Key Points
-
•
Macrophages regulate Tf-iron availability in the duodenum to control systemic iron metabolism.
-
•
Tf levels are controlled in the duodenum by digestive proteases during inflammation.
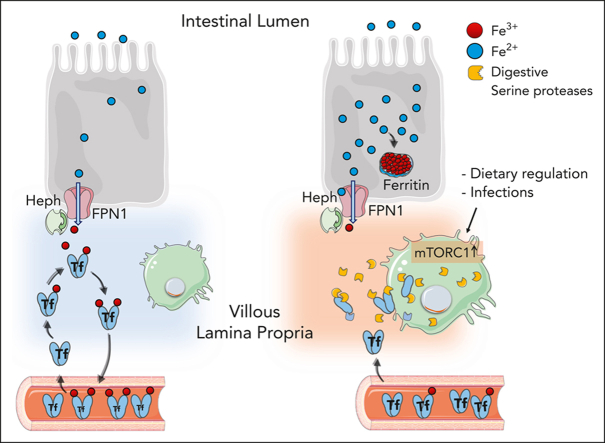
Visual Abstract
Activation of mTORC1 in villous duodenal macrophages induces the expression of serine protease, which locally degrade Trasferrin to inhibit efficient iron export from enterocytes.
Abstract
Iron is an essential cellular metal that is important for many physiological functions including erythropoiesis and host defense. It is absorbed from the diet in the duodenum and loaded onto transferrin (Tf), the main iron transport protein. Inefficient dietary iron uptake promotes many diseases, but mechanisms regulating iron absorption remain poorly understood. By assessing mice that harbor a macrophage-specific deletion of the tuberous sclerosis complex 2 (Tsc2), a negative regulator of mechanistic target of rapamycin complex 1 (mTORC1), we found that these mice possessed various defects in iron metabolism, including defective steady-state erythropoiesis and a reduced saturation of Tf with iron. This iron deficiency phenotype was associated with an iron import block from the duodenal epithelial cells into the circulation. Activation of mTORC1 in villous duodenal CD68+ macrophages induced serine protease expression and promoted local degradation of Tf, whereas the depletion of macrophages in mice increased Tf levels. Inhibition of mTORC1 with everolimus or serine protease activity with nafamostat restored Tf levels and Tf saturation in the Tsc2-deficient mice. Physiologically, Tf levels were regulated in the duodenum during the prandial process and Citrobacter rodentium infection. These data suggest that duodenal macrophages determine iron transfer to the circulation by controlling Tf availability in the lamina propria villi.
Sukhbaatar and colleagues investigated the role of mTORC1 signaling in iron metabolism. They report a novel pathway regulating iron metabolism, demonstrating that mTORC1 regulates macrophages in the lamina propria to induce local degradation of transferrin, limiting iron transfer from intestinal epithelial cells by decreasing availability of transferrin.
Introduction
Iron is an essential metal that plays a key role in many basic cellular processes including DNA synthesis, cellular metabolism, cell growth, and oxygen transport in erythrocytes. Because of the lack of a regulated mechanism for iron excretion in the body, iron absorption is critical to the control of iron homeostasis.1
Iron deficiency and iron-deficiency anemia are major health problems associated with different pathophysiological conditions of gastrointestinal diseases and inflammatory conditions.2,3 Iron absorption is known to be impaired in a range of these pathophysiological complications, which also limits the success of oral iron supplementation therapies, necessitating a better understanding of iron uptake physiology.4, 5, 6, 7
Dietary iron is absorbed at the brush border of duodenal epithelial cells. After the iron is absorbed from the diet, its export from the epithelial cells into circulation is facilitated by the iron exporter SLC40A1 (also known as ferroportin-1 [FPN1]) and the major iron transport protein transferrin (Tf) that safely carries iron to the target tissues.8,9 Tf has 2 high-affinity binding sites for the metal, and iron saturated holo-Tf is internalized by target cells by TfR1-mediated endocytosis.10,11 Current knowledge suggests that iron export from the duodenal epithelial cells is mainly controlled by a concerted expression/degradation of FPN1 as a consequence of the circulating iron hormone hepcidin,7,9 whereas Tf is mainly expressed in the liver and is reportedly not critical to the control of iron absorption.12
Macrophages are innate immune cells implicated in various steps of systemic iron metabolism.13 Splenic red pulp macrophages and Kupffer cells in the liver play critical roles in maintaining iron homeostasis by controlling iron recycling and storage, whereas central nurse macrophages in the bone marrow (BM) support erythrocyte differentiation.14,15 In addition, macrophages can sequester iron to inhibit growth of infectious bacteria.16, 17, 18, 19 Macrophages are part of the intestinal niche,20 however, whether duodenal macrophages contribute to iron metabolism has, to our knowledge, not been explored.
Mechanistic target of rapamycin complex 1 (mTORC1) is an evolutionarily conserved serine/threonine kinase that is involved in sensing and responding to the nutrition status to control cell growth, inflammatory response, and metabolic adaption.21,22 Tuberous sclerosis complex 2 (Tsc2) is a negative regulator of mTORC1, and its deletion in macrophages can promote granuloma formation in mice.23 Although mTORC1 has been implicated in the regulation of cellular iron metabolism,24,25 such a function for mTORC1 in macrophages is unknown.
In this study, we uncovered a novel physiological role of duodenal lamina propria macrophages in iron absorption. Our results indicate that mTORC1 activation in CD68+ villous lamina propria macrophages inhibit iron uptake by decoupling iron passage from epithelial cells to Tf in the duodenum. We identified upregulated digestive serine proteases in macrophages that locally degraded Tf in the duodenal interstitium, which was associated with reduced Tf saturation in the circulation.
Methods
All methods are described in the supplemental Materials, available on the Blood website.
Animals
Tsc2fl/flLyz2cre/+ (denoted as cre/+) mice were used as previously described.23 All animal experiments were approved by the official Austrian institutional ethics board.
55Fe absorption
For measuring whole-body iron distribution, a single dose of 2.5 μCi (9.25 × 104 Bq) 55FeCl3 per animal was administered to mice via oral gavage.
In vivo Fe dextran treatment
Control (+/+) and cre/+ mice, between 9 and 12 weeks old, were injected IV with a bolus of 1g/kg iron dextran in 1× phosphate-buffered saline (PBS). To maintain iron loading, mice received 3 further IV injections of 0.2 g/kg iron dextran for 4 weeks.
Intestinal macrophage depletion
Mice between 8 and 12 weeks old were injected intraperitoneally with 1.2 mg M279 anti-muCfms ralgG1 monoclonal antibody in 1× PBS 3 times a week for 6 weeks.
Protease inhibition in vivo with nafamostat
Mice between 9 and 16 weeks old were injected intraperitoneally with 10 mg/kg nafamostat mesylate every other day for 1 week.
Mouse whole-blood count analysis
Mouse blood was collected from the facial vein into a heparinized microhematocrit tube. Hematological values were determined via complete blood cell analyses, using a hematology analyzer.
Prussian blue staining
Paraffin-embedded tissue sections were stained with Perls’ Prussian blue solution and 2% hydrochloric acid.
Total iron concentration measurements with atomic absorption spectrometry
Total iron levels in serum and digested tissues were determined via flame atomic absorption spectrometry.
Tf saturation
Tf saturation in mouse serum was measured using an iron/total iron binding capacity (TIBC) kit (POINTE Scientific) per the manufacturer’s protocol.
Visualization of Tf saturation by urea–polyacrylamide gel electrophoresis
Visualization of Tf saturation by urea–polyacrylamide gel electrophoresis was performed as previously described.26
Colorimetric ferrozine assay for quantification of total Fe and Fe2+ in tissues
Quantification of iron concentration in tissues were determined via a colorimetric ferrozine assay, as previously described.27,28
Immunofluorescence of mouse cryosections and immunohistochemistry of formalin-fixed, paraffin-embedded mouse tissues
Flow cytometry analysis of erythropoiesis
BM was isolated freshly, and erythroid cells in the cell suspensions were stained with CD71 and Ter119C antibodies.
Flow cytometry analysis of duodenum macrophages
Cell suspensions from the duodenum of mice was prepared using a mouse lamina propria dissociation kit (MACS Miltenyi Biotec) per the manufacturer’s protocol. Isolated cell suspensions were further used for flow cytometry.
Tf uptake by duodenum macrophages
To analyze pH-sensitive Tf-iron endocytosis, cells suspensions from the duodenum of mice were prepared, and lamina propria (LP) macrophages (Cd45+, Cd11c+, Cd11b+, Ly6C−, Cd64+/−, and Cd68+) were identified. Cells were incubated with pHrodo-Red Tf conjugate, and the uptake of pHrodo-Tf in macrophages was measured via flow cytometry.
Bulk RNA-seq
Duodenal LP macrophages (Cd45+, Ly6G−, Cd11c+, Cd11b+, Ly6C−, and Cd64+) and Cd45−EpCAM+ epithelial cells were isolated and subjected to bulk RNA sequencing (RNA-seq).30
Single-cell RNA-seq
CD45+ cells were sorted. Single-cell next-generation sequencing libraries were generated using the 10x Genomic Single Cell 3' version 3 workflow and sequenced on a NovaSeq 6000 instrument (Illumina) in paired-end configuration, as recommended by the manufacturer.
Statistical analysis
Detailed statistical methods are described in the supplemental Information.
Results
Myeloid deletion of Tsc2 causes an erythropoiesis defect
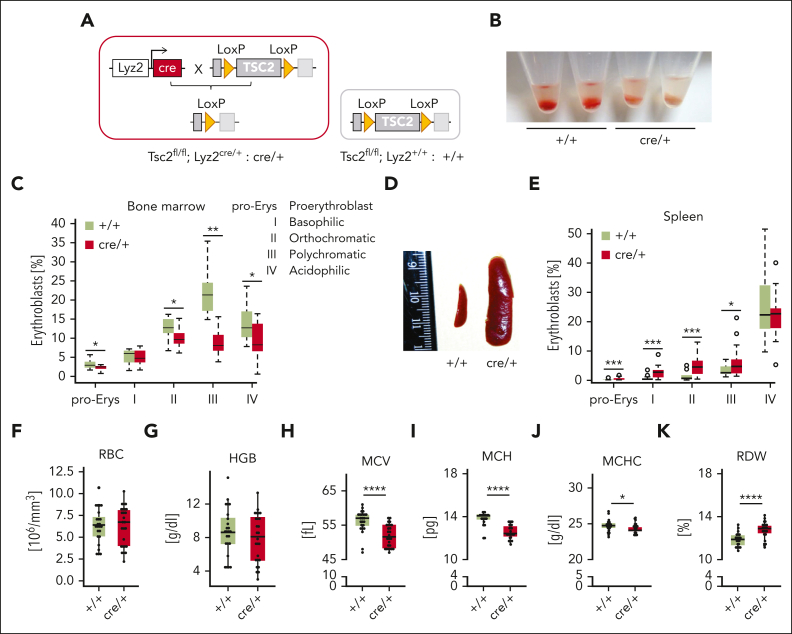
We previously established a mouse model in which we inactivated TSC2, a negative regulator of mTORC1 in Lyz2-expressing myeloid cells (cre/+), to constitutively activate mTORC1 in macrophages (Figure 1A).23 By visual inspection, we observed that the total BM was pale in cre/+ mice compared with that in littermate controls (Tsc2fl/flLyz2+/+; designated as +/+), indicating a failure in red blood cell (RBC) production (Figure 1B). To investigate whether the overactivation of mTORC1 by deletion of Tsc2 was directly responsible for this phenotype, cre/+ mice were treated with the mTORC1 inhibitor everolimus (supplemental Figure 1A). Indeed, after 5 days, the BM of cre/+ mice appeared red again (supplemental Figure 1B).
Figure 1.
TSC2 deletion in myeloid cells impairs medullary erythropoiesis. (A) Schematic representation of the genetic background of cre/+ mice vs +/+ controls. TSC2 knockout mice are generated by LysZ-cre system cross-targeting TSC2 gene with loxP sites (cre/+ mice). Control littermates contain the TSC2 gene flanked by LoxP sites (+/+). (B) Representative image showing defect in medullary erythropoiesis in the BM isolated from control (+/+) and cre/+ mice. (C) Bar graph of flow cytometric analysis showing erythroblast counts in the BM (∗P < .05; ∗∗P < .01). Different maturation stages of erythroblasts (pro-Erys, proerythroblasts; I, basophilic erythroblasts; II, orthochromatic erythroblasts; III, polychromatic erythroblasts; and IV, acidophilic erythroblasts) were identified based on CD71 (Tf receptor) and Ter119 (erythrocyte marker) expression and cell size (SSC). (D) Photography of the spleen of cre/+ mice compared with controls (+/+) represents splenomegaly in cre/+ mice. (E) Bar graph of flow cytometric analysis of erythroblast counts in different maturation stages in the spleen identified via CD71 (Tf receptor) and Ter119 (erythrocyte marker) expression (∗P < .05; ∗∗∗P < .001). (F-K) Complete blood cell count analysis showing hematological parameters in whole blood from control (+/+) and cre/+ mice: RBC count (F), blood hemoglobin (HGB) level (G), mean corpuscular volume (MCV) of RBCs (∗∗∗∗P < .0001) (H), mean corpuscular hemoglobin (MCH) of RBCs (∗∗∗∗P < .0001) (I), mean corpuscular hemoglobin concentration of RBCs (∗P < .05) (J), and the red cell distribution width (RDW) of RBCs (∗∗∗∗P < .0001) (K).
A detailed analysis of the BM erythroid progenitors using Ter119 and CD71 (TfR1) antibodies showed that the number of more mature erythrocytic progenitor cells (proerythroblasts, orthochromatic, polychromatic, and acidophilic erythroblasts) were markedly reduced in the BM of cre/+ mice (Figure 1C; supplemental Figure 1C). Specifically, the number of polychromic erythroid progenitors (stage III) that are characterized by hemoglobin accumulation, was significantly reduced in the cre/+ mice (Figure 1C). Parallely, we found massive spleen enlargement and significantly enhanced splenic erythroid progenitors in the cre/+ mice (Figure 1D-E), suggestive of compensatory extramedullary erythropoiesis. The analysis of the complete blood count of the cre/+ mice showed that the RBC number, hemoglobin level, and other blood parameters were similar to that of the +/+ littermates (Figure 1F-G; supplemental Figure 1D-G), indicative of no anemia. However, many erythrocyte-related values, such as the mean corpuscular volume, mean corpuscular hemoglobin, and mean corpuscular hemoglobin concentration were significantly lower, whereas the RBC distribution width was higher in the cre/+ mice than in the +/+ littermates (Figure 1H-K). These alterations revealed that erythrocytes are hypochromic (less hemoglobin content per RBC) and microcytic (RBC size reduced), a phenotype indicating iron limitation for erythroid progenitor cells. Furthermore, the RBCs of the cre/+ mice had a prolonged half-life but a decreased osmotic resistance, which suggests that the erythrocytes are older in cre/+ mice, compensating for the erythropoiesis failure in the BM (supplemental Figure 1H-I). BM-derived macrophages of cre/+ mice showed no obvious defect in erythrophagocytosis (supplemental Figure 1J). Overall, these data show that deletion of Tsc2 in macrophages significantly affects RBC production.
The erythropoiesis defect of cre/+ mice is caused by iron deficiency
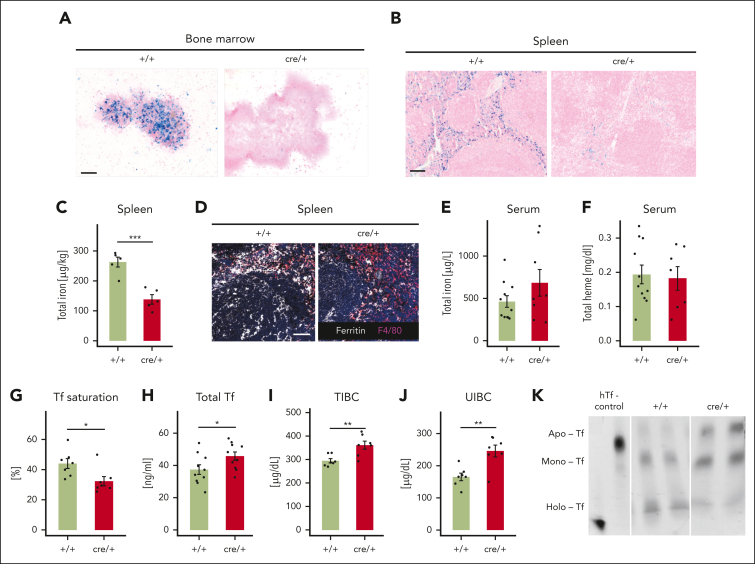
The aforementioned findings point to alterations in iron metabolism as a cause for the erythropoiesis defect in cre/+ mice. Indeed, tissue iron (Fe3+) pools in the BM and the spleen were completely depleted in cre/+ mice, as indicated by Prussian blue staining (Figure 2A-B). After an everolimus treatment of the cre/+ mice, iron levels were restored in the spleen (supplemental Figure 2A), suggesting that the alteration of tissue iron disruption in the cre/+ mice is dependent on mTORC1 signaling in macrophages. Further evaluation of total iron as well as a differentiated analysis of nonheme and labile iron (Fe2+) in the spleen confirmed that tissue iron levels in these organs were highly reduced (Figure 2C; supplemental Figure 2B). Iron concentration in the liver was not different between the mice (supplemental Figure 2C-D). Moreover, we did not observe obvious differences of iron deposition in other organs (supplemental Figure E-F). Ferritin is the major iron storage protein and a direct marker of tissue iron levels. Consistently, we found decreased ferritin levels in the spleen, but ferritin levels in the liver and serum of cre/+ mice were similar (Figure 2D; supplemental Figure 2G-H).
Figure 2.
TSC2 deletion in macrophages causes iron deficiency. (A-B) Representative image showing ferric iron staining using Prussian blue, counterstained with nuclear fast red in the BM (A) and on splenic formalin-fixed paraffin-embedded (FFPE) sections (B) of +/+ and cre/+ mice. Scale bar, 100 μm. (C) The total iron concentration was measured via an atomic absorption spectrometry (AAS) using the spleen of control (+/+) and cre/+ mice aged between 8 and 12 weeks old (∗∗∗P < .001). Total iron levels were normalized with respect to the tissue weight. (D) Representative image of immunofluorescent staining for ferritin (iron storage protein) and F4/80 (macrophage marker) on mouse spleen cryosections. Scale bar, 50 μm. (E) Total serum iron concentration was measured via AAS. (F) Total serum heme concentration was measured spectrometrically. (G) Tf saturation was determined in serum of control (+/+) and cre/+ mice (∗P < .05). (H) Total Tf concentration in the serum was determined using an enzyme-linked immunosorbent assay. (I-J) The TIBC (I) and unsaturated iron binding capacity (J) of Tf in the serum from control (+/+) and cre/+ mice were determined (∗∗ P < .01). (K) Steady-state levels of the Fe-free Apo-Tf (Apo), monoferric-Tf (Mono), and Fe-saturated holo-Tf (Holo) in control (+/+) and cre/+ mice as visualized on urea gel electrophoresis. Human apo-Tf and holo-Tf (hTf) were used as controls (lane1).
Levels of total serum iron and the heme concentration in serum were comparable in cre/+ and +/+ mice (Figure 2E-F). Interestingly, Tf saturation in serum, which is a main iron transporter for erythroid progenitor cells, was reduced in cre/+ mice, although total Tf levels in serum were increased compared with the controls (Figure 2G-H). Accordingly, the TIBC and unsaturated iron binding capacity of the serum were increased, which is in line with reduced Tf- bound iron in cre/+ mice (Figure 2I-J). In accordance, we detected more unsaturated mono- and apo-Tfs in the serum of the cre/+ mice, whereas the serum of +/+ control mice contained more saturated holo-Tf (Figure 2K). These findings imply that the erythropoiesis defect in cre/+ mice is due to iron deficiency that manifests in reduced Tf-iron saturation.
Iron is retained in duodenal epithelial cells in cre/+ mice
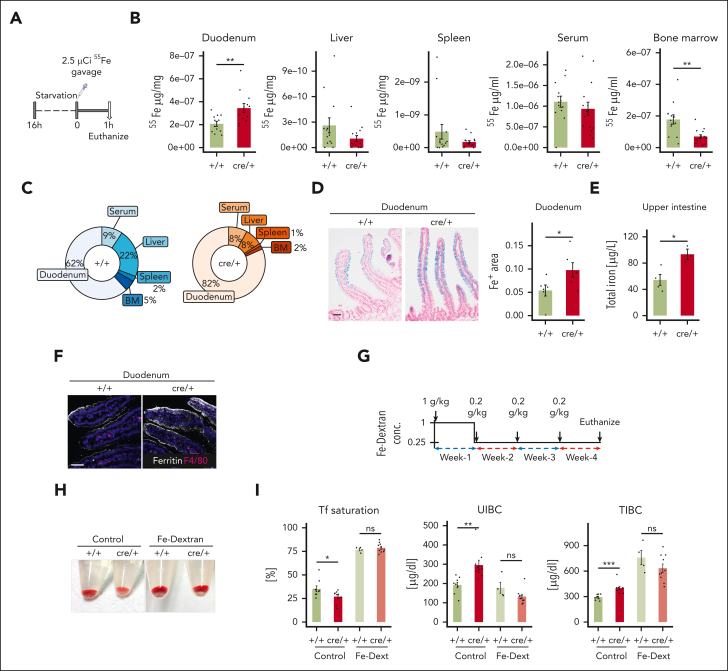
To elucidate the cause of reduced Tf loading with iron, we infused mice with radiolabeled iron (55Fe) via oral gavage (Figure 3A) and traced the route of dietary iron from the duodenum into the circulatory system and other tissues. Strikingly, 1 hour after the gavage, significantly more iron was detected in the duodenum of the mutant cre/+ mice, whereas lower amounts of 55Fe disseminated to the serum, spleen, liver, and BM in cre/+ mice (Figure 3B-C). Indeed, Prussian blue staining of the duodenum showed that iron storage levels in the duodenal epithelial cells were increased in the cre/+ mice compared with that in the controls (Figure 3D). Furthermore, in cre/+ mice, we measured higher iron levels in the proximal 5 cm of the upper small intestine containing the duodenum (Figure 3E), whereas in the remaining part of the small intestine, termed the lower intestine, we detected similar iron levels between +/+ and cre/+ mice (supplemental Figure 3A). In parallel, the expression of ferritin was elevated in the duodenal epithelial cells in cre/+ mice (Figure 3F; supplemental Figure 3B). Therefore, we concluded that iron is retained in the duodenal epithelium in cre/+ mice resulting in lower Tf-iron flux into the circulation, causing the reduced Tf saturation, and iron deficiency.
Figure 3.
Iron transport from duodenal epithelial cells into the circulation is impaired in cre/+ mice. (A) Graphical presentation of 55Fe administration via gavage in control (+/+) and cre/+ mice aged between 9 and 15 weeks old. One hour after administration, indicated tissues were collected and analyzed. (B) Quantification of radioactivity of 55Fe after 1 hour absorption in the duodenum (∗∗P = .008), liver (P = .09), spleen (P = .59), serum (P = .23), and BM (∗∗P = .002). (C) Pie chart illustrating 55Fe distribution in the tissues in percentage. Fe retention is expressed as a percentage of the total 55Fe measured in all the collected samples. (D) Representative image of ferric iron Prussian blue staining on duodenum FFPE sections obtained from +/+ and cre/+ mice, counterstained with nuclear fast red. Quantification of the blue-stained area in the duodenal LP (∗P < .05). Scale bar, 50 μm. (E) Total iron concentration was measured using AAS in the upper intestine including the duodenum of 8- to 12-week-old control (+/+) and cre/+ mice. (∗P < .05). (F) Representative image of immunofluorescent staining for ferritin (iron storage protein) and F4/80 (macrophage marker) on mouse duodenum cryosections obtained from +/+ and cre/+ mice. Scale bar, 50 μm. (G) Graphical presentation of IV iron dextran (Fe-dext) treatment of +/+ and cre/+ mice aged between 9 and 15 weeks old, as indicated. Control (+/+) mice were treated with sterile 1× PBS. After 4 weeks of treatment, tissues were collected and analyzed. (H) Representative image showing the BM isolated from controls (+/+) and cre/+ in mice treated with Fe-dext. (I) Tf saturation, unsaturated iron binding capacity, and TIBC of transferrin in serum were determined in mice treated IV with Fe-dext, and compared with controls (+/+) treated with 1× PBS (∗P < .05; ∗∗∗P < .001).
Next, we assessed whether IV iron supplementation, bypassing the duodenal iron absorption, restores the reduced Tf-iron loading and ameliorates erythropoiesis in cre/+ mice (Figure 3G). Notably, after 4 weeks of IV treatment with iron dextran, the BM of the cre/+ mice became red again (Figure 3H) and the reduced numbers of polychromic erythroid progenitors (stage III) in the BM were normalized (supplemental Figure 3C). In addition, the differences between +/+ and cre/+ mice in serum Tf saturation, unsaturated iron binding capacity, and, surprisingly, TIBC levels were abrogated by iron dextran treatment (Figure 3I; supplemental Figure 3D). Prussian blue staining showed that iron deposits in the storage tissues were recovered in the cre/+ mice (supplemental Figure 3E). Hence, the iron deficiency could be overcome by iron supplementation that bypasses duodenal iron absorption, supporting our observation that iron export from the epithelial cells is blocked in the duodenum of the cre/+ mice.
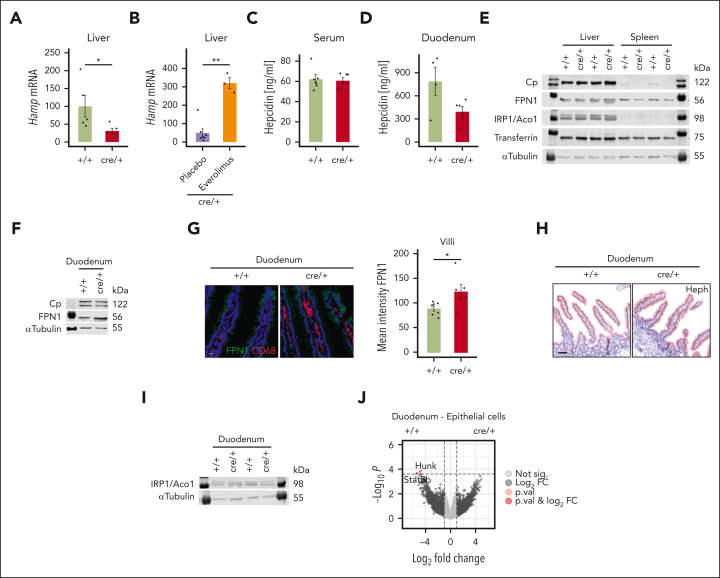
Iron retention in duodenum is not due to hepcidin or other known regulatory mechanisms
A key regulator of iron homeostasis is the peptide hepcidin that controls systemic iron levels by binding to FPN1 to induce its degradation.9,31 Thereby, iron remains sequestered in duodenal epithelial cells and tissue macrophages.1,7,9,32, 33, 34 In response to reduced serum iron levels, hepcidin production is inhibited in the liver to increase iron absorption.35 In contrast, inflammatory stimuli induce hepcidin expression to limit iron availability for microbes.36 To clarify whether hepcidin is involved in the tissue iron loss, we measured messenger RNA (mRNA) expression of its encoding gene Hamp in the liver. We found that the hepatic expression of Hamp was reduced (Figure 4A), whereas Hamp expression increased upon everolimus treatment in cre/+ mice (Figure 4B; supplemental Figures 1B and 2A). Expression thus paralleled low (cre/+) or high (cre/+ with everolimus treatment) iron levels, being in line with secondary regulation of Hamp by iron availability.35 Interestingly, protein expression of hepcidin in serum and the liver was similar between the genotypes, whereas we found a trend toward reduced levels in the duodenum (Figure 4C-D; supplemental Figure 4A). Interestingly, FPN1 protein levels were unchanged in the liver and spleen (Figure 4E; supplemental Figure 4B) but increased in the duodenal enterocytes (Figure 4F-G).The ferroxidases hephaestin and ceruloplasmin are required for the efficient transfer of FPN1-exported iron to Tf.8,35 However, levels and activity as well as IRP1 expression were similar in the tissues and serum (Figure 4E-F,H-I; supplemental Figure 4C,E-H). Moreover, expression of the TfR1 was comparable between the genotypes in the liver and the spleen (supplemental Figure 4D). To investigate other mechanisms that could explain the iron retention, we performed bulk RNA-seq on sorted duodenal epithelial cells (supplemental Figure 4I). However, we only found 2 genes differently expressed between cre/+ and +/+ controls (Figure 4J). Overall, several well-known regulatory mechanisms of iron metabolism do not account for the iron retention in duodenal epithelial cells in cre/+ mice.
Figure 4.
Iron deficiency is independent of hepcidin and other known regulatory mechanisms in cre/+ mice. (A) Liver Hamp (hepcidin) mRNA expression in control (+/+) and cre/+ mice (∗P < .05), and (B) in cre/+ mice after everolimus treatment (∗∗P < .01). (C-D) Hepcidin concentration measured in the serum (C) and in duodenum lysates (D) of control (+/+) and cre/+ mice via enzyme-linked immunosorbent assay. (E) Representative western blot analysis of Cp (ceruloplasmin [ferroxidase], 122 kDa), FPN1 (ferroportin-1 [cellular iron exporter], 56 kDa), iron regulatory protein/acotinase1 (iron regulatory protein [IRP1/Aco1], 98 kDa), transferrin (75 kDa), and the housekeeping protein α-tubulin (55 kDa) in the liver and spleen lysates from control (+/+) and cre/+ mice. (F) Representative western blot of Cp (122 kDa), FPN1 (56 kDa), and the housekeeping protein α-tubulin (55 kDa) in duodenum lysates from control (+/+) and cre/+ mice. (G) Representative image of immunofluorescent staining for FPN1 and F4/80 (macrophage marker) on mouse duodenum cryosections obtained from control (+/+) and cre/+ mice. Scale bar, 50 μm. Quantification of FPN1 mean fluorescence intensity in LP of duodenum. (H) Representative image of immunohistochemistry staining for hephaestin on mouse duodenum FFPE sections obtained from +/+ and cre/+ mice. Scale bar, 100 μm. (I) Representative western blot analysis of iron regulatory protein IRP1/Aco1 (iron regulatory protein/acotinase 1, 98 kDa) and the housekeeping protein α-tubulin (55 kDa) in duodenum lysates from control (+/+) and cre/+ mice. (J) Volcano-plot of normalized gene expression profiles in duodenum epithelial cells from control (+/+) and cre/+ mice.
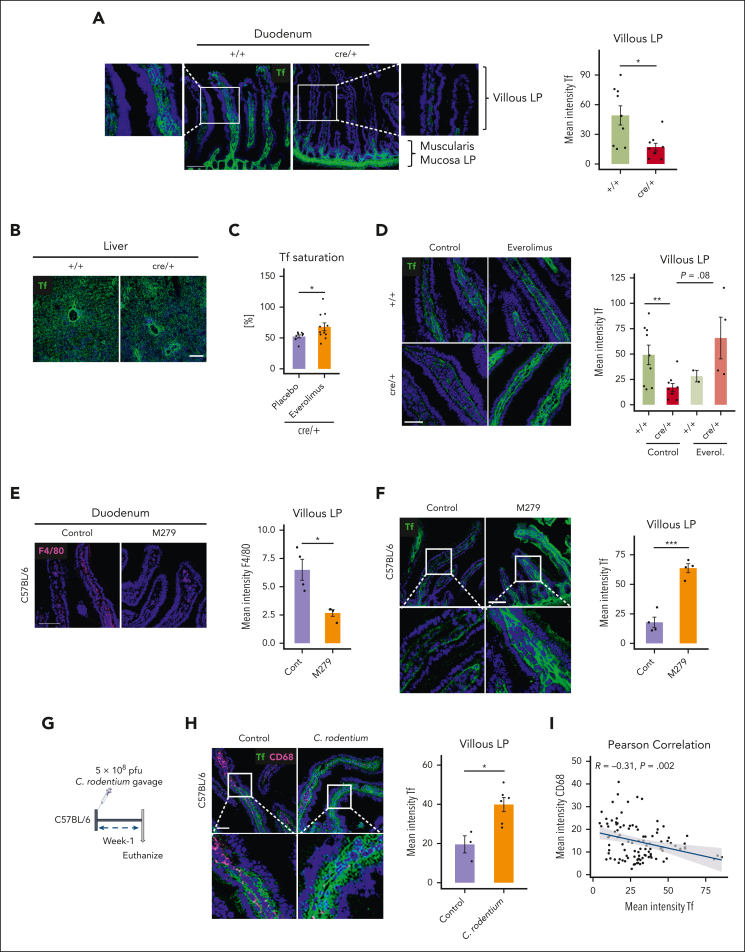
Local depletion of Tf in the duodenum by macrophages
We, therefore, looked for alternative explanations as to why iron is retained in the duodenum in cre/+ mice. Because of the reduced Tf saturation in the animals, we stained for Tf in the tissues. Strikingly, we found substantially decreased Tf levels in the villous interstitium of the LP but not in the muscularis mucosa LP in cre/+ mice compared with controls (Figure 5A). In contrast, Tf staining intensity in the LP regions near the muscularis mucosa as well as the total Tf concentration in the duodenum was not considerably different (Figure 5A; supplemental Figure 5A). Moreover, Tf expression was not altered in the liver or in the spleen of cre/+ mice (Figure 5B; supplemental Figure 5B). To evaluate, whether mTORC1 signaling in cre/+ macrophages mediates the local reduction of Tf, we treated the mice with everolimus (supplemental Figure 1A). Indeed, mTORC1 inhibition significantly increased Tf saturation in the cre/+ mice and restored Tf levels in the duodenal villous LP, albeit it was not statistically significant (P = .08; Figure 5C-D). Therefore, we postulated that the lack of Tf in the villous LP interstitium of the cre/+ mice disrupts Tf-mediated iron loading.
Figure 5.
Villous LP macrophages control local transferrin levels in the duodenum. (A) Representative image of immunofluorescent staining for Tf on mouse duodenum cryosections obtained from control (+/+) and cre/+ mice showing villous LP and muscularis mucosa LP regions. Scale bar, 100 μm. Quantification of Tf mean fluorescence intensity in duodenal LP (∗P < .05). (B) Representative image of immunofluorescent staining for Tf on liver cryosections of control (+/+) and cre/+ mice. Scale bar, 100 μm. (C) Tf saturation was determined in the serum of cre/+ mice treated with everolimus (inhibitor mTORC1 signal) compared with control cre/+ mice treated with 1× PBS (∗P < .05). (D) Representative image of immunofluorescent staining for Tf on mouse duodenum cryosections of +/+ and cre/+ mice treated with everolimus. Scale bar, 50 μm. Quantification of Tf fluorescence intensity in the duodenum LP. (E-F) Representative image of immunofluorescent staining for F4/80 (macrophage marker) and quantification of its mean fluorescence intensity (∗P < .05), (E) and for Tf (F) and quantification (∗∗∗P < .001) on mouse duodenum cryosections obtained from C57BL/6J mice treated with CSFR1 receptor inhibitor (M279) to deplete LP macrophages. Scale bar, 100 μm. (G) Graphical depiction of C rodentium treatment of 9- to 12-week-old C57BL/6J mice. One week after oral C rodentium inoculation, tissues were collected and analyzed. (H) Representative image of immunofluorescent staining for Tf and CD68 (macrophage marker) on mouse duodenum cryosections obtained from C57BL/6J mice treated with 1× PBS, and C57BL/6J mice treated with C rodentium. Scale bar, 50 μm. Quantification of Tf mean fluorescence intensity in LP (∗P < .05). (I) Pearson correlation between Tf and CD68 mean fluorescence intensity per villi in C57BL/6J mice treated with C rodentium.
To determine whether macrophages, in general, are involved in controlling local Tf levels, we depleted macrophages in the duodenum with a colony stimulating factor 1 receptor (CSF1R; M279)-blocking antibody. We found reduced numbers of F4/80+ and CD68+ macrophages, indicating successful depletion of macrophages in the LP of the duodenum (Figure 5E; supplemental Figure 5D). Importantly, Tf levels in the LP were significantly increased in macrophage-depleted mice, suggesting that wild-type macrophages also regulate Tf availability in the villous LP (Figure 5F).
In addition, we analyzed whether local Tf levels are physiologically controlled during the prandial process. Therefore, we starved +/+ and cre/+ mice overnight from food and refed them for 1 hour or 3 hours (supplemental Figure 5D). Remarkably, although cre/+ mice had reduced Tf levels during normal housing conditions compared with +/+ mice, as shown earlier, Tf levels substantially increased in both genotypes after starvation (supplemental Figure 5E). Refeeding the mice for 1 or 3 hours quickly decreased Tf levels in the duodenal LP villi (supplemental Figure 5E). Next, we wanted to assess whether alterations in Tf may occur in a pathophysiological setting. We hypothesized that infection with an enteric pathogen that colonizes the duodenum might affect Tf expression. To this end, we infected wild-type mice orally with Citrobacter rodentium to induce acute mucosal inflammation (Figure 5G). We found that higher Tf levels in the duodenum of infected animals compared with that in the duodenum of uninfected controls (Figure 5H). Furthermore, we observed a highly negative association between CD68+ macrophages and Tf, that is, in villous LP regions, areas in which high numbers of CD68+ macrophages were present, we detected low levels of Tf, and vice versa (Figure 5I). Thus, local regulation of Tf levels occurs with enteric infections and during the prandial process. Taken together, these results suggest an unexpected role of mTORC1 in duodenal villous LP macrophages in controlling iron transfer from epithelial cells through the local regulation of Tf.
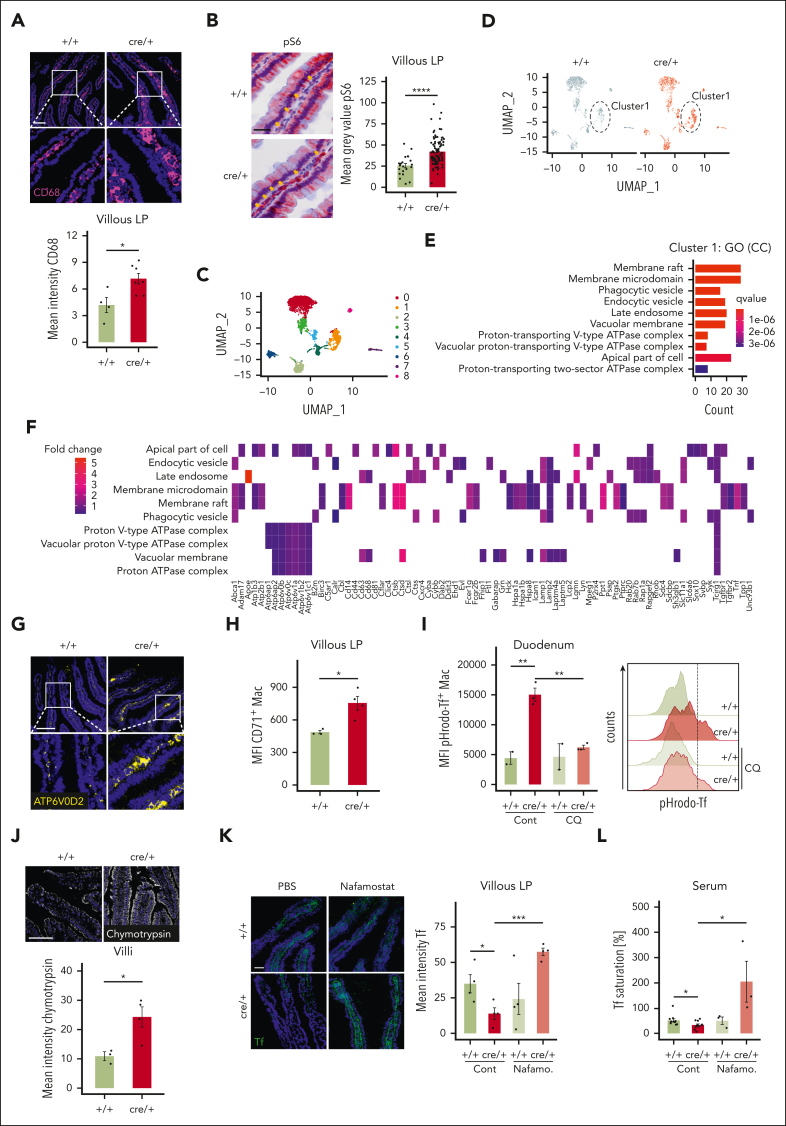
CD68+ LP macrophages restrict iron absorption through regulation of protease activity
To understand how LP macrophages affect Tf levels, we further characterized the duodenal macrophages. Macrophages in the villous LP regions of cre/+ mice expressed higher levels of CD68, a macrophage activation marker (Figure 6A), whereas F4/80+ expression was similar between +/+ and cre/+ mice (supplemental Figure 6A). In cre/+ mice, CD68 levels decreased after everolimus treatment, indicating mTORC1-dependent activation of duodenal macrophages (supplemental Figure 6B). Furthermore, we discovered an increased phosphorylation of S6 (pS6), a marker of activated mTORC1 signaling, in the villous LP regions of cre/+ mice, as expected (Figure 6B). To elucidate whether duodenal macrophages directly regulate Tf levels, we sorted macrophages from the LP and performed a bulk RNA-seq analysis (supplemental Figure 6C). We found that the number of CD64+ LP macrophages was similar and only few genes were altered (supplemental Figure 6D-E). Furthermore, we could not detect significant differences in the expression of known iron metabolism genes (supplemental Figure 6F). Recently, the involvement of mTOR signaling in the regulation of hepcidin via BMP-SMAD signaling was described.37,38 However, none of the target genes were altered in our transcriptomics data (supplemental Figure 6G). Nevertheless, pathway analysis of RNA-seq data revealed a significant enrichment of protein digestion pathways and genes encoding various proteases such as elastase-like proteases (Cela2a and Cela3b), carboxypeptidases, or trypsin (supplemental Figure 6H-I).
Figure 6.
Digestive proteases in villous CD68+ LP macrophages locally degrade transferrin in cre/+ mice. (A) Representative image of immunofluorescent staining for CD68 (macrophage activation marker) on mouse duodenum cryosections obtained from control (+/+) and cre/+ mice. Scale bar, 100 μm. Quantification of CD68 mean fluorescence intensity in the LP (∗P < .05). (B) Representative image of immunohistochemistry staining for pS6 (phosphorylated S6 ribosomal protein [Ser240/244]), mTORC1 signal activation marker) on FFPE sections obtained from +/+ and cre/+ mice, and its quantification in the LP. Scale bar, 25 μm (∗∗∗∗P < .0001). Arrows indicate LP region with macrophages. (C-F) Single-cell RNA-seq analysis of CD68+ cells: Umap plots of gene expression levels in CD68+ cell clusters (C) and differentially expressed genes between cre/+ and +/+ mice (D). Significantly enriched pathways in GO terms, category cellular components with differentially expressed genes from cluster 1 (E). Heatmap of identified genes in enriched GO pathways (F). (G) Representative image of immunofluorescent staining for ATP6V0D2 (vATPases) on mouse duodenum cryosections obtained from control (+/+) and cre/+ mice. Scale bar, 100 μm. (H) Flow cytometry quantitative analysis of CD71 (Trf1, transferrin receptor) mean fluorescence intensity of LP macrophages (CD64+ Ly6C− CD11b+ CD11cint) (I) and of pH-sensitive pHrodo-Red Tf uptake in LP macrophages from +/+ and cre/+ mice. Histogram of pHrodo-Red Tf+ LP macrophages compared between +/+ and cre/+ mice with or without lysosomal pH-disrupting drug hydrochloroquine. (J) Representative images of chymotrypsin enzyme activity on duodenum cryosections with an activity-based probe (ABP; fluorophore-labeled covalent inhibitor). Scale bar, 100 μm. Quantification of enzyme activity based on mean fluorescence intensity in the duodenal villi. (K) Representative image of immunofluorescent staining for Tf in LP of control (+/+) and cre/+ mice after protease inhibitor nafamostat (FUT-175) treatment for 1 week. Quantification of Tf mean fluorescence intensity in the LP (∗P < .05; ∗∗∗P < .001). Scale bar, 50 μm. (L) Tf saturation was determined in serum of mice treated with protease inhibitor nafamostat (FUT-175) for 1 week (∗P < .05). Control animals were treated with 1× PBS.
To analyze the duodenal macrophages more precisely, we performed single-cell RNA-seq on CD45+ LP cells and focused our analysis on CD68+ cells. We found 9 clusters of CD68+ cells; among them, clusters 1 to 8 were identified as myeloid populations (Figure 6C; supplemental Figure 7A). Examination of genes related to iron metabolism in LP macrophages confirmed our observation that their expression is not affected (supplemental Figure 7B). Furthermore, cluster 1 showed the highest expression of CD68 and other macrophage-specific markers such as Csf1r, Adgre1 (F4/80), and Cx3cr1 (supplemental Figure 7C), and its gene expression profile was mostly different between +/+ and cre/+ groups (Figure 6D; supplemental Figure 7A). Therefore, we further analyzed cluster 1 to assess how these macrophages might affect Tf levels in cre/+ mice. mRNA expression of Tf was not significantly different between the cluster 1 macrophages of the +/+ and cre/+ mice (supplemental Figure 7B). Gene Ontology (GO) enrichment analysis of cluster 1 genes resulted in the enrichment of pathways related to endosomal and phagocytic processes (Figure 6E). KEGG enrichment analysis of cluster 1 genes revealed lysosome- and phagosome-related pathways in cre/+ mice (supplemental Figure 7D). Gene profiling of the genes in the enriched pathways revealed that several cathepsin proteases were enriched in cre/+ macrophages (Figure 6F; supplemental Figure 7E-F). Legumain, an endopeptidase that targets several cleavage sites of Tf was also identified in cre/+ macrophages.39
Activation of lysosomal digestive proteases in endosomes are dependent on lysosomal acidification that is regulated via vacuolar-H+-adenosine triphosphatases (vATPases).40,41 Indeed, GO enrichment helped identify the pathways associated with upregulated vATPases in cre/+ mice (Figure 6E-F; supplemental Figure 7G). The macrophage-specific vATPase subunit Atp6v0d2 was also one of the most differentially expressed genes in the cre/+ macrophages in the bulk RNA-seq experiment (supplemental Figure 6E).42 Atp6v0d2 immunostaining helped confirm the significantly increased expression of the subunit in the duodenal LP macrophages in the cre/+ mice (Figure 6G). Analysis of LP macrophages revealed increased mRNA expression of TfR1 (CD71) in the duodenal LP (Figure 6H; supplemental Figure 7H). In cluster 1 of the cre/+ animals, expression of Zfp36, which encodes thymidine 5′-triphosphate (TTP) and has been shown to induce TfR1 expression in a mTORC1-dependent way,24 was significantly higher (fold change = 2.1; adjP = 1.29E−10). Consistently, LP macrophages from the cre/+ mice were able to engulf more Tf iron ex vivo, whereas the lysosomal pH-disrupting drug hydrochloroquine reduced Tf-iron uptake (Figure 6I). Because our data indicated increased protease expression and activity in the duodenum of cre/+ mice, we wanted to confirm this with an activity-based protease probe in situ.43 Indeed, we found increased elastase-like and chymotrypsin activity in cre/+ mice (Figure 6J; supplemental Figure 7I-J). Finally, to examine the contribution of protease activities downstream of mTORC1 in macrophages in regulating LP Tf levels, we treated mice with the serine protease inhibitor nafamostat mesylate, which did not interfere with CD68 expression by macrophages (supplemental Figure 7K-L). Remarkably, in vivo inhibition of proteases with nafamostat restored Tf levels in the LP of cre/+ animals (Figure 6K). In addition, nafamostat induced iron release from enterocytes and rescued serum Tf saturation in the cre/+ mice (Figure 6L; supplemental Figure 7M).
Taken together, these results imply that increased endosomal and lysosomal acidification favors subsequent proteolytic degradation of interstitial Tf in CD68+ villous LP macrophages to regulate dietary iron absorption and systemic iron availability.
Discussion
Here, we propose a novel mechanism in the duodenum to control systemic iron homeostasis. Specifically, we hypothesize that villous CD68+ LP macrophages control dietary iron uptake by locally modifying Tf stability in the duodenum, thereby affecting efficient transfer of iron from duodenal epithelial cells to Tf. We further suggest that this system is controlled by mTORC1 activity in macrophages.
In a myeloid-specific Tsc2-deficient mouse model, we found that hyperactivation of mTORC1 signaling in macrophages causes iron retention in duodenal epithelial cells and leads to a systemic iron deficiency and iron-limited BM erythropoiesis. Neither hepcidin nor other known iron regulatory pathways in the epithelial cells and in the LP macrophages seemed to be involved in the iron retention phenotype of the cre/+ mice.44 Instead, we found reduced Tf levels specifically in the interstitium of the villous LP. Intestinal Tf deficiency was associated with reduced Tf saturation in the serum. The lower Tf saturation that presented with more apoferric and monoferric Tfs, which has a lower affinity for the TfR1, may potentially affect the erythropoiesis defect in the BM.11,45 Nevertheless, we propose that Tsc2-deficient central nurse and red pulp macrophages, may also reinforce the systemic iron deficiency phenotype in the BM and spleen of the mice, respectively. This assumption is further supported by the observation that mice who are heterozygous hypotransferrinemic have low systemic Tf concentrations but no overt iron deficiency in the BM or spleen.46 Despite the reduced Tf saturation and the clear retention of iron in the enterocytes in the cre/+ mice, the similar total iron levels in the serum and liver between the genotypes suggested that some non–Tf-bound iron may be secreted and loaded onto other high-affinity iron binding proteins, which may be taken up by liver hepatocytes.
Furthermore, our results demonstrate that macrophages directly control Tf levels in the duodenum, because deletion of macrophages in the intestine increased duodenal Tf. Starvation of mice also increased local Tf levels in the duodenum, whereas refeeding decreased Tf, suggesting regulation of iron absorption during the prandial process. During C rodentium infection in mice, Tf levels were increased and were negatively associated with the presence of CD68+ macrophages, indicating a potential new role of duodenal macrophage conditions associated with intestinal infectious disease that involves the alteration of Tf-iron availability as a host defense mechanism. Chronic inflammation and infection cause functional iron deficiency as a consequence of an evolutionary conserved mechanism of nutritional immunity with the purpose of restricting iron availability to invading pathogens.15, 16, 17,19 Given the high binding affinities of Tf,47 our finding that Tf levels in LP increase with intestinal infections are in a line with the host’s strategy of restricting iron availability to intestinal bacteria, pointing to the importance of macrophage iron control of Tf for host immune defense against invading microbes.
Our findings show that Tsc2-deficient macrophages increase the expression of enzymatic proteases in the LP. In line with this, inhibition of protease activity by nafamostat restored Tf levels in the duodenum, induced the release of storage iron from the enterocytes, and increased serum Tf saturation in the cre/+ mice. These findings imply that mTORC1 activation in LP macrophages affects Tf -mediated iron transport into the blood circulation by controlling local protease activity. The production and activation of digestive proteases depend on high endosomal acidification maintained by vATPase proton pumps.48,49 mTORC1 can promote lysosome biogenesis and affects lysosomal protein expression.50 Accordingly, in duodenal LP we found increased expression of vATPases, which maintain low pH in the endosomes, necessary for enzymatic protease activity. Overall, our results suggest that mTORC1-activated macrophages locally degrade Tf dependent on endosomal/lysosomal acidification. Early reports demonstrated that Tfs internalized by macrophages can be degraded.51 Generally, our findings provide insight into a novel iron regulatory function of intestinal macrophages and suggest local control of Tf availability as an important mechanism to regulate efficient dietary iron intake.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Acknowledgments
The authors thank Herbert Stangl and Peter Chiba for their support and guidance with the radiolabeled 55Fe experiment. The authors also thank Clarissa Braun, Kamil Pabis, Clarice Lim, and Chrysoula Vraka for their excellent technical assistance. The authors thank the team of the Biomedical Sequencing Facility at CeMM for assistance with next-generation sequencing and single-cell RNA-seq.
This research was supported by funding from the following sources: the Austrian Science Fund (FWF) grants P30857-B28 (T.W.), P34023-B (T.W.), P34266-B (T.W.), FWF Sonderforschungsbereich F83 (T.W.), SFB F6101 (B.S., M. Müller), SFB F6106 (B.S., M. Müller), the Vienna Science and Technology Fund grant LS18-058 (T.W.), and the Christian Doppler Laboratory for Iron Metabolism and Anemia Research (G.W.). N.S. was supported by a DOC fellowship of the Austrian Academy of Sciences. J.P.K. was supported by a doctoral fellowship of The Research Foundation–Flanders (no. 1S18520N).
Authorship
Contribution: N.S. and T.W. conceived the project; N.S., M.S., S.D.F., M.L., S.H., M.T., M. Mazić, S.F., K.G., J.P.K., C.B., and C.L. conducted experiments and analyzed the data; B.S.-M., C.G., S.D., M.H., R.K., S.H.L.V., G.W., I.T., M. Müller, and B.S. analyzed/interpreted results and provided reagents; T.W. supervised the project; and N.S. and T.W. wrote the manuscript with input from all other authors.
Footnotes
Bulk RNA sequencing data has been deposited in the Gene Expression Omnibus database (accession number GSE175992).
Original data are available upon request to the corresponding author, Thomas Weichhart (thomas.weichhart@meduniwien.ac.at).
The online version of this article contains a data supplement.
There is a Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Supplementary Material
References
- 1.Hentze MW, Muckenthaler MU, Galy B, Camaschella C. Two to tango: regulation of mammalian iron metabolism. Cell. 2010;142(1):24–38. doi: 10.1016/j.cell.2010.06.028. [DOI] [PubMed] [Google Scholar]
- 2.Pasricha S-R, Tye-Din J, Muckenthaler MU, Swinkels DW. Iron deficiency. Lancet. 2021;397(10270):233–248. doi: 10.1016/S0140-6736(20)32594-0. [DOI] [PubMed] [Google Scholar]
- 3.Camaschella C. Iron deficiency. Blood. 2019;133(1):30–39. doi: 10.1182/blood-2018-05-815944. [DOI] [PubMed] [Google Scholar]
- 4.Gonçalves C, Oliveira ME, Palha AM, Ferrão A, Morais A, Lopes AI. Autoimmune gastritis presenting as iron deficiency anemia in childhood. World J Gastroenterol. 2014;20(42):15780–15786. doi: 10.3748/wjg.v20.i42.15780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kaitha S, Bashir M, Ali T. Iron deficiency anemia in inflammatory bowel disease. World J Gastrointest Pathophysiol. 2015;6(3):62–72. doi: 10.4291/wjgp.v6.i3.62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rustgi SD, Bijlani P, Shah SC. Autoimmune gastritis, with or without pernicious anemia: epidemiology, risk factors, and clinical management. Therap Adv Gastroenterol. 2021;14 doi: 10.1177/17562848211038771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Theurl I, Aigner E, Theurl M, et al. Regulation of iron homeostasis in anemia of chronic disease and iron deficiency anemia: diagnostic and therapeutic implications. Blood. 2009;113(21):5277–5286. doi: 10.1182/blood-2008-12-195651. [DOI] [PubMed] [Google Scholar]
- 8.Vulpe CD, Kuo YM, Murphy TL, et al. Hephaestin, a ceruloplasmin homologue implicated in intestinal iron transport, is defective in the sla mouse. Nat Genet. 1999;21(2):195–199. doi: 10.1038/5979. [DOI] [PubMed] [Google Scholar]
- 9.Nemeth E, Tuttle MS, Powelson J, et al. Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science. 2004;306(5704):2090–2093. doi: 10.1126/science.1104742. [DOI] [PubMed] [Google Scholar]
- 10.van Renswoude J, Bridges KR, Harford JB, Klausner RD. Receptor-mediated endocytosis of transferrin and the uptake of fe in K562 cells: identification of a nonlysosomal acidic compartment. Proc Natl Acad Sci U S A. 1982;79(20):6186–6190. doi: 10.1073/pnas.79.20.6186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dautry-Varsat A, Ciechanover A, Lodish HF. pH and the recycling of transferrin during receptor-mediated endocytosis. Proc Natl Acad Sci U S A. 1983;80(8):2258–2262. doi: 10.1073/pnas.80.8.2258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gkouvatsos K, Papanikolaou G, Pantopoulos K. Regulation of iron transport and the role of transferrin. Biochim Biophys Acta. 2012;1820(3):188–202. doi: 10.1016/j.bbagen.2011.10.013. [DOI] [PubMed] [Google Scholar]
- 13.Sukhbaatar N, Weichhart T. Iron regulation: macrophages in control. Pharmaceuticals (Basel) 2018;11(4):137. doi: 10.3390/ph11040137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Soares MP, Hamza I. Macrophages and iron metabolism. Immunity. 2016;44(3):492–504. doi: 10.1016/j.immuni.2016.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nairz M, Theurl I, Swirski FK, Weiss G. “Pumping iron”-how macrophages handle iron at the systemic, microenvironmental, and cellular levels. Pflugers Arch. 2017;469(3-4):397–418. doi: 10.1007/s00424-017-1944-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nairz M, Schroll A, Demetz E, Tancevski I, Theurl I, Weiss G. ‘Ride on the ferrous wheel’--the cycle of iron in macrophages in health and disease. Immunobiology. 2015;220(2):280–294. doi: 10.1016/j.imbio.2014.09.010. [DOI] [PubMed] [Google Scholar]
- 17.Weiss G, Ganz T, Goodnough LT. Anemia of inflammation. Blood. 2019;133(1):40–50. doi: 10.1182/blood-2018-06-856500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Soares MP, Weiss G. The iron age of host-microbe interactions. EMBO Rep. 2015;16(11):1482–1500. doi: 10.15252/embr.201540558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ganz T, Nemeth E. Iron homeostasis in host defence and inflammation. Nat Rev Immunol. 2015;15(8):500–510. doi: 10.1038/nri3863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chiaranunt P, Tai SL, Ngai L, Mortha A. Beyond immunity: underappreciated functions of intestinal macrophages. Front Immunol. 2021;12:749708. doi: 10.3389/fimmu.2021.749708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Weichhart T, Hengstschläger M, Linke M. Regulation of innate immune cell function by mTOR. Nat Rev Immunol. 2015;15(10):599–614. doi: 10.1038/nri3901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Battaglioni S, Benjamin D, Wälchli M, Maier T, Hall MN. mTOR substrate phosphorylation in growth control. Cell. 2022;185(11):1814–1836. doi: 10.1016/j.cell.2022.04.013. [DOI] [PubMed] [Google Scholar]
- 23.Linke M, Pham HTT, Katholnig K, et al. Chronic signaling via the metabolic checkpoint kinase mTORC1 induces macrophage granuloma formation and marks sarcoidosis progression. Nat Immunol. 2017;18(3):293–302. doi: 10.1038/ni.3655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bayeva M, Khechaduri A, Puig S, et al. mTOR regulates cellular iron homeostasis through tristetraprolin. Cell Metab. 2012;16(5):645–657. doi: 10.1016/j.cmet.2012.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guan P, Wang N. Mammalian target of rapamycin coordinates iron metabolism with iron-sulfur cluster assembly enzyme and tristetraprolin. Nutrition. 2014;30(9):968–974. doi: 10.1016/j.nut.2013.12.016. [DOI] [PubMed] [Google Scholar]
- 26.Praschberger M, Haider K, Cornelius C, et al. Iron sucrose and ferric carboxymaltose: no correlation between physicochemical stability and biological activity. Biometals. 2015;28(1):35–50. doi: 10.1007/s10534-014-9801-0. [DOI] [PubMed] [Google Scholar]
- 27.Riemer J, Hoepken HH, Czerwinska H, Robinson SR, Dringen R. Colorimetric ferrozine-based assay for the quantitation of iron in cultured cells. Anal Biochem. 2004;331(2):370–375. doi: 10.1016/j.ab.2004.03.049. [DOI] [PubMed] [Google Scholar]
- 28.Fish WW. Rapid colorimetric micromethod for the quantitation of complexed iron in biological samples. Methods Enzymol. 1988;158:357–364. doi: 10.1016/0076-6879(88)58067-9. [DOI] [PubMed] [Google Scholar]
- 29.Gorvel JP, Joly I, Rigal A, Chambraud L, Bourges F, Maroux S. Expression of the human A, B and related antigens in the jejunum of human and rabbit small intestines, in particular on the brush border membrane glycoproteins. Rev Fr Transfus Immunohematol. 1987;30(5):471–483. doi: 10.1016/s0338-4535(87)80107-1. [DOI] [PubMed] [Google Scholar]
- 30.Picelli S, Faridani OR, Björklund AK, Winberg G, Sagasser S, Sandberg R. Full-length RNA-seq from single cells using Smart-seq2. Nat Protoc. 2014;9(1):171–181. doi: 10.1038/nprot.2014.006. [DOI] [PubMed] [Google Scholar]
- 31.Ganz T, Nemeth E. Iron metabolism: interactions with normal and disordered erythropoiesis. Cold Spring Harb Perspect Med. 2012;2(5):a011668. doi: 10.1101/cshperspect.a011668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ma S, Dubin AE, Zhang Y, et al. A role of PIEZO1 in iron metabolism in mice and humans. Cell. 2021;184(4):969–982.e13. doi: 10.1016/j.cell.2021.01.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Donovan A, Lima CA, Pinkus JL, et al. The iron exporter ferroportin/Slc40a1 is essential for iron homeostasis. Cell Metab. 2005;1(3):191–200. doi: 10.1016/j.cmet.2005.01.003. [DOI] [PubMed] [Google Scholar]
- 34.Ganz T, Nemeth E. Hepcidin and iron homeostasis. Biochim Biophys Acta. 2012;1823(9):1434–1443. doi: 10.1016/j.bbamcr.2012.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Girelli D, Nemeth E, Swinkels DW. Hepcidin in the diagnosis of iron disorders. Blood. 2016;127(23):2809–2813. doi: 10.1182/blood-2015-12-639112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Prentice AM, Bah A, Jallow MW, et al. Respiratory infections drive hepcidin-mediated blockade of iron absorption leading to iron deficiency anemia in African children. Sci Adv. 2019;5(3) doi: 10.1126/sciadv.aav9020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mleczko-Sanecka K, Roche F, da Silva AR, et al. Unbiased RNAi screen for hepcidin regulators links hepcidin suppression to proliferative Ras/RAF and nutrient-dependent mTOR signaling. Blood. 2014;123(10):1574–1585. doi: 10.1182/blood-2013-07-515957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Colucci S, Pagani A, Pettinato M, et al. The immunophilin FKBP12 inhibits hepcidin expression by binding the BMP type I receptor ALK2 in hepatocytes. Blood. 2017;130(19):2111–2120. doi: 10.1182/blood-2017-04-780692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Manoury B, Hewitt EW, Morrice N, Dando PM, Barrett AJ, Watts C. An asparaginyl endopeptidase processes a microbial antigen for class II MHC presentation. Nature. 1998;396(6712):695–699. doi: 10.1038/25379. [DOI] [PubMed] [Google Scholar]
- 40.Pejler G, Hu Frisk JM, Sjöström D, Paivandy A, Öhrvik H. Acidic pH is essential for maintaining mast cell secretory granule homeostasis. Cell Death Dis. 2017;8(5) doi: 10.1038/cddis.2017.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yoshimori T, Yamamoto A, Moriyama Y, Futai M, Tashiro Y. Bafilomycin A1, a specific inhibitor of vacuolar-type H(+)-ATPase, inhibits acidification and protein degradation in lysosomes of cultured cells. J Biol Chem. 1991;266(26):17707–17712. [PubMed] [Google Scholar]
- 42.Xia Y, Liu N, Xie X, et al. The macrophage-specific v-ATPase subunit ATP6V0D2 restricts inflammasome activation and bacterial infection by facilitating autophagosome-lysosome fusion. Autophagy. 2019;15(6):960–975. doi: 10.1080/15548627.2019.1569916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kahler JP, Verhelst SHL. Phosphinate esters as novel warheads for activity-based probes targeting serine proteases. RSC Chem Biol. 2021;2(4):1285–1290. doi: 10.1039/d1cb00117e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Muckenthaler MU, Rivella S, Hentze MW, Galy B. A red carpet for iron metabolism. Cell. 2017;168(3):344–361. doi: 10.1016/j.cell.2016.12.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yersin A, Osada T, Ikai A. Exploring transferrin-receptor interactions at the single-molecule level. Biophys J. 2008;94(1):230–240. doi: 10.1529/biophysj.107.114637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bu JT, Bartnikas TB. The use of hypotransferrinemic mice in studies of iron biology. Biometals. 2015;28(3):473–480. doi: 10.1007/s10534-015-9833-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Barber MF, Elde NC. Escape from bacterial iron piracy through rapid evolution of transferrin. Science. 2014;346(6215):1362–1366. doi: 10.1126/science.1259329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Li SC, Kane PM. The yeast lysosome-like vacuole: endpoint and crossroads. Biochim Biophys Acta. 2009;1793(4):650–663. doi: 10.1016/j.bbamcr.2008.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Forgac M. Vacuolar ATPases: rotary proton pumps in physiology and pathophysiology. Nat Rev Mol Cell Biol. 2007;8(11):917–929. doi: 10.1038/nrm2272. [DOI] [PubMed] [Google Scholar]
- 50.Alesi N, Akl EW, Khabibullin D, et al. TSC2 regulates lysosome biogenesis via a non-canonical RAGC and TFEB-dependent mechanism. Nat Commun. 2021;12(1):4245. doi: 10.1038/s41467-021-24499-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rama R, Sánchez J. Transferrin uptake by bone marrow macrophages is independent of the degree of iron saturation. Br J Haematol. 1992;82(2):455–459. doi: 10.1111/j.1365-2141.1992.tb06444.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.