MWCNTs were employed in these studies (high-nickel MWCNTs (Girtsman et al., 2014) vs. mitusi MWCNTs (Mercer et al., 2011) vs. hydroxyl functionalized MWCNTs in our experiment) which likely play an important role in the varied pulmonary toxicity responses observed.
In contrast, a few studies have documented significantly altered cytokine and chemokine levels in BALF after MWCNT exposure as early as 24-h post exposure in mice (e.g. TNFα, TGFβ, IL-6, IL-10 and MCP-1) (Grecco et al., 2011; Girtsman et al., 2014) and after chronic and constant exposure of rats to MWCNTs for 24 weeks (IP-10, RANTES, IL-2, IL-18, MIP-1α. MIP-2, MCP-1, KC, IP-10, RANTES, among others) (Xu et al., 2014). Besides variations in exposure times, different exposure methods between studies have also been shown to impact immune-related outcomes. For example, more severe inflammatory was exhibited in mouse lungs after inhalation of aerosolized MWCNTs 4-d post-exposure, which progressed into more severe tissue damage, compared to exposure via intratracheal aspiration (Ma-Hock et al., 2013; Porter et al., 2013).
As expected, IAV challenge induced significant changes in BALF immune cell profiles, modulation of pro-inflammatory cytokine and chemokine levels, and mRNA expression of antiviral genes, that have also been observed in other studies where mice were exposed to IAV (Bauer et al., 2010; Harata et al., 2011; Otte et al., 2011; Chen et al., 2017). The significant changes in cytokines and chemokines represent proteins that are responsible for mediating innate and adaptive immune responses (e.g. IL-1α, IL-1β, IL-6), recruiting immune cells (e.g. G-CSF, IP-10, KC, MIP-1, RANTES), and initiating antiviral immune responses (e.g. IFNβ1 and IFNγ) (Kline et al., 2011; Knipe and Howley, 2013). The decreased levels of a few cytokines (IL-1α, IL-2, and IL-10) in BALF are consistent with our previous study (Chen et al., 2017) but are inconsistent with some other reports; for example, IL-2 and IL-10 levels were not changed, while the levels of IL-1α have been reported to both increase and decrease in IAV exposed mouse lungs, depending on study conditions (Otte et al., 2011; Kim et al., 2012; Han et al., 2014). These discrepancies are likely a result of different strains of viruses, infection time, and host physiology.
Multiple markers of the signaling pathways related to innate immunity, and previously shown to be impacted by pristine SWCNTs (Chen et al. 2017; Chen et al. 2019), were also investigated in this study. Tlr3, Rig-i, and Mda5 are responsible for detecting viral RNA and subsequently initiate downstream signaling cascades (Kawai and Akira, 2009) that include Mavs, Nlrx1 (Yoneyama et al., 2016), Ifnβ1, Ifit2, Ifit3, Il-8, and Ccl5 that are essential for commencing antiviral and pro-inflammatory immune responses (Kawai and Akira, 2009; Chen et al., 2017). No significant induction of Mavs and Nlrx1 expression was observed after IAV exposure, likely because these are constitutively expressed proteins. However, other more inducible genes including Ifnβ1, Ifit2, Ifit3, and Ccl5, were significantly increased at the mRNA level in response to IAV infection. Interestingly, the IAV-induced expression of several of these antiviral genes (Tlr3, Rig-i, Mda5, and Ifit2) was significantly reduced by the presence of MWCNTs. This was surprising as repression of anti-viral defenses should logically lead to enhanced titers – an observation we noted for SWCNTs (Chen et al., 2017). We previously reported that the expression of several of these genes was also affected by the cellular production of ROS (Chen et al. 2019), which we now show to be produced by both SWCNTs and MWCNTs in human lung epithelial cells. It is possible that some of the similar biological responses triggered by MWCNT and SWCNT exposures are driven by ROS production, while the differential effects are independent of ROS and may be due to varied physicochemical properties of the CNTs. These observations suggest that the specific types of ROS that are induced by CNT exposures should be further investigated to better define the role in CNT-induced injury related to viral infections.
In addition to differences in anti-viral gene expression, a comparison of the effects of exposure to SWCNTs in a mouse model of IAV infection published earlier (Chen et al. 2017) and the effects of MWCNTs determined in this study are summarized in Table 1. While the exposures to MWCNTS and SWCNT were performed in separate studies and therefore are not exactly comparable, it is important to note that the animal model (strain, age, sex), experimental design (dose, time), and viral response (titers) were very consistent. In general, MWCNTs+IAV exposure caused only mild lung tissue injury, did not cause an increase in viral titers or immune cells in BALF compared to IAV alone. This is different from the response observed for SWCNTs where we noted more severe lung tissue injury and pneumonia and increased viral titers and immune cell increases in the BALF, with a noted increase in lymphocytes by SWCNTs alone that was also reflected in the SWCNTs+IAV group. Interestingly, increased levels of many cytokines and chemokines in BALF by MWCNTs were similar to what we observed for SWCNTs (TNFa, MIP-1α, MIP-1β and RANTES), although a number of differences were also noted (IL-1β, IL-12 (p70), GM-CSF, IP-10, KC, MCP-1, and MIP-2) (Fig. S3).
Table 1.
Comparison of the effects of MWCNT and SWCNT exposure on IAV infection. SWCNT data was previously published (Chen et al., 2017).
| Compared to IAV only | ||
|---|---|---|
| Measurement | Eff ects of MWCNTs | Effects of SWCNTs |
| Tissue injury | Mild | Moderate; pneumonia increased |
| Viral titer | No change | Increased |
| Cell differentials in BALF | No change | Increased Lymphocytes, Monocytes, Neutrophils |
| Cytokines increased | IL-1β, GM-CSF*, TNFα, KC, MIP-1α, MIP-1β, MIP-2*, RANTES | IL-4, IL-12(P70)*, TNFα, IP-10*, MCP-1, MIP-1α, MIP-1β, RANTES |
| Inflammatory and antiviral genes increased | Tlr3, Rig-i, Mda5, Ifit2, | No change |
The differences in these immune profiles might partially explain the varied titers and pathology observed. The significant increase of IL-1β, GM-CSF, KC, and MIP-2 by MWCNTs+IAV exposure indicates that there might be a higher degree of involvement of the NLRP3 inflammasome pathway and viral clearance by macrophages and neutrophils based on their known functions. Significantly higher levels of IL-12(p70), IP-10, and MCP-1 by SWCNTS+IAV, but not MWCNTs+IAV, might be responsible for inducing differentiation of T cells into Th1 cells (Hsieh et al., 1993) and recruiting macrophages, T cells, and NK cells (Xu et al., 1996; Romero et al., 2006). This may lead to differential clearance between the SWCNTs and MWCNTs. In fact, the histopathology analysis supports that MWCNTs can be phagocytosed by macrophages, an observation not observed for SWCNTs. In another study, we showed that SWCNTs remain in mouse lung tissues 21 days post exposure using a custom near-infrared fluorescence (NIRF) tracking method which led to early granuloma-like injury (Nicholas et al., 2019). Interestingly, one study showed that MWCNTs were cleared from the airway 7 days post pharyngeal aspiration (Mercer et al., 2011). While we identified MWCNTs in macrophages that were well distributed in the lungs on day 7 in our study, it is possible that MWCNTs may be more easily cleared from the lungs than SWCNTs, which is an excellent area for future research.
Inherent differences in physicochemical properties of the CNTs, such as agglomerate size (average 151 nm for SWCNTs vs. 58.3 nm for MWCNTs) and surface chemistry (pristine SWCNTs vs. hydroxyl-functionalized MWCNTs) probably also influence their ability to cause physical damage, become phagocytosed and cleared by the immune system, and activate/repress immune defense and repair pathways (Palomaki et al., 2011; Dong and Ma, 2015; Oner et al., 2018). These responses directly impact the host’s ability to mount an immune response and consequently impact viral life cycles. Other studies have noted varied infection outcomes from combined exposures to carbon-based NMs. For example, carbon nanodots functionalized with boronic acid or amine functional groups were shown to interfere with herpes virus entry and lead to inhibited infection in in vitro models (Barras et al., 2016). SWCNTs have been shown to bind and inhibit the innate immune agonist protein (VP35) of Ebola virus using molecular dynamics simulation methods (Zhang et al., 2017), attenuate Caco-2 cells from damage induced by E. Coli or S. Aureus by inhibiting inflammasome activation and IL-1β production (Chen et al., 2015), and increase disease severity with reduced bacterial clearance to Listeria monocytogenes in mice (Shvedova et al., 2005; Shvedova et al., 2008). Similar to our findings, Hashiguchi and colleagues found that intranasal exposure of mice to MWCNTs did not affect the titer of respiratory syncytial virus (RSV) but did increase the severity of pneumonia (Hashiguchi et al., 2015). We quantified the mRNA expression level of influenza matrix protein 2 (M2), a highly conserved and tetrameric type III integral membrane protein (Gannage et al., 2009; World Health Organization, 2011; Ma et al., 2012; Knipe and Howley, 2013), as a measure of viral replication. In both SWCNT (Fig. S4) and MWCNT exposures, M2 gene expression was highly induced by IAV alone as expected. Interestingly, M2 expression was not significantly changed by the pre-treatment of either type of CNTs. These data suggest that there may be greater impacts of CNTs on IAV entry or egress, rather than replication (Chen et al., 2020).
5. Conclusions
In this study, we found that exposure of mice to hydroxyl functionalized MWCNTs can modulate immune responses (antiviral and inflammatory markers) to IAV, but with no effect on the viral titer outcomes and modest pulmonary injury, a result different from a previous study which showed pristine SWCNT exposures significantly increased viral titers in concert with severe lung tissue injury in the same mouse model. Variations in cytokine and chemokine responses that control virus clearance and other aspects of host defenses may explain that MWCNTs did not exacerbate IAV viral titers in mice. Further research should focus on the mechanisms and the contribution of physicochemical properties of CNTs which lead to varied outcomes of respiratory virus infections.
Supplementary Material
Highlights.
Multi-walled carbon nanotubes (MWCNTs) cause mild lung inflammation and injury
MWCNTs do not significantly enhance influenza A virus (IAV) titers in mouse lungs
MWCNTs alter levels of cytokines and gene expression related to anti-viral signaling
Acknowledgements
This work was supported by the National Institute of Health (R01HL114907 to TSA) and the University of Florida Research Foundation (to TSA) for funding.
Footnotes
Declaration of Competing Interest
The authors declare there are no conflicts of interest.
Declaration of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could a?ect the content, and all legal disclaimers that apply to the journal pertain.
References
- Afrooz AR, Khan IA, Hussain SM, Saleh NB, 2013a. Mechanistic heteroaggregation of gold nanoparticles in a wide range of solution chemistry. Environ Sci Technol 47, 1853–1860. [DOI] [PubMed] [Google Scholar]
- Afrooz ARMN, Khan IA, Hussain SM, Saleh NB, 2013b. Mechanistic Heteroaggregation of Gold Nanoparticles in a Wide Range of Solution Chemistry. Environ Sci Technol 47, 1853–1860. [DOI] [PubMed] [Google Scholar]
- Aich N, Boateng LK, Sabaraya IV, Das D, Flora JR, Saleh NB, 2016. Aggregation Kinetics of Higher-Order Fullerene Clusters in Aquatic Systems. Environ Sci Technol 50, 3562–3571. [DOI] [PubMed] [Google Scholar]
- Aschberger K, Johnston HJ, Stone V, Aitken RJ, Hankin SM, Peters SA, Tran CL, Christensen FM, 2010. Review of carbon nanotubes toxicity and exposure--appraisal of human health risk assessment based on open literature. Critical reviews in toxicology 40, 759–790. [DOI] [PubMed] [Google Scholar]
- Barras A, Pagneux Q, Sane F, Wang Q, Boukherroub R, Hober D, Szunerits S, 2016. High Efficiency of Functional Carbon Nanodots as Entry Inhibitors of Herpes Simplex Virus Type 1. ACS applied materials & interfaces 8, 9004–9013. [DOI] [PubMed] [Google Scholar]
- Bauer CM, Zavitz CC, Botelho FM, Lambert KN, Brown EG, Mossman KL, Taylor JD, Stampfli MR, 2010. Treating viral exacerbations of chronic obstructive pulmonary disease: insights from a mouse model of cigarette smoke and H1N1 influenza infection. PloS one 5, e13251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen H, Humes ST, Robinson SE, Loeb JC, Sabaraya IV, Saleh NB, Khattri RB, Merritt ME, Martyniuk CJ, Lednicky JA, Sabo-Attwood T, 2019. Single-walled carbon nanotubes repress viral-induced defense pathways through oxidative stress. Nanotoxicology 13, 1176–1196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen H, Humes ST, Saleh NB, Lednicky JA, Sabo-Attwood T, 2020. Nanomaterial Effects on Viral Infection. In Bonner JC, Brown JM, (Eds.), Interaction of Nanomaterials with the Immune System. Springer International Publishing, Cham, pp. 167–195. [Google Scholar]
- Chen H, Wang B, Zhao R, Gao D, Guan M, Zheng L, Zhou X, Chai Z, Zhao Y, Feng W, 2015. Coculture with Low-Dose SWCNT Attenuates Bacterial Invasion and Inflammation in Human Enterocyte-like Caco-2 Cells. Small 11, 4366–4378. [DOI] [PubMed] [Google Scholar]
- Chen H, Zheng X, Nicholas J, Humes ST, Loeb JC, Robinson SE, Bisesi JH Jr., Das D, Saleh NB, Castleman WL, Lednicky JA, Sabo-Attwood T, 2017. Single-walled carbon nanotubes modulate pulmonary immune responses and increase pandemic influenza a virus titers in mice. Virology journal 14, 242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das D, Plazas-Tuttle J, Sabaraya I, Jain S, Sabo-Attwood T, Saleh N, 2016. An Elegant Method for Large Scale Synthesis of Metal Oxide-Carbon Nanotube Nanohybrids for Nano-environmental Application and Implication Studies. Environmental science. Nano. 4. [Google Scholar]
- Das D, Sabaraya IV, Zhu T, Sabo-Attwood T, Saleh NB, 2018. Aggregation Behavior of Multiwalled Carbon Nanotube-Titanium Dioxide Nanohybrids: Probing the Part-Whole Question. Environ Sci Technol 52, 8233–8241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Volder MF, Tawfick SH, Baughman RH, Hart AJ, 2013. Carbon nanotubes: present and future commercial applications. Science 339, 535–539. [DOI] [PubMed] [Google Scholar]
- Donaldson K, Poland CA, Murphy FA, MacFarlane M, Chernova T, Schinwald A, 2013. Pulmonary toxicity of carbon nanotubes and asbestos - similarities and differences. Advanced drug delivery reviews 65, 2078–2086. [DOI] [PubMed] [Google Scholar]
- Dong J, Ma Q, 2015. Advances in mechanisms and signaling pathways of carbon nanotube toxicity. Nanotoxicology 9, 658–676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erdely A, Dahm M, Chen BT, Zeidler-Erdely PC, Fernback JE, Birch ME, Evans DE, Kashon ML, Deddens JA, Hulderman T, Bilgesu SA, Battelli L, Schwegler-Berry D, Leonard HD, McKinney W, Frazer DG, Antonini JM, Porter DW, Castranova V, Schubauer-Berigan MK, 2013. Carbon nanotube dosimetry: from workplace exposure assessment to inhalation toxicology. Particle and fibre toxicology 10, 53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujita K, Fukuda M, Endoh S, Maru J, Kato H, Nakamura A, Shinohara N, Uchino K, Honda K, 2015. Size effects of single-walled carbon nanotubes on in vivo and in vitro pulmonary toxicity. Inhalation toxicology 27, 207–223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujita K, Fukuda M, Endoh S, Maru J, Kato H, Nakamura A, Shinohara N, Uchino K, Honda K, 2016. Pulmonary and pleural inflammation after intratracheal instillation of short single-walled and multi-walled carbon nanotubes. Toxicol Lett 257, 23–37. [DOI] [PubMed] [Google Scholar]
- Gannage M, Dormann D, Albrecht R, Dengjel J, Torossi T, Ramer PC, Lee M, Strowig T, Arrey F, Conenello G, Pypaert M, Andersen J, Garcia-Sastre A, Munz C, 2009. Matrix protein 2 of influenza A virus blocks autophagosome fusion with lysosomes. Cell host & microbe 6, 367–380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerlach RL, Camp JV, Chu YK, Jonsson CB, 2013. Early Host Responses of Seasonal and Pandemic Influenza A Viruses in Primary Well-Differentiated Human Lung Epithelial Cells. PloS one 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Girtsman TA, Beamer CA, Wu N, Buford M, Holian A, 2014. IL-1R signalling is critical for regulation of multi-walled carbon nanotubes-induced acute lung inflammation in C57Bl/6 mice. Nanotoxicology 8, 17–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grand View Research, 2015. Carbon Nanotubes Market Size, Share & Trends Analysis Report By Product (SWNT, MWNT), By Application (Polymers, Energy, Electrical & Electronics), And Segment Forecasts, 2015 – 2022. Grand View Research, pp. 157. hhttps://www.grandviewresearch.com/industry-analysis/carbon-nanotubes-cnt-market
- Grecco AC, Paula RF, Mizutani E, Sartorelli JC, Milani AM, Longhini AL, Oliveira EC, Pradella F, Silva VD, Moraes AS, Peterlevitz AC, Farias AS, Ceragioli HJ, Santos LM, Baranauskas V, 2011. Up-regulation of T lymphocyte and antibody production by inflammatory cytokines released by macrophage exposure to multi-walled carbon nanotubes. Nanotechnology 22, 265103. [DOI] [PubMed] [Google Scholar]
- Guzman KA, Taylor MR, Banfield JF, 2006. Environmental risks of nanotechnology: National Nanotechnology Initiative funding, 2000–2004. Environ Sci Technol 40, 1401–1407. [DOI] [PubMed] [Google Scholar]
- Han Y, Ling MT, Mao H, Zheng J, Liu M, Lam KT, Liu Y, Tu W, Lau Y-L, 2014. Influenza virus-induced lung inflammation was modulated by cigarette smoke exposure in mice. PloS one 9, e86166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harata G, He F, Hiruta N, Kawase M, Kubota A, Hiramatsu M, Yausi H, 2011. Intranasally administered Lactobacillusgasseri TMC0356 protects mice from H1N1 influenza virus infection by stimulating respiratory immune responses. World Journal of Microbiology and Biotechnology 27, 411–416. [Google Scholar]
- Hashiguchi S, Yoshida H, Akashi T, Hirose A, Sugita C, Kurokawa M, Watanabe W, 2015. Effects of multi-walled carbon nanotubes on the pneumonia in respiratory syncytial virus-infected mice. Toxicology Letters 238, S201. [Google Scholar]
- Hoa LTM, 2018. Characterization of multi-walled carbon nanotubes functionalized by a mixture of HNO3/H2SO4. Diam Relat Mater 89, 43–51. [Google Scholar]
- Hsieh C, Macatonia S, Tripp C, Wolf S, O'Garra A, Murphy K, 1993. Development of TH1 CD4+ T cells through IL-12 produced by Listeria-induced macrophages. Science 260, 547–549. [DOI] [PubMed] [Google Scholar]
- Kawai T, Akira S, 2009. The roles of TLRs, RLRs and NLRs in pathogen recognition. International immunology 21, 317–337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan IA, Afrooz AR, Flora JR, Schierz PA, Ferguson PL, Sabo-Attwood T, Saleh NB, 2013a. Chirality affects aggregation kinetics of single-walled carbon nanotubes. Environ Sci Technol 47, 1844–1852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan IA, Aich N, Afrooz ARMN, Flora JRV, Schierz PA, Ferguson PL, Sabo-Attwood T, Saleh NB, 2013b. Fractal structures of single-walled carbon nanotubes in biologically relevant conditions: Role of chirality vs. media conditions. Chemosphere 93, 1997–2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan IA, Berge ND, Sabo-Attwood T, Ferguson PL, Saleh NB, 2013c. Single-walled carbon nanotube transport in representative municipal solid waste landfill conditions. Environ Sci Technol 47, 8425–8433. [DOI] [PubMed] [Google Scholar]
- Khan IA, Flora JR, Nabiul Afrooz AR, Aich N, Schierz PA, Ferguson PL, Sabo-Attwood T, Saleh NB, 2015. Change in Chirality of Semiconducting Single-Walled Carbon Nanotubes Can Overcome Anionic Surfactant Stabilization: A Systematic Study of Aggregation Kinetics. Environ Chem 12, 652–661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim HM, Kang YM, Song BM, Kim HS, Seo SH, 2012. The 2009 pandemic H1N1 influenza virus is more pathogenic in pregnant mice than seasonal H1N1 influenza virus. Viral immunology 25, 402–410. [DOI] [PubMed] [Google Scholar]
- Kline KA, Schwartz DJ, Lewis WG, Hultgren SJ, Lewis AL, 2011. Immune activation and suppression by group B streptococcus in a murine model of urinary tract infection. Infection and immunity 79, 3588–3595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knipe DM, Howley PM, 2013. Fields virology. Wolters Kluwer/Lippincott Williams & Wilkins Health, Philadelphia, PA. [Google Scholar]
- Lange JH, Mastrangelo G, Cegolon L, 2012. Infectious disease risk in asbestos abatement workers. BMC public health 12, 665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X, Castleman WL, 1991. Effects of 4-ipomeanol on bovine parainfluenza type 3 virus-induced pneumonia in calves. Veterinary pathology 28, 428–437. [DOI] [PubMed] [Google Scholar]
- Liao HY, Chung YT, Lai CH, Wang SL, Chiang HC, Li LA, Tsou TC, Li WF, Lee HL, Wu WT, Lin MH, Hsu JH, Ho JJ, Chen CJ, Shih TS, Lin CC, Liou SH, 2014. Six-month follow-up study of health markers of nanomaterials among workers handling engineered nanomaterials. Nanotoxicology 8 Suppl 1, 100–110. [DOI] [PubMed] [Google Scholar]
- Ling XL, Wei YZ, Zou LM, Xu S, 2013. Preparation and characterization of hydroxylated multi-walled carbon nanotubes. Colloid Surface A 421, 9–15. [Google Scholar]
- Liou SH, Tsai CS, Pelclova D, Schubauer-Berigan MK, Schulte PA, 2015. Assessing the first wave of epidemiological studies of nanomaterial workers. Journal of nanoparticle research: an interdisciplinary forum for nanoscale science and technology 17, 413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livak KJ, Schmittgen TD, 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408. [DOI] [PubMed] [Google Scholar]
- Long J, Ma W, Yu Z, Liu H, Cao Y, 2019. Multi-walled carbon nanotubes (MWCNTs) promoted lipid accumulation in THP-1 macrophages through modulation of endoplasmic reticulum (ER) stress. Nanotoxicology 13, 938–951. [DOI] [PubMed] [Google Scholar]
- Ma-Hock L, Strauss V, Treumann S, Kuttler K, Wohlleben W, Hofmann T, Groters S, Wiench K, van Ravenzwaay B, Landsiedel R, 2013. Comparative inhalation toxicity of multi-wall carbon nanotubes, graphene, graphite nanoplatelets and low surface carbon black. Particle and fibre toxicology 10, 23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma H, Kien F, Maniere M, Zhang Y, Lagarde N, Tse KS, Poon LL, Nal B, 2012. Human annexin A6 interacts with influenza a virus protein M2 and negatively modulates infection. Journal of virology 86, 1789–1801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mercer RR, Hubbs AF, Scabilloni JF, Wang L, Battelli LA, Friend S, Castranova V, Porter DW, 2011. Pulmonary fibrotic response to aspiration of multi-walled carbon nanotubes. Particle and fibre toxicology 8, 21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicholas J, Chen H, Liu KR, Venu I, Bolser D, Saleh NB, Bisesi JH, Castleman W, Ferguson PL, Sabo-Attwood T, 2019. Utilization of near infrared fluorescence imaging to track and quantify the pulmonary retention of single-walled carbon nanotubes in mice. Nanoimpact 14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oner D, Ghosh M, Bove H, Moisse M, Boeckx B, Duca RC, Poels K, Luyts K, Putzeys E, Van Landuydt K, Vanoirbeek JA, Ameloot M, Lambrechts D, Godderis L, Hoet PH, 2018. Differences in MWCNT- and SWCNT-induced DNA methylation alterations in association with the nuclear deposition. Particle and fibre toxicology 15, 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otte A, Sauter M, Alleva L, Baumgarte S, Klingel K, Gabriel G, 2011. Differential host determinants contribute to the pathogenesis of 2009 pandemic H1N1 and human H5N1 influenza A viruses in experimental mouse models. The American journal of pathology 179, 230–239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palomaki J, Valimaki E, Sund J, Vippola M, Clausen PA, Jensen KA, Savolainen K, Matikainen S, Alenius H, 2011. Long, needle-like carbon nanotubes and asbestos activate the NLRP3 inflammasome through a similar mechanism. ACS nano 5, 6861–6870. [DOI] [PubMed] [Google Scholar]
- Park EJ, Khaliullin TO, Shurin MR, Kisin ER, Yanamala N, Fadeel B, Chang J, Shvedova AA, 2018. Fibrous nanocellulose, crystalline nanocellulose, carbon nanotubes, and crocidolite asbestos elicit disparate immune responses upon pharyngeal aspiration in mice. Journal of immunotoxicology 15, 12–23. [DOI] [PubMed] [Google Scholar]
- Plazas-Tuttle J, Das D, Sabaraya I, Saleh N, 2018. Harnessing the power of microwaves for inactivating Pseudomonas aeruginosa with nanohybrids. Environmental Science: Nano 5, 72–82. [Google Scholar]
- Porter DW, Hubbs AF, Chen BT, McKinney W, Mercer RR, Wolfarth MG, Battelli L, Wu N, Sriram K, Leonard S, Andrew M, Willard P, Tsuruoka S, Endo M, Tsukada T, Munekane F, Frazer DG, Castranova V, 2013. Acute pulmonary dose-responses to inhaled multi-walled carbon nanotubes. Nanotoxicology 7, 1179–1194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porter DW, Hubbs AF, Mercer RR, Wu N, Wolfarth MG, Sriram K, Leonard S, Battelli L, Schwegler-Berry D, Friend S, Andrew M, Chen BT, Tsuruoka S, Endo M, Castranova V, 2010. Mouse pulmonary dose- and time course-responses induced by exposure to multi-walled carbon nanotubes. Toxicology 269, 136–147. [DOI] [PubMed] [Google Scholar]
- Reed LJ, Muench H, 1938. A SIMPLE METHOD OF ESTIMATING FIFTY PER CENT ENDPOINTS12. American Journal of Epidemiology 27, 493–497. [Google Scholar]
- Romero AI, Lagging M, Westin J, Dhillon AP, Dustin LB, Pawlotsky JM, Neumann AU, Ferrari C, Missale G, Haagmans BL, Schalm SW, Zeuzem S, Negro F, Verheij-Hart E, Hellstrand K, 2006. Interferon (IFN)-gamma-inducible protein-10: association with histological results, viral kinetics, and outcome during treatment with pegylated IFN-alpha 2a and ribavirin for chronic hepatitis C virus infection. The Journal of infectious diseases 194, 895–903. [DOI] [PubMed] [Google Scholar]
- Saleh NB, Pfefferle LD, Elimelech M, 2008. Aggregation kinetics of multiwalled carbon nanotubes in aquatic systems: measurements and environmental implications. Environ Sci Technol 42, 7963–7969. [DOI] [PubMed] [Google Scholar]
- Sanpui P, Zheng X, Loeb JC, Bisesi JH Jr., Khan IA, Afrooz AR, Liu K, Badireddy AR, Wiesner MR, Ferguson PL, Saleh NB, Lednicky JA, Sabo-Attwood T, 2014. Single-walled carbon nanotubes increase pandemic influenza A H1N1 virus infectivity of lung epithelial cells. Particle and fibre toxicology 11, 66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sattler C, Moritz F, Chen S, Steer B, Kutschke D, Irmler M, Beckers J, Eickelberg O, Schmitt-Kopplin P, Adler H, Stoeger T, 2017. Nanoparticle exposure reactivates latent herpesvirus and restores a signature of acute infection. Particle and fibre toxicology 14, 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shvedova AA, Fabisiak JP, Kisin ER, Murray AR, Roberts JR, Tyurina YY, Antonini JM, Feng WH, Kommineni C, Reynolds J, Barchowsky A, Castranova V, Kagan VE, 2008. Sequential exposure to carbon nanotubes and bacteria enhances pulmonary inflammation and infectivity. American journal of respiratory cell and molecular biology 38, 579–590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shvedova AA, Kisin ER, Mercer R, Murray AR, Johnson VJ, Potapovich AI, Tyurina YY, Gorelik O, Arepalli S, Schwegler-Berry D, Hubbs AF, Antonini J, Evans DE, Ku BK, Ramsey D, Maynard A, Kagan VE, Castranova V, Baron P, 2005. Unusual inflammatory and fibrogenic pulmonary responses to single-walled carbon nanotubes in mice. American journal of physiology. Lung cellular and molecular physiology 289, L698–708. [DOI] [PubMed] [Google Scholar]
- Shvedova AA, Pietroiusti A, Fadeel B, Kagan VE, 2012. Mechanisms of carbon nanotube-induced toxicity: focus on oxidative stress. Toxicology and applied pharmacology 261, 121–133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ursini CL, Cavallo D, Fresegna AM, Ciervo A, Maiello R, Buresti G, Casciardi S, Tombolini F, Bellucci S, Iavicoli S, 2012a. Comparative cyto-genotoxicity assessment of functionalized and pristine multiwalled carbon nanotubes on human lung epithelial cells. Toxicology in vitro: an international journal published in association with BIBRA 26, 831–840. [DOI] [PubMed] [Google Scholar]
- Ursini CL, Cavallo D, Fresegna AM, Ciervo A, Maiello R, Casciardi S, Tombolini F, Buresti G, Iavicoli S, 2012b. Study of Cytotoxic and Genotoxic Effects of Hydroxyl-Functionalized Multiwalled Carbon Nanotubes on Human Pulmonary Cells. J Nanomater. [Google Scholar]
- Walling BE, Kuang Z, Hao Y, Estrada D, Wood JD, Lian F, Miller LA, Shah AB, Jeffries JL, Haasch RT, Lyding JW, Pop E, Lau GW, 2013. Helical carbon nanotubes enhance the early immune response and inhibit macrophage-mediated phagocytosis of Pseudomonas aeruginosa. PloS one 8, e80283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Katwa P, Podila R, Chen P, Ke PC, Rao AM, Walters DM, Wingard CJ, Brown JM, 2011. Multi-walled carbon nanotube instillation impairs pulmonary function in C57BL/6 mice. Particle and fibre toxicology 8, 24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Health Organization, 2011. Manual for the laboratory diagnosis and virological surveillance of influenza. World Health Organization, Geneva. [Google Scholar]
- World Health Organization, 2018. Influenza (Seasonal). World Health Organization, pp. Fact sheets of seasonal influenza. In. http://www.who.int/news-room/fact-sheets/detail/influenza-(seasonal) Accessed May 10th 2018
- Xu J, Alexander DB, Futakuchi M, Numano T, Fukamachi K, Suzui M, Omori T, Kanno J, Hirose A, Tsuda H, 2014. Size- and shape-dependent pleural translocation, deposition, fibrogenesis, and mesothelial proliferation by multiwalled carbon nanotubes. Cancer science 105, 763–769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu LL, Warren MK, Rose WL, Gong W, Wang JM, 1996. Human recombinant monocyte chemotactic protein and other C-C chemokines bind and induce directional migration of dendritic cells in vitro. Journal of leukocyte biology 60, 365–371. [DOI] [PubMed] [Google Scholar]
- Yoneyama M, Jogi M, Onomoto K, 2016. Regulation of antiviral innate immune signaling by stress-induced RNA granules. Journal of biochemistry 159, 279–286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang YJ, Ding JN, Zhong H, Sun CP, Han JG, 2017. Molecular dynamics exploration of the binding mechanism and properties of single-walled carbon nanotube to WT and mutant VP35 FBP region of Ebola virus. Journal of biological physics 43, 149–165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou LL, Forman HJ, Ge Y, Lunec J, 2017. Multi-walled carbon nanotubes: A cytotoxicity study in relation to functionalization, dose and dispersion. Toxicology in Vitro 42, 292–298. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.