Abstract
Background
Prescription of testosterone replacement therapy (TRT) has increased in the United States in recent years, and though anabolic steroids have been associated with tendon rupture, there is a paucity of literature evaluating the risk of Achilles tendon injury with TRT. This study aims to evaluate the associative relationship between consistent TRT, Achilles tendon injury, and subsequent surgery.
Methods
This is a one-to-one matched retrospective cohort study utilizing the PearlDiver database. Records were queried for patients aged 35–75 who were prescribed at least 3 consecutive months of TRT between January 1, 2010 and December 31, 2019. Achilles tendon injuries and subsequent surgeries were identified using ICD-9, ICD-10, and CPT billing codes. Multivariable logistic regression was used to compare odds of Achilles tendon injury, Achilles tendon surgery, and revision surgery, with a p-value < 0.05 representing statistical significance.
Results
A sample of 423,278 patients who filled a TRT prescription for a minimum of 3 consecutive months was analyzed. The 2-year incidence of Achilles tendon injury was 377.8 (95% CI, 364.8–391.0) per 100,000 person-years in the TRT cohort, compared to 245.8 (95% CI, 235.4–256.6) in the control (p < 0.001). The adjusted analysis demonstrated TRT to be associated with a significantly increased likelihood of being diagnosed with Achilles tendon injury (aOR = 1.24, 95% CI, 1.15–1.33, p < 0.001). Of those diagnosed with Achilles tendon injury, 287/3,198 (9.0%) of the TRT cohort subsequently underwent surgery for their injury, compared to 134/2,081 (6.4%) in the control cohort (aOR = 1.54, 95% CI, 1.19–1.99, p < 0.001).
Conclusions
There is a significant association between Achilles tendon injury and prescription TRT, with a concomitantly increased rate of undergoing surgical management. These results provide insight into the risk profile of TRT and further research into the science of tendon pathology in the setting of TRT is an area of continued interest.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13047-023-00678-0.
Keywords: Achilles, Testosterone, Tendon Injury, Tendinopathy, Surgery
Background
Achilles tendon injuries are common musculoskeletal pathologies that are characterized by pain and swelling about the heel and decreased athletic performance and mobility [1]. While prevalent among athletes, the condition also considerably affects the general population [2, 3]. Risk factors for Achilles tendon injury include excessive mechanical overload and training errors, along with factors such as increased age, hypertension, obesity, diabetes, inflammatory arthropathies, and corticosteroid use [4]. Non-surgical management is generally pursued first and has reasonably high success rates. This involves a tailored combination of rest, orthotics, non-steroidal anti-inflammatory drugs, and eccentric exercise. However, for unknown reasons, nearly a third of patients continue to have persistent symptoms despite undergoing conservative care and proceed to undergo surgery [5].
Low levels of serum sex hormones can have detrimental impacts on patient’s health, with presentation including sexual dysfunction and decreases in muscle mass, strength, bone density, libido, and concentration [6, 7]. In patients with persistently low hormone concentration, testosterone replacement therapy (TRT) can be prescribed to restore physiologic levels and treat the symptoms of testosterone insufficiency [8]. Rate of TRT prescription increased more than threefold in the United States from 2001 to 2011, with most patients having no clear indication for therapy [9].
In addition to the well-understood risk of cardiovascular disease with the use of TRT, hepatic dysfunction, infertility, and tendon pathologies have also been associated with TRT [10]. Specifically, androgenic-anabolic steroids (AASs) have been associated with tendon rupture. Two hypotheses are proposed in the literature: AAS-induced rapid muscle hypertrophy is not accompanied by corresponding tendon adaptation and causes injury or AASs directly damage the structure of tendons, thereby leading to injury or rupture [10, 11]. In rodents, AASs have demonstrated negative effects on collagen synthesis, a critical aspect of tendon maintenance and repair, and in a cross-sectional cohort study, tendon rupture in AAS users was found to be 9 times more likely than in nonusers [12, 13]. Additionally, in a recently published study, TRT has been associated with increased rates of primary rotator cuff tears and secondary, ipsilateral cuff tears, making the relationship between TRT and Achilles tendon injury of significant interest [14]. Thus, this study aimed to determine the risk of Achilles tendon injury and subsequent surgery in patients who had received TRT. Our hypothesis was that patients who had previously been prescribed exogenous testosterone would have an increased likelihood of Achilles tendon injury and subsequently need increased rates of surgical management in comparison to controls, with these associations most notable among the older adult populations and similar between men and women.
Methods
Data source
This retrospective comparative analysis was performed using the de-identified data from M157Ortho dataset within the larger PearlDiver database (PearlDiver Technologies, Colorado Springs, CO). This database is generated using insurance claims from Humana Inc. and contains longitudinal data from over 150 million patients from January 2010 through December 2021, and has been an established resource used for orthopedic surgery research questions that require large datasets to answer [15]. These data allow researchers to characterize and analyze short-, medium-, and long-term rates and trends of various conditions, postoperative complications, and sequalae using the International Classifications of Disease, ninth revision (ICD-9) and tenth revision (ICD-10) and Current Procedural Terminology (CPT) codes. This dataset also includes prescription medication information, including the duration and time of prescriptions filled. For the current study, these data were used to calculate the rates of Achilles tendon injury and subsequent surgery in patients who filled prescriptions of exogenous testosterone for at least three consecutive months and compare it to a control [14].
Creating the cohorts
The testosterone group was generated by querying the M157Ortho dataset for patients matching the inclusion criteria, and then subsequently filtering out patients based on exclusion criteria. Inclusion criteria for the testosterone group were patients aged 35 to 75 who filled a prescription for exogenous testosterone for a minimum of 3 consecutive months with a minimum of two years follow up within the time period. The time range restriction allowed for a minimum of 2 years of follow-up for each patient. Exclusion criteria were patients with a diagnosis of rheumatologic disease, cancer, or mitochondrial disease, and those who were not active within the dataset (i.e., they dropped their coverage or changed insurance) within the two years after their prescription was filled, which facilitated a more accurate calculation of rate. The control cohort was created by first generating a random sample of patients between the ages of 35 and 75 who had never filled a prescription for testosterone before, and then utilizing one-to-one exact matching to yield a final cohort of patients matched with the testosterone cohort on age, sex, Charlson comorbidity index, and a diagnosis of tobacco use or diabetes. All codes used can be referenced in the Supplemental Table.
Identifying injuries and surgery
The rate and incidence (per 100,000 person-years) of index Achilles tendon injury were calculated over a two-year period following 3 consecutive months of testosterone and compared to the control cohort over the same time period. This analysis was performed for the combined cohort, as well as sex- (male or female) and age-specific (35 to 45, 46 to 55, 56 to 65, and 66 to 75 years) cohorts. These injuries were identified using ICD-9 and ICD-10 diagnosis codes. The primary Achilles tendon surgery rate was calculated by tracking the initial Achilles tendon injury diagnosis and determining the number of patients who filed a claim with an associated CPT code for surgery of the Achilles tendon. The rate of Achilles surgery was used as a proxy for the severity of the initially identified injury as more severe cases, including complete Achilles tendon rupture, are more likely to undergo surgery [16].
Statistical analysis
The incidence of index Achilles tendon injury was calculated for all cohorts and compared using the exact Poisson method. Chi-square tests were used for bivariate comparisons between the two matched cohorts. Multivariable logistic regression was used to compare the two cohorts while adjusting for other specific comorbidities, including a diagnosis of hypogonadism, chronic kidney disease, osteoporosis, overweight/obesity (body mass index > 25), class III obesity (body mass index > 40), alcohol use, osteoarthritis, lung disease, congestive heart failure, and dementia. These variables were included in the regression model to adjust for other factors that may influence either the rate of initial injury or rate of undergoing surgical management (i.e., poor surgical candidate secondary to concomitant lung or heart disease) [17–20]. The number needed to harm, a metric used to quantify the number of patients needed to be treated to cause harm to one patient, was calculated for the overall cohort by dividing 1 by the absolute risk increase. The absolute risk increase is the absolute difference in the testosterone event rate and the control event rate. Adjusted odds ratios (aOR) and their respective 95% confidence intervals were generated and reported for each comparison. To protect patient identity, cohorts comprised of less than 11 patients are reported as “-1” by PearlDiver and thus were reported as “< 11” throughout this manuscript. Statistical analyses are still able to be performed on these smaller cohorts, but the specific value is simply not reported. A p-value of < 0.05 was determined to represent statistical significance a priori. All statistical analyses were performed using the R Statistical Package (v4.2.1; R Core Team 2022, Vienna, Austria) embedded within PearlDiver.
Results
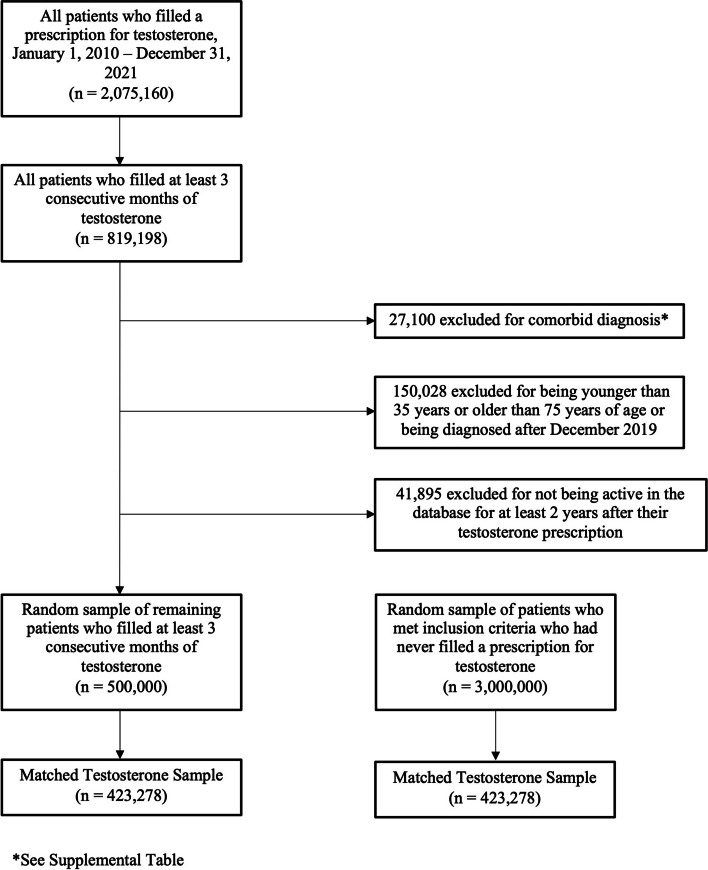
A total of 2,075,160 patients filled a prescription for exogenous testosterone, with 819,198 of those patients filling the prescription for a minimum of 3 months. Following exclusions and the 1:1 exact matching process, 423,278 patients comprised both the testosterone and control cohorts (Fig. 1). Prior to matching, both cohorts had significant demographic differences, including prior tobacco use and diabetes (Table 1). However, following the matching process, these differences no longer persisted. Table 2 depicts the number of patients in each of the age- and sex-specific cohorts.
Fig. 1.
Flow diagram depicting inclusion and exclusion criteria and the number of patients excluded at each point
Table 1.
Demographics comparison of the unmatched and matched cohorts
| Characteristic | Unmatched Cohorts | Matched Cohorts | ||||
|---|---|---|---|---|---|---|
| Prior Testosterone Use | Control | p-value | Prior Testosterone Use | Control | p-value | |
| Sex, n male (%) | 421,362 (84.3) | 1,222,831 (40.8) | < 0.001 | 345,369 (81.6) | 345,369 (81.6) | 1.000 |
| Age, mean ± SD | 56.3 ± 11.7 | 55.4 ± 10.5 | < 0.001 | 54.8 ± 10.4 | 54.8 ± 10.4 | 1.000 |
| CCI, mean ± SD | 0.8 ± 1.5 | 0.1 ± 0.5 | < 0.001 | 0.4 ± 0.8 | 0.4 ± 0.8 | 1.000 |
| Tobacco Use, n (%) | 164,212 (32.8) | 872,296 (29.1) | < 0.001 | 130,130 (30.7) | 130,130 (30.7) | 1.000 |
| Diabetes, n (%) | 203,436 (40.7) | 1,106,875 (36.9) | < 0.001 | 157,449 (37.2) | 157,449 (37.2) | 1.000 |
Bold indicates statistical significance
CCI Charlson comorbidity index, SD Standard deviation
Table 2.
A breakdown of the number of patients in both the experimental and control cohort following the matching process
| Population | Testosterone Use, n (%) | Control Cohort, n (%) | |
|---|---|---|---|
| Sex | Age | ||
| All Patients | 423,278 (100.0) | 423,278 (100.0) | |
| Male | 345,369 (81.6) | 345,369 (81.6) | |
| 35 to 45 | 80,608 (19.0) | 80,608 (19.0) | |
| 46 to 55 | 103,754 (24.5) | 103,754 (24.5) | |
| 56 to 65 | 92,961 (22.0) | 92,961 (22.0) | |
| 66 to 75 | 68,046 (16.1) | 68,046 (16.1) | |
| Female | 77,909 (18.4) | 77,909 (18.4) | |
| 35 to 45 | 10,120 (2.4) | 10,120 (2.4) | |
| 46 to 55 | 28,320 (6.7) | 28,320 (6.7) | |
| 56 to 65 | 28,185 (6.7) | 28,185 (6.7) | |
| 66 to 75 | 11,284 (2.7) | 11,284 (2.7) | |
Over the two-year follow-up period, the incidence of Achilles tendinopathy was 377.8 (95% CI, 364.8–391.0) injuries per 100,000 person-years, compared to 245.8 (95% CI, 235.4–256.6) injuries per 100,000 person-years (p < 0.001). The adjusted multivariable regression demonstrated that patients who previously filled prescriptions for testosterone were significantly more likely to be diagnosed with Achilles tendon injuries when compared to the control (aOR = 1.24, 95% CI, 1.15–1.33, p < 0.001), with an overall number needed to harm of 371. For male patients, there was a significantly increased likelihood of developing an Achilles tendon injury in each age cohort. Female patients had a significantly increased likelihood in all age cohorts except the 35 to 45 cohort. The two sex- and age-specific cohorts with the greatest increase in adjusted odds were the males aged 66 to 75 (aOR = 1.66, 95% CI, 1.35–2.03, p < 0.001) and the females aged 66 to 75 (aOR = 1.82, 95% CI, 1.21–2.75, p = 0.004). See Table 3 for all unadjusted and adjusted comparisons.
Table 3.
Comparison of the likelihood of being diagnosed with Achilles tendon injury over a two-year period
| Population and Time Period of Interest | Number of Injuries, n (%) | Regression Analysis | ||
|---|---|---|---|---|
| Testosterone Use | Control | aORa (95% CI) | p - value | |
| All Patients | 3,198 (0.76) | 2,081 (0.49) | 1.24 (1.15 - 1.33) | < 0.001 |
| Male | 2,565 (0.74) | 1,630 (0.47) | 1.38 (1.29 - 1.49) | < 0.001 |
| 35 to 45 | 651 (0.81) | 441 (0.55) | 1.40 (1.21 - 1.61) | < 0.001 |
| 46 to 55 | 846 (0.82) | 567 (0.55) | 1.28 (1.13 - 1.46) | < 0.001 |
| 56 to 65 | 710 (0.76) | 435 (0.47) | 1.51 (1.31 - 1.74) | < 0.001 |
| 66 to 75 | 358 (0.53) | 187 (0.27) | 1.66 (1.35 - 2.03) | < 0.001 |
| Female | 633 (0.81) | 451 (0.58) | 1.44 (1.27 - 1.64) | < 0.001 |
| 35 to 45 | 74 (0.73) | 58 (0.57) | 1.29 (0.89 - 1.87) | 0.175 |
| 46 to 55 | 241 (0.85) | 185 (0.65) | 1.33 (1.08 - 1.64) | 0.007 |
| 56 to 65 | 244 (0.87) | 167 (0.59) | 1.54 (1.25 - 1.90) | < 0.001 |
| 66 to 75 | 74 (0.66) | 41 (0.36) | 1.82 (1.21 - 2.75) | 0.004 |
Bold indicated statistical significance (p < 0.05)
aOR Adjusted odds ratio, CI Confidence interval
aadjusted used multivariable logistic regression to additionally control for hypogonadism, chronic kidney disease, osteoporosis, overweight/obesity (body mass index > 25), class III obesity (body mass index > 40), alcohol use, osteoarthritis, lung disease, congestive heart failure, and dementia
The testosterone cohort was also more likely to undergo surgery on their Achilles tendon injury when compared to the control cohort (Table 4.) Of the patients diagnosed with Achilles tendon injury, 287/3,198 (9.0%) of the testosterone cohort subsequently underwent surgery for their injury, compared to just 134/2,081 (6.4%) in the control cohort (aOR = 1.54, 95% CI, 1.19–1.99, p < 0.001). The two sex- and age-specific cohorts with the greatest increase in odds were the males aged 56 to 65 (aOR = 2.07, 95% CI, 1.17–3.75, p = 0.014) and the females aged 56 to 65 (aOR = 3.13, 95% CI, 1.33–8.25, p = 0.013).
Table 4.
Comparison of the likelihood of subsequently undergoing surgery for an Achilles tendon injury
| Population and Time Period of Interest | Number of Repairs, n (%) | Regression Analysis | ||
|---|---|---|---|---|
| Testosterone Use | Control | aORa (95% CI) | p - value | |
| All Patients | 287 (9.0) | 134 (6.4) | 1.54 (1.19 - 1.99) | 0.001 |
| Male | 231 (9.0) | 115 (7.1) | 1.24 (0.95 - 1.64) | 0.118 |
| 35 to 45 | 67 (10.3) | 47 (10.7) | 1.00 (0.62 - 1.62) | 0.989 |
| 46 to 55 | 71 (8.4) | 36 (6.3) | 1.23 (0.74 - 2.02) | 0.421 |
| 56 to 65 | 68 (9.6) | 21 (4.8) | 2.07 (1.17 - 3.75) | 0.014 |
| 66 to 75 | 25 (7.0) | 11 (5.9) | 1.08 (0.45 - 2.62) | 0.863 |
| Female | 56 (8.8) | 19 (4.2) | 2.57 (1.50 - 4.55) | < 0.001 |
| 35 to 45 | < 11 (N/A) | < 11 (N/A) | 1.89 (0.24 - 17.85) | 0.545 |
| 46 to 55 | 21 (8.7) | < 11 (N/A) | 2.25 (0.95 - 5.77) | 0.074 |
| 56 to 65 | 24 (9.8) | < 11 (N/A) | 3.13 (1.33 - 8.25) | 0.013 |
| 66 to 75 | < 11 (N/A) | < 11 (N/A) | 2.11 (0.35 - 17.55) | 0.432 |
Bold indicated statistical significance (p < 0.05)
aOR Adjusted odds ratio, CI Confidence interval
aadjusted used multivariable logistic regression to additionally control for hypogonadism, chronic kidney disease, osteoporosis, overweight/obesity (body mass index > 25), class III obesity (body mass index > 40), alcohol use, osteoarthritis, lung disease, congestive heart failure, and dementia
Discussion
The purpose of the current study was to evaluate the relationship between exogenous testosterone administration and Achilles tendon injury. Our results demonstrate that, when compared to a matched control cohort, the rate of Achilles tendon injury and subsequent surgery was significantly higher in patients prescribed TRT. Furthermore, this trend was found in both males and females even after statistical control for several medical comorbidities, most notably hypogonadism. These results demonstrate an important correlation between testosterone use and Achilles tendon injury and point to future clinical implications of testosterone prescription and usage. Much of prior literature has focused on the effects of anabolic steroids on tendon structure and health, but this study is the first to retrospectively analyze rates of Achilles tendon injuries and subsequent surgery in patients prescribed TRT.
In the Achilles tendon, testosterone has been linked to increased tendon stiffness, a reduced relaxin response, and inhibited matrix metalloproteinase (MMPs) activity, all of which negatively affect tendon integrity after repeated strain [21, 22]. At a molecular level, tendon health is maintained by a balance of MMPs and tissue inhibitors of matrix metalloproteinases (TIMPs) that is crucial for tissue healing, and the use of exogenous steroids has been demonstrated to significantly alter this balance, leading to collagen dysplasia in rats and a consequent increase in Achilles tendon injury [23–25]. In multiple studies on human AAS users, increased rates of tendon rupture were discovered compared to non-AAS users [12, 13, 26]. Multiple case reports on rare tendon avulsions and ruptures have also linked AAS usage of the patients to the cause of the tendon injury [27–29]. In the context of testosterone’s biochemical effects on tendon health and structure and its rising usage and prescription, it imperative to better understand the musculoskeletal complications associated with exogenous testosterone in order to minimize patient harm.
This study reports an increased odds of 1.38 in Achilles tendon injury in males and an increased odds of 1.44 in females with a minimum of three consecutive months of prescription TRT. While the current body of literature regarding prescription TRT and musculotendinous injury is scarce, a recent study by Testa et al. in a similar analysis demonstrated a significant increase in rotator cuff tears and revision rotator cuff repair [14]. A second similar study published by Smith et al. evaluated rates of rotator cuff repair (RCR) in patients that were sex-hormone deficient and they found an 89% increase in rates of RCR in males who were testosterone deficient and a 48% increase in estrogen-deficient females [30]. Although Smith et al. do not account for testosterone supplementation in sex-hormone-deficient patients, their study does allude to the complex relationship between sex-hormone derangements and musculotendinous health, which is in accordance with the present study. The present study builds on this literature by adjusting for previously diagnosed hypogonadism and other comorbidities associated with musculoskeletal injury when evaluating and comparing rates of Achilles tendon injury and subsequent surgery in patients prescribed consistent TRT, identifying increased odds of Achilles tendon injury and subsequent surgery regardless of hypogonadism status and after controlling for other comorbidities.
Prior studies observed increased rates of injury in male patients compared to female patients. Testa et al. found increased odds of rotator cuff tears in both male and female patients prescribed TRT, with males having a higher aOR of 4.73 in comparison to 3.49 for females [14]. In another study on the association of TRT with rates of distal biceps tendon injury (BTI), Rebello et al. demonstrated that male patients prescribed TRT were 4.68 times more likely to experience distal BTI, while females prescribed TRT were 1.82 times more likely [31]. There are many speculative explanations for these findings, including the prevalence weight training, strain from higher force loads across the tendon, or rates of aerobic exercise participation [32–35]. Our study observed increased odds of general Achilles tendon injury in both men and women prescribed TRT, but in contrast to the above studies, the association was similar between men and women. However, when only considering full-thickness Achilles tendon ruptures, previous literature has identified a 5:1 ratio of men to women in the occurrence of rupture, which may be more consistent with other published literature on tendon injuries and TRT [32]. Additionally, other studies have demonstrated the decreased stiffness of the Achilles tendon in women, including Intziegianni et al. who measured an increased tendon cross-sectional area deformation under a lower applied force in female tendons, pointing to a more compliant and less injury-prone tendon that could be associated with the decreased rates of Achilles tendon rupture in women [32, 33]. Nevertheless, it remains difficult to truly explain this association without further understanding of the interplay between testosterone and estrogen in maintaining tendon health and as the effects of estrogen on tendon size, stiffness, and collagen content are still debated, this interconnection is likely multifaceted and remains a topic for additional research [36–38].
Within both male and female cohorts in this study, the highest adjusted odds were seen in the aged 66 to 75 cohorts. As hypogonadism increases with age, the presence of this trend in the oldest age cohort regardless of patient hypogonadism status indicates that there is likely a separate explanation for this observation. One such reason is that tendon physiology and structure can change significantly due to aging. In a study by Pardes et al., Achilles tendon properties were measured and compared among rats of various ages, with increased passive stiffness and decreased range of motion seen in older rats. Older rats were also found to have Achilles tendons with lower maximum stresses and lower elasticities compared to those of younger rats, additional properties that make a tendon more susceptible to injury [39]. Aging has a similar impact on tendons in humans, and causes additional biological changes such as decreased cellularity, reduced proliferation and activity of tenocytes, and decreased organization of collagen fibers that all weaken tendon structure [40]. The association of TRT with decreased obesity, improved energy levels, and increased self-esteem in patients with hypogonadism implicates a possibility for increased physical activity in these patients as well, which, in combination with the biomechanical and physiologic changes in tendon structure, could explain the increased odds of Achilles tendon injury in the oldest cohorts [41]. The interplay between degenerative tendon changes and participation in physical activity of each age group could also determine the acuity of the Achilles tendon injury sustained, thereby affecting the odds of undergoing surgery.
The decision to undergo surgery is also heavily impacted by the physician’s evaluation of a patient’s healing potential, which decreases with age [40]. Thus, despite having the highest odds of Achilles tendon injury, the aged 66 to 75 TRT cohorts did not have significant increases in likelihood of surgery possibly due to physician preference of nonsurgical treatment for older patients regardless of TRT prescription. Among the remaining age-sex TRT cohorts, both males and females aged 56 to 65 had significant odds of undergoing surgery for their Achilles tendon injury, which, speculatively, may be a result of an increase in participation in high-intensity exercise along with less physician preference for nonsurgical treatment out of concern for age-related complications of surgery in comparison to the oldest cohorts [42]. As this trend is not present for the male cohorts aged 56–65, in which the odds were significantly higher than the control cohorts, there likely is an additional layer of complexity in the relationship among sex, comorbidities, and TRT prescription that could explain the increased odds of surgical intervention for Achilles tendon injury for specific age-sex cohorts.
Limitations
As with all retrospective administrative claims database studies, this study is not without limitations. As a retrospective and observational study, the results of this study cannot be used to make any causal inference between TRT and Achilles tendon injury. In addition, while this study does use one-to-one matching on patient demographics and the Charlson comorbidity index and subsequently controls for several comorbid medical conditions in the multivariable analyses, there are several other factors that place patients at increased risk of injury that could not be captured in this study, such as patient baseline activity level. Similarly, this study could not control for the specific Achilles tendon injury each patient incurred; there is no specific CPT code for Achilles tendon rupture and therefore we could not directly look at these injuries, which is an important entity to be considered more closely in future studies. We were unable to address additional informative factors including degree of injury, muscle atrophy, and degree of muscle retraction in potential ruptures, as well as details regarding the surgery, including specific method used, surgeon experience, or the location of the operation. We are likewise unable to determine the specific dosing of TRT, route of administration, changes in dosing over time, compliance with the prescribed dosing, or the testosterone levels of the patient over time. Therefore, this study is unable to draw any conclusions on the dose-related effect of TRT or the effect of route of administration on Achilles tendon injury and is only able to determine that there is an association between a minimum of three months of testosterone replacement therapy and increased rates of Achilles tendon injury. Finally, while this study does capture over 400,000 patients who filled prescriptions for TRT, this study only includes data from a single insurer, Humana Inc., and may not be representative of the greater national population that would include patients insured through other private companies, Medicare, or Medicaid. Notably, the much smaller sample sizes of female cohorts make it difficult to obtain a comprehensive perspective on the female population that is prescribed TRT, limiting insight into the sex differences in TRT- associated Achilles tendon injury.
Conclusion
By using a national administrative claims database, this study observed a significant increase in the likelihood of both Achilles tendon injury and Achilles tendon injury requiring surgery in patients who filled a minimum of three consecutive months of prescription TRT. While prior studies have documented musculotendinous injury in the context of exogenous steroid administration, this is the first study to specifically associate prescription TRT with Achilles tendon injury and subsequent surgery. These results are important for the prescribing physicians and patients considering TRT to recognize when formulating treatment plans and are a valuable addition to the growing body of literature regarding the effects of testosterone therapy and musculoskeletal health.
Supplementary Information
Additional file 1: Supplemental Table. All International Classifications of Disease (ICD), ninth and tenth revision and Current Procedural Terminology (CPT) codes used.
Acknowledgements
None.
Abbreviations
- TRT
Testosterone replacement therapy
- OR
Odds ratio
- aOR
Adjusted odds ratio
- CI
Confidence interval
- AAS
Androgenic-anabolic steroids
- MMP
Matrix metalloproteinases
- TIMP
Tissue inhibitors of matrix metalloproteinases
- RCR
Rotator cuff repair
Authors’ contributions
JAA introduced the idea, analyzed and interpreted the data, assisted in generating the first draft of the manuscript, and revised the manuscript. ML assisted in generating the first draft of the manuscript and revised the manuscript. ER assisted in generating the first draft of the manuscript. JG assisted in generating the first draft of the manuscript. EJT assisted in generating the first draft of the manuscript, revised the manuscript, and served in a supervising capacity for the project. AHD served as a supervisor, provided the resources to complete the project, and revised and edited the manuscript. MA served as a supervisor, provided the resources to complete the project, and revised and edited the manuscript.
Funding
None.
Availability of data and materials
The data that support the findings of this study are available from PearlDiver, Inc. but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. However, data are available from the authors upon reasonable request and with permission of PearlDiver, Inc.
Declarations
Ethics approval and consent to participate
All data used was extracted from a publicly accessible database with no patient identifying information, so the study was not required to undergo IRB review at our institution.
Consent for publication
Not applicable.
Competing interests
JAA, ML, ER, JG, EJT, and MA have no competing interests to report. AHD reports the following competing interests: EOS, paid consultant; Orthofix, Inc., paid consultant, research support; SpineArt, paid consultant; Medtronic/Medicrea, paid consultant; Springer: publishing royalties, financial or material support; Stryker: paid consultant, all outside submitted work.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Maffulli N, Longo UG, Kadakia A, Spiezia F. Achilles tendinopathy. Foot Ankle Surg. 2020;26(3):240–249. doi: 10.1016/j.fas.2019.03.009. [DOI] [PubMed] [Google Scholar]
- 2.Zafar MS, Mahmood A, Maffulli N. Basic science and clinical aspects of achilles tendinopathy. Sports Med Arthrosc Rev. 2009;17(3):190–197. doi: 10.1097/JSA.0b013e3181b37eb7. [DOI] [PubMed] [Google Scholar]
- 3.Magnan B, Bondi M, Pierantoni S, Samaila E. The pathogenesis of achilles tendinopathy: a systematic review. Foot Ankle Surg. 2014;20(3):154–159. doi: 10.1016/j.fas.2014.02.010. [DOI] [PubMed] [Google Scholar]
- 4.Li HY, Hua YH. Achilles tendinopathy: current concepts about the basic science and clinical treatments. Biomed Res Int. 2016;2016:1–9. doi: 10.1155/2016/6492597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.von Rickenbach KJ, Borgstrom H, Tenforde A, Borg-Stein J, McInnis KC. Achilles tendinopathy: evaluation, rehabilitation, and prevention. Curr Sports Med Rep. 2021;20(6):327–334. doi: 10.1249/JSR.0000000000000855. [DOI] [PubMed] [Google Scholar]
- 6.Bassil N, Alkaade S, Morley JE. The benefits and risks of testosterone replacement therapy: a review. Ther Clin Risk Manag. 2009;5:427. doi: 10.2147/tcrm.s3025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bhasin S, Brito JP, Cunningham GR, Hayes FJ, Hodis HN, Matsumoto AM, et al. Testosterone therapy in men with hypogonadism: an endocrine society clinical practice guideline. J Clin Endocrinol Metabolism. 2018;103(5):1715–1744. doi: 10.1210/jc.2018-00229. [DOI] [PubMed] [Google Scholar]
- 8.Wang C, Swerdloff RS. Testosterone replacement therapy in hypogonadal men. Endocrinol Metab Clin North Am. 2022;51(1):77–98. doi: 10.1016/j.ecl.2021.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Baillargeon J, Kuo YF, Westra JR, Urban RJ, Goodwin JS. Testosterone prescribing in the United States, 2002–2016. JAMA. 2018;320(2):200. doi: 10.1001/jama.2018.7999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jones IA, Togashi R, Hatch GFR, Weber AE, Vangsness CT. Anabolic steroids and tendons: a review of their mechanical, structural, and biologic effects. J Orthop Res. 2018;36(11):2830–2841. doi: 10.1002/jor.24116. [DOI] [PubMed] [Google Scholar]
- 11.Horwitz H, Andersen JT, Dalhoff KP. Health consequences of androgenic anabolic steroid use. J Intern Med. 2019;285(3):333–340. doi: 10.1111/joim.12850. [DOI] [PubMed] [Google Scholar]
- 12.Weber AE, Gallo MC, Bolia IK, Cleary EJ, Schroeder TE, Rick Hatch GF. Anabolic androgenic steroids in orthopaedic surgery: current concepts and clinical applications. JAAOS Glob Res Rev. 2022;6(1). Available from: https://journals.lww.com/10.5435/JAAOSGlobal-D-21-00156. Cited 2023 Feb 23. [DOI] [PMC free article] [PubMed]
- 13.Kanayama G, DeLuca J, Meehan WP, Hudson JI, Isaacs S, Baggish A, et al. Ruptured tendons in anabolic-androgenic steroid users: a cross-sectional cohort study. Am J Sports Med. 2015;43(11):2638–2644. doi: 10.1177/0363546515602010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Testa EJ, Albright JA, Hartnett D, Lemme NJ, Daniels AH, Owens BD, et al. The relationship between testosterone therapy and rotator cuff tears, repairs, and revision repairs. J Am Acad Orthop Surg. 2023. Publish Ahead of Print. Available from: https://journals.lww.com/10.5435/JAAOS-D-22-00554. Cited 2023 Feb 12. [DOI] [PubMed]
- 15.Bolognesi MP, Habermann EB. Commercial claims data sources: pearldiver and individual payer databases. J Bone Joint Surg. 2022;104(Suppl 3):15–17. doi: 10.2106/JBJS.22.00607. [DOI] [PubMed] [Google Scholar]
- 16.Medina Pabon M, Naqvi U. Achilles tendinitis. Treasure Island (FL): StatPearls Publishing; 2022. [PubMed] [Google Scholar]
- 17.Golds G, Houdek D, Arnason T. Male hypogonadism and osteoporosis: the effects, clinical consequences, and treatment of testosterone deficiency in bone health. Int J Endocrinol. 2017;2017:1–15. doi: 10.1155/2017/4602129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shapses SA, Pop LC, Wang Y. Obesity is a concern for bone health with aging. Nutr Res. 2017;39:1–13. doi: 10.1016/j.nutres.2016.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Deme S, Fisseha B, Kahsay G, Melese H, Alamer A, Ayhualem S. Musculoskeletal disorders and associated factors among patients with chronic kidney disease attending at Saint Paul Hospital, Addis Ababa, Ethiopia. Int J Nephrol Renovasc Dis. 2021;14:291–300. doi: 10.2147/IJNRD.S319991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wieczorek M, Gwinnutt JM, Ransay-Colle M, Balanescu A, Bischoff-Ferrari H, Boonen A, et al. Smoking, alcohol consumption and disease-specific outcomes in rheumatic and musculoskeletal diseases (RMDs): systematic reviews informing the 2021 EULAR recommendations for lifestyle improvements in people with RMDs. RMD Open. 2022;8(1):e002170. doi: 10.1136/rmdopen-2021-002170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hansen M, Kjaer M. Sex hormones and tendon. Adv Exp Med Biol. 2016;920:139–149. doi: 10.1007/978-3-319-33943-6_13. [DOI] [PubMed] [Google Scholar]
- 22.Marqueti RC, Parizotto NA, Chriguer RS, Perez SEA, Selistre-de-Araujo HS. Androgenic-anabolic steroids associated with mechanical loading inhibit matrix metallopeptidase activity and affect the remodeling of the achilles tendon in rats. Am J Sports Med. 2006;34(8):1274–1280. doi: 10.1177/0363546506286867. [DOI] [PubMed] [Google Scholar]
- 23.Tsitsilonis S, Chatzistergos PE, Mitousoudis AS, Kourkoulis SK, Vlachos IS, Agrogiannis G, et al. Anabolic androgenic steroids reverse the beneficial effect of exercise on tendon biomechanics: an experimental study. Foot Ankle Surg. 2014;20(2):94–99. doi: 10.1016/j.fas.2013.12.001. [DOI] [PubMed] [Google Scholar]
- 24.Michna H. Tendon injuries induced by exercise and anabolic steroids in experimental mice. Int Orthop. 1987;11(2):157–162. doi: 10.1007/BF00266702. [DOI] [PubMed] [Google Scholar]
- 25.Vincenti MP. Matrix metalloproteinase protocols. New Jersey: Humana Press; 2000. The Matrix Metalloproteinase (MMP) and Tissue Inhibitor of Metalloproteinase (TIMP) genes: transcriptional and posttranscriptional regulation, signal transduction and cell-type-specific expression; pp. 121–48. [DOI] [PubMed] [Google Scholar]
- 26.Morgenthaler M, Weber M. [Pathological rupture of the distal biceps tendon after long-term androgen substitution] Z Orthop Ihre Grenzgeb. 1999;137(4):368–370. doi: 10.1055/s-2008-1039728. [DOI] [PubMed] [Google Scholar]
- 27.Gould HP, Hawken JB, Duvall GT, Hammond JW. Asynchronous bilateral achilles tendon rupture with selective androgen receptor modulators: a case report. JBJS Case Connect. 2021;11(2):e20. doi: 10.2106/JBJS.CC.20.00635. [DOI] [PubMed] [Google Scholar]
- 28.Sollender JL, Rayan GM, Barden GA. Triceps tendon rupture in weight lifters. J Shoulder Elbow Surg. 1998;7(2):151–153. doi: 10.1016/S1058-2746(98)90227-0. [DOI] [PubMed] [Google Scholar]
- 29.Liow RY, Tavares S. Bilateral rupture of the quadriceps tendon associated with anabolic steroids. Br J Sports Med. 1995;29(2):77–79. doi: 10.1136/bjsm.29.2.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Smith KM, Hotaling JM, Presson AP, Zhang C, Horns JJ, Cannon-Albright LA, et al. The effect of sex hormone deficiency on the incidence of rotator cuff repair: analysis of a large insurance database. J Bone Joint Surg. 2022;104(9):774–779. doi: 10.2106/JBJS.21.00103. [DOI] [PubMed] [Google Scholar]
- 31.Rebello E, Albright JA, Testa EJ, Alsoof D, Daniels AH, Arcand M. The use of prescription testosterone is associated with an increased likelihood of experiencing a distal biceps tendon injury and subsequently requiring surgical repair. J Shoulder Elbow Surg. 2023;32(6):1254–1261. doi: 10.1016/j.jse.2023.02.122. [DOI] [PubMed] [Google Scholar]
- 32.Vosseller JT, Ellis SJ, Levine DS, Kennedy JG, Elliott AJ, Deland JT, et al. Achilles tendon rupture in women. Foot Ankle Int. 2013;34(1):49–53. doi: 10.1177/1071100712460223. [DOI] [PubMed] [Google Scholar]
- 33.Intziegianni K, Cassel M, Hain G, Mayer F. Gender differences of achilles tendon cross-sectional area during loading. Sports Med Int Open. 2017;1(04):E135–140. doi: 10.1055/s-0043-113814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.2020 U.S. Running Trends. Running USA; 2020:1–10. Available from: https://www.wpr.org/sites/default/files/running_usa_trends_report_2019-r4.pdf. Cited 2023 Aug 6.
- 35.Chen W, Cloosterman KLA, Bierma-Zeinstra SMA, van Middelkoop M, de Vos RJ. Epidemiology of insertional and midportion Achilles tendinopathy in runners: A prospective cohort study. J Sport Health Sci. 2023;S2095-2546(23)00037-6. 10.1016/j.jshs.2023.03.007. Accessed 6 Aug 2023. [DOI] [PubMed]
- 36.Chidi-Ogbolu N, Baar K. Effect of estrogen on musculoskeletal performance and injury risk. Front Physiol. 2018;9:1834. doi: 10.3389/fphys.2018.01834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hansen M, Kjaer M. Influence of sex and estrogen on musculotendinous protein turnover at rest and after exercise. Exerc Sport Sci Rev. 2014;42(4):183–192. doi: 10.1249/JES.0000000000000026. [DOI] [PubMed] [Google Scholar]
- 38.Leblanc DR, Schneider M, Angele P, Vollmer G, Docheva D. The effect of estrogen on tendon and ligament metabolism and function. J Steroid Biochem Mol Biol. 2017;172:106–116. doi: 10.1016/j.jsbmb.2017.06.008. [DOI] [PubMed] [Google Scholar]
- 39.Pardes AM, Beach ZM, Raja H, Rodriguez AB, Freedman BR, Soslowsky LJ. Aging leads to inferior achilles tendon mechanics and altered ankle function in rodents. J Biomech. 2017;60:30–38. doi: 10.1016/j.jbiomech.2017.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lui PPY, Wong CM. Biology of tendon stem cells and tendon in aging. Front Genet. 2019;10:1338. doi: 10.3389/fgene.2019.01338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kelly DM, Jones TH. Testosterone and obesity: testosterone and obesity. Obes Rev. 2015;16(7):581–606. doi: 10.1111/obr.12282. [DOI] [PubMed] [Google Scholar]
- 42.Ho G, Tantigate D, Kirschenbaum J, Greisberg JK, Vosseller JT. Increasing age in achilles rupture patients over time. Injury. 2017;48(7):1701–1709. doi: 10.1016/j.injury.2017.04.007. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Supplemental Table. All International Classifications of Disease (ICD), ninth and tenth revision and Current Procedural Terminology (CPT) codes used.
Data Availability Statement
The data that support the findings of this study are available from PearlDiver, Inc. but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. However, data are available from the authors upon reasonable request and with permission of PearlDiver, Inc.