Abstract
Osteoporosis is a prevalent bone disorder characterized by low bone mineral density (BMD) and deteriorated bone microarchitecture, leading to an increased risk of fractures. Vitamin D (VD), an essential nutrient for skeletal health, plays a vital role in maintaining bone homeostasis. The biological effects of VD are primarily mediated through the vitamin D receptor (VDR), a nuclear receptor that regulates the transcription of target genes involved in calcium and phosphate metabolism, bone mineralization, and bone remodeling. In this review article, we conduct a thorough literature search of the PubMed and EMBASE databases, spanning from January 2000 to September 2023. Utilizing the keywords “vitamin D,” “vitamin D receptor,” “osteoporosis,” and “therapy,” we aim to provide an exhaustive overview of the role of the VD/VDR system in osteoporosis pathogenesis, highlighting the most recent findings in this field. We explore the molecular mechanisms underlying VDR’s effects on bone cells, including osteoblasts and osteoclasts, and discuss the impact of VDR polymorphisms on BMD and fracture risk. Additionally, we examine the interplay between VDR and other factors, such as hormonal regulation, genetic variants, and epigenetic modifications, that contribute to osteoporosis susceptibility. The therapeutic implications of targeting the VDR pathway for osteoporosis management are also discussed. By bringing together these diverse aspects, this review enhances our understanding of the VD/VDR system’s critical role in the pathogenesis of osteoporosis and highlights its significance as a potential therapeutic target.
Keywords: Osteoporosis, Vitamin D receptor, Bone mineral density, Bone remodeling, Calcium metabolism, Genetic variations
Introduction
Osteoporosis is a significant and growing health concern worldwide, particularly among the aging population [1, 2]. It is characterized by reduced bone mineral density (BMD) and deteriorated bone microarchitecture, resulting in increased skeletal fragility and a heightened risk of fractures [1, 2]. According to the International Osteoporosis Foundation, osteoporosis affects an estimated 200 million individuals globally, and this number is projected to rise as the population continues to age [3]. The overall prevalence of osteoporosis among adults aged 50 and over is nearly 13% while it rises to approximately 49% among women over 50 ages [2, 3]. The present rapid growth of the elderly population has made osteoporosis a major healthcare burden that has resulted in enormous and growing costs to national healthcare systems [1–3].
The pathogenesis of osteoporosis is complex and multifactorial, involving a delicate interplay between genetic, hormonal, environmental, and lifestyle factors (Fig. 1A) [4, 5]. Importantly, a deficiency of vitamin D (VD) is an important cause that leads to osteoporosis [6]. It is crucial to understand the underlying mechanisms that contribute to bone loss and compromised bone quality in order to develop effective preventive and therapeutic strategies.
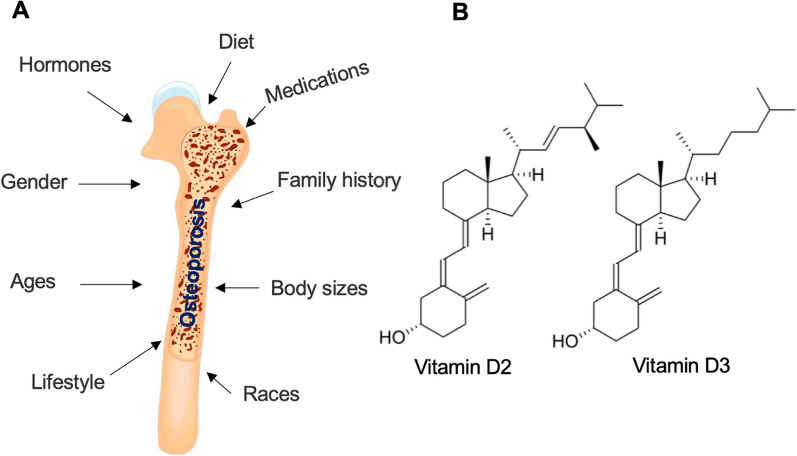
Fig. 1.
Factors involved in the pathogenesis of osteoporosis and chemical structures of vitamin D2/D3. A Factors contributing to pathogenesis of osteoporosis. Osteoporosis can be affected by multiple factors, such as gender, age, race, body size, family history, diet, changes in hormones, lifestyle, and special medicines. B Chemical structures of vitamin D2 (C28H44O) and D3 (C27H44O) are shown
VD, in particular VD2 and VD3 (Fig. 1B), is a group of fat-soluble secosteroids vital for skeletal health, and it plays a key role in maintaining bone homeostasis [7, 8]. Its biological effects are primarily mediated via the vitamin D receptor (VDR), a nuclear receptor that governs the transcription of target genes [7, 8]. These genes are involved in a wide array of processes, including calcium and phosphate metabolism, bone mineralization, and bone remodeling, all critical to the preservation of bone health [9]. Dysregulation of the VD/VDR system has been linked to several bone pathologies, including osteoporosis [10]. Recent advancements have shed light on how the VD/VDR system influences the behavior of bone cells, such as osteoblasts and osteoclasts, and how VDR polymorphisms might affect BMD and fracture risk [1, 11, 12]. Moreover, the interplay between the VDR and other factors, such as hormonal regulation, genetic variants, and epigenetic modifications, has become a topic of intense investigation [13, 14].
In this review, we aim to provide a comprehensive overview of the current understanding of the VD/VDR system's role in osteoporosis pathogenesis. By consolidating recent findings, we underscore the relevance of the VD/VDR system in the onset and progression of osteoporosis, while also highlighting potential therapeutic avenues for disease management.
Uptake, synthesis, and metabolism of VD
VD can be sourced from natural sun exposure and certain foods, including fatty fish, fish liver oils, beef liver, egg yolks, and mushrooms. Additionally, various foods, such as cheeses, milk, butter, cereal, and whole grains are often fortified with VD supplements to enhance dietary intake (Fig. 2) [7, 15]. The synthesis of VD begins in the skin, where exposure to ultraviolet B (UVB) radiation converts 7-dehydrocholesterol, a compound found in significant amounts in the skin, into previtamin D3 (Fig. 2) [7, 15]. Previtamin D3 is then transformed into vitamin D3 (VD3), or cholecalciferol, via a heat-dependent process (Fig. 2) [7, 15]. Following its synthesis, VD3 is metabolized in the liver by the enzyme 25-hydroxylase to 25-hydroxyvitamin D3, also known as calcifediol (Fig. 2) [7, 15].
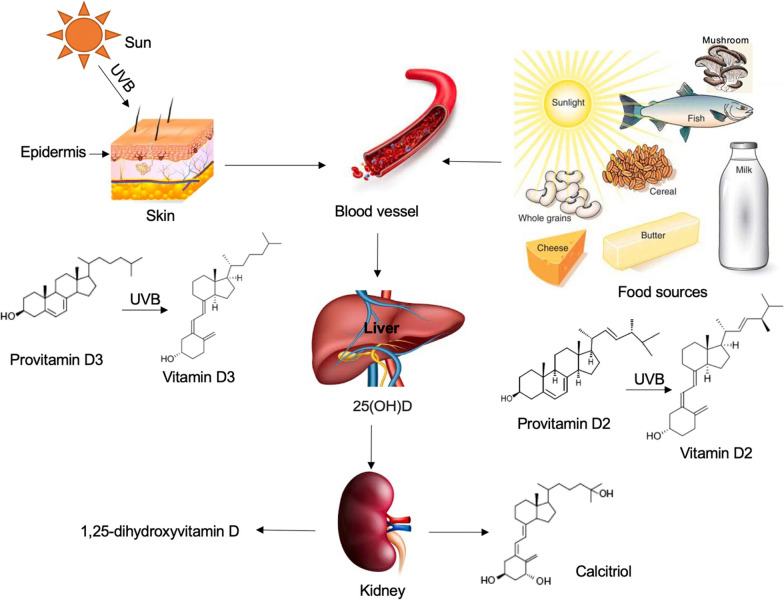
Fig. 2.
Uptake, synthesis, and metabolism of vitamin D2 and D3. Vitamin D can be obtained through natural sun exposure and from dietary sources such as fish, mushrooms, cheeses, milk, butter, cereal, and whole grains. Vitamin D3 is synthesized in the skin when pro-vitamin D3 (7-dehydrocholesterol) is converted to pre-vitamin D3 in response to sunlight exposure (ultraviolet B radiation). Alternatively, Vitamin D3 can be obtained from natural or fortified foods and supplements from vitamin D2 (ergocalciferol) and vitamin D3. After entering the bloodstream, vitamin D2 and D3 bind to vitamin D-binding protein (DBP) and are transported to the liver where they are hydroxylated by liver 25-hydroxylases to form 25-hydroxycholecalciferol [25(OH)D] (calcifediol or calcidiol). 25(OH)D is then transported to the kidney where it is further hydroxylated by 1α-hydroxylase to produce the active secosteroid 1,25(OH)2D (calcitriol). The synthesis of 1,25(OH)2D is regulated by parathyroid hormone and is suppressed by calcium, phosphate, and 1,25(OH)2D itself
VD metabolism occurs primarily in the kidneys but also in other tissues, where the enzyme 1α-hydroxylase converts calcifediol into its active form, calcitriol (1,25-dihydroxyvitamin D3) (Fig. 2) [7, 15, 16]. This conversion is tightly regulated by several factors including serum levels of parathyroid hormone, calcium, and phosphate [11]. Recent studies have found that dysregulation in this final conversion step could lead to inadequate levels of active VD, contributing to the pathogenesis of various health conditions including osteoporosis [17].
In addition to endogenous synthesis, dietary intake also contributes to the body's VD status [7, 8]. The primary dietary forms of VD are vitamin D3, found in foods like fatty fish and egg yolks, and vitamin D2, or ergocalciferol, which is obtained from plant sources and fortified foods (Fig. 2) [7, 8]. Both forms are metabolized in the same manner within the body [7, 8].
Structure and functions of VDR
Molecular structure of VDR
VDR is a nuclear hormone receptor that regulates gene expression in response to VD [18]. The VDR protein consists of several domains that contribute to its structure and function [19, 20]. The N-terminal domain (NTD) contains transcriptional activation functions and interacts with coactivators and corepressors (Fig. 3A) [19, 20]. The DNA-binding domain (DBD) allows the VDR to bind to specific DNA sequences called vitamin D response elements (VDREs) (Fig. 3A) [19, 20]. The ligand-binding domain (LBD) is responsible for ligand recognition and dimerization with the retinoid X receptor (RXR) (Fig. 3A) [19, 20]. Together, these domains enable the VDR to interact with DNA and other proteins to modulate gene expression [19, 20].
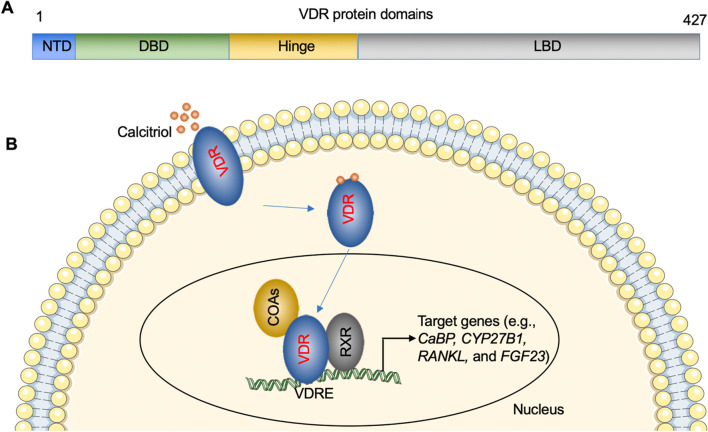
Fig. 3.
Functional domains of VDR protein and activation of VDR. A Functional domains of VDR proteins. Different domains, including NTD, DBD, and LBD, are shown. B The classic VDR signaling. The VDR-ligand complex forms a heterodimer with RXR, and the complex translocates to the nucleus. In the nucleus, the VDR-RXR heterodimer binds to VDREs located in the promoter regions of target genes, leading to the recruitment of coactivators and the initiation of gene transcription
Ligand binding and activation of VDR
VDR is activated by binding to its ligand, calcitriol [21]. Upon ligand binding, the VDR undergoes conformational changes that facilitate its interaction with coactivator proteins [22]. The VDR-ligand complex then forms a heterodimer with RXR, and the complex translocates to the nucleus [22]. In the nucleus, the VDR-RXR heterodimer binds to VDREs located in the promoter regions of target genes, leading to the recruitment of coactivators and the initiation of gene transcription (Fig. 3B) [22]. The ligand-binding process is crucial for the activation of VDR and the regulation of target gene expression [22].
VDR signaling pathways
Activation of VDR can trigger a multitude of signaling pathways that contribute to its myriad physiological effects, ranging from maintaining bone homeostasis to modulating immune responses [23–25]. The binding of VDR–RXR heterodimer to VDREs on gene promoters represents the classic genomic pathway of VDR, which has been shown to regulate numerous genes [e.g., Calbindin (CaBP), Cytochrome P450, Family 27, Subfamily B, Polypeptide 1 (CYP27B1), nuclear factor kappa-Β ligand (RANKL), fibroblast growth factor 23 (FGF23)] involved in calcium and phosphate metabolism (Fig. 3B), crucial for bone health [9, 23, 26].
Besides the classic pathway, VDR can also initiate rapid non-genomic actions, which take effect within minutes to hours of VD administration [27, 28]. In these pathways, VDR interacts with and is phosphorylated by various protein kinases, such as protein kinase A (PKA), mitogen-activated protein kinase (MAPK), protein kinase C beta (PKC-β), and casein kinase 2 (CK2) or with phospholipase C to modulate intracellular calcium and phosphate levels (Fig. 4A) [27, 29, 30].
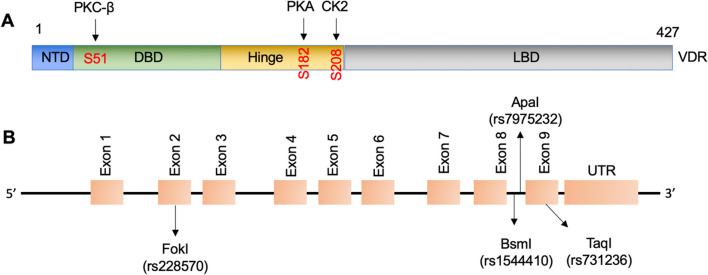
Fig. 4.
Phosphorylation sites of VDR by kinases and chromosomal positions of VDR polymorphisms. A Phosphorylation sites of VDR by kinases (PKC-β, PKA, and CK2). B Chromosomal positions of four VDR polymorphisms (rs7975232, rs1544410, rs228570, and rs731236) are shown
Moreover, VDR can interact with other signaling molecules, exemplified by its crosstalk with the Wnt/β-catenin signaling pathway, which is instrumental for bone formation [31, 32]. In addition, VDR has been found to interact with other nuclear receptors, such as estrogen receptor (ER) and peroxisome proliferator-activated receptor gamma (PPARγ), indicating complex interactions in regulating bone health and inflammation [33].
Emerging evidence indicates a pivotal role for the VD/VDR signaling pathway in modulating inflammatory responses as well. Some studies have demonstrated the inhibitory effect of VD/VDR signaling on the activation of the NF-κB pathway, a key player in inflammation, in specific cell types such as embryonic fibroblasts and intestinal epithelial cells [34–37]. Moreover, VD has been found to downregulate the expression of hypoxia-inducible factor-1α (HIF-1α) in osteoclasts, while tumor necrosis factor-alpha (TNFα) appears to reciprocally decrease VDR levels [38]. Additionally, VDR signaling has been implicated in immune regulation, cell proliferation, and differentiation [39, 40]. The VDR is expressed in immune cells, and its activation can modulate immune responses, such as the production of antimicrobial peptides and the regulation of immune cell differentiation [39, 40].
Regulation of VDR expression
The expression of VDR can be regulated at multiple levels. Transcriptional regulation plays a crucial role in determining VDR abundance. Factors such as VD status, calcium levels, and hormonal signals can influence VDR gene transcription [25, 41]. Additionally, posttranscriptional mechanisms, including mRNA stability and microRNA-mediated regulation, can impact VDR expression [25, 41]. Furthermore, epigenetic modifications, such as DNA methylation and histone modifications, can affect VDR expression by altering chromatin structure and accessibility [25, 41]. These modifications can be influenced by environmental factors, including diet, lifestyle, and exposure to sunlight [25, 41].
Effects of VD/VDR on osteoblasts and osteoclasts
VDR regulation of osteoblast function and mineralization
Osteoblasts, the cells responsible for bone formation, play a crucial role in maintaining skeletal integrity and bone remodeling [42]. Emerging evidence has highlighted the importance of VDR in regulating osteoblast function and differentiation [43, 44]. After binding of calcitriol, VDR activation promotes osteoblast differentiation, a tightly regulated process involving the transition of mesenchymal stem cells into mature osteoblasts [43, 44]. Studies have demonstrated that VDR activation enhances the expression of key osteogenic markers, such as runt-related transcription factor 2 (Runx2), osterix (Osx), and alkaline phosphatase (ALP) [45–47]. These factors are essential for osteoblast commitment and maturation, as well as the subsequent synthesis and mineralization of the bone matrix [48].
Beyond its role in osteoblast differentiation, VDR exerts profound effects on osteoblast function and mineralization [49]. VDR activation stimulates the production of various extracellular matrix proteins, including type I collagen, osteopontin, and osteocalcin, which are essential for bone formation and maintenance of bone strength [43, 44]. Moreover, VDR activation modulates the balance between osteoblast-mediated bone formation and osteoclast-mediated bone resorption by influencing the expression of receptor activator of nuclear factor kappa-B ligand (RANKL) and osteoprotegerin (OPG) [50–52]. RANKL is a key osteoclastogenic factor, while OPG acts as a decoy receptor, inhibiting RANKL and preventing osteoclast activation [50–52]. VDR activation in osteoblasts leads to decreased RANKL expression and increased OPG expression, thereby suppressing osteoclastogenesis and maintaining bone homeostasis [50–52].
VDR regulation of osteoclast function and bone resorption
Osteoclasts, the cells responsible for bone resorption, play a crucial role in maintaining bone homeostasis through the removal of old or damaged bone tissue [53]. Emerging evidence suggests that VDR is involved in the regulation of osteoclast differentiation and activation. Studies have demonstrated that VDR activation inhibits osteoclast differentiation by suppressing the expression of key osteoclastogenic factors such as RANKL and macrophage colony-stimulating factor (M-CSF) [54]. VDR activation reduces the production of RANKL and M-CSF, thereby inhibiting osteoclast formation and subsequent bone resorption [54].
In addition to its effects on osteoclast differentiation, VDR has been shown to regulate the function of mature osteoclasts and the process of bone resorption [54, 55]. VDR activation has been reported to suppress the expression of genes involved in osteoclast activity, such as cathepsin K, matrix metalloproteinase-9 (MMP-9), and tartrate-resistant acid phosphatase (TRAP) [56]. Cathepsin K is a lysosomal protease involved in the degradation of the organic matrix of bone, while MMP-9 participates in the breakdown of the collagenous matrix [56]. TRAP is an enzyme associated with osteoclast function and is used as a marker for osteoclast activity [56]. By downregulating the expression of these genes, VDR activation reduces the resorptive capacity of osteoclasts [56]. By increasing the OPG-to-RANKL ratio and Wnt/β-catenin signaling, VDR activation helps maintain a balance between bone formation and resorption [32].
Genetic variations in the VDR gene and osteoporosis
Polymorphisms in the VDR gene and osteoporosis susceptibility
The human VDR gene is located on the short arm of chromosome 12q13.1 [57]. This gene is more than 100 kb in length and consists of 6 untranslated exons (exons 1a–1f) and 8 protein-coding exons (exons 2–9) [58]. More than 60 polymorphisms of the VDR gene have been reported, ranging from sites in the promoter, exons, and introns to the 3’-untranslated region (UTR) [59]. Genetic variations in the VDR gene have been extensively studied in relation to osteoporosis susceptibility. Several single nucleotide polymorphisms (SNPs) have been identified in the VDR gene, and these variations can influence VDR function and subsequent effects on bone health. Commonly studied VDR SNPs include FokI (rs2228570), ApaI (rs7975232), BsmI (rs1544410), and TaqI (rs731236). Of these polymorphisms, ApaI and BsmI are located in the intron between exon 8 and exon 9, FokI is present in exon 2, and TaqI is located in exon 9 (Fig. 4B) [60].
Extensive research has been carried out globally on the relationship between VDR gene polymorphisms, particularly ApaI, TaqI, and BsmI, and osteoporosis (Table 1) [61–75]. Although there are variations in study results, a significant association between these polymorphisms and osteoporosis risk has been predominantly reported (Table 1). Notably, studies conducted in various ethnic populations including Saudis, British, and Chinese revealed significant associations between these polymorphisms and BMD or osteoporosis risk, despite some discrepancies (Table 1) [61–75].
Table 1.
Association of different VDR polymorphisms with osteoporosis risks
| Polymorphism | Osteoporosis Risk | Ethnicity | References |
|---|---|---|---|
|
ApaI TaqI |
Significantly associated with osteoporosis incidence risk | Saudi, White British males, Postmenopausal Chinese women, Korea Saudi Arabia | [61, 62, 66, 67, 69] |
|
ApaI TaqI |
No correlation with BMD | Austria, Spanish women aged over 60 | [64, 71] |
| BsmI | Significant association with osteoporosis susceptibility | Caucasians, Asian | [65, 70] |
| BsmI | No statistical association with osteoporosis risk and BMD | Postmenopausal Spanish women undergoing osteoporosis treatment, Han Chinese | [71, 74] |
| FokI | Potential biomarker for osteoporosis development | Postmenopausal Thai, Italian postmenopausal women, Chinese, Caucasian | [63, 68, 72, 75] |
| FokI | Association with osteoporosis risk in Asians, not in Caucasians | Meta-analysis | [73] |
| rs11568820 | Association with risk of fracture and osteoporosis | Young, healthy, postmenopausal Spanish women | [76] |
| rs11568820 | No significant association with osteoporosis risk | Meta-analysis | [12] |
|
p.Gly14Ala p.His305Gln |
Association with osteoporosis risk | Postmenopausal Chinese women | [77] |
In addition to the mentioned polymorphisms, others like rs11568820, and genetic variants p.Gly14Ala and p.His305Gln have been identified as potential osteoporosis predictors in certain populations (Table 1) [12, 76, 77]. Nevertheless, their association with osteoporosis risk remains inconclusive, as results varied among different studies.
Influence of VDR polymorphisms on BMD
BMD is a key indicator of bone health and osteoporosis risk [2]. Multiple studies have reported that the FokI, BsmI, TaqI, and ApaI polymorphisms significantly impact BMD in different populations, thereby influencing osteoporosis risk. However, these findings have been inconsistent, with some studies revealing significant associations between these polymorphisms and BMD, while others show no such relationship [61–75]. This disparity has been attributed to factors such as ethnic diversity and genetic heterogeneity.
Recent advances in genomic research have further shed light on how VDR polymorphisms might interact with environmental factors to influence BMD. For instance, the impact of VDR polymorphisms on BMD may be modulated by VD levels, dietary calcium intake, and sun exposure [78, 79]. Several studies have reported that the association between VDR polymorphisms and BMD becomes more pronounced under conditions of low calcium intake or VD deficiency [80, 81]. Additionally, research has started to unravel the mechanisms through which these polymorphisms might influence BMD, with data suggesting that they can affect the efficiency of VD signaling, and therefore the regulation of calcium and phosphate homeostasis [79–81]. However, further research is needed to fully understand the complex interactions between VDR polymorphisms, BMD, and osteoporosis risk.
Interplay between VDR and other key players in bone metabolism
VDR and parathyroid hormone
The interplay between VDR and parathyroid hormone (PTH) is crucial for maintaining calcium homeostasis and bone health [82]. PTH plays a central role in regulating serum calcium levels, while VD and its receptor are involved in calcium absorption and utilization in the body [82]. The VDR is expressed in the parathyroid glands, where it modulates the production and secretion of PTH [83]. PTH acts on target tissues, such as the kidneys and bones, to increase calcium levels [83]. Through its interaction with the VDR, PTH regulates the expression of genes involved in calcium metabolism and bone remodeling [18, 82, 83].
VDR and calcium-sensing receptor
The calcium-sensing receptor (CaSR) is another key player in calcium homeostasis [84]. It is expressed in various tissues, including the parathyroid glands, kidneys, and bones [84]. The CaSR detects changes in extracellular calcium levels and regulates PTH secretion accordingly [85]. The VDR and CaSR have a complex interplay in maintaining calcium balance. VD, acting through the VDR, stimulates intestinal calcium absorption, which indirectly affects the activity of the CaSR [84, 85]. The CaSR, in turn, modulates PTH secretion and calcium reabsorption in the kidneys [84, 85]. Both the VDR and CaSR are involved in feedback mechanisms that regulate PTH production and calcium levels in the body [84, 85].
VDR and estrogen receptor
Estrogen plays a critical role in bone health, and its deficiency contributes to the development of osteoporosis [86]. The estrogen receptor (ER) and VDR have interactions that influence bone metabolism [87, 88]. Estrogen stimulates calcium absorption in the intestines and inhibits bone resorption by osteoclasts [89]. Estrogen and VD signaling pathways can cross-regulate each other, highlighting the interconnectedness between these receptors in maintaining bone health.
Understanding the interplay between the VDR, PTH, CaSR, and ER is crucial for unraveling the complex mechanisms involved in bone metabolism and calcium homeostasis. Dysregulation or disruption of these interactions can lead to imbalances in bone remodeling, impaired calcium absorption, and increased osteoporosis risk. Further research is needed to elucidate the intricate molecular pathways and regulatory mechanisms underlying the interplay between the VDR and other key players in bone metabolism. This knowledge may open new avenues for targeted therapeutic interventions for bone-related disorders, including osteoporosis.
Therapeutic implications of targeting the VD/VDR signaling
VD supplementation in osteoporosis
VD supplementation is a widely used therapeutic approach in the management of osteoporosis [90, 91]. As VD is essential for calcium absorption and bone mineralization, inadequate levels can contribute to bone loss and increased fracture risk [90, 91]. Supplementation with VD aims to optimize serum VD levels and enhance calcium absorption, thereby improving bone health [90, 91].
Clinical studies have shown that VD supplementation, especially when combined with calcium, can reduce the risk of fractures in osteoporotic individuals [90, 91]. The optimal dosage and duration of VD supplementation vary depending on individual factors such as age, sunlight exposure, and baseline VD levels [90, 91]. Regular monitoring of serum VD levels is recommended to ensure adequate supplementation [90, 91].
Selective VDR modulators
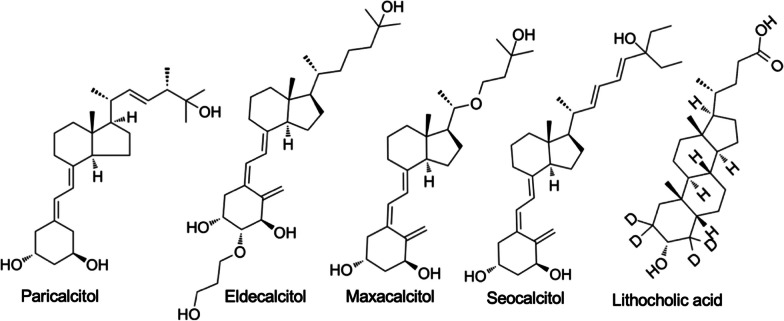
Selective vitamin D receptor modulators (VDRMs) (e.g., paricalcitol, eldecalcitol, maxacalcitol, seocalcitol, and lithocholic acid, Fig. 5) are a class of drugs that target the VDR pathway [92, 93]. These compounds interact with the VDR and selectively modulate its activity, offering the potential to enhance bone health without the side effects associated with systemic VD [92, 93].
Fig. 5.
Chemical structures of VDRMs. Chemical structures of dfferent VDRMs, such as paricalcitol, eldecalcitol, maxacalcitol, seocalcitol, and lithocholic acid, are shown
VDRMs have shown promise in preclinical and clinical studies for their ability to promote bone formation, reduce bone resorption, and improve BMD [94, 95]. These compounds exhibit tissue-selective effects, acting specifically on bone and reducing the risk of hypercalcemia and other adverse effects associated with high-dose VD supplementation [94–96]. However, further research is needed to evaluate their long-term efficacy, safety, and potential benefits compared to traditional VD supplementation.
Combination therapies targeting VDR pathway
Combination therapies that target the VDR pathway have gained attention as potential approaches to optimize bone health and reduce fracture risk in osteoporotic individuals. These therapies involve combining VD supplementation or VDRMs with other agents that enhance bone formation or inhibit bone resorption. For example, combining vitamin K, VD supplementation with calcium and bisphosphonates, which inhibit bone resorption, has been shown to have synergistic effects in improving BMD and reducing fracture risk [97, 98]. Other agents, such as denosumab (a monoclonal antibody targeting bone resorption) [99], teriparatide (a parathyroid hormone analog promoting bone formation) [100], and selective estrogen receptor modulators (SERMs) [101], can also be used in combination with VD-based therapies to optimize bone health.
The rationale behind combination therapies is to target multiple aspects of bone metabolism, including calcium absorption, bone formation, and bone resorption, to achieve a comprehensive and synergistic effect on bone health. However, further research is needed to determine the optimal combinations, dosages, and treatment durations, as well as to assess the long-term safety and efficacy of these approaches.
Discrepancies in findings: VD supplementation and fracture risk reduction
In recent years, randomized controlled trials in the general population examining the effects of supplemental VD on fracture outcomes have yielded inconsistent results. Some studies have demonstrated benefits, whereas others showed no significant effects [102–107]. Several factors might account for these inconsistent findings, including the use of bolus dosing, limited sample sizes in some studies, co-administration of calcium, and baseline VD levels [102–107]. A critical aspect yet to be fully addressed is the potential variability in VDR functionality among the participants. To more comprehensively understand the correlation between VD supplementation and fractures, forthcoming experimental designs should address certain limitations. These encompass the heterogeneity of study populations, ambiguities related to the optimal blood concentration of VD, the unavailability of data on participants’ baseline VD levels, and the absence of insights into genetic variations in the VDR gene.
Conclusion and future perspectives
In this review, we have explored the role of the VD/VDR system in the pathogenesis of osteoporosis. The VDR is a key regulator of bone metabolism, mediating the effects of VD on calcium absorption, bone mineralization, and bone remodeling. Genetic variations in the VDR gene have been associated with osteoporosis susceptibility and BMD. Furthermore, the VDR interacts with other key players in bone metabolism, including the parathyroid hormone, the calcium-sensing receptor, and the estrogen receptor, contributing to the complex regulation of bone homeostasis.
Therapeutically, targeting the VDR pathway has shown promise in the management of osteoporosis [90–93]. VD supplementation, particularly in combination with calcium, has been widely used to improve bone health and reduce fracture risk. Selective VDR modulators offer a more targeted approach, selectively modulating VDR activity to enhance bone formation and reduce bone resorption [90–93]. Combination therapies that combine VDR-targeting agents with other bone-modifying drugs have also shown potential synergistic effects [97, 98].
Despite significant advancements in our understanding of the VDR and its role in osteoporosis, several areas warrant further investigation. First, more studies are needed to elucidate the mechanisms underlying the interplay between the VDR and other key players in bone metabolism, such as PTH, CaSR, and ER. Understanding these interactions at a molecular level may uncover novel therapeutic targets for osteoporosis. Furthermore, the influence of genetic variations in the VDR gene on osteoporosis susceptibility and treatment response requires further exploration. Large-scale genetic studies, including genome-wide association studies and functional analyses, can provide insights into the specific VDR polymorphisms and their implications for personalized medicine in osteoporosis. In terms of therapeutics, future research should focus on optimizing the use of VDR-targeting agents, including selective VDR modulators and combination therapies. This includes identifying the most effective combinations, determining optimal dosages and treatment durations, and assessing long-term safety and efficacy in diverse patient populations. Additionally, the potential role of VDR-based therapies in other bone-related conditions, such as osteomalacia and secondary osteoporosis, should be explored. Lastly, the integration of emerging technologies, such as omics approaches and advanced imaging techniques, can enhance our understanding of the VDR pathway and its interactions with other molecular networks in bone metabolism. These technologies can provide a comprehensive and multidimensional view of the molecular mechanisms underlying osteoporosis, paving the way for personalized and precision medicine approaches.
In conclusion, the VDR pathway plays a critical role in the pathogenesis of osteoporosis, and targeting this pathway holds significant therapeutic potential. Continued research efforts are needed to unravel the complexities of the VDR network, explore genetic influences, optimize therapeutic strategies, and leverage emerging technologies. These advancements will contribute to the development of novel treatments and improve outcomes for individuals with osteoporosis in the future.
Acknowledgements
We would like to acknowledge the contributions of all the researchers, scientists, and clinicians whose work has significantly contributed to our understanding of VDR and its role in osteoporosis. Their dedication and commitment to advancing knowledge in this field are greatly appreciated.
Author contribution
GL, KL, and YT conceived the structures of the article and revised the manuscript. YL searched the publications and wrote the manuscript. PZ, BJ, KL, LZ, and HW revised the manuscript.
Funding
This work is supported by the grant of the High-end Foreign Expert Program of the Ministry of Science and Technology of China (G2021003005L) and the Hebei Provincial Science and Technology Plan Project (223777118D).
Availability of data and materials
The data underlying the present study are available on request (corresponding author).
Declarations
Ethics approval and consent to participate
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Yansheng Tian, Email: tianyansheng2008@163.com.
Kun Li, Email: 285501202@qq.com.
Guoqi Liu, Email: guoqi.liu22@outlook.com.
References
- 1.Foger-Samwald U, Dovjak P, Azizi-Semrad U, Kerschan-Schindl K, Pietschmann P. Osteoporosis: pathophysiology and therapeutic options. EXCLI J. 2020;19:1017–1037. doi: 10.17179/excli2020-2591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sozen T, Ozisik L, Basaran NC. An overview and management of osteoporosis. Eur J Rheumatol. 2017;4(1):46–56. doi: 10.5152/eurjrheum.2016.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Salari N, Ghasemi H, Mohammadi L, Behzadi MH, Rabieenia E, Shohaimi S, Mohammadi M. The global prevalence of osteoporosis in the world: a comprehensive systematic review and meta-analysis. J Orthop Surg Res. 2021;16(1):609. doi: 10.1186/s13018-021-02772-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pouresmaeili F, Kamalidehghan B, Kamarehei M, Goh YM. A comprehensive overview on osteoporosis and its risk factors. Ther Clin Risk Manag. 2018;14:2029–2049. doi: 10.2147/TCRM.S138000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Raterman HG, Bultink IE, Lems WF. Osteoporosis in patients with rheumatoid arthritis: an update in epidemiology, pathogenesis, and fracture prevention. Expert Opin Pharmacother. 2020;21(14):1725–1737. doi: 10.1080/14656566.2020.1787381. [DOI] [PubMed] [Google Scholar]
- 6.De Martinis M, Allegra A, Sirufo MM, Tonacci A, Pioggia G, Raggiunti M, Ginaldi L, Gangemi S. Vitamin D deficiency, osteoporosis and effect on autoimmune diseases and hematopoiesis: a review. Int J Mol Sci. 2021;22(16):8855. doi: 10.3390/ijms22168855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dominguez LJ, Farruggia M, Veronese N, Barbagallo M. Vitamin D sources, metabolism, and deficiency: available compounds and guidelines for its treatment. Metabolites. 2021;11(4):255. doi: 10.3390/metabo11040255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mungai LNW, Mohammed Z, Maina M, Anjumanara O. Vitamin D review: the low hanging fruit for human health. J Nutr Metab. 2021;2021:6335681. doi: 10.1155/2021/6335681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Carlberg C. Vitamin D and its target genes. Nutrients. 2022;14(7):1354. doi: 10.3390/nu14071354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bouillon R, Marcocci C, Carmeliet G, Bikle D, White JH, Dawson-Hughes B, Lips P, Munns CF, Lazaretti-Castro M, Giustina A, Bilezikian J. Skeletal and extraskeletal actions of vitamin D: current evidence and outstanding questions. Endocr Rev. 2019;40(4):1109–1151. doi: 10.1210/er.2018-00126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Christakos S, Dhawan P, Verstuyf A, Verlinden L, Carmeliet G. Vitamin D: metabolism, molecular mechanism of action, and pleiotropic effects. Physiol Rev. 2016;96(1):365–408. doi: 10.1152/physrev.00014.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mu YY, Liu B, Chen B, Zhu WF, Ye XH, Li HZ, He XF. Evaluation of association studies and an updated meta-analysis of VDR polymorphisms in osteoporotic fracture risk. Front Genet. 2021;12:791368. doi: 10.3389/fgene.2021.791368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bahrami A, Sadeghnia HR, Tabatabaeizadeh SA, Bahrami-Taghanaki H, Behboodi N, Esmaeili H, Esmaeili H, Ferns GA, Mobarhan MG, Avan A. Genetic and epigenetic factors influencing vitamin D status. J Cell Physiol. 2018;233(5):4033–4043. doi: 10.1002/jcp.26216. [DOI] [PubMed] [Google Scholar]
- 14.Lee SM, Meyer MB, Benkusky NA, O'Brien CA, Pike JW. Mechanisms of enhancer-mediated hormonal control of vitamin D receptor gene expression in target cells. J Biol Chem. 2015;290(51):30573–30586. doi: 10.1074/jbc.M115.693614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Al Mheid I, Patel RS, Tangpricha V, Quyyumi AA. Vitamin D and cardiovascular disease: Is the evidence solid? Eur Heart J. 2013;34(48):3691–3698. doi: 10.1093/eurheartj/eht166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jeon SM, Shin EA. Exploring vitamin D metabolism and function in cancer. Exp Mol Med. 2018;50(4):1–14. doi: 10.1038/s12276-018-0038-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bikle DD. Vitamin D metabolism, mechanism of action, and clinical applications. Chem Biol. 2014;21(3):319–329. doi: 10.1016/j.chembiol.2013.12.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Haussler MR, Haussler CA, Whitfield GK, Hsieh JC, Thompson PD, Barthel TK, Bartik L, Egan JB, Wu Y, Kubicek JL, Lowmiller CL, Moffet EW, Forster RE, Jurutka PW. The nuclear vitamin D receptor controls the expression of genes encoding factors which feed the “Fountain of Youth” to mediate healthful aging. J Steroid Biochem Mol Biol. 2010;121(1–2):88–97. doi: 10.1016/j.jsbmb.2010.03.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pike JW, Meyer MB. The vitamin D receptor: new paradigms for the regulation of gene expression by 1,25-dihydroxyvitamin D3. Rheum Dis Clin N Am. 2012;38(1):13–27. doi: 10.1016/j.rdc.2012.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wan LY, Zhang YQ, Chen MD, Liu CB, Wu JF. Relationship of structure and function of DNA-binding domain in vitamin D receptor. Molecules. 2015;20(7):12389–12399. doi: 10.3390/molecules200712389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Barminko J, Reinholt BM, Emmanuelli A, Lejeune AN, Baron MH. Activation of the vitamin D receptor transcription factor stimulates the growth of definitive erythroid progenitors. Blood Adv. 2018;2(11):1207–1219. doi: 10.1182/bloodadvances.2018017533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rochel N. Vitamin D and its receptor from a structural perspective. Nutrients. 2022;14(14):2847. doi: 10.3390/nu14142847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Haussler MR, Livingston S, Sabir ZL, Haussler CA, Jurutka PW. Vitamin D receptor mediates a myriad of biological actions dependent on its 1,25-dihydroxyvitamin D ligand: distinct regulatory themes revealed by induction of klotho and fibroblast growth factor-23. JBMR Plus. 2021;5(1):e10432. doi: 10.1002/jbm4.10432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pike JW, Christakos S. Biology and mechanisms of action of the vitamin D hormone. Endocrinol Metab Clin N Am. 2017;46(4):815–843. doi: 10.1016/j.ecl.2017.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pike JW, Meyer MB, Lee SM, Onal M, Benkusky NA. The vitamin D receptor: contemporary genomic approaches reveal new basic and translational insights. J Clin Invest. 2017;127(4):1146–1154. doi: 10.1172/JCI88887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Costa PLF, Franca MM, Katayama ML, Carneiro ET, Martin RM, Folgueira MAK, Latronico AC, Ferraz-de-Souza B. Transcriptomic response to 1,25-dihydroxy vitamin D in human fibroblasts with or without a functional vitamin D receptor (VDR): novel target genes and insights into VDR basal transcriptional activity. Cells. 2019;8(4):318. doi: 10.3390/cells8040318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Donati S, Palmini G, Aurilia C, Falsetti I, Miglietta F, Iantomasi T, Brandi ML. Rapid nontranscriptional effects of calcifediol and calcitriol. Nutrients. 2022;14(6):1291. doi: 10.3390/nu14061291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zmijewski MA. Nongenomic activities of vitamin D. Nutrients. 2022;14(23):5104. doi: 10.3390/nu14235104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Irazoqui AP, Boland RL, Buitrago CG. Actions of 1,25(OH)2-vitamin D3 on the cellular cycle depend on VDR and p38 MAPK in skeletal muscle cells. J Mol Endocrinol. 2014;53(3):331–343. doi: 10.1530/JME-14-0102. [DOI] [PubMed] [Google Scholar]
- 30.Zenata O, Vrzal R. Fine tuning of vitamin D receptor (VDR) activity by post-transcriptional and post-translational modifications. Oncotarget. 2017;8(21):35390–35402. doi: 10.18632/oncotarget.15697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.van Driel M, Pols HA, van Leeuwen JP. Osteoblast differentiation and control by vitamin D and vitamin D metabolites. Curr Pharm Des. 2004;10(21):2535–2555. doi: 10.2174/1381612043383818. [DOI] [PubMed] [Google Scholar]
- 32.Xu D, Gao HJ, Lu CY, Tian HM, Yu XJ. Vitamin D inhibits bone loss in mice with thyrotoxicosis by activating the OPG/RANKL and Wnt/beta-catenin signaling pathways. Front Endocrinol (Lausanne) 2022;13:1066089. doi: 10.3389/fendo.2022.1066089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Haussler MR, Jurutka PW, Mizwicki M, Norman AW. Vitamin D receptor (VDR)-mediated actions of 1alpha,25(OH)(2)vitamin D(3): genomic and non-genomic mechanisms. Best Pract Res Clin Endocrinol Metab. 2011;25(4):543–559. doi: 10.1016/j.beem.2011.05.010. [DOI] [PubMed] [Google Scholar]
- 34.Chen Y, Zhang J, Ge X, Du J, Deb DK, Li YC. Vitamin D receptor inhibits nuclear factor kappaB activation by interacting with IkappaB kinase beta protein. J Biol Chem. 2013;288(27):19450–19458. doi: 10.1074/jbc.M113.467670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chen Z, Huang D, Yongyut P, Li G, Esteban MA, Jintasataporn O, Deng J, Zhang W, Ai Q, Mai K, Zhang Y. Vitamin D(3) deficiency induced intestinal inflammatory response of turbot through nuclear factor-kappaB/inflammasome pathway, accompanied by the mutually exclusive apoptosis and autophagy. Front Immunol. 2022;13:986593. doi: 10.3389/fimmu.2022.986593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.He L, Liu T, Shi Y, Tian F, Hu H, Deb DK, Chen Y, Bissonnette M, Li YC. Gut epithelial vitamin D receptor regulates microbiota-dependent mucosal inflammation by suppressing intestinal epithelial cell apoptosis. Endocrinology. 2018;159(2):967–979. doi: 10.1210/en.2017-00748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Szeto FL, Sun J, Kong J, Duan Y, Liao A, Madara JL, Li YC. Involvement of the vitamin D receptor in the regulation of NF-kappaB activity in fibroblasts. J Steroid Biochem Mol Biol. 2007;103(3–5):563–566. doi: 10.1016/j.jsbmb.2006.12.092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ge X, Wang L, Li M, Xu N, Yu F, Yang F, Li R, Zhang F, Zhao B, Du J. Vitamin D/VDR signaling inhibits LPS-induced IFNgamma and IL-1beta in Oral epithelia by regulating hypoxia-inducible factor-1alpha signaling pathway. Cell Commun Signal. 2019;17(1):18. doi: 10.1186/s12964-019-0331-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kongsbak M, Levring TB, Geisler C, von Essen MR. The vitamin d receptor and T cell function. Front Immunol. 2013;4:148. doi: 10.3389/fimmu.2013.00148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fernandez Lahore G, Raposo B, Lagerquist M, Ohlsson C, Sabatier P, Xu B, Aoun M, James J, Cai X, Zubarev RA, Nandakumar KS, Holmdahl R. Vitamin D3 receptor polymorphisms regulate T cells and T cell-dependent inflammatory diseases. Proc Natl Acad Sci U S A. 2020;117(40):24986–24997. doi: 10.1073/pnas.2001966117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Saccone D, Asani F, Bornman L. Regulation of the vitamin D receptor gene by environment, genetics and epigenetics. Gene. 2015;561(2):171–180. doi: 10.1016/j.gene.2015.02.024. [DOI] [PubMed] [Google Scholar]
- 42.Kim JM, Lin C, Stavre Z, Greenblatt MB, Shim JH. Osteoblast–osteoclast communication and bone homeostasis. Cells. 2020;9(9):2073. doi: 10.3390/cells9092073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bikle DD. Vitamin D and bone. Curr Osteoporos Rep. 2012;10(2):151–159. doi: 10.1007/s11914-012-0098-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.van de Peppel J, van Leeuwen JP. Vitamin D and gene networks in human osteoblasts. Front Physiol. 2014;5:137. doi: 10.3389/fphys.2014.00137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Atkins GJ, Anderson PH, Findlay DM, Welldon KJ, Vincent C, Zannettino AC, O'Loughlin PD, Morris HA. Metabolism of vitamin D3 in human osteoblasts: evidence for autocrine and paracrine activities of 1 alpha,25-dihydroxyvitamin D3. Bone. 2007;40(6):1517–1528. doi: 10.1016/j.bone.2007.02.024. [DOI] [PubMed] [Google Scholar]
- 46.Kogawa M, Findlay DM, Anderson PH, Ormsby R, Vincent C, Morris HA, Atkins GJ. Osteoclastic metabolism of 25(OH)-vitamin D3: a potential mechanism for optimization of bone resorption. Endocrinology. 2010;151(10):4613–4625. doi: 10.1210/en.2010-0334. [DOI] [PubMed] [Google Scholar]
- 47.Kitazawa S, Kajimoto K, Kondo T, Kitazawa R. Vitamin D3 supports osteoclastogenesis via functional vitamin D response element of human RANKL gene promoter. J Cell Biochem. 2003;89(4):771–777. doi: 10.1002/jcb.10567. [DOI] [PubMed] [Google Scholar]
- 48.van Driel M, Koedam M, Buurman CJ, Roelse M, Weyts F, Chiba H, Uitterlinden AG, Pols HA, van Leeuwen JP. Evidence that both 1alpha,25-dihydroxyvitamin D3 and 24-hydroxylated D3 enhance human osteoblast differentiation and mineralization. J Cell Biochem. 2006;99(3):922–935. doi: 10.1002/jcb.20875. [DOI] [PubMed] [Google Scholar]
- 49.van Leeuwen JP, van Driel M, van den Bemd GJ, Pols HA. Vitamin D control of osteoblast function and bone extracellular matrix mineralization. Crit Rev Eukaryot Gene Expr. 2001;11(1–3):199–226. [PubMed] [Google Scholar]
- 50.Nakamichi Y, Udagawa N, Horibe K, Mizoguchi T, Yamamoto Y, Nakamura T, Hosoya A, Kato S, Suda T, Takahashi N. VDR in osteoblast-lineage cells primarily mediates vitamin D treatment-induced increase in bone mass by suppressing bone resorption. J Bone Miner Res. 2017;32(6):1297–1308. doi: 10.1002/jbmr.3096. [DOI] [PubMed] [Google Scholar]
- 51.Shymanskyi I, Lisakovska O, Mazanova A, Labudzynskyi D, Veliky M. Vitamin D(3) modulates impaired crosstalk between RANK and glucocorticoid receptor signaling in bone marrow cells after chronic prednisolone administration. Front Endocrinol (Lausanne) 2018;9:303. doi: 10.3389/fendo.2018.00303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Yamamoto Y, Yoshizawa T, Fukuda T, Shirode-Fukuda Y, Yu T, Sekine K, Sato T, Kawano H, Aihara K, Nakamichi Y, Watanabe T, Shindo M, Inoue K, Inoue E, Tsuji N, Hoshino M, Karsenty G, Metzger D, Chambon P, Kato S, Imai Y. Vitamin D receptor in osteoblasts is a negative regulator of bone mass control. Endocrinology. 2013;154(3):1008–1020. doi: 10.1210/en.2012-1542. [DOI] [PubMed] [Google Scholar]
- 53.Florencio-Silva R, Sasso GR, Sasso-Cerri E, Simoes MJ, Cerri PS. Biology of bone tissue: structure, function, and factors that influence bone cells. Biomed Res Int. 2015;2015:421746. doi: 10.1155/2015/421746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zarei A, Morovat A, Javaid K, Brown CP. Vitamin D receptor expression in human bone tissue and dose-dependent activation in resorbing osteoclasts. Bone Res. 2016;4:16030. doi: 10.1038/boneres.2016.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Menaa C, Barsony J, Reddy SV, Cornish J, Cundy T, Roodman GD. 1,25-Dihydroxyvitamin D3 hypersensitivity of osteoclast precursors from patients with Paget’s disease. J Bone Miner Res. 2000;15(2):228–236. doi: 10.1359/jbmr.2000.15.2.228. [DOI] [PubMed] [Google Scholar]
- 56.Deng J, Yang Y, He J, Xie Z, Luo F, Xu J, et al. Vitamin D receptor activated by vitamin D administration alleviates Mycobacterium tuberculosis-induced bone destruction by inhibiting NFkappaB-mediated aberrant osteoclastogenesis. FASEB J. 2021;35(6):e21543. doi: 10.1096/fj.202100135R. [DOI] [PubMed] [Google Scholar]
- 57.Taymans SE, Pack S, Pak E, Orban Z, Barsony J, Zhuang Z, et al. The human vitamin D receptor gene (VDR) is localized to region 12cen-q12 by fluorescent in situ hybridization and radiation hybrid mapping: genetic and physical VDR map. J Bone Miner Res. 1999;14(7):1163–1166. doi: 10.1359/jbmr.1999.14.7.1163. [DOI] [PubMed] [Google Scholar]
- 58.Penna-Martinez M, Badenhoop K. Inherited variation in vitamin D genes and type 1 diabetes predisposition. Genes (Basel) 2017;8(4):125. doi: 10.3390/genes8040125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kostner K, Denzer N, Koreng M, Reichrath S, Graber S, Klein R, Tilgen W, Vogt T, Reichrath J. Association of genetic variants of the vitamin D receptor (VDR) with cutaneous squamous cell carcinomas (SCC) and basal cell carcinomas (BCC): a pilot study in a German population. Anticancer Res. 2012;32(1):327–333. [PubMed] [Google Scholar]
- 60.Santoro D, Lucisano S, Gagliostro G, Alibrandi A, Benvenga S, Ientile R, Bellinghieri G, Buemi M, Caccamo D. Vitamin D receptor polymorphism in chronic kidney disease patients with complicated cardiovascular disease. J Ren Nutr. 2015;25(2):187–193. doi: 10.1053/j.jrn.2014.10.022. [DOI] [PubMed] [Google Scholar]
- 61.Ansari MGA, Mohammed AK, Wani KA, Hussain SD, Alnaami AM, Abdi S, Aljohani NJ, Al-Daghri NM. Vitamin D receptor gene variants susceptible to osteoporosis in arab post-menopausal women. Curr Issues Mol Biol. 2021;43(3):1325–1334. doi: 10.3390/cimb43030094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Banjabi AA, Al-Ghafari AB, Kumosani TA, Kannan K, Fallatah SM. Genetic influence of vitamin D receptor gene polymorphisms on osteoporosis risk. Int J Health Sci (Qassim) 2020;14(4):22–28. [PMC free article] [PubMed] [Google Scholar]
- 63.Becherini L, Gennari L, Masi L, Mansani R, Massart F, Morelli A, Falchetti A, Gonnelli S, Fiorelli G, Tanini A, Brandi ML. Evidence of a linkage disequilibrium between polymorphisms in the human estrogen receptor alpha gene and their relationship to bone mass variation in postmenopausal Italian women. Hum Mol Genet. 2000;9(13):2043–2050. doi: 10.1093/hmg/9.13.2043. [DOI] [PubMed] [Google Scholar]
- 64.Boschitsch E, Suk EK, Mayr WR, Lang T, Schwartz WM, Panzer S. Genotypes of the vitamin-D-receptor gene and bone mineral density in Caucasoid postmenopausal females. Maturitas. 1996;24(1–2):91–96. doi: 10.1016/0378-5122(95)01023-8. [DOI] [PubMed] [Google Scholar]
- 65.Jiang LL, Zhang C, Zhang Y, Ma F, Guan Y. Associations between polymorphisms in VDR gene and the risk of osteoporosis: a meta-analysis. Arch Physiol Biochem. 2022;128(6):1637–1644. doi: 10.1080/13813455.2020.1787457. [DOI] [PubMed] [Google Scholar]
- 66.Kim SW, Lee JM, Ha JH, Kang HH, Rhee CK, Kim JW, Moon HS, Baek KH, Lee SH. Association between vitamin D receptor polymorphisms and osteoporosis in patients with COPD. Int J Chron Obstr Pulm Dis. 2015;10:1809–1817. doi: 10.2147/COPD.S91576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kow M, Akam E, Singh P, Singh M, Cox N, Bhatti JS, Tuck SP, Francis RM, Datta H, Mastana S. Vitamin D receptor (VDR) gene polymorphism and osteoporosis risk in White British men. Ann Hum Biol. 2019;46(5):430–433. doi: 10.1080/03014460.2019.1659851. [DOI] [PubMed] [Google Scholar]
- 68.Lau EM, Lam V, Li M, Ho K, Woo J. Vitamin D receptor start codon polymorphism (Fok I) and bone mineral density in Chinese men and women. Osteoporos Int. 2002;13(3):218–221. doi: 10.1007/s001980200017. [DOI] [PubMed] [Google Scholar]
- 69.Li Y, Xi B, Li K, Wang C. Association between vitamin D receptor gene polymorphisms and bone mineral density in Chinese women. Mol Biol Rep. 2012;39(5):5709–5717. doi: 10.1007/s11033-011-1380-3. [DOI] [PubMed] [Google Scholar]
- 70.Liao JL, Qin Q, Zhou YS, Ma RP, Zhou HC, Gu MR, Feng YP, Wang BY, Yang L. Vitamin D receptor Bsm I polymorphism and osteoporosis risk in postmenopausal women: a meta-analysis from 42 studies. Genes Nutr. 2020;15(1):20. doi: 10.1186/s12263-020-00679-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Pedrera-Canal M, Moran JM, Vera V, Roncero-Martin R, Lavado-Garcia JM, Aliaga I, Pedrera-Zamorano JDI. Lack of influence of vitamin D receptor BsmI (rs1544410) polymorphism on the rate of bone loss in a cohort of postmenopausal Spanish women affected by osteoporosis and followed for five years. PLoS ONE. 2015;10(9):e0138606. doi: 10.1371/journal.pone.0138606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Techapatiphandee M, Tammachote N, Tammachote R, Wongkularb A, Yanatatsaneejit P. VDR and TNFSF11 polymorphisms are associated with osteoporosis in Thai patients. Biomed Rep. 2018;9(4):350–356. doi: 10.3892/br.2018.1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Wang S, Ai Z, Song M, Yan P, Li J, Wang S. The association between vitamin D receptor FokI gene polymorphism and osteoporosis in postmenopausal women: a meta-analysis. Climacteric. 2021;24(1):74–79. doi: 10.1080/13697137.2020.1775806. [DOI] [PubMed] [Google Scholar]
- 74.Yu M, Chen GQ, Yu F. Lack of association between vitamin D receptor polymorphisms ApaI (rs7975232) and BsmI (rs1544410) and osteoporosis among the Han Chinese population: A meta-analysis. Kaohsiung J Med Sci. 2016;32(12):599–606. doi: 10.1016/j.kjms.2016.10.002. [DOI] [PubMed] [Google Scholar]
- 75.Zintzaras E, Rodopoulou P, Koukoulis GN. BsmI, TaqI, ApaI and FokI polymorphisms in the vitamin D receptor (VDR) gene and the risk of osteoporosis: a meta-analysis. Dis Markers. 2006;22(5–6):317–326. doi: 10.1155/2006/921694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Casado-Diaz A, Cuenca-Acevedo R, Navarro-Valverde C, Diaz-Molina C, Caballero-Villarraso J, Santiago-Mora R, Dorado G, Quesada-Gomez JM. Vitamin D status and the Cdx-2 polymorphism of the vitamin D receptor gene are determining factors of bone mineral density in young healthy postmenopausal women. J Steroid Biochem Mol Biol. 2013;136:187–189. doi: 10.1016/j.jsbmb.2012.09.026. [DOI] [PubMed] [Google Scholar]
- 77.He W, Liu M, Huang X, Qing Z, Gao W. The influence of vitamin D receptor genetic variants on bone mineral density and osteoporosis in Chinese postmenopausal women. Dis Markers. 2015;2015:760313. doi: 10.1155/2015/760313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ferrari S, Rizzoli R, Manen D, Slosman D, Bonjour JP. Vitamin D receptor gene start codon polymorphisms (FokI) and bone mineral density: interaction with age, dietary calcium, and 3′-end region polymorphisms. J Bone Min Res. 1998;13(6):925–930. doi: 10.1359/jbmr.1998.13.6.925. [DOI] [PubMed] [Google Scholar]
- 79.Uitterlinden AG, Fang Y, Van Meurs JB, Pols HA, Van Leeuwen JP. Genetics and biology of vitamin D receptor polymorphisms. Gene. 2004;338(2):143–156. doi: 10.1016/j.gene.2004.05.014. [DOI] [PubMed] [Google Scholar]
- 80.Al Anouti F, Taha Z, Shamim S, Khalaf K, Al Kaabi L, Alsafar H. An insight into the paradigms of osteoporosis: from genetics to biomechanics. Bone Rep. 2019;11:100216. doi: 10.1016/j.bonr.2019.100216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Morrison NA, Qi JC, Tokita A, Kelly PJ, Crofts L, Nguyen TV, Sambrook PN, Eisman JA. Prediction of bone density from vitamin D receptor alleles. Nature. 1994;367(6460):284–287. doi: 10.1038/367284a0. [DOI] [PubMed] [Google Scholar]
- 82.Veldurthy V, Wei R, Oz L, Dhawan P, Jeon YH, Christakos S. Vitamin D, calcium homeostasis and aging. Bone Res. 2016;4:16041. doi: 10.1038/boneres.2016.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Latic N, Erben RG. Interaction of vitamin D with peptide hormones with emphasis on parathyroid hormone, FGF23, and the renin-angiotensin-aldosterone system. Nutrients. 2022;14(23):5186. doi: 10.3390/nu14235186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Iamartino L, Brandi ML. The calcium-sensing receptor in inflammation: Recent updates. Front Physiol. 2022;13:1059369. doi: 10.3389/fphys.2022.1059369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Goodman WG. Calcium-sensing receptors. Semin Nephrol. 2004;24(1):17–24. doi: 10.1053/j.semnephrol.2003.08.017. [DOI] [PubMed] [Google Scholar]
- 86.Emmanuelle NE, Marie-Cecile V, Florence T, Jean-Francois A, Francoise L, Coralie F, Alexia V. Critical role of estrogens on bone homeostasis in both male and female: from physiology to medical implications. Int J Mol Sci. 2021;22(4):1568. doi: 10.3390/ijms22041568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Brown MA, Haughton MA, Grant SF, Gunnell AS, Henderson NK, Eisman JA. Genetic control of bone density and turnover: role of the collagen 1alpha1, estrogen receptor, and vitamin D receptor genes. J Bone Min Res. 2001;16(4):758–764. doi: 10.1359/jbmr.2001.16.4.758. [DOI] [PubMed] [Google Scholar]
- 88.Long J, Liu P, Zhang Y, Shen H, Liu Y, Dvornyk V, Deng HW. Interaction effects between estrogen receptor alpha gene, vitamin D receptor gene, age, and sex on bone mineral density in Chinese. J Hum Genet. 2003;48(10):514–519. doi: 10.1007/s10038-003-0068-5. [DOI] [PubMed] [Google Scholar]
- 89.Mills EG, Yang L, Nielsen MF, Kassem M, Dhillo WS, Comninos AN. The relationship between bone and reproductive hormones beyond estrogens and androgens. Endocr Rev. 2021;42:691–719. doi: 10.1210/endrev/bnab015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.LeBoff MS, Greenspan SL, Insogna KL, Lewiecki EM, Saag KG, Singer AJ, et al. The clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2022;33(10):2049–2102. doi: 10.1007/s00198-021-05900-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Sunyecz JA. The use of calcium and vitamin D in the management of osteoporosis. Ther Clin Risk Manag. 2008;4(4):827–836. doi: 10.2147/TCRM.S3552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Campbell MJ, Adorini L. The vitamin D receptor as a therapeutic target. Expert Opin Ther Targets. 2006;10(5):735–748. doi: 10.1517/14728222.10.5.735. [DOI] [PubMed] [Google Scholar]
- 93.Lason W, Jantas D, Leskiewicz M, Regulska M, Basta-Kaim A. The vitamin D receptor as a potential target for the treatment of age-related neurodegenerative diseases such as Alzheimer’s and Parkinson’s diseases: a narrative review. Cells. 2023;12(4):660. doi: 10.3390/cells12040660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Appelman-Dijkstra NM, Papapoulos SE. Modulating bone resorption and bone formation in opposite directions in the treatment of postmenopausal osteoporosis. Drugs. 2015;75(10):1049–1058. doi: 10.1007/s40265-015-0417-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Plum LA, DeLuca HF. Vitamin D, disease and therapeutic opportunities. Nat Rev Drug Discov. 2010;9(12):941–955. doi: 10.1038/nrd3318. [DOI] [PubMed] [Google Scholar]
- 96.Noguchi Y, Kawate H, Nomura M, Takayanagi R. Eldecalcitol for the treatment of osteoporosis. Clin Interv Aging. 2013;8:1313–1321. doi: 10.2147/CIA.S49825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Moore AE, Dulnoan D, Voong K, Ayis S, Mangelis A, Gorska R, Harrington DJ, Tang JCY, Fraser WD, Hampson G. The additive effect of vitamin K supplementation and bisphosphonate on fracture risk in post-menopausal osteoporosis: a randomised placebo controlled trial. Arch Osteoporos. 2023;18(1):83. doi: 10.1007/s11657-023-01288-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Golden NH, Abrams SA. Committee on N. Optimizing bone health in children and adolescents. Pediatrics. 2014;134:e122943. doi: 10.1542/peds.2014-2173. [DOI] [PubMed] [Google Scholar]
- 99.Bone HG, Bolognese MA, Yuen CK, Kendler DL, Miller PD, Yang YC, et al. Effects of denosumab treatment and discontinuation on bone mineral density and bone turnover markers in postmenopausal women with low bone mass. J Clin Endocrinol Metab. 2011;96(4):972–980. doi: 10.1210/jc.2010-1502. [DOI] [PubMed] [Google Scholar]
- 100.Gomberg SJ, Wustrack RL, Napoli N, Arnaud CD, Black DM. Teriparatide, vitamin D, and calcium healed bilateral subtrochanteric stress fractures in a postmenopausal woman with a 13-year history of continuous alendronate therapy. J Clin Endocrinol Metab. 2011;96(6):1627–1632. doi: 10.1210/jc.2010-2520. [DOI] [PubMed] [Google Scholar]
- 101.Liu HJ, Kim SA, Shim DJ, Jung JM, Lee EJ. Influence of supplementary vitamin D on bone mineral density when used in combination with selective estrogen receptor modulators. J Menopausal Med. 2019;25(2):94–99. doi: 10.6118/jmm.19193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Shapses SA, Kendler DL, Robson R, Hansen KE, Sherrell RM, Field MP, Woolf E, Berd Y, Mantz AM, Santora AC. Effect of alendronate and vitamin D(3) on fractional calcium absorption in a double-blind, randomized, placebo-controlled trial in postmenopausal osteoporotic women. J Bone Miner Res. 2011;26(8):1836–1844. doi: 10.1002/jbmr.395. [DOI] [PubMed] [Google Scholar]
- 103.Bischoff-Ferrari HA, Vellas B, Rizzoli R, Kressig RW, da Silva JAP, Blauth M, Felson DT, McCloskey EV, Watzl B, Hofbauer LC, Felsenberg D, Willett WC, Dawson-Hughes B, Manson JE, Siebert U, Theiler R, Staehelin HB, DO-HEALTH Research Group Effect of vitamin D supplementation, omega-3 fatty acid supplementation, or a strength-training exercise program on clinical outcomes in older adults: the DO-HEALTH randomized clinical trial. JAMA. 2020;324(18):1855–1868. doi: 10.1001/jama.2020.16909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Sanders KM, Stuart AL, Williamson EJ, Simpson JA, Kotowicz MA, Young D, Nicholson GC. Annual high-dose oral vitamin D and falls and fractures in older women: a randomized controlled trial. JAMA. 2010;303(18):1815–1822. doi: 10.1001/jama.2010.594. [DOI] [PubMed] [Google Scholar]
- 105.Khaw KT, Stewart AW, Waayer D, Lawes CMM, Toop L, Camargo CA, Jr, Scragg R. Effect of monthly high-dose vitamin D supplementation on falls and non-vertebral fractures: secondary and post-hoc outcomes from the randomised, double-blind, placebo-controlled ViDA trial. Lancet Diabetes Endocrinol. 2017;5(6):438–447. doi: 10.1016/S2213-8587(17)30103-1. [DOI] [PubMed] [Google Scholar]
- 106.Jackson RD, LaCroix AZ, Gass M, Wallace RB, Robbins J, Lewis CE, Bassford T, Beresford SA, Black HR, Blanchette P, Bonds DE, Brunner RL, Brzyski RG, Caan B, Cauley JA, Chlebowski RT, Cummings SR, Granek I, Hays J, Heiss G, Hendrix SL, Howard BV, Hsia J, Hubbell FA, Johnson KC, Judd H, Kotchen JM, Kuller LH, Langer RD, Lasser NL, Limacher MC, Ludlam S, Manson JE, Margolis KL, McGowan J, Ockene JK, O'Sullivan MJ, Phillips L, Prentice RL, Sarto GE, Stefanick ML, Van Horn L, Wactawski-Wende J, Whitlock E, Anderson GL, Assaf AR, Barad D, Women’s Health Initiative Investigators Calcium plus vitamin D supplementation and the risk of fractures. N Engl J Med. 2006;354(7):669–683. doi: 10.1056/NEJMoa055218. [DOI] [PubMed] [Google Scholar]
- 107.LeBoff MS, Chou SH, Ratliff KA, Cook NR, Khurana B, Kim E, Cawthon PM, Bauer DC, Black D, Gallagher JC, Lee IM, Buring JE, Manson JE. Supplemental vitamin D and incident fractures in midlife and older adults. N Engl J Med. 2022;387(4):299–309. doi: 10.1056/NEJMoa2202106. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data underlying the present study are available on request (corresponding author).