Abstract
Historically, biological research has relied primarily on animal models. While this led to the understanding of numerous human biological processes, inherent species-specific differences make it difficult to answer certain liver-related developmental and disease-specific questions. The advent of 3D organoid models that are either derived from pluripotent stem cells or generated from healthy or diseased tissue-derived stem cells have made it possible to recapitulate the biological aspects of human organs. Organoid technology has been instrumental in understanding the disease mechanism and complements animal models. This review underscores the advances in organoid technology and specifically how liver organoids are used to better understand human-specific biological processes in development and disease. We also discuss advances made in the application of organoid models in drug screening and personalized medicine.
Keywords: human pluripotent stem cell, liver development, organoids, metabolic liver disease, viral infection, primary liver cancer, regenerative medicine, organ-on-chip
1. Introduction—Historical Overview
Stem cells have revolutionized how we perceive developmental biology, disease modeling, and the development of possible therapies. However, the main focus has been to obtain a considerably pure population of a specific cell type [1]. However, the advent of organoid technology (Figure 1), a three-dimensional (3D) culture system that accurately emulates an organ, has opened new avenues in the field of cell culture [2,3,4].
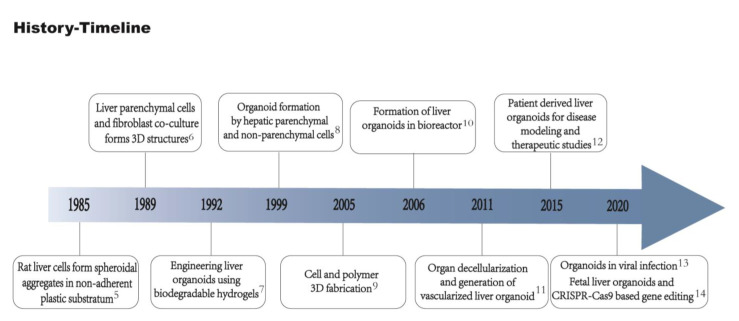
Figure 1.
Timeline of important events in liver organoid technology. Rat liver cells successfully formed spheroidal aggregates in non-adherent plastic substratum [5]. Hepatocytes and fibroblasts co-culture formed 3D structures [6]. Biodegradable hydrogels used to generate liver organoids in vitro [7]. Liver parenchymal and non-parenchymal cells isolated and co-cultured to form organoids [8]. 3D Cell and biomaterial complex formation [9]. Development of bioreactor for the generation of liver organoids using bio-artificial liver [10]. Generation of vascularized liver organoids [11]. Generation of organoids from patient-derived cells [12]. Generation of liver ductal organoids to recapitulate SAR-CoV-2 infection [13]. Development of fetal liver organoids for gene editing using CRISPR-Cas9 [14].
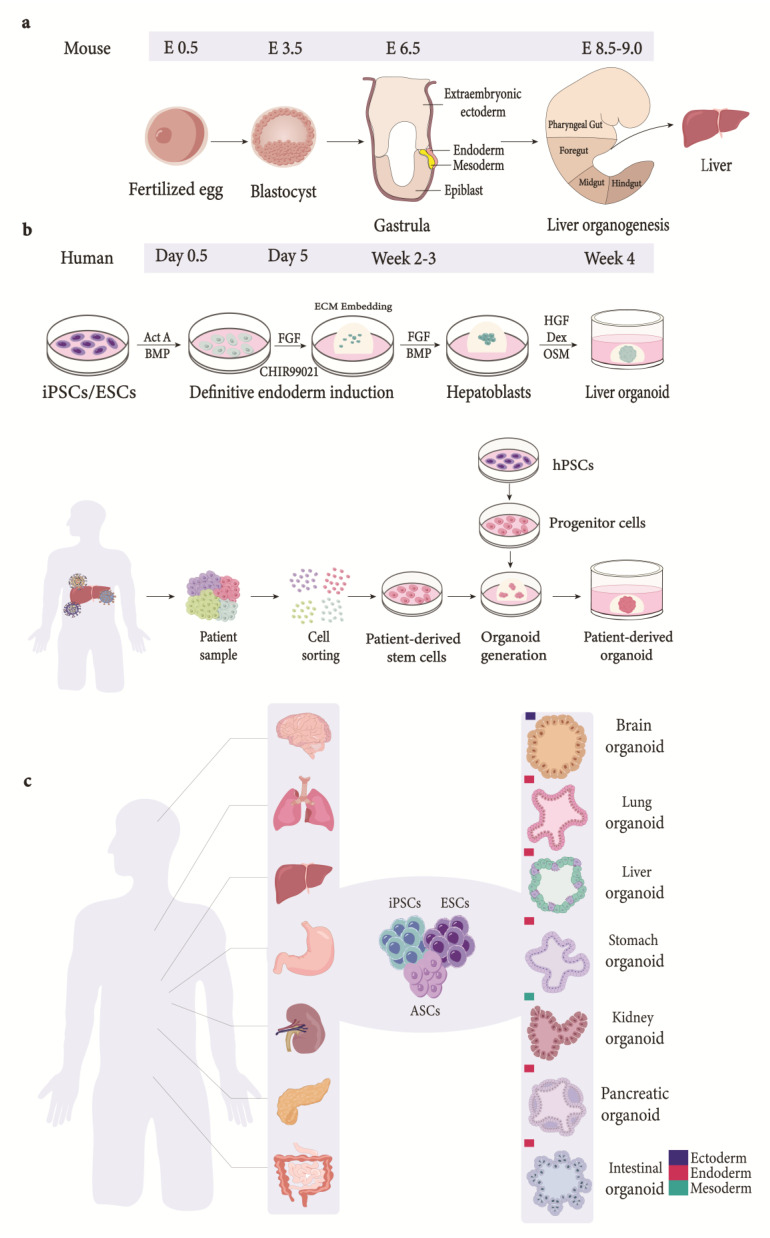
Organoids possess key characteristics of organs, such as two or more types of cells of the organ it mimics, displaying similarity in function and resembling cellular organization. Hence, an organoid can be defined as a miniature organ containing several types of cells obtained via the differentiation of stem cells or progenitor cells that self-organize and embody lineage commitment [15]. So far, organoids have been developed for organs originating from all three germ layers (Figure 2) and are being used as model systems in basic and translational research.
Figure 2.
Organogenesis and generation of liver organoids from various sources. (a) Schematic depiction of stages involved in organogenesis. Embryo formation is followed by blastocyst stage. The blastocyst consists of an outer layer of trophectodermal cells and ICM. The ICM, consisting of embryonic stem cells, further specializes into either epiblast lineage or primitive endoderm lineage during the late stages of its formation. Blastocyst is followed by gastrulation, where morphological rearrangements transform epiblast into the three germ layers: ectoderm, endoderm, and mesoderm [16,17]. The endoderm becomes patterned into anterior foregut (AF), posterior foregut (PF), midgut (M), and hindgut (H). As illustrated above, the liver is derived from PF domain of the endoderm. (b) Organoids derived from pluripotent stem cells follow a stage-wise differentiation process that recapitulates signaling pathways observed during development. The differentiation process starts by directing iPSCs/ESCs towards endodermal fate when exposed to Act A and Wnt. The cells are embedded in ECM and differentiated into hepatoblasts-like cells (progenitor cells) using FGF and BMP. Hepatoblast-like cells differentiate into hepatocyte-like cells via exposure to OSM [18,19]. Moreover, ductal organoids can be generated by modulating FGF, EGF, and Act A signaling [16]. Hepatoblasts embedded in ECM give rise to hepatic organoids. Several tissue sources have been used to generate patient-derived organoids using a number of different techniques for tissue processing. The variation in techniques used has led to non-standardized patient-derived organoid culture techniques [20]. In general, tissue-derived stem cells are dissociated into single cells and embedded in extracellular matrix to generate organoids [21,22]. (c) Generation of organoids. Organoids generated for various organs derived from the three germ layers. ICM, inner cell mass; Act A, Activin A; BMP, bone morphogenetic protein; FGF, fibroblast growth factor; ECM, extracellular matrix; HGF, hepatocyte growth factor; OSM, Oncostatin M; Dex, dexamethasone; iPSCs, induced pluripotent stem cells; ESCs, embryonic stem cells; hPSCs, human pluripotent stem cells; ASCs, adult stem cells.
The advancement in the organoid technology has its roots in the fields of stem cell biology, extracellular matrix (ECM) biology, and the reorganization of tissues dissociated ex vivo [23]. Bissel and colleagues [24] observed improved hepatocellular function in rat hepatocytes in the presence of ECM. Furthermore, mammary gland epithelia were able to develop tubules and ducts when embedded in hydrogels containing ECM [24,25]. Interestingly, coculturing gastric epithelia and fibroblast in hydrogels in the presence of ECM enabled a successful, albeit short-term, culture of stomach tissues and their differentiation into gastric mucosal cells [26]. Although these cultures had hallmarks of organoids and signified the importance of 3D culture systems, they were not self-renewable; hence, sustained long-term cultures were not possible. However, these shortcomings were overcome by culturing leucine-rich repeat-containing G-protein-coupled receptor 5 (Lgr5)-positive adult stem cells isolated from mouse intestinal tissue. These cells were able to self-renew and demonstrate morphological and functional characteristics of intestinal crypt-villus [27]. The addition of the LGR ligand, R-spondin, in the culture media mediated the up-regulation of the Wnt signaling pathway, thus facilitating stem cell maintenance and organoid formation from Lgr5+ intestinal progenitor cells [28]. Further optimization of the R-spondin-containing media led to the generation of organoids from lgr5+ progenitor cells from the colon [28], stomach [29], and liver [30]. In addition to the progenitor cells, embryonic stem cells (ESCs) have also been shown to differentiate into cortical tissues in a 3D culture [31]. This has led to the generation of cerebral organoid models that recapitulate the developmental aspects of the human brain [32]. Subsequent studies have successfully generated organoids using PSCs and tissue-specific progenitor cells to model organs derived from the ectoderm, mesoderm, and endoderm [33] (Figure 2), including the liver [12,30,34,35] (Table 1).
Ever since the human embryonic stem cells (hESCs) and induced pluripotent stem cells (iPSCs) were established [36,37,38], numerous cell types have been generated via differentiation protocols recapitulating the essence of organogenesis in vivo [39] (Table 1). The establishment of organoids over the years has added a new paradigm to modelling human diseases (Table 2). Organoids can be defined as three-dimensional structures that originate from stem/progenitor or differentiated cells and self-organize via cell-to-cell and cell-to-matrix interactions, thus recapitulating the tissue-specific structure and function in vitro [40] in a way that the traditional cell cultures and animal models lack. In this review, we discuss the importance of liver organoids and the generation of organoids from hPSCs and tissue-resident stem cells. We also discuss how organoids can further our understanding of the mechanisms involved in disease progression and provide opportunities in developing personalized therapies.
Table 1.
A general overview of the organoid technology in disease modeling for different organs.
| Organ | Organoid | Model System | Cell Source | Media and Supplements | Salient Features | Species | Refs. |
|---|---|---|---|---|---|---|---|
| Brain | Cerebral organoid | Microcephaly | hPSCs/mouse ES cells | D0; human ES media, bFGF, and ROCK inhibitor. D6; DMEM/F12, N2, Glutamax. MEM-NEAA and heparin. D11; DMEM/F12 and Neurobasal containing N2, B27 without vitamin A, 2-mercaptoethanol, insulin, Glutamax and MEM-NEAA. D15; DMEM/F12 and Neurobasal containing N2, B27 with vitamin A, 2-mercaptoethanol, insulin, Glutamax and MEM-NEAA | Cerebral organoids that recapitulate cortical development | Human/Mouse | [32] |
| Lungs | Organoids consisting of epithelia and alveoli-like structures | Fetal lung | hPSCs | D0; RPMI 1640, Activin A, and dFBS. D4; anterior foregut, Advanced DMEM/F12, N-2 and B27, Hepes, L-Glutamine, Penicillin–streptomycin, Noggin, SB431542 and CHIR99021 | Development of fetal-like lung organoids with proximal airway structures | Human | [41] |
| Liver | Functional liver bud | Liver failure model | iPSCs, MSCs, HUVECs | D0; RPMI 1640, B27 and Activin A. D6; RPMI1640, B27, human bFGF, human BMP4 | iPSCs-derived liver bud transplantation in liver failure model | Human | [42] |
| Stomach | Gastric organoids | Pyloric epithelial organoids | ASCs | Gastric culture medium; Advanced DMEM/F12, B27, N2, nAcetylcysteine, and Gastrin containing EGF, R-spondin1, Noggin, FGF10 and Wnt3A. Enteroendocrine lineage; Exendin 4 | Lgr5+ stem cells at the base of pyloric glands drive epithelial regeneration | Mouse | [29] |
| Kidney | Kidney organoids | Fetal kidney organoid | iPSCs | D0; APEL basal medium, CHIR99021 and Antibiotic–Antimycotic. D5; APEL basal medium, FGF9 and heparin. D7; APEL and CHIR99021 (1h), APEL, FGF9 and heparin. D13; APEL basal medium | Generation of multicellular fetal-like kidney organoids using small molecules | Human | [43] |
| Pancreas | Pancreatic organoids consisting of ductal and acinar progeny | Cystic fibrosis model | iPSCs | D0; BE1 (MCDB13, glucose, sodium bicarbonate, fatty acid free, BSA, L-glutamine) supplemented with CHIR99021 and Activin A. D1; BE1 with Activin A. D4; BE1 with KFG. D6; BE3 (MCDB131 with 0.44 g/L glucose, 1.754 g/L sodium bicarbonate, FAF-BSA, L-glutamine, L-ascorbic acid, ITS-X) with SANT-1, retinoic acid, LDN-193189 and PD0325901. D10; BE3 with FGF10, Indolactam V, SB431542 and glucose. D12; BE3 with Rock inhibitor. D14; BE3, FGF2 and Rock inhibitor. D18; BE3 with FGF2 and nicotinamide | Patient-derived pancreatic organoids as in vitro cystic fibrosis model | Human | [44] |
| Intestine | Intestinal organoids | Crypt-villus organoids recapitulating intestinal crypts | ASCs | Culture media; Advanced DMEM/F12, EGF, R-spondin 1, Noggin and Jagged-1 peptide. Overlay media; Advanced DMEM/F12, EGF, R-spondin 1, Noggin and Y-27632 | Lgr5+ cells can form crypt-villus organoids in vitro | Mouse | [27] |
Organoids have been generated for a number of organs as depicted in the table using pluripotent stem cells and tissue-resident stem cells. Direct differentiation of PSC-derived organoids require the induction of germ-layer specification following induction of specific cell types and finally maturation of cells via the introduction of growth factors. Organoids derived from adult stem cells require the isolation of tissue-specific stem cells. iPSCs, induced pluripotent stem cells; ES, embryonic stem cells; ASCs, adult stem cells; MSCs, mesenchymal stem cells; hPSCs, human pluripotent stem cells; HUVECs, human umbilical vein endothelial cells. bFGF, basic fibroblast growth factor; ROCK, Rho-associated protein kinase; DMEM/F12, Dulbecco’s modified eagle medium/F12; MEM-NEAA, minimum essential media–nonessential amino acids; dFBS, defined fetal bovine serum; FGF, fibroblast growth factor; BMP, bone morphogenetic protein; EGF, epidermal growth factor; KFG, keratinocyte growth factor; FAF-BSA, fatty acid-free bovine serum albumin; ITS-X, insulin–transferrin–selenium–ethanolamine.
Table 2.
Organoids and organ-on-chip in modeling liver diseases.
| Organ | Disease Category | Disease Modelled | Platform | Cell Source | Cell Repertoire in Organoid | Study Objective | Advantages | Constraints | Refs. |
|---|---|---|---|---|---|---|---|---|---|
| Liver | Metabolic diseases | NAFLD/MASLD | Organoid | ESCs/iPSCs (Human)|Fetal hepatocytes |
Hepatocytes, Stellate cells, and Kupffer cells|Hepatocytes | Investigate steatohepatitis | Patient-derived organoids in precision medicine|Precise modeling | Batch to batch variability|CRISPR-based gene editing | [18,45] |
| Organ-on-chip | Hepatocytes, Stellate cells, Kupffer cells, Sinusoidal cells (Human primary) |
Hepatocytes, Stellate cells, Kupffer cells, Sinusoidal endothelial cells | NAFLD progression to NASH | Non-invasive model consisting of all the major cells involved in NASH progression as a preclinical system | Maintain dynamic flow conditions. | [46] | |||
| ALD | Organoid | ESCs, hFLMCs (Human) |
Hepatocytes, Cholangiocytes | Development of alcoholic liver injury model that improved survivability of FRG mice after transplantation | Developed robust functional and transplantable organoids | Fetal-like characteristics | [47] | ||
| Organ-on-chip | PHH, LSECs, Kupffer cells (Human) |
Hepatocytes, Sinusoidal endothelial cells, and Kupffer cells |
Alcoholic liver disease model that recapitulates blood alcohol concentrations | Primary cells recapitulating alcoholic liver disease in a triculture system | Study lacks long-term culture | [48] | |||
| Viral infections | SARS-CoV-2 | Organoid | Progenitor cells (Human) |
Human ductal cells | SARS-CoV-2 ductal organoid model | To test antivirals | Low infection rate | [13] | |
| Organ-on-chip | - | - | - | - | - | - | |||
| HEV | Organoid | Bipotent progenitor cells (Human) |
Hepatocytes and Cholangiocytes | Development of experimental model for hepatotropic virus (HEV) infection | Identification of antiviral drugs | Liver biopsies | [49] | ||
| Organ-on-chip | - | - | - | - | - | - | |||
| HBV | Organoid | iPSCs, HUVECs, and BM-MSCs (Human) |
Hepatocytes, Mesenchymal stem cells, and endothelial cells | Development of patient-derived organoid model system for HBV infection | Compared to 2D culture systems the organoids were more prone to HBV infection | Lack of adult phenotype | [50] | ||
| Organ-on-chip | PHH and KC (Human) |
Primary hepatocytes and Kupffer cells | Microfluidic system that recapitulates HBV life cycle and innate immunity | The system provides an in-depth analysis of HBV and human immune response | Activation of Kupffer cells is required via exogenous stimulant | [51] | |||
| HCV | Organoid | - | - | - | - | - | - | ||
| Organ-on-chip | Hepatoblasts and CD8+ T cells (Human) |
Hepatocytes and T Cells | Microfluidic model to study adaptive immune response against HCV | An alternative to surrogate animal models | Lack of other immune cells. Extracellular matrix impedes T Cell and organoid contact | [52] | |||
| Primary Liver Cancer | CC | Organoid | Tumor cells | Cholangiocarcinoma cells | Biomarker identification and drug screening | Personalized drug testing | Lack of immune and stromal cells | [53] | |
| Organ-on-chip | - | - | - | - | - | - | |||
| HCC | Organoid | Tumor cells | Primary liver cancer cells | Biomarker identification and drug screening | Patient-derived organoids for personalized medicine | Model deficit in immune cells | [53] | ||
| Organ-on-chip | - | - | - | - | - | - | |||
| CHC | Organoid | Tumor cells | Cholangiocarcinoma cells and Hepatocellular carcinoma cells | Biomarker identification and drug screening | In vivo model for drug screening that mimics tumor architecture and gene expression | Lack of stromal and immune cells | [53] | ||
| Organ-on-chip | - | - | - | - | - | - |
Liver diseases modeled using organoids and organ-on-chip. Liver organoids and organ-on-chips have been successfully used to model liver diseases, identify biomarkers, and further our understanding of personalized medicine. NAFLD, non-alcoholic fatty liver disease; MASLD, Metabolic dysfunction-associated steatotic liver disease; ALD, alcoholic liver disease; SARS-CoV-2, severe acute respiratory syndrome coronavirus disease 2; HEV, Hepatitis E virus; HBV, Hepatitis B virus; HCV, Hepatitis C virus; CC, Cholangiocarcinoma; HCC, Hepatocellular carcinoma; CHC, combined hepatocellular–cholangiocarcinoma.
2. Animal Models and Divergences in Developmental Trajectories
Many aspects of biological principles are evolutionarily conserved across species; this enables animal models, such as C. elegans, D. melanogaster, and Mus musculus, which is resourceful in understanding developmental processes and disease [54]. Mice have been especially useful as disease-related models due to their physiological relevance, availability of genetic tools, small size, and rapid reproduction. Nevertheless, fundamental differences exist between species which necessitates the need for human-specific model systems in understanding human development and disease. The need for such models is ever so important in understanding the genetic variability and susceptibility to genetic diseases; for instance, neurological and neurodevelopmental disorders, and devising therapeutics. Developmental processes such as embryonic and fetal development are species-specific and differ greatly from species to species (Figure 2) with different life spans. The complex nature of the human brain is considered to be a main factor for its prolonged developmental time scale [55,56], as evident by the fact that the synapse formation and dendritic development takes place from months to years in humans. Moreover, the process of synaptic elimination continues over decades in humans, whereas cortical neurons reach maturity after 5 weeks in the mouse brain and 4 months in macaques [55].
Apart from the complexity and slower pace of the developmental process of the human brain, other aspects of human development occur at a slower pace as well. Somite development in vertebrates is controlled via the oscillation of a gene expression pattern termed as the “somite segmentation clock”, which lasts 5–6 h in humans as compared to 2–3 h in mouse [57]. Likewise, the motor neurons take 2.5 times longer to develop in humans in comparison to mice [58], which is attributed to increased protein stability and cell cycle duration in human cells [57,58]. This underlies the disparity in metabolic processes that could influence the developmental time scale in species.
The prolonged time scale for development in humans is also attributed to diversity in progeny cells [59], as evident by the evolution of the human neocortex [60], especially in the supragranular layers, which shows increased diversification in glutamatergic neurons compare to the mouse neocortex [61]. The highly divergent neuronal subtypes, especially in cortical layer 3, could render increased susceptibility to Alzheimer’s disease [62], consequently making species-specific disparities ever so important in the analysis of human diseases.
Animal models have been extensively used to help determine pharmacological and biological actions of drugs. However, in the past 20 years, numerous candidate drugs showing encouraging results in preclinical models of liver diseases, such as NASH, failed during clinical trials [63,64]. Additionally, there seems to be a lack of consensus on which NAFLD model recapitulates human liver disease more closely [65]. Furthermore, genetic models of human diseases fail to recapitulate the mechanisms found in patients [66]. The inconsistency in study design and interlaboratory variability severely limits the reproducibility of in vivo experiments [67,68].
Inter-species differences also dictate susceptibility to pathogenic diseases. For instance, Helicobacter pylori, which causes gastritis and is a known factor for causing adenocarcinoma in humans [69], can infect mice but does not cause adenocarcinoma in wild-type mice, thus limiting their use as animal models. Viruses are also known to be host-specific. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which causes COVID-19 in humans (Figure 3), does not infect wild-type mice [70]. Mosquito-borne Zika virus, which causes birth defects and microcephaly in human fetuses, only causes microcephaly in the offspring of wild-type mice when the virus is injected in the placenta or fetal brain and not when the pregnant mice are injected with the virus subcutaneously [71,72]. Lack of a proper murine infection model impedes the in vivo assessment of HBV drugs [73]. The expression of hNTCP in the murine model helps facilitate viral entry but fails to establish viral infection [74]. On the other hand, mice are resistant to HCV infection and several transgenic mouse lines have to be developed to study different aspects of HCV infection [75]. Consequently, this necessitates the use of organoid models to overcome the limiting factors that species-specific differences pose, such as developmental timing, diversity in cell types, and genetic predisposition.
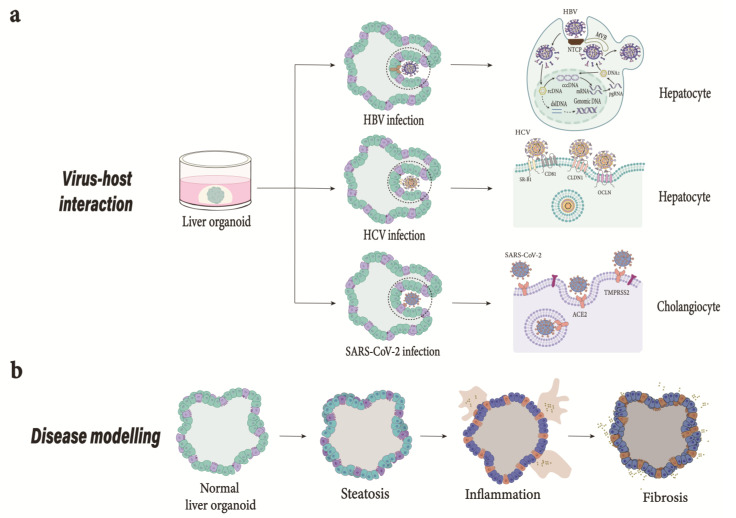
Figure 3.
Application of liver organoid models. (a) Virus host interaction. HBV; Modeling HBV infection and drug screening. The model developed can successfully recapitulate HBV infection [21]. Depiction of HBV life cycle from attachment to secretion of viral particles. HBV enters hepatocyte via a mechanism involving NTCP receptor. Once internalized, the HBV’s rcDNA genome liberated into the nucleus is converted into cccDNA, which serves as a template for transcription. The dslDNA produced can either integrate into cellular genome or convert into cccDNA. The viral mRNA transported to cytoplasm is translated into viral proteins. The pgRNA and viral polymerase are encapsulated and reverse transcribed into progeny rcDNA within the nucleocapsid. HCV; The importance of new models to study HCV is crucial due to limited animal models [52]. Organoids derived from adult stem cells and hPSCs can be used to further our understanding of HCV infection and develop antiviral drugs. HCV interaction with cell surface receptors initiates viral entry. HEV; Generation of liver-derived organoids support HEV infection and life cycle and could help develop new therapies. SARS-CoV-2; The interaction of spike protein with ACE2 receptor in the presence of TMPRSS2 facilitates the entry of SARS-CoV-2 into the host cell. COVID-19 caused by SARS-CoV-2 could mediate cell damage, dysregulate RAAS leading to decreased cleavage of angiotensin I and angiotensin II, thromboinflammation, and endothelial cell damage, and inhibit interferon signaling, i.e., depletion of T lymphocytes and production of cytokines such as IL-6 and TNFα [76]. (b) Modeling steatohepatitis in vitro. The failure of animal models in identifying translatable therapies highlights the need for improved models. Generation of organoids from patient-derived hPSCs consisting of multiple hepatic cell types successfully emulates liver-in-a-dish and can be utilized to study liver inflammation and fibrosis and identify effective drug treatments [18]. NTCP, Sodium taurocholate cotransporting polypeptide; rcDNA, relaxed circular DNA; cccDNA, closed circular DNA; dslDNA, double-stranded linear DNA; pgRNA, pregenomic RNA; ACE2, Angiotensin-converting enzyme 2; TMPRSS2, transmembrane protease, serine 2; RAAS, renin-angiotensin-aldosterone system; IL-6, interleukin 6; TNFα, tumor necrosis factor alpha.
3. Organoid Technology: The Inception of an Organ in a Dish
Understanding how ECM can impact cellular organization and behavior has been instrumental in discovering the self-organizing potential of stem cells. This is evident by alveolar structures formed by mammary epithelia in the presence of a reconstituted basement membrane [77,78]. In addition to their structural resemblance, epithelial polarization, and compartmentalization to alveoli, they remarkably emulated alveolar function, evident by the tissue-specific vectorial secretion of milk into the lumina. Although these alveolar models recapitulating the alveolar architecture could be considered as early examples of organoids, it took over two decades for the field of organoids as seen today, when a single Lgr5+ adult stem cell (ASC) was shown to give rise to miniature tissue that resembled the stomach and small intestine [27,29]. Advancement in the field of stem cell biology has led to many pioneering work using embryonic stem cells (ESCs) in developing organoids for the brain [31], optic cup [79], and pituitary gland [80]. The field of organoids really took pace when induced-pluripotent stem cells (iPSCs) and adult stem cell-derived organoids were utilized as model systems for human diseases [32,81]. Numerous organoid models have been developed since then for human organs. However, PSC- (pluripotent stem cells) and ASC-derived organoids are used in different context. Since ESCs/iPSCs give rise to all the three germ layers, PSC-derived organoids are considered developmental models. The organoid cultures follow the developmental process that an embryo goes through, such as the induction of the endoderm, ectoderm, or mesoderm mimicking embryonic and fetal development. This allows us to study the human developmental process to answer questions that have been eluding us thus far. However, PSCs fail to emulate the adult stages of tissue development, thus they can be deemed more suitable for understanding embryonic and fetal development and diseases. Organoids derived from ESCs and iPSCs show remarkable histological similarities to the developmental stages of an organ they represent [54]. Multiple developmental stages of an organ in vivo are emulated in a step-by-step protocol for the development of PSC-derived organoids [31,32,54,82]. Generally, the differentiation process starts with the expansion of PSCs and the formation of embryoid bodies [31,32,82]. This is followed by the addition of exogenous morphogens to simulate the developmental process step by step (Figure 2). For instance, the development of brain organoids starts with the formation of embryoid bodies that are introduced to a morphogen-containing medium for neuronal differentiation [31,32,83,84]. Once the fate of the cells is decided and the cells in the progenitor state proliferate, the medium is switched again to promote maturation of the neurons [83,85]. Consequently, the stage wise exposure of organoids to different conditions in vitro recapitulates the development stages of human fetal organ developmental.
How cells interact with the surrounding ECM is instrumental in the establishment of organoids. ECM for organoid cultures is generally animal-based and derived from ECM-producing tumors providing structural support in addition to growth factors and cell-matrix signals for the optimal development of organoids. There are different ways of administering ECM components; dissolve the matrix in the medium used for the organoid culture or embed the organoids in the basement membrane matrix [83,85,86]. It is worth mentioning here that some organoids have also been developed in suspension cultures in the absence of a reconstituted basement membrane [31,87,88]. These 3D structures have also been referred to spheroids due to the absence of a cell–substrate interaction. However, these structures could be referred to as organoids, as long as the aforementioned requirements are fulfilled [40].
PSC-derived organoids are used for modeling development, whereas ASC-derived organoids are crucial for understanding the process of tissue repair and maintenance in adult tissues [82]. In this regard, PSC- and ASC-derived organoids are complementary systems that represent the developmental and adult tissue stages, respectively. However adult stem cells can only be isolated from certain organs with a high regenerative capacity, such as intestinal epithelia that form organoids resembling polarized cysts; these organoids are not as complex as the organoids derived from PSCs. Nevertheless, the relative ease in establishing ASC-derived organoids makes them extremely useful for basic and clinical research (Table 3).
Table 3.
Organoids in clinical trials.
| Organ | NCT Number | Study Title | Study Status | Conditions | Study Objective |
|---|---|---|---|---|---|
| Liver | NCT05183425 | Patient-derived Organoids Predicts the Clinical Efficiency of Colorectal Liver Metastasis | Recruiting | Explore the Consistency of Drug Sensitivity Between Primary Colorectal Cancer and Liver Metastases | Evaluate consistency of drug sensitivity of primary tumor and its matched liver metastasis |
| Pancreas | NCT04777604 | Development of a Prediction Platform for Neoadjuvant Treatment and Prognosis in Pancreatic Cancer Using Organoid | Not yet recruiting | Pancreas Cancer | Survival rate of pancreatic cancer patients after they were started adjuvant chemotherapy |
| NCT04736043 | Development of a Prediction Platform for Adjuvant Treatment and Prognosis in Resected Pancreatic Cancer Using Organoid | Recruiting | Pancreatic Cancer Resectable | Overall survival rate after adjuvant chemotherapy for pancreatic cancer was started | |
| NCT05351983 | Patient-derived Organoids Drug Screen in Pancreatic Cancer | Recruiting | Pancreas Cancer |Pancreas Neoplasm|Pancreas Adenocarcinoma|Pancreatic Cancer|Pancreatic Neoplasms|Pancreatic Adenocarcinoma |
Feasibility of patient-derived organoids that can be successfully used for drug screening | |
| NCT04931381 | Organoid-Guided Chemotherapy for Advanced Pancreatic Cancer | Recruiting | Advanced Pancreatic Cancer | Measure patient response to cancer treatment, complete response (CR), partial response (PR), or stable disease (SD) | |
| NCT04931394 | Organoid-Guided Adjuvant Chemotherapy for Pancreatic Cancer | Recruiting | Pancreatic Cancer | Radiology assessment of response to adjuvant therapy | |
| NCT05842187 | In Vitro Organoid Drug Sensitivity-Guided Treatment for Metastatic Pancreatic and Gastric Cancer | Recruiting | Pancreatic Cancer|Gastric Cancer | Analyze PFS (Progressive-free survival) from initiation of treatment to the occurrence of disease progression or death | |
| Brain | NCT05772741 | Grafts of GSCs Into Brain Organoids for Testing Anti-invasion Drugs | Recruiting | Glioblastoma|Glioma, Malignant | Study anti-invasive drugs against invasive glioma stem cells co-cultured with brain organoids |
| Kidney | NCT04342286 | To Establish a Reproducible Organoid Culture Model with Human Kidney Cancer | Completed | Kidney Cancer | Establishment of patient-derived kidney tumor organoids with phenotypic stability |
| Intestine | NCT04497727 | Gut Organoid Study | Active, not recruiting | Gut Inflammation | Gut–microbiota interaction in hypertensive and normotensive reference subjects |
| NCT03256266 | Effect of Antigens or Therapeutic Agents on in Vitro Human Intestinal Organoids | Recruiting | Intestine Disease | Gluten peptide provocation to determine proliferation, apoptosis, histology, and cytokine expression | |
| NCT05294107 | Intestinal Organoids | Recruiting | Digestive System Diseases |Inflammatory Bowel Disease, Ulcerative Colitis Type|Crohn Disease | Patient-derived organoids cultured on 3D matrix gel for molecular screening to assess the effects of intestinal stress and healing | |
| NCT02874365 | Intestinal Stem Cells Characterization | Recruiting | Inflammatory Bowel Diseases | Generation of organoid models for stem cell characterization | |
| NCT05832398 | Precision Chemotherapy Based on Organoid Drug Sensitivity for Colorectal Cancer | Recruiting | Colorectal Cancer | Organoid generation for drug screening | |
| NCT05425901 | Preclinical Evaluation of Multimodal Therapeutic Strategies in Intestinal Irradiation and Inflammatory Bowel Disease from Organoids | Not yet recruiting | Radiation Enteritis|Inflammatory Bowel Diseases | Optimize organoid generation to mimic human intestine by performing scRNA-seq | |
| NCT04371198 | Patient-Derived Organoids for Rectal Cancer | Completed | Rectum Cancer | Patient-derived organoid to study rectal cancer | |
| NCT04996355 | Organoids-on-a-chip for Colorectal Cancer and in Vitro Screening of Chemotherapeutic Drugs | Recruiting | Colorecta l Neoplasms|Organoids | Chemotherapeutic drug screening based on organoid chips to predict the effect of chemotherapy against Colorectal cancer | |
| NCT05352165 | The Clinical Efficacy of Drug Sensitive Neoadjuvant Chemotherapy Based on Organoid Versus Traditional Neoadjuvant Chemotherapy in Advanced Rectal Cancer | Not yet recruiting | Neoadjuvant Therapy | Neoadjuvant therapy in organoids | |
| NCT05183425 | Patient-derived Organoids Predicts the Clinical Efficiency of Colorectal Liver Metastasis | Recruiting | Explore the Consistency of Drug Sensitivity Between Primary Colorectal Cancer and Liver Metastases | Organoids in testing drug sensitivity to primary tumor and its matched liver metastasis | |
| NCT05304741 | The Culture of Advanced/Recurrent/Metastatic Colorectal Cancer Organoids and Drug Screening | Recruiting | Colorectal Cancer | Generate patient-derived organoids to evaluate overall survival | |
| NCT04906733 | Cetuximab Sensitivity Correlation Between Patient-Derived Organoids and Clinical Response in Colon Cancer Patients. | Recruiting | Colon Cancer|Organoids|Metastatic Cancer | Association of drug sensitivity tests in patient-derived organoids and clinical outcomes | |
| Lungs | NCT04859166 | Prospective Primary Human Lung cancer Organoids to Predict Treatment Response | Completed | Lung Cancer | To successfully establish PDX model and biobanks of lung cancer organoids and determine the frequency of primary, secondary, and tertiary organoid formation |
| NCT05669586 | Organoids Predict Therapeutic Response in Patients with Multi-line Drug-resistant Non-small Cell Lung Cancer | Recruiting | Lung Cancer|Organoid|NSCLC | Generate non-small cell lung cancer organoids to predict therapeutic response in multi-drug resistant cancer and evaluate progression-free survival | |
| NCT03655015 | Patient-derived Organoid Model and Circulating Tumor Cells for Treatment Response of Lung Cancer | Recruiting | Lung Neoplasm | Establish biobank of patient-derived organoids (PDOs) to recapitulate ex vivo responses observed clinically | |
| NCT05092009 | Lung Cancer Organoids and Patient-Derived Tumor Xenografts | Recruiting | Lung Cancer | Generate normal and patient tumor-derived organoids for long-term culture and bio-banking | |
| NCT03979170 | Patient-derived Organoids of Lung Cancer to Test Drug Response | Recruiting | Lung Cancer|Organoid | Establish patient-derived organoids that are phenotypically and genetically identical to the source tumor | |
| NCT05136014 | Evaluation of the Response to Tyrosine Kinase Inhibitors in Localized Non-small Cell Lung Cancer (NSCLC) Patients with EGFR Mutation in a Patient-derived Organoid Model | Enrolling by invitation | Lung Cancer|Lung Adenocarcinoma|EGFR Activating Mutation| KRAS Mutation-Related Tumors|Non-Small Cell Lung Cancer | In vivo evaluation of osimertinib alone or in combination in patient-derived organoids | |
| Gastric | NCT05842187 | In Vitro Organoid Drug Sensitivity-Guided Treatment for Metastatic Pancreatic and Gastric Cancer | Recruiting | Pancreatic Cancer|Gastric Cancer | Pancreatic and gastric metastatic cancer organoids to study drug sensitivity and PFS (Progressive-free survival) |
| NCT05652348 | Response Prediction of Hyperthermic Intraperitoneal Chemotherapy in Gastro- Intestinal Cancer | Recruiting | Gastric Cancer|Colon Cancer|Peritoneal Carcinomatosis | Ex vivo response to dose dependent chemotherapeutic agents in organoid models | |
| NCT05351398 | The Clinical Efficacy of Drug Sensitive Neoadjuvant Chemotherapy Based on Organoid Versus Traditional Neoadjuvant Chemotherapy in Advanced Gastric Cancer | Not yet recruiting | Advanced Gastric Carcinoma | Determine clinical efficacy based on studies in advanced gastric cancer organoids by evaluating Objective Response Rate (ORR) | |
| NCT05203549 | Consistency Between Treatment Responses in PDO Models and Clinical Outcomes in Gastric Cancer | Recruiting | Gastric Cancer | Establish and optimize generation of patient-derived organoids from gastric cancer tumors |
Data taken from clinicaltrials.org *. Patient-derived organoids can identify patient-specific drug targets and thus work as a tool for personalized medicine. Links to the studies can be found in Supplementary Table S1. * URL last accessed on 20 October 2023.
4. Liver Development and Regeneration: Cues from Nature
The human liver mainly consists of epithelial cells (hepatocytes and cholangiocytes) that work synergistically with stromal, endothelial, and mesenchymal cells to perform metabolic, exocrine, and endocrine functions crucial for homeostasis. In spite of fundamental biological differences, our understanding of human liver development and in vitro differentiation of liver progenitor/stem cells come from studies in mice. The posterior foregut endoderm gives rise to embryonic liver progenitor cells (hepatoblasts) during organogenesis. Furthermore, the surrounding mesenchyme secretes growth factors such as FGF, HGF, Wnt, and BMP, which facilitates hepatoblasts to proliferate and migrate to the adjacent mesoderm-forming liver bud [89]. Moreover, hepatoblasts also specify into hepatocytes and cholangiocytes [90] as Lgr5+ hepatoblasts are bipotent in nature [22]. The lineage commitment is influenced by local signals: hepatocytes adjacent to the portal mesenchyme give rise to cholangiocytes, whereas those further away are influenced by the signals from hematopoietic cells giving rise to hepatocytes. Furthermore, the adult liver remains under homeostatic condition to maintain normal physiological functioning. However, unlike the intestine with 3–5 days of cellular turnover, it takes 60 days for cholangiocytes and 150 days for hepatocytes to self-renew [91]. Moreover, homeostatic conditions are normally maintained via the division of mature cells [92,93]. The liver has a remarkable regenerative capacity, although repeated assaults could lead to fibrosis [94]. Nevertheless, when the liver is surgically resected, the remaining healthy tissue induces liver regeneration via tumor necrosis factor-alpha (TNFα) and interleukin-6 [95,96]. Hepatocytes lose their ability to undergo cell division and proliferation during toxin-mediated damage, such as viruses, alcohol-related fatty liver disease (ALD), and non-alcoholic fatty liver disease. However, the liver still maintains regenerative capabilities by activating ductal cells to repopulate the liver [97,98,99,100]. Clearly, these studies have substantially increased our understanding of liver biology, thus facilitating the establishment of in vitro models using stem/liver progenitor cells (Figure 2).
5. Liver Organoid Models
Various human cell and tissue models have been developed such as immortalized cell lines, xenografts, and organoids derived from differentiated stem cells to overcome the limitations posed by animal models. Every model has advantages and disadvantages; for instance, cells cultured in a conventional 2D system, where cells grow in a monolayer, are easy to maintain and respond well to genetic engineering tools, making them suitable for drug discovery and understanding disease progression. Nevertheless, 2D models lack cell–cell and cell–niche interactions that are crucial to the functioning of human organs in vivo [101]. These limitations could be addressed by using patient-derived xenografts that essentially recapitulate the heterogeneity and intricacy of the tissue of origin. But, xenografts are expensive to establish and are not suitable for high-throughput screening experiments [102]. This has led to a great deal of interest in organoids, which have been in use for almost two decades largely in the field of regenerative medicine [103,104].
In one of the earliest studies of liver organoids, cells isolated from newborn rats aggregated together and formed an organoid on a low-attachment plate. The organoid consisted of hepatocytes, forming central lumen-like structures, and cholangiocytes, forming bile ducts like structures. Interestingly the organoid also managed to increase in diameter over a period of time [5].
Progenitor cells split up from the foregut endoderm and from the liver bud during hepatogenesis, which is subsequently vascularized. To recapitulate the spatial arrangement of cells during organogenesis such as hepatogenesis, Taniguchi and colleagues co-cultured human-induced pluripotent stem cells specified into hepatocyte-like cells with mesenchymal and endothelial cells to form a liver bud. The human iPSCs after differentiation into hepatic endodermal cells using activin A followed by bFGF/BMP4 formed organoids after plating with endothelial cells and mesenchymal stem cells on Matrigel [42] (Figure 2). These organoids also developed blood vessels that connected to the host vessels within 48 h when transplanted into mouse models. Moreover, they were able to perform human-specific liver function and rescued mice from drug-induced liver failure [105].
5.1. Adult Stem Cell-Derived Liver Organoids
Ever since the establishment of crypt-villus organoids from LGR5+ stem cells [27], organoids have been developed from tissue-specific stem cells for various organs using a similar approach. For instance, hepatotoxin (carbon tetrachloride)-induced liver injury causes the Wnt-regulated LGR5+ stem cells to appear in the surroundings of bile ducts [30]. These cells give rise to cyst-like structures when cultured in the presence of Wnt agonist RSPO1. The cells could differentiate into hepatocytes in vitro and repopulate Fah−/− mice upon transplantation [30]. Moreover, EpCAM+ biliary cell-derived human adult liver organoids were found to be genetically stable even after expansion for over a year in vitro [12]; consequently, these models could be used as indispensable tools in regenerative medicine, drug discovery, and understanding liver diseases. Nevertheless, the organoids are essentially epithelial stem cells that need to be differentiated into hepatocyte-like or cholangiocyte-like cells to be functional in vivo or in vitro [12].
5.2. Liver Organoids Derived from Primary Hepatocytes
The ability of hepatocytes to proliferate in vivo has been exploited to generate organoids that expand for months in vitro [34,106]. In one such study, organoids generated from mouse primary hepatocytes and human fetal hepatocytes formed ‘grape like’ structures normally seen in organoids developed from hepatocytes, as opposed to cyst-like structures formed by biliary epithelial organoids [34]. Interestingly, the organoids developed from hepatocytes exhibited regenerative characteristics after partial hepatectomy and had functional similarity to primary hepatocytes [34]. In another study, organoid models were developed to recapitulate liver cancer [107]. Intriguingly, inflammatory cytokines have been shown to expand organoids in vitro [35]. The expression profile of these organoids exhibited similarity to proliferating hepatocytes and repopulated livers of Fah−/− mice [35]. Perhaps these protocols could be used to further optimize the culture conditions [34,35] for the expansion of organoids derived from an adult human liver. Moreover, hepatic organoids have also been derived from fibroblasts differentiated into hepatocytes by inducing the expression of hepatocyte-specific transcription factors [107]. However, PSC-derived hepatic organoids with increased generative capacity and patient specificity are advantageous over ASC-derived organoids for disease modelling and drug discovery [16].
5.3. hPSC-Derived Liver Buds
The ability of hPSCs to self-renew and differentiate into different types of cells has led to the development of numerous organoid models for liver and multi-organ organoids, where multiple cell types represent different human organs [40]. The methodology that was first used to develop liver organoids in vitro involved the generation of liver buds [42], where iPSC differentiated into hepatic endoderm cells were co-cultured with MSCs and human umbilical vein endothelial cells to emulate liver organogenesis [89]. Interestingly, the cells organized into a three dimensional liver bud on Matrigel had a gene expression profile similar to mouse embryonic liver buds [42]. The buds were able to vascularize and rescue a liver failure mouse model when engrafted [42]. Later, the same protocol was further improved and liver organoids were developed entirely from iPSC-derived endoderm, mesenchymal, and endothelial progenitor cells [108]. The organoids could be used as a potential source of cells for patients in regenerative medicine using iPSCs from HLA-matched donors and a differentiation medium that is of clinical grade. Nonetheless, the complexity of the co-culture system and a lack of maturity are limiting the clinical application of liver buds [42,108].
5.4. Cholangiocyte Organoids
Numerous studies have utilized signaling cues to direct the differentiation of hPSCs into cholangiocyte organoids, hepatic organoids, or hepatobiliary organoids having multiple types of cells [109]. The developmental signaling cues have been used to guide the differentiation of hPSCs into definitive endoderm followed by differentiation into definitive endoderm and ultimately into cholangiocytes with the induction of the notch signaling pathway [110]. The cholangiocytes were able to self-organize into organoids with structural similarities to the ductal structures found in the liver and expressed biliary marker genes [111,112,113] such as apical sodium-dependent bile acid transporter and cystic fibrosis transmembrane conductance regulator-mediated fluid secretion when embedded in Matrigel [112]. Interestingly, the organoids derived from human primary cholangiocytes retain plasticity and resume the expression of the transcriptional signature genes found in mature cholangiocytes when injected in the intrahepatic ducts of a liver undergoing ex vivo normothermic perfusion (EVNP), thus regenerating the functional biliary tree [114]. This proof-of-concept study opens new avenues in the field of regenerative medicine and disease modeling.
5.5. hPSC-Derived Hepatobiliary Organoids
Multi-cellular hepatobiliary organoids that consist of hepatocytes and cholangiocytes exhibiting bile duct-like morphology have also been developed by several groups [19,115,116], recapitulating the hepatic developmental process of an embryo. These organoids exhibiting the expression of hepatogenic genes and hepatobiliary functions were used to identify the role of JAG1 mutations in the development of liver disease [19]. In another study, iPSC-derived EpCAM+ endodermal cells were used to generate hepatic organoids that were able to expand for over a year and were used to characterize the role of the argininosuccinate synthetase (ASS1) enzyme in citrullinemia type 1 disease [117]. Hepatobiliary organoids have also been generated in a novel 3D suspension culture where the organoids self-organize and form functional bile canaliculi [116] and are thus useful models for studying cholestasis induced by drug or disease. The fact that these organoids are generated in a suspension culture in the absence of a matrix broadens their scope in the realm of cell therapy and drug screening [116].
5.6. hPSC-Derived Complex Liver Organoids
In a study aimed at recapitulating the complexity of the human liver by modeling steatohepatitis and fibrosis, pluripotent stem cell-derived organoids consisting of hepatocyte-like cells, Kupffer-like cells, and stellate-like cells were generated [18]. The organoids were able characterize steatohepatitis and fibrosis when treated with free fatty acids (FFA) for 5 days [18]. The protocol was further optimized to generate organoids with distinct bile canalicular lumen-like structures [118]. High-throughput screening was performed to assess the effects of drugs on liver injury [118]. The fact that these organoids were generated from PSCs differentiated into foregut progenitor cells which can be cryopreserved, and the induction of multiple cell types that took place simultaneously under the same culture conditions are the main advantages of this protocol. Further characterization using high-throughput screening can help us to better understand the developmental process of the human embryonic liver.
Hepato-biliary-pancreatic (HBP) organoids generated by co-culturing SOX2+ anterior spheroids in close proximity with CDX2+ posterior spheroids from hPSCs in the absence of signal cues were used to understand multi-organ communications during organogenesis [119]. The HBP organoids developed into multi-organ anlages consisting of hepatic and pancreatic tissues with connecting ducts [119]. This model provides an efficient and accessible way to understand the complex process of endoderm organogenesis in vitro. Models like these in combination with technologies such as organ-on-chip and tissue engineering could be developed to recapitulate organ-to-organ interaction and communication during the developmental process and disease onset [120,121,122].
6. Liver Organoid Validation
Both human and mice tissue samples have been used to establish cholangiocyte [12,30] and hepatocyte [34] organoids. Liver organoids are assessed based on measuring passaging time and proliferating organoid cells. The expression of hepatocyte-related markers (for example ALB, HNF4A, and MRP4) and cholangiocyte-related markers (such as KRT19, KRT7, and SOX9) are assessed using real-time quantitative PCR, and scRNA-seq on the RNA level and immunofluorescence staining is performed on the protein level. Liver progenitor makers (including LGR5) are often used to evaluate the differentiation state of organoids and CYP3A4 and CYP3A11 are used to assess organoid functionality on the RNA level. Moreover, low-density lipoprotein uptake, albumin secretion, assessment of glycogen accumulation with acid–Schiff staining, bile acid production, and CYP3A4 activity are observed to corroborate hepatocyte-specific functionality. Hepatocyte and cholangiocyte morphology is also examined using a transmission electron microscope (TEM). Ultimately, the integration and functionality of organoids observed in FRG immune-deficient mice potentially determines the functionality of generated organoids [30,35].
7. Validation of Tumor Organoids
It is imperative for the patient tissue-derived tumor organoids to maintain genomic, functional, and physiological profiles of the tissue of origin when compared. Comparing histological and immunohistochemistry profiles [123] of tumor-derived organoids with the tissue of origin as well as validation using genomic and transcriptomic profiles [124,125]. Tumor-derived organoids can be easily compared with origin tissue for cellular organization, protein expression, and tissue structure. Likewise, genomic and immunohistological analyses are used to validate tumor types and subtypes in PLC and other tumors [53,126,127]. Moreover, the confirmation of whether organoids have diverged at the genomic level is also important. In this regard, several studies conducted have shown that organoids retain mutations and copy number variations observed in parent tissue [124,125]. Nevertheless, sampling bias, spatial diversity, and intra-tumor heterogeneity could purportedly lead to greater genomic divergences [128,129]. Moreover, culture conditions might selectively favor certain clones, thus altering clonality over a period of time [127].
8. Application of Liver Organoids in Disease
The presence of hepatocytes, cholangiocytes, Kupffer cells, and endothelial cells makes liver a heterogenous organ [130]. Liver diseases are caused by the interplay between the environment and genetic factors, which is a manifestation of impaired communication between cell-cell and cell-environment [131,132,133]. Therefore, generating liver organoids with multiple cell types is imperative for modeling liver diseases, such as non-alcoholic fatty liver disease (NAFLD) and alcoholic liver disease (ALD).
8.1. Modeling Alcoholic Liver Disease
Consumption of high levels of alcohol has led to an increase in alcohol liver disease (ALD) and its related comorbidities globally. A wide range of liver pathologies including steatosis, fibrosis, cirrhosis, and even hepatocellular carcinoma are manifestations of ALD [134]. Unfortunately, due to limited access to liver tissue samples and a lack of reliable in vitro models until recently, there is no FDA-approved treatment for ADL [135]. However, a model system to recapitulate ALD in vitro was developed in 2019 by co-culturing hepatic organoids derived from hESCs with mesenchymal cells from a human fetal liver. In addition to the complex architecture displayed by these organoids, they exhibited functional similarity to mature hepatocytes by expressing enzymes involved in ethanol metabolism, inflammation, and fibrosis when treated with ethanol for a week [47]. ALD manifestations like oxidative stress and dysfunctional mitochondria were also observed in these organoids [47], making this system an efficient tool for drug discovery. Nevertheless, the addition of resident macrophages such as Kupffer cells would further improve the system.
8.2. Modeling Non-Alcoholic Fatty Liver Disease
The lack of approved pharmacological interventions to treat NAFLD (non-alcoholic fatty liver disease) and NASH (non-alcoholic steatohepatitis) could be attributed to the absence of proper animal and cellular models and limited access to liver biopsies from patients. To recapitulate the liver architecture and liver pathologies such as inflammation and fibrosis as observed in NAFLD, multicellular liver organoids consisting of stellate-like, hepatocyte-like, and Kupffer-like cells were developed and treated with FFA [18]. Numerous other models have been developed to model liver diseases (Table 2).
Moreover, the treatment of organoids derived from a patient with Wolman disease, a genetic disorder which leads to steatohepatitis, with FGF19 rescued the cells [18]. Treatment with FFA induces a gene expression pattern similar to NASH in hepatobiliary organoids consisting of functional bile canaliculi. Furthermore, disruption of canaliculi system is observed in organoids recapitulating NASH [116]. The model was further improved when hiPSC-derived vascularized organoids consisting of hepatoblasts, MSCs, endothelial cells, and stellate cells were developed and exposed to FFA to mimic steatosis, inflammation, and fibrosis, as observed in NAFLD [136].
Generation of bipotent ductal organoids has also been achieved from liver biopsies of patients with NASH. These organoids exhibited increased lipid accumulation, lower levels of albumin, decreased ability to proliferate, and higher expression of cytochrome p450-related genes in comparison to the organoids derived from healthy livers [137]. The organoids thus generated mimicked hallmarks of NASH. Additionally, organoids from NASH mouse models with varying degrees of disease severity exhibited fibrosis and pro-inflammatory responses [138]. Patient-specific organoids generated from biopsies can potentially open new avenues in the field of precision medicine for liver diseases.
Interestingly, an individual’s predisposition to steatosis and its severity is dependent on the interplay between the environment and genetics [131,139]. Numerous genetic variations are associated with the progression of NAFLD [140], of which patatin-like phospholipase domain-containing protein 3 (PNPLA3) Ile148Met polymorphism is the most significant one [141]. Liraglutide, elafibranor, or momelotinib are shown to rescue spheroids consisting of immortalized hepatic stellate cells (LX-2) and HepG2, that are homozygous for the PNPLA3 Ile148Met variant, from fat and collagen accumulation when exposed to FFA [142,143]. A similar strategy was used to understand the association of NAFLD with transmembrane 6 superfamily member 2 (TM6SF2) Glu167Lys and membrane-bound O-acyltransferase 7 (MBOAT7) [144,145]. Patient-derived organoids can potentially be used to assess the effects of genetic polymorphism solely on NAFLD progression to NASH without the interference of environmental factors.
8.3. Organoids in Viral Infection
Microbial diseases are taking a toll on human health globally. The health emergency caused by Zika virus in addition to the recent COVID-19 pandemic are stark reminders of how infectious agents pose a threat to human health worldwide. Organoids can be instrumental in understanding infectious diseases where research on animal models is hindered by species-specific differences. Since wild-type mice are not susceptible to SARS-CoV-2, the COVID-19 pandemic resulting in almost 7 million deaths has increased the prospects of using organoid models for disease modeling [70] (Figure 3). This is attributed to the fact that organoids are ideally suited to study the implications of viral infection on multiple organs [146]. Although COVID-19 caused by SARS-CoV-2 mostly affects the lungs and upper respiratory tract, it has also been found to affect the liver, blood vessels, heart, intestine, kidneys, and even the brain. A number of organoid and organ-on-chip models have been developed to mimic the human respiratory tract for studying COVID-19 and tissue response [146] (Table 2). While the angiotensin-converting enzyme 2 (ACE2) receptor for SARS-CoV-2 is ubiquitous in the respiratory tract, the expression is varied with a higher expression found in the nasal epithelial of the upper respiratory tract compared to bronchioles and alveoli [147]. Organoids consisting of cholangiocyte progenitors expressing ACE2 and transmembrane serine protease 2 (TMPRSS2), an activator of the viral spike protein, are shown to be susceptible to SARS-CoV-2 infection [13]. A section of patients have exhibited multi-organ damage [148,149,150]; notably, liver damage has emerged as a co-existing symptom, as observed in a recent epidemiological study carried out in Shanghai (China), where 75 of 148 COVID-19 patients had abnormal liver functioning evident by higher amounts of alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and total bilirubin (TBIL) [151]. The fact that a significant proportion of cholangiocytes have been found to be ACE2+ in healthy livers [152] renders them susceptible to SARS-CoV-2. In a recent study by Zhao et al., human liver ductal organoids were found to be susceptible to SARS-COV-2. Moreover, the organoids also supported viral replication [13]. Consequently, the infection induced apoptosis in cholangiocytes via the CD40 molecule (CD40), caspase recruitment domain family member 8 (CARD8), and serine/threonine kinase 4 (STK4). Moreover, performing gene set enrichment analysis (GSEA) showed disruption in cell junction enrichment genes, thus interfering with the role of ductal epithelia as a permeability barrier [13]. Interestingly, SARS-CoV-2 also significantly decreases the expression of solute carrier family 10 member 2 (SLC10A2) and the cystic fibrosis transmembrane conductance regulator (CFTR), which are involved in bile acid transport. This implies the damage to cholangiocytes as a major factor in liver injury in COVID-19 patients [13].
Mild-to-moderate steatosis and lobular/portal activity has also been observed in liver biopsy specimens obtained from patients deceased due to severe COVID-19 infection, suggesting COVID-19 or drugs as a source of liver injury [153]. The injury could be attributed to a number of reasons. Inflammation due to COVID-19 could potentially trigger the immune system causing liver damage [154], as higher levels of the C reactive protein (CRP), serum ferritin, LDH (lactate dehydrogenase), D-dimer, IL-2, and IL-6 are found in patients with severe COVID-19 infection [155]. The presence of ACE2+ cells in liver makes it a potential target, thus leading to viral-associated cellular cytotoxicity [152]. However, injury due to direct viral replication has yet to be observed. Anoxia-induced hypoxic hepatitis has been frequently observed in severe cases. Finally, drug-induced liver injury has also been observed in patients. The lack of knowledge about the effectiveness of anti-viral drugs being used against COVID-19 has led to the administration of numerous anti-viral drugs such as lopinavir/ritonavir, remdesivir, chloroquine, tocilizumab, uminefovir, and Traditional Chinese medications. This has led to an increase in drug-related liver injuries [156]. SARS-CoV-2 may also exacerbate pre-existing chronic liver diseases, making patients with these conditions more prone to liver damage [157]. Nevertheless, severe liver injuries are rare [156]. The implication of non-alcoholic fatty liver disease (NAFLD) in patients has also been observed in a recent study [158]. Over 50–75% of patients with COVID-19 and NAFLD have some form of liver injury, although most of the injuries were mild patients with NAFLD who are more prone to progress to the severe form of COVID-19 infection and take longer to recover [158].
Organoids have also been used for testing antiviral drugs for hepatic viruses with narrow tropism. Organoids derived from fetal and adult liver have been successfully used for the replication of hepatitis E virus (HEV), a single-stranded RNA virus. Liver-derived organoids fully supported the life cycle of HEV virus and the apical secretion of HEV particles was observed when the organoids in the transwell system were directed toward polarized monolayers. Robust host responses were triggered by the viral replication when genome-wide transcriptomic analyses were performed. The successful recapitulation of HEV infection in host-derived organoids and drug screening identified brequinar and homoharringtonine as potent inhibitors of even a ribavirin-resistant HEV variant with G1634R mutation [49]. Additionally, in vitro host virus interaction was also observed in iPSC-derived liver organoids and hepatitis B virus (HBV), leading to the downregulation of hepatic genes such as HNF1A, ALB, G6PC, CYP3A7, CYP2C9, and RBP4 in organoids infected with HBV [50]. Moreover, EpCAM+ stem cells isolated from liver resections used for the generation of liver organoids have been successfully co-cultured with patient-derived CD8+ T cells using a microfluidic technique to recapitulate the patient-specific immunological response against HCV. CD8+ T cells successfully killed liver organoids that were pulsed with HCV non-structural protein 3 (NS3)-specific peptide [52].
8.4. Tumor-Derived Organoids
A wide variation has been observed in the past decade in organoid derivation, including preferred tumor source and the downstream processes. Primary tumors [159], metastasized tumors [160], circulating tumor cells [161], and tumor cells isolated from liquid effusions [162], including liquid and solid biopsies, autopsies, and surgical resections [163], have all been used to generate tumor organoids. Samples once collected are processed before encapsulation in the 3D matrix for the downstream cultures (Figure 2 and Figure 3). The two most prevalent strategies for pre-processing the tissue samples currently used are (1) the dissociation of tissue samples into single cells followed by encapsulation and (2) mechanical and enzymatic mincing of tissue samples into smaller fragments and encapsulation of tumor fragments that are a few millimeters in size. An alternate method used to generate cancer organoids is to induce driver mutations that recapitulate cancer onset and progression. Although each strategy for the derivation of the organoid culture potentially helps in demystifying certain unknown aspects of cancer biology, a lack of standardization puts their use in clinical research under jeopardy [20]. Liver cancer lacks dependable in vitro models to recapitulate cancer pathophysiology. Culture systems as mentioned above have been used for the long-term expansion of primary liver cancer (PLC) organoids derived from common PLC subtypes: cholangiocarcinoma (CC), hepatocellular carcinoma (HCC), and combined hepatocellular–cholangiocarcinoma (CHC) tumors (Table 2). These PLC-derived organoids maintain the genetic landscape and histological architecture of the parent tumors even after a long-term culture. This has also been observed in xenograft studies for PLC-derived organoids. The organoids possessed tumor-specific biomarkers and were instrumental in the identification of ERK inhibitor SCH772984 as a therapeutic agent for liver cancer [53].
9. Organ-on-Chip
The fact that the data obtained from animal models regularly fail to predict the outcome of human trials and the lack of preclinical models that are human-relevant call for finding alternatives to animal models [164,165,166,167,168]. This has also led to fewer effective drugs and an unsustainable rise in healthcare [169]. Organ-on-chip, a microfluidic culture device that recapitulates organ physiology and function in vitro, represents an alternative to the animal model. These microphysiological systems come in various shapes and sizes that consist of hollow channels lined by organ-specific cells under dynamic fluid flow (Figure 4). Multi-organ systems, such as body-on-chip, can also be created by coupling two or more chips fluidically to mimic the physiology of the human body. Advancement in stem cell technology and patient-derived organoids has opened new possibilities in the field of personalized medicine. As the design and functionality of microfluidic devices improve, the next step would be to prove the advantage of microfluidic systems over animal models [169].
Numerous liver chip designs to model hepatotoxicity, inflammation, drug–drug interactions, and infection have been developed in this regard. Organ-on-chip consisting of multiwells lined by hepatocytes and Kupffer cells mimicked the glucocorticoid hydrocortisone breakdown to phase I and phase II metabolites in correspondence with the human data [170]. In another similar system based on microfluidic liver chip IL-6-induced inflammation was shown to suppress cytochrome P4503A4 isoform (CYP3A4) activity, increase secretion of the C-reactive protein, and reduce IL-6 receptor shedding [171]. Treatment with tocilizumab (anti-IL-6 receptor monoclonal antibody) modulated the activity of CYP3A4, therefore altering the metabolism of simvastatin hydroxy acid and thus emulating the human drug–drug interaction which is otherwise not observable in 2D models. Another interesting study explored the effects of human population variability on hepatic drug metabolism using multiple liver chips each lined by hepatocytes from a different donor [172]. Analysis of metabolic depletion profiles of six drugs confirmed the existence of substantial inter-donor variability with respect to the gene expression levels, drug metabolism, and other hepatocyte functions. Importantly, clearance values predicted on-chip correlated well with those observed in vivo, and a physiologically based pharmacokinetics model developed for lidocaine successfully predicted the observed clinical concentration–time profiles and associated population variability. The liver chip has also been used for modeling viral infection. Hepatitis B virus infection of the hepatocyte-lined liver chip successfully modelled the viral life cycle including the replication and maintenance of covalently closed circular DNA (cccDNA) [51]. Additionally, the model recapitulated the host cytokine and innate immune response observed in patients infected with HBV [169].
10. Organoids in Precision Medicine
Human tumors can be used for the generation of organoids as an individualized precision medicine approach in cancer treatment [173] (Figure 4). The advancement in the field of 3D culture has made it possible to generate organoids for many epithelial-adult and pediatric tumors. Patient cancer-derived organoids successfully retain the genetic, histopathological, and drug response of original tumors and can be generated with a higher success rate compared to 2D cultures [173,174]. This allows for individualized targeted therapies [175]. Nevertheless, limitations like higher costs and varying success rate need to be overcome for cancer organoids to transition from the bench to the bedside [173,174]. Additionally, a lack of tumor microenvironment that includes endothelial cells, fibroblasts, stromal, and immune cells potentially limits their use in predicting patient responses. On the other hand, species-specific differences in immune response limits the use of xenografts due to their reliance on the immune system of the mouse. Therefore, complementing tumor organoids by immune cells could potentially overcome the shortcomings of organoid technology and help better understand the role of the immune system in human cancer [176].
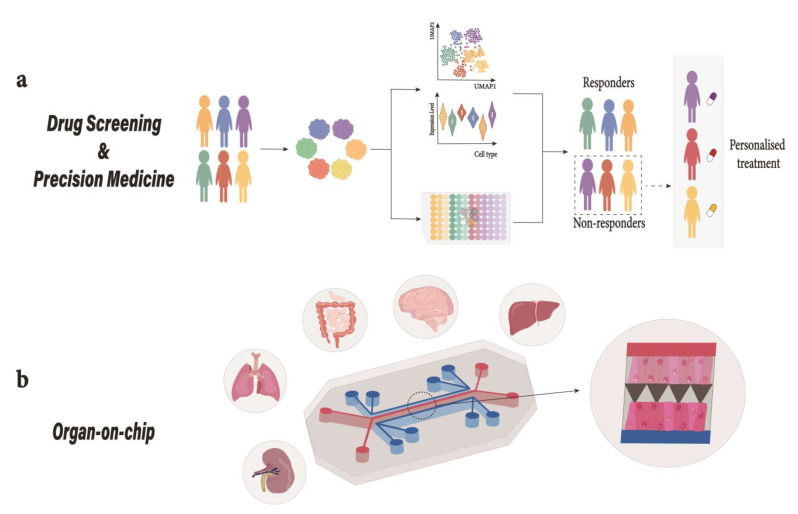
Figure 4.
Organoids in translational medicine. (a) Drug-Screening; Patient-derived organoids can be used in a high-throughput drug screening platform to study DILI. These pre-clinical platforms can be used as diagnostic tools and predictive models for drug discovery [177]. (b) Organ-on-chip; Depiction of a mechanically actuable two channel organ chip fabricated using soft lithography from PDMS with two channels parallel to each other separated by a microporous membrane. Cells from different tissues are cultured on top and bottom of the extracellular matrix (ECM)-coated microporous membrane to emulate tissue–tissue interaction where air introduction above the epithelium can take place to recreate air-liquid interface (as observed in lungs) or the channel could be used for fluid perfusion [169]. Human liver organoid-based chips have recently been used for DILI risk assessment [177]. DILI, drug induced liver injury; PDMS, polydimethylsiloxane.
There are two main approaches to co-culturing immune cells with cancer organoids. One is a holistic approach where endogenous immune cells present in the tumor are preserved and truly depict a patient immune response to a particular tumor; alternatively, the other is a reductionist approach that involves patient tumor organoids co-cultured with immune cells propagated separately [176,178], allowing for better understanding of tumor–immune interaction even though the immune cell repertoire is limited. One such example includes the co-culturing of peripheral blood-derived T cells with colorectal and non-small cell lung cancer organoids [179]. A potential use of these complex models is to study immunotherapy regimens, a new class of therapy that utilizes patient’s immune system to fight tumor cells [178]. Organoids generated from patient tumor have been co-cultured with genetically engineered chimeric antigen receptor (CAR) T cells expressing antigen receptors specific to the antigens present on cancer cells [180]. CAR-T therapy has so far been successful against hematological malignancies; however, treatment of solid tumors, including liver cancer, remains a challenge due to a lack of specific antigenic targets, tumor heterogeneity, and the immunosuppressive tumor microenvironment [181,182]. Organoid technology could also be harnessed to produce immune cells for immunotherapy in addition to modeling different cancer types [183]. Organoid-derived cells can integrate, and under certain circumstances, restore organ function [184]. Disease treatment using organoid-derived cells could potentially be a reality in the near future; in fact, exciting new opportunities await us with the beginning of the first clinical trial for head and neck cancer treatment using organoid-derived salivary gland cells (ClinicalTrials.gov Identifier: NCT04593589).
An approach to predicting the efficacy of individualized treatments is to use patient-derived organoids (Table 3). For instance, organoids derived from patients with cystic fibrosis (CF) have been used to predict the effectiveness of the treatment [81,82,185]. Owing to the self-renewable characteristics of organoids, they could potentially serve as cryopreserved biobanks of both healthy and diseased individuals. This could be made possible with the establishment of biobanks, such as HUB organoids by Hubrecht Institute. Such biobanks can help with the development of personalized treatments, drug discovery, and identifying new diagnostic markers for liver diseases [159,186]. As a matter of fact, NAFLD progression differs among humans [131,139], and so individuals at risk of developing NAFLD could be identified by generating organoid models with genetic variations from different sources (Figure 3). Moreover, the organoids could also be used for developing precision medicine and predicting drug-induced genetic and nongenetic risk factors that lead to acute liver failure [187,188].
11. Gene Therapy and Regenerative Medicine
Gene editing tools such as CRISPR-Cas9 used in cell/animal models could potentially prevent or rollback phenotypes associated with genetic diseases [189]. Modern genome editing techniques in conjunction with stem cell technology are being used to generate diseased organoid models from isogenic cell lines as well as organoids to treat metabolic diseases due to genetic mutations [190,191]. One of the key advantages of generating patient-specific organoids is to overcome allograft rejection. The expression of UCP1 in human white preadipocytes using CRISPR-Cas9 led to the generation of brown-like adipocyte cells that improved glucose tolerance and insulin sensitivity in obese mice upon transplantation [192]. Likewise, the deletion of NRIP1, a thermogenic suppressor gene, using CRISPR-Cas9 and subsequent implantation into obese mice enhanced glucose tolerance and decreased liver triglyceride levels in addition to decreasing adiposity [193]. Interestingly, Alagille syndrome and Wilson disease-related mutations have been successfully reverted to the wild type using CRISPR-Cas9 base editing technology in patient-derived organoids [19,194], thus opening new avenues in approaching liver-related metabolic disorders. Even though an orthotopic liver transplant is marred by the high risk of immune rejection and a lack of donors, it is still the only viable option for late-stage liver failure [195]. A viable alternative approach is to generate liver organoids consisting of complex structures that are able to imitate liver function. In fact, liver organoids have shown promising results when engrafted into the mice liver [34,35,42,108].
12. Current Limitations of the Panacea Illusion
Even though organoid models have been instrumental in providing insights into human-centric aspects of development and disease and have the potential to radically revolutionize drug discovery and contribute toward clinical treatment, albeit with some caveats that need consideration. Hypoxia and a lack of access to nutrients result in necrotic cores in large organoids; this is mainly due to the absence of a functional vascular system in organoids [196]. Efforts are underway to increase the supply of oxygen and nutrients to the organoid core by introducing blood vessels [197]. Apart from being a source of transportation, blood vessels play key role in delivering signaling cues to facilitate cell migration and differentiation [198]. Varying degrees of success has been achieved in generating networks of blood vessels and endothelial cells by employing different strategies including organoid transplantation into mice models [197], thus leveraging endogenous cells or by co-culturing organoids with exogenous endothelial cells [198]. However, unless these co-cultures could be established in a robust and cost-effective way, it is essential to remove stressed cells to avoid bias from oxidative stress [199].
A disadvantage of organoid models is the requirement of exogenously supplemented animal-derived ECM which severely limits their clinical use and hinders transplantation into humans. Matrigel, which is purified from Engelbreth–Holm–Swarm (EHS) sarcoma, is the most widely used basement membrane that contains a considerable amount of ECM components, primarily collagen IV, laminin, and entactin. Additionally, it contains growth factors such as insulin-like growth factor 1 (IGF-1), EGF, TGF-b, and platelet-derived growth factor (PDGF). The presence of thousands of proteins, lot-to-lot variations in composition, and differences in the mechanical aspects of Matrigel preparations results in batch-to-batch divergences in organoid cultures [88]. Moreover, there are ethical considerations regarding Matrigel production which requires growing tumors in mice. This has led to efforts being made to replace Matrigel with synthetic hydrogels, decellularized ECM from organs, and isolated ECM proteins [88,200]. Decellularized ECM purportedly retains intrinsic signaling and mechanical cues; however, the availability of human organs limits their use [88]. Synthetic hydrogels or specific components of ECM are also used as more defined approaches [200]. Particularly, unprecedented flexibility is introduced in organoid cultures via synthetic hydrogels as multiple strategies could be used to modulate their chemical and mechanical attributes; for instance, the generation of localized complex gradients consisting of growth factors. Nevertheless, further technological development is required to utilize the full potential of synthetic hydrogels [88].
Altogether, organoids models have led to a great degree of understanding into the developmental processes and disease, especially the mechanisms of genetic diseases. They have been vital in demonstrating how human organs are affected by infectious diseases and have been instrumental in generating valuable insights during Zika virus and the COVID-19 pandemic. Organoids have more recently entered clinical setups. The first organoid-based personalized medicine regimen in the Netherlands consisted of rectal organoids from CF patients to predict their response to various drugs [185]. Such approaches would more likely be used for other diseases including cancer in the near future. Similarly, transplantation of salivary gland-derived cells (ClinicalTrials.gov Identifier: NCT04593589) will open new opportunities in the field of cell replacement therapy. The future looks bright for the field of transplantation and regenerative medicine. Transplantation of neuronal progenitor cells, skin grafts for burns, and endless other possibilities could revolutionize the field of regenerative medicine.
13. Conclusions
A lack of in vitro models that reliably recapitulate human liver physiology has been a limiting factor in the transition from basic to translational research. Organoids derived from hPSCs and/or patient biopsies are excellent platforms to study disease development and progression and provide technological advancement in the realm of personalized medicine. Further advancements are required in current organoid technology by improving cellular composition to better emulate liver heterogeneity and function. The insights gained from organoids can further our understanding of liver development and have the potential to improve personalized therapy and drug development.
Acknowledgments
All the authors thank NNSFC for funding this study.
Supplementary Materials
The supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms242115921/s1.
Author Contributions
W.I., Y.W. and X.Z. contributed to the gathering of the data and the writing of the manuscript; W.I., Y.W., X.Z. and P.S. contributed to the editing and revising of the manuscript; W.I. and Y.W. drafted the figures; W.I., X.Z. and P.S. contributed to the funding acquisition. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Informed consent was obtained from all the subjects involved in this study.
Data Availability Statement
The links to data available in Table 3 can be found in Supplementary Table S1. Any other information related to the article can be obtained upon request.
Conflicts of Interest
Authors declare no competing interest. The funding body played no role in the design, data collection, interpretation, the analysis of the study, and the writing of the manuscript.
Funding Statement
The study was funded by the National Natural Science Foundation of China, Grants no. 81950410640 and 81870432.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Lanza R., Gearhart J., Hogan B., Melton D., Pedersen R., Thomas E.D., Thomson J., Wilmut S.I. Essentials of Stem Cell Biology. Elsevier Inc.; Amsterdam, The Netherlands: 2009. [Google Scholar]
- 2.Hachitanda Y., Tsuneyoshi M. Neuroblastoma with a distinct organoid pattern: A clinicopathologic, immunohistochemical, and ultrastructural study. Hum. Pathol. 1994;25:67–72. doi: 10.1016/0046-8177(94)90173-2. [DOI] [PubMed] [Google Scholar]
- 3.Zimmermann B. Lung organoid culture. Differentiation. 1987;36:86–109. doi: 10.1111/j.1432-0436.1987.tb00183.x. [DOI] [PubMed] [Google Scholar]
- 4.Xinaris C., Benedetti V., Rizzo P., Abbate M., Corna D., Azzollini N., Conti S., Unbekandt M., Davies J.A., Morigi M., et al. In vivo maturation of functional renal organoids formed from embryonic cell suspensions. J. Am. Soc. Nephrol. 2012;23:1857–1868. doi: 10.1681/ASN.2012050505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Landry J., Bernier D., Ouellet C., Goyette R., Marceau N. Spheroidal aggregate culture of rat liver cells: Histotypic reorganization, biomatrix deposition, and maintenance of functional activities. J. Cell Biol. 1985;101:914–923. doi: 10.1083/JCB.101.3.914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Senoo H., Tsukada Y., Sato T., Hata R.I. Co-culture of fibroblasts and hepatic parenchymal cells induces metabolic changes and formation of a three-dimensional structure. Cell Biol. Int. Rep. 1989;13:197–206. doi: 10.1016/0309-1651(89)90066-0. [DOI] [PubMed] [Google Scholar]
- 7.Takezawa T., Yamazaki M., Mori Y., Yonaha T., Yoshizato K. Morphological and immuno-cytochemical characterization of a hetero-spheroid composed of fibroblasts and hepatocytes. J. Cell Sci. 1992;101:495–501. doi: 10.1242/JCS.101.3.495. [DOI] [PubMed] [Google Scholar]
- 8.Mitaka T., Sato F., Mizuguchi T., Yokono T., Mochizuki Y. Reconstruction of hepatic organoid by rat small hepatocytes and hepatic nonparenchymal cells. Hepatology. 1999;29:111–125. doi: 10.1002/HEP.510290103. [DOI] [PubMed] [Google Scholar]
- 9.Yan Y., Wang X., Pan Y., Liu H., Cheng J., Xiong Z., Lin F., Wu R., Zhang R., Lu Q. Fabrication of viable tissue-engineered constructs with 3D cell-assembly technique. Biomaterials. 2005;26:5864–5871. doi: 10.1016/J.BIOMATERIALS.2005.02.027. [DOI] [PubMed] [Google Scholar]
- 10.Saito M., Matsuura T., Masaki T., Maehashi H., Shimizu K., Hataba Y., Iwahori T., Suzuki T., Braet F. Reconstruction of liver organoid using a bioreactor. World J. Gastroenterol. 2006;12:1881–1888. doi: 10.3748/WJG.V12.I12.1881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Baptista P.M., Siddiqui M.M., Lozier G., Rodriguez S.R., Atala A., Soker S. The use of whole organ decellularization for the generation of a vascularized liver organoid. Hepatology. 2011;53:604–617. doi: 10.1002/HEP.24067. [DOI] [PubMed] [Google Scholar]
- 12.Huch M., Gehart H., Van Boxtel R., Hamer K., Blokzijl F., Verstegen M.M.A., Ellis E., Van Wenum M., Fuchs S.A., De Ligt J., et al. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell. 2015;160:299–312. doi: 10.1016/j.cell.2014.11.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhao B., Ni C., Gao R., Wang Y., Yang L., Wei J., Lv T., Liang J.J., Zhang Q., Xu W., et al. Recapitulation of SARS-CoV-2 infection and cholangiocyte damage with human liver ductal organoids. Protein Cell. 2020;11:771–775. doi: 10.1007/s13238-020-00718-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hendriks D., Artegiani B., Hu H., Chuva de Sousa Lopes S., Clevers H. Establishment of human fetal hepatocyte organoids and CRISPR–Cas9-based gene knockin and knockout in organoid cultures from human liver. Nat. Protoc. 2020;16:182–217. doi: 10.1038/s41596-020-00411-2. [DOI] [PubMed] [Google Scholar]
- 15.Lancaster M.A., Knoblich J.A. Organogenesis in a dish: Modeling development and disease using organoid technologies. Science. 2014;345:1247125. doi: 10.1126/science.1247125. [DOI] [PubMed] [Google Scholar]
- 16.Prior N., Inacio P., Huch M. Liver organoids: From basic research to therapeutic applications. Gut. 2019;68:2228–2237. doi: 10.1136/GUTJNL-2019-319256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Van den Brink S.C., van Oudenaarden A. 3D gastruloids: A novel frontier in stem cell-based in vitro modeling of mammalian gastrulation. Trends Cell Biol. 2021;31:747–759. doi: 10.1016/j.tcb.2021.06.007. [DOI] [PubMed] [Google Scholar]
- 18.Ouchi R., Togo S., Kimura M., Shinozawa T., Koido M., Koike H., Thompson W., Karns R.A., Mayhew C.N., McGrath P.S., et al. Modeling Steatohepatitis in Humans with Pluripotent Stem Cell-Derived Organoids. Cell Metab. 2019;30:374–384.e6. doi: 10.1016/j.cmet.2019.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Guan Y., Xu D., Garfin P.M., Ehmer U., Hurwitz M., Enns G., Michie S., Wu M., Zheng M., Nishimura T., et al. Human hepatic organoids for the analysis of human genetic diseases. J. Clin. Investig. 2017;2:e94954. doi: 10.1172/jci.insight.94954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.LeSavage B.L., Suhar R.A., Broguiere N., Lutolf M.P., Heilshorn S.C. Next-generation cancer organoids. Nat. Mater. 2021;21:143–159. doi: 10.1038/s41563-021-01057-5. [DOI] [PubMed] [Google Scholar]
- 21.De Crignis E., Hossain T., Romal S., Carofiglio F., Moulos P., Khalid M.M., Rao S., Bazrafshan A., Verstegen M.M.A., Pourfarzad F., et al. Application of human liver organoids as a patient-derived primary model for HBV infection and related hepatocellular carcinoma. eLife. 2021;10:e60747. doi: 10.7554/ELIFE.60747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Prior N., Hindley C.J., Rost F., Meléndez E., Lau W.W.Y., Göttgens B., Rulands S., Simons B.D., Huch M. Lgr5+ stem and progenitor cells reside at the apex of a heterogeneous embryonic hepatoblast pool. Development. 2019;146:dev174557. doi: 10.1242/dev.174557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Simian M., Bissell M.J. Organoids: A historical perspective of thinking in three dimensions. J. Cell Biol. 2017;216:31–40. doi: 10.1083/jcb.201610056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bissell D.M., Arenson D.M., Maher J.J., Roll F.J. Support of cultured hepatocytes by a laminin-rich gel. Evidence for a functionally significant subendothelial matrix in normal rat liver. J. Clin. Investig. 1987;79:801–812. doi: 10.1172/JCI112887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Roskelley C.D., Desprez P.Y., Bissell M.J. Extracellular matrix-dependent tissue-specific gene expression in mammary epithelial cells requires both physical and biochemical signal transduction. Proc. Natl. Acad. Sci. USA. 1994;91:12378–12382. doi: 10.1073/pnas.91.26.12378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ootani A., Toda S., Fujimoto K., Sugihara H. Foveolar differentiation of mouse gastric mucosa in vitro. Am. J. Pathol. 2003;162:1905–1912. doi: 10.1016/S0002-9440(10)64324-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sato T., Vries R.G., Snippert H.J., Van De Wetering M., Barker N., Stange D.E., Van Es J.H., Abo A., Kujala P., Peters P.J., et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 2009;459:262–265. doi: 10.1038/nature07935. [DOI] [PubMed] [Google Scholar]
- 28.Sato T., Stange D.E., Ferrante M., Vries R.G.J., Van Es J.H., Van Den Brink S., Van Houdt W.J., Pronk A., Van Gorp J., Siersema P.D., et al. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology. 2011;141:1762–1772. doi: 10.1053/j.gastro.2011.07.050. [DOI] [PubMed] [Google Scholar]
- 29.Barker N., Huch M., Kujala P., van de Wetering M., Snippert H.J., van Es J.H., Sato T., Stange D.E., Begthel H., van den Born M., et al. Lgr5+ve Stem Cells Drive Self-Renewal in the Stomach and Build Long-Lived Gastric Units In Vitro. Cell Stem Cell. 2010;6:25–36. doi: 10.1016/j.stem.2009.11.013. [DOI] [PubMed] [Google Scholar]
- 30.Huch M., Dorrell C., Boj S.F., Van Es J.H., Li V.S.W., Van De Wetering M., Sato T., Hamer K., Sasaki N., Finegold M.J., et al. In vitro expansion of single Lgr5 + liver stem cells induced by Wnt-driven regeneration. Nature. 2013;494:247–250. doi: 10.1038/nature11826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Eiraku M., Watanabe K., Matsuo-Takasaki M., Kawada M., Yonemura S., Matsumura M., Wataya T., Nishiyama A., Muguruma K., Sasai Y. Self-Organized Formation of Polarized Cortical Tissues from ESCs and Its Active Manipulation by Extrinsic Signals. Cell Stem Cell. 2008;3:519–532. doi: 10.1016/j.stem.2008.09.002. [DOI] [PubMed] [Google Scholar]
- 32.Lancaster M.A., Renner M., Martin C.A., Wenzel D., Bicknell L.S., Hurles M.E., Homfray T., Penninger J.M., Jackson A.P., Knoblich J.A. Cerebral organoids model human brain development and microcephaly. Nature. 2013;501:373–379. doi: 10.1038/nature12517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rossi G., Manfrin A., Lutolf M.P. Progress and potential in organoid research. Nat. Rev. Genet. 2018;19:671–687. doi: 10.1038/s41576-018-0051-9. [DOI] [PubMed] [Google Scholar]
- 34.Hu H., Gehart H., Artegiani B., LÖpez-Iglesias C., Dekkers F., Basak O., van Es J., Chuva de Sousa Lopes S.M., Begthel H., Korving J., et al. Long-Term Expansion of Functional Mouse and Human Hepatocytes as 3D Organoids. Cell. 2018;175:1591–1606.e19. doi: 10.1016/j.cell.2018.11.013. [DOI] [PubMed] [Google Scholar]
- 35.Peng W.C., Logan C.Y., Fish M., Anbarchian T., Aguisanda F., Álvarez-Varela A., Wu P., Jin Y., Zhu J., Li B., et al. Inflammatory Cytokine TNFα Promotes the Long-Term Expansion of Primary Hepatocytes in 3D Culture. Cell. 2018;175:1607–1619.e15. doi: 10.1016/j.cell.2018.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Thomson J.A., Itskovitz-Eldor J., Shapiro S.S., Waknitz M.A., Swiergiel J.J., Marshall V.S., Jones J.M. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282:1145–1147. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- 37.Takahashi K., Tanabe K., Ohnuki M., Narita M., Ichisaka T., Tomoda K., Yamanaka S. Induction of Pluripotent Stem Cells from Adult Human Fibroblasts by Defined Factors. Cell. 2007;131:861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 38.Takahashi K., Yamanaka S. Induction of Pluripotent Stem Cells from Mouse Embryonic and Adult Fibroblast Cultures by Defined Factors. Cell. 2006;126:663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 39.Sharma A., Sances S., Workman M.J., Svendsen C.N. Multi-lineage Human iPSC-Derived Platforms for Disease Modeling and Drug Discovery. Cell Stem Cell. 2020;26:309–329. doi: 10.1016/j.stem.2020.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Marsee A., Roos F.J.M., Verstegen M.M.A., Roos F.J.M., Verstegen M.M.A., Clevers H., Vallier L., Takebe T., Huch M., Peng W.C., et al. Building consensus on definition and nomenclature of hepatic, pancreatic, and biliary organoids. Cell Stem Cell. 2021;28:816–832. doi: 10.1016/j.stem.2021.04.005. [DOI] [PubMed] [Google Scholar]
- 41.Dye B.R., Hill D.R., Ferguson M.A., Tsai Y.H., Nagy M.S., Dyal R., Wells J.M., Mayhew C.N., Nattiv R., Klein O.D., et al. In Vitro generation of human pluripotent stem cell derived lung organoids. eLife. 2015;4:e05098. doi: 10.7554/eLife.05098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Takebe T., Sekine K., Enomura M., Koike H., Kimura M., Ogaeri T., Zhang R.R., Ueno Y., Zheng Y.W., Koike N., et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature. 2013;499:481–484. doi: 10.1038/nature12271. [DOI] [PubMed] [Google Scholar]
- 43.Takasato M., Er P.X., Chiu H.S., Maier B., Baillie G.J., Ferguson C., Parton R.G., Wolvetang E.J., Roost M.S., Chuva de Sousa Lopes S.M., et al. Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis. Nature. 2015;526:564–568. doi: 10.1038/nature15695. [DOI] [PubMed] [Google Scholar]
- 44.Hohwieler M., Illing A., Hermann P.C., Mayer T., Stockmann M., Perkhofer L., Eiseler T., Antony J.S., Müller M., Renz S., et al. Human pluripotent stem cell-derived acinar/ductal organoids generate human pancreas upon orthotopic transplantation and allow disease modelling. Gut. 2017;66:473–486. doi: 10.1136/gutjnl-2016-312423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hendriks D., Brouwers J.F., Hamer K., Geurts M.H., Luciana L., Massalini S., López-Iglesias C., Peters P.J., Rodríguez-Colman M.J., Chuva de Sousa Lopes S., et al. Engineered human hepatocyte organoids enable CRISPR-based target discovery and drug screening for steatosis. Nat. Biotechnol. 2023:1–15. doi: 10.1038/s41587-023-01680-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Freag M.S., Namgung B., Reyna Fernandez M.E., Gherardi E., Sengupta S., Jang H.L. Human Nonalcoholic Steatohepatitis on a Chip. Hepatol. Commun. 2021;5:217–233. doi: 10.1002/hep4.1647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wang S., Wang X., Tan Z., Su Y., Liu J., Chang M., Yan F., Chen J., Chen T., Li C., et al. Human ESC-derived expandable hepatic organoids enable therapeutic liver repopulation and pathophysiological modeling of alcoholic liver injury. Cell Res. 2019;29:1009–1026. doi: 10.1038/s41422-019-0242-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Nawroth J.C., Petropolis D.B., Manatakis D.V., Maulana T.I., Burchett G., Schlünder K., Witt A., Shukla A., Kodella K., Ronxhi J., et al. Modeling alcohol-associated liver disease in a human Liver-Chip. Cell Rep. 2021;36:109393. doi: 10.1016/j.celrep.2021.109393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Li P., Li Y., Wang Y., Liu J., Lavrijsen M., Li Y., Zhang R., Verstegen M.M.A., Wang Y., Li T.C., et al. Recapitulating hepatitis E virus-host interactions and facilitating antiviral drug discovery in human liver-derived organoids. Sci. Adv. 2022;8:eabj5908. doi: 10.1126/sciadv.abj5908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nie Y.Z., Zheng Y.W., Miyakawa K., Murata S., Zhang R.R., Sekine K., Ueno Y., Takebe T., Wakita T., Ryo A., et al. Recapitulation of hepatitis B virus–host interactions in liver organoids from human induced pluripotent stem cells. EBioMedicine. 2018;35:114–123. doi: 10.1016/j.ebiom.2018.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ortega-Prieto A.M., Skelton J.K., Wai S.N., Large E., Lussignol M., Vizcay-Barrena G., Hughes D., Fleck R.A., Thursz M., Catanese M.T., et al. 3D microfluidic liver cultures as a physiological preclinical tool for hepatitis B virus infection. Nat. Commun. 2018;9:682. doi: 10.1038/s41467-018-02969-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Natarajan V., Simoneau C.R., Erickson A.L., Meyers N.L., Baron J.L., Cooper S., McDevitt T.C., Ott M. Modelling T-cell immunity against hepatitis C virus with liver organoids in a microfluidic coculture system. Open Biol. 2022;12:210320. doi: 10.1098/rsob.210320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Broutier L., Mastrogiovanni G., Verstegen M.M.A., Francies H.E., Gavarró L.M., Bradshaw C.R., Allen G.E., Arnes-Benito R., Sidorova O., Gaspersz M.P., et al. Human primary liver cancer-derived organoid cultures for disease modeling and drug screening. Nat. Med. 2017;23:1424–1435. doi: 10.1038/nm.4438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kim J., Koo B.K., Knoblich J.A. Human organoids: Model systems for human biology and medicine. Nat. Rev. Mol. Cell Biol. 2020;21:571–584. doi: 10.1038/s41580-020-0259-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Petanjek Z., Judaš M., Šimić G., Rašin M.R., Uylings H.B.M., Rakic P., Kostović I. Extraordinary neoteny of synaptic spines in the human prefrontal cortex. Proc. Natl. Acad. Sci. USA. 2011;108:13281–13286. doi: 10.1073/pnas.1105108108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Stepien B.K., Naumann R., Holtz A., Helppi J., Huttner W.B., Vaid S. Lengthening Neurogenic Period during Neocortical Development Causes a Hallmark of Neocortex Expansion. Curr. Biol. 2020;30:4227–4237.e5. doi: 10.1016/j.cub.2020.08.046. [DOI] [PubMed] [Google Scholar]
- 57.Matsuda M., Hayashi H., Garcia-Ojalvo J., Yoshioka-Kobayashi K., Kageyama R., Yamanaka Y., Ikeya M., Toguchida J., Alev C., Ebisuya M. Species-specific segmentation clock periods are due to differential biochemical reaction speeds. Science. 2020;369:1450–1455. doi: 10.1126/science.aba7668. [DOI] [PubMed] [Google Scholar]
- 58.Rayon T., Stamataki D., Perez-Carrasco R., Garcia-Perez L., Barrington C., Melchionda M., Exelby K., Lazaro J., Tybulewicz V.L.J., Fisher E.M.C., et al. Species-specific pace of development is associated with differences in protein stability. Science. 2020;369:eaba7667. doi: 10.1126/science.aba7667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Subramanian L., Calcagnotto M.E., Paredes M.F. Cortical Malformations: Lessons in Human Brain Development. Front. Cell. Neurosci. 2020;13:576. doi: 10.3389/fncel.2019.00576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Rakic P. Evolution of the neocortex: A perspective from developmental biology. Nat. Rev. Neurosci. 2009;10:724–735. doi: 10.1038/nrn2719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hodge R.D., Bakken T.E., Miller J.A., Smith K.A., Barkan E.R., Graybuck L.T., Close J.L., Long B., Johansen N., Penn O., et al. Conserved cell types with divergent features in human versus mouse cortex. Nature. 2019;573:61–68. doi: 10.1038/s41586-019-1506-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Berg J., Sorensen S.A., Ting J.T., Miller J.A., Chartrand T., Buchin A., Bakken T.E., Budzillo A., Dee N., Ding S.L., et al. Human neocortical expansion involves glutamatergic neuron diversification. Nature. 2021;598:151–158. doi: 10.1038/s41586-021-03813-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ramos M.J., Bandiera L., Menolascina F., Fallowfield J.A. In vitro models for non-alcoholic fatty liver disease: Emerging platforms and their applications. iScience. 2022;25:103549. doi: 10.1016/j.isci.2021.103549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Colca J.R. Discontinued drugs in 2008: Endocrine and metabolic. Expert Opin. Investig. Drugs. 2009;18:1243–1255. doi: 10.1517/13543780903132673. [DOI] [PubMed] [Google Scholar]
- 65.Brennan P.N., Elsharkawy A.M., Kendall T.J., Loomba R., Mann D.A., Fallowfield J.A. Antifibrotic therapy in nonalcoholic steatohepatitis: Time for a human-centric approach. Nat. Rev. Gastroenterol. Hepatol. 2023;20:679–688. doi: 10.1038/s41575-023-00796-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Im Y.R., Hunter H., de Gracia Hahn D., Duret A., Cheah Q., Dong J., Fairey M., Hjalmarsson C., Li A., Lim H.K., et al. A Systematic Review of Animal Models of NAFLD Finds High-Fat, High-Fructose Diets Most Closely Resemble Human NAFLD. Hepatology. 2021;74:1884–1901. doi: 10.1002/hep.31897. [DOI] [PubMed] [Google Scholar]
- 67.von Herrath M., Pagni P.P., Grove K., Christoffersson G., Tang-Christensen M., Karlsen A.E., Petersen J.S. Case Reports of Pre-clinical Replication Studies in Metabolism and Diabetes. Cell Metab. 2019;29:795–802. doi: 10.1016/j.cmet.2019.02.004. [DOI] [PubMed] [Google Scholar]
- 68.Oldham S., Rivera C., Boland M.L., Trevaskis J.L. Incorporation of a Survivable Liver Biopsy Procedure in Mice to Assess Non-alcoholic Steatohepatitis (NASH) Resolution. J. Vis. Exp. 2019;146:e59130. doi: 10.3791/59130. [DOI] [PubMed] [Google Scholar]
- 69.Peek R.M., Blaser M.J. Helicobacter pylori and gastrointestinal tract adenocarcinomas. Nat. Rev. Cancer. 2002;2:28–37. doi: 10.1038/nrc703. [DOI] [PubMed] [Google Scholar]
- 70.Bao L., Deng W., Huang B., Gao H., Liu J., Ren L., Wei Q., Yu P., Xu Y., Qi F., et al. The pathogenicity of SARS-CoV-2 in hACE2 transgenic mice. Nature. 2020;583:830–833. doi: 10.1038/s41586-020-2312-y. [DOI] [PubMed] [Google Scholar]
- 71.Gladwyn-Ng I., Cordón-Barris L., Alfano C., Creppe C., Couderc T., Morelli G., Thelen N., America M., Bessières B., Encha-Razavi F., et al. Stress-induced unfolded protein response contributes to Zika virus–associated microcephaly. Nat. Neurosci. 2017;21:63–71. doi: 10.1038/s41593-017-0038-4. [DOI] [PubMed] [Google Scholar]
- 72.Miner J.J., Cao B., Govero J., Smith A.M., Fernandez E., Cabrera O.H., Garber C., Noll M., Klein R.S., Noguchi K.K., et al. Zika Virus Infection during Pregnancy in Mice Causes Placental Damage and Fetal Demise. Cell. 2016;165:1081–1091. doi: 10.1016/j.cell.2016.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Mason W.S. Animal Models and the Molecular Biology of Hepadnavirus Infection. Cold Spring Harb. Perspect. Med. 2015;5:a021352. doi: 10.1101/cshperspect.a021352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Lempp F.A., Wiedtke E., Qu B., Roques P., Chemin I., Vondran F.W.R., Le Grand R., Grimm D., Urban S. Sodium taurocholate cotransporting polypeptide is the limiting host factor of hepatitis B virus infection in macaque and pig hepatocytes. Hepatology. 2017;66:703–716. doi: 10.1002/hep.29112. [DOI] [PubMed] [Google Scholar]
- 75.Kremsdorf D., Brezillon N. New animal models for hepatitis C viral infection and pathogenesis studies. World J. Gastroenterol. 2007;13:2427–2435. doi: 10.3748/wjg.v13.i17.2427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Gupta A., Madhavan M.V., Sehgal K., Nair N., Mahajan S., Sehrawat T.S., Bikdeli B., Ahluwalia N., Ausiello J.C., Wan E.Y., et al. Extrapulmonary manifestations of COVID-19. Nat. Med. 2020;26:1017–1032. doi: 10.1038/s41591-020-0968-3. [DOI] [PubMed] [Google Scholar]
- 77.Barcellos-Hoff M.H., Aggeler J., Ram T.G., Bissell M.J. Functional differentiation and alveolar morphogenesis of primary mammary cultures on reconstituted basement membrane. Development. 1989;105:223–235. doi: 10.1242/dev.105.2.223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Petersen O.W., Ronnov-Jessen L., Howlett A.R., Bissell M.J. Interaction with basement membrane serves to rapidly distinguish growth and differentiation pattern of normal and malignant human breast epithelial cells. Proc. Natl. Acad. Sci. USA. 1992;89:9064–9068. doi: 10.1073/pnas.89.19.9064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Eiraku M., Takata N., Ishibashi H., Kawada M., Sakakura E., Okuda S., Sekiguchi K., Adachi T., Sasai Y. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature. 2011;472:51–56. doi: 10.1038/nature09941. [DOI] [PubMed] [Google Scholar]
- 80.Suga H., Kadoshima T., Minaguchi M., Ohgushi M., Soen M., Nakano T., Takata N., Wataya T., Muguruma K., Miyoshi H., et al. Self-formation of functional adenohypophysis in three-dimensional culture. Nature. 2011;480:57–62. doi: 10.1038/nature10637. [DOI] [PubMed] [Google Scholar]
- 81.Dekkers J.F., Wiegerinck C.L., De Jonge H.R., Bronsveld I., Janssens H.M., De Winter-De Groot K.M., Brandsma A.M., De Jong N.W.M., Bijvelds M.J.C., Scholte B.J., et al. A functional CFTR assay using primary cystic fibrosis intestinal organoids. Nat. Med. 2013;19:939–945. doi: 10.1038/nm.3201. [DOI] [PubMed] [Google Scholar]
- 82.Schutgens F., Clevers H. Human Organoids: Tools for Understanding Biology and Treating Diseases. Annu. Rev. Pathol. Mech. Dis. 2020;15:211–234. doi: 10.1146/annurev-pathmechdis-012419-032611. [DOI] [PubMed] [Google Scholar]
- 83.Giandomenico S.L., Mierau S.B., Gibbons G.M., Wenger L.M.D., Masullo L., Sit T., Sutcliffe M., Boulanger J., Tripodi M., Derivery E., et al. Cerebral organoids at the air–liquid interface generate diverse nerve tracts with functional output. Nat. Neurosci. 2019;22:669–679. doi: 10.1038/s41593-019-0350-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Sidhaye J., Knoblich J.A. Brain organoids: An ensemble of bioassays to investigate human neurodevelopment and disease. Cell Death Differ. 2020;28:52–67. doi: 10.1038/s41418-020-0566-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Eichmüller O.L., Corsini N.S., Vértesy Á., Morassut I., Scholl T., Gruber V.E., Peer A.M., Chu J., Novatchkova M., Hainfellner J.A., et al. Amplification of human interneuron progenitors promotes brain tumors and neurological defects. Science. 2022;375:eabf5546. doi: 10.1126/science.abf5546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Pellegrini L., Bonfio C., Chadwick J., Begum F., Skehel M., Lancaster M.A. Human CNS barrier-forming organoids with cerebrospinal fluid production. Science. 2020;369:eaaz5626. doi: 10.1126/science.aaz5626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Birey F., Li M.Y., Gordon A., Thete M.V., Valencia A.M., Revah O., Paşca A.M., Geschwind D.H., Paşca S.P. Dissecting the molecular basis of human interneuron migration in forebrain assembloids from Timothy syndrome. Cell Stem Cell. 2022;29:248–264.e7. doi: 10.1016/j.stem.2021.11.011. [DOI] [PubMed] [Google Scholar]
- 88.Kozlowski M.T., Crook C.J., Ku H.T. Towards organoid culture without Matrigel. Commun. Biol. 2021;4:1387. doi: 10.1038/s42003-021-02910-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ober E.A., Lemaigre F.P. Development of the liver: Insights into organ and tissue morphogenesis. J. Hepatol. 2018;68:1049–1062. doi: 10.1016/j.jhep.2018.01.005. [DOI] [PubMed] [Google Scholar]
- 90.Zorn A.M. Liver Development. StemBook. 2008 doi: 10.3824/stembook.1.25.1. [DOI] [Google Scholar]
- 91.Magami Y., Azuma T., Inokuchi H., Kokuno S., Moriyasu F., Kawai K., Hattori T. Cell proliferation and renewal of normal hepatocytes and bile duct cells in adult mouse liver. Liver Int. 2002;22:419–425. doi: 10.1034/j.1600-0676.2002.01702.x. [DOI] [PubMed] [Google Scholar]
- 92.Malato Y., Naqvi S., Schürmann N., Ng R., Wang B., Zape J., Kay M.A., Grimm D., Willenbring H. Fate tracing of mature hepatocytes in mouse liver homeostasis and regeneration. J. Clin. Investig. 2011;121:4850–4860. doi: 10.1172/JCI59261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Wang B., Zhao L., Fish M., Logan C.Y., Nusse R. Self-renewing diploid Axin2 + cells fuel homeostatic renewal of the liver. Nature. 2015;524:180–185. doi: 10.1038/nature14863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Cordero-Espinoza L., Huch M. The balancing act of the liver: Tissue regeneration versus fibrosis. J. Clin. Investig. 2018;128:85–96. doi: 10.1172/JCI93562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Michalopoulos G.K., DeFrances M.C. Liver regeneration. Science. 1997;276:60–65. doi: 10.1126/science.276.5309.60. [DOI] [PubMed] [Google Scholar]
- 96.Kang L.-I., Mars W., Michalopoulos G. Signals and Cells Involved in Regulating Liver Regeneration. Cells. 2012;1:1261–1292. doi: 10.3390/cells1041261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Choi T.Y., Ninov N., Stainier D.Y.R., Shin D. Extensive conversion of hepatic biliary epithelial cells to hepatocytes after near total loss of hepatocytes in zebrafish. Gastroenterology. 2014;146:776–788. doi: 10.1053/j.gastro.2013.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Raven A., Lu W.Y., Man T.Y., Ferreira-Gonzalez S., O’Duibhir E., Dwyer B.J., Thomson J.P., Meehan R.R., Bogorad R., Koteliansky V., et al. Cholangiocytes act as facultative liver stem cells during impaired hepatocyte regeneration. Nature. 2017;547:350–354. doi: 10.1038/nature23015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Deng X., Zhang X., Li W., Feng R.X., Li L., Yi G.R., Zhang X.N., Yin C., Yu H.Y., Zhang J.P., et al. Chronic Liver Injury Induces Conversion of Biliary Epithelial Cells into Hepatocytes. Cell Stem Cell. 2018;23:114–122.e3. doi: 10.1016/j.stem.2018.05.022. [DOI] [PubMed] [Google Scholar]
- 100.Russell J.O., Lu W.Y., Okabe H., Abrams M., Oertel M., Poddar M., Singh S., Forbes S.J., Monga S.P. Hepatocyte-Specific β-Catenin Deletion During Severe Liver Injury Provokes Cholangiocytes to Differentiate Into Hepatocytes. Hepatology. 2019;69:742–759. doi: 10.1002/hep.30270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Armingol E., Officer A., Harismendy O., Lewis N.E. Deciphering cell–cell interactions and communication from gene expression. Nat. Rev. Genet. 2020;22:71–88. doi: 10.1038/s41576-020-00292-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Jung J., Seol H.S., Chang S. The Generation and Application of Patient-Derived Xenograft Model for Cancer Research. Cancer Res. Treat. 2018;50:1–10. doi: 10.4143/crt.2017.307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Zhang X., Ma Z., Song E., Xu T. Islet organoid as a promising model for diabetes. Protein Cell. 2022;13:239–257. doi: 10.1007/s13238-021-00831-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Bakhti M., Böttcher A., Lickert H. Modelling the endocrine pancreas in health and disease. Nat. Rev. Endocrinol. 2019;15:155–171. doi: 10.1038/s41574-018-0132-z. [DOI] [PubMed] [Google Scholar]
- 105.Clevers H. Modeling Development and Disease with Organoids. Cell. 2016;165:1586–1597. doi: 10.1016/j.cell.2016.05.082. [DOI] [PubMed] [Google Scholar]
- 106.Campana L., Esser H., Huch M., Forbes S. Liver regeneration and inflammation: From fundamental science to clinical applications. Nat. Rev. Mol. Cell Biol. 2021;22:608–624. doi: 10.1038/s41580-021-00373-7. [DOI] [PubMed] [Google Scholar]
- 107.Sun L., Wang Y., Cen J., Ma X., Cui L., Qiu Z., Zhang Z., Li H., Yang R.Z., Wang C., et al. Modelling liver cancer initiation with organoids derived from directly reprogrammed human hepatocytes. Nat. Cell Biol. 2019;21:1015–1026. doi: 10.1038/s41556-019-0359-5. [DOI] [PubMed] [Google Scholar]
- 108.Takebe T., Sekine K., Kimura M., Yoshizawa E., Ayano S., Koido M., Funayama S., Nakanishi N., Hisai T., Kobayashi T., et al. Massive and Reproducible Production of Liver Buds Entirely from Human Pluripotent Stem Cells. Cell Rep. 2017;21:2661–2670. doi: 10.1016/j.celrep.2017.11.005. [DOI] [PubMed] [Google Scholar]
- 109.Lam D.T.U.H., Dan Y.Y., Chan Y.S., Ng H.H. Emerging liver organoid platforms and technologies. Cell Regen. 2021;10:27. doi: 10.1186/s13619-021-00089-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Zong Y., Panikkar A., Xu J., Antoniou A., Raynaud P., Lemaigre F., Stanger B.Z. Notch signaling controls liver development by regulating biliary differentiation. Development. 2009;136:1727–1739. doi: 10.1242/dev.029140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Dianat N., Dubois-Pot-Schneider H., Steichen C., Desterke C., Leclerc P., Raveux A., Combettes L., Weber A., Corlu A., Dubart-Kupperschmitt A. Generation of functional cholangiocyte-like cells from human pluripotent stem cells and HepaRG cells. Hepatology. 2014;60:700–714. doi: 10.1002/hep.27165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Ogawa M., Ogawa S., Bear C.E., Ahmadi S., Chin S., Li B., Grompe M., Keller G., Kamath B.M., Ghanekar A. Directed differentiation of cholangiocytes from human pluripotent stem cells. Nat. Biotechnol. 2015;33:853–861. doi: 10.1038/nbt.3294. [DOI] [PubMed] [Google Scholar]
- 113.Sampaziotis F., De Brito M.C., Geti I., Bertero A., Hannan N.R.F., Vallier L. Directed differentiation of human induced pluripotent stem cells into functional cholangiocyte-like cells. Nat. Protoc. 2017;12:814–827. doi: 10.1038/nprot.2017.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Sampaziotis F., Muraro D., Tysoe O.C., Sawiak S., Beach T.E., Godfrey E.M., Upponi S.S., Brevini T., Wesley B.T., Garcia-Bernardo J., et al. Cholangiocyte organoids can repair bile ducts after transplantation in the human liver. Science. 2021;371:839–846. doi: 10.1126/science.aaz6964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Wu F., Wu D., Ren Y., Huang Y., Feng B., Zhao N., Zhang T., Chen X., Chen S., Xu A. Generation of hepatobiliary organoids from human induced pluripotent stem cells. J. Hepatol. 2019;70:1145–1158. doi: 10.1016/j.jhep.2018.12.028. [DOI] [PubMed] [Google Scholar]
- 116.Bin Ramli M.N., Lim Y.S., Koe C.T., Demircioglu D., Tng W., Gonzales K.A.U., Tan C.P., Szczerbinska I., Liang H., Soe E.L., et al. Human Pluripotent Stem Cell-Derived Organoids as Models of Liver Disease. Gastroenterology. 2020;159:1471–1486.e12. doi: 10.1053/j.gastro.2020.06.010. [DOI] [PubMed] [Google Scholar]
- 117.Akbari S., Sevinç G.G., Ersoy N., Basak O., Kaplan K., Sevinç K., Ozel E., Sengun B., Enustun E., Ozcimen B., et al. Robust, Long-Term Culture of Endoderm-Derived Hepatic Organoids for Disease Modeling. Stem Cell Rep. 2019;13:627–641. doi: 10.1016/j.stemcr.2019.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Shinozawa T., Kimura M., Cai Y., Saiki N., Yoneyama Y., Ouchi R., Koike H., Maezawa M., Zhang R.R., Dunn A., et al. High-Fidelity Drug-Induced Liver Injury Screen Using Human Pluripotent Stem Cell-Derived Organoids. Gastroenterology. 2021;160:831–846. doi: 10.1053/j.gastro.2020.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Koike H., Iwasawa K., Ouchi R., Maezawa M., Giesbrecht K., Saiki N., Ferguson A., Kimura M., Thompson W.L., Wells J.M., et al. Modelling human hepato-biliary-pancreatic organogenesis from the foregut-midgut boundary. Nature. 2019;574:112–116. doi: 10.1038/s41586-019-1598-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Xiang Y., Tanaka Y., Cakir B., Patterson B., Kim K.Y., Sun P., Kang Y.J., Zhong M., Liu X., Patra P., et al. hESC-Derived Thalamic Organoids Form Reciprocal Projections When Fused with Cortical Organoids. Cell Stem Cell. 2019;24:487–497.e7. doi: 10.1016/j.stem.2018.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Xiang Y., Tanaka Y., Patterson B., Kang Y.J., Govindaiah G., Roselaar N., Cakir B., Kim K.Y., Lombroso A.P., Hwang S.M., et al. Fusion of Regionally Specified hPSC-Derived Organoids Models Human Brain Development and Interneuron Migration. Cell Stem Cell. 2017;21:383–398.e7. doi: 10.1016/j.stem.2017.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Miura Y., Li M.Y., Birey F., Ikeda K., Revah O., Thete M.V., Park J.Y., Puno A., Lee S.H., Porteus M.H., et al. Generation of human striatal organoids and cortico-striatal assembloids from human pluripotent stem cells. Nat. Biotechnol. 2020;38:1421–1430. doi: 10.1038/s41587-020-00763-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Al Shihabi A., Davarifar A., Lam Nguyen H.T., Tavanaie N., Nelson S.D., Yanagawa J., Federman N., Bernthal N., Hornicek F., Soragni A. Personalized chordoma organoids for drug discovery studies. Sci. Adv. 2022;8:3674. doi: 10.1126/sciadv.abl3674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Kim M., Mun H., Sung C.O., Cho E.J., Jeon H.J., Chun S.M., Jung D.J., Shin T.H., Jeong G.S., Kim D.K., et al. Patient-derived lung cancer organoids as in vitro cancer models for therapeutic screening. Nat. Commun. 2019;10:3991. doi: 10.1038/s41467-019-11867-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Lee S.H., Hu W., Matulay J.T., Silva M.V., Owczarek T.B., Kim K., Chua C.W., Barlow L.M.J., Kandoth C., Williams A.B., et al. Tumor Evolution and Drug Response in Patient-Derived Organoid Models of Bladder Cancer. Cell. 2018;173:515–528.e17. doi: 10.1016/j.cell.2018.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Yu L., Li Z., Mei H., Li W., Chen D., Liu L., Zhang Z., Sun Y., Song F., Chen W., et al. Patient-derived organoids of bladder cancer recapitulate antigen expression profiles and serve as a personal evaluation model for CAR-T cells in vitro. Clin. Transl. Immunol. 2021;10:e1248. doi: 10.1002/cti2.1248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Li X., Francies H.E., Secrier M., Perner J., Miremadi A., Galeano-Dalmau N., Barendt W.J., Letchford L., Leyden G.M., Goffin E.K., et al. Organoid cultures recapitulate esophageal adenocarcinoma heterogeneity providing a model for clonality studies and precision therapeutics. Nat. Commun. 2018;9:2983. doi: 10.1038/s41467-018-05190-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Honkala A., Malhotra S.V., Kummar S., Junttila M.R. Harnessing the predictive power of preclinical models for oncology drug development. Nat. Rev. Drug Discov. 2021;21:99–114. doi: 10.1038/s41573-021-00301-6. [DOI] [PubMed] [Google Scholar]
- 129.Gerlinger M., Rowan A.J., Horswell S., Larkin J., Endesfelder D., Gronroos E., Martinez P., Matthews N., Stewart A., Tarpey P., et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N. Engl. J. Med. 2012;366:883–892. doi: 10.1056/NEJMoa1113205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Aizarani N., Saviano A., Sagar, Mailly L., Durand S., Herman J.S., Pessaux P., Baumert T.F., Grün D. A human liver cell atlas reveals heterogeneity and epithelial progenitors. Nature. 2019;572:199–204. doi: 10.1038/s41586-019-1373-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Albhaisi S., Sanyal A.J. Gene-Environmental Interactions as Metabolic Drivers of Nonalcoholic Steatohepatitis. Front. Endocrinol. 2021;12:665987. doi: 10.3389/fendo.2021.665987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Deczkowska A., David E., Ramadori P., Pfister D., Safran M., Li B., Giladi A., Jaitin D.A., Barboy O., Cohen M., et al. XCR1+ type 1 conventional dendritic cells drive liver pathology in non-alcoholic steatohepatitis. Nat. Med. 2021;27:1043–1054. doi: 10.1038/s41591-021-01344-3. [DOI] [PubMed] [Google Scholar]
- 133.Seidman J.S., Troutman T.D., Sakai M., Gola A., Spann N.J., Bennett H., Bruni C.M., Ouyang Z., Li R.Z., Sun X., et al. Niche-Specific Reprogramming of Epigenetic Landscapes Drives Myeloid Cell Diversity in Nonalcoholic Steatohepatitis. Immunity. 2020;52:1057–1074.e7. doi: 10.1016/j.immuni.2020.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Lamas-Paz A., Hao F., Nelson L.J., Vázquez M.T., Canals S., del Moral M.G., Martínez-Naves E., Nevzorova Y.A., Cubero F.J. Alcoholic liver disease: Utility of animal models. World J. Gastroenterol. 2018;24:5063–5075. doi: 10.3748/wjg.v24.i45.5063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Frazier T.H., Mcclain C.J., Kershner N.A., Marsano L.S. Treatment of alcoholic liver disease. Therap. Adv. Gastroenterol. 2011;4:63–81. doi: 10.1177/1756283X10378925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Tsang H.Y., Lo P.H.Y., Lee K.K.H. Generation of liver organoids from human induced pluripotent stem cells as liver fibrosis and steatosis models. bioRxiv. 2021:preprint. doi: 10.1101/2021.06.29.450347. [DOI] [Google Scholar]
- 137.McCarron S., Bathon B., Conlon D.M., Abbey D., Rader D.J., Gawronski K., Brown C.D., Olthoff K.M., Shaked A., Raabe T.D. Functional Characterization of Organoids Derived from Irreversibly Damaged Liver of Patients with NASH. Hepatology. 2021;74:1825–1844. doi: 10.1002/hep.31857. [DOI] [PubMed] [Google Scholar]
- 138.Elbadawy M., Yamanaka M., Goto Y., Hayashi K., Tsunedomi R., Hazama S., Nagano H., Yoshida T., Shibutani M., Ichikawa R., et al. Efficacy of primary liver organoid culture from different stages of non-alcoholic steatohepatitis (NASH) mouse model. Biomaterials. 2020;237:119823. doi: 10.1016/J.BIOMATERIALS.2020.119823. [DOI] [PubMed] [Google Scholar]
- 139.Younossi Z.M., Koenig A.B., Abdelatif D., Fazel Y., Henry L., Wymer M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64:73–84. doi: 10.1002/hep.28431. [DOI] [PubMed] [Google Scholar]
- 140.Al-Goblan A.S., Al-Alfi M.A., Khan M.Z. Mechanism linking diabetes mellitus and obesity. Diabetes Metab. Syndr. Obes. 2014;7:587–591. doi: 10.2147/DMSO.S67400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Romeo S., Kozlitina J., Xing C., Pertsemlidis A., Cox D., Pennacchio L.A., Boerwinkle E., Cohen J.C., Hobbs H.H. Genetic variation in PNPLA3 confers susceptibility to nonalcoholic fatty liver disease. Nat. Genet. 2008;40:1461–1465. doi: 10.1038/ng.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Pingitore P., Sasidharan K., Ekstrand M., Prill S., Lindén D., Romeo S. Human Multilineage 3D Spheroids as a Model of Liver Steatosis and Fibrosis. Int. J. Mol. Sci. 2019;20:1629. doi: 10.3390/ijms20071629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Schwartz B.E., Rajagopal V., Smith C., Cohick E., Whissell G., Gamboa M., Pai R., Sigova A., Grossman I., Bumcrot D., et al. Discovery and Targeting of the Signaling Controls of PNPLA3 to Effectively Reduce Transcription, Expression, and Function in Pre-Clinical NAFLD/NASH Settings. Cells. 2020;9:2247. doi: 10.3390/cells9102247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Prill S., Caddeo A., Baselli G., Jamialahmadi O., Dongiovanni P., Rametta R., Kanebratt K.P., Pujia A., Pingitore P., Mancina R.M., et al. The TM6SF2 E167K genetic variant induces lipid biosynthesis and reduces apolipoprotein B secretion in human hepatic 3D spheroids. Sci. Rep. 2019;9:11585. doi: 10.1038/S41598-019-47737-W. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Tanaka Y., Shimanaka Y., Caddeo A., Kubo T., Mao Y., Kubota T., Kubota N., Yamauchi T., Mancina R.M., Baselli G., et al. LPIAT1/MBOAT7 depletion increases triglyceride synthesis fueled by high phosphatidylinositol turnover. Gut. 2021;70:180–193. doi: 10.1136/gutjnl-2020-320646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Van der Vaart J., Lamers M.M., Haagmans B.L., Clevers H. Advancing lung organoids for COVID-19 research. DMM Dis. Model. Mech. 2021;14:dmm049060. doi: 10.1242/dmm.049060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Hou Y.J., Okuda K., Edwards C.E., Martinez D.R., Asakura T., Dinnon K.H., Kato T., Lee R.E., Yount B.L., Mascenik T.M., et al. SARS-CoV-2 Reverse Genetics Reveals a Variable Infection Gradient in the Respiratory Tract. Cell. 2020;182:429–446.e14. doi: 10.1016/j.cell.2020.05.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Chen N., Zhou M., Dong X., Qu J., Gong F., Han Y., Qiu Y., Wang J., Liu Y., Wei Y., et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet. 2020;395:507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Zhu N., Zhang D., Wang W., Li X., Yang B., Song J., Zhao X., Huang B., Shi W., Lu R., et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Fan Z., Chen L., Li J., Cheng X., Yang J., Tian C., Zhang Y., Huang S., Liu Z., Cheng J. Clinical Features of COVID-19-Related Liver Functional Abnormality. Clin. Gastroenterol. Hepatol. 2020;18:1561–1566. doi: 10.1016/j.cgh.2020.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Chai X., Hu L., Zhang Y., Han W., Lu Z., Ke A., Zhou J., Shi G., Fang N., Fan J.J., et al. Specific ACE2 Expression in Cholangiocytes May Cause Liver Damage After 2019-nCoV Infection. bioRxiv. 2020:preprint. doi: 10.1101/2020.02.03.931766. [DOI] [Google Scholar]
- 153.Xu Z., Shi L., Wang Y., Zhang J., Huang L., Zhang C., Liu S., Zhao P., Liu H., Zhu L., et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020;8:420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Liu J., Li S., Liu J., Liang B., Wang X., Wang H., Li W., Tong Q., Yi J., Zhao L., et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55:102763. doi: 10.1016/j.ebiom.2020.102763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Sun J., Aghemo A., Forner A., Valenti L. COVID-19 and liver disease. Liver Int. 2020;40:1278–1281. doi: 10.1111/liv.14470. [DOI] [PubMed] [Google Scholar]
- 157.Mantovani A., Beatrice G., Dalbeni A. Coronavirus disease 2019 and prevalence of chronic liver disease: A meta-analysis. Liver Int. 2020;40:1316–1320. doi: 10.1111/liv.14465. [DOI] [PubMed] [Google Scholar]
- 158.Ji D., Qin E., Xu J., Zhang D., Cheng G., Wang Y., Lau G. Non-alcoholic fatty liver diseases in patients with COVID-19: A retrospective study. J. Hepatol. 2020;73:451–453. doi: 10.1016/j.jhep.2020.03.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Van De Wetering M., Francies H.E., Francis J.M., Bounova G., Iorio F., Pronk A., Van Houdt W., Van Gorp J., Taylor-Weiner A., Kester L., et al. Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell. 2015;161:933–945. doi: 10.1016/j.cell.2015.03.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Vlachogiannis G., Hedayat S., Vatsiou A., Jamin Y., Fernández-Mateos J., Khan K., Lampis A., Eason K., Huntingford I., Burke R., et al. Patient-derived organoids model treatment response of metastatic gastrointestinal cancers. Science. 2018;359:920–926. doi: 10.1126/science.aao2774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Gao D., Vela I., Sboner A., Iaquinta P.J., Karthaus W.R., Gopalan A., Dowling C., Wanjala J.N., Undvall E.A., Arora V.K., et al. Organoid Cultures Derived from Patients with Advanced Prostate Cancer. Cell. 2014;159:176–187. doi: 10.1016/j.cell.2014.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Kopper O., de Witte C.J., Lõhmussaar K., Valle-Inclan J.E., Hami N., Kester L., Balgobind A.V., Korving J., Proost N., Begthel H., et al. An organoid platform for ovarian cancer captures intra- and interpatient heterogeneity. Nat. Med. 2019;25:838–849. doi: 10.1038/s41591-019-0422-6. [DOI] [PubMed] [Google Scholar]
- 163.Tiriac H., Belleau P., Engle D.D., Plenker D., Deschênes A., Somerville T.D.D., Froeling F.E.M., Burkhart R.A., Denroche R.E., Jang G.H., et al. Organoid profiling identifies common responders to chemotherapy in pancreatic cancer. Cancer Discov. 2018;8:1112–1129. doi: 10.1158/2159-8290.CD-18-0349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Fabre K., Berridge B., Proctor W.R., Ralston S., Will Y., Baran S.W., Yoder G., Van Vleet T.R. Introduction to a manuscript series on the characterization and use of microphysiological systems (MPS) in pharmaceutical safety and ADME applications. Lab Chip. 2020;20:1049–1057. doi: 10.1039/C9LC01168D. [DOI] [PubMed] [Google Scholar]
- 165.Golding H., Khurana S., Zaitseva M. What Is the Predictive Value of Animal Models for Vaccine Efficacy in Humans? The Importance of Bridging Studies and Species-Independent Correlates of Protection. Cold Spring Harb. Perspect. Biol. 2018;10:a028902. doi: 10.1101/cshperspect.a028902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Barrile R., van der Meer A.D., Park H., Fraser J.P., Simic D., Teng F., Conegliano D., Nguyen J., Jain A., Zhou M., et al. Organ-on-Chip Recapitulates Thrombosis Induced by an anti-CD154 Monoclonal Antibody: Translational Potential of Advanced Microengineered Systems. Clin. Pharmacol. Ther. 2018;104:1240–1248. doi: 10.1002/cpt.1054. [DOI] [PubMed] [Google Scholar]
- 167.Seok J., Warren H.S., Cuenca A.G., Mindrinos M.N., Baker H.V., Xu W., Richards D.R., McDonald-Smith G.P., Gao H., Hennessy L., et al. Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc. Natl. Acad. Sci. USA. 2013;110:3507–3512. doi: 10.1073/pnas.1222878110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Franco R., Cedazo-Minguez A. Successful therapies for Alzheimer’s disease: Why so many in animal models and none in humans? Front. Pharmacol. 2014;5:146. doi: 10.3389/fphar.2014.00146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Ingber D.E. Human organs-on-chips for disease modelling, drug development and personalized medicine. Nat. Rev. Genet. 2022;23:467–491. doi: 10.1038/s41576-022-00466-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Sarkar U., Rivera-Burgos D., Large E.M., Hughes D.J., Ravindra K.C., Dyer R.L., Ebrahimkhani M.R., Wishnok J.S., Griffith L.G., Tannenbaum S.R. Metabolite profiling and pharmacokinetic evaluation of hydrocortisone in a perfused three-dimensional human liver bioreactor. Drug Metab. Dispos. 2015;43:1091–1099. doi: 10.1124/dmd.115.063495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Long T.J., Cosgrove P.A., Dunn R.T., Stolz D.B., Hamadeh H., Afshari C., McBride H., Griffith L.G. Modeling Therapeutic Antibody-Small Molecule Drug-Drug Interactions Using a Three-Dimensional Perfusable Human Liver Coculture Platform. Drug Metab. Dispos. 2016;44:1940–1948. doi: 10.1124/dmd.116.071456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Tsamandouras N., Kostrzewski T., Stokes C.L., Griffith L.G., Hughes D.J., Cirit M. Quantitative Assessment of Population Variability in Hepatic Drug Metabolism Using a Perfused Three-Dimensional Human Liver Microphysiological System. J. Pharmacol. Exp. Ther. 2017;360:95–105. doi: 10.1124/jpet.116.237495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Veninga V., Voest E.E. Tumor organoids: Opportunities and challenges to guide precision medicine. Cancer Cell. 2021;39:1190–1201. doi: 10.1016/j.ccell.2021.07.020. [DOI] [PubMed] [Google Scholar]
- 174.Wensink G.E., Elias S.G., Mullenders J., Koopman M., Boj S.F., Kranenburg O.W., Roodhart J.M.L. Patient-derived organoids as a predictive biomarker for treatment response in cancer patients. NPJ Precis. Oncol. 2021;5:30. doi: 10.1038/s41698-021-00168-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Sachs N., de Ligt J., Kopper O., Gogola E., Bounova G., Weeber F., Balgobind A.V., Wind K., Gracanin A., Begthel H., et al. A Living Biobank of Breast Cancer Organoids Captures Disease Heterogeneity. Cell. 2018;172:373–386.e10. doi: 10.1016/j.cell.2017.11.010. [DOI] [PubMed] [Google Scholar]
- 176.Bar-Ephraim Y.E., Kretzschmar K., Clevers H. Organoids in immunological research. Nat. Rev. Immunol. 2019;20:279–293. doi: 10.1038/s41577-019-0248-y. [DOI] [PubMed] [Google Scholar]
- 177.Zhang C.J., Meyer S.R., O’Meara M.J., Huang S., Capeling M.M., Ferrer-Torres D., Childs C.J., Spence J.R., Fontana R.J., Sexton J.Z. A human liver organoid screening platform for DILI risk prediction. J. Hepatol. 2023;78:998–1006. doi: 10.1016/j.jhep.2023.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Yuki K., Cheng N., Nakano M., Kuo C.J. Organoid Models of Tumor Immunology. Trends Immunol. 2020;41:652–664. doi: 10.1016/j.it.2020.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Dijkstra K.K., Cattaneo C.M., Weeber F., Chalabi M., van de Haar J., Fanchi L.F., Slagter M., van der Velden D.L., Kaing S., Kelderman S., et al. Generation of Tumor-Reactive T Cells by Co-culture of Peripheral Blood Lymphocytes and Tumor Organoids. Cell. 2018;174:1586–1598.e12. doi: 10.1016/j.cell.2018.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Jacob F., Ming G.-L., Song H. Generation and biobanking of patient-derived glioblastoma organoids and their application in CAR T cell testing. Nat. Protoc. 2020;15:4000–4033. doi: 10.1038/s41596-020-0402-9. [DOI] [PubMed] [Google Scholar]
- 181.Guo J., Tang Q. Recent updates on chimeric antigen receptor T cell therapy for hepatocellular carcinoma. Cancer Gene Ther. 2021;28:1075–1087. doi: 10.1038/s41417-020-00259-4. [DOI] [PubMed] [Google Scholar]
- 182.Li D., English H., Hong J., Liang T., Merlino G., Day C.P., Ho M. A novel PD-L1-targeted shark VNAR single-domain-based CAR-T cell strategy for treating breast cancer and liver cancer. Mol. Ther.-Oncolytics. 2022;24:849–863. doi: 10.1016/j.omto.2022.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Wang Z., McWilliams-Koeppen H.P., Reza H., Ostberg J.R., Chen W., Wang X., Huynh C., Vyas V., Chang W.C., Starr R., et al. 3D-organoid culture supports differentiation of human CAR+ iPSCs into highly functional CAR T cells. Cell Stem Cell. 2022;29:515–527.e8. doi: 10.1016/j.stem.2022.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Turksen K., editor. Cell Biology and Translational Medicine. Volume 14. Springer; Cham, Switzerland: 2021. [DOI] [Google Scholar]
- 185.Berkers G., van Mourik P., Vonk A.M., Kruisselbrink E., Dekkers J.F., de Winter-de Groot K.M., Arets H.G.M., Marck-van der Wilt R.E.P., Dijkema J.S., Vanderschuren M.M., et al. Rectal Organoids Enable Personalized Treatment of Cystic Fibrosis. Cell Rep. 2019;26:1701–1708.e3. doi: 10.1016/j.celrep.2019.01.068. [DOI] [PubMed] [Google Scholar]
- 186.Drost J., Clevers H. Organoids in cancer research. Nat. Rev. Cancer. 2018;18:407–418. doi: 10.1038/s41568-018-0007-6. [DOI] [PubMed] [Google Scholar]
- 187.Brooks A., Liang X., Zhang Y., Zhao C.X., Roberts M.S., Wang H., Zhang L., Crawford D.H.G. Liver organoid as a 3D in vitro model for drug validation and toxicity assessment. Pharmacol. Res. 2021;169:105608. doi: 10.1016/j.phrs.2021.105608. [DOI] [PubMed] [Google Scholar]
- 188.Matsui T., Shinozawa T. Human Organoids for Predictive Toxicology Research and Drug Development. Front. Genet. 2021;12:767621. doi: 10.3389/fgene.2021.767621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Kennedy E.M., Cullen B.R. Gene Editing: A New Tool for Viral Disease. Annu. Rev. Med. 2017;68:401–411. doi: 10.1146/annurev-med-051215-031129. [DOI] [PubMed] [Google Scholar]
- 190.Roper J., Yilmaz Ö.H. Breakthrough Moments: Genome Editing and Organoids. Cell Stem Cell. 2019;24:841–842. doi: 10.1016/j.stem.2019.05.008. [DOI] [PubMed] [Google Scholar]
- 191.Melton D. The promise of stem cell-derived islet replacement therapy. Diabetologia. 2021;64:1030–1036. doi: 10.1007/s00125-020-05367-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Wang C.H., Lundh M., Fu A., Kriszt R., Huang T.L., Lynes M.D., Leiria L.O., Shamsi F., Darcy J., Greenwood B.P., et al. CRISPR-engineered human brown-like adipocytes prevent diet-induced obesity and ameliorate metabolic syndrome in mice. Sci. Transl. Med. 2020;12:eaaz8664. doi: 10.1126/scitranslmed.aaz8664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Tsagkaraki E., Nicoloro S.M., DeSouza T., Solivan-Rivera J., Desai A., Lifshitz L.M., Shen Y., Kelly M., Guilherme A., Henriques F., et al. CRISPR-enhanced human adipocyte browning as cell therapy for metabolic disease. Nat. Commun. 2021;12:6931. doi: 10.1038/s41467-021-27190-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Schene I.F., Joore I.P., Oka R., Mokry M., van Vugt A.H.M., van Boxtel R., van der Doef H.P.J., van der Laan L.J.W., Verstegen M.M.A., van Hasselt P.M., et al. Prime editing for functional repair in patient-derived disease models. Nat. Commun. 2020;11:5352. doi: 10.1038/s41467-020-19136-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Zarrinpar A., Busuttil R.W. Liver transplantation: Past, present and future. Nat. Rev. Gastroenterol. Hepatol. 2013;10:434–440. doi: 10.1038/nrgastro.2013.88. [DOI] [PubMed] [Google Scholar]
- 196.Bhaduri A., Andrews M.G., Mancia Leon W., Jung D., Shin D., Allen D., Jung D., Schmunk G., Haeussler M., Salma J., et al. Cell stress in cortical organoids impairs molecular subtype specification. Nature. 2020;578:142–148. doi: 10.1038/s41586-020-1962-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Mansour A.A., Gonçalves J.T., Bloyd C.W., Li H., Fernandes S., Quang D., Johnston S., Parylak S.L., Jin X., Gage F.H. An in vivo model of functional and vascularized human brain organoids. Nat. Biotechnol. 2018;36:432–441. doi: 10.1038/nbt.4127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Mansour A.A.F., Schafer S.T., Gage F.H. Cellular complexity in brain organoids: Current progress and unsolved issues. Semin. Cell Dev. Biol. 2021;111:32–39. doi: 10.1016/j.semcdb.2020.05.013. [DOI] [PubMed] [Google Scholar]
- 199.Vértesy Á., Eichmueller O.L., Naas J., Novatchkova M., Esk C., Balmaña M., Ladstaetter S., Bock C., von Haeseler A., Knoblich J.A. Cellular stress in brain organoids is limited to a distinct and bioinformatically removable subpopulation. bioRxiv. 2022:preprint. doi: 10.1101/2022.03.11.483643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Hofer M., Lutolf M.P. Engineering organoids. Nat. Rev. Mater. 2021;6:402–420. doi: 10.1038/s41578-021-00279-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The links to data available in Table 3 can be found in Supplementary Table S1. Any other information related to the article can be obtained upon request.